Performance of a Handheld Chlorophyll-a Fluorometer: Potential Use for Rapid Algae Monitoring
Abstract
1. Introduction
2. Methods
2.1. Apparatus Description
2.2. Sampling and Laboratory Experiment
2.3. Statistical Analyses
3. Results
3.1. Algal Taxa and Concentrations in Lakeside and Silverbell Lakes
3.2. Sensitivity to Ambient Light
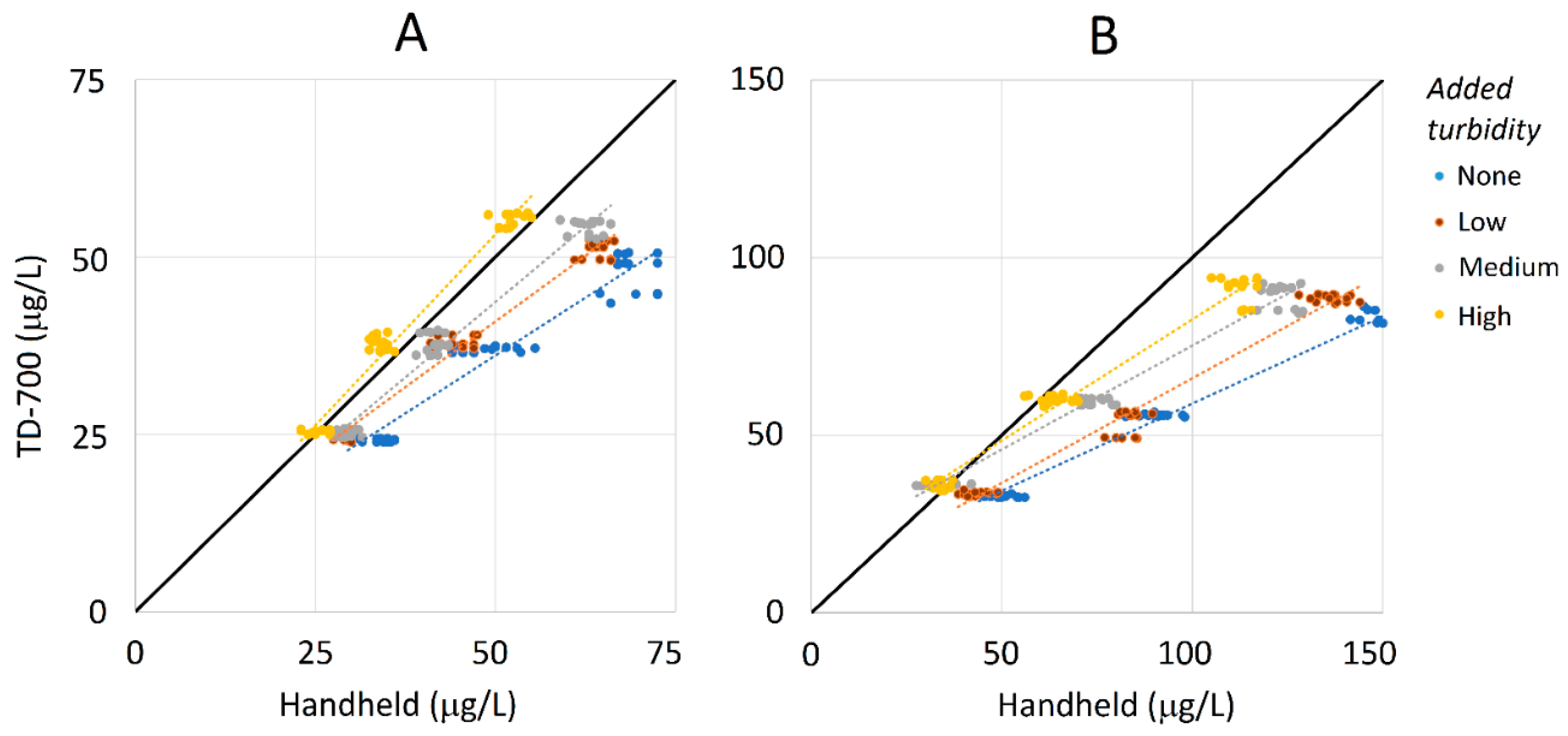
3.3. Sensitivity to Turbidity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elliott, J.A.; Jones, I.D.; Thackeray, S.J. Testing the Sensitivity of Phytoplankton Communities to Changes in Water Temperature and Nutrient Load, in a Temperate Lake. Hydrobiologia 2006, 559, 401–411. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Otten, T.G. Harmful Cyanobacterial Blooms: Causes, Consequences, and Controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef]
- Zhu, Y.; McCowan, A.; Cook, P.L. Effects of changes in nutrient loading and composition on hypoxia dynamics and internal nutrient cycling of a stratified coastal lagoon. Biogeosciences 2017, 14, 4423–4433. [Google Scholar] [CrossRef]
- Ibelings, B.W.; Backer, L.C.; Kardinaal, W.E.A.; Chorus, I. Current approaches to cyanotoxin risk assessment and risk management around the globe. Harmful Algae 2014, 40, 63–74. [Google Scholar] [CrossRef]
- Hollister, J.W.; Kreakie, B.J. Associations between chlorophyll a and various microcystin health advisory concentrations. F1000Research 2016, 5, 151. [Google Scholar] [PubMed]
- Arar, E.J.; Collins, G.B. Method 445.0 In Vitro Determination of Chlorophyll a and Pheophytin a in Marine and Freshwater Algae by Fluorescence; United States Environmental Protection Agency, Office of Research and Development, National Exposure Research Laboratory: Cincinnati, OH, USA, 1997; p. 22.
- Almomani, F.A.; Örmeci, B. Monitoring and measurement of microalgae using the first derivative of absorbance and comparison with chlorophyll extraction method. Environ. Monit. Assess. 2018, 190, 90. [Google Scholar] [CrossRef]
- Loftus, M.E.; Seliger, H.H. Some Limitations of the In Vivo Fluorescence Technique. Chesap. Sci. 1975, 16, 79–92. [Google Scholar] [CrossRef]
- Porter, G.; Synowiec, J.A.; Tredwell, C.J. Intensity effects on the fluorescence of in vivo chlorophyll. Biochim. Biophys. Acta BBA Bioenerg. 1977, 459, 329–336. [Google Scholar] [CrossRef]
- Ghadouani, A.; Smith, R.E. Phytoplankton distribution in Lake Erie as assessed by a new in situ spectrofluorometric technique. J.Gt. Lakes Res. 2005, 31, 154–167. [Google Scholar] [CrossRef]
- Lu, J.; Yuan, Y.; Duan, Z.; Zhao, G.; Svanberg, S. Short-range remote sensing of water quality by a handheld fluorosensor system. Appl. Opt. 2020, 59, C1–C7. [Google Scholar] [CrossRef] [PubMed]
- Salonen, K.; Sarvala, J.; Järvinen, M.; Langenberg, V.; Nuottajärvi, M.; Vuorio, K.; Chitamwebwa, D.B.R. Phytoplankton in Lake Tanganyika—Vertical and horizontal distribution of in vivo fluorescence. In From Limnology to Fisheries: Lake Tanganyika and Other Large Lakes; Springer: Dordrecht, The Netherlands, 1999; pp. 89–103. [Google Scholar]
- Cremella, B.; Huot, Y.; Bonilla, S. Interpretation of total phytoplankton and cyanobacteria fluorescence from cross-calibrated fluorometers, including sensitivity to turbidity and colored dissolved organic matter. Limnol. Oceanogr. Methods 2018, 16, 881–894. [Google Scholar] [CrossRef]
- Zamyadi, A.; McQuaid, N.; Dorner, S.; Bird, D.F.; Burch, M.; Baker, P.; PRÉvost, M. Cyanobacterial detection using in vivo fluorescence probes: Managing interferences for improved decision–making. J. Am. Water Work. Assoc. 2012, 104, E466–E479. [Google Scholar] [CrossRef]
- Cui, J.S.; Lv, P.Y. Turbidity effect on the fluorescence determination of chlorophyll-a in water. In Applied Mechanics and Materials; Trans Tech Publications Ltd.: Bach, Switzerland, 2014; Volume 522, pp. 62–63. [Google Scholar]
- Laney, S.R. In Situ Measurement of Variable Fluorescence Transients. In Chlorophyll a Fluorescence in Aquatic Sciences: Methods and Applications; Springer: Dordrecht, The Netherlands, 2010; pp. 19–30. [Google Scholar]
- Leeuw, T.; Boss, E.S.; Wright, D.L. In Situ Measurements of Phytoplankton Fluorescence Using Low Cost Electronics. Sensors 2013, 13, 7872–7883. [Google Scholar] [CrossRef] [PubMed]
- Strass, V. On the calibration of large-scale fluorometric chlorophyll measurements from towed undulating vehicles. Deep. Sea Res. Part A Oceanogr. Res. Pap. 1990, 37, 525–540. [Google Scholar] [CrossRef]
- Vincent, W.F. Photosynthetic capacity measured by DCMU-induced chlorophyll fluorescence in an oligotrophic lake. Freshw. Biol. 1981, 11, 61–78. [Google Scholar] [CrossRef]
- Eppehimer, D.E.; Hamdhani, H.; Hollien, K.D.; Bogan, M.T. Evaluating the potential of treated effluent as novel habitats for aquatic invertebrates in arid regions. Hydrobiologia 2020, 847, 3381–3396. [Google Scholar] [CrossRef]
- Wehr, J.D.; Sheath, R.G.; Kociolek, J.P. (Eds.) Freshwater Algae of North America: Ecology and Classification; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Stevenson, R.J.; Bahls, L.L. Periphyton protocols. In Rapid Bioassessment Protocols for Use in Wadeable Streams and Rivers: Periphyton, Benthic Macroinvertebrates, and Fish; US Environmental Protection Agency, Office of Water: Washington, DC, USA, 1999. [Google Scholar]
- Chang, D.-W.; Hobson, P.; Burch, M.; Lin, T.-F. The limitation of measurement in cyanobacteria using in-vivo fluoroscopy. In Proceedings of the 2011 Seventh International Conference on Intelligent Sensors, Adelaide, Australia, 6–9 December 2011; pp. 184–188. [Google Scholar]
- Stata Statistical Software; Release 15; StataCorp LLC: College Station, TX, USA, 2017.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 10 July 2020).
- Rovati, L.; Docchio, F. Low-noise front-end electronics for solid-state fluorometers. Rev. Sci. Instrum. 1999, 70, 3759–3764. [Google Scholar] [CrossRef]
- Brient, L.; Lengronne, M.; Bertrand, E.; Rolland, D.; Sipel, A.; Steinmann, D.; Baudin, I.; Legeas, M.; Le Rouzic, B.; Bormans, M. A phycocyanin probe as a tool for monitoring cyanobacteria in freshwater bodies. J. Environ. Monit. 2008, 10, 248–255. [Google Scholar] [CrossRef]
- Zamyadi, A.; Choo, F.; Newcombe, G.; Stuetz, R.; Henderson, R.K. A review of monitoring technologies for real-time management of cyanobacteria: Recent advances and future direction. TrAC Trends Anal. Chem. 2016, 85, 83–96. [Google Scholar] [CrossRef]
- Van Nieuwenhuyse, E.E.; Jones, J.R. Phosphorus chlorophyll relationship in temperate streams and its variation with stream catchment area. Can. J. Fish. Aquat. Sci. 1996, 53, 99–105. [Google Scholar] [CrossRef]
- Istvánovics, V. Eutrophication of lakes and reservoirs. In Lake Ecosystem Ecology; Elsevier: San Diego, CA, USA, 2010; pp. 47–55. [Google Scholar]
- Hudson, N.; Baker, A.; Reynolds, D. Fluorescence analysis of dissolved organic matter in natural, waste and polluted waters—A review. River Res. Appl. 2007, 23, 631–649. [Google Scholar] [CrossRef]
- Pip, E.; Bowman, L. Microcystin and Algal Chlorophyll in Relation to Nearshore Nutrient Concentrations in Lake Winnipeg, Canada. Environ. Pollut. 2014, 3, 36–47. [Google Scholar] [CrossRef][Green Version]
- Yuan, L.L.; Pollard, A.I.; Pather, S.; Oliver, J.L.; Anglada, L.D. Managing microcystin: Identifying national–scale thresholds for total nitrogen and chlorophyll a. Freshw. Biol. 2014, 59, 1970–1981. [Google Scholar] [CrossRef]


| Phylum | Genus | Quantity (Units/mL) |
|---|---|---|
| Lakeside Lake | ||
| Chlorophyta | Dictyosphaerium | 3400 |
| Chlorophyta | Chlamydomonas | 3000 |
| Chlorophyta | Scenedesmus | 2800 |
| Pyrrophyta | Gymnodinium | 2200 |
| Chrysophyta | Fragilaria | 2000 |
| Chrysophyta | Cymbella | 800 |
| Cyanobacteria | Microcystis | 400 |
| Cyanobacteria | Oscillatoria | 200 |
| Silverbell Lake | ||
| Cyanobacteria | Microcystis | 32,600 |
| Sample Origin | Algal Concentration | Turbidity Added | Turbidity | Light Treatment | Dark Treatment | t-Test (p-Value) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (NTU) | Std. Dev. | Mean | Std. Dev. | Min | Max | Mean | Std. Dev. | Min | Max | ||||
| Lakeside Lake | High | None | 21.48 | 0.18 | 71.00 | 3.08 | 67 | 74 | 66.20 | 3.27 | 62 | 71 | >0.05 |
| Low | 45.39 | 1.61 | 66.00 | 2.45 | 63 | 69 | 63.60 | 2.97 | 59 | 67 | >0.05 | ||
| Medium | 61.00 | 1.58 | 63.80 | 1.79 | 62 | 66 | 62.40 | 3.65 | 58 | 67 | >0.05 | ||
| High | 82.20 | 2.86 | 51.20 | 1.30 | 49 | 52 | 54.00 | 2.74 | 50 | 57 | >0.05 | ||
| Medium | None | 16.65 | 0.74 | 47.00 | 2.55 | 44 | 50 | 50.60 | 5.86 | 43 | 57 | >0.05 | |
| Low | 42.64 | 0.48 | 44.00 | 3.55 | 40 | 48 | 44.80 | 3.56 | 39 | 48 | >0.05 | ||
| Medium | 72.00 | 1.87 | 42.00 | 2.24 | 39 | 45 | 40.40 | 1.67 | 39 | 43 | >0.05 | ||
| High | 90.80 | 1.30 | 34.60 | 1.52 | 33 | 37 | 33.20 | 1.30 | 32 | 35 | >0.05 | ||
| Low | None | 12.13 | 1.19 | 34.40 | 2.07 | 31 | 36 | 32.60 | 2.70 | 28 | 35 | <0.05 * | |
| Low | 34.00 | 0.38 | 29.20 | 0.84 | 28 | 30 | 28.20 | 1.10 | 27 | 30 | >0.05 | ||
| Medium | 52.80 | 0.84 | 30.20 | 1.64 | 28 | 32 | 28.40 | 1.52 | 27 | 30 | >0.05 | ||
| High | 71.00 | 0.71 | 25.00 | 2.00 | 23 | 27 | 26.20 | 1.10 | 25 | 27 | >0.05 | ||
| Silverbell Lake | High | None | 12.28 | 0.46 | 151.80 | 8.24 | 139 | 170 | 148.60 | 7.24 | 133 | 156 | >0.05 |
| Low | 34.03 | 0.46 | 136.07 | 7.37 | 122 | 150 | 136.67 | 6.86 | 126 | 148 | >0.05 | ||
| Medium | 56.60 | 0.55 | 121.67 | 5.84 | 114 | 134 | 125.13 | 7.38 | 112 | 138 | >0.05 | ||
| High | 89.00 | 1.00 | 112.73 | 3.71 | 105 | 118 | 111.33 | 5.02 | 104 | 124 | >0.05 | ||
| Medium | None | 4.408 | 0.08 | 91.93 | 7.31 | 78 | 102 | 88.80 | 5.28 | 77 | 100 | >0.05 | |
| Low | 35.68 | 1.05 | 83.47 | 3.58 | 78 | 90 | 82.40 | 5.05 | 74 | 89 | >0.05 | ||
| Medium | 61.40 | 0.55 | 75.73 | 4.25 | 71 | 85 | 72.20 | 6.06 | 66 | 85 | >0.05 | ||
| High | 97.80 | 1.48 | 64.67 | 6.22 | 55 | 76 | 62.67 | 4.43 | 55 | 70 | >0.05 | ||
| Low | None | 3.08 | 0.12 | 51.53 | 6.00 | 41 | 63 | 47.33 | 5.42 | 39 | 61 | >0.05 | |
| Low | 46.16 | 0.32 | 43.07 | 2.76 | 39 | 48 | 43.47 | 4.75 | 37 | 50 | >0.05 | ||
| Medium | 95.00 | 1.58 | 33.93 | 4.18 | 29 | 43 | 31.73 | 3.73 | 26 | 41 | <0.05 * | ||
| High | 118.00 | 1.00 | 34.47 | 2.29 | 30 | 38 | 33.27 | 2.60 | 30 | 38 | >0.05 | ||
| Coefficients | Std. Error | T-Value | p-Value | |
|---|---|---|---|---|
| Intercept | 1.796 | 0.563 | 3.189 | 0.0015 |
| FluoroSense reading | 0.590 | 0.005 | 107.204 | <2 × 10−16 |
| Turbidity | 0.186 | 0.006 | 30.565 | <2 × 10−16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamdhani, H.; Eppehimer, D.E.; Walker, D.; Bogan, M.T. Performance of a Handheld Chlorophyll-a Fluorometer: Potential Use for Rapid Algae Monitoring. Water 2021, 13, 1409. https://doi.org/10.3390/w13101409
Hamdhani H, Eppehimer DE, Walker D, Bogan MT. Performance of a Handheld Chlorophyll-a Fluorometer: Potential Use for Rapid Algae Monitoring. Water. 2021; 13(10):1409. https://doi.org/10.3390/w13101409
Chicago/Turabian StyleHamdhani, Hamdhani, Drew E. Eppehimer, David Walker, and Michael T. Bogan. 2021. "Performance of a Handheld Chlorophyll-a Fluorometer: Potential Use for Rapid Algae Monitoring" Water 13, no. 10: 1409. https://doi.org/10.3390/w13101409
APA StyleHamdhani, H., Eppehimer, D. E., Walker, D., & Bogan, M. T. (2021). Performance of a Handheld Chlorophyll-a Fluorometer: Potential Use for Rapid Algae Monitoring. Water, 13(10), 1409. https://doi.org/10.3390/w13101409






