Effects of Spatial Resolution on the Satellite Observation of Floating Macroalgae Blooms
Abstract
:1. Introduction
2. Data and Methods
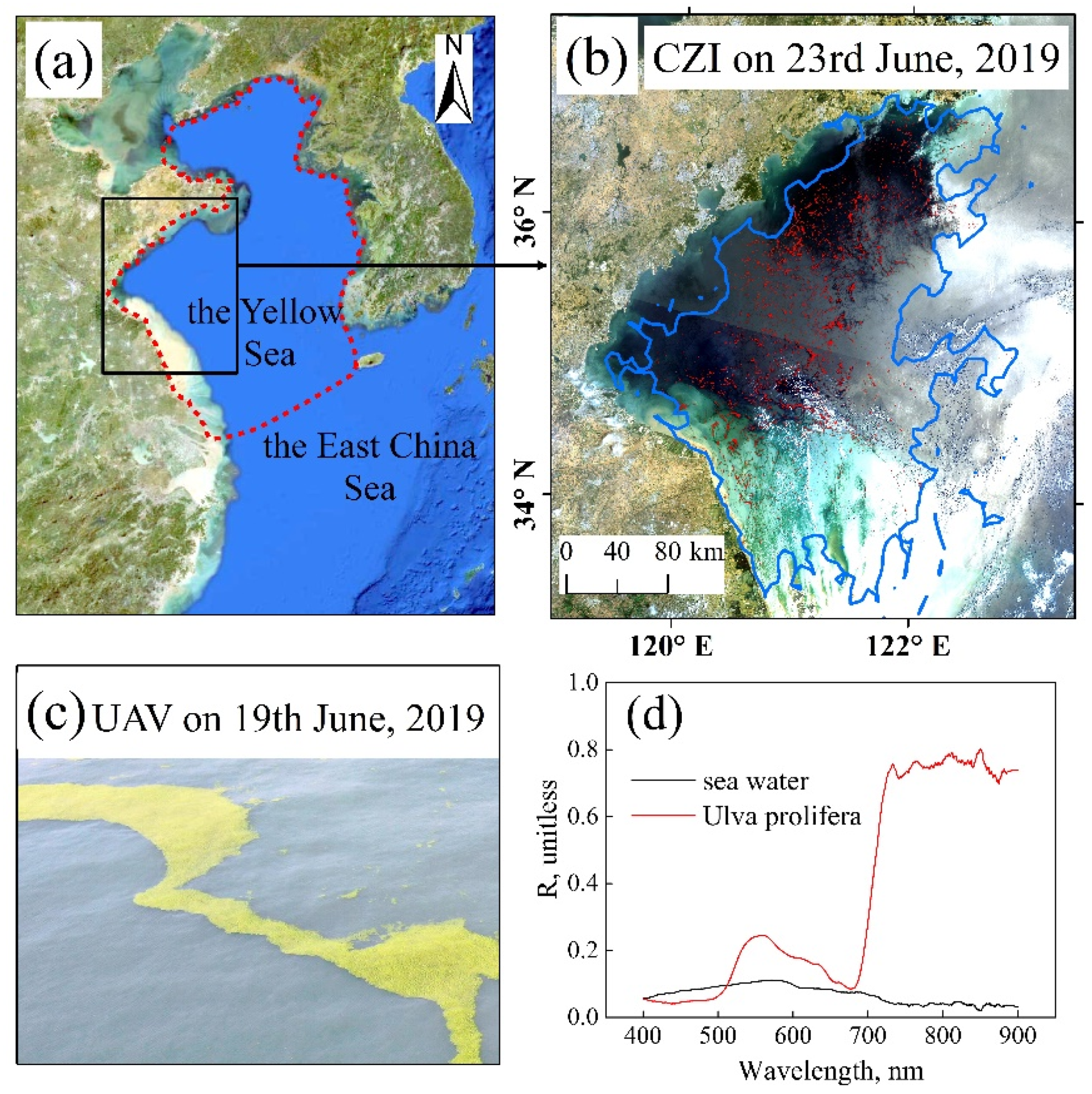
2.1. The Study Area
2.2. Satellite Images and Data Processing
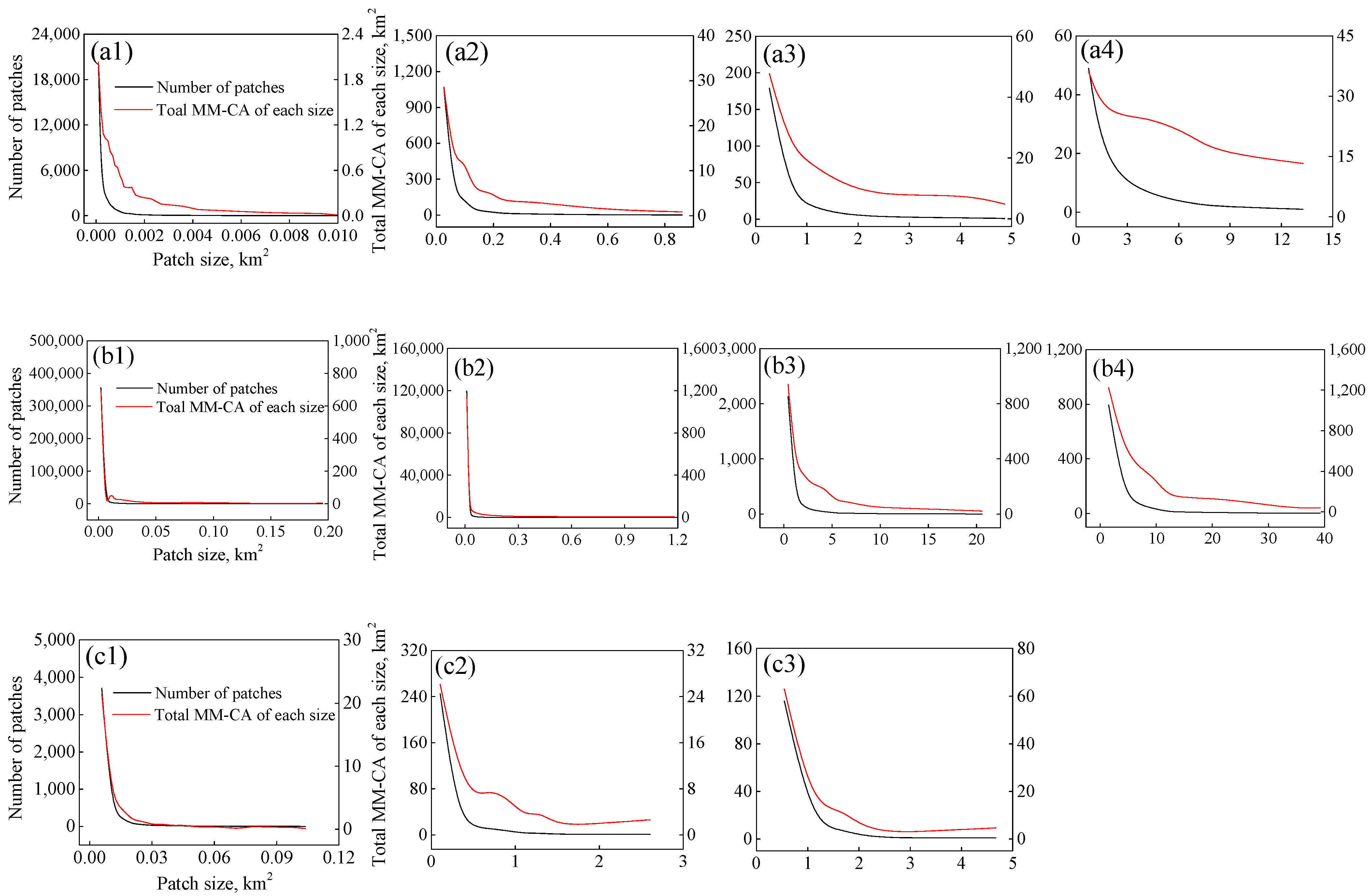
2.3. Estimation of the Area of Floating Macroalgae
3. Results and Analysis
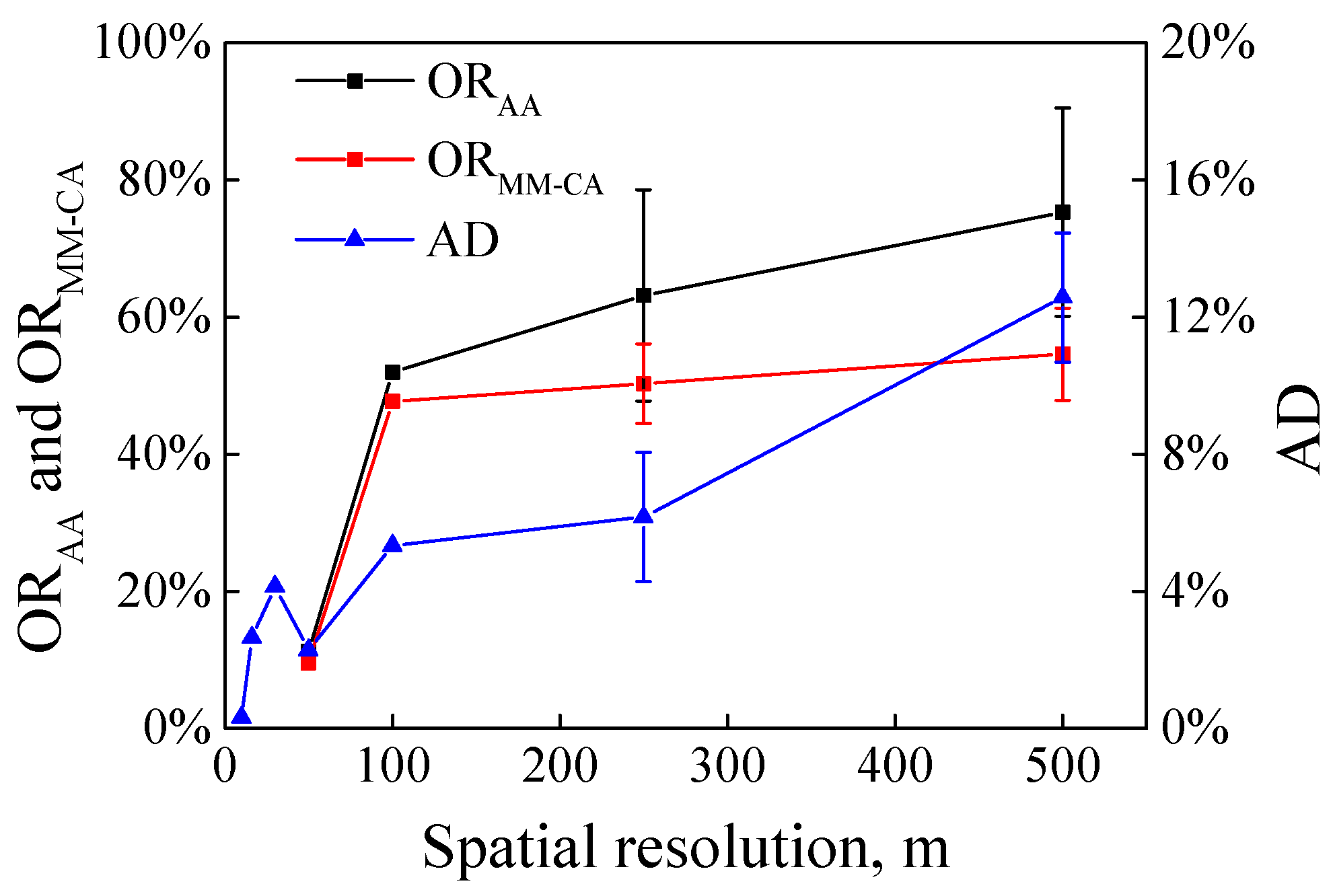
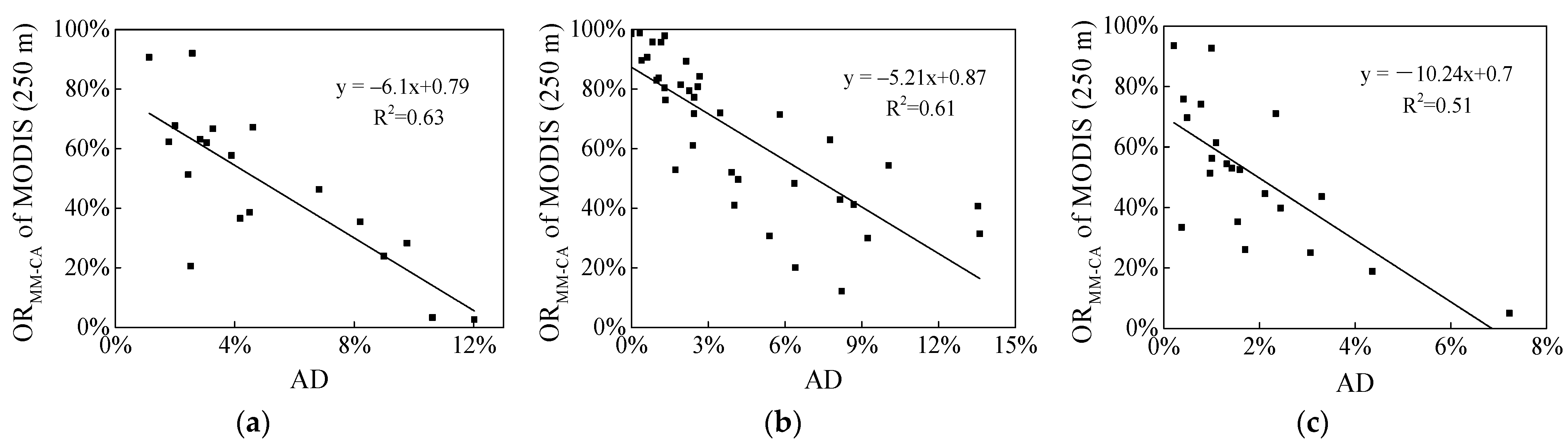
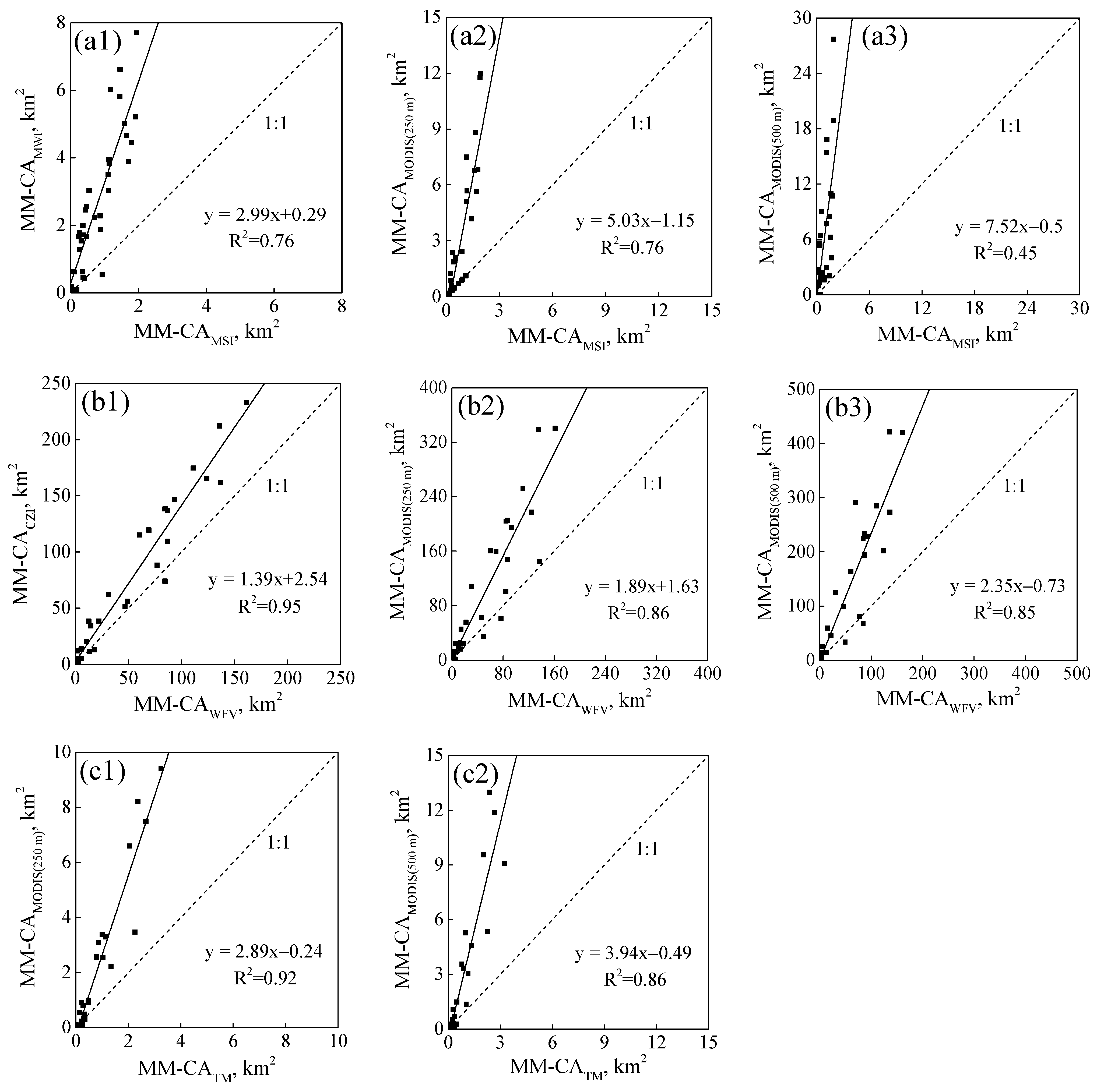
3.1. Variations of MM-CA, AA and AD Derived from Different Resolution Images
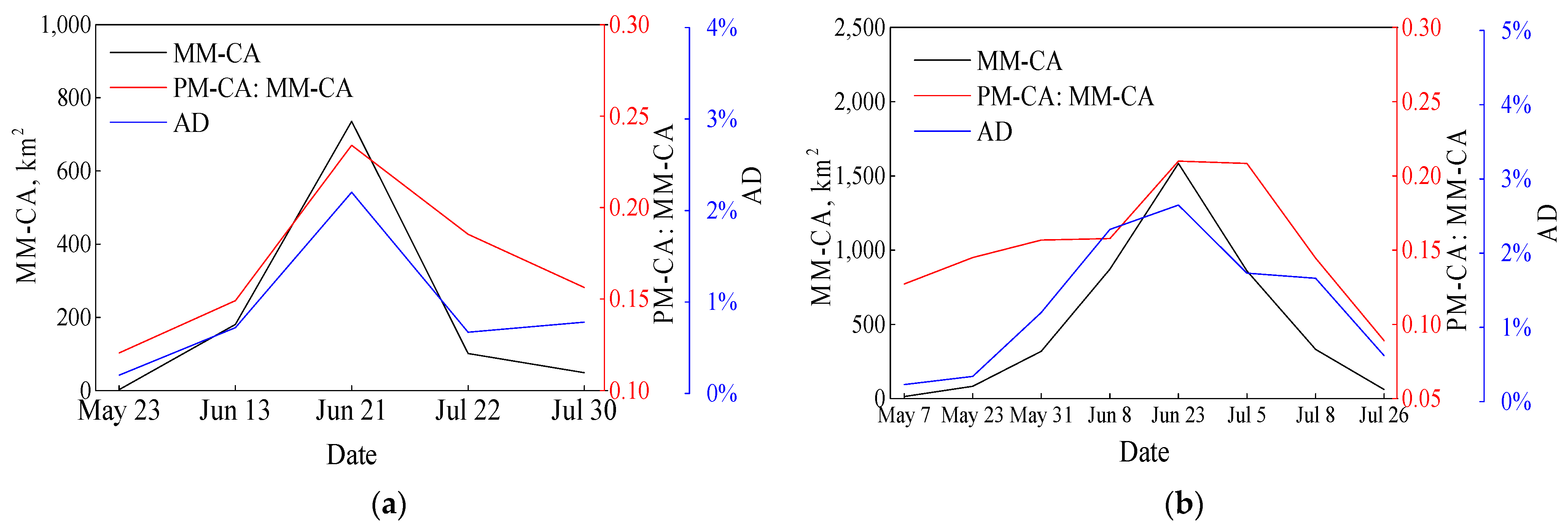
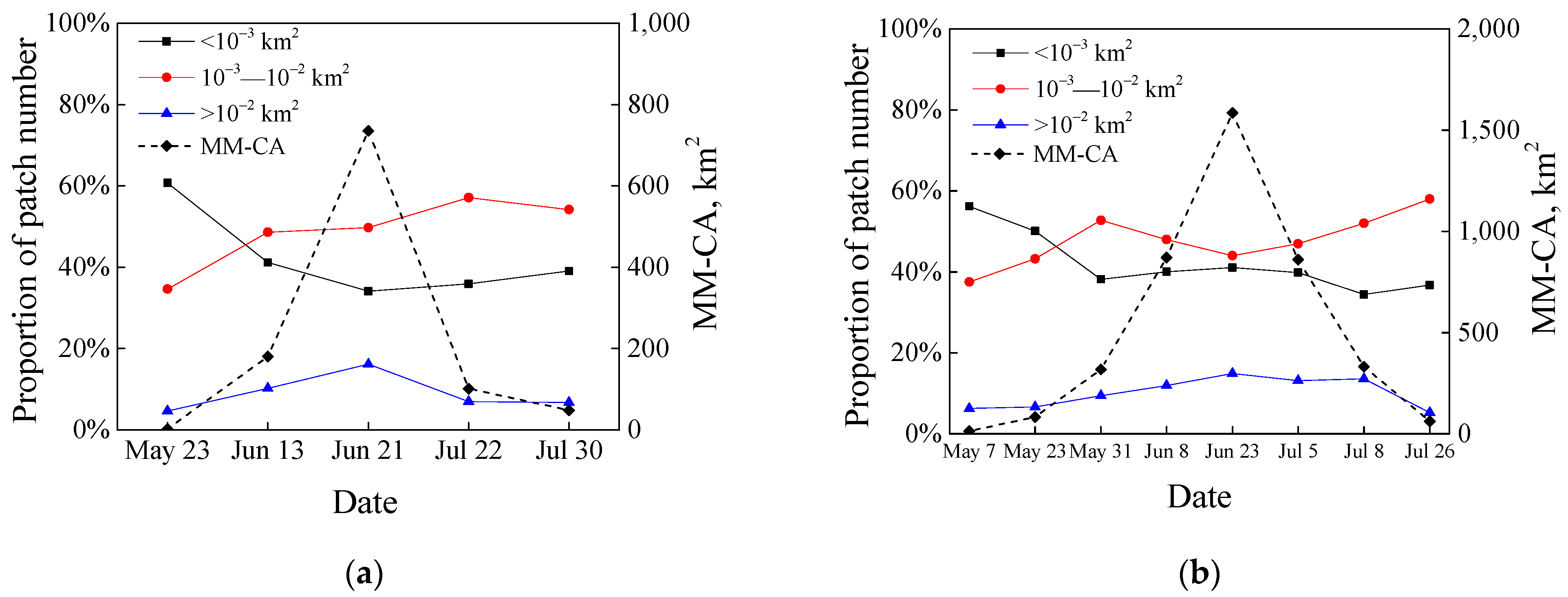
3.2. Seasonal Changes in Floating Macroalgae Coverages Estimated by High Resolution Images
4. Conclusions and Prospect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gower, J.; Hu, C.M.; Borstad, G.; King, S. Ocean color satellites show extensive lines of floating sargassum in the Gulf of Mexico. IEEE Trans. Geosci. Remote Sens. 2006, 44, 3619–3625. [Google Scholar] [CrossRef]
- Smetacek, V.; Zingone, A. Green and golden seaweed tides on the rise. Nature 2013, 504, 84–88. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.Q.; Hu, C.M.; Barnes, B.B.; Mitchum, G.; Lapointe, B.; Montoya, J.P. The great Atlantic Sargassum belt. Science 2019, 365, 83–87. [Google Scholar] [CrossRef]
- Gower, J.; King, S. Seaweed, seaweed everywhere. Science 2019, 365, 27. [Google Scholar] [CrossRef] [PubMed]
- Lyons, D.A.; Arvanitidis, C.; Blight, A.J.; Chatzinikolaou, E.; Guy-Haim, T.; Kotta, J.; Orav-Kotta, H.; Queiros, A.M.; Rilov, G.; Somerfield, P.J.; et al. Macroalgal blooms alter community structure and primary productivity in marine ecosystems. Glob. Chang. Biol. 2014, 20, 2712–2724. [Google Scholar] [CrossRef]
- Son, Y.B.; Min, J.E.; Ryu, J.H. Detecting Massive Green Algae (Ulva prolifera) Blooms in the Yellow Sea and East China Sea using Geostationary Ocean Color Imager (GOCI) Data. Ocean Sci. J. 2012, 47, 359–375. [Google Scholar] [CrossRef]
- Hu, L.B.; Zeng, K.; Hu, C.M.; He, M.X. On the remote estimation of Ulva prolifera areal coverage and biomass. Remote Sens. Environ. 2019, 223, 194–207. [Google Scholar] [CrossRef]
- Wang, W.C.; Zhang, G.T.; Sun, X.X.; Zhang, F.; Zhang, X. Temporal variability in zooplankton community in the western Yellow Sea and its possible links to green tides. PeerJ 2019, 7, e6641. [Google Scholar] [CrossRef]
- Liu, D.Y.; Keesing, J.K.; Xing, Q.G.; Shi, P. World’s largest macroalgal bloom caused by expansion of seaweed aquaculture in China. Mar. Pollut. Bull. 2009, 58, 888–895. [Google Scholar] [CrossRef]
- Hu, C.M.; Li, D.Q.; Chen, C.S.; Ge, J.Z.; Muller-Karger, F.E.; Liu, J.P.; Yu, F.; He, M.X. On the recurrent Ulva prolifera blooms in the Yellow Sea and East China Sea. J. Geophys. Res. Ocean. 2010, 115. [Google Scholar] [CrossRef] [Green Version]
- Xing, Q.G.; Wu, L.L.; Tian, L.Q.; Cui, T.W.; Li, L.; Kong, F.Z.; Gao, X.L.; Wu, M.Q. Remote sensing of early-stage green tide in the Yellow Sea for floating-macroalgae collecting campaign. Mar. Pollut. Bull. 2018, 133, 150–156. [Google Scholar] [CrossRef]
- Xing, Q.G.; An, D.Y.; Zheng, X.Y.; Wei, Z.N.; Wang, X.H.; Li, L.; Tian, L.Q.; Chen, J. Monitoring seaweed aquaculture in the Yellow Sea with multiple sensors for managing the disaster of macroalgal blooms. Remote Sens. Environ. 2019, 231, 111279. [Google Scholar] [CrossRef]
- Xing, Q.G.; Hu, C.M. Mapping macroalgal blooms in the Yellow Sea and East China Sea using HJ-1 and Landsat data: Application of a virtual baseline reflectance height technique. Remote Sens. Environ. 2016, 178, 113–126. [Google Scholar] [CrossRef]
- Xing, Q.G.; Guo, R.H.; Wu, L.L.; An, D.Y.; Cong, M.; Qin, S.; Li, X.R. High-Resolution Satellite Observations of a New Hazard of Golden Tides Caused by Floating Sargassum in Winter in the Yellow Sea. IEEE Geosci. Remote Sens. Lett. 2017, 14, 1815–1819. [Google Scholar] [CrossRef]
- Lu, T.; Lu, Y.C.; Hu, L.B.; Jiao, J.N.; Zhang, M.W.; Liu, Y.X. Uncertainty in the optical remote estimation of the biomass of Ulva prolifera macroalgae using MODIS imagery in the Yellow Sea. Opt. Express 2019, 27, 18620–18627. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.B.; Hu, C.M.; He, M.X. Remote estimation of biomass of Ulva prolifera macroalgae in the Yellow Sea. Remote Sens. Environ. 2017, 192, 217–227. [Google Scholar] [CrossRef]
- Qiu, Z.F.; Li, Z.X.; Bilal, M.; Wang, S.Q.; Sun, D.Y.; Chen, Y.L. Automatic method to monitor floating macroalgae blooms based on multilayer perceptron: Case study of Yellow Sea using GOCI images. Opt. Express 2018, 26, 26810–26829. [Google Scholar] [CrossRef]
- Harun-Al-Rashid, A.; Yang, C.S. Improved Detection of Tiny Macroalgae Patches in Korea Bay and Gyeonggi Bay by Modification of Floating Algae Index. Remote Sens. 2018, 10, 1478. [Google Scholar] [CrossRef] [Green Version]
- Xing, Q.G.; Zheng, X.Y.; Shi, P.; Hao, J.J.; Yu, D.F.; Liang, S.Z.; Liu, D.Y.; Zhang, Y.Z. Monitoring “Green Tide” in the Yellow Sea and the East China Sea Using Multi-Temporal and Multi-Source Remote Sensing Images. Spectrosc. Spectr. Anal. 2011, 31, 1644–1647. [Google Scholar]
- Xing, Q.G.; Hu, C.M.; Tang, D.L.; Tian, L.Q.; Tang, S.L.; Wang, X.H.; Lou, M.J.; Gao, X.L. World’s Largest Macroalgal Blooms Altered Phytoplankton Biomass in Summer in the Yellow Sea: Satellite Observations. Remote Sens. 2015, 7, 12297–12313. [Google Scholar] [CrossRef] [Green Version]
- Qi, L.; Hu, C.M. To what extent can Ulva and Sargassum be detected and separated in satellite imagery? Harmful Algae 2021, 103, 102001. [Google Scholar] [CrossRef]
- He, X.Q.; Bai, Y.; Wei, J.; Ding, J.; Shanmugam, P.; Wang, D.F.; Song, Q.J.; Huang, X.X. Ocean color retrieval from MWI onboard the Tiangong-2 Space Lab: Preliminary results. Opt. Express 2017, 25, 23955–23973. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.G.; Duan, H.T.; Song, Q.J.; Shen, M.; Ma, R.H.; Liu, D. Evaluation of the sensitivity of China’s next-generation ocean satellite sensor MWI onboard the Tiangong-2 space lab over inland waters. Int. J. Appl. Earth Obs. Geoinf. 2018, 71, 109–120. [Google Scholar]
- Wang, X.H.; Liu, J.Q.; Xing, Q.G.; Chen, Y.L. Monitoring of Porphyra Cultivation Dynamics in Lianyungang Based on Coastal Zone Imager (CZI). Mar. Sci. 2021, in press. [Google Scholar]
- Hu, C.M. A novel ocean color index to detect floating algae in the global oceans. Remote Sens. Environ. 2009, 113, 2118–2129. [Google Scholar] [CrossRef]
- Garcia, R.A.; Fearns, P.; Keesing, J.K.; Liu, D.Y. Quantification of floating macroalgae blooms using the scaled algae index. J. Geophys. Res. Ocean. 2013, 118, 26–42. [Google Scholar] [CrossRef] [Green Version]
- Son, Y.B.; Choi, B.J.; Kim, Y.H.; Park, Y.G. Tracing floating green algae blooms in the Yellow Sea and the East China Sea using GOCI satellite data and Lagrangian transport simulations. Remote Sens. Environ. 2015, 156, 21–33. [Google Scholar] [CrossRef]
- Shen, F.; Tang, R.G.; Sun, X.R.; Liu, D.Y. Simple methods for satellite identification of algal blooms and species using 10-year time series data from the East China Sea. Remote Sens. Environ. 2019, 235, 111484. [Google Scholar] [CrossRef]
- Zhang, H.L.; Qiu, Z.F.; Devred, E.; Sun, D.Y.; Wang, S.Q.; He, Y.J.; Yu, Y. A simple and effective method for monitoring floating green macroalgae blooms: A case study in the Yellow Sea. Opt. Express 2019, 27, 4528–4548. [Google Scholar] [CrossRef]
- Qi, L.; Hu, C.M.; Wang, M.Q.; Shang, S.L.; Wilson, C. Floating Algae Blooms in the East China Sea. Geophys. Res. Lett. 2017, 44, 11501–11509. [Google Scholar] [CrossRef]
- Xiao, Y.F.; Zhang, J.; Cui, T.W.; Gong, J.L.; Liu, R.J.; Chen, X.Y.; Liang, X.J. Remote sensing estimation of the biomass of floating Ulva prolifera and analysis of the main factors driving the interannual variability of the biomass in the Yellow Sea. Mar. Pollut. Bull. 2019, 140, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Zhang, H.Y.; Cheng, Y.C. Multi-sensor monitoring of Ulva prolifera blooms in the Yellow Sea using different methods. Front. Earth Sci. 2016, 10, 378–388. [Google Scholar] [CrossRef]
- Xing, Q.G.; Tosi, L.G.; Braga, F.; Gao, X.L.; Gao, M. Interpreting the progressive eutrophication behind the world’s largest macroalgal blooms with water quality and ocean color data. Nat. Hazards 2015, 78, 7–21. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Xing, Q.G.; Li, X.R.; Yu, D.F.; Zhang, J.; Zou, J.Q. Assessment of the Impacts From the World’s Largest Floating Macroalgae Blooms on the Water Clarity at the West Yellow Sea Using MODIS Data (2002–2016). IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2018, 11, 1397–1402. [Google Scholar] [CrossRef]
- Qi, L.; Hu, C.M.; Xing, Q.G.; Shang, S.L. Long-term trend of Ulva prolifera blooms in the western Yellow Sea. Harmful Algae 2016, 58, 35–44. [Google Scholar] [CrossRef]
- Cui, T.W.; Liang, X.J.; Gong, J.L.; Tong, C.; Xiao, Y.F.; Liu, R.J.; Zhang, X.; Zhang, J. Assessing and refining the satellite-derived massive green macro-algal coverage in the Yellow Sea with high resolution images. ISPRS J. Photogramm. Remote Sens. 2018, 144, 315–324. [Google Scholar] [CrossRef]
- Li, L.; Zheng, X.Y.; Wei, Z.N.; Zou, J.Q.; Xing, Q.G. A spectral-mixing model for estimating sub-pixel coverage of sea-surface floating macroalgae. Atmos. Ocean 2018, 56, 296–302. [Google Scholar] [CrossRef]
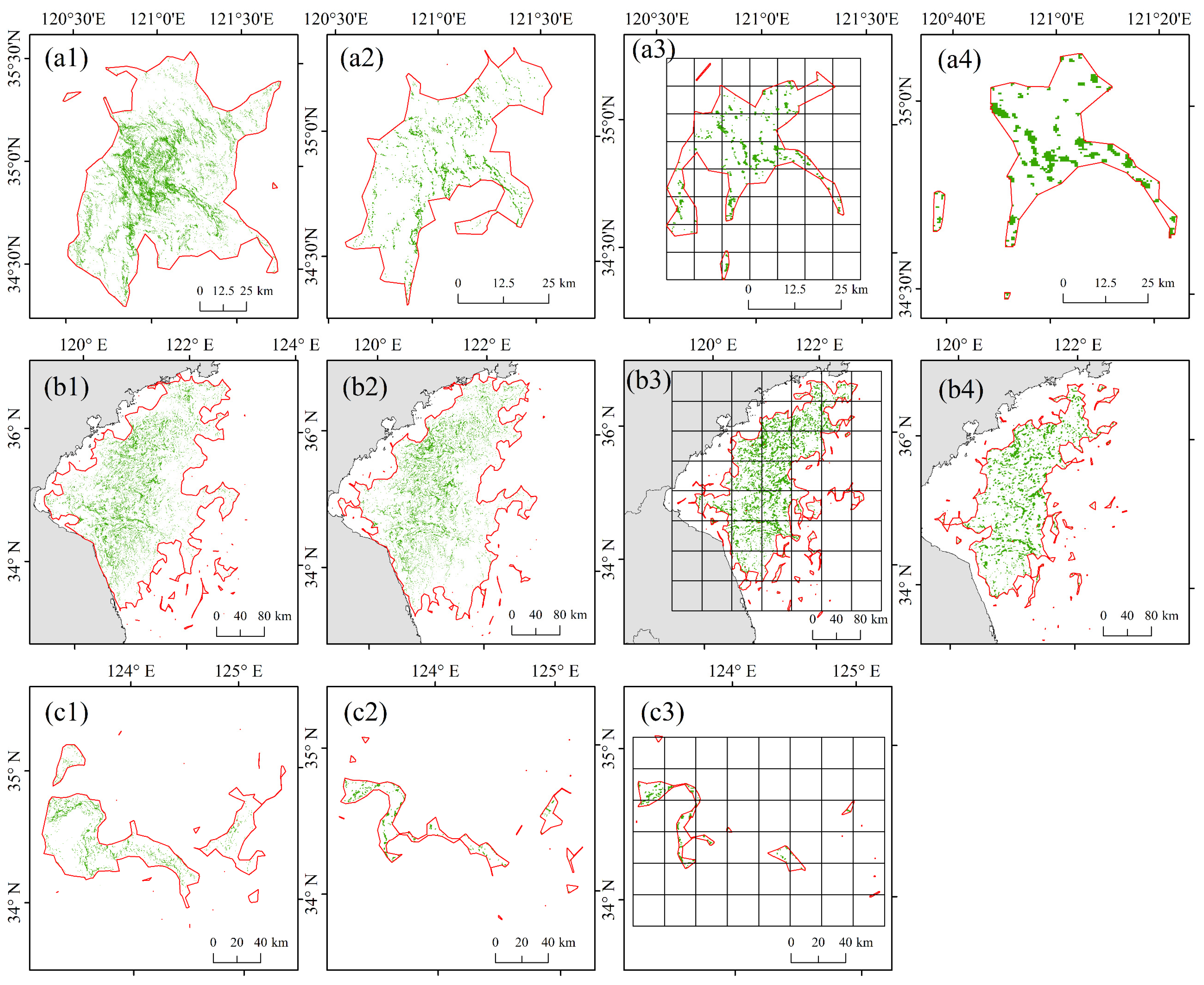
| Satellite Images | Sensing Date and Time | Spatial Resolution (m) |
|---|---|---|
| A1, Sentinel-2 MSI | 3 June 2018 10:36:51 | 10 |
| A2, Tiangong-2 MWI | 3 June 2018 14:30:33 | 100 |
| A3, Terra MODIS | 3 June 2018 11:10:00 | 250 |
| A4, Terra MODIS | 3 June 2018 11:10:00 | 500 |
| B1, GF-1 WFV | 23 June 2019 11:05:11 | 16 |
| B2, HY-1C CZI | 23 June 2019 11:43:25 | 50 |
| B3, Aqua MODIS | 23 June 2019 13:30:00 | 250 |
| B4, Aqua MODIS | 23 June 2019 13:30:00 | 500 |
| C1, Landsat-5 TM | 24 June 2009 10:06:30 | 30 |
| C2, Aqua MODIS | 24 June 2009 13:05:00 | 250 |
| C3, Aqua MODIS | 24 June 2009 13:05:00 | 500 |
| Satellite Images | MM-CA (km2) | AA (km2) | AD | ORAA | ORMM-CA |
|---|---|---|---|---|---|
| A1. Sentinel-2 MSI (10 m) | 27.13 | 8243.24 | 0.33% | - | - |
| A2. TG-2 MWI (100 m) | 93.17 | 4073.31 | 2.29% | 51.98% | 47.71% |
| A3. Terra MODIS (250 m) | 108.83 | 2038.39 | 5.34% | 75.33% | 58.04% |
| A4. Terra MODIS (500 m) | 158.07 | 1089.39 | 14.51% | 86.78% | 64.09% |
| B1. GF-1 WFV (16 m) | 1584.75 | 59,728.31 | 2.65% | - | - |
| B2. HY-1C CZI (50 m) | 2293.66 | 55,235.83 | 4.15% | 11.21% | 9.53% |
| B3. Aqua MODIS (250 m) | 3103.49 | 35,327.71 | 8.78% | 41.42% | 48.82% |
| B4. Aqua MODIS (500 m | 3644.28 | 27,658.22 | 13.18% | 53.84% | 49.33% |
| C1. Landsat-5 TM (30 m) | 29.26 | 6389.28 | 0.46% | - | - |
| C2. Aqua MODIS (250 m) | 78.34 | 1785.27 | 4.39% | 72.77% | 44.05% |
| C3. Aqua MODIS (500 m) | 96.01 | 956.84 | 10.03% | 85.3% | 50.34% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Xing, Q.; An, D.; Meng, L.; Zheng, X.; Jiang, B.; Liu, H. Effects of Spatial Resolution on the Satellite Observation of Floating Macroalgae Blooms. Water 2021, 13, 1761. https://doi.org/10.3390/w13131761
Wang X, Xing Q, An D, Meng L, Zheng X, Jiang B, Liu H. Effects of Spatial Resolution on the Satellite Observation of Floating Macroalgae Blooms. Water. 2021; 13(13):1761. https://doi.org/10.3390/w13131761
Chicago/Turabian StyleWang, Xinhua, Qianguo Xing, Deyu An, Ling Meng, Xiangyang Zheng, Bo Jiang, and Hailong Liu. 2021. "Effects of Spatial Resolution on the Satellite Observation of Floating Macroalgae Blooms" Water 13, no. 13: 1761. https://doi.org/10.3390/w13131761
APA StyleWang, X., Xing, Q., An, D., Meng, L., Zheng, X., Jiang, B., & Liu, H. (2021). Effects of Spatial Resolution on the Satellite Observation of Floating Macroalgae Blooms. Water, 13(13), 1761. https://doi.org/10.3390/w13131761






