Association between Drinking Water Nitrate and Adverse Reproductive Outcomes: A Systematic PRISMA Review
Abstract
1. Introduction
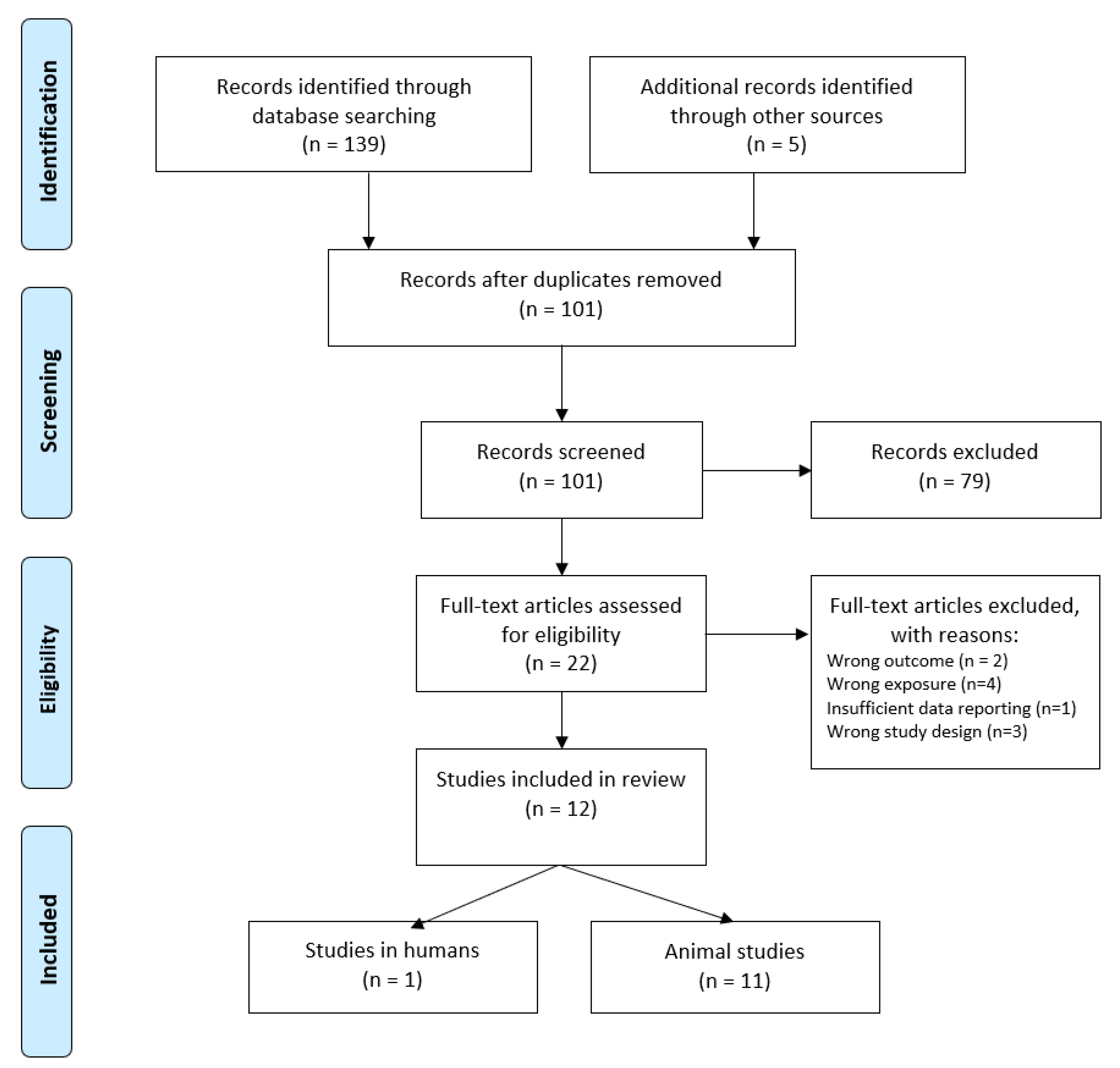
2. Methods
2.1. Search Strategy and Study Selection
- Studies containing a relevant population:
- Women of fertile age (15–51 years)
- Men of fertile age (15–60 years)
- Animal population
- Studies reporting a numerical exposure range for drinking water nitrate
- Studies containing a relevant control group
- Studies investigating at least one of the outcomes:
- Subfecundity or fertility (TTP, pregnancy rates, assisted reproductive technology treatment (ART))
- Spontaneous abortion
- Semen quality
- Original studies
- Studies with one of the following designs:
- Case-control study
- Cohort study
- Randomized control trials (RCT) study
2.2. Data Extraction and Quality Assessment
3. Results
3.1. Studies Reporting on Human Outcomes
3.2. Studies Reporting on Animal Outcomes
3.2.1. Studies on Female Animals
3.2.2. Studies on Male Animals
3.3. Quality of Included Studies
4. Discussion
4.1. Strengths and Limitations
4.2. Studies Reporting on Human Outcomes
4.3. Studies Reporting on Animal Outcomes
4.4. Comparisons: Studies in Humans and Animals
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- WHO (World Health Organization) Nitrate and nitrite in drinking-water: Background document for development of WHO guidelines for drinking-water quality. Available online: https://www.who.int/water_sanitation_health/dwq/chemicals/nitratenitrite2ndadd.pdf (accessed on 4 March 2020).
- Keshari, V.; Adeeb, B.; Simmons, A.E.; Simmons, T.W.; Diep, C.Q. Zebrafish as a model to assess the teratogenic potential of nitrite. J. Vis. Exp. 2016, 11, e53615. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.H.; deKok, T.M.; Levallois, P.; Brender, J.; Gulis, G.; Nolan, B.T.; VanDerslice, J. Workgroup report: Drinking-water nitrate and health - Recent findings and research needs. Environ. Health Perspect. 2005, 113, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Schullehner, J.; Hansen, B.; Thygesen, M.; Pedersen, C.B.; Sigsgaard, T. Nitrate in drinking water and colorectal cancer risk: A nationwide population-based cohort study. Int. J. Cancer 2018, 143, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Bruning-Fann, C.S.; Kaneene, J.B. The effects of nitrate, nitrite and N-nitroso compounds on human health: A review. Vet. Hum. Toxicol. 1993, 35, 521–538. [Google Scholar] [PubMed]
- Manassaram, D.M.; Backer, L.C.; Moll, D.M. A review of nitrates in drinking water: Maternal exposure and adverse reproductive and developmental outcomes. Environ. Health Perspect. 2006, 114, 320–327. [Google Scholar] [CrossRef]
- Ward, M.H.; Jones, R.R.; Brender, J.D.; de Kok, T.M.; Weyer, P.J.; Nolan, B.T.; Villanueva, C.M.; van Breda, S.G. Drinking water nitrate and human health: An updated review. Int. J. Environ. Res. Public Health 2018, 15, 1557. [Google Scholar] [CrossRef]
- van Grinsven, H.J.M.; Ward, M.H.; Benjamin, N.; de Kok, T.M. Does the evidence about health risks associated with nitrate ingestion warrant an increase of the nitrate standard for drinking water? Environ. Heal. A Glob. Access Sci. Source 2006, 5, 26. [Google Scholar] [CrossRef]
- Van Grinsven, H.J.; Rabl, A.; De Kok, T.M. Estimation of incidence and social cost of colon cancer due to nitrate in drinking water in the EU: A tentative cost-benefit assessment. Environ. Health A Glob. Access Sci. Source 2010, 9, 1–12. [Google Scholar] [CrossRef]
- Weselak, M.; Arbuckle, T.E.; Walker, M.C.; Krewski, D. The influence of the environment and other exogenous agents on spontaneous abortion risk. J. Toxicol. Environ. Heal. Part B Crit. Rev. 2008, 11, 221–241. [Google Scholar] [CrossRef]
- Slama, R.; Hansen, O.K.H.; Ducot, B.; Bohet, A.; Sorensen, D.; Giorgis Allemand, L.; Eijkemans, M.J.C.; Rosetta, L.; Thalabard, J.C.; Keiding, N.; et al. Estimation of the frequency of involuntary infertility on a nation-wide basis. Hum. Reprod. 2012, 27, 1489–1498. [Google Scholar] [CrossRef]
- Thoma, M.E.; McLain, A.C.; Louis, J.F.; King, R.B.; Trumble, A.C.; Sundaram, R.; Buck Louis, G.M. Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil. Steril. 2013, 99, 1324–1331.e1. [Google Scholar] [CrossRef] [PubMed]
- Sund, J.M.; Wright, M.J.; Simon, J. Weeds containing nitrates cause abortion in cattle. Agron. J. 1957, 49, 278–279. [Google Scholar] [CrossRef]
- Fan, A.M.; Steinberg, V.E. Health implications of nitrate and nitrite in drinking water: An update on methemoglobinemia occurrence and reproductive and developmental toxicity. Regul. Toxicol. Pharmacol. 1996, 23, 35–43. [Google Scholar] [CrossRef]
- Fan, A.M.; Willhite, C.C.; Book, S.A. Evaluation of the Nitrate Drinking Water Standard with Reference to Infant Methemoglobinemia and Potential Reproductive Toxicity. Regul. Toxicol. Pharmacol. 1987, 7, 135–148. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1–e28. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugweel, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 30 May 2019).
- Hooijmans, C.R. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 1–19. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, 1–8. [Google Scholar] [CrossRef]
- Aschengrau, A.; Zierler, S.; Cohen, A. Quality of community drinking water and the occurrence of spontaneous abortion. Arch. Environ. Health 1989, 44, 283–290. [Google Scholar] [CrossRef]
- Anderson, L.M.; Giner-Sorolla, A.; Ebeling, D.; Budinger, J.M. Effects of imipramine, nitrite, and dimethylnitrosamine on reproduction in mice. Res. Commun. Chem. Pathol. Pharmacol. 1978, 19, 311–327. [Google Scholar]
- Anderson, L.M.; Giner-Sorolla, A.; Haller, I.M.; Budinger, J.M. Effects of cimetidine, nitrite, cimetidine plus nitrite, and nitrosocimetidine on tumors in mice following transplacental plus chronic lifetime exposure. Cancer Res. 1985, 45, 3561–3566. [Google Scholar]
- Greenlee, A.R.; Ellis, T.M.; Berg, R.L. Low-dose agrochemicals and lawn-care pesticides induce developmental toxicity in murine preimplantation embryos. Environ. Health Perspect. 2004, 112, 703–709. [Google Scholar] [CrossRef] [PubMed]
- National Toxicology Program Toxicology and carcinogenesis studies of sodium nitrite (CAS NO. 7632-00-0) in F344/N rats and B6C3F1 mice (drinking water studies). Natl. Toxicol. Program Tech. Rep. Ser. 2001, 495, 7–273.
- Sleight, S.D.; Atallah, O.A. Reproduction in the Guinea Pig as Affected by Chronic Administration of Potassium Nitrate and Potassium Nitrite. Toxicol. Appl. Pharmacol. 1968, 12, 179–185. [Google Scholar] [CrossRef]
- Aly, H.A.A.; Mansour, A.M.; Abo-Salem, O.M.; Abd-Ellah, H.F.; Abdel-Naim, A.B. Potential testicular toxicity of sodium nitrate in adult rats. Food Chem. Toxicol. 2010, 48, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Amini, S.; Nikravesh, M.R.; Fazel, A.; Jalali, M.; Nabavi, A.S. Evaluation of the laminin α5 expression in mice testicular parenchyma following an exposure to water contaminated with sodium nitrite. Int. J. Adv. Biotechnol. Res. 2016, 7, 513–522. [Google Scholar]
- Amini, S.; Nikravesh, M.R.; Jalali, M.; Fazel, A.; Nabavi, A.S. Effects of oral administration of sodium nitrite on laminin expression in mice testicular interstitium: An immunohistochemical study. Biomed. Res. 2017, 28, 8671–8676. [Google Scholar]
- Amini, S.; Nikravesh, M.R.; Jalali, M.; Fazel, A.; Nabavi, A.S. Effect of chronic sodium nitrite administration on the expression of fibronectin in interstitial tissue of mice testis: An immunohistochemical study. Biomed. Res. 2018, 29, 91–95. [Google Scholar] [CrossRef]
- Attia, Y.A.; Abd El Hamid, E.A.; Ismaiel, A.M.; El-Nagar, A. The detoxication of nitrate by two antioxidants or a probiotic, and the effects on blood and seminal plasma profiles and reproductive function of New Zealand White rabbit bucks. Animal 2013, 7, 591–601. [Google Scholar] [CrossRef]
- Pant, N.; Srivastava, S.P. Testicular and spermatotoxic effect of nitrate in mice. Hum. Exp. Toxicol. 2002, 37–41. [Google Scholar] [CrossRef]
- Prager, S.; Micks, E.; Dalton, V.K. Pregnancy loss (miscarriage): Risk factors, etiology, clinical manifestations, and diagnostic evaluation – UpToDate. Available online: https://www.uptodate.com/contents/pregnancy-loss-miscarriage-risk-factors-etiology-clinical-manifestations-and-diagnostic-evaluation (accessed on 16 November 2019).
- Laegehaandbogen 2. trimester abort eller dødfødsel. Available online: https://www.sundhed.dk/sundhedsfaglig/laegehaandbogen/obstetrik/tilstande-og-sygdomme/komplikationer-i-svangerskabet/anden-trimester-abort-eller-doedfoedsel/ (accessed on 16 November 2019).
- Smith, R.B.; Toledano, M.B.; Wright, J.; Raynor, P.; Nieuwenhuijsen, M.J. Tap water use amongst pregnant women in a multi-ethnic cohort. Environ. Health A Glob. Access Sci. Source 2009, 8, S7. [Google Scholar] [CrossRef]
- Ruckart, P.Z.; Henderson, A.K.; Black, M.L.; Flanders, W.D. Are nitrate levels in groundwater stable over time? J. Expo. Sci. Environ. Epidemiol. 2008, 18, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, A.M.L.; Liew, Z.; Riis, A.H.; Stayner, L.T.; Ramlau-Hansen, C.H.; Sigsgaard, T.; Olsen, J. Nitrosatable drug exposure during pregnancy and risk of stillbirth. Pharmacoepidemiol. Drug Saf. 2019, 28, 1204–1210. [Google Scholar] [CrossRef] [PubMed]
- Swan, S.H.; Neutra, R.R.; Wrensch, M.; Hertz-picciotto, I.; Windham, G.C.; Fenster, L.; Epstein, D.M.; Deane, M. Is Drinking Water Related to Spontaneous Abortion? Reviewing the Evidence from the California Department of Health Services Studies. Epidemiology 1992, 3, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.; Steele, G.; Isiorho, S. Spontaneous abortions possibly related to ingestion of nitrate-contaminated well water--LaGrange County, Indiana, 1991–1994. MMWR. Morb. Mortal. Wkly. Rep. 1996, 45, 569–572. [Google Scholar]
- Super, M.; Heese, H.D.V.; MacKenzie, D., Dempster; Du Plessis, J.; Ferreira, J.J. An epidemiological study of well-water nitrates in a group of south west african/namibian infants. Water Res. 1981, 15, 1265–1270. [Google Scholar] [CrossRef]
- Gelperin, A.; Moses, V.K.; Bridger, C. Relationship of high nitrate community water supply to infant and fetal mortality. IMJ. Ill. Med. J. 1975, 147, 155–157, 186. [Google Scholar]
- Scragg, R.K.; Dorsch, M.M.; McMichael, A.J.; Baghurst, P.A. Birth defects and household water supply. Epidemiological studies in the Mount Gambier region of South Australia. Med. J. Aust. 1982, 2, 577–579. [Google Scholar] [CrossRef]
- Dorsch, M.M.; Scragg, R.K.; McMichael, A.J.; Baghurst, P.A.; Dyer, K.F. Congenital malformations and maternal drinking water supply in rural South Australia: A case-control study. Am. J. Epidemiol. 1984, 119, 473–486. [Google Scholar] [CrossRef]
- Alexander, E.K.; Pearce, E.N.; Brent, G.A.; Brown, R.S.; Chen, H.; Dosiou, C.; Grobman, W.A.; Laurberg, P.; Lazarus, J.H.; Mandel, S.J.; et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid 2017, 27, 315–389. [Google Scholar] [CrossRef]
- Schmitz, J.T. Methemoglobinemia - A Cause of Abortions? Preliminary Report. Obstet. Gynecol. 1961, 17, 413–415. [Google Scholar]
- Woodruff, T.J.; Sutton, P. The navigation guide systematic review methodology: A rigorous and transparent method for translating environmental health science into better health outcomes. Environ. Health Perspect. 2014, 122, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Sargeant, J.M.; O’Connor, A.M.; Gardner, I.A.; Dickson, J.S.; Torrence, M.E.; Dohoo, I.R.; Lefebvre, S.L.; Morley, P.S.; Ramirez, A.; Snedeker, K. The REFLECT statement: Reporting guidelines for randomized controlled trials in livestock and food safety: Explanation and elaboration. J. Food Prot. 2010, 73, 579–603. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.K.; Chapin, R.E.; Gulati, D.K.; George, J.D.; Price, C.J.; Marr, M.C.; Myers, C.B.; Barnes, L.H.; Fail, P.A.; Grizzle, T.B.; et al. Assessment of the reproductive and developmental toxicity of pesticide/fertilizer mixtures based on confirmed pesticide contamination in California and Iowa groundwater. Fundam. Appl. Toxicol. 1994, 22, 605–621. [Google Scholar] [CrossRef] [PubMed]
- Keyhanmanesh, R.; Hamidian, G.; Alipour, M.R.; Ranjbar, M.; Oghbaei, H. Protective effects of sodium nitrate against testicular apoptosis and spermatogenesis impairments in streptozotocin-induced diabetic male rats. Life Sci. 2018, 211, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Kammerer, M. [Influence of nitrates on reproduction in domestic mammals]. Contracept. Fertil. Sex. 1993, 21, 642–647. [Google Scholar]
- Bruning-Fann, C.S.; Kaneene, J.B.; Lloyd, J.W.; Stein, A.D.; Thacker, B.; Hurd, H.S. Associations between drinking-water nitrate and the productivity and health of farrowing swine. Prev. Vet. Med. 1996, 26, 33–46. [Google Scholar] [CrossRef]
- Frisbie, S.H.; Mitchell, E.J.; Sarkar, B. Urgent need to reevaluate the latest World Health Organization guidelines for toxic inorganic substances in drinking water. Environ. Health 2015, 14, 1–15. [Google Scholar] [CrossRef]
| First Author, Year, Country | Study Design | Study Population (Size and Selection) | Exposure Description | Outcome | Control for Maternal Age | Control for Other Confounding Factors | Effect of Nitrate Exposure | Main Results | Total NOS Score |
|---|---|---|---|---|---|---|---|---|---|
| Aschengrau, 1989, US [20] | Case-control | 286 cases, 1391 controls | Public water supply | Spontaneous abortion (<28 weeks gestation a) | Yes | Water quality, educational level, history of prior spontaneous abortion. | ↓ | Results from any detectable level: | 9 |
| 1: Undetectable level of nitrate (<0.1 mg-N/L) or nitrite (<0.01 mg-N/L) | All subjects, crude: Nitrate: OR (95% CI) = 0.4 (0.3, 0.6); Nitrite: OR (95% CI) = 1.1 (0.8, 1.6) | ||||||||
| 2: Any detectable level of nitrate (0.1–5.5 mg-N/L) | All subjects, adjusted: Nitrate: OR (95%CI) = 0.5 (0.2, 0.9) | ||||||||
| 3: Any detectable level of nitrite (0.01–0.03 mg-N/L) |
| First Author, Year, Country | Study Design | Animal Age, Species and Source | Groups | Study Duration | Outcome | Control for Feed and/or Water Consumption | Control for Other Factors | Effects of Nitrate Exposure | Main Results |
|---|---|---|---|---|---|---|---|---|---|
| Anderson, 1978, US [21] | RCT | 5–6 weeks old female and male swiss CD-1 mice from breeding farm | Drinking water containing: Experiment 1: 1: Control group (10 females); 2: NaNO2 (1 g/L/1000 ppm) in drinking water (10 females) Experiment 2: 1: Control group (20 females) 2: NaNO2 (1 g/L/1000 ppm) in drinking water (20 females) | Months | Experiment 1: No. of females with surviving litters, no. of offspring. | Yes | Housing (environmental and hygienic conditions, no. per cage) | ↑ | Experiment 1: Lower no. offspring in treated group; 30% vs. 0% in control group had no surviving litters (p < 0.01). Experiment 2: On average 5 days longer to conception, 3 more mice littered more than 30 days after males were added, smaller litters. None of these results had p < 0.05 comparing to control group. |
| Experiment 2: Conception time, infertility, litter size, rates of stillbirth, neonatal death. | |||||||||
| Anderson, 1985, US [22] | RCT | 7–8 weeks old female C57BL/6 and male BALB/c mice from laboratory | Drinking water containing: | Median 27 months (depending on survival) | No. of litters, effective no., number of females becoming pregnant, average time from introduction of the male until birth, no. of stillborn litters, average litter size at birth. | Yes | Housing (bedding, environmental conditions) | (↑) | No. of litters: 20 in control group, 14 in 184 ppm group, 15 in 1840 ppm group. Data not shown for the other outcomes. All outcomes described as non-significant. |
| 1: Control, no treatment (66 females, 51 males) | |||||||||
| 2: 184 ppm NaNO2 in drinking water (39 females, 52 males) | |||||||||
| 3: 1840 ppm NaNO2 in drinking water (65 females, 54 males) | |||||||||
| Greenlee, 2004, US [23] a | RCT | Mice embryos from CD-1 female mice (21–26 days old), from laboratory. Incubated in vitro | Drinking water containing: | 96 h (corresponding to the first 5–7 days after human conception) | Development to blastocyst, percentage of apoptosis, mean cell number per embryo. | Not applicable | Same incubation conditions | ↑ | NH4NO3: Reduced mean cell number per embryo (p ≤ 0.0005), increased percentage of apoptosis (p ≤ 0.05) compared to control. |
| 1: Control, 0.1% ethanol (20–25 embryos) | |||||||||
| 2: 1 ppm NH4NO3 in drinking water, which is based on RfD b (20–25 embryos) | |||||||||
| National Toxicology program, 2001, US [24] | RCT | Male and female B6C3F1 mice from animal farm, average age 6 weeks | Drinking water containing: | 98 days | Estrous cycle length, estrous stages. | Yes | Weight at baseline, Housing (no. per cage, environmental and hygienic conditions) | ↑ | Estrous cycle length was prolonged in 1500 ppm (p ≤ 0.05) and 5000 ppm (p ≤ 0.01) groups. |
| 1: 0 ppm NaNO3 (10 males, 10 females) | |||||||||
| 2: 375 ppm NaNO3 (10 males, 10 females) | |||||||||
| 3: 750 ppm NaNO3 (10 males, 10 females) | |||||||||
| 4: 1500 ppm NaNO3 (10 males, 10 females) | |||||||||
| 5: 3000 ppm NaNO3 (10 males, 10 females) | |||||||||
| 6: 5000 ppm NaNO3 (10 males, 10 females) | |||||||||
| National Toxicology program, 2001, US [24] | RCT | Male and female F344/N rats from animal farm, average age 7 weeks | Drinking water containing: | 98 days | Estrous cycle length, estrous stages. | Yes | Weight at baseline, Housing (no. per cage, environmental and hygienic conditions) | → | Non. |
| 1: 0 ppm NaNO3 (10 males, 10 females) | |||||||||
| 2: 375 ppm NaNO3 (10 males, 10 females) | |||||||||
| 3: 750 ppm NaNO3 (10 males, 10 females) | |||||||||
| 4: 1500 ppm NaNO3 (10 males, 10 females) | |||||||||
| 5: 3000 ppm NaNO3 (10 males, 10 females) | |||||||||
| 6: 5000 ppm NaNO3 (10 males, 10 females) | |||||||||
| Sleight, 1968, US [25] | RCT | Female and male guinea pigs of unknown age | Drinking water containing: | 100–240 days | No. of litters, relative percent reproductive performance, aborted fetuses, mummified or absorbed fetuses, stillborn fetuses, percent of fetal loss. Tissue samples from: Ovaries, uterus, cervix, thyroid. | Yes | Housing (no. per cage) | (↑) | KNO3: Poor reproductive performance at 30,000 ppm. KNO2: No live births were seen at 5000 or 10,000 ppm levels due to abortion or mummification. Dose-response relationship. |
| 1: 0 ppm KNO3/KNO2 | |||||||||
| 2: 300 ppm KNO3 | |||||||||
| 3: 2500 ppm KNO3 | |||||||||
| 4: 10,000 ppm KNO3 | |||||||||
| 5: 30,000 ppm KNO3 | |||||||||
| 6: 300 ppm KNO2 | |||||||||
| 7: 1000 ppm KNO2 | |||||||||
| 8: 2000 ppm KNO2 | |||||||||
| 9: 3000 ppm KNO2 | |||||||||
| 10: 4000 ppm KNO2 | |||||||||
| 11: 5000 ppm KNO2 | |||||||||
| 12: 10,000 ppm KNO2 | |||||||||
| 3–6 females per group + at least 1 male |
| First Author, Year, Country | Study Design | Animal Age, Species and Source | Groups | Study Duration | Outcome | Control for Feed and/or Water Consumption | Control for Other Factors | Effects of Nitrate Exposure | Main Results |
|---|---|---|---|---|---|---|---|---|---|
| Aly, 2010, Egypt [26] | RCT | Adult male swiss albino rats from animal facility | Drinking water containing: | 60 days | Sperm: Sperm count, sperm motility, daily sperm production.Testis: testicular lactic dehydrogenase-X (LDH-X), glucose-6-phosphatate dehydrogenase (G6PD), acid phosphatase (AP), testis weight, histopathological examination of testis, hydrogen peroxide generation (H2O2), lipid peroxidation (LPO), antioxidant enzymes activity, reduced glutathione (GSH). | No | Weight at baseline, housing (“standard laboratory conditions”) | ↑ | Significant (p < 0.05) negative effect in a dose-response relationship was seen in all treatment groups compared to controls on: sperm count, sperm motility, daily sperm production, testicular enzymes activity, hydrogen peroxide generation (H2O2), lipid peroxidation (LPO), reduced glutathione (GSH). Significant (p < 0.05) lower testis weight in treatment group: 100 and 200 mg/kg/day compared to controls. Induction of histopathological changes. Antioxidant activity significantly (p < 0.05) decreased in some groups compared to controls. |
| 1: 0 mg mg/kg/day NaNO3 (6 males) | |||||||||
| 2: 50 mg/kg/day NaNO3 (6 males) | |||||||||
| 3: 100 mg/kg/day NaNO3 (6 males) | |||||||||
| 4: 200 mg/kg/day NaNO3 (6 males) | |||||||||
| Amini, 2016, Iran [27] | RCT | Adult male mice from animal care unit | Drinking water containing: | 60 days | Expression of laminin α5 in basal level, middle level and elongated spermatid (apical compartment) of seminiferous epithelium. | No | Weight at baseline, housing (“controlled environment”) | ↑ | Basal and middle levels of seminiferous epithelium: Differences in expression of laminin but not significant. Elongated spermatid of seminiferous epithelium: Dose-response relationship difference in expression, significant (p = 0.032) increase in 50 mg/L treatment group compared to control. Real time PCR showed significant (p = 0.001) increase in laminin ratio in control group compared to experimental groups. |
| 1: 0 mg/L NaNO2 (5 males) | |||||||||
| 2: 3 mg/L NaNO2 (5 males) | |||||||||
| 3: 10 mg/L NaNO2 (5 males) | |||||||||
| 4: 50 mg/L NaNO2 (5 males) | |||||||||
| Amini, 2017, Iran [28] | RCT | Fertile male mice | Drinking water containing: | 60 days | Expression of laminin α5 as total amount in testicular tissue and in extracellular matrix of mice testicular interestitium. | No | No information | ↑ | Significant (p = 0.003) reduction in total level of laminin in testicular tissue in 50 mg/L group. No changes in expression of laminin in extracellular matrix. |
| 1: Control, free of contaminants (6 males) | |||||||||
| 2: 3 mg/L NaNO2 (6 males) | |||||||||
| 3: 50 mg/L NaNO2 (6 males) | |||||||||
| Amini, 2018, Iran [29] | RCT | Fertile male mice from animal house | Drinking water containing: | 60 days | Testis weight. Expression of fibronectin in testicular interstitial tissue. | No | Weight at baseline, housing (“suitable conditions”) | (↑) | Insignificantly (p = 0.094) weaker expression of fibronectin in 50 mg/L group. |
| 1: Control, pollutant-free (8 males) | |||||||||
| 2: 3 mg/L NaNO2 (8 males) | |||||||||
| 3: 50 mg/L NaNO2 (8 males) | |||||||||
| Attia, 2013, Egypt [30] | RCT | 16 weeks old New Zealand white male rabbits | Drinking water containing: | 322 days | Sperm: Sperm concentration, total sperm, total live sperm, total dead sperm, total abnormal sperm. Age at first ejaculate, male fertility, number of offspring, testosterone (seminal and blood plasma), histopathology of testis. | Yes | Weight at baseline, housing (no. per cage, environmental and hygienic conditions) | ↑ | Significant negative effect was seen in 700 ppm group compared to 350 ppm and control (dose-response relationship) on: Blood plasma testosterone (p = 0.004), seminal plasma testosterone (p = 0.0001), sperm concentration (p = 0.03), total sperm output (p = 0.008), total live sperm (p = 0.002), total dead sperm (p = 0.006), total abnormal sperm (p = 0.01), age at first ejaculate (p = 0.01), fertility (p = 0.03), no. of offspring (p = 0.02). |
| 1: 14 ppm/tap water (7/4/3 males) | |||||||||
| 2: 350 ppm NaNO3 (7/4/3 males) | |||||||||
| 3: 700 ppm NaNO3 (7/4/3 males) | |||||||||
| National Toxicology Program, 2001, US [24] | RCT | Male and female B6C3F1 mice from animal farm, average age 6 weeks | Drinking water containing: | 98 days | Organ weights: Cauda epididymis, epididymis, testis. Sperm: Spermatid heads, spermatid count, sperm motility, sperm concentration. | Yes | Weight at baseline, housing (no. per cage, environmental and hygienic conditions) | ↑ | Sperm motility in 5000 ppm (p ≤ 0.01) males was significantly decreased. |
| 1: 0 ppm NaNO3 in (10 males, 10 females) | |||||||||
| 2: 375 ppm NaNO3 (10 males, 10 females) | |||||||||
| 3: 750 ppm NaNO3 (10 males, 10 females) | |||||||||
| 4: 1500 ppm NaNO3 (10 males, 10 females) | |||||||||
| 5: 3000 ppm NaNO3 (10 males, 10 females) | |||||||||
| 6 5000 ppm NaNO3 (10 males, 10 females) | |||||||||
| National Toxicology Program, 2001, US [24] | RCT | Male and female F344/N rats from animal farm, average age 7 weeks | Drinking water containing: | 98 days | Organ weights: Cauda epididymis, epididymis, testis. Sperm: Spermatid heads, spermatid count, sperm motility, sperm concentration. | Yes | Weight at baseline, housing (no. per cage, environmental and hygienic conditions) | ↑ | Sperm motility lower in 1500 ppm (p ≤ 0.05) and 5000 ppm (p ≤ 0.01) groups. Epididymis weight lower in 5000 ppm group (p ≤ 0.05). |
| 1: 0 ppm NaNO3 in (10 males, 10 females) | |||||||||
| 2: 375 ppm NaNO3 (10 males, 10 females) | |||||||||
| 3: 750 ppm NaNO3 (10 males, 10 females) | |||||||||
| 4: 1500 ppm NaNO3 (10 males, 10 females) | |||||||||
| 5: 3000 ppm NaNO3 (10 males, 10 females) | |||||||||
| 6 5000 ppm NaNO3 (10 males, 10 females) | |||||||||
| Pant, 2002, India [31] | RCT | 7 weeks old male Swiss white mice from animal colony | Drinking water containing: | 35 days | Organ weights: Testis, epididymis, seminal vesicle, ventral prostate, coagulating gland. Sperm: Sperm count, sperm motility, morphological abnormalities in sperm. Testicular enzymes activity: Sorbitol dehydrogenase (SDH), lactate dehydrogenase (LDH), 17-β hydroxysteroid dehydrogenase (17-βHSD), acid phosphatase (AP), β glucuronidase (β-G), γ glutamyl transpeptidase (γ-GT). | Yes | Weight at baseline, Housing (“standard laboratory conditions”) | ↑ | Significant (p < 0.05) effect in 900 ppm exposure group; declined activity of 17-β hydroxysteroid dehydrogenase (17-βHSD, increased activity of γ glutamyl transpeptidase (γ-GT), decrease in sperm count and motility, increase in total abnormal sperm. |
| 1: Control, tap water (5 males) | |||||||||
| 2: 90 ppm KNO3 (5 males) | |||||||||
| 3: 200 ppm KNO3 (5 males) | |||||||||
| 4: 500 ppm KNO3 (5 males) | |||||||||
| 5: 700 ppm KNO3 (5 males) | |||||||||
| 6: 900 ppm KNO3 (5 males) | |||||||||
| Sleight, 1968, US [25] | RCT | Female and male guinea pigs of unknown age | Drinking water containing: | 100–240 days | Reproductive performance. | Yes | Housing (no. per cage) | → | Conception at all levels of treatment; male fertility apparently not impaired. |
| 1: 0 ppm KNO3/KNO2 | |||||||||
| 2: 300 ppm KNO3 | |||||||||
| 3: 2500 ppm KNO3 | |||||||||
| 4: 10,000 ppm KNO3 | |||||||||
| 5: 30,000 ppm KNO3 | |||||||||
| 6: 300 ppm KNO2 | |||||||||
| 7: 1000 ppm KNO2 | |||||||||
| 8: 2000 ppm KNO2 | |||||||||
| 9: 3000 ppm KNO2 | |||||||||
| 10: 4000 ppm KNO2 | |||||||||
| 11: 5000 ppm KNO2 | |||||||||
| 12: 10,000 ppm KNO2 | |||||||||
| 3–6 females per group + at least 1 male |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clausen, H.S.; Ebdrup, N.H.; Barsøe, I.M.; Lyngsø, J.; Schullehner, J.; Ramlau-Hansen, C.H.; Bay, B.; Knudsen, U.B. Association between Drinking Water Nitrate and Adverse Reproductive Outcomes: A Systematic PRISMA Review. Water 2020, 12, 2287. https://doi.org/10.3390/w12082287
Clausen HS, Ebdrup NH, Barsøe IM, Lyngsø J, Schullehner J, Ramlau-Hansen CH, Bay B, Knudsen UB. Association between Drinking Water Nitrate and Adverse Reproductive Outcomes: A Systematic PRISMA Review. Water. 2020; 12(8):2287. https://doi.org/10.3390/w12082287
Chicago/Turabian StyleClausen, Hannah S., Ninna H. Ebdrup, Ida M. Barsøe, Julie Lyngsø, Jörg Schullehner, Cecilia H. Ramlau-Hansen, Bjørn Bay, and Ulla B. Knudsen. 2020. "Association between Drinking Water Nitrate and Adverse Reproductive Outcomes: A Systematic PRISMA Review" Water 12, no. 8: 2287. https://doi.org/10.3390/w12082287
APA StyleClausen, H. S., Ebdrup, N. H., Barsøe, I. M., Lyngsø, J., Schullehner, J., Ramlau-Hansen, C. H., Bay, B., & Knudsen, U. B. (2020). Association between Drinking Water Nitrate and Adverse Reproductive Outcomes: A Systematic PRISMA Review. Water, 12(8), 2287. https://doi.org/10.3390/w12082287





