Understanding Microbial Loads in Wastewater Treatment Works as Source Water for Water Reuse
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and Processing
2.2. Microbial Analyses
2.2.1. Fecal Indicator Organisms
2.2.2. Protozoan Parasites
2.2.3. Adenovirus Infectivity Assay
2.3. Data Presentation and Statistical Analyses
3. Results and Discussion
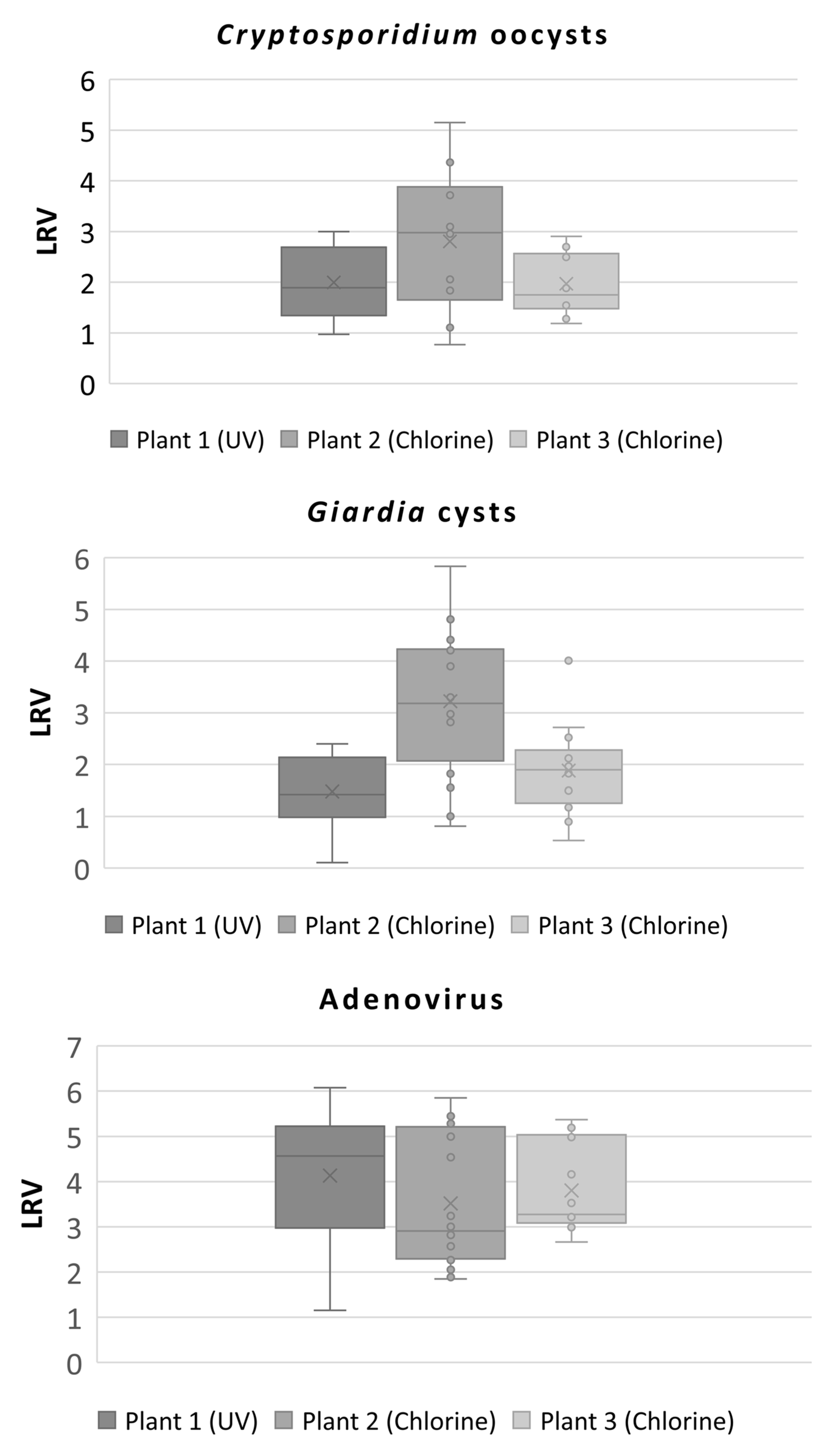
3.1. Enteric Protozoan Parasites
3.2. Human Adenovirus
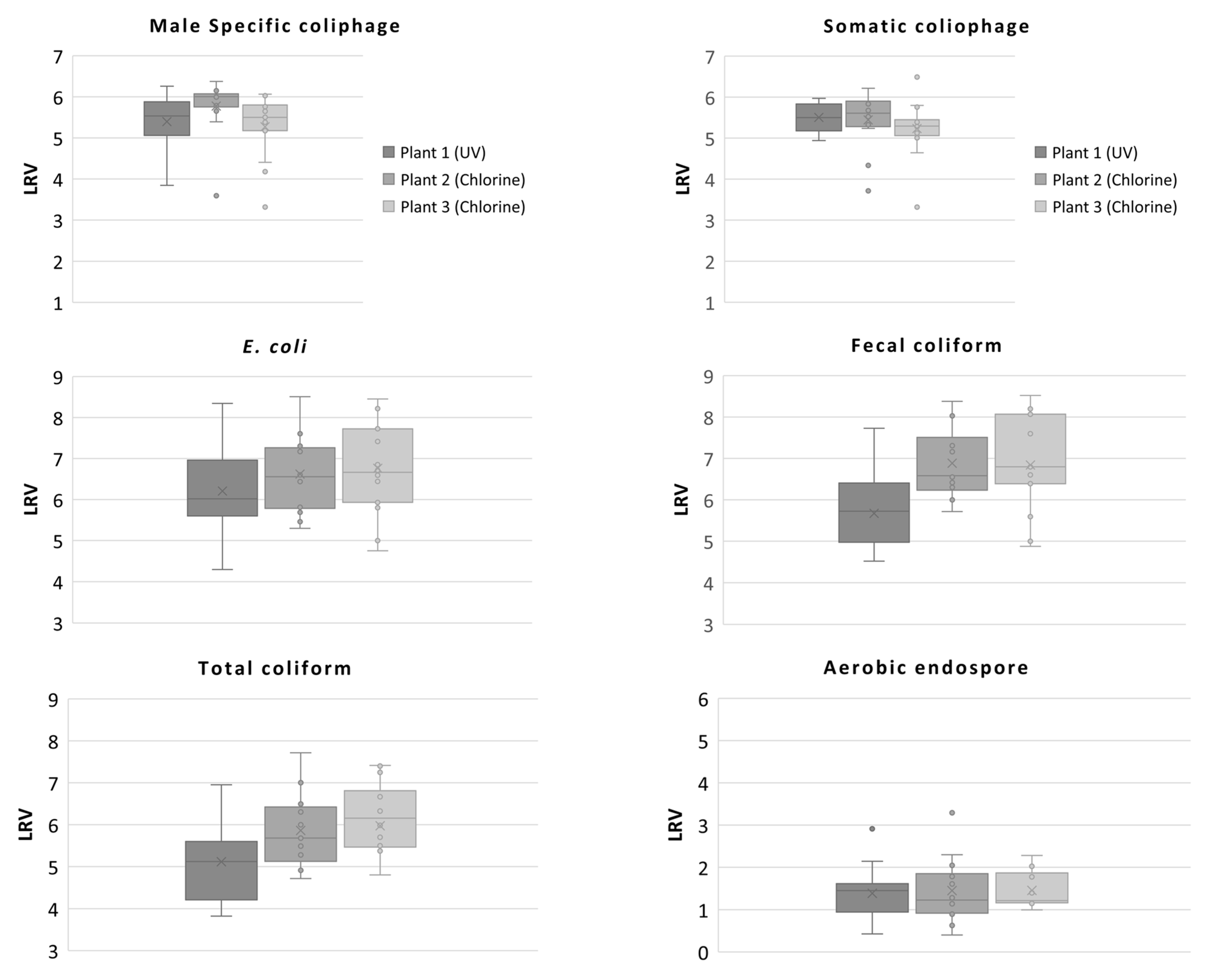
3.3. Microbial Indicators
3.4. Calculated Log10 Reduction Values (LRVs) Obtained from the Three Wastewater Treatment Plants
3.5. Engineering Implications for DPR on Microbial Quality
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cohen, M. Municipal Deliveries of Colorado River Basin Water; Pacific Institute: Oakland, CA, USA, 2011. [Google Scholar]
- U.S. Department of the Interior (USDOI); Bureau of Reclamation. Water 2025: Preventing Conflict and Crisis in the West. 2003. Available online: www.doi.gov/sites/doi.gov/files/archive/news/archive/03_News_Releases/030502a.htm (accessed on 20 May 2021).
- Garfin, G.; Crimmins, M.A.; Jacobs, K.L. Drought, Climate Variability, and Implications for Water Supply. In Arizona Water Policy: Management Innovations in an Urbanizing Arid Region; Colby, B.G., Jacobs, K.L., Eds.; Resources for the Future: Washington, DC, USA, 2007. [Google Scholar]
- Creel, L. Ripple Effects: Population and Coastal Regions; Population Reference Bureau: Washington, DC, USA, 2003. [Google Scholar]
- U.S. Environmental Protection Agency (USEPA). Guidelines for Water Reuse; EPA/600/R-12/618; U.S. Environmental Protection Agency; U.S. Agency for International Development: Washington, DC, USA, 2012.
- Rice, J.; Wutich, A.; Westerhoff, P. Assessment of De Facto Wastewater Reuse across the U.S.: Trends between 1980 and 2008. Environ. Sci. Technol. 2013, 47, 11099–11105. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Potable Reuse Compendium; Office of Ground Water and Drinking Water; Office of Water: Washington, DC, USA, 2017. Available online: https://www.epa.gov/sites/production/files/2018-01/documents/potablereusecompendium_3.pdf (accessed on 20 May 2021).
- World Health Organization (WHO). Guidelines of the Safe Use of Wastewater, Excreta and Greywater: Excreta and Greywater Use in Agriculture. 2006. Available online: www.who.int/water_sanitation_health/publications/gsuweg4/en/ (accessed on 20 May 2021).
- Crook, J. Regulatory Aspects of Direct Potable Reuse in California; National Water Research Institute: Fountain Valley, CA, USA, 2010. [Google Scholar]
- Leverenz, H.L.; Tchobanoglous, G.; Asano, T. Direct potable reuse: A future imperative. J. Water Reuse Desalin. 2011, 1, 2–10. [Google Scholar] [CrossRef]
- National Research Council (NRC). Water Reuse: Potential for Expanding the Nation’s Water Supply through Reuse of Municipal Wastewater; The National Academies Press: Washington, DC, USA, 2012. [Google Scholar]
- Schroeder, E.; Tchobanoglous, G.; Leverenz, H.L.; Asano, T. Direct Potable Reuse: Benefits for Public Water Supplies, Agriculture, the Environment, and Energy Conservation; National Water Research Institute (NWRI) White Paper, NWRI-2012-01; National Water Research Institute: Fountain Valley, CA, USA, 2012. [Google Scholar]
- Tchobanoglous, G.; Leverenz, H.L.; Nellor, M.H.; Crook, J. Direct Potable Reuse: The Path Forward; Water Reuse Research Foundation; Water Reuse California: Washington, DC, USA, 2011. [Google Scholar]
- Huang, C.; Hu, Y.; Wang, L.; Wang, Y.; Li, N.; Guo, Y.; Feng, Y.; Xiao, L. Environmental Transport of Emerging Human-Pathogenic Cryptosporidium Species and Subtypes through Combined Sewer Overflow and Wastewater. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef]
- Rose, J.B.; Dickson, L.J.; Farrah, S.R.; Carnahan, R.P. Removal of pathogenic and indicator microorganisms by a full-scale water reclamation facility. Water Res. 1996, 30, 2785–2797. [Google Scholar] [CrossRef]
- Tanaka, H.; Asano, T.; Schroeder, E.D.; Tchobanoglous, G. Estimating the safety of wastewater reclamation and reuse using enteric virus monitoring data. Water Environ. Res. 1998, 70, 39–51. [Google Scholar] [CrossRef]
- Ryu, H.; Alum, A.; Abbaszadegan, M. Microbial Characterization and Population Changes in Nonpotable Reclaimed Water Distribution Systems. Environ. Sci. Technol. 2005, 39, 8600–8605. [Google Scholar] [CrossRef]
- Ryu, H.; Alum, A.; Mena, K.; Abbaszadegan, M. Assessment of the risk of infection by Cryptosporidium and Giardia in non-potable reclaimed water. Water Sci. Technol. 2007, 55, 283–290. [Google Scholar] [CrossRef]
- Chaudhry, R.M.; Hamilton, K.A.; Haas, C.N.; Nelson, K.L. Drivers of Microbial Risk for Direct Potable Reuse and de Facto Reuse Treatment Schemes: The Impacts of Source Water Quality and Blending. Int. J. Environ. Res. Public Health 2017, 14, 635. [Google Scholar] [CrossRef]
- Soller, J.A.; Eftim, S.E.; Warren, I.; Nappier, S.P. Evaluation of microbiological risks associated with direct potable reuse. Microb. Risk Anal. 2017, 5, 3–14. [Google Scholar] [CrossRef]
- Nappier, S.P.; Soller, J.A.; Eftim, S.E. Potable water reuse: What are the microbiological risks? Curr. Environ. Health Rep. 2018, 5, 283–292. [Google Scholar] [CrossRef]
- Soller, J.A.; Eftim, S.E.; Nappier, S.P. Direct potable reuse microbial risk assessment methodology: Sensitivity analysis and application to State log credit allocations. Water Res. 2018, 128, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, E.R.; Huff, E.M.; Hamilton, D.W.; Jones, J.L. The evaluation of hollow-fiber ultrafiltration and celite concentration of enteroviruses, adenoviruses and bacteriophage from different water matrices. J. Virol. Methods 2016, 228, 31–38. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Cryptosporidium and Giardia in Water by Filtration/IMS/FA; EPA/816/R-12/001; U.S. Environmental Protection Agency, Office of Water: Washington, DC, USA, 2012.
- Ware, M.; Keely, S.; Villegas, E. Development and evaluation of an off-the-slide genotyping technique for identifying Giardia cysts and Cryptosporidium oocysts directly from US EPA Method 1623 slides. J. Appl. Microbiol. 2013, 115, 298–309. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency (USEPA). Most Probable Number (MPN) Calculator. 2012. Available online: https://mostprobablenumbercalculator.epa.gov (accessed on 20 May 2021).
- U.S. Environmental Protection Agency (USEPA). Method 1681: Fecal Coliform in Sewage Sludge (Biosolids) by Multiple Tube Fermentation Using A-1 Medium; EPA/821/R-06/013; U.S. Environmental Protection Agency, Office of Water: Washington, DC, USA, 2006.
- American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF). Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- U.S. Environmental Protection Agency (USEPA). Method 1602: Male-Specific (F+) and Somatic Coliphage in Water by Single Agar Layer (SAL) Procedure; EPA/821/R-01/029; U.S. Environmental Protection Agency, Office of Water: Washington, DC, USA, 2001.
- Ware, M.W.; Wymer, L.; Lindquist, H.D.; Schaefer, F.W. Evaluation of an Alternative IMS Dissociation Procedure for Use with Method 1622: Detection of Cryptosporidium in Water. J. Microbiol. Methods 2003, 55, 575–583. [Google Scholar] [CrossRef]
- McMinn, B.R.; Cashdollar, J.L.; Grimm, A.C.; Fout, G.S. Evaluation of the celite secondary concentration procedure and an alternate elution buffer for the recovery of enteric adenoviruses 40 and 41. J. Virol. Methods 2012, 179, 423–428. [Google Scholar] [CrossRef]
- Cashdollar, J.L.; Brinkman, N.E.; Griffin, S.M.; McMinn, B.R.; Rhodes, E.R.; Varughese, E.A.; Grimm, A.C.; Parshionikar, S.U.; Wymer, L.; Fout, G.S. Development and Evaluation of EPA Method 1615 for Detection of Enterovirus and Norovirus in Water. Appl. Environ. Microbiol. 2012, 79, 215–223. [Google Scholar] [CrossRef]
- Ryu, H.; Cashdollar, J.L.; Fout, G.S.; Schrantz, K.A.; Hayes, S. Applicability of integrated cell culture quantitative PCR (ICC-qPCR) for the detection of infectious adenovirus type 2 in UV disinfection studies. J. Environ. Sci. Health 2015, 50, 777–787. [Google Scholar] [CrossRef]
- Gerrity, D.; Ryu, H.; Crittenden, J.; Abbaszadegan, M. UV inactivation of Adenovirus Type 4 measured by integrated cell culture qPCR. J. Environ. Sci. Health 2008, 43, 1628–1638. [Google Scholar] [CrossRef]
- Sykora, J.L.; Sorber, C.A.; Jakubowski, W.; Casson, L.W.; Gavaghan, P.D.; Shapiro, M.A.; Schott, M.J. Distribution of Giardia Cysts in Wastewater. Water Sci. Technol. 1991, 24, 187–192. [Google Scholar] [CrossRef]
- Wallis, P.M.; Erlandsen, S.L.; Isaac-Renton, J.L.; Olson, M.E.; Robertson, W.J.; Van Keulen, H. Prevalence of Giardia cysts and Cryptosporidium oocysts and characterization of Giardia spp. isolated from drinking water in Canada. Appl. Environ. Microbiol. 1996, 62, 2789–2797. [Google Scholar] [CrossRef] [PubMed]
- Harwood, V.J.; Levine, A.D.; Scott, T.M.; Chivukula, V.; Lukasik, J.; Farrah, S.R.; Rose, J.B. Validity of the indicator organism paradigm for pathogen reduction in reclaimed water and public health protection. Appl. Environ. Microbiol. 2005, 71, 3163–3170. [Google Scholar] [CrossRef] [PubMed]
- Crockett, C.S. The Role of Wastewater Treatment in Protecting Water Supplies against Emerging Pathogens. Water Environ. Res. 2007, 79, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Schneider, O.D.; Jjemba, P.K.; Lechevallier, M.W. Microbial Risk Modeling for Main Breaks. J. Am. Water Works Assoc. 2015, 107, E97–E108. [Google Scholar] [CrossRef]
- Nasser, A.M. Removal of Cryptosporidium by wastewater treatment processes: A review. J. Water Health 2015, 14, 1–13. [Google Scholar] [CrossRef]
- Dungeni, M.; Momba, M. The abundance of Cryptosporidium and Giardia spp. in treated effluents produced by four wastewater treatment plants in the Gauteng Province of South Africa. Water SA 2010, 36, 425–432. [Google Scholar] [CrossRef][Green Version]
- Taran-Benshoshan, M.; Ofer, N.; Dalit, V.O.; Aharoni, A.; Revhun, M.; Nitzan, Y.; Nasser, A.M. Cryptosporidium and Giardia removal by secondary and tertiary wastewater treatment. J. Environ. Sci. Health 2015, 50, 1265–1273. [Google Scholar] [CrossRef]
- Robertson, L.J.; Hermansen, L.; Gjerde, B.K. Occurrence of Cryptosporidium Oocysts and Giardia Cysts in Sewage in Norway. Appl. Environ. Microbiol. 2006, 72, 5297–5303. [Google Scholar] [CrossRef]
- Hurst, C.J.; McClellan, K.A.; Benton, W.H. Comparison of cytopathogenicity, immunofluorescence and in situ DNA hybridization as methods for the detection of adenoviruses. Water Res. 1988, 22, 1547–1552. [Google Scholar] [CrossRef]
- Hewitt, J.; Leonard, M.; Greening, G.E.; Lewis, G.D. Influence of wastewater treatment process and the population size on human virus profiles in wastewater. Water Res. 2011, 45, 6267–6276. [Google Scholar] [CrossRef]
- Eftim, S.; Hong, T.; Ichida, A.; Warren, I.; Soller, J.; Nappier, S.P. Systematic Literature Reviews and Development of Distribution Curves for Viral Densities in Raw Wastewater; University of North Carolina: Chapel Hill, NC, USA, 2016. [Google Scholar]
- Boczek, L.; Rhodes, E.; Cashdollar, J.; Ryu, J.; Popovici, J.; Hoelle, J.; Hayes, S.; Rodgers, M.; Ryu, H. Applicability of UV resistant Bacillus pumilus spore as a human adenovirus surrogate for evaluating the effectiveness of virus inactivation in low-pressure UV treatment systems. J. Microbiol. Methods 2016, 122, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-W.A.; Lucy, F.E.; Graczyk, T.K.; Broaders, M.A.; Tamang, L.; Connolly, M. Fate of Cryptosporidium parvum and Cryptosporidium hominis oocysts and Giardia duodenalis cysts during secondary wastewater treatments. Parasitol. Res. 2009, 105, 689–696. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Drinking water; national primary drinking water regulations; filtration, disinfection; turbidity, Giardia lamblia, viruses, Legionella, and heterotrophic bacteria; final rule. Fed. Regist. 1989, 54, 27486–27541. [Google Scholar]
- Clark, R.M.; Regli, S. Development of Giardia C · t values for the surface water treatment rule. J. Environ. Sci. Health 1993, 28, 1081–1097. [Google Scholar] [CrossRef]
- Finch, G.R.; Gyurek, L.L.; Liyanage, L.R.J.; Belosevic, M. Effect of Various Disinfection Methods on the Inactivation of Cryptosporidium; American Water Works Association Research Foundation: Denver, CO, USA, 1997. [Google Scholar]
- Clancy, J.L.; Bukhari, Z.; Hargy, T.M.; Bolton, J.R.; Dussert, B.W.; Marshall, M.M. Using UV to inactivate Cryptosporidium. J. Am. Water Works Assoc. 2000, 92, 97–104. [Google Scholar] [CrossRef]
- Morita, S.; Namikoshi, A.; Hirata, T.; Oguma, K.; Katayama, H.; Ohgaki, S.; Motoyama, N.; Fujiwara, M. Efficacy of UV irradiation in inactivating Cryptosporidium parvum oocysts. Appl. Environ. Microbiol. 2002, 68, 5387–5393. [Google Scholar] [CrossRef]
- Zimmer, J.; Slawson, R.; Huck, P. Inactivation and potential repair of Cryptosporidium parvum following low- and medium-pressure ultraviolet irradiation. Water Res. 2003, 37, 3517–3523. [Google Scholar] [CrossRef]
- Qian, S.S.; Donnelly, M.; Schmelling, D.C.; Messner, M.; Linden, K.G.; Cotton, C. Ultraviolet light inactivation of protozoa in drinking water: A Bayesian meta-analysis. Water Res. 2004, 38, 317–326. [Google Scholar] [CrossRef]
- Ryu, H.; Abbaszadegan, M. Long-term study of Cryptosporidium and Giardia occurrence and quantitative microbial risk assessment in surface waters of Arizona in the USA. J. Water Health 2008, 6, 263–273. [Google Scholar] [CrossRef][Green Version]
- Leoni, F.; Amar, C.; Nichols, G.; Pedraza-Diaz, S.; McLauchlin, J. Genetic analysis of Cryptosporidium from 2414 humans with diarrhoea in England between 1985 and 2000. J. Med. Microbiol. 2006, 55, 703–707. [Google Scholar] [CrossRef]
- Xiao, L.; Fayer, R. Molecular characterisation of species and genotypes of Cryptosporidium and Giardia and assessment of zoonotic transmission. Int. J. Parasitol. 2008, 38, 1239–1255. [Google Scholar] [CrossRef]
- Francy, D.S.; Stelzer, E.A.; Bushon, R.N.; Brady, A.M.; Williston, A.G.; Riddell, K.R.; Borchardt, M.A.; Spencer, S.K.; Gellner, T.M. Comparative effectiveness of membrane bioreactors, conventional secondary treatment, and chlorine and UV disinfection to remove microorganisms from municipal wastewaters. Water Res. 2012, 46, 4164–4178. [Google Scholar] [CrossRef]
- Olivieri, A.W.; Crook, J.; Anderson, M.A.; Bull, R.J.; Drewes, J.E.; Haas, C. Expert Panel Final Report: Evaluation of the Feasibility of Developing Uniform Water Recycling Criteria for Direct Potable Reuse; National Water Research Institute for the State Water Resources Control Board: Sacramento, CA, USA, 2016. Available online: http://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/rw_dpr_criteria.shtml (accessed on 20 May 2021).
- Gerba, C.P.; Gramos, D.M.; Nwachuku, N. Comparative inactivation of enteroviruses and adenovirus type 2 by UV light. Appl. Environ. Microbiol. 2002, 68, 5167–5169. [Google Scholar] [CrossRef] [PubMed]
- Thurston-Enriquez, J.A.; Haas, C.N.; Jacangelo, J.; Riley, K.; Gerba, C.P. Inactivation of feline calicivirus and adenovirus type 40 by UV irradiation. Appl. Environ. Microbiol. 2003, 69, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Ko, G.; Cromeans, T.L.; Sobsey, M.D. UV inactivation of adenovirus type 41 measured by cell culture mRNA RT-PCR. Water Res. 2005, 39, 3643–3649. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Ultraviolet Disinfection Guidance Manual for the Final Long Term 2 Enhanced Surface Water Treatment Rule; EPA: Washington, DC, USA, 2006.
- U.S. Environmental Protection Agency (USEPA). National Primary Drinking Water Regulations: Stage 2 Disinfectants and Disinfection Byproducts Rule; Final Rule. Part II. EPA, Washington, DC; 40 CFR Parts 9, 141 and 142. Fed. Regist. 2006, 71, 388. [Google Scholar]
- U.S. Environmental Protection Agency (USEPA). National Primary Drinking Water Rregulations: Long Term 2 Enhanced Surface Water Treatment Rule; Final Rule. Part II. EPA, Washington, DC; 40 CFR Parts 9, 141 and 142. Fed. Regist. 2006, 71, 654. [Google Scholar]
- Hijnen, W.A.M.; Beerendonk, E.F.; Medema, G.J. Inactivation credit of UV radiation for viruses, bacteria and protozoan (oo)cysts in water: A review. Water Res. 2006, 40, 3–22. [Google Scholar] [CrossRef]
| WWTP | Customer Base | Processing Size (m3/Day) | Secondary Treatment Processes * | Disinfection Type |
|---|---|---|---|---|
| Plant 1 | 180,000 | 41.6 thousand | Aeration, Disc filter microscreens | Ultraviolet |
| Plant 2 | 28,000 | 9.1 thousand | Aeration, Trickling filters | Chlorine |
| Plant 3 | 20,000 | 8.0 thousand | Aeration | Chlorine |
| Plant | Adenovirus (MPN/L) | Cryptosporidium (Oocysts/L) | Giardia (Cysts/L) | ||||
|---|---|---|---|---|---|---|---|
| Influent-Grab | Effluent-HFUF a | Influent-Grab | Effluent-HFUF a | Influent-Grab | Effluent-HFUF a | ||
| 1 | Prevalence | 16/16 (100%) | 9/16 (56%) | 10/16 (63%) | 13/16 (81%) | 16/16 (100%) | 16/16 (100%) |
| Median [min-max] | 3025 [8.64–65,480] | 0.07 [0.05–511] | 30.0 [10.0–190] | 0.34 [0.11–8.55] | 270 [50.0–2840] | 10.0 [0.72–80.0] | |
| 2 | Prevalence | 16/16 (100%) | 13/16 (81%) | 11/16 (69%) | 12/16 (75%) | 16/16 (100%) | 14/16 (88%) |
| Median [min-max] | 64.1 [3.17–14,878] | 0.07 [0.02–0.54] | 1150 [10.0–3420] | 0.17 [0.02–49.8] | 2400 [20.0–35,000] | 1.45 [0.01–52.1] | |
| 3 | Prevalence | 14/15 (93%) | 6/15 (40%) | 10/15 (67%) | 14/15 (93%) | 14/15 (93%) | 15/15 (100%) |
| Median [min-max] | 25.9 [10.23–5797] | 0.02 [0.02–0.04] | 15.0 [8.00–150] | 0.23 [0.01–1.42] | 717 [80.0–3140] | 4.74 [0.10–77.0] | |
| Plant | Male Specific (PFU/mL) | Somatic (PFU/mL) | |||||
|---|---|---|---|---|---|---|---|
| Influent-Grab | Effluent-Grab | Effluent-HFUF a | Influent-Grab | Effluent-Grab | Effluent-HFUF a | ||
| 1 | Prevalence | 16/16 (100%) | 0/16 (0%) | 3/16 (19%) | 16/16 (100%) | 0/16 (0%) | 5/16 (31%) |
| Median [min-max] | 1433 [52–7400] | ND | 0.004, 0.005, 0.043 | 1458 [355–3850] | ND | 0.005 [0.004–0.009] | |
| 2 | Prevalence | 14/14 (100%) | 1/14 (7%) | 1/14 (7%) | 13/13 (100%) | 1/14 (7%) | 2/14 (14%) |
| Median [min-max] | 4700 [20–11,700] | 100 | 0.038 | 1865 [110–7300] | 2 | 0.004, 0.25 | |
| 3 | Prevalence | 15/15 (100%) | 1/15 (6.7%) | 1/15 (6.7%) | 15/15 (100%) | 0/15 (0%) | 2/15 (13.3%) |
| Median [min-max] | 1555 [52–6050] | 100 | 1.11 | 985 [225–15, 150] | ND | 0.005, 1.01 | |
| Plant [Log10 CFU/100 mL] | E. coli | Fecal Coliform | Total Coliform | Endospore | |||||
|---|---|---|---|---|---|---|---|---|---|
| Influent | Effluent | Influent | Effluent | Influent | Effluent | Influent | Effluent | ||
| 1 | Prevalence | 16/16 (100%) | 16/16 (100%) | 16/16 (100%) | 16/16 (100%) | 16/16 (100%) | 16/16 (100%) | 16/16 (100%) | 16/16 (100%) |
| Median [min-max] | 6.99 [5.80–8.84] | 0.96 [0.30–1.68] | 7.30 [5.30–9.38] | 1.69 [0.99–2.81] | 8.01 [6.92–9.38] | 2.91 [2.31–3.38] | 4.34 [3.30–5.35] | 2.96 [2.31–3.38] | |
| 2 | Prevalence | 14/14 (100%) | 7/14 (50%) | 14/14 (100%) | 10/14 (71%) | 14/14 (100%) | 12/14 (86%) | 14/14 (100%) | 14/14 (100%) |
| Median [min-max] | 6.94 [5.30–8.81] | 0.61 [0.30–2.39] | 7.37 [6.30–8.94] | 0.76 [0.30–2.04] | 8.16 [6.99–9.38] | 2.77 [0.49–3.38] | 4.20 [2.70–5.47] | 2.82 [1.60–4.30] | |
| 3 | Prevalence | 15/15 (100%) | 2/15 (13%) | 15/15 (100%) | 5/15 (33%) | 15/15 (100%) | 14/15 (93%) | 15/15 (100%) | 15/15 (100%) |
| Median [min-max] | 6.72 [5.00–8.45] | 0.30, 2.29 | 6.93 [5.00–8.94] | 0.61 [0.30–2.38] | 7.51 [5.30–9.38] | 1.83 [0.80–3.19] | 4.42 [3.79–5.48] | 2.93 [2.40–3.99] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryu, H.; Addor, Y.; Brinkman, N.E.; Ware, M.W.; Boczek, L.; Hoelle, J.; Mistry, J.H.; Keely, S.P.; Villegas, E.N. Understanding Microbial Loads in Wastewater Treatment Works as Source Water for Water Reuse. Water 2021, 13, 1452. https://doi.org/10.3390/w13111452
Ryu H, Addor Y, Brinkman NE, Ware MW, Boczek L, Hoelle J, Mistry JH, Keely SP, Villegas EN. Understanding Microbial Loads in Wastewater Treatment Works as Source Water for Water Reuse. Water. 2021; 13(11):1452. https://doi.org/10.3390/w13111452
Chicago/Turabian StyleRyu, Hodon, Yao Addor, Nichole E. Brinkman, Michael W. Ware, Laura Boczek, Jill Hoelle, Jatin H. Mistry, Scott P. Keely, and Eric N. Villegas. 2021. "Understanding Microbial Loads in Wastewater Treatment Works as Source Water for Water Reuse" Water 13, no. 11: 1452. https://doi.org/10.3390/w13111452
APA StyleRyu, H., Addor, Y., Brinkman, N. E., Ware, M. W., Boczek, L., Hoelle, J., Mistry, J. H., Keely, S. P., & Villegas, E. N. (2021). Understanding Microbial Loads in Wastewater Treatment Works as Source Water for Water Reuse. Water, 13(11), 1452. https://doi.org/10.3390/w13111452








