Agricultural Conservation Practices and Aquatic Ecological Responses
Abstract
1. Introduction
2. Methods
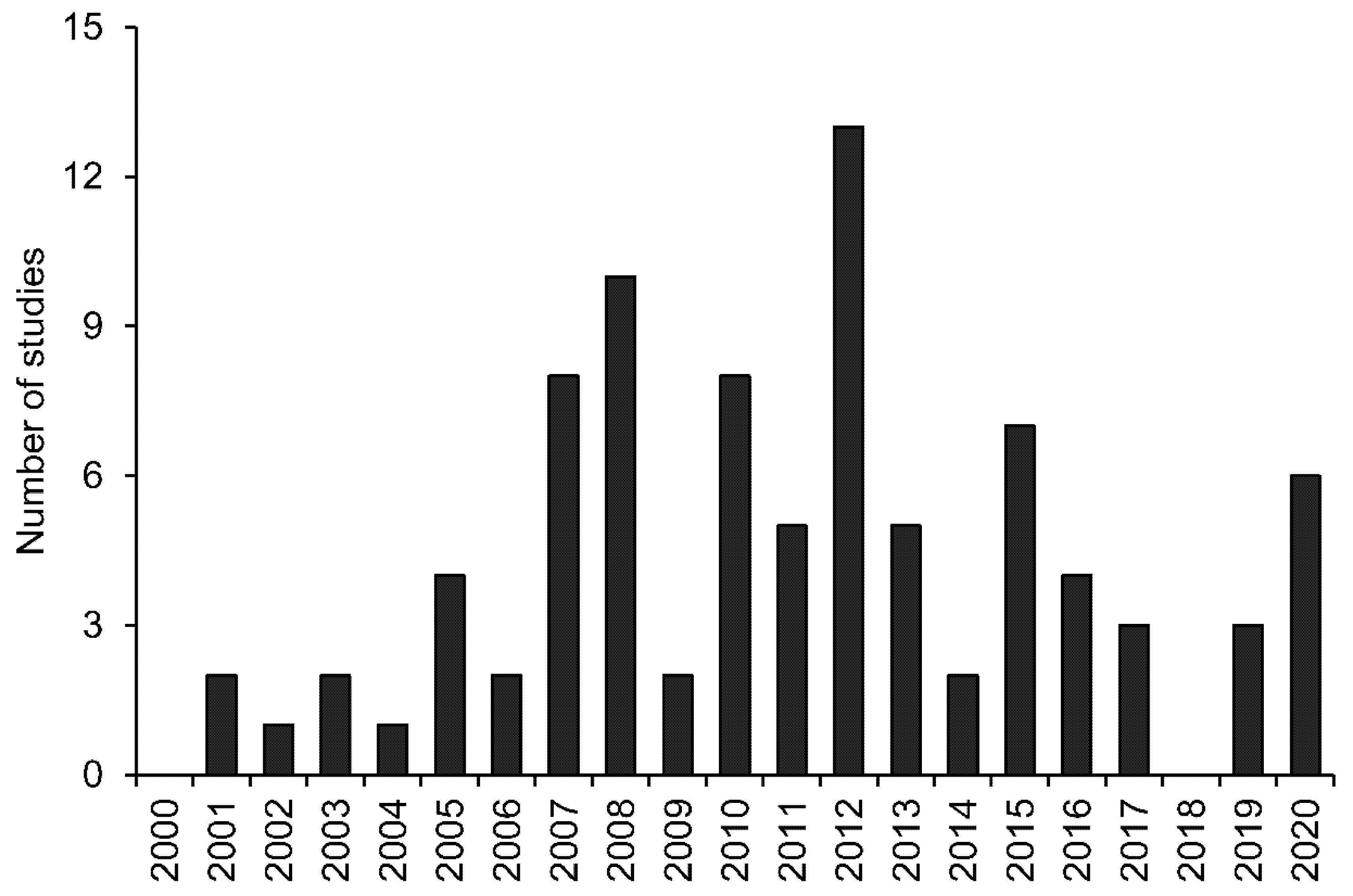
3. Results and Discussion
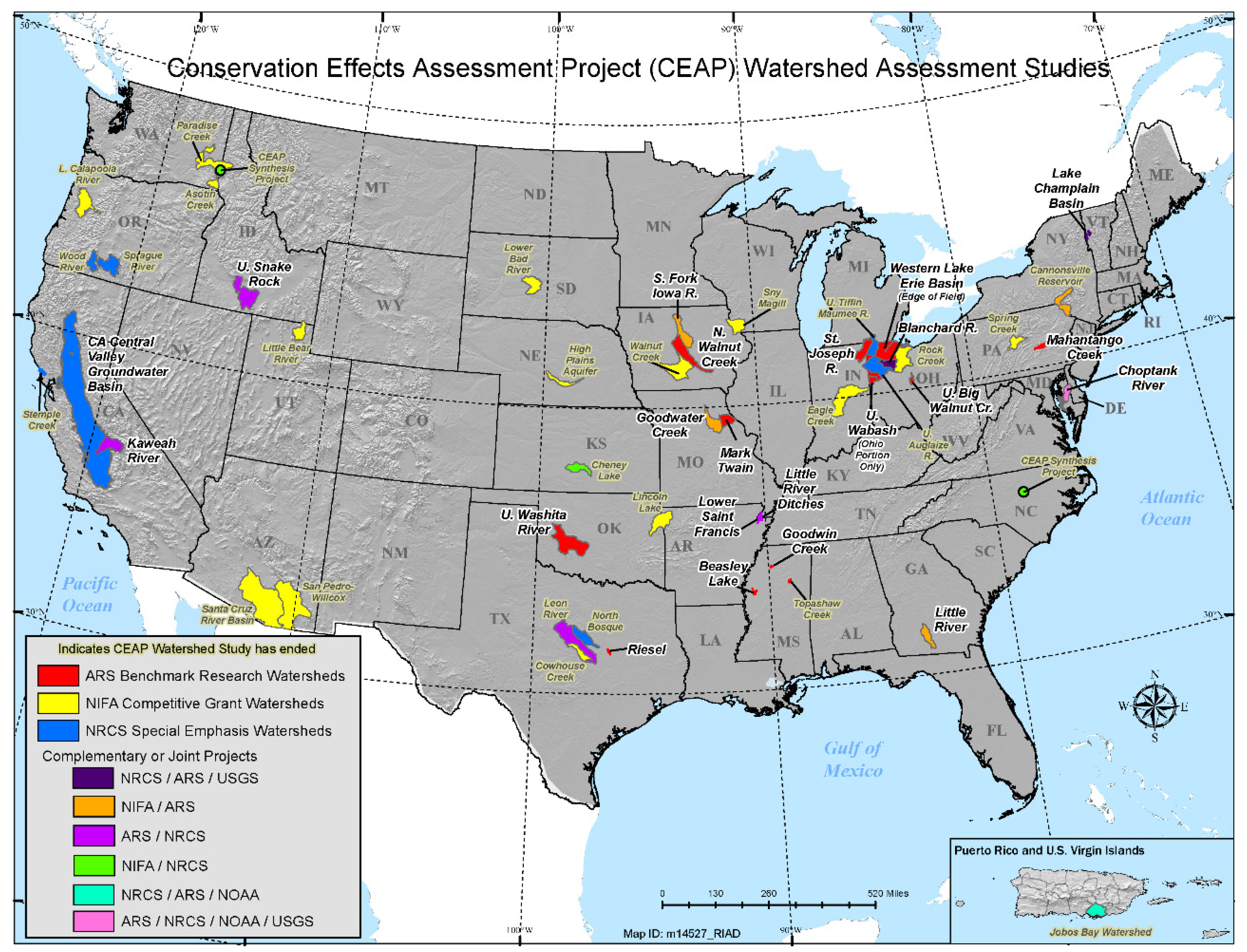
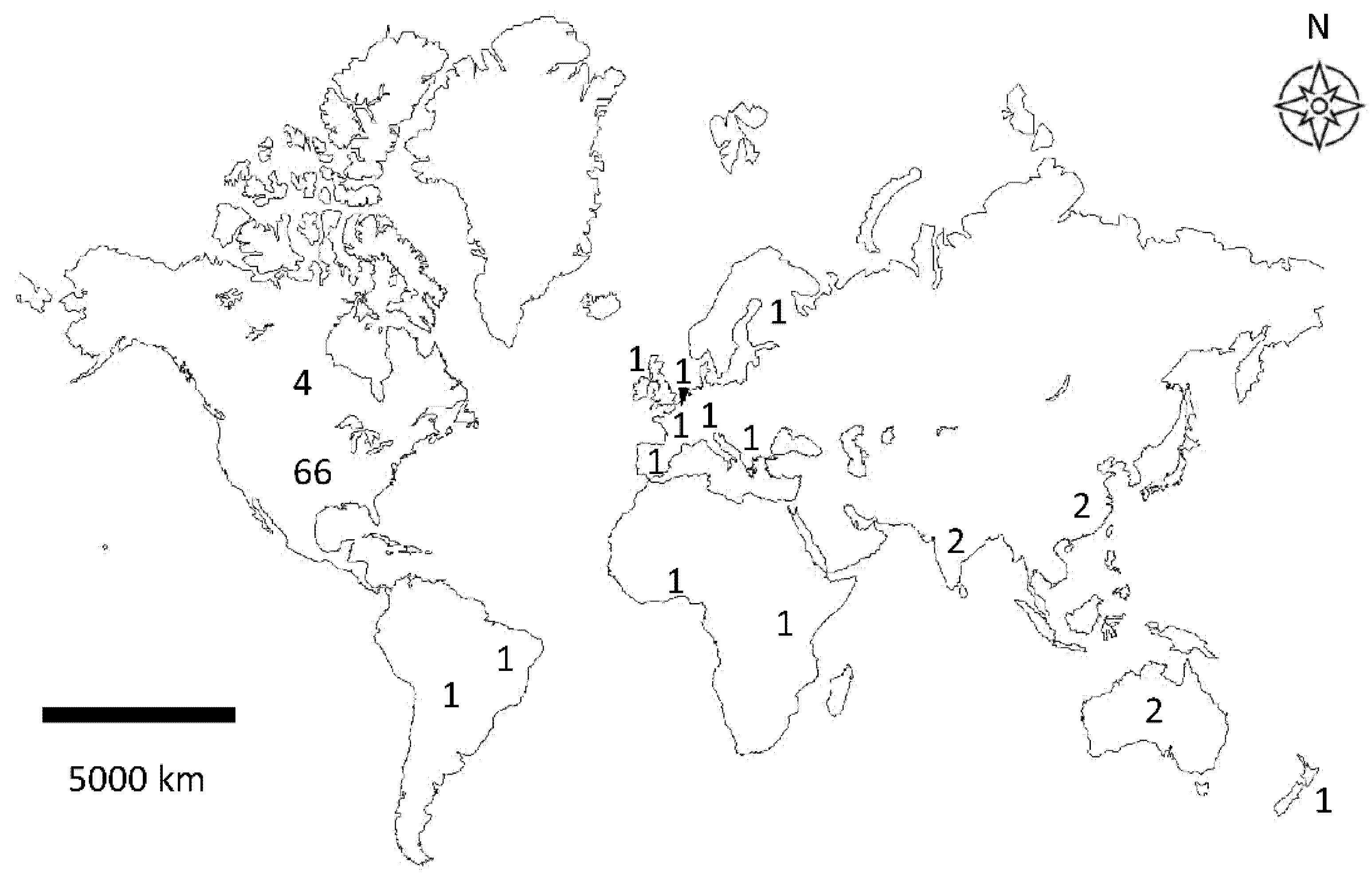
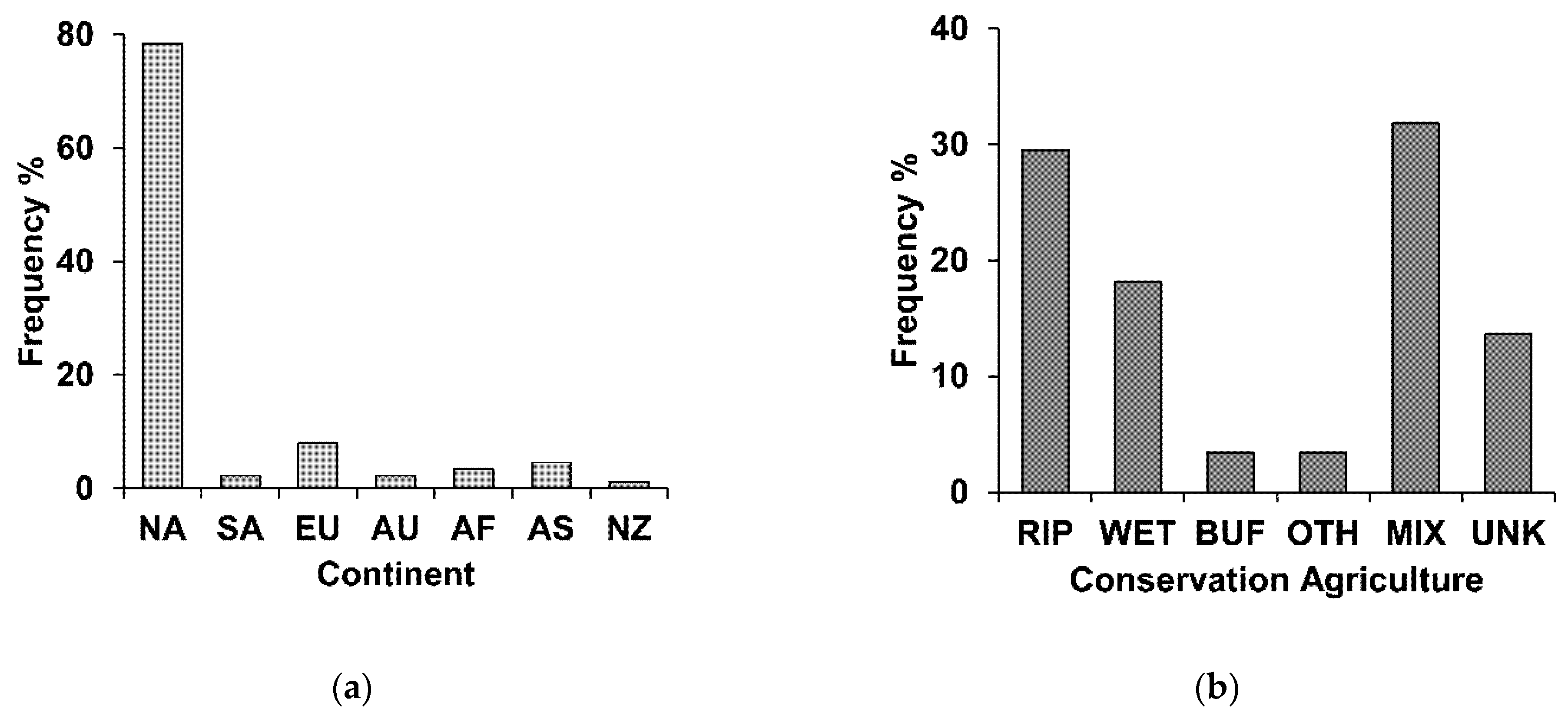
3.1. Study Locations and Conservation Agriculture Practices
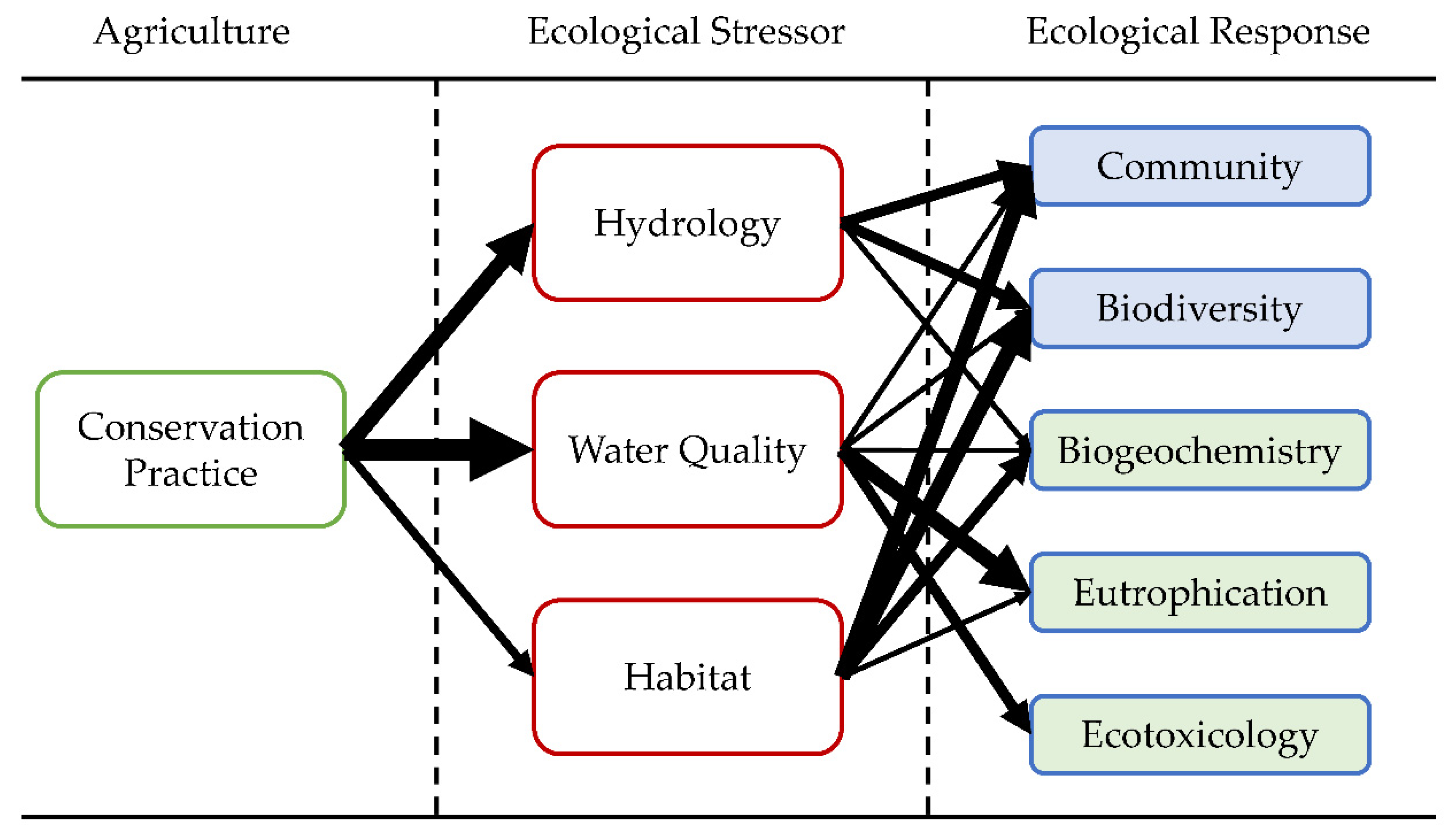
3.2. Ecological Responses to Conservation Agriculture
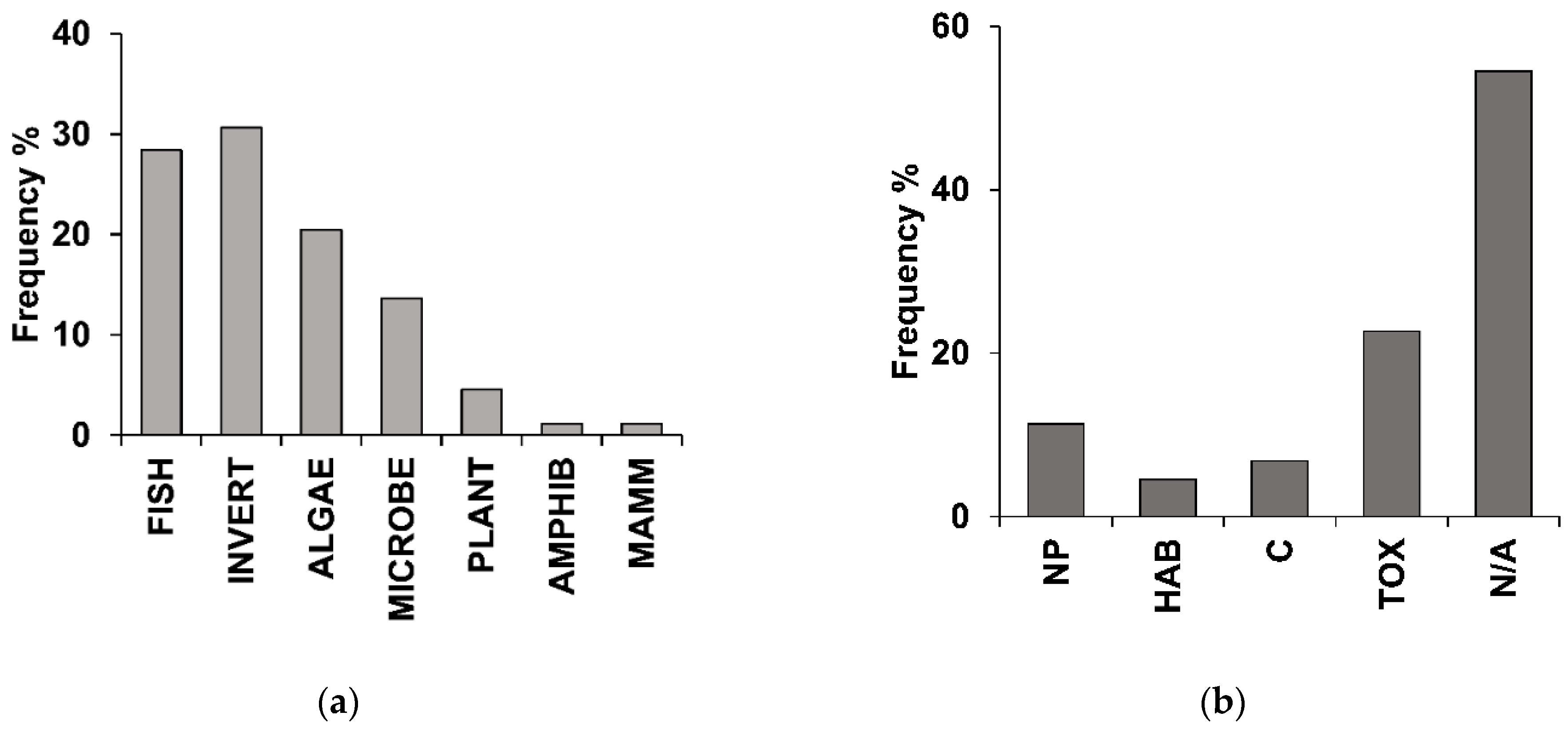
3.3. Structural Ecological Responses
3.4. Functional Ecological Responses
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Costanza, R.; Mageau, M. What is a healthy ecosystem? Aquat. Ecol. 1999, 33, 105–115. [Google Scholar] [CrossRef]
- Watzin, M.C.; McIntosh, A.W. Aquatic ecosystems in agricultural landscapes: A review of ecological indicators and achievable ecological outcomes. J. Soil Water Conserv. 1999, 54, 636–644. [Google Scholar]
- Tscharntke, T.; Klein, A.M.; Kruess, A.; Steffan-Dewenter, I.; Thies, C. Landscape perspectives on agricultural intensification and biodiversity—Ecosystem service management. Ecol. Lett. 2005, 8, 857–874. [Google Scholar] [CrossRef]
- Zhang, W.; Ricketts, T.H.; Kremen, C.; Carney, K.; Swinton, S.M. Ecosystem services and dis-services to agriculture. Ecol Econ. 2007, 64, 253–260. [Google Scholar] [CrossRef]
- Bennett, E.M.; Peterson, G.D.; Gordon, L.J. Understanding relationships among multiple ecosystem services. Ecol. Lett. 2009, 12, 1394–1404. [Google Scholar] [CrossRef]
- Schmutz, S.; Sendzimir, J. Riverine Ecosystem Management. Science for Governing Towards a Sustainable Future; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Stehle, S.; Elsaesser, D.; Gregoire, C.; Imfeld, G.; Niehaus, E.; Passeport, E.; Payraudeau, S.; Schäfer, R.B.; Tournebize, J.; Schulz, R. Pesticide risk mitigation by vegetated treatment systems: A meta-analysis. J. Environ. Qual. 2011, 40, 1068–1080. [Google Scholar] [CrossRef]
- Yu, J.; Wu, J. The sustainability of agricultural development in China: The agriculture–environment nexus. Sustainability 2018, 10, 1776. [Google Scholar] [CrossRef]
- Withers, P.J.A.; Vadas, P.A.; Uusitalo, R.; Forber, K.J.; Hart, M.; Foy, R.H.; Delgado, A.; Dougherty, W.; Lilja, H.; Burkitt, L.L.; et al. A global perspective on integrated strategies to manage soil phosphorus status for eutrophication control without limiting land productivity. J. Environ. Qual. 2019, 48, 1234–1246. [Google Scholar] [CrossRef] [PubMed]
- Syabeera, N.; Ahmad, B.N.; Mustafa, F.B.; Yusoff, S.Y.M.; Didams, G. A systematic review of soil erosion control practices on the agricultural land in Asia. Int. Soil Water Conserv. Res. 2020, 8, 103–115. [Google Scholar] [CrossRef]
- Wato, T.; Amare, M. The agricultural water pollution and its minimization strategies—A review. J. Resour. Dev. Manag. 2020, 64, 10–22. [Google Scholar] [CrossRef]
- Shively, D.; Rothlisberger, J.D.; Gillespie, N.; Dombek, M.; Roghair, C.; Demario, D.; McCormick, F.; Weldon, L. Historical perspectives and a new U.S. Forest Service Strategy for Fish and Aquatic Stewardship. Fisheries 2018, 43, 386–395. [Google Scholar] [CrossRef]
- Barling, R.D.; Moore, I.D. Role of buffer strips in management of waterway pollution: A review. Environ. Manag. 1994, 18, 543–558. [Google Scholar] [CrossRef]
- Broadmeadow, S.; Nisbet, T.R. The effects of riparian forest management on the freshwater environment: A literature review of best management practice. Hydrol. Earth Syst. Sci. 2004, 8, 286–305. [Google Scholar] [CrossRef]
- Henle, K.; Alard, D.; Clitherow, J.; Cobb, P.; Firbank, L.; Kull, T.; McCracken, D.; Moritz, R.F.A.; Niemelä, J.; Rebane, M.; et al. Identifying and managing the conflicts between agriculture and biodiversity conservation in Europe—A review. Agric. Ecosyst. Environ. 2008, 124, 60–71. [Google Scholar] [CrossRef]
- Maret, T.R.; MacCoy, D.E.; Carlisle, D.M. Long-term water quality and biological responses to multiple best management practices in Rock Creek, Idaho. J. Am. Water Resour. Assoc. 2008, 44, 1248–1269. [Google Scholar] [CrossRef]
- Smiley, P.C.; Gillespie, R.B. Influence of physical habitat and agricultural contaminants on fishes in agricultural drainage ditches. In Agricultural Drainage Ditches: Mitigation Wetlands of the 21st Century; Moore, M., Kroger, R., Eds.; Research Signpost: Kerula, India, 2010; pp. 37–73. [Google Scholar]
- Pierce, S.C.; Kröger, R.; Pezeshki, R. Managing artificially drained low-gradient agricultural headwaters for enhanced ecosystem functions. Biology 2012, 1, 794–856. [Google Scholar] [CrossRef]
- Smiley, P.C., Jr.; Shields, F.D., Jr.; Knight, S.S. Designing impact assessments for evaluating ecological effects of agricultural conservation practices on streams. J. Am. Water Resour. Assoc. 2009, 45, 867–878. [Google Scholar] [CrossRef]
- Larned, S.T.; Schallengberg, M. Stressor-response relationships and the prospective management of aquatic ecosystems. N. Z. J. Mar. Freshw. Res. 2019, 53, 489–512. [Google Scholar] [CrossRef]
- McPhee, C.; Bancerz, M.; Mambrini-Doudet, M.; Chrétien, F.; Huyghe, C.; Gracia-Garza, J. The defining characteristics of agroecosystem living Labs. Sustainability 2021, 13, 1718. [Google Scholar] [CrossRef]
- Jiao, W.; Min, Q. Reviewing the progress in the identification, conservation and management of China-Nationally important agricultural heritage systems (China-NIAHS). Sustainability 2017, 9, 1698. [Google Scholar] [CrossRef]
- Townsend, P.V.; Harper, R.J.; Brennan, P.D.; Dean, C.; Wu, S.; Smettem, K.R.J.; Cook, S.E. Multiple environmental services as an opportunity for watershed restoration. For. Policy Econ. 2012, 17, 45–58. [Google Scholar] [CrossRef]
- McDowell, R.W.; Dils, R.M.; Collins, A.L.; Flahive, K.A.; Sharpley, A.N.; Quinn, J. A review of the policies and implementation of practices to decrease water quality impairment by phosphorus in New Zealand, the UK, and the US. Nutr. Cycl. Agroecosyst. 2016, 104, 289–305. [Google Scholar] [CrossRef]
- Tomer, M.D.; Locke, M.A. The challenge of documenting water quality benefits of conservation practices: A review of USDA-ARS’s conservation effects assessment project watershed studies. Water Sci. Technol. 2011, 64, 300–310. [Google Scholar] [CrossRef]
- Moriasi, D.N.; Duriancik, L.F.; Sadler, E.J.; Tsegaye, T.; Steiner, J.L.; Locke, M.A.; Strickland, T.C.; Osmond, D.L. Quantifying the impacts of the Conservation Effects Assessment Project watershed assessments: The first fifteen years. J. Soil Water Conserv. 2020, 75, 57A–74A. [Google Scholar] [CrossRef]
- Álvarez, X.; Valero, E.; Santos, R.M.B.; Varandas, S.G.P.; Sanches Fernandes, L.F.; Pacheco, F.A.L. Anthropogenic nutrients and eutrophication in multiple land use watersheds: Best management practices and policies for the protection of water resources. Land Use Policy 2017, 69, 1–11. [Google Scholar] [CrossRef]
- Barletta, M.; Jaureguizar, A.J.; Baigun, C.; Fontoura, N.F.; Agostinho, A.A.; Almeida-Val, V.M.F.; Val, A.L.; Torres, R.A.; Jimenes-Segura, L.F.; Giarrizzo, T.; et al. Fish and aquatic habitat conservation in South America: A continental overview with emphasis on neotropical systems. J. Fish Biol. 2010, 76, 2118–2176. [Google Scholar] [CrossRef]
- Beketov, M.A.; Kefford, B.J.; Schäfer, R.B.; Liess, M. Pesticides reduce regional biodiversity of stream invertebrates. Proc. Natl. Acad. Sci. USA 2013, 110, 11039–11043. [Google Scholar] [CrossRef] [PubMed]
- Boesch, D.F.; Brinsfield, R.B.; Magnien, R.F. Chesapeake Bay eutrophication: Scientific understanding, ecosystem restoration, and challenges for agriculture. J. Environ. Qual. 2001, 30, 303–320. [Google Scholar] [CrossRef]
- Brooks, R.P.; Yetter, S.E.; Carline, R.F.; Shortle, J.S.; Bishop, J.A.; Ingram, H.; Weller, D.; Boomer, K.; Stedman, R.; Armstrong, A.; et al. Analysis of BMP Implementation Performance and Maintenance in Spring Creek, an Agriculturally-Influenced Watershed in Pennsylvania; USDA Project Number: PENW-2006-03984; USDA-NIFA Report; USDA: Washington, DC, USA, 2011; 66p.
- Brooks, B.W.; Lazorchak, J.M.; Howard, M.D.A.; Johnson, M.V.; Morton, S.L.; Perkins, D.A.K.; Reavie, E.D.; Scott, G.I.; Smith, S.A.; Steevens, J.A. Are harmful algal blooms becoming the greatest inland water quality threat to public health and aquatic ecosystems? Environ. Toxicol. Chem. 2016, 35, 6–13. [Google Scholar] [CrossRef]
- Bullerjahn, G.S.; McKay, R.M.; Davis, T.W.; Baker, D.B.; Boyer, G.L.; D’Anglada, L.V.; Doucette, G.J.; Ho, J.C.; Irwin, E.G.; Kling, C.L.; et al. Global solutions to regional problems: Collecting global expertise to address the problem of harmful cyanobacterial blooms. A Lake Erie case study. Harmful Algae 2016, 54, 223–238. [Google Scholar] [CrossRef]
- Carey, R.; Vellidis, G.; Lowrance, R.; Pringle, C. Nutrient Enrichment and Stream Periphyton Growth in the Southern Coastal Plain of Georgia; ASAE Paper No. 052197; ASAE: St. Joseph, MI, USA, 2005; 13p. [Google Scholar]
- Carey, R.; Vellidis, G.; Lowrance, R.; Pringle, C. Do nutrients limit algal periphyton in small blackwater coastal plain streams? J. Am. Water Resour. Assoc. 2007, 43, 1183–1193. [Google Scholar] [CrossRef]
- Chapman, L.J.; Chapman, C.A.; Kaufman, L.; Witte, F.; Balirwa, J. Biodiversity conservation in African inland waters: Lessons of the Lake Victoria region. SIL Proc. Int. Ver. Theor. Angew. Limnol. Verh. 2008, 30, 16–34. [Google Scholar] [CrossRef]
- Christensen, V.G.; Lee, K.E.; McLees, J.M.; Niemela, S.L. Relations between retired agricultural land, water quality, and aquatic-community health, Minnesota River Basin. J. Environ. Qual. 2012, 41, 1459–1472. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Minns, C.K.; Lester, N.P.; Mandrak, N.E. An updated assessment of human activities, the environment, and freshwater fish biodiversity in Canada. Can. J. Fish Aquat. Sci. 2015, 72, 135–148. [Google Scholar] [CrossRef]
- Cullum, R.F.; Knight, S.S.; Cooper, C.M.; Smith, S. Combined effects of best management practices on water quality in oxbow lakes from agricultural watersheds. Soil Tillage Res. 2006, 90, 212–221. [Google Scholar] [CrossRef]
- Dembkowski, D.J.; Miranda, L.E. Hierarchy in factors affecting fish biodiversity in floodplain lakes of the Mississippi Alluvial Valley. Environ. Biol. Fishes 2012, 93, 357–368. [Google Scholar] [CrossRef]
- Deegan, D.J. Endocrine Disrupting Effects on Creek Chubs (Semotilus atromaculatus) Exposed to Agricultural Contaminants in the Cedar Creek Watershed, Northeast Indiana. Master’s Thesis, Purdue University, Fort Wayne, IN, USA, May 2010. [Google Scholar]
- De Steven, D.; Lowrance, R. Agricultural conservation practices and wetland ecosystem services in the wetland-rich Piedmont-Coastal Plain region. Ecol. Appl. 2011, 21, S3–S17. [Google Scholar] [CrossRef]
- Dudgoen, D. River rehabilitation for conservation of fish biodiversity in monsoonal Asia. Ecol. Soc. 2005, 10, 15. Available online: http://www.ecologyandsociety.org/vol10/iss2/art15/ (accessed on 7 February 2021). [CrossRef]
- Ellison, C.A.; Skinner, Q.D.; Hicks, L.S. Assessment of best-management practice effects on rangeland stream water quality using multivariate statistical techniques. Rangel. Ecol. Manag. 2009, 62, 371–386. [Google Scholar] [CrossRef]
- Fraker, M.E.; Keitzer, S.C.; Sinclair, J.S.; Aloysius, N.R.; Dippold, D.A.; Yen, H. Projecting the effects of agricultural conservation practices on stream fish communities in a changing climate. Sci. Total Environ. 2020, 747, 141112. [Google Scholar] [CrossRef]
- Fu, C.; Wu, J.; Chen, J.; Wu, Q.; Lei, G. Freshwater fish biodiversity in the Yangtze River basin of China: Patterns, threats and conservation. Biodivers. Conserv. 2003, 12, 1649–1685. [Google Scholar] [CrossRef]
- Gagliardi, B.; Pettigrove, V. Removal of intensive agriculture from the landscape improves aquatic ecosystem health. Agric. Ecosyst. Environ. 2013, 176, 1–8. [Google Scholar] [CrossRef]
- Gbaguidi, H.M.A.G.; Adite, A.; Sossoukpe, E. Ecology and fish biodiversity of man-made lakes of Southern Benin (West Africa): Implications for species conservation and fisheries management. J. Environ. Prot. 2016, 7, 874–894. [Google Scholar] [CrossRef][Green Version]
- Goeller, B.C.; Febria, C.M.; McKergow, L.A.; Harding, J.S. Combining tools from edge-of-field to in-stream to attenuate reactive nitrogen along small agricultural waterways. Water 2020, 12, 383. [Google Scholar] [CrossRef]
- Gilbert, P.M. From hogs to HABs: Impacts of industrial farming in the US on nitrogen and phosphorus and greenhouse gas pollution. Biogeochemistry 2020, 150, 139–180. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.R.; Herbert, M.E.; Sowa, S.P.; Mysorekar, S.; Woznicki, S.A.; Nejadhashemi, P.A.; Wang, L. Reducing current and future risks: Using climate change scenarios to test an agricultural conservation framework. J. Great Lakes Res. 2017, 43, 59–68. [Google Scholar] [CrossRef]
- Holmes, R.; Armanini, D.G.; Yates, A.G. Effects of best management practice on ecological condition: Does location matter? Environ. Manag. 2016, 57, 1062–1076. [Google Scholar] [CrossRef] [PubMed]
- Hunt, L.; Marrochi, N.; Bonetto, C.; Liess, M.; Buss, D.F.; Vieira da Silva, C.; Chiu, M.-C.; Resh, V.H. Do riparian buffers protect stream invertebrate communities in South American Atlantic Forest agricultural areas? Environ. Manag. 2017, 60, 1155–1170. [Google Scholar] [CrossRef]
- James, R.T.; Havens, K.E.; McOrmick, P.; Jones, B.; Ford, C. Water quality trends in shallow South Florida lakes and assessment of regional versus local forcing functions. Crit. Rev. Environ. Sci. Technol. 2011, 41 (Suppl. S1), 576–607. [Google Scholar] [CrossRef]
- Jordan, M.A.; Patel, D.; Sanders, K.E.; Gillespie, R.B. The relative roles of contemporary and ancient processes in shaping genetic variation of a generalist fish in a catchment dominated by agriculture. Freshw. Biol. 2013, 58, 1660–1671. [Google Scholar] [CrossRef]
- King, K.W.; Smiley, P.C.; Baker, B.J.; Fausey, N.R. Validation of paired watersheds for assessing conservation practices in the Upper Big Walnut Creek watershed, Ohio. J. Soil Water Conserv. 2008, 63, 380–395. [Google Scholar] [CrossRef]
- Knight, S.S.; Welch, T.D. Ecological and water quality effects of the Mississippi Delta management systems evaluation area on oxbow lakes. In Proceedings of the Mississippi Water Resources Conference, Raymond, MS, USA, 23 April 2002; pp. 131–143. [Google Scholar]
- Knight, S.S.; Welch, T.D. Evaluation of watershed management practices on oxbow lake ecology and water quality. In Water Quality Assessments in the Mississippi Delta: Regional Solutions, National Scope; Nett, M.T., Locke, M.A., Pennington, D.A., Eds.; ACS Symposium Series 877; American Chemical Society: Washington, DC, USA, 2004; pp. 119–133. [Google Scholar]
- Knight, S.S.; Cullum, R.F.; Cooper, C.M.; Lizotte, R.E. Effects of suspended sediments on the chlorophyll-phosphorus relationship in oxbow lakes. Int. J. Ecol. Environ. Sci. 2008, 34, 1–6. [Google Scholar]
- Knight, S.S.; Lizotte, R.E.; Smith, S.; Bryant, C.T. Responses of Hyalella azteca to chronic exposure of Mississippi Delta sediments. J. Environ. Sci. Eng. 2010, 4, 1–12. [Google Scholar]
- Knight, S.S.; Locke, M.A.; Smith, S. Effects of agricultural conservation practices on oxbow lake watersheds in the Mississippi River alluvial plain. Soil Water Res. 2013, 8, 113–123. [Google Scholar] [CrossRef]
- Knight, S.S.; Cullum, R.F. Effects of conservation practices on fisheries management. J. Agric. Biodivers. Res. 2014, 3, 1–8. [Google Scholar]
- Knight, S.; Cullum, R.; Lizotte, R. The limnology of a Mississippi River Alluvial Plain oxbow lake following the application of conservation practices. J. Water Resour. Prot. 2015, 7, 707–714. [Google Scholar] [CrossRef][Green Version]
- Kovalenko, K.E.; Johnson, L.B.; Brady, V.J.; Ciborowski, J.J.H.; Cooper, M.J.; Gathman, J.P.; Lamberti, G.A.; Moerke, A.H.; Ruetz, C.R.; Uzarski, D.G. Hotspots and bright spots in functional and taxonomic fish diversity. Freshw. Sci. 2019, 38, 480–490. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Knight, S.S.; Cooper, C.M. Toxicity evaluation of a conservation effects assessment program watershed, Beasley Lake, in the Mississippi Delta, USA. Bull. Environ. Contam. Toxicol. 2010, 84, 422–426. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Knight, S.S.; Bryant, C.T. Sediment quality assessment of Beasley Lake: Bioaccumulation and effects of pesticides in Hyalella azteca. Chem. Ecol. 2010, 26, 411–424. [Google Scholar] [CrossRef]
- Lizotte, R.; Knight, S.; Locke, M.; Stienriede, W.; Testa, S.; Bryant, C. Water quality monitoring of an agricultural watershed lake: The effectiveness of agricultural best management practices. In Design and Nature IV: Comparing Design in Nature with Science and Engineering; Hernandez, S., Brebbia, C.A., Eds.; WIT transactions in Ecology and the Environment Series; WIT Press: Southampton, UK, 2012; Volume 160, pp. 283–294. [Google Scholar]
- Lizotte, R.E.; Shields, F.D.; Testa, S. Effects of a simulated agricultural runoff event on sediment toxicity in a managed backwater wetland. Water Air Soil Pollut. 2012, 223, 5375–5389. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Shields, F.D.; Murdock, J.N.; Knight, S.S. Responses of Hyalella azteca and phytoplankton to a simulated agricultural runoff event in a managed backwater wetland. Chemosphere 2012, 87, 684–691. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Knight, S.S.; Locke, M.A.; Steinriede, R.W. Ten-year assessment of agricultural management and land-use practices on pesticide loads and risk to aquatic biota of an oxbow lake in the Mississippi Delta, USA. In Agricultural Research Updates; Hendricks, B.P., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2012; Volume 2, pp. 349–371. [Google Scholar]
- Lürling, M.; Mucci, M. Mitigating eutrophication nuisance: In-lake measures are becoming inevitable in eutrophic waters in the Netherlands. Hydrobiologia 2020, 847, 4447–4467. [Google Scholar] [CrossRef]
- McKinney, E.N. Relative Contribution of Water Auality and Habitat to Macroinvertebrate Community Composition in Streams Influenced by Agricultural Land Use in the Cedar Creek Watershed, Indiana. Master’s Thesis, Purdue University Fort Wayne, Fort Wayne, IN, USA, May 2012. [Google Scholar]
- Mehring, A.S.; Vellidis, G.; Pringle, C.M.; Kuehn, K.A.; Lowrance, R.R.; Rosemond, A.D. The effects of tree species on microbial respiration and leaf breakdown in a coastal plain blackwater stream. In Proceedings of the 2007 Georgia Water Resources Conference, Athens, GA, USA, 27–29 March 2007. [Google Scholar]
- Moore, A.A.; Palmer, M.A. Invertebrate biodiversity in agricultural and urban headwater streams: Implications for conservation and management. Ecol. Appl. 2005, 15, 1169–1177. [Google Scholar] [CrossRef]
- Moore, M.T.; Lizotte, R.E.; Knight, S.S.; Smith, S.; Cooper, C.M. Assessment of pesticide contamination in three Mississippi Delta oxbow lakes using Hyalella azteca. Chemosphere 2007, 67, 2184–2191. [Google Scholar] [CrossRef]
- Moore, M.T.; Lizotte, R.E.; Smith, S. Responses of Hyalella azteca to a pyrethroid mixture in a constructed wetland. Bull. Environ. Contam. Toxicol. 2007, 78, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.T.; Lizotte, R.E.; Smith, S. Toxicity evaluation of diazinon contaminated leaf litter. Bull. Environ. Contam. Toxicol. 2007, 78, 168–171. [Google Scholar] [CrossRef]
- Moran, K.; Anderson, B.; Phillips, B.; Luo, Y.; Singhasemanon, N.; Breuer, R.; Tadesse, D. Water quality impairments due to aquatic life pesticide toxicity: Prevention and mitigation in California, USA. Environ. Toxicol. Chem. 2019, 39, 953–966. [Google Scholar] [CrossRef]
- Pearce, N.J.T.; Yates, A.G. Agricultural best management practice abundance and location does not influence stream ecosystem function or water quality in the summer season. Water 2015, 7, 6861–6876. [Google Scholar] [CrossRef]
- Porter, P.A.; Mitchell, R.B.; Moore, K.J. Reducing hypoxia in the Gulf of Mexico: Reimagining a more resilient agricultural landscape in the Mississippi River Watershed. J. Soil Water Conserv. 2015, 70, 63A–68A. [Google Scholar] [CrossRef]
- Sanders, K.E. Relative Importance of Water Quality and Habitat to Fish Communities in Streams Influenced by Agricultural Land Use in the Cedar Creek Watershed, Indiana. Master’s Thesis, Purdue University Fort Wayne, Fort Wayne, IN, USA, May 2012. [Google Scholar]
- Sarkar, U.K.; Pathak, A.K.; Sinha, R.K.; Sivakumar, K.; Pandian, A.K.; Pandey, A.; Dubey, V.K.; Lakra, W.S. Freshwater fish biodiversity in the River Ganga (India): Changing pattern, threats and conservation perspectives. Rev. Fish Biol. Fish. 2012, 22, 251–272. [Google Scholar] [CrossRef]
- Sarkar, U.K.; Bakshi, S.; Lianthuamluaia, L.; Mishal, P.; Das Ghosh, B.; Saha, S.; Karnatak, G. Understanding enviro-climatological impact on fish biodiversity of the tropical floodplain wetlands for their sustainable management. Sustain. Water Resour. Manag. 2020, 6, 96. [Google Scholar] [CrossRef]
- Schäfer, R.B.; Caquet, T.; Siimes, K.; Mueller, R.; Lagadic, L.; Liess, M. Effects of pesticides on community structure and ecosystem functions in agricultural streams of three biogeographical regions in Europe. Sci. Total Environ. 2007, 382, 272–285. [Google Scholar] [CrossRef]
- Seger, K.R.; Smiley, P.C.; King, K.W. Influence of riparian habitat on aquatic macroinvertebrate community colonization within riparian zones of agricultural headwater streams. J. Freshw. Ecol. 2012, 27, 393–407. [Google Scholar] [CrossRef]
- Smiley, P.C.; Gillespie, R.B.; King, K.W.; Huang, C. Contribution of habitat and water quality to the integrity of fish communities in agricultural drainage ditches. J. Soil Water Conserv. 2008, 63, 218A–219A. [Google Scholar] [CrossRef]
- Smiley, P.C.; King, K.W.; Fausey, N.R. Public health perspectives of channelized and unchannelized headwater streams in central Ohio: A case study. J. Water Health 2010, 8, 577–592. [Google Scholar] [CrossRef][Green Version]
- Smiley, P.C.; King, K.W.; Fausey, N.R. Influence of herbaceous riparian buffers on physical habitat, water chemistry, and stream communities within channelized agricultural headwater streams. Ecol. Eng. 2011, 37, 1314–1323. [Google Scholar] [CrossRef]
- Smiley, P.C.; King, K.W.; Gillespie, R.B.; Fausey, N.R. Influence of watershed scale atrazine reduction practices on pesticides and fishes within channelized agricultural headwater streams. J. Sustain. Watershed Sci. Manag. 2012, 1, 61–75. [Google Scholar] [CrossRef]
- Smiley, P.C.; Rumora, K.R. Planting grass filter strips: Does it influence the structure and function of riparian habitats of agricultural headwater streams? Riparian Ecol. Conserv. 2015, 2, 58–71. [Google Scholar] [CrossRef]
- Smith, S.; Lizotte, R.E.; Knight, S.S. Pesticide body residues of Hyalella azteca exposed to Mississippi Delta sediments. Bull. Environ. Contam. Toxicol. 2007, 78, 26–29. [Google Scholar] [CrossRef]
- Smith, S.; Lizotte, R.E.; Moore, M.T. Toxicity assessment of diazinon in a constructed wetland using Hyalella azteca. Bull. Environ. Contam. Toxicol. 2007, 79, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.G.; Ferrell, G.M.; Harned, D.A.; Cuffney, T.F. A Study of the Effects of Implementing Agricultural Best Management Practices and In-Stream Restoration on Suspended Sediment, Stream Habitat, and Benthic Macroinvertebrates at Three Stream Sites in Surry County, North Carolina, 2004–2007—Lessons Learned; Scientific Investigations Report, 2011–5098; U.S. Department of the Interior: Washington, DC, USA; U.S. Geological Survey: Reston, VA, USA, 2011.
- Smith, A.; Tetzlaff, D.; Gelbrecht, J.; Kleine, L.; Soulsby, C. Riparian wetland rehabilitation and beaver re-colonization impacts on hydrological processes and water quality in a lowland agricultural catchment. Sci. Total Environ. 2020, 699, 134302. [Google Scholar] [CrossRef]
- Stephens, W.W.; Moore, M.T.; Farris, J.L.; Bouldin, J.L.; Cooper, C.M. Considerations for assessments of wadable drainage systems in the agriculturally dominated Deltas of Arkansas and Mississippi. Arch. Environ. Contam. Toxicol. 2008, 55, 432–441. [Google Scholar] [CrossRef]
- Todd, M.J.; Vellidis, G.; Lowrance, R.R.; Pringle, C.M. High sediment oxygen demand within an instream swamp in southern Georgia: Implications for low dissolved oxygen levels in coastal blackwater streams. J. Am. Water Resour. Assoc. 2009, 45, 1493–1507. [Google Scholar] [CrossRef]
- Todd, M.J.; Lowrance, R.R.; Goovaerts, P.; Vellidis, G.; Pringle, C.M. Geostatistical modeling of the spatial distribution of sediment oxygen demand within a Coastal Plain blackwater watershed. Geoderma 2010, 159, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Tsaboula, A.; Menexes, G.; Papadakis, E.-N.; Vryzas, Z.; Kotopoulou, A.; Kintzikoglou, K.; Papadopoulou-Mourkidou, E. Assessment and management of pesticide pollution at a river basin level part II: Optimization of pesticide monitoring networks on surface aquatic ecosystems by data analysis methods. Sci. Total Environ. 2019, 653, 1612–1622. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Breitenbeck, G.A.; Faulkner, S.P. Denitrification and N2O emission from forested and cultivated alluvial clay soil. Biogeochemistry 2005, 73, 499–513. [Google Scholar] [CrossRef]
- Keating, M.P.; Ochs, C.A.; Balachandran, D.; Holland, M.M.; Lizotte, R.E.; Yu, K. Spatial variation related to hydrologic patterns and vegetation in greenhouse gas fluxes from the Mississippi Delta agricultural region. Appl. Soil Ecol. 2016, 98, 278–281. [Google Scholar] [CrossRef]
- Ullah, S.; Faulkner, S.P. Denitrification potential of different land-use types in an agricultural watershed, lower Mississippi valley. Ecol. Eng. 2006, 28, 131–140. [Google Scholar] [CrossRef]
- Utley, B.C.; Vellidis, G.; Lowrance, R.; Smith, M.C. Factors affecting sediment oxygen demand dynamics in blackwater streams of Georgia’s coastal plain. J. Am. Water Resour. Assoc. 2008, 44, 742–753. [Google Scholar] [CrossRef]
- Wainger, L.; Loomis, J.; Johnston, R.; Hansen, L.; Carlisle, D.; Lawrence, D.; Gollehon, N.; Duriancik, L.; Schwartz, G.; Ribaudo, M.; et al. Ecosystem service benefits generated by improved water quality from conservation practices. In The Valuation of Ecosystem Services from Farms and Forests: Informing a Systematic Approach to Quantifying Benefits of Conservation Programs; The Council on Food, Agricultural and Resource Economics (C-FARE) Report No. 0114-301; Wainger, L., Ervin, D., Eds.; C-FARE: Washington, DC, USA, 2017; pp. 2–57. [Google Scholar]
- Weaver, M.A.; Zablotowicz, R.M.; Krutz, L.J.; Bryson, C.T.; Locke, M.A. Microbial and vegetative changes associated with development of a constructed wetland. Ecol. Indic. 2012, 13, 37–45. [Google Scholar] [CrossRef]
- Whittaker, G.; Barnhart, B.L.; Srinivasan, R.; Arnold, J.G. Cost of areal reduction of gulf hypoxia through agricultural practice. Sci. Total Environ. 2015, 505, 149–153. [Google Scholar] [CrossRef]
- Withers, P.J.A.; Neal, C.; Jarvie, H.P.; Doody, D.G. Agriculture and eutrophication: Where do we go from here? Sustainability 2014, 6, 5853–5875. [Google Scholar] [CrossRef]
- Wronski, T.; Dusabe, M.C.; Apio, A.; Hausdorf, B.; Albrecht, C. Biological assessment of water quality and biodiversity in Rwandan rivers draining into Lake Kivu. Aquat. Ecol. 2015, 49, 309–320. [Google Scholar] [CrossRef]
- Zablotowicz, R.M.; Locke, M.A.; Hoagland, R.E.; Knight, S.S.; Cash, B. Fluorescent Pseudomonas Isolates from Mississippi Delta Oxbow Lakes: In Vitro Herbicide Biotransformations. Environ. Toxicol. 2001, 16, 9–19. [Google Scholar] [CrossRef]
- Zablotowicz, R.M.; Zimba, P.V.; Locke, M.A.; Knight, S.S.; Lizotte, R.E.; Gordon, R.E. Effects of land management practices on water quality in Mississippi Delta oxbow lakes: Biochemical and microbiological aspects. Agric. Ecosyst. Environ. 2010, 139, 214–223. [Google Scholar] [CrossRef]
- Paerl, H.W.; Dyble, J.; Moisander, P.H.; Noble, R.T.; Piehler, M.F.; Pinckney, J.L.; Steppe, T.F.; Twomey, L.; Valdes, L.M. Microbial indicators of aquatic ecosystem change: Current applications to eutrophication studies. FEMS Microbiol. Ecol. 2003, 46, 233–246. [Google Scholar] [CrossRef]
- Zedler, J.B. Wetlands at your service: Reducing impacts of agriculture at the watershed scale. Front. Ecol. Environ. 2003, 1, 65–72. [Google Scholar] [CrossRef]
- Benbow, M.E.; Pechal, J.L.; Ward, A.K. Heterotrophic bacteria production and microbial community assessment. In Methods in Stream Ecology Volume 1: Ecosystem Structure, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 1, pp. 161–176. [Google Scholar]
- Lowe, R.L.; LaLiberte, G.D. Benthic stream algae: Distribution and structure. In Methods in Stream Ecology Volume 1: Ecosystem Structure, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 1, pp. 193–222. [Google Scholar]
- Hauer, F.R.; Resh, V.H. Macroinvertebrates. In Methods in Stream Ecology Volume 1: Ecosystem Structure, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 1, pp. 297–320. [Google Scholar]
- McGarvey, D.J.; Falke, J.A.; Li, H.W.; Li, J.L. Fish assemblages. In Methods in Stream Ecology Volume 1: Ecosystem Structure, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 1, pp. 321–354. [Google Scholar]
- Carter, J.L.; Resh, V.H.; Hannaford, M.J. Macroinvertebrates as biotic indicators of environmental quality. In Methods in Stream Ecology Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 2, pp. 293–318. [Google Scholar]
- Simon, T.P.; Evans, N.T. Environmental quality assessment using stream fishes. In Methods in Stream Ecology Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 2, pp. 319–334. [Google Scholar]
- Sanders, K.E.; Smiley, P.C.; Gillespie, R.B.; King, K.W.; Smith, D.R.; Pappas, E.A. Conservation implications of fish-habitat relationships in channelized agricultural headwater streams. J. Environ. Qual. 2020, 49, 1585–1598. [Google Scholar] [CrossRef] [PubMed]
- Smiley, P.C.; Gillespie, R.B.; King, K.W.; Huang, C. Management implications of the relationships between water chemistry and fishes within channelized headwater streams in the Midwestern United States. Ecohydrology 2009, 2, 294–302. [Google Scholar] [CrossRef]
- Smiley, P.C.; King, K.W.; Fausey, N.R. Influence of different habitat factors on creek chub (Semotilus atromaculatus) within channelized agricultural headwater streams. Northeast. Nat. 2017, 24, 18–44. [Google Scholar] [CrossRef]
- Troy, J.L. Importance of Total Suspended Solids in Explaining Fish Community Structure in Agricultural Headwater Streams. Master’s Thesis, Purdue University Fort Wayne, Fort Wayne, IN, USA, August 2019. [Google Scholar]
- Lizotte, R.; Knight, S.; Locke, M.; Bingner, R. Influence of integrated watershed-scale agricultural conservation practices on lake water quality. J. Soil Water Conserv. 2014, 69, 160–170. [Google Scholar] [CrossRef]
- Smiley, P.C.; King, K.W.; Fausey, N.R. Annual and seasonal differences in pesticide mixtures within channelized agricultural headwater streams in central Ohio. Agric. Ecosyst. Environ. 2014, 193, 83–95. [Google Scholar] [CrossRef]
- Findlay, S.E.G.; Parr, T.B. Dissolved organic matter. In Methods in Stream Ecology Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 2, pp. 21–36. [Google Scholar]
- Dodds, W.K.; Burgin, A.M.; Marcarelli, A.M.; Strauss, E.A. Nitrogen transformations. In Methods in Stream Ecology Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 2, pp. 173–196. [Google Scholar]
- Hall, R.O.; Hotchkiss, E.R. Stream metabolism. In Methods in Stream Ecology Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 2, pp. 219–234. [Google Scholar]
- Culp, J.M.; Yates, A.G.; Armanini, D.G.; Baird, D.J. Establishing cause-effect relationships in multistressor environments. In Methods in Stream Ecology Volume 2: Ecosystem Function, 3rd ed.; Lamberti, G.A., Hauer, F.R., Eds.; Academic Press: San Diego, CA, USA, 2017; Volume 2, pp. 335–352. [Google Scholar]
- Locke, M.A.; Knight, S.S.; Smith, S.; Cullum, R.F.; Zablotowicz, R.M.; Yuan, Y.; Bingner, R.L. Environmental quality research in the Beasley Lake watershed, 1995 to 2007: Succession from conventional to conservation practices. J. Soil Water Conserv. 2008, 63, 430–442. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lizotte, R.E.; Smiley, P.C.; Gillespie, R.B.; Knight, S.S. Agricultural Conservation Practices and Aquatic Ecological Responses. Water 2021, 13, 1687. https://doi.org/10.3390/w13121687
Lizotte RE, Smiley PC, Gillespie RB, Knight SS. Agricultural Conservation Practices and Aquatic Ecological Responses. Water. 2021; 13(12):1687. https://doi.org/10.3390/w13121687
Chicago/Turabian StyleLizotte, Richard E., Peter C. Smiley, Robert B. Gillespie, and Scott S. Knight. 2021. "Agricultural Conservation Practices and Aquatic Ecological Responses" Water 13, no. 12: 1687. https://doi.org/10.3390/w13121687
APA StyleLizotte, R. E., Smiley, P. C., Gillespie, R. B., & Knight, S. S. (2021). Agricultural Conservation Practices and Aquatic Ecological Responses. Water, 13(12), 1687. https://doi.org/10.3390/w13121687






