Sediment Nutrient Flux Rates in a Shallow, Turbid Lake Are More Dependent on Water Quality Than Lake Depth
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Sampling
2.1.1. Water Quality
2.1.2. Sediment Cores
2.2. Sediment Core Incubations
2.3. Temperature Experiment
2.4. Data Analysis
3. Results
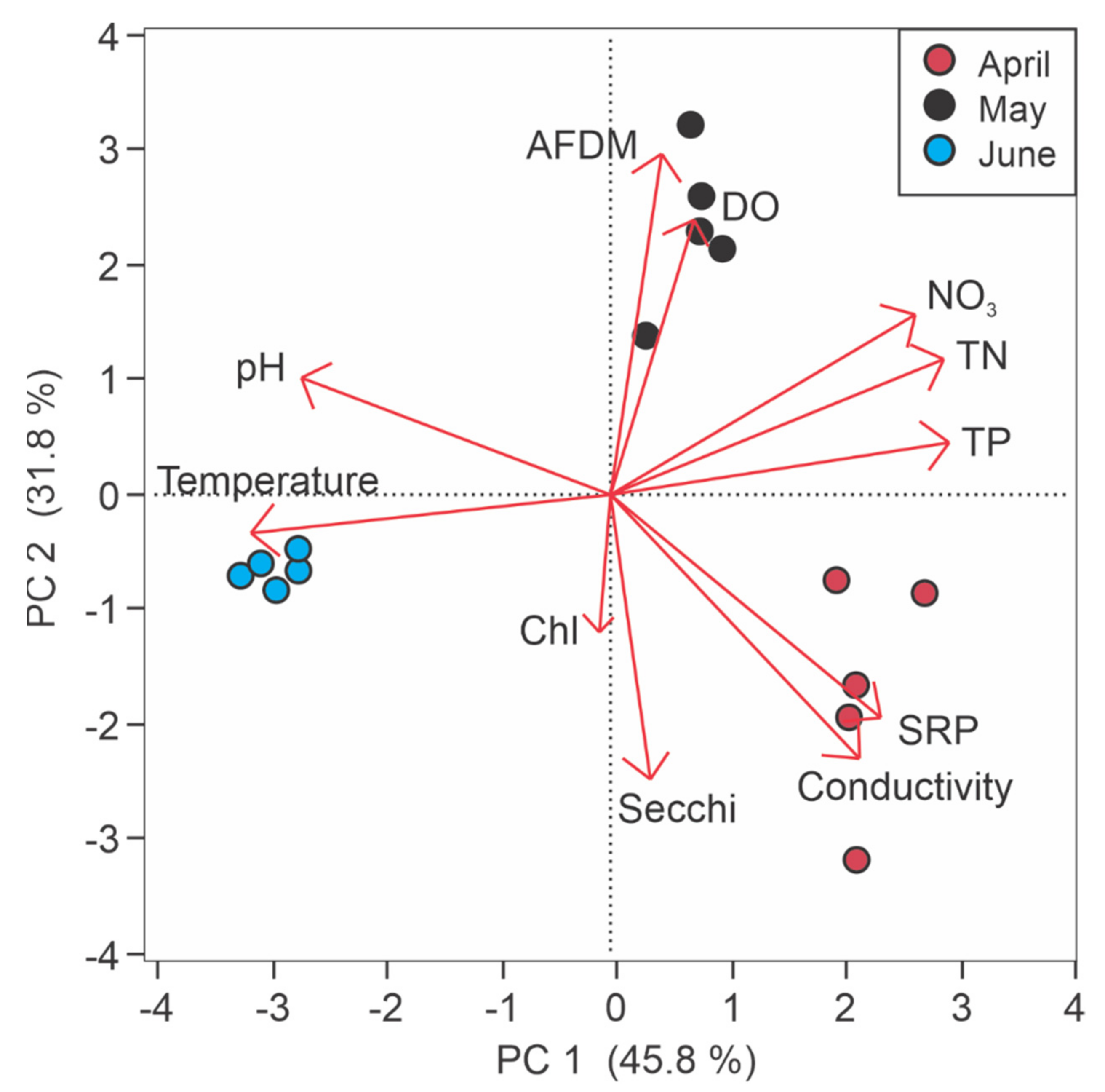
3.1. Depth and Water Quality Variability
3.2. Nutrient Flux Rates
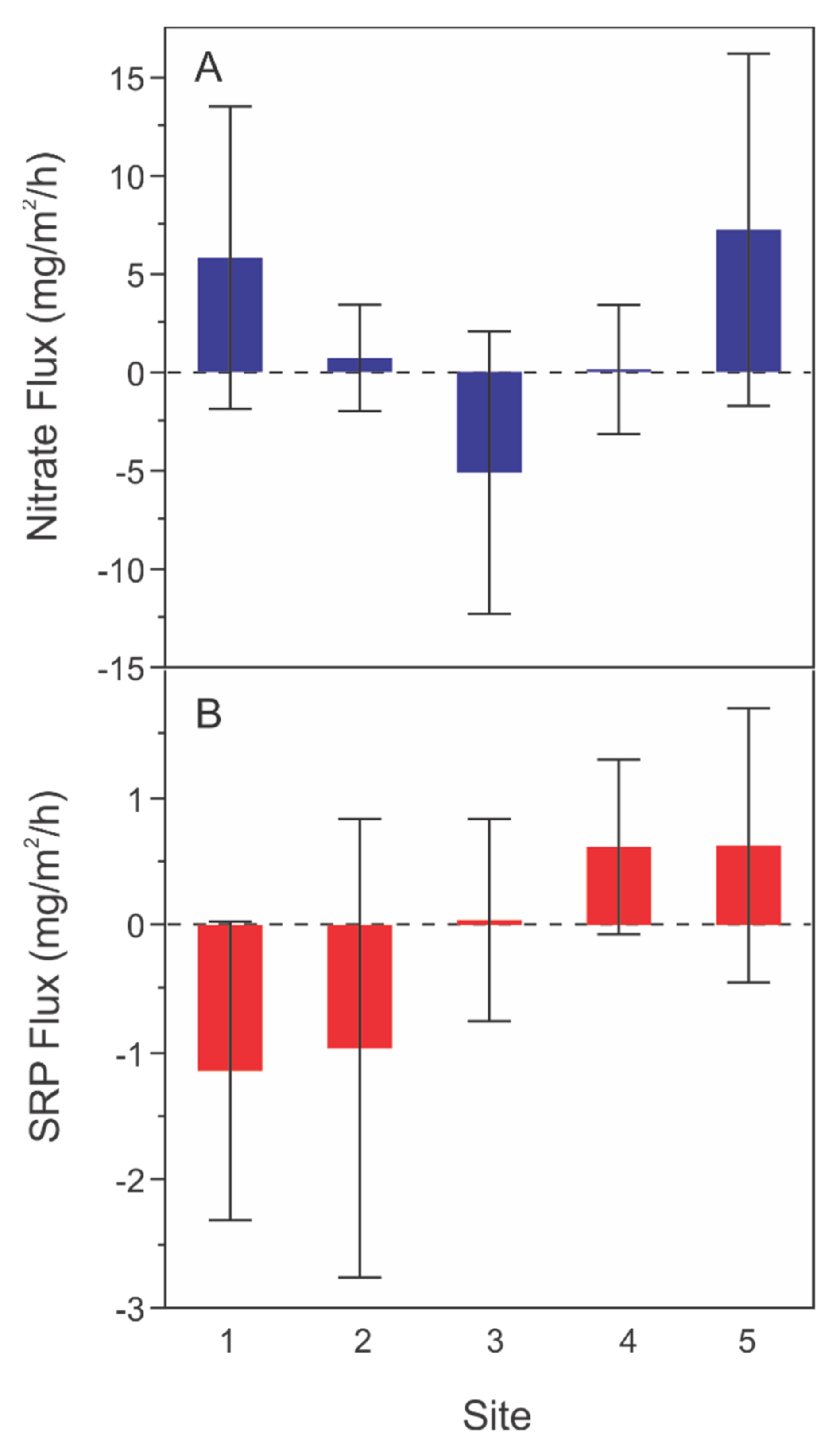
3.2.1. Spatial Variation in Sediment Flux Rates
3.2.2. Water Quality and Sediment Nutrient Flux Rates
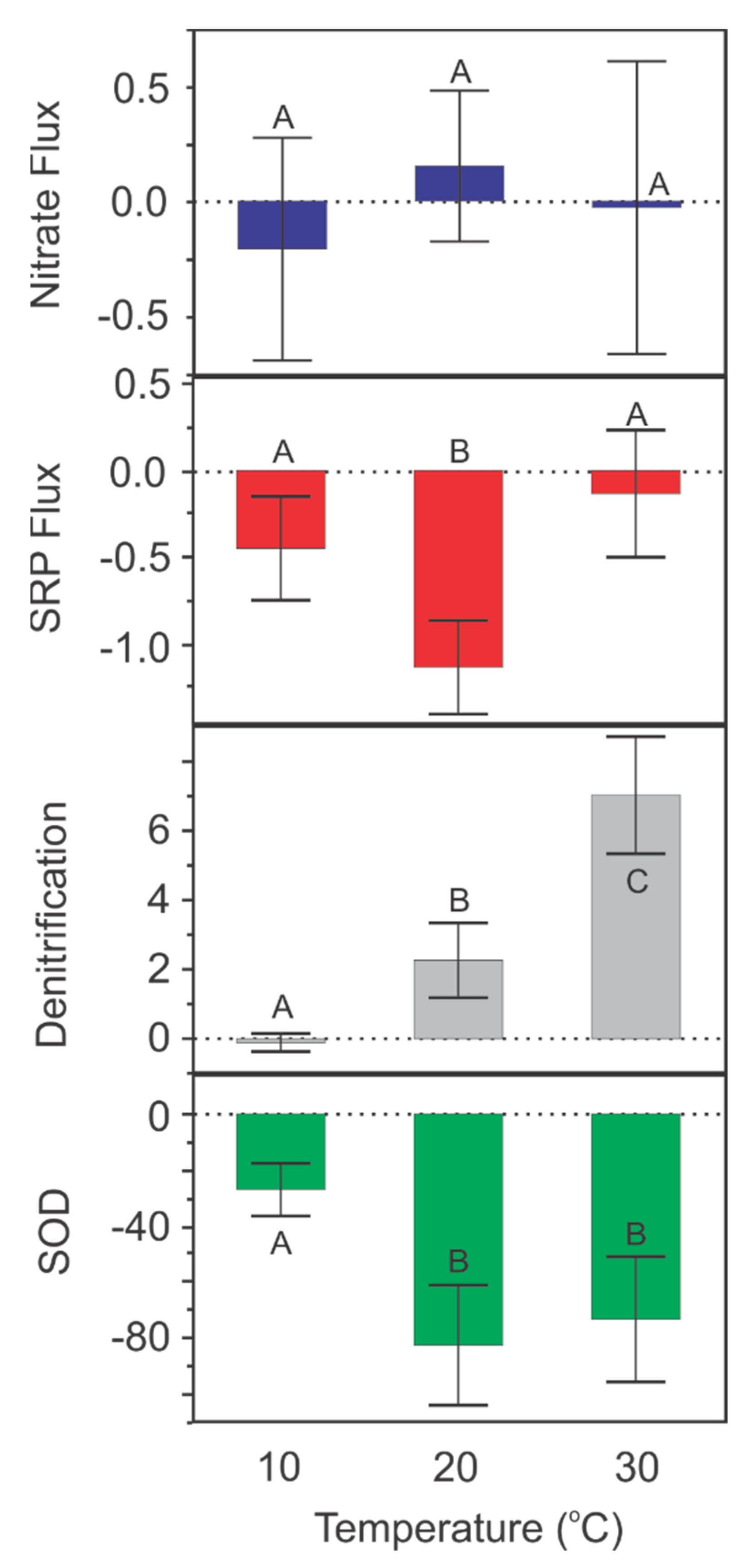
3.3. Temperature Experiment
4. Discussion
4.1. Sediment Flux Trends
4.2. Water Quality and Sediment Nutrient Flux
4.3. Water Depth, Spatial Location, and Sediment Nutrient Flux
4.4. Season as A Controlling Variable
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smith, V.H. Eutrophication of freshwater and coastal marine ecosystems: A global problem. Environ. Sci. Pollut. Res. 2003, 10, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S. Solutions for a cultivated planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Galloway, J.; Cowling, E. Reactive nitrogen and the world: 200 years of change. AMBIO J. Hum. Environ 2002, 31, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Daniel, T.C.; Sharpley, A.N.; Lemunyon, J.L. Agricultural Phosphorus and Eutrophication: A Symposium Overview. J. Environ. Qual. 1998, 27, 251–257. [Google Scholar] [CrossRef]
- Bennett, E.; Carpenter, S.; Caraco, N. Human Impact on Erodable Phosphorus and Eutrophication: A Global Perspective. Bioscience 2001, 51, 227–234. [Google Scholar] [CrossRef]
- Berggren, M.; Laudon, H.; Jonsson, A.; Jansson, M. Nutrient constraints on metabolism affect the temperature regulation of aquatic bacterial growth efficiency. Microb. Ecol. 2010, 60, 894–902. [Google Scholar] [CrossRef]
- Hessen, D.O.; Anderson, T.R. Excess carbon in aquatic organisms and ecosystems: Physiological, ecological, and evolutionary implications. Limnol. Oceanogr. 2008, 53, 1685–1696. [Google Scholar] [CrossRef]
- Rabalais, N.N.; Turner, R.E.; Dortch, Q.; Wiseman, W.J., Jr.; Gupta, B.K.S. Nutrient changes in the Mississippi river and system responses on the adjacent continental shelf. Estuaries 1996, 19, 386–407. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Day, J.W.; Gilliam, J.W.; Groffman, P.M.; Hey, D.L.; Randall, G.W.; Wang, N. Reducing nitrogen loading to the Gulf of Mexico from the Mississippi River Basin: Strategies to counter a persistent ecological problem. Bioscience 2001, 51, 373–388. [Google Scholar] [CrossRef]
- Turner, R.E.; Rabalais, N.N.; Justic, D. Gulf of Mexico hypoxia: Alternate states and a legacy. Environ. Sci. Technol. 2008, 42, 2323–2327. [Google Scholar] [CrossRef]
- Killgore, K.J.; Hoover, J.J.; Murphy, C.E.; Parrish, K.D.; Johnson, D.R.; Myers, K.F. Restoration of Delta Streams: A Case History and Conceptual Model; Engineer Research and Development Center Vicksburg, Mississippi Coastal and Hydraulics Lab: Vicksburg, MS, USA, 2008. [Google Scholar]
- Fisk, H.N. Geological Investigation of the Alluvial Valley of the Lower Mississippi River, War Department, Corps of Engineers; US Army, Mississippi River Commission: Vicksburg, MS, USA, 1944. [Google Scholar]
- Saucier, R. Geomorphology and Quaternary Geologic History of the Lower Mississippi River Valley; U.S. Army Engineers Waterways Experiment, Mississippi River Commission: Vicksburg, MS, USA, 1994. [Google Scholar]
- Hudson, P.F.; Kessel, R.H. Channel migration and meander-bend curvature in the lower Mississippi River prior to major human modification. Geology 2000, 28, 531–534. [Google Scholar] [CrossRef]
- Wren, D.G.; Davidson, G.R.; Walker, W.G.; Galicki, S.J. The evolution of an oxbow lake in the Mississippi alluvial floodplain. J. Soil Water Conserv. 2008, 63, 129–135. [Google Scholar] [CrossRef]
- Wondim, Y.K.; Mosa, H.M. Spatial variation of sediment physicochemical characteristics of Lake Tana, Ethiopia. J. Environ. Earth Sci. 2015, 5, 95–109. [Google Scholar]
- Janssen, A.B.G.; van Wijk, D.; van Gervena, L.P.A.; Bakker, E.S.; Brederveld, R.J.; DeAngelis, D.L.; Janse, J.H.; Mooij, W.M. Success of lake restoration depends on spatial aspects of nutrient loading and hydrology. Sci. Total Environ. 2019, 679, 249–259. [Google Scholar] [CrossRef]
- Shaughnessy, A.R.; Sloan, J.J.; Corcoran, M.J.; Hasenmueller, E.A. Sediments in agricultural reservoirs act as sinks and sources for nutrients over various timescales. Water Resour. Res. 2019, 55, 5985–6000. [Google Scholar] [CrossRef]
- Wang, Y.; Kong, X.; Peng, Z.; Zhang, H.; Liu, G.; Hu, W.; Zhou, X. Retention of nitrogen and phosphorus in Lake Chaohu, China: Implications for eutrophication management. Environ. Sci. Poll. Res. 2020, 27, 41488–41502. [Google Scholar] [CrossRef]
- Harrison, J.A.; Maranger, R.J.; Alexander, R.B.; Giblin, A.E.; Jacinthe, P.A.; Mayorga, E.; Seitzinger, S.; Sobota, D.; Wollheim, W. The regional and global significance of nitrogen removal in lakes and reservoirs. Biogeochemistry 2009, 93, 143–157. [Google Scholar] [CrossRef]
- Knowles, R. Denitrification. Microbiol. Rev. 1982, 46, 43. [Google Scholar] [CrossRef]
- Saunders, D.L.; Kalff, J. Nitrogen retention in wetlands, lakes and rivers. Hydrobiologia 2001, 443, 205–212. [Google Scholar] [CrossRef]
- Seitzinger, S.P. Denitrification in freshwater and coastal marine ecosystems: Ecological and geochemical significance. Limnol. Oceanogr. 1988, 33, 702–724. [Google Scholar] [CrossRef]
- Seitzinger, S.P. Denitrification in Aquatic Sediments. In Denitrification in Soil and Sediment; Springer: Boston, MA, USA, 1990; pp. 301–322. [Google Scholar]
- Stall, J.B. Effects of sediment on water quality. J. Environ. Qual. 1972, 1, 353–360. [Google Scholar] [CrossRef]
- Nichols, D.S. Capacity of natural wetlands to remove nutrients from wastewater. J. Water Pollut. Control Fed. 1983, 55, 495–505. [Google Scholar]
- Hammer, D.A.; Knight, R.L. Designing constructed wetlands for nitrogen removal. Water Sci. Technol. 1994, 29, 15–27. [Google Scholar] [CrossRef]
- Huang, L.; Li, Z.; Bai, X.; Li, R.; Wu, H.; Wei, D.; Yu, L. Laboratory study of phosphorus retention and release by eutrophic lake sediments: Modeling and implications for P release assessments. Ecol. Eng. 2016, 95, 438–446. [Google Scholar] [CrossRef]
- Orihel, D.M.; Baulch, H.M.; Casson, N.J.; North, R.L.; Parsons, C.T.; Seckar, D.C.M.; Venkiteswaran, J.J. Internal phosphorus loading in Canadian fresh waters: A critical review and data analysis. Can. J. Fish. Aquat. Sci. 2017, 74, 2005–2029. [Google Scholar] [CrossRef]
- Randall, M.C.; Carling, G.T.; Dastrup, D.B.; Miller, T.; Nelson, S.T.; Rey, K.A.; Hansen, N.C.; Bickmore, B.R.; Aanderud, Z.T. Sediment potentially controls in-lake phosphorus cycling and harmful cyanobacteria in shallow, eutrophic Utah Lake. PLoS ONE 2019, 14, e0212238. [Google Scholar] [CrossRef]
- Jin, G.; Onodera, S.; Saito, M.; Shimizu, Y. Sediment phosphorus cycling in a nutrient-rich embayment in relation to sediment phosphorus pool and release. Limnology 2020, 21, 415–425. [Google Scholar] [CrossRef]
- Saikku, M. This Delta, This Land: An Environmental History of the Yazoo-Mississippi Floodplain; University of Georgia Press: Athens, GA, USA, 2005. [Google Scholar]
- Shields, D.F., Jr.; Lizotte, R.E.; Knight, S.S. Spatial and temporal water quality variability in aquatic habitats of a cultivated floodplain. River Res. Appl. 2013, 29, 313–329. [Google Scholar] [CrossRef]
- Sartory, D.P.; Grobbelaar, J.U. Extraction of chlorophyll a from freshwater phytoplankton for spectrophotometric analysis. Hydrobiologia 1984, 114, 177–187. [Google Scholar] [CrossRef]
- Welschmeyer, N.A. Fluorometric analysis of chlorophyll a in the presence of chlorophyll b and pheopigments. Limnol. Oceanogr. 1994, 39, 1985–1992. [Google Scholar] [CrossRef]
- Crumpton, W.G.; Isenhart, T.M.; Mitchell, P.D. Nitrate and organic N analyses with second-derivative spectroscopy. Limnol. Oceanogr. 1992, 37, 907–913. [Google Scholar] [CrossRef]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Stainton, M.; Capel, M.; Armstrong, F. The Chemical Analysis of Fresh Water, 2nd ed.; Department of the Environment (Canada): Winnipeg, MB, Canada, 1977. [Google Scholar]
- American Public Health Association. Standard Methods of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Nifong, R.L.; Taylor, J.M.; Moore, M.T. Mulch-derived organic carbon stimulates high denitrification fluxes from agricultural ditch sediments. J. Environ. Qual. 2019, 48, 476–484. [Google Scholar] [CrossRef]
- Speir, S.L.; Taylor, J.M.; Scott, J.T. Seasonal Differences in Relationships between Nitrate Concentration and Denitrification Rates in Ditch Sediments Vegetated with Rice Cutgrass. J. Environ. Qual. 2017, 46, 1500–1509. [Google Scholar] [CrossRef]
- Holtan-Hartwig, L.; Dörsch, P.; Bakken, L.R. Low temperature control of soil denitrifying communities: Kinetics of N2O production and reduction. Soil Biol. Biochem. 2002, 34, 1797–1806. [Google Scholar] [CrossRef]
- Rysgaard, S.; Glud, R.N.; Risgaard-Petersen, N.; Dalsgaard, T. Denitrification and anammox activity in Arctic marine sediments. Limnol. Oceanogr. 2004, 49, 1493–1502. [Google Scholar] [CrossRef]
- Kana, T.M.; Darkangelo, C.; Hunt, M.D.; Oldham, J.B.; Bennett, G.E.; Cornwell, J.C. Membrane Inlet Mass Spectrometer for Rapid High-Precision Determination of N2, O2, and Ar in Environmental Water Samples. Anal. Chem. 1994, 66, 4166–4170. [Google Scholar] [CrossRef]
- Nakagawa, S.; Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Pinheiro, J.; Bates, D.; Deb-Roy, S.; Sarkar, D.; Heisterkamp, S.; van Willigen, D.; Matintainer, R. Package ‘nlme’. Linear Nonlinear Mixed Effects Models; R Core Team: Vienna, Austria, 2021; p. 3. [Google Scholar]
- Bartoń, K. MuMIn: Multi-Model Inference; R Package Version 1.42.1; R Core Team: Vienna, Austria, 2018. [Google Scholar]
- Ogle, D.; Ogle, M.D. Package ‘FSA’. CRAN Repos 2017, 1–206. [Google Scholar]
- Locke, M.A.; Lizotte, R.E.; Yasarer LM, W.; Bingner, R.L.; Moore, M.T. Moore. Surface runoff in Beasley Lake watershed: Effect of land management practices in a Lower Mississippi River Basin watershed. J. Soil Water Conserv. 2020, 75, 278–290. [Google Scholar] [CrossRef]
- Kõiv, T.; Nõges, T.; Laas, A. Phosphorus retention as a function of external loading, hydraulic turnover time, area and relative depth in 54 lakes and reservoirs. Hydrobiologia 2011, 660, 105–115. [Google Scholar] [CrossRef]
- Arango, C.P.; Tank, J.L.; Johnson, L.T.; Hamilton, S.K. Assimilatory uptake rather than nitrification and denitrification determines nitrogen removal patterns in streams of varying land use. Limnol. Oceanogr. 2008, 53, 2558–2572. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Zhang, L.; Anderson, C.J.; Altor, A.E.; Hernandez, M.E. Creating riverine wetlands: Ecological succession, nutrient retention, and pulsing effects. Ecol. Eng. 2005, 25, 510–527. [Google Scholar] [CrossRef]
- Bernot, M.J.; Dodds, W.K. Nitrogen retention, removal, and saturation in lotic ecosystems. Ecosystems 2005, 8, 442–453. [Google Scholar] [CrossRef]
- Hill, A.R. Phosphorus and major cation mass balance for two rivers during low summer flows. Freshw. Biol. 1982, 12, 293–304. [Google Scholar] [CrossRef]
- Munn, N.L.; Meyer, J.L. Habitat-specific solute retention in two small streams: An intersite comparison. Ecology 1990, 71, 2351–2357. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Locke, M.A. Assessment of runoff water quality for an integrated best management practice system in an agricultural watershed. J. Soil Water Conserv. 2018, 73, 247–256. [Google Scholar] [CrossRef]
- Dodds, W.K. Misuse of inorganic N and soluble reactive P concentrations to indicate nutrient status of surface waters. J. North Am. Benthol. Soc. 2003, 22, 171–181. [Google Scholar] [CrossRef]
- Dodds, W.K.; Smith, V.H.; Lohman, K. Nitrogen and phosphorus relationships to benthic algal biomass in temperate streams. Can. J. Fish. Aquat. Sci. 2002, 59, 865–874. [Google Scholar] [CrossRef]
- Hall, R.O., Jr.; Tank, J.L.; Sobota, D.J.; Mulholland, P.J.; O’Brien, J.M.; Dodds, W.K.; Arangob, C.P. Nitrate removal in stream ecosystems measured by 15N addition experiments: Total uptake. Limnol. Oceanogr. 2009, 54, 653–665. [Google Scholar] [CrossRef]
- Spieles, D.J.; Mitsch, W.J. The effects of season and hydrologic and chemical loading on nitrate retention in constructed wetlands: A comparison of low- and high-nutrient riverine systems. Ecol. Eng. 1999, 14, 77–91. [Google Scholar] [CrossRef]
- Haggard, B.E.; Storm, D.E.; Stanley, E.H. Effect of a point source input on stream nutrient retention. J. Am. Water Resour. Assoc. 2001, 37, 1291–1299. [Google Scholar] [CrossRef]
- Marchant, H.K.; Holtappels, M.; Lavik, G.; Ahmerkamp, S.; Winter, C.; Kuypers, M.M. Coupled nitrification–denitrification leads to extensive N loss in subtidal permeable sediments. Limnol. Oceanogr. 2016, 61, 1033–1048. [Google Scholar] [CrossRef]
- Lotti, T.; Kleerebezem, R.; van Loosdrecht, M.C.M. Effect of temperature change on anammox activity. Biotechnol. Bioeng. 2015, 112, 98–103. [Google Scholar] [CrossRef]
- Wu, Y.; Wen, Y.; Zhou, J.; Wu, Y. Phosphorus release from lake sediments: Effects of pH, temperature and dissolved oxygen. KSCE J. Civ. Eng. 2014, 18, 323–329. [Google Scholar] [CrossRef]
- McDowell, R.W.; Elkin, K.R.; Kleinman, P.J. Temperature and nitrogen effects on phosphorus uptake by agricultural stream-bed sediments. J. Environ. Qual. 2017, 46, 295–301. [Google Scholar] [CrossRef]
- Spears, B.M.; Carvalho, L.; Perkins, R.; Paterson, D.M. Effects of light on sediment nutrient flux and water column nutrient stoichiometry in a shallow lake. Water Res. 2008, 42, 977–986. [Google Scholar] [CrossRef]
- Pettry, D.E.; Switzer, R.E. Sharkey Soils in Mississippi. Mississippi State: Office of Agricultural Communications (Publications Section); Division of Agriculture, Forestry, and Veterinary Medicine, Mississippi State University: Starkville, MS, USA, 1996; Volume 1057. [Google Scholar]
- Hermans, M.; Risgaard-Petersen, N.; Meysman, F.J.; Slomp, C.P. Biogeochemical impact of cable bacteria on coastal Black Sea sediment. Biogeosciences 2020, 17, 5919–5938. [Google Scholar] [CrossRef]
- Wren, D.G.; Davidson, G.R. Using lake sedimentation rates to quantify the effectiveness of erosion control in watersheds. J. Soil Water Conserv. 2011, 66, 313–322. [Google Scholar] [CrossRef]
- Van Luijn, F.; Boers, P.C.M.; Lijklema, L.; Sweerts, J.P. Nitrogen fluxes and processes in sandy and muddy sediments from a shallow eutrophic lake. Water Res. 1999, 33, 33–42. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Z.; Gulati, R.D.; Jeppesen, E. The effect of benthic algae on phosphorus exchange between sediment and overlying water in shallow lakes: A microcosm study using 32 P as a tracer. Hydrobiologia 2013, 710, 109–116. [Google Scholar] [CrossRef]
- Wren, D.G.; Taylor, J.M.; Rigby, J.R.; Locke, M.A.; Yasarer, L.M. Short term sediment accumulation rates reveal seasonal time lags between sediment delivery and deposition in an oxbow lake. Agric. Ecosyst. Environ. 2019, 281, 92–99. [Google Scholar] [CrossRef]
- Aryal, N.; Reba, M.L.; Straitt, N.; Teague, T.G.; Bouldin, J.; Dabney, S. Impact of cover crop and season on nutrients and sediment in runoff water measured at the edge of fields in the Mississippi Delta of Arkansas. J. Soil Water Conserv. 2018, 73, 24–34. [Google Scholar] [CrossRef]


| Nitrate Flux | |||||
|---|---|---|---|---|---|
| Variation | df | Type II Sums of Squares | Mean Square | F Statistic | p |
| Site | 4 | 496.8 | 124.2 | 8.15 | <0.001 |
| Habitat | 2 | 68.4 | 34.2 | 2.24 | 0.130 |
| Month | 2 | 744.4 | 372.2 | 24.41 | <0.001 |
| Site × Month | 8 | 1128.9 | 141.1 | 9.26 | <0.001 |
| Habitat × Month | 4 | 28.3 | 7.1 | 0.46 | 0.761 |
| Residuals | 21 | 335.4 | 15.2 | ||
| SRP Flux | |||||
| Variation | df | Type II Sums of Squares | Mean Square | F Statistic | p |
| Site | 4 | 25.8 | 6.45 | 3.61 | 0.019 |
| Habitat | 2 | 0.1 | 0.05 | 0.03 | 0.973 |
| Month | 2 | 16.1 | 3.07 | 1.76 | 0.201 |
| Site × Month | 8 | 40.3 | 5.04 | 2.82 | 0.024 |
| Habitat × Month | 4 | 3.4 | 0.86 | 0.48 | 0.751 |
| Residuals | 21 | 42.9 | 1.79 | ||
| Model Parameters | AICc | Δi | wi | K | R2 Marginal | R2 Conditional | |
|---|---|---|---|---|---|---|---|
| Core Nitrate Flux | DO, Month, Temp, TN, TP, Secchi | 297.1 | 0 | 0.313 | 10 | 0.57 | 0.82 |
| DO, Month, PO4, Secchi, Temp, TN | 297.9 | 0.79 | 0.211 | 10 | 0.53 | 0.84 | |
| AFDM (water), DO, Month, Secchi, Temp, TN | 297.9 | 0.82 | 0.208 | 10 | 0.55 | 0.85 | |
| Core SRP Flux | AFDM (sediment), DO, Secchi, Temp, TP | 168.0 | 0 | 0.103 | 8 | 0.38 | 0.38 |
| Model Parameter | Sediment Nitrate | Sediment SRP |
|---|---|---|
| Month | 1.00 | 0.42 |
| Distance to Shore | 0.19 | 0.17 |
| Nitrate | 0.18 | 0.28 |
| Phosphate | 0.35 | 0.27 |
| Total Nitrogen | 0.86 | 0.30 |
| Total Phosphorus | 0.37 | 0.77 |
| Water Depth | 0.20 | 0.18 |
| Temperature | 1.00 | 0.44 |
| Dissolved Oxygen | 0.95 | 0.27 |
| Secchi Depth | 1.00 | 0.59 |
| pH | 0.24 | 0.39 |
| Sediment AFDM | 0.14 | 0.77 |
| Water AFDM | 0.33 | 0.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evans, J.L.; Murdock, J.N.; Taylor, J.M.; Lizotte, R.E., Jr. Sediment Nutrient Flux Rates in a Shallow, Turbid Lake Are More Dependent on Water Quality Than Lake Depth. Water 2021, 13, 1344. https://doi.org/10.3390/w13101344
Evans JL, Murdock JN, Taylor JM, Lizotte RE Jr. Sediment Nutrient Flux Rates in a Shallow, Turbid Lake Are More Dependent on Water Quality Than Lake Depth. Water. 2021; 13(10):1344. https://doi.org/10.3390/w13101344
Chicago/Turabian StyleEvans, Jordan L., Justin N. Murdock, Jason M. Taylor, and Richard E. Lizotte, Jr. 2021. "Sediment Nutrient Flux Rates in a Shallow, Turbid Lake Are More Dependent on Water Quality Than Lake Depth" Water 13, no. 10: 1344. https://doi.org/10.3390/w13101344
APA StyleEvans, J. L., Murdock, J. N., Taylor, J. M., & Lizotte, R. E., Jr. (2021). Sediment Nutrient Flux Rates in a Shallow, Turbid Lake Are More Dependent on Water Quality Than Lake Depth. Water, 13(10), 1344. https://doi.org/10.3390/w13101344







