Precipitant Effects on Aggregates Structure of Asphaltene and Their Implications for Groundwater Remediation
Abstract
1. Introduction
2. Experiments
2.1. Materials
2.2. Experiments
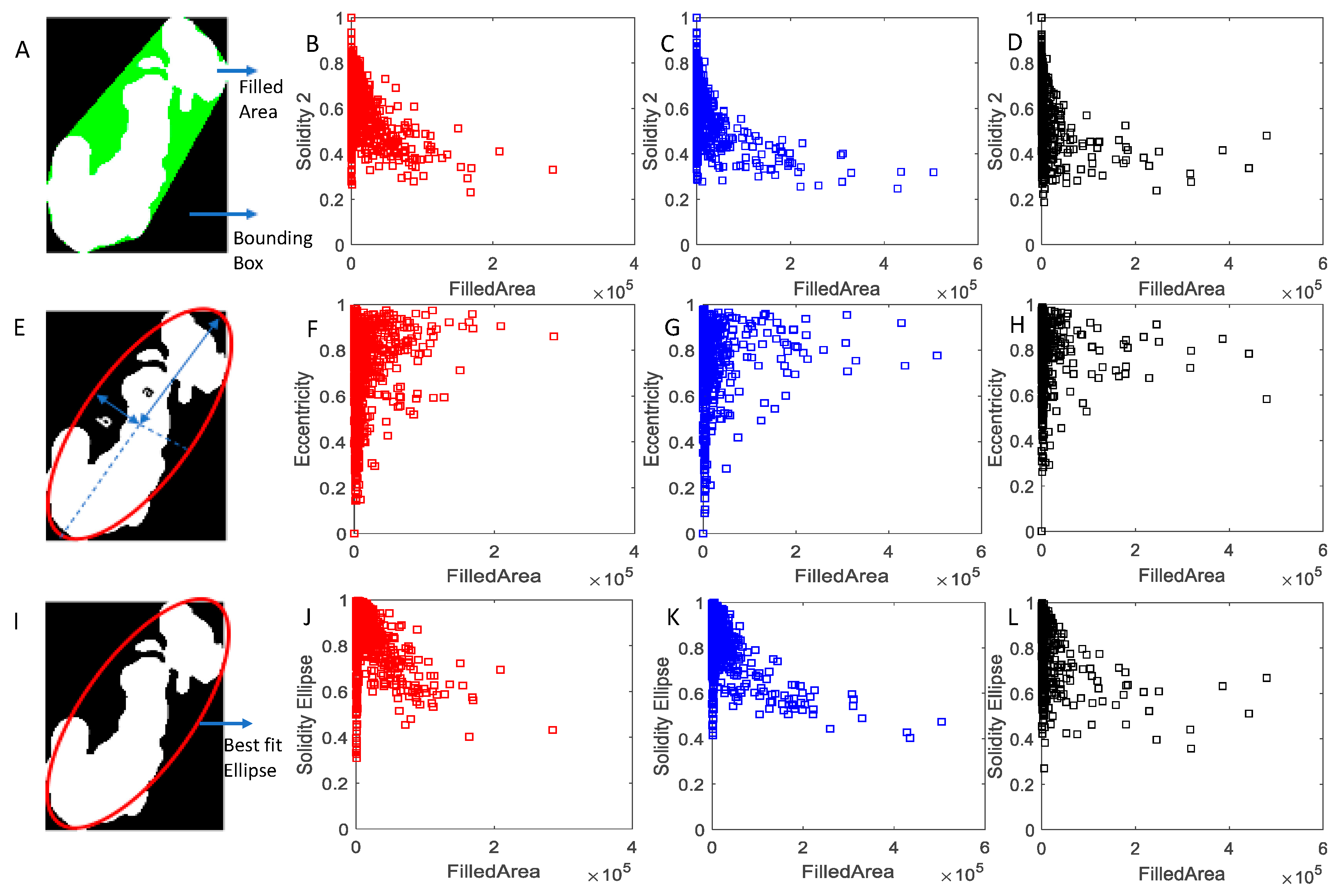
2.3. Image Analysis
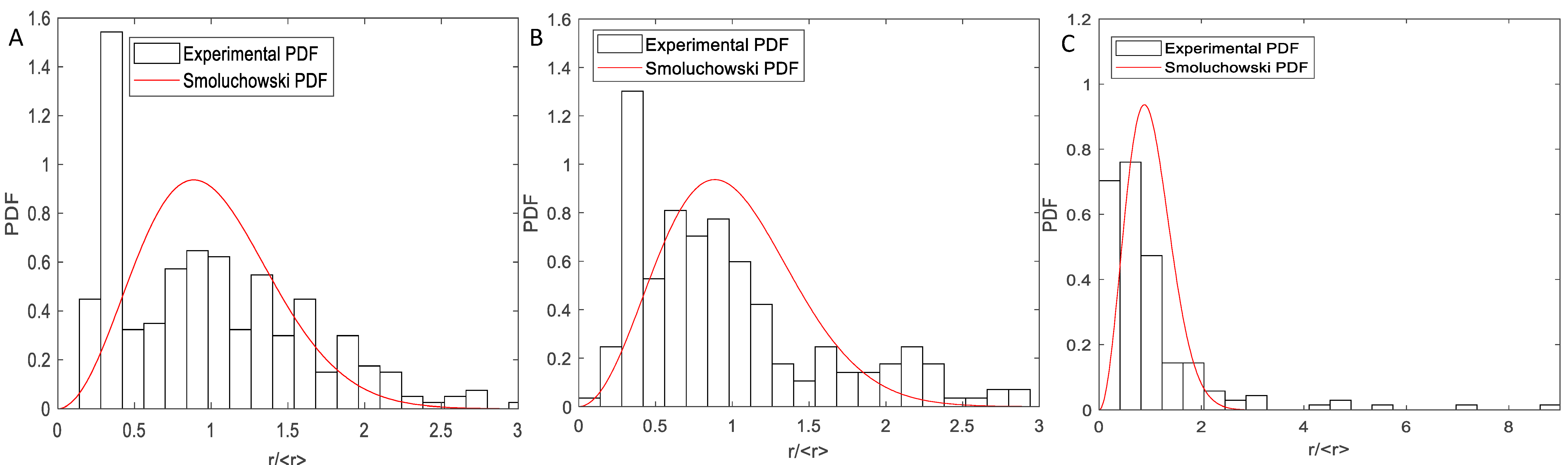
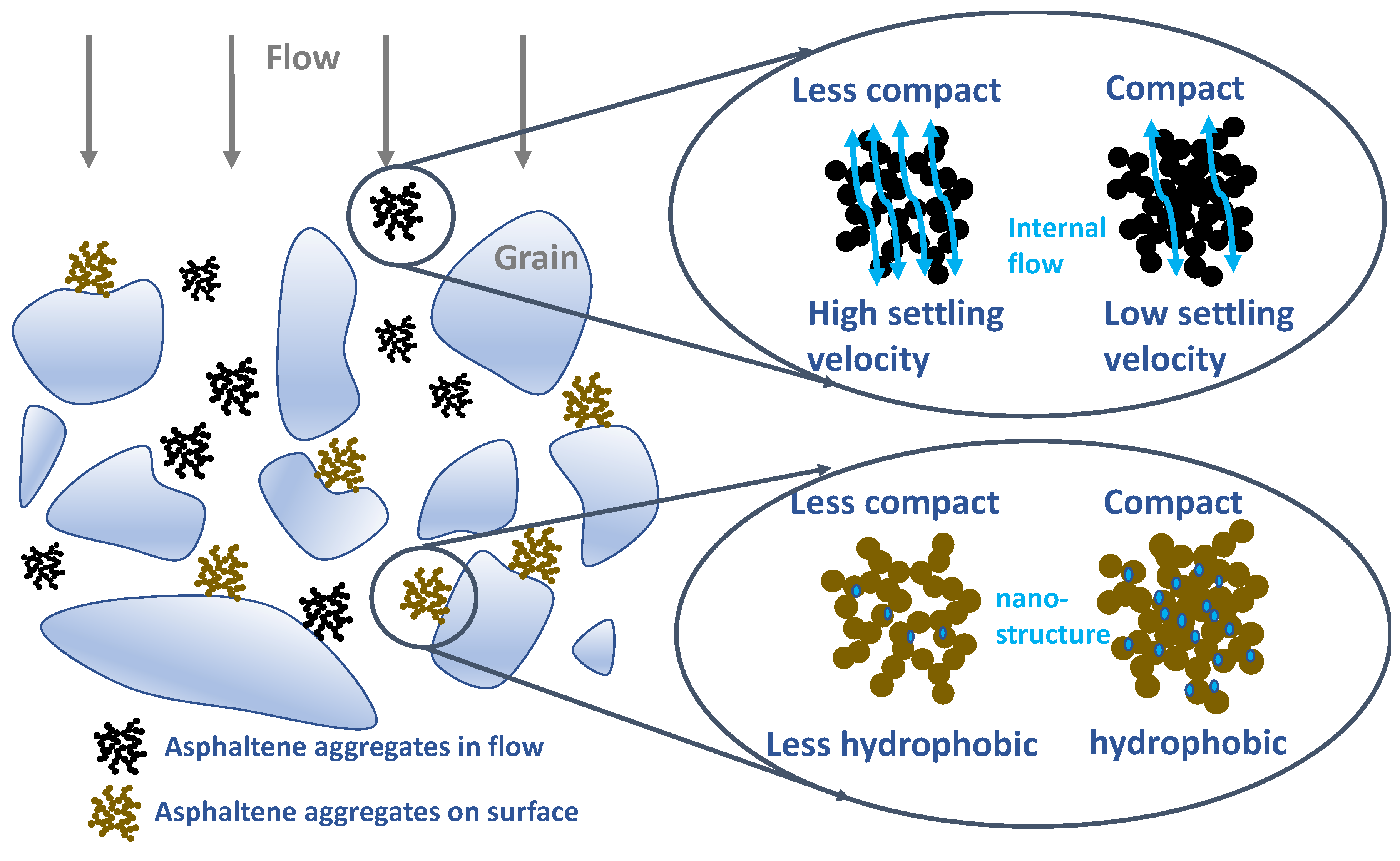
3. Results and Discussion
4. Conclusions and Environmental Implications
Author Contributions
Funding
Conflicts of Interest
References
- Javanbakht, G.; Goual, L. Mobilization and Micellar Solubilization of Napl Contaminants in Aquifer Rocks. J. Contam. Hydrol. 2016, 185–186, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Javanbakht, G.; Goual, L. Impact of Surfactant Structure on NAPL Mobilization and Solubilization in Porous Media. Ind. Eng. Chem. Res. 2016, 55, 11736–11746. [Google Scholar] [CrossRef]
- Lowry, E.; Sedghi, M.; Goual, L. Molecular Simulations of Napl Removal from Mineral Surfaces Using Microemulsions and Surfactants. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 485–494. [Google Scholar] [CrossRef]
- Whittaker, M.; Pollard, S.J.T.; Fallick, T.E. Characterisation of Refractory Wastes at Heavy Oil-Contaminated Sites: A Review of Conventional and Novel Analytical Methods. Environ. Technol. 1995, 16, 1009–1033. [Google Scholar] [CrossRef]
- Ghosh, J.; Tick, G.R. A Pore Scale Investigation of Crude Oil Distribution and Removal from Homogeneous Porous Media During Surfactant-Induced Remediation. J. Contam. Hydrol. 2013, 155, 20–30. [Google Scholar] [CrossRef]
- EPA SW-846. Test Methods for Evaluating Solid Waste, Physical/Chemical Methods. Final Update, 3rd ed.; U.S. Environmental Protection Agency: Washington, DC, USA, 1996.
- Eskin, D.; Mohammadzadeh, O.; Akbarzadeh, K.; Taylor, S.D.; Ratulowski, J. Reservoir Impairment by Asphaltenes: A Critical Review. Can. J. Chem. Eng. 2016, 94, 1202–1217. [Google Scholar] [CrossRef]
- Hung, J.; Castillo, J.; Reyes, A. Kinetics of Asphaltene Aggregation in Toluene-Heptane Mixtures Studied by Confocal Microscopy. Energy Fuel 2005, 19, 898–904. [Google Scholar] [CrossRef]
- Zheng, J.; Behrens, S.H.; Borkovec, M.; Powers, S.E. Predicting the Wettability of Quartz Surfaces Exposed to Dense Nonaqueous Phase Liquids. Environ. Sci. Technol. 2001, 35, 2207–2213. [Google Scholar] [CrossRef]
- Zheng, J.; Shao, J.; Powers, S.E. Asphaltenes from Coal Tar and Creosote: Their Role in Reversing the Wettability of Aquifer Systems. J. Colloid Interface Sci. 2001, 244, 365–371. [Google Scholar] [CrossRef]
- Ghadimi, M.; Amani, M.J.; Ghaedi, M.; Malayeri, M.R. Modeling of Formation Damage Due to Asphaltene Deposition in near Wellbore Region Using a Cylindrical Compositional Simulator. J. Pet. Sci. Eng. 2019, 173, 630–639. [Google Scholar] [CrossRef]
- Cruz, A.A.; Amaral, M.; Santos, D.; Palma, A.; Franceschi, E.; Borges, G.R.; Coutinho, J.A.; Palácio, J.; Dariva, C. CO2 Influence on Asphaltene Precipitation. J. Supercrit. Fluid. 2019, 143, 24–31. [Google Scholar] [CrossRef]
- Dashti, H.; Zanganeh, P.; Kord, S.; Ayatollahi, S.; Amiri, A. Mechanistic Study to Investigate the Effects of Different Gas Injection Scenarios on the Rate of Asphaltene Deposition: An Experimental Approach. Fuel 2020, 262, 116615. [Google Scholar] [CrossRef]
- Duran, J.A.; Casas, Y.A.; Xiang, L.; Zhang, L.; Zeng, H.; Yarranton, H.W. Nature of Asphaltene Aggregates. Energy Fuel 2019, 33, 3694–3710. [Google Scholar] [CrossRef]
- Seifried, C.M.; Crawshaw, J.; Boek, E.S. Kinetics of Asphaltene Aggregation in Crude Oil Studied by Confocal Laser-Scanning Microscopy. Energy Fuel 2013, 27, 1865–1872. [Google Scholar] [CrossRef]
- Rassamdana, H.; Sahimi, M. Asphalt Flocculation and Deposition: Ii. Formation and Growth of Fractal Aggregates. AIChE J. 1996, 42, 3318–3332. [Google Scholar] [CrossRef]
- Hoepfner, M.P.; Fávero, C.V.; Haji-Akbari, N.; Fogler, H.S. The Fractal Aggregation of Asphaltenes. Langmuir 2013, 29, 8799–8808. [Google Scholar] [CrossRef]
- Yudin, I.K.; Nikolaenko, G.L.; Gorodetskii, E.E.; Kosov, V.I.; Melikyan, V.R.; Markhashov, E.L.; Frot, D.; Briolant, Y. Mechanisms of Asphaltene Aggregation in Toluene-Heptane Mixtures. J. Pet. Sci. Eng. 1998, 20, 297–301. [Google Scholar] [CrossRef]
- Ashoori, S.; Abedini, A.; Saboorian, H.; Qorbani Nasheghi, K.; Abedini, R. Mechanisms of Asphaltene Aggregation in Toluene and Heptane Mixtures. J.Jpn. Pet. Inst. 2009, 52, 283–287. [Google Scholar] [CrossRef][Green Version]
- Wu, L.; Ortiz, C.P.; Jerolmack, D.J. Aggregation of Elongated Colloids in Water. Langmuir 2017, 33, 622–629. [Google Scholar] [CrossRef]
- Wu, L.; Ortiz, C.P.; Xu, Y.; Willenbring, J.; Jerolmack, D.J. In Situ Liquid Cell Observations of Asbestos Fiber Diffusion in Water. Environ. Sci. Technol. 2015, 49, 13340–13349. [Google Scholar] [CrossRef]
- Meakin, P. Formation of Fractal Clusters and Networks by Irreversible Diffusion-Limited Aggregation. Phys. Rev. Lett. 1983, 51, 1119. [Google Scholar] [CrossRef]
- Robison Fernlund, J. Image Analysis Method for Determining 3-D Size Distribution of Coarse Aggregates. Bull. Eng. Geol. Environ. 2005, 64, 159–166. [Google Scholar] [CrossRef]
- Sholl, D.S.; Skodje, R.T. Late-Stage Coarsening of Adlayers by Dynamic Cluster Coalescence. Physica A 1996, 231, 631–647. [Google Scholar] [CrossRef]
- Johnson, C.P.; Li, X.; Logan, B.E. Settling Velocities of Fractal Aggregates. Environ. Sci. Technol. 1996, 30, 1911–1918. [Google Scholar] [CrossRef]
- Lord, D.L.; Buckley, J.S. An Afm Study of the Morphological Features That Affect Wetting at Crude Oil-Water-Mica Interfaces. Colloids Surf. A Physicochem. Eng. Asp. 2002, 206, 531–546. [Google Scholar] [CrossRef]
- Li, X.; Du, Y.; Wu, G.; Li, Z.; Li, H.; Sui, H. Solvent Extraction for Heavy Crude Oil Removal from Contaminated Soils. Chemosphere 2012, 88, 245–249. [Google Scholar] [CrossRef] [PubMed]


| Experiments | Cluster | Goodness of Fit | |||||
|---|---|---|---|---|---|---|---|
| Best fit df | R2 | SSE | DFE | Adjusted R2 | RMSE | ||
| 1 | Large | 1.66 ± 0.17 | 0.96 | 5.77 | 203 | 0.96 | 0.17 |
| Small | 1.99 ± 0.01 | 0.99 | 0.009 | 82 | 0.99 | 0.01 | |
| 2 | Large | 1.73 ± 0.17 | 0.97 | 4.88 | 163 | 0.97 | 0.17 |
| Small | 2.00 ± 0.01 | 0.99 | 0.006 | 34 | 0.99 | 0.013 | |
| 3 | Large | 1.83 ± 0.28 | 0.96 | 10.87 | 138 | 0.96 | 0.28 |
| Small | 2.00 ± 0.01 | 0.99 | 0.004 | 28 | 0.99 | 0.012 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hammond, C.B.; Wang, D.; Wu, L. Precipitant Effects on Aggregates Structure of Asphaltene and Their Implications for Groundwater Remediation. Water 2020, 12, 2116. https://doi.org/10.3390/w12082116
Hammond CB, Wang D, Wu L. Precipitant Effects on Aggregates Structure of Asphaltene and Their Implications for Groundwater Remediation. Water. 2020; 12(8):2116. https://doi.org/10.3390/w12082116
Chicago/Turabian StyleHammond, Christian B., Dengjun Wang, and Lei Wu. 2020. "Precipitant Effects on Aggregates Structure of Asphaltene and Their Implications for Groundwater Remediation" Water 12, no. 8: 2116. https://doi.org/10.3390/w12082116
APA StyleHammond, C. B., Wang, D., & Wu, L. (2020). Precipitant Effects on Aggregates Structure of Asphaltene and Their Implications for Groundwater Remediation. Water, 12(8), 2116. https://doi.org/10.3390/w12082116







