Reactivation of Frozen Stored Microalgal-Bacterial Granular Sludge under Aeration and Non-Aeration Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthetic Municipal Wastewater
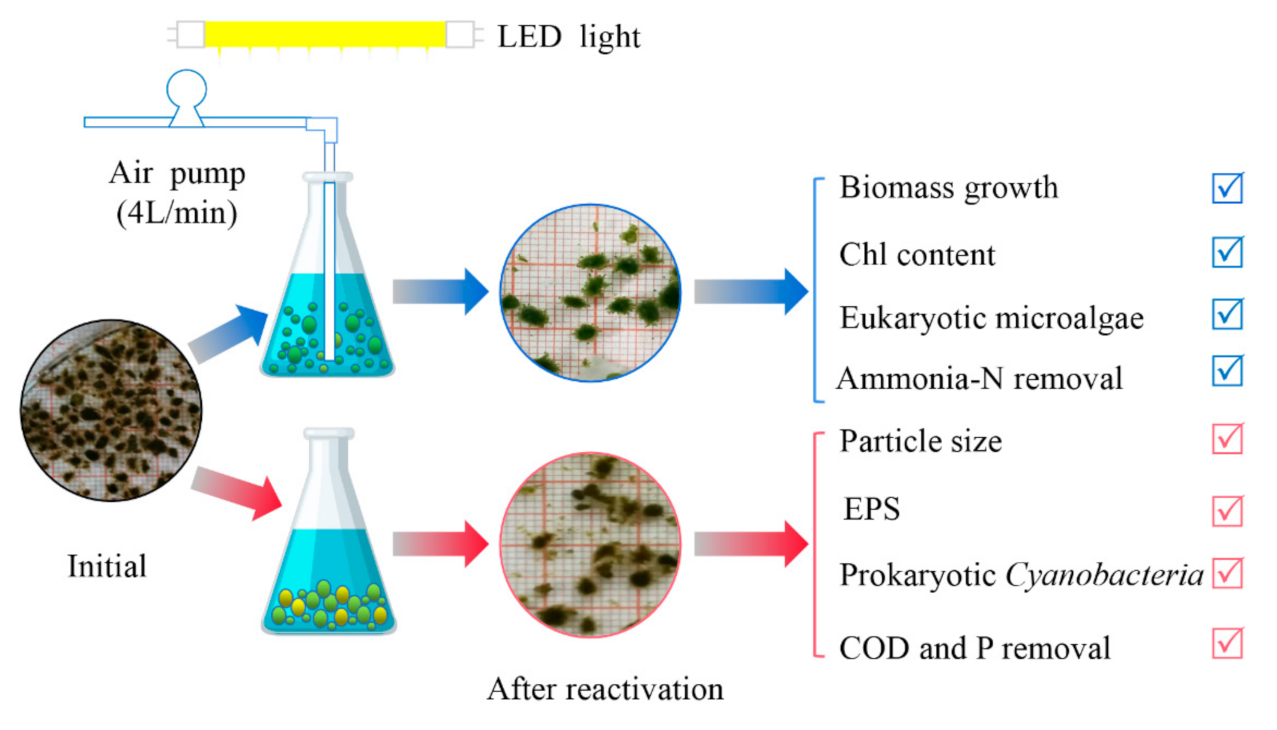
2.2. Experimental Setup
2.3. Microbial Community Analysis
2.4. Analytical Methods
2.5. Statistical Analysis
3. Results and Discussion
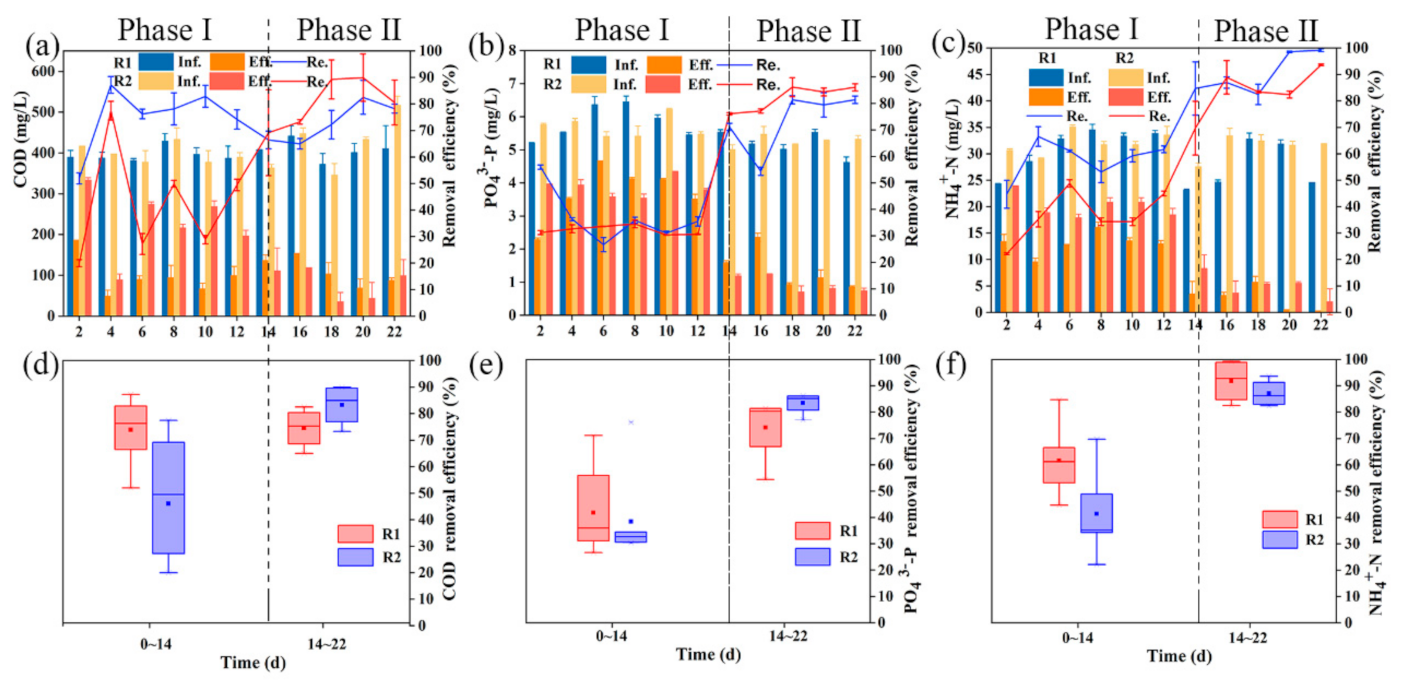
3.1. Reactor Performance
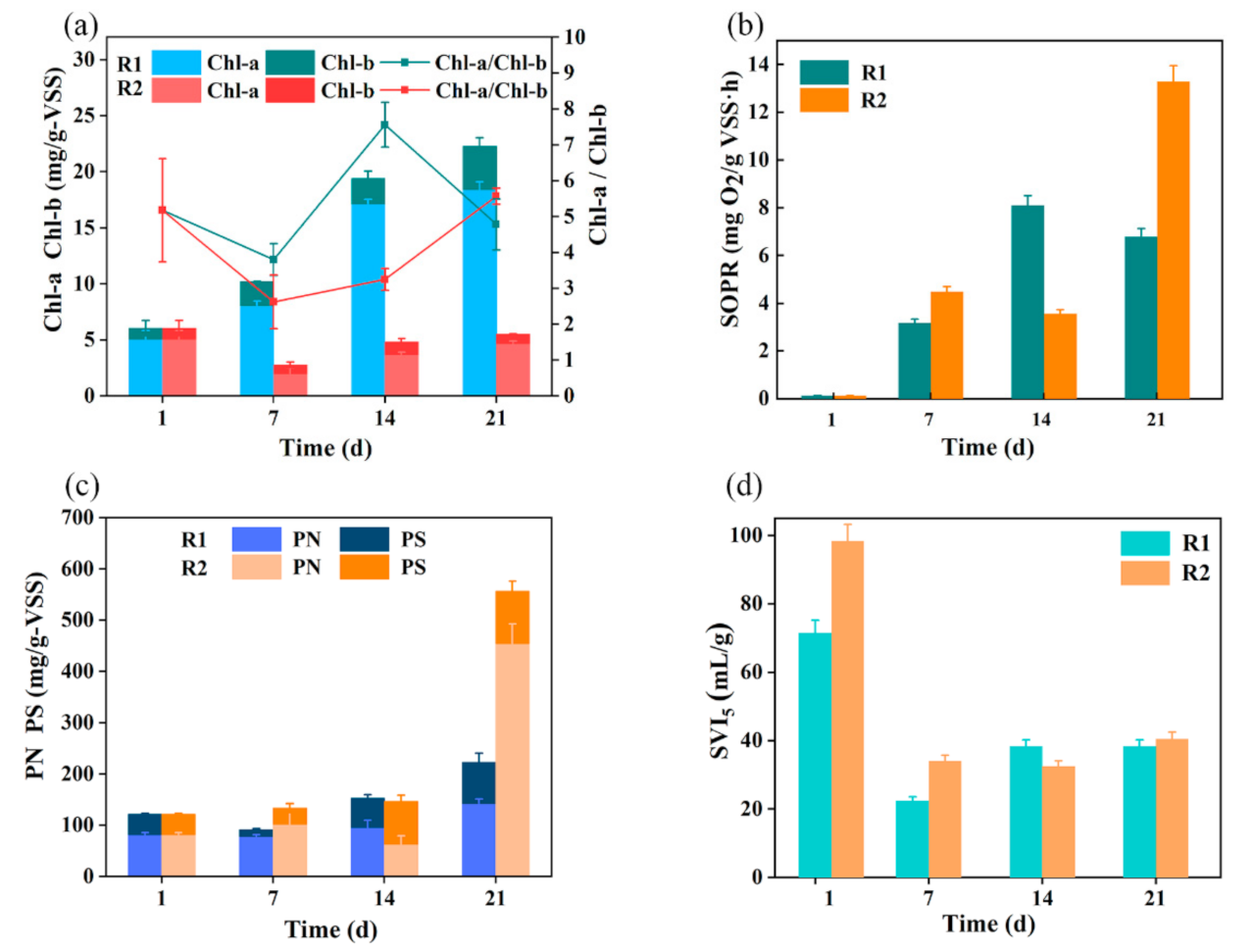
3.2. Morphological and Characteristic of MBGS
3.3. Microbial Community Succession
3.4. Potential Functional Characteristics of Microorganisms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, X.; Zhang, M.; Liu, H.; Gu, J.; Liu, Y. Environmental sustainability: A pressing challenge to biological sewage treatment processes. Curr. Opin. Environ. Sci. Health 2019, 12, 1–5. [Google Scholar] [CrossRef]
- Nordlander, E.; Olsson, J.; Thorin, E.; Nehrenheim, E. Simulation of energy balance and carbon dioxide emission for microalgae introduction in wastewater treatment plants. Algal Res. 2017, 24, 251–260. [Google Scholar] [CrossRef] [Green Version]
- Guo, W.-Q.; Yang, S.-S.; Xiang, W.-S.; Wang, X.-J.; Ren, N.-Q. Minimization of excess sludge production by in-situ activated sludge treatment processes—A comprehensive review. Biotechnol. Adv. 2013, 31, 1386–1396. [Google Scholar] [CrossRef]
- Franca, R.D.; Pinheiro, H.M.; van Loosdrecht, M.C.; Lourenço, N.D. Stability of aerobic granules during long-term bioreactor operation. Biotechnol. Adv. 2018, 36, 228–246. [Google Scholar] [CrossRef]
- Ji, B.; Zhang, M.; Gu, J.; Ma, Y.; Liu, Y. A self-sustaining synergetic microalgal-bacterial granular sludge process towards energy-efficient and environmentally sustainable municipal wastewater treatment. Water Res. 2020, 179, 115884. [Google Scholar] [CrossRef]
- Zhang, M.; Ji, B.; Liu, Y. Microalgal-bacterial granular sludge process: A game changer of future municipal wastewater treatment? Sci. Total Environ. 2021, 752, 141957. [Google Scholar] [CrossRef]
- Huang, W.; Liu, D.; Huang, W.; Cai, W.; Zhang, Z.; Lei, Z. Achieving partial nitrification and high lipid production in an algal-bacterial granule system when treating low COD/NH4–N wastewater. Chemosphere 2020, 248, 126106. [Google Scholar] [CrossRef]
- Wang, J.; Lei, Z.; Wei, Y.; Wang, Q.; Tian, C.; Shimizu, K.; Zhang, Z.; Adachi, Y.; Lee, D. Behavior of algal-bacterial granular sludge in a novel closed photo-sequencing batch reactor under no external O2 supply. Bioresour. Technol. 2020, 318, 124190. [Google Scholar] [CrossRef]
- Wang, S.; Ji, B.; Cui, B.; Ma, Y.; Guo, D.; Liu, Y. Cadmium-effect on performance and symbiotic relationship of microalgal-bacterial granules. J. Clean. Prod. 2021, 282, 125383. [Google Scholar] [CrossRef]
- Wang, S.; Ji, B.; Zhang, M.; Ma, Y.; Gu, J.; Liu, Y. Defensive responses of microalgal-bacterial granules to tetracycline in municipal wastewater treatment. Bioresour. Technol. 2020, 312, 123605. [Google Scholar] [CrossRef]
- Huang, W.; Li, B.; Zhang, C.; Zhang, Z.; Lei, Z. Effect of algae growth on aerobic granulation and nutrients removal from synthetic wastewater by using sequencing batch reactors. Bioresour. Technol. 2015, 179, 187–192. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.; Yang, X.; Cai, W.; Lei, Z.; Shimizu, K.; Zhang, Z.; Utsumi, M.; Lee, D. Response of algal-bacterial granular system to low carbon wastewater: Focus on granular stability, nutrients removal and accumulation. Bioresour. Technol. 2018, 268, 221–229. [Google Scholar] [CrossRef]
- Meng, F.; Liu, D.; Huang, W.; Lei, Z.; Zhang, Z. Effect of salinity on granulation, performance and lipid accumulation of algal-bacterial granular sludge. Bioresour. Technol. Rep. 2019, 7, 100228. [Google Scholar] [CrossRef]
- Ji, B.; Zhang, M.; Wang, L.; Wang, S.; Liu, Y. Removal mechanisms of phosphorus in non-aerated microalgal-bacterial granular sludge process. Bioresour. Technol. 2020, 312, 123531. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gu, J.; Zhang, M. AB Processes: Towards Energy Self-Sufficient Municipal Wastewater Treatment; IWA Publishing: London, UK, 2019. [Google Scholar]
- Ji, B.; Yang, K.; Zhu, L.; Jiang, Y.; Wang, H.; Zhou, J.; Zhang, H. Aerobic denitrification: A review of important advances of the last 30 years. Biotechnol. Bioprocess Eng. 2015, 20, 643–651. [Google Scholar] [CrossRef]
- Ji, B.; Zhu, L.; Wang, S.; Liu, Y. Temperature-effect on the performance of non-aerated microalgal-bacterial granular sludge process in municipal wastewater treatment. J. Environ. Manag. 2021, 282, 111955. [Google Scholar] [CrossRef]
- Zhang, B.; Guo, Y.; Lens, P.N.L.; Zhang, Z.; Shi, W.; Cui, F.; Tay, J.H. Effect of light intensity on the characteristics of algal-bacterial granular sludge and the role of N-acyl-homoserine lactone in the granulation. Sci. Total Environ. 2019, 659, 372–383. [Google Scholar] [CrossRef]
- Lv, Y.; Wan, C.; Liu, X.; Zhang, Y.; Lee, D.; Tay, J. Freezing of aerobic granules for storage and subsequent recovery. J. Taiwan Inst. Chem. Eng. 2013, 44, 770–773. [Google Scholar] [CrossRef]
- Gao, D.; Yuan, X.; Liang, H. Reactivation performance of aerobic granules under different storage strategies. Water Res. 2012, 46, 3315–3322. [Google Scholar] [PubMed]
- Ji, B.; Zhu, L.; Wang, S.; Qin, H.; Ma, Y.; Liu, Y. A novel micro-ferrous dosing strategy for enhancing biological phosphorus removal from municipal wastewater. Sci. Total Environ. 2020, 704, 135453. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Lens, P.N.L.; Shi, W.; Zhang, R.; Zhang, Z.; Guo, Y.; Bao, X.; Cui, F. Enhancement of aerobic granulation and nutrient removal by an algal-bacterial consortium in a lab-scale photobioreactor. Chem. Eng. J. 2018, 334, 2373–2382. [Google Scholar] [CrossRef]
- Federation, Water Environmental, and APH Association. Standard Methods for the Examination of Water and Wastewater, 21th ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- He, Q.; Chen, L.; Zhang, S.; Chen, R.; Wang, H.; Zhang, W.; Song, J. Natural sunlight induced rapid formation of water-born algal-bacterial granules in an aerobic bacterial granular photo-sequencing batch reactor. J. Hazard. Mater. 2018, 359, 222–230. [Google Scholar] [CrossRef]
- Lee, C.S.; Lee, S.; Ko, S.; Oh, H.; Ahn, C. Effects of photoperiod on nutrient removal, biomass production, and algal-bacterial population dynamics in lab-scale photobioreactors treating municipal wastewater. Water Res. 2015, 68, 680–691. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Fadaei, H.; Hu, Z. Rapid evaluation of algal and cyanobacterial activities through specific oxygen production rate measurement. Ecol. Eng. 2014, 73, 439–445. [Google Scholar] [CrossRef]
- Sayara, T.; Khayat, S.; Saleh, J.; Abu-Khalaf, N.; van der Steen, P. Algal–bacterial symbiosis for nutrients removal from wastewater: The application of multivariate data analysis for process monitoring and control. Environ. Technol. Innov. 2021, 23, 101548. [Google Scholar] [CrossRef]
- Yang, J.; Li, Z.; Lu, L.; Fang, F.; Guo, J.; Ma, H. Model-based evaluation of algal-bacterial systems for sewage treatment. J. Water Process Eng. 2020, 38, 101568. [Google Scholar] [CrossRef]
- Boelee, N.C.; Temmink, H.; Janssen, M.; Buisman, C.J.N.; Wijffels, R.H. Balancing the organic load and light supply in symbiotic microalgal–bacterial biofilm reactors treating synthetic municipal wastewater. Ecol. Eng. 2014, 64, 213–221. [Google Scholar] [CrossRef]
- Abouhend, A.S.; Milferstedt, K.; Hamelin, J.; Ansari, A.A.; Butler, C.; Carbajal-González, B.I.; Park, C. Growth progression of oxygenic photogranules and its impact on bioactivity for aeration-free wastewater treatment. Environ. Sci. Technol. 2019, 54, 486–496. [Google Scholar] [CrossRef]
- Liu, L.; Fan, H.; Liu, Y.; Liu, C.; Huang, X. Development of algae-bacteria granular consortia in photo-sequencing batch reactor. Bioresour. Technol. 2017, 232, 64–71. [Google Scholar] [CrossRef]
- He, Q.; Zhang, W.; Zhang, S.; Zou, Z.; Wang, H. Performance and microbial population dynamics during stable operation and reactivation after extended idle conditions in an aerobic granular sequencing batch reactor. Bioresour. Technol. 2017, 238, 116–121. [Google Scholar] [CrossRef]
- Arcila, J.S.; Buitrón, G. Influence of solar irradiance levels on the formation of microalgae-bacteria aggregates for municipal wastewater treatment. Algal Res. 2017, 27, 190–197. [Google Scholar] [CrossRef]
- Zhang, L.; Long, B.; Wu, J.; Cheng, Y.; Zhang, B.; Zeng, Y.; Huang, S.; Zeng, M. Evolution of microbial community during dry storage and recovery of aerobic granular sludge. Heliyon 2019, 5, e3023. [Google Scholar] [CrossRef] [Green Version]
- Wan, C.; Zhang, Q.; Lee, D.; Wang, Y.; Li, J. Long-term storage of aerobic granules in liquid media: Viable but non-culturable status. Bioresour. Technol. 2014, 166, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.; Zhu, H.; Shutes, B.; Yan, B. Treatment of microcystin (MC-LR) and nutrients in eutrophic water by constructed wetlands: Performance and microbial community. Chemosphere 2021, 263, 128139. [Google Scholar] [CrossRef] [PubMed]
- Szabó, E.; Liébana, R.; Hermansson, M.; Modin, O.; Persson, F.; Wilén, B. Microbial Population Dynamics and Ecosystem Functions of Anoxic/Aerobic Granular Sludge in Sequencing Batch Reactors Operated at Different Organic Loading Rates. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Meng, F.; Xi, L.; Liu, D.; Huang, W.; Lei, Z.; Zhang, Z.; Huang, W. Effects of light intensity on oxygen distribution, lipid production and biological community of algal-bacterial granules in photo-sequencing batch reactors. Bioresour. Technol. 2019, 272, 473–481. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, W.; Ye, Y.; He, Q.; Zhang, S. Isolation and Characterization of Pseudoxanthomonas sp. Strain YP1 Capable of Denitrifying Phosphorus Removal (DPR). Geomicrobiol. J. 2018, 35, 537–543. [Google Scholar] [CrossRef]
- Wang, Q.; He, J. Complete nitrogen removal via simultaneous nitrification and denitrification by a novel phosphate accumulating Thauera sp. strain SND5. Water Res. 2020, 185, 116300. [Google Scholar] [CrossRef] [PubMed]
- Izadi, P.; Izadi, P.; Eldyasti, A. Enhancement of simultaneous nitrogen and phosphorus removal using intermittent aeration mechanism. J. Environ. Sci. 2021, 109, 1–14. [Google Scholar] [CrossRef]
- Ren, T.T.; Jin, C.Z.; Jin, F.J.; Li, T.; Kim, C.J.; Oh, H.M.; Lee, H.G.; Jin, L. Flavihumibacter profundi sp. nov., isolated from eutrophic freshwater sediment. J. Microbiol. 2018, 56, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sekiguchi, Y.; Hanada, S.; Hugenholtz, P.; Kim, H.; Kamagata, Y.; Nakamura, K. Gemmatimonas aurantiaca gen. nov., sp. nov., a gram-negative, aerobic, polyphosphate-accumulating micro-organism, the first cultured representative of the new bacterial phylum Gemmatimonadetes phyl. nov. Int. J. Syst. Evol. Microbiol. 2003, 53, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, Y. Turning food waste to energy and resources towards a great environmental and economic sustainability: An innovative integrated biological approach. Biotechnol. Adv. 2019, 37, 107414. [Google Scholar] [CrossRef] [PubMed]


| Key Functional Groups | Relative Abundance (%) | Reference | |||
|---|---|---|---|---|---|
| R0 | R1 | R2 | |||
| Heterotrophic Bacteria | Pseudomonas | 0.43 | 1.36 | 15.79 | [39] |
| Thauera | 0 | 0.02 | 7.01 | [40] | |
| Acinetobacter | 12.47 | 0.32 | 3.21 | [41] | |
| Flavihumibacter | 0.02 | 11.5 | 0.02 | [42] | |
| Pseudoxanthomonas | 0.04 | 6.84 | 1.04 | [39] | |
| Aquimonas | 0.01 | 0.15 | 7.9 | [34] | |
| Phosphorus-Accumulating Organisms | Pseudomonas | 0.43 | 1.36 | 15.79 | [39] |
| Thauera | 0 | 0.02 | 7.01 | [40] | |
| Acinetobacter | 12.47 | 0.32 | 3.21 | [41] | |
| Gemmatimonas | 0 | 2.39 | 0.01 | [43] | |
| Leptolyngbyales | 0.02 | 1.8 | 0.61 | [14] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Y.; Zhu, L.; Ji, B.; Fan, S.; Xiao, Y.; Ma, Y. Reactivation of Frozen Stored Microalgal-Bacterial Granular Sludge under Aeration and Non-Aeration Conditions. Water 2021, 13, 1974. https://doi.org/10.3390/w13141974
Shen Y, Zhu L, Ji B, Fan S, Xiao Y, Ma Y. Reactivation of Frozen Stored Microalgal-Bacterial Granular Sludge under Aeration and Non-Aeration Conditions. Water. 2021; 13(14):1974. https://doi.org/10.3390/w13141974
Chicago/Turabian StyleShen, Yao, Lin Zhu, Bin Ji, Siqi Fan, Yabin Xiao, and Yingqun Ma. 2021. "Reactivation of Frozen Stored Microalgal-Bacterial Granular Sludge under Aeration and Non-Aeration Conditions" Water 13, no. 14: 1974. https://doi.org/10.3390/w13141974





