TiO2-Powdered Activated Carbon (TiO2/PAC) for Removal and Photocatalytic Properties of 2-Methylisoborneol (2-MIB) in Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sample Collection and Preparation
2.3. Synthesis of TiO2/PAC
2.4. 2-MIB Quantification
2.5. Experimental Studies for 2-MIB Removal
2.5.1. Batch Adsorption Experiments
2.5.2. Photodegradation Experiments
2.6. Characterization of the Adsorbent
2.7. Modeling for Competing NOM Analysis
3. Results and Discussion
3.1. Characterization of Adsorbent
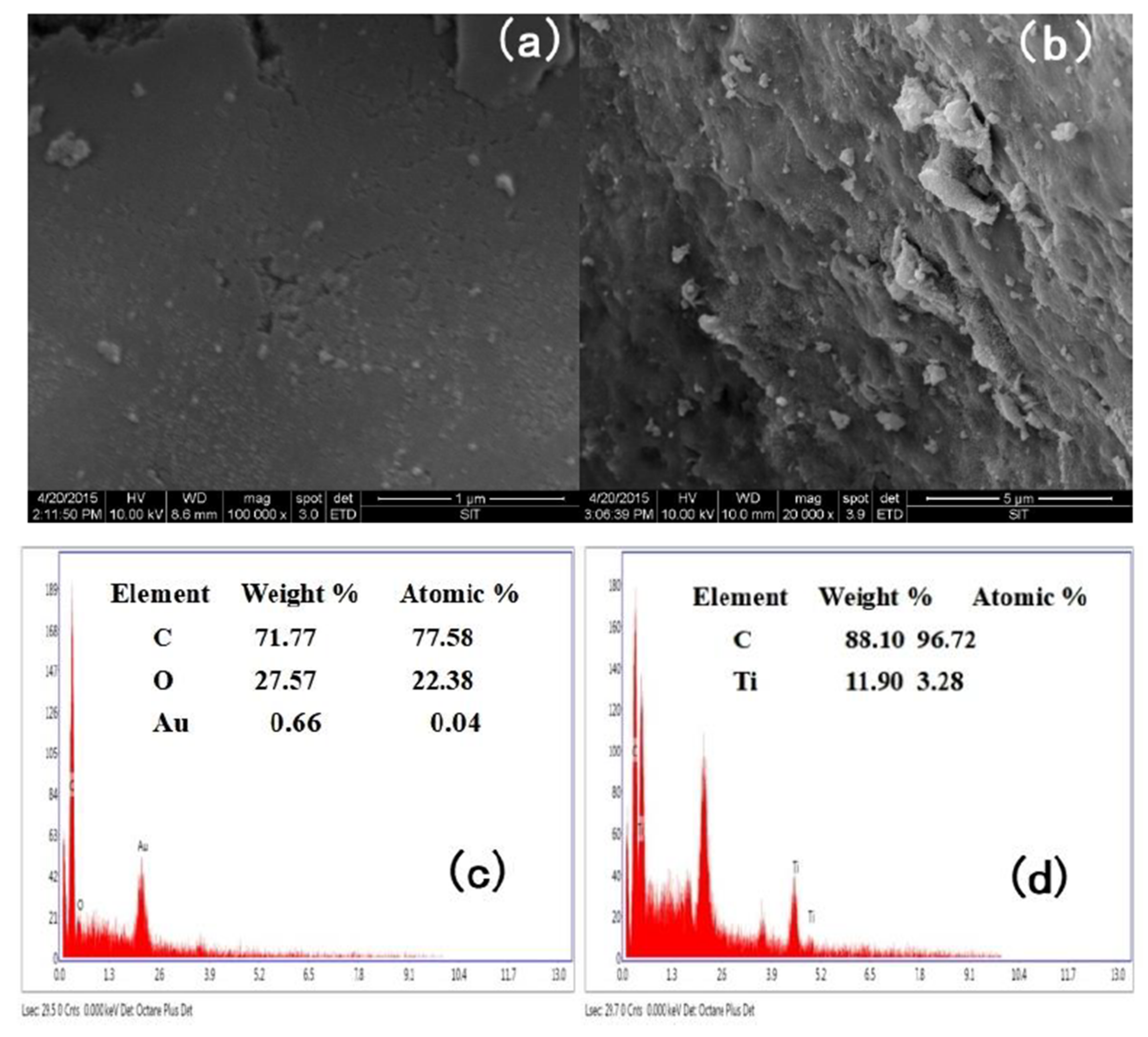
3.1.1. SEM Images
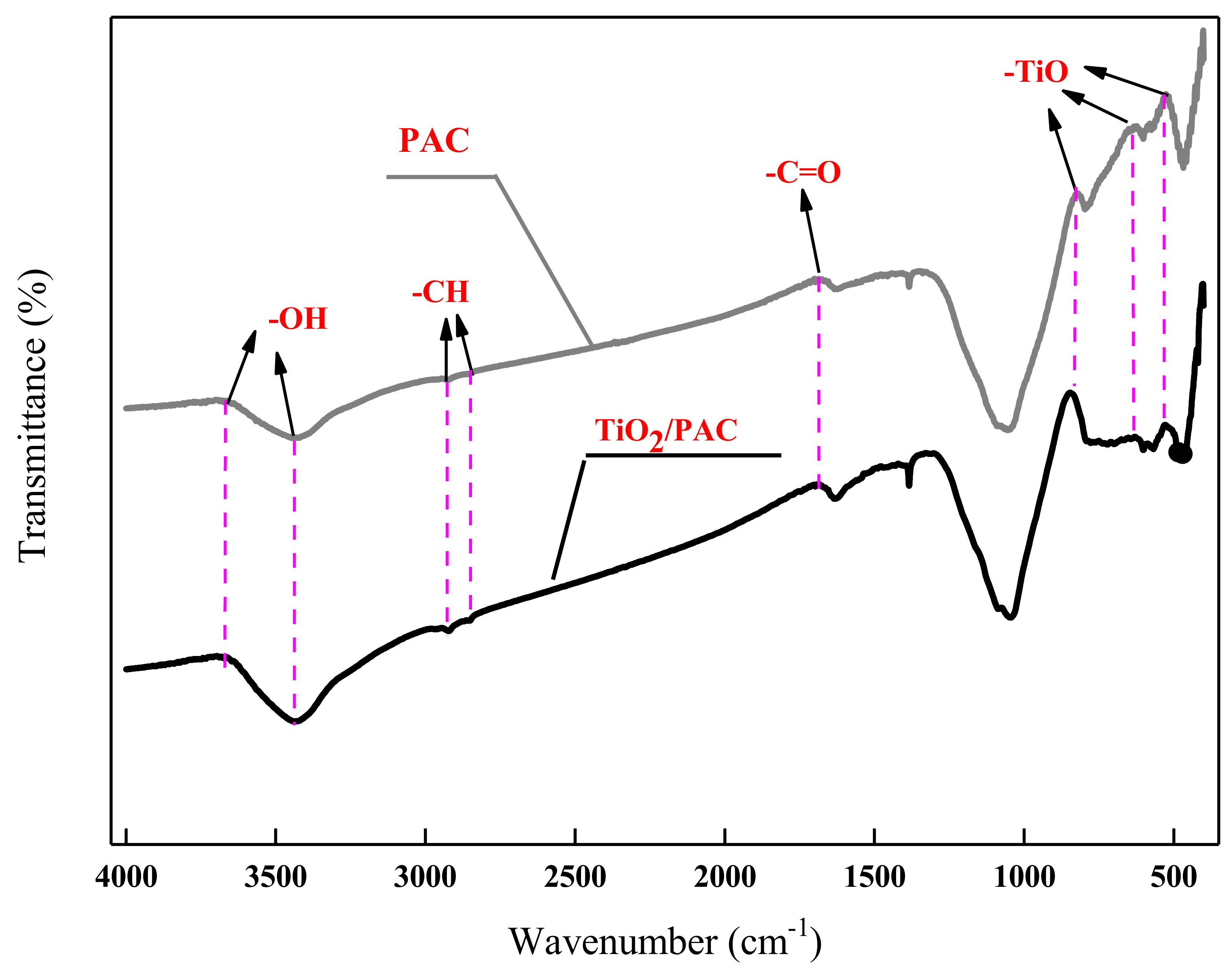
3.1.2. FTIR Analysis
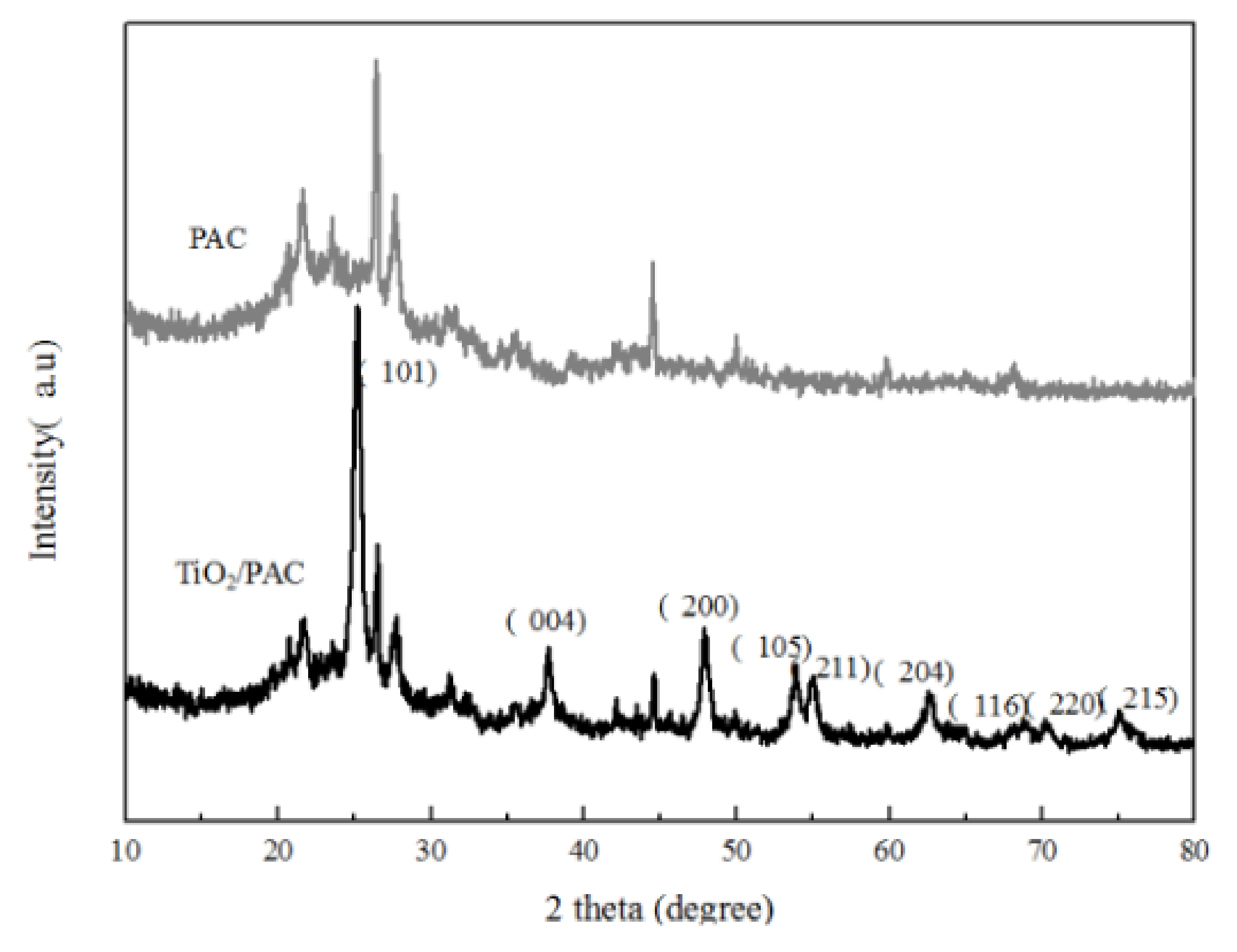
3.1.3. XRD Analysis
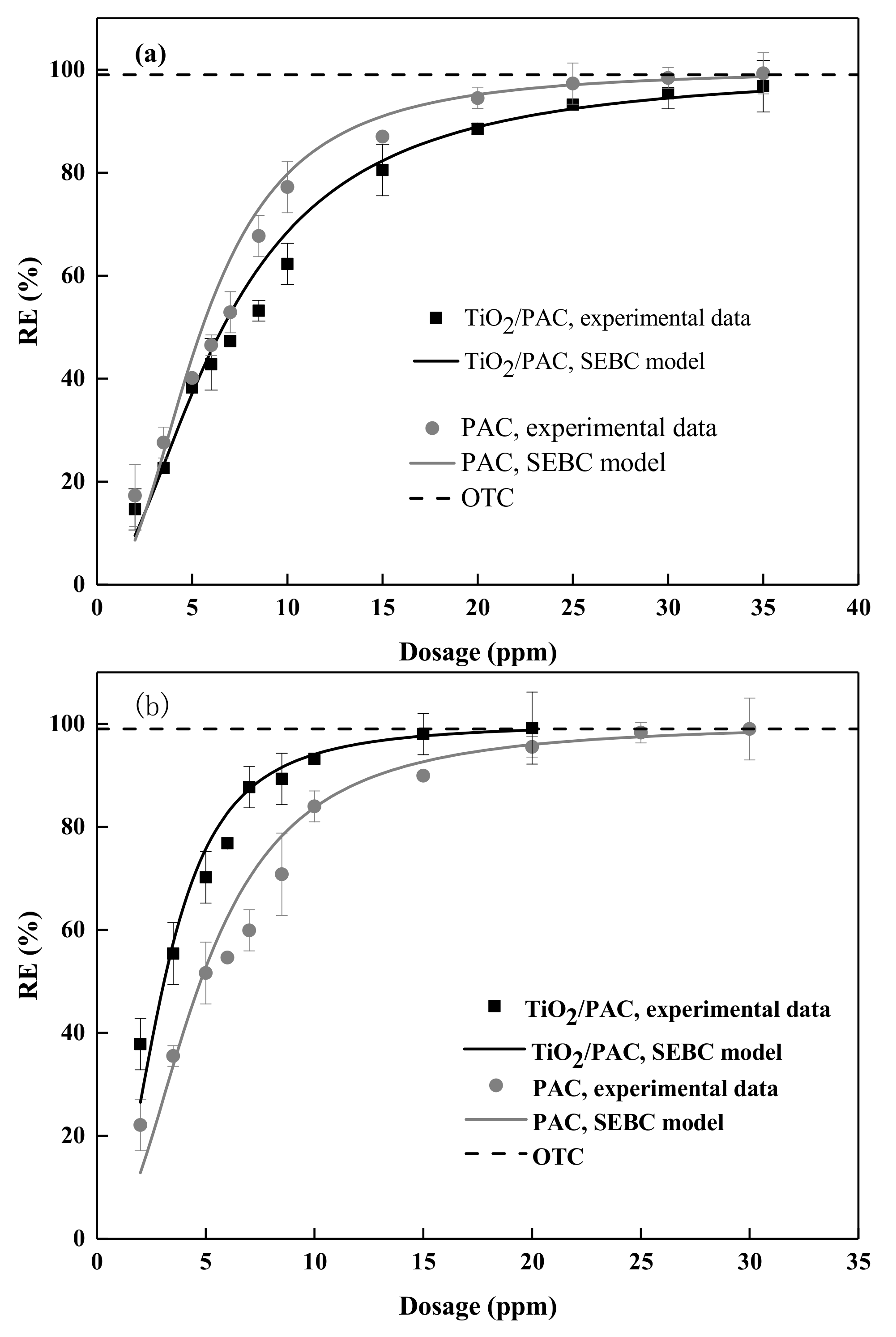
3.2. Adsorption Experiments
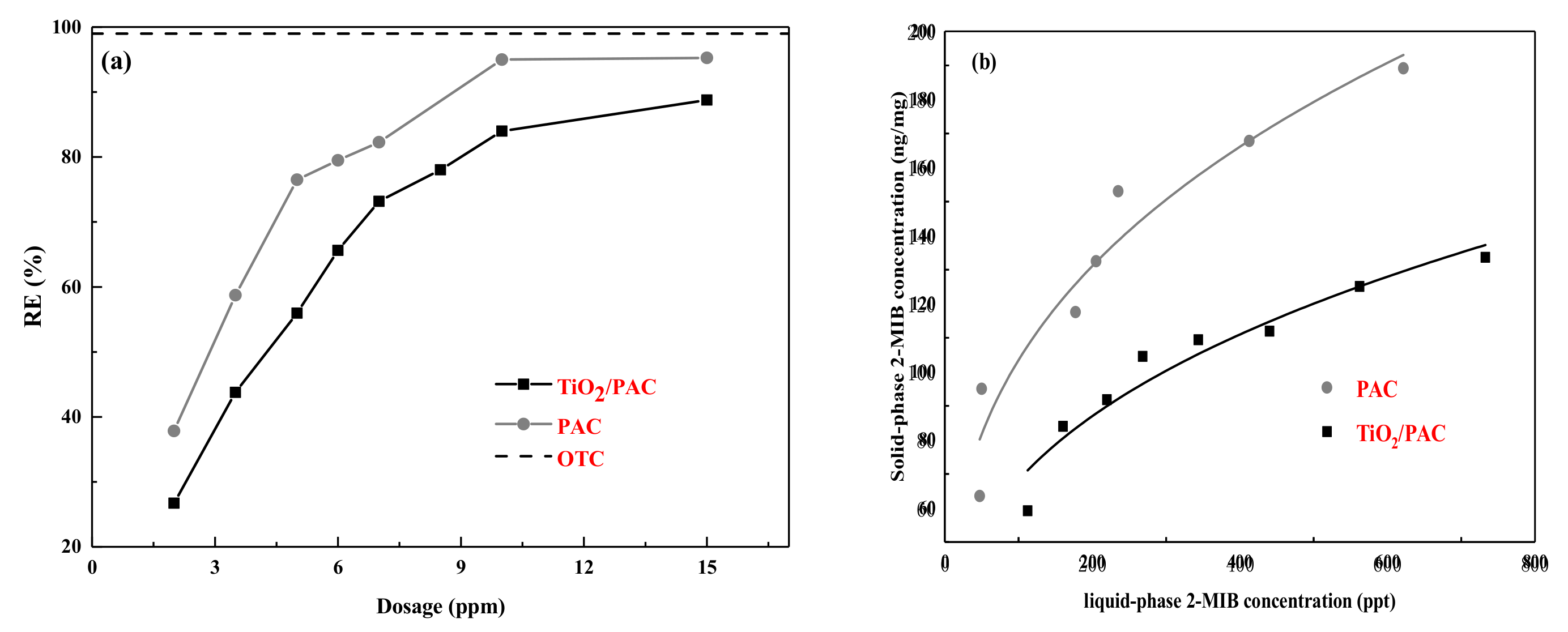
3.2.1. Adsorption Isotherm
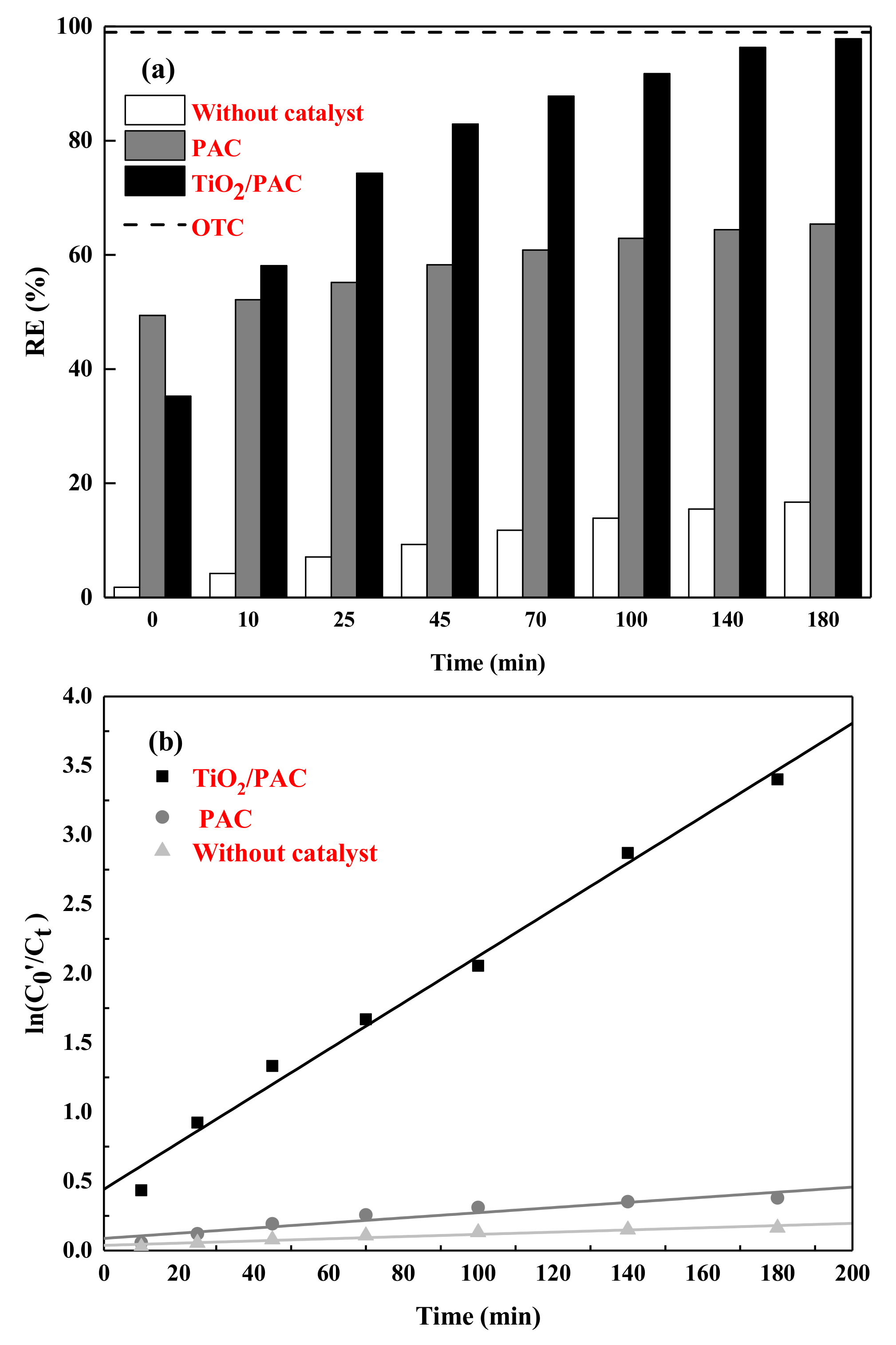
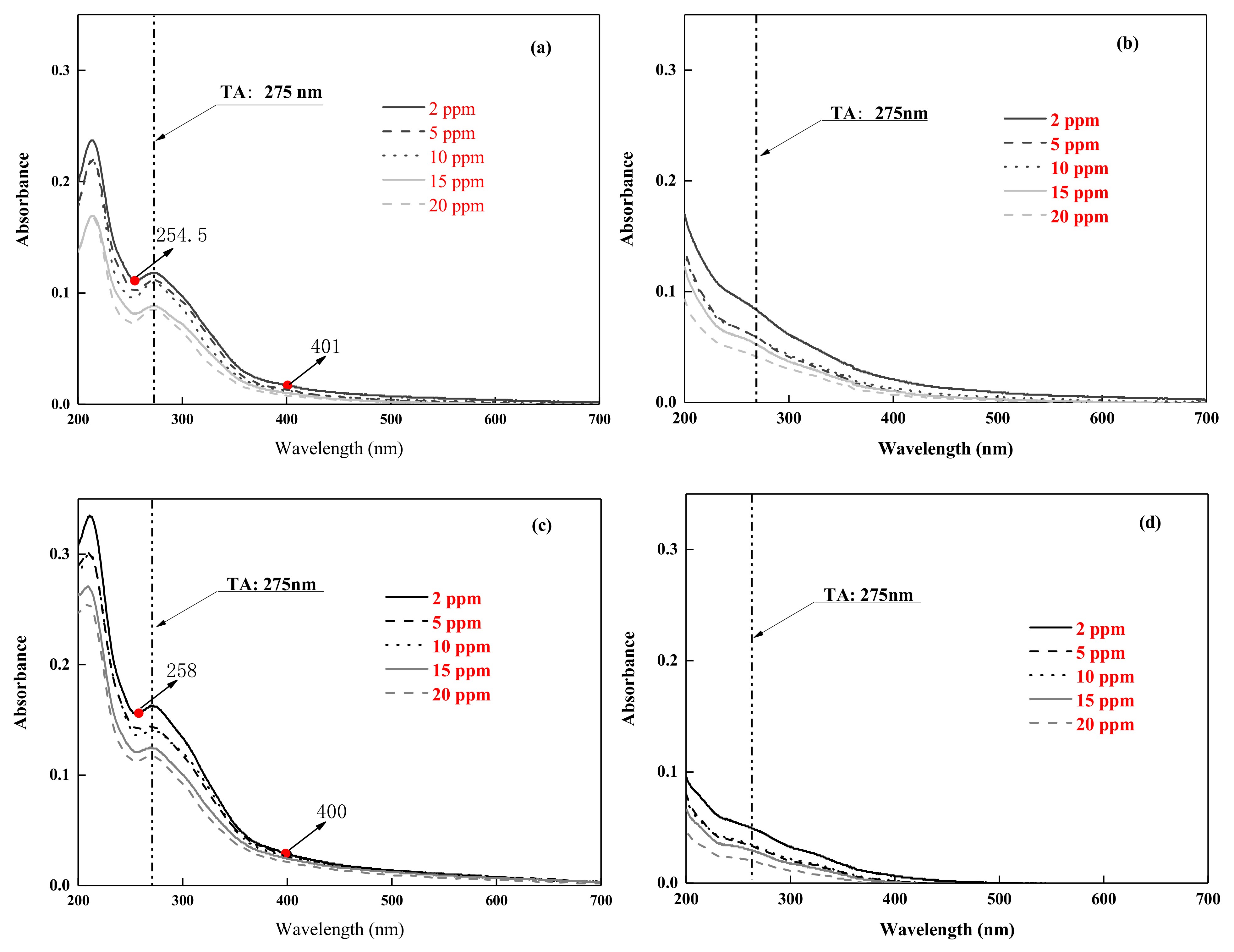
3.2.2. Photodegradation Performance
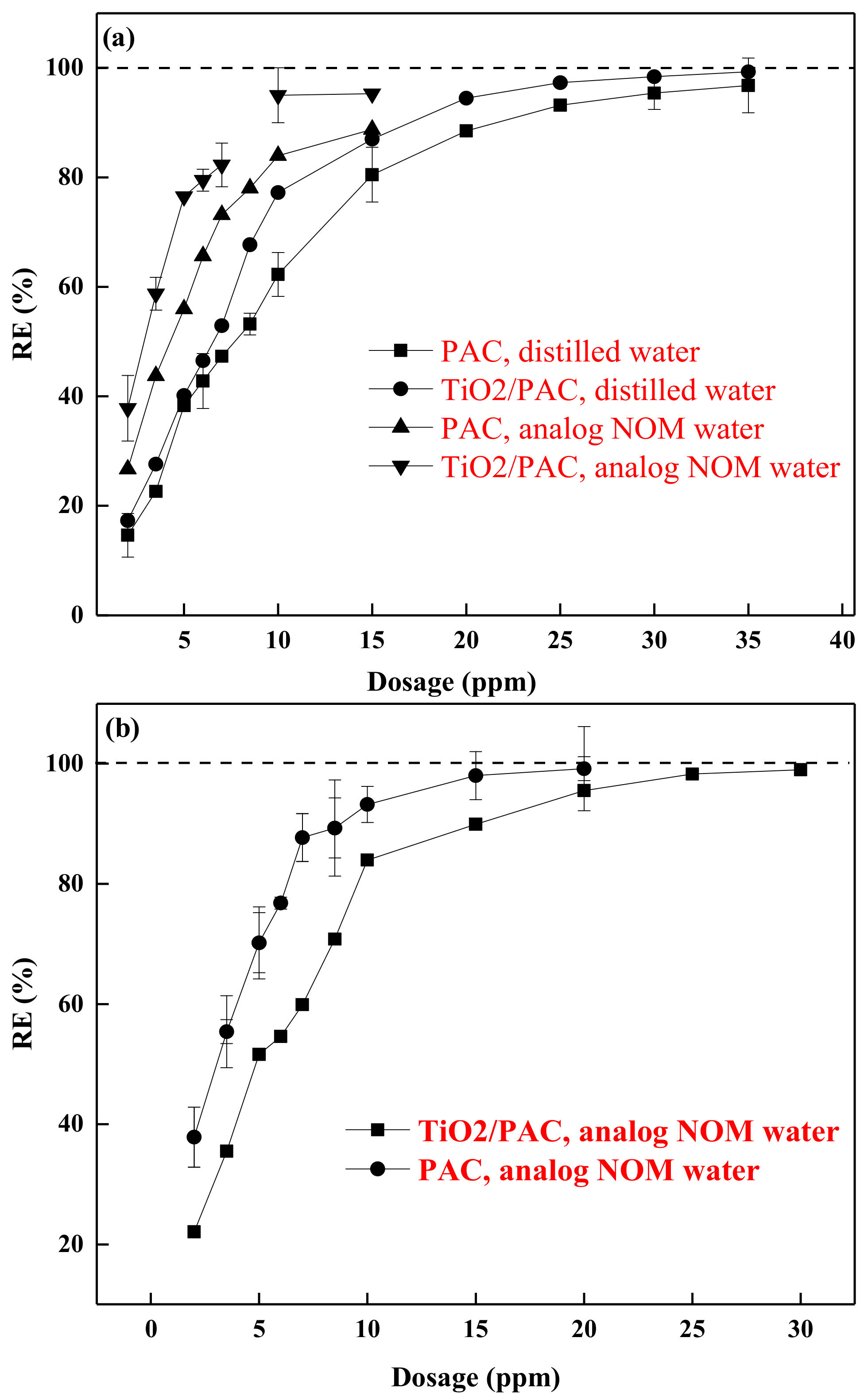
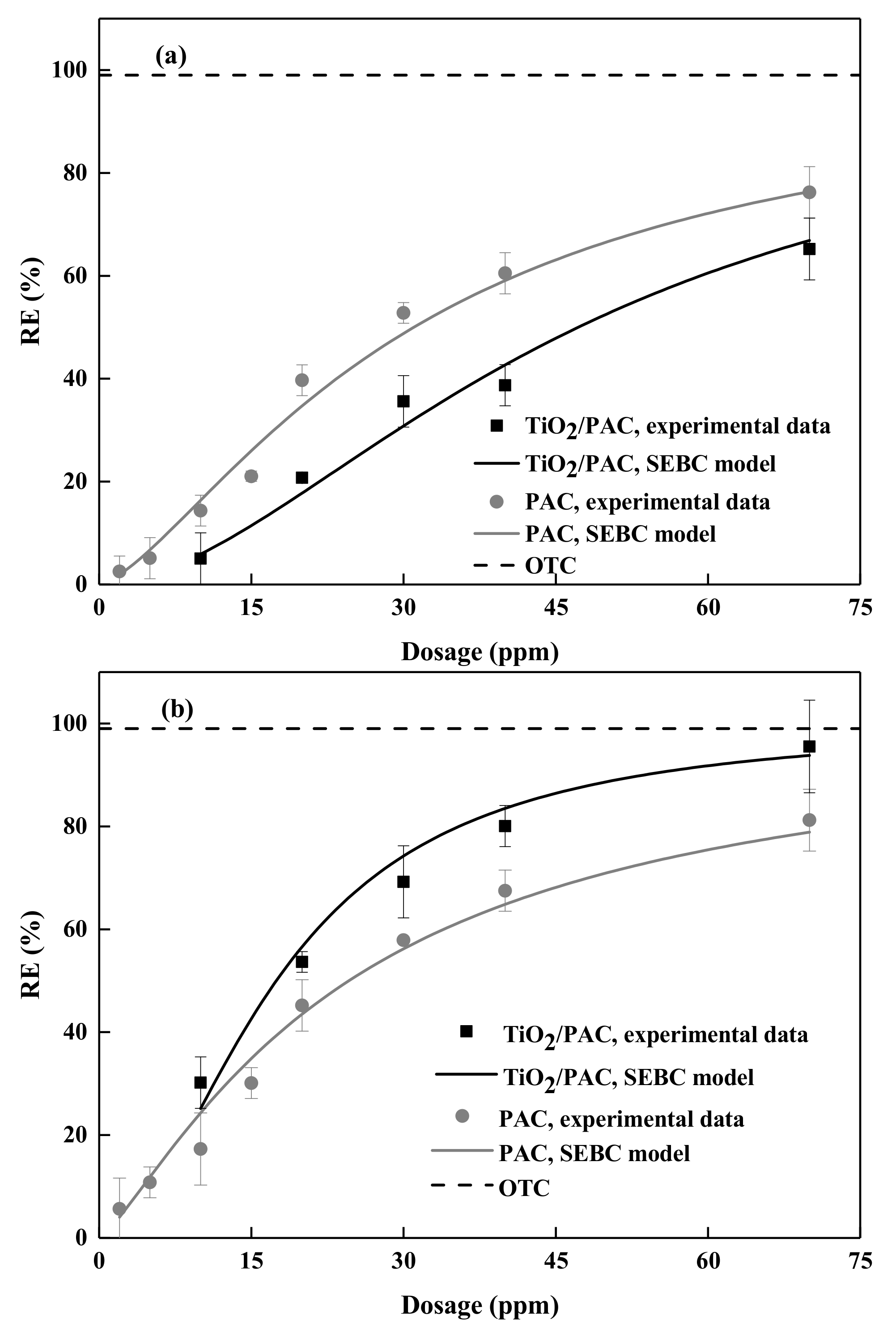
3.3. Influence of NOM on Photodegradation Performance
3.4. Competitive Adsorption
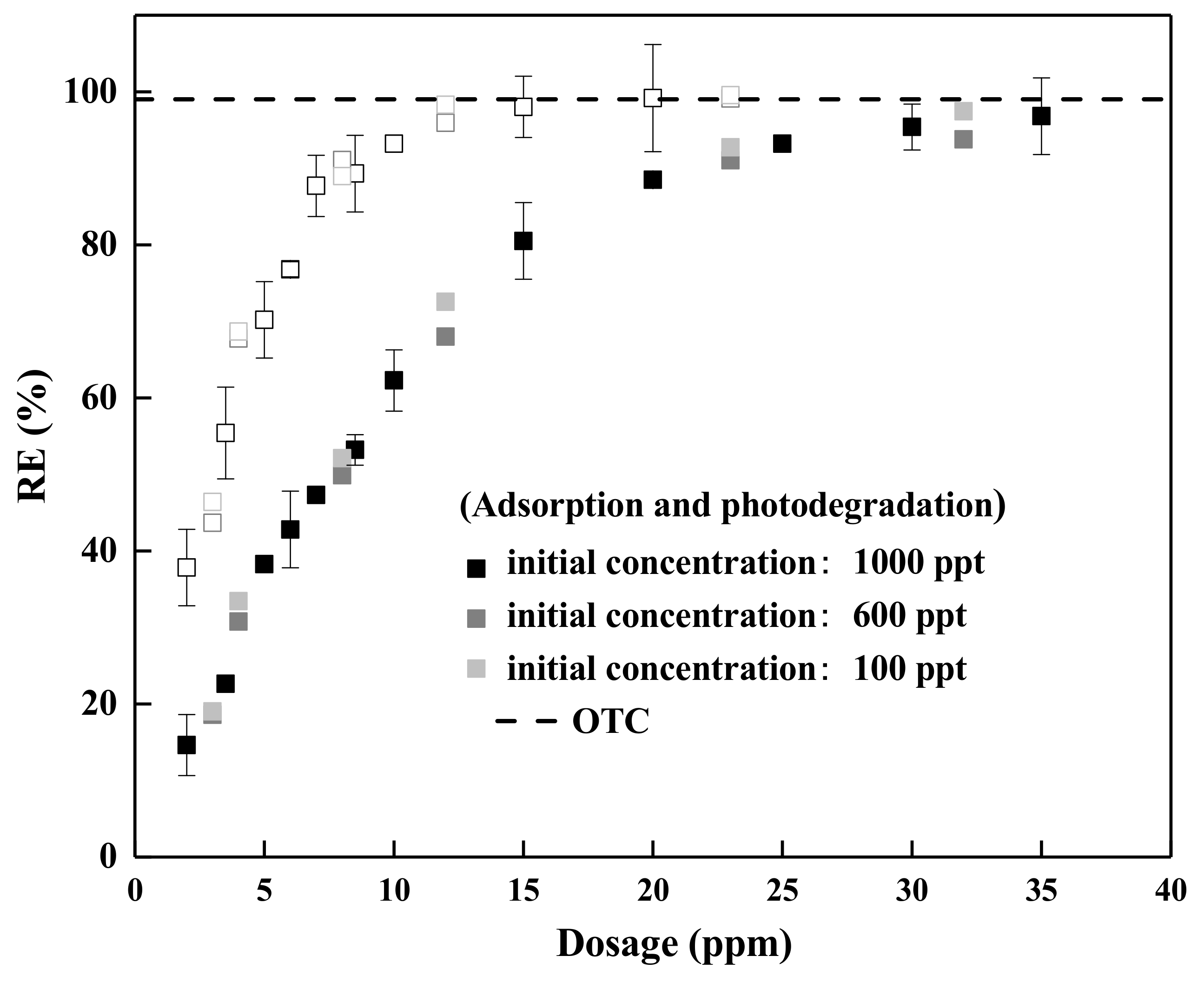
3.5. Adsorption Performance in Realistic Water Matrices
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| qi | the loading of the adsorbate surface of a single substance; (ng/mg) |
| qj | the total surface loading of the adsorbent; (ng/mg) |
| Ci | the concentration of a single adsorbate in the system; (ng/L) |
| ki, n | a Freundlich isotherm equation constant and exponent ((ng/mg)/(ng/L)1/n) of component i |
| mA | adsorbent dosage; (mg/L) |
| CT,0 | initial concentration of tracer; (ng/L) |
| CE,0 | initial concentration of EBC; (ng/L) |
| qT | solid equilibrium concentration of the tracer; (ng/mg) |
| qE | solid equilibrium concentration of EBC; (ng/mg) |
| V | the volume of the solution |
| m | amount of the PAC or TiO2/PAC used (mg) |
| PAR0 and PARt | initial peak area ratio and post- adsorption peak area ratio at time t of the solution |
| C0, Ct, C0 | the initial concentration (ng/L), post-adsorption concentration after selected time interval (ng/L), equilibrium concentration of 2-MIB after adsorption in the dark (ng/L) |
| Ce | equilibrium concentration (ng/L); |
| qe | equilibrium adsorption amount (ng/mg) |
| K, kapp | Freundlich constant (ng/mg)/(ng/L)1/n, apparent first-order reaction (min-1) constant. |
| n | the related parameters between the adsorbent molecules and the adsorbent surface strength |
| R2 | the correlation coefficient |
| R | the removal efficiency |
References
- Zaitlin, B.; Watson, S.B. Actinomycetes in relation to taste and odour in drinking water: Myths, tenets and truths. Water. Res. 2006, 40, 1741–1753. [Google Scholar] [CrossRef]
- Wang, C.M.; Yu, J.W.; Guo, Q.Y.; Sun, D.L.; Su, M.; An, W.; Zhang, Y.; Yang, M. Occurrence of swampy/septic odor and possible odorants in source and finished drinking water of major cities across China. Environ. Pollut. 2019, 249, 305–310. [Google Scholar] [CrossRef]
- Matsui, Y.; Yoshida, T.; Nakao, S.; Knappe, D.R.U.; Matsushita, T. Characteristics of competitive adsorption between 2-methylisoborneol and natural organic matter on superfine and conventionally sized powdered activated carbons. Water. Res. 2012, 46, 4741–4749. [Google Scholar] [CrossRef] [PubMed]
- Devi, A.; Chiu, Y.T.; Hsueh, H.T.; Lin, T.F. Quantitative PCR based detection system for cyanobacterial geosmin/2-methylisoborneol (2-MIB) events in drinking water sources: Current status and challenges. Water. Res. 2021, 188, 116478. [Google Scholar] [CrossRef]
- Zoschke, K.; Engel, C.; Börnick, H.; Worch, E. Adsorption of geosmin and 2-methylisoborneol onto powdered activated carbon at non-equilibrium conditions: Influence of NOM and process modeling. Water Res. 2011, 45, 4544–4550. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Ma, J.; Liu, W.; Zou, J.; Yue, S.; Li, X.; Wiesner, M.R.; Fang, J. Removal of 2-MIB and geosmin using UV/persulfate: Contributions of hydroxyl and sulfate radicals. Water Res. 2015, 69, 223–233. [Google Scholar] [CrossRef]
- Ho, L.; Hoefel, D.; Bock, F.; Saint, C.P.; Newcombe, G. Biodegradation rates of 2-methylisoborneol (MIB) and geosmin through sand filters and in bioreactors. Chemosphere 2007, 66, 2210–2218. [Google Scholar] [CrossRef]
- Tennant, M.F.; Mazyck, D.W. The role of surface acidity and pore size distribution in the adsorption of 2-methylisoborneol via powdered activated carbon. Carbon 2007, 45, 858–864. [Google Scholar] [CrossRef]
- Sidney Seckler, F.F.; Margarida, M.; Rosemeire, A.L. Interference of iron as a coagulant on MIB removal by powdered activated carbon adsorption for low turbidity waters. J. Environ. Sci. 2013, 25, 1575–1582. [Google Scholar] [CrossRef]
- Rangel-Mendez, J.R.; Cannon, F.S. Improved activated carbon by thermal treatment in methane and steam: Physicochemical influences on MIB sorption capacity. Carbon 2005, 43, 467–479. [Google Scholar] [CrossRef]
- Meunier, L.; Canonica, S.; von Gunten, U. Implications of sequential use of UV and ozone for drinking water quality. Water Res. 2006, 40, 1864–1876. [Google Scholar] [CrossRef]
- Yu, J.; Yang, M.; Lin, T.F.; Guo, Z.; Zhang, Y.; Gu, J.; Zhang, S. Effects of surface characteristics of activated carbon on the adsorption of 2-methylisobornel (MIB) and geosmin from natural water. Sep. Purif. Technol. 2007, 56, 363–370. [Google Scholar] [CrossRef]
- Drikas, M.; Dixon, M.; Morran, J. Removal of MIB and geosmin using granular activated carbon with and without MIEX pre-treatment. Water Res. 2009, 43, 5151–5159. [Google Scholar] [CrossRef]
- Summers, R.S.; Kim, S.M.; Shimabuku, K.; Chae, S.H.; Corwin, C.J. Granular activated carbon adsorption of MIB in the presence of dissolved organic matter. Water Res. 2013, 47, 3507–3513. [Google Scholar] [CrossRef]
- Kim, C.; Lee, S.I.; Hwang, S.; Cho, M.; Kim, H.S.; Noh, S.H. Removal of geosmin and 2-methylisoboneol (2-MIB) by membrane system combined with powdered activated carbon (PAC) for drinking water treatment. J. Water Process Eng. 2014, 4, 91–98. [Google Scholar] [CrossRef]
- Azaria, S.; Nir, S.; van Rijn, J. Combined adsorption and degradation of the off-flavor compound 2-methylisoborneol in sludge derived from a recirculating aquaculture system. Chemosphere 2017, 169, 69–77. [Google Scholar] [CrossRef]
- Wu, J.Y.; Jiang, R.F.; Liu, Q.L.; Ouyang, G.F. Impact of different modes of adsorption of natural organic matter on the environmental fate of nanoplastics. Chemosphere 2021, 263, 127967. [Google Scholar] [CrossRef]
- Park, J.A.; Nam, H.L.; Choi, J.W.; Ha, J.; Lee, S.H. Oxidation of geosmin and 2-methylisoborneol by the photo-Fenton process: Kinetics, degradation intermediates, and the removal of microcystin-LR and trihalomethane from Nak-Dong River water, South Korea. Chem. Eng. J. 2017, 313, 345–354. [Google Scholar] [CrossRef]
- Lindholm-Lehto, P.C.; Vielma, J. Controlling of geosmin and 2-methylisoborneol induced off-flavours in recirculating aquaculture system farmed fish—A review. Aquac. Res. 2019, 50, 9–28. [Google Scholar] [CrossRef]
- Cordero, T.; Duchamp, C.; Chovelon, J.M.; Ferronato, C.; Matos, J. Influence of L-type activated carbons on photocatalytic activity of TiO2 in 4-chlorophenol photodegradation. J. Photoch. Photobio. A 2007, 191, 122–131. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Y.; Hu, Z.; Chen, Y.; Liu, W.; Zhao, G. Degradation of methyl orange by composite photocatalysts nano-TiO2 immobilized on activated carbons of different porosities. J. Hazard. Mater. 2009, 169, 1061–1067. [Google Scholar] [CrossRef]
- In, S.I.; Vesborg, P.C.K.; Abrams, B.L.; Hou, Y.D.; Chorkendorff, I. A comparative study of two techniques for determining photocatalytic activity of nitrogen doped TiO2 nanotubes under visible light irradiation: Photocatalytic reduction of dye and photocatalytic oxidation of organic molecules. J. Photochem. Photobiol. A Chem. 2011, 222, 258–262. [Google Scholar] [CrossRef]
- Ocampo-Pérez, R.; Sánchez-Polo, M.; Rivera-Utrilla, J.; Leyva-Ramos, R. Enhancement of the catalytic activity of TiO2 by using activated carbon in the photocatalytic degradation of cytarabine. Appl. Catal. B. Environ. 2011, 104, 177–184. [Google Scholar] [CrossRef]
- Souza, I.P.A.; Crespo, L.H.S.; Spessato, L.; Melo, S.A.R.; Martins, A.F.; Cazetta, A.L.; Almeida, V.C. Optimization of thermal conditions of sol-gel method for synthesis of TiO2 using RSM and its influence on photodegradation of tartrazine yellow dye. J. Environ. Chem. Eng. 2021, 9, 104753. [Google Scholar] [CrossRef]
- He, X.; Liu, Q.; Li, Q.; Tang, K.; Gao, N.; Ma, X. 2-methylisoborneol removal by activated carbon fiber, chlorine dioxide and their combined process. J. Cent. South Univ. (Sci. Technol.) 2014, 45, 973–978. [Google Scholar]
- Nakayama, A.; Sakamoto, A.; Matsushita, T.; Matsui, Y.; Shirasaki, N. Effects of pre, post, and simultaneous loading of natural organic matter on 2-methylisoborneol adsorption on superfine powdered activated carbon: Reversibility and external pore-blocking. Water Res. 2020, 182, 115992. [Google Scholar] [CrossRef] [PubMed]
- Truong, H.B.; Ike, I.A.; Ok, Y.S.; Hur, J. Polyethyleneimine modification of activated fly ash and biochar for enhanced removal of natural organic matter from water via adsorption. Chemosphere 2020, 243, 125454. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zietzschmann, F.; Yu, J.W.; Hofman, R.; An, W.; Yang, M.; Rietveld, L.C. Projecting competition between 2-methylisoborneol and natural organic matter in adsorption onto activated carbon from ozonated source waters. Water Res. 2020, 173, 115574. [Google Scholar] [CrossRef]
- Newcombe, G.; Morrison, J.; Hepplewhite, C. Simultaneous adsorption of MIB and NOM onto activated carbon. I. Characterisation of the system and NOM adsorption. Carbon 2002, 40, 2135–2146. [Google Scholar] [CrossRef]
- Matsui, Y.; Nakao, S.; Taniguchi, T.; Matsushita, T. Geosmin and 2-methylisoborneol removal using superfine powdered activated carbon: Shell adsorption and branched-pore kinetic model analysis and optimal particle size. Water Res. 2013, 47, 2873–2880. [Google Scholar] [CrossRef] [PubMed]
- Knappe, D.R.U.; Matsui, Y.; Snoeyink, V.L. Predicting the capacity of powdered activated carbon for trace organic compounds in natural waters. Environ. Sci. Technol. 1998, 32, 1694–1698. [Google Scholar] [CrossRef]
- Qi, S.; Schideman, L.; Mariñas, B.J.; Snoeyink, V.L.; Campos, C. Simplification of the IAST for activated carbon adsorption of trace organic compounds from natural water. Water Res. 2007, 41, 440–448. [Google Scholar] [CrossRef]
- Worch, E. Competitive adsorption of micropollutants and NOM onto activated carbon: Comparison of different model approaches. J. Water Supply Res. Technol. 2010, 59, 285–297. [Google Scholar] [CrossRef]
- Mishra, A.; Mehta, A.; Kainth, S.; Basu, S. Effect of g-C3N4 loading on TiO2/Bentonite nanocomposites for efficient heterogeneous photocatalytic degradation of industrial dye under visible light. J. Alloy Compd. 2018, 764, 406–415. [Google Scholar] [CrossRef]
- Chandrabose, G.; Dey, A.; Gaur, S.S.; Pitchaimuthu, S.; Jagadeesan, H.; Braithwaite, N.S.J.; Selvaraj, V.; Kumar, V.; Krishnamurthy, S. Removal and degradation of mixed dye pollutants by integrated adsorption-photocatalysis technique using 2-D MoS2/TiO2 nanocomposite. Chemosphere 2021, 279, 130467. [Google Scholar] [CrossRef] [PubMed]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. B. 1906, 57, 384–471. [Google Scholar]
- Bi, L.B.; Chen, Z.L.; Li, L.H.; Kang, J.; Zhao, S.X.; Wang, B.Y.; Yan, P.W.; Li, Y.B.; Zhang, X.X.; Shen, J.M. Selective adsorption and enhanced photodegradation of diclofenac in water by molecularly imprinted TiO2. J. Hazard. Mater. 2021, 407, 124759. [Google Scholar] [CrossRef]
- Mahmood, A.; Shi, G.S.; Wang, Z.; Rao, Z.P.; Xiao, W.; Xie, X.F.; Sun, J. Carbon quantum dots-TiO2 nanocomposite as an efficient photocatalyst for the photodegradation of aromatic ring-containing mixed VOCs: An experimental and DFT studies of adsorption and electronic structure of the interface. J. Hazard. Mater. 2021, 401, 123402. [Google Scholar] [CrossRef] [PubMed]
- Omri, A.; Lambert, S.D.; Geens, J.; Bennour, F.; Benzina, M. Synthesis, surface characterization and photocatalytic activity of TiO2 supported on almond shell activated carbon. J. Mater. Sci. Technol. 2014, 30, 894–902. [Google Scholar] [CrossRef]
- Ragupathy, S.; Raghu, K.; Prabu, P. Synthesis and characterization of TiO2 loaded cashew nut shell activated carbon and photocatalytic activity on BG and MB dyes under sunlight radiation. Spectrochim. Acta A 2015, 138, 314–320. [Google Scholar] [CrossRef]
- Ho, L.; Newcombe, G. Effect of NOM, turbidity and floc size on the PAC adsorption of MIB during alum coagulation. Water Res. 2005, 39, 3668–3674. [Google Scholar] [CrossRef] [PubMed]
| Chromaticity | DO | Conductivity | NH3-N | COD | pH |
|---|---|---|---|---|---|
| 20 (PCU) | 2.9 (mg/L) | 1375 (μs/cm) | 6.75 (mg/L) | 137.72 (mg/L) | 7.56 |
| Adsorbents | K((ng/mg)/(ng/L)1/n) | 1/n | R2 |
|---|---|---|---|
| PAC | 21.4713 | 0.3414 | 0.9202 |
| TiO2/PAC | 13.5444 | 0.3510 | 0.9231 |
| Photocatalyst | kapp ( min−1) | Correlation Coefficient, R2 |
|---|---|---|
| PAC | 1.850 × 10−3 | 0.892 |
| TiO2/PAC | 16.820 × 10−3 | 0.987 |
| Without catalyst | 0.794 × 10−3 | 0.913 |
| Parameters | PAC | TiO2/PAC | ||
|---|---|---|---|---|
| Adsorption | Photocatalytic | Adsorption | Photocatalytic | |
| n | 0.4317 | 0.4528 | 0.5320 | 0.4251 |
| lnB | 3.9636 | 3.4460 | 3.5520 | 2.6498 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, X.-P.; Zang, P.; Li, Y.-M.; Bi, D.-S. TiO2-Powdered Activated Carbon (TiO2/PAC) for Removal and Photocatalytic Properties of 2-Methylisoborneol (2-MIB) in Water. Water 2021, 13, 1622. https://doi.org/10.3390/w13121622
Guo X-P, Zang P, Li Y-M, Bi D-S. TiO2-Powdered Activated Carbon (TiO2/PAC) for Removal and Photocatalytic Properties of 2-Methylisoborneol (2-MIB) in Water. Water. 2021; 13(12):1622. https://doi.org/10.3390/w13121622
Chicago/Turabian StyleGuo, Xiao-Pin, Peng Zang, Yong-Mei Li, and Dong-Su Bi. 2021. "TiO2-Powdered Activated Carbon (TiO2/PAC) for Removal and Photocatalytic Properties of 2-Methylisoborneol (2-MIB) in Water" Water 13, no. 12: 1622. https://doi.org/10.3390/w13121622
APA StyleGuo, X.-P., Zang, P., Li, Y.-M., & Bi, D.-S. (2021). TiO2-Powdered Activated Carbon (TiO2/PAC) for Removal and Photocatalytic Properties of 2-Methylisoborneol (2-MIB) in Water. Water, 13(12), 1622. https://doi.org/10.3390/w13121622






