E. coli CB390 as an Indicator of Total Coliphages for Microbiological Assessment of Lime and Drying Bed Treated Sludge
Abstract
:1. Introduction
2. Materials and Methods
2.1. WWTPs Location and Treatment Plant Description
2.2. Sampling
2.3. Microbiological Analysis of Sludge and Wastewater Samples
2.4. Thermotolerant Coliform (TTC)
2.5. Salmonella spp.
2.6. Helminth Eggs (HE)
2.7. Total Coliforms (TC) and E. coli
2.8. Somatic Coliphages, F-Specific Coliphages and CB390 Phage Analysis
2.9. Chiquinquirá WWTP Sludge Chemical Analysis
2.10. Evaluation of Microbiological Indicators in Wastewater Samples
2.11. Data Analysis
3. Results
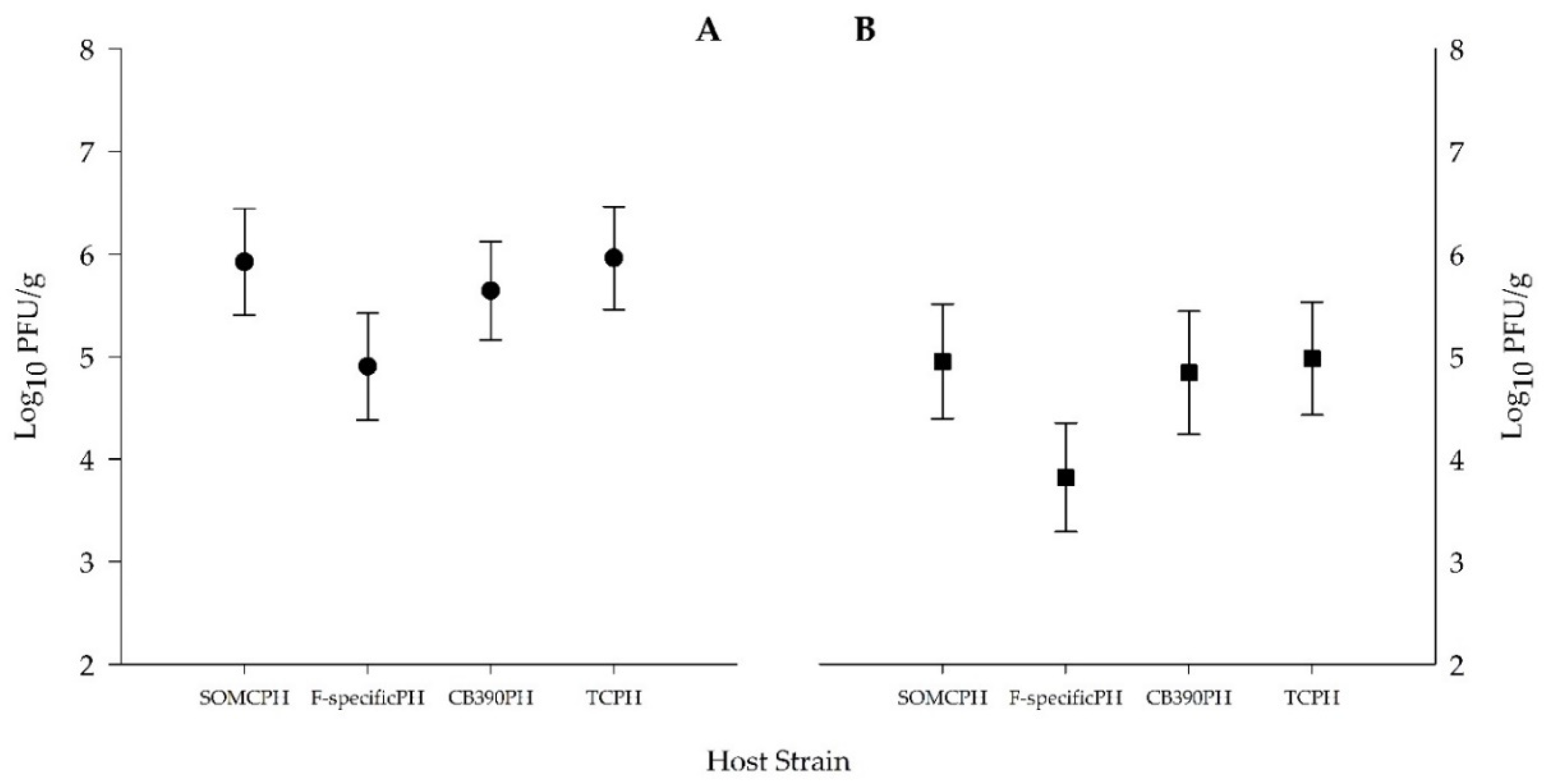
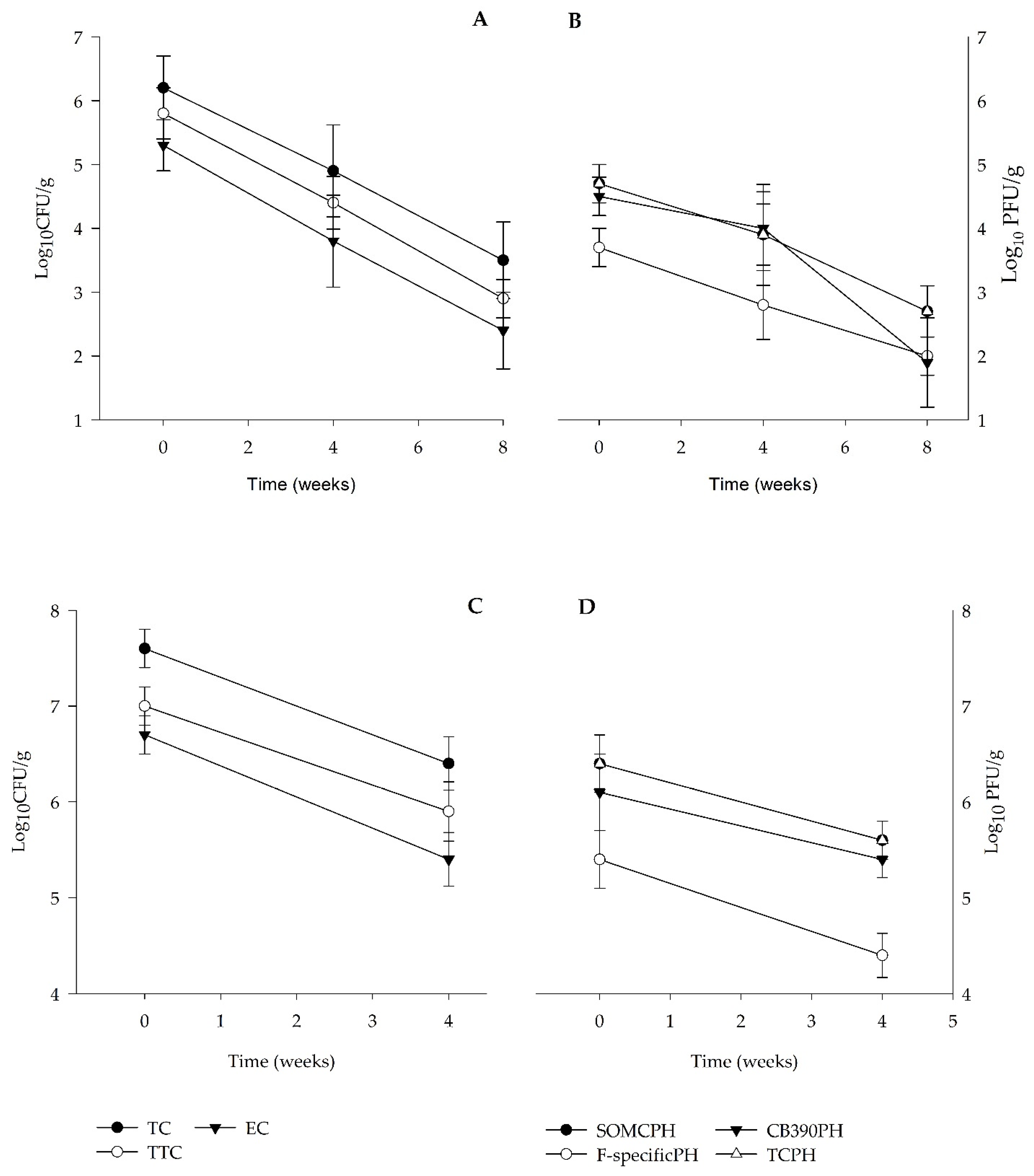
3.1. Sludge Bacterial and Viral Indicators
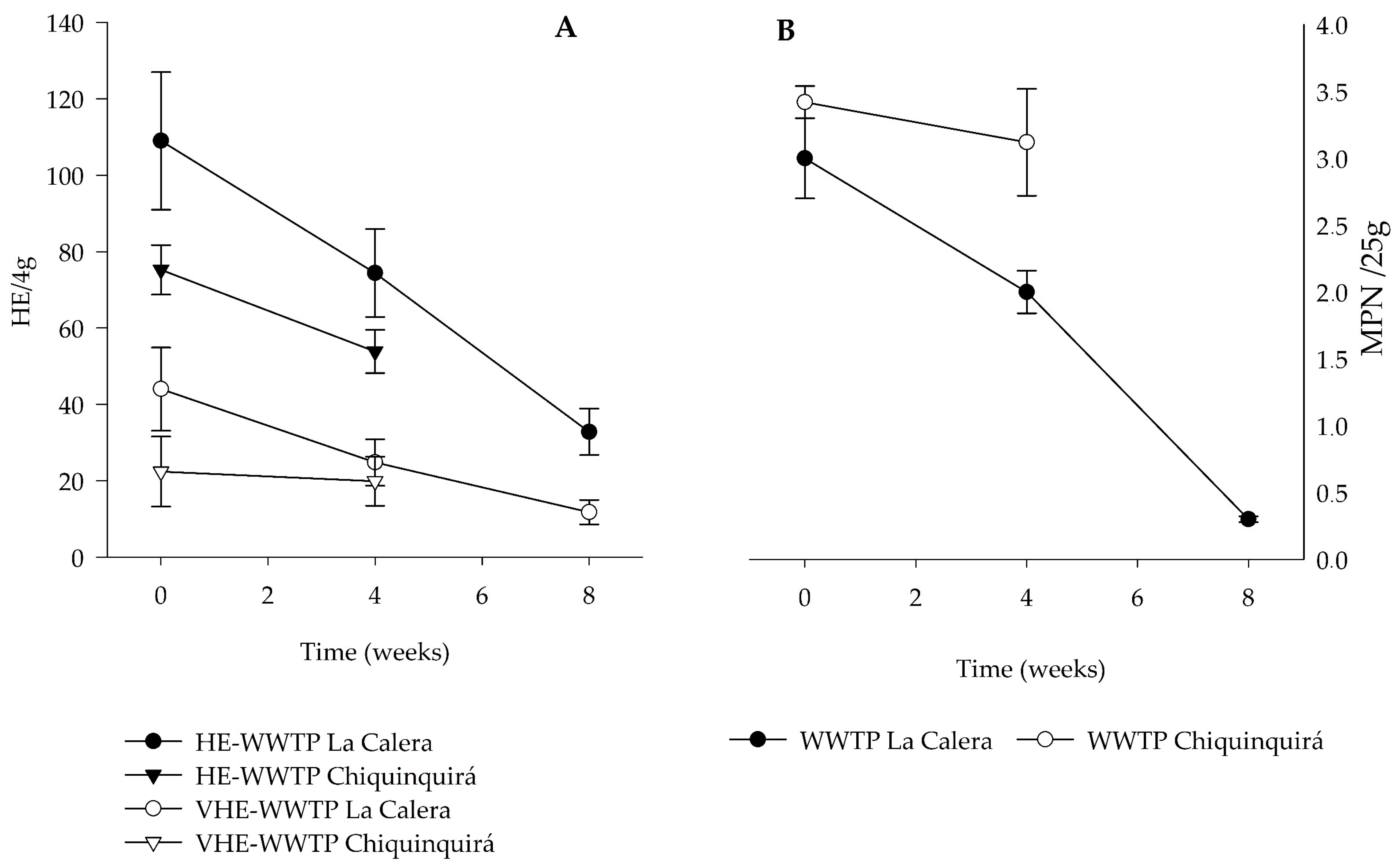
3.2. Concentration of Salmonella spp and Helminth Egg in Sludge
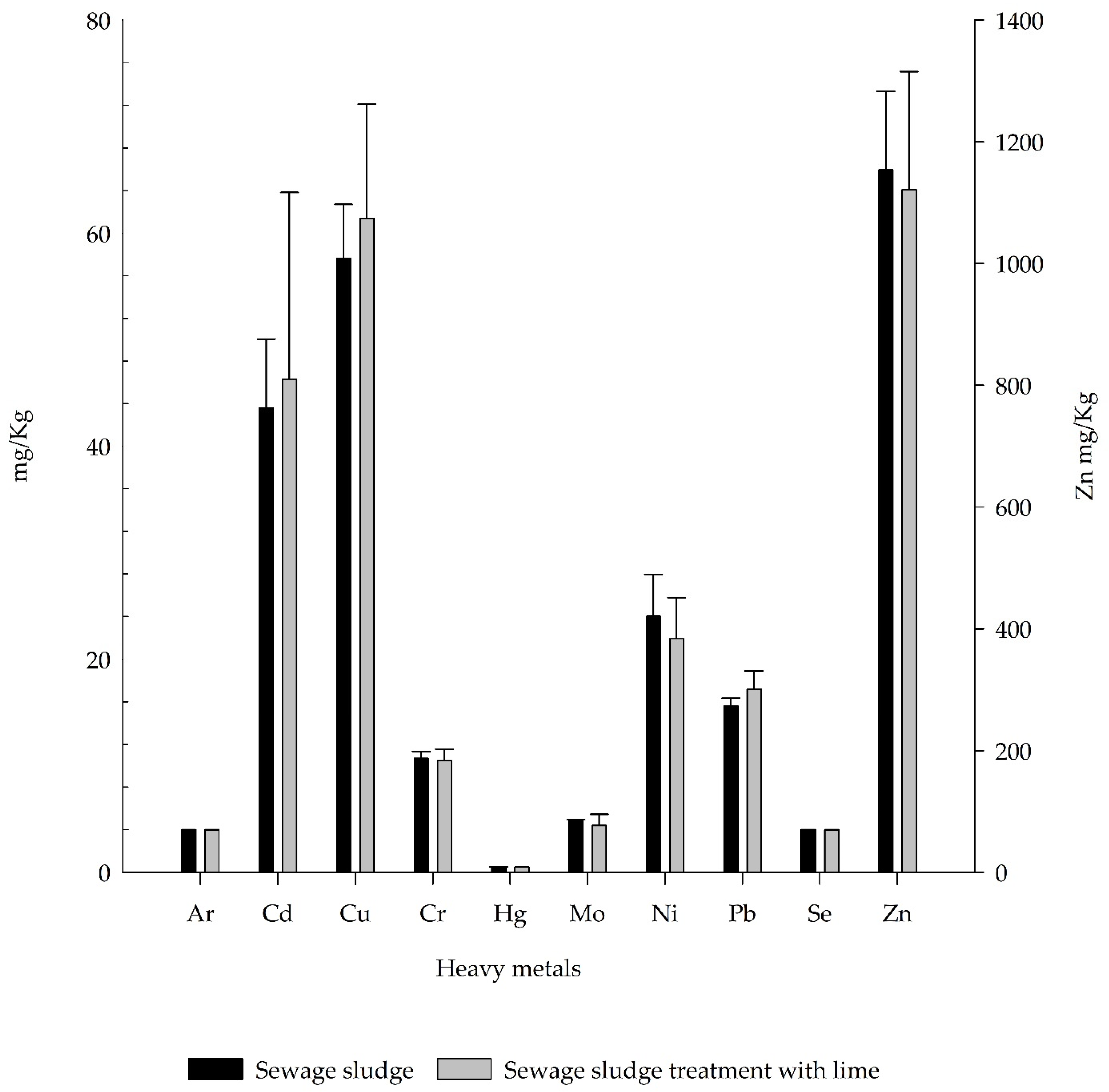
3.3. Chiquinquirá WTTP Heavy Metal Concetration
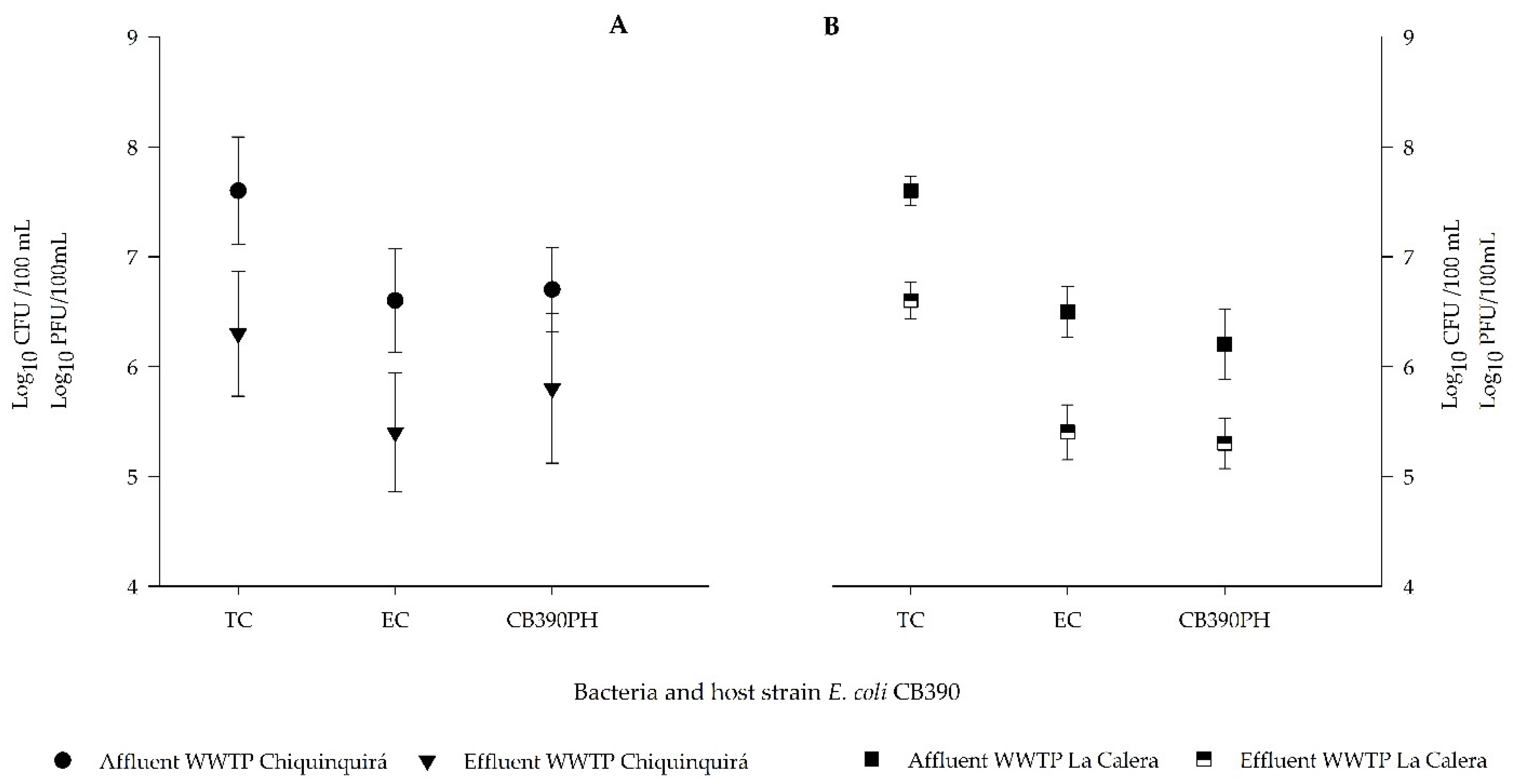
3.4. Bacterial and Viral Indicators in Domestic Waste Water
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Indicator | WWTP La Calera | WTTP Chiquinquirá | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sludge n: 9 | Sludge Dryingbed Treatment n: 5 (4 Weeks) | Sludge Dryingbed Treatment n: 5 (8 Weeks) | Sludge n: 24 | Lime-Terated Sludge n: 27 (4 Weeks) | ||||||
| Average (SD) | Range | Average (SD) | Range | Average (SD) | Range | Average (SD) | Range | Average (SD) | Range | |
| TC Log10 (UFC/g) | 6.2 (0.5) | 5.7–6.9 | 4.9 (0.7) | 4.1–5.7 | 3.5 (0.6) | 2.5–4.0 | 7.6 (0.2) | 7.2–8 | 6.4 (0.3) | 5.7–6.8 |
| TTC Log10 (UFC/g) | 5.8 (0.4) | 5.3–6.4 | 4.4 (0.4) | 3.9–4.9 | 2.9 (0.3) | 2.3–3.1 | 7.0 (0.2) | 6.5–7.4 | 5.9 (0.3) | 5.2–6.3 |
| E. coli Log10 (UFC/g) | 5.3 (0.4) | 4.7–5.9 | 3.8 (0.7) | 3.0–4.6 | 2.4 (0.6) | 1.4–3.0 | 6.7 (0.2) | 6.2–7.1 | 5.4 (0.3) | 4.9–5.8 |
| SOMCPH Log10 (UFC/g) | 4.7 (0.3) | 4.3–5.1 | 3.9 (0.5) | 3.4–4.3 | 2.7 (0.4) | 2.1–3.1 | 6.4 (0.3) | 5.9–7.1 | 5.6 (0.2) | 5.3–6.0 |
| F-specificPH Log10 (UFC/g) | 3.7 (0.3) | 3.3–4.1 | 2.8 (0.5) | 2.2–3.4 | 1.6 (0.3) | 1.1–2.0 | 5.4 (0.3) | 4.8–6.1 | 4.4 (0.2) | 4.0–4.9 |
| PHCB390 Log10 (UFC/g) | 4.5 (0.3) | 3.9–5 | 4.0 (0.6) | 3.3–4.6 | 2.6 (0.7) | 1.4–3.1 | 6.1 (0.4) | 5.6–7.0 | 5.4 (0.2) | 5.2–5.9 |
| TCPH Log10 (UFC/g) | 4.7 (0.3) | 4.4–5.2 | 3.9 (0.5) | 3.4–4.4 | 2.7 (0.4) | 2.1–3.1 | 6.4 (0.3) | 6.0–7.1 | 5.6 (0.2) | 5.4–6.1 |
| Salmonella MPN/25 g | 2.5 (0.3) | 2.1–2.9 | 2.0 (0.2) | 1.9–2.2 | 0.3 (0.0) | 0.3–0.3 | 3.4 (0.1) | 3.2–3.6 | 3.1 (0.4) | 2.4–3.5 |
| HE/4 g | 109 (18) | 82–136 | 74.4 (11.5) | 56–84 | 32.8 (6.1) | 24–40 | 75.3 (6.5) | 64–88 | 53.9 (5.7) | 40–66 |
| VHE/4 g | 43.8 (10.9) | 24–58 | 24.8 (6.1) | 20–34 | 11.8 (3.2) | 8.0–16 | 22.4 (9.2) | 4.0–34 | 19.9 (6.4) | 8.0–34 |
References
- UNICEF; World Health Organization. Progress on Sanitation and Drinking Water 2015 Update and MDG Assessment; World Health Organization: Geneva, Switzerland, 2015; pp. 1–90. ISBN 978-92-4-150914-5. [Google Scholar]
- United Nations World Water Assessment Programme. The United Nations World Water Development Report 2017. Wastewater: The Untapped Resource; UNESCO: Paris, France, 2017; ISBN 978-92-3-100201-4. [Google Scholar]
- MinVivienda. Plan Director de Agua y Saneamiento Básico 2018–2030; Ministerio de Vivienda, Ciudad y Territorio: Bogotá, Colombia, 2018; pp. 1–103.
- Porcentaje de Aguas Residuales Urbanas Domésticas Tratadas de Manera Segura Indicador. Available online: https://www.ods.gov.co/es/objetivos/agua-limpia-y-saneamiento (accessed on 15 February 2021).
- Agua Limpia y Saneamiento, La Agenda 2030 en Colombia y Objetivos de Desarrollo Sostenible. Available online: https://www.ods.gov.co/es/data-explorer?state=%7B%22goal%22%3A%226%22%2C%22indicator%22%3A%226.1.1.P%22%2C%22dimension%22%3A%22DES_GEO_DEPTOS%22%2C%22view%22%3A%22line%22%7D (accessed on 25 February 2021).
- Departamento Nacional de Planeación. CONPES 3918. Estrategia para la Implementación de los Objetivos de Desarrollo Sostenible (ODS) en Colombia; Departamento Nacional de Planeación: Bogotá, Colombia, 2018; pp. 1–74.
- Superintendencia de Servicios Públicos Domiciliarios. Estudio Sectorial de los Servicios Públicos Domiciliarios de Acueducto y Alcantarillado 2014–2017; Superintendencia de Servicios Públicos Domiciliarios: Bogotá, Colombia, 2018; pp. 1–88.
- Wiśniowska, E.; Grobelak, A.; Kokot, P.; Kacprzak, M. Sludge legislation-comparison between different countries. In Industrial and Municipal Sludge: Emerging Concerns and Scope for Resource Recovery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 201–224. ISBN 978-0-12-815907-1. [Google Scholar]
- Margot, J.; Rossi, L.; Barry, D.A.; Holliger, C. A Review of the Fate of Micropollutants in Wastewater Treatment Plants. Wiley Interdiscip. Rev. Water 2015, 2, 457–487. [Google Scholar] [CrossRef] [Green Version]
- Mihelcic, J. Sludge Management: Biosolids and Fecal Sludge. In Water and Sanitation for the 21st Century: Health and Microbiological Aspects of Excreta and Wastewater Management (Global Water Pathogen Project); Mihelcic, J., Verbyla, M., Eds.; Michigan State University: Lansing, MI, USA; UNESCO: Paris, France, 2018. [Google Scholar]
- Basic Information about Biosolids. Available online: https://www.epa.gov/biosolids/basic-information-about-biosolids (accessed on 25 February 2021).
- Collard, M.; Teychené, B.; Lemée, L. Comparison of Three Different Wastewater Sludge and Their Respective Drying Processes: Solar, Thermal and Reed Beds–Impact on Organic Matter Characteristics. J. Environ. Manag. 2017, 203, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Teoh, S.K.; Li, L.Y. Feasibility of Alternative Sewage Sludge Treatment Methods from a Lifecycle Assessment (LCA) Perspective. J. Clean. Prod. 2020, 247. [Google Scholar] [CrossRef]
- Lu, Q.; He, Z.L.; Stoffella, P.J. Land Application of Biosolids in the USA: A Review. Appl. Environ. Soil Sci. 2012, 2012, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Gianico, A.; Braguglia, C.M.; Gallipoli, A.; Montecchio, D.; Mininni, G. Land Application of Biosolids in Europe: Possibilities, Con-Straints and Future Perspectives. Water 2021, 13, 103. [Google Scholar] [CrossRef]
- Gutiérrez-Rosero, J.A.; Ramírez-Fajardo, Á.I.; Rivas, R.; Linares, B.; Paredes, D. Tratamiento de Lodos Generados en el Proceso Convencional de Potabilización de Agua. Rev. Ing. Univ. Medellín 2014, 13, 13–27. [Google Scholar] [CrossRef] [Green Version]
- Velez, J.A. Los Biosólidos: ¿Una Solución o un Problema? Prod. Más Limpia 2007, 2, 57–70. [Google Scholar]
- Melo Cerón, A.R.; Rodríguez González, A.; González Guzmán, J.M. Manejo de Biosólidos y su Posible Aplicación al Suelo, Caso Colombia y Uruguay. Rev. Investig. Agrar. Ambient. 2017, 8, 217–226. [Google Scholar] [CrossRef]
- Spinosa, L. Wastewater Sludge: A Global Overview of the Current Status and Future Prospects, 2nd ed.; IWA Publishing: London, UK, 2011; pp. 1–41. ISBN 978-1-78040-119-5. [Google Scholar]
- Venegas, C. Aprovechamiento de Los Biosólidos para la Agricultura a Través del Fortalecimiento de Estrategias de Gestión Ambiental para el municipio de Chiquinquirá, Boyacá, Colombia. (Unpublished; Manuscript in Preparation). Master’s Thesis, Pontificia Universidad Javeriana, Bogotá, Colombia, 2021. [Google Scholar]
- Dáguer, G.P. Gestión de Biosólidos en Colombia. Rev. ACODAL 2003, 46, 1–7. [Google Scholar]
- Standards for the Use or Disposal of Sewage Sludge 40 CFR Part 503. Available online: https://www.law.cornell.edu/cfr/text/40/part-503 (accessed on 6 February 2021).
- About Biosolids. Available online: https://www.nebiosolids.org/about-biosolids (accessed on 15 March 2021).
- Chávez Porras, Á.; Velásquez Castiblanco, Y.L.; Casallas Ortega, N.D. Características Físico-Químicas de Humus Obtenido de Biosólidos Provenientes de Procesos de Tratamiento de Aguas Residuales. Inf. Técnico 2017, 81, 122–130. [Google Scholar] [CrossRef] [Green Version]
- Eriksson, E.; Christensen, N.; Ejbye Schmidt, J.; Ledin, A. Potential Priority Pollutants in Sewage Sludge. Desalination 2008, 226, 371–388. [Google Scholar] [CrossRef]
- Viau, E.; Bibby, K.; Paez-Rubio, T.; Peccia, J. Toward a Consensus View on the Infectious Risks Associated with Land Application of Sewage Sludge. Environ. Sci. Technol. 2011, 45, 5459–5469. [Google Scholar] [CrossRef]
- Turek, V.; Kilkovský, B.; Jegla, Z.; Stehlík, P. Proposed EU Legislation to Force Changes in Sewage Sludge Disposal: A Case Study. Front. Chem. Sci. Eng. 2018, 12, 660–669. [Google Scholar] [CrossRef]
- Turunen, V.; Sorvari, J.; Mikola, A. A Decision Support Tool for Selecting the Optimal Sewage Sludge Treatment. Chemosphere 2018, 193, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Rulkens, W.H. Sustainable Sludge Management—What Are the Challenges for the Future? Water Sci. Technol. 2004, 49, 11–19. [Google Scholar] [CrossRef]
- Tytła, M. Assessment of Heavy Metal Pollution and Potential Ecological Risk in Sewage Sludge from Municipal Wastewater Treatment Plant Located in the Most Industrialized Region in Poland—Case Study. Int. J. Environ. Res. Public Health 2019, 16, 2430. [Google Scholar] [CrossRef] [Green Version]
- Crini, G.; Lichtfouse, E. Advantages and Disadvantages of Techniques Used for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Mohamed Samer Biological and Chemical Wastewater Treatment Processes. In Wastewater Treatment Engineering; IntechOpen: Rijeka, Croatia, 2015; pp. 1–49.
- Xu, Y.-N.; Chen, Y. Advances in Heavy Metal Removal by Sulfate-Reducing Bacteria. Water Sci. Technol. 2020, 81, 1797–1827. [Google Scholar] [CrossRef]
- van den Brand, T.; Snip, L.; Palmen, L.; Weij, P.; Sipma, J.; van Loosdrecht, M. Sulfate Reducing Bacteria Applied to Domestic Wastewater. Water Pract. Technol. 2018, 13, 542–554. [Google Scholar] [CrossRef]
- Liu, Z.; Li, L.; Li, Z.; Tian, X. Removal of Sulfate and Heavy Metals by Sulfate-Reducing Bacteria in an Expanded Granular Sludge Bed Reactor. Environ. Technol. 2018, 39, 1814–1822. [Google Scholar] [CrossRef] [PubMed]
- Disposal and Recycling Routes for Sewage Sludge Part 1—Sludge Use Acceptance. Available online: https://ec.europa.eu/environment/archives/waste/sludge/pdf/sludge_disposal1.pdf (accessed on 25 February 2021).
- Disposal and Recycling Routes for Sewage Sludge Part 2—Regulatory Report. Available online: http://ec.europa.eu/environment/archives/waste/sludge/pdf/sludge_disposal2.pdf (accessed on 25 February 2021).
- Hudcová, H.; Vymazal, J.; Rozkošný, M. Present Restrictions of Sewage Sludge Application in Agriculture within the European Union. Soil Water Res. 2019, 14, 104–120. [Google Scholar] [CrossRef]
- Ministry of Environment and Sustainable Development. Technical Standard for the Sustainable Management of Sludge and Biosolids Generated in Treatment Plants of Cloacal and Cloacal-Industrial Liquid Effluents in Argentina, Resolution 410/2018; Ministry of Environment and Sustainable Development: Buenos Aires, Argentina, 2018; pp. 1–18.
- Ministry of Environment and Sustainable Development. Sustainable Management of Sludge and Biosolids Generated in Sewage and Mixed Sewage-Industrial Liquid Effluent Treatment Plants in Argentina, Annex IF-2018-19428692-APN-DCAYR # MAD. IF-2018-19428692-APN-DCAYR # MAD; Ministry of Environment and Sustainable Development: Buenos Aires, Argentina, 2018; pp. 1–17.
- Mininni, G.; Blanch, A.R.; Lucena, F.; Berselli, S. EU Policy on Sewage Sludge Utilization and Perspectives on New Approaches of Sludge Management. Environ. Sci. Pollut. Res. 2015, 22, 7361–7374. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Housing, City and Territory. Criteria Are Established for the Use of Biosolids Generated in Municipal Wastewater Treatment Plants in Colombia, Decree 1287; Ministry of Housing, City and Territory: Bogotá, Colombia, 2014; pp. 1–15. [Google Scholar]
- Ministry General Secretariat of the Presidency of Chile. Regulation for the Management of Sludge Generated in Sewage Treatment Plants in Chile, Decree 4; Ministry General Secretariat of the Presidency of Chile: Santiago, Chile, 2009; pp. 1–15.
- Ministry of the Environment National Council of the Environment. Defines Criteria and Procedures for the Agricultural Use of Sewage Sludge Generated in Sanitary Sewage Treatment Plants and Their Derived Products, and Provides Other Measures in Brazil, Resolution Resolución N° 375; Ministry of the Environment National Council of the Environment: Brasilia, Brazil, 2006; pp. 1–32.
- Ministry of the Environment and Natural Resources. Official Mexican Standard, Environmental Protection, Sludge and Biosolids, NOM-004-SEMARNAT-2002; Ministry of the Environment and Natural Resources: Mexico City, Mexico, 2003; pp. 1–37.
- Spinosa, L. Characterization: A Necessary Tool in Sludge Management. Water Sci. Technol. 2013, 68, 748–755. [Google Scholar] [CrossRef]
- Ministry of Housing, Construction and Sanitation. Biosolids Monitoring Protocol in Peru, Resolution N° 093-2018; Ministry of Housing, Construction and Sanitation: Lima, Peru, 2018; pp. 1–34.
- Ministry of Housing, Construction and Sanitation. Regulation for the Reuse of Sludge Generated in Wastewater Treatment Plants in Peru, Decree N° 015-2017; Ministry of Housing, Construction and Sanitation: Lima, Peru, 2017; pp. 1–9.
- Ministry of Commerce and Industry. Water Uses and Final Disposal of Sludge in Panama. Resolution 2000, 352, 12. [Google Scholar]
- Ministry of the Environment National Council of the Environment. Defines criteria and procedures for the production and application of biosolids in soils, and takes other measures in Brazil. Resolution 2020, 498, 1–10. [Google Scholar]
- Regli, S.; Rose, J.B.; Haas, C.N.; Gerba, C.P. Modeling the Risk from Giardia and Viruses in Drinking Water. J. AWWA 1991, 83. [Google Scholar] [CrossRef]
- Funderburg, S.W.; Sorber, C.A. Coliphages as Indicators of Enteric Viruses in Activated Sludge. Water Res. 1985, 19, 547–555. [Google Scholar] [CrossRef]
- Sidhu, J.; Toze, S. Human Pathogens and Their Indicators in Biosolids: A Literature Review. Environ. Int. 2009, 35, 187–201. [Google Scholar] [CrossRef]
- Ballesté, E.; Blanch, A.R.; Mendez, J.; Sala-Comorera, L.; Maunula, L.; Monteiro, S.; Farnleitner, A.H.; Tiehm, A.; Jofre, J.; García-Aljaro, C. Bacteriophages Are Good Estimators of Human Viruses Present in Water. Front. Microbiol. 2021, 12, 973. [Google Scholar] [CrossRef]
- Toribio-Avedillo, D.; Blanch, A.R.; Muniesa, M.; Rodríguez-Rubio, L. Bacteriophages as Fecal Pollution Indicators. Viruses 2021, 13, 1089. [Google Scholar] [CrossRef]
- McMinn, B.R.; Ashbolt, N.J.; Korajkic, A. Bacteriophages as Indicators of Faecal Pollution and Enteric Virus Removal. Lett. Appl. Microbiol. 2017, 65, 11–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McMinn, B.R.; Rhodes, E.R.; Huff, E.M.; Korajkic, A. Decay of Infectious Adenovirus and Coliphages in Freshwater Habitats Is Differentially Affected by Ambient Sunlight and the Presence of Indigenous Protozoa Communities. Virol. J. 2020, 17, 1. [Google Scholar] [CrossRef] [PubMed]
- Jofre, J.; Lucena, F.; Blanch, A.R.; Muniesa, M. Coliphages as Model Organisms in the Characterization and Management of Water Resources. Water 2016, 8, 199. [Google Scholar] [CrossRef] [Green Version]
- Skraber, S.; Gassilloud, B.; Schwartzbrod, L.; Gantzer, C. Survival of Infectious Poliovirus-1 in River Water Compared to the Persistence of Somatic Coliphages, Thermotolerant Coliforms and Poliovirus-1 Genome. Water Res. 2004, 38, 2927–2933. [Google Scholar] [CrossRef] [PubMed]
- Martín-Díaz, J.; Lucena, F.; Blanch, A.R.; Jofre, J. Review: Indicator Bacteriophages in Sludge, Biosolids, Sediments and Soils. Environ. Res. 2020, 182. [Google Scholar] [CrossRef]
- Lasobras, J.; Dellunde, J.; Jofre, J.; Lucena, F. Occurrence and Levels of Phages Proposed as Surrogate Indicators of Enteric Viruses in Different Types of Sludges. J. Appl. Microbiol. 1999, 86, 723. [Google Scholar] [CrossRef]
- Mandilara, G.; Mavridou, A.; Lambiri, M.; Vatopoulos, A.; Rigas, F. The Use of Bacteriophages for Monitoring the Microbiological Quality of Sewage Sludge. Environ. Technol. 2006, 27, 367–375. [Google Scholar] [CrossRef]
- Mocé-Llivina, L.; Muniesa, M.; Pimenta-Vale, H.; Lucena, F.; Jofre, J. Survival of Bacterial Indicator Species and Bacteriophages after Thermal Treatment of Sludge and Sewage. Appl. Environ. Microbiol. 2003, 69, 1452–1456. [Google Scholar] [CrossRef] [Green Version]
- Guzmán, C.; Jofre, J.; Montemayor, M.; Lucena, F. Occurrence and Levels of Indicators and Selected Pathogens in Different Sludges and Biosolids. J. Appl. Microbiol. 2007, 103, 2420–2429. [Google Scholar] [CrossRef]
- Mignotte-Cadiergues, B.; Gantzer, C.; Schwartzbrod, L. Evaluation of Bacteriophages during the Treatment of Sludge. In Proceedings of the Water Science and Technology. Water Sci. Technol. 2002, 46, 189–194. [Google Scholar] [CrossRef]
- Bailey, E.S.; Price, M.; Casanova, L.M.; Sobsey, M.D. E. coli CB390: An Alternative, E. coli Host for Simultaneous Detection of Somatic and F+ Coliphage Viruses in Reclaimed and Other Waters. J. Virol. Methods 2017, 250, 25–28. [Google Scholar] [CrossRef]
- Campos, C.; Méndez, J.; Venegas, C.; Riaño, L.F.; Castaño, P.; Leiton, N.; Riaño, E. Aptness of Escherichia coli Host Strain CB390 to Detect Total Coliphages in Colombia. Sci. Rep. 2019, 9, 9246. [Google Scholar] [CrossRef]
- Agulló-Barceló, M.; Galofré, B.; Sala, L.; García-Aljaro, C.; Lucena, F.; Jofre, J. Simultaneous Detection of Somatic and F-Specific Coliphages in Different Settings by Escherichia coli Strain CB390. FEMS Microbiol. Lett. 2016, 363, fnw180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guzmán, C.; Mocé-Llivina, L.; Lucena, F.; Jofre, J. Evaluation of Escherichia coli Host Strain CB390 for Simultaneous Detection of Somatic and F-Specific Coliphages. Appl. Environ. Microbiol. 2008, 74, 531–534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korajkic, A.; McMinn, B.; Herrmann, M.P.; Sivaganesan, M.; Kelty, C.A.; Clinton, P.; Nash Maliha, S.; Shanks Orin, C.; Schaffner Donald, W. Viral and Bacterial Fecal Indicators in Untreated Wastewater across the Contiguous United States Exhibit Geospatial Trends. Appl. Environ. Microbiol. 2020, 86, e02967-19. [Google Scholar] [CrossRef] [PubMed]
- Jebri, S.; Jofre, J.; Barkallah, I.; Saidi, M.; Hmaied, F. Presence and Fate of Coliphages and Enteric Viruses in Three Wastewater Treatment Plants Effluents and Activated Sludge from Tunisia. Environ. Sci. Pollut. Res. 2012, 19, 2195–2201. [Google Scholar] [CrossRef] [PubMed]
- Harwood, V.J.; Levine, A.D.; Scott, T.M.; Chivukula, V.; Lukasik, J.; Farrah, S.R.; Rose, J.B. Validity of the Indicator Organism Paradigm for Pathogen Reduction in Reclaimed Water and Public Health Protection. Appl. Environ. Microbiol. 2005, 71, 3163–3170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costán-Longares, A.; Montemayor, M.; Payán, A.; Méndez, J.; Jofre, J.; Mujeriego, R.; Lucena, F. Microbial Indicators and Pathogens: Removal, Relationships and Predictive Capabilities in Water Reclamation Facilities. Water Res. 2008, 42, 4439–4448. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (EPA). Control of Pathogens and Vector Attraction in Sewage Sludge—(Including Domestic Septage): Under 40 CFR Part 503; (EPA 625/R-92/013); EPA: Cincinnati, OH, USA, 2003.
- United States Environmental Protection Agency (EPA). Method 1682: Salmonella in Sewage Sludge (Biosolids) by Modified Semisolid Rappaport-Vassiliadis (MSRV) Medium; (EPA-821-R-06-14); EPA: Washington, DC, USA, 2006.
- International Standardization Organization (ISO). Water Quality. Enumeration of Escherichia coli and Coliform Bacteria—Part 1: Membrane Filtration Method for Waters with Low Bacterial Background Flora; ISO 9308-1:2014; International Organization for Standardization: Geneva, Switzerland, 2014. [Google Scholar]
- International Standardization Organization (ISO). Water Quality. Detection and Enumeration of Bacteriophages. Pt. 2: Enumeration of Somatic Coliphages; ISO-10705-2; International Standardization Organization: Geneva, Switzerland, 2000. [Google Scholar]
- International Organization for Standardization (ISO). Water Quality. Detection and Enumeration of Bacteriophages—Part 1: Enumeration of F-Specific RNA Bacteriophages; ISO 10705-1; International Organization for Standardization: Geneva, Switzerland, 1995. [Google Scholar]
- U.S. Environmental Protection Agency. Method 200.7 Trace Elements in Water, Solids, and Biosolids by Inductively Coupled Plasma-Atomic Emission Spectrometry; U.S. Environmental Protection Agency: Washington, DC, USA, 2001.
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017; p. 1323. ISBN 978-0-87553-287-5. [Google Scholar]
- Maya, C.; Ortiz, M.; Jiménez, B. Viability of Ascaris and Other Helminth Genera Non Larval Eggs in Different Conditions of Temperature, Lime (pH) and Humidity. Water Sci. Technol. 2010, 62, 2616–2624. [Google Scholar] [CrossRef]
- Castellanos-Rozo, J.; Galvis-López, J.A.; Merchán Castellanos, N.A.; Manjarres-Hernández, E.H.; Rojas, A.L. Assessment of Two Sludge Stabilization Methods in a Wastewater Treatment Plant in Sotaquirá, Colombia. Univ. Sci. (Bogota) 2020, 25, 17–36. [Google Scholar] [CrossRef]
- Bean, C.; Hansen, J.; Margolin, A.; Balkin, H.; Batzer, G.; Widmer, G. Class B Alkaline Stabilization to Achieve Pathogen Inactivation. Int. J. Environ. Res. Public Health 2007, 4, 53–60. [Google Scholar] [CrossRef] [Green Version]
- Campos, C.; Beltrán, M.; Duarte, M.; Medina, L.; Lucena, F.; Jofre, J. Abatement of Helminth Eggs and Bacterial and Viral Indicators in Soil after Land Application of Treated Sludges. J. Water Resour. Prot. 2013, 5, 1155–1164. [Google Scholar] [CrossRef]
- Biosolids Technology Fact Sheet. Use of Land Filling for Biosolids Management. Available online: https://www.epa.gov/biosolids/fact-sheet-use-landfilling-biosolids-management (accessed on 10 January 2021).
- Santos, D.S.; Teshima, E.; Dias, S.M.F.; Araújo, R.A.; Silva, C.M.R. da Efeito Da Secagem Em Leito Nas Características Físico-Químicas e Microbiológicas de Lodo de Reator Anaeróbio de Fluxo Ascendente Usado no Tratamento de Esgoto Sanitário. Eng. Sanit. Ambient. 2016, 22, 341–349. [Google Scholar] [CrossRef] [Green Version]
- Pompeo, R.P.; Andreoli, C.V.; de Castro, E.A.; Aisse, M.M. Influence of Long-Term Storage Operating Conditions on the Reduction of Viable Ascaris Eggs in Sewage Sludge for Agricultural Reuse. Water Air Soil Pollut. 2016, 227, 144. [Google Scholar] [CrossRef]
- Fernanda García, L.; Diaz, F.E. Manejo de Biosólidos de la Planta de Tratamiento de Aguas Residuales, PTAR, del Municipio de La Calera. Master´s Thesis, Universidad Industrial de Santander, Bucaramanga, Colombia, 2015. [Google Scholar]
- Silveira, M.L.A.; Alleoni, L.R.F.; Guilherme, L.R.G. Biosolids and Heavy Metals in Soils. Sci. Agric. 2003, 60, 793–806. [Google Scholar] [CrossRef] [Green Version]
- Venegas, C.; Diez, H.; Blanch, A.R.; Jofre, J.; Campos, C. Microbial Source Markers Assessment in the Bogotá River Basin (Colombia). J. Water Health 2015, 13, 801–810. [Google Scholar] [CrossRef] [Green Version]
- Empresa de Servicios Públicos de La Calera. Informes Análisis de Resultados de Efluente y Afluente PTAR La Calera. Available online: http://www.espucal.gov.co/tema/estudios-e-investigaciones (accessed on 26 February 2021).
- Empochiquinquirá, E.S.P. Informe Semestral de operaciones PTAR Chiquinquirá-Colombia; Empresa Industrial y Comercial de Servicios Públicos de Chiquinquirá: Boyacá, Colombia, 2020; pp. 1–56.
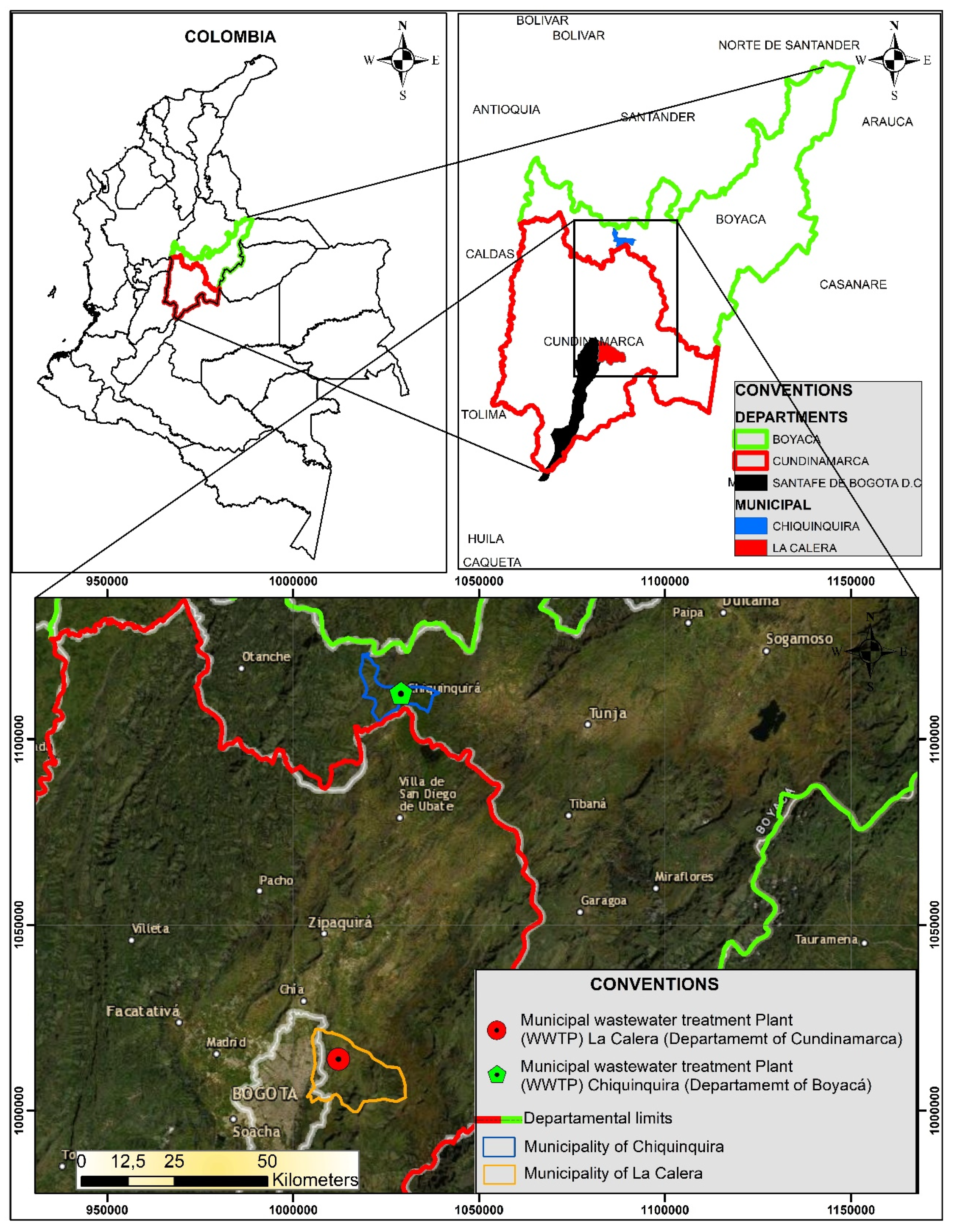
| WWTPs Flow Treatment | Population Served | Water Line | Sludge Treatment | Type of Sludge Stabilization | Time of Treatment or Stabilization | Quantity of Treated Sludge Generated |
|---|---|---|---|---|---|---|
| La Calera 32 L/s | ~18,000 people | Pretreatment Primary treatment Secondary treatment | Digester Drybeds | Drybed | ~2 months | ~4 to 7 Ton/year |
| Chiquinquirá 240 L/s to 252 L/s | ~72,770 people | Thickeners and Dewatering | Lime-treated | ~1 month | ~480 Ton/year |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venegas, C.; Sánchez-Alfonso, A.C.; Celis Zambrano, C.; González Mendez, M.; Vesga, F.-J. E. coli CB390 as an Indicator of Total Coliphages for Microbiological Assessment of Lime and Drying Bed Treated Sludge. Water 2021, 13, 1833. https://doi.org/10.3390/w13131833
Venegas C, Sánchez-Alfonso AC, Celis Zambrano C, González Mendez M, Vesga F-J. E. coli CB390 as an Indicator of Total Coliphages for Microbiological Assessment of Lime and Drying Bed Treated Sludge. Water. 2021; 13(13):1833. https://doi.org/10.3390/w13131833
Chicago/Turabian StyleVenegas, Camilo, Andrea C. Sánchez-Alfonso, Crispín Celis Zambrano, Mauricio González Mendez, and Fidson-Juarismy Vesga. 2021. "E. coli CB390 as an Indicator of Total Coliphages for Microbiological Assessment of Lime and Drying Bed Treated Sludge" Water 13, no. 13: 1833. https://doi.org/10.3390/w13131833
APA StyleVenegas, C., Sánchez-Alfonso, A. C., Celis Zambrano, C., González Mendez, M., & Vesga, F.-J. (2021). E. coli CB390 as an Indicator of Total Coliphages for Microbiological Assessment of Lime and Drying Bed Treated Sludge. Water, 13(13), 1833. https://doi.org/10.3390/w13131833






