Effects of Zeolitic Urea on Nitrogen Leaching (NH4-N and NO3-N) and Volatilization (NH3) in Spodosols and Alfisols
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Sampling
2.2. Zeolitic Fertilizer Formulations
2.3. Column Leaching Experiment
2.4. Statistical Analysis
3. Results
3.1. Effect on Ammonium Leaching Loss
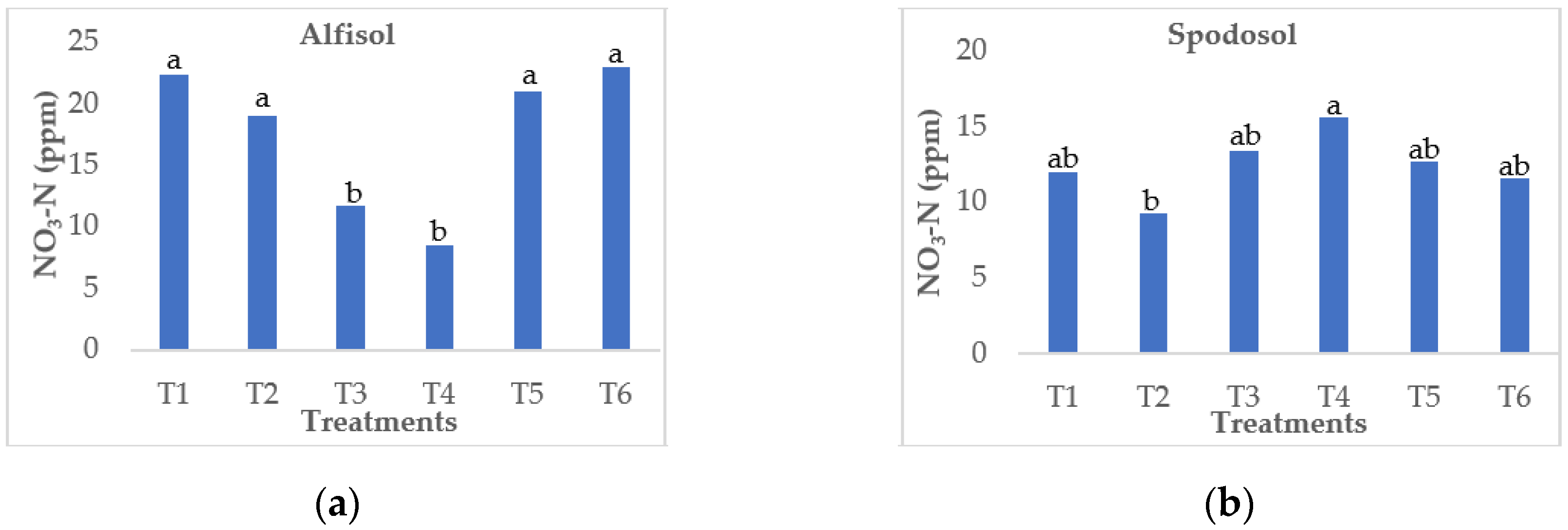
3.2. Effect on Nitrate Leaching Loss
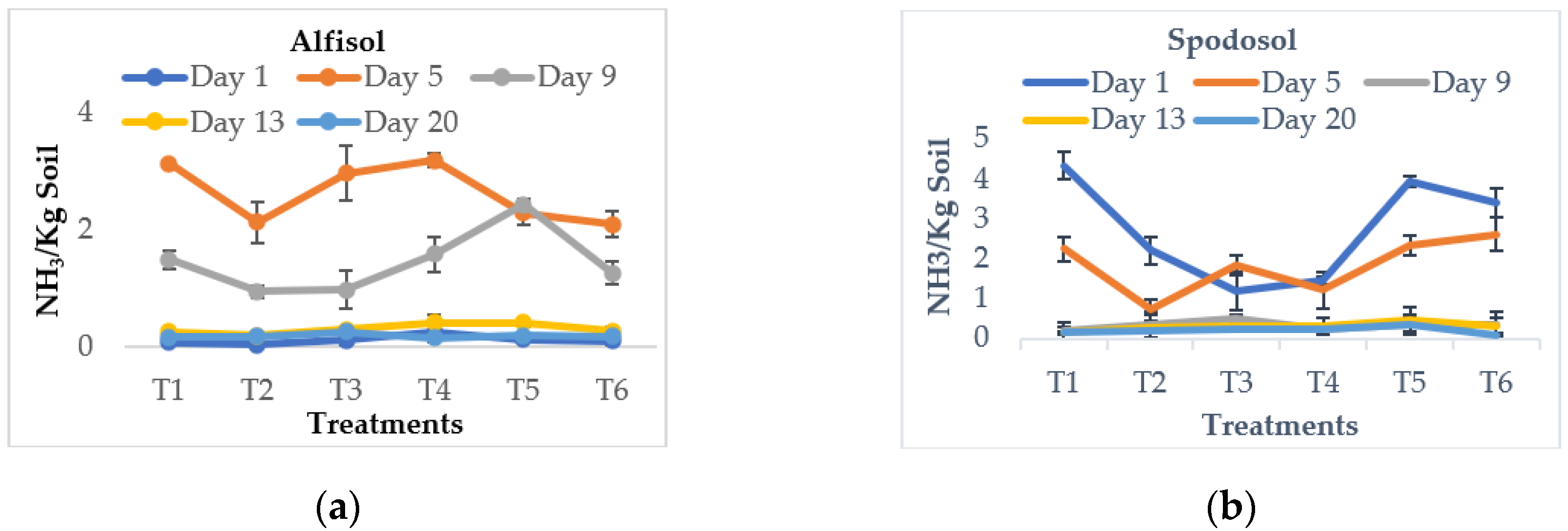
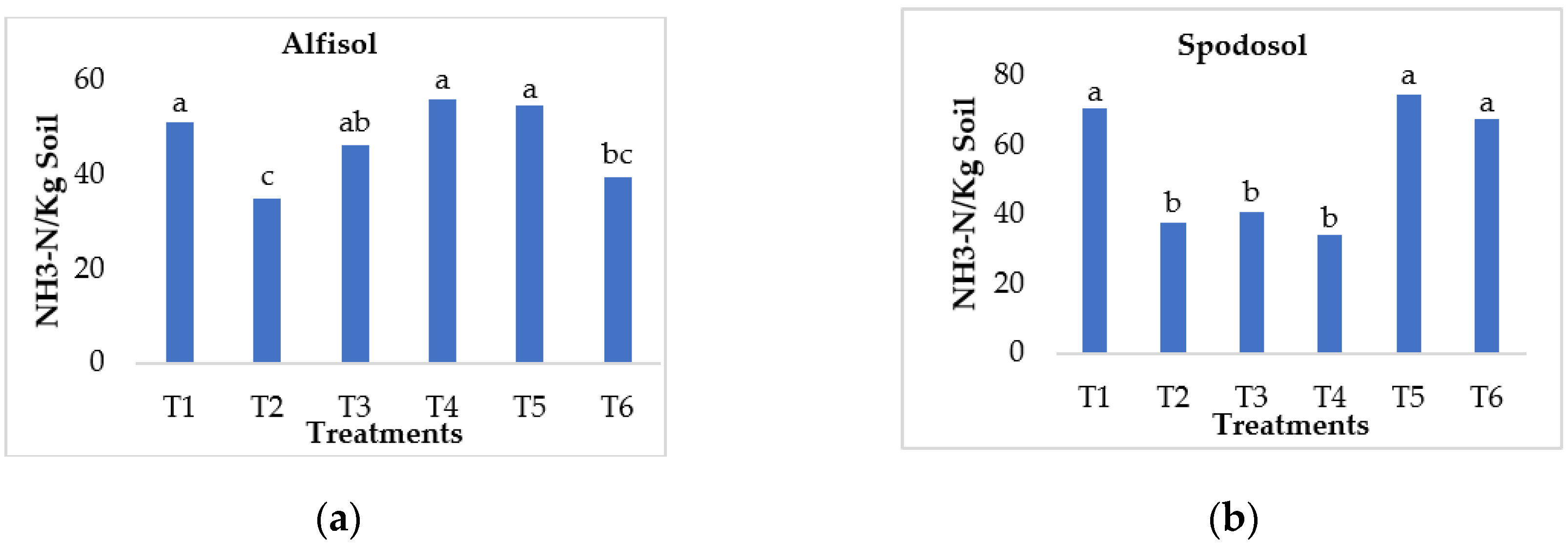
3.3. Effect on Ammonia Volatilization Loss
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, M.; Farooq, U. State of food security in Pakistan: Future challenges and coping strategies. Pak. Dev. Rev. 2010, 49, 903–923. [Google Scholar] [CrossRef] [Green Version]
- Hunter, M.C.; Smith, R.G.; Schipanski, M.E.; Atwood, L.W.; Mortensen, D.A. Agriculture in 2050: Recalibrating targets for sustainable intensification. Bio. Sci. 2017, 67, 386–391. [Google Scholar] [CrossRef] [Green Version]
- Sigurdarson, J.J.; Svane, S.; Karring, H. The molecular processes of urea hydrolysis in relation to ammonia emissions from agriculture. Rev. Environ. Sci. Biotechnol. 2018, 17, 241–258. [Google Scholar] [CrossRef] [Green Version]
- Heffer, P.; Prud, M. Fertilizer Outlook 2019–2023. In Proceedings of the 87th IFA Annual Conference, Montreal, QC, Canada, 11–13 June 2019; pp. 11–13. [Google Scholar]
- Glibert, P.M.; Harrison, J.; Heil, C.; Seitzinger, S. Scalating worldwide use of urea–A global change contributing to coastal eutrophication. Biogeochemistry 2006, 77, 441–463. [Google Scholar] [CrossRef]
- Wallace, A.J.; Armstrong, R.D.; Grace, P.R.; Scheer, C.; Partington, D.L. Nitrogen use efficiency of 15N urea applied to wheat based on fertilizer timing and use of inhibitors. Nutr. Cycl. Agroecosyst. 2020, 116, 41–56. [Google Scholar] [CrossRef] [Green Version]
- Salazar, F.; Martínez, J.; Lagos, M.; Alfaro, M.; Misselbrook, T. Ammonia emissions from urea application to permanent pasture on a volcanic soil. Atmos. Environ. 2012, 61, 395–399. [Google Scholar] [CrossRef]
- Cantarella, H.; Trivelin, P.C.O.; Contin, T.L.M.; Dias, F.L.F.; Rossetto, R.; Marcelino, R.; Coimbra, R.B.; Quaggio, J.A. Ammonia volatilization from urease inhibitor-treated urea applied to sugarcane trash blankets. Sci. Agric. 2008, 65, 397–401. [Google Scholar] [CrossRef] [Green Version]
- Sahu, S.K.; Samant, P.K. Nitrogen loss from rice soils in Orissa. Orissa Rev. Ind. 2006, 1–3. [Google Scholar]
- Ti, C.; Xia, L.; Chang, S.X.; Yan, X. Potential for mitigating global agricultural ammonia emission: A meta-analysis. Environ. Pollut. 2019, 245, 141–148. [Google Scholar] [CrossRef]
- Smith, W.; Grant, B.; Qi, Z.; He, W.; VanderZaag, A.; Drury, C.F.; Vergè, X.; Balde, H.; Gordon, R.; Helmers, M.J. Assessing the impacts of climate variability on fertilizer management decisions for reducing nitrogen losses from corn silage production. J. Environ. Qual. 2019, 48, 1006–1015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atwood, J.L.; Davies, J.E.D.; MacNicol, D.D. Inclusion Compounds: Physical Properties and Applications; Academic Press: London, UK, 1984; Volume 3, pp. 260–300. [Google Scholar]
- Lebedynets, M.; Sprynskyy, M.; Sakhnyuk, I.; Zbytniewski, R.; Golembiewski, R.; Buszewski, B. Adsorption of ammonium ions onto a natural zeolite: Transcarpathian Clinoptilolite. Ads. Sci. Tech. 2004, 22, 732–741. [Google Scholar] [CrossRef] [Green Version]
- Bernardi, A.C.C.; Polidoro, J.C.; Monte, M.B.M.; Pereira, E.I.; Oliveira, C.R.; Ramesh, K. Enhancing nutrient use efficiency using zeolites minerals. A review. Adv. Chem. Eng. Sci. 2016, 6, 295–304. [Google Scholar] [CrossRef]
- Karol, M. Natural vs. synthetic zeolites. Crystals 2020, 10, 622. [Google Scholar] [CrossRef]
- Jakkula, V.S.; Wani, S.P. Zeolites: Potential soil amendments for improving nutrient and water use efficiency and agriculture productivity. Sci. Revs. Chem. Commun. 2018, 8, 1–15. [Google Scholar]
- Eslami, M.; Khorassani, R.; Fotovat, A.; Halajnia, A. NH4+-K+ co-loaded clinoptilolite as a binary fertilizer. Arch. Agron. Soil Sci. 2019, 66, 33–45. [Google Scholar] [CrossRef]
- Mumpton, F.A. Using zeolites in agriculture. In Innovative Biological Technologies for Lesser Developed Countries; Workshop proceedings ; US Congress, Office of Technology Assessment: Washington, DC, USA, 1985; pp. 125–158. [Google Scholar]
- Sloan, J.J.; Ampim, P.A.Y.; Jaber, F. Phosphorus and nitrogen adsorption by clinoptilolite zeolite coated with iron-oxide. Commun. Soil Sci. Plant Anal. 2019, 50, 2669–2681. [Google Scholar] [CrossRef]
- Gholamhoseini, M.; AghaAlikhani, M.; Dolatabadian, A.; Khodaei-Joghan, A.; Zakikhani, H. Decreasing nitrogen leaching and increasing canola forage yield in a sandy soil by application of natural zeolite. Agron. J. 2012, 104, 1467–1475. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, O.; Aminuddin, H.; Husni, M. Reducing ammonia loss from urea and improving soil exchangeable ammonium retention through mixing triple super-phosphate, humic acid and zeolite. Soil Use Manag. 2006, 22, 315–319. [Google Scholar] [CrossRef]
- Palanivell, P.; Ahmed, O.H.; Ab Majid, N.M. Minimizing ammonia volatilization from urea, improving lowland rice (cv. MR219) seed germination, plant growth variables, nutrient uptake, and nutrient recovery using clinoptilolite zeolite. Arch. Agron. Soil Sci. 2016, 62, 708–724. [Google Scholar] [CrossRef]
- Stanley-Wood, N.G.; Lines, R.W. Particle Size Analysis; Royal Society of Chemistry: London, UK, 2007. [Google Scholar]
- Gillman, G.P.; Bruce, R.C.; Davey, B.G.; Kimble, J.M.; Searle, P.L.; Skjemstad, J.O. A comparison of methods used for determination of cation exchange capacity. Comm. Soil Sci. Plant Anal. 1983, 14, 1005–1014. [Google Scholar] [CrossRef]
- Chang, H.J. Method of Preparing a Slow Release Fertilizer. U.S. Patent 5695542, 9 December 1997. [Google Scholar]
- He, Z.L.; Alva, A.K.; Calvert, D.V.; Banks, D.J. Ammonia volatilization from different fertilizer sources and effects of temperature and soil pH. Soil Sci. 1999, 164, 750–758. [Google Scholar] [CrossRef]
- Astuti, D.W.; Mudasir; Aprilita, N.H. Preparation and characterization adsorbent based on zeolite from Klaten, Central Java, Indonesia. J. Phys. Conf. Ser. 2019, 1–6. [Google Scholar] [CrossRef]
- Sarkar, B.; Naidu, R. Nutrient and water use efficiency in soil: The influence of geological mineral amendments. In Nutrient Use Efficiency: From Basics to Advances; Springer: Berlin, Germany, 2015; pp. 29–44. [Google Scholar]
- Wasielewski, S.; Eduard, R.; Ralf, M.; Heidrun, S. Evaluation of different clinoptilolite zeolites as adsorbent for ammonium removal from highly concentrated synthetic wastewater. Water 2018, 10, 584. [Google Scholar] [CrossRef] [Green Version]
- Rech, I.; Polidoro, J.C.; Pavinato, P.S. Additives incorporated into urea to reduce nitrogen losses after application to the soil. Pesq. Agropec. Bras. 2017, 52, 194–204. [Google Scholar] [CrossRef] [Green Version]
- Swingmann, N.; Singh, B.; Mackinnon, I.D.R.; Gilkes, R.J. Zeolite from alkali modified kaolin increases NH4+ retention by sandy soil: Column experiments. Appl. Clay Sci. 2009, 46, 7–12. [Google Scholar] [CrossRef]
- Colombani, N.; Mastrocicco, M.; Giuseppe, D.D.; Faccini, B.; Coltorti, M. Batch and column experiments on nutrient leaching in soils amended with Italian natural zeolitites. Catena 2015, 127, 64–71. [Google Scholar] [CrossRef]
- Mahboubeh, E.; Khorassani, R.; Coltorti, M.; Malferrari, D.; Faccini, B.; Ferretti, G.; Giuseppe, D.D.; Fotovat, A.; Halajnia, A. Leaching behaviour of a sandy soil amended with natural and NH4-N and K+ saturated clinoptilolite and chabazite. Arch. Agron. Soil Sci. 2018, 64, 1142–1151. [Google Scholar]
- Omar, L.; Ahmed, O.H.; Majid, N.M.A. Improving ammonium and nitrate release from urea using clinoptilolite zeolite and compost produced from agricultural wastes. Sci. World J. 2015, 2005, 1–12. [Google Scholar] [CrossRef]
- Aghaalikhani, M.; Gholamhoseini, M.; Dolatabadian, A.; Joghan, A.K.; Asilan, K.S. Zeolite influences on nitrate leaching, nitrogen-use efficiency, yield and yield components of canola in sandy soil. Arch. Agron. Soil Sci. 2012, 58, 1149–1169. [Google Scholar] [CrossRef]
- Ahmed, O.H.; Yap, C.H.B.; Muhamad, A.M.N. Minimizing ammonia loss from urea through mixing with zeolite and acid sulphate soil. Int. J. Phys. Sci. 2010, 5, 2198–2202. [Google Scholar]
- Carlos, G.W.; Breda, F.A.; Zonta, E.; Lima, E.; Polidoro, J.C.; Balieiro, F.C.; Bernardi, A.C.C. Ammonia volatilization from urea with natural zeolite. Pesq. Agropec. Bras. 2012, 47, 466–470. [Google Scholar]
- Campana, M.; Alves, A.C.; Oliveira, P.P.A.; Bernardi, A.C.C.; Santos, E.A.; Herling, V.R.; Morais, J.P.G.; Junior, W.B. Ammonia volatilization from exposed soil and Tanzania grass pasture fertilized with urea and zeolite mixture. Commun. Soil Sci. Plant Anal. 2015, 46, 1024–1033. [Google Scholar] [CrossRef]
- He, Z.L.; Calvert, D.V.; Alva, A.K.; Li, Y.C.; Banks, D.J. Clinoptilolite zeolite and cellulose amendments to reduce ammonia volatilization in a calcareous sandy soil. Plant Soil 2002, 247, 253–260. [Google Scholar] [CrossRef]
- Bernardi, A.C.C.; Souza, G.B.; Polidoro, J.C.; Paiva, P.R.P.; Monte, M.B.d.M. Yield quality components and nitrogen levels of silage corn fertilized with urea and zeolite. Commun. Soil Sci. Plant Anal. 2011, 42, 1–10. [Google Scholar] [CrossRef]
- Faria, L.A.; Nascimento, C.A.C.; Vitti, G.C.; Luz, P.H.C.; Guedes, E.M.S. Loss of ammonia from nitrogen fertilizers applied to maize and soybean straw. R. Bras. Ci. Solo. 2013, 37, 969–975. [Google Scholar] [CrossRef] [Green Version]
- Dubey, A.; Mailapalli, D.R. Zeolite coated urea fertilizer using different binders: Fabrication, material properties and nitrogen release studies. Environ. Technol. Innov. 2019, 16, 100452. [Google Scholar] [CrossRef]
- Tarkalson, D.D.; Ippolito, J.A. Clinoptilolite zeolite influence on inorganic nitrogen in silt loam and sandy agricultural soils. Soil Sci. 2010, 175, 357–362. [Google Scholar] [CrossRef] [Green Version]
- Mehrab, N.; Chorom, M.; Hojati, S. Effect of raw and NH4-enriched zeolite on nitrogen uptake by wheat and nitrogen leaching in soils with different textures. Commun. Soil Sci. Plant Anal. 2016, 47, 1306–1316. [Google Scholar] [CrossRef]
- Nieder, R.; Dinesh, K.B.; Heinrich, W.S. Fixation and defixation of ammonium in soils: A review. Biol. Fertil. Soils 2011, 47, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.Z.; Huang, S.H.; Wan, D.J.; Huang, Y.X.; Zhou, W.J.; Zou, Y.B. Fixed ammonium content and maximum capacity of ammonium fixation in major types of tilled soils in Hunan Province, China. Agric. Sci. China 2007, 6, 466–474. [Google Scholar] [CrossRef]
- Malekian, R.; Abedi-Koupai, J.; Eslamian, S.S. Influences of clinoptilolite and surfactant-modified clinoptilolite zeolite on nitrate leaching and plant growth. J. Haz. Mat. 2011, 185, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Sahrawat, K.L. Factors affecting nitrification in soils. Commun. Soil Sci. Plant Anal. 2008, 39, 1436–1446. [Google Scholar] [CrossRef] [Green Version]
- Jadon, P.; Selladurai, R.; Yadav, S.S.; Coumar, M.V.; Dotaniya, M.L.; Singh, A.K.; Bhadouriya, J.; Kundu, S. Volatilization and leaching losses of nitrogen from different coated urea fertilizers. J. Soil Sci. Plant Nutr. 2018, 18, 1036–1047. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.; Xiaa, G.; Heb, Z.; Wua, Q.; Zhenga, J.; Lia, Y.; Wanga, Y.; Chena, T.; Chia, D. Zeolite amendment coupled with alternate wetting and drying to reduce nitrogen loss and enhance rice production. Field Crop. Res. 2019, 235, 95–103. [Google Scholar] [CrossRef]
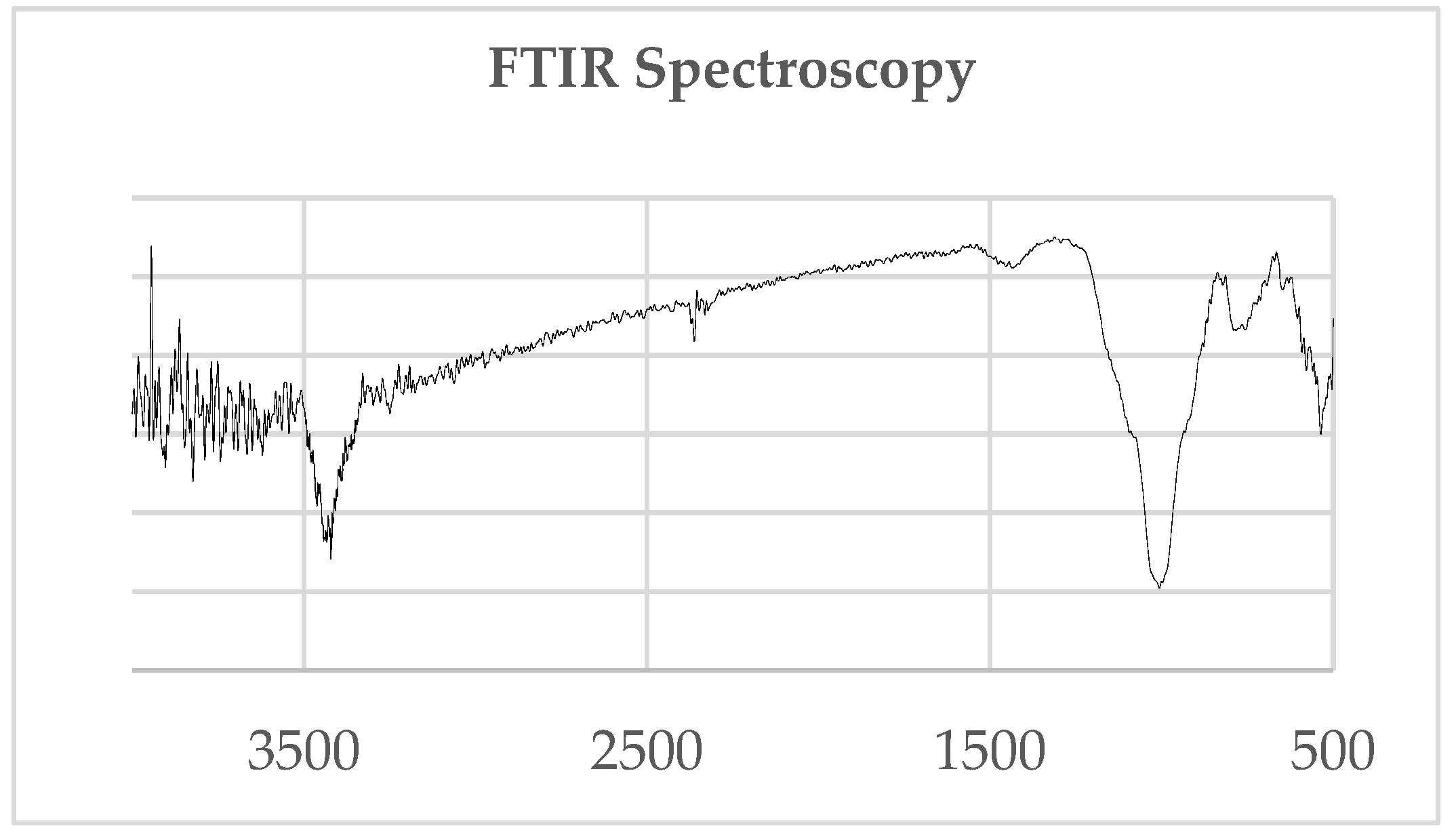
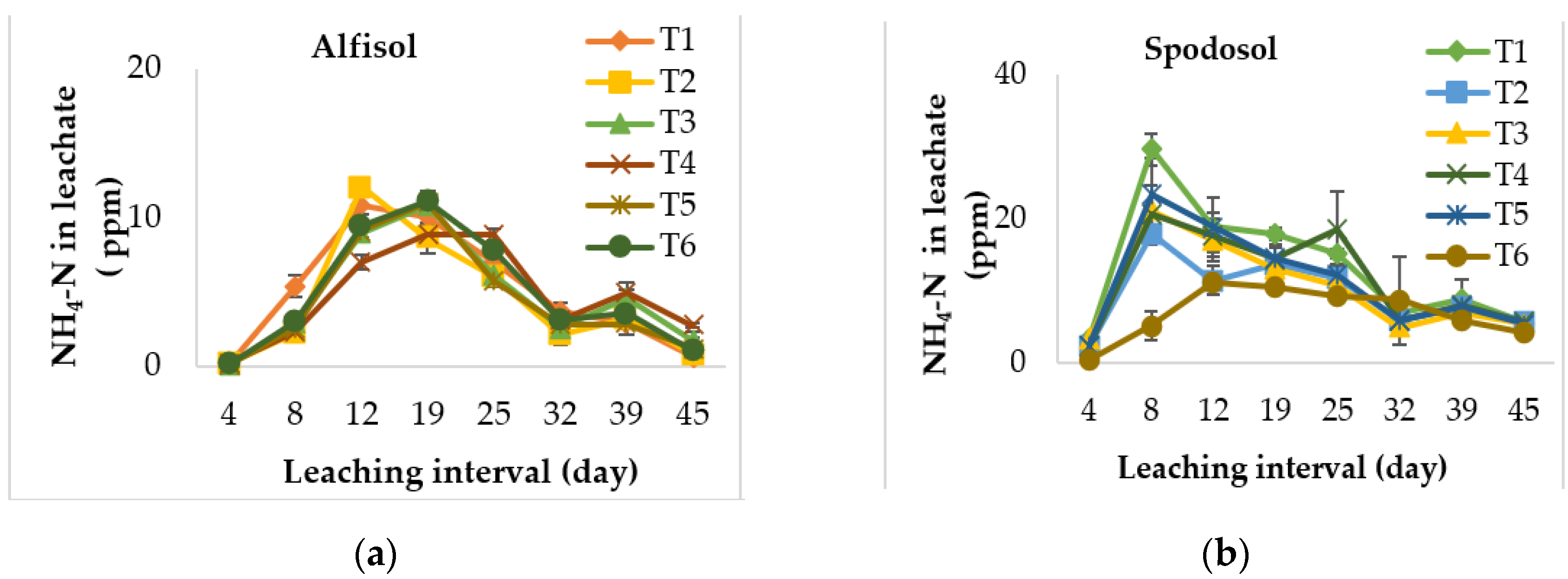
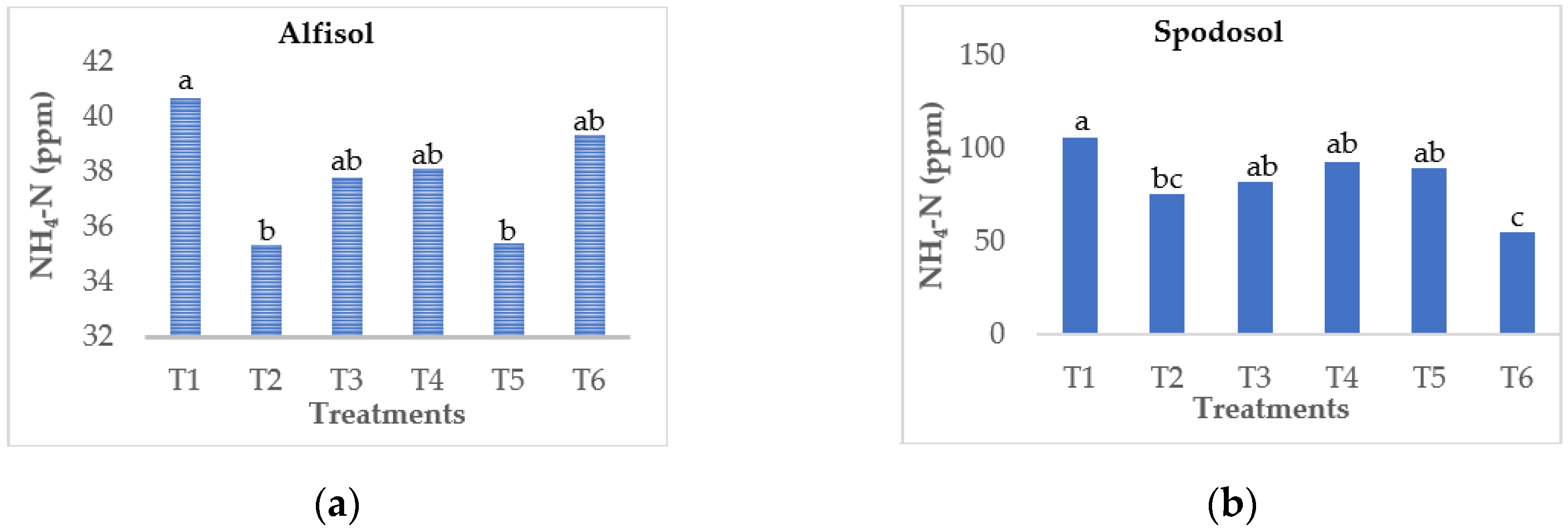
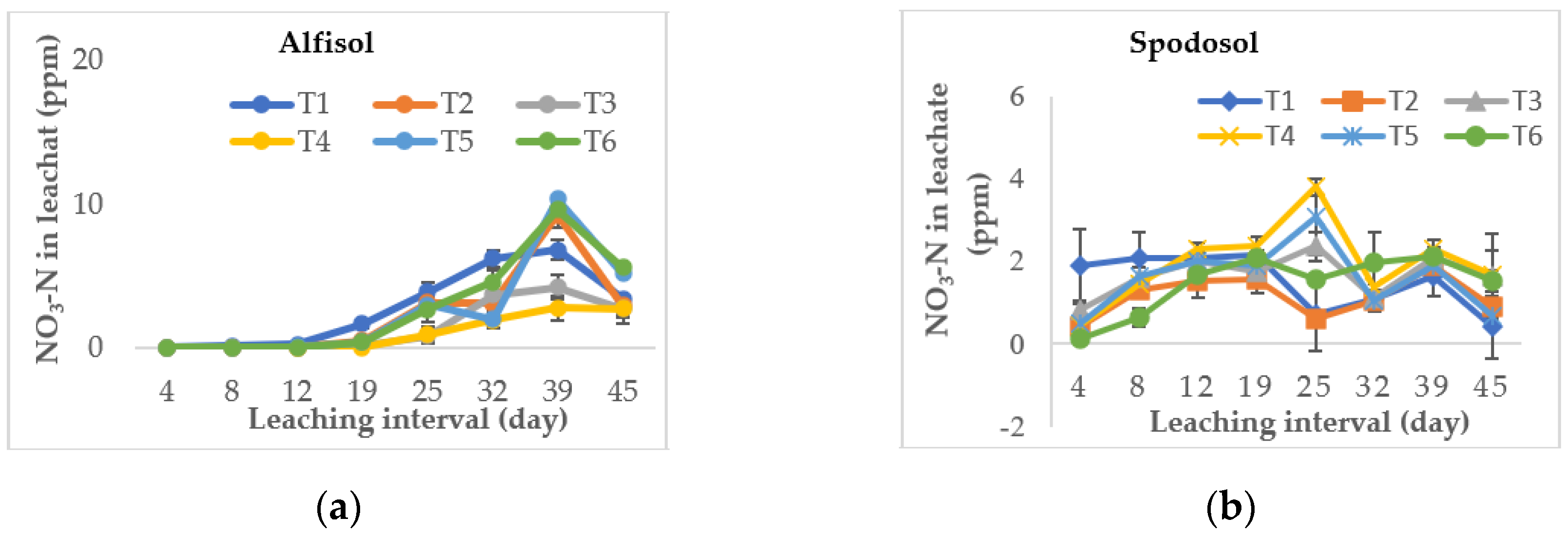
| Soil Type | Alfisol (Riviera Series) | Spodosol (Ankona Series) |
|---|---|---|
| Sand (%) | 83 | 92 |
| Silt (%) | 5 | 3 |
| Clay (%) | 12 | 5 |
| Texture | Loamy | Sandy |
| pH | 6.1 | 5.2 |
| CEC (meq/100 g) | 15.1 | 5.7 |
| Organic Matter (g kg−1) | 18.2 | 8.3 |
| Type of Fertilizer Formulation | Nitrogen Content (%) |
|---|---|
| Urea | 46 |
| Zeo-urea (1:1) | 21.60 |
| Zeo-urea (2:1) | 14.13 |
| Zeo-urea (3:1) | 10.80 |
| Zeo-urea (1:2) | 29.20 |
| Zeo-urea (1:3) | 34.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, A.; Ijaz, S.S.; He, Z. Effects of Zeolitic Urea on Nitrogen Leaching (NH4-N and NO3-N) and Volatilization (NH3) in Spodosols and Alfisols. Water 2021, 13, 1921. https://doi.org/10.3390/w13141921
Ahmad A, Ijaz SS, He Z. Effects of Zeolitic Urea on Nitrogen Leaching (NH4-N and NO3-N) and Volatilization (NH3) in Spodosols and Alfisols. Water. 2021; 13(14):1921. https://doi.org/10.3390/w13141921
Chicago/Turabian StyleAhmad, Ayaz, Shahzada Sohail Ijaz, and Zhenli He. 2021. "Effects of Zeolitic Urea on Nitrogen Leaching (NH4-N and NO3-N) and Volatilization (NH3) in Spodosols and Alfisols" Water 13, no. 14: 1921. https://doi.org/10.3390/w13141921







