Hausdorff Fractal Derivative Model to Characterize Transport of Inorganic Arsenic in Porous Media
Abstract
1. Introduction
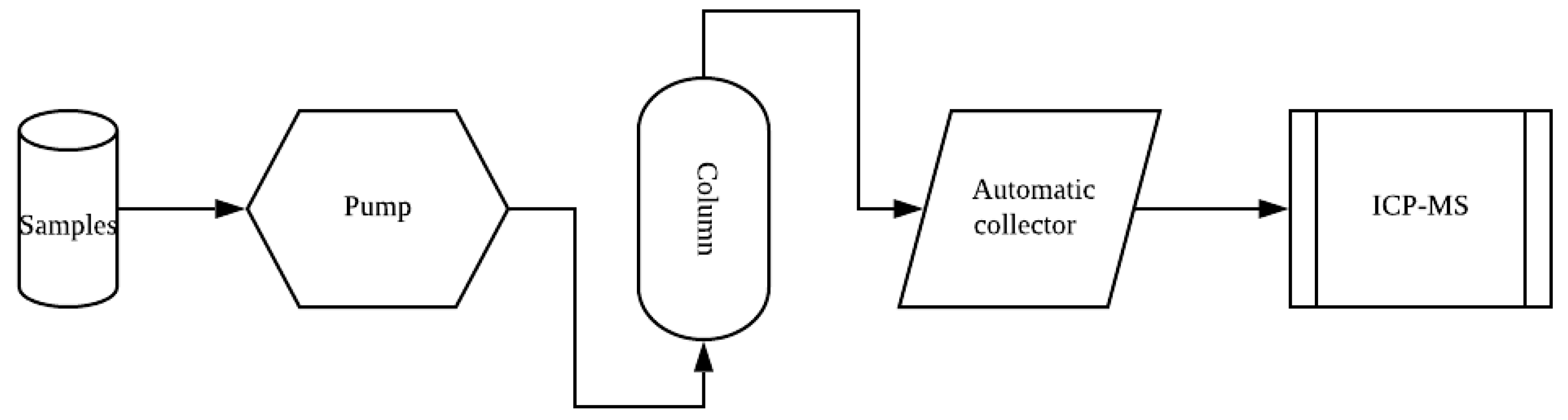
2. Experimental Method
2.1. Preparation of the Experiment
2.2. Arsenate Transport Experiments
3. Hausdorff Fractal Derivative Model
4. Results
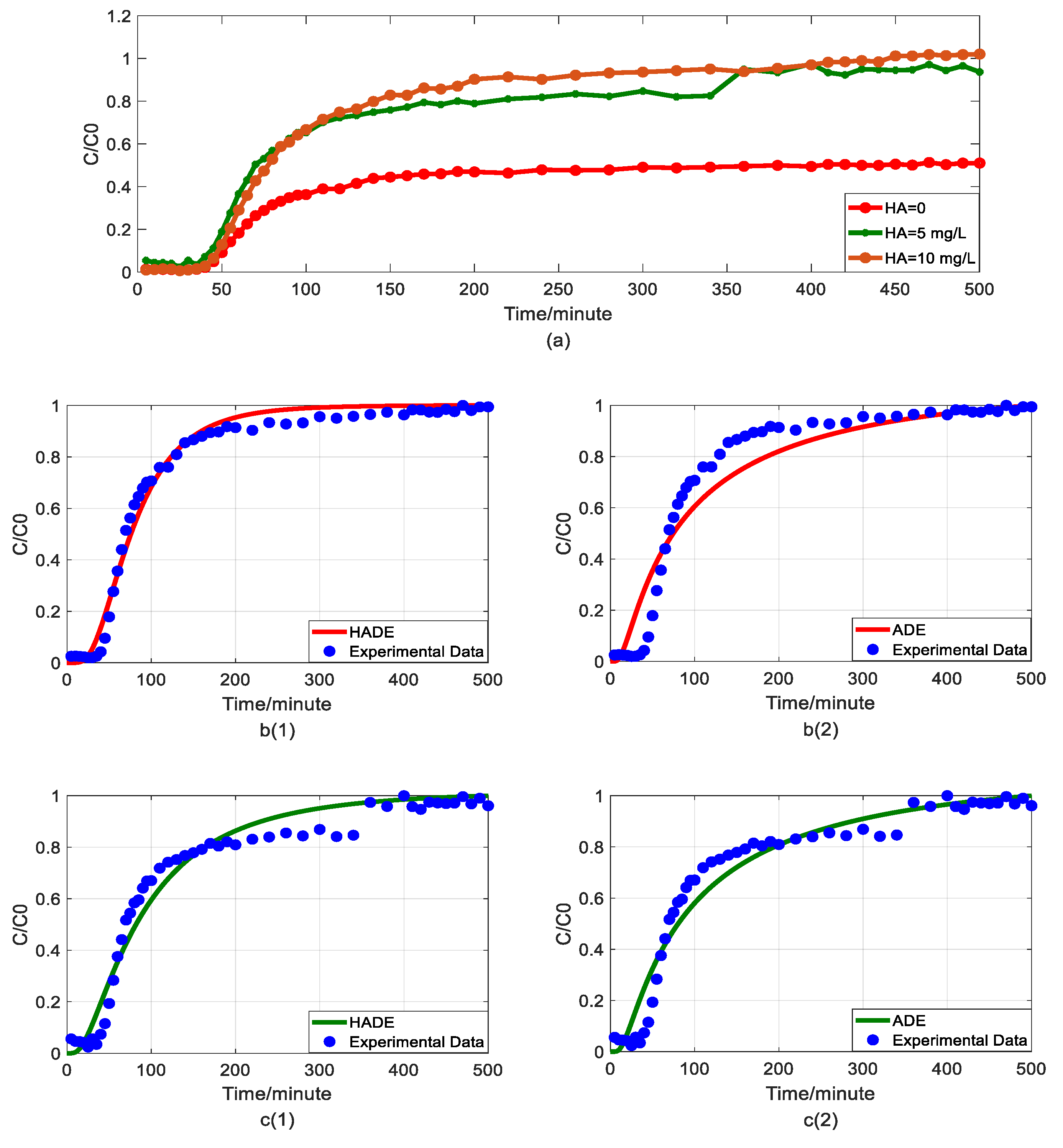
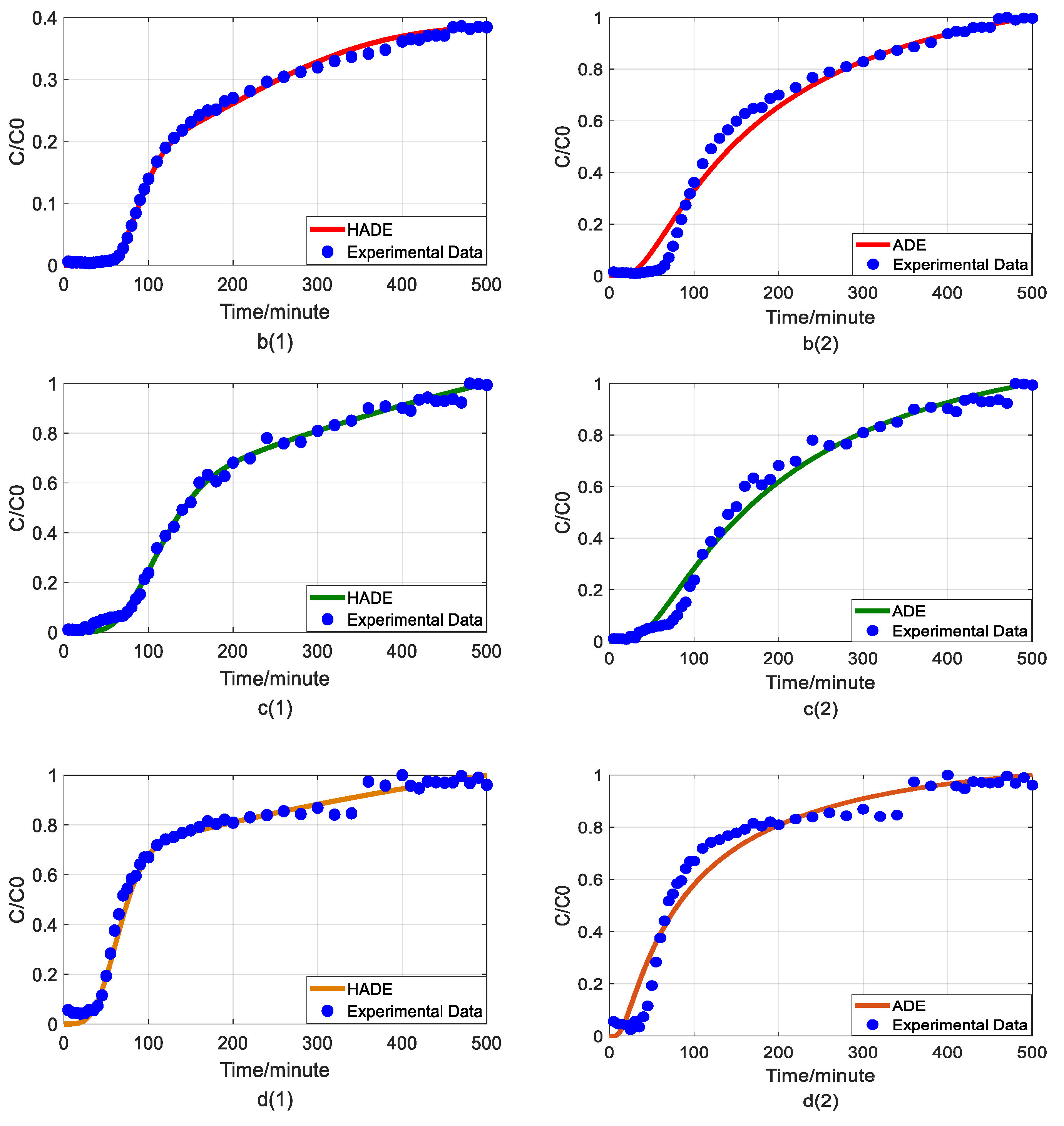
4.1. The Effect of HA on Arsenic Transport
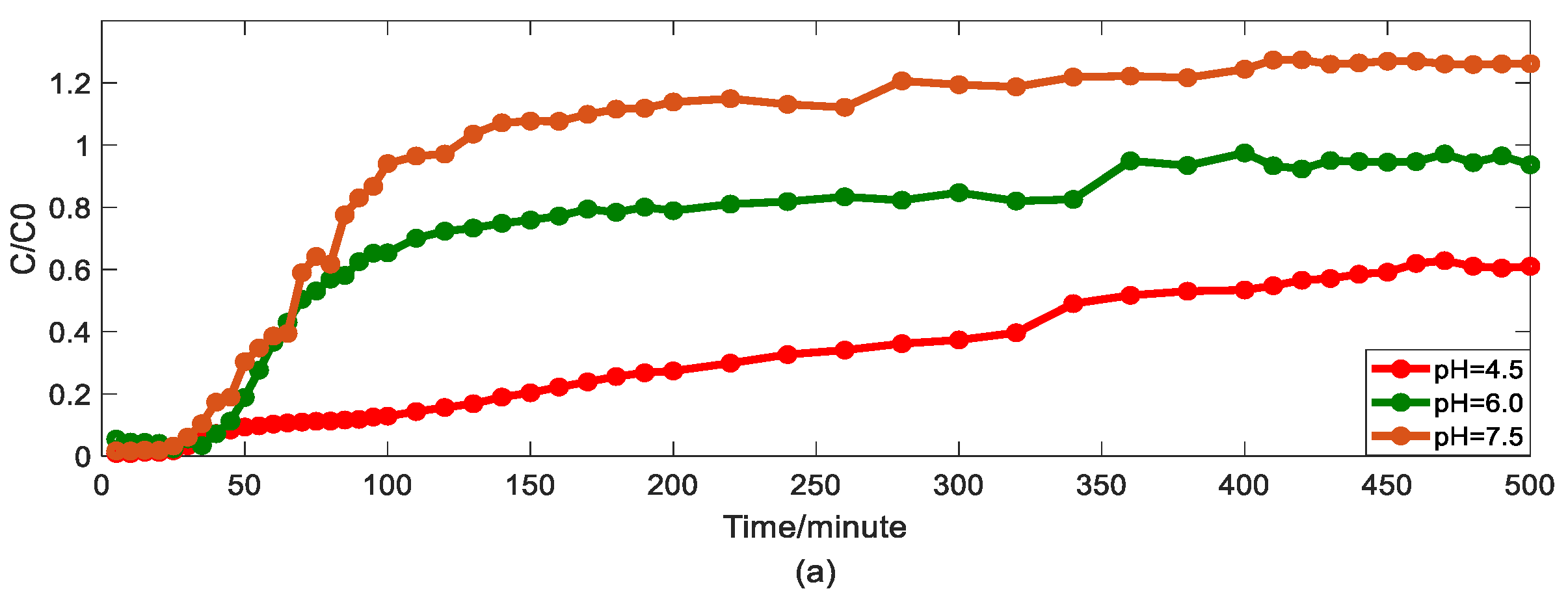
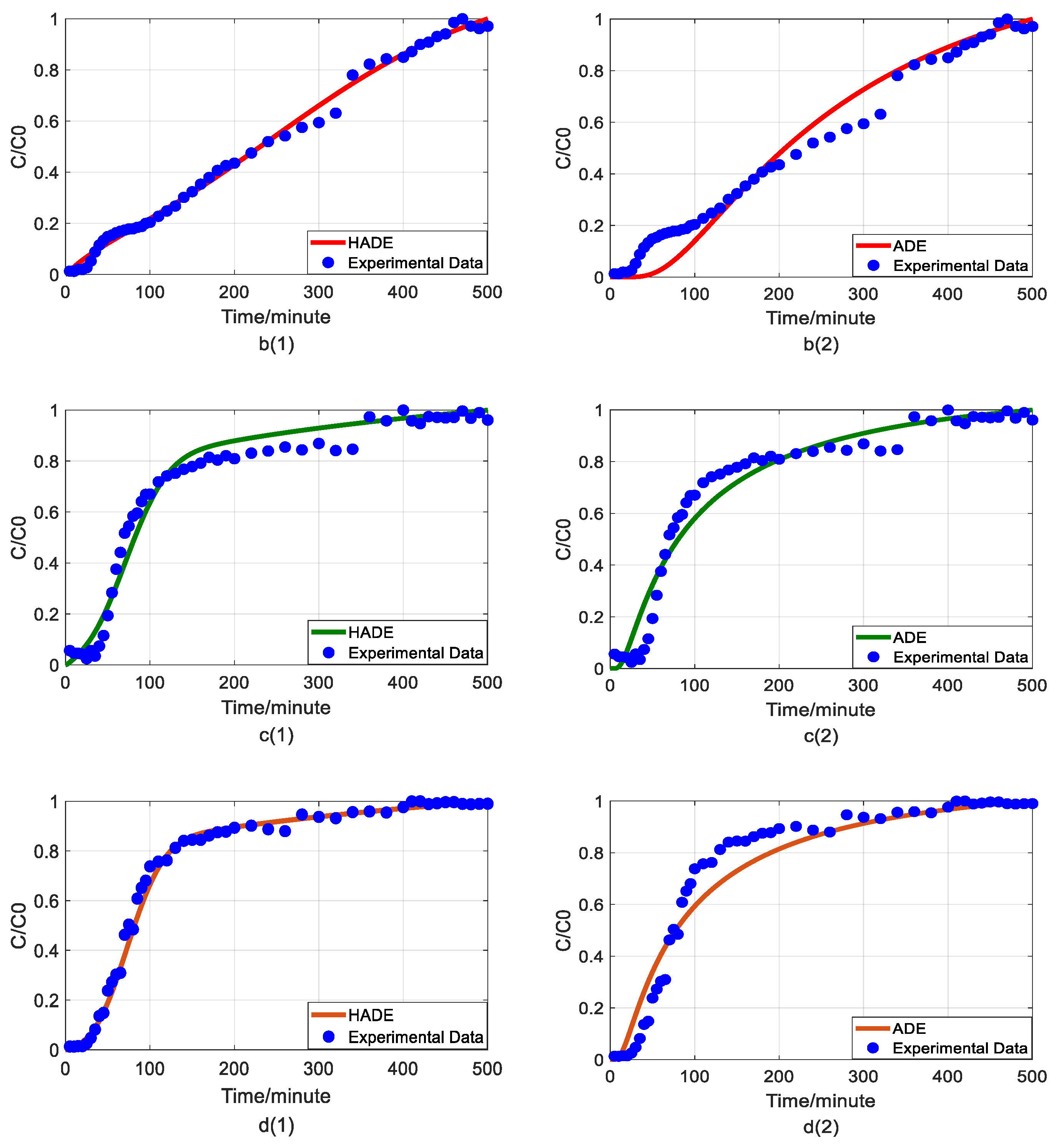
4.2. The Effect of pH on Arsenic Transport
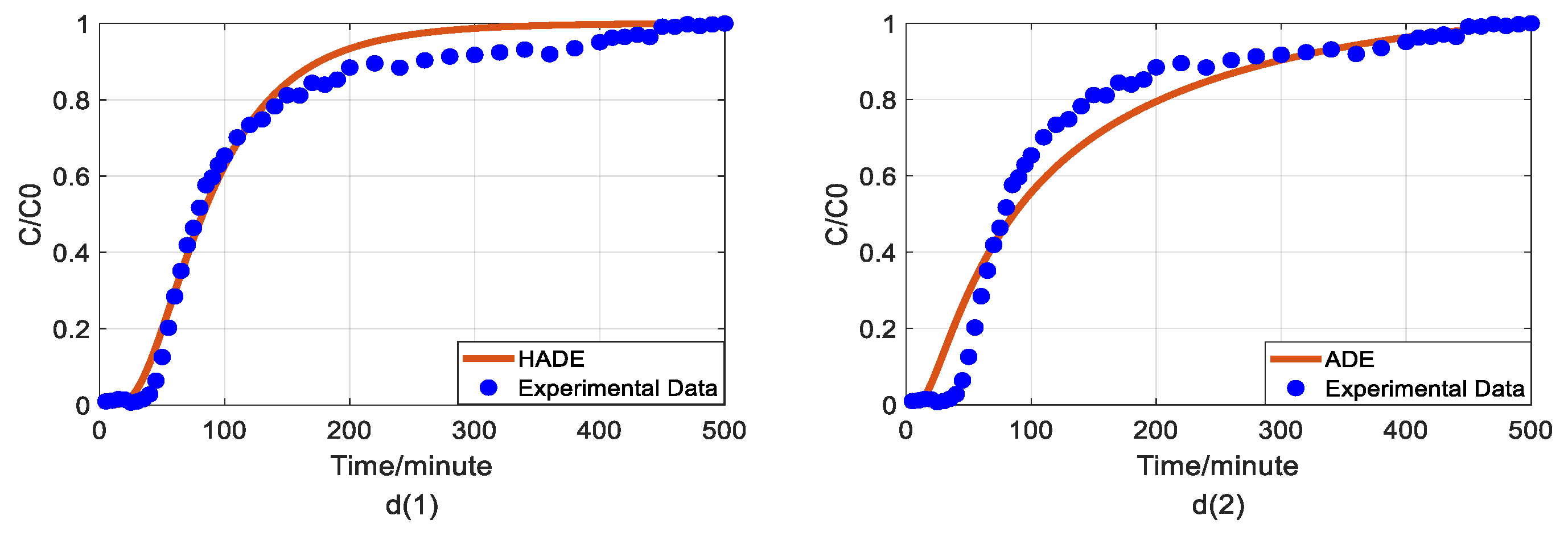
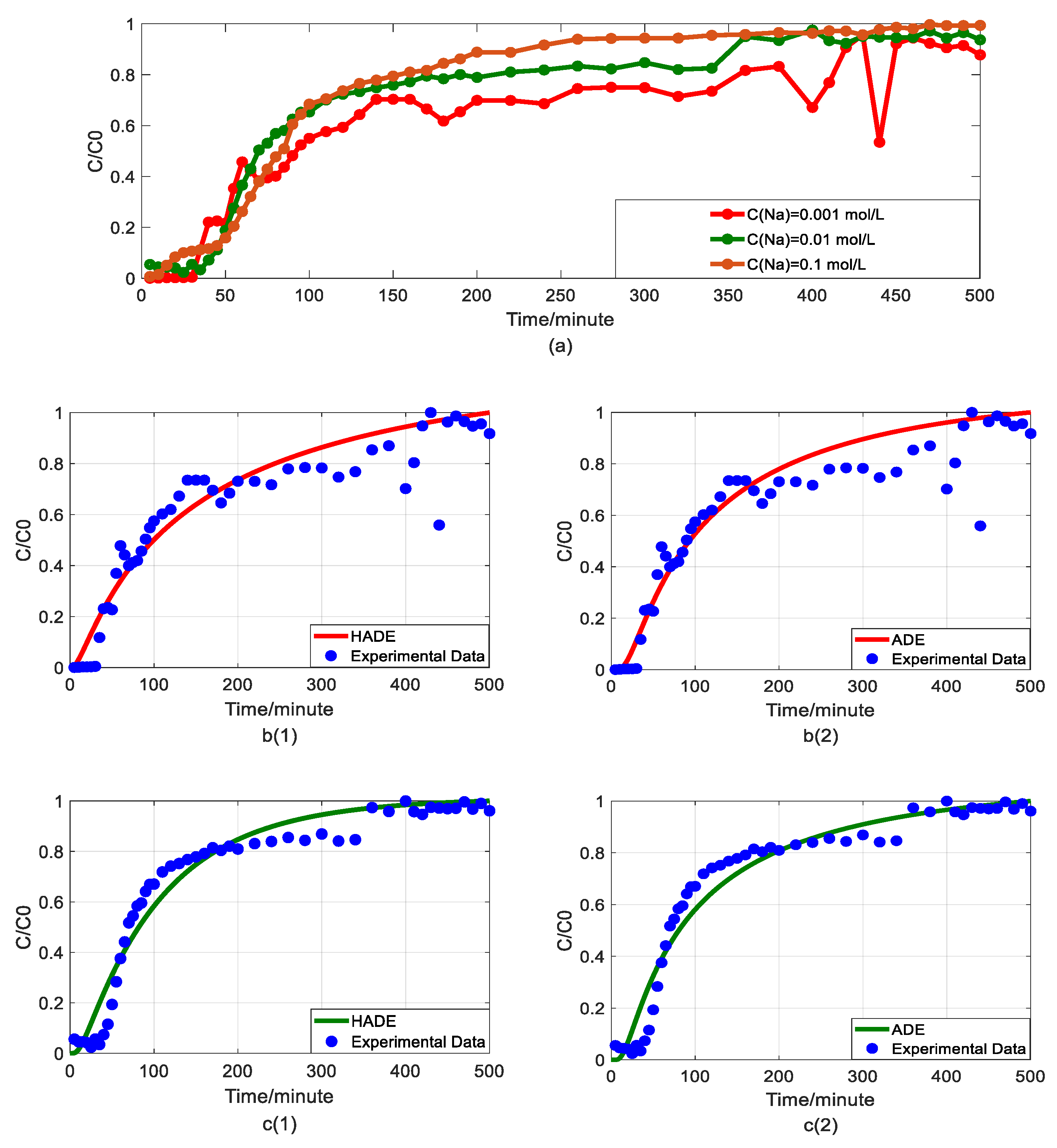
4.3. The Effect of Sodium on Arsenic Transport
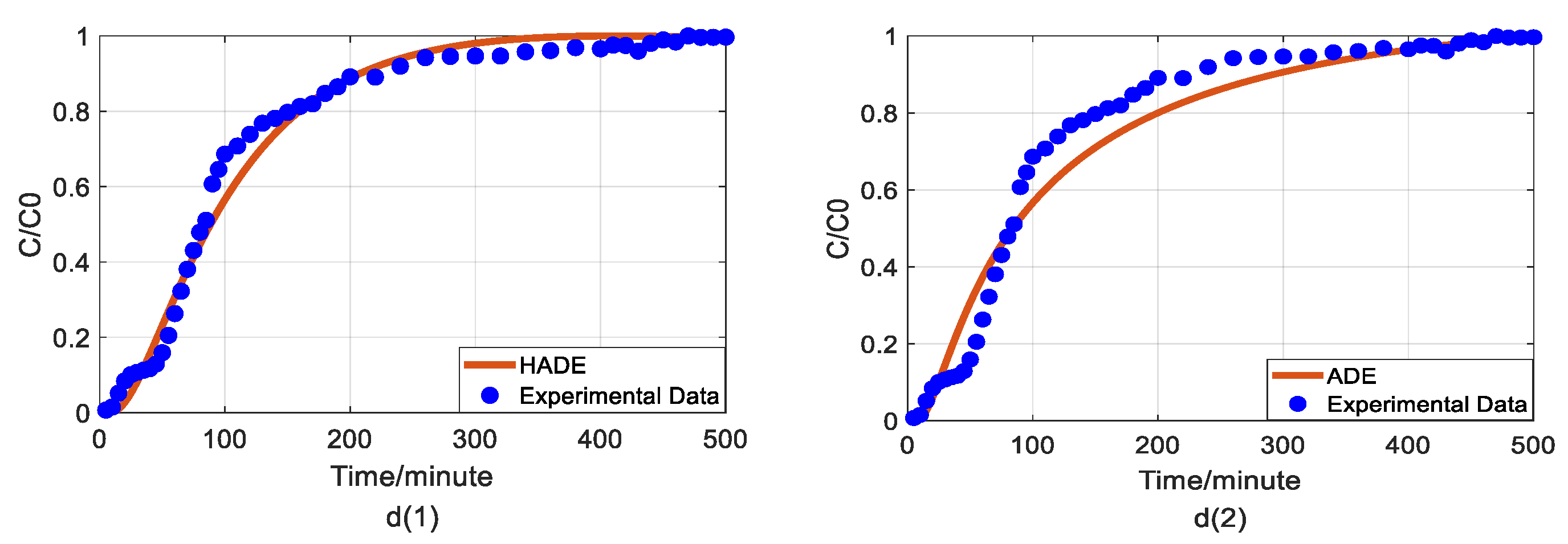
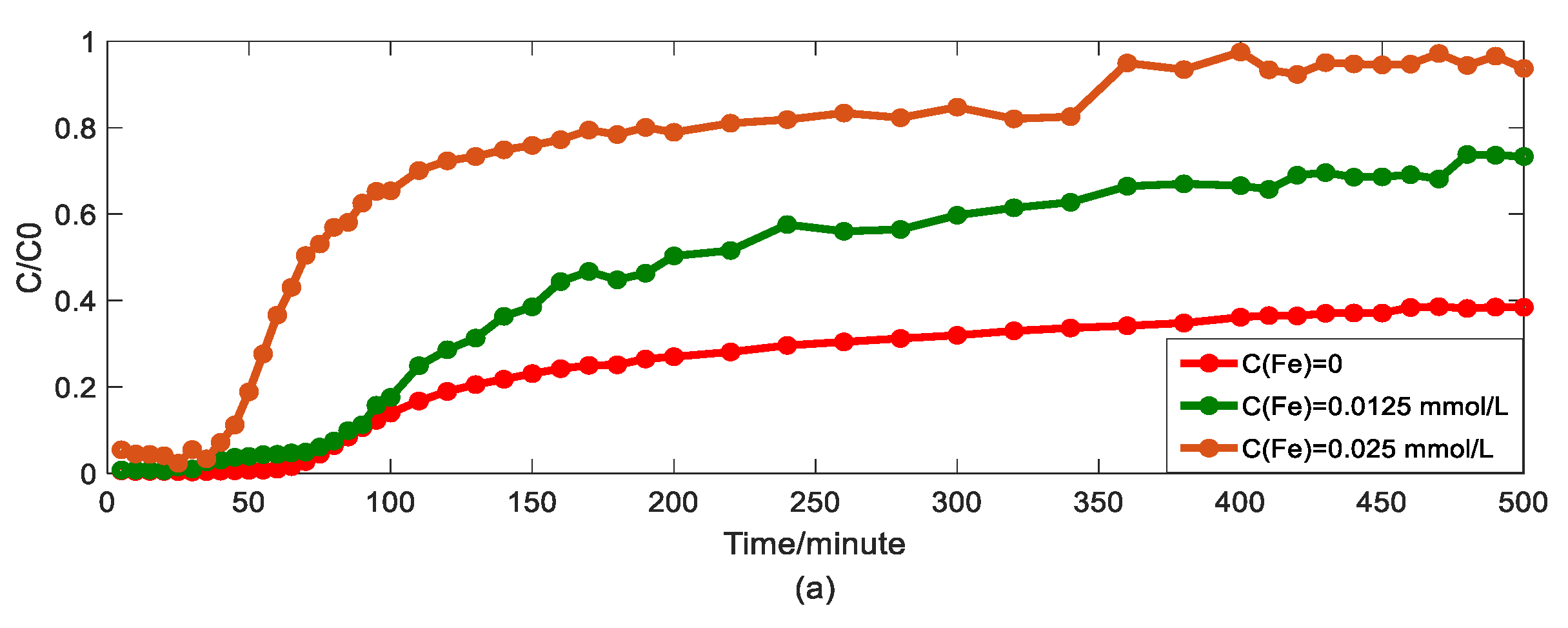
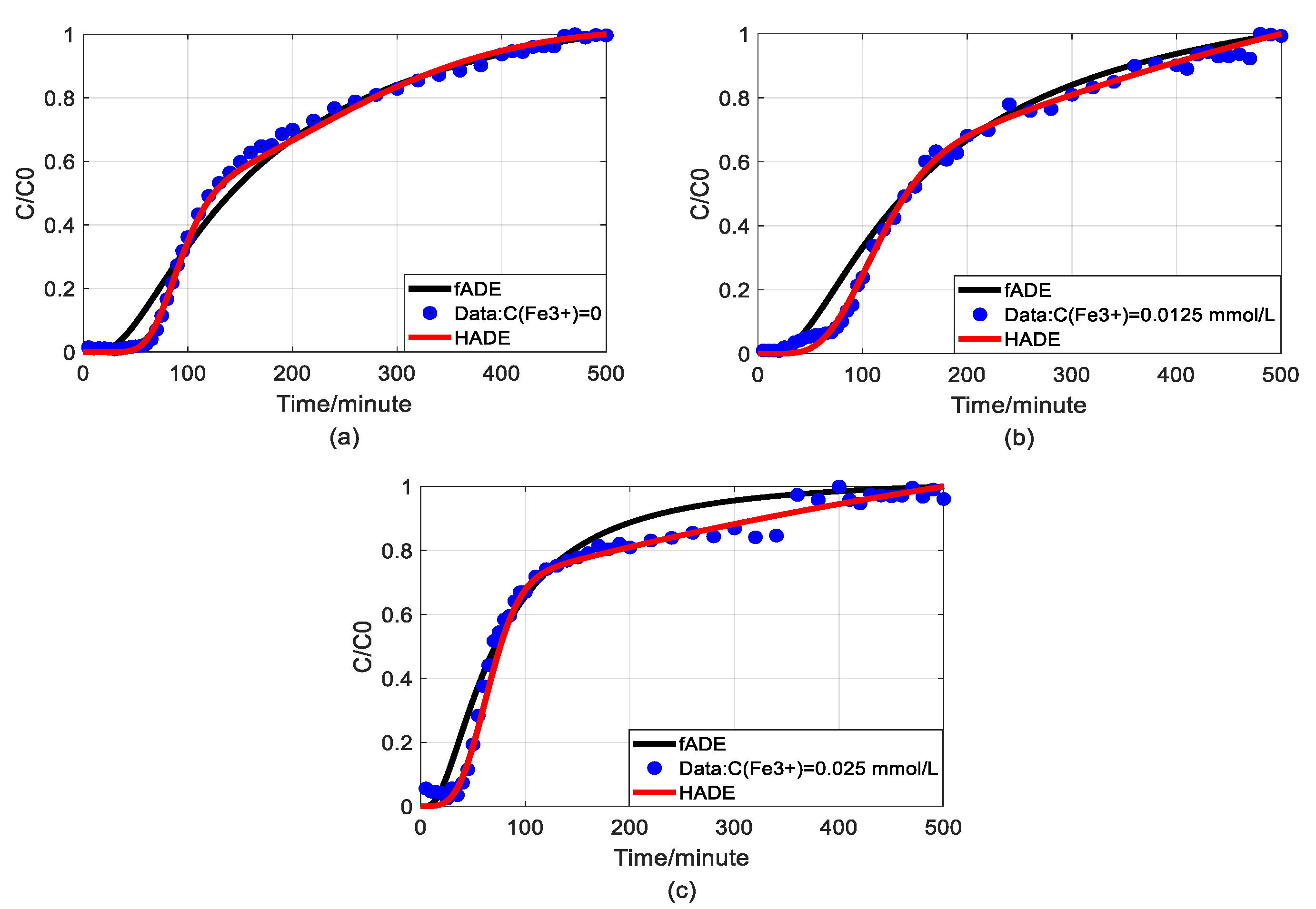
4.4. The Effect of Ferric Nitrate on Arsenic Transport
4.5. The Comparison between Fractional Model and HADE
5. Conclusions
- The physical mechanism in As(V) transport in this heterogeneous porous medium can be explained by the time HADE model. From the results above, the intensity of the sub-diffusion behavior of arsenic migration was negatively correlated with the increase of pH, HA concentration, and sodium ion concentration, and positively correlated with the increase of ferric ion concentration, which can be well reflected by the change of α.
- The HADE model has more obvious advantages than the ADE model in simulating arsenic migration. Compared with the fADE model (used in [33]), the advantages of the HADE model include an accurate description (especially at the early stage), simple expression, and easy-to-implement.
- By comparing the mean global errors in the simulation results of four variables (Fe3+, pH, HA, and Na+) in affecting arsenic migration, the best sorting based on the HADE fitting effect is Fe3+ > pH > HA > Na+.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brantley, S.L.; Lebedeva, M.I.; Balashov, V.N.; Singha, K.; Sullivan, P.L.; Stinchcomb, G. Toward a conceptual model relating chemical reaction fronts to water flow paths in hills. Geomorphology 2017, 277, 100–117. [Google Scholar] [CrossRef]
- Zheng, X.S.; Lu, A.H.; Gao, X.; Zhao, J.; Zheng, D.S. Contamination of heavy metals in soil present situation and method. Soil Environ. Sci. 2002, 11, 79–84. [Google Scholar]
- Rudnick, R.; Gao, S. Composition of the Continental Crust. Treatise Geochem. 2003, 3, 1–64. [Google Scholar]
- Wedepohl, K.H. The composition of the continental crust. Geochim. Cosmochim. Acta 1995, 59, 1217–1232. [Google Scholar] [CrossRef]
- Jomova, K.; Jenisova, Z.; Feszterova, M.; Baros, S.; Liska, J.; Hudecova, D.; Rhodes, C.J.; Valko, M. Arsenic: Toxicity, oxidative stress and human disease. J. Appl. Toxicol. 2011, 31, 95–107. [Google Scholar] [CrossRef]
- Zong, G.M.; Tang, Z.Z. Environmental cadmium, lead and arsenic pollution and health impact-research progress in China. J. Environ. Health 2006, 23, 562–565. [Google Scholar]
- Wu, Y.; Zhou, X.Y.; Lei, M.; Yang, J.; Ma, J.; Qiao, P.W.; Chen, T.B. Migration and transformation of arsenic: Contamination control and remediation in realgar mining areas. Appl. Geochem. 2017, 77, 44–51. [Google Scholar] [CrossRef]
- Tang, X.Y.; Zhu, Y.G.; Shan, X.Q.; McLaren, R.; Duan, J. The ageing effect on the bioaccessibility and fractionation of arsenic in soils from China. Chemosphere 2007, 66, 1183–1190. [Google Scholar] [CrossRef]
- Yang, J.; Mosby, D.E.; Casteel, S.W.; Blanchar, R.W. In vitro lead bioacceesibility and phosphate leaching as affected by surface application of phosphoric acid in lead-contaminated soil. Arch. Environ. Contam. Toxicol. 2002, 43, 399–405. [Google Scholar] [CrossRef]
- Girouard, E.; Zagury, G.J. Arsenic bioaccessibility in CCA-contaminated soils: Influence of soil properties, arsenic fractionation, and particle-size fraction. Sci. Total. Environ. 2009, 407, 2576–2585. [Google Scholar] [CrossRef]
- Wang, Q.; Cheng, T.; Yang, W. Influence of mineral colloids and humic substances on uranium(VI) transport in water-saturated geologic porous media. J. Contam. Hydrol. 2014, 170, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Nath, B.; Sarkar, S.; Chatterjee, D.; Ross, G.R.; Hidalgo, M. Size-fractionation of groundwater arsenic in alluvial aquifers of West Bengal, India: The role of organic and inorganic colloids. Sci. Total Environ. 2014, 468, 804–812. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Xie, Y.; Li, F.; Jiang, T.; Wang, Q.; Jiang, Z.M.; Wei, S.Q. Adsorption of arsenate on iron oxides as influenced by humic acids. J. Environ. Qual. 2015, 44, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Pajany, Y.M.; Hurel, C.; Marmier, N.; Roméo, M. Arsenic adsorption onto hematite and goethite. C. R. Chim. 2009, 12, 876–881. [Google Scholar] [CrossRef]
- Sharma, P.; Rolle, M.; Kocar, B.; Fendorf, S.; Kappler, A. Influence of natural organic matter on As transport and retention. Environ. Sci. Technol. 2011, 45, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Guo, H.; Lei, M.; Zhou, X.Y.; Li, F.L.; Yu, T.; Wei, R.F.; Zhang, H.Z.; Zhang, X.; Wu, Y. Arsenic adsorption and its fractions on aquifer sediment: Effect of pH, arsenic species, and iron/manganese minerals. Water Air Soil Pollut. 2015, 226, 260. [Google Scholar] [CrossRef]
- Zhang, H.; Selim, H.M. Modeling the Transport and Retention of Arsenic (V) in Soils. Soil Sci. Soc. Am. J. 2006, 70, 1677–1687. [Google Scholar] [CrossRef]
- Lim, M.S.; Yeo, I.W.; Clement, T.P.; Roh, Y.; Lee, K.K. Mathematical model for predicting microbial reduction and transport of arsenic in groundwater systems. Water Res. 2007, 41, 2079–2088. [Google Scholar] [CrossRef]
- Huang, X.; Luo, Y.; Zhang, J.; Liu, J.; Yang, J.; Liu, J. Study on convective diffusion model of migration behavior of As(III) and As (V) in bone charcoal research. J. Saf. Environ. 2014, 14, 210–215. [Google Scholar]
- Giménez, J.; Pablo, J.; Martínez, M.; Rovira, M.; Valderrama, C. Reactive transport of arsenic(III) and arsenic(V) on natural hematite: Experimental and modeling. J. Colloid Interface Sci. 2010, 348, 293–297. [Google Scholar] [CrossRef]
- Nikolaidis, N.P.; Dobbs, G.M.; Chen, J.; Lackovic, J.A. Arsenic mobility in contaminated lake sediments. Environ. Pollut. 2004, 129, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Eljamal, O.; Sasaki, K.; Hirajima, T. Numerical simulation for reactive solute transport of arsenic in permeable reactive barrier column including zero-valent iron. App. Math. Model. 2011, 35, 5198–5207. [Google Scholar] [CrossRef]
- Seuront, L.; Lagadeuc, Y. Characterisation of space–time variability in stratified and mixed coastal waters (Baie des Chaleurs, Quebec, Canada): Application of fractal theory. Mar. Ecol. Prog. Ser. 1997, 159, 81–95. [Google Scholar] [CrossRef]
- Helalizadeh, A.; Steinhagen, H.M.; Jamialahmadi, M. Application of fractal theory for characterisation of crystalline deposits. Chem. Eng. Sci. 2006, 61, 2069–2078. [Google Scholar] [CrossRef]
- Meng, F.G.; Zhang, H.M.; Li, Y.S.; Zhang, X.W.; Yang, F.L. Application of fractal permeation model to investigate membrane fouling in membrane bioreactor. J. Membr. Sci. 2005, 262, 107–116. [Google Scholar] [CrossRef]
- Liu, X.T.; Sun, H.G.; Zhang, Y.; Fu, Z.J. A scale-dependent finite difference approximation for time fractional differential equation. Comput. Mech. 2018, 63, 429–442. [Google Scholar] [CrossRef]
- Heymans, N.; Bauwens, J. Fractal rheological models and fractional differential equations for viscoelastic behavior. Rheol. Acta 1994, 33, 210–219. [Google Scholar] [CrossRef]
- Liang, Y.J.; Ye, A.Q.; Chen, W.; Gatto, R.G.; Perez, L.C.; Mareci, T.H.; Magin, R.L. A fractal derivative model for the characterization of anomalous diffusion in magnetic resonance imaging. Commun. Nonlinear Sci. 2016, 39, 529–537. [Google Scholar] [CrossRef]
- Ryutaro, K. Representation of random walk in fractal space-time. Physica A 1998, 248, 165–175. [Google Scholar]
- Chen, W. Time-space fabric underlying anomalous diffusion. Chaos Soliton Fract. 2006, 28, 923–929. [Google Scholar] [CrossRef]
- Chen, W.; Sun, H.G.; Zhang, X.D.; Korošak, D. Anomalous diffusion modeling by fractal and fractional derivatives. Comput. Math. Appl. 2009, 59, 1754–1759. [Google Scholar] [CrossRef]
- Sun, H.G.; Meerschaert, M.M.; Zhang, Y.; Zhu, J.T.; Chen, W. A fractal Richards’ equation to capture the non-Boltzmann scaling of water transport in unsaturated media. Adv. Water Resour. 2013, 52, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.R.; Mi, N.; He, C.; Yin, L.; Zhou, D.B.; Zhang, Y.; Sun, H.G.; Yang, S.; Li, S.; He, H. Transport of arsenic loaded by ferric humate colloid in saturated porous media. Chemosphere 2019, 240, 124987. [Google Scholar] [CrossRef]
- Pi, K.; Wang, Y.; Xie, X.; Huang, S.; Yu, Q.; Yu, M. Geochemical effects of dissolved organic matter biodegradation on arsenic transport in groundwater systems. J. Geochem. Explor. 2015, 149, 8–21. [Google Scholar] [CrossRef]
- Fogg, G.E.; Zhang, Y. Debates-Stochastic subsurface hydrology from theory to practice: A geologic perspective. Water Resour. Res. 2016, 53, 9235–9245. [Google Scholar] [CrossRef]
- Zhang, Y.; Meerschaert, M.M. Gaussian setting time for solute transport in fluvial systems. Water Resour. Res. 2011, 47, W08601. [Google Scholar] [CrossRef]
- Chen, D.; Sun, H.G.; Zhang, Y. Fractional dispersion equation for sediment suspension. J. Hydrol. 2013, 491, 13–22. [Google Scholar] [CrossRef]
- Guymon, G.L. A finite element solution of the one-dimensional diffusion-convection equation. Water Resour. Res. 1970, 6, 204–210. [Google Scholar] [CrossRef]
- Redman, A.D.; Macalady, D.L.; Ahmann, D. Natural organic matter affects arsenic speciation and sorption onto hematite. Environ. Sci. Technol. 2002, 36, 2889–2896. [Google Scholar] [CrossRef]
- Chen, Z.R.; Cai, Y.; Liu, G.L.; Gabriele, H.S.; Snyder, G.H.; Cisar, J.L. Role of soil-derived dissolved substances in arsenic transport and transformation in laboratory experiments. Sci. Total Environ. 2008, 406, 180–189. [Google Scholar] [CrossRef]
- Ma, J.; Guo, H.M.; Weng, L.P.; Li, Y.T.; Lei, M.; Chen, Y.L. Distinct effect of humic acid on ferrihydrite colloid-facilitated transport of arsenic in saturated media at different pH. Chemosphere 2018, 212, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.W.; Lu, Y.; Peng, L.F.; Wang, P.; Zhu, M.Q.; Dohnalkova, A.C.; Chen, H.; Lin, Z.; Dang, Z.; Shi, Z. Coupled kinetics of ferrihydrite transformation and As (V) sequestration under the effect of humic acids: A mechanistic and quantitative study. Environ. Sci. Technol. 2018, 52, 11632–11641. [Google Scholar] [CrossRef] [PubMed]
| Variables | Run 1 | Run 2 | Run 3 | Run 4 |
|---|---|---|---|---|
| Humic Acid (mg/L) | 0, 5, 10 | 5 | 5 | 5 |
| pH | 6.0 | 4.5, 6.0, 7.5 | 6.0 | 6.0 |
| Ironic Strength (mol/L) | 0.01 | 0.01 | 0.001, 0.01, 0.1 | 0.01 |
| Ferric nitrate (mmol/L) | 0.025 | 0.025 | 0.025 | 0, 0.0125, 0.025 |
| C (HA) | 0 | 5 mg/L | 10 mg/L |
|---|---|---|---|
| α (HADE) | 0.4 | 0.48 | 0.49 |
| D (HADE) | 0.04 | 0.08 | 0.04 |
| GE (HADE) | 0.054167 | 0.087855 | 0.062635 |
| D (ADE) | 0.057 | 0.051 | 0.046 |
| GE (ADE) | 0.12359 | 0.098227 | 0.1083 |
| pH | 4.5 | 6.0 | 7.5 |
|---|---|---|---|
| α (HADE) | 0.13 | 0.48 | 0.49 |
| D (HADE) | 0.15 | 0.08 | 0.05 |
| GE (HADE) | 0.052165 | 0.087855 | 0.052928 |
| D (ADE) | 0.01 | 0.051 | 0.054 |
| GE (ADE) | 0.13004 | 0.098227 | 0.10396 |
| C (Sodium) | 0.001 mol/L | 0.01 mol/L | 0.1 mol/L |
|---|---|---|---|
| α (HADE) | 0.41 | 0.48 | 0.49 |
| D (HADE) | 0.18 | 0.08 | 0.05 |
| GE (HADE) | 0.14957 | 0.087855 | 0.050204 |
| D (ADE) | 0.041 | 0.051 | 0.048 |
| GE (ADE) | 0.15134 | 0.098227 | 0.092982 |
| C (Iron) | 0 | 0.0125 mmol/L | 0.025 mmol/L |
|---|---|---|---|
| α (HADE) | 0.72 | 0.6 | 0.4 |
| D (HADE) | 0.011 | 0.015 | 0.1 |
| GE (HADE) | 0.0269 | 0.0355 | 0.0444 |
| D (ADE) | 0.02 | 0.017 | 0.051 |
| GE (ADE) | 0.080665 | 0.072521 | 0.098227 |
| Concentration (Iron, mmol/L) | 0 | 0.0125 | 0.025 |
|---|---|---|---|
| RMSE (fADE) | 0.0257 | 0.0225 | 0.0363 |
| α (fADE) | 0.72 | 0.6 | 0.4 |
| RMSE (HADE) | 0.00665 | 0.0081 | 0.0124 |
| α (HADE) | 0.4125 | 0.6480 | 0.7647 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, X.; Sun, H.; Zhang, Y.; Li, S.; Song, J.; Salsky, K. Hausdorff Fractal Derivative Model to Characterize Transport of Inorganic Arsenic in Porous Media. Water 2020, 12, 2353. https://doi.org/10.3390/w12092353
Hao X, Sun H, Zhang Y, Li S, Song J, Salsky K. Hausdorff Fractal Derivative Model to Characterize Transport of Inorganic Arsenic in Porous Media. Water. 2020; 12(9):2353. https://doi.org/10.3390/w12092353
Chicago/Turabian StyleHao, Xiaoxiao, HongGuang Sun, Yong Zhang, Shiyin Li, Jia Song, and Kate Salsky. 2020. "Hausdorff Fractal Derivative Model to Characterize Transport of Inorganic Arsenic in Porous Media" Water 12, no. 9: 2353. https://doi.org/10.3390/w12092353
APA StyleHao, X., Sun, H., Zhang, Y., Li, S., Song, J., & Salsky, K. (2020). Hausdorff Fractal Derivative Model to Characterize Transport of Inorganic Arsenic in Porous Media. Water, 12(9), 2353. https://doi.org/10.3390/w12092353





