Effects of Land Use Types on CH4 and CO2 Production Potentials in Subtropical Wetland Soils
Abstract
1. Introduction
2. Materials and Methods
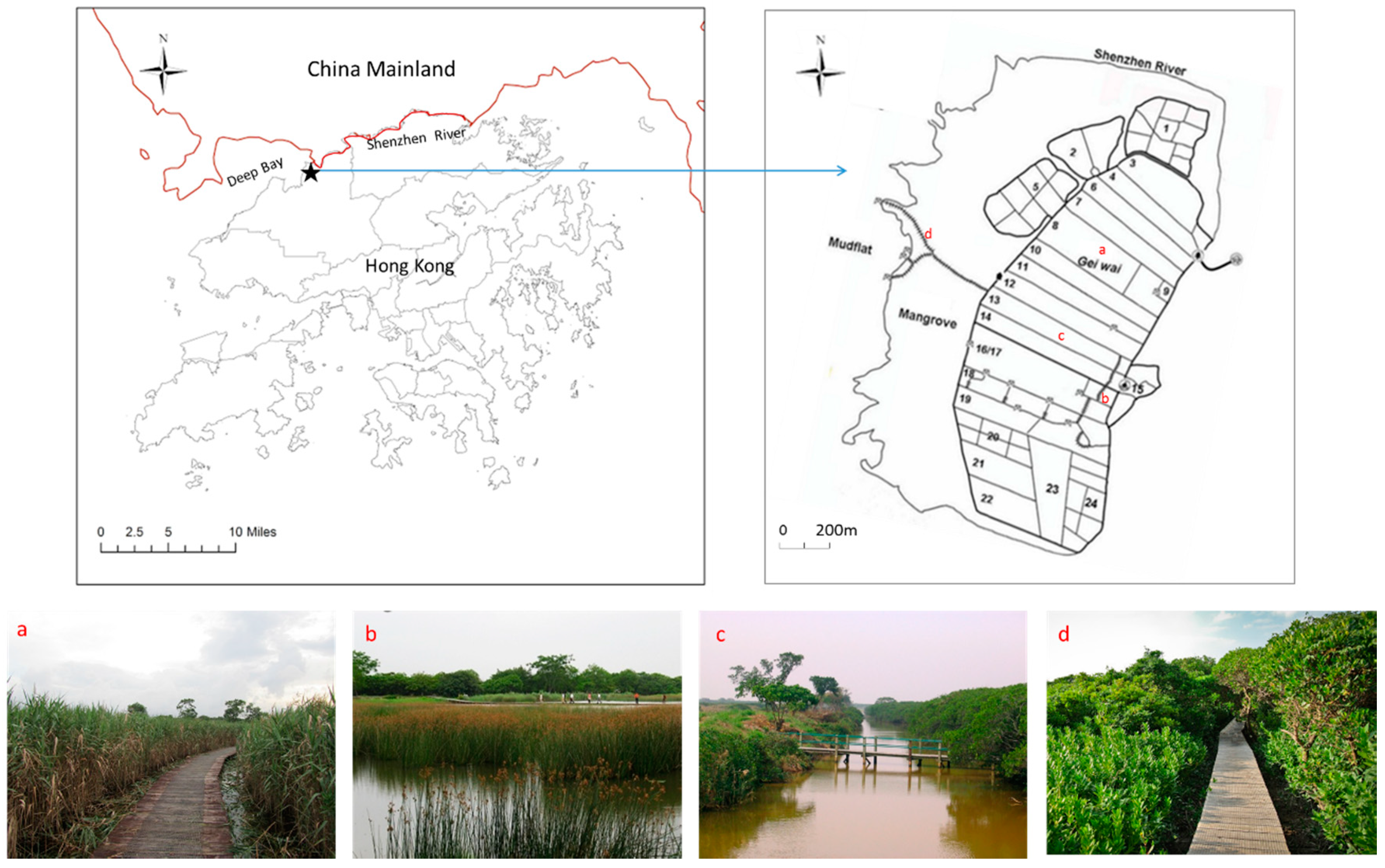
2.1. Study Site
- (1)
- Mangrove: The natural mangrove is dominated by Kandelia obovata, which forms a continuous belt of approximately 500 m in width and 3 km in length [40]. It represents the original land use type at this site before human disturbances. The tallest K. obovata community in this mangrove has a mean canopy height of 6.5 m, a mean diameter at breast height of 7.6 cm, and a mean tree density of 0.7 m−2 [41];
- (2)
- Gei Wai: The tidal shrimp ponds (locally called Gei Wais) were converted from the natural mangrove in the 1940s. These brackish shrimp ponds, each with a size of around 10 ha, are surrounded by a water channel (~1.5 m deep, 10 m wide) around the four inner edges. Each pond has a central vegetated platform which is dominated by K. obovata (referred to as Gei Wai forest thereafter) and is occasionally flooded, similar to the high intertidal zone of the mangrove [42]. The pond is operated by a sluice gate that controls the water level, stocking of shrimp fries, and harvesting of shrimps;
- (3)
- Reedbed: The reedbeds are dominated by Phragmites australis, and are mostly found in the central platform of some Gei Wais. The reeds became dominant after the tidal Gei Wais were converted to permanently flooded ponds in the 1980s, leading to the drowning of existing mangrove vegetation [42]. The reedbeds in Mai Po now cover a total of 46 ha and is one of the largest in the Guangdong province in southern China [43];
- (4)
- Freshwater pond: It was converted from mangrove vegetation to shrimp pond in the 1940s, and then subsequently to a rain-fed freshwater pond dominated by Scirpus subulatus in 1997 to provide a freshwater habitat for enhancing biodiversity in this coastal wetland.
2.2. Soil Sampling and Incubation
2.3. Measurement of Soil Properties
2.4. Determination of Soil C Fractions
2.5. Statistical Analysis
3. Results
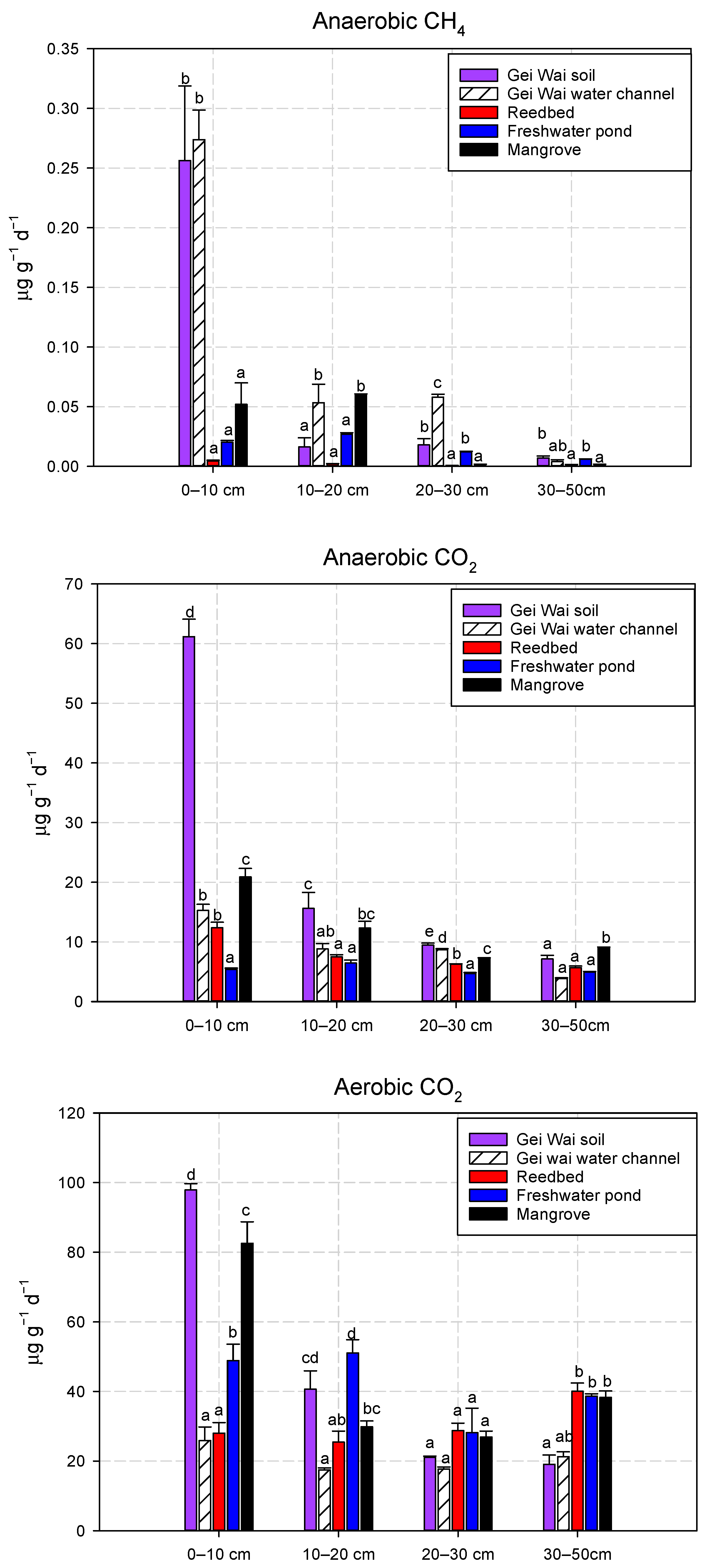
3.1. Effects of Land Use Types on CH4 and CO2 Production Potentials
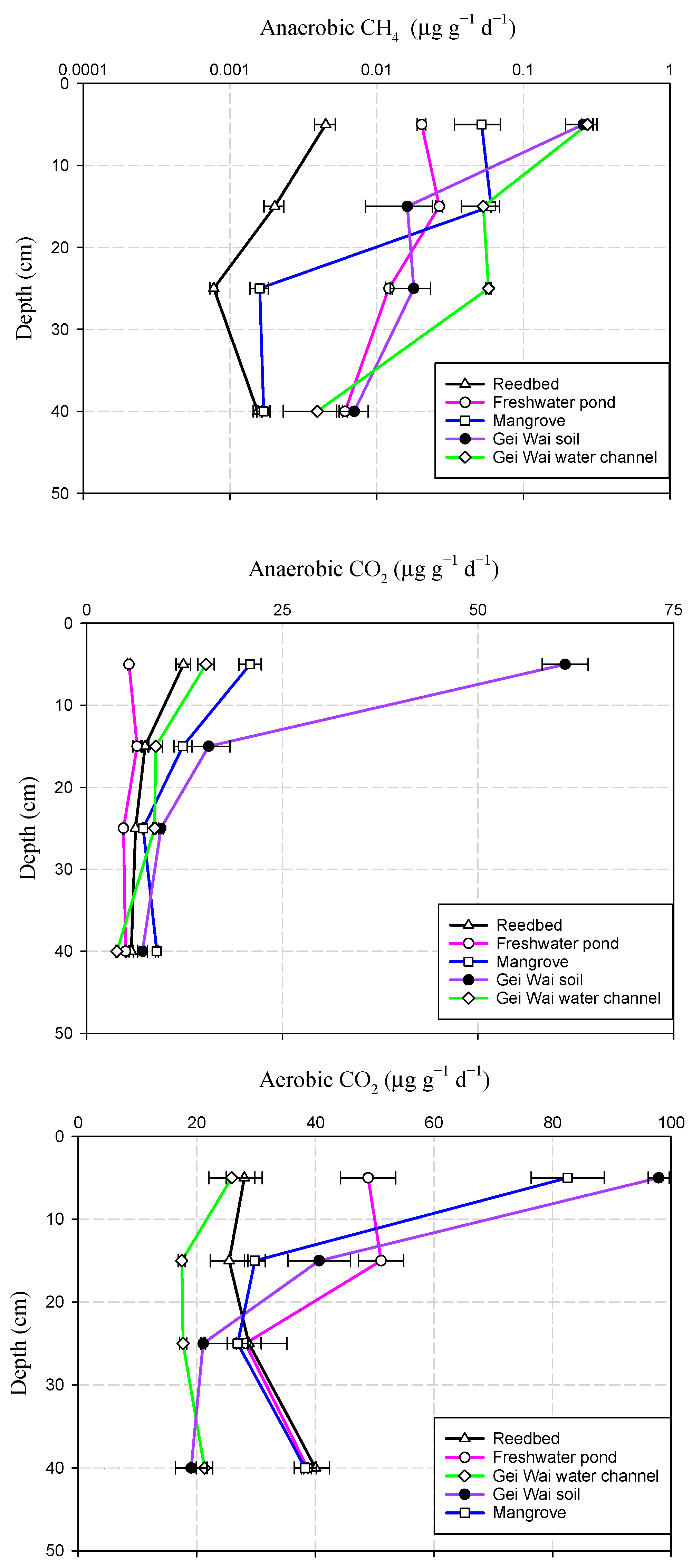
3.2. Variations of Soil CH4 and CO2 Production Potentials with Depth
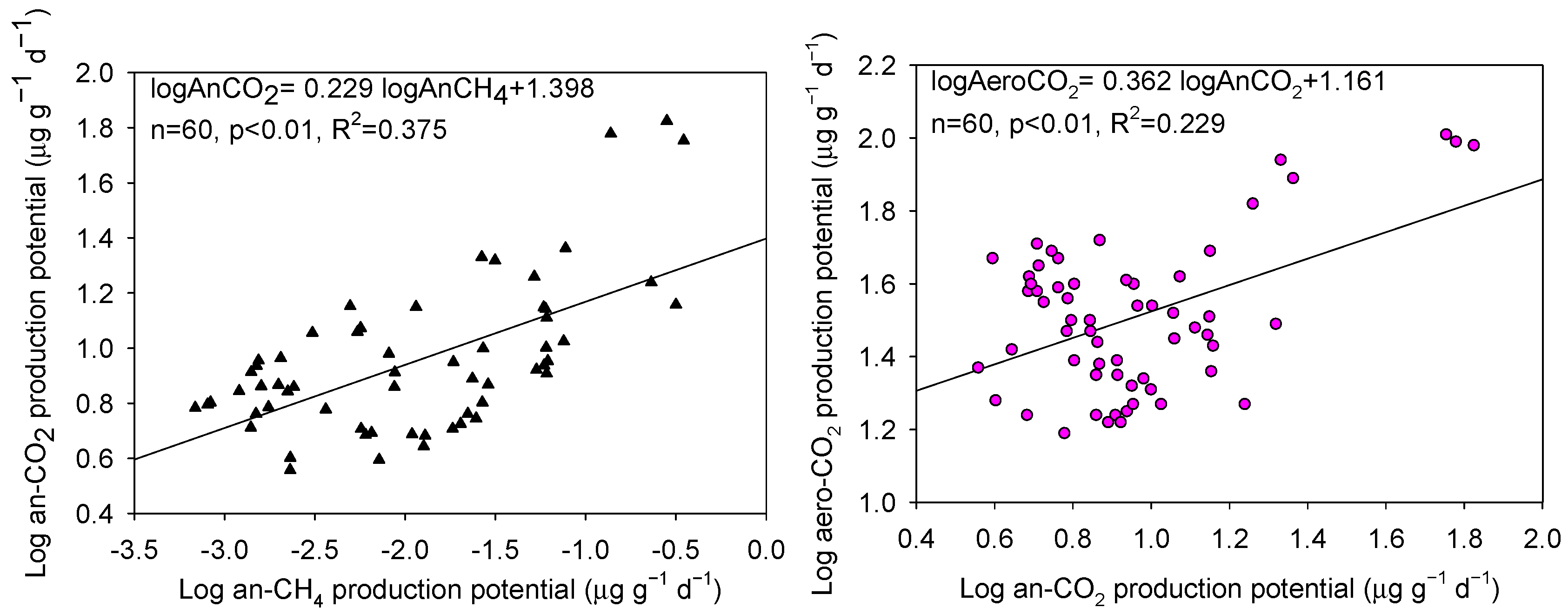
3.3. Relationships between Soil CH4 and CO2 Production Potentials
3.4. Effects of Land Use Types on Soil Physico-Chemical Properties
3.5. Relationships between Soil Properties and GHG Production Potentials
4. Discussion
4.1. CH4 and CO2 Production Potentials
4.2. Effects of Land Use Types on Soil CH4 and CO2 Production Potentials
4.3. Variations of CH4 and CO2 Production Potential with Depth
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Forster, P.; Ramaswamy, V.; Artaxo, P.; Berntsen, T.; Betts, R.; Fahey, D.W.; Haywood, J.; Lean, J.; Lowe, D.C.; Myhre, G. Changes in atmospheric constituents and in radiative forcing. In Climate Change 2007: The Physical Science Basis; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Lai, D. Methane dynamics in northern peatlands: A review. Pedosphere 2009, 19, 409–421. [Google Scholar] [CrossRef]
- Le Mer, J.; Roger, P. Production, oxidation, emission and consumption of methane by soils: A review. Eur. J. Soil Biol. 2001, 37, 25–50. [Google Scholar] [CrossRef]
- Sundh, I.; Nilsson, M.; Granberg, G.; Svensson, B. Depth distribution of microbial production and oxidation of methane in northern boreal peatlands. Microb. Ecol. 1994, 27, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Bergman, I.; Svensson, B.H.; Nilsson, M. Regulation of methane production in a Swedish acid mire by PH, temperature and substrate. Soil Biol. Biochem. 1998, 30, 729–741. [Google Scholar] [CrossRef]
- Yavitt, J.B.; Lang, G.E. Methane production in contrasting wetland sites: Response to organic-chemical components of peat and to sulfate reduction. Geomicrobiol. J. 1990, 8, 27–46. [Google Scholar] [CrossRef]
- Valentine, D.W.; Holland, E.A.; Schimel, D.S. Ecosystem and physiological controls over methane production in northern wetlands. J. Geophys. Res. Atmos. 1994, 99, 1563–1571. [Google Scholar] [CrossRef]
- Garcia, J.-L.; Patel, B.K.; Ollivier, B. Taxonomic, phylogenetic, and ecological diversity of methanogenic archaea. Anaerobe 2000, 6, 205–226. [Google Scholar] [CrossRef]
- Dunfield, P.; Dumont, R.; Moore, T.R. Methane production and consumption in temperate and subarctic peat soils: Response to temperature and PH. Soil Biol. Biochem. 1993, 25, 321–326. [Google Scholar] [CrossRef]
- Westermann, P.; Ahring, B.K. Dynamics of methane production, sulfate reduction, and denitrification in a permanently waterlogged alder swamp. Appl. Environ. Microbiol. 1987, 53, 2554–2559. [Google Scholar] [CrossRef]
- Bridgham, S.D.; Johnston, C.A.; Pastor, J.; Updegraff, K. Potential feedbacks of northern wetlands on climate change. BioScience 1995, 45, 262–274. [Google Scholar] [CrossRef]
- Yavitt, J.B.; Williams, C.J.; Wieder, R.K. Production of methane and carbon dioxide in peatland ecosystems across North America: Effects of temperature, aeration, and organic chemistry of peat. Geomicrobiol. J. 1997, 14, 299–316. [Google Scholar] [CrossRef]
- Inglett, K.; Inglett, P.; Reddy, K.; Osborne, T. Temperature sensitivity of greenhouse gas production in wetland soils of different vegetation. Biogeochemistry 2012, 108, 77–90. [Google Scholar] [CrossRef]
- Coles, J.R.; Yavitt, J.B. Control of methane metabolism in a forested northern wetland, New York state, by aeration, substrates, and peat size fractions. Geomicrobiol. J. 2002, 19, 293–315. [Google Scholar] [CrossRef]
- Poffenbarger, H.J.; Needelman, B.A.; Megonigal, J.P. Salinity influence on methane emissions from tidal marshes. Wetlands 2011, 31, 831–842. [Google Scholar] [CrossRef]
- Conrad, R.; Rothfuss, F. Methane oxidation in the soil surface layer of a flooded rice field and the effect of ammonium. Biol. Fertil. Soils 1991, 12, 28–32. [Google Scholar] [CrossRef]
- Treat, C.C.; Natali, S.M.; Ernakovich, J.; Iversen, C.M.; Lupascu, M.; McGuire, A.D.; Norby, R.J.; Roy Chowdhury, T.; Richter, A.; Šantrůčková, H. A pan-arctic synthesis of CH4 and CO2 production from anoxic soil incubations. Glob. Change Biol. 2015, 21, 2787–2803. [Google Scholar] [CrossRef]
- Bridgham, S.D.; Richardson, C.J. Mechanisms controlling soil respiration (CO2 and CH4) in southern peatlands. Soil Biol. Biochem. 1992, 24, 1089–1099. [Google Scholar] [CrossRef]
- Moore, T.R.; Dalva, M. Methane and carbon dioxide exchange potentials of peat soils in aerobic and anaerobic laboratory incubations. Soil Biol. Biochem. 1997, 29, 1157–1164. [Google Scholar] [CrossRef]
- Schlesinger, W.H.; Andrews, J.A. Soil respiration and the global carbon cycle. Biogeochemistry 2000, 48, 7–20. [Google Scholar] [CrossRef]
- Bergman, I.; Lundberg, P.; Nilsson, M. Microbial carbon mineralisation in an acid surface peat: Effects of environmental factors in laboratory incubations. Soil Biol. Biochem. 1999, 31, 1867–1877. [Google Scholar] [CrossRef]
- Kim, Y.; Ullah, S.; Roulet, N.T.; Moore, T.R. Effect of inundation, oxygen and temperature on carbon mineralization in boreal ecosystems. Sci. Total Environ. 2015, 511, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Alongi, D.M.; Tirendi, F.; Clough, B.F. Below-ground decomposition of organic matter in forests of the mangroves Rhizophora stylosa and Avicennia marina along the arid coast of Western Australia. Aquat. Bot. 2000, 68, 97–122. [Google Scholar] [CrossRef]
- Sainju, U.M.; Jabro, J.D.; Stevens, W.B. Soil carbon dioxide emission and carbon content as affected by irrigation, tillage, cropping system, and nitrogen fertilization. J. Environ. Qual. 2008, 37, 98–106. [Google Scholar] [CrossRef]
- Poeplau, C.; Don, A. Sensitivity of soil organic carbon stocks and fractions to different land-use changes across Europe. Geoderma 2013, 192, 189–201. [Google Scholar] [CrossRef]
- Aulakh, M.S.; Wassmann, R.; Bueno, C.; Rennenberg, H. Impact of root exudates of different cultivars and plant development stages of rice (Oryza sativa L.) on methane production in a paddy soil. Plant Soil 2001, 230, 77–86. [Google Scholar] [CrossRef]
- Alongi, D.M. Carbon sequestration in mangrove forests. Carbon Manag. 2012, 3, 313–322. [Google Scholar] [CrossRef]
- Donato, D.C.; Kauffman, J.B.; Murdiyarso, D.; Kurnianto, S.; Stidham, M.; Kanninen, M. Mangroves among the most carbon-rich forests in the tropics. Nat. Geosci. 2011, 4, 293–297. [Google Scholar] [CrossRef]
- Mcleod, E.; Chmura, G.L.; Bouillon, S.; Salm, R.; Björk, M.; Duarte, C.M.; Lovelock, C.E.; Schlesinger, W.H.; Silliman, B.R. A blueprint for blue carbon: Toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front. Ecol. Environ. 2011, 9, 552–560. [Google Scholar] [CrossRef]
- Pendleton, L.; Donato, D.C.; Murray, B.C.; Crooks, S.; Jenkins, W.A.; Sifleet, S.; Craft, C.; Fourqurean, J.W.; Kauffman, J.B.; Marbà, N. Estimating global “blue carbon” emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 2012, 7, e43542. [Google Scholar] [CrossRef]
- Duke, N.C.; Meynecke, J.O.; Dittmann, S.; Ellison, A.M.; Anger, K.; Berger, U.; Cannicci, S.; Diele, K.; Ewel, K.C.; Field, C.D. A world without mangroves? Science 2007, 317, 41–42. [Google Scholar] [CrossRef]
- Ouyang, X.; Lee, S.Y. Improved estimates on global carbon stock and carbon pools in tidal wetlands. Nat. Commun. 2020, 11, 1–7. [Google Scholar] [CrossRef]
- Sidik, F.; Lovelock, C.E. CO2 efflux from shrimp ponds in Indonesia. PLoS ONE 2013, 8, e66329. [Google Scholar] [CrossRef] [PubMed]
- Granek, E.; Ruttenberg, B.I. Changes in biotic and abiotic processes following mangrove clearing. Estuar. Coast. Shelf Sci. 2008, 80, 555–562. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Ruess, R.W.; Feller, I.C. CO2 efflux from cleared mangrove peat. PLoS ONE 2011, 6, e21279. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Sun, W.; Tong, C.; Zeng, C.; Yu, X.; Mou, X. China’s coastal wetlands: Conservation history, implementation efforts, existing issues and strategies for future improvement. Environ. Int. 2015, 79, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Lai, D.Y.F.; Lam, K.C. Phosphorus retention and release by sediments in the eutrophic Mai Po Marshes, Hong Kong. Mar. Pollut. Bull. 2008, 57, 349–356. [Google Scholar] [CrossRef]
- Chen, G.C.; Tam, N.F.Y.; Ye, Y. Spatial and seasonal variations of atmospheric N2O and CO2 fluxes from a subtropical mangrove swamp and their relationships with soil characteristics. Soil Biol. Biochem. 2012, 48, 175–181. [Google Scholar] [CrossRef]
- Hong Kong Observatory. Monthly Meteorological Normals for Hong Kong. Available online: http://www.weather.gov.hk/en/cis/normal/1981_2010/normals.htm# (accessed on 24 June 2020).
- Duke, N.C.; Khan, M.A. Structure and composition of the seaward mangrove forest at Mai Po Marshes Nature Reserve, Hong Kong. In The Mangrove Ecosystem of Deep Bay and the Mai Po Marshes; Lee, S.Y., Ed.; Hong Kong University Press: Hong Kong, China, 1999. [Google Scholar]
- Liu, J.; Lai, D.Y.F. Subtropical mangrove wetland is a stronger carbon dioxide sink in the dry than wet seasons. Agric. Forest Meteorol. 2019, 278, 107644. [Google Scholar] [CrossRef]
- Fitzgerald, W.J., Jr. Silvofisheries: Integrated mangrove forest aquaculture systems. In Ecological Aquaculture; Costa-Pierce, B.A., Ed.; Blackwell Publishing: Oxford, UK, 2002; pp. 161–262. [Google Scholar]
- Cha, M.W.; Young, L.; Wong, K.M. The fate of traditional extensive (Gei Wai) shrimp farming at the Mai Po Marshes Nature Reserve, Hong Kong. Hydrobiologia 1997, 352, 295–303. [Google Scholar] [CrossRef]
- Saarnio, S.; Alm, J.; Silvola, J.; Lohila, A.; Nykänen, H.; Martikainen, P.J. Seasonal variation in CH4 emissions and production and oxidation potentials at microsites on an oligotrophic pine fen. Oecologia 1997, 110, 414–422. [Google Scholar] [CrossRef]
- Black, C.A.; Evans, D.D.; White, J.L.; Ensminger, L.E.; Clark, F.E.; Rinauer, R.C. Methods of Soil Analysis, Part 2—Chemical and Microbiological Properties; American Society of Agronomy: Madison, WI, USA, 1965. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis, Part 3—Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America, Inc.: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Qian, P.; Schoenau, J. Availability of nitrogen in solid manure amendments with different C:N ratios. Can. J. Soil Sci. 2002, 82, 219–225. [Google Scholar] [CrossRef]
- Witt, C.; Biker, U.; Galicia, C.C.; Ottow, J.C. Dynamics of soil microbial biomass and nitrogen availability in a flooded rice soil amended with different C and N sources. Biol. Fertil. Soils 2000, 30, 520–527. [Google Scholar] [CrossRef]
- Haynes, R.; Swift, R. Stability of soil aggregates in relation to organic constituents and soil water content. J. Soil Sci. 1990, 41, 73–83. [Google Scholar] [CrossRef]
- Inubushi, K.; Brookes, P.; Jenkinson, D. Soil microbial biomass C, N and ninhydrin-N in aerobic and anaerobic soils measured by the fumigation-extraction method. Soil Biol. Biochem. 1991, 23, 737–741. [Google Scholar] [CrossRef]
- Vance, E.; Brookes, P.; Jenkinson, D. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Angers, D.; Mehuys, G. Effects of cropping on carbohydrate content and water-stable aggregation of a clay soil. Can. J. Soil Sci. 1989, 69, 373–380. [Google Scholar] [CrossRef]
- Neue, H.U.; Roger, P.A. Potential of methane emission in major rice ecologies. In Climate Biosphere Interaction: Biogenic Emissions and Environmental Effects of Climate Change; Zepp, R.G., Ed.; John Wiley and Sons, Inc.: Chichester, UK, 1994; pp. 65–94. [Google Scholar]
- Tong, C.; Huang, J.F.; Hu, Z.Q.; Jin, Y.F. Diurnal variations of carbon dioxide, methane, and nitrous oxide vertical fluxes in a subtropical estuarine marsh on neap and spring tide days. Estuar. Coasts 2013, 36, 633–642. [Google Scholar] [CrossRef]
- Prieme, A. Production and emission of methane in a brackish and a freshwater wetland. Soil Biol. Biochem. 1994, 26, 7–18. [Google Scholar] [CrossRef]
- Marton, J.M.; Herbert, E.R.; Craft, C.B. Effects of salinity on denitrification and greenhouse gas production from laboratory-incubated tidal forest soils. Wetlands 2012, 32, 347–357. [Google Scholar] [CrossRef]
- Glatzel, S.; Basiliko, N.; Moore, T. Carbon dioxide and methane production potentials of peats from natural, harvested and restored sites, Eastern Québec, Canada. Wetlands 2004, 24, 261–267. [Google Scholar] [CrossRef]
- Chowdhury, T.R.; Dick, R.P. Ecology of aerobic methanotrophs in controlling methane fluxes from wetlands. Appl. Soil Ecol. 2013, 65, 8–22. [Google Scholar] [CrossRef]
- Serrano-Silva, N.; Sarria-Guzmán, Y.; Dendooven, L.; Luna-Guido, M. Methanogenesis and methanotrophy in soil: A review. Pedosphere 2014, 24, 291–307. [Google Scholar] [CrossRef]
- Caldwell, S.L.; Laidler, J.R.; Brewer, E.A.; Eberly, J.O.; Sandborgh, S.C.; Colwell, F.S. Anaerobic oxidation of methane: Mechanisms, bioenergetics, and the ecology of associated microorganisms. Environ. Sci. Technol. 2008, 42, 6791–6799. [Google Scholar] [CrossRef] [PubMed]
- Thauer, R.K.; Shima, S. Methane as fuel for anaerobic microorganisms. Ann. N. Y. Acad. Sci. 2008, 1125, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Van den Pol-van Dasselaar, A.; Oenema, O. Methane production and carbon mineralisation of size and density fractions of peat soils. Soil Biol. Biochem. 1999, 31, 877–886. [Google Scholar] [CrossRef]
- Yavitt, J.B.; Lang, G.E.; Wieder, R.K. Control of carbon mineralization to CH4 and CO2 in anaerobic, sphagnum-derived peat from Big Run Bog, West Virginia. Biogeochemistry 1987, 4, 141–157. [Google Scholar] [CrossRef]
- Yavitt, J.B.; Basiliko, N.; Turetsky, M.R.; Hay, A.G. Methanogenesis and methanogen diversity in three peatland types of the Discontinuous Permafrost Zone, Boreal Western Continental Canada. Geomicrobiol. J. 2006, 23, 641–651. [Google Scholar] [CrossRef]
- Liu, D.; Ding, W.; Jia, Z.; Cai, Z. Relation between methanogenic archaea and methane production potential in selected natural wetland ecosystems across China. Biogeosciences 2011, 8, 329–338. [Google Scholar] [CrossRef]
- Allen, D.E.; Dalal, R.C.; Rennenberg, H.; Meyer, R.L.; Reeves, S.; Schmidt, S. Spatial and temporal variation of nitrous oxide and methane flux between subtropical mangrove sediments and the atmosphere. Soil Biol. Biochem. 2007, 39, 622–631. [Google Scholar] [CrossRef]
- Martin, R.M.; Moseman-Valtierra, S. Greenhouse gas fluxes vary between phragmites australis and native vegetation zones in coastal wetlands along a salinity gradient. Wetlands 2015, 35, 1021–1031. [Google Scholar] [CrossRef]
- Treat, C.; Wollheim, W.M.; Varner, R.; Grandy, A.S.; Talbot, J.; Frolking, S. Temperature and peat type control CO2 and CH4 production in Alaskan permafrost peats. Glob. Change Biol. 2014, 20, 2674–2686. [Google Scholar] [CrossRef] [PubMed]
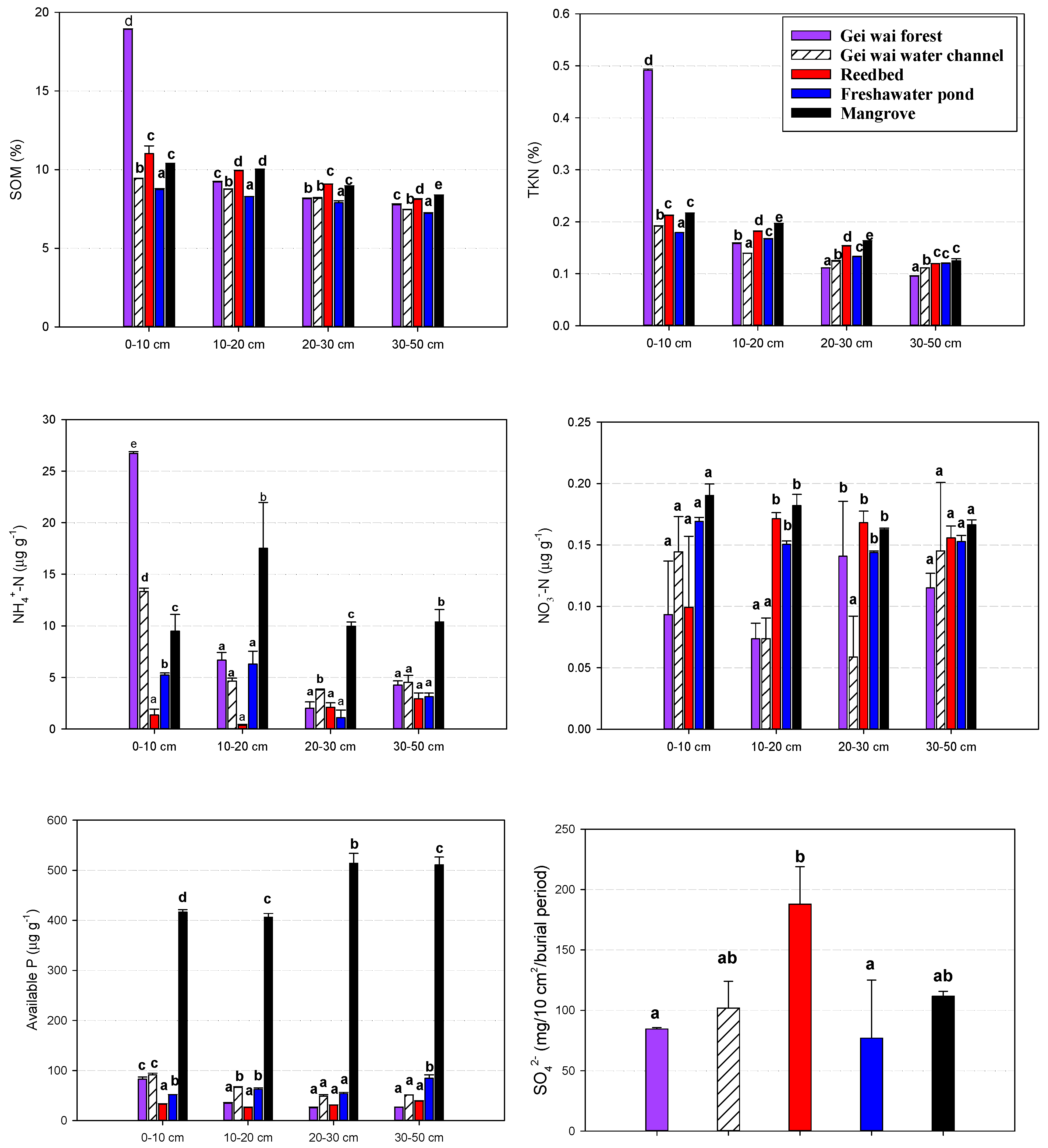
| Land Use Type | Water Table | pH | Salinity |
|---|---|---|---|
| cm | ‰ | ||
| Gei Wai forest | −18.00 ± 1.53 | 7.46 ± 0.14 | 5.33 ± 0.20 |
| Gei Wai water channel | 74.67 ± 2.40 | 8.10 ± 0.08 | 0.77 ± 0.00 |
| Reedbed | 0.70 ± 0.35 | 7.24 ± 0.12 | 7.10 ± 0.06 |
| Freshwater pond | 18.00 ± 2.52 | 6.91 ± 0.05 | 0.16 ± 0.00 |
| Mangrove | −2.33 ± 0.67 | 7.23 ± 0.01 | 11.73 ± 0.27 |
| Land Use Type | MBC | WSC | AHC | RMC | SOC |
|---|---|---|---|---|---|
| mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | % | |
| Gei Wai forest | 1724 ± 86.0a | 394 ± 26.0a | 1519 ± 64.9a | 699 ±22.6a | 4.90 ± 0.06a |
| Gei Wai water channel | 856 ± 258.3b | 151 ± 46.8b | 587 ± 116.1b | 308 ± 94.1b | 2.50 ± 0.42b |
| Reedbed | 1218 ± 103.0c | 276 ± 51.2c | 1016 ± 218.4c | 510 ± 47.4c | 3.73 ± 0.16c |
| Freshwater pond | 796 ± 55.7b | 150 ± 17.0b | 664 ± 106.1b | 316 ± 20.6b | 2.28 ± 0.13b |
| Mangrove | 1134 ± 60.9c | 163 ± 19.2b | 975 ± 164.9c | 507 ± 49.4c | 2.58 ± 0.18b |
| Land Use Type | Production Potential | Available P | NH4+-N | NO3−-N | SOM | TKN |
|---|---|---|---|---|---|---|
| Gei Wai forest | An-CH4 | 0.950 ** | 0.908 ** | n.s. | 0.918 ** | 0.917 ** |
| An-CO2 | 0.985 ** | 0.981 ** | n.s. | 0.991 ** | 0.992 ** | |
| Aero-CO2 | 0.972 ** | 0.982 ** | n.s. | 0.979 ** | 0.982 ** | |
| Gei Wai water channel | An-CH4 | 0.804 ** | 0.725 ** | n.s. | 0.722 ** | 0.788 ** |
| An-CO2 | 0.841 ** | 0.808 ** | n.s. | 0.924 ** | 0.930 ** | |
| Aero-CO2 | n.s. | n.s. | 0.639 * | n.s. | n.s. | |
| Reedbed | An-CH4 | n.s. | n.s. | −0.663 * | 0.744 ** | 0.736 ** |
| An-CO2 | n.s. | n.s. | −0.691 * | 0.780 ** | 0.861 ** | |
| Aero-CO2 | 0.697 * | n.s. | n.s. | n.s. | −0.663 * | |
| Freshwater pond | An-CH4 | n.s. | n.s. | n.s. | 0.792 ** | 0.875 ** |
| An-CO2 | n.s. | n.s. | n.s. | n.s. | n.s. | |
| Aero-CO2 | n.s. | 0.645 * | n.s. | n.s. | n.s. | |
| Mangrove | An-CH4 | −0.887 ** | n.s. | 0.632 * | 0.874 ** | 0.816 ** |
| An-CO2 | −0.707 * | n.s. | 0.600 * | 0.815 ** | 0.771 ** | |
| Aero-CO2 | n.s. | n.s. | n.s. | n.s. | n.s. |
| Land Cover | Country | Depth | Incubation Period | Incubation Temperature (°C) | an-CH4 | an-CO2 | aero-CO2 | Source |
|---|---|---|---|---|---|---|---|---|
| Sphagnum- and sedge-dominated boreal peatland | Sweden | 0–60 cm | 8 d | 16 | 0–6 μg g−1 soil day−1 | - | - | [4] |
| Peatlands of natural, harvested and restored sites | Canada | 0–10 cm and depth near water table | 4 d and 30 d for aerobic and anaerobic incubation, respectively | 20 | 0.00–816, average of 21 μg g−1 soil day−1 | 10–290 μg g−1 soil day−1 | 40–150 μg g−1 soil day−1 | [57] |
| Freshwater marsh with different vegetation | USA | 0–10 | 28 d | 20 | 5.3–19.6 μg g−1 soil day−1 | 50–95 μg g-1 soil day-1 | 400–660 μg g−1 soil day−1 | [13] |
| Boreal forest, peatland, lakes, rivers and exposed bedrocks | Canada | 0–5 and 5–15 cm | 16 weeks | 22 | 0.11–20.69 μg C g−1 C day−1 | 290–1500 μg C g-1 C day-1 | 160–2560 μg C g−1 C day−1 | [22] |
| Peatlands of bogs, fens and swamps | Canada | 0–110 cm | 5 d | 15 and 20 | 0.01–100, average of 11.37 μg g−1 soil day−1 | 70–5000, average of 1800 μg g−1 soil day−1 | 580–5000, average of 3700 μg g−1 soil day−1 | [19] |
| Sphagnum-derived peat from Big Run Bog | USA | 5–10, 20–25 and 40–45 cm depth | 10 d | 19 | 1.68 to 249.6 μg g−1 soil day−1 for surface soil | 120–720 μg g−1 soil day−1 for surface soil | - | [63] |
| Peatlands of bogs, internal lawns, and permafrost mounds | Canada | 0–100 cm | 60 d | 25 | 0.16 and 0.32–9.6 μg g−1 soil day−1 for permafrost and no permafrost bogs, 12.8 for lawns | 120–260 μg g−1 soil day−1 | - | [64] |
| Peatlands of bog and fen | Canada | 0–10, 10–20 cm | 3 d | 0–25 | 0.10–0.27 μg g−1 soil day−1 for 20 and 25 °C | - | - | [9] |
| Freshwater marsh of wetland | China | 0–60 cm | 20 | 0.011–0.66 μg g−1 soil day−1 | - | - | [65] | |
| Estuarine brackish marshes | China | 0–30 cm | 3 d | 20 | 0.058–0.142 μg g−1 soil day−1 at 30 cm | - | - | [54] |
| Coastal meadow, freshwater fen | Denmark | 0–80 cm | 6 d | 24 | 0–0.8 μg g−1 C day−1 for meadow 0–3.84 μg g−1 C day−1 for fen | - | - | [55] |
| Hong Kong, China | China | 0–10, 10–20, 20–30, 30–50 cm | 14 d and 7 d for anaerobic and aerobic incubation, respectively | 25 | 0.00–0.27 μg g−1 soil day−1 | 4.96–61.14 μg g−1 soil day−1 | 19.06–97.91 μg g−1 soil day−1 | Our study |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, J.; Lai, D.Y.F.; Neogi, S. Effects of Land Use Types on CH4 and CO2 Production Potentials in Subtropical Wetland Soils. Water 2020, 12, 1856. https://doi.org/10.3390/w12071856
Xu J, Lai DYF, Neogi S. Effects of Land Use Types on CH4 and CO2 Production Potentials in Subtropical Wetland Soils. Water. 2020; 12(7):1856. https://doi.org/10.3390/w12071856
Chicago/Turabian StyleXu, Jiaxing, Derrick Y. F. Lai, and Suvadip Neogi. 2020. "Effects of Land Use Types on CH4 and CO2 Production Potentials in Subtropical Wetland Soils" Water 12, no. 7: 1856. https://doi.org/10.3390/w12071856
APA StyleXu, J., Lai, D. Y. F., & Neogi, S. (2020). Effects of Land Use Types on CH4 and CO2 Production Potentials in Subtropical Wetland Soils. Water, 12(7), 1856. https://doi.org/10.3390/w12071856






