Geochemical Characteristics of Alluvial Aquifer in the Varaždin Region
Abstract
:1. Introduction
2. Description of the Study Area
3. Materials and Methods
4. Results
5. Discussion
6. Conclusions
- (a)
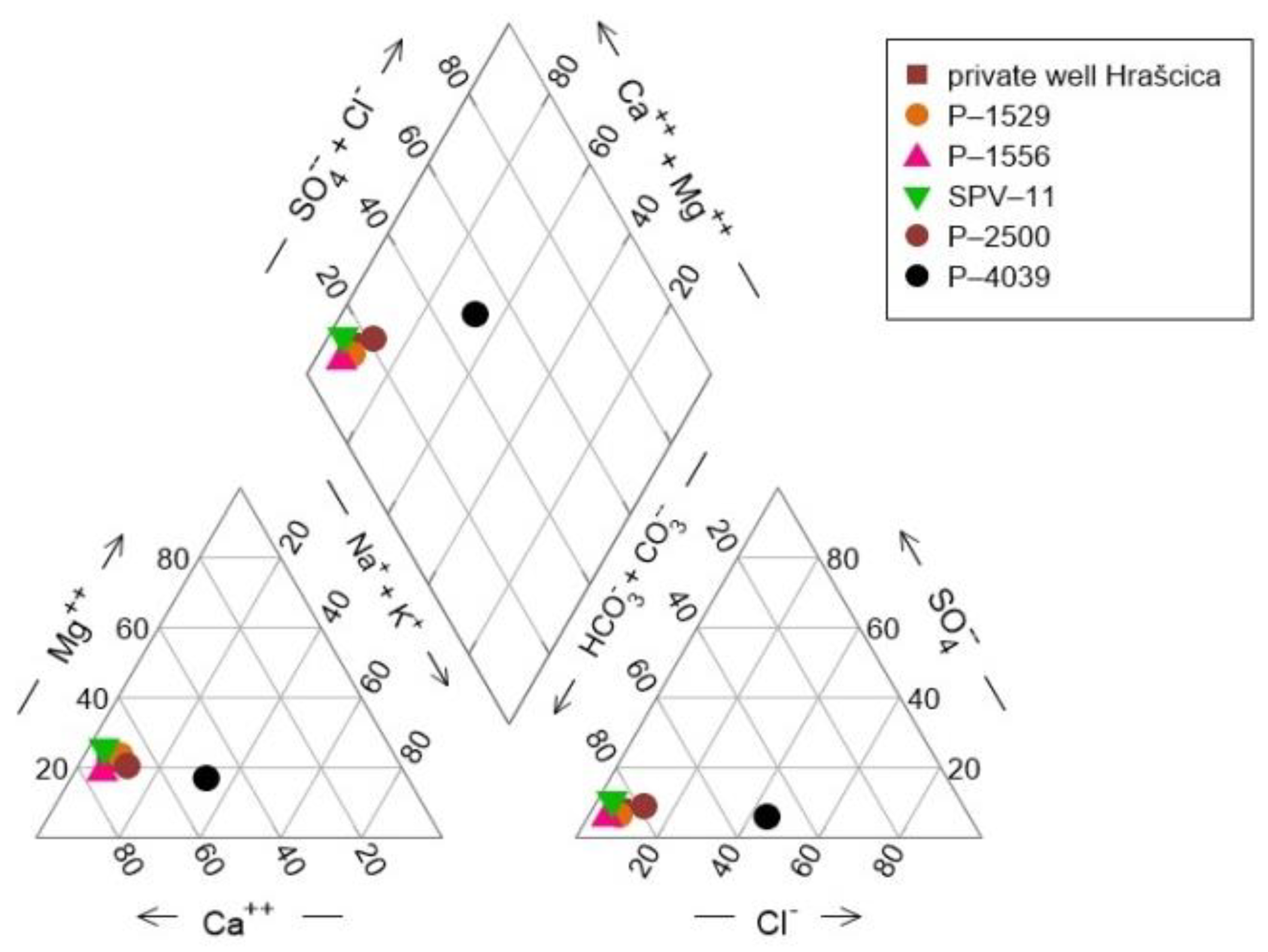
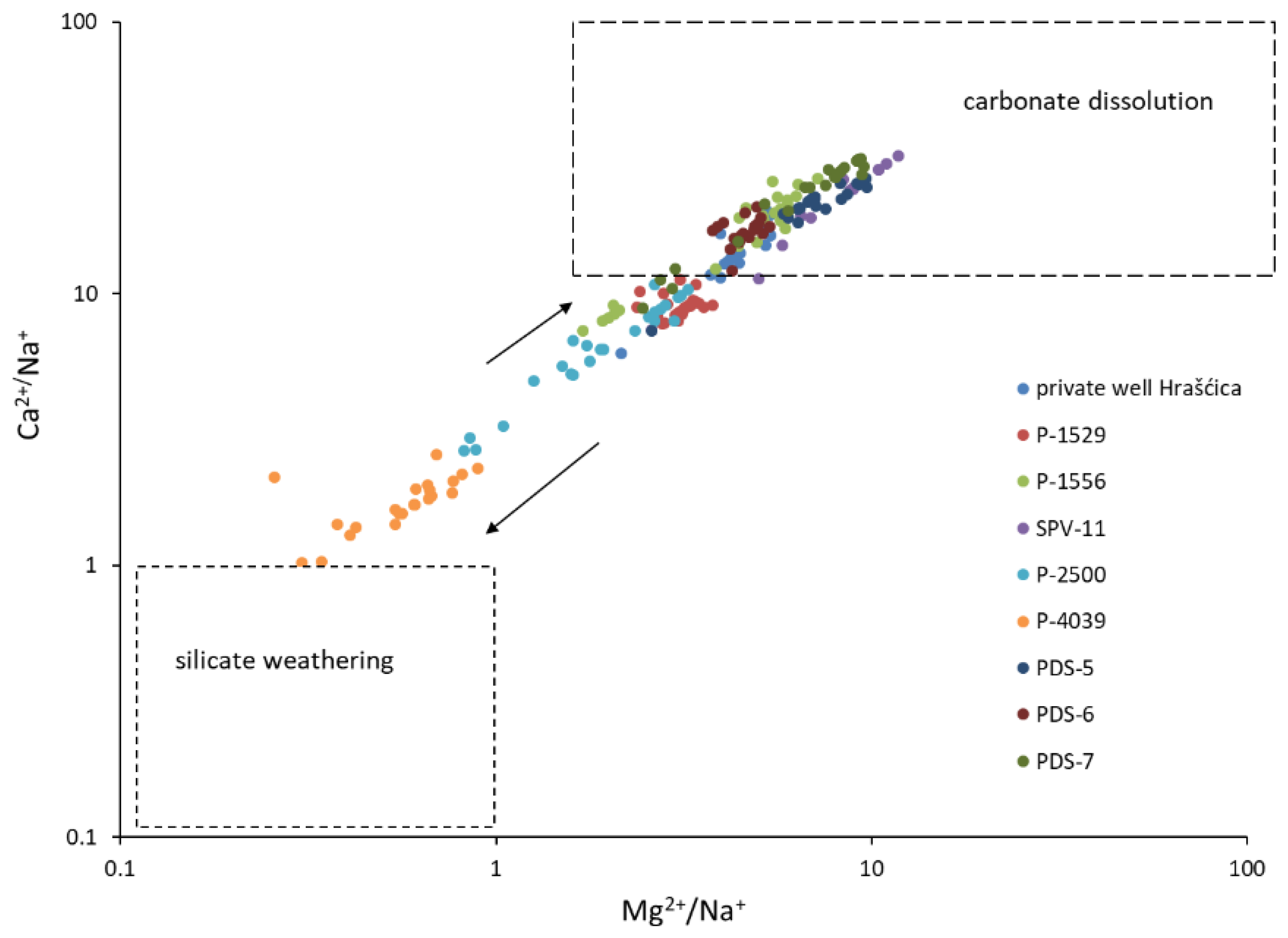
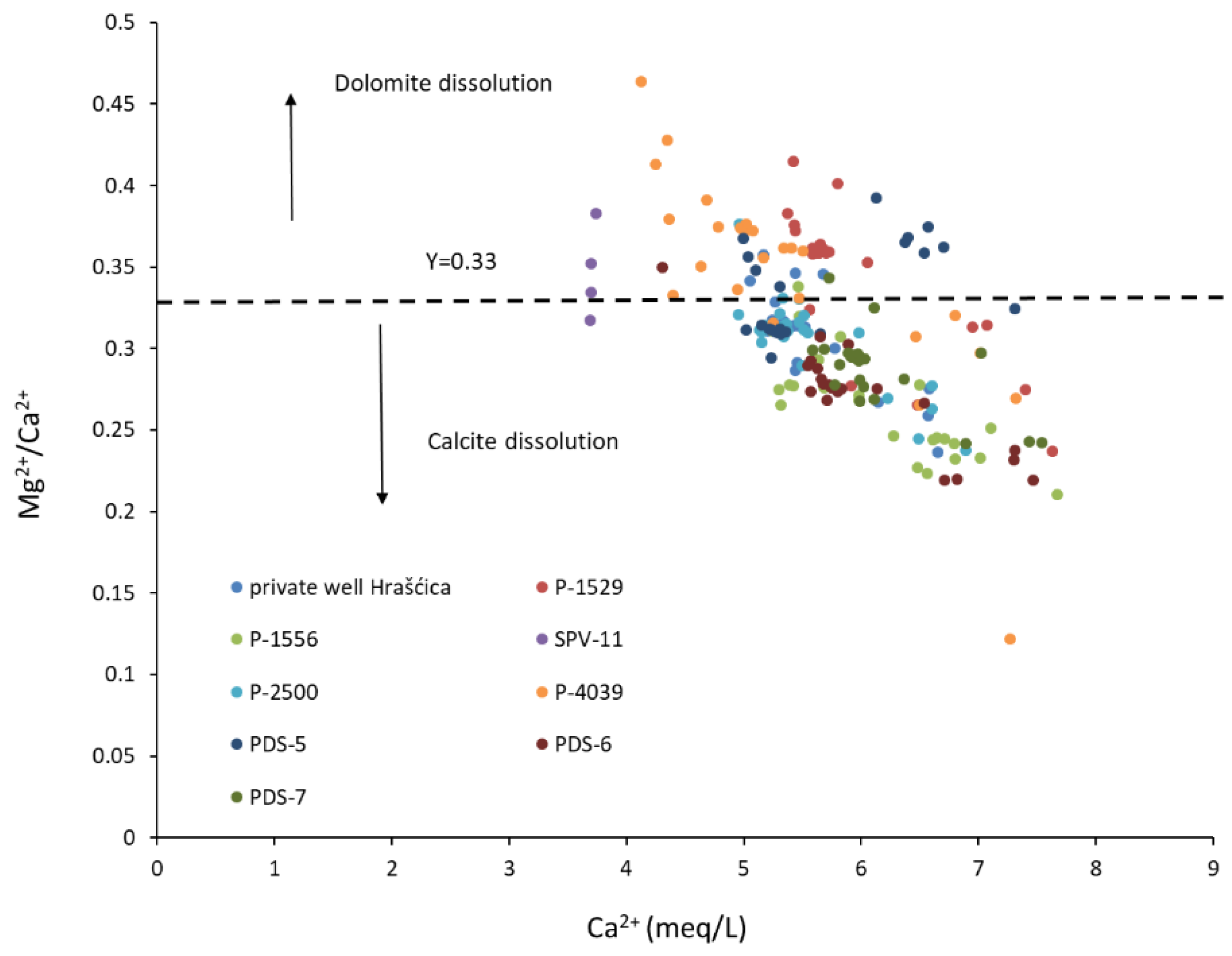
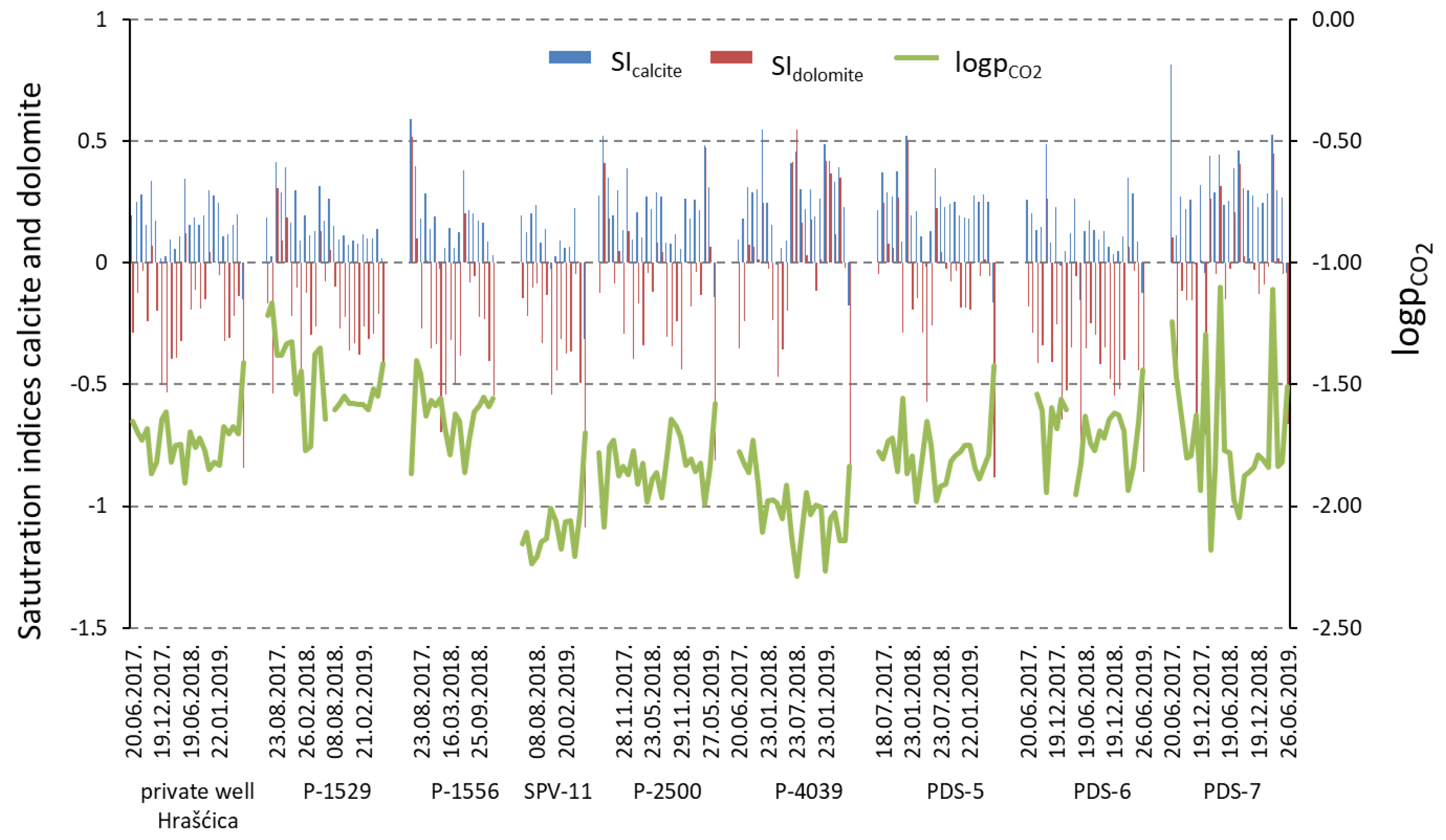
- The dissolution and precipitation of carbonate minerals represents the main mechanism controlling the groundwater chemistry. Although the aquifer is composed of carbonate and silicate minerals, carbonate dissolution is dominant against silicate weathering, due to the great difference in their weathering rates. Most of the time, sampled groundwater is saturated with respect to calcite, which enables the precipitation of calcite, and undersaturated with respect to dolomite.
- (b)
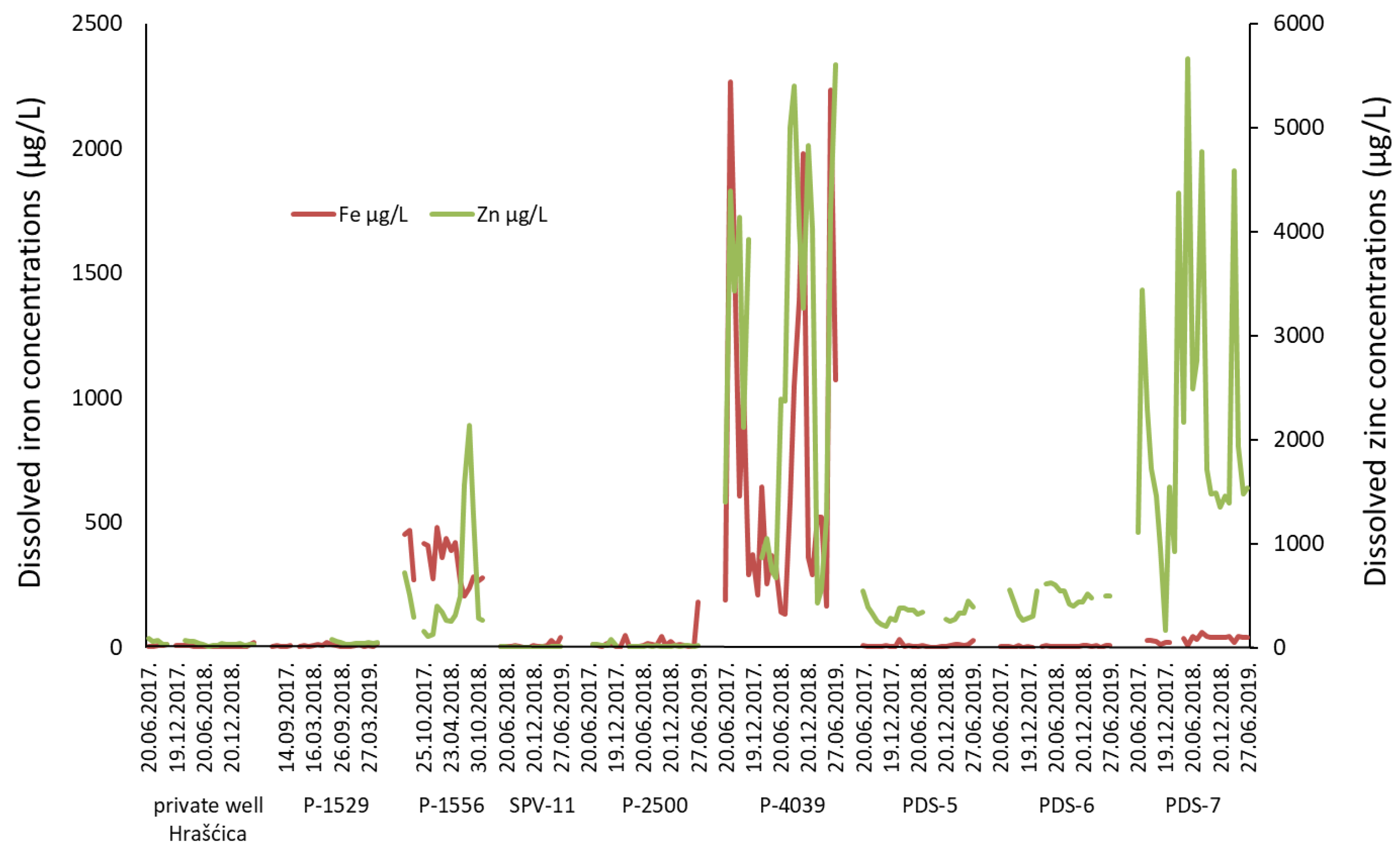
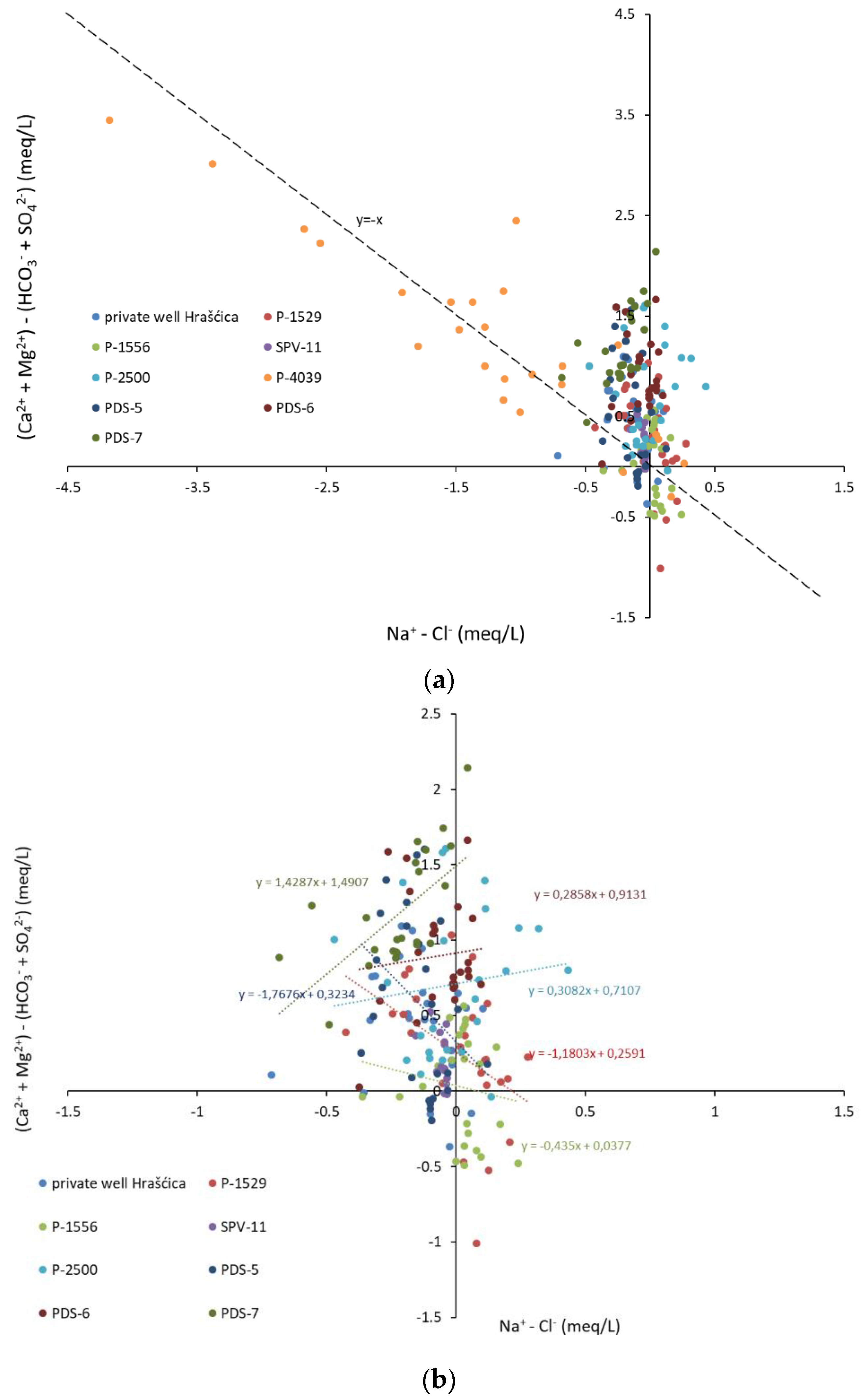
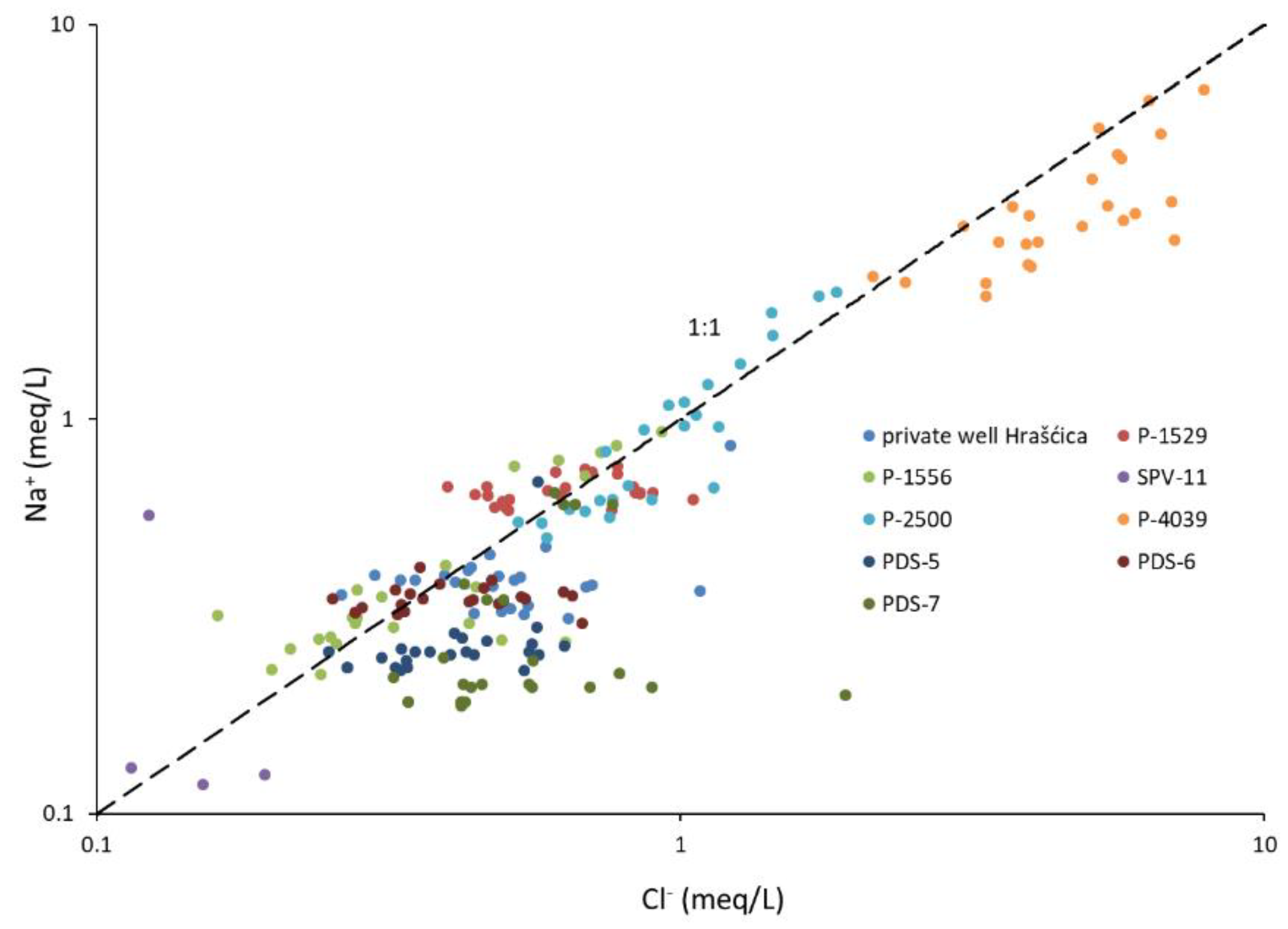
- The cation exchange process is well documented in the catchment area of well P-4039, while other observation wells do not show the signs of this process.
- (c)
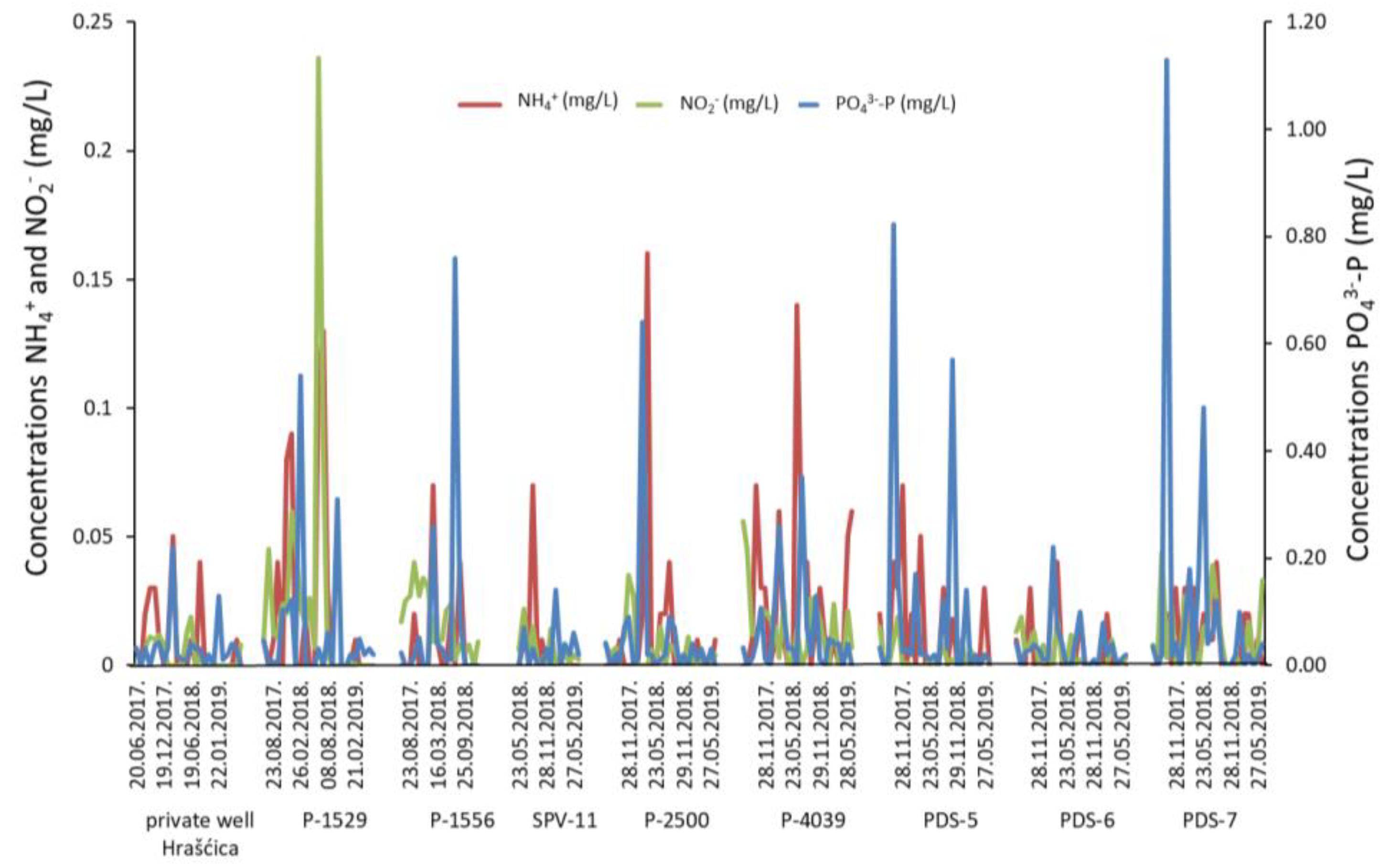
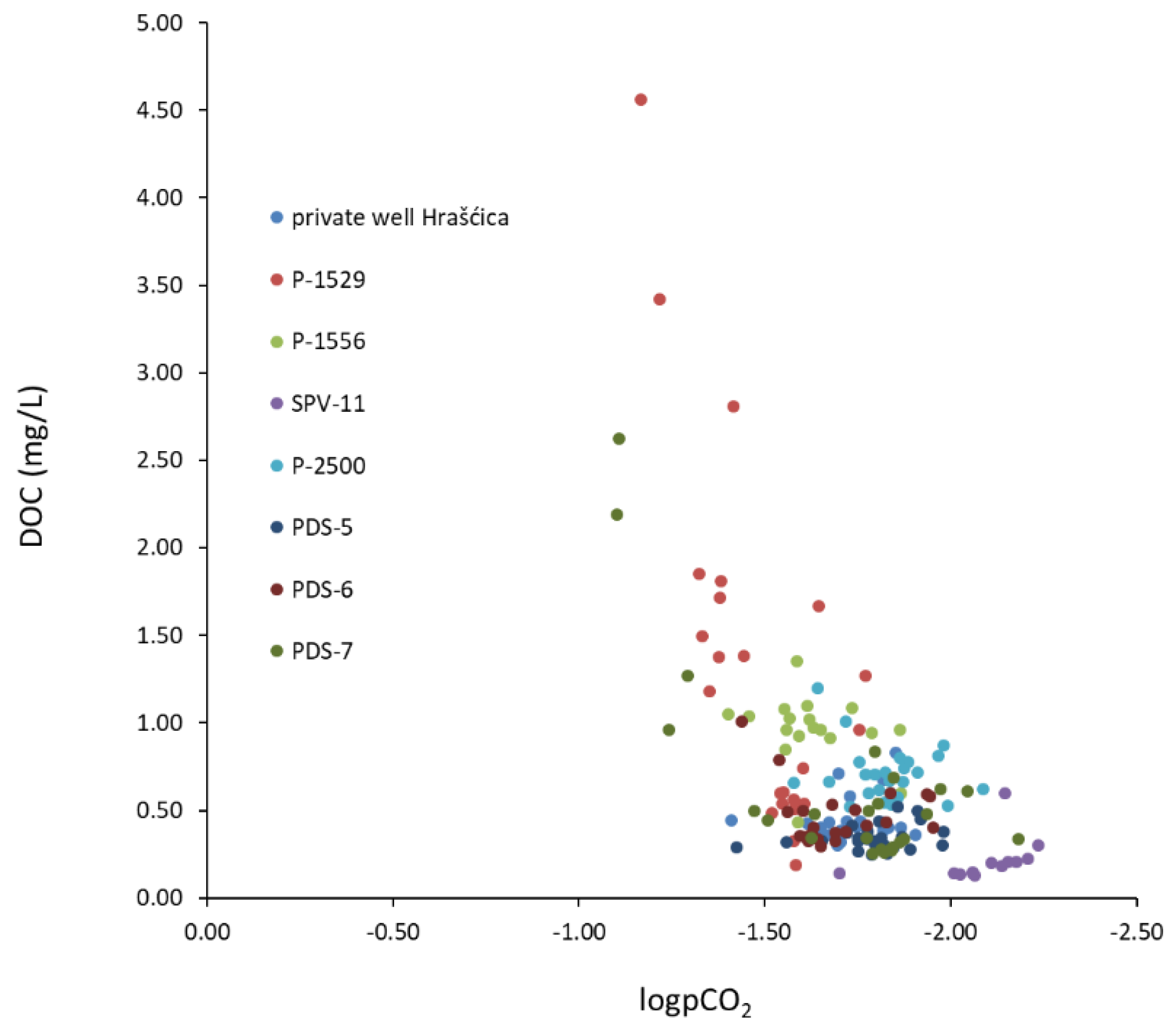
- The transformation of organic matter is observed in the catchment area of the observation wells P-1529, PDS-7, PDS-5, and PDS-6. High DOC concentrations in water are followed by high partial pressure of CO2, which is a consequence of flushing organic matter from the soil and unsaturated zone into the aquifer.
- (d)
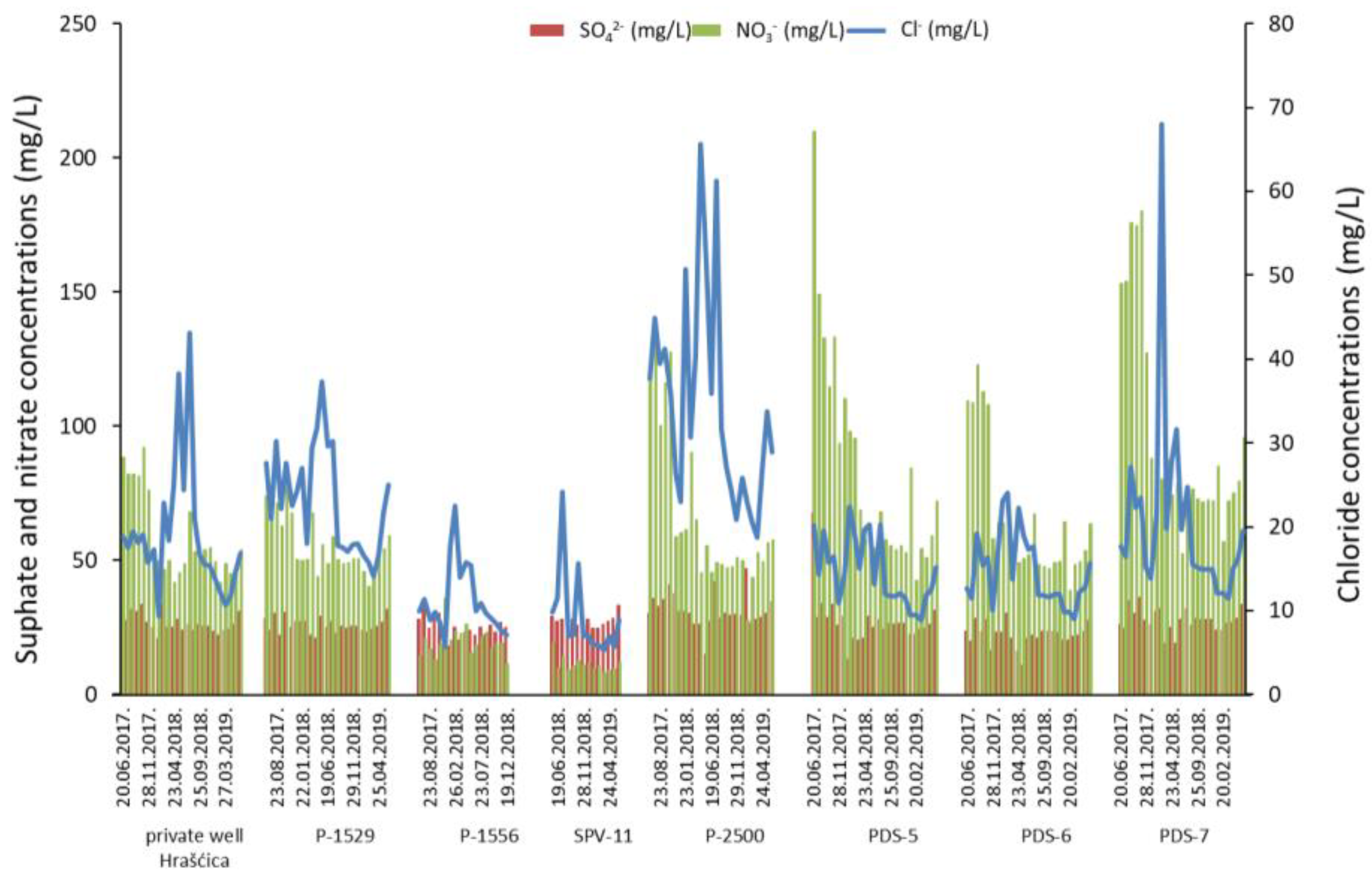
- An anthropogenic influence is recognized through high nitrate concentrations in groundwater. The application of synthetic fertilizers and manure in agricultural production is considered the main source of nitrate contamination. However, changes in land use and recent urbanization caused a more significant impact of waste water on nitrate content in the Varaždin aquifer.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; John Wiley & Sons, INC: New Jersey, NJ, USA, 1995; p. 625. [Google Scholar]
- Malcom, R.; Soulsby, C. Hydrogeochemistry of groundwater in coastal wetlands: Implications for coastal conservation in Scotland. Sci. Total. Environ. 2001, 265, 269–280. [Google Scholar] [CrossRef]
- Robins, N.S. Groundwater quality in Scotland: Major ion chemistry of the key groundwater bodies. Sci. Total. Environ. 2002, 294, 41–56. [Google Scholar] [CrossRef] [Green Version]
- Edmunds, W.M.; Shand, P.; Hart, P.; Ward, R.S. The natural (baseline) quality of groundwater: A UK pilot study. Sci. Total. Environ. 2003, 310, 25–35. [Google Scholar] [CrossRef] [Green Version]
- Rosen, M.; Jones, S. Controls on the chemical composition of groundwater from alluvial aquifers in the Wanaka and Wakatipu basins, Central Otago, New Zealand. Hydrogeol. J. 2007, 6, 264–281. [Google Scholar]
- Marković, T.; Miko, S.; Kapelj, S.; Buljan, R.; Larva, O.; Peh, Z. Behaviour of metals and nutrients in soils and groundwater of a karst polje. J. Geochem. Explor. 2006, 88, 124–129. [Google Scholar] [CrossRef]
- Marković, T.; Brkić, Ž.; Larva, O. Using hydrochemical data and modelling to enhance the knowledge of groundwater flow and quality in an alluvial aquifer of Zagreb, Croatia. Sci. Total. Environ. 2013, 458–460, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Marković, T.; Borović, S.; Larva, O. Geochemical characteristics of thermal waters of Hrvatsko zagorje. Geol. Croat. 2015, 68, 67–77. [Google Scholar] [CrossRef]
- Marković, T.; Karlović, I.; Perčec Tadić, M.; Larva, O. Application of Stable Water Isotopes to Improve Conceptual Model of Alluvial Aquifer in the Varaždin Area. Water 2020, 12, 379. [Google Scholar] [CrossRef] [Green Version]
- Brkić, Ž.; Briški, M.; Marković, T. Use of hydrochemistry and isotopes for improving the knowledge of groundwater flow in a semiconfined aquifer system of the Eastern Slavonia (Croatia). Catena 2016, 142, 153–165. [Google Scholar] [CrossRef]
- Pandey, S.K.; Singh, A.K.; Hasnain, S.I. Hydrochemical characteristics of meltwater draining from Pindari glacier, Kumon Himalaya. J. Geol. Soc. India 2001, 57, 519–527. [Google Scholar]
- McGinleya, P.M.; Masarika, K.C.; Gotkowitzb, M.B.; Mechenicha, D.J. Impact of anthropogenic geochemical change and aquifer geology on groundwater phosphorus concentrations. Appl. Geochem. 2016, 72, 1–9. [Google Scholar] [CrossRef]
- Sajil Kumar, P.J. Hydrogeochemical and multivariate statistical appraisal of pollution sources in the groundwater of the lower Bhavani River basin in Tamil Nadu. GeologyEcolog. Landsc. 2020, 4, 40–51. [Google Scholar] [CrossRef] [Green Version]
- Hallburg, G.R.; Keeney, D.R. Nitrate. In Regional Groundwater Quality; Alley, W.M., Ed.; Van Norstrand Reinhold: New York, NY, USA, 1993; pp. 297–322. [Google Scholar]
- Spalding, R.F.; Exner, M.E. Occurence of nitrate in groundwater: A review. J. Environ. Qual. 1993, 22, 392–402. [Google Scholar] [CrossRef]
- Nolan, B.T. Relating nitrogen sources and aquifer susceptibility of nitrate in shallow ground waters of the United States. Ground Water 2001, 39, 290–299. [Google Scholar] [CrossRef]
- Walton, B. Survey of literature relating infant methemoglobinemia due to nitrate contaminated water. Am. J. Public Health 1951, 41, 986–996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winneberger, J.H.T. Nitrogen, Public Health and the Environment; Ann Arbor Science Publishers Inc.: Ann Arbor, MI, USA, 1982. [Google Scholar]
- World Health Organization. Health Hazards from Nitrates in Drinking Water, Report on a WHO Meeting in Copenhagen, Denmark, 5–9 March 1984; Regional Office for Europe: Copenhagen, Denmark, 1985; Volume 3, pp. 49–66. [Google Scholar]
- Zaninović, K.; Gajić-Čapka, M.; Perčec Tadić, M. Klimatski Atlas Hrvatske. Climate Atlas of Croatia: 1961–1990, 1971–2000; Državni hidrometeorološki zavod: Zagreb, Croatia, 2008; p. 200. [Google Scholar]
- Nimac, I.; Perčec Tadić, M. New 1981–2010 Climatological Normals for Croatia and Comparison to Previous 1961–1990 and 1971–2000 Normals. In Proceedings of the GeoMLA Conference, Belgrade, Serbia, 21–24 June 2016; pp. 79–85. [Google Scholar]
- Babić, Ž.; Čakarun, I.; Sokač, A.; Mraz, V. On geological features of quaternary sediments of Drava basin on Croatian territory. Geol. Vjesn. 1978, 30, 43–61. [Google Scholar]
- Urumović, K. On quaternary aquifer complex in Varaždin area. Geol. Vjesn. 1971, 24, 183–191. [Google Scholar]
- Urumović, K.; Hlevnjak, B.; Prelogović, E.; Mayer, D. Hydrogeological conditions of Varaždin aquifer. Geol. Vjesn. 1990, 43, 149–158. [Google Scholar]
- Prelogović, E.; Velić, I. Quaternary tectonic activity in western part of Drava basin. Geol. Vjesn. 1988, 41, 237–253. [Google Scholar]
- Gazarek, M.; Crnički, J.; Premur, V.; Kreč, D. Granulometrijski sastav šljunka i pijesaka i teški minerali u pijescima dravskog bazena od Ormoža do Đurđevca. Rudarsko Geološko Naftni Zbornik 1990, 2, 67–73. [Google Scholar]
- Crnički, J. Mineralne Sirovine Varaždinske Regije i Njihovo Privredno Značenje. Varaždinski Zbornik 1181–1981, Varaždin, Croatia, 1–3 October 1981; Mohorovičić, A., Ed.; Jugoslavenska akademija znanosti i umjetnosti: Zagreb, Hrvatska, 1983. [Google Scholar]
- Mioč, P.; Marković, S. Osnovna Geološka Karta SFRJ 1:100.000. In List Čakovec L33–57 (Basic Geological Map of SFRY, Scale 1:100.000. Sheet Čakovec L33–57—in Croatian); Geological Survey, Zagreb and Geological, Geotechnical and Geophysical Survey: Ljubljana, Slovenia, 1998. [Google Scholar]
- Mioč, P.; Marković, S. Osnovna Geološka Karta SFRJ 1:100.000. In Tumač za list Čakovec L33–57 (Basic Geological Map of SFRY, Scale 1:100.000. The Map Book for Sheet Čakovec L33–57—in Croatian); Geological Survey, Zagreb and Geological, Geotechnical and Geophysical Survey: Ljubljana, Slovenia, 1998. [Google Scholar]
- Karlović, I.; Marković, T.; Vujnović, T.; Larva, O. Development of a Hydrogeological Conceptual Model of the Varaždin Alluvial Aquifer. Hydrology 2021, 8, 19. [Google Scholar] [CrossRef]
- Mandel, S.; Shiftan, Z.L. Groundwater Resources: Investigation and Development; Academic Press: New York, NY, USA, 1981; p. 269. [Google Scholar]
- Domenico, P.A.; Schwartz, F.W. Physical and Chemical Hydrogeology; John Willey and Sons: New York, NY, USA, 1990; p. 824. [Google Scholar]
- HRN EN ISO 17294-2:2016. Determination of Selected Elements Including Uranium Isotopes. Available online: https://repozitorij.hzn.hr/norm/HRN+EN+ISO+17294-2%3A2016 (accessed on 21 April 2021).
- Parkhurst, D.L.; Appelo, C.A.J. Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; U.S. Geological Survey Techniques and Methods, Book 6; USA; 2013; Chapter A43; p. 497. Available online: https://pubs.usgs.gov/tm/06/a43/ (accessed on 21 April 2021).
- McMahon, P.B.; Chapelle, F.H. Redox processes and water quality of selected principal aquifer systems. Ground Water 2008, 46, 259–271. [Google Scholar] [CrossRef]
- OG 125/17. Rulebook on Compliance Parameters, Methods of Analysis, Monitoring and Water Safety Plans for Human Consumption. Available online: https://narodne-novine.nn.hr/clanci/sluzbeni/2017_12_125_2848.html (accessed on 21 April 2021).
- Meybeck, M. Global chemical weathering from surficial rocks estimated from river dissolved loads. Am. J. Sci. 1987, 287, 401–428. [Google Scholar] [CrossRef]
- Faure, G. Principles and Applications of Geochemistry, 2nd ed.; Prentice Hall: New Jersey, NJ, USA, 1998; p. 600. [Google Scholar]
- Mukherjee, A.; Bhattacharya, P.; Shi, F.; Fryar, A.E.; Mukherjee, A.B.; Xie, Z.M.; Jacks, G.; Bundschuh, J. Chemical evolution in the high arsenic groundwater of the Huhhot basin (Inner Mongolia, PR China) and its difference from the western Bengal basin (India). Appl. Geochem. 2009, 24, 1835–1851. [Google Scholar] [CrossRef]
- McLean, W.; Jankowski, J.; Lavitt, N. Groundwater quality and sustainability in an alluvial aquifer, Australia. In Groundwater, Past Achievements and Future Challenges, Proceedings of the XXX IAH congress, Cape Town, South Africa, 26 November–1 December 2000; Balkema: Rotterdam, The Nederland, 2000; pp. 567–573. [Google Scholar]
- Jankowski, J.; Acworth, R.I.; Shekarforoush, S. Reverse ion exchange in deeply weathered porphyritic dacite fractured aquifer system, Yass, New South Wales, Australia. In Proceedings of the 9th International Symposium on Water-Rock Interaction, Taupo, New Zealand, 30 March–3 April 1998; pp. 243–246. [Google Scholar]
- Deutsch, W.J. Groundwater Geochemistry: Fundamentals and Application to Contamination, 1st ed.; CRC Press: Boca Raton, FL, USA, 1997; p. 232. [Google Scholar]
- Macpherson, G.L.; Roberts, J.A.; Blair, J.M.; Townsend, M.A.; Fowle, D.A.; Beisner, K.R. Increasing shallow groundwater CO2 and limestone weathering, Konza Prairie, USA. Geochim. Cosmochim. Acta 2008, 72, 5581–5599. [Google Scholar] [CrossRef]
- Keeney, D. Sources of Nitrate to Ground Water. Crit. Rev. Environ. Contr. 1986, 32, 1039–1050. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Groffman, P.M.; Brand, L.E.; Elliott, E.M.; Shields, C.A.; Kendall, C. Tracking Nonpoint Source Nitrogen Pollution in Human-Impacted Watersheds. Environ. Sci. Technol. 2011, 45, 8225–8232. [Google Scholar] [CrossRef]
- Jung, H.; Koh, D.-C.; Kim, Y.S.; Jeen, S.-W.; Lee, J. Stable Isotopes of Water and Nitrate for the Identification of Groundwater Flowpaths: A Review. Water 2020, 12, 138. [Google Scholar] [CrossRef] [Green Version]
- Jogun, K.; Pavlek, K.; Belić, T.; Buhin, S.; Malešić, N. Land cover changes in northern Croatia from 1981 to 2011. Hrvat. Geogr. Glas. 2017, 79, 33–59. [Google Scholar] [CrossRef] [Green Version]
- Kacaroglu, F.; Gunay, G. Impacts of Human Activities on Groundwater Quality of an Alluvial Aquifer: A Case Study of the Eskisehir plain, Turkey. Hydrogeol. J. 1997, 5, 60–70. [Google Scholar] [CrossRef]
- Fernando, W.A.M.; Ilankoon, I.; Syed, T.H.; Yellishetty, M. Challenges and opportunities in the removal of sulphate ions in contaminated mine water: A review. Miner. Eng. 2018, 117, 74–90. [Google Scholar] [CrossRef]
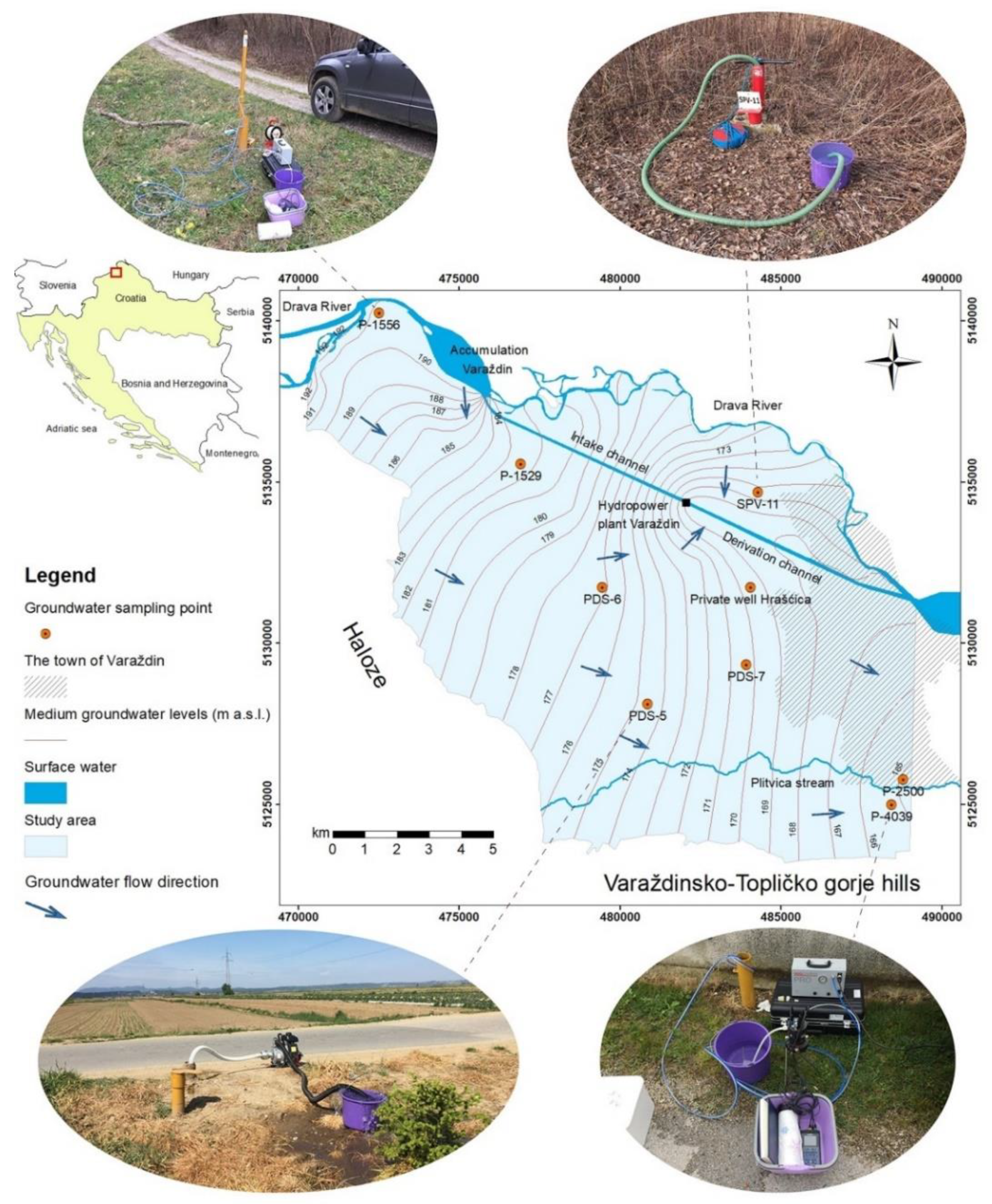
| Observation Well | Elevation (m a.s.l.) | Depth of the Well (m) | Depth Interval of the Screen (m) |
|---|---|---|---|
| Private well Hrašćica | 176.00 | 15.0 | 5–15 |
| PDS-5 | 178.36 | 31.0 | 13.7–19.7 |
| PDS-6 | 184.07 | 25.0 | 11.7–17.7 |
| PDS-7 | 175.71 | 42.5 | 29.3–32.3 |
| P-1529 | 187.32 | 8.0 | n.a. 1 |
| P-1556 | 193.03 | 15.6 | n.a. 1 |
| P-2500 | 167.81 | 5.20 | n.a. 1 |
| P-4039 | 167.76 | 8.0 | n.a. 1 |
| SPV-11 | 177.69 | 40.0 | 24.5–35.8 |
| EC (µS/cm) | T (°C) | pH | O2 (mg/L) | HCO3− (mg/L) | PO43−-P (mg/L) | NH4+ (mg/L) | NO2− (mg/L) | Cl− (mg/L) | SO42− (mg/L) | NO3− (mg/L) | TN (mg/L) | Br− (mg/L) | Ca2+ (mg/L) | Mg2+ (mg/L) | Na+ (mg/L) | K+ (mg/L) | DOC (mg/L) | SiO2 (mg/L) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Private well | min | 673 | 10.8 | 6.91 | 3.1 | 342 | <0.01 | <0.01 | <0.01 | 9.3 | 21.0 | 41.8 | 10.6 | <0.10 | 101 | 18.9 | 7.2 | 3.9 | 0.30 | 11.4 |
| max | 747 | 13.8 | 7.40 | 8.5 | 414 | 0.22 | 0.05 | 0.02 | 43.1 | 34.0 | 91.9 | 20.9 | 4.0 | 133 | 23.8 | 19.7 | 5.3 | 0.83 | 14.4 | |
| average | 713 | 13.1 | 7.22 | 6.7 | 382 | 0.04 | 0.02 | 0.01 | 19.0 | 26.6 | 58.4 | 14.4 | 2.5 | 111 | 20.7 | 9.2 | 4.5 | 0.45 | 12.5 | |
| P-1529 | min | 755 | 10.3 | 6.86 | 1.5 | 388 | <0.01 | <0.01 | <0.01 | 14.1 | 21.0 | 40.1 | 11.2 | <0.10 | 108 | 19.9 | 13.5 | 5.1 | 0.19 | 10.1 |
| max | 814 | 14.5 | 7.36 | 9.2 | 512 | 0.54 | 0.13 | 0.24 | 37.3 | 31.8 | 77.1 | 17.5 | 4.0 | 153 | 28.3 | 17.5 | 12.5 | 4.6 | 18.4 | |
| average | 787 | 12.6 | 7.15 | 6.1 | 437 | 0.10 | 0.05 | 0.04 | 23.1 | 25.8 | 55.7 | 14.0 | 2.9 | 119 | 24.6 | 15.1 | 6.1 | 1.3 | 12.7 | |
| P-1556 | min | 658 | 9.40 | 6.93 | 0.6 | 381 | <0.01 | <0.01 | <0.01 | 5.7 | 9.4 | 5.3 | 2.2 | <0.10 | 105 | 17.2 | 5.2 | 4.2 | 0.43 | 9.2 |
| max | 877 | 16.0 | 7.45 | 7.8 | 512 | 0.76 | 0.07 | 0.04 | 32.9 | 34.8 | 35.6 | 6.7 | <0.10 | 154 | 22.5 | 21.3 | 10.1 | 5.4 | 15.1 | |
| average | 743 | 13.1 | 7.15 | 4.9 | 442 | 0.10 | 0.03 | 0.02 | 14.7 | 23.8 | 17.4 | 4.4 | <0.10 | 123 | 19.6 | 9.9 | 5.8 | 1.2 | 11.8 | |
| SPV-11 | min | 490 | 12.0 | 7.03 | 1.4 | 249 | <0.01 | <0.01 | <0.01 | 5.3 | 24.4 | 8.0 | 2.2 | <0.10 | 73.9 | 15.5 | 2.4 | 0.94 | 0.13 | 10.3 |
| max | 496 | 14.4 | 7.56 | 7.7 | 278 | 0.14 | 0.07 | 0.02 | 24.2 | 33.2 | 18.9 | 5.00 | <0.10 | 82.6 | 20.6 | 13.1 | 3.8 | 0.60 | 16.1 | |
| average | 494 | 12.3 | 7.43 | 2.2 | 267 | 0.05 | 0.03 | 0.01 | 9.1 | 27.3 | 11.2 | 2.7 | <0.10 | 75.8 | 16.8 | 3.9 | 1.6 | 0.59 | 12.7 | |
| P-2500 | min | 696 | 10.8 | 7.01 | 0.9 | 325 | <0.01 | <0.01 | <0.01 | 18.7 | 15.0 | 27.5 | 11.3 | <0.10 | 99.4 | 19.0 | 11.5 | 1.0 | 0.52 | 10.5 |
| max | 802 | 17.9 | 7.55 | 9.2 | 405 | 0.64 | 0.16 | 0.04 | 65.6 | 46.8 | 137 | 31.3 | 5.8 | 138 | 22.7 | 48.1 | 2.9 | 1.2 | 15.6 | |
| average | 737 | 13.7 | 7.29 | 6.5 | 361 | 0.07 | 0.04 | 0.01 | 34.6 | 31.3 | 66.4 | 15.9 | 3.6 | 112 | 20.6 | 22.3 | 1.7 | 0.71 | 12.4 | |
| P-4039 | min | 766 | 11.0 | 7.01 | 0.2 | 238 | <0.01 | <0.01 | <0.01 | 75.8 | 6.6 | <0.10 | <1.0 | <0.10 | 82.6 | 10.7 | 47.1 | 1.5 | 0.69 | 9.6 |
| max | 1091 | 14.7 | 7.71 | 4.2 | 410 | 0.35 | 0.14 | 0.06 | 279 | 40.2 | 21.9 | 2.4 | 4.2 | 147 | 26.5 | 157 | 3.7 | 2.4 | 22.0 | |
| average | 975 | 13.2 | 7.40 | 1.5 | 324 | 0.08 | 0.04 | 0.02 | 170 | 28.2 | 5.1 | 1.4 | 2.8 | 107 | 21.9 | 81.5 | 2.8 | 1.3 | 13.1 | |
| PDS-5 | min | 661 | 11.8 | 6.91 | 3.6 | 322 | <0.01 | <0.01 | <0.01 | 8.8 | 13.0 | 42.5 | 12.1 | <0.10 | 100 | 18.7 | 5.3 | 11.4 | 0.25 | 10.7 |
| max | 694 | 13.9 | 7.45 | 9.9 | 456 | 0.82 | 0.07 | 0.02 | 22.4 | 67.5 | 210 | 47.7 | 3.0 | 147 | 29.9 | 15.9 | 88.8 | 0.52 | 19.7 | |
| average | 683 | 12.6 | 7.28 | 8.3 | 387 | 0.12 | 0.03 | 0.01 | 14.7 | 27.3 | 83.0 | 20.0 | 1.5 | 113 | 22.7 | 6.2 | 31.3 | 0.35 | 13.4 | |
| PDS-6 | min | 708 | 11.4 | 6.92 | 7.3 | 260 | <0.01 | <0.01 | <0.01 | 9.0 | 11.0 | 38.5 | 11.8 | <0.10 | 86.3 | 17.9 | 7.0 | 1.5 | 0.30 | 10.8 |
| max | 744 | 13.0 | 7.45 | 9.1 | 456 | 0.22 | 0.04 | 0.02 | 24.0 | 30.0 | 123 | 27.9 | 4.0 | 150 | 21.7 | 9.7 | 3.0 | 2.5 | 29.9 | |
| average | 730 | 12.4 | 7.17 | 8.7 | 381 | 0.05 | 0.03 | 0.01 | 14.7 | 22.2 | 65.4 | 15.4 | 2.5 | 120 | 19.6 | 8.0 | 2.2 | 0.61 | 14.4 | |
| PDS-7 | min | 730 | 11.2 | 6.99 | 6.6 | 280 | <0.01 | <0.01 | <0.01 | 11.4 | 19.0 | 52.5 | 12.6 | <0.10 | 112 | 19.5 | 4.3 | 0.40 | 0.25 | 10.9 |
| max | 777 | 13.0 | 7.76 | 10.9 | 435 | 1.1 | 0.04 | 0.04 | 68.0 | 35.9 | 180 | 41.0 | 4.0 | 151 | 25.4 | 15.0 | 8.9 | 2.6 | 23.5 | |
| average | 758 | 12.3 | 7.32 | 8.9 | 374 | 0.14 | 0.02 | 0.02 | 20.4 | 27.7 | 96.7 | 23.2 | 3.0 | 123 | 21.4 | 6.7 | 1.9 | 0.64 | 15.1 |
| As (µg/L) | Cd (µg/L) | Cr (µg/L) | Cu (µg/L) | Fe (µg/L) | Li (µg/L) | Mn (µg/L) | Mo (µg/L) | Ni (µg/L) | Pb (µg/L) | Sr (µg/L) | Zn (µg/L) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Private well | min | 0.11 | <0.01 | 0.28 | 0.49 | 1.1 | 0.68 | 0.08 | 0.51 | <0.02 | 0.06 | 135 | 13.2 |
| max | 0.44 | 0.06 | 0.57 | 7.3 | 19.8 | 3.9 | 1.7 | 0.99 | 1.1 | 0.46 | 211 | 88.4 | |
| average | 0.18 | 0.02 | 0.43 | 1.40 | 6.6 | 2.1 | 0.54 | 0.77 | 0.51 | 0.20 | 181 | 40.2 | |
| P-1529 | min | 0.07 | <0.01 | 0.25 | 0.55 | 2.1 | 1.2 | 0.06 | 0.36 | 0.22 | 0.11 | 171 | 28.0 |
| max | 0.36 | 0.10 | 0.89 | 6.1 | 21.4 | 4.5 | 9.8 | 0.64 | 1.1 | 1.7 | 267 | 78.0 | |
| average | 0.16 | 0.05 | 0.45 | 2.3 | 7.3 | 2.7 | 2.7 | 0.51 | 0.52 | 0.53 | 235 | 45.9 | |
| P-1556 | min | 0.34 | <0.01 | <0.02 | 0.30 | 208 | 1.2 | 0.29 | 0.33 | 0.05 | 0.10 | 242 | 104 |
| max | 1.1 | 0.05 | 0.26 | 3.5 | 483 | 3.6 | 237 | 0.71 | 3.2 | 0.60 | 371 | 2134 | |
| average | 0.81 | 0.03 | 0.08 | 0.96 | 349 | 2.7 | 158 | 0.52 | 1.1 | 0.27 | 303 | 557 | |
| SPV-11 | min | 0.10 | <0.01 | 0.06 | 0.07 | 1.7 | <0.01 | <0.05 | 0.53 | 0.12 | 0.05 | 243 | 3.5 |
| max | 0.39 | 0.06 | 0.46 | 1.3 | 41.8 | 1.1 | 0.98 | 0.95 | 5.3 | 0.41 | 327 | 13.5 | |
| average | 0.19 | 0.02 | 0.18 | 0.57 | 9.3 | 0.69 | 0.42 | 0.78 | 0.69 | 0.17 | 296 | 6.2 | |
| P-2500 | min | 0.09 | <0.01 | 0.03 | 0.15 | 1.8 | 0.14 | 0.27 | 0.12 | 0.11 | 0.05 | 126 | 7.6 |
| max | 0.30 | 0.08 | 0.42 | 2.5 | 184 | 2.1 | 8.8 | 0.34 | 4.6 | 0.76 | 212 | 76.3 | |
| average | 0.15 | 0.02 | 0.13 | 1.0 | 19.4 | 1.3 | 1.6 | 0.21 | 0.61 | 0.19 | 172 | 18.6 | |
| P-4039 | min | 0.12 | 0.01 | <0.02 | 0.13 | 135 | 0.95 | 11.1 | 0.28 | 0.06 | 0.11 | 146 | 427 |
| max | 0.85 | 0.09 | 0.44 | 4.1 | 2265 | 2.9 | 30.5 | 0.94 | 46.7 | 4.6 | 251 | 5600 | |
| average | 0.27 | 0.03 | 0.11 | 1.1 | 744 | 1.8 | 19.8 | 0.52 | 2.6 | 1.2 | 205 | 2873 | |
| PDS-5 | min | 0.11 | <0.01 | 0.37 | 0.11 | 1.5 | 0.10 | 0.29 | 0.19 | <0.02 | 0.06 | 143 | 205 |
| max | 0.39 | 0.05 | 0.63 | 6.7 | 34.0 | 2.9 | 1.6 | 0.34 | 1.7 | 0.60 | 223 | 543 | |
| average | 0.17 | 0.02 | 0.51 | 0.69 | 8.2 | 1.6 | 0.68 | 0.26 | 0.32 | 0.19 | 188 | 330 | |
| PDS-6 | min | 0.08 | <0.01 | 0.27 | 0.15 | 2.0 | 0.89 | 0.25 | 0.16 | <0.02 | 0.41 | 192 | 265 |
| max | 0.35 | 0.11 | 0.66 | 3.2 | 9.6 | 4.2 | 4.5 | 0.71 | 0.84 | 1.7 | 309 | 627 | |
| average | 0.14 | 0.03 | 0.47 | 0.88 | 4.9 | 2.6 | 1.1 | 0.25 | 0.34 | 0.76 | 260 | 466 | |
| PDS-7 | min | 0.05 | <0.01 | 0.15 | 0.02 | 10.2 | 0.31 | 2.2 | 0.29 | 0.04 | 0.01 | 116 | 169 |
| max | 0.33 | 0.09 | 0.82 | 1.6 | 59.3 | 2.7 | 16.5 | 0.58 | 1.3 | 1.5 | 268 | 5665 | |
| average | 0.11 | 0.04 | 0.49 | 0.72 | 34.1 | 1.6 | 7.8 | 0.44 | 0.44 | 0.48 | 227 | 2169 |
| Observation Well | General Redox Category | Redox Process |
|---|---|---|
| Private well Hrašćica | Oxic | O2 |
| P-1529 | Oxic | O2 |
| P-1556 | Mixed (oxic-anoxic) | O2-Fe(III)/SO4 or O2-Mn(IV) |
| SPV-11 | Oxic | O2 |
| P-2500 | Oxic | O2 |
| P-4039 | Anoxic or Mixed (oxic-anoxic) | NO3-Fe(III)/SO4 or O2-Fe(III)/SO4 |
| PDS-5 | Oxic | O2 |
| PDS-6 | Oxic | O2 |
| PDS-7 | Oxic | O2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karlović, I.; Marković, T.; Šparica Miko, M.; Maldini, K. Geochemical Characteristics of Alluvial Aquifer in the Varaždin Region. Water 2021, 13, 1508. https://doi.org/10.3390/w13111508
Karlović I, Marković T, Šparica Miko M, Maldini K. Geochemical Characteristics of Alluvial Aquifer in the Varaždin Region. Water. 2021; 13(11):1508. https://doi.org/10.3390/w13111508
Chicago/Turabian StyleKarlović, Igor, Tamara Marković, Martina Šparica Miko, and Krešimir Maldini. 2021. "Geochemical Characteristics of Alluvial Aquifer in the Varaždin Region" Water 13, no. 11: 1508. https://doi.org/10.3390/w13111508
APA StyleKarlović, I., Marković, T., Šparica Miko, M., & Maldini, K. (2021). Geochemical Characteristics of Alluvial Aquifer in the Varaždin Region. Water, 13(11), 1508. https://doi.org/10.3390/w13111508







