Abstract
To date, the management of high-strength wastewater represents a serious problem. This work aims to evaluate the performance on chemical pollutants and on sludge production of one of the two full-scale thermophilic membrane bioreactors (ThMBRs) currently operational in Italy, based on monitoring data of the last two and a half years. Removal yields on COD, N-NOx, non-ionic and anionic surfactants (TAS and MBAS), increased with the input load up to 81.9%, 97.6%, 94.7%, and 98.4%, respectively. In the period of stability, a very low value of sludge production (0.052 kgVS kgCOD−1) was observed. Oxygen uptake rate (OUR) tests allowed us to exclude the possibility that mesophilic biomass generally exhibited any acute inhibition following contact with the aqueous residues (ARs), except for substrates that presented high concentrations of perfluoro alkyl substances (PFAS), cyanides and chlorides. In one case, nitrifying activity was partially inhibited by high chlorides and PFAS concentration, while in another the substrate determined a positive effect, stimulating the phenomenon of nitrification. Nitrogen uptake rate (NUR) tests highlighted the feasibility of reusing the organic carbon contained in the substrate as a source in denitrification, obtaining a value comparable with that obtained using the reference solution with methanol. Therefore, respirometric tests proved to be a valid tool to assess the acute effect of AR of ThMBR on the activity of mesophilic biomass in the case of recirculation.
1. Introduction
To date, the management of high-strength wastewater (WW) represents a very serious problem [1]. Conventional wastewater treatment plants (WWTPs) do not present suitable treatments for treating WW with high organic content and possible recalcitrant pollutants such as color [2,3], surfactants [4,5], solvents [6,7], perfluoro alkyl substances (PFAS) [8,9,10] and heavy metals [11,12]. These WWs must be treated in special treatment lines built on site or located in authorized WWTPs.
Chemical–physical treatments (e.g., coagulation–flocculation) are the most commonly used because of the recalcitrance of these pollutants which tend to inhibit the activity of the biomass with which they come into contact [13,14,15]. However, these treatments have significant disadvantages. For instance, coagulation–flocculation required the use of chemical reagents and produced large quantities of chemical sludge in which some of these pollutants (e.g., heavy metals) are concentrated [16,17]. This aspect causes an increase in costs and is against the principle of the circular economy of minimizing the use of resources and optimizing the recovery of materials [18,19,20].
For some years, it has been known that it is possible to biologically treat high-strength WW by exploiting the advantages provided by operating at thermophilic temperatures [21,22,23]. Recalcitrant pollutants can pass unaltered by the conventional active sludge (CAS) systems because they are not biologically degradable at mesophilic temperatures, e.g., perfluorooctanesulfonic acid (PFOS) [24] but also cause the inhibition of the biomass present [13,14,15]. However, the high resistance of thermophilic systems combined with the excellent degradative capacity of the thermophilic biomass make these treatments an excellent alternative to traditional physicochemical processes [25,26]. For instance, Pugazhendhi et al. [27] proved that thermophilic biological treatment can reduce the level of tetracycline in manure to about 96% in 72 h. In our previous study on a pilot plant, thermophilic aerobic treatments removed 10–56% and 85–96% of non-ionic and anionic surfactants (TAS and MBAS), respectively, starting from a total surfactant concentration up to 222 mg L−1 and 753 mg L−1 [28]. Furthermore, in thermophilic biological systems, the excess sludge is generally less than an order of magnitude compared to that produced in CAS systems [29]. This is a strong advantage considering the importance of minimizing the production of sewage sludge at the source [30,31] and the increasingly stringent legislation on the disposal of sludge, especially in agriculture [32,33,34].
The main disadvantage of thermophilic biological systems is the poor sedimentability of the sludge compared to CAS [22]. One of the possible solutions is to couple ultrafiltration (UF) membranes to the biological treatment [21]. A full-scale thermophilic membrane bioreactor (ThMBR), operating in aerobic conditions, for the treatment of high-strength WW has been operational in Italy for several years, adopting this solution [35,36].
In Italy, a second ThMBR become operational 2018. Data from July 2018 to December 2020 were analyzed and removal yields on COD, N-NOx, TAS, and MBAS in relation to the loading rate of pollutants were investigated. Sludge production has been calculated and compared with literature data of other mesophilic and thermophilic processes. Moreover, respirometric tests were used as tool to assess the effect on activity of mesophilic biomass of AR of ThMBR. Diverse WWs were separately treated in pilot ThMBR to selectively evaluate the influence of pollutants in ARs (such as cyanides, PFAS and chlorides) on oxygen uptake rate (OUR), ammonia uptake rate (AUR) and nitrate uptake rate (NUR) in the case of permeate recirculation in a mesophilic CAS system.
2. Materials and Methods
2.1. Thermophilic Membrane Bioreactor (ThMBR): Characteristics and Monitoring
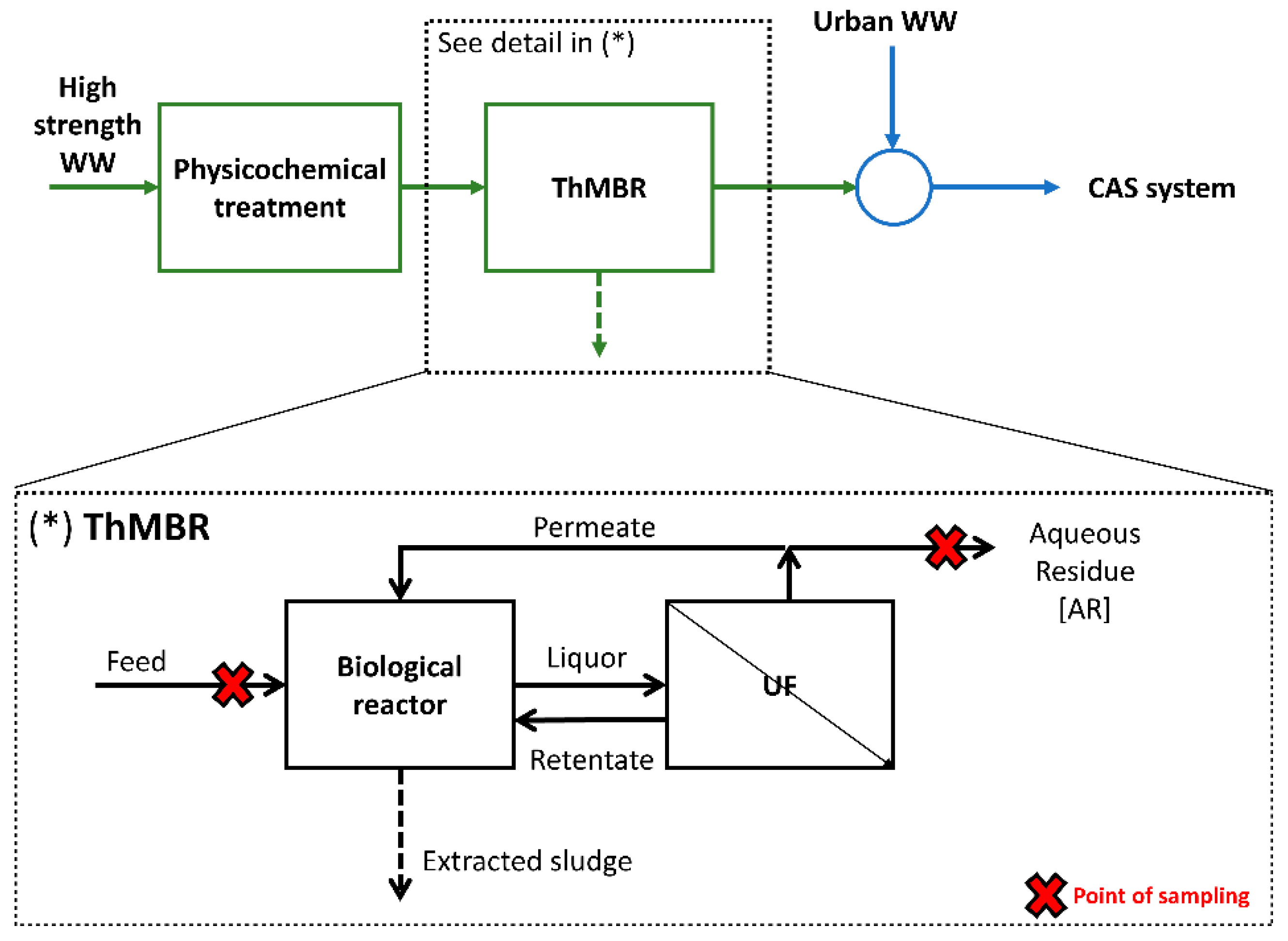
The full-scale ThMBR is located in a WWTP authorized to treat both high-strength and urban WW in Lombardy (Italy). High-strength WW was pre-treated with physicochemical process to effectively remove heavy metals and increase the biological treatability, and then by ThMBR (Figure 1). The ThMBR is composed by: (i) a biological reactor where pure oxygen is injected to maintain the O2 concentration equal to 1.5–3 mg L−1 (our previous results indicated a consumption equal to 1.1–1.2 kgO2 kgCODremoved−1 [25]), and (ii) a recirculation line where a UF unit (six vessels with 99 ceramic membranes; cut-off: 300 kDa) operating with 3–5 bar of pressure is located. The total volume of the system is 1700 m3. Temperature (48–50 °C) is maintained thanks to exothermic reactions produced by biomass during the degradation of the organic substance, and it is controlled by a heating/cooling system. To date, only one other ThMBR with similar characteristics is operational in Italy, as described in our previous study [35].
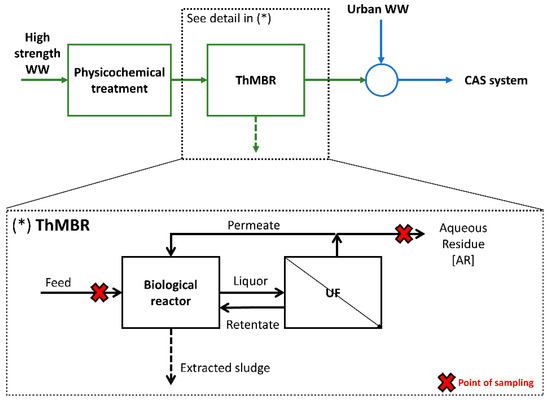
Figure 1.
Localization and details of ThMBR in the pretreatment line for high-strength WW.
The AR of the process (permeate) is mixed with urban WW and treated by a CAS system. Therefore, to avoid an excessive overload of the CAS located downstream of the pre-treatment station, it is important that the ThMBR system operates efficiently and effectively in the removal of chemical pollutants. For this reason, monitoring is essential to understand and verify what the real-scale performance of the process actually is.
In Table 1, the main characteristics of high-strength WW treated in ThMBR are reported. The ThMBR was activated in January 2018 to treat industrial WW with highly recalcitrant organic substances (e.g., surfactants, solvents, pharmaceutical products). To ensure significant results, only data from July 2018 (after the system reached complete stability) to December 2020 have been analyzed. For chemical and physicochemical analysis, samples were taken weekly. In 2018–2020, about 62 ± 3 m3 d−1 of high strength WW was fed to the ThMBR, with an average hydraulic retention time (HRT) of the system equal to 30 ± 1.5 d. COD reached 47,913 ± 2568 mg L−1 in 2018–2020 with the highest average value (53,220 ± 4911 mg L−1) in 2019. High strength WW fed to the reactor also showed a high concentration of TAS, MBAS, N-NOx and chlorides. The high conductivity (22,692 ± 1537 µS cm−1) is effectively endured by the ThMBR due to strong resistance of thermophilic biological systems [37].

Table 1.
Summary of flowrate, HRT, chemical and physicochemical parameters of high-strength WW fed to the ThMBR during the monitoring period. HRT: hydraulic retention time; TAS: non-ionic surfactants; MBAS: anionic surfactants; TP: total phosphorus. n: number of data points.
2.2. Respirometric Tests
2.2.1. Aqueous Residues
These tests aim to evaluate the effect on mesophilic biomass of the AR of the ThMBR, after the treatment of diverse matrices, not only in this specific situation but also in the case of a future application of the process in other WWTPs.
In this case, usually, high-strength WW products fed to the real ThMBR were previously mixed and homogenized making it impossible to separate the effect of the different polluting matrices. Therefore, a continuous flow pilot plant (1 m3) with the same characteristics of the full-scale scale ThMBR was used to treat four diverse high-strength WW products. The management conditions were kept similar to those used in the real scale (HRT = 25–30 d; O2= 1–3 mg L−1; T= 48–50 °C) and the AR extracted from the recirculation line was studied by respirometric tests. Chemical and physicochemical characteristics of the ARs are reported in Table 2. In the case of WW1 and WW2, diverse ARs, due to diverse phases of experimentation and diverse fed concentrations, were considered (ARX-a,b,c). AR3-a represents the residue after WW3 was preatreated in UASB and then in ThMBR. AR3-b refers to WW3 directly treated in ThMBR.

Table 2.
Summary of chemical and physicochemical characteristics of AR extracted from the pilot-scale ThMBR.
2.2.2. Practical Procedure
As reported by several authors [38,39,40,41], respirometric tests (OUR, AUR, and NUR) can be used to evaluate the effect of a substrate on the heterotrophic and autotrophic mesophilic biomass. In our study, respirometric analyses were used to assess the effect of AR of ThMBR on mesophilic biological activity of the CAS system. All tests were conducted at 20 ± 1 °C.
Biomass used in OUR and AUR analysis was withdrawn directly from the oxidation/nitrification reactor of the same WWTP. In order to later express oxygen uptake as a function of COD, diverse organic substrate solutions were prepared by diluting AR with distilled water in various ratios [40]. In exogenous OUR tests, 500 mL of biomass was aerated for 30 min (up to O2 concentration of 6.5–7.5 mg L−1) and then mixed with 500 mL of an organic substrate solution. Then, the aeration was stopped, and the system with the 1L mixture was hermetically closed to avoid the dispersion of the oxygen into the atmosphere. Continuous stirring was maintained (300–400 RPM) and nitrification phenomena were inhibited by addition of allylthiourea. pH was maintained at 7.0–7.5 adding H2SO4 drop by drop as necessary. The specific oxygen uptake rate (SOUR) was evaluated considering the VSS concentration in the batch reactor and the slope of the curve of oxygen consumption [38]. To evaluate the respiration of the biomass, no substrate was added in endogenous OUR. The SOUR was calculated according to the following equation:
SOUR (mgO2 gVSS−1 h−1) = SOURexogenous − SOURendogenous
To better highlight the behavior trend of the SOUR as a function of the COD and evaluate the acute effect of the substrate on the mesophilic biomass, the results were linear fitted.
In AUR, 500 mL of biomass was aerated for 30 min, mixed with 500 mL of AR and enriched of ammonia using (NH4)2SO4 to obtain an initial N-NH4+ concentration in the 1L blend of 50–60 mg L−1. Continuous stirring (300–400 RPM) and aeration (O2 concentration of 6.5–7.5 mg L−1) were maintained in the mixture. pH was maintained at 7.0–7.5 adding H2SO4 adding H2SO4 drop by drop as necessary. Tests were conducted for 6 h and 20 mL was sampled every hour. AUR was evaluated considering the VSS concentration in the batch reactor and the slope of the curve of N-NO2− and N-NO3− production, according to Holm et al. [38]. The WW that usually enters the oxidation/nitrification reactor of the same WWTP, properly enriched by ammonia as previously described, was used as reference in AUR tests.
NUR tests aim to evaluate the feasibility of using the permeate as an external source of organic carbon for denitrifying bacteria. Biomass was withdrawn directly from a full-scale denitrification reactor of the same WWTP. A volume of 500 mL of biomass was aerated for 30 min and mixed with 500 mL of an organic substrate solution. To be able to later express NUR as a function of COD, diverse 500 mL organic substrate solutions were prepared by diluting AR with distilled water in various ratios. The 1L blend was enriched with nitrates using KNO3 to obtain an initial N-NOx concentration of 40–50 mg L−1. Continuous stirring (300–400 RPM) was maintained and the system was hermetically closed to avoid the solubilization of the atmospheric oxygen. pH was maintained at 7.0–7.5 adding H2SO4 drop by drop as necessary. Tests were conducted for 6 h and 20 mL was sampled every hour and filtered. NUR was evaluated considering the VSS concentration in the batch reactor and the slope of the curve of N-NO2− and N-NO3− consumption. Methanol, properly diluted with distilled water, was used as reference organic substrate solution in NUR tests.
2.3. Analytical Procedures
COD was measured according to ISPRA 5135 method [42]. Total solids (TS), volatile solids (VS) and volatile suspended solids (VSS) were determined following APAT-IRSA-CNR 2090 method [43]. N-NH4+ and N-NO2− were studied following APAT-IRSA-CNR 4030 [44] and APAT-IRSA-CNR 4050 [45] methods, respectively. N-NO3− concentrations were studied according to EPA 300.1 1997 [46]. N-NOX was calculated as the sum of N-NO3− and N-NO2−. Cyanides were measured following APAT IRSA CNR 4070 method [47]. Total phosphorus (TP) was analyzed according to EPA 3051A 2007 [48] and EPA 6010D 2018 [49]. PFAS were determined according to EPA 3550C 2007 [50] and EPA 537 2009 [51] methods. MBAS were analyzed using ISO 7875-1 1996 [52] and ISO 7875-2 1996 [53]. TAS were measured following UNI 10511-1 1996 method [54].
3. Results and Discussion
3.1. Performance on Chemical Pollutants and Sludge Production
The performance of the ThMBR on the removal of COD, TAS and MBAS was evaluated. The main aspect that can be highlighted is that the removal of chemical pollutants strongly depended on the pollutant load rate. In the monitored period, the food:microorganism (F/M) ratio was 0.030 ± 0.003 gCOD gVS−1 d−1. This low ratio is related to the high recalcitrance of the organic substance (e.g., surfactants, solvents, pharmaceutical products) present in the high-strength WW fed to ThMBR, which, on the other hand, would not be treatable with a conventional mesophilic process.
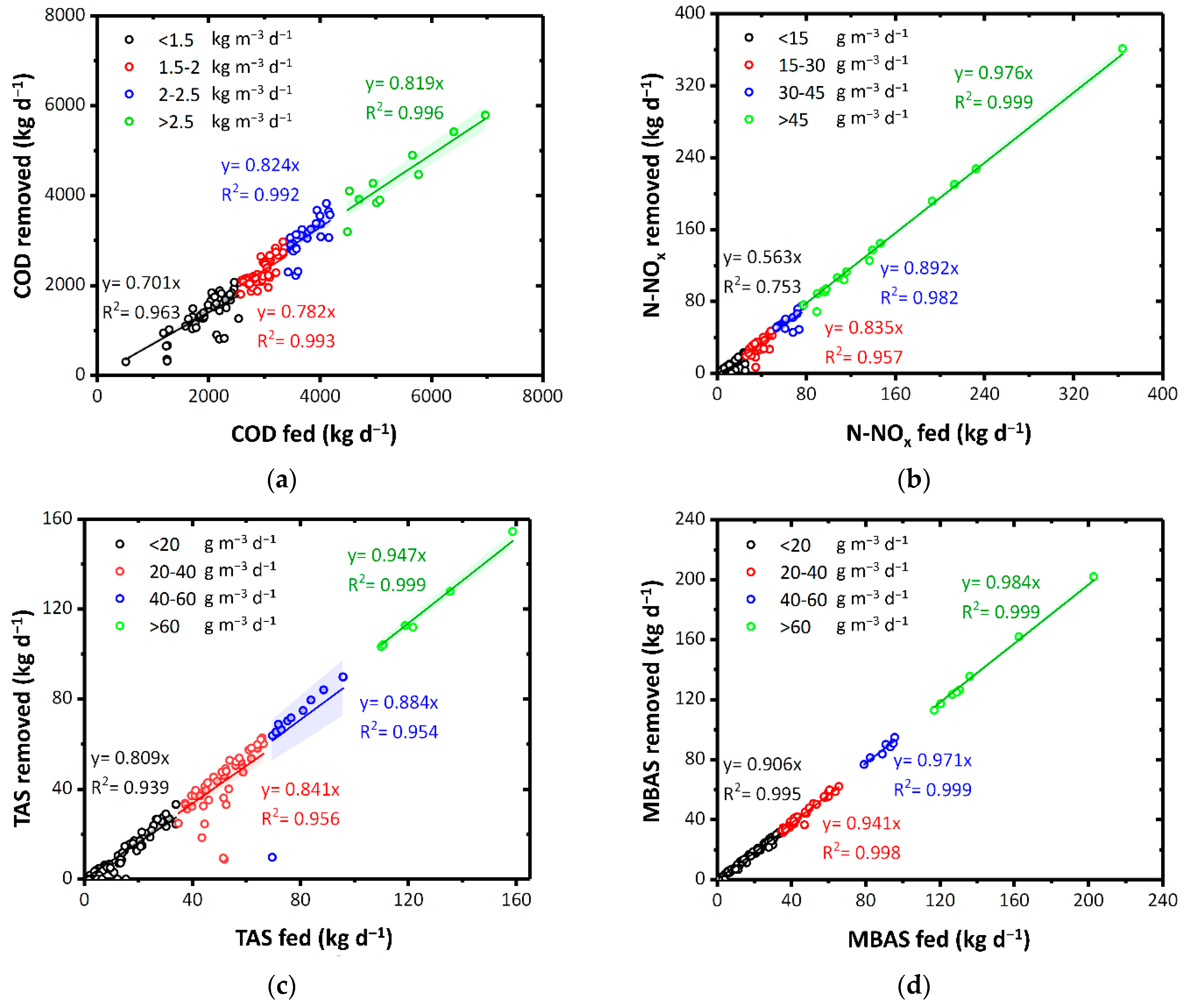
With an organic loading rate (OLR) between 1.5 and 2 kg m−3 d−1, the COD removal efficiency was on average 78.2%. However, this efficiency increased up to 81.9% in the presence of an OLR greater than 3 kg m−3 d−1 (Figure 2a). These results confirm our previous findings: almost 77% of COD fed was effectively removed by the other real-scale ThMBR plant operating in Italy [35]. Moreover, Simstich et al. [21] reported 83% of COD removal when treating industrial wastewater by a thermophilic MBR operating in aerobic conditions.
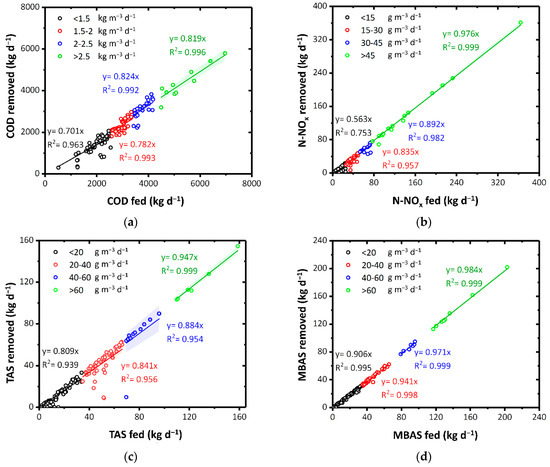
Figure 2.
(a) COD, (b) N-NOx (c) TAS, and (d) MBAS removal as a function of pollutants fed. Colors highlight diverse load rates. The colored bands represent the 95% confidence interval of the linear fitting.
N-NOx was significantly removed according to the increase in loading rate. In the case of a load rate higher than 45 g m−3 d−1, an average of 97.6% of N-NO3 and N-NO2 were denitrified (Figure 2b). At thermophilic temperatures (>45 °C), denitrification takes place by a microbial biomass comparable or even superior to the mesophilic one in terms of diversity and resistance [55]. Although pure oxygen is dosed in the ThMBR, the denitrification phenomenon can be attributed to the concomitant presence of (i) a high quantity of organic substance given the very high COD of the fed matrices and (ii) some areas of the reactor that remain without adequate oxygenation. These results highlight that a study of the hydrodynamic behavior of the reactor by a residence time distribution (RTD) analysis and/or a computational fluid dynamic (CFD) analysis, as already done in conventional WWTP [56,57], is greatly required. The presence of the same denitrification phenomena was also observed in the other full-scale ThMBR in our previous study [35].
A similar result to COD and N-NOx was also observed for TAS (Figure 2c) and MBAS (Figure 2d), with maximum removal (94.7% and 98.4%, respectively) corresponding with a fed load rate greater than 60 g m−3 d−1. In thermophilic membrane biological processes, the direct proportionality between the COD removal yields and the OLR is in accordance with what was reported in our previous studies on pilot plants [28]. However, in this work the same relationship applied to full scale plants has been shown and not only for COD but also for N-NOx, and surfactants (TAS and MBAS). Precisely, the TAS that could be usually difficult to degrade by biological systems in mesophilic conditions [25,58,59] were instead effectively removed by ThMBR (>84.1%) with fed load rate higher than 20 g m−3 d−1. This aspect should not be attributed to the presence of a UF membrane. In fact, due to their molecular weights (from 200 to 400 Da), surfactants can pass through the pores [60]. Instead, these results can be related to the greater ability of thermophilic microorganisms to degrade recalcitrant chemical pollutants such as TAS [25]. MBAS were almost totally removed (94.1–98.4%) by ThMBR. Treating real laundry WW in the other full-scale ThMBR, lower removal yields for TAS and MBAS were obtained (47.8% and 49.5%, respectively) probably due to lower initial surfactant concentrations [25].
Furthermore, even in the presence of significant incoming TAS load rate (up to 100 g m−3 d−1), no inhibition of the biomass degradation activity was highlighted, unlike what has generally been observed in mesophilic systems in the presence of surfactants even in low concentrations (30 mg L−1) [13]. This result can be related to the greater resilience of thermophilic biomass towards potentially inhibiting substances for mesophilic biomass present in CAS systems [28].
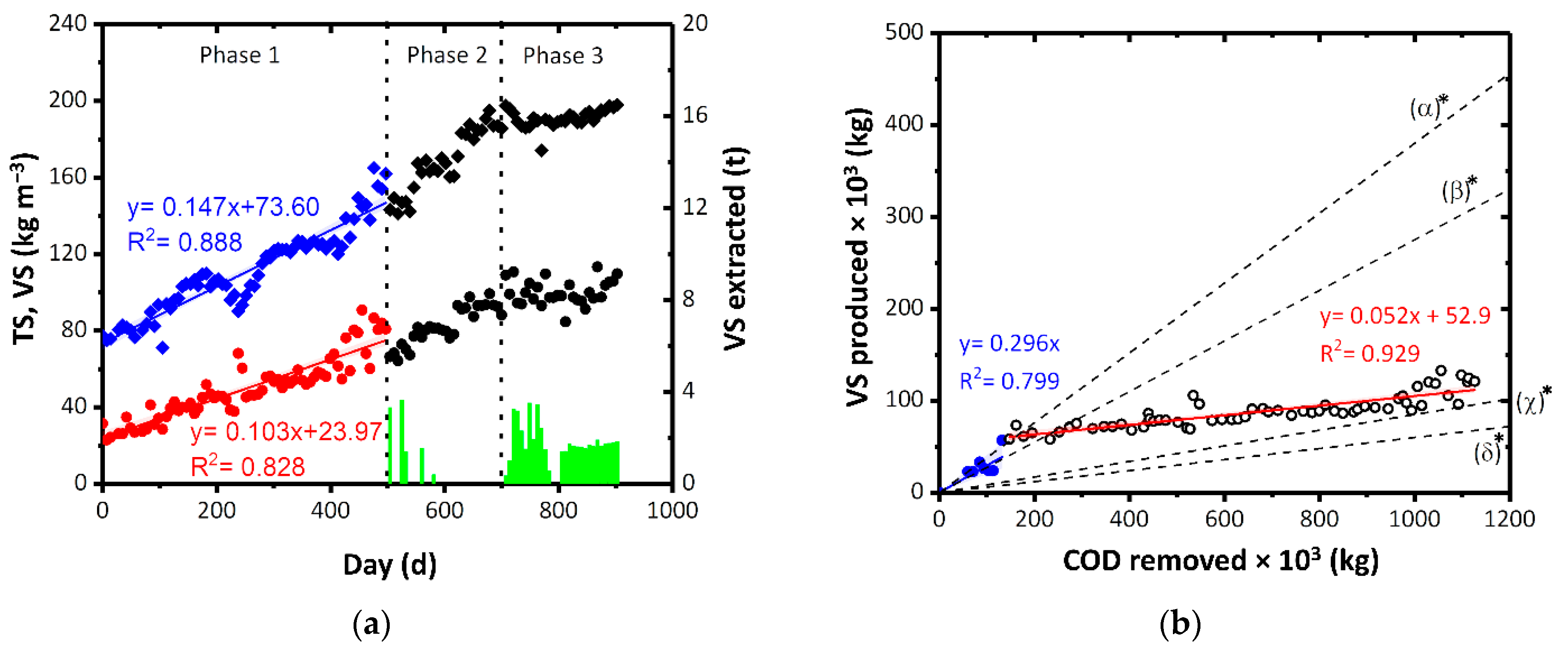
The concentration of TS and VS in the biological reactor was monitored. In Figure 3a, the concentrations of solids in the tank and the sludge extractions are reported. Three different periods can be identified. In period 1 (duration: 500 days), no extractions were carried out and the concentration of TS and VS in the reactor reached 160 kg m−3 and 80 kg m−3, respectively. In period 2, occasional extractions were carried out and the increase in the concentration of TS and VS up to 185 kg m−3 and 90 kg m−3, respectively, was observed. For management reasons, in period 3, the concentration of solids in the tank was kept almost constant thanks to periodic extraction of sludge (about 20 t per week).
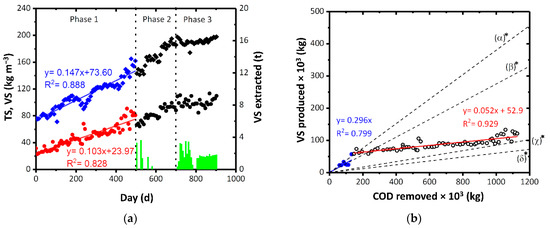
Figure 3.
(a) Concentration of TS and VS in the biological reactor during the monitoring period and amount of VS extracted. ♦ indicates TS, • represents VS and green bars indicate VS extracted. (b) VS produced by the process as a function of COD removed and comparison with theoretical value of (α) CAS [61,62], (β) mesophilic MBR [63,64,65], (γ) thermophilic MBR [29], and (δ) value of the other full-scale ThMBR [35]. The specific productions of VS used as comparison were the mean values of those reported in the literature. Blue • represents VS in the start-up phase while ○ represents VS in the subsequent phase. The colored bands represent the 95% confidence interval of the linear fitting. *: values are expressed in kgVSS kgCOD−1.
To determine the specific production of VS in relation to the COD removed, period 1 was chosen as the reference as the concentration of sludge accumulated in the tank was not influenced by the extractions (as in periods 2 and 3). The daily production of TS and VS was 0.147 kgTS d−1 and 0.103 kgVS d−1, respectively (Figure 3a). Comparing COD removed with the production of VS, the specific sludge production can be approximated with two linear functions. A specific production of 0.296 kgVS kgCOD−1 has been observed that corresponds with very low COD removals (substantially in the first period after the start-up phase) while subsequently, the production of VS dropped to 0.052 kgVS kgCOD−1 (Figure 3b).
In the literature, specific sludge production is generally expressed considering the VSS produced. Therefore, the comparison VSS-VS is then made by overestimating the specific production obtained in the present study. The value obtained in this study was lower by one order of magnitude than 0.25–0.51 kgVSS kgCOD−1 produced by CAS systems [61,62], and 0.19–0.36 kgVSS kgCOD−1 [63,64,65] produced by mesophilic membrane biological reactors (MBRs). The present result was comparable with the value of sludge production by thermophilic MBRs reported in the literature (0.02–0.1 kgVSS kgCOD−1) [29] and with of the other real ThMBR reported in our previous study (0.08–0.09 kgVSS kgCOD−1) [35].
3.2. Influence of Aqueous Residues on Mesophilic Biological Activity
3.2.1. Effect on Decomposing and Nitrifying Biomass
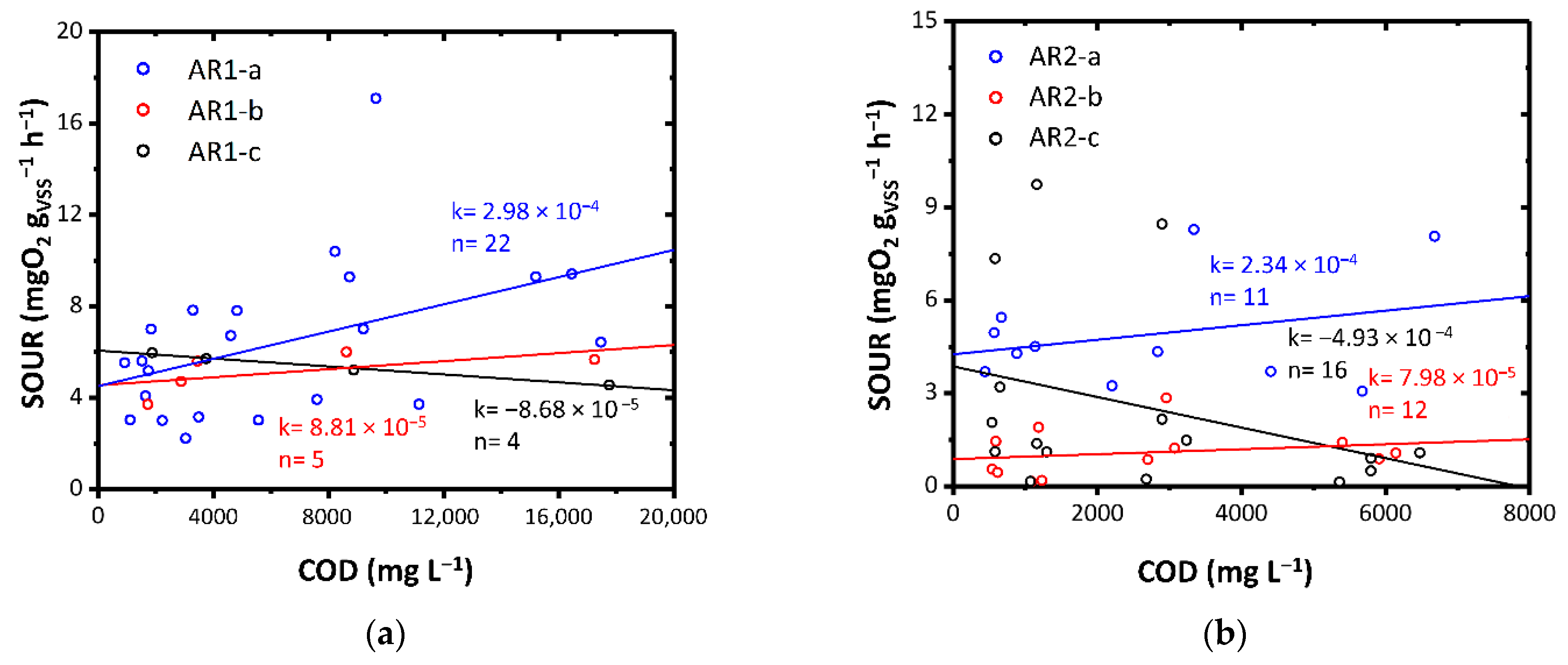
The effect of aqueous residues of ThMBR on mesophilic biomass was first evaluated by means of respirometric tests. The results show a different behavior depending on the initial matrix treated by the ThMBR and therefore on the characteristics of the AR tested. The AR1-a and AR1-b substrates, although rich in cyanides and chlorides, showed a substantial good degradability by the mesophilic biomass, not showing evident acute inhibitory effects of biological activity (Figure 4a). Instead, the AR1-c resulted in minimal toxic-inhibiting activity on biomass. This can be evidenced by the average increase in SOUR with decreasing COD, following dilution of the substrate. This result can be attributed to the fact that this matrix had a similar concentration of cyanides but about double the concentration of chlorides compared to the other AR1s tested. In our study, the initial ratios of SOUR with non-diluted AR were 0.45–1.2 gCl− gVSS−1 for AR1-a, 0.05–1.75 gCl− gVSS−1 for AR1-b and 0.6–2.5 gCl− gVSS−1 for AR1-c. The presence of salinity in WW is a factor that strongly influences the respiration of mesophilic biomass, especially when the biomass is not acclimatized. For example, Pernetti and Palma [14] found that in batch mode, salt/VSS ratios between 0.37 gsalt gVSS−1 and 30.7 gsalt gVSS−1 produced respiration inhibition between 4% and 84%, respectively. On the contrary, Bassin et al. found that Cl− had a positive effect on the settling properties as antagonist filamentous bacteria were inhibited by high salt concentrations, but only when the increase in Cl− concentration was gradual [66].
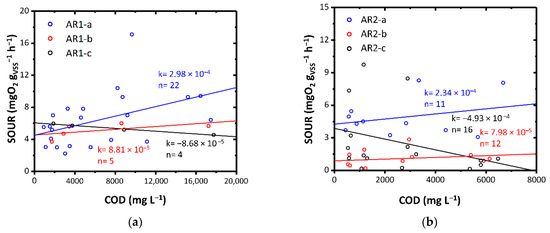
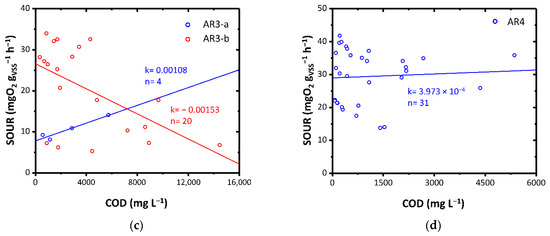
Figure 4.
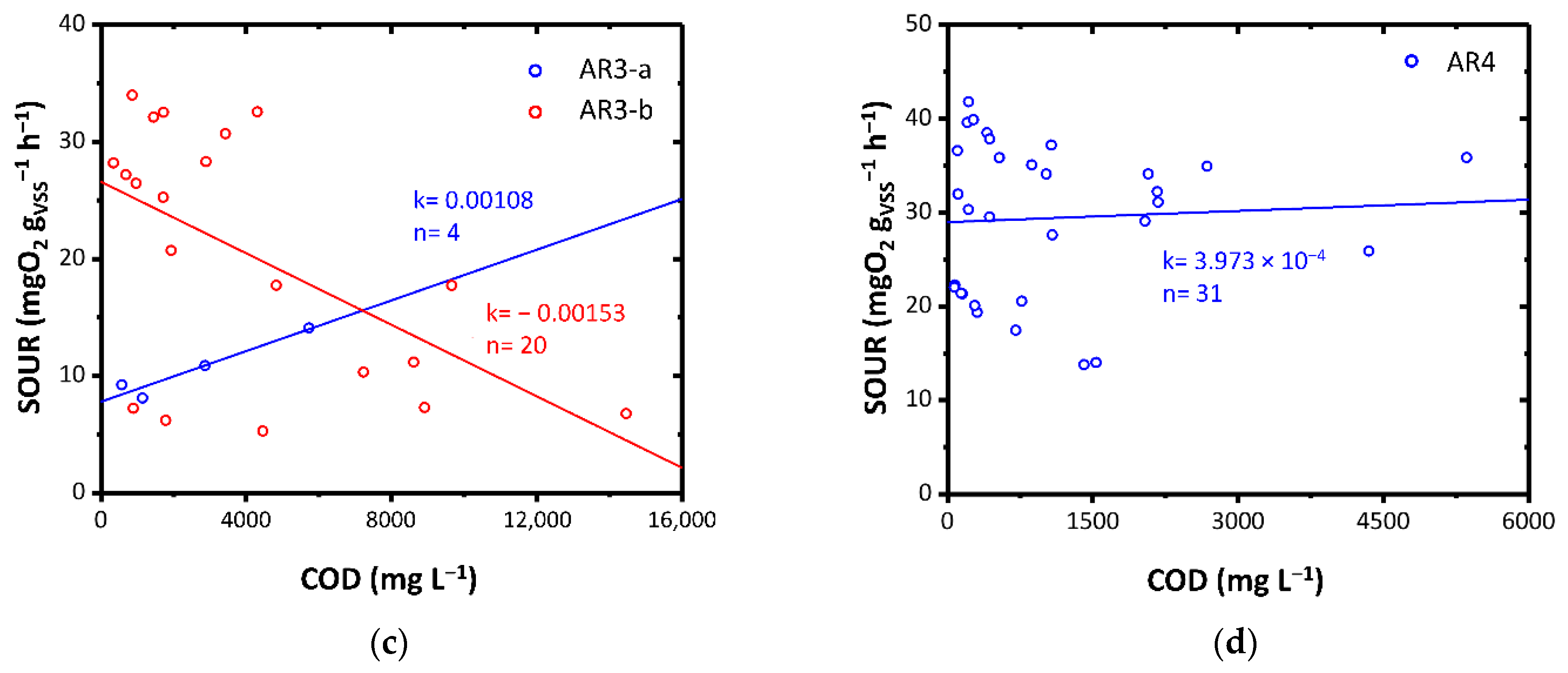
SOUR as a function of COD for: (a) AR1-a, AR1-b and AR1-c; (b) AR2-a, AR2-b and AR2-c; (c) AR3-a, and AR3-b; (d) AR4. Colors highlight diverse load rates. k: angular coefficient of the linear fitting expressed as mgO2 L gVSS−1 h−1 mgCOD−1; n: number of data points.
AR2 had a significant concentration of chlorides and PFAS. AR2-a and AR2-b, however, did not cause acute inhibition phenomena (Figure 4b). Instead, AR2-c produced a toxic-inhibiting effect on biomass, even more evident than that obtained with the AR1-c substrate. This result could be attributed to two aspects: (i) the high concentration of chlorides (1.1–2.6 gCl− gVSS−1 in SOUR with non-diluted AR2-c); (ii) the very high concentration of PFAS (0.4–1.9 gPFAS gVSS−1 in SOUR with non-diluted AR2-c). While some types of PFAS (e.g., PFOS) would seem not to have a toxic effect on the mesophilic biomass of CAS [67], exposure to high concentrations of other PFAS (e.g., Perfluorooctanoic acid-PFOA) has shown negative impacts on microbial growth and organic substance removal performance [15].
AR3-a and AR3-b exhibited a diverse effect (Figure 4c). While AR3-a showed excellent biodegradability in the mesophilic conditions, AR3-b showed an acute inhibitory toxic effect. This result could be attributed to the fact that in the case of the AR3-a, the WW3 had been pre-treated through an anaerobic biological process and then fed to the ThMBR while in the case of the AR3-b, it was directly fed to the ThMBR. The first case may have favored the removal of some interfering substances which instead remained in the AR3-c. This aspect will be the subject of future studies.
AR4 showed a good biodegradability in the mesophilic field highlighted by the average decrease in SOUR with decreasing COD, following dilution of the substrate (Figure 4d). The good biodegradability was also favored by the low concentration of chlorides within the initial matrix (0.01–0.03 gCl− gVSS−1 in SOUR with non-diluted AR4).
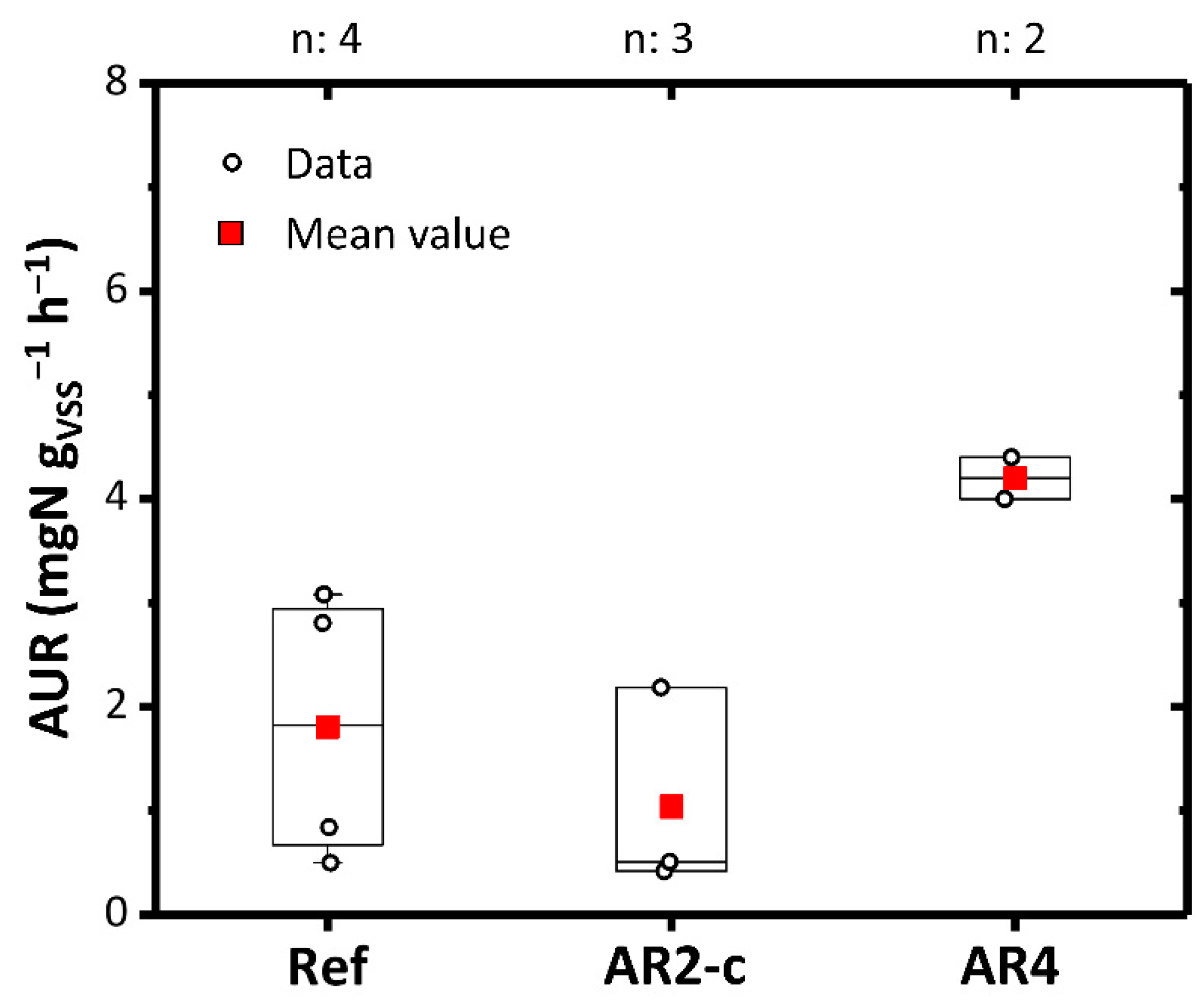
The impact of AR2-c and AR4 on nitrification activity was also investigated. The WW entering the oxidation/nitrification reactor, properly enriched with N-NH4+, was used as the reference substrate. AR2-c showed a slight acute inhibition (1.35 ± 0.84 mgN gVSS−1 h−1) compared to the reference solution (1.81 ± 1.15 mgN gVSS−1 h−1) (Figure 5). This could be traced back to the high concentration of chlorides and PFAS which also had a minimal inhibitory effect in the decomposing microorganisms. Recent studies have shown that PFOS and PFOA can reduce nitrifying activity [68]. An absence of inhibitory phenomena and an increase in nitrification kinetics up to 4.2 ± 0.1 mgN gVSS−1 h−1 was observed with AR4. Therefore, in this case the substrate had a positive effect of stimulating the mesophilic biomass and the phenomenon of nitrification.
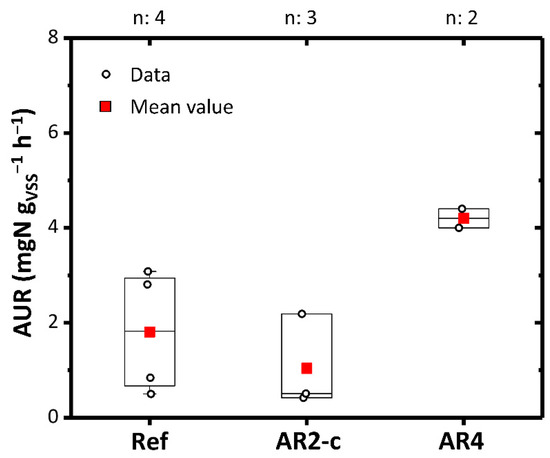
Figure 5.
AUR with AR2-c and AR4. Ref: Reference value. Boxplots represent the distance between the first and third quartiles while whiskers are set as the most extreme (lower and upper) data point not exceeding 1.5 times the quartile range from the median. n: number of data points.
3.2.2. Impact on Denitrifying Biomass
The effect of the AR on denitrifying microorganisms was tested only for the AR4. The goal was to identify any acute toxic effect of the substrate against the denitrifying heterotrophic biomass and determine the feasibility of reuse the organic substance, contained within the aqueous residue, as an external source of carbon in denitrification.
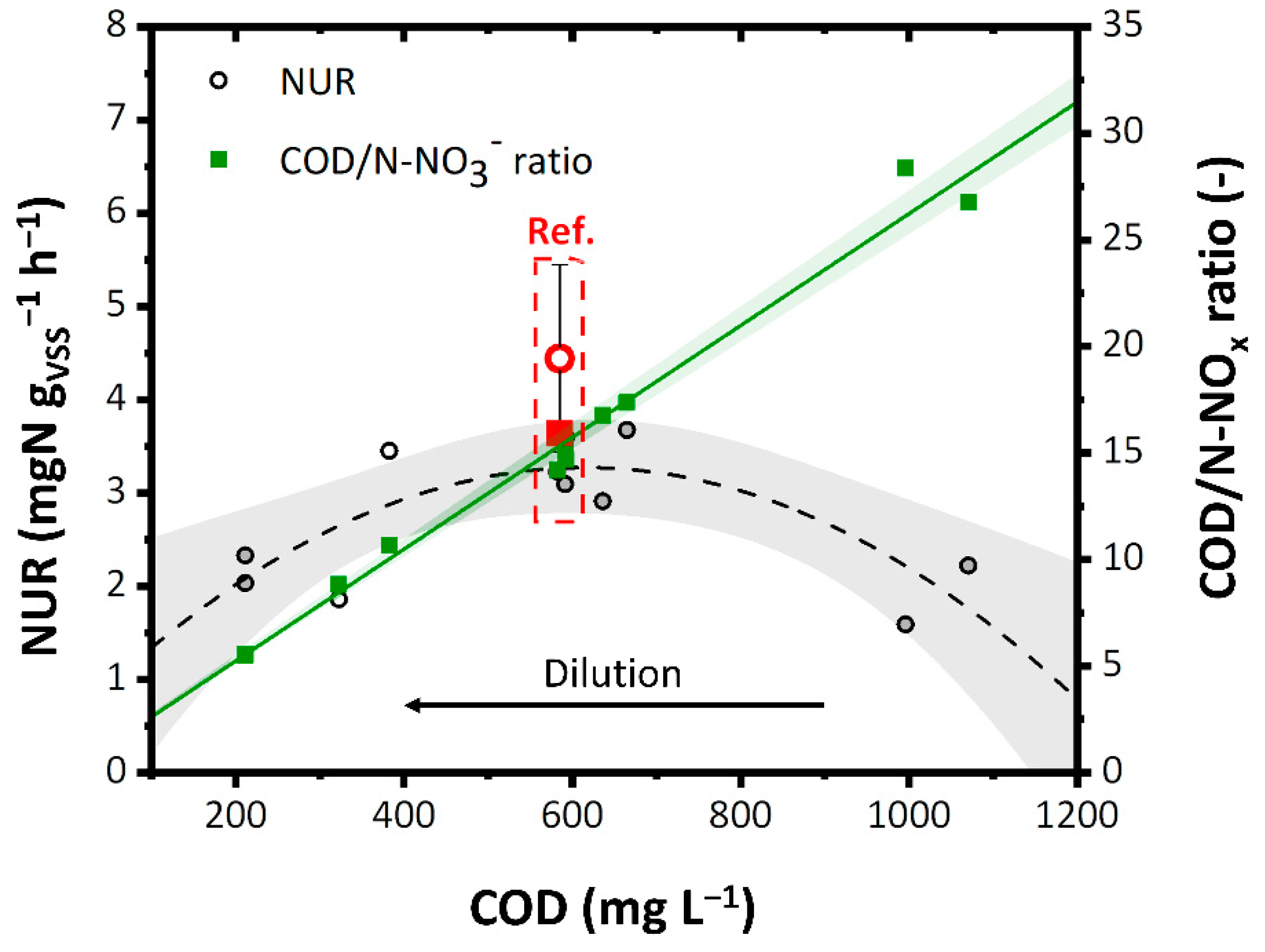
As the dilution increased, the NUR increased until it reached the value of 3.5 mgN gVSS−1 h−1 that corresponded to the COD/N-NOx ratio equal to 15–17.5 (Figure 6). This can be attributed to a slight acute inhibitory effect on the denitrifying biomass of the undiluted aqueous residue. However, the further increase in dilution reduced the NUR (2–2.5 mgN gVSS−1 h−1 with 200 mg L−1 of COD), due to the significant lowering of the COD/N-NOx ratio. The maximum NUR is comparable to that obtained with the reference solution 4.5 ± 1 mgN gVSS−1 h−1 (source of carbon: methanol) and to that reported in the literature for other industrial WW. For instance, Liwarska-Bizukojć et al. [69] reported around 2.0–6.0 mgN gVSS−1 h−1 as the denitrification rate using industrial WW and around 3.2 mgN gVSS−1 h−1 using methanol as an external carbon source.
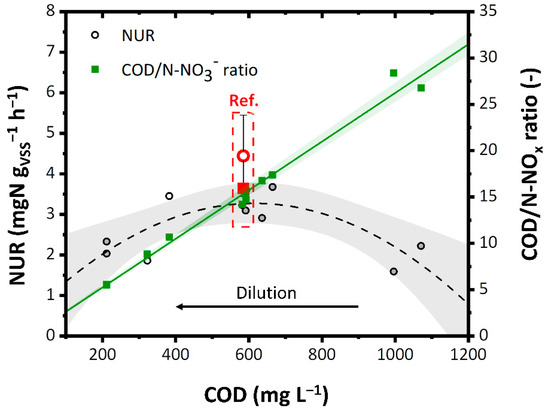
Figure 6.
NUR as a function of COD and COD/N-NOx ratio. Ref: Reference value. The colored bands represent the 95% confidence interval of the linear fitting.
4. Conclusions
Full-scale ThMBR proved to be very effective in pollutants removal, demonstrating an increase in performance as the input load increases. COD, N-NOx, TAS, and MBAS removal yields were up to 81.9%, 97.6%, 94.7%, and 98.4%, respectively. The denitrification phenomenon can be attributed to some areas of the reactor that remain without adequate oxygenation and the presence of a high quantity of organic substance given the very high COD of the fed matrices. Very low daily sludge production has also been evaluated (TS and VS were 0.147 kgTS d−1 and 0.103 kgVS d−1, respectively). Comparing COD removed and VS production, a specific production of 0.296 kgVS kgCOD−1 has been observed, especially corresponding to the first period after the start-up phase, while subsequently, the production of VS drops down to 0.052 kgVS kgCOD−1.
OUR tests allowed us to exclude the possibility that mesophilic biomass generally exhibited any acute inhibition following contact with the tested substrates except for AR1-c, AR2-c and AR3-b. In these cases, due to the presence of high concentrations of cyanides and chlorides (AR1-c), PFAS and chlorides (AR2-c) and probably other interfering substances (AR3-b), an acute toxic-inhibiting effect was evidenced. The AR2-c matrix itself showed a slight decrease in nitrifying activity (1.35 ± 0.84 mgN gVSS−1 h−1) compared to the reference solution (1.81 ± 1.15 mgN gVSS−1 h−1) probably due to high PFAS and chlorides concentration, while the substrate AR5 determined a positive effect, stimulating the mesophilic biomass and the phenomenon of nitrification. NUR tests on AR4 highlighted the feasibility of reusing the organic carbon contained in the substrate as a source in denitrification and obtaining a value comparable with that obtained when using the reference solution with methanol.
Although this work focuses only on the evaluation of the acute effect of the aqueous residue on the mesophilic biological activity, respirometric tests proved to be a valid tool to assess the impact of AR on the activity of mesophilic biomass in the case of recirculation. As a future perspective, aspects such as the OUR as a function of time with continuous feeding could be evaluated, to also consider the physiological acclimatization of the biomass to the organic substrate, and thus determine the medium-term effects of RA on mesophilic biomass.
Author Contributions
Conceptualization, M.C.C., M.C.M. and A.A.; methodology, M.C.C., M.C.M., M.B. and A.A.; validation, M.C.C. and M.B., formal analysis, M.C.M. and A.A.; investigation, F.M.C.; resources, M.C.C.; data curation, M.C.M. and F.M.C.; writing—original draft preparation, M.C.M. and F.M.C.; writing—review and editing, M.C.C., M.C.M., M.B. and A.A.; visualization, F.M.C.; supervision, M.C.C. and A.A.; All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by ASMia S.r.l. (Lombardy, Italy).
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Acknowledgments
The authors wish to thank ASMia S.r.l. (Lombardy, Italy) for providing the data. The authors also acknowledge Angela Durante for the technical support to the research activities.
Conflicts of Interest
The authors declare no conflict of interest.
Nomenclature
AR: Aqueous residue; AUR: Ammonia uptake rate; CAS: Conventional active sludge; CFD: Computational fluid dynamic; HRT: Hydraulic retention time; MBAS: anionic surfactants; MBR: Membrane biological reactor; NUR: Nitrate uptake rate; OLR: Organic loading rate; OUR: Oxygen uptake rate; PFAS: perfluoro alkyl substances; PFOA: Perfluorooctanoic acid; PFOS: perfluorooctanesulfonic acid; RTD: Residence time distribution; SOUR: Specific oxygen uptake rate; TAS: Non-ionic surfactants; ThMBR: Thermophilic membrane bioreactor; TP: Total phosphorus; TS: Total solids; UF: Ultrafiltration; VS: Volatile solids; VSS: Volatile suspended solids; WW: Wastewater; WWTP: Wastewater treatment plant.
References
- Hamza, R.A.; Iorhemen, O.T.; Tay, J.H. Advances in biological systems for the treatment of high-strength wastewater. J. Water Process Eng. 2016, 10, 128–142. [Google Scholar] [CrossRef]
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A critical review on textile wastewater treatments: Possible approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, D.; Sharma, N.R.; Singh, J.; Kanwar, R.S. Biological methods for textile dye removal from wastewater: A review. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1836–1876. [Google Scholar] [CrossRef]
- Siyal, A.A.; Shamsuddin, M.R.; Low, A.; Rabat, N.E. A review on recent developments in the adsorption of surfactants from wastewater. J. Environ. Manag. 2020, 254, 109797. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.; Hatley, H. The role of surfactants in wastewater treatment: Impact, removal and future techniques: A critical review. Water Res. 2018, 147, 60–72. [Google Scholar] [CrossRef]
- Bukalova, N.P. Statistically distributed characteristics of wastewater discharged from Kamskiy Kabel, LTD and containing organic phenolic compounds. In Proceedings of the IOP Conference Series: Materials Science and Engineering; International Conference on Construction, Architecture and Technosphere Safety (ICCATS 2020), Sochi, Russia, 6–12 September 2020; Volume 962, p. 042090. [Google Scholar]
- Miazek, K.; Kratky, L.; Sulc, R.; Jirout, T.; Aguedo, M.; Richel, A.; Goffin, D. Effect of Organic Solvents on Microalgae Growth, Metabolism and Industrial Bioproduct Extraction: A Review. Int. J. Mol. Sci. 2017, 18, 1429. [Google Scholar] [CrossRef] [Green Version]
- Phong Vo, H.N.; Ngo, H.H.; Guo, W.; Hong Nguyen, T.M.; Li, J.; Liang, H.; Deng, L.; Chen, Z.; Hang Nguyen, T.A. Poly-and perfluoroalkyl substances in water and wastewater: A comprehensive review from sources to remediation. J. Water Process Eng. 2020, 36, 101393. [Google Scholar] [CrossRef]
- Lenka, S.P.; Kah, M.; Padhye, L.P. A Review of the Occurrence, Transformation, and Removal of Poly- and Perfluoroalkyl Substances (PFAS) in Wastewater Treatment Plants. Water Res. 2021, 199, 117187. [Google Scholar] [CrossRef]
- Vu, C.T.; Wu, T. Recent progress in adsorptive removal of per- and poly-fluoroalkyl substances (PFAS) from water/wastewater. Crit. Rev. Environ. Sci. Technol. 2020, 2020, 1–40. [Google Scholar] [CrossRef]
- Iloms, E.; Ololade, O.O.; Ogola, H.J.O.; Selvarajan, R. Investigating Industrial Effluent Impact on Municipal Wastewater Treatment Plant in Vaal, South Africa. Int. J. Environ. Res. Public Health 2020, 17, 1096. [Google Scholar] [CrossRef] [Green Version]
- Burakov, A.E.; Galunin, E.V.; Burakova, I.V.; Kucherova, A.E.; Agarwal, S.; Tkachev, A.G.; Gupta, V.K. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotoxicol. Environ. Saf. 2018, 148, 702–712. [Google Scholar] [CrossRef]
- Othman, M.Z.; Ding, L.; Jiao, Y. Effect of Anionic and Non-ionic Surfactants on Activated Sludge Oxygen Uptake Rate and Nitrification. Int. J. Chem. Mol. Nucl. Mater. Metall. Eng. 2009, 3, 596–602. [Google Scholar]
- Pernetti, M.; Palma, L. Di Experimental Evaluation of Inhibition Effects of Saline Wastewater on Activated Sludge. Environ. Technol. 2005, 26, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Nishimura, F.; Hidaka, T. Impact of Long-Term Perfluorooctanoic Acid (PFOA) Exposure on Activated Sludge Process. Water Air Soil Pollut. 2018, 229, 134. [Google Scholar] [CrossRef]
- Ramos, C.; Suárez-Ojeda, M.E.; Carrera, J. Biodegradation of a high-strength wastewater containing a mixture of ammonium, aromatic compounds and salts with simultaneous nitritation in an aerobic granular reactor. Process Biochem. 2016, 51, 399–407. [Google Scholar] [CrossRef] [Green Version]
- Collivignarelli, M.C.; Abbà, A.; Bestetti, M.; Crotti, B.M.; Carnevale Miino, M. Electrolytic Recovery of Nickel and Copper from Acid Pickling Solutions Used to Treat Metal Surfaces. Water Air Soil Pollut. 2019, 230, 101. [Google Scholar] [CrossRef]
- Rada, E.C.; Ragazzi, M.; Torretta, V.; Castagna, G.; Adami, L.; Cioca, L.I. Circular economy and waste to energy. AIP Conf. Proc. 2018, 1968, 030050. [Google Scholar] [CrossRef]
- European Commission Closing the Loop—An EU Action Plan for the Circular Economy. 2015. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52015DC0614 (accessed on 10 April 2021).
- European Commission EU Circular Economy Action Plan. Available online: https://ec.europa.eu/environment/pdf/circular-economy/new_circular_economy_action_plan.pdf (accessed on 10 April 2021).
- Simstich, B.; Beimfohr, C.; Horn, H. Lab scale experiments using a submerged MBR under thermophilic aerobic conditions for the treatment of paper mill deinking wastewater. Bioresour. Technol. 2012, 122, 11–16. [Google Scholar] [CrossRef]
- Duncan, J.; Bokhary, A.; Fatehi, P.; Kong, F.; Lin, H.; Liao, B. Thermophilic membrane bioreactors: A review. Bioresour. Technol. 2017, 243, 1180–1193. [Google Scholar] [CrossRef]
- Baker, B.A.; Tawabini, B.; Nazal, M.; AlThaqfi, J.; Khalil, A. Efficiency of Thermophilic Bacteria in Wastewater Treatment. Arab. J. Sci. Eng. 2021, 46, 123–128. [Google Scholar] [CrossRef]
- Dhangar, K.; Kumar, M. Perfluorooctanesulfonate (PFOS), Its Occurrence, Fate, Transport and Removal in Various Environmental Media: A Review. In Contaminants in Drinking and Wastewater Sources; Kumar, M., Snow, D., Honda, R., Mukherjee, S., Eds.; Springer Transactions in Civil and Environmental Engineering: Singapore, 2021; pp. 405–436. [Google Scholar]
- Collivignarelli, M.C.; Carnevale Miino, M.; Baldi, M.; Manzi, S.; Abbà, A.; Bertanza, G. Removal of non-ionic and anionic surfactants from real laundry wastewater by means of a full-scale treatment system. Process Saf. Environ. Prot. 2019, 132, 105–115. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Bertanza, G.; Baldi, M.; Setti, M.; Frattarola, A.; Carnevale Miino, M. Treatment of high strength wastewater by thermophilic aerobic membrane reactor and possible valorisation of nutrients and organic carbon in its residues. J. Clean. Prod. 2021, 280, 124404. [Google Scholar] [CrossRef]
- Pugazhendhi, A.; Theivaraj, S.D.; Boovaragamoorthy, G.M.; Veeramani, V.; Brindhadevi, K.; Al-Dhabi, N.A.; Arasu, M.V.; Kaliannan, T. Impact on degradation of antibiotics from poultry litter using Autothermal Thermophilic Aerobic Digestion (ATAD). Saudi J. Biol. Sci. 2021, 28, 988–992. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Bertanza, G.; Barbieri, G. Treatment of high strength aqueous wastes in a thermophilic aerobic membrane reactor (TAMR): Performance and resilience. Water Sci. Technol. 2017, 76, 3236–3245. [Google Scholar] [CrossRef] [PubMed]
- Collivignarelli, M.C.; Abbà, A.; Bertanza, G.; Frattarola, A. The upgrading of conventional activated sludge processes with thermophilic aerobic membrane reactor: Alternative solutions for sludge reduction. J. Environ. Manag. 2020, 264, 110490. [Google Scholar] [CrossRef]
- Bertanza, G.; Papa, M.; Canato, M.; Collivignarelli, M.C.; Pedrazzani, R. How can sludge dewatering devices be assessed? Development of a new DSS and its application to real case studies. J. Environ. Manag. 2014, 137, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Joo, S.H.; Monaco, F.D.; Antmann, E.; Chorath, P. Sustainable approaches for minimizing biosolids production and maximizing reuse options in sludge management: A review. J. Environ. Manag. 2015, 158, 133–145. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abba, A.; Padovani, S.; Frascarolo, M.; Sciunnach, D.; Turconi, M.; Orlando, M. Recovery of Sewage Sludge on agricultural land in Lombardy: Current issues and regulatory scenarios. Environ. Eng. Manag. J. 2015, 14, 1477–1486. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Frattarola, A.; Carnevale Miino, M.; Padovani, S.; Katsoyiannis, J.; Torretta, V. Legislation for the Reuse of Biosolids on Agricultural Land in Europe: Overview. Sustainability 2019, 11, 6015. [Google Scholar] [CrossRef] [Green Version]
- Bauer, T.; Andreas, L.; Lagerkvist, A.; Burgman, L.E. Effect of the different implementation of legislation relating to sewage sludge disposal in the EU. Detritus 2020, 10, 92–99. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Frattarola, A.; Manenti, S.; Todeschini, S.; Bertanza, G.; Pedrazzani, R. Treatment of aqueous wastes by means of Thermophilic Aerobic Membrane Reactor (TAMR) and nanofiltration (NF): Process auditing of a full-scale plant. Environ. Monit. Assess. 2019, 191, 708. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Bertanza, G.; Sordi, M.; Pedrazzani, R. High-strength wastewater treatment in a pure oxygen thermophilic process: 11-year operation and monitoring of different plant configurations. Water Sci. Technol. 2015, 71, 588–596. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Bertanza, G. Treatment of high strength pharmaceutical wastewaters in a Thermophilic Aerobic Membrane Reactor (TAMR). Water Res. 2014, 63, 190–198. [Google Scholar] [CrossRef]
- Holm Kristensen, G.; Elberg Jørgensen, P.; Henze, M. Characterization of Functional Microorganism Groups and Substrate in Activated Sludge and Wastewater by AUR, NUR and OUR. Water Sci. Technol. 1992, 25, 43–57. [Google Scholar] [CrossRef]
- Surmacz-Gorska, J.; Gernaey, K.; Demuynck, C.; Vanrolleghem, P.; Verstraete, W. Nitrification monitoring in activated sludge by oxygen uptake rate (OUR) measurements. Water Res. 1996, 30, 1228–1236. [Google Scholar] [CrossRef]
- Collivignarelli, C.; Bertanza, G.; Collivignarelli, M.; Zanaboni, S.; Abbà, A. Optimization of the Urban Waste Water Treatment Service: Maximization of Resource Recovery (Water and Sludge) and Reduction of Energy Consumption; Italian Higher Institute for Environmental Protection and Research: Rome, Italy, 2009. (In Italian) [Google Scholar]
- Hagman, M.; La Cour Jansen, J. Oxygen uptake rate measurements for application at wastewater treatment plants. Vatten 2007, 63, 131–138. [Google Scholar]
- ISPRA. Measurement Procedure for the Determination of the Chemical Oxygen Demand (COD) by Cuvette Test: Method 5135; ISPRA: Rome, Italy, 2014. (In Italian) [Google Scholar]
- APAT-IRSA-CNR. Analytical Methods for Water—n. 2090: Solids; Italian Agency for the Protection of the Environment and for Technical Services: Rome, Italy, 2003. (In Italian) [Google Scholar]
- APAT-IRSA-CNR. Analytical Methods for Water—n. 4030: Ammonia Nitrogen (Spectrophotometric with Indophenol; Spectrophotometric with Nessler’s Reagent; Potentiometric; Spectrophotometric or Titrimetric, After Distillation); Italian Agency for the Protection of the Environment and for Technical Services: Rome, Italy, 2003. (In Italian) [Google Scholar]
- APAT-IRSA-CNR. Analytical Methods for Water—n. 4050: Nitrous Nitrogen; Italian Agency for the Protection of the Environment and for Technical Services: Rome, Italy, 2003. (In Italian) [Google Scholar]
- EPA. Method 300.1: Determination of Inorganic Anions in Drinking Water by Ion Chromatography—Revision 1.0; United States Environmental Protection Agency: Cincinnati, OH, USA, 1997.
- APAT-IRSA-CNR. Analytical Methods for Water—n. 4070: Cyanide; Italian Agency for the Protection of the Environment and for Technical Services: Rome, Italy, 2003. (In Italian) [Google Scholar]
- EPA. Method 3051A (SW-846): Microwave Assisted Acid Digestion of Sediments, Sludges, and Oils—Revision 1.0; United States Environmental Protection Agency: Cincinnati, OH, USA, 2007.
- EPA. Method 6010D (SW-846): Inductively Coupled Plasma—Atomic Emission Spectrometry—Revision 5.0; United States Environmental Protection Agency: Washington, DC, USA, 2018.
- EPA. Method 3550C (SW-846): Ultrasonic Extraction; United States Environmental Protection Agency: Cincinnati, OH, USA, 2007.
- EPA. Method 537: Determination of Selected Perfluorinated Alkyl Acids in Drinking Water by SPE and LC/MS/MS—Version 1; United States Environmental Protection Agency: Cincinnati, OH, USA, 2009.
- ISO 7875-1 ISO 7875-1:1996. Water Quality—Determination of Surfactants—Part 1: Determination of Anionic Surfactants by Measurement of the Methylene Blue Index (MBAS). Available online: https://www.iso.org/standard/24784.html (accessed on 15 April 2021).
- ISO 7875-2 ISO 7875-2:1984. Water Quality—Determination of Surfactants—Part 2: Determination of Non-Ionic Surfactants Using Dragendorff Reagent. Available online: https://www.iso.org/standard/14809.html (accessed on 15 April 2021).
- UNI. Method 10511–1:1996 + A1:2000. Water Quality—Determination of Non-Ionic Surfactants. Method for Biphasic Titration with Tetrakis (4-Fluorophenyl) Sodium Borate (TAS); UNI: Milan, Italy, 1996. (In Italian) [Google Scholar]
- Courtens, E.N.P.; Vlaeminck, S.E.; Vilchez-Vargas, R.; Verliefde, A.; Jauregui, R.; Pieper, D.H.; Boon, N. Trade-off between mesophilic and thermophilic denitrification: Rates vs. sludge production, settleability and stability. Water Res. 2014, 63, 234–244. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Carnevale Miino, M.; Manenti, S.; Todeschini, S.; Sperone, E.; Cavallo, G.; Abbà, A. Identification and Localization of Hydrodynamic Anomalies in a Real Wastewater Treatment Plant by an Integrated Approach: RTD-CFD Analysis. Environ. Process. 2020, 7, 563–578. [Google Scholar] [CrossRef]
- Sánchez, F.; Rey, H.; Viedma, A.; Nicolás-Pérez, F.; Kaiser, A.S.; Martínez, M. CFD simulation of fluid dynamic and biokinetic processes within activated sludge reactors under intermittent aeration regime. Water Res. 2018, 139, 47–57. [Google Scholar] [CrossRef]
- Gomez, V.; Ferreres, L.; Pocurull, E.; Borrull, F. Determination of non-ionic and anionic surfactants in environmental water matrices. Talanta 2011, 84, 859–866. [Google Scholar] [CrossRef]
- Kruszelnicka, I.; Ginter-Kramarczyk, D.; Wyrwas, B.; Idkowiak, J. Evaluation of surfactant removal efficiency in selected domestic wastewater treatment plants in Poland. J. Environ. Health Sci. Eng. 2019, 17, 1257–1264. [Google Scholar] [CrossRef] [Green Version]
- Chang, I.-S.; Chung, C.-M.; Han, S.-H. Treatment of oily wastewater by ultrafiltration and ozone. Desalination 2001, 133, 225–232. [Google Scholar] [CrossRef]
- Foladori, P.; Andreottola, G.; Ziglio, G. Sludge Reduction Technologies in Wastewater Treatment Plants; IWA Publishing: London, UK, 2010. [Google Scholar]
- Tchobanoglou, G.; Burton, F.L.; Stensel, H.D.; Eddy, M. Wastewater Engineering: Treatment and Resource Recovery; McGraw Hill Education: New York, NY, USA, 2014. [Google Scholar]
- Acharya, C.; Nakhla, G.; Bassi, A.; Kurian, R. Treatment of High-Strength Pet Food Wastewater Using Two-Stage Membrane Bioreactors. Water Environ. Res. 2006, 78, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Abeynayaka, A.; Visvanathan, C. Performance comparison of mesophilic and thermophilic aerobic sidestream membrane bioreactors treating high strength wastewater. Bioresour. Technol. 2011, 102, 5345–5352. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Liu, Y.; Wang, C. Modeling of enhanced denitrification capacity with microbial storage product in MBR systems. Sep. Purif. Technol. 2014, 126, 1–6. [Google Scholar] [CrossRef]
- Bassin, J.P.; Kleerebezem, R.; Muyzer, G.; Rosado, A.S.; van Loosdrecht, M.C.M.; Dezotti, M. Effect of different salt adaptation strategies on the microbial diversity, activity, and settling of nitrifying sludge in sequencing batch reactors. Appl. Microbiol. Biotechnol. 2012, 93, 1281–1294. [Google Scholar] [CrossRef] [Green Version]
- Ochoa-Herrera, V.; Field, J.A.; Luna-Velasco, A.; Sierra-Alvarez, R. Microbial toxicity and biodegradability of perfluorooctane sulfonate (PFOS) and shorter chain perfluoroalkyl and polyfluoroalkyl substances (PFASs). Environ. Sci. Process. Impacts 2016, 18, 1236–1246. [Google Scholar] [CrossRef] [PubMed]
- Chiavola, A.; Di Marcantonio, C.; Boni, M.R.; Biagioli, S.; Frugis, A.; Cecchini, G. PFOA and PFOS Removal Processes in Activated Sludge Reactor at Laboratory Scale; Springer: Berlin/Heidelberg, Germany, 2020; pp. 375–377. [Google Scholar]
- Liwarska-Bizukojć, E.; Chojnacki, J.; Klink, M.; Olejnik, D. Effect of the type of the external carbon source on denitrification kinetics of wastewater. Desalin. Water Treat. 2018, 101, 143–150. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).