Vegetation and Residence Time Interact to Influence Metabolism and Net Nutrient Uptake in Experimental Agricultural Drainage Systems
Abstract
1. Introduction
2. Materials and Methods
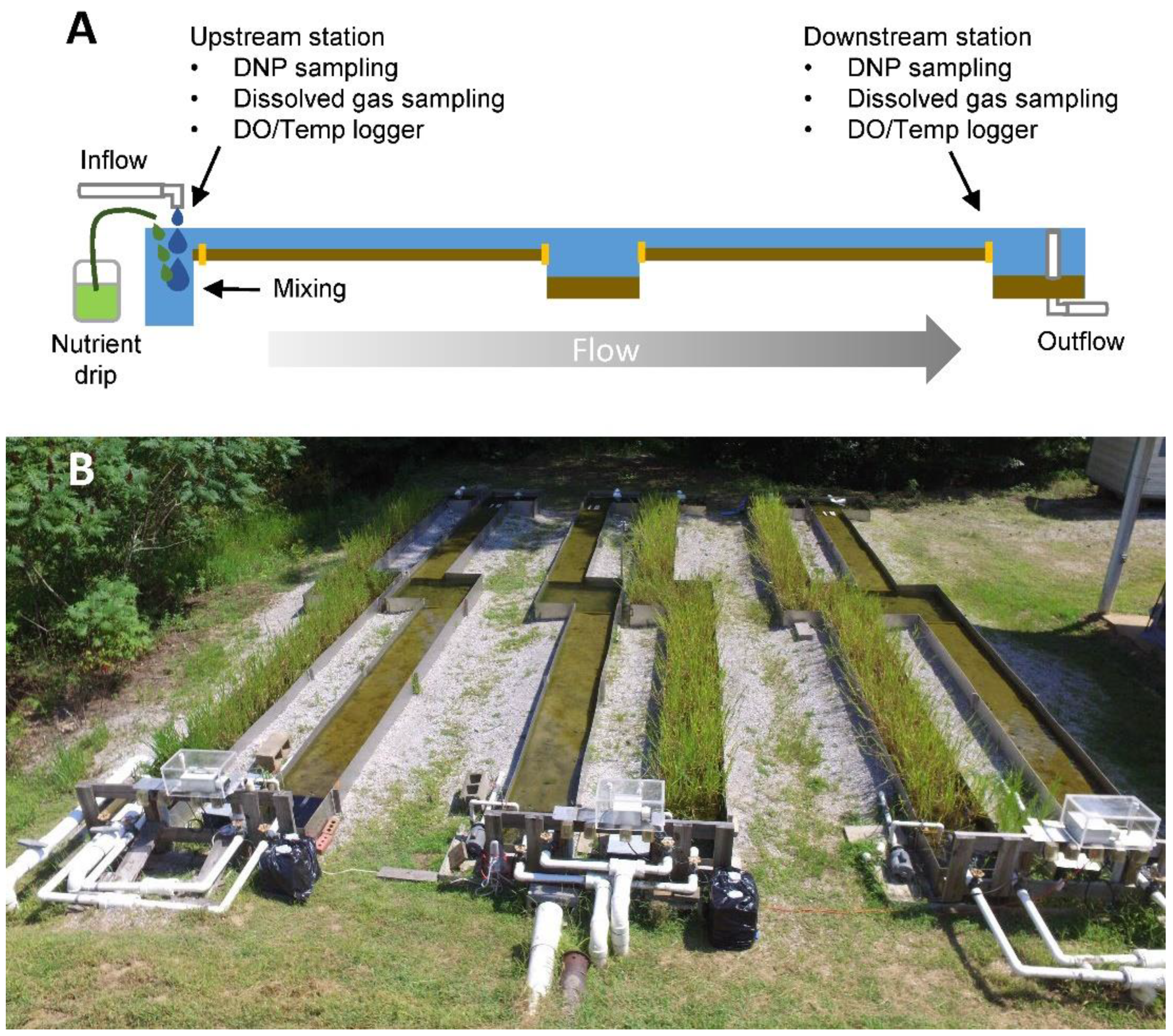
2.1. Study Site
2.2. Dissolved Nutrient Sampling
2.3. Reach-Scale Metabolism Estimates
2.4. Denitrification Estimates
2.5. Vegetation and Sediment Data Analysis
2.6. Statistical Analyses
3. Results
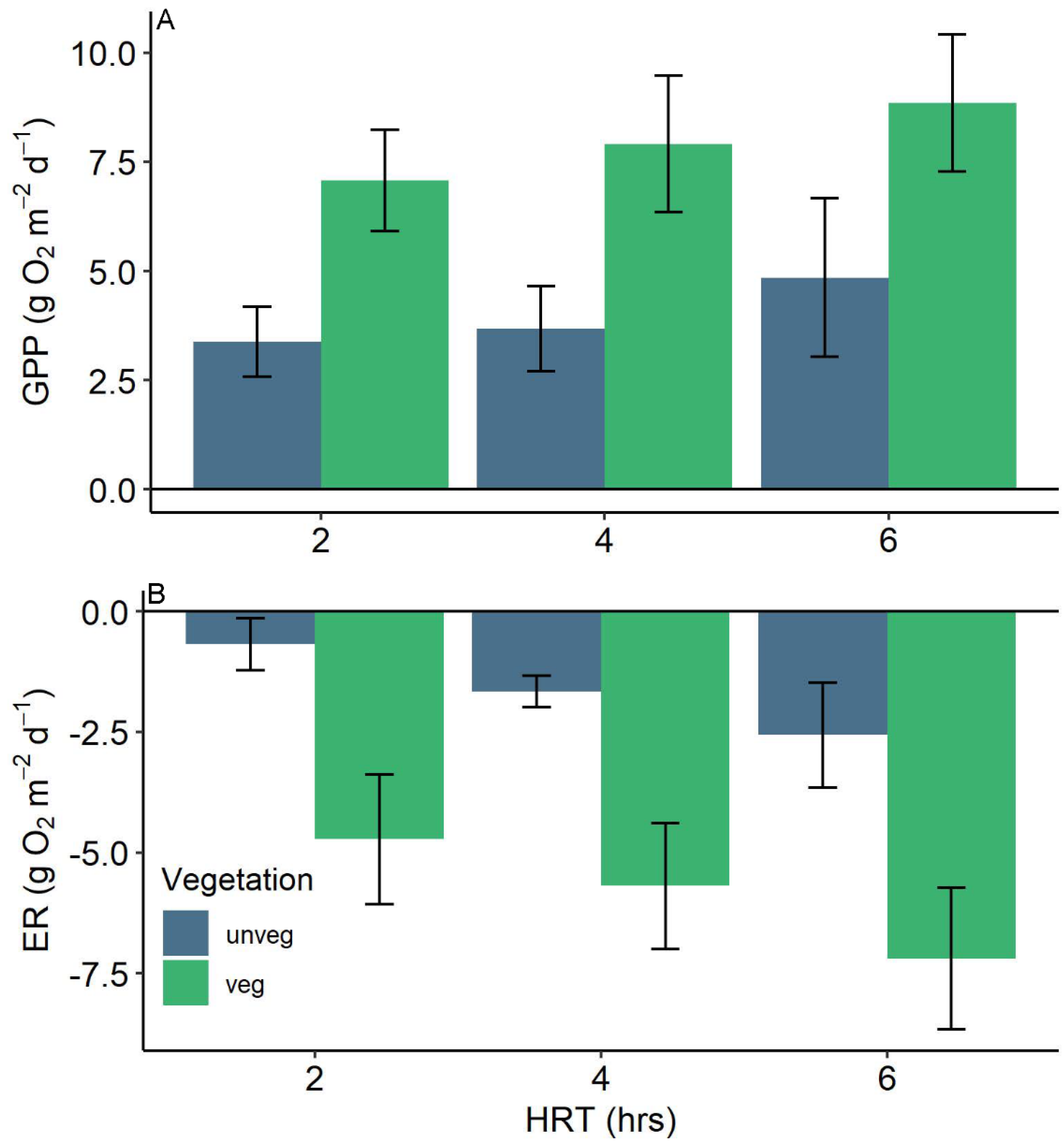
3.1. Agroecosystem Gas Fluxes
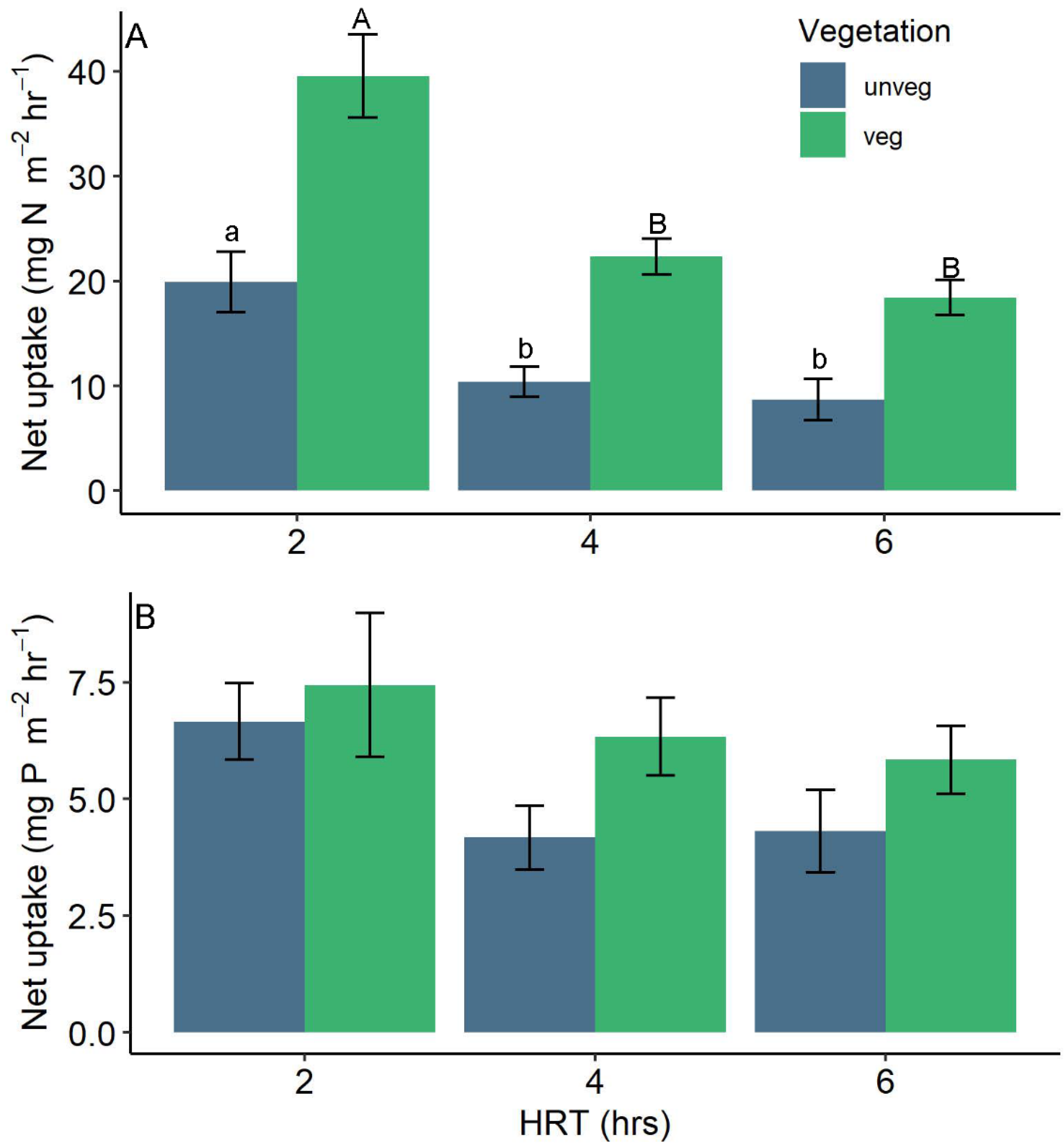
3.2. Nutrient Uptake
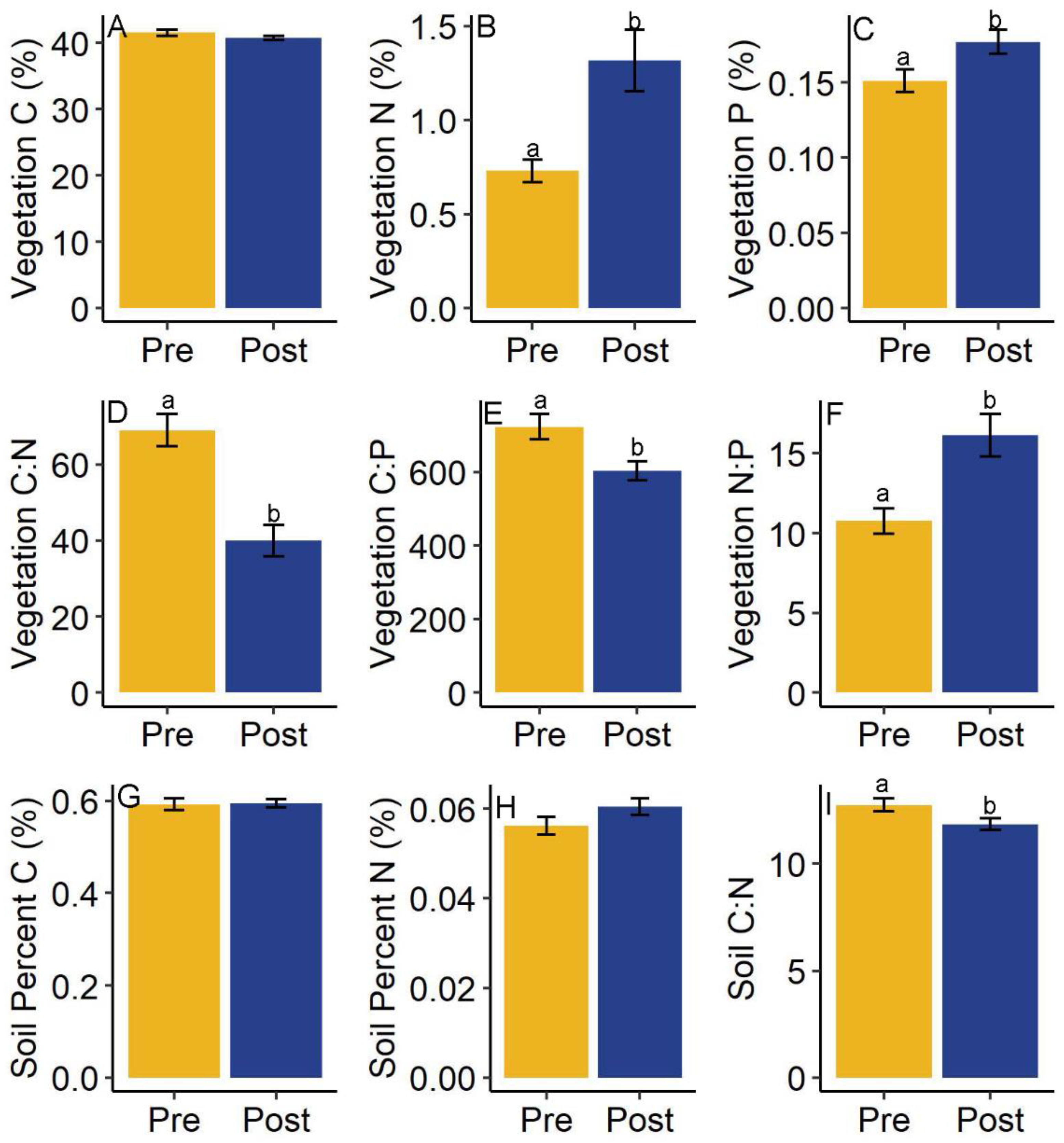
3.3. Vegetation and Sediment
4. Discussion
4.1. Vegetation
4.2. Residence Time
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Alexander, R.B.; Smith, R.A.; Schwarz, G.E.; Boyer, E.W.; Nolan, J.V.; Brakebill, J.W. Differences in phosphorus and nitrogen delivery to the Gulf of Mexico from the Mississippi River basin. Environ. Sci. Technol. 2008, 42, 822–830. [Google Scholar] [CrossRef]
- Stoddard, J.L.; Van Sickle, J.; Herlihy, A.T.; Brahney, J.; Paulsen, S.; Peck, D.V.; Mitchell, R.; Pollard, A.I. Continental-scale increase in lake and stream phosphorus: Are oligotrophic systems disappearing in the United States? Environ. Sci. Technol. 2016, 50, 3409–3415. [Google Scholar] [CrossRef]
- Rabalais, N.N.; Turner, R.E. Gulf of Mexico hypoxia: Past, present, and future. Limnol. Oceano. Bull. 2019, 28, 117–124. [Google Scholar] [CrossRef]
- Pierce, S.C.; Kröger, R.; Pezeshki, R. Managing artificially drained low-gradient agricultural headwaters for enhanced ecosystem functions. Biology 2012, 1, 794–856. [Google Scholar] [CrossRef]
- King, S.L.; Keeland, B.D. Evaluation of reforestation in the Lower Mississippi River Alluvial Valley. Restor. Ecol. 1999, 7, 348–359. [Google Scholar] [CrossRef]
- Lassaletta, L.; Billen, G.; Grizzetti, B.; Anglade, J.; Garnier, J. 50 year trends in nitrogen use efficiency of world cropping systems: The relationship between yield and nitrogen input to cropland. Environ. Res. Lett. 2014, 9, 105011. [Google Scholar] [CrossRef]
- Canfield, D.E.; Glazer, A.N.; Falkowski, P.G. The evolution and future of Earth’s N cycle. Science 2010, 330, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.; Acreman, M.C. Wetland nutrient removal: A review of the evidence. Hydrol. Earth Syst. Sci. 2004, 8, 673–685. [Google Scholar] [CrossRef]
- Vymazal, J. Plants used in constructed wetlands with horizontal subsurface flow: A review. Hydrobiologia 2011, 674, 133–156. [Google Scholar] [CrossRef]
- Vymazal, J. The use of constructed wetlands for nitrogen removal from agricultural drainage: A review. Sci. Agric. Bohem. 2017, 48, 82–91. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, D.; Dong, J.; Tan, S.K. Application of constructed wetlands for treating agricultural runoff and agro-industrial wastewater: A review. Hydrobiologia 2018, 805, 1–31. [Google Scholar] [CrossRef]
- Li, J.; Yang, X.; Wang, Z.; Shan, Y.; Zheng, Z. Comparison of four aquatic plant treatment systems for nutrient removal from eutrophied water. Bioresour. Technol. 2015, 179, 1–7. [Google Scholar] [CrossRef]
- Soana, E.; Balestrini, R.; Vincenzi, F.; Bartoli, M.; Castaldelli, G. Mitigation of nitrogen pollution in vegetated ditches fed by nitrate-rich spring waters. Agric. Ecosyst. Environ. 2017, 243, 74–82. [Google Scholar] [CrossRef]
- Lu, B.; Xu, Z.; Li, J.; Chai, X. Removal of water nutrients by different aquatic plant species: An alternative way to remediate polluted rural rivers. Ecol. Eng. 2018, 110, 18–26. [Google Scholar] [CrossRef]
- Kröger, R.; Holland, M.M.; Moore, M.T.; Cooper, C.M. Hydrological variability and agricultural drainage ditch inorganic nitrogen reduction capacity. J. Environ. Qual. 2007, 36, 1646–1652. [Google Scholar] [CrossRef]
- Herzon, I.; Helenius, J. Agricultural drainage ditches, their biological importance and functioning. Biol. Conserv. 2008, 141, 1171–1183. [Google Scholar] [CrossRef]
- Dollinger, J.; Dagès, C.; Bally, J.; Lagacherie, P.; Voltz, M. Managing ditches for agroecological engineering of landscape. A review. Agron. Sustain. Dev. 2015, 35, 999–1020. [Google Scholar] [CrossRef]
- Kroger, R.; Moore, M.T.; Locke, M.A.; Cullum, R.F.; Steinreide, R.W., Jr.; Testa, S., III; Bryant, C.T.; Cooper, C.M. Evaluating the influence of wetland vegetation on chemical residence time in Mississippi Delta drainage ditches. Agric. Water Manag. 2009, 96, 1175–1179. [Google Scholar] [CrossRef]
- Baker, D.W.; Bledsoe, D.P.; Price, J.M. Stream nitrate uptake and transient storage over a gradient of geomorphic complexity, north-central Colorado, USA. Hydrol. Process. 2012, 26, 3241–3252. [Google Scholar] [CrossRef]
- Cunha, D.G.F.; Finkler, N.R.; Calijuri, M.C.; Covino, T.P.; Tromboni, F.; Dodds, W.K. Nutrient uptake in a simplified stream channel: Experimental manipulation of hydraulic residence time and transient storage. Ecohydrology 2018, 11, e2012. [Google Scholar] [CrossRef]
- Davis, R.T.; Tank, J.L.; Mahl, U.H.; Winikoff, S.G.; Roley, S.S. The Influence of two-stage ditches with constructed floodplains on water column nutrients and sediments in agricultural streams. J. Am. Water Resour. Assoc. 2015, 51, 941–955. [Google Scholar] [CrossRef]
- Hodaj, A.; Bowling, L.C.; Frankenberger, J.R.; Chaubey, I. Impact of a two-stage ditch on channel water quality. Agric. Water Manag. 2017, 192, 126–137. [Google Scholar] [CrossRef]
- Kröger, R.; Cooper, C.M.; Moore, M.T. A preliminary study of an alternative controlled drainage strategy in surface drainage ditches: Low-grade weirs. Agric. Water Manag. 2008, 95, 678–684. [Google Scholar] [CrossRef]
- Jadhav, R.S.; Buchberger, S.G. Effects of vegetation on flow through free water surface wetlands. Ecol. Eng. 1995, 5, 481–496. [Google Scholar] [CrossRef]
- Bouldin, J.L.; Farris, J.L.; Moore, M.T.; Smith, S., Jr.; Stephens, W.W.; Cooper, C.M. Evaluated fate and effects of atrazine and lambda-cyhalothrin in vegetated and unvegetated microcosms. Environ. Tox. 2005, 20, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Darby, S.E. Effect of riparian vegetation on flow resistance and flood potential. J. Hydraul. Eng. 1999, 125, 443–454. [Google Scholar] [CrossRef]
- Nepf, H.M. Drag, turbulence, and diffusion in flow through emergent vegetation. J. Water Resour. Res. 1999, 35, 479–489. [Google Scholar] [CrossRef]
- Madsen, J.D.; Chambers, P.A.; James, W.F.; Koch, E.W.; Westlake, D.F. The interaction between water movement, sediment dynamics and submersed macrophytes. Hydrobiologia 2001, 444, 71–84. [Google Scholar] [CrossRef]
- Cooper, C.M.; Moore, M.T.; Bennett, E.R.; Smith, S., Jr.; Farris, J.L.; Milam, C.D.; Shields, F.D., Jr. Innovative uses of vegetated drainage ditches for reducing agricultural runoff. Water Sci. Technol. 2004, 49, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Strock, J.S.; Dell, C.J.; Schmidt, J.P. Managing natural processes in drainage ditches for nonpoint source nitrogen control. J. Soil Water Conserv. 2007, 62, 188–196. [Google Scholar]
- Sharpley, A.N.; Krogstad, T.; Kleinman, P.J.A.; Haggard, B.; Shigaki, F.; Saporito, L.S. Managing natural processes in drainage ditches for nonpoint source phosphorus control. J. Soil Water Conserv. 2007, 62, 197–206. [Google Scholar]
- Moore, M.T.; Kröger, R.; Locke, M.A.; Cullum, R.F.; Steinriede, R.W., Jr.; Testa, S., III; Lizotte, R.E., Jr.; Bryant, C.T.; Cooper, C.M. Nutrient mitigation capacity in Mississippi Delta, USA drainage ditches. Environ. Pollut. 2010, 158, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Durand, P.; Breuer, L.; Johnes, P.J.; Billen, G.; Butturini, A.; Pinay, G.; van Grinsven, H.; Garnier, J.; Rivett, M.; Reay, D.S.; et al. Nitrogen processes in aquatic ecosystems. In European Nitrogen Assessment; Cambridge University Press: Cambridge, UK, 2011; pp. 126–146. [Google Scholar]
- Hall, R.O.; Tank, J.L. Ecosystem metabolism controls nitrogen uptake in streams in Grand Teton National Park, Wyoming. Limnol. Oceanogr. 2003, 48, 1120–1128. [Google Scholar] [CrossRef]
- Kaenel, B.R.; Buehrer, H.; Uehlinger, U. Effects of aquatic plant management on stream metabolism and oxygen balance in streams. Freshw. Biol. 2001, 45, 85–95. [Google Scholar] [CrossRef]
- Taylor, J.M.; Moore, M.T.; Scott, J.T. Contrasting nutrient mitigation and denitrification potential of agricultural drainage environments with different emergent aquatic macrophytes. J. Environ. Qual. 2015, 44, 1304–1314. [Google Scholar] [CrossRef]
- Speir, S.L.; Taylor, J.M.; Scott, J.T. Seasonal differences in relationships between nitrate concentration and denitrification rates in ditch sediments vegetated with rice cutgrass. J. Environ. Qual. 2017, 46, 1500–1509. [Google Scholar] [CrossRef]
- Needelman, B.A.; Kleinman, P.J.A.; Strock, J.S.; Allen, A.L. Improved management of agricultural drainage ditches for water quality protection: An overview. J. Soil Water Conserv. 2007, 62, 171–178. [Google Scholar]
- Taylor, J.M.; Moore, M.T.; Speir, S.L.; Testa, S., III. Vegetated ditch habitats provide net nitrogen sink and phosphorus storage capacity in agricultural drainage networks despite senescent plant leaching. Water 2020, 12, 875. [Google Scholar] [CrossRef]
- Nguyen, L.; Sukias, J. Phosphorus fractions and retention in drainage ditch sediments receiving surface runoff and subsurface drainage from agricultural catchments in the North Island, New Zealand. Agric. Ecosyst. Environ. 2002, 92, 49–69. [Google Scholar] [CrossRef]
- Collins, S.D.; Shukla, S.; Shresta, N.K. Drainage ditches have sufficient adsorption capacity but inadequate residence time for phosphorus retention in the Everglades. Ecol. Eng. 2016, 92, 218–228. [Google Scholar] [CrossRef]
- Vzmazal, J.; Březinová, T.D. Removal of nutrients, organics and suspended solids in vegetated agricultural drainage ditch. Ecol. Eng. 2018, 118, 97–103. [Google Scholar] [CrossRef]
- Nifong, R.L.; Taylor, J.M.; Moore, M.T. To model or measure: Estimating gas exchange to measure metabolism in shallow, low-gradient stream habitats. Freshw. Sci. 2020, 39, 70–85. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association; American Water Works Association; Water Environment Federation: Washington, DC, USA, 2005.
- Heffernan, J.B.; Cohen, M.J. Direct and indirect coupling of primary production and diel nitrate dynamics in a subtropical spring-fed river. Limnol. Oceanogr. 2010, 55, 677–688. [Google Scholar] [CrossRef]
- Marzolf, E.R.; Mulholland, P.J.; Steinman, A.D. Improvements to the diurnal upstream–downstream dissolved oxygen change technique for determining whole-stream metabolism in small streams. Can. J. Fish. Aquat. Sci. 1994, 51, 1591–1599. [Google Scholar] [CrossRef]
- Young, R.G.; Huryn, A.D. Comment: Improvements to the diurnal upstream-downstream dissolved oxygen change technique for determing whole-stream metabolism in small streams. Can. J. Fish. Aquat. Sci. 1998, 55, 1784–1785. [Google Scholar] [CrossRef]
- Hall, R.O.; Hotchkiss, E.R. Stream Metabolism. In Methods in Stream Ecology; Academic Press: Cambridge, MA, USA, 2017; pp. 219–233. [Google Scholar]
- Nifong, R.L.; Taylor, J.M.; Adams, G.; Moore, M.T.; Farris, J.L. Recognizing both denitrification and nitrogen consumption improves performance of stream diel N2 flux models. Limno. Oceanogr. Meth. 2020, 18, 169–182. [Google Scholar] [CrossRef]
- Hall, R.O.; Tank, J.L.; Baker, M.A.; Rosi-Marshall, E.J.; Hotchkiss, E.R. Metabolism, gas exchange, and carbon spiraling in rivers. Ecosystems 2016, 19, 73–86. [Google Scholar] [CrossRef]
- Geyer, C.J.; Johnson, L.T. MCMC: Markov Chain Monte Carlo. Package Version 0.9-2. 2019. Available online: https://cran.r-project.org/web/packages/mcmc/mcmc.pdf (accessed on 30 March 2021).
- Appling, A.P.; Hall, R.O.; Yackulic, C.B.; Arroita, M. Overcoming equifinality: Leveraging long time series for stream metabolism estimation. J. Geophys. Res. Biogeosci. 2018, 123, 624–645. [Google Scholar] [CrossRef]
- Reisinger, A.J.; Tank, J.L.; Hoellein, T.J.; Hall, R.O., Jr. Sediment, water column, and open-channel denitrification in rivers measured using membrane-inlet mass spectrometry. J. Geophys. Res. Biogeosci. 2016, 121, 1258–1274. [Google Scholar] [CrossRef]
- Hall, R.O.; Madinger, H.L. Use of argon to measure gas exchange in turbulent mountain streams. Biogeosciences 2018, 15, 3085–3092. [Google Scholar] [CrossRef]
- Kana, T.M.; Darkangelo, C.; Hunt, M.D.; Oldham, J.B.; Bennett, G.E.; Cornwell, J.C. Membrane inlet mass spectrometer for rapid high-precision determination of N2, O2, and Ar in environmental water samples. Anal. Chem. 1994, 66, 4166–4170. [Google Scholar] [CrossRef]
- Grantz, E.M.; Kogo, A.; Scott, J.T. Partitioning whole-lake denitrification using in situ dinitrogen gas accumulation and intact sediment core experiments. Limnol. Oceanogr. 2012, 57, 925–935. [Google Scholar] [CrossRef]
- Weiss, R.F. The solubility of nitrogen, oxygen and argon in water and seawater. Deep-Sea Res. 1970, 17, 721–735. [Google Scholar] [CrossRef]
- Eyre, B.D.; Rysgaard, S.; Dalsgaard, T.; Christensen, P.B. Comparison of isotope pairing and N2:Ar methods for measuring sediment denitrification: Assumptions, modifications and implications. Estuaries 2002, 25, 1077–1087. [Google Scholar] [CrossRef]
- Jähne, B.; Heinz, G.; Dietrich, W. Measurement of the diffusion coefficients of sparingly soluble gases in water. J. Geophys. Res. 1987, 92, 10767–10776. [Google Scholar] [CrossRef]
- Reichert, P.; Uehlinger, U.; Acuna, V. Estimating stream metabolism from oxygen concentrations: Effect of spatial heterogeneity. J. Geophys. Res. 2009, 114, G03016. [Google Scholar] [CrossRef]
- Halvorson, H.M.; White, G.; Scott, J.T.; Evans-White, M.A. Dietary and taxonomic controls on incorporation of microbial carbon and phosphorus by detritivorous caddisflies. Oecologia 2016, 180, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Zuur, A.F.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; Heisterkamp, S.; Van Willigen, B. Nlme: Linear and Nonlinear Mixed Effects Models. R Project for Statistical Computing: Vienna, Austria. Available online: https://cran.r-project.org/web/packages/nlme (accessed on 27 July 2018).
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. 2019. Available online: https://cran.r-project.org/web/packages/emmeans/emmeans.pdf (accessed on 31 March 2021).
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Rogers, K.H.; Breen, P.F.; Chick, A.J. Nitrogen removal in experimental wetland treatment systems: Evidence for the role of aquatic plants. J. Water Pollut. Control Fed. 1991, 63, 934–941. [Google Scholar]
- Kroger, R.; Holland, M.M.; Moore, M.T.; Cooper, C.M. Plant senescence: A mechanism for nutrient release in temperate agricultural wetlands. Environ. Pollut. 2007, 146, 114–119. [Google Scholar] [CrossRef]
- Kadlec, R.H. Pond and wetland treatment. Water Sci. Technol. 2003, 48, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tester, M.; Leigh, R.A. Partitioning of nutrient transport processes in roots. J. Exp. Bot. 2001, 52, 445–457. [Google Scholar] [CrossRef]
- Alnoee, A.B.; Riis, T.; Baatrrup-Pedersen, A. Comparison of metabolic rates among macrophyte and nonmacrophyte habitats in streams. Freshw. Sci. 2016, 35, 834–844. [Google Scholar] [CrossRef]
- Alnoee, A.B.; Levi, P.S.; Baattrup-Pedersen, A.B.; Riis, T. Macrophytes enhance reach-scale metabolism on a daily, seasonal and annual basis in agricultural lowland streams. Aquat. Sci. 2021, 83, 11. [Google Scholar] [CrossRef]
- Tyler, H.L.; Moore, M.T.; Locke, M.A. Influence of three aquatic macrophytes on mitigation of nitrogen species from agricultural runoff. Water Air Soil Pollut. 2012, 223, 3227–3236. [Google Scholar] [CrossRef]
- Roley, S.S.; Tank, J.L.; Griffiths, N.A.; Hall, R.O., Jr.; Davis, R.T. The influence of floodplain restoration on whole-stream metabolism in an agricultural stream: Insights from a 5-year continuous data set. Freshw. Sci. 2014, 33, 1043–1059. [Google Scholar] [CrossRef]
- Reilly, J.F.; Horne, A.J.; Miller, C.D. Nitrate removal from a drinking water supply with large free-surface constructed wetlands prior to groundwater recharge. Ecol. Eng. 2000, 14, 33–47. [Google Scholar] [CrossRef]
- Chang, J.; Wu, S.; Dai, Y.; Liang, W.; Wu, Z. Nitrogen removal from nitrate-laden wastewater by integrated vertical-flow constructed wetland systems. Ecol. Eng. 2013, 58, 192–201. [Google Scholar] [CrossRef]
- Acuna, V.; Vilches, C.; Giorgi, A. As productive and slow as a stream can be—The metabolism of a Pampean stream. Freshw. Sci. 2011, 30, 71–83. [Google Scholar]
- O’Donnell, B.; Hotchkiss, E.R. Coupling concentration- and process-discharge relationships integrates water chemistry and metabolism in streams. Water Resour. Res. 2019, 55, 10179–10190. [Google Scholar] [CrossRef]
- Poor, C.J.; McDonnell, J.J. The effects of land use on stream nitrate dynamics. J. Hydrol. 2007, 332, 54–68. [Google Scholar] [CrossRef]
- Carey, R.O.; Wollheim, W.M.; Mulukutla, G.K.; Mineau, M.M. Characterizing storm-event nitrate fluxes in a fifth order suburbanizing watershed using in situ sensors. Environ. Sci. Technol. 2014, 48, 7756–7765. [Google Scholar] [CrossRef] [PubMed]
- Castadelli, G.; Aschonitis, V.; Vincenzi, F.; Fano, E.A.; Soana, E. The effect of water velocity on nitrate removal in vegetated waterways. J. Environ. Manag. 2018, 215, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.; Ghisalberti, M.; Nepf, H. Model and laboratory study of dispersion in flows with submerged vegetation. J. Water Resour. Res. 2007, 43, W05438. [Google Scholar] [CrossRef]
- Usborne, E.L.; Kröger, R.; Pierce, S.C.; Brandt, J.; Goetz, D. Preliminary evidence of sediment and phosphorus dynamics behind newly installed low-grade weirs in agricultural drainage ditches. Water Air Soil Pollut. 2013, 224, 1520. [Google Scholar] [CrossRef]
- Sheibley, R.W.; Duff, J.H.; Tesoriero, A.J. Low transient storage and uptake efficiencies in seven agricultural streams. J. Environ. Qual. 2014, 43, 1980–1990. [Google Scholar] [CrossRef] [PubMed]
- Kröger, R.; Moore, M.T.; Farris, J.L.; Gopalan, M. Evidence for the use of flow-grade weirs in drainage ditches to improve nutrient reductions from agriculture. Water Air Soil Pollut. 2011, 221, 223–234. [Google Scholar] [CrossRef]
- Zhao, S.; Cui, Y.; Luo, Y.; Peifeng, L. Experimental study on wetland hydraulic characteristics of vegetated drainage ditches. Water 2017, 9, 311. [Google Scholar] [CrossRef]
- Yasarer, L.M.W.; Bingner, R.L.; Garbrecht, J.D.; Locke, M.A.; Lizotte, R., Jr.; Momm, H.G.; Busteed, P.R. Climate change impacts on runoff, sediment, and nutrient loads in an agricultural watershed in the lower Mississippi River Basin. App. Eng. Agric. 2017, 33, 379–392. [Google Scholar] [CrossRef]
- Lizotte, R.E., Jr.; Yasarer, L.M.W.; Locke, M.A.; Bingner, R.L.; Knight, S.S. Lake nutrient responses to integrated conservation practices in an agricultural watershed. J. Environ. Qual. 2017, 46, 330–338. [Google Scholar] [CrossRef] [PubMed]
| 2 h HRT | 4 h HRT | 6 h HRT | ||||
|---|---|---|---|---|---|---|
| Stream Fluxes (g) | Veg | UnVeg | Veg | UnVeg | Veg | UnVeg |
| NO3-N load | 15.5 ± 0.6 | 15.5 ± 0.6 | 15.5 ± 0.6 | 15.5 ± 0.6 | 15.5 ± 0.6 | 15.5 ± 0.6 |
| NO3-N net uptake | 8.4 ± 0.9 | 4.2 ± 0.6 | 4.8 ± 0.4 | 2.2 ± 0.3 | 3.9 ± 0.4 | 1.9 ± 0.4 |
| Denitrification | 1.3 ± 0.3 | 0.7 ± 0.0 | 2.0 ± 0.8 | 1.0 ± 0.1 | 1.1 ± 0.2 | 0.8 ± 0.2 |
| % N removal | 63% | 32% | 44% | 21% | 32% | 17% |
| PO4-P load | 9.9 ± 0.4 | 9.9 ± 0.4 | 9.9 ± 0.4 | 9.9 ± 0.4 | 9.9 ± 0.4 | 9.9 ± 0.4 |
| PO4-P net uptake | 1.6 ± 0.3 | 1.4 ± 0.2 | 1.4 ± 0.2 | 0.9 ± 0.3 | 1.3 ± 0.2 | 0.8 ± 0.2 |
| % P removal | 16% | 14% | 14% | 10% | 13% | 8% |
| Nutrient | Pre | Post | p |
|---|---|---|---|
| Percent C | 30.23 ± 2.32 | 34.92 ± 1.63 | 0.12 |
| Percent N | 0.75 ± 0.09 | 0.90 ± 0.04 | 0.18 |
| Percent P | 0.17 ± 0.01 | 0.18 ± 0.01 | 0.40 |
| C:N | 51.64 ± 5.49 | 45.98 ± 2.62 | 0.37 |
| C:P | 475.92 ± 34.90 | 506.43 ± 23.70 | 0.48 |
| N:P | 10.02 ± 1.27 | 11.29 ± 0.81 | 0.41 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
DuBose, R.L.; Taylor, J.M. Vegetation and Residence Time Interact to Influence Metabolism and Net Nutrient Uptake in Experimental Agricultural Drainage Systems. Water 2021, 13, 1416. https://doi.org/10.3390/w13101416
DuBose RL, Taylor JM. Vegetation and Residence Time Interact to Influence Metabolism and Net Nutrient Uptake in Experimental Agricultural Drainage Systems. Water. 2021; 13(10):1416. https://doi.org/10.3390/w13101416
Chicago/Turabian StyleDuBose, Rachel L., and Jason M. Taylor. 2021. "Vegetation and Residence Time Interact to Influence Metabolism and Net Nutrient Uptake in Experimental Agricultural Drainage Systems" Water 13, no. 10: 1416. https://doi.org/10.3390/w13101416
APA StyleDuBose, R. L., & Taylor, J. M. (2021). Vegetation and Residence Time Interact to Influence Metabolism and Net Nutrient Uptake in Experimental Agricultural Drainage Systems. Water, 13(10), 1416. https://doi.org/10.3390/w13101416






