Characterization of Bottled Waters by Multielemental Analysis, Stable and Radiogenic Isotopes
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples and Sampling Procedure
2.2. Analytical Procedures
2.2.1. Determination of Major and Trace Elements
2.2.2. Determination of 87Sr/86Sr Isotope Ratio
2.2.3. Determination of the Isotope Composition of Hydrogen and Oxygen
2.2.4. Determination of Alkalinity
2.2.5. Determination of Stable Isotope Composition of Dissolved Inorganic Carbon (δ13CDIC)
2.2.6. Thermodynamic Modeling
2.2.7. Statistical Analysis
3. Results and Discussion
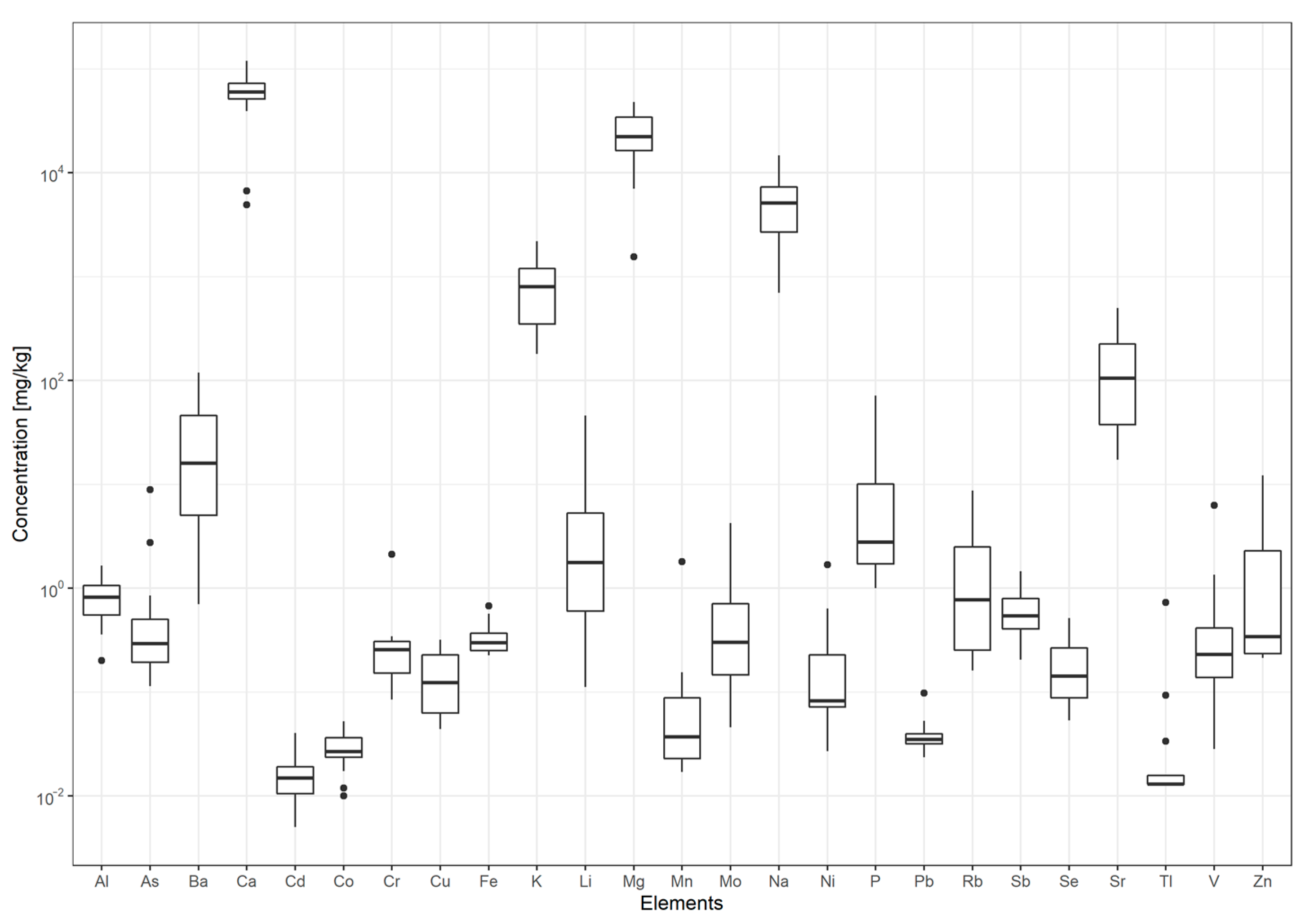
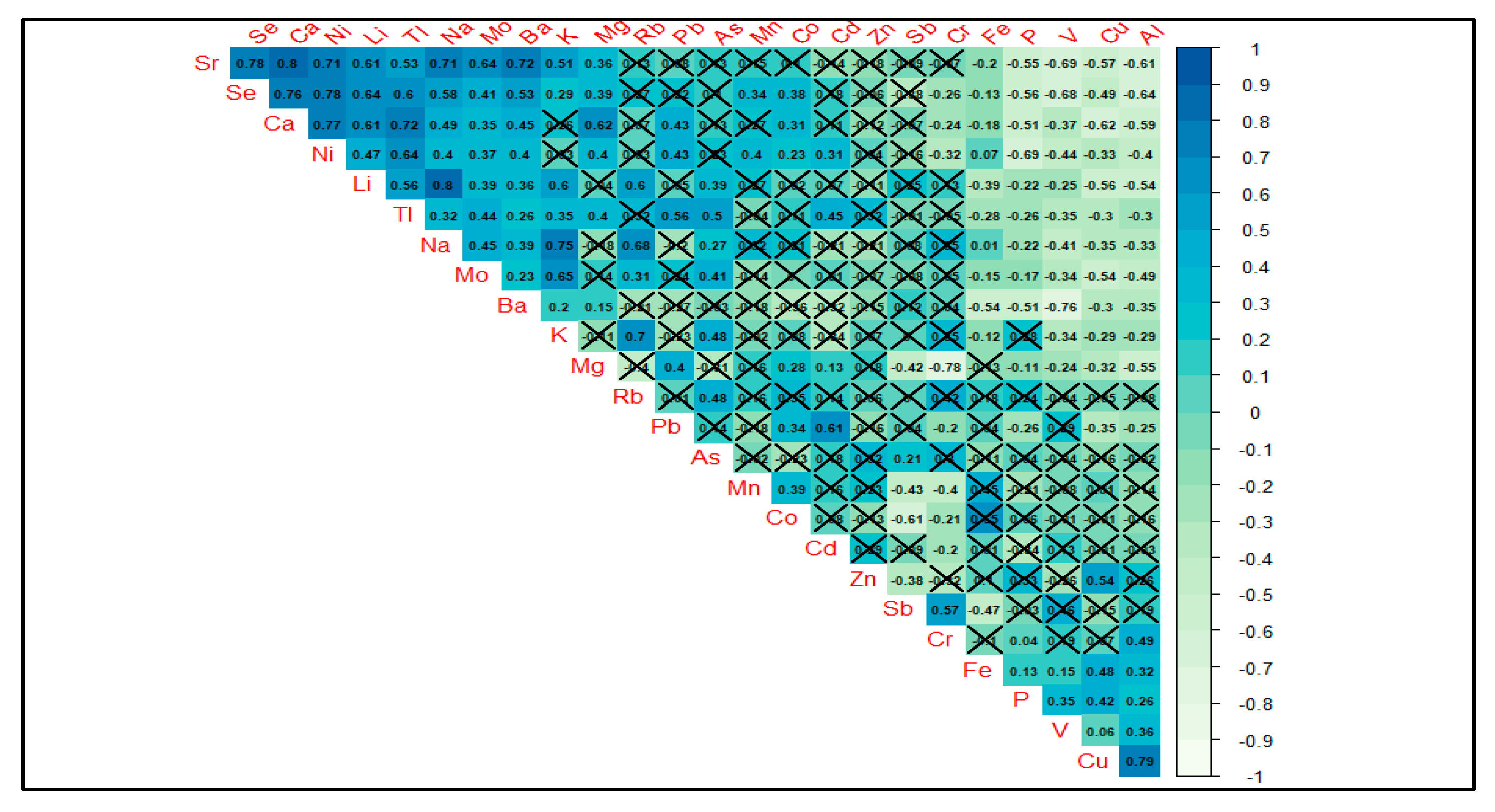
3.1. Content of Major and Trace Elements in Bottled Waters
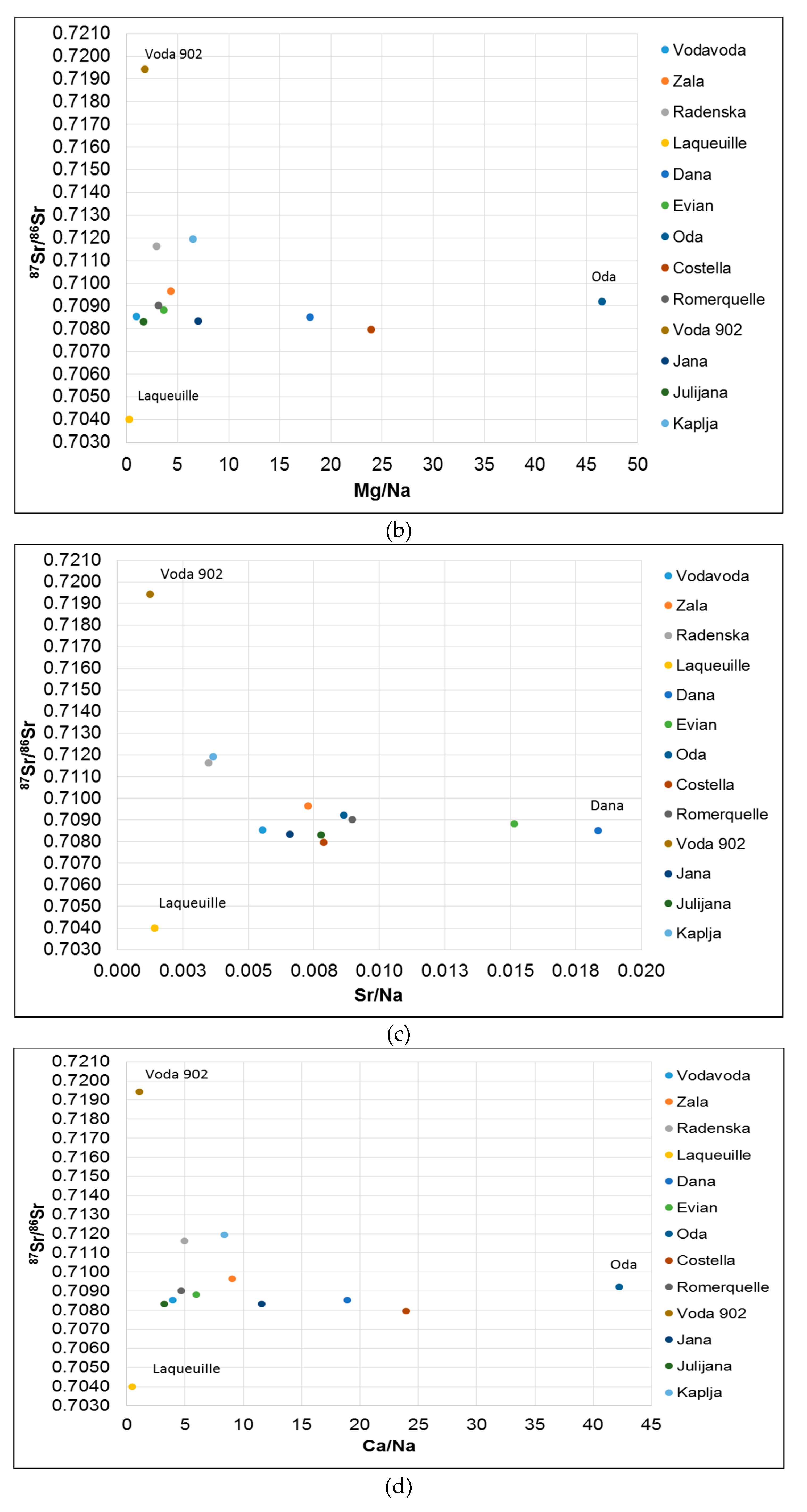
3.2. 87Sr/86Sr Isotope Ratio
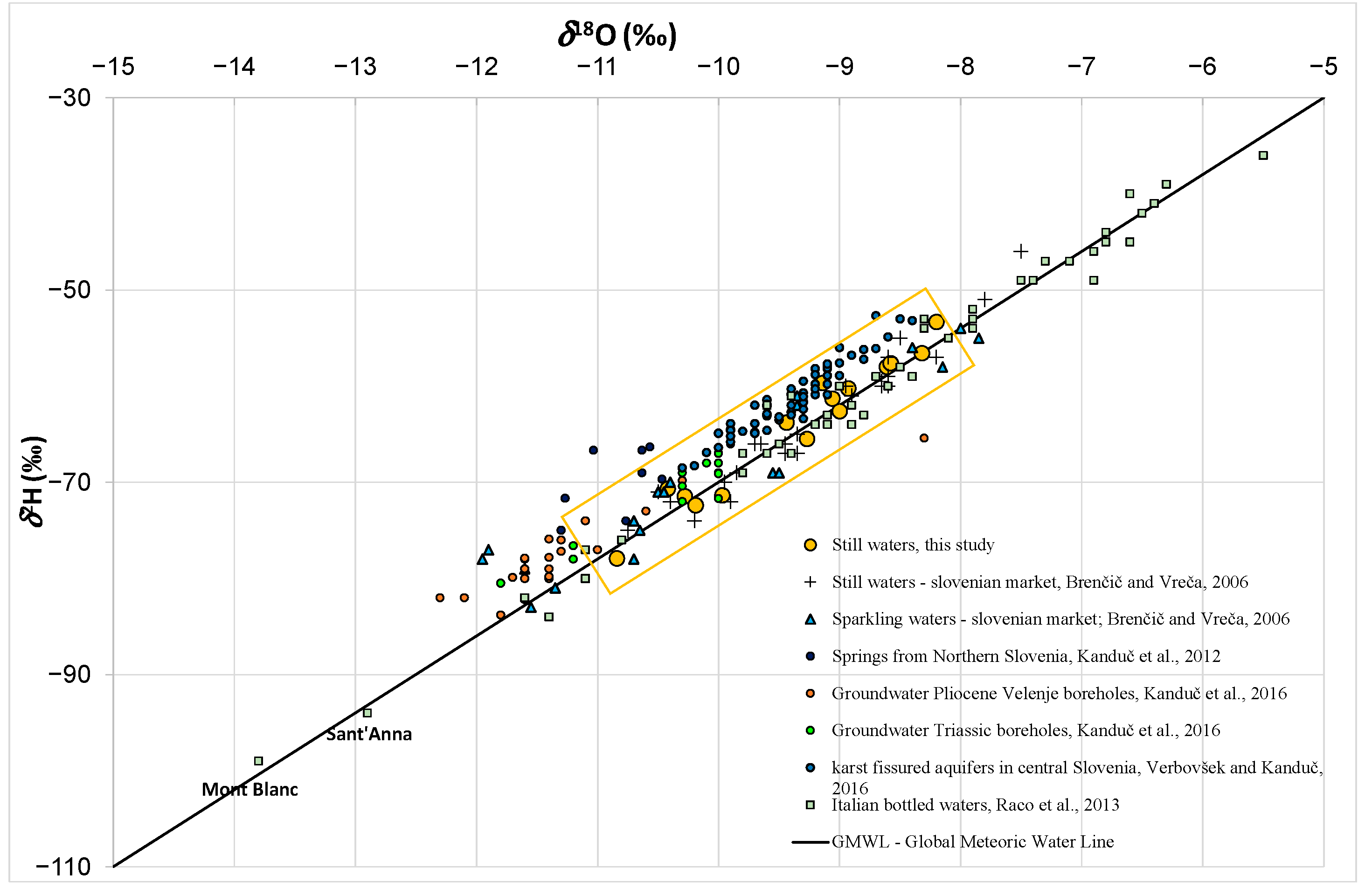
3.3. Hydrogen (δ2H) and Oxygen (δ18O) Analysis
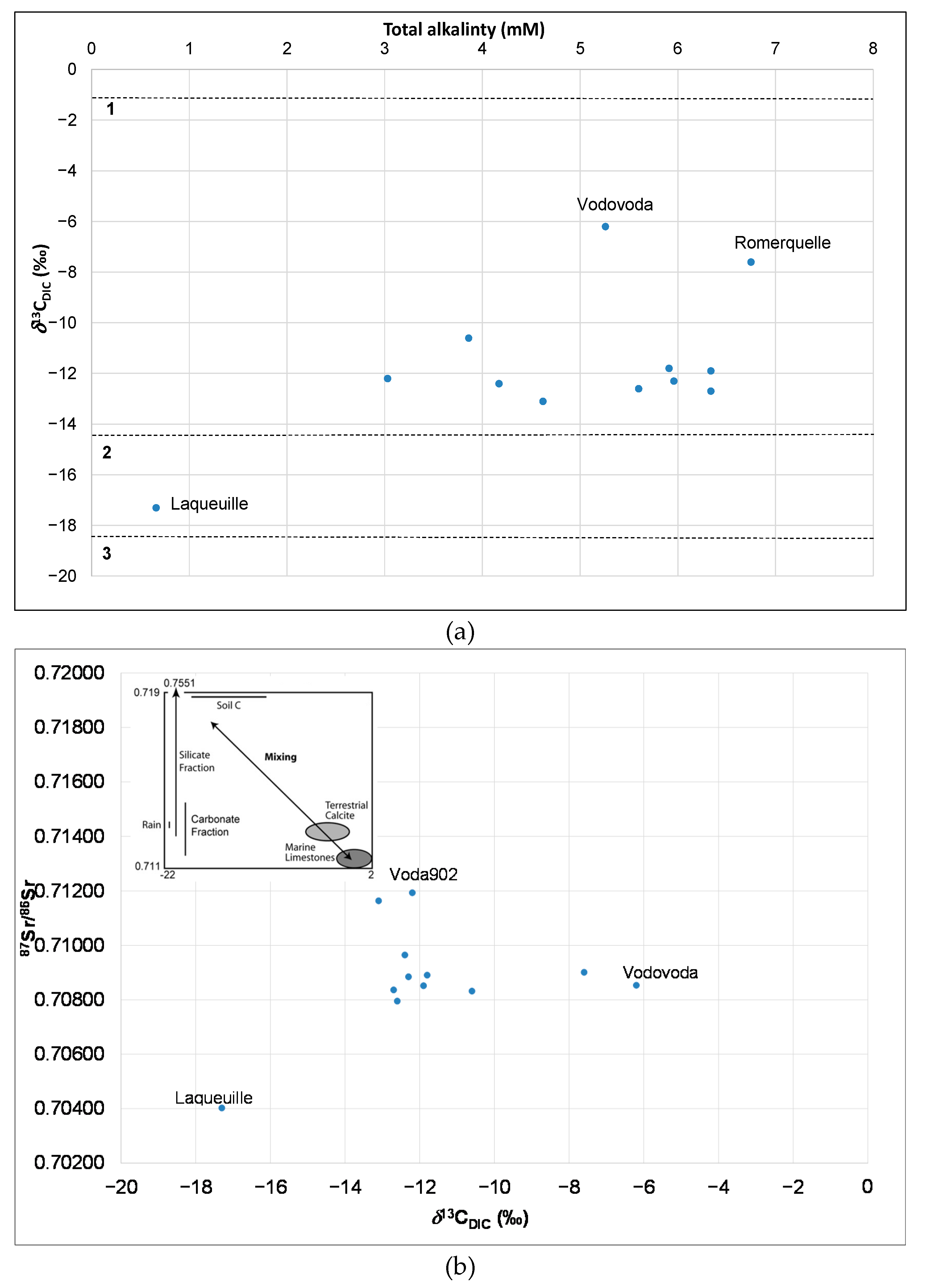
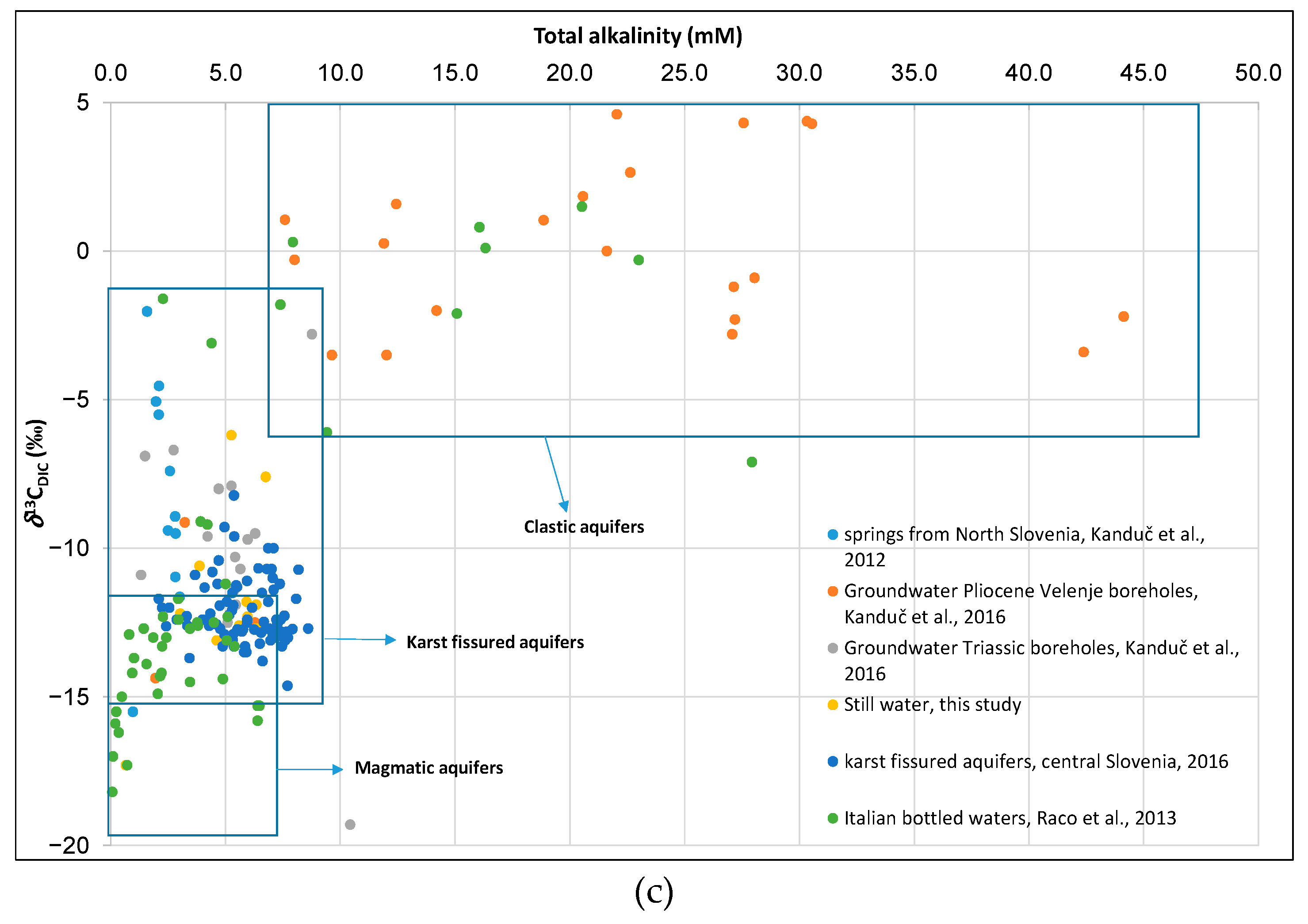
3.4. Evaluation of Geochemical Processes with Geochemical Parameters (Ca2+/Mg2+ Ratios, Total Alkalinity) and δ13CDIC in Bottled Waters
3.4.1. Geochemical Processes
3.4.2. Origin of Carbon in the Dissolved Inorganic Carbon (δ13CDIC) in Groundwater
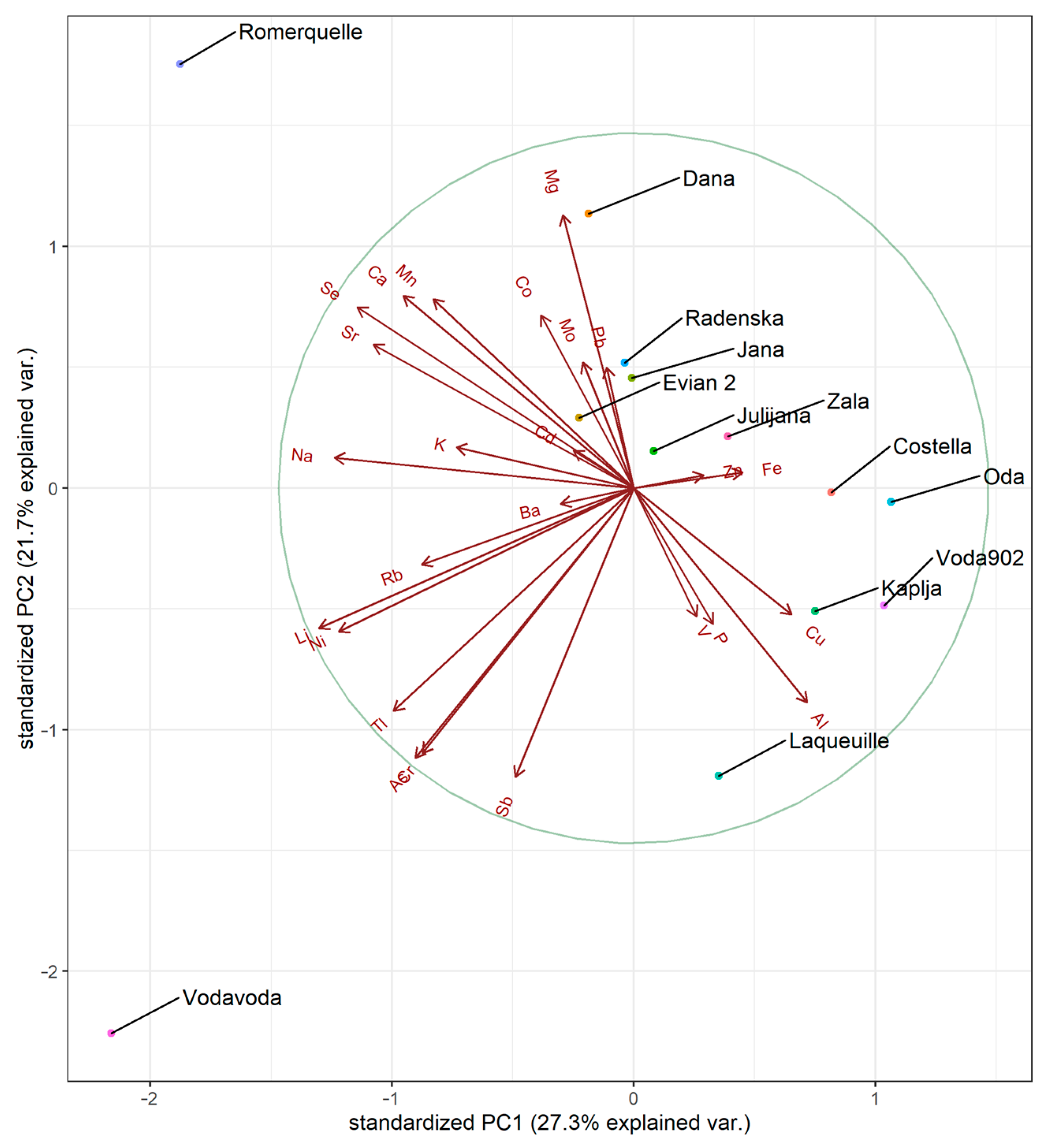
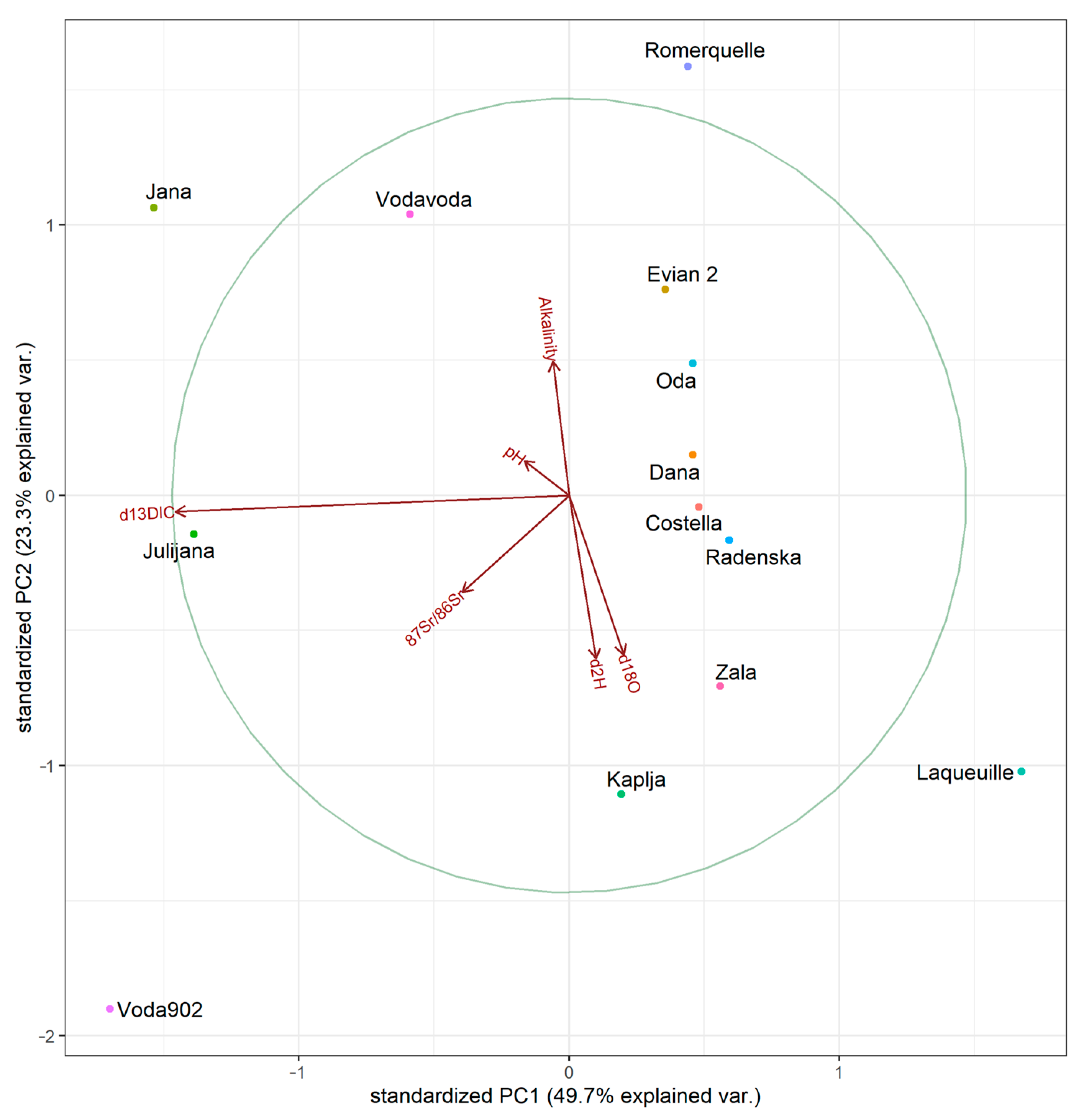
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Statista. Available online: www.statista.com (accessed on 14 September 2018).
- Natural Mineral & Spring Waters The Natural Choice For Hydration. Available online: www.efbw.org (accessed on 14 September 2018).
- European Commission. Council Directive 80/777/EEC of 15 July 1980 on the Approximation of the Laws of the Member States Relating to the Exploiting and Marketing of Natural Waters. Off. J. L. 1980, 229, 1–10. [Google Scholar]
- European Commission. Directive 2009/54/EC of the European Parliament and of the Council of 18 June 2009 on the Exploitation and Marketing of Natural Mineral Waters. Off. J. Eur. Union 2009, 164, 45–58. [Google Scholar]
- European Commission. Council Directive 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption. Off. J. L. 1998, 330, 32–54. [Google Scholar]
- European Commission. Commission Directive 2003/40/EC of 16 May 2003 Establishing the List Concentration Limits and Labelling Requirements for the Constituents of Natural Mineral Waters and the Conditions for Using Ozone-Enriched Air for the Treatment of Natural Mineral Waters and Spring Waters. Off. J. L. 2003, 126, 34–39. [Google Scholar]
- Brenčič, M.; Vreča, P. Identification of sources and production processes of bottled waters by stable hydrogen and oxygen isotope ratios. Rapid Commun. Mass Spectrom. 2006, 20, 3205–3212. [Google Scholar] [CrossRef] [PubMed]
- Felipe-Sotelo, M.; Henshall-Bell, E.R.; Evans, N.D.M.; Read, D. Comparison of the chemical composition of British and continental European bottled waters by multivariate analysis. J. Food Compos. Anal. 2015, 39, 33–42. [Google Scholar] [CrossRef]
- Pip, E. Survey of the bottled drinking water available in Manitoba, Canada. Environ. Health Perspect. 2000, 108, 863–866. [Google Scholar] [CrossRef]
- Spangenbery, J.E.; Vennemann, T.W. The stable hydrogen and oxygen isotope variation of water stored in polyethylene terephthalate (PET) bottles. Rapid Commun. Mass Spectrom. 2008, 22, 672–676. [Google Scholar] [CrossRef]
- Ferjan, T.; Brenčič, M.; Vreča, P. Changes in isotopic composition of bottled natural mineral waters due to different storage conditions. In Proceedings of the V: International Symposium on Isotopes in Hydrology, Marine Ecosystems, and Climate Change Studies, Monaco, Monaco, 27 March–1 April 2011; International Atomic Energy Agency: Vienna, Austria, 2011; p. 153. [Google Scholar]
- Ferjan, T. Determination of Sources of Bottled Waters; in Slovene. Ph.D. Thesis, University of Ljubljana, Ljubljana, Slovenia, 2012. [Google Scholar]
- Cartwright, I.; Weaver, T.; Petrides, B. Controls on 87Sr/86Sr ratios of groundwater in silicate-dominated aquifers: SE Murray Basin, Australia. Chem. Geol. 2007, 246, 107–123. [Google Scholar] [CrossRef]
- Kim, G.E.; Shin, W.J.; Ryu, J.S.; Choi, M.S.; Lee, K.S. Identification of the origin and water type of various Korean bottled waters using strontium isotopes. J. Geochem. Explor. 2013, 132, 1–5. [Google Scholar] [CrossRef]
- Frei, K.; Frei, R. The geographic distribution of strontium isotopes in Danish surface waters–A base for provenance studies in archaeology, hydrology and agriculture. Appl. Geochem. 2011, 26, 326–340. [Google Scholar] [CrossRef]
- Dansgaard, W. Stable isotopes in precipitation. Tellus 1964, 16, 436–468. [Google Scholar] [CrossRef]
- Atekwana, E.A.; Krishnamurthy, R.V. Seasonal variations of dissolved inorganic carbon and δ13C of surface waters: Application of a modified gas evaluation technique. J. Hydrol. 1998, 205, 260–278. [Google Scholar] [CrossRef]
- Frengstad, B.S.; Lax, K.; Tarvainen, T.; Jæger, Ø.; Wigum, B.J. The chemistry of bottled mineral and spring waters from Norway, Sweden, Finland and Iceland. J. Geochem. Explor. 2010, 107, 350–361. [Google Scholar] [CrossRef]
- Birke, M.; Rauch, U.; Harazim, B.; Lorenz, H.; Glatte, W. Major and trace elements in German Bottled water, their regional distribution, and accordance with national and international standards. J. Geochem. Explor. 2010, 107, 245–271. [Google Scholar] [CrossRef]
- Montgomery, J.; Evans, J.A.; Wildman, G. 87Sr/86Sr isotope composition of bottled British mineral waters for environmental and forensic purposes. Appl. Geochem. 2006, 21, 1626–1634. [Google Scholar] [CrossRef]
- Voerkelius, S.; Lorenz Gesine, D.; Rummel, S.; Quétel, C.R.; Heiss, G.; Baxter, M.; Brach-Papa, C.; Deters-Itzelsberger, P.; Hoelzl, S.; Hoogewerff, J.; et al. Strontium isotopic signatures of natural mineral waters, the reference to a simple geological map and its potential for authentication of food. Food Chem. 2010, 118, 933–940. [Google Scholar] [CrossRef]
- Brenčič, M.; Vreča, P. Isotopic composition of dissolved inorganic carbon in bottled waters on the Slovene market. Food Chem. 2007, 101, 1516–1525. [Google Scholar] [CrossRef]
- Redondo, R.; Yélamas, J.G. Determination of CO2 origin (natural and industrial) in sparkling bottled waters by 13C/12C isotope ratio analysis. Food Chem. 2005, 92, 507–514. [Google Scholar] [CrossRef]
- Raco, B.; Dotsika, E.; Cerrina Feroni, A.; Battaglini, R.; Poutoukis, D. Stable isotope composition of Italian bottled waters. J. Geochem. Explor. 2013, 124, 203–211. [Google Scholar] [CrossRef]
- ARSO. 2019. Available online: www.arso.gov.si/en/water (accessed on 26 April 2019).
- Brenčič, M.; Vreča, P. General chemistry of bottled waters on the Slovene market. Mater. Geoenviron. 2005, 52, 549–560. [Google Scholar]
- Brenčič, M.; Ferjan, T.; Gosar, M. Geochemical survey of Slovenian bottled waters. J. Geochem. Explor. 2010, 107, 400–409. [Google Scholar] [CrossRef]
- Coplen, T.; Wildman, J.; Chen, J. Improvements in the gaseous hydrogen-water equilibration technique for hydrogen isotope ratio analysis. Anal. Chem. 1991, 63, 910–912. [Google Scholar] [CrossRef]
- Epstein, S.; Mayeda, T. Variations of 18O content of waters from natural sources. Geochim. Cosmochim. Acta 1953, 4, 213–224. [Google Scholar] [CrossRef]
- Avak, H.; Brand, W.A. The finning MAT HDO-equilibration—A fully automated H2O/gas phase equilibration system for hydrogen and oxygen isotope analyses. Thermo Electron. Corp. Appl. News 1995, 11, 1–13. [Google Scholar]
- Clesceri, L.S.; Greenberg, A.E.; Eaton, A.D.; American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF). Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Organization: Washington, DC, USA, 1998. [Google Scholar]
- Miyajima, T.; Yamada, Y.; Hanba, Y.T. Determining the stable isotope ratio of total dissolved inorganic carbon in lake water by GC/C/IRMS. Limnol. Oceanogr. 1995, 40, 994–1000. [Google Scholar] [CrossRef]
- Spötl, C. A robust and fast method of sampling and analysis of δ13C of dissolved inorganic carbon in ground waters. Isot. Environ Health Stud. 2005, 41, 217–221. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. User’s Guide to PHREEQC (Version 2)—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; Water Resources Investigations Report 99-4259; U.S. DEPARTMENT OF THE INTERIOR: Denver, CO, USA, 1999.
- R Core Team 2019: R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: https://www.R-project.org/ (accessed on 1 October 2019).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Petrović, T.; Zlokolica-Mandić, M.; Veljković, N.; Vidojević, D. Hydrogeological conditions for the forming and quality of mineral waters in Serbia. J. Geochem. Explor. 2010, 107, 373–381. [Google Scholar] [CrossRef]
- Demetriades, A.; Reimann, C.; Birke, M.; The Eurogeosurveys Geochemistry EGG Team. European ground water geochemistry using bottled water as a sampling medium. In Clean Soil and Safe Water; NATO Science for Peace and Security Series; Quercia, F.F., Vidojevic, D., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 115–139. [Google Scholar]
- Republic of Slovenia, Ministry of Health. Drinking Water Regulations of 1 March 2004; Official Gazette of the Republic Slovenia: Ljubljana, Slovenia, 2004; Volume 19, pp. 2155–2158.
- US EPA, Safe Drinking Water Act. 2019. Available online: www.epa.gov/ground-water-and-drinking-water (accessed on 26 April 2019).
- World Health Organization. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Lapajne, A. Hydrogeological properties of the thermal source Rimske Toplice. Geologija 2002, 45, 451–456. [Google Scholar]
- Kanduč, T.; Mori, N.; Kocman, D.; Stibilj, V.; Grassa, F. Hydrogeochemistry of Alpine springs from North Slovenia: Insights from stable isotopes. Chem. Geol. 2012, 300, 40–54. [Google Scholar] [CrossRef]
- Kanduč, T.; Mori, N.; Koceli, A.; Verbovšek, T. Hydrogeochemistry and isotopic geochemistry of the Velenje basin groundwater. Geologija 2016, 51, 7–22. [Google Scholar] [CrossRef]
- Verbovšek, T.; Kanduč, T. Isotope geochemistry of groundwater from fractured dolomite aquifers in central Slovenia. Aquat. Geochem. 2016, 22, 131–151. [Google Scholar] [CrossRef]
- Romanek, C.S.; Grossman, E.L.; Morse, J.W. Carbon isotopic fractionation in synthetic aragonite and calcite: Effects temperature and precipitation rate. Geochim. Cosmochim. Acta 1992, 46, 419–430. [Google Scholar] [CrossRef]
- Clarke, I.D.; Fritz, P. Environmental Isotopes in Hydrogeology; Lewis: New York, NY, USA, 1997; p. 328. [Google Scholar]



| Bottled by | Brand | Type | Source | Country of Origin |
|---|---|---|---|---|
| Radenska | Naturelle | Natural mineral water | Radenci | Slovenia |
| Dana | Dana | Natural mineral water | Mirna | |
| Union Brewery | Zala | Spring water | Ljubljana | |
| Voda Julijana | Julijana | Spring water | Tržič | |
| Laško Brewery | Oda | Spring water | Rimske Toplice | |
| Plastenka | Kaplja | Natural mineral water | Radomlje | |
| Voda 902 | Voda 902 | Spring water | Črnivec | |
| Costella | Costella | Natural mineral water | Fara | |
| Jamnica | Jana | Natural mineral water | Gorice Svetojanske | Croatia |
| Vodavoda | Vodavoda | Spring water | Gornja Toplica | Serbia |
| Aquamark | Laqueuille | Spring water | Pay-de-Dôme | France |
| Danone | Evian | Natural mineral water | Evian-les-Bains | |
| Coca-Cola HBC | Römerquelle | Natural mineral water | Edelstal and Prellenkirchen | Austria |
| Sample Name | 87Sr/86Sr |
|---|---|
| Radenska | 0.71164 ± 0.00035 |
| Dana | 0.70852 ± 0.00028 |
| Zala | 0.70965 ± 0.00025 |
| Julijana | 0.70832 ± 0.00034 |
| Oda | 0.70921 ± 0.00021 |
| Kaplja | 0.71194 ± 0.00020 |
| Voda 902 | 0.71942 ± 0.00026 |
| Costella | 0.70795 ± 0.00020 |
| Jana | 0.70833 ± 0.00022 |
| Vodavoda | 0.70854 ± 0.00018 |
| Laqueuille | 0.70400 ± 0.00015 |
| Evian | 0.70885 ± 0.00024 |
| Römerquelle | 0.70901 ± 0.00025 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuliani, T.; Kanduč, T.; Novak, R.; Vreča, P. Characterization of Bottled Waters by Multielemental Analysis, Stable and Radiogenic Isotopes. Water 2020, 12, 2454. https://doi.org/10.3390/w12092454
Zuliani T, Kanduč T, Novak R, Vreča P. Characterization of Bottled Waters by Multielemental Analysis, Stable and Radiogenic Isotopes. Water. 2020; 12(9):2454. https://doi.org/10.3390/w12092454
Chicago/Turabian StyleZuliani, Tea, Tjaša Kanduč, Rok Novak, and Polona Vreča. 2020. "Characterization of Bottled Waters by Multielemental Analysis, Stable and Radiogenic Isotopes" Water 12, no. 9: 2454. https://doi.org/10.3390/w12092454
APA StyleZuliani, T., Kanduč, T., Novak, R., & Vreča, P. (2020). Characterization of Bottled Waters by Multielemental Analysis, Stable and Radiogenic Isotopes. Water, 12(9), 2454. https://doi.org/10.3390/w12092454







