Abstract
Freshwater organisms are facing threats from various natural and anthropogenic disturbances. Using data sampled on a nationwide scale from streams in South Korea, we identified the crucial environmental factors influencing the distribution and abundance of freshwater gastropods. We used nonmetric multidimensional scaling and the random forest model to evaluate the relationships between environmental factors and gastropod assemblages. Among the 30 recorded species, two invasive gastropod species (Pomacea canaliculata and Physa acuta) have enlarged their distribution (10.4% and 57.3% frequency of occurrence, respectively), and were found to be widespread in streams and rivers. Our results revealed that the most influential factor in the distribution of gastropod assemblages was the ratio of cobble (%) in the substrate composition, although meteorological and physiographical factors were also important. However, the main environmental factors influencing species distribution varied among species according to habitat preference and environmental tolerance. Additionally, anthropogenic disturbance caused a decrease in the distribution of endemic species and an increase in the spatial distribution of invasive species. Finally, the results of the present study provide baseline information for planning successful strategies to maintain and conserve gastropod diversity when facing anthropogenic disturbance, as well as understanding the factors associated with the establishment of invasive species.
1. Introduction
Freshwater gastropods comprise 5% of the global gastropod fauna but are facing a disproportionately high degree of threat according to the 2019 IUCN Red List of Threatened Species (http://www.redlist.org, Cambridge, UK) [1]. Furthermore, 74% of gastropod species have been classified as vulnerable, endangered, threatened, or already extinct in the United States and Canada [2]. Similarly, in mollusks, 40% of freshwater bivalve species in the world are near threatened, threatened, or extinct [3]. In South Korea, two rare species (Clithon retropictus and Koreanomelania nodifila) are listed in the Red Book as endangered, and five species (K. nodifila, Koreoleptoxis globus ovalis, Semisulcospira coreana, Semisulcospira forticosta, and Semisulcospira tegulata) are endemic in the database of the National Aquatic Ecological Monitoring Program (National Institute of Environmental Research, Incheon, South Korea) [4]. These threats are of particular global concern because most freshwater gastropods are endemic with small geographic ranges [2,5,6,7].
The distribution and structure of gastropod assemblages are influenced by various environmental factors [8,9]. Across a large-scale area that includes several basins, climate-related factors, such as temperature and precipitation as well as physiographical factors, are important in influencing the structure of freshwater gastropod assemblages [10]. Other environmental factors, such as hydrological, physicochemical, and biological factors, are important within the same climate region. Differences in vegetation, land use, and flood disturbance are important in explaining variation in species composition among streams [11,12]. In addition, within-stream and/or -microhabitat differences (e.g., differences between flow regimes, substrate composition, and riparian vegetation) contribute to variations among gastropod assemblages [13,14,15].
Gastropods are sessile and have a very limited ability to avoid unfavorable environments, making it difficult to recover the heterogeneity of a freshwater ecosystem once it has been disrupted [10,16]. Therefore, research on the multiscale regulation of gastropod assemblage structures in freshwater ecosystems is the first step in planning a successful strategy for either conserving or restoring freshwater diversity. However, most studies on species distribution and assemblage structure have had a small-scale focus or have been mainly conducted in lentic habitats [17,18], or considered only a limited number of environmental factors [19,20,21].
Therefore, the present study aimed to identify environmental factors influencing gastropod distribution patterns, focusing on lotic habitats on a nationwide scale (South Korea). Specifically, we tested two hypotheses. First, large-scale factors, including temperature-related and physiographical factors, are more influential than other factors in our research area to determine gastropod assemblage structure. Second, major influential environmental factors differ depending on species. Finally, we considered strategies for the conservation and management of freshwater gastropods based on our results.
2. Materials and Methods
2.1. Ecological Data
Gastropod data were obtained from the database of the NAEMP (http://water.nier.go.kr/). Since 2008, NAEMP has conducted nationwide surveys of the freshwater organisms of South Korea, including periphyton, benthic macroinvertebrates, and fish, together with associated environmental factors, twice per year (i.e., spring and autumn, when natural disturbances such as heavy rain and drought are at a minimum). The river systems in South Korea form five major river basins (i.e., the Han, Nakdong, Yeongsan, Seomjin, and Geum River basins). Among these, the Han River basin (basin area: 41,957 km2) in the north of South Korea is the largest, occupying one-third of the country. The Nakdong River basin (31,785 km2) is in the southeast, and the Geum River basin (17,537 km2) is in the midwest. Lastly, the Yeongsan River (3,467 km2) and Seomjin River (4,912 km2) basins are located close to each other in the southwest of South Korea [4,22,23,24].
Gastropods were collected at 714 sampling sites from all the South Korean river basins twice per year from 2008 to 2013, using a Surber net (30 × 30 cm, 1 mm mesh size, Table 1). Three replicates were collected from each sampling site at each sampling time and then were transformed into abundance/m2 for the further analyses, based on NAEMP guidelines [25]. Detailed information regarding the sampling protocol is given in previous studies [26,27,28]. The samples were preserved in 95% ethanol in the field, and then placed in 70% ethanol in the laboratory. They were sorted and identified, mostly to species level, and the number of individuals per species was counted using naked-eye or microscope examination [4].

Table 1.
Average values and standard deviations (SD) of the environmental variables characterizing the sampling sites.
2.2. Data Analyses
We analyzed the data in three steps. First, we classified the sampling sites by conducting a hierarchical cluster analysis (CA) based on gastropod abundance. The CA was calculated based on the Bray–Curtis dissimilarity with Ward’s linkage method [29], using the ‘vegan’ package [30] in R [31]. Then, multiresponse permutation procedures (MRPP) was considered to evaluate the significant differences among the clusters defined through CA. We defined the indicator species in each cluster using an indicator species analysis [32]. The indicator species was selected based on the indicator value (IndVal), by considering relative species abundance and its associated relative frequency of occurrence within the defined clusters. The IndVal range was from 0 to 100 (all individuals of a species are included only within a single cluster). Species with a statistically significant (p < 0.05) IndVal higher than 25% were selected as indicator species [32]. A site randomization procedure that reallocates samples among sample groups (9999 permutations) was used to test for significance. Indicator species analysis was performed using the ‘indval’ function in the ‘labdsv’ package [33] in R [31].
Second, to describe the gastropod assemblage patterns, we applied nonmetric multidimensional scaling (NMDS) based on the Bray–Curtis dissimilarity between sampling sites, using the ‘vegan’ package [34] in R [31]. In order to identify the best NMDS solution (i.e., the lowest STRESS value), the ‘metaMDS’ function was applied. Then, we used the ‘envfit’ function to evaluate the relationships between gastropod assemblages and environmental factors [34,35]. All the analyses related to NMDS were conducted using the ‘vegan’ package in R [31].
Lastly, a random forest (RF) model was used to predict the distribution of gastropod species based on gastropod abundance and environmental variables, and to evaluate the contribution of each environmental variable to species distribution. The RF model, a machine learning model, is computed using a combination of a large set of decision trees [4,36], and does not require assumptions, such as linear or nonlinear relationships between predictors (environmental variables in the present study) and response factors (gastropod species) [27,37,38]. We used the ‘randomForest’ package [39] in R [31], with the three default training parameters: ntree (number of trees = 500), mtry (number of variables = 3), and node size (5). The importance of environmental factors to gastropod distribution was computed based on the mean decrease in accuracy, and importance values were then rescaled from 0 to 100 [40,41]. The RF model was applied to dominant gastropod species (here, more than 10% occurrence frequency, Table 2). Then, we used partial dependence analysis [42] to evaluate the relationship between environmental factors and 11 gastropod species presenting more than a 10% frequency of occurrence in all sampling sites.

Table 2.
Gastropod species and their frequency of occurrence (%) in the dataset. Species with a frequency of occurrence of over 10% are indicated in bold.
The abundance of each gastropod species was averaged at each site after pooling yearly data and transformed with natural logarithm to reduce variation in abundance prior to further analyses. In environmental factors, the extremes and outliers were removed before analyzing the data and the values were averaged after pooling yearly data like gastropod abundance.
3. Results
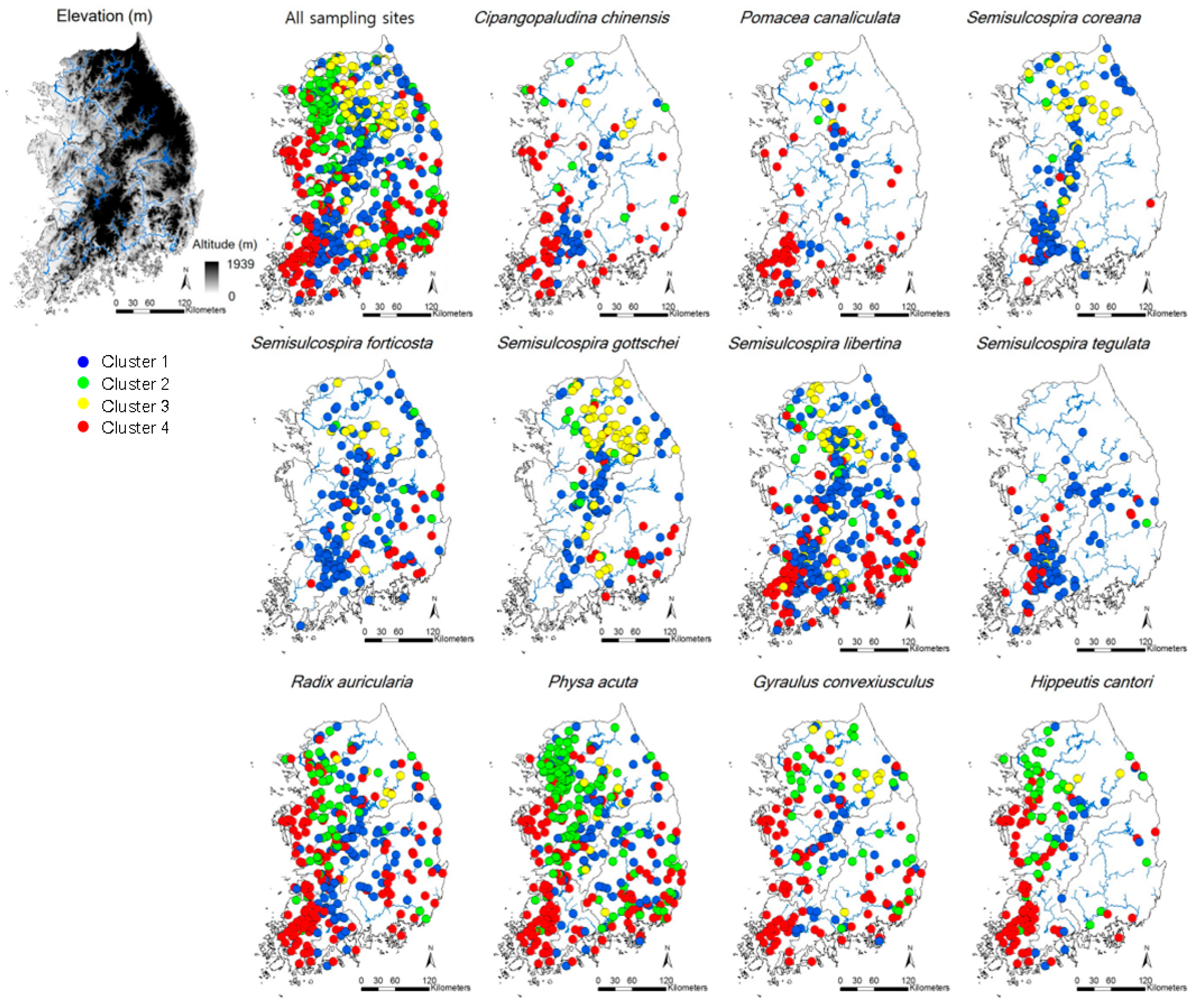
Thirty species, belonging to three orders and 13 families, were recorded in the study area (Table 2). Physa acuta, an invasive species, was the most commonly observed (57.3% occurrence frequency of all the sites), followed by Semisulcospira libertina (49.0%), Radix auricularia (44.4%), and S. forticosta (26.8%, Figure 1 and Table 2). Two species, Clithon retropictus and Koreanomelania nodifila, listed in the Red Book of Korea (National Institute of Biological Resources, Incheon, South Korea) as endangered had low occurrence frequencies (1.5% and 4.9%, respectively), and Pomacea canaliculata, an invasive species, was recorded in 10.4% of the sites.
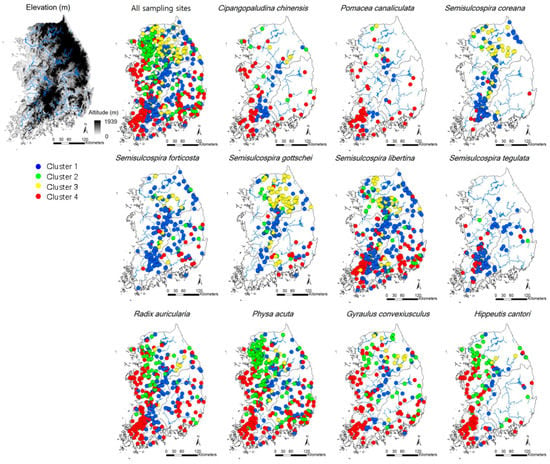
Figure 1.
Sampling sites and occurrence patterns of 11 gastropod species in Korea that present more than a 10% frequency of occurrence. Classification of sampling sites were defined in Figure 2. The clusters reflected the differences of environmental condition at each sampling site.
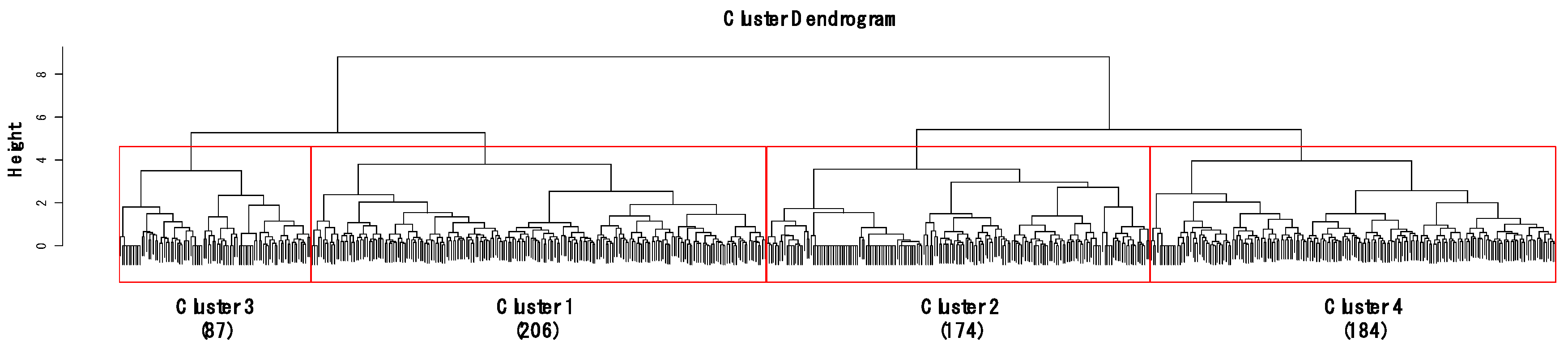
CA classified the sites into four clusters (1–4) based on similarities in gastropod assemblage composition (Figure 2) and four clusters were significantly different based on MRPP (A = 0.18–0.21, p < 0.05). A total of 21 species were selected as indicator species for four clusters based on their IndVal (p < 0.05, Table S1). Cluster 4 contained the highest number of indicator species (10), followed by Cluster 1 (8), Cluster 3 (2), and Cluster 2 (1). In Clusters 2 and 4, the species (i.e., Hippeutis cantori and Austropeplea ollula) mainly found in organic enriched streams and/or invasive species (i.e., P. canaliculata and P. acuta) were selected as an indicator species. On the other hand, in Clusters 1 and 3, the species (i.e., S. gottschei) mostly found in the less disturbed area and endangered and/or endemic species (i.e., K. globus and K. nodifila) were mainly selected.
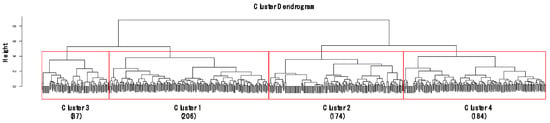
Figure 2.
Dendrogram of cluster analysis based on gastropod assemblages using Ward’s linkage method with the Bray–Curtis dissimilarity. Height on the y-axis indicates distances of merging clusters, reflecting the distance between the samples. Numbers in parenthesis represent the number of sampling sites in each cluster.
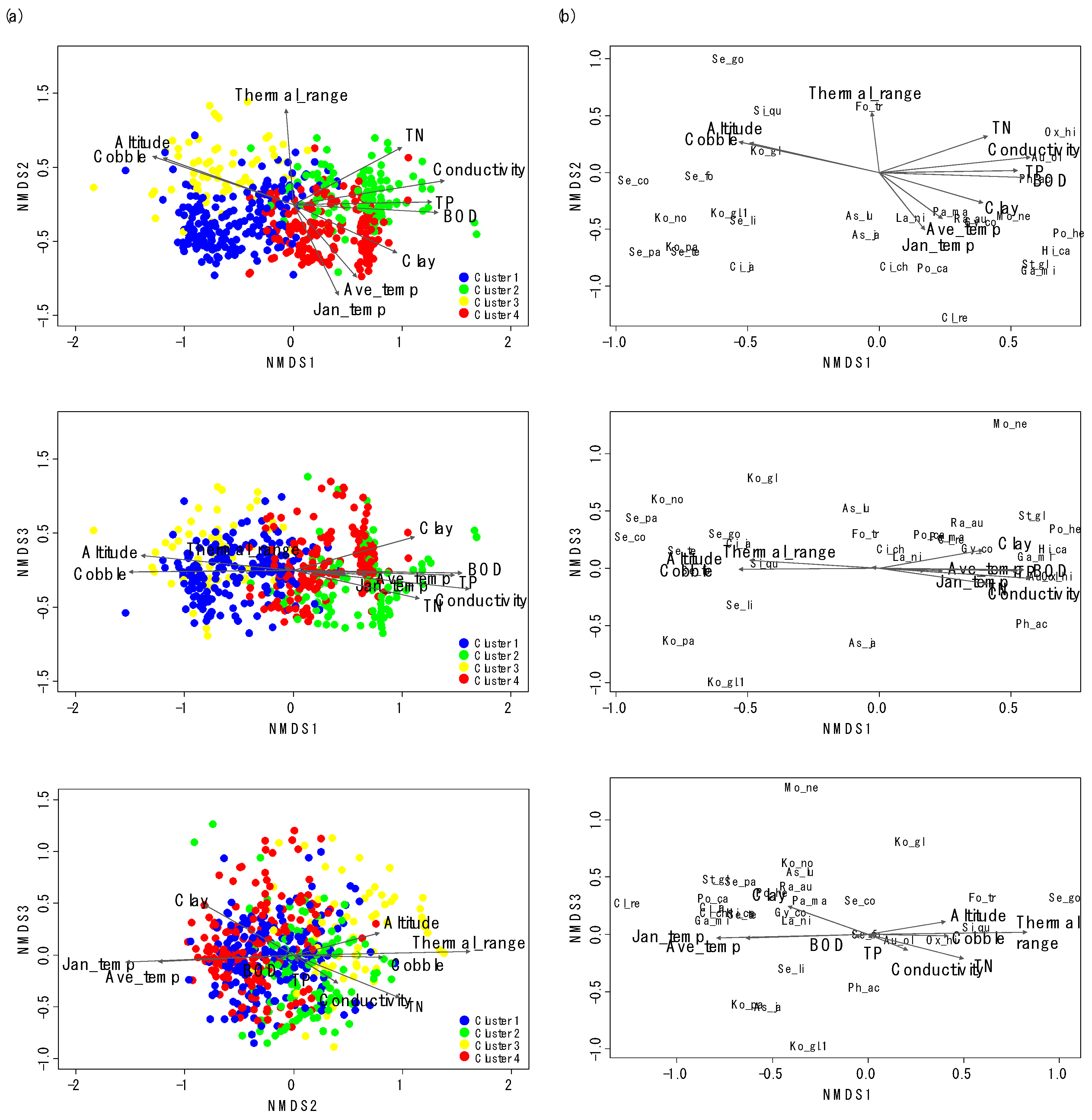
NMDS also showed differences in gastropod assemblage composition (Figure 3), reflecting the classification of sampling sites in CA. After the NMDS ordination with gastropod assemblage, environmental variables were visualized with sampling sites and gastropod taxa with biplot. We selected the first three NMDS axes based on the Kruskal’s stress value (the first three axes stress = 13.8). Clusters 1 and 3 and Clusters 2 and 4 were differentiated on the NMDS 1, whereas Clusters 2 and 3 and Clusters 1 and 4 were divided on the NMDS 2 (Figure 3a). However, the differentiation of clusters was not clear on NMDS 3. NMDS 1 reflected the gradient of water quality, whereas NMDS2 presented the gradient of temperature. On the ordination with NMDS 1 and NMDS 2, sampling sites with high values of cobble (%) and altitude were located on the left side of NMDS 1, whereas sites with high values of TN, conductivity, BOD, and TP were located on the right side of NMDS 1 (Figure 3a). Sampling sites with high values of average temperature in January were located on the lower part of NMDS 2, whereas sampling sites with high values of TP and conductivity were located on the lower part of NMDS 3. Species which prefer riffle areas with a large-sized substrate and good water quality, such as Koreoleptoxis globus ovalis, K. nodifila, S. coreana, and S. forticosta, were on the left section of NMDS 1 (Figure 3b). Conversely, the high values of TN, conductivity, BOD, and TP strongly influenced the distributions of R. auricularia, P. acuta, Gyraulus convexiusculus, and H. cantori. The average temperature in January was influential to the distributions of P. canaliculata and C. retropictus.
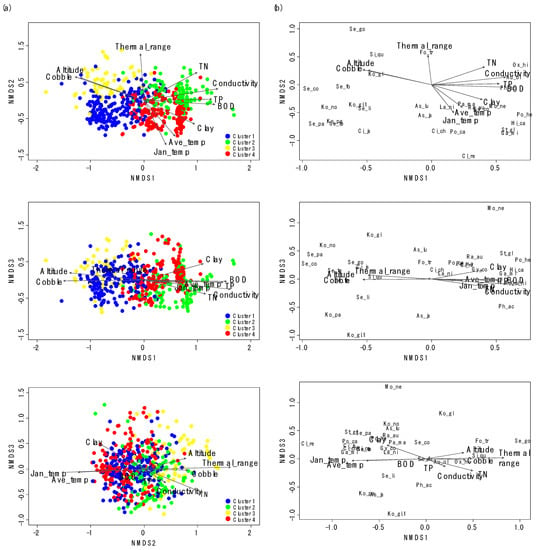
Figure 3.
Nonmetric multidimensional scaling (NMDS) ordination of sampling sites (a) with gastropod assemblages (b). The first three axes (Kruskal’s stress = 13.8) were used to visualize the ordination. Arrows correspond to environmental variables significantly related to assemblage composition (only environmental variables with R2 > 0.2 are presented). Arrow length is proportional to the correlation magnitude (r). The abbreviations for the environmental variables and species names are explained in Table 1 and Table 2, respectively.
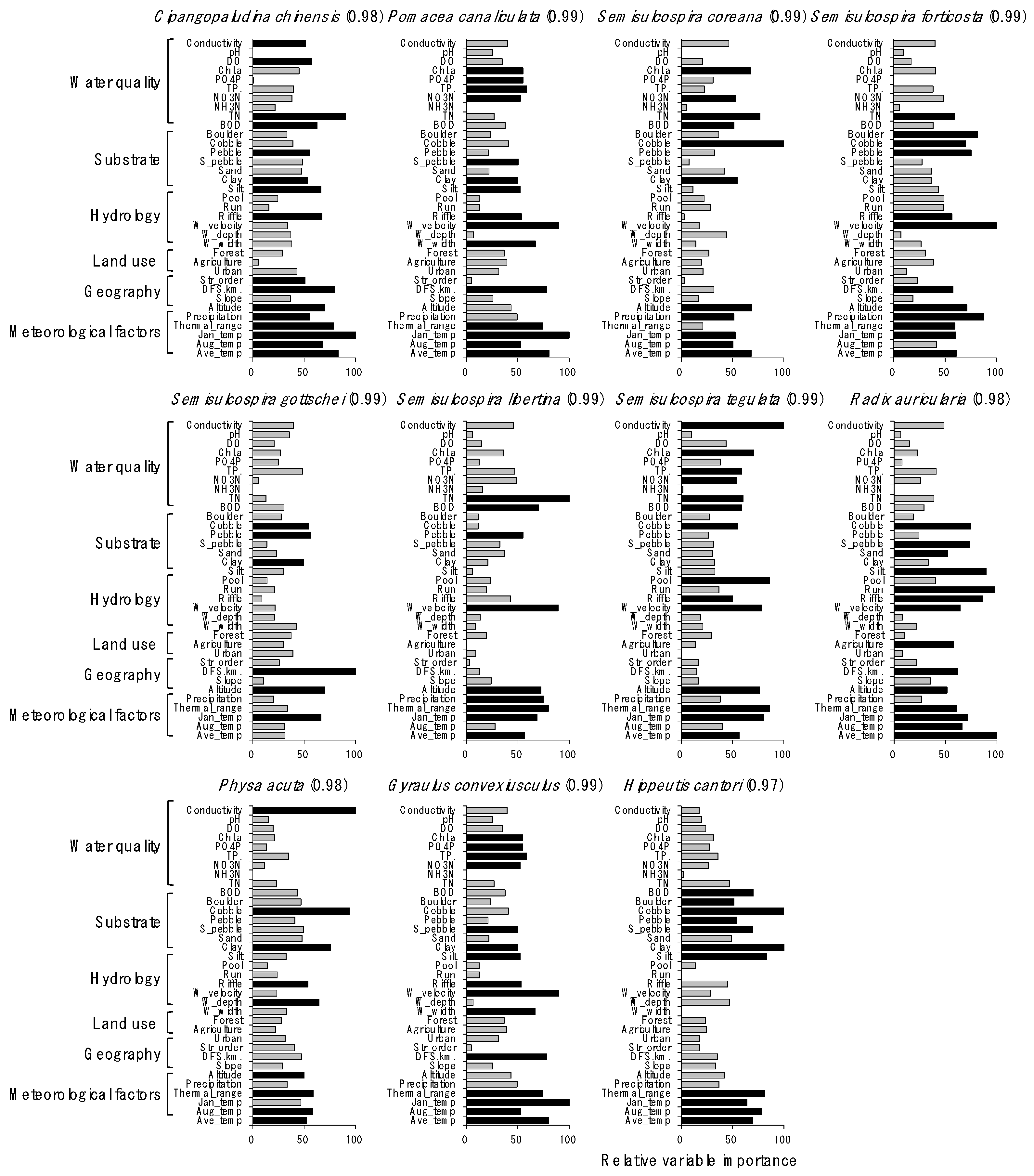
Distributions of species were well predicted by the RF models, with a high prediction power ranging from 0.97 to 0.99 (Figure 4). Overall, meteorological and physiographical variables were included in the main factors influencing the distribution of gastropod species. However, the contribution of other environmental factors for predicting species distribution differed depending on species. For example, conductivity was the most important factor (100) for predicting the occurrence of P. acuta, followed by cobble (94), and water depth (65). TN (100), silt (89), and riffle (86) were important for predicting S. libertine abundance. Average temperature in January (100) was the most important factor for P. canaliculata, an invasive species in South Korea, followed by water velocity (90), annual average temperature (80), and distance from the source (78). The ratio (%) of cobble (100) and TN (77) were influential in determining the occurrence of S. coreana.
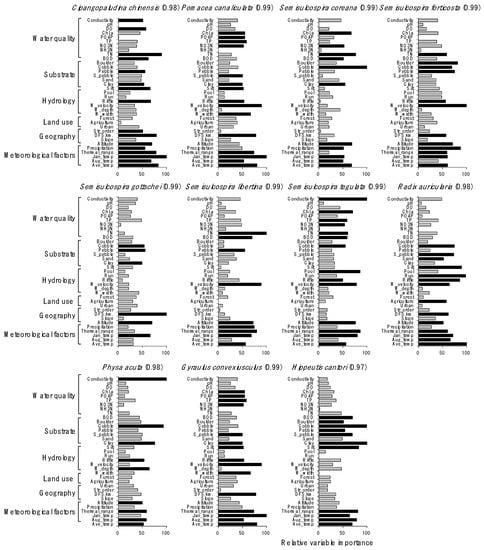
Figure 4.
Relative importance of environmental variables for predicting the distribution of gastropod species in the random forest model. Numbers in parentheses indicate the predictability of the model for each species. black bar: ≥50 of the relative importance value and gray bar: <50 of the relative importance value.
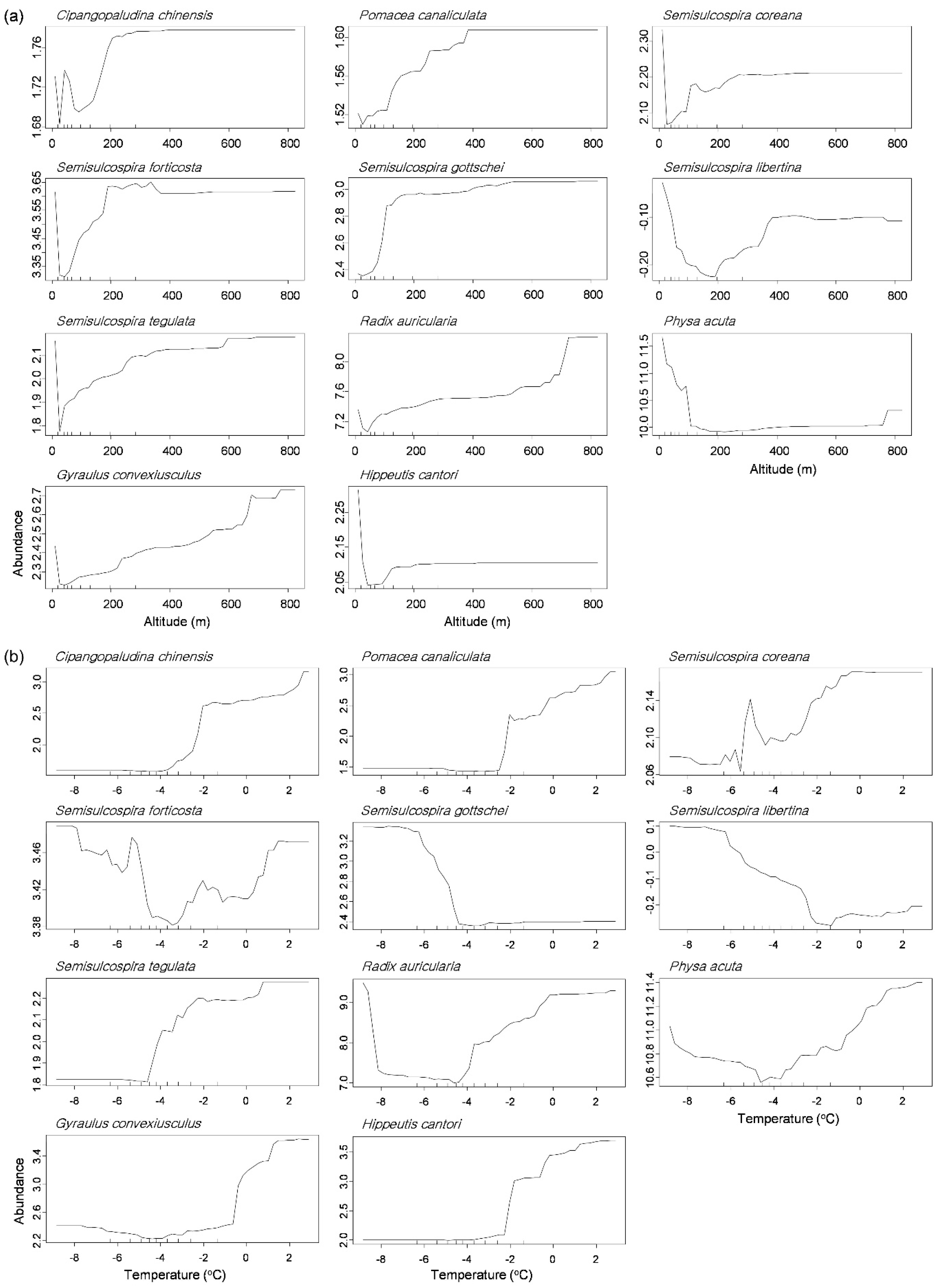
The partial dependence plot displayed that each gastropod species responded differently to the environmental factors in the RF models (Figure 5 and Figure S1). We visualized two variables showing a strong gradient in the NMDS plot: altitude on NMDS 1 and the average temperature in January on NMDS 2 (Figure 3). Most of genus Semisulcospira, C. chinensis, and P. canaliculata displayed increase of their abundance as increase of altitude, whereas P. acuta was abundant mostly at low altitude (≤160 m, Figure 5a). The high abundance of P. acuta was also related to high concentrations of TN (≥1.8 mg/L, Figure S1). Meanwhile, responding to temperature, the abundance of P. canaliculata increased with an increasing average temperature in January above −2.0 °C, whereas abundances of S. gottschei and S. libertina were high at a lower temperature in January (−4.2 °C and −2.2 °C, respectively, Figure 5b).
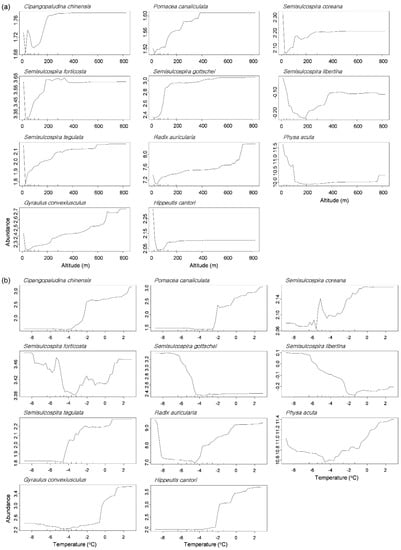
Figure 5.
Partial dependence plots of 11 abundant species responding to altitude (a) and average temperature in January (b) in the random forest model.
4. Discussion
4.1. Environmental Factors Influencing Gastropod Assemblages
The structure and distribution of the gastropod assemblage were differentiated by various environmental factors in our study. Meteorological (e.g., temperature-related factors), physiographical (e.g., altitude), substrate composition (e.g., the ratio of cobble) and water quality (e.g., conductivity, TN, TP, and BOD) gradients were important in structuring gastropod assemblages. Among them, the ratio of cobble (%) was the most influential factor in gastropod assemblage. Substrate composition is closely related to habitat complexity and resource availability (e.g., the number of algae, aquatic macrophytes, and microorganisms), especially for gastropods [43]. For instance, the distribution of Semisulcospira which mainly feed on periphytic algae attached to large-sized substrate materials, such as boulders and cobbles, was highly related to the ratio of cobble (%) [44]. In addition to substrate composition, gastropod assemblages showed differentiation from the least polluted to anthropogenically disturbed streams (i.e., the gradient of water quality). Species such as R. auricularia, P. acuta, G. convexiusculus, and H. cantori occurred frequently at sites with high values of conductivity, BOD, TN, and TP. Because these species can assimilate atmospheric air through their vascularized mantle cavity, they can thrive under harsh conditions [45], for example, in areas with silt substrates and high conductivity [46]. Physidae, in particular, have a high tolerance to organic pollution [47]; the occurrence of the genus Physa is associated with relatively higher values of pH, calcium hardness, total hardness, total alkalinity, conductivity, and total dissolved solids than other species [48,49].
4.2. Conservation of Gastropoda Species
In South Korea, the number of species in Pleuroceridae (10 species) was the highest among gastropod taxa, especially in the genus Semisulcospira (6 species). The distribution of Semisulcospira was related to cobble percentage in substrate composition. This might be linked to food resources and habitat characteristics, as Semisulcospira mainly is a scraper feeding on periphytic algae attached to large-sized substrates like boulders and cobbles without dense canopy cover in riparian areas (Karube et al., 2012). Semisulcospira is a key food resource for many freshwater organisms, especially for species with high conservation priority in South Korea, such as Spotted barbel (Hemibarbus mylodon; fish, Korean National Monument No. 259 and endemic species) as well as firefly (Luciola lateralis and Lychnuris rufa, insect). Especially, the main population of the latter two species (i.e., L. lateralis and L. rufa) inhabit only within a confined area in Muju-gun (Jeollabuk-do, South, Korea) with their food sources (i.e., Semisulcospira). Therefore, this area is designated as the Korean National Monument (No 332) to conserve their population as well as Semisulcospira population. However, in spite of their important roles including the critical food sources in the freshwater ecosystem, Semisulcospira has been excessively collected by human beings for domestic consumption (i.e., snail soup) because Semisulcospira is well known to people about its the high-protein food sources. In addition, collecting Semisulcospira in streams is one of the general and popular family leisure activities in Korea, especially in the summer season. However, because people cannot distinguish the genus Semisulcospira into species level and just collect them without considering their ecological importance, resulting in the dramatic reduction of its abundance and diversity.
Currently, the habitable area for most species, including endangered species, is being consistently lost [50]. Endangered and/or endemic species distributed in only one basin should, therefore, be selected as the first priority for management and conservation [51]. Fortunately, in our study, no endemic and/or endangered species were found to be inhabiting a single watershed, with limited distribution. However, anthropogenic disturbances could threaten gastropod species, especially those that are endemic and/or endangered, or which have a limited distribution. For instance, we found that the occurrence frequency of endemic and/or endangered species was less than 5%. Among three species recorded as endemic and/or endangered, C. nodifila attracts a particularly high conservation concern. This is because this species currently has a limited and narrow distribution, being found only in some of the southern parts of South Korea (1.5% frequency of occurrence), such as the Yeongsan and Seumjin River basins, which are in the least disturbed area with a shallow water depth and low current velocity. C. retropictus is distributed mainly within tropical and subtropical and some temperate regions [52]; the distribution of these taxa is, therefore, also sensitive to low temperature and large thermal ranges. Moreover, bank and dam construction could destroy the habitable environment of C. retropictus in various ways. This species is designated within the second grade of endangered wild fauna and flora species by the Ministry of Environment in the Republic of Korea, and its populations require active conservation and management.
K. nodifila is another endangered species that is endemic to South Korea. In our study, it was found mainly to be restricted (4.9% frequency of occurrence) to the least disturbed upstream areas in the northern part of South Korea (i.e., in the Imjin, Hantan, and Dong Rivers). The distribution of K. nodifila is strongly influenced by small variations in stream habitat conditions. For instance, this species prefers natural habitats that have a high water velocity, riffle (%), and the ratio of cobble (%), as well as good water quality. However, its original habitat has suffered continuous disruption and habitat loss, due to the construction and reorganization of flood control dams and weirs. These constructions alter the flow regime from lotic to lentic, a factor that is influential in the occurrence of this species. Furthermore, K. nodifila may be gathered with Semisulcospira species when the latter is collected during the snail hand-picking season for use in cooking snail soup and/or leisure activities in Korea. This is because it is difficult for the general public to distinguish Semisulcospira species from K. nodifila. In this sense, ongoing public relations and education need to cover the importance and key characteristics of K. nodifila to protect the species from excessive collection.
4.3. Management of Invasive Species
In this study, the distribution of P. canaliculata (10.4% frequency of occurrence) was determined by two critical factors: average temperature in January and percentage of agricultural area (Figure 4). P. canaliculata is native to tropical areas of South America and is one of the world’s 100 worst invasive species [53]. In South Korea, this species has been introduced as a food resource, and as a herbicide substitute to control weeds in environmentally friendly paddy fields. In fact, the increased usage of this snail is mostly due to its high efficiency in controlling weeds (99%) [54]. P. canaliculata was introduced to South Korea on the assumption that it would be unable to overwinter there because of the low winter temperatures [55]. Currently, however, 30 years after its first introduction to South Korea, there are frequent reports of P. canaliculata overwintering in open freshwater ecosystems, especially in the southern part of South Korea [56]. In addition, the geographic range over which P. canaliculata can overwinter is continuously increasing due to global warming and the biological adaptation of this invasive species, resulting in severe impacts on aquatic ecosystems [57]; based on our database, it is already found in all the South Korean river basins. The invasion of Thailand’s natural wetlands by P. canaliculata is causing aquatic plants to disappear from riparian systems, resulting in high nutrient concentrations and phytoplankton biomass [58]. Therefore, there has been a complete shift in the state of the ecosystem and functions in areas where P. canaliculata has become established. Ongoing and systematic management of this species is, therefore, essential if it is to be eradicated and to prevent its further expansion and establishment in the aquatic ecosystems of South Korea.
In addition, there are many studies reporting that land use disturbance alters landscapes, ecosystem structure, and functions [40]. The continuous increase in agricultural areas and urbanization is leading to an increase in the introduction of invasive species and homogenization of gastropod assemblages [59]. In our study, species indicating greater tolerance of organic pollution and urbanization possessed the highest frequencies of occurrence. In particular, P. acuta had a frequency of occurrence of 57.3%, the highest of all species in our study; similarly, P. canaliculata, which has a high tolerance to organic enrichment, had a frequency of occurrence of 10.4%. Therefore, habitat degradation can induce changes in the structure and distribution of gastropod assemblages and cause the distribution of both tolerant and invasive species to expand.
5. Conclusions
Our evaluation of the two questions examined in this study revealed the following results. (1) The ratio of cobble in the substrate composition was the most influential factor in gastropod assemblage distribution on a national scale. (2) Nonetheless, the major environmental factors influencing the distribution of each species varied according to habitat preference and environmental tolerance. This study is the first to quantify the distribution ranges of all gastropod species on the national scale, and to evaluate the influential factors determining that distribution based on a modeling approach. Even though no endemic and/or endangered species were found inhabiting only a single basin, they nonetheless have a limited distribution (less than 5%). Furthermore, invasive gastropod species have extended their distribution (P. canaliculata, 10.4%; and P. acuta, 57.3%) and are easily found within all the river basins. The habitats occupied by gastropods have been continuously disrupted by various factors, resulting in reductions to available suitable habitat. Local pollution, hydrologic alteration, agriculture, global warming, and the introduction of invasive species have had severe impacts on aquatic ecosystems. Detailed information on gastropod assemblages and the factors influencing their assemblage structure and distribution is, therefore, required for the successful conservation of aquatic gastropods. In particular, the distribution and abundance of invasive and endangered species should be evaluated to prevent ecosystem disruption and enhance species conservation strategies. Finally, the results of the present study would contribute to the development of adequate and systematic management policies for the conservation and management of freshwater gastropods.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4441/12/7/1908/s1, Table S1: Indicator species selected in four clusters based on the Indval. Only taxa with significant values are shown. Figure S1: Partial dependence plots of 11 abundant species responding to (a) total nitrogen, (b) agriculture (%) in land cover, (c) water current velocity and (d) cobble (%).
Author Contributions
Conceptualization, M.-J.B. and Y.-S.P.; formal analysis, M.-J.B.; investigation, M.-J.B. and Y.-S.P.; writing—original draft preparation, M.-J.B.; writing—review and editing, Y.-S.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Nakdonggang National Institute of Biological Resources (NNIBR), funded by the Ministry of Environment (MOE) of the Republic of Korea (grant number NNIBR201901111), and by the National Research Foundation of Korea (NRF) funded by the Korean government (MSIP, grant numbers NRF-2016R1A2B4011801 and NRF-2019R1A2C1087099).
Acknowledgments
We would like to thank the National Aquatic Ecological Monitoring Program (NAEMP) operated by the Ministry of Environment and National Institute of Environmental Research, Korea, for providing part of the dataset used.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Strong, E.E.; Gargominy, O.; Ponder, W.F.; Bouchet, P. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. In Freshwater Animal Diversity Assessment. Developments in Hydrobiology; Balian, E.V., Lévêque, C., Segers, H., Martens, K., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 198, pp. 149–166. [Google Scholar]
- Johnson, P.D.; Bogan, A.E.; Brown, K.M.; Burkhead, N.M.; Cordeiro, J.R.; Garner, J.T.; Hartfield, P.D.; Lepitzki, D.A.; Mackie, G.L.; Pip, E.; et al. Conservation status of freshwater gastropods of Canada and the United States. Fisheries 2013, 38, 247–282. [Google Scholar] [CrossRef]
- Lopes-Lima, M.; Burlakova, L.E.; Karatayev, A.Y.; Mehler, K.; Seddon, M.; Sousa, R. Conservation of freshwater bivalves at the global scale: Diversity, threats and research needs. Hydrobiologia 2018, 810, 1–14. [Google Scholar] [CrossRef]
- Bae, M.-J.; Kwon, Y.; Hwang, S.-J.; Chon, T.-S.; Yang, H.-J.; Kwak, I.-S.; Park, J.-H.; Ham, S.-A.; Park, Y.-S. Relationships between three major stream assemblages and their environmental factors in multiple spatial scales. Ann. Limnol. Int. J. Lim. 2011, 47, S91–S105. [Google Scholar] [CrossRef]
- Williams, J.D.; Warren, M.L., Jr.; Cummings, K.S.; Harris, J.L.; Neves, R.J. Conservation status of freshwater mussels of the United States and Canada. Fisheries 1993, 18, 6–22. [Google Scholar] [CrossRef]
- Abramovitzm, J.N. Imperiled Waters Impoverished Future the Decline of Freshwater Ecosystems; Worldwatch Institute: Washington, DC, USA, 1996; Volume Paper No. 128. [Google Scholar]
- Clements, R.; Koh, L.P.; Lee, T.M.; Meier, R.; Li, D. Importance of reservoirs for the conservation of freshwater molluscs in a tropical urban landscape. Biol. Conserv. 2006, 128, 136–146. [Google Scholar] [CrossRef]
- Oehlmann, J.; Schulte-Oehlmann, U. Molluscs as bioindicators. In Trace Metals and Other Contaminants in the Environment; Markert, B.A., Breure, A.M., Zechmeister, H.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; pp. 577–635. [Google Scholar]
- Waykar, B.; Deshmukh, G. Evaluation of bivalves as bioindicators of metal pollution in freshwater. Bull. Environ. Contam. Toxicol. 2012, 88, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Heino, J.; Muotka, T.; Paavola, R.; Hämäläinen, H.; Koskenniemi, E. Correspondence between regional delineations and spatial patterns in macroinvertebrate assemblages of boreal headwater streams. J. N. Am. Benthol. Soc. 2002, 21, 397–413. [Google Scholar] [CrossRef]
- Richards, C.; Haro, R.; Johnson, L.; Host, G. Catchment and reach-scale properties as indicators of macroinvertebrate species traits. Freshw. Biol. 1997, 37, 219–230. [Google Scholar] [CrossRef]
- Townsend, C.R.; Doledec, S.; Scarsbrooks, M.R. Species traits in relation to temporal and spatial heterogeneity in streams: A test of habitat templet theory. Freshw. Biol. 1997, 37, 367–387. [Google Scholar] [CrossRef]
- Barmuta, L.A. Interaction between the effects of substratum, velocity and location on stream benthos: An experiment. Aust. J. Mar. Freshw. Res. 1990, 41, 557–573. [Google Scholar] [CrossRef]
- Downes, B.J.; Lake, P.S.; Schreiber, E.S.G. Habitat structure and invertebrate assemblages on stream stones: A multivariate view from the riffle. Austral Ecol. 1995, 20, 502–514. [Google Scholar] [CrossRef]
- Wohl, D.L.; Wallace, J.B.; Meyer, J.L. Benthic macroinvertebrate community structure, function and production with respect to habitat type, reach and drainage basin in the southern Appalachians. Freshw. Biol. 1995, 34, 447–464. [Google Scholar] [CrossRef]
- Poff, N.L. Landscape filters and species traits: Towards mechanistic understanding and prediction in stream ecology. J. N. Am. Benthol. Soc. 1997, 16, 19. [Google Scholar] [CrossRef]
- Strzelec, M.; Spyra, A.; Krodkiewska, M.; Serafinski, W. The long-term transformations of Gastropod communities in dam-reservoirs of Upper Silesia (Southern Poland). Malacol. Bohemoslov. 2005, 4, 41–47. [Google Scholar]
- Lewin, I.; Smoliński, A. Rare, threatened and alien species in the gastropod communities in the clay pit ponds in relation to the environmental factors (The Ciechanowska Upland, Central Poland). Biodivers. Conserv. 2006, 15, 3617–3635. [Google Scholar] [CrossRef]
- Greenwood, K.S.; Thorp, J.H. Aspects of ecology and conservation of sympatric, prosobranch snails in a large river. Hydrobiologia 2001, 455, 229–236. [Google Scholar] [CrossRef]
- Antoine, C.; Castella, E.; Castella-Müller, J.; Lachavanne, J.B. Habitat requirements of freshwater gastropod assemblages in a lake fringe wetland (Lake Neuchâtel, Switzerland). Arch. Für Hydrobiol. 2004, 15, 377–394. [Google Scholar] [CrossRef]
- Çabuk, Y.; Arslan, N.; Yılmaz, V. Species composition and seasonal variations of the Gastropoda in Upper Sakarya River System (Turkey) in relation to water quality. Clean–Soilairwater 2004, 32, 393–400. [Google Scholar]
- Li, F.; Chung, N.; Bae, M.-J.; Kwon, Y.-S.; Park, Y.-S. Relationships between stream macroinvertebrates and environmental variables at multiple spatial scales. Freshw. Biol. 2012, 57, 2107–2124. [Google Scholar] [CrossRef]
- Li, F.; Chung, N.; Bae, M.-J.; Kwon, Y.-S.; Kwon, T.-S.; Park, Y.-S. Temperature change and macroinvertebrate biodiversity: Assessments of organism vulnerability and potential distributions. Clim. Chang. 2013, 119, 421–434. [Google Scholar] [CrossRef]
- Kang, H.; Bae, M.-J.; Lee, D.-S.; Hwang, S.-J.; Moon, J.-S.; Park, Y.-S. Distribution patterns of the freshwater oligochaete Limnodrilus hoffmeisteri influenced by environmental factors in streams on a Korean nationwide scale. Water 2017, 9, 921. [Google Scholar] [CrossRef]
- Ministry of Environment/National Institute of Environmental Research. The Survey and Evaluation of Aquatic Ecosystem Health in Korea; The Ministry of Environment/National Institute of Environmental Research: Incheon, Korea, 2008; (In Korean with English Summary).
- Kwak, I.-S.; Lee, D.-S.; Hong, C.; Park, Y.-S. Distribution patterns of benthic macroinvertebrates in streams of Korea. Korean J. Ecol. Environ. 2018, 51, 60–70. [Google Scholar] [CrossRef]
- Lee, D.Y.; Lee, D.S.; Bae, M.J.; Hwang, S.J.; Noh, S.Y.; Moon, J.S.; Park, Y.S. Distribution patterns of odonate assemblages in relation to environmental variables in streams of South Korea. Insects 2018, 9, 152. [Google Scholar] [CrossRef] [PubMed]
- Bae, M.-J.; Li, F.; Kwon, Y.-S.; Chung, N.; Choi, H.; Hwang, S.-J.; Park, Y.-S. Concordance of diatom, macroinvertebrate and fish assemblages in streams at nested spatial scales: Implications for ecological integrity. Ecol. Indic. 2014, 47, 89–101. [Google Scholar] [CrossRef]
- Ward, J.H. Hierarchical grouping to optimize an objective function. J. Am. Stat. Assoc. 1963, 58, 236–244. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.B.; Simpson, G.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version. 2011. Available online: https://www.researchgate.net/publication/258996515_Vegan_Community_Ecology_Package_R_Package_Version_117-8 (accessed on 23 June 2018).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 23 June 2018).
- Dufrene, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345. [Google Scholar] [CrossRef]
- Roberts, D.W. Labdsv: Ordination and Multivariate Analysis for Ecology. R package version 1.8-0. 2016. Available online: https://CRAN.R-project.org/package=labdsv (accessed on 23 June 2018).
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Simpson, G.L.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version 1.13-1. 2008. Available online: http://vegan.r-forge.r-project.org/ (accessed on 23 June 2018).
- Virtanen, R.; Luoto, M.; Rämä, T.; Mikkola, K.; Hjort, J.; Grytnes, J.A.; Birks, H.J.B. Recent vegetation changes at the high-latitude tree line ecotone are controlled by geomorphological disturbance, productivity and diversity. Glob. Ecol. Biogeogr. 2010, 19, 810–821. [Google Scholar] [CrossRef]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Cutler, D.R.; Edwards, T.C., Jr.; Beard, K.H.; Cutler, A.; Hess, K.T.; Gibson, J.; Lawler, J.J. Random forests for classification in ecology. Ecology 2007, 88, 2783–2792. [Google Scholar] [CrossRef]
- Prasad, A.M.; Iverson, L.R.; Liaw, A. Newer classification and regression tree techniques: Bagging and random forests for ecological predictions. Ecosystems 2007, 9, 181–199. [Google Scholar] [CrossRef]
- Liaw, A.; Wiener, M. Classification and Regression by randomForest. R News 2002, 2/3, 18–22. [Google Scholar]
- Bae, M.-J.; Park, Y.-S. Diversity and distribution of endemic stream insects on a nationwide scale, South Korea: Conservation perspectives. Water 2017, 9, 833. [Google Scholar] [CrossRef]
- Kwon, Y.-S.; Bae, M.-J.; Hwang, S.-J.; Kim, S.-H.; Park, Y.-S. Predicting potential impacts of climate change on freshwater fish in Korea. Ecol. Inform. 2015, 29, 156–165. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R.; Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef]
- Stewart, P.M.; Butcher, J.T.; Swinford, T.O. Land use, habitat, and water quality effects on macroinvertebrate communities in three watersheds of a Lake Michigan associated marsh system. Aquat. Ecosyst. Health Manag. 2000, 3, 179–189. [Google Scholar] [CrossRef]
- Karube, Z.I.; Okada, N.; Tayasu, I. Sulfur stable isotope signature identifies the source of reduced sulfur in benthic communities in macrophyte zones of Lake Biwa, Japan. Limnology 2012, 13, 269–280. [Google Scholar] [CrossRef]
- Lodge, D.M.; Kelly, P. Habitat disturbance and the stability of freshwater gastropod populations. Oecologia 1985, 68, 111–117. [Google Scholar] [CrossRef]
- Brown, K.M.; George, G.; Daniel, W. Urbanization and a threatened freshwater mussel: Evidence from landscape scale studies. Hydrobiologia 2010, 655, 189–196. [Google Scholar] [CrossRef]
- Tchakonté, S.; Ajeagah, G.A.; Diomandé, D.; Camara, A.I.; Ngassam, P. Diversity, dynamic and ecology of freshwater snails related to environmental factors in urban and suburban streams in Douala-Cameroon (Central Africa). Aquat. Ecol. 2014, 48, 379–395. [Google Scholar] [CrossRef]
- Camara, I.A.; Bony, Y.K.; Diomandé, D.; Edia, O.E.; Konan, F.K.; Kouassi, C.N.; Gourene, G.; Pointier, J.P. Freshwater snail distribution related to environmental factors in Banco National Park, an urban reserve in the Ivory Coast (West Africa). Afr. Zool. 2012, 47, 160–168. [Google Scholar] [CrossRef]
- Evans, R.R.; Ray, S.J. Distribution and environmental influences on freshwater gastropods from lotic systems and springs in Pennsylvania, U.S.A., with conservation recommendations. Am. Malacol. Bull. 2010, 28, 135–150. [Google Scholar] [CrossRef]
- Venter, O.; Brodeur, N.N.; Nemiroff, L.; Belland, B.; Dolinsek, I.J.; Grant, J.W. Threats to endangered species in Canada. Aibs Bull. 2006, 56, 903–910. [Google Scholar] [CrossRef]
- Burlakova, L.E.; Karatayev, A.Y.; Karatayev, V.A.; May, M.E.; Bennett, D.L.; Cook, M.J. Endemic species: Contribution to community uniqueness, effect of habitat alteration, and conservation priorities. Biol. Conserv. 2011, 144, 155–165. [Google Scholar] [CrossRef]
- Matsuoka, K.; Kimura, T.; Kimura, S.; Yamaguchi, K.; Takayasu, K. Molluscan Fauna of the lower reaches of the Toyogawa River. Sci. Rep. Toyohashi Mus. Nat. Hist. 1999, 9, 15–24, (In Japanese with English Abstract). [Google Scholar]
- Lowe, S.; Browne, M.; Boudjelas, S. 100 of the World’s Worst Invasive Alien Species: A Selection from the Global Invasive Species Database; Invasive Species Specialist Group: Auckland, New Zealand, 2000. [Google Scholar]
- Bae, M.-J.; Kwon, Y.-S.; Park, Y.-S. Effects of global warming on the distribution of overwintering Pomacea canaliculata (Gastropoda: Ampullariidae) in Korea. Korean J. Limnol. 2012, 45, 453–458. [Google Scholar]
- Lee, D.-S.; Park, Y.-S. Factors affecting distribution and dispersal of Pomacea canaliculata in South Korea. Korean J. Ecol. Environ. 2020, 53, 185–194. [Google Scholar] [CrossRef]
- Bae, M.-J.; Chon, T.-S.; Park, Y.-S. Modeling behavior control of golden apple snails at different temperatures. Ecol. Model. 2015, 306, 86–94. [Google Scholar] [CrossRef]
- Seuffert, M.E.; Martín, P.R. Thermal limits for the establishment and growth of populations of the invasive apple snail Pomacea canaliculata. Biol. Invasions 2017, 19, 1169–1180. [Google Scholar] [CrossRef]
- Carlsson, N.O.L.; Brönmark, C.; Hansson, L.-A. Invading herbivory: The golden apple snail alters ecosystem functioning in Asian wetlands. Ecology 2004, 85, 1575–1580. [Google Scholar] [CrossRef]
- Clavero, M.; Brotons, L.; Pons, P.; Sol, D. Prominent role of invasive species in avian biodiversity loss. Biol. Conserv. 2009, 142, 2043–2049. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).