Responses of Freshwater Diatoms and Macrophytes Rely on the Stressor Gradient Length across the River Systems
Abstract
1. Introduction
2. Materials and Methods
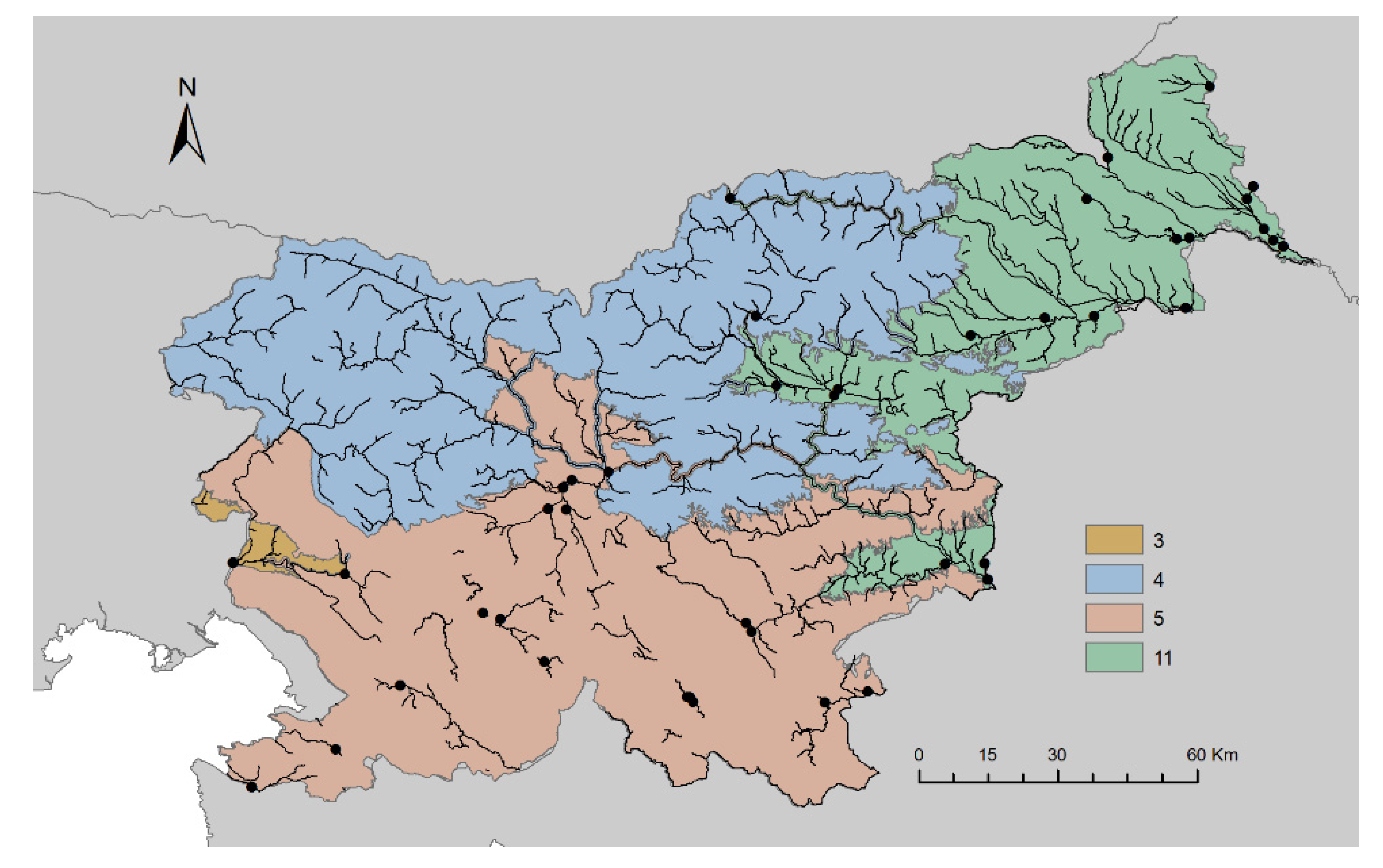
2.1. Study Area and Sampling
- QAi = abundance of the taxon i from the group A;
- QABi = abundance of the taxon i from the group AB;
- QBCi = abundance of the taxon i from the group BC;
- QCi = abundance of the taxon i from the group C;
- QSi = abundance of the taxon i from the groups A, AB, B, BC, and C;
- nA = total number of taxa in group A;
- nAB = total number of taxa in group AB;
- nBC = total number of taxa in group BC;
- nC = total number of taxa in group C;
- nS = total number of taxa in groups A, AB, B, BC, and C.
- TI = Trophic index;
- TWi = trophic value of the taxon i;
- Gi = indicative weight of the taxon i;
- Hi = abundance of the taxon i.
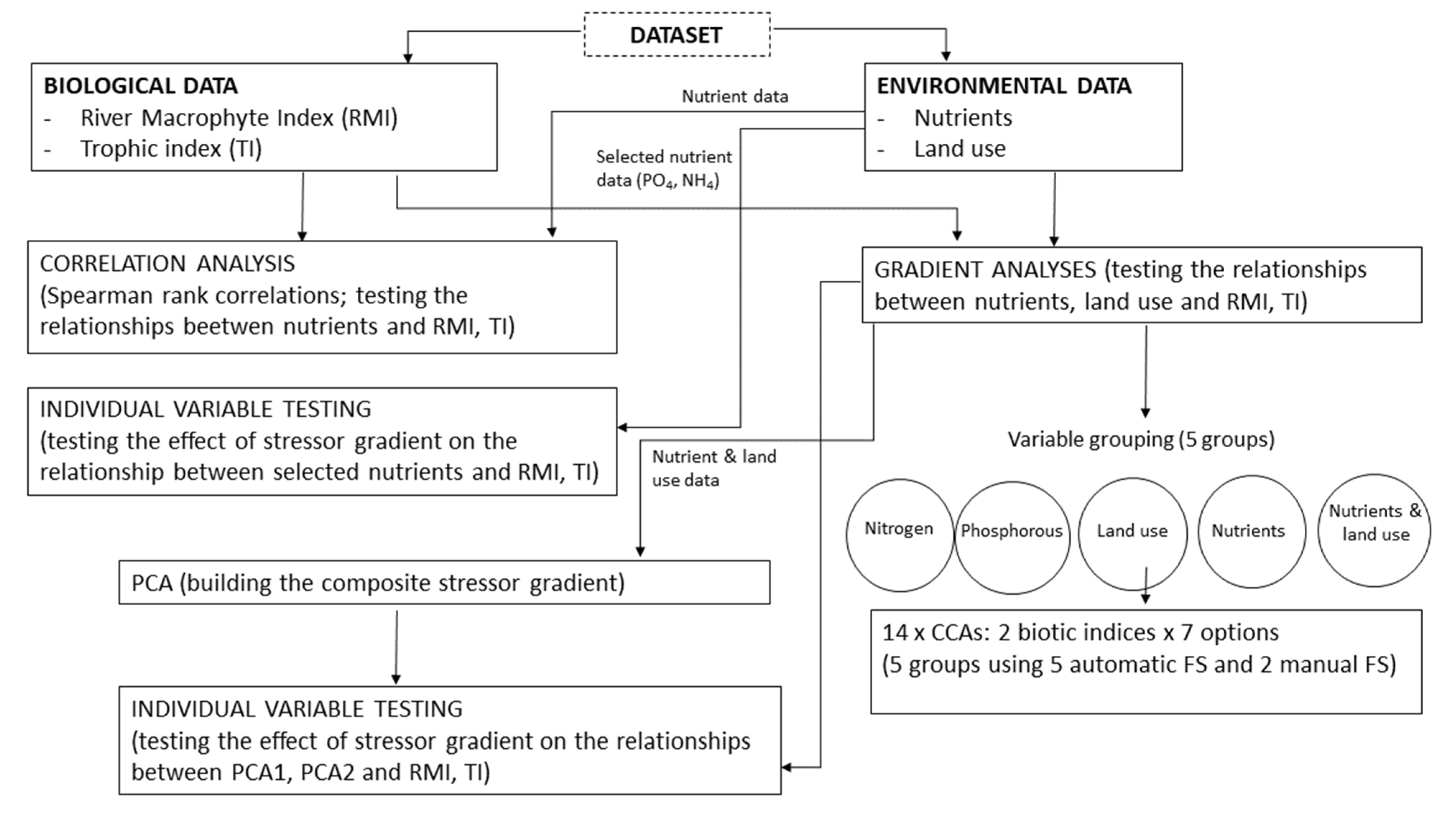
2.2. Data Analyses
3. Results
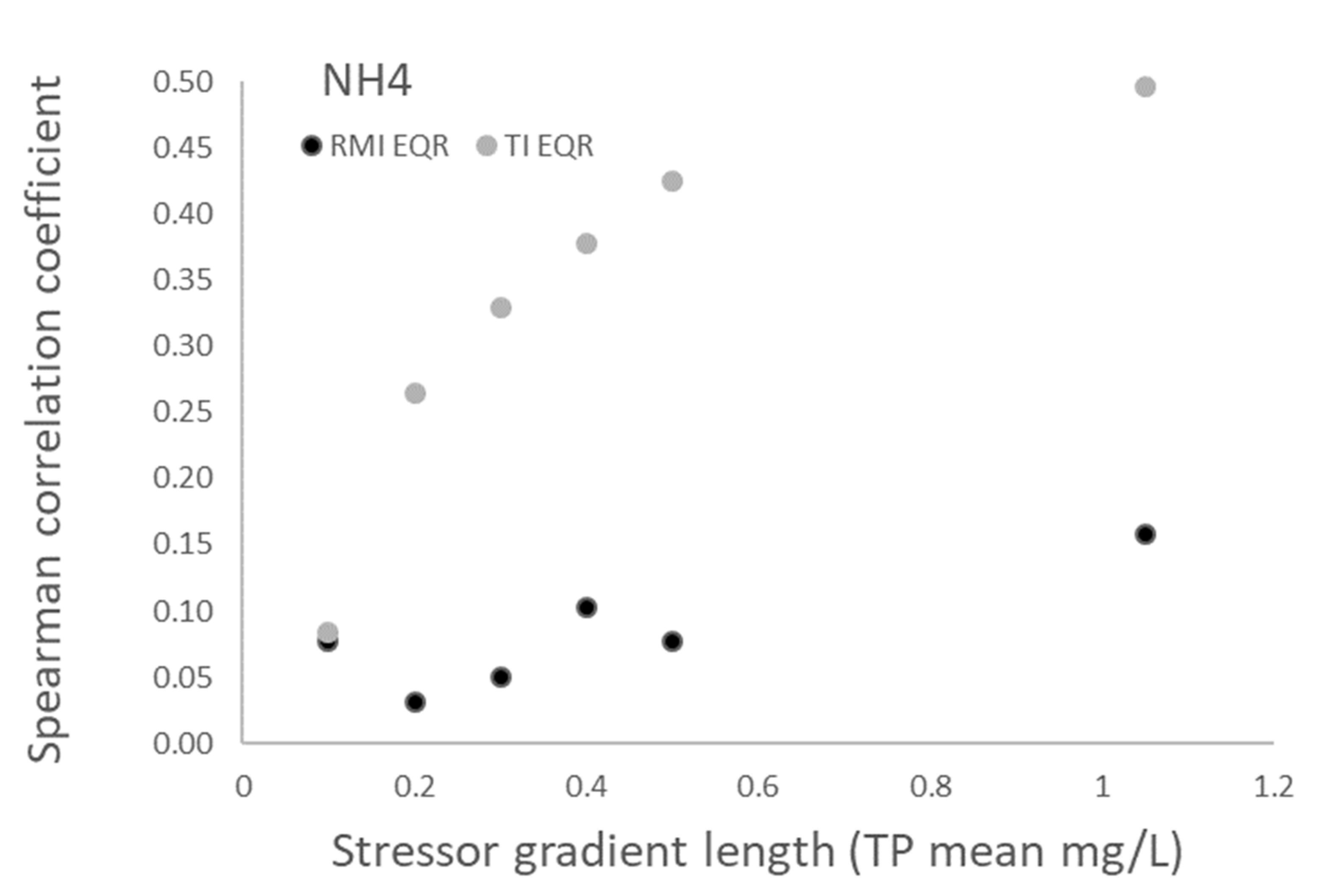
3.1. Relationships between Nutrient Variables and Biotic Indices
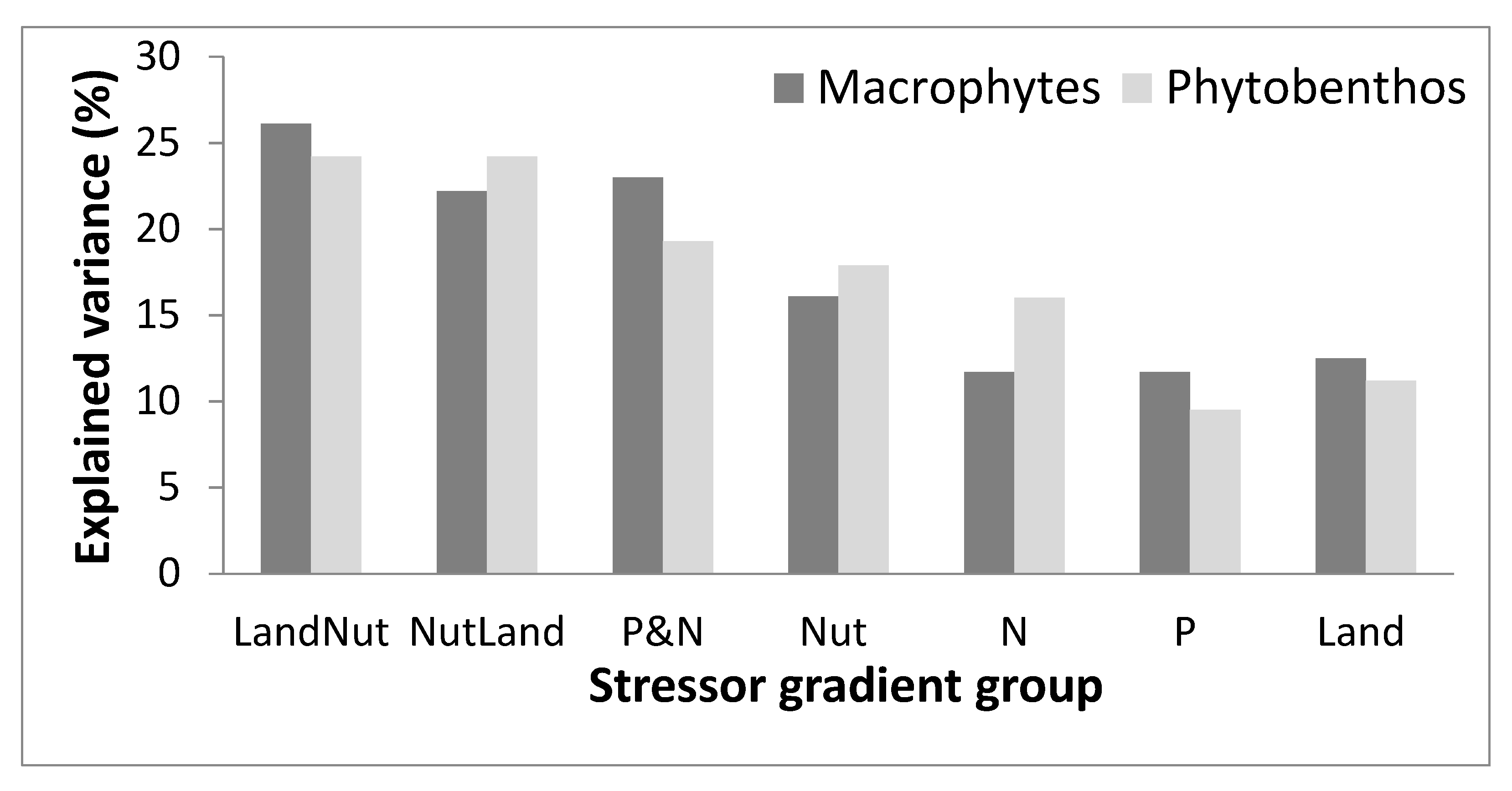
3.2. Relationships between Nutrient, Land Use Variables, and Biotic Communities
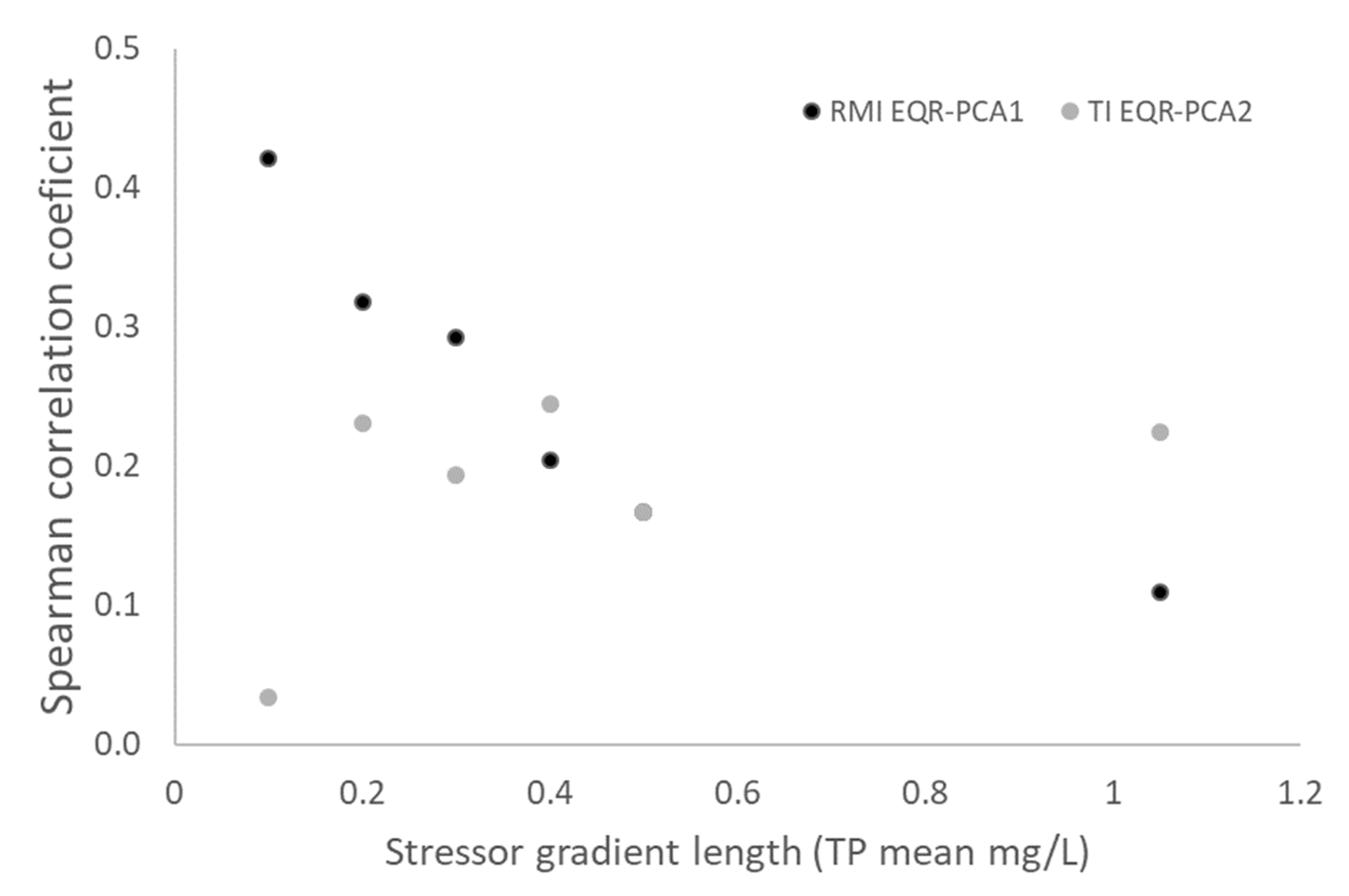
3.3. Composite Stressor Gradient
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schaumburg, J.; Schranz, C.; Foerster, J.; Gutowski, A.; Hofmann, G.; Meilinger, P.; Schneider, S.; Schmedtje, U. Ecological classification of macrophytes and phytobenthos for rivers in Germany according to the water framework directive. Limnologica 2004, 34, 283–301. [Google Scholar] [CrossRef]
- Spitale, D.; Scalfi, A.; Cantonati, M. Niche partitioning, shape of species response, and diversity in the phytobenthos across the rocky shoreline of a large peri-Alpine lake. J. Great Lakes Res. 2012, 38, 620–627. [Google Scholar] [CrossRef]
- Horner, R.R.; Welch, E.B.; Seeley, M.R.; Jacoby, J.M. Responses of periphyton to changes in current velocity, suspended sediment and phosphorus concentration. Freshw. Biol. 1990, 24, 215–232. [Google Scholar] [CrossRef]
- Tanabe, Y.; Ohtani, S.; Kasamatsu, N.; Fukuchi, M.; Kudoh, S. Photophysiological responses of phytobenthic communities to the strong light and UV in Antarctic shallow lakes. Polar Biol. 2010, 33, 85–100. [Google Scholar] [CrossRef]
- Spitale, D.; Scalfi, A.; Cantonati, M. Urbanization effects on shoreline phytobenthos: A multiscale approach at lake extent. Aquat. Sci. 2014, 76, 17–28. [Google Scholar] [CrossRef]
- Allan, J.D.; Castillo, M.M. Stream Ecology; Springer: Dordrecht, The Netherlands, 2007; p. 372. [Google Scholar]
- Graham, L.E.; Graham, J.M.; Graham, J.E.; Wilcox, L.W. Algae; Benjamin Cummings: San Francisco, CA, USA; University of Wisconsin: Madison, WI, USA, 2009; p. 616. [Google Scholar]
- Danilov, R.A.; Ekelund, N.G. The use of epiphyton and epilithon data as a base for calculating ecological indices in monitoring of eutrophication in lakes in central Sweden. Sci. Total Environ. 2000, 248, 63–70. [Google Scholar] [CrossRef]
- Lavoie, I.; Vincent, W.F.; Pienitz, R.; Painchaud, J. Benthic algae as bioindicators of agricultural pollution in the streams and rivers of southern Québec (Canada). Aquat. Ecosyst. Health Manag. 2004, 7, 43–58. [Google Scholar] [CrossRef]
- Hering, D.; Feld, C.K.; Moog, O.; Ofenböck, T. Cook book for the development of a Multimetric Index for biological condition of aquatic ecosystems: Experiences from the European AQEM and STAR projects and related initiatives. In The Ecological Status of European Rivers: Evaluation and Intercalibration of Assessment Methods; Springer: Dordrecht, The Netherlands, 2006; pp. 311–324. [Google Scholar]
- Townsend, S.A.; Garcia, E.A.; Douglas, M.M. The response of benthic algal biomass to nutrient addition over a range of current speeds in an oligotrophic river. Freshw. Sci. 2012, 31, 1233–1243. [Google Scholar] [CrossRef]
- Kelly, M.G.; Phillips, G.; Juggins, S.; Willby, N.J. Re-evaluating expectations for river phytobenthos assessment and understanding the relationship with macrophytes. Ecol. Indic. 2020, 117, 106582. [Google Scholar] [CrossRef]
- Franklin, P.; Dunbar, M.; Whitehead, P. Flow controls on lowland river macrophytes: A review. Sci. Total Environ. 2008, 400, 369–378. [Google Scholar] [CrossRef]
- Kuhar, U.; Germ, M.; Gaberščik, A. Macrophytes of watercourses in the Slovenian Danube Basin. In Macrophytes of Watercourses in the Slovenian Danube Basin; Academia: Praha, Czech Republic, 2018; pp. 234–251. [Google Scholar]
- Baattrup-Pedersen, A.; Riis, T. Macrophyte diversity and composition in relation to substratum characteristics in regulated and unregulated Danish streams. Freshw. Biol. 1999, 42, 375–385. [Google Scholar] [CrossRef]
- Madsen, J.D.; Chambers, P.A.; James, W.F.; Koch, E.W.; Westlake, D.F. The interaction between water movement, sediment dynamics and submersed macrophytes. Hydrobiologia 2001, 444, 71–84. [Google Scholar] [CrossRef]
- Thomaz, S.M.; Cunha, E.R.D. The role of macrophytes in habitat structuring in aquatic ecosystems: Methods of measurement, causes and consequences on animal assemblages’ composition and biodiversity. Acta Limnol. Bras. 2010, 22, 218–236. [Google Scholar] [CrossRef]
- Ambrožič, Š.; Gaberščik, A.; Vrezec, A.; Germ, M. Hydrophyte community structure affects the presence and abundance of the water beetle family Dytiscidae in water bodies along the Drava River. Ecol. Eng. 2018, 120, 397–404. [Google Scholar] [CrossRef]
- Hansen, J.P.; Wikström, S.A.; Axemar, H.; Kautsky, L. Distribution differences and active habitat choices of invertebrates between macrophytes of different morphological complexity. Aquat. Ecol. 2011, 45, 11–22. [Google Scholar] [CrossRef]
- Schneider, S.; Melzer, A. The Trophic Index of Macrophytes (TIM)—A New Tool for Indicating the Trophic State of Running Waters. Int. Rev. Hydrobiol. 2003, 88, 49–67. [Google Scholar] [CrossRef]
- Haslam, S.M. River Plants of Western Europe: The Macrophytic Vegetation of Watercourses of the European Economic Community; Cambridge University Press: Cambridge, UK, 1987; p. 512. [Google Scholar]
- Kuhar, U.; Germ, M.; Gaberščik, A.; Urbanič, G. Development of a River Macrophyte Index (RMI) for assessing river ecological status. Limnologica 2011, 41, 235–243. [Google Scholar] [CrossRef]
- Szoszkiewicz, K.; Budka, A.; Pietruczuk, K.; Kayzer, D.; Gebler, D. Is the macrophyte diversification along the trophic gradient distinct enough for river monitoring? Environ. Monit. Assess. 2017, 189, 4. [Google Scholar] [CrossRef] [PubMed]
- Chambers, P.A.; Prepas, E.E.; Bothwell, M.L.; Hamilton, H.R. Roots versus Shoots in Nutrient Uptake by Aquatic Macrophytes in Flowing Waters. Can. J. Fish. Aquat. Sci. 1989, 46, 435–439. [Google Scholar] [CrossRef]
- Barko, J.W.; Gunnison, D.; Carpenter, S.R. Sediment interactions with submersed macrophyte growth and community dynamics. Aquat. Bot. 1991, 41, 41–65. [Google Scholar] [CrossRef]
- Carr, G.M. Macrophyte growth and sediment phosphorus and nitrogen in a Canadian prairie river. Freshw. Biol. 1998, 39, 525–536. [Google Scholar] [CrossRef]
- Robach, F.; Hajnsek, I.; Eglin, I.; Trémolières, M. Phosphorus sources for aquatic macrophytes in running waters: Water or sediment? Acta Bot. Gall. 1995, 142, 719–731. [Google Scholar] [CrossRef]
- Clarke, S.J. Vegetation growth in rivers: Influences upon sediment and nutrient dynamics. Prog. Phys. Geogr. Earth Environ. 2002, 26, 159–172. [Google Scholar] [CrossRef]
- Madsen, T.V.; Cedergreen, N. Sources of nutrients to rooted submerged macrophytes growing in a nutrient-rich stream. Freshw. Biol. 2002, 47, 283–291. [Google Scholar] [CrossRef]
- Janauer, G.A.; Gaberščik, A.; Kvet, J.; Germ, M.; Exler, N. Macrophytes of the River Danube Basin; Academia: Praha, Czech Republic, 2018; p. 408. [Google Scholar]
- EEA. European Waters—Assessment of Status and Pressures; European Environment Agency: Copenhagen, Denmark, 2012; p. 96. [Google Scholar]
- Poikane, S.; Salas Herrero, F.; Kelly, M.G.; Borja, A.; Birk, S.; van de Bund, W. European aquatic ecological assessment methods: A critical review of their sensitivity to key pressures. Sci. Total Environ. 2020, 740, 140075. [Google Scholar] [CrossRef]
- Rott, E.; Pipp, E.; Pfister, P. Diatom methods developed for river quality assessment in Austria and a cross-check against numerical trophic indication methods used in Europe. Algol. Stud. Für Hydrobiol. Suppl. Vol. 2003, 110, 91–115. [Google Scholar] [CrossRef]
- Lebkuecher, J.G.; Tuttle, E.N.; Johnson, J.L.; Willis, N.K.S. Use of algae to assess the trophic state of a stream in Middle Tennessee. J. Freshw. Ecol. 2015, 30, 349–376. [Google Scholar] [CrossRef][Green Version]
- Turner, R.E.; Rabalais, N.N.; Justic, D.; Dortch, Q. Global patterns of dissolved N, P and Si in large rivers. Biogeochemistry 2003, 64, 297–317. [Google Scholar] [CrossRef]
- Dodkins, I.; Aguiar, F.; Rivaes, R.; Albuquerque, A.; Rodríguez-González, P.; Ferreira, M.T. Measuring ecological change of aquatic macrophytes in Mediterranean rivers. Limnologica 2012, 42, 95–107. [Google Scholar] [CrossRef]
- Aguiar, F.C.; Segurado, P.; Urbanič, G.; Cambra, J.; Chauvin, C.; Ciadamidaro, S.; Dörflinger, G.; Ferreira, J.; Germ, M.; Manolaki, P.; et al. Comparability of river quality assessment using macrophytes: A multi-step procedure to overcome biogeographical differences. Sci. Total Environ. 2014, 476–477, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Dodds, W.K. Eutrophication and trophic state in rivers and streams. Limnol. Oceanogr. 2006, 51, 671–680. [Google Scholar] [CrossRef]
- Francoeur, S.N. Meta-analysis of lotic nutrient amendment experiments: Detecting and quantifying subtle responses. J. North Am. Benthol. Soc. 2001, 20, 358–368. [Google Scholar] [CrossRef]
- Germ, M.; Urbanič, G.; Gaberščik, A.; Kuhar, U.; Šiško, M. Prilagoditev Trofičnega Indeksa Zahtevam Vodne Direktive (Direktiva 2000/60/ES) Za Vrednotenje Ekološkega Stanja Rek V Sloveniji Na Podlagi Makrofitov: Končno Poročilo; Nacionalni inštitut za biologijo: Ljubljana, Slovenia, 2007. [Google Scholar]
- Urbanič, G.; Germ, M. River Macrophyte Index (RMI)—An Update on Index Application with New Boundary Values for Assessment of Rivers in Slovenia; Institute for Water of the Republic of Slovenia: Ljubljana, Slovenia, 2012; p. 18. [Google Scholar]
- Kosi, G.; Šiško, M.; Bricelj, M.; Urbanič, G.; Grbovič, J.; Rotar, B.; Stanič, K. Prilagoditev Trofičnega Indeksa Zahtevam Vodne Direktive (Direktiva 2000/60/ES) Za Vrednotenje Ekološkega Stanja Rek V Sloveniji Na Podlagi Fitobentosa; Nacionalni inštitut za biologijo: Ljubljana, Slovenia, 2006; p. 81. [Google Scholar]
- Kosi, G.; Šiško, M.; Bricelj, M.; Urbanič, G.; Grbovič, J.; Rotar, B.; Stanič, K. Prilagoditev Saprobnega Indeksa Zahtevam Vodne Direktive (Direktiva 2000/60/ES) Za Vrednotenje Ekološkega Stanja Rek V Sloveniji Na Podlagi Fitobentosa; Univerza v Ljubljani: Ljubljana, Slovenia, 2006; p. 83. [Google Scholar]
- Urbanič, G.; Kosi, G. Interkalibracija Metod Vrednotenja Ekološkega Stanja—Fitobentos. Poročilo O Delu Za Leto 2012; Inštitut za vode Republike Slovenije: Ljubljana, Slovenia, 2012; p. 32. [Google Scholar]
- Rott, E.; Hofmann, G.; Pall, K.; Pfister, P.; Pipp, E. Indikationslisten Für Aufwuchsalgen. Teil 1: Saprobielle Indikation; Bundesministerium für Land-und Forstwirtschaft: Wien, Austria, 1997; p. 73. [Google Scholar]
- Rott, E.; Pipp, E.; Pfister, P.; van Dam, H.; Ortler, K.; Binder, N.; Pall, K. Indikationslisten Für Aufwuchsalgen. Teil 2: Trophieindikation; Bundesministerium für Land-und Forstwirtschaft: Wien, Austria, 1999. [Google Scholar]
- Štupnikar, N.; Urbanič, G. Metodologija Vrednotenja Ekološkega Stanja S Podpornimi Splošnimi Fizikalno-Kemijskimi Elementi, Za Vrednotenje Stanja Hranil (Celotni Fosfor); Institute for Water of the Republic of Slovenia: Ljubljana, Slovenia, 2012; p. 46.
- Urbanič, G. Reka Ščavnica—Pomembna dobrina. In Sveta Ana Skozi Čas: Zbornik Občine Sveta Ana; Toš, M., Zemljič, I., Eds.; Ostroga: Maribor, Slovenia, 2009; pp. 29–46. [Google Scholar]
- Šraj-Kržič, N.; Germ, M.; Urbanc-Berčič, O.; Kuhar, U.; Janauer, G.A.; Gaberščik, A. The quality of the aquatic environment and macrophytes of karstic watercourses. Plant Ecol. 2007, 192, 107–118. [Google Scholar] [CrossRef]
- Klančnik, K.; Mlinar, M.; Gaberščik, A. Heterophylly results in a variety of “spectral signatures” in aquatic plant species. Aquat. Bot. 2012, 98, 20–26. [Google Scholar] [CrossRef]
- Klančnik, K.; Vogel-Mikuš, K.; Kelemen, M.; Vavpetič, P.; Pelicon, P.; Kump, P.; Jezeršek, D.; Gianoncelli, A.; Gaberščik, A. Leaf optical properties are affected by the location and type of deposited biominerals. J. Photochem. Photobiol. B Biol. 2014, 140, 276–285. [Google Scholar] [CrossRef]
- Granéli, W.; Solander, D. Influence of aquatic macrophytes on phosphorus cycling in lakes. Hydrobiologia 1988, 170, 245–266. [Google Scholar] [CrossRef]
- Lukács, B.A.; Dévai, G.; Tóthmérész, B. Aquatic macrophytes as bioindicators of water chemistry in nutrient rich backwaters along the Upper-Tisza river (in Hungary). Phytocoenologia 2009, 39, 287–293. [Google Scholar] [CrossRef]
- Illies, J. Limnofauna Europaea: Eine Zusammenstellung Aller Die Europäischen Binnengewässer Bewohnenden Mehrzelligen Tierarten mit Angaben Über Ihre Verbreitung Und Ökologie; Fischer: Stuttgart, Germany, 1978. [Google Scholar]
- Urbanic, G. Redelineation of European inland water ecoregions in Slovenia. Rev. Hydrobiol. 2008, 1, 17–25. [Google Scholar]
- Ministrstvo Za Okolje in Prostor. Metodologija Vrednotenja Ekološkega Stanja Vodotokov Na Podlagi Fitobentosa in Makrofitov; Ministrstvo za okolje in prostor: Ljubljana, Slovenia, 2016; p. 83. [Google Scholar]
- Kohler, A. Methoden der Kartierung von Flora und Vegetation von Süβwasserbiotopen. Landsch. Stadt 1978, 10, 73–85. [Google Scholar]
- Kohler, A.; Janauer, G.A. Zur Methodik der Untersuchung von aquatischen Makrophyten in Fließgewässern. In Handbuch Angewandte Limnologie; Steinberg, C., Bernhardt, H., Klapper, H., Eds.; Ecomed: Landsberg am Lech, Germany, 1995; Volume III-1.1.3, pp. 3–22. [Google Scholar]
- OGRS. Rules on Monitoring of Surface Water. In Pravilnik o Monitoringu Stanja Površinskih Voda; 2009; Volume 10, pp. 832–839. Available online: http://pisrs.si/Pis.web/pregledPredpisa?id=PRAV9315 (accessed on 15 April 2018).
- Almeida, S.F.P.; Elias, C.; Ferreira, J.; Tornés, E.; Puccinelli, C.; Delmas, F.; Dörflinger, G.; Urbanič, G.; Marcheggiani, S.; Rosebery, J.; et al. Water quality assessment of rivers using diatom metrics across Mediterranean Europe: A methods intercalibration exercise. Sci. Total Environ. 2014, 476, 768–776. [Google Scholar] [CrossRef]
- Pavlin, M.; Birk, S.; Hering, D.; Urbanič, G. The role of land use, nutrients, and other stressors in shaping benthic invertebrate assemblages in Slovenian rivers. Hydrobiologia 2011, 678, 137–153. [Google Scholar] [CrossRef]
- Corine Land Cover. 2012. Available online: https://land.copernicus.eu/pan-european/corine-land-cover/clc-2012 (accessed on 12 April 2018).
- IBM. IBM SPSS Statistics 21 Core System User’s Guide; International Business Machines Corporation: Armonk, NY, USA, 2012. [Google Scholar]
- ter Braak, C.J.F.; Šmilauer, P. Canoco Reference Manual and User’s Guide: Software for Ordination; Version 5.0; Microcomputer Power: Houston, TX, USA, 2012; p. 496. [Google Scholar]
- Miltner, R.J.; Rankin, A.E.T. Primary nutrients and the biotic integrity of rivers and streams. Freshw. Biol. 1998, 40, 145–158. [Google Scholar] [CrossRef]
- O’Hare, M.T.; Clarke, R.T.; Bowes, M.J.; Cailes, C.; Henville, P.; Bissett, N.; McGahey, C.; Neal, M. Eutrophication impacts on a river macrophyte. Aquat. Bot. 2010, 92, 173–178. [Google Scholar] [CrossRef]
- D’Aiuto, P.E.; Makarewicz, J.C.; Bosch, I. The impact of stream nutrient loading on macrophytes and metaphyton in Conesus Lake, USA. SIL Proc. 1922–2010 2006, 29, 1373–1376. [Google Scholar] [CrossRef]
- Mebane, C.A.; Simon, N.S.; Maret, T.R. Linking nutrient enrichment and streamflow to macrophytes in agricultural streams. Hydrobiologia 2014, 722, 143–158. [Google Scholar] [CrossRef]
- Hill, B.H. Uptake and release of nutrients by aquatic macrophytes. Aquat. Bot. 1979, 7, 87–93. [Google Scholar] [CrossRef]
- Bornette, G.; Puijalon, S. Response of aquatic plants to abiotic factors: A review. Aquat. Sci. 2011, 73, 1–14. [Google Scholar] [CrossRef]
- O’Hare, M.T.; Baattrup-Pedersen, A.; Baumgarte, I.; Freeman, A.; Gunn, I.D.M.; Lázár, A.N.; Sinclair, R.; Wade, A.J.; Bowes, M.J. Responses of Aquatic Plants to Eutrophication in Rivers: A Revised Conceptual Model. Front. Plant Sci. 2018, 9, 451. [Google Scholar] [CrossRef]
- Larcher, W. Physiological Plant Ecology, 4th ed.; Springer: Berlin, Germany, 2003; p. 513. [Google Scholar]
- Fabris, M.; Schneider, S.; Melzer, A. Macrophyte-based bioindication in rivers—A comparative evaluation of the reference index (RI) and the trophic index of macrophytes (TIM). Limnologica 2009, 39, 40–55. [Google Scholar] [CrossRef]
- Squires, M.M.; Lesack, L.F.W. Benthic algal response to pulsed versus distributed inputs of sediments and nutrients in a Mackenzie Delta lake. J. N. Am. Benthol. Soc. 2001, 20, 369–384. [Google Scholar] [CrossRef]
- Veraart, A.J.; Romaní, A.M.; Tornés, E.; Sabater, S. Algal response to nutrient enrichment in forested oligotrophic stream. J. Phycol. 2008, 44, 564–572. [Google Scholar] [CrossRef]
- Hutchinson, G.E. A Treatise on Limnology. Volume III. Limnological Botany; John Wiley & Sons: New York, NY, USA, 1975; p. 660. [Google Scholar]
- Haslam, S.M. River Plants: The Macrophytic Vegetation of Watercourses; Cambridge University Press: Cambridge, UK, 2006; p. 450. [Google Scholar]
- Barko, J.W.; James, W.F. Effects of Submerged Aquatic Macrophytes on Nutrient Dynamics, Sedimentation, and Resuspension. In The Structuring Role of Submerged Macrophytes in Lakes; Springer: New York, NY, USA, 1998; pp. 197–214. [Google Scholar]
- Thiébaut, G. Phosphorus and aquatic plants. In The Ecophysiology of Plant-Phosphorus Interactions; White, P.J., Hammond, J.P., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 31–49. [Google Scholar]
- Sponseller, R.A.; Benfield, E.F.; Valett, H.M. Relationships between land use, spatial scale and stream macroinvertebrate communities. Freshw. Biol. 2001, 46, 1409–1424. [Google Scholar] [CrossRef]
- Kovalenko, K.E.; Brady, V.J.; Brown, T.N.; Ciborowski, J.J.H.; Danz, N.P.; Gathman, J.P.; Host, G.E.; Howe, R.W.; Johnson, L.B.; Niemi, G.J.; et al. Congruence of community thresholds in response to anthropogenic stress in Great Lakes coastal wetlands. Freshw. Sci. 2014, 33, 958–971. [Google Scholar] [CrossRef]
- Mackay, S.; Marsh, N. The Response of Aquatic Macrophytes to Riparian Shading in a Stream Rehabilitation Site; Monash University: Melbourne, Australia, 2005; p. 41. [Google Scholar]
- Giling, D.P.; Reich, P.; Thompson, R.M. Riparian vegetation removal alters consumer—Resource stoichiometry in an Australian lowland stream. Mar. Freshw. Res. 2012, 63, 1–8. [Google Scholar] [CrossRef]
- Brazner, J.C.; Danz, N.P.; Trebitz, A.S.; Niemi, G.J.; Regal, R.R.; Hollenhorst, T.; Host, G.E.; Reavie, E.D.; Brown, T.N.; Hanowski, J.A.M.; et al. Responsiveness of Great Lakes wetland indicators to human disturbances at multiple spatial scales: A multi-assemblage assessment. J. Great Lakes Res. 2007, 33, 42–66. [Google Scholar] [CrossRef]
- Birk, S.; Bonne, W.; Borja, A.; Brucet, S.; Courrat, A.; Poikane, S.; Solimini, A.; van de Bund, W.; Zampoukas, N.; Hering, D. Three hundred ways to assess Europe’s surface waters: An almost complete overview of biological methods to implement the Water Framework Directive. Ecol. Indic. 2012, 18, 31–41. [Google Scholar] [CrossRef]
- Strayer, D.L.; Lutz, C.; Malcom, H.M.; Munger, K.; Shaw, W.H. Invertebrate communities associated with a native (Vallisneria americana) and an alien (Trapa natans) macrophyte in a large river. Freshw. Biol. 2003, 48, 1938–1949. [Google Scholar] [CrossRef]
- Kuhar, U.; Gregorc, T.; Renčelj, M.; Šraj-Kržič, N.; Gaberščik, A. Distribution of macrophytes and condition of the physical environment of streams flowing through agricultural landscape in north-eastern Slovenia. Limnologica 2007, 37, 146–154. [Google Scholar] [CrossRef]


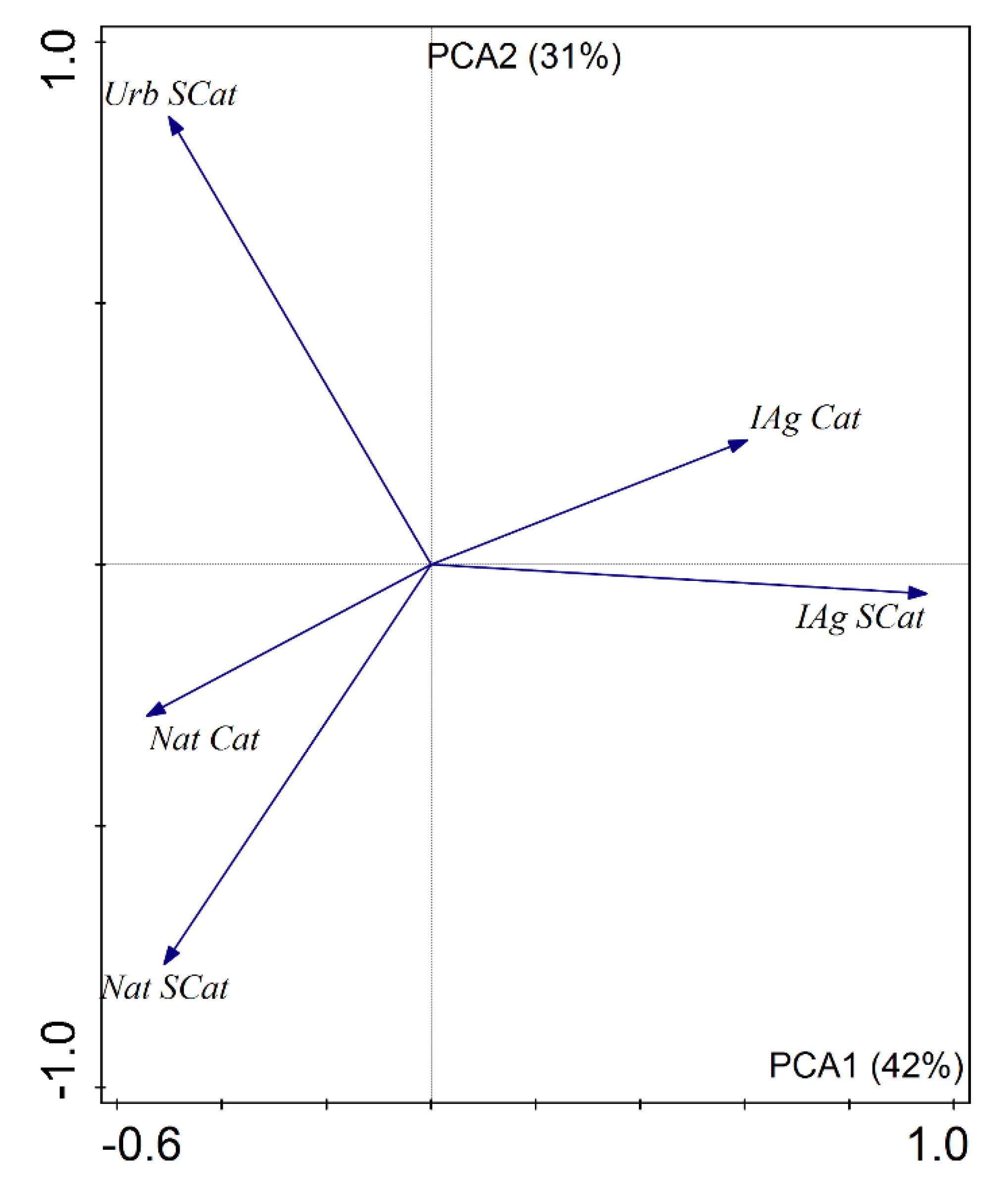
| Variable | Code | Mean | Median | Minimum | Maximum |
|---|---|---|---|---|---|
| Total nitrogen—maximum | TN max | 2.4 | 1.8 | 0.7 | 7.7 |
| Total nitrogen—annual mean | TN mean | 1.7 | 1.4 | 0.5 | 6.5 |
| Total nitrogen—summer mean | TN Smean | 1.6 | 1.2 | 0.4 | 7.5 |
| Ammonium—maximum | NH4 max | 0.3 | 0.1 | 0.0 | 3.2 |
| Ammonium—annual mean | NH4 mean | 0.1 | 0.1 | 0.0 | 1.5 |
| Ammonium—summer mean | NH4 Smean | 0.1 | 0.0 | 0.0 | 3.2 |
| Nitrite—maximum | NO2 max | 0.1 | 0.1 | 0.0 | 0.8 |
| Nitrite—annual mean | NO2 mean | 0.1 | 0.0 | 0.0 | 0.3 |
| Nitrite—summer mean | NO2 Smean | 0.1 | 0.0 | 0.0 | 0.8 |
| Nitrate—maximum | NO3 max | 8.6 | 6.7 | 2.2 | 32.0 |
| Nitrate—annual mean | NO3 mean | 6.1 | 5.0 | 1.2 | 26.7 |
| Nitrate—summer mean | NO3 Smean | 4.8 | 4.0 | 0.5 | 28.6 |
| Total phosphorous—maximum | TP max | 0.5 | 0.3 | 0.1 | 3.1 |
| Total phosphorous—annual mean | TP mean | 0.3 | 0.2 | 0.0 | 1.1 |
| Total phosphorous—summer mean | TP Smean | 0.3 | 0.2 | 0.0 | 1.8 |
| Orthophosphate—maximum | PO4 max | 0.2 | 0.1 | 0.0 | 2.0 |
| Orthophosphate—annual mean | PO4 mean | 0.1 | 0.1 | 0.0 | 0.8 |
| Orthophosphate—summer mean | PO4 Smean | 0.1 | 0.1 | 0.0 | 1.1 |
| CLC Natural—subcatchment | Nat SCat | 34.1 | 35.1 | 0.0 | 95.4 |
| CLC Natural—catchment | Nat Cat | 57.9 | 64.1 | 26.2 | 93.2 |
| CLC Intensive agriculture—subcatchment | IAg SCat | 40.7 | 39.4 | 0.0 | 100.0 |
| CLC Intensive agriculture—catchment | IAg Cat | 26.2 | 18.7 | 3.3 | 58.8 |
| CLC Extensive agriculture—subcatchment | EAg SCat | 11.9 | 9.2 | 0.0 | 39.0 |
| CLC Extensive agriculture—catchment | EAg Cat | 13.5 | 11.9 | 2.7 | 30.0 |
| CLC Urban—subcatchment | Urb SCat | 13.2 | 0.2 | 0.0 | 98.2 |
| CLC Urban—catchment | Urb Cat | 2.5 | 2.8 | 0.0 | 7.0 |
| TP Mean (mg/L) | N |
|---|---|
| 0.1 | 29 |
| 0.2 | 36 |
| 0.3 | 40 |
| 0.4 | 50 |
| 0.5 | 60 |
| 1.1 | 70 |
| CCA Option Code | Stressor Group | Forward Selection Option |
|---|---|---|
| N | Nitrogen | automatic |
| P | Phosphorous | automatic |
| Nut | Nitrogen and Phosphorous | automatic |
| P&N | Nitrogen and Phosphorous | manual |
| Land | Land Use | automatic |
| NutLand | Nutrients and Land use | automatic |
| LandNut | Nutrients and Land use | manual |
| Variable | RMI EQR | TI EQR |
|---|---|---|
| Total nitrogen—maximum | 0.21 | −0.51 ** |
| Total nitrogen—annual mean | 0.19 | −0.49 ** |
| Total nitrogen—summer mean | 0.30 * | −0.40 ** |
| Ammonium—maximum | 0.16 | −0.50 ** |
| Ammonium—annual mean | 0.15 | −0.57 ** |
| Ammonium—summer mean | 0.09 | −0.51 ** |
| Nitrite—maximum | −0.07 | −0.49 ** |
| Nitrite—annual mean | −0.03 | −0.58 ** |
| Nitrite—summer mean | −0.06 | −0.59 ** |
| Nitrate—maximum | 0.09 | −0.33 ** |
| Nitrate—annual mean | 0.07 | −0.37 ** |
| Nitrate—summer mean | 0.06 | −0.22 |
| Total phosphorous—maximum | −0.03 | −0.40 ** |
| Total phosphorous—annual mean | 0.05 | −0.40 ** |
| Total phosphorous—summer mean | 0.13 | −0.40 ** |
| Orthophosphate—maximum | −0.07 | −0.34 ** |
| Orthophosphate—annual mean | −0.01 | −0.37 ** |
| Orthophosphate—summer mean | 0.06 | −0.37 ** |
| Stressor Gradient | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Environmental Variable | Variable Group | λ1 (%) | p-Value | LandNut | NutLand | P&N | Nut | N | P | Land | |||||||
| λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | ||||
| Nitrite—annual mean | Nutrients | 5.3 | 0.001 | 11.8 | 0.001 | 11.8 | 0.001 | 7.3 | 0.001 | 15.2 | 0.001 | 20.3 | 0.001 | ||||
| Total nitrogen—maximum | Nutrients | 4.9 | 0.001 | 7.8 | 0.001 | 6.8 | 0.001 | 10.1 | 0.001 | 13.5 | 0.001 | ||||||
| Total nitrogen—annual mean | Nutrients | 4.9 | 0.001 | ||||||||||||||
| Nitrate—maximum | Nutrients | 4.8 | 0.001 | ||||||||||||||
| Nitrite—maximum | Nutrients | 4.7 | 0.001 | 4.1 | 0.036 | 4.2 | 0.033 | 5.3 | 0.034 | 5.4 | 0.03 | 7.3 | 0.026 | ||||
| Nitrate—annual mean | Nutrients | 4.7 | 0.001 | 6.6 | 0.001 | 6.5 | 0.001 | 6.8 | 0.007 | 8.3 | 0.001 | 11.1 | 0.001 | ||||
| CLC Intensive agriculture—catchment | Land Use | 4.6 | 0.001 | 8 | 0.001 | 4.3 | 0.009 | 30.3 | 0.001 | ||||||||
| Total phosphorous—annual mean | Nutrients | 4.6 | 0.001 | 13 | 0.001 | 32.4 | 0.001 | ||||||||||
| CLC Natural—catchment | Land Use | 4.5 | 0.001 | ||||||||||||||
| Orthophosphate—maximum | Nutrients | 4.2 | 0.001 | 4.3 | 0.025 | 8.3 | 0.001 | 20.7 | 0.001 | ||||||||
| Total phosphorous—summer mean | Nutrients | 4.2 | 0.001 | 4.1 | 0.031 | 5.8 | 0.012 | 14.6 | 0.018 | ||||||||
| Orthophosphate—annual mean | Nutrients | 4 | 0.001 | ||||||||||||||
| Ammonium—annual mean | Nutrients | 4 | 0.001 | 4.7 | 0.011 | 5.3 | 0.001 | 6 | 0.007 | 6.8 | 0.001 | 9.2 | 0.001 | ||||
| Total phosphorous—maximum | Nutrients | 3.8 | 0.001 | 6.3 | 0.004 | 15.7 | 0.007 | ||||||||||
| CLC Urban—catchment | Land Use | 3.7 | 0.001 | 5.1 | 0.002 | 4.5 | 0.013 | 21.6 | 0.001 | ||||||||
| Orthophosphate—summer mean | Nutrients | 3.5 | 0.001 | ||||||||||||||
| Nitrite—summer mean | Nutrients | 3.2 | 0.001 | ||||||||||||||
| Total nitrogen—summer mean | Nutrients | 3.2 | 0.001 | ||||||||||||||
| Ammonium—maximum | Nutrients | 3 | 0.001 | ||||||||||||||
| CLC Intensive agriculture—subcatchment | Land Use | 2.8 | 0.001 | 4 | 0.042 | 12.6 | 0.037 | ||||||||||
| CLC Natural—subcatchment | Land Use | 2.6 | 0.005 | 5 | 0.005 | 4.7 | 0.011 | 17.6 | 0.001 | ||||||||
| Nitrate—summer mean | Nutrients | 2.4 | 0.017 | ||||||||||||||
| Ammonium—summer mean | Nutrients | 2.3 | 0.011 | ||||||||||||||
| CLC Urban—subcatchment | Land Use | 2.1 | 0.065 | ||||||||||||||
| CLC Extensive agriculture—catchment | Land Use | 1.8 | 0.102 | ||||||||||||||
| CLC Extensive agriculture—subcatchment | Land Use | 1.8 | 0.129 | ||||||||||||||
| Environmental Variable | Variable Group | Stressor Gradient | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| λ1 (%) | p-Value | LandNut | NutLand | P&N | Nut | N | P | Land | |||||||||
| λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | λa (%) | p-Value | ||||
| Nitrite—annual mean | Nutrients | 6.5 | 0.001 | 14.8 | 0.001 | 14.8 | 0.001 | 10.0 | 0.001 | 18.9 | 0.001 | 25 | 0.001 | ||||
| CLC Intensive agriculture—catchment | Land Use | 5.9 | 0.001 | 10.6 | 0.001 | 6.0 | 0.001 | 38.2 | 0.001 | ||||||||
| CLC Natural—catchment | Land Use | 5.9 | 0.001 | 4.4 | 0.010 | 4.5 | 0.002 | ||||||||||
| Total nitrogen—annual mean | Nutrients | 5.8 | 0.001 | ||||||||||||||
| Total phosphorous—annual mean | Nutrients | 5.8 | 0.001 | 4.4 | 0.009 | 7.1 | 0.001 | 16.9 | 0.001 | 9.0 | 0.001 | 43.7 | 0.001 | ||||
| Nitrate—annual mean | Nutrients | 5.6 | 0.001 | 5.2 | 0.001 | 4.3 | 0.016 | 4.9 | 0.048 | 5.5 | 0.013 | 11.7 | 0.001 | ||||
| Total nitrogen—maximum | Nutrients | 5.4 | 0.001 | 9.0 | 0.001 | ||||||||||||
| Nitrate—maximum | Nutrients | 5.2 | 0.001 | 3.9 | 0.018 | 5.8 | 0.001 | 7.3 | 0.001 | 7.4 | 0.001 | 8.3 | 0.001 | ||||
| Nitrite—maximum | Nutrients | 5.2 | 0.001 | ||||||||||||||
| Total phosphorous—summer mean | Nutrients | 5.1 | 0.001 | ||||||||||||||
| Ammonium—annual mean | Nutrients | 5.0 | 0.001 | ||||||||||||||
| Orthophosphate—annual mean | Nutrients | 4.9 | 0.001 | 5.4 | 0.028 | 14.1 | 0.030 | ||||||||||
| Orthophosphate—summer mean | Nutrients | 4.7 | 0.001 | ||||||||||||||
| Total phosphorous—maximum | Nutrients | 4.6 | 0.001 | 3.7 | 0.036 | 3.8 | 0.038 | 5.3 | 0.036 | 4.9 | 0.043 | 13.7 | 0.033 | ||||
| Orthophosphate—maximum | Nutrients | 4.4 | 0.001 | ||||||||||||||
| Total nitrogen—summer mean | Nutrients | 4.1 | 0.001 | ||||||||||||||
| Nitrite—summer mean | Nutrients | 4.1 | 0.001 | ||||||||||||||
| Ammonium—maximum | Nutrients | 4.0 | 0.001 | ||||||||||||||
| CLC Urban—catchment | Land Use | 3.7 | 0.001 | 21.1 | 0.001 | ||||||||||||
| Nitrate—summer mean | Nutrients | 3.5 | 0.001 | 4.3 | 0.015 | 4.9 | 0.003 | 6.3 | 0.002 | 6.3 | 0.003 | 7.5 | 0.01 | ||||
| Ammonium—summer mean | Nutrients | 2.3 | 0.007 | ||||||||||||||
| CLC Extensive agriculture—catchment | Land Use | 2.1 | 0.027 | ||||||||||||||
| CLC Intensive agriculture—subcatchment | Land Use | 1.9 | 0.05 | 3.9 | 0.028 | 3.9 | 0.017 | 13.1 | 0.010 | ||||||||
| CLC Natural—subcatchment | Land Use | 1.8 | 0.089 | ||||||||||||||
| CLC Extensive agriculture—subcatchment | Land Use | 1.5 | 0.319 | ||||||||||||||
| CLC Urban—catchment | Land Use | 1.4 | 0.453 | ||||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbanič, G.; Debeljak, B.; Kuhar, U.; Germ, M.; Gaberščik, A. Responses of Freshwater Diatoms and Macrophytes Rely on the Stressor Gradient Length across the River Systems. Water 2021, 13, 1814. https://doi.org/10.3390/w13131814
Urbanič G, Debeljak B, Kuhar U, Germ M, Gaberščik A. Responses of Freshwater Diatoms and Macrophytes Rely on the Stressor Gradient Length across the River Systems. Water. 2021; 13(13):1814. https://doi.org/10.3390/w13131814
Chicago/Turabian StyleUrbanič, Gorazd, Barbara Debeljak, Urška Kuhar, Mateja Germ, and Alenka Gaberščik. 2021. "Responses of Freshwater Diatoms and Macrophytes Rely on the Stressor Gradient Length across the River Systems" Water 13, no. 13: 1814. https://doi.org/10.3390/w13131814
APA StyleUrbanič, G., Debeljak, B., Kuhar, U., Germ, M., & Gaberščik, A. (2021). Responses of Freshwater Diatoms and Macrophytes Rely on the Stressor Gradient Length across the River Systems. Water, 13(13), 1814. https://doi.org/10.3390/w13131814








