Enhanced Adsorptive Removal of Dyes Using Mandarin Peel Biochars via Chemical Activation with NH4Cl and ZnCl2
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Preparation of Mandarin Peel Biochars
2.3. Characterization of Mandarin Peel Biochars
2.4. Batch Adsorption Experiments
2.4.1. Optimal Adsorbent Dosages
2.4.2. Adsorption Kinetics Experiments
2.4.3. Adsorption Isotherm Experiments
2.4.4. Influence of Solution Temperature and pH on Adsorption of Dyes
3. Results and Discussions
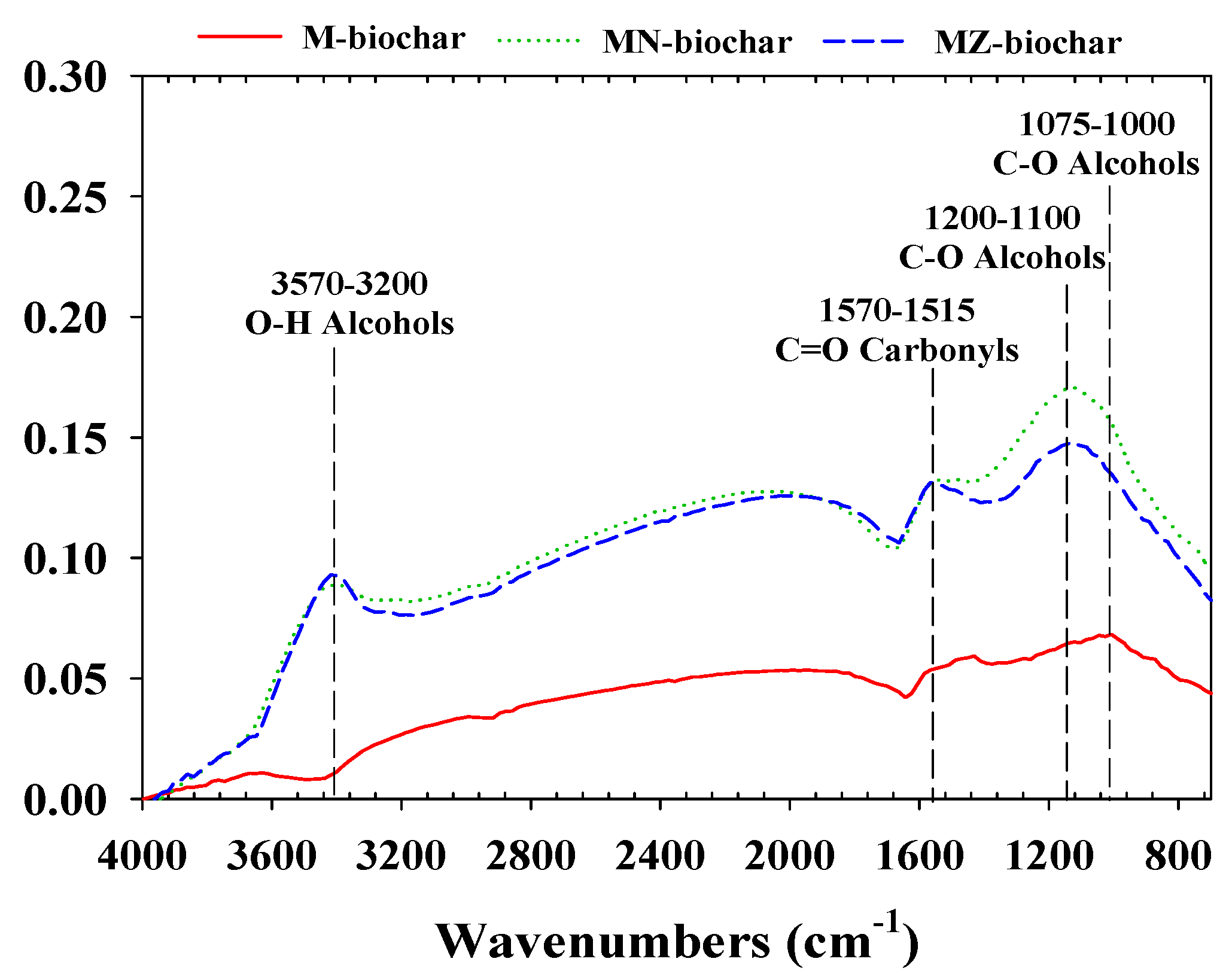
3.1. Physicochemical Characteristics of Mandarin Peel Biochars
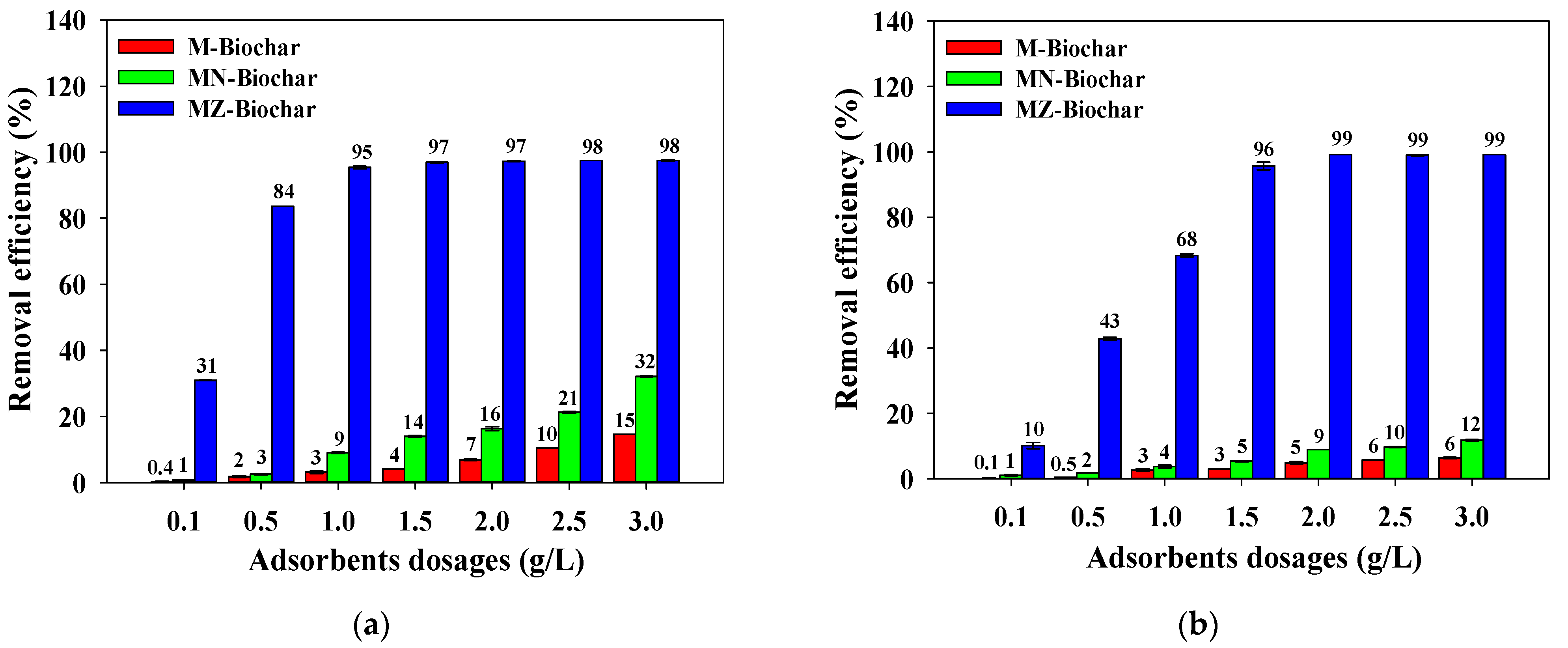
3.2. Influence of Mandarin Peel Biochar Dosage
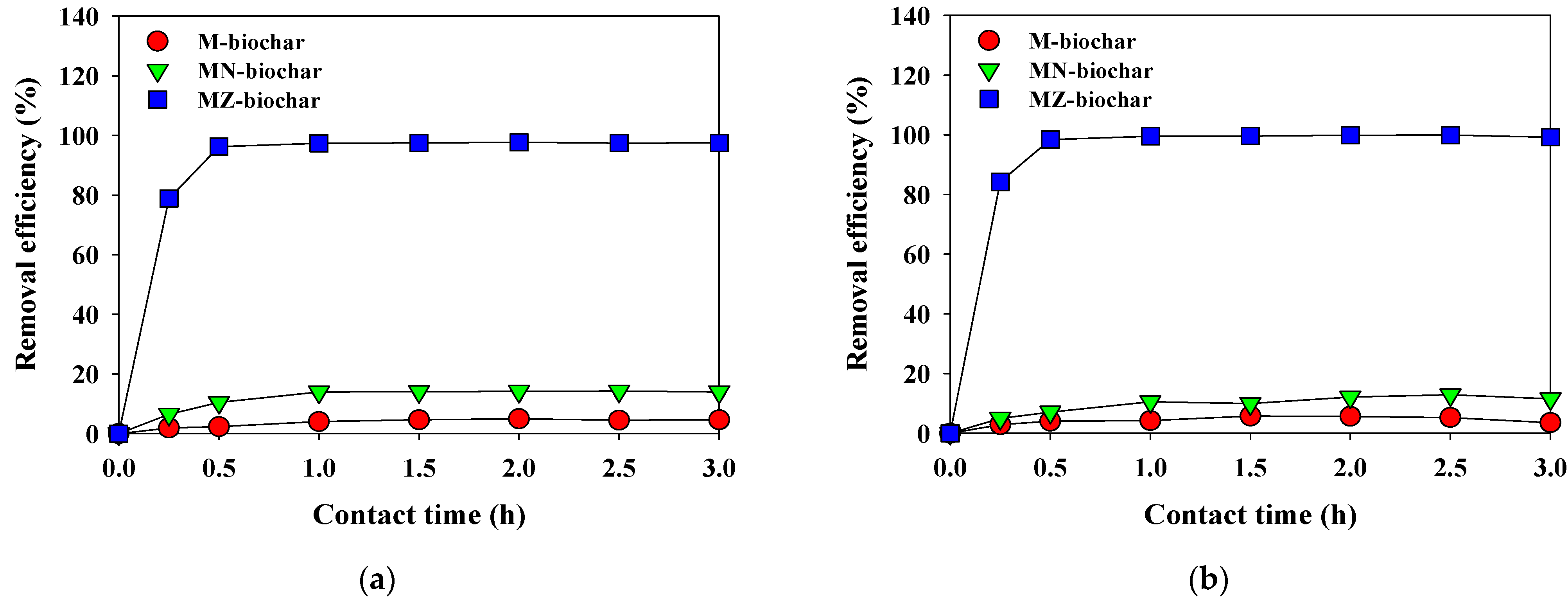
3.3. Adsorption Kinetics of Dyes
3.4. Adsorption Isotherms of Dyes
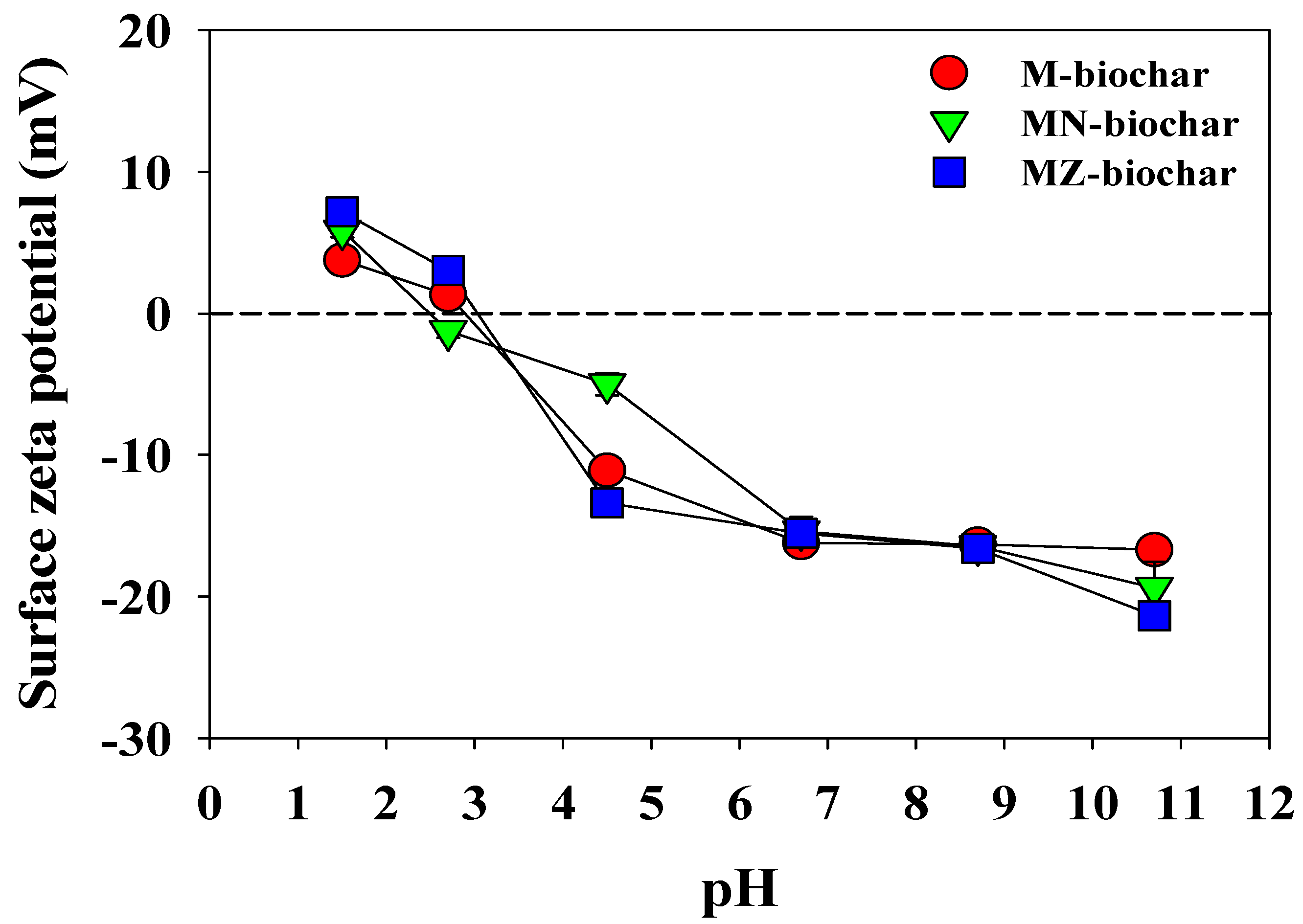
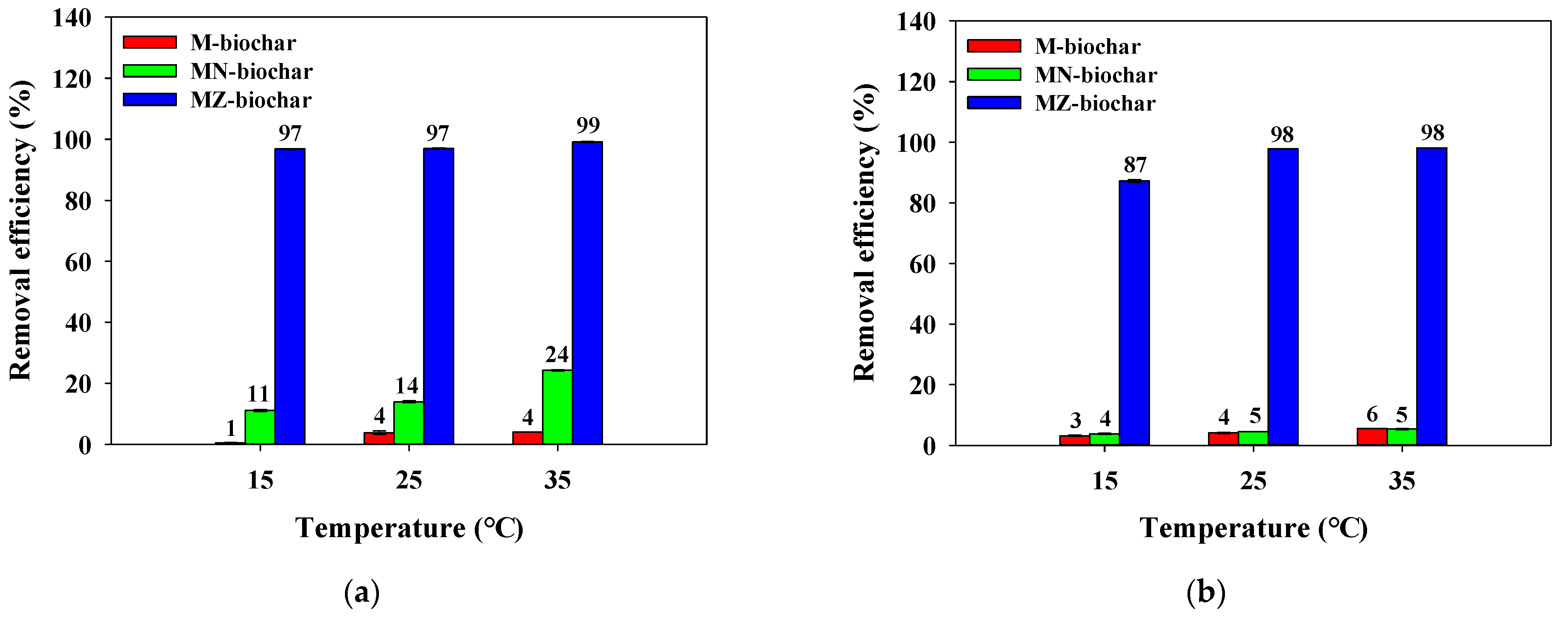
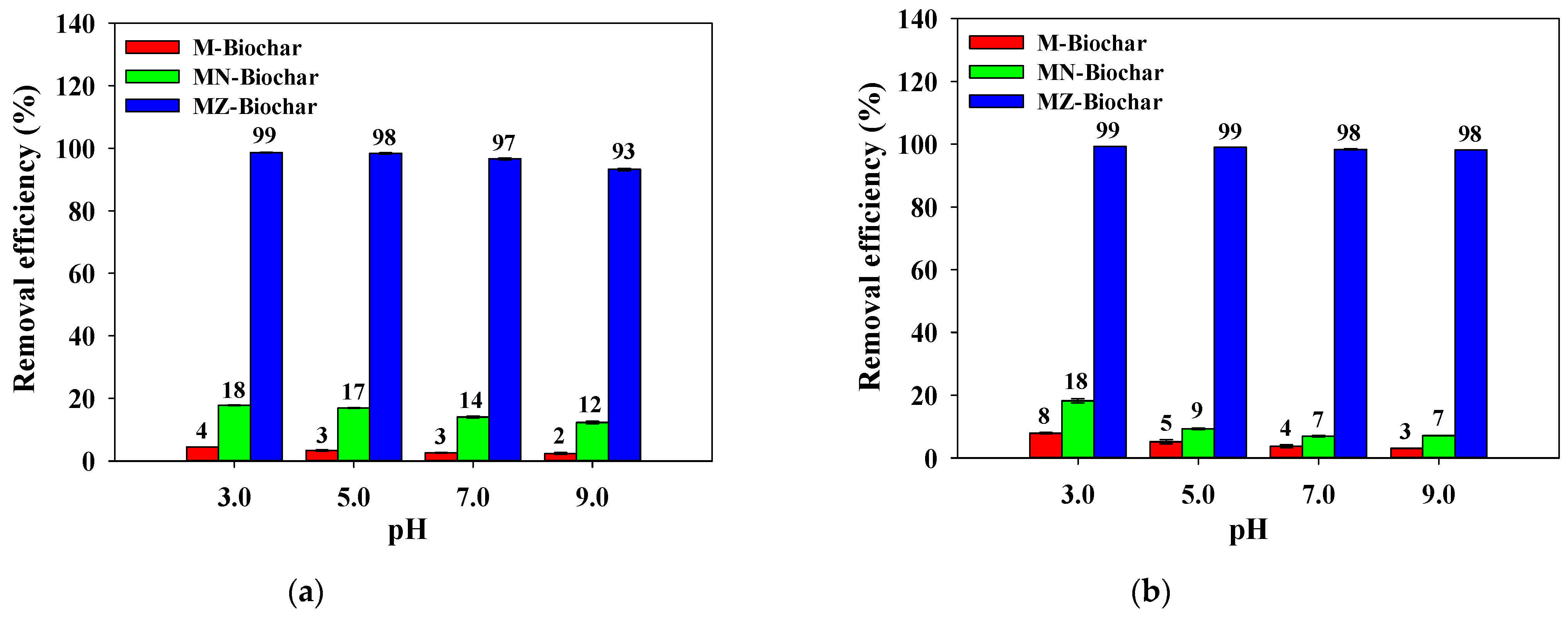
3.5. Influence of Temperature and pH on Adsorption of Dyes
4. Conclusions
- Pretreatment with ZnCl2 was the most effective for increasing the specific surface area of mandarin peel biochars. Specific surface area was closely related to the adsorption of MO and FG (MZ–biochar (1085.0 m2/g) > MN–biochar (181.1 m2/g) > M–biochar (8.4 m2/g)).
- The adsorption of MO and FG using M–biochar, MN–biochar, and MZ–biochar was described better by the pseudo-second-order model for chemical adsorption (R2 = 0.952–0.999) than by the pseudo-first-order model (R2 = 0.008–0.575).
- The adsorption of MO and FG by M–biochar, MN–biochar, and MZ–biochar was better modeled by the Freundlich isotherm equation (R2 = 0.997–0.999), with multilayer adsorption characteristics, than the Langmuir isotherm equation (R2 = 0.791–0.893), which has monolayer adsorption characteristics.
- The correlation of temperature increases with increases of MO and FG removal efficiencies onto the pristine and chemical activated mandarin peel biochars indicated that the adsorption reaction was a spontaneous and endothermic reaction.
- The adsorption efficiencies of the dyes using the M–biochar, MN–biochar, and MZ–biochar in acidic pH conditions were effective compared with the neutral and alkali pH conditions. These results suggest that the mandarin peel biochars may be a promising option in improving the dye removal from a real-scale acidic wastewater treatment plant.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Ce | Concentration of dyes at equilibrium (mg/L) |
| C0 | Initial concentrations of dyes (mg/L) |
| ∆G° | The Gibbs free energy (kJ/mol) |
| ∆H° | The enthalpy (kJ/mol) |
| ∆S° | The entropy (kJ/mol·K) |
| FG | Fast Green FCF |
| k1 | Pseudo-first-order rate constant (1/h) |
| k2 | Pseudo-second-order rate constant (g/mg·hr) |
| KF | Freundlich isotherm capacity factor (mg1−(1/n) L1/n/g) |
| KL | The adsorption energy (L/mg) |
| Kd | The distribution coefficient (L/g) |
| Qe | The quantities of the adsorbed dyes at equilibrium (mg/g) |
| Qt | The amounts of the adsorbed dyes at time t (mg/g) |
| Qe, exp | The adsorption capacities of the dyes at equilibrium (mg/g) |
| Qmax | The maximum adsorption capacity (mg/g) |
| M-biochar | Pristine mandarin peel biochar |
| MN-biochar | NH4Cl activated mandarin peel biochar |
| MZ-biochar | ZnCl2 activated mandarin peel biochar |
| MO | Methyl orange |
| n | The adsorption affinity of dyes |
| T | The absolute temperature (K) |
| V | Volume of dyes solution (L) |
References
- Namasivayam, C.; Muniasamy, N.; Gayatri, K.; Rani, M.; Ranganathan, K. Removal of dyes from aqueous solutions by cellulosic waste orange peel. Bioresour. Technol. 1996, 57, 37–43. [Google Scholar] [CrossRef]
- El-Sayed, G.O. Removal of methylene blue and crystal violet from aqueous solutions by palm kernel fiber. Desalination 2011, 272, 225–232. [Google Scholar] [CrossRef]
- Stolz, A. Basic and applied aspects in the microbial degradation of azo dyes. Appl. Microbiol. Biotechnol. 2001, 56, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Mittal, A.; Kaur, D.; Mittal, J. Batch and bulk removal of a triarylmethane dye, fast green FCF, from wastewater by adsorption over waste materials. J. Hazard. Mater. 2009, 163, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, N.; Khani, H.; Gupta, V.K.; Amereh, E.; Agarwal, S. Adsorption process of methyl orange dye onto mesoporous carbon material–kinetic and thermodynamic studies. J. Colloid Interface Sci. 2011, 362, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Weldegebrieal, G.K. Synthesis method, antibacterial and photocatalytic activity of ZnO nanoparticles for azo dyes in wastewater treatment: A review. Inorg. Chem. Commun. 2020, 120. [Google Scholar] [CrossRef]
- Pagga, U.; Brown, D. The degradation of dyestuffs: Part II behaviour of dyestuffs in aerobic biodegradation tests. Chemosphere 1986, 15, 479–491. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Ho, S.-H.; Zhou, Y.; Ren, N. Highly efficient adsorption of dyes by biochar derived from pigments-extracted macroalgae pyrolyzed at different temperature. Bioresour. Technol. 2018, 259, 104–110. [Google Scholar]
- Khadhraoui, M.; Trabelsi, H.; Ksibi, M.; Bouguerra, S.; Elleuch, B. Discoloration and detoxicification of a Congo red dye solution by means of ozone treatment for a possible water reuse. J. Hazard. Mater. 2009, 161, 974–981. [Google Scholar] [CrossRef]
- Mailler, R.; Gasperi, J.; Coquet, Y.; Derome, C.; Buleté, A.; Vulliet, E.; Bressy, A.; Varrault, G.; Chebbo, G.; Rocher, V. Removal of emerging micropollutants from wastewater by activated carbon adsorption: Experimental study of different activated carbons and factors influencing the adsorption of micropollutants in wastewater. J. Environ. Chem. Eng. 2016, 4, 1102–1109. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Bing, H.; Feifei, X.; Xiaofeng, C. Equilibrium and kinetic studies of methyl orange adsorption on multiwalled carbon nanotubes. Chem. Eng. J. 2011, 170, 82–89. [Google Scholar] [CrossRef]
- Yang, S.-T.; Chen, S.; Chang, Y.; Cao, A.; Liu, Y.; Wang, H. Removal of methylene blue from aqueous solution by graphene oxide. J. Colloid Interface Sci. 2011, 359, 24–29. [Google Scholar] [CrossRef]
- Yao, Y.; Miao, S.; Yu, S.; Ping Ma, L.; Sun, H.; Wang, S. Fabrication of Fe3O4/SiO2 core/shell nanoparticles attached to graphene oxide and its use as an adsorbent. J. Colloid Interface Sci. 2012, 379, 20–26. [Google Scholar] [CrossRef]
- Vakili, M.; Cagnetta, G.; Huang, J.; Yu, G.; Yuan, J. Synthesis and regeneration of A MXene-based pollutant adsorbent by mechanochemical methods. Molecules 2019, 24, 2478. [Google Scholar] [CrossRef] [Green Version]
- Vakili, M.; Zwain, H.M.; Mojiri, A.; Wang, W.; Gholami, F.; Gholami, Z.; Giwa, A.S.; Wang, B.; Cagnetta, G.; Salamatinia, B. Effective adsorption of reactive black 5 onto hybrid hexadecylamine impregnated chitosan-powdered activated carbon beads. Water 2020, 12, 2242. [Google Scholar] [CrossRef]
- Yuan, M.; Tong, S.; Zhao, S.; Jia, C.Q. Adsorption of polycyclic aromatic hydrocarbons from water using petroleum coke-derived porous carbon. J. Hazard. Mater. 2010, 181, 1115–1120. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Li, Z.; Feng, R.; Zhang, Y. Characterization of corncob-derived biochar and pyrolysis kinetics in comparison with corn stalk and sawdust. Bioresour. Technol. 2014, 170, 76–82. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Abdelhafez, A.A.; Li, J. Removal of Pb (II) from aqueous solution by using biochars derived from sugar cane bagasse and orange peel. J. Taiwan Inst. Chem. Eng. 2016, 61, 367–375. [Google Scholar] [CrossRef]
- Dhillon, S.S.; Gill, R.K.; Gill, S.S.; Singh, M. Studies on the utilization of citrus peel for pectinase production using fungus Aspergillus Niger. Int. J. Environ. Stud. 2004, 61, 199–210. [Google Scholar] [CrossRef]
- Ahiduzzaman, M.; Sadrul Islam, A.K.M. Preparation of porous bio-char and activated carbon from rice husk by leaching ash and chemical activation. SpringerPlus 2016, 5. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Chen, G.; Guo, X.; Abbasi, A.M.; Liu, R.H. Influence of the stage of ripeness on the phytochemical profiles, antioxidant and antiproliferative activities in different parts of Citrus reticulata Blanco cv. Chachiensis. LWT Food Sci. Technol. 2016, 69, 67–75. [Google Scholar] [CrossRef]
- Boluda-Aguilar, M.; García-Vidal, L.; del Pilar González-Castañeda, F.; López-Gómez, A. Mandarin peel wastes pretreatment with steam explosion for bioethanol production. Bioresour. Technol. 2010, 101, 3506–3513. [Google Scholar] [CrossRef] [PubMed]
- Unugul, T.; Nigiz, F.U. Synthesis of acid treated carbonized mandarin peel for purification of copper. Water Pract. Technol. 2020, 15, 460–471. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.; Kwak, J.; Lee, Y.-G.; Kim, S.; Choi, M.; Bae, S.; Lee, S.-H.; Park, Y.; Chon, K. Competitive adsorption of pharmaceuticals in lake water and wastewater effluent by pristine and NaOH-activated biochars from spent coffee wastes: Contribution of hydrophobic and π-π interactions. Environ. Pollut. 2021, 270. [Google Scholar] [CrossRef] [PubMed]
- Aljerf, L. High-efficiency extraction of bromocresol purple dye and heavy metals as chromium from industrial effluent by adsorption onto a modified surface of zeolite: Kinetics and equilibrium study. J. Environ. Manag. 2018, 225, 120–132. [Google Scholar] [CrossRef]
- Kumar, P.; Govindaraju, M.; Senthamilselvi, S.; Premkumar, K. Photocatalytic degradation of methyl orange dye using silver (Ag) nanoparticles synthesized from Ulva lactuca. Colloids Surf. B Biointerfaces 2013, 103, 658–661. [Google Scholar] [CrossRef]
- Abdi, S.; Nasiri, M. Removal of fast green FCF dye from aqueous solutions using flower gel as a low-cost adsorbent. Water Sci. Technol. 2017, 77, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Ko, R.-A.; Lee, S.; Chon, K. Removal efficiencies of manganese and iron using pristine and phosphoric acid pre-treated biochars made from banana peels. Water 2020, 12, 1173. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Wang, Z.; Guo, J.; Li, Y.; Zhang, H.; Zhu, J.; Xie, X. Enhanced adsorption of ciprofloxacin by KOH modified biochar derived from potato stems and leaves. Water Sci. Technol. 2017, 77, 1127–1136. [Google Scholar] [CrossRef] [Green Version]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef] [Green Version]
- Freundlich, H. Colloid & Capillary Chemistry; EP Dutton & Company: Boston, MA, USA, 1922. [Google Scholar]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Thermodynamic parameters of cadmium adsorption onto orange peel calculated from various methods: A comparison study. J. Environ. Chem. Eng. 2016, 4, 2671–2682. [Google Scholar] [CrossRef]
- Tsai, W.-T.; Hsien, K.-J.; Lai, C.-W. Chemical activation of spent diatomaceous earth by alkaline etching in the preparation of mesoporous adsorbents. Ind. Eng. Chem. Res. 2004, 43, 7513–7520. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Yorgun, S.; Vural, N.; Demiral, H. Preparation of high-surface area activated carbons from Paulownia wood by ZnCl2 activation. Microporous Mesoporous Mater. 2009, 122, 189–194. [Google Scholar] [CrossRef]
- Uğurlu, M.; Gürses, A.; Açıkyıldız, M. Comparison of textile dyeing effluent adsorption on commercial activated carbon and activated carbon prepared from olive stone by ZnCl2 activation. Microporous Mesoporous Mater. 2008, 111, 228–235. [Google Scholar] [CrossRef]
- Shin, J.; Lee, S.-H.; Kim, S.; Ochir, D.; Park, Y.; Kim, J.; Lee, Y.-G.; Chon, K. Effects of physicochemical properties of biochar derived from spent coffee grounds and commercial activated carbon on adsorption behavior and mechanisms of strontium ions (Sr2+). Environ. Sci. Pollut. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chon, K.; Cho, J.; Shon, H.K. A pilot-scale hybrid municipal wastewater reclamation system using combined coagulation and disk filtration, ultrafiltration, and reverse osmosis: Removal of nutrients and micropollutants, and characterization of membrane foulants. Bioresour. Technol. 2013, 141, 109–116. [Google Scholar] [CrossRef]
- Unugul, T.; Nigiz, F.U. Preparation and characterization an active carbon adsorbent from waste mandarin peel and determination of adsorption behavior on removal of synthetic dye solutions. Water Air Soil Pollut. 2020, 231. [Google Scholar] [CrossRef]
- Chia, C.H.; Gong, B.; Joseph, S.D.; Marjo, C.E.; Munroe, P.; Rich, A.M. Imaging of mineral-enriched biochar by FTIR, Raman and SEM–EDX. Vib. Spectrosc. 2012, 62, 248–257. [Google Scholar] [CrossRef]
- Chen, T.; Zhou, Z.; Han, R.; Meng, R.; Wang, H.; Lu, W. Adsorption of cadmium by biochar derived from municipal sewage sludge: Impact factors and adsorption mechanism. Chemosphere 2015, 134, 286–293. [Google Scholar] [CrossRef]
- Mahmoudi, K.; Hamdi, N.; Kriaa, A.; Srasra, E. Adsorption of methyl orange using activated carbon prepared from lignin by ZnCl2 treatment. Rus. J. Phys. Chem. A 2012, 86, 1294–1300. [Google Scholar] [CrossRef]
- Son, C.; An, W.; Lee, G.; Jeong, I.; Lee, Y.-G.; Chon, K. Adsorption characteristics of phosphate ions by pristine, CaCl2 and FeCl3-activated biochars originated from tangerine peels. Separations 2021, 8, 32. [Google Scholar] [CrossRef]
- Lee, Y.-G.; Shin, J.; Kwak, J.; Kim, S.; Son, C.; Cho, K.H.; Chon, K. Effects of NaOH activation on adsorptive removal of herbicides by biochars prepared from ground coffee residues. Energies 2021, 14, 1297. [Google Scholar] [CrossRef]
- Shin, J.; Lee, Y.-G.; Lee, S.-H.; Kim, S.; Ochir, D.; Park, Y.; Kim, J.; Chon, K. Single and competitive adsorptions of micropollutants using pristine and alkali-modified biochars from spent coffee grounds. J. Hazard. Mater. 2020, 400. [Google Scholar] [CrossRef]
- Konicki, W.; Aleksandrzak, M.; Moszyński, D.; Mijowska, E. Adsorption of anionic azo-dyes from aqueous solutions onto graphene oxide: Equilibrium, kinetic and thermodynamic studies. J. Colloid Interface Sci. 2017, 496, 188–200. [Google Scholar] [CrossRef]
- Fan, S.; Tang, J.; Wang, Y.; Li, H.; Zhang, H.; Tang, J.; Wang, Z.; Li, X. Biochar prepared from co-pyrolysis of municipal sewage sludge and tea waste for the adsorption of methylene blue from aqueous solutions: Kinetics, isotherm, thermodynamic and mechanism. J. Mol. Liq. 2016, 220, 432–441. [Google Scholar] [CrossRef]
- Chowdhury, S.; Mishra, R.; Saha, P.; Kushwaha, P. Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination 2011, 265, 159–168. [Google Scholar] [CrossRef]
- Esmaeli, A.; Jokar, M.; Kousha, M.; Daneshvar, E.; Zilouei, H.; Karimi, K. Acidic dye wastewater treatment onto a marine macroalga, Nizamuddina zanardini (Phylum: Ochrophyta). Chem. Eng. J. 2013, 217, 329–336. [Google Scholar] [CrossRef]
- Elizalde-González, M.; García-Díaz, L. Application of a taguchi L16 orthogonal array for optimizing the removal of acid orange 8 using carbon with a low specific surface area. Chem. Eng. J. 2010, 163, 55–61. [Google Scholar] [CrossRef]


| Methyl Orange | Fast Green FCF | |
|---|---|---|
| Structure a |  |  |
| M–Biochar | MN–Biochar | MZ–Biochar | |
|---|---|---|---|
| C (%) | 79.53 ± 0.71 | 80.05 ± 0.55 | 79.21 ± 0.46 |
| H (%) | 1.92 ± 0.015 | 1.72 ± 0.028 | 1.78 ± 0.040 |
| O (%) | 5.09 ± 0.078 | 4.88 ± 0.061 | 3.63 ± 0.089 |
| N (%) | 2.07 ± 0.019 | 1.76 ± 0.023 | 1.44 ± 0.029 |
| S (%) | 0.13 ± 0.010 | 0.25 ± 0.037 | 0.15 ± 0.011 |
| H/C | 0.29 ± 0.003 | 0.26 ± 0.002 | 0.27 ± 0.003 |
| O/C | 0.048 ± 0.008 | 0.046 ± 0.009 | 0.034 ± 0.010 |
| N/C | 0.022 ± 0.003 | 0.019 ± 0.001 | 0.016 ± 0.002 |
| Specific surface area (m2/g) | 8.5 | 181.1 | 1085.0 |
| Total pore volume (cm3/g) | 0.016 | 0.031 | 0.19 |
| Pore size (nm) | 8.74 | 4.58 | 3.62 |
| Adsorbents | M–Biochar | MN–Biochar | MZ–Biochar | ||||
|---|---|---|---|---|---|---|---|
| Dyes | MO | FG | MO | FG | MO | FG | |
| Qe, exp (mg/g) | 0.95 ± 0.098 | 0.78 ± 0.039 | 2.52 ± 0.42 | 2.13 ± 0.38 | 16.27 ± 0.91 | 12.44 ± 0.88 | |
| Pseudo-first- order | Qe, cal (mg/g) | 2.06 ± 0.19 | 15.85 ± 0.23 | 1.78 ± 0.12 | 2.07 ± 0.66 | 16.00 ± 0.38 | 1.90 ± 0.62 |
| k1 (1/h) | 0.49 ± 0.057 | 0.063 ± 0.004 | 0.56 ± 0.092 | 0.48 ± 0.083 | 0.063 ± 0.009 | 0.52 ± 0.017 | |
| R2 | 0.575 | 0.008 | 0.465 | 0.565 | 0.482 | 0.206 | |
| Pseudo-second- order | Qe, cal (mg/g) | 0.97 ± 0.10 | 0.80 ± 0.022 | 2.53 ± 0.28 | 2.22 ± 0.67 | 16.27 ± 0.096 | 12.45 ± 0.88 |
| k2 (g/mg·h) | 2.81 ± 0.19 | 3.14 ± 0.44 | 3.45 ± 0.58 | 0.98 ± 0.020 | 12.56 ± 0.29 | 12.81 ± 0.23 | |
| R2 | 0.981 | 0.962 | 0.996 | 0.952 | 0.999 | 0.999 | |
| Adsorbents | M–Biochar | MN–Biochar | MZ–Biochar | ||||
|---|---|---|---|---|---|---|---|
| Dyes | MO | FG | MO | FG | MO | FG | |
| Langmuir | Qmax (mg/g) | 2.00 ± 0.18 | 14.47 ± 0.57 | 0.14 ± 0.008 | 14.39 ± 0.67 | 14.25 ± 0.91 | 45.87 ± 2.19 |
| KL (L/mg) | 0.01 ± 0.002 | 0.35 ± 0.022 | 0.85 ± 0.096 | 0.42 ± 0.22 | 0.069 ± 0.003 | 0.18 ± 0.097 | |
| R2 | 0.959 | 0.791 | 0.893 | 0.843 | 0.813 | 0.841 | |
| Freundlich | n | 4.71 ± 0.39 | 0.96 ± 0.019 | 1.81 ± 0.57 | 0.98 ± 0.44 | 1.68 ± 0.096 | 1.38 ± 0.022 |
| KF (mg1−(1/n) L1/n/g) | 0.42 ± 0.046 | 12.35 ± 0.36 | 1.13 ± 0.38 | 12.87 ± 0.84 | 38.21 ± 2.87 | 3.67 ± 0.22 | |
| R2 | 0.999 | 0.999 | 0.999 | 0.998 | 0.999 | 0.997 | |
| Adsorbents | Dyes | Thermodynamic Parameters | |||
|---|---|---|---|---|---|
| Temperature (K) | ∆G° (kJ/mol) | ∆H° (kJ/mol) | ∆S° (kJ/mol·K) | ||
| M–biochar | MO | 288 | −2.84 ± 0.17 | 0.002 ± 0.0005 | 0.032 ± 0.002 |
| 298 | −3.57 ± 0.14 | ||||
| 308 | −4.35 ± 0.11 | ||||
| FG | 288 | −4.24 ± 0.09 | 0.008 ± 0.0007 | 0.043 ± 0.003 | |
| 298 | −4.50 ± 0.12 | ||||
| 308 | −4.81 ± 0.10 | ||||
| MN–biochar | MO | 288 | −5.59 ± 0.21 | 0.050 ± 0.001 | 0.039 ± 0.009 |
| 298 | −5.80 ± 0.34 | ||||
| 308 | −6.45 ± 0.20 | ||||
| FG | 288 | −4.38 ± 0.38 | 0.012 ± 0.002 | 0.052 ± 0.003 | |
| 298 | −4.58 ± 0.25 | ||||
| 308 | −4.75 ± 0.28 | ||||
| MZ–biochar | MO | 288 | −11.27 ± 0.23 | 0.003 ± 0.002 | 0.045 ± 0.009 |
| 298 | −11.35 ± 0.13 | ||||
| 308 | −12.86 ± 0.42 | ||||
| FG | 288 | −9.69 ± 0.33 | 0.002 ± 0.0003 | 0.037 ± 0.005 | |
| 298 | −11.35 ± 0.32 | ||||
| 308 | −11.75 ± 0.25 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, H.; Kim, J.; Lee, Y.-G.; Chon, K. Enhanced Adsorptive Removal of Dyes Using Mandarin Peel Biochars via Chemical Activation with NH4Cl and ZnCl2. Water 2021, 13, 1495. https://doi.org/10.3390/w13111495
Park H, Kim J, Lee Y-G, Chon K. Enhanced Adsorptive Removal of Dyes Using Mandarin Peel Biochars via Chemical Activation with NH4Cl and ZnCl2. Water. 2021; 13(11):1495. https://doi.org/10.3390/w13111495
Chicago/Turabian StylePark, Hyunjun, Jiseok Kim, Yong-Gu Lee, and Kangmin Chon. 2021. "Enhanced Adsorptive Removal of Dyes Using Mandarin Peel Biochars via Chemical Activation with NH4Cl and ZnCl2" Water 13, no. 11: 1495. https://doi.org/10.3390/w13111495
APA StylePark, H., Kim, J., Lee, Y.-G., & Chon, K. (2021). Enhanced Adsorptive Removal of Dyes Using Mandarin Peel Biochars via Chemical Activation with NH4Cl and ZnCl2. Water, 13(11), 1495. https://doi.org/10.3390/w13111495







