Particulate Organic Carbon in the Tropical Usumacinta River, Southeast Mexico: Concentration, Flux, and Sources
Abstract
1. Introduction
2. Materials and Methods
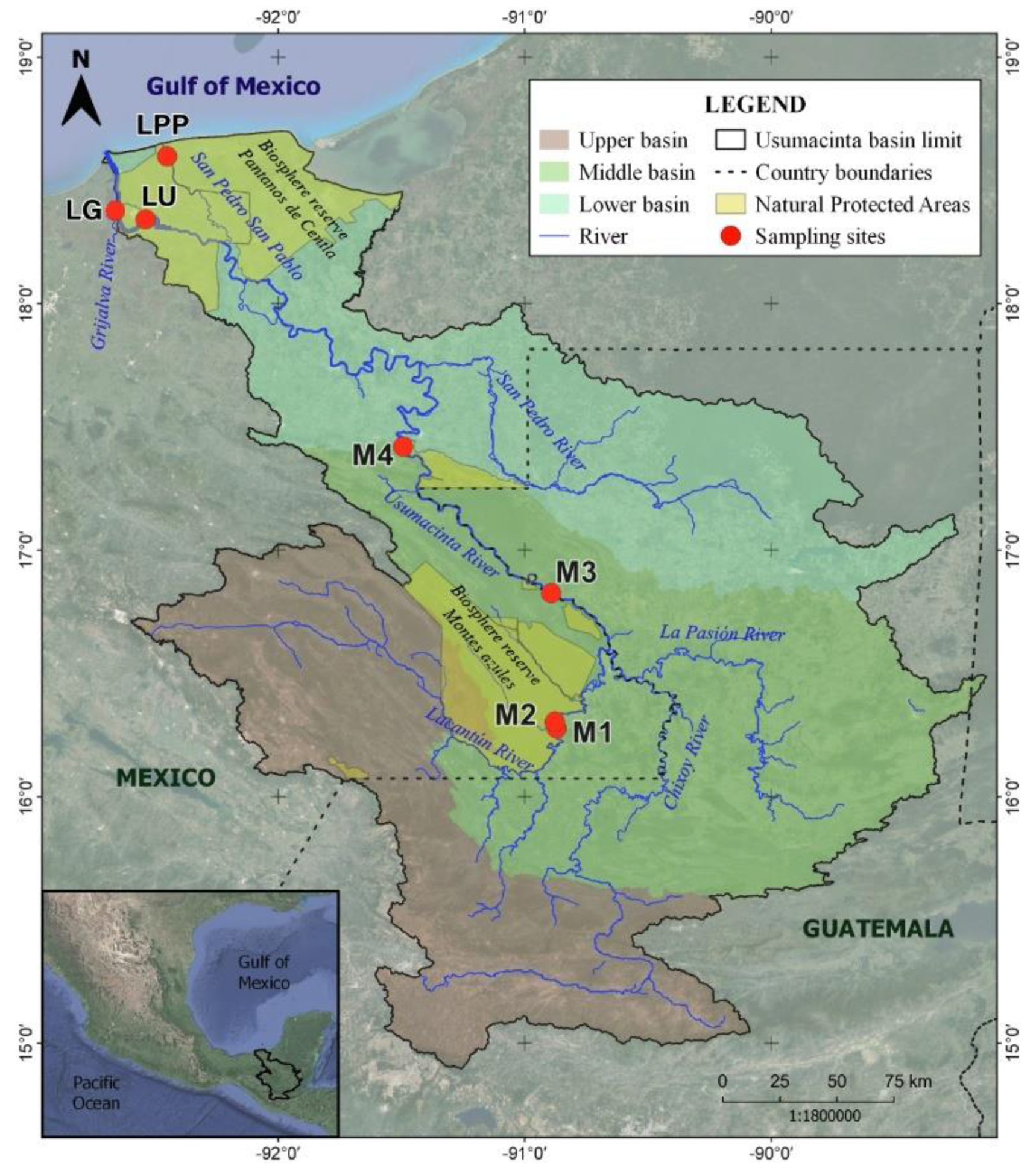
2.1. Study Site
2.2. Sampling
2.3. Analytical Methods
2.4. Data Analysis: POC Sources, Mass Balance, and Statistical and Geographical Analyses
3. Results
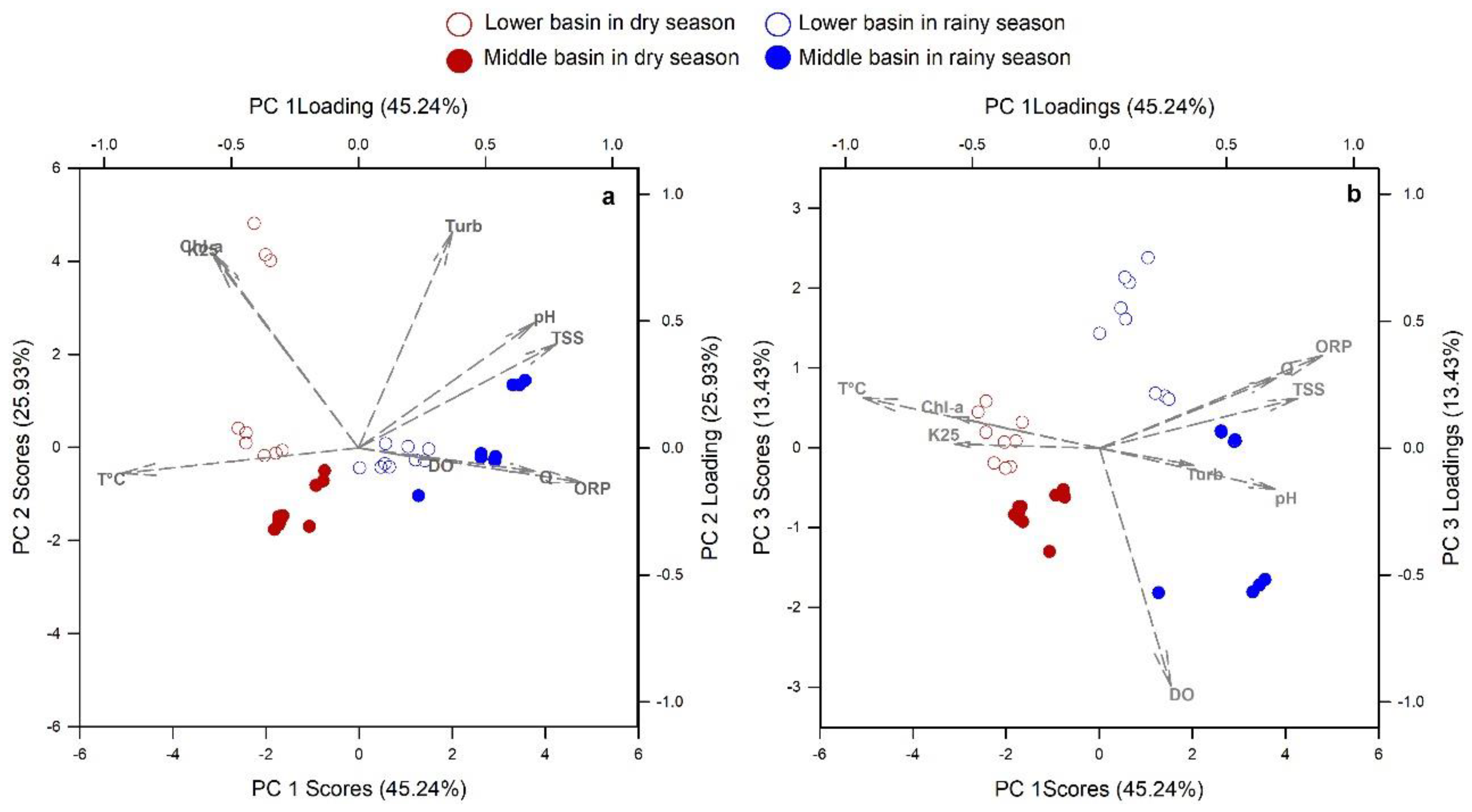
3.1. River Water Quality Dynamics
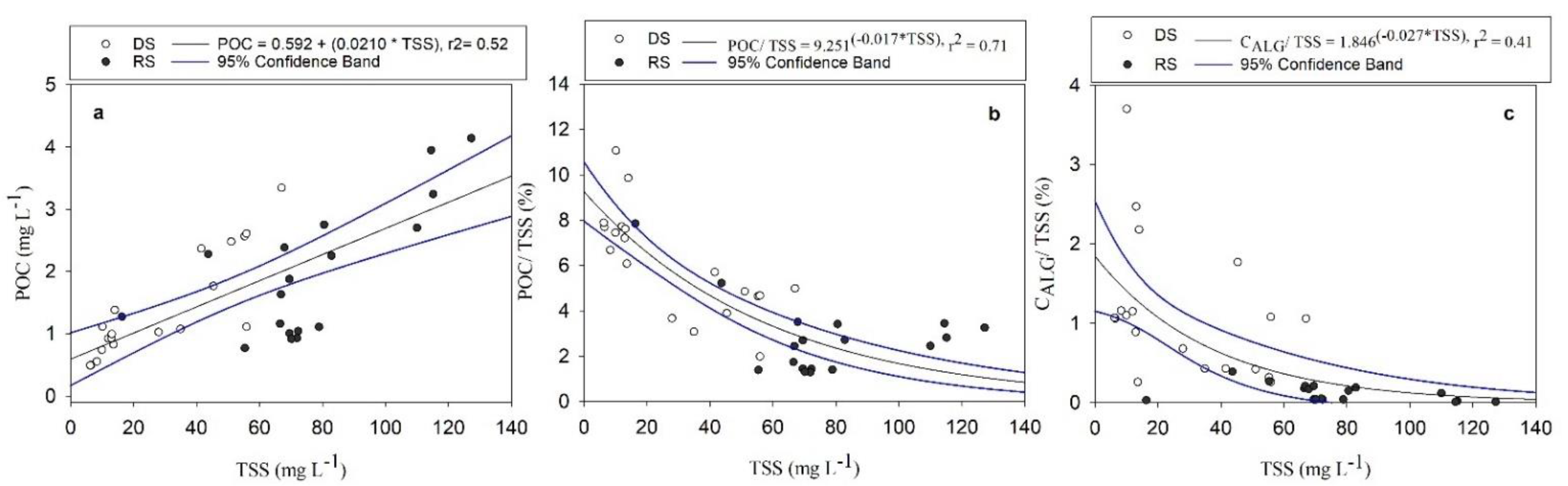
3.2. POC Relationship with Water Quality Variables
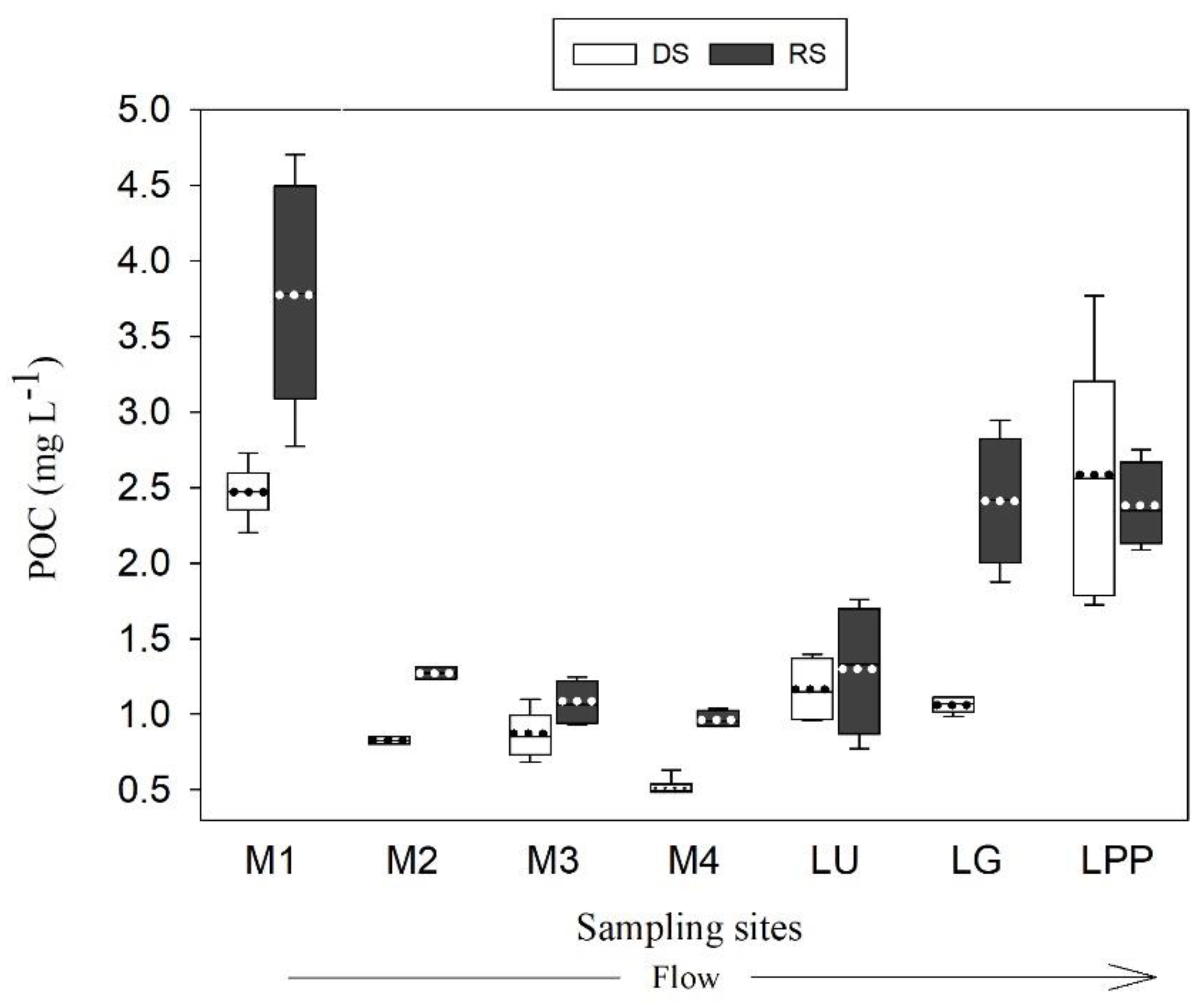
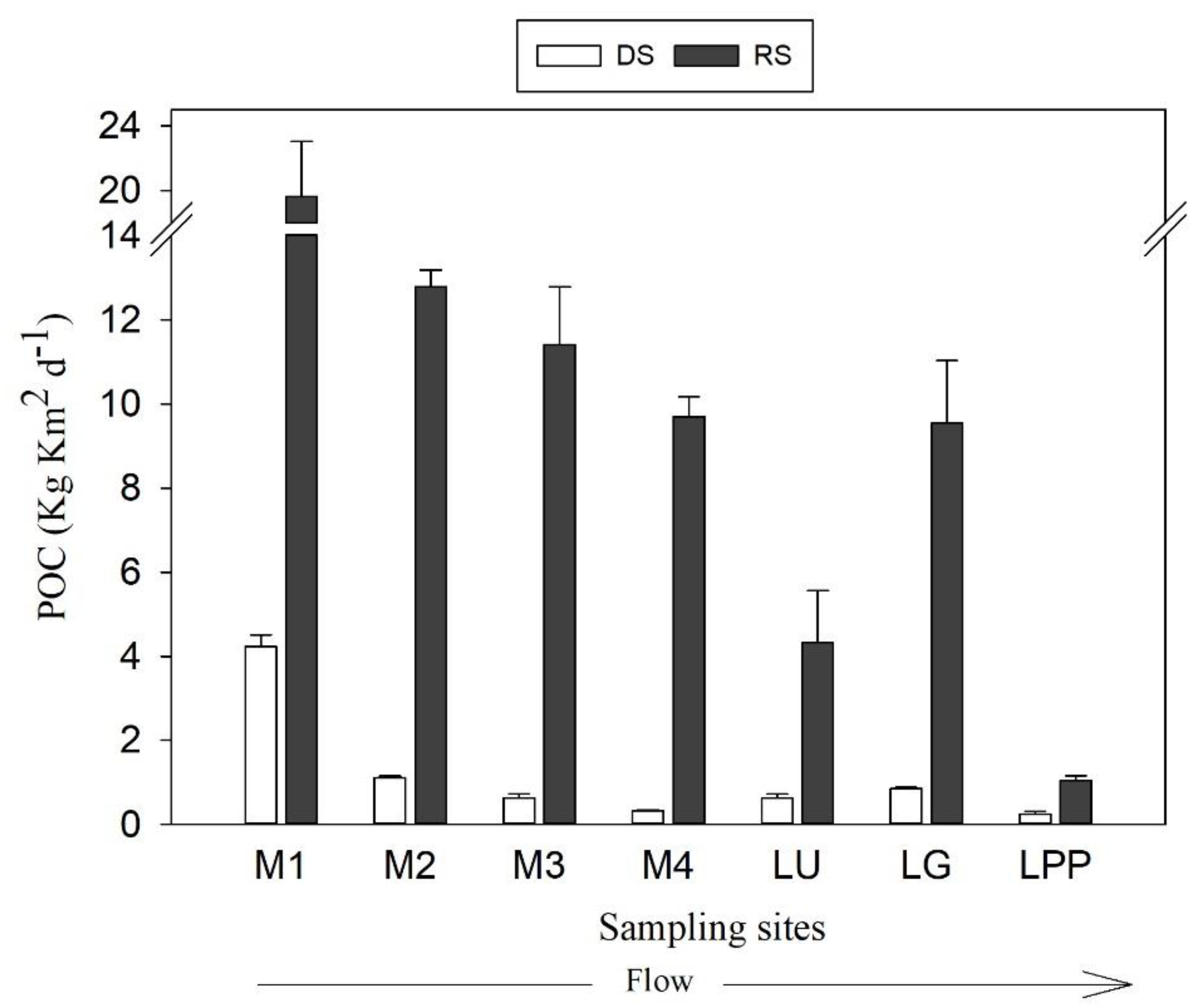
3.3. Variation of POC Concentration and Yield
3.4. Mass Balance of POC in the Usumacinta Lower Basin and POC Export to the Southern Gulf of Mexico
4. Discussion
4.1. Seasonal Hydrologic Differences in the Usumacinta River Basin and its Implication in POC Sources
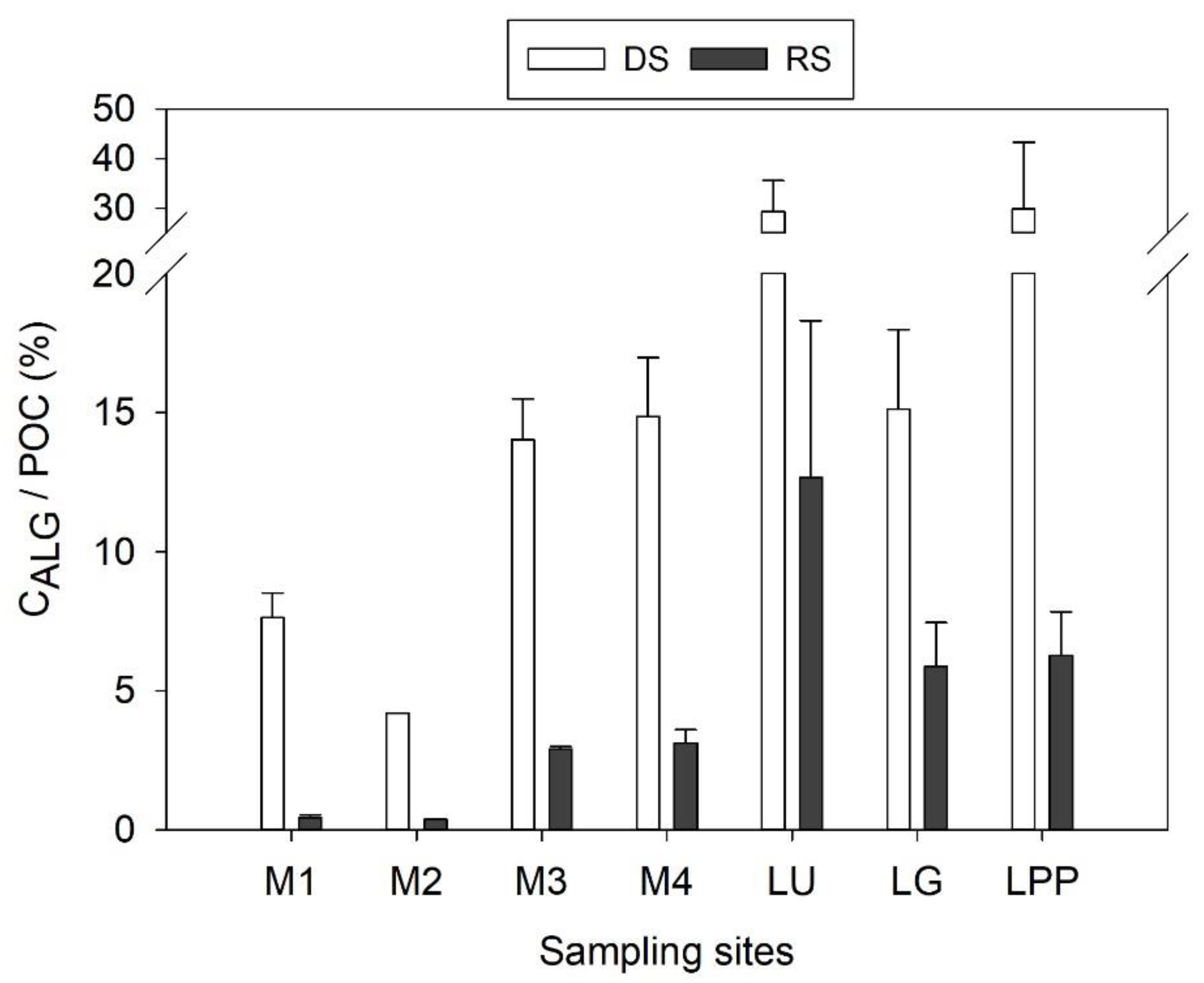
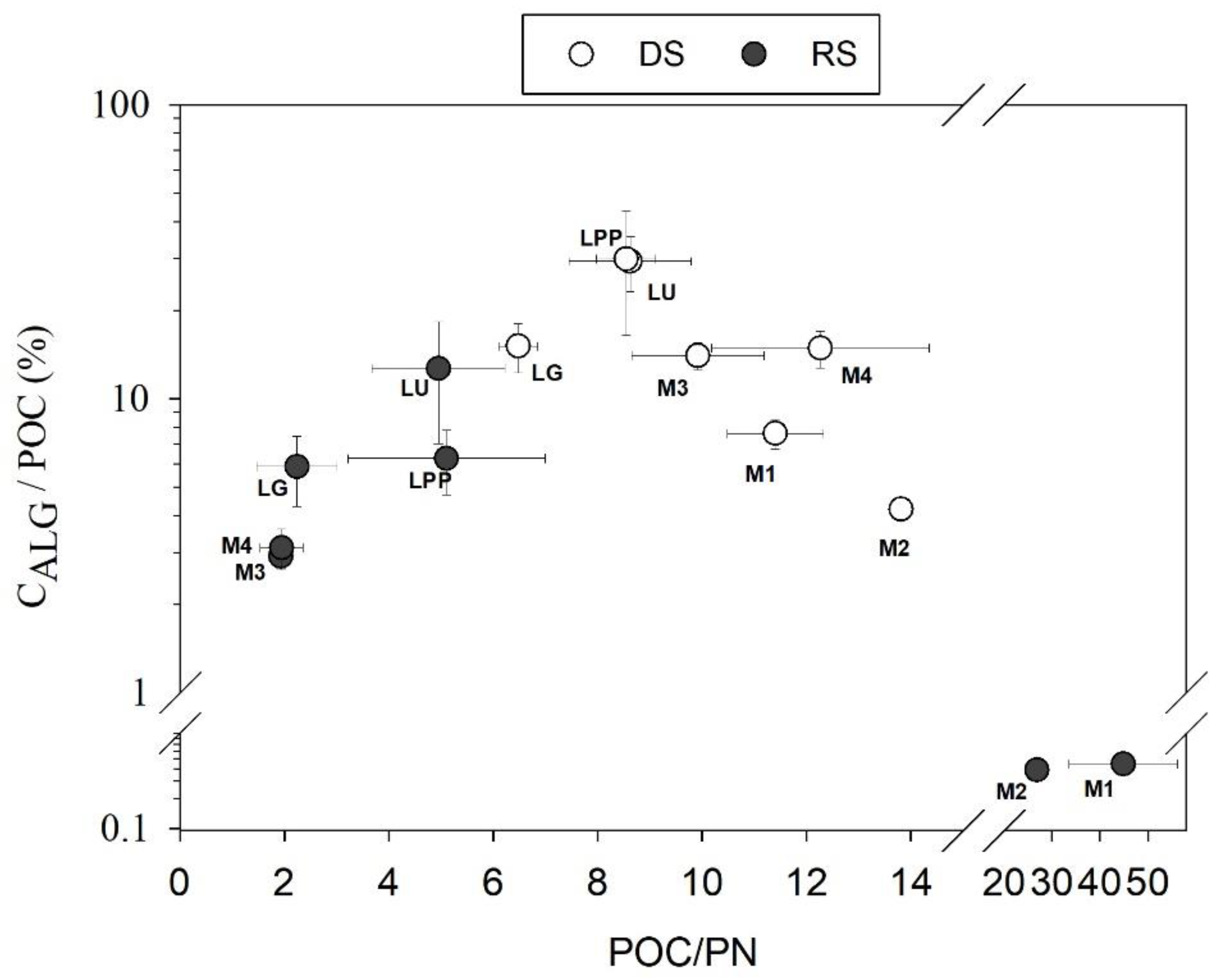
4.2. Spatial Variation of POC Sources
4.3. POC Concentration and Yield Variabilities in the Usumacinta River Basin
4.4. POC Export to the Gulf of Mexico
| River | Country | Zone | POC | Q | Ref. | |
|---|---|---|---|---|---|---|
| (mg L−1) | (t C d−1) | (m3 s−1) | ||||
| Usumacinta | Mexico | Trop | 0.48–4.7 | 109–926 | 23–5924 | This study |
| Maroni | Guyana | Trop | 0.45–4.56 | 17–1266 | 178–4634 | a |
| Oyapock | Guyana | Trop | 0.64–3.16 | 9–279 | 141–2564 | a |
| Orinoco | Venezuela | Trop | 0.17–2.29 | 200–7500 | 500–70,000 | b |
| Amazon | Brazil | Trop | 0.27–26.8 | 4060–33,523 | 28,000–209,000 | c |
| Senegal | Africa | Trop | 0.2–4 | ≤1700 | d | |
| Tana | Kenya | Trop | 0.23–119.8 | 123–208 | e | |
| Mississippi | USA | Temp | 1.9–9.7 | 1013–5287 | 13,196–29,733 | f |
| Yellow | China | Temp | 4.6–92.4 | g | ||
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cole, J.J.; Prairie, Y.T.; Caraco, N.F.; McDowell, W.H.; Tranvik, L.J.; Striegl, R.G.; Duarte, C.M.; Kortelainen, P.; Downing, J.A.; Middelburg, J.J.; et al. Plumbing the Global Carbon Cycle: Integrating Inland Waters into the Terrestrial Carbon Budget. Ecosystems 2007, 10, 172–185. [Google Scholar] [CrossRef]
- Tranvik, L.J.; Cole, J.J.; Prairie, Y.T. The study of carbon in inland waters-from isolated ecosystems to players in the global carbon cycle. Limnol. Oceanogr. Lett. 2018, 3, 41–48. [Google Scholar] [CrossRef]
- Aufdenkampe, A.K.; Mayorga, E.; Raymond, P.A.; Melack, J.M.; Doney, S.C.; Alin, S.R.; Aalto, R.E.; Yoo, K. Riverine coupling of biogeochemical cycles between land, oceans, and atmosphere. Front. Ecol. Environ. 2011, 9, 53–60. [Google Scholar] [CrossRef]
- Schlesinger, W.H.; Melack, J.M. Transport of organic carbon in the world’s rivers. Tellus 1981, 33, 172–187. [Google Scholar] [CrossRef]
- Ludwig, W.; Probst, J.-L.L.; Kempe, S. Predicting the oceanic input of organic carbon by continental erosion. Global Biogeochem. Cycles 1996, 10, 23–41. [Google Scholar] [CrossRef]
- Li, M.; Peng, C.; Wang, M.; Xue, W.; Zhang, K.; Wang, K.; Shi, G.; Zhu, Q. The carbon flux of global rivers: A re-evaluation of amount and spatial patterns. Ecol. Indic. 2017, 80, 40–51. [Google Scholar] [CrossRef]
- Galy, V.; Peucker-Ehrenbrink, B.; Eglinton, T. Global carbon export from the terrestrial biosphere controlled by erosion. Nature 2015, 521, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.-H.; Fu, Y.-H.; Pan, P.-Y.; Chen, C.-T.A. Fluvial carbon fluxes in tropical rivers. Curr. Opin. Environ. Sustain. 2012, 4, 162–169. [Google Scholar] [CrossRef]
- Meybeck, M. Riverine transport of atmospheric carbon: Sources, global typology and budget. Water Air Soil Pollut. 1993, 70, 443–463. [Google Scholar] [CrossRef]
- Bouillon, S.; Yambélé, A.; Spencer, R.G.M.; Gillikin, D.P.; Hernes, P.J.; Six, J.; Merckx, R.; Borges, A.V. Organic matter sources, fluxes and greenhouse gas exchange in the Oubangui River (Congo River basin). Biogeosciences 2012, 9, 2045–2062. [Google Scholar] [CrossRef]
- Bouchez, J.; Galy, V.; Hilton, R.G.; Gaillardet, J.; Moreira-Turcq, P.; Pérez, M.A.; France-Lanord, C.; Maurice, L. Source, transport and fluxes of Amazon River particulate organic carbon: Insights from river sediment depth-profiles. Geochim. Cosmochim. Acta 2014, 133, 280–298. [Google Scholar] [CrossRef]
- Mbaye, M.L.; Gaye, A.T.; Spitzy, A.; Dähnke, K.; Afouda, A.; Gaye, B. Seasonal and spatial variation in suspended matter, organic carbon, nitrogen, and nutrient concentrations of the Senegal River in West Africa. Limnologica 2016, 57, 1–13. [Google Scholar] [CrossRef]
- Ziegler, A.D.; Benner, S.G.; Kunkel, M.L.; Phang, V.X.H.; Lupascu, M.; Tantasirin, C. Particulate carbon and nitrogen dynamics in a headwater catchment in Northern Thailand: Hysteresis, high yields, and hot spots. Hydrol. Process. 2016, 30, 3339–3360. [Google Scholar] [CrossRef]
- Shi, J.; Wang, B.; Wang, F.; Peng, X. Sources and fluxes of particulate organic carbon in the Wujiang cascade reservoirs, southwest China. Inland Waters 2018, 8, 141–147. [Google Scholar] [CrossRef]
- Lloret, E.; Dessert, C.; Lajeunesse, E.; Crispi, O.; Pastor, L.; Gaillardet, J.; Benedetti, M.F. Tropical oceanic islands carbon export Are small mountainous tropical watersheds of oceanic islands important for carbon export? Tropical oceanic islands carbon export. Biogeosci. Discuss. 2012, 9, 7117–7163. [Google Scholar] [CrossRef]
- Hedges, J.I.; Clark, W.A.; Quay, P.D.; Richey, J.E.; Devol, A.H.; Santos, M. Compositions and fluxes of particulate organic material in the Amazon River. Limnol. Oceanogr. 1986, 31, 717–738. [Google Scholar] [CrossRef]
- Richey, J.E.; Hedges, J.I.; Devol, A.H.; Quay, P.D.; Victoria, R.; Martinelli, L.; Forsberg, B.R. Biogeochemistry of carbon in the Amazon River. Limnol. Oceanogr. 1990, 35, 352–371. [Google Scholar] [CrossRef]
- Moreira-Turcq, P.; Seyler, P.; Guyot, J.L.; Etcheber, H. Exportation of organic carbon from the Amazon River and its main tributaries. Hydrol. Process. 2003, 17, 1329–1344. [Google Scholar] [CrossRef]
- Geeraert, N.; Omengo, F.O.; Tamooh, F.; Marwick, T.R.; Borges, A.V.; Govers, G.; Bouillon, S. Seasonal and inter-annual variations in carbon fluxes in a tropical river system (Tana River, Kenya). Biogeosci. Discuss. 2017, 14, 1–27. [Google Scholar] [CrossRef]
- Benke, A. Streams and Rivers of North America: Western, Northern and Mexican Basins. In Encyclopedia of Inland Waters; Likens, G.E., Ed.; Elsevier: Boston, MA, USA, 2009; pp. 414–424. ISBN 0120882531. [Google Scholar]
- Hudson, P.F.; Hendrickson, D.A.; Benke, A.C.; Varela-Rmero, A.; Rodiles-Hernández, R.; Minckley, W.L. Rivers of Mexico. In Rivers of North America; Benke, A., Cushing, C., Eds.; Elsevier: Burlington, MA, USA, 2005; pp. 1030–1084. [Google Scholar]
- Tapia-Silva, F.-O.; Contreras-Silva, A.-I.; Rosales-Arriaga, E.-R. Hydrological characterization of the Usumacinta River Basin towards the preservation of environmental services. ISPRS Int. Arch. Photogramm. Remote Sens. Spat. Inf. Sci. 2015, XL-7/W3, 1505–1509. [Google Scholar] [CrossRef]
- Waibel, L. (Ed.) Sierra Madre de Chiapas, 1st ed.; Miguel Ángel Porrúa: Mexico City, Mexico, 1998; ISBN 968-842-716-7. [Google Scholar]
- Saavedra Guerrero, A.; López López, D.M.; Castellanos Fajardo, L.A. Descripción del medio físico de la cuenca media del río Usumacinta en México. In Conservación y Desarrollo Sustentable en la Selva Lacandona. 25 años de Actividades y Experiencias; Carabias, J., de La Maza, J., Cadena, R., Eds.; (coordinadores); Natura y Ecosistemas Mexicanos: Mexico City, Mexico, 2015; pp. 19–34. ISBN 9786079710200. [Google Scholar]
- Díaz Gallegos, J.R.; Mas, J.-F.; Velázquez, A.; Díaz-Gallegos, J.R.; Mas, J.-F.; Velázquez, A. Trends of tropical deforestation in Southeast Mexico. Singap. J. Trop. Geogr. 2010, 31, 180–196. [Google Scholar] [CrossRef]
- CONAGUA Programas de Medidas Preventivas y de Mitigación a la Sequía (PMPMS) por Consejo de Cuenca. Available online: https://www.gob.mx/cms/uploads/attachment/file/99961/PMPMS_CC_R_os_Grijalva_y_Usumacinta.pdf (accessed on 18 February 2020).
- CCGSS Servicios de Información Geoespacial Plataforma GeoWeb IDEGeo. Available online: https://idegeo.centrogeo.org.mx/layers/geonode:cuenca_17/layer_info_metadata (accessed on 18 March 2021).
- Plascencia Vargas, J.H.; González Espinosa, M.; Ramírez Marcial, N.; Musálem Castillejos, K. Características físico-bióticas de la cuenca del río Grijalva. In Montañas, Pueblos y Agua. Dimenciones y Realidades de la Cuenca Grijalva; González Espinosa, M., Brunel Manse, M.C., Eds.; El Colegio de la Frontera Sur: Mexico City, Mexico, 2014; Volume 1, pp. 29–79. ISBN 978-607-8429-01-1. [Google Scholar]
- Mendoza, A.; Soto-Cortes, G.; Priego-Hernandez, G.; Rivera-Trejo, F. Historical description of the morphology and hydraulic behavior of a bifurcation in the lowlands of the Grijalva River Basin, Mexico. CATENA 2019, 176, 343–351. [Google Scholar] [CrossRef]
- CONAGUA. Estadísticas del Agua en México, 2018th ed.; Secretaría de Medio Ambiente y Recursos Naturales, Comisión Nacional del Agua: Mexico City, Mexico, 2018. [Google Scholar]
- Arreguín-Cortés, F.I.; Rubio-Gutiérrez, H.; Domínguez-Mora, R.; de Luna-Cruz, F. Analysis of Floods in the Tabasco Plains from 1995–2010. Ecnología y Ciencias del Agua 2014, 5, 5–32. [Google Scholar]
- Soares, D.; García, A. La cuenca del río Usumacinta desde la Perspectiva del cambio Climático; Instituto Mexicano de Tecnología del Agua: Jiutepec, Mexico, 2017; ISBN 9786079368807. [Google Scholar]
- Grodsky, S.A.; Carton, J.A. The Intertropical Convergence Zone in the South Atlantic and the Equatorial Cold Tongue. J. Clim. 2003, 16, 723–733. [Google Scholar] [CrossRef]
- Muñoz-Salinas, E.; Castillo, M. Streamflow and sediment load assessment from 1950 to 2006 in the Usumacinta and Grijalva Rivers (Southern Mexico) and the influence of ENSO. Catena 2015, 127, 270–278. [Google Scholar] [CrossRef]
- Alcérreca-Huerta, J.C.; Callejas-Jiménez, M.E.; Carrillo, L.; Castillo, M.M. Dam implications on salt-water intrusion and land use within a tropical estuarine environment of the Gulf of Mexico. Sci. Total Environ. 2018, 652, 1102–1112. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Salinas, E.; Castillo, M.; Sanderson, D.; Kinnaird, T.; Cruz-Zaragoza, E. Using three different approaches of OSL for the study of young fluvial sediments at the coastal plain of the Usumacinta-Grijalva River Basin, southern Mexico. Earth Surf. Process. Landforms 2016, 41, 823–834. [Google Scholar] [CrossRef]
- de la Maza, J. Caracterización de la subcuenca del Lacantún. In Conservación y Desarrollo Sustentable de la Selva Lacandona; Natura y Ecosistemas Mexicanos, A.C.: Mexico City, Mexico, 2015; pp. 79–83. [Google Scholar]
- Peralta-Carreta, C.; Gallardo-Cruz, J.A.; Solórzano, J.V.; Hernandez-Gómez, M. Clasificación del uso de suelo y vegetación en áreas de pérdida de cobertura arbórea (2000–2016) en la cuenca del río Usumacinta. Madera y Bosques 2019, 25, 1–19. [Google Scholar] [CrossRef]
- García, A.G.; Kauffer, M.E. Las cuencas compartidas entre México, Guatemala y Belice: Un acercamiento a su delimitación y problemática general. Front. Norte 2011, 23, 131–162. [Google Scholar]
- Sánchez-Hernández, R.; De Dios Mendoza-Palacios, J.; De, J.C.; Reyes, C.; Mendoza Martínez, J.E.; Ramos-Reyes, R. Erosión potencial en la cuenca baja Grijalva-Usumacinta. Univ. y Cienc. 2013, 29, 153–161. [Google Scholar]
- Castillo, M.M. Suspended sediment, nutrients, and chlorophyll in tropical floodplain lakes with different patterns of hydrological connectivity. Limnologica 2020, 82, 125767. [Google Scholar] [CrossRef]
- Olivera, M. de los A.G.; Gil, G.O. Análisis de la flora de la Reserva de la Biosfera de los Pantanos de Centla, Tabasco, México. Univ. y Cienc. 2000, 15, 67–104. [Google Scholar]
- Arar, E.J.; Collins, G.B. Method 445.0 In Vitro Determination of Chlorophyll a and Pheophytin ain Marine and Freshwater Algae by Fluorescence; Environmental Protection Agency: Washington, DC, USA, 1997. [Google Scholar]
- Ran, L.; Lu, X.X.; Sun, H.; Han, J.; Li, R.; Zhang, J. Spatial and seasonal variability of organic carbon transport in the Yellow River, China. J. Hydrol. 2013, 498, 76–88. [Google Scholar] [CrossRef]
- Meybeck, M. Origins and Behaviors of Carbon Species in World Rivers. In Soil Erosion and Carbon Dynamics; Roose, E.J., Lal, R., Feller, C., Barthes, B., Stewart, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 2005; pp. 209–238. [Google Scholar]
- Del Giorgio, P.A.; Gasol, J.M. Biomass Distribution in Freshwater Plankton Communities. Am. Nat. 1995, 146, 135–152. [Google Scholar] [CrossRef]
- Brust, G.E. Management strategies for organic vegetable fertility. In Safety and Practice for Organic Food; Elsevier: San Diego, CA, USA, 2019; pp. 193–212. ISBN 9780128120606. [Google Scholar]
- DelDuco, E.M.; Xu, Y.J. Dissolved carbon transport and processing in North America’s largest swamp river entering the Northern Gulf of Mexico. Water 2019, 11, 1395. [Google Scholar] [CrossRef]
- U.S. Geological Survey USGS EROS Archive—Digital Elevation—Shuttle Radar Topography Mission (SRTM). Available online: https://doi.org/10.5066/F7PR7TFT (accessed on 31 March 2021).
- QGIS.org QGIS Geographic Information System 2021. Available online: http://www.qgis.org (accessed on 21 March 2021).
- Benedetti, M.F.; Mounier, S.; Filizola, N.; Benaim, J.; Seyler, P. Carbon and metal concentrations, size distributions and fluxes in major rivers of the Amazon basin. Hydrol. Process. 2003, 17, 1363–1377. [Google Scholar] [CrossRef]
- Seyler, P.; Coynel, A.; Moreira-Turcq, P.; Etcheber, H.; Colas, C.; Orange, D.; Bricquet, J.-P.; Laraque, A.; Luc Guyot, J.; Olivry, J.-C.; et al. Organic Carbon Transported by the Equatorial Rivers: Example of Congo-Zaire and Amazon Basins. In Soil Erosion and Carbon Dynamics; CRC Press: Boca Raton, FL, USA, 2005; pp. 255–274. [Google Scholar]
- Tamooh, F.; Van Den Meersche, K.; Meysman, F.; Marwick, T.R.; Borges, A.V.; Merckx, R.; Dehairs, F.; Schmidt, S.; Nyunja, J.; Bouillon, S. Distribution and origin of suspended matter and organic carbon pools in the Tana River Basin, Kenya. Biogeosciences 2012, 9, 2905–2920. [Google Scholar] [CrossRef]
- Cai, Y.; Guo, L.; Wang, X.; Aiken, G. Abundance, stable isotopic composition, and export fluxes of DOC, POC, and DIC from the Lower Mississippi River during 2006-2008. J. Geophys. Res. Biogeosciences 2015, 120, 2273–2288. [Google Scholar] [CrossRef]
- Webster, J.R.; Benfield, E.F.; Golladay, S.W.; Hill, B.H.; Hornick, L.E.; Kazmierczak, R.F.; Perry, W.B. Experimental studies of physical factors affecting seston transport in streams1. Limnol. Oceanogr. 1987, 32, 848–863. [Google Scholar] [CrossRef]
- Zou, J. Geochemical characteristics and organic carbon sources within the upper reaches of the Xi River, southwest China during high flow. J. Earth Syst. Sci. 2017, 126, 6. [Google Scholar] [CrossRef][Green Version]
- Tao, S.; Eglinton, T.I.; Zhang, L.; Yi, Z.; Montluçon, D.B.; McIntyre, C.; Yu, M.; Zhao, M. Temporal variability in composition and fluxes of Yellow River particulate organic matter. Limnol. Oceanogr. 2018, 63, S119–S141. [Google Scholar] [CrossRef]
- Ittekkot, V. Global trends in the nature of organic matter in river suspensions. Nature 1988, 332, 436–438. [Google Scholar] [CrossRef]
- Coynel, A.; Seyler, P.; Etcheber, H.; Meybeck, M.; Orange, D. Spatial and seasonal dynamics of total suspended sediment and organic carbon species in the Congo River. Global Biogeochem. Cycles 2005, 19, 1–17. [Google Scholar] [CrossRef]
- Kiran, S.; Reddy, K.; Gupta, H.; Badimela, U.; Reddy, D.V.; Kurakalva, R.M.; Kumar, D. Export of particulate organic carbon by the mountainous tropical rivers of Western Ghats, India: Variations and controls. Sci. Total Environ. 2021, 751, 142115. [Google Scholar] [CrossRef]
- Tao, F.-X.; Liu, C.-Q.; Li, S.-L. Source and flux of POC in two subtropical karstic tributaries with contrasting land use practice in the Yangtze River Basin. Appl. Geochemistry 2009, 24, 2102–2112. [Google Scholar] [CrossRef]
- Beck, H. Tropical ecology. Encycl. Ecol. 2018, 2, 671–678. [Google Scholar] [CrossRef]
- Syvitski, J.P.M.; Cohen, S.; Kettner, A.J.; Brakenridge, G.R. How important and different are tropical rivers?-An overview. Geomorphology 2014, 227, 5–17. [Google Scholar] [CrossRef]
- Kendall, C.; Silva, S.R.; Kelly, V.J. Carbon and nitrogen isotopic compositions of particulate organic matter in four large river systems across the United States. Hydrol. Process. 2001, 15, 1301–1346. [Google Scholar] [CrossRef]
- Cleveland, C.C.; Liptzin, D. C:N:P stoichiometry in soil: Is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 2007, 85, 235–252. [Google Scholar] [CrossRef]
- Bird, M.I.; Robinson, R.A.J.; Oo, N.W.; Aye, M.M.; Lu, X.X.; Higgitt, D.L.; Swe, A.; Tun, T.; Lhaing Win, S.; Sandar Aye, K.; et al. A preliminary estimate of organic carbon transport by the Ayeyarwady (Irrawaddy) and Thanlwin (Salween) Rivers of Myanmar. Quat. Int. 2008, 186, 113–122. [Google Scholar] [CrossRef]
- Navarrete-Segueda, A.; Martínez-Ramos, M.; Ibarra-Manríquez, G.; Vázquez-Selem, L.; Siebe, C. Variation of main terrestrial carbon stocks at the landscape-scale are shaped by soil in a tropical rainforest. Geoderma 2018, 313, 57–68. [Google Scholar] [CrossRef]
- Meyers, P.A.; Ishiwatari, R. Lacustrine organic geochemistryman overview of indicators of organic matter sources and diagenesis in lake sediments. Org. Geochem. 1993, 20, 867–900. [Google Scholar] [CrossRef]
- Weiguo, L.; Zisheng, A.; Weijian, Z.; Head, M.; Delin, C. Carbon isotope and C/N ratios of suspended matter in rivers: An indicator of seasonal change in C4/C3 vegetation. Appl. Geochem. 2003, 18, 1241–1249. [Google Scholar] [CrossRef]
- Meyers, P.A. Preservation of elemental and isotopic source identification of sedimentary organic matter. Chem. Geol. 1994, 114, 289–302. [Google Scholar] [CrossRef]
- Onstad, G.D.; Canfield, D.E.; Quay, P.D.; Hedges, J.I. Sources of particulate organic matter in rivers from the continental usa: Lignin phenol and stable carbon isotope compositions. Geochim. Cosmochim. Acta 2000, 64, 3539–3546. [Google Scholar] [CrossRef]
- Kohler, T.J.; Heatherly, T.N.; El-Sabaawi, R.W.; Zandonà, E.; Marshall, M.C.; Flecker, A.S.; Pringle, C.M.; Reznick, D.N.; Thomas, S.A. Flow, nutrients, and light availability influence Neotropical epilithon biomass and stoichiometry. Freshw. Sci. 2012, 31, 1019–1034. [Google Scholar] [CrossRef]
- Ward, N.D.; Bianchi, T.S.; Medeiros, P.M.; Seidel, M.; Richey, J.E.; Keil, R.G.; Sawakuchi, H.O. Where Carbon Goes When Water Flows: Carbon Cycling across the Aquatic Continuum. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Lewis, W.M. Physical and Chemical Features of Tropical Flowing Waters. In Tropical Stream Ecology; Dudgeon, D., Ed.; Academic Press: London, UK, 2008; pp. 1–21. ISBN 9780120884490. [Google Scholar]
- Moreira-Turcq, P.; Bonnet, M.P.; Amorim, M.; Bernardes, M.; Lagane, C.; Maurice, L.; Perez, M.; Seyler, P. Seasonal variability in concentration, composition, age, and fluxes of particulate organic carbon exchanged between the floodplain and Amazon River. Global Biogeochem. Cycles 2013, 27, 119–130. [Google Scholar] [CrossRef]
- Jobbágy, E.G.; Jackson, R.B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl. 2000, 10, 423–436. [Google Scholar] [CrossRef]
- Keller, C.K. Carbon Exports from Terrestrial Ecosystems: A Critical-Zone Framework. Ecosystems 2019, 22, 1–15. [Google Scholar] [CrossRef]
- Ludwig, W. Continental Erosion and River Transport of Organic Carbon to the World’s Oceans; Mémoire, 9.; Sciences Géologiques, University Louis-Pasteur: Strasbourg, France, 1997. [Google Scholar]
- Selva, E.C.; Couto, E.G.; Johnson, M.S.; Lehmann, J. Litterfall production and fluvial export in headwater catchments of the southern Amazon. J. Trop. Ecol. 2007, 23, 329–335. [Google Scholar] [CrossRef][Green Version]
- Nooren, K.; Hoek, W.Z.; Winkels, T.; Huizinga, A.; Van der Plicht, H.; Van Dam, R.L.; Van Heteren, S.; Van Bergen, M.J.; Prins, M.A.; Reimann, T.; et al. The Usumacinta–Grijalva beach-ridge plain in southern Mexico: A high-resolution archive of river discharge and precipitation. Earth Surf. Dyn. 2017, 5, 529–556. [Google Scholar] [CrossRef]
- Noe, G.B.; Hupp, C.R. Carbon, nitrogen, and phosphorus accumulation in floodplains of Atlantic Coastal Plain rivers, USA. Ecol. Appl. 2005, 15, 1178–1190. [Google Scholar] [CrossRef]
- Sutfin, N.A.; Wohl, E.E.; Dwire, K.A. Banking carbon: A review of organic carbon storage and physical factors influencing retention in floodplains and riparian ecosystems. Earth Surf. Process. Landforms 2016, 41, 38–60. [Google Scholar] [CrossRef]
- Hedges, J.I.; Mayorga, E.; Tsamakis, E.; McClain, M.E.; Aufdenkampe, A.; Quay, P.; Richey, J.E.; Benner, R.; Opsahl, S.; Black, B.; et al. Organic matter in Bolivian tributaries of the Amazon River: A comparison to the lower mainstream. Limnol. Oceanogr. 2000, 45, 1449–1466. [Google Scholar] [CrossRef]
- Milliman, J.D.; Farnsworth, K.L. River Discharge to the Coastal Ocean: A Global Synthesis; Cambridge University Press: Cambridge, UK, 2011; ISBN 9780511781247. [Google Scholar]
- Gallay, M.; Mora, A.; Martinez, J.-M.; Gardel, A.; Laraque, A.; Sarrazin, M.; Beaucher, E.; Doudou, J.-C.; Lagane, C. Dynamics and fluxes of organic carbon and nitrogen in two Guiana Shield river basins impacted by deforestation and mining activities. Hydrol. Process. 2018, 32, 17–29. [Google Scholar] [CrossRef]
- Tamooh, F.; Meysman, F.J.R.; Borges, A.V.; Marwick, T.R.; Van Den Meersche, K.; Dehairs, F.; Merckx, R.; Bouillon, S. Sediment and carbon fluxes along a longitudinal gradient in the lower Tana River (Kenya). J. Geophys. Res. G Biogeosci. 2014, 119, 1340–1353. [Google Scholar] [CrossRef]
- Liu, J.; Song, X.; Wang, Z.; Yang, L.; Sun, Z.; Wang, W. Variations of carbon transport in the Yellow River, China. Hydrol. Res. 2015, 46, 746–762. [Google Scholar] [CrossRef]
- Mora, A.; Laraque, A.; Moreira-Turcq, P.; Alfonso, J.A. Temporal variation and fluxes of dissolved and particulate organic carbon in the Apure, Caura and Orinoco rivers, Venezuela. J. S. Am. Earth Sci. 2014, 54, 47–56. [Google Scholar] [CrossRef]
- Wang, X.-C.; Chen, R.F.; Gardner, G.B. Sources and transport of dissolved and particulate organic carbon in the Mississippi River estuary and adjacent coastal waters of the northern Gulf of Mexico. Mar. Chem. 2004, 89, 241–256. [Google Scholar] [CrossRef]
- Liu, Z.; Xue, J. The Lability and Source of Particulate Organic Matter in the Northern Gulf of Mexico Hypoxic Zone. J. Geophys. Res. Biogeosciences 2020, 125, 1–16. [Google Scholar] [CrossRef]
- Signoret, M.; Monreal-Gómez, M.A.; Aldeco, J.; Salas-de-León, D.A. Hydrography, oxygen saturation, suspended particulate matter, and chlorophyll-a fluorescence in an oceanic region under freshwater influence. Estuar. Coast. Shelf Sci. 2006, 69, 153–164. [Google Scholar] [CrossRef]
- Cardoso-Mohedano, J.G.; Canales-Delgadillo, J.C.; Machain-Castillo, M.L.; Hernández-Hernández, J.G.; Sanchez-Cabeza, J.A.; Ruiz-Fernández, A.C.; Alonso-Rodríguez, R.; Gómez-Ponce, M.A.; Esqueda-Lara, K.; Merino-Ibarra, M.; et al. Absence of hypoxia events in the adjacent coastal waters of Grijalva-Usumacinta river, Southern Gulf of Mexico. Mar. Pollut. Bull. 2020, 156, 111174. [Google Scholar] [CrossRef] [PubMed]
| Season | River/Site | Q | Zmax | T | DO | pH | K25 | ORP | Turb | Chl-a | TSS | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dry Season | Lacantún/M1 + | X− | 312 | 3.5 | 27.3 | 6.3 | 6.9 * | 395 | 208 | 46 | 4.7 | 49 |
| σ | 0 | <0.1 | 1.1 | 2 | 2 | 8 | 0.5 | 7 | ||||
| Tzendales/M2 + | X− | 23 | 1.5 | 27.8 | 7.2 | 7.1 * | 931 | 202 | 10 | 0.9 | 14 | |
| σ | 0 | 0 | 1.0 | 0 | <1 | 1 | 0.1 | 1 | ||||
| Usumacinta/M3 | X− | 396 | 4.2 | 30.7 | 6.5 | 7.1 * | 706 | 197 | 11 | 3.0 | 12 | |
| σ | 0.1 | <0.1 | 1.1 | <1 | 8 | 1 | 0.4 | 1 | ||||
| Usumacinta/M4 | X− | 369 | 20.1 | 30.7 | 6.4 | 7.2 * | 839 | 169 | 7 | 1.9 | 7 | |
| σ | 0.1 | <0.1 | 1.2 | 3 | 3 | 1 | 0.4 | 1 | ||||
| Usumacinta/LU | X− | 432 | 12.5 | 29.9 | 3.3 | 7.4 * | 33,368 | 153 | 8 | 8.3 | 12 | |
| σ | 0.9 | 2.1 | 1.1 | 18,806 | 14 | 3 | 0.8 | 2 | ||||
| Grijalva/LG + | X− | 527 | 11 | 30.3 | 4.0 | 7.4 * | 27,610 | 142 | 6 | 4.0 | 40 | |
| σ | 0.8 | 2.2 | 1.2 | 15,126 | 5 | 1 | 1.0 | 15 | ||||
| San Pedro–San Pablo/LPP ─ | X− | 78 | 4.8 | 29.8 | 5.1 | 7.6 * | 57,098 | 142 | 132 | 17.6 | 54 | |
| σ | 0.3 | 0.8 | 1.1 | 1148 | 5 | 85 | 2.2 | 11 | ||||
| Total | X− | 305 | 8.2 | 29.6 | 5.5 | 7.2 * | 13,019 | 174 | 23 | 6.3 | 28 | |
| σ | 187 | 6.6 | 2.2 | 1.9 | 1.6 | 19,866 | 26 | 36 | 5.5 | 20 | ||
| Rainy Season | Lacantún/M1 + | X− | 948 | 6.3 | 22.2 | 8.5 | 8.1 * | 374 | 346 | 100 | 0.4 | 119 |
| σ | <0.1 | <0.1 | 1.1 | <1 | 16 | 4 | 0.1 | 14 | ||||
| Tzendales/M2 + | X− | 173 | 3.8 | 23.4 | 8.0 | 8.1 * | 539 | 355 | 8 | 0.1 | 16 | |
| σ | <0.1 | <0.1 | 1.0 | <1 | 1 | 1 | 0.0 | |||||
| Usumacinta/M3 | X− | 5715 | 15.6 | 24.3 | 5.8 | 7.7 * | 316 | 357 | 55 | 0.8 | 76 | |
| σ | <0.1 | 0.1 | 1.1 | 5 | 9 | 1 | 0.1 | 6 | ||||
| Usumacinta/M4 | X− | 5934 | 35.2 | 24.9 | 6.6 | 7.8 * | 359 | 428 | 52 | 0.7 | 71 | |
| σ | <0.1 | <0.1 | 1.0 | <1 | 4 | 2 | 0.1 | 2 | ||||
| Usumacinta/L1 | X− | 2709 | 17.1 | 26.7 | 4.6 | 7.7 * | 361 | 409 | 44 | 3.4 | 63 | |
| σ | <0.1 | 0.1 | 1.0 | <1 | 2 | 8 | 0.3 | 7 | ||||
| Grijalva/LG + | X− | 2618 | 12.3 | 27.9 | 1.4 | 7.3 * | 337 | 381 | 49 | 3.3 | 82 | |
| σ | <0.1 | 0.1 | 1.1 | 1 | 10 | 9 | 0.3 | 22 | ||||
| San Pedro–San Pablo/LPP ─ | X− | 368 | 4.8 | 27.5 | 1.9 | 7.4 * | 394 | 390 | 43 | 3.7 | 69 | |
| σ | 0.1 | 0.2 | 1.1 | 14 | 4 | 15 | 0.5 | 20 | ||||
| Total | X− | 2638 | 14 | 25.4 | 5.2 | 7.7 * | 360 | 390 | 55 | 2.0 | 78 | |
| σ | 2395 | 11 | 1.9 | 2.3 | 1.7 | 37 | 31 | 21 | 1.5 | 25 |
| POC | POC/TSS (%) | POC/PN | CALG/POC (%) | |||||
|---|---|---|---|---|---|---|---|---|
| Season | River/Site | Concentration (mg L−1) | Flux (t C d−1) | Yield (kg C km2 d−1) | ||||
| Dry Season | Lacantún/M1 + | X− | 2.47 | 66.7 | 4.2 | 5.1 | 14 | 7.6 |
| σ | 0.16 | 4.4 | 0.3 | 0.6 | 2 | 0.9 | ||
| Tzendales/M2 + | X− | 0.83 | 1.7 | 1.1 | 6.1 | 11 | 4.2 | |
| σ | 0.03 | 0.1 | 0.0 | 1 | ||||
| Usumacinta/M3 | X− | 0.86 | 29.5 | 0.6 | 7.5 | 10 | 14.0 | |
| σ | 0.14 | 4.7 | 0.1 | 0.3 | 1 | 1.5 | ||
| Usumacinta/M4 | X− | 0.51 | 16.4 | 0.3 | 7.4 | 12 | 14.9 | |
| σ | 0.05 | 1.7 | 0.0 | 0.6 | 2 | 2.1 | ||
| Usumacinta/LU | X− | 1.16 | 43.4 | 0.6 | 9.5 | 9 | 29.4 | |
| σ | 0.18 | 6.8 | 0.1 | 1.8 | 1 | 6.2 | ||
| Grijalva/LG + | X− | 1.06 | 48.4 | 0.8 | 2.9 | 6 | 15.1 | |
| σ | 0.05 | 2.1 | 0.0 | 0.9 | 1 | 2.9 | ||
| San Pedro–San Pablo/LPP ─ | X− | 2.57 | 17.3 | 0.2 | 4.5 | 9 | 29.9 | |
| σ | 0.73 | 4.9 | 0.1 | 0.6 | 0 | 13.4 | ||
| Total | X− | 1.42 | 33.3 | 1.1 | 3.7 | 10 | 16.4 | |
| σ | 0.86 | 21.1 | 1.4 | 3.2 | 2 | 4.8 | ||
| Rainy Season | Lacantún/M1 + | X− | 3.78 | 309.3 | 19.6 | 3.2 | 45 | 0.4 |
| σ | 0.66 | 54.3 | 3.4 | 0.3 | 11 | 0.1 | ||
| Tzendales/M2 + | X− | 1.27 | 19 | 12.8 | 7.8 | 27 | 0.4 | |
| σ | 0.04 | 0.6 | 0.4 | 1 | ||||
| Usumacinta/M3 | X− | 1.07 | 530.7 | 11.4 | 1.4 | 2 | 2.9 | |
| σ | 0.13 | 63.6 | 1.4 | 0.0 | 0 | 0.1 | ||
| Usumacinta/M4 | X− | 0.96 | 494.7 | 9.7 | 1.4 | 2 | 3.1 | |
| σ | 0.05 | 23.9 | 0.5 | 0.1 | 0 | 0.5 | ||
| Usumacinta/LU | X− | 1.30 | 304 | 4.3 | 1.9 | 5 | 12.7 | |
| σ | 0.37 | 87.1 | 1.2 | 0.5 | 1 | 5.6 | ||
| Grijalva/LG + | X− | 2.42 | 546.3 | 9.5 | 2.9 | 2 | 5.9 | |
| σ | 0.38 | 85.6 | 1.5 | 0.6 | 1 | 1.6 | ||
| San Pedro–San Pablo/LPP ─ | X− | 2.38 | 75.9 | 1.0 | 3.8 | 5 | 6.3 | |
| σ | 0.24 | 7.7 | 0.1 | 1.3 | 2 | 1.6 | ||
| Total | X− | 2.06 | 323.0 | 9.8 | 1.9 | 10 | 4.5 | |
| σ | 1.14 | 198.9 | 6.0 | 2.1 | 14 | 2.1 | ||
| Site | Q (m3 s−1) | POC Flux (t C d−1) | |||
|---|---|---|---|---|---|
| DS | RS | DS | RS | ||
| Input | M4 | 369 | 5934 | 16.4 | 494.7 |
| Output | LU | 432 | 2709 | 43.4 | 304.0 |
| LPP | 78 | 368 | 17.3 | 75.9 | |
| Lower Usumacinta basin balance(input-output) | M4 − (LU + LPP) | −141 | 2857 | −44.3 | 114.9 |
| Usumacinta basin export | LU + LPP | 510 | 3077 | 60.7 | 379.9 |
| Usumacinta–Grijalva basin export | LU + LG + LPP | 1037 | 5695 | 109.1 | 926.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuevas-Lara, D.; Alcocer, J.; Cortés-Guzmán, D.; Soria-Reinoso, I.F.; García-Oliva, F.; Sánchez-Carrillo, S.; Oseguera, L.A. Particulate Organic Carbon in the Tropical Usumacinta River, Southeast Mexico: Concentration, Flux, and Sources. Water 2021, 13, 1561. https://doi.org/10.3390/w13111561
Cuevas-Lara D, Alcocer J, Cortés-Guzmán D, Soria-Reinoso IF, García-Oliva F, Sánchez-Carrillo S, Oseguera LA. Particulate Organic Carbon in the Tropical Usumacinta River, Southeast Mexico: Concentration, Flux, and Sources. Water. 2021; 13(11):1561. https://doi.org/10.3390/w13111561
Chicago/Turabian StyleCuevas-Lara, Daniel, Javier Alcocer, Daniela Cortés-Guzmán, Ismael F. Soria-Reinoso, Felipe García-Oliva, Salvador Sánchez-Carrillo, and Luis A. Oseguera. 2021. "Particulate Organic Carbon in the Tropical Usumacinta River, Southeast Mexico: Concentration, Flux, and Sources" Water 13, no. 11: 1561. https://doi.org/10.3390/w13111561
APA StyleCuevas-Lara, D., Alcocer, J., Cortés-Guzmán, D., Soria-Reinoso, I. F., García-Oliva, F., Sánchez-Carrillo, S., & Oseguera, L. A. (2021). Particulate Organic Carbon in the Tropical Usumacinta River, Southeast Mexico: Concentration, Flux, and Sources. Water, 13(11), 1561. https://doi.org/10.3390/w13111561











