Crustaceans in the Meiobenthos and Plankton of the Thermokarst Lakes and Polygonal Ponds in the Lena River Delta (Northern Yakutia, Russia): Species Composition and Factors Regulating Assemblage Structures
Abstract
1. Introduction
2. Materials and Methods
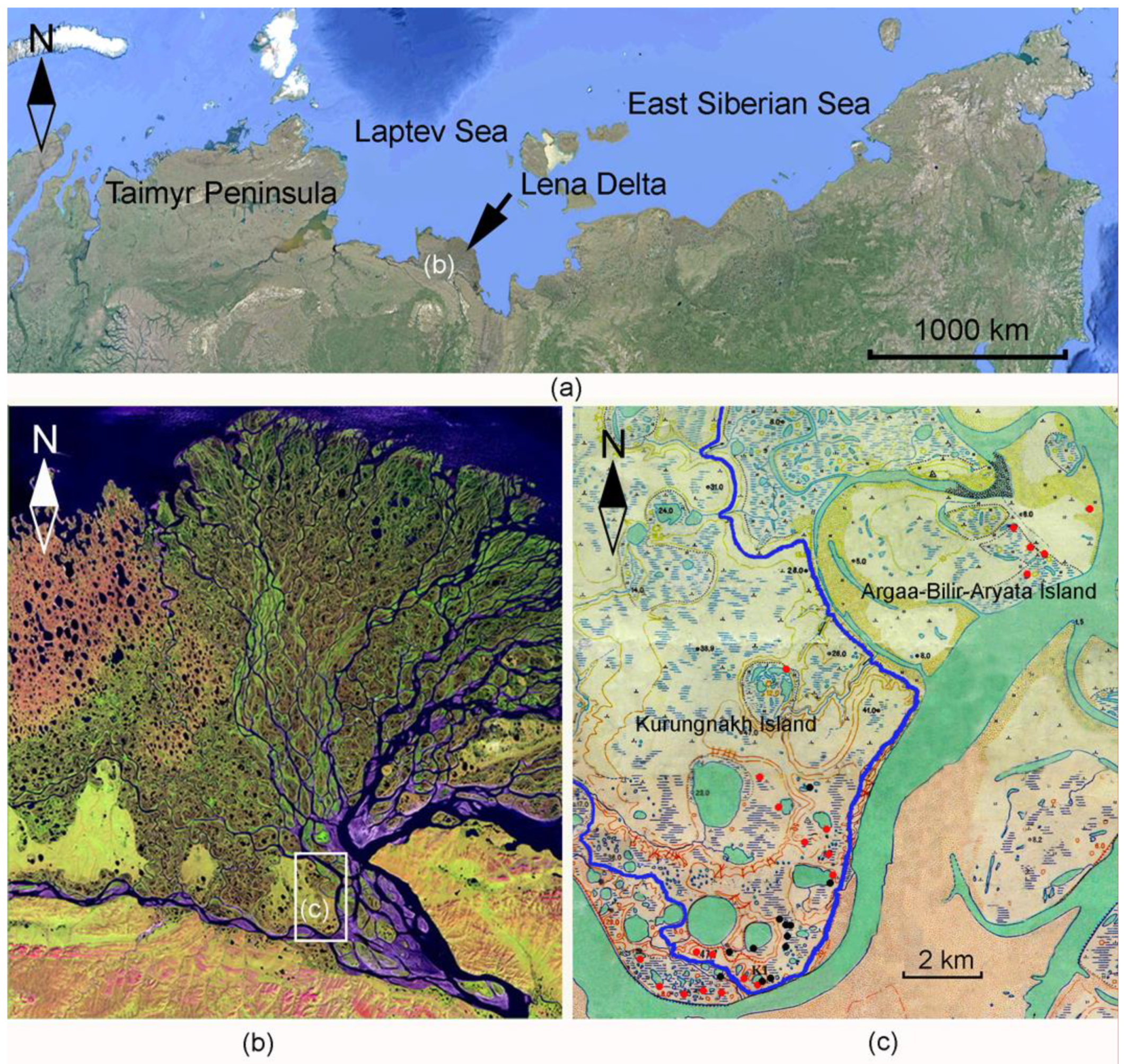
2.1. Studied Area
2.2. Types of Waterbodies
2.3. Sampling
2.4. Statistical Analysis
3. Results
3.1. Species Richness and Abundance
3.2. Patterns in Species Richness of Different Crustacean Groups
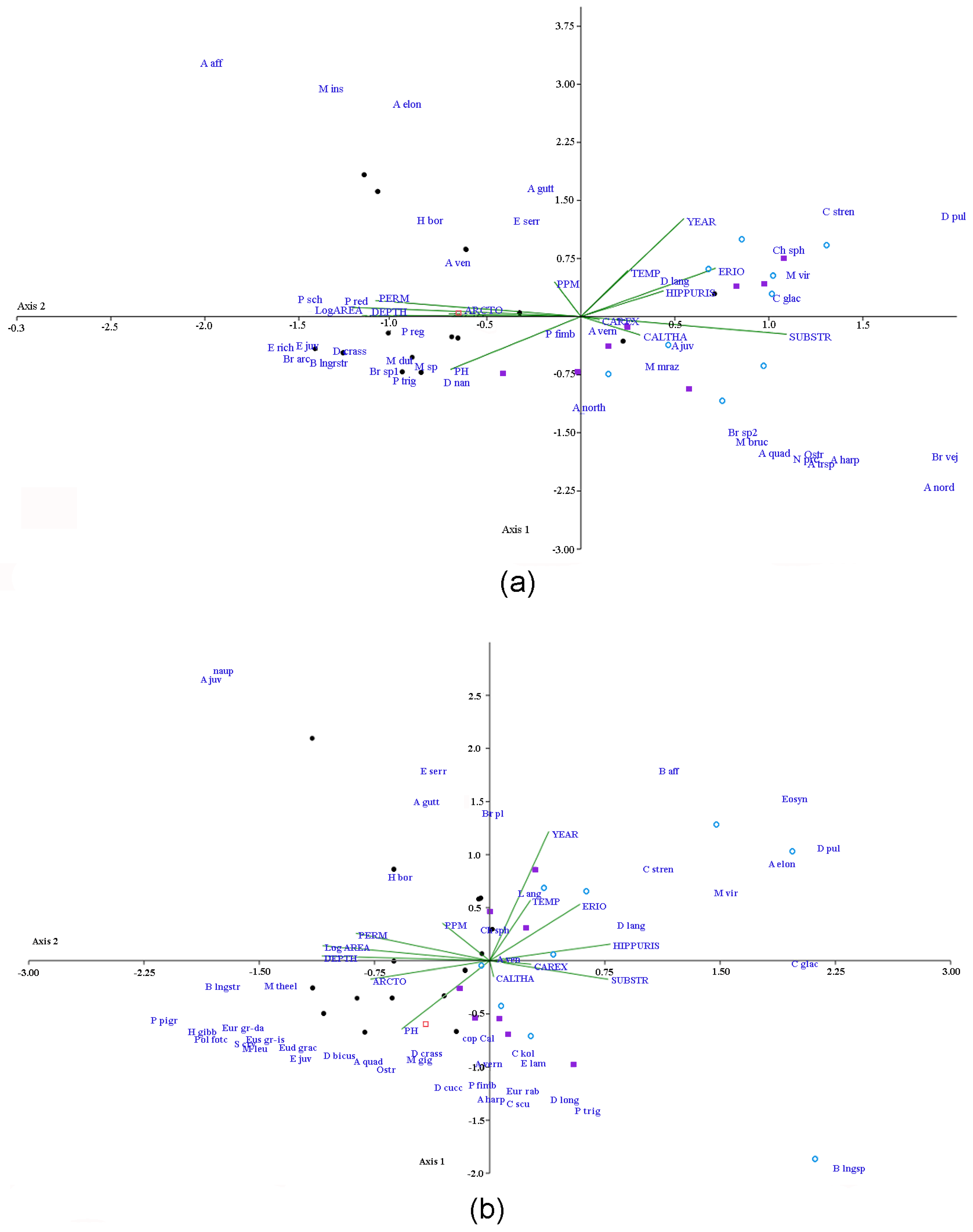
3.3. Variations in Species Composition and Assemblage Structure
4. Discussion
4.1. Fauna and New Records for the Region
4.2. Species Richness and Abundance
4.3. Crustacean Assemblage Structure and Regulating Factors
5. Conclusions
- In the present study, 59 crustacean species and taxa were found in the water bodies of the southern Lena River Delta: 39 Copepoda, 16 Cladocera, 2 Anostraca, 1 Amphipoda and Ostracoda (not identified). Five of these crustaceans (Al. quadrangularis, Pa. pigra, At. cf. trispinosa, Br. arcticus, and N. procerus) are new for the region.
- The species richness of crustaceans was higher in the thermokarst lakes than in the single polygonal ponds due to the freezing of small reservoirs during the winter. In contrast, the abundance of crustaceans was lower in the lakes than in the shallow ponds, which warms more in summer and lacks fish.
- Variations in the Cladocera assemblage structure are due to annual differences in the water temperature (connected to the year of research) and, to a lesser degree, hydrochemical features of the water bodies. The structure of non-harpacticoid Copepoda was generally determined by hydrochemical factors and less affected by the year of research. The main factor that was sensitive to Harpacticoida was the composition of macrophytes.
- Three types of crustacean assemblages characteristic of different stages of the development of tundra water bodies were distinguished. At the single polygonal pond stage, large species of Cladocera were mostly dominant in the plankton; at the complex polygonal pond stage, the most abundant species were Copepoda in the Cyclopoida family; and in thermokarst lakes, large Calanoida copepods prevailed. The meiobenthic crustacean fauna of single polygonal lakes consisted of several Harpacticoida and Cyclopoida species together with the cladoceran Ch. sphaericus, while the complex of species in thermokarst lakes included only harpacticoids.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bolshiyanov, D.Y.; Makarov, A.S.; Shnayder, V.; Shtof, G. Origins and Development of the Lena Delta; Arctic and Antarctic Research Institute: St. Petersburg, Russia, 2013; p. 268. (In Russian) [Google Scholar]
- Schwamborn, G.; Rachold, V.; Grigoriev, M.N. Late quaternary sedimentation history of the Lena Delta. Quat. Int. 2002, 89, 119–134. [Google Scholar] [CrossRef]
- Morgenstern, A.; Grosse, G.; Schirrmeister, L. Genetic, morphological, and statistical characterization of lakes in the permafrost-dominated Lena Delta. In Proceedings of the 9th International Conference on Permafrost, Inst. Northern Engineering, Univ, Fairbanks, AK, USA, 29 June–3 July 2008; pp. 1239–1244. [Google Scholar]
- Rylov, V.M. Materials on the fauna of freshwater copepods (Copepoda, Calanoida) of the Northern Siberia. Tr. Kom. Po Iizucheniyu Yakutsk. ASSR. 1928, 11, 1–32. (In Russian) [Google Scholar]
- Behning, A.L. About the Lena River plankton. Izv. Biol.-Geogr. Nauchno-Issledovatelskogo Inst. Pri Vostochno-Sib. Gos. Universitete. 1942, 6, 217–230. (In Russian) [Google Scholar]
- Pirozhnikov, P.L.; Shulga, E.L. The main features of the zooplankton of the lower reaches of the Lena River. Tr. Vsesoyuznogo Gidrobiol. Obshchestva. 1957, 8, 219–230. (In Russian) [Google Scholar]
- Martynov, A.V. On Freshwater Amphipoda and Isopoda of North Yakutia. Ezhegodnik Zool. Muzeya Akad. Nauk. SSSR 1931, 32, 523–540. (In Russian) [Google Scholar]
- Gukov, A.Y. Hydrobiologie of the Lena Mouth Area; Scientific World: Moscow, Russia, 2001; p. 285. (In Russian) [Google Scholar]
- Nigamatzyanova, G.R.; Frolova, L.A.; Chetverova, A.A.; Fedorova, I.V. Hydrobiological investigation of channels in the mouth region of thr Lena River. Uchenye Zap. Kazan. Universiteta. 2015, 4, 96–108. (In Russian) [Google Scholar]
- Nikanorov, A.M.; Bryzgalo, V.A.; Kosmenko, L.S.; Reshetnyak, O.S. Anthropogenic transformation of biocenosis structural organization in Lena River mouth area. Water Resour. 2011, 38, 306–314. [Google Scholar] [CrossRef]
- Serkina, R.A. Plankton and benthos of the Lena Delta and adjacent coastal waters. Tr. Yakutsk. Otd. Sib. Nauchno-Issledovatel’skogo Inst. Rybn. Hozyaistva 1969, 3, 188–196. (In Russian) [Google Scholar]
- Kerer, E.F. Zooplankton of the flood plain lakes in the lower reaches of the Lena River. Ucheniye Zapizki LGPI 1968, 311, 37–60. [Google Scholar]
- Abramova, E.N.; Zhulay, I.A. The occurrence of the new zooplankton species in the Lena River Delta. Proc. Zool. Inst. RAS 2016, 320, 473–487. [Google Scholar] [CrossRef]
- Abramova, E.; Vishnyakova, I. New data about zooplankton species composition and distribu-tion in the lakes of the Lena Delta. Ber. Polar Meerforsch. Russ. Ger. Coop. System Laptev Sea Exped. LENA 2008 2012, 642, 30–35. (In Russian) [Google Scholar]
- Abramova, E.; Vishnyakova, I.; Boike, J.; Abramova, A.; Solovyev, G.; Martynov, F. Structure of freshwater zooplankton communities from tundra waterbodies in the Lena River Delta, Russian Arctic, with a discussion on new records of glacial relict copepods. Polar Biol. 2017, 40, 1629–1643. [Google Scholar] [CrossRef]
- Frolova, L.A.; Nigamatzayanova, G.R. Structural and functional characteristics of zooplankton communities of thermokarst lakes of Somoilovskii island (Lena Delta, Sakha Republic (Yakutia)). Uchenye Zap. Kazan. Univ. 2019, 1, 158–172. [Google Scholar] [CrossRef]
- Nigamatzyanova, G.R.; Frolova, L.A.; Abramova, E.N. Zooplankton spatial distribution in thermokarst lake of The Lena River Delta (Republic of Sakha (Yakutia). Res. J. Pharmac. Biol. Chem. Scien. 2016, 7, 1288–1297. [Google Scholar]
- Aanes, R.; Sæther, B.E.; Smith, F.M.; Cooper, E.J.; Wookey, P.A.; Øritsland, N.A. The Arctic oscillation predicts effects of climate change in two trophic levels in a high arctic ecosystem. Ecol. Lett. 2002, 5, 445–453. [Google Scholar] [CrossRef]
- Culp, J.M.; Goedkoop, W.; Lento, J.; Christoffersen, K.S.; Frenzel, S.; Gubergsson, G.; Liljaniemi, P.; Sandy, S.; Svoboda, M.; Brittain, J.; et al. The Arctic Freshwater Biodiversity Monitoring Plan. CAFF Int. Secr. CAFF Monit. Ser. Rep. CAFF Int. Secr. Akureyri 2012, 7, 151. [Google Scholar]
- Novikov, A.A.; Abramova, E.N.; Sabirov, R.M. Faunistic complexes of copepods in water bodies of the lower reaches of the Lena River. In Abstracts of the International Conference Ecology of Aquatic Invertebrates, Dedicated to the 110-th Anniversary of the Birth of F.D.; Krylov, A.V., Ed.; Filigran: Yaroslavl, Russia, 2020; p. 110. [Google Scholar]
- Novikov, A.A.; Abramova, E.N.; Sabirov, R.M. Fauna of the freshwater Harpacticoida (Copepoda) Lena River Delta. Russ. J. Zool. 2021, 100, 264–274. (In Russian) [Google Scholar]
- Boike, J.; Kattenstroth, B.; Abramova, K.; Bornemann, N.; Chetverova, A.; Fedorova, I.; Fröb, K.; Grigoriev, M.; Grüber, M.; Kutzbach, L.; et al. Baseline characteristics of climate, permafrost and land cover from a new permafrost observatory in the Lena River Delta, Siberia (1998–2011). Biogeosciences 2013, 10, 2105–2128. [Google Scholar] [CrossRef]
- Ogorodnikov, N.N.; Fedorova, I.V. Calculation of the thawing depth of permafrost rocks on the example of Samoilovsky Island (Lena River Delta). In Abstracts of the International Conference Aquatic Environment and Natural-Territorial Complexes: Research, Use, Protection; Semenov, I.K., Ed.; KarSC RAS: Petrosavodsk, Russia, 2011; pp. 29–33. (In Russian) [Google Scholar]
- Schneider, J.; Grosse, G.; Wagner, D. Land cover classification or tundra environments in the Arctic Lena Delta based in Landsat ETM+ data and its application for upscaling of methane emissions. Remote Sens. Environ. 2009, 113, 380–391. [Google Scholar] [CrossRef]
- Morgenstern, A.; Röhr, C.; Grosse, G.; Grigoriev, M.N. The Lena River Delta—Inventory of Lakes and Geomorphological Terraces; Alfred Wegener Institute—Research Unit Potsdam, PANGAEA: Potsdam, Germany, 2011. [Google Scholar] [CrossRef]
- Shchukin, I.S. Fundamentals of Geomorphology, 2; MGU: Moscow, Russia, 1964; pp. 485–561. (In Russian) [Google Scholar]
- Vishnyakova, I.; Abramova, E. Zooplankton community structure in the polygonal lakes of the south part of the Lena Delta. In System of the Laptev Sea and the Adjacent Arctic Seas Modern and Past Environments; Kassens, H., Lisitzin, A.P., Thiede, J., Eds.; Moskow University Press: Moscow, Russia, 2009; pp. 223–236. (In Russian) [Google Scholar]
- Alekseev, V.R.; Tsalolikhin, S.Y. Guide of Freshwater Zooplankton and Zoobenthos of European Russia. Zooplankton, 1; KMK Press: Moscow, Russia, 2010; p. 495. (In Russian) [Google Scholar]
- Borutsky, E.V. Crustaceans Freshwater Harpacticoids. In Fauna of USSR Crustacea 3; AN USSR: Moscow-Leningrad, Russia, 1952; p. 425. (In Russian) [Google Scholar]
- Borutsky, E.V.; Stepanova, L.A.; Kos, M.S. Key to Identification of Calanoida from Fresh Waters; Nauka: St. Petersburg, Russia, 1991; p. 504. (In Russian) [Google Scholar]
- Brtek, J.; Mura, G. Revised key to families and genera of the Anostraca with notes on their geographical distribution. Crustaceana 2000, 73, 1037–1088. [Google Scholar] [CrossRef]
- Dussart, B.H.; Defaye, D. Répertoire mondial des Crustacés Copépodes des eaux Intérieures. Calanoïdes; CNRS Bordeaux: Paris, France, 1983; p. 224. [Google Scholar]
- Fefilova, E.B. Copepods (Copepoda). Fauna of the European North-East of Russia; KMK Scientific Press: Moscow, Russia, 2015; p. 319. [Google Scholar]
- Garibian, P.G.; Neretina, A.N.; Klimovsky, A.I.; Kotov, A.A. A new case of West-East differentiation of the freshwater fauna in Northern Eurasia: The Pleuroxus trigonellus species group (Crustacea: Cladocera: Chydoridae). Zootaxa 2018, 4532, 451–482. [Google Scholar] [CrossRef]
- Kotov, A.A.; Bekker, E.I. Cladocera: Family Eurycercidae (Branchiopoda: Cladocera: Anomopoda). In Dumont; Identification Guides to the Plankton and Benthos of Inland Waters; Dumont, H.J., Ed.; Backhuys Publishers & Weikersheim, Margraf Publishers: Leiden, The Netherlands, 2016; p. 89. [Google Scholar]
- Kotov, A.A.; Ishida, S.; Taylor, D.J. Revision of the genus Bosmina Baird, 1845 (Cladocera: Bosminidae), based on evidence from male morphological characters and molecular phylogenies. Zool. J. Linn. Soc. 2009, 156, 1–51. [Google Scholar] [CrossRef][Green Version]
- Lieder, U. Crustacea: Cladocera/Bosminidae. In Süsswasserfauna von Mitteleuropa; Fischer Verlag: Stuttgart, Germany, 1996; p. 80. [Google Scholar]
- Sinev, A.Y.u. A key to identifying cladocerans of the genus Alona (Anomopoda, Chydoridae) from the Russian European part and Siberia. Russ. J. Zool. 2002, 81, 926–939. [Google Scholar]
- Sinev, A.Y. Notes on morphology and taxonomic status of some North American species of the genus Alona Baird, 1843 (Cladocera: Anomopoda: Chydoridae). Hydrobiologia 2009, 175, 59–77. [Google Scholar] [CrossRef]
- Smirnov, N.N. Chydoridae of the world fauna. Fauna SSSR. In Rakoobraznye (Crustacea), 1; Smirnov, N.N., Ed.; Nauka: Leningrad, Russia, 1971; p. 529. (In Russian) [Google Scholar]
- Clarke, K.R.; Gorley, R.N. Primer, User Manual/Tutorial, 5th ed.; PRIMER-E Ltd.: Plymouth, UK, 2001; p. 91. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electronoca. 2001, 4, 9. [Google Scholar]
- Novichkova, A.A.; Kotov, A.A.; Chertoprud, E.S. Freshwater crustaceans of Bykovsky Peninsula and neighboring territory (Northern Yakutia, Russia). Arthropoda Sel. 2020, 29, 1–12. [Google Scholar] [CrossRef]
- Novichkova, A.A.; Chertoprud, E.S. Freshwater crustaceans (Cladocera, Copepoda) of Bering Island (Commander Islands, Russian Far East): Species richness and taxocenes structure. J. Nat. Hist. 2015, 50, 1357–1368. [Google Scholar] [CrossRef]
- Deasley, K.; Korosi, J.B.; Thienpont, J.R.; Kokelj, S.V.; Pisaric, M.F.J.; Smol, J.P. Investigating the response of Cladocera to a major saltwater intrusion event in an Arctic lake from the outer Mackenzie Delta (NT, Canada). J. Paleolimnol. 2012, 48, 287–296. [Google Scholar] [CrossRef]
- Fiers, F.; Kotwicki, L. The multiple faces of Nannopus palustris auct. reconsidered: Amorphological approach (Copepoda: Harpacticoida: Nannopodidae). Zool. Anz. 2013, 253, 36–65. [Google Scholar] [CrossRef]
- Novichkova, A.A.; Chertoprud, E.S. Cladocera and Copepoda of Shokalsky Island: New data from Northwest Siberia. J. Nat. Hist. 2017, 51, 1781–1793. [Google Scholar] [CrossRef]
- Ishida, T. Freshwater harpacticoids copepods of Hokkaido, northern Japan. Sci. Rep. Hokkaido Salmon Hatch. 1987, 47, 77–119. [Google Scholar]
- Sinev, A.Y.; Karabanov, D.P.; Kotov, A.A. A new North Eurasian species of the Alona affinis complex (Cladocera: Chydoridae). Zootaxa 2020, 4767, 115–137. [Google Scholar] [CrossRef] [PubMed]
- Hebert, P.D.; Hann, B.J. Patterns in the composition of arctic tundra pond microcrustacean communities. Can. J. Fish. Aquat. Scien. 1986, 43, 1416–1425. [Google Scholar] [CrossRef]
- Weider, L.J.; Hobaek, A. Glacial refugia, haplotype distributions, and clonal richness of the Daphnia pulex complex in arctic Canada. Mol. Ecol. 2003, 12, 463–473. [Google Scholar] [CrossRef]
- De Moor, F.; Ivanov, V. Global diversity of caddisflies (Trichoptera: Insecta) in freshwater. In Freshwater Animal Diversity Assessment; Balian, E.V., Leveque, C., Segers, H., Martens, K., Eds.; Springer: Cham, Switzerland; Berlin, Germany, 2007; pp. 393–407. [Google Scholar]
- Neretina, A.N.; Gololobova, M.A.; Neplyukhina, A.A.; Zharov, A.A.; Rogers, C.D.; Horne, D.J.; Protopopov, A.V.; Kotov, A.A. Crustacean remains from the Yuka mammoth raise questions about non-analogue freshwater communities in the Beringian region during the Pleistocene. Sci. Rep. 2020, 10, 859. [Google Scholar] [CrossRef]
- Zuykova, E.I.; Bochkarev, N.A.; Taylor, D.J.; Kotov, A.A. Unexpected endemism in the Daphnia longispina complex (Crustacea: Cladocera) in Southern Siberia. PLoS ONE 2019, 14, e0221527. [Google Scholar] [CrossRef]
- Taylor, D.J.; Connolly, S.J.; Kotov, A.A. The Intercontinental phylogeography of neustonic daphniids. Sci. Rep. 2020, 10, 1818. [Google Scholar] [CrossRef]
- Kotov, A.A.; Garibian, P.G.; Bekker, E.I.; Taylor, D.J.; Karabanov, D.P. A new species group from the Daphnia curvirostris species complex (Cladocera: Anomopoda) from the Eastern Palearctic: Taxonomy, phylogeny and phylogeography. Zool. J. Linn. Soc. 2021, 191, 772–822. [Google Scholar] [CrossRef]
- Korovchinsky, N.M. Cladocerans of the Order Ctenopoda of the World Fauna (Morphology, Systematics, Ecology, Biogeography); KMK Press: Moscow, Russia, 2004; p. 410. [Google Scholar]
- Vincent, W.F.; Hobbie, J.E. Ecology of Arctic Lakes and Rivers. In The Arctic: Environment, People, Policies; Harwood Academic Publishers: Reading, UK, 2019; pp. 197–231, Chapter 8. [Google Scholar]
- Dubovskaya, O.P.; Kotov, A.A.; Korovchinsky, N.M.; Smirnov, N.N.; Sinev, A.Y. Zooplankton of lakes in the spurs of the Putorana Plateau and adjacent territories (North of Krasnoyarsk Krai). Contemp. Probl. Ecol. 2010, 3, 401–434. [Google Scholar] [CrossRef]
- Novichkova, A.A.; Azovsky, A.I. Factors affecting regional diversity and distribution of freshwater microcrustaceans (Cladocera, Copepoda) at high latitudes. Polar Biol. 2017, 40, 185–198. [Google Scholar] [CrossRef]
- Sweetman, J.N.; Smol, J.P. A guide to the identification of cladoceran remains (Crustacea, Branchiopoda) in Alaskan lake sediments. Arch. Hydrobiol. 2006, 151, 353–394. [Google Scholar]
- Hicks, G.R.F.; Coull, B.C. The ecology of marine meiobenthic harpacticoid copepods. Oceanogr. Mar. Biol. Ann. Rev. 1983, 21, 67–175. [Google Scholar]
- Callaghan, T.V.; Jonasson, S. Arctic ecosystems and environmental change. Phil. Trans. Roy. Soc. Lond. 1995, 352, 259–276. [Google Scholar] [CrossRef]
- Kirpotin, S.N.; Polishchuk, Y.M.; Bryksina, N.A. Thermokarst lakes square dynamics of Western Siberian continuous and discontinuous permafrost under conditions of global warming. Tomsk State Univ. J. 2008, 311, 185–189. [Google Scholar]

| Taxa | Number of Waterbodies | Lakes | Complex Polygon Ponds | Single Polygon Ponds | Oxbow | |
|---|---|---|---|---|---|---|
| Class Malacostraca | ||||||
| Order Amphipoda | ||||||
| Family Crangonyctidae Bousfield, 1973 | ||||||
| Eosynurella jakutana (Martynov, 1931) | 10 | + | + | + | - | |
| Class Branchiopoda | ||||||
| Order Anostraca | ||||||
| Family Chirocephalidae Daday, 1910 | ||||||
| Polyartemia forcipata (Fischer, 1851) | 3 | + | - | - | - | |
| Family Branchinectidae Daday, 1910 | ||||||
| Branchinecta paludosa (O.F. Müller, 1788) | 16 | + | + | + | - | |
| Superorder Cladocera | ||||||
| Order Ctenopoda | ||||||
| Family Holopedidae G.O. Sars, 1865 | ||||||
| Holopedium gibberum Zaddach, 1855 | 1 | + | - | - | - | |
| Family Sididae Baird, 1850 | ||||||
| Sida crystallina (O.F. Müller, 1776) | 3 | + | - | - | - | |
| Order Anomopoda | ||||||
| Family Bosminidae Baird, 1845 | ||||||
| Bosmina longirostris (O.F. Müller, 1785) | 8 | + | + | - | + | |
| Bo. cf. longispina (O.F. Müller, 1785) | 4 | + | + | + | - | |
| Family Chydoridae Dybowski and Grochowski, 1894 | ||||||
| Acroperus harpae (Baird, 1834) | 7 | + | + | + | + | |
| Alona guttata G.O. Sars, 1862 | 10 | + | + | + | - | |
| * Al. quadrangularis (O.F. Müller, 1776) | 6 | + | + | + | + | |
| Alonopsis elongatus G.O. Sars, 1862 | 20 | + | + | + | - | |
| Biapertura affinis (Leydig, 1860) | 10 | + | + | + | - | |
| Chydorus cf. sphaericus (O.F. Müller, 1785) | 27 | + | + | + | - | |
| *Paralona pigra G.O. Sars, 1862 | 2 | + | - | - | - | |
| Pleuroxus cf. trigonellus (O.F. Müller, 1776) | 5 | + | - | + | - | |
| Family Daphniidae Straus, 1820 | ||||||
| Daphnia cucullata G.O. Sars, 1862 | 3 | + | + | - | - | |
| D. cf. longispina (O.F. Müller, 1776) | 15 | + | + | + | + | |
| D. cf. pulex Leydig, 1860 | 6 | + | + | + | - | |
| Family Eurycercidae Kurz, 1875 | ||||||
| Eurycercus lamellatus (O.F. Müller, 1776) | 5 | + | + | + | + | |
| Class Hexanauplia | ||||||
| Subclass Copepoda | ||||||
| Order Calanoida | ||||||
| Family Temoridae Giesbrecht, 1893 | ||||||
| Eurytemora gracilicauda Akatova, 1949 | 1 | - | - | - | + | |
| Eur. gracilis (G.O. Sars, 1863) | 6 | + | + | - | - | |
| Eur. cf. raboti Richard, 1897 | 1 | - | + | - | - | |
| Heterocope borealis (Fischer, 1851) | 24 | + | + | + | + | |
| Family Diaptomidae Baird, 1850 | ||||||
| Eudiaptomus graciloides (Lilljeborg, 1888) | 9 | + | + | + | - | |
| Leptodiaptomus angustilobius (G.O. Sars, 1898) | 17 | + | + | + | - | |
| Mixodiaptomus theeli (Lilljeborg in Guerne et Richard, 1889) | 16 | + | + | + | + | |
| Order Cyclopoida | ||||||
| Family Cyclopidae Rafinesque, 1815 | ||||||
| Acanthocyclops venustus (Norman and Scott, 1906) | 25 | + | + | + | + | |
| Ac. vernalis vernalis (Fischer, 1853) | 13 | + | + | + | - | |
| Cyclops scutifer scutifer G.O.Sars, 1863 | 13 | + | + | + | - | |
| C. kolensis Lilljeborg, 1901 | 19 | + | + | + | + | |
| C. cf. strenuus Fischer, 1851 | 16 | + | + | + | + | |
| Diacyclops bicuspidatus (Claus, 1857) | 6 | + | + | + | - | |
| Di. crassicaudis (G.O. Sars, 1863) | 8 | + | - | + | + | |
| Di. languidoides (Lilljeborg, 1901) | 13 | + | + | + | + | |
| Di. nanus (G.O. Sars, 1863) | 2 | + | + | - | - | |
| Eucyclops gr. serrulatus (Fischer, 1851) | 8 | + | + | + | - | |
| Megacyclops gigas gigas (Claus, 1857) | 10 | + | + | + | - | |
| Me. viridis viridis (Jurine, 1820) | 13 | + | + | + | - | |
| Mesocyclops leuckarti (Claus, 1857) | 2 | + | - | - | - | |
| Paracyclops fimbriatus (Fischer, 1853) | 7 | + | + | + | + | |
| Order Harpacticoida | ||||||
| Family Canthocamptidae Brady, 1880 | ||||||
| Attheyella dentata (Poggenpool, 1874) | 3 | + | + | + | - | |
| At. nordenskioldii (Lilljeborg, 1902) | 1 | - | - | + | - | |
| * At. cf. trispinosa (Brady, 1880) | 1 | - | - | + | - | |
| * Bryocamptus arcticus (Lilljeborg, 1902) | 1 | + | - | - | - | |
| Br. vejdovskyi (Mrazek, 1893) | 3 | + | - | + | - | |
| Br. sp. 1 | 2 | + | - | - | - | |
| Br. sp. 2 | 10 | + | + | + | + | |
| Canthocamptus glacialis (Lilljeborg, 1902) | 17 | + | + | + | - | |
| Epactophanes richardi Mrazek, 1893 | 2 | + | - | - | - | |
| Maraenobiotus brucei (Ricard, 1898) | 6 | + | + | + | - | |
| Moraria duthiei (Scott, 1896) | 18 | + | + | + | - | |
| Mo. insularis Fefilova, 2008 | 3 | + | - | - | - | |
| Mo. mrazeki Scott, 1903 | 20 | + | + | + | - | |
| M. sp. | 13 | + | + | - | - | |
| Pesceus reductus (M.S. Wilson, 1956) | 4 | + | + | - | - | |
| Pe. schmeili (Mrazek, 1893) | 4 | + | - | - | - | |
| Pe. cf. reggiae (M.S. Wilson, 1958) | 7 | + | + | - | - | |
| Family Nannopodidae Brady, 1880 | ||||||
| * Nannopus procerus Fiers and Kotwicki, 2013 | 1 | - | + | - | - | |
| Class Ostracoda | ||||||
| Ostracoda spp. | 7 | + | + | + | + | |
| Total species richness: | 54 | 43 | 39 | 16 | ||
| Group | AIC | P | Prop. | Cumul. |
|---|---|---|---|---|
| Total number of species | ||||
| +YEAR | 91.218 | 0.001 | 0.37953 | 0.37953 |
| +PH | 86.279 | 0.012 | 0.11767 | 0.4972 |
| Cladocera number of species | ||||
| +YEAR | 26.473 | 0.004 | 0.27455 | 0.27455 |
| +TEMP | 23.819 | 0.044 | 0.09541 | 0.36996 |
| +PH | 20.342 | 0.023 | 0.09637 | 0.46633 |
| Non-harpacticoid Copepoda number of species | ||||
| +YEAR | 59.766 | 0.018 | 0.15939 | 0.15939 |
| +PH | 54.685 | 0.009 | 0.16235 | 0.32173 |
| +DEPTH | 53.668 | 0.103 | 0.05925 | 0.38099 |
| +AREA | 51.12 | 0.057 | 0.0797 | 0.46069 |
| +PPM | 44.792 | 0.01 | 0.12029 | 0.58097 |
| +ISLAND | 43.354 | 0.109 | 0.041455 | 0.62243 |
| Harpacticoida number of species | ||||
| +MACR | 47.234 | 0.01 | 0.36699 | 0.36699 |
| +YEAR | 45.828 | 0.118 | 0.062083 | 0.42908 |
| Cladocera/Copepoda number of species | ||||
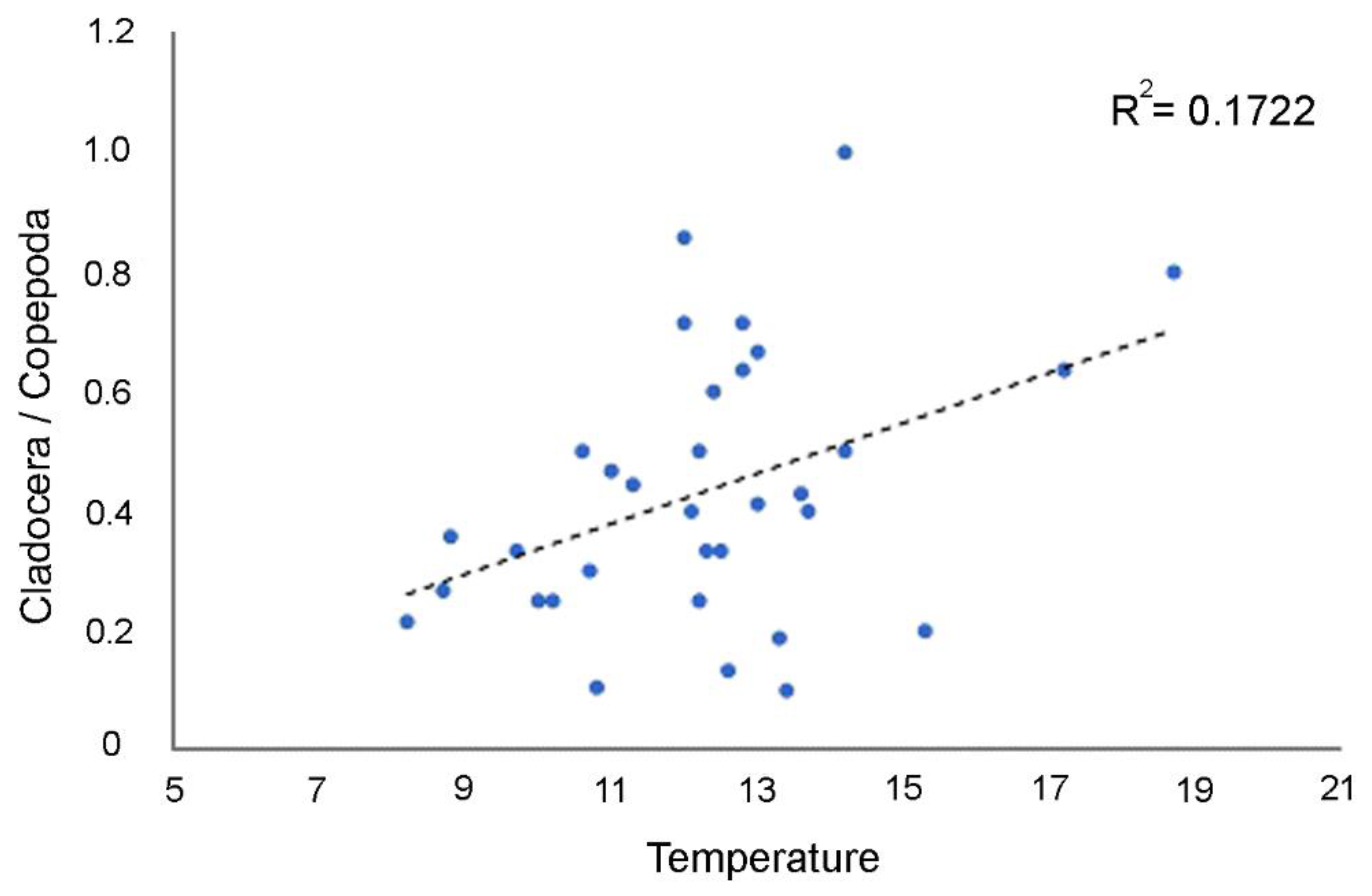
| +TEMP | −101.52 | 0.016 | 0.1722 | 0.1722 |
| Species Group A | Species Group B | Species Group C | |
|---|---|---|---|
| Wb Group A | 63 (46–81) | 31 (4–53) | 6 (0.3–15) |
| Wb Group B | 12 (0–31) | 76 (52–95) | 12 (2–48) |
| Wb Group C | 4 (0–17) | 38 (1–70) | 58 (27–99) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chertoprud, E.S.; Novichkova, A.A. Crustaceans in the Meiobenthos and Plankton of the Thermokarst Lakes and Polygonal Ponds in the Lena River Delta (Northern Yakutia, Russia): Species Composition and Factors Regulating Assemblage Structures. Water 2021, 13, 1936. https://doi.org/10.3390/w13141936
Chertoprud ES, Novichkova AA. Crustaceans in the Meiobenthos and Plankton of the Thermokarst Lakes and Polygonal Ponds in the Lena River Delta (Northern Yakutia, Russia): Species Composition and Factors Regulating Assemblage Structures. Water. 2021; 13(14):1936. https://doi.org/10.3390/w13141936
Chicago/Turabian StyleChertoprud, Elena S., and Anna A. Novichkova. 2021. "Crustaceans in the Meiobenthos and Plankton of the Thermokarst Lakes and Polygonal Ponds in the Lena River Delta (Northern Yakutia, Russia): Species Composition and Factors Regulating Assemblage Structures" Water 13, no. 14: 1936. https://doi.org/10.3390/w13141936
APA StyleChertoprud, E. S., & Novichkova, A. A. (2021). Crustaceans in the Meiobenthos and Plankton of the Thermokarst Lakes and Polygonal Ponds in the Lena River Delta (Northern Yakutia, Russia): Species Composition and Factors Regulating Assemblage Structures. Water, 13(14), 1936. https://doi.org/10.3390/w13141936







