Development of a Regeneration Technique for Aluminum-Rich and Iron-Rich Phosphorus Sorption Materials
Abstract
1. Introduction
1.1. Sorption of P by Al/Fe-Rich Materials
1.2. Desorption of P from Al/Fe-Rich Materials
2. Materials and Methods
2.1. Characterization of PSMs
2.2. Preliminary Evaluation: Flow-through Experiments and Batch Isotherms
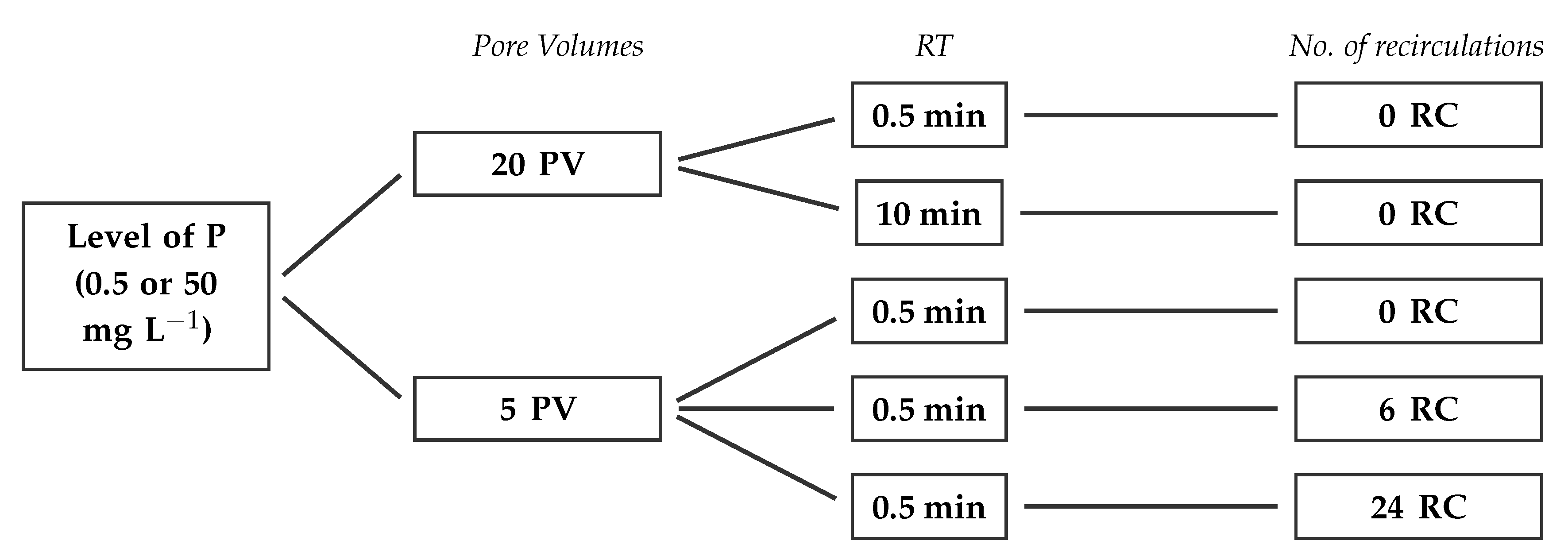
2.3. Sorption-Desorption Cycles: Testing Regeneration Treatments
3. Results and Discussion
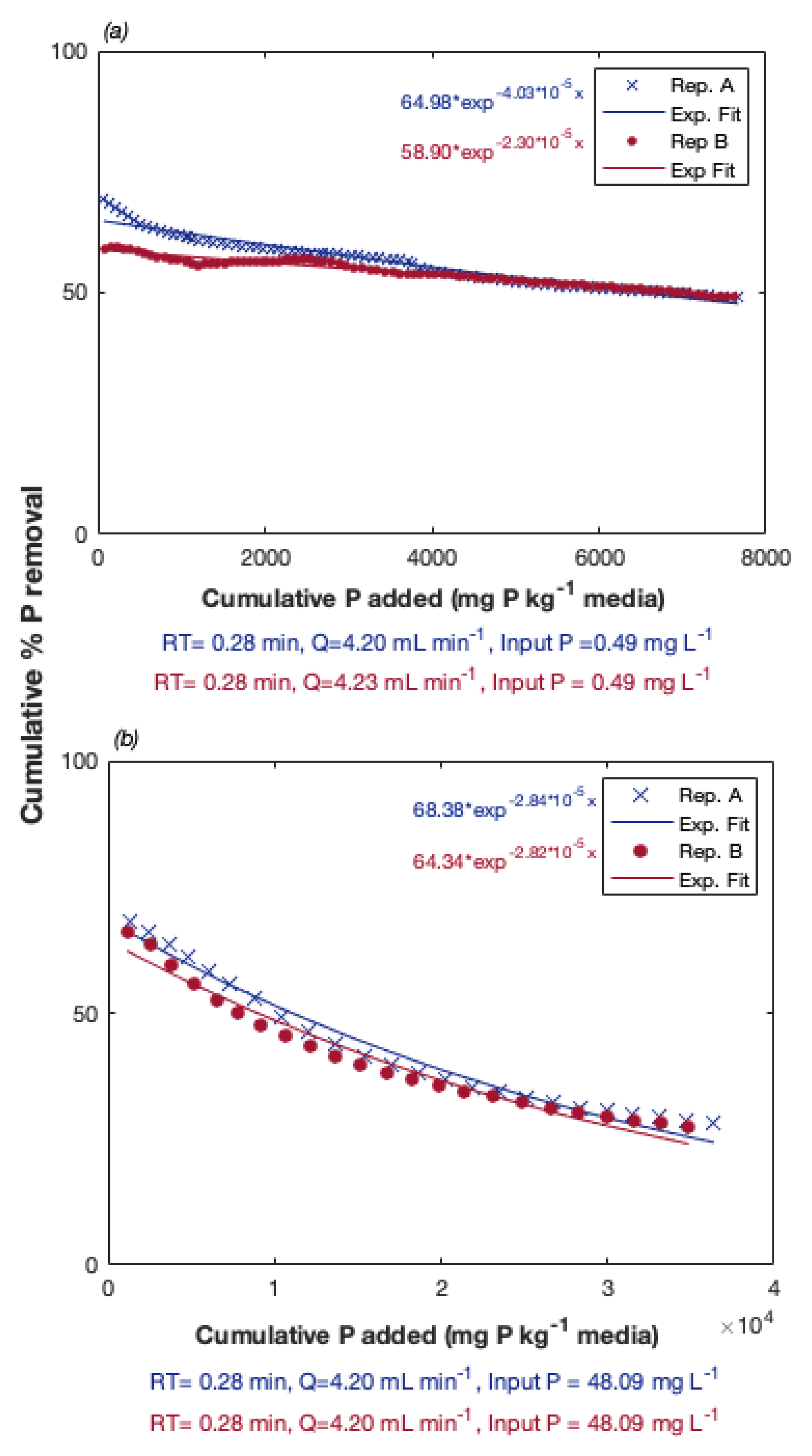
3.1. Preliminary Experiments: Evaluation of P Removal Ability of Al/Fe-Rich PSMs
3.2. Regeneration Treatments
3.2.1. Effect of P Concentration on Regeneration
3.2.2. Effect of Residence Time
3.2.3. Effect of Volume of Regenerative Solution
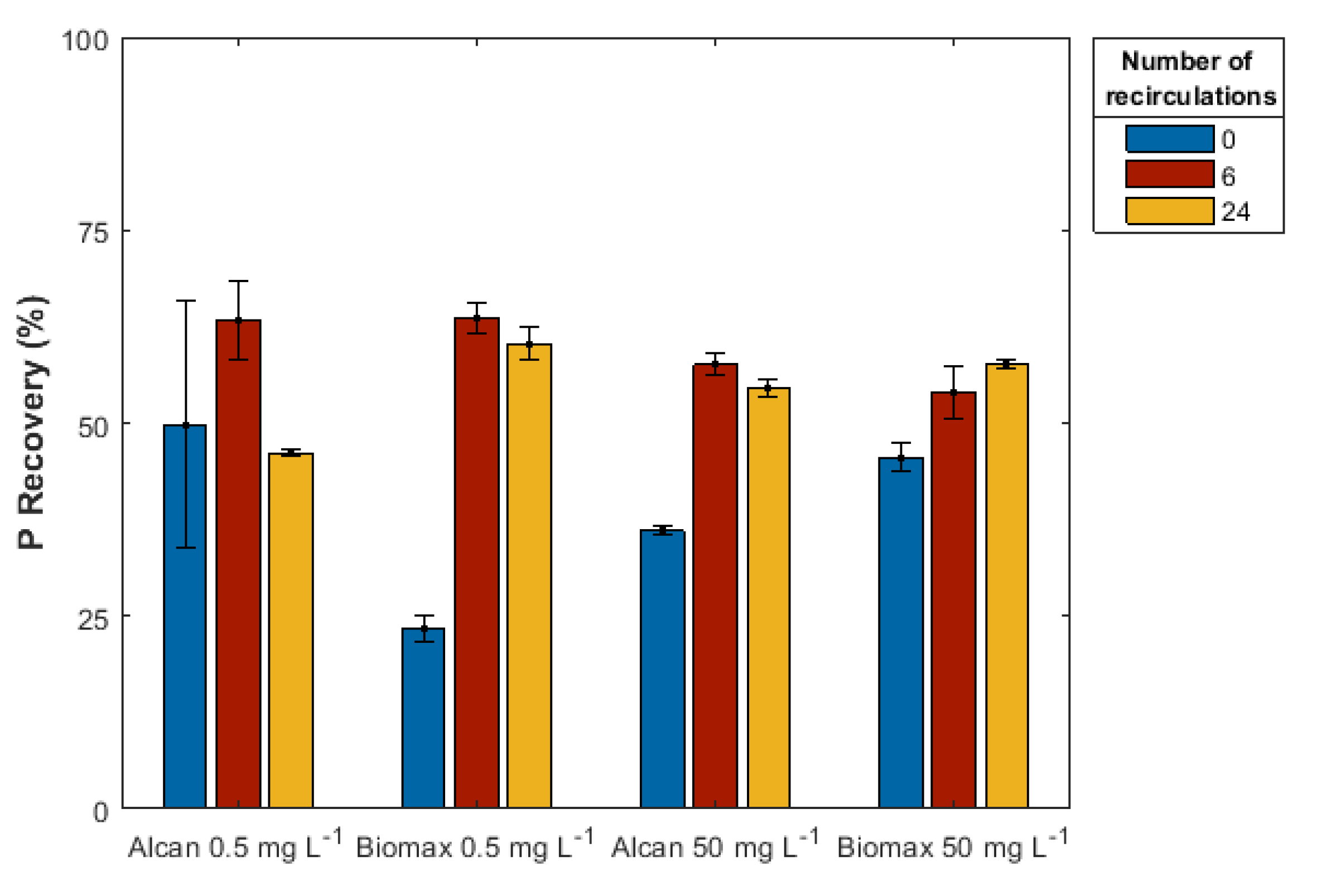
3.2.4. Effect of Number of Recirculations
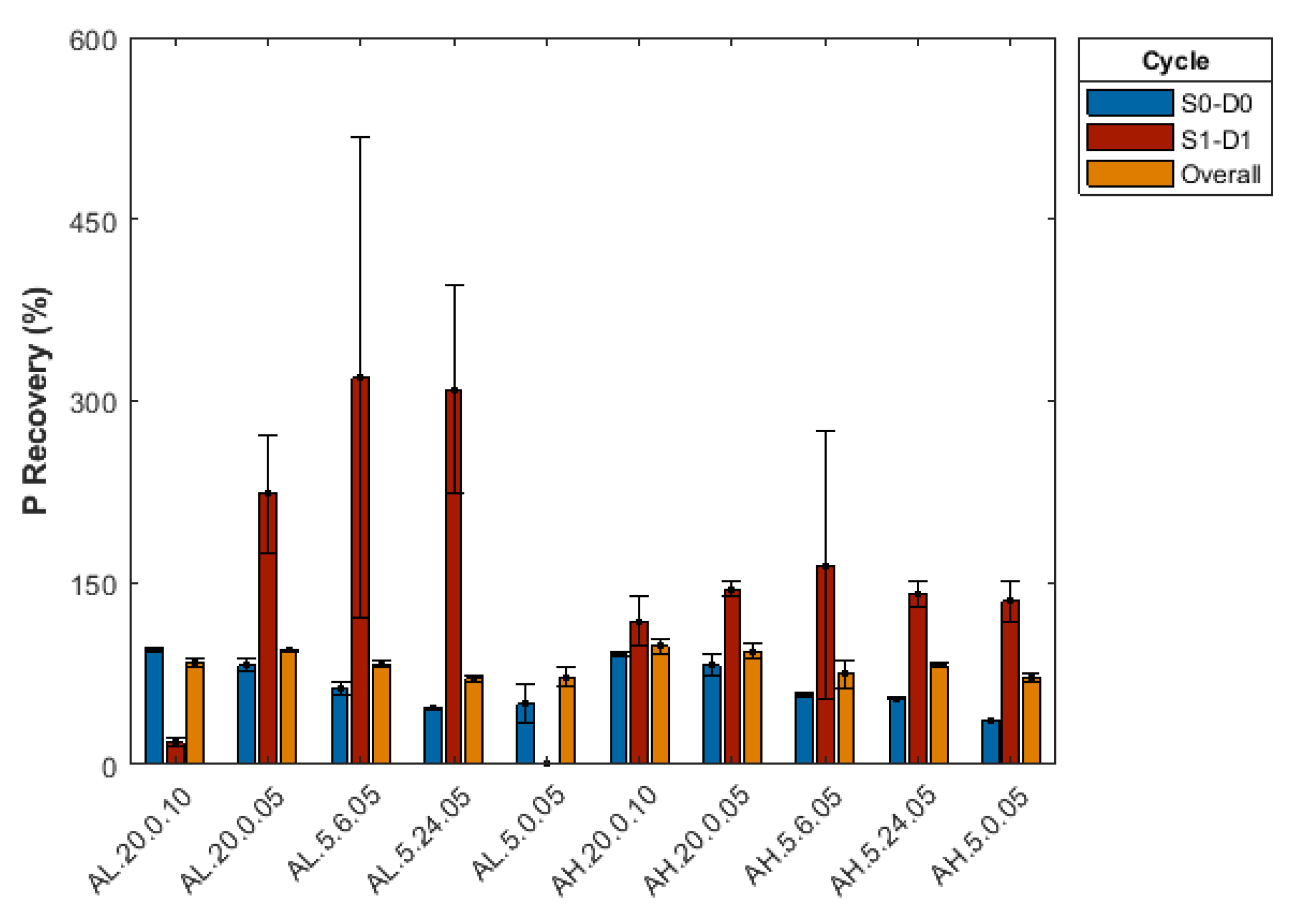
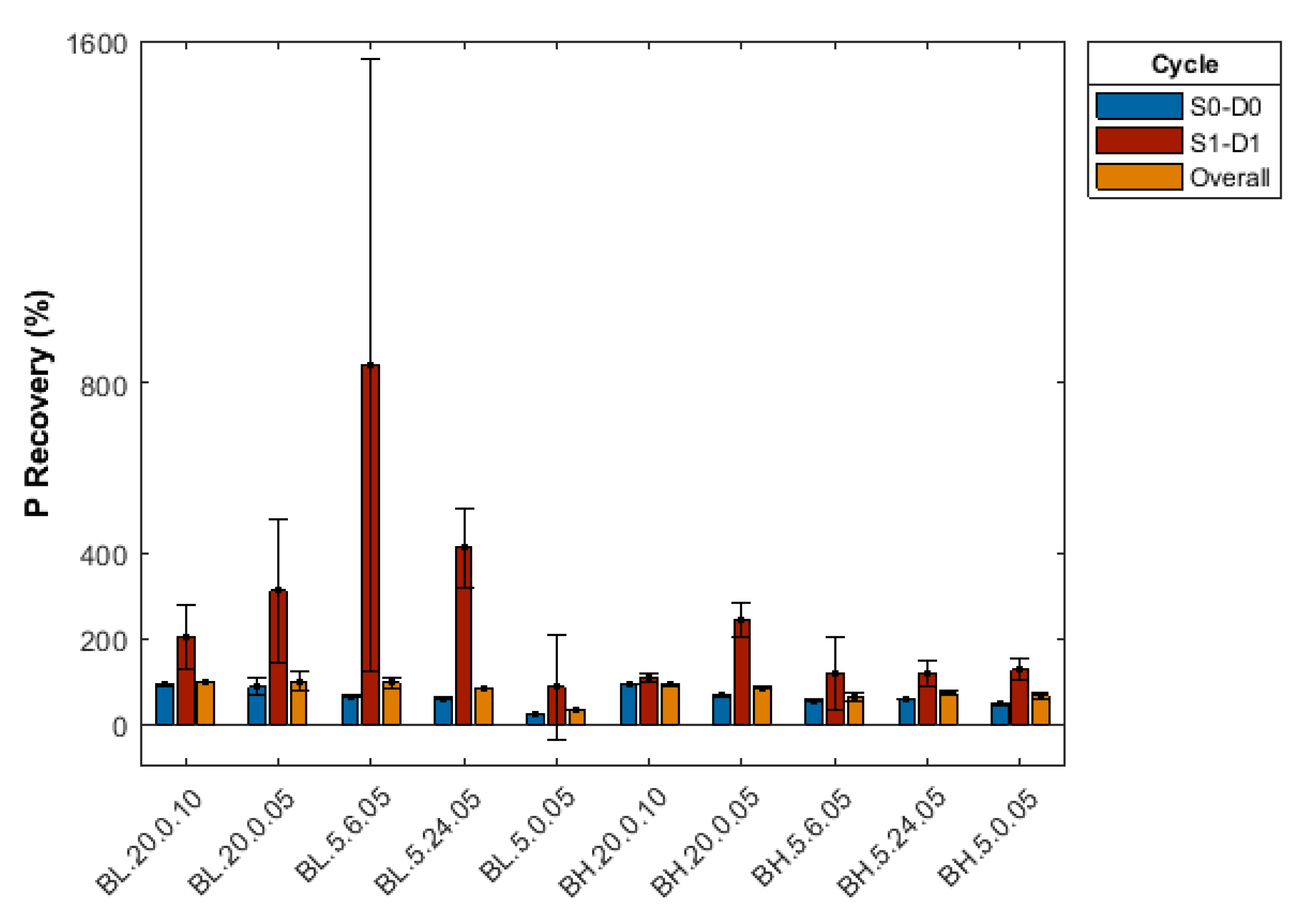
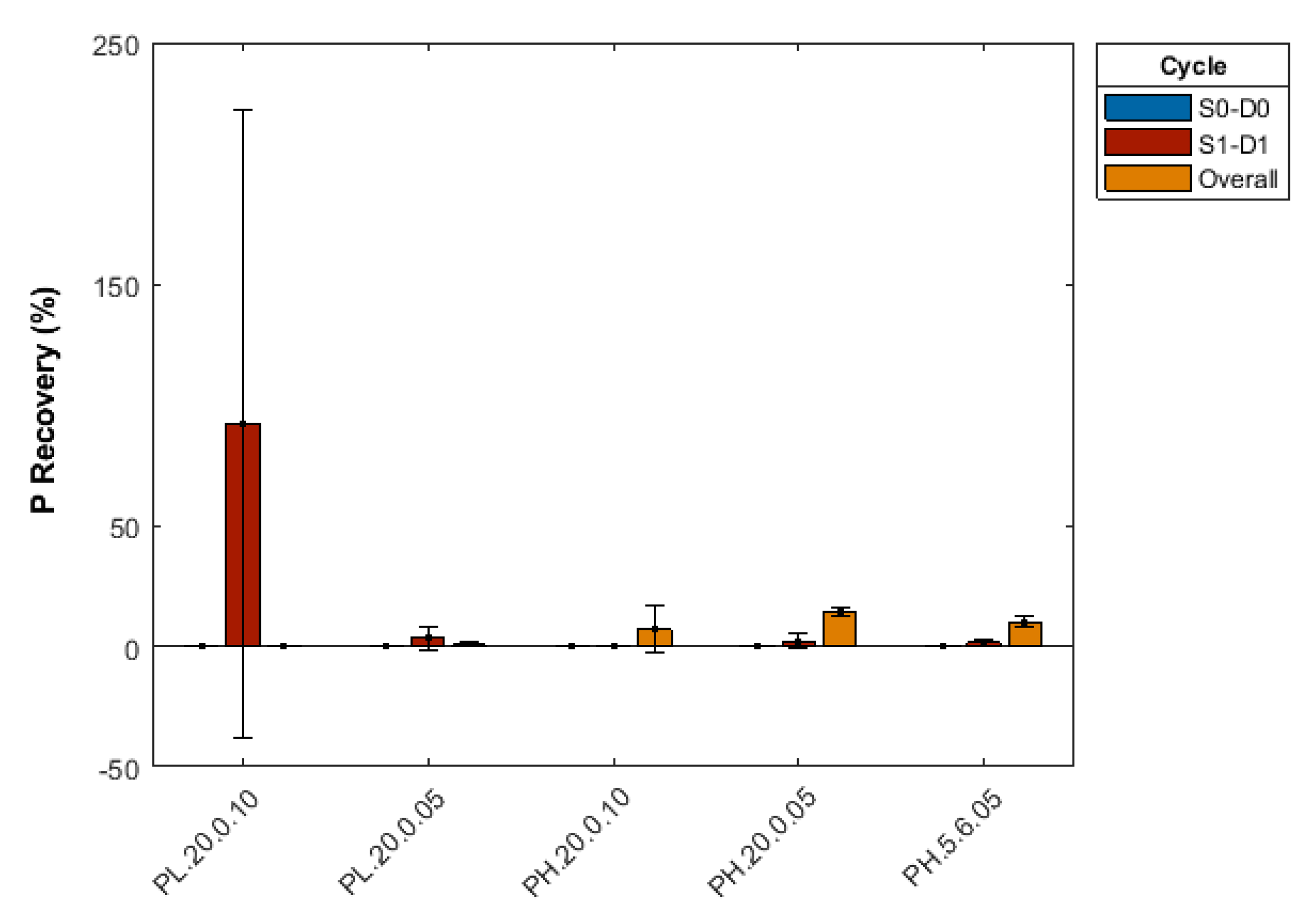
3.2.5. Effect of Repeated Desorption Cycles
4. Conclusions and Implications
Author Contributions
Funding
Conflicts of Interest
Appendix A. Effect of Residence Time on PhosRedeem
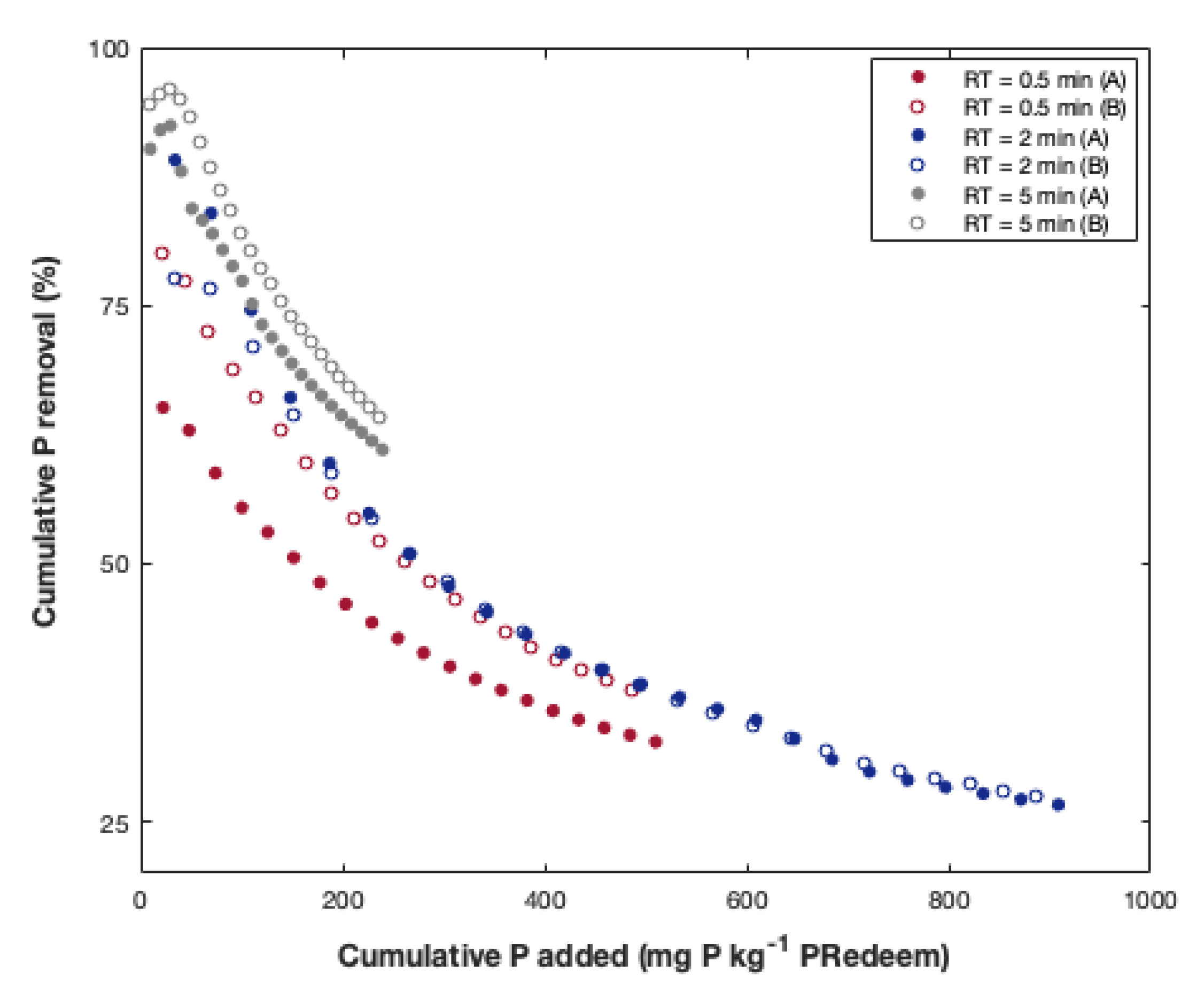
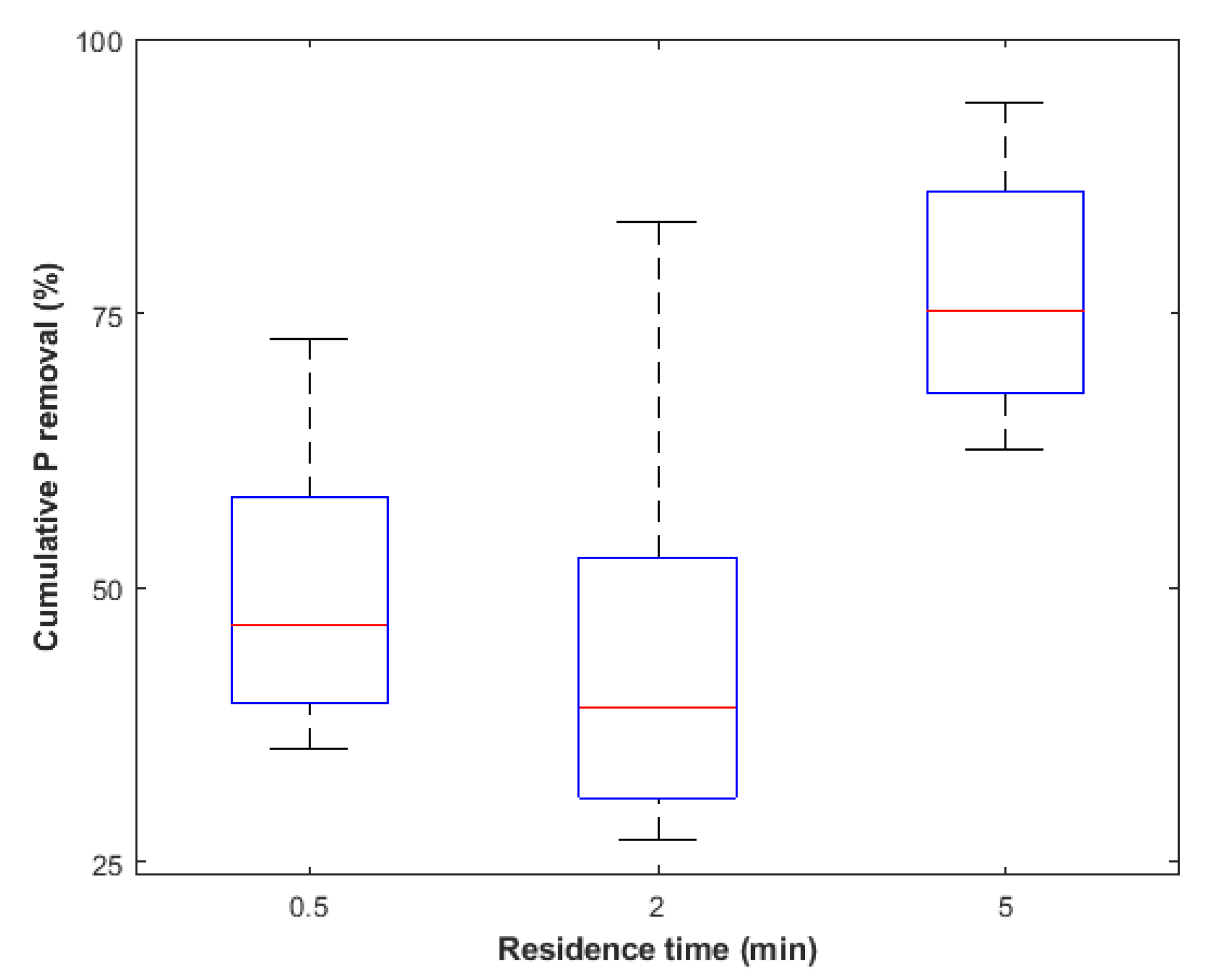
Appendix B. Summary of Sorption-Desorption Cycles
| Replicate | S0 | D0 | Net P Cycle 0 | S1 | D1 | Net P Cycle 1 | S2 | Net P | ||
|---|---|---|---|---|---|---|---|---|---|---|
| P Sorbed | P Desorbed | P Added | P Sorbed | P Desorbed | P Added | P Sorbed | ||||
| mg P kgPSM | ||||||||||
| A.20.0.10.1 | 11,463 | 10,704 | 759 | 2453 | 1343 | 276 | 834 | 3362 | 2377 | 4124 |
| A.20.0.10.2 | 11,463 | 10,884 | 579 | 2533 | 2377 | 377 | −221 | 2984 | 1791 | 4290 |
| A.20.0.05.1 | 11,463 | 9752 | 1711 | 2821 | 1123 | 2127 | −429 | 2986 | 1349 | 1976 |
| A.20.0.05.2 | 11,463 | 8860 | 2604 | 2848 | 1055 | 2725 | −932 | 2936 | 1728 | 2582 |
| A.5.0.05.1 | 11,463 | 4385 | 7079 | 2634 | 51 | 3190 | −607 | 2610 | 1189 | 5049 |
| A.5.0.05.2 | 11,463 | 6956 | 4508 | 2701 | 1188 | 2800 | −1287 | 2773 | 1538 | 4354 |
| A.5.6.05.1 | 11,463 | 6809 | 4654 | 2688 | 744 | 3422 | −1478 | 2951 | 1618 | 3515 |
| A.5.6.05.2 | 11,463 | 7617 | 3846 | 2821 | 1618 | 2896 | −1692 | 2999 | 1670 | 4159 |
| A.5.24.05.1 | 11,463 | 5293 | 6170 | 2596 | 1011 | 3739 | −2155 | 2609 | 1497 | 4858 |
| A.5.24.05.2 | 11,463 | 5210 | 6254 | 2559 | 1496 | 3718 | −2655 | 2582 | 1579 | 5530 |
| A.20.0.10.1 | 14,493 | 12,916 | 1577 | 19,496 | 4767 | 4980 | 9749 | 19,635 | 4414 | 5643 |
| A.20.0.10.2 | 14,493 | 13,246 | 1246 | 21,034 | 4313 | 5844 | 10,878 | 20,479 | 4720 | 4300 |
| A.20.0.05.1 | 14,493 | 12,641 | 1852 | 18,872 | 3513 | 4958 | 10,400 | 19,142 | 3143 | 3415 |
| A.20.0.05.2 | 14,493 | 10,877 | 3616 | 19,844 | 3123 | 4684 | 12,037 | 20,366 | 3158 | 5077 |
| A.5.0.05.1 | 14,493 | 5130 | 9363 | 17,579 | 7206 | 10,850 | −477 | 21,049 | 8599 | 14,183 |
| A.5.0.05.2 | 14,493 | 5232 | 9261 | 18,066 | 8473 | 10,548 | −955 | 18,195 | 6190 | 13,242 |
| A.5.6.05.1 | 14,493 | 8132 | 6361 | 18,196 | 2313 | 5686 | 10,197 | 18,592 | 5940 | 8791 |
| A.5.6.05.2 | 14,493 | 8406 | 6087 | 17,455 | 5909 | 5094 | 6452 | 17,463 | 5524 | 12,292 |
| A.5.24.05.1 | 14,493 | 7729 | 6764 | 18,781 | 6921 | 9345 | 2515 | 17,921 | 5883 | 10,088 |
| A.5.24.05.2 | 14,493 | 7933 | 6560 | 23,141 | 5767 | 8739 | 8635 | 17,795 | 7561 | 11,015 |
| Replicate | S0 | D0 | Net P Cycle 0 | S1 | D1 | Net P Cycle 1 | S2 | Net P | ||
|---|---|---|---|---|---|---|---|---|---|---|
| P Sorbed | P Desorbed | P Added | P Sorbed | P Desorbed | P Added | P Sorbed | ||||
| mg P kgPSM | ||||||||||
| B.20.0.10.1 | 7744 | 6871 | 941 | 947 | 548 | 1401 | −1002 | 1156 | 881 | 900 |
| B.20.0.10.2 | 7744 | 7294 | 518 | 1030 | 881 | 1329 | −1180 | 1064 | 847 | 849 |
| B.20.0.05.1 | 7812 | 7920 | −108 | 1040 | 357 | 1537 | −854 | 1203 | 722 | −635 |
| B.20.0.05.2 | 7812 | 5689 | 2123 | 1063 | 722 | 1386 | −1045 | 1170 | 538 | 1928 |
| B.5.0.05.1 | 7812 | 1721 | 6091 | 1059 | −274 | 779 | 555 | 1075 | 365 | 5335 |
| B.5.0.05.2 | 7812 | 1903 | 5909 | 1050 | 365 | 630 | 55 | 1134 | 467 | 6042 |
| B.5.6.05.1 | 7812 | 4812 | 3000 | 1231 | 270 | 3647 | −2687 | 1249 | 760 | 316 |
| B.5.6.05.2 | 7812 | 5041 | 2771 | 1322 | 760 | 2533 | −1971 | 1393 | 549 | 1480 |
| B.5.24.05.1 | 7812 | 4784 | 3028 | 1107 | 447 | 2141 | −1481 | 1221 | 744 | 2010 |
| B.5.24.05.2 | 7812 | 4558 | 3254 | 458 | 744 | 2578 | −2864 | 1203 | 813 | 2164 |
| B.20.0.10.1 | 18,675 | 16,110 | 2565 | 22,872 | 4995 | 5753 | 12,124 | 22,332 | 5350 | 1105 |
| B.20.0.10.2 | 18,675 | 16,210 | 2465 | 17,296 | 5344 | 5394 | 6558 | 18,124 | 3845 | −142 |
| B.20.0.05.1 | 18,675 | 11,547 | 7128 | 22,254 | 2929 | 6293 | 13,032 | 19,547 | 2232 | 2009 |
| B.20.0.05.2 | 18,675 | 12,110 | 6565 | 17,923 | 2203 | 6017 | 9703 | 23,586 | 5578 | 5068 |
| B.5.0.05.1 | 18,675 | 7800 | 10,875 | 20,310 | 8509 | 9417 | 2385 | 21,775 | 7169 | 7569 |
| B.5.0.05.2 | 18,675 | 8245 | 10,430 | 19,032 | 6982 | 10,116 | 1934 | 21,711 | 8297 | 7553 |
| B.5.6.05.1 | 18,675 | 9097 | 9578 | 23,939 | 3427 | 6147 | 14,365 | 24,318 | 9822 | 12,196 |
| B.5.6.05.2 | 18,675 | 9931 | 8744 | 20,803 | 9765 | 5680 | 5358 | 21,197 | 5606 | 7612 |
| B.5.24.05.1 | 18,675 | 10,237 | 8438 | 21,140 | 7182 | 10,131 | 3827 | 21,162 | 10,014 | 7263 |
| B.5.24.05.2 | 18,675 | 10,096 | 8579 | 20,053 | 9836 | 9436 | 781 | 23,148 | 10,903 | 8988 |
| Replicate | S0 | D0 | Net P Cycle 0 | S1 | D1 | Net P Cycle 1 | S2 | Net P | ||
|---|---|---|---|---|---|---|---|---|---|---|
| P Sorbed | P Desorbed | P Added | P Sorbed | P Desorbed | P Added | P Sorbed | ||||
| mg P kgPSM | ||||||||||
| P.20.0.10.1 | 1376 | 0 | 1376 | 1190 | 1036 | 0 | 1035 | 1277 | 129 | 2540 |
| P.20.0.10.2 | 1376 | 3 | 1373 | 1150 | 1021 | 0 | 1021 | 1154 | 102 | 2496 |
| P.20.0.05.1 | 1376 | 0 | 1376 | 1125 | 862 | 21 | 842 | 1261 | 212 | 2429 |
| P.20.0.05.2 | 1376 | 0 | 1376 | 1098 | 887 | 0 | 887 | 1242 | 201 | 2463 |
| P.20.0.10.1 | 15,292 | 2198 | 13,095 | 19,671 | 18,048 | 145 | 17,903 | 19,781 | 1333 | 30,746 |
| P.20.0.10.2 | 15,292 | 2402 | 12,891 | 19,627 | 18,294 | 164 | 18,129 | 18,885 | 1251 | 30,686 |
| P.20.0.05.1 | 15,292 | 1555 | 13,737 | 19,306 | 20,967 | 197 | 20,816 | 19,096 | 776 | 33,698 |
| P.20.0.05.2 | 15,292 | 1355 | 13,937 | 19,783 | 19,008 | 34 | 19,027 | 15,899 | 1370 | 32,697 |
| P.5.6.05.1 | 15,292 | 1522 | 13,770 | 18,489 | 18,791 | 195 | 18,679 | 17,473 | 294 | 31,075 |
| P.5.6.05.2 | 15,292 | 1463 | 13,829 | 18,753 | 18,459 | 181 | 18,380 | 18,187 | 349 | 30,871 |
References
- Vohla, C.; Kõiv, M.; Bavor, H.J.; Chazarenc, F.; Mander, Ü. Filter materials for phosphorus removal from wastewater in treatment wetlands—A review. Ecol. Eng. 2011, 37, 70–89. [Google Scholar] [CrossRef]
- Penn, C.; Chagas, I.; Klimeski, A.; Lyngsie, G. A review of phosphorus removal structures: How to assess and compare their performance. Water 2017, 9, 583. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Wallace, S.; Comeau, Y. Model of phosphorus precipitation and crystal formation in electric arc furnace steel slag filters. Environ. Sci. Technol. 2012, 46, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Allred, B.J.; Martinez, L.R.; Gamble, D.L. Phosphate removal from agricultural drainage water using an iron oxyhydroxide filter material. Water Air Soil Pollut. 2017, 228, 240. [Google Scholar] [CrossRef]
- Dobbie, K.; Heal, K.; Aumonier, J.; Smith, K.; Johnston, A.; Younger, P. Evaluation of iron ochre from mine drainage treatment for removal of phosphorus from wastewater. Chemosphere 2009, 75, 795–800. [Google Scholar] [CrossRef]
- McCobb, T.D.; LeBlanc, D.R.; Massey, A.J. Monitoring the removal of phosphate from ground water discharging through a pond-bottom permeable reactive barrier. Groundw. Monit. Remediat. 2009, 29, 43–55. [Google Scholar] [CrossRef]
- Wood, R.; McAtamney, C. Constructed wetlands for waste water treatment: The use of laterite in the bed medium in phosphorus and heavy metal removal. Hydrobiologia 1996, 340, 323–331. [Google Scholar] [CrossRef]
- Bastin, O.; Janssens, F.; Dufey, J.; Peeters, A. Phosphorus removal by a synthetic iron oxide–gypsum compound. Ecol. Eng. 1999, 12, 339–351. [Google Scholar] [CrossRef]
- Boujelben, N.; Bouzid, J.; Elouear, Z.; Feki, M.; Jamoussi, F.; Montiel, A. Phosphorus removal from aqueous solution using iron coated natural and engineered sorbents. J. Hazard. Mater. 2008, 151, 103–110. [Google Scholar] [CrossRef]
- Clayton, M.E.; Liegeois, S.; Brown, E.J. Phosphorus sequestration in lake sediment with iron mine tailings. Soil Sediment Contam. 2004, 13, 421–431. [Google Scholar] [CrossRef]
- Ayoub, G.M.; Koopman, B.; Pandya, N. Iron and aluminum hydroxy (oxide) coated filter media for low-concentration phosphorus removal. Water Environ. Res. 2001, 73, 478–485. [Google Scholar] [CrossRef]
- Li, M.; Liu, J.; Xu, Y.; Qian, G. Phosphate adsorption on metal oxides and metal hydroxides: A comparative review. Environ. Rev. 2016, 24, 319–332. [Google Scholar] [CrossRef]
- Mao, Y.; Yue, Q. Kinetic modeling of phosphate adsorption by preformed and in situ formed hydrous ferric oxides at circumneutral pH. Sci. Rep. 2016, 6, 35292. [Google Scholar] [CrossRef]
- De Vicente, I.; Jensen, H.S.; Andersen, F.Ø. Factors affecting phosphate adsorption to aluminum in lake water: Implications for lake restoration. Sci. Total Environ. 2008, 389, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Wilfert, P.; Kumar, P.S.; Korving, L.; Witkamp, G.J.; van Loosdrecht, M.C. The relevance of phosphorus and iron chemistry to the recovery of phosphorus from wastewater: A review. Environ. Sci. Technol. 2015, 49, 9400–9414. [Google Scholar] [CrossRef] [PubMed]
- Siwek, H.; Bartkowiak, A.; Włodarczyk, M. Adsorption of Phosphates from Aqueous Solutions on Alginate/Goethite Hydrogel Composite. Water 2019, 11, 633. [Google Scholar] [CrossRef]
- Tanada, S.; Kabayama, M.; Kawasaki, N.; Sakiyama, T.; Nakamura, T.; Araki, M.; Tamura, T. Removal of phosphate by aluminum oxide hydroxide. J. Colloid Interface Sci. 2003, 257, 135–140. [Google Scholar] [CrossRef]
- Sibrell, P.L.; Kehler, T. Phosphorus removal from aquaculture effluents at the Northeast Fishery Center in Lamar, Pennsylvania using iron oxide sorption media. Aquac. Eng. 2016, 72, 45–52. [Google Scholar] [CrossRef]
- Sibrell, P.L.; Montgomery, G.A.; Ritenour, K.L.; Tucker, T.W. Removal of phosphorus from agricultural wastewaters using adsorption media prepared from acid mine drainage sludge. Water Res. 2009, 43, 2240–2250. [Google Scholar] [CrossRef]
- Kunaschk, M.; Schmalz, V.; Dietrich, N.; Dittmar, T.; Worch, E. Novel regeneration method for phosphate loaded granular ferric (hydr) oxide–A contribution to phosphorus recycling. Water Res. 2015, 71, 219–226. [Google Scholar] [CrossRef]
- Axens Solutions. ActiGuard®—Water Treatment Series—Arsenic & Metals Removal from Water. 2017. Available online: https://www.axens.net/product/catalysts-a-adsorbents/228/actiguard-aafs50.html (accessed on 21 October 2019).
- ABS Materials. BioMaxTM Media Application for Bioswales. 2016. Available online: http://abswastewater.com/markets-served/stormwater-treatment/ (accessed on 21 October 2019).
- Penn, C.J.; Bowen, J.M. Design and Construction of Phosphorus Removal Structures for Improving Water Quality; Springer International Publishing AG: Cham, Switzerland, 2017. [Google Scholar]
- Awual, M.R.; Jyo, A.; Ihara, T.; Seko, N.; Tamada, M.; Lim, K.T. Enhanced trace phosphate removal from water by zirconium (IV) loaded fibrous adsorbent. Water Res. 2011, 45, 4592–4600. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- USEPA, Environment and Climate Change Canada. Annex 4 Objects and Targets Task Team, 2015: Recommended Phosphorus Loading Targets for Lake Erie. Available online: http://scavia.seas.umich.edu/wp-content/uploads/2015/06/FINAL-Objectives-TT-report-en-150624-3.pdf (accessed on 20 October 2019).
- McBride, M.B. Environmental Chemistry of Soils; Oxford University Press: New York, NY, USA, 1994. [Google Scholar]
- Cabrera, F.; De Arambarri, P.; Madrid, L.; Toga, C. Desorption of phosphate from iron oxides in relation to equilibrium pH and porosity. Geoderma 1981, 26, 203–216. [Google Scholar] [CrossRef]
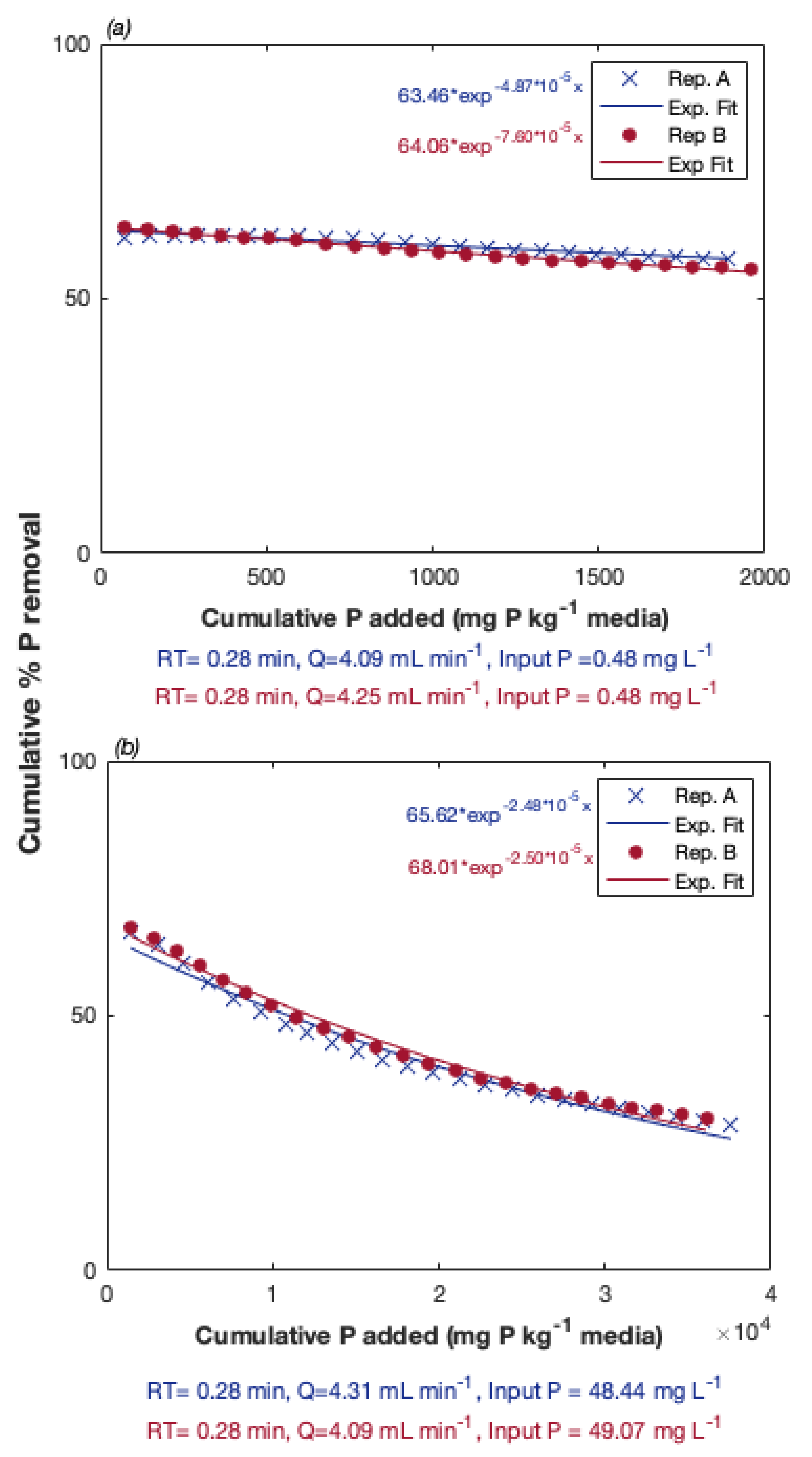
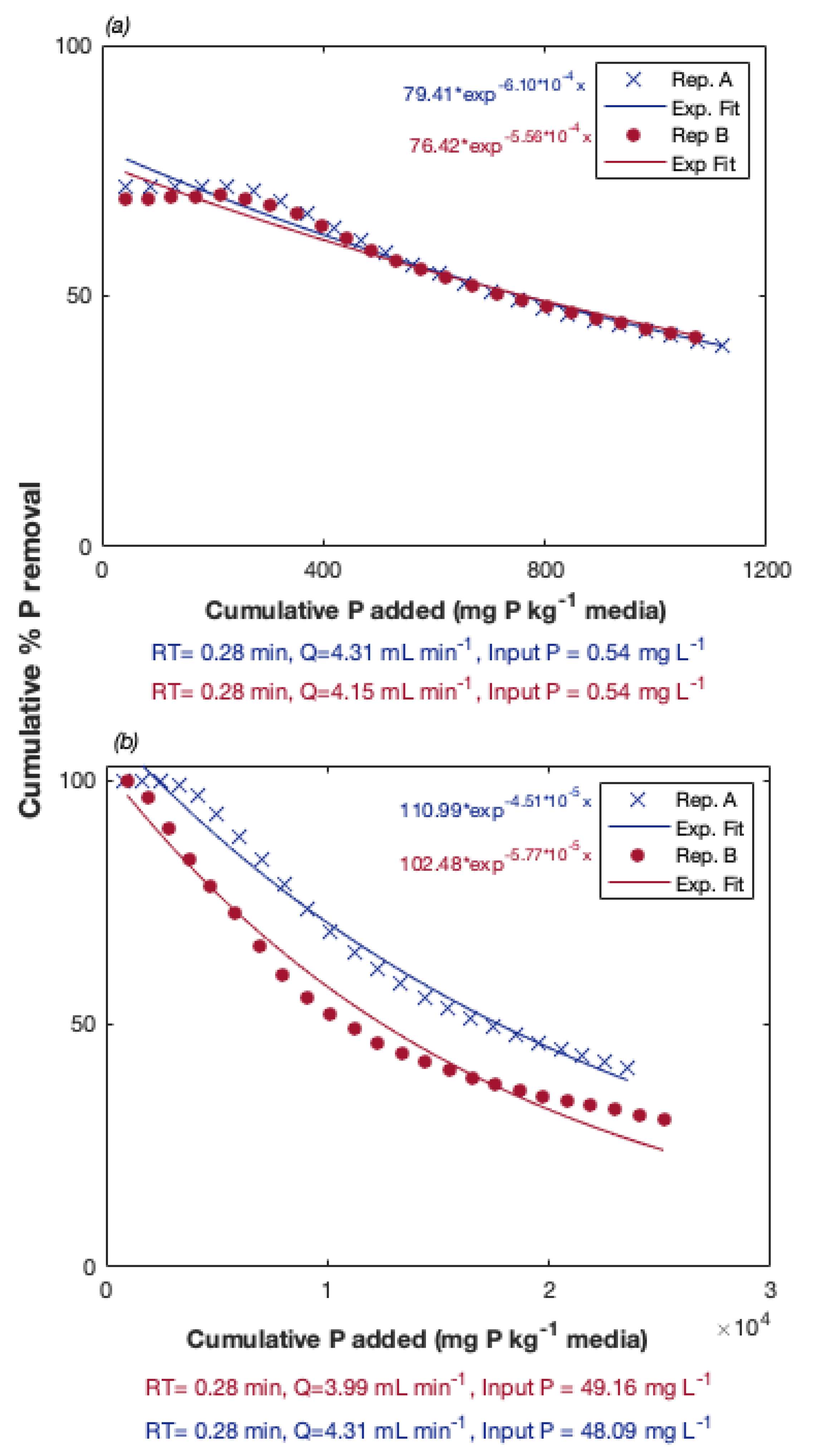
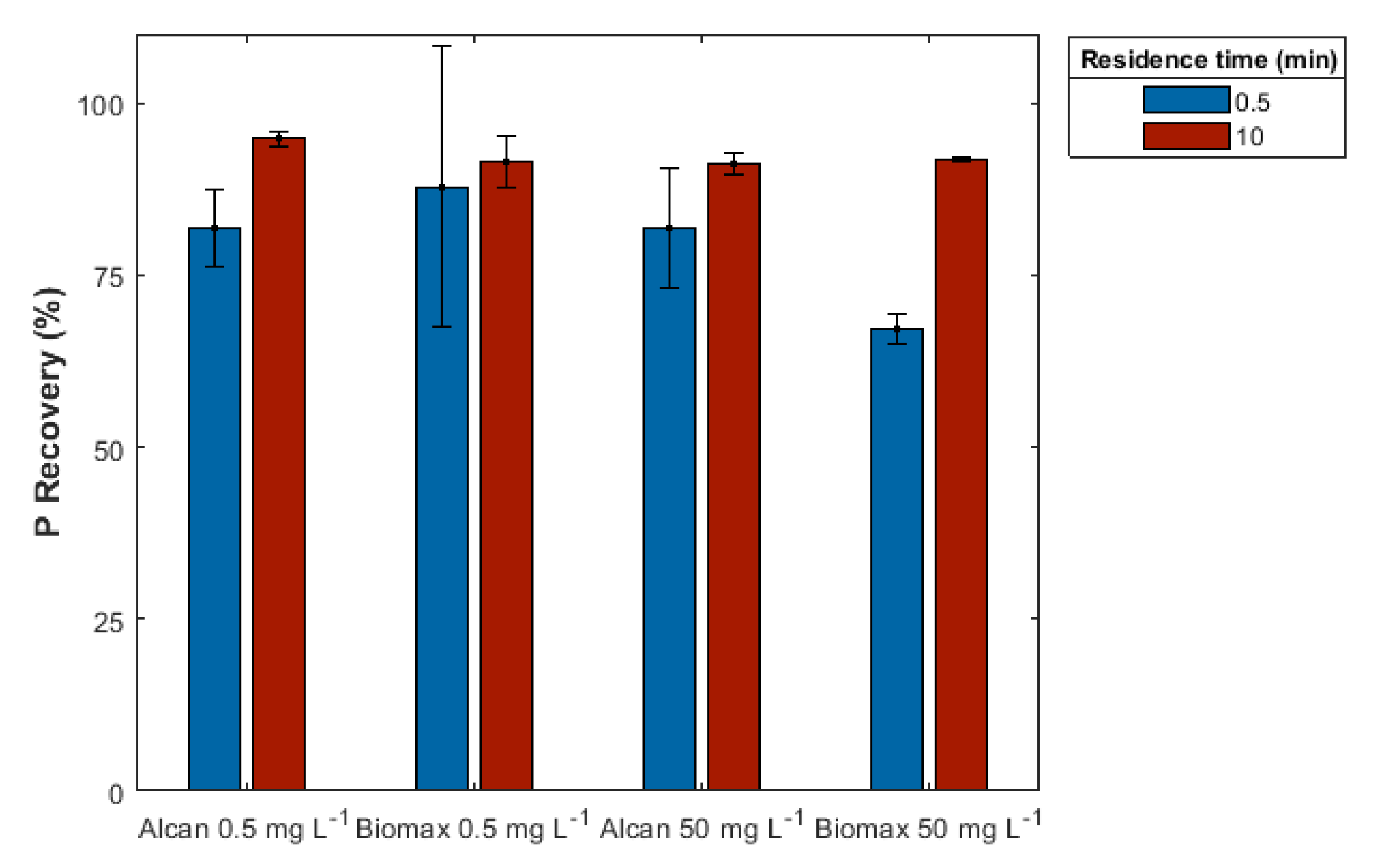
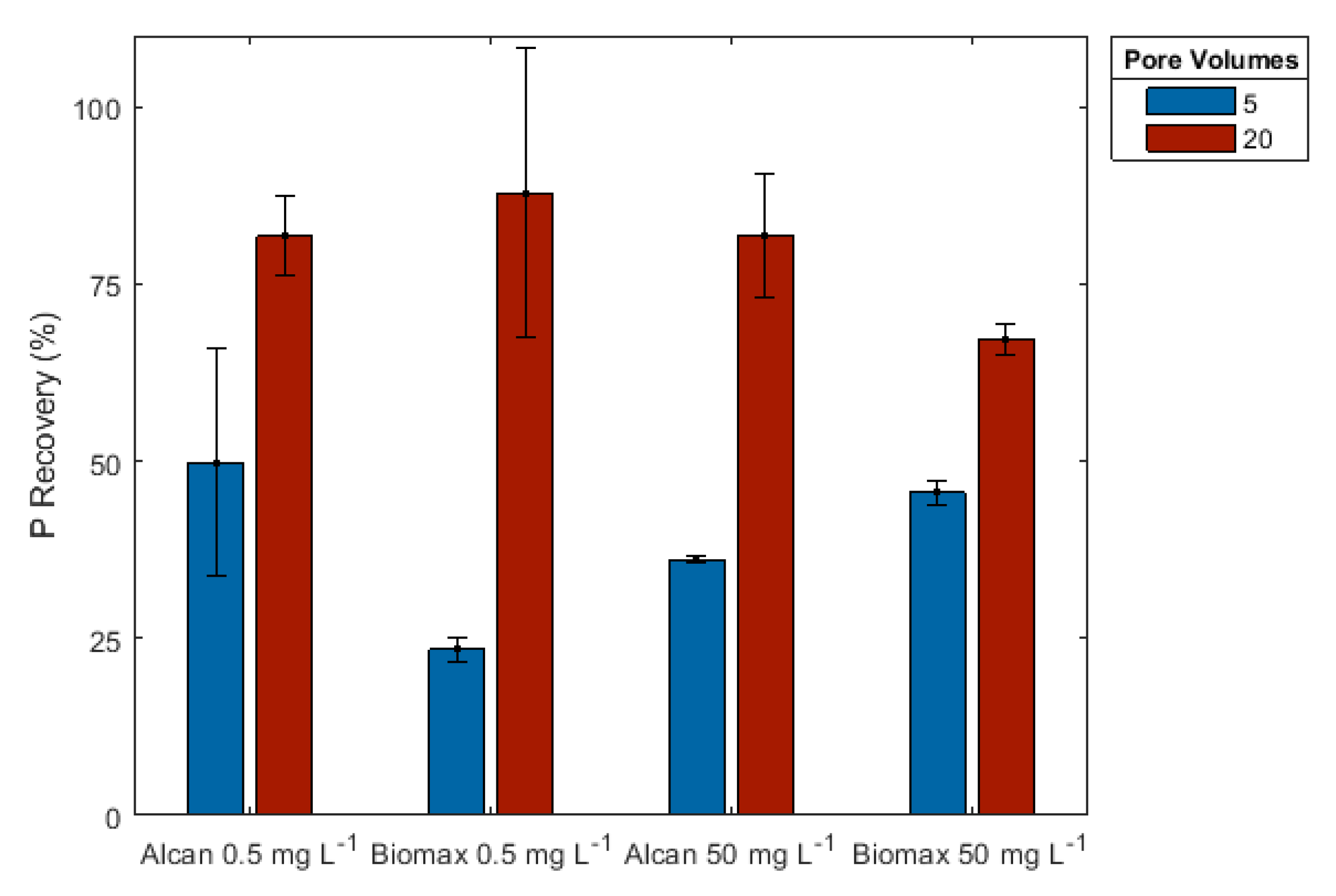
| Sample | Aluminum | Calcium | Iron | Magnesium | pH |
|---|---|---|---|---|---|
| mg kg | |||||
| Alcan | 350,975 | 348 | 20,286 | 0 | 6.49 |
| Biomax | 297,508 | 356 | 56,502 | 0 | 6.85 |
| PhosRedeem | 7567 | 86,726 | 335,646 | 2917 | 11.49 |
| PSM | Sample | Flow-Through Model | P |
|---|---|---|---|
| Alcan 0.5 mg L | A05A | 12,043 | |
| A05B | 16,817 | ||
| AVERAGE | 14,430 | ||
| STD DEV | 3376 | ||
| Biomax 0.5 mg L | B05A | 9469 | |
| B05B | 6193 | ||
| AVERAGE | 7831 | ||
| STD DEV | 2322 | ||
| PhosRedeem 0.5 mg L | P05A | 1123 | |
| P05B | 1164 | ||
| AVERAGE | 1144 | ||
| STD DEV | 16 | ||
| Alcan 50 mg L | A50A | 18,874 | |
| A50B | 16,849 | ||
| AVERAGE | 17,861 | ||
| STD DEV | 1432 | ||
| Biomax 50 mg L | B50A | 19,992 | |
| B50B | 21,231 | ||
| AVERAGE | 20,612 | ||
| STD DEV | 876 | ||
| PhosRedeem 50 mg L | P50A | 22,616 | |
| P50B | 16,308 | ||
| AVERAGE | 18,884 | ||
| STD DEV | 2829 |
| Cycle 0 | SORPTION PHASE | ⇒ | DESORPTION PHASE/REGENERATION |
| Phase S0 | Phase D0 | ||
| Batch sorption isotherm | Flow-through system | ||
| Cycle 1 | SORPTION PHASE | ⇒ | DESORPTION PHASE/REGENERATION |
| Phase S1 | Phase D1 | ||
| Flow-through system | |||
| Cycle 2 | SORPTION PHASE | ||
| Phase S2 | |||
| Flow-through system | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
S. P. C. Scott, I.; J. Penn, C.; Huang, C.-h. Development of a Regeneration Technique for Aluminum-Rich and Iron-Rich Phosphorus Sorption Materials. Water 2020, 12, 1784. https://doi.org/10.3390/w12061784
S. P. C. Scott I, J. Penn C, Huang C-h. Development of a Regeneration Technique for Aluminum-Rich and Iron-Rich Phosphorus Sorption Materials. Water. 2020; 12(6):1784. https://doi.org/10.3390/w12061784
Chicago/Turabian StyleS. P. C. Scott, Isis, Chad J. Penn, and Chi-hua Huang. 2020. "Development of a Regeneration Technique for Aluminum-Rich and Iron-Rich Phosphorus Sorption Materials" Water 12, no. 6: 1784. https://doi.org/10.3390/w12061784
APA StyleS. P. C. Scott, I., J. Penn, C., & Huang, C.-h. (2020). Development of a Regeneration Technique for Aluminum-Rich and Iron-Rich Phosphorus Sorption Materials. Water, 12(6), 1784. https://doi.org/10.3390/w12061784






