Drivers of Benthic Macroinvertebrate Assemblages in Equatorial Alpine Rivers of the Rwenzoris (Uganda)
Abstract
1. Introduction
2. Materials and Methods
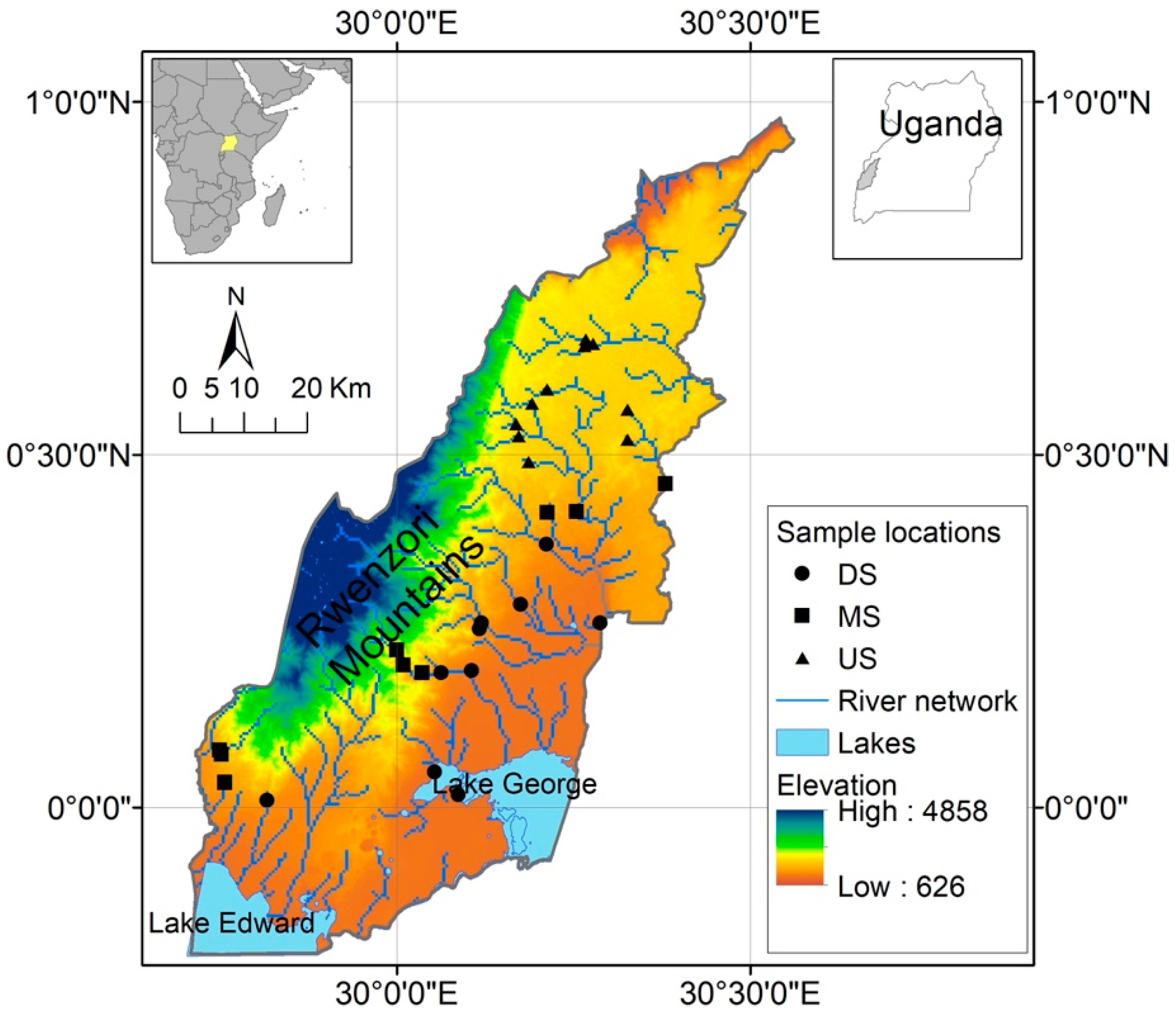
2.1. Study Area
Site Selection
2.2. Collection of Environmental Data
2.3. Aquatic Macroinvertebrate Sampling and Identification
2.4. Data Analysis
3. Results
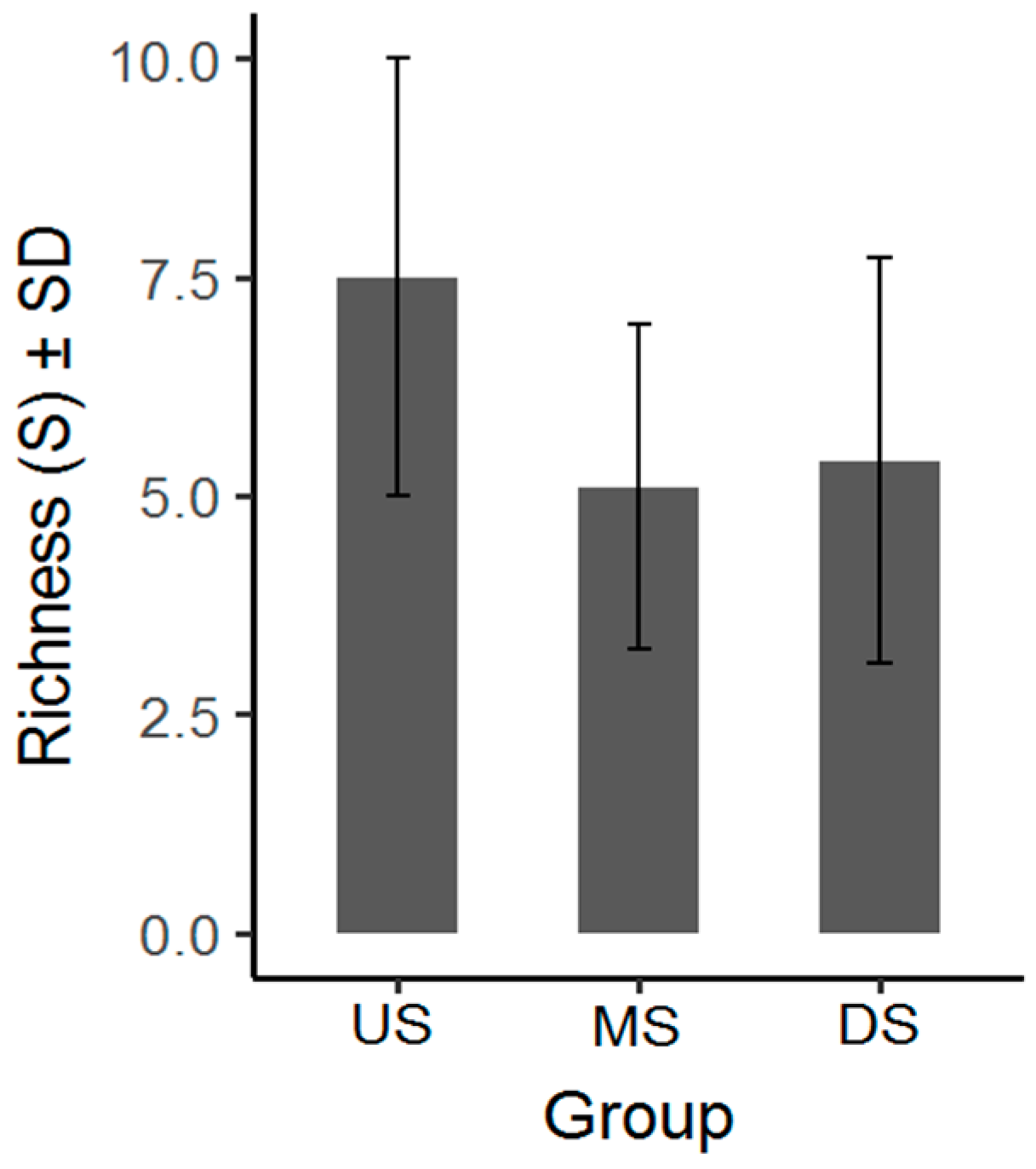
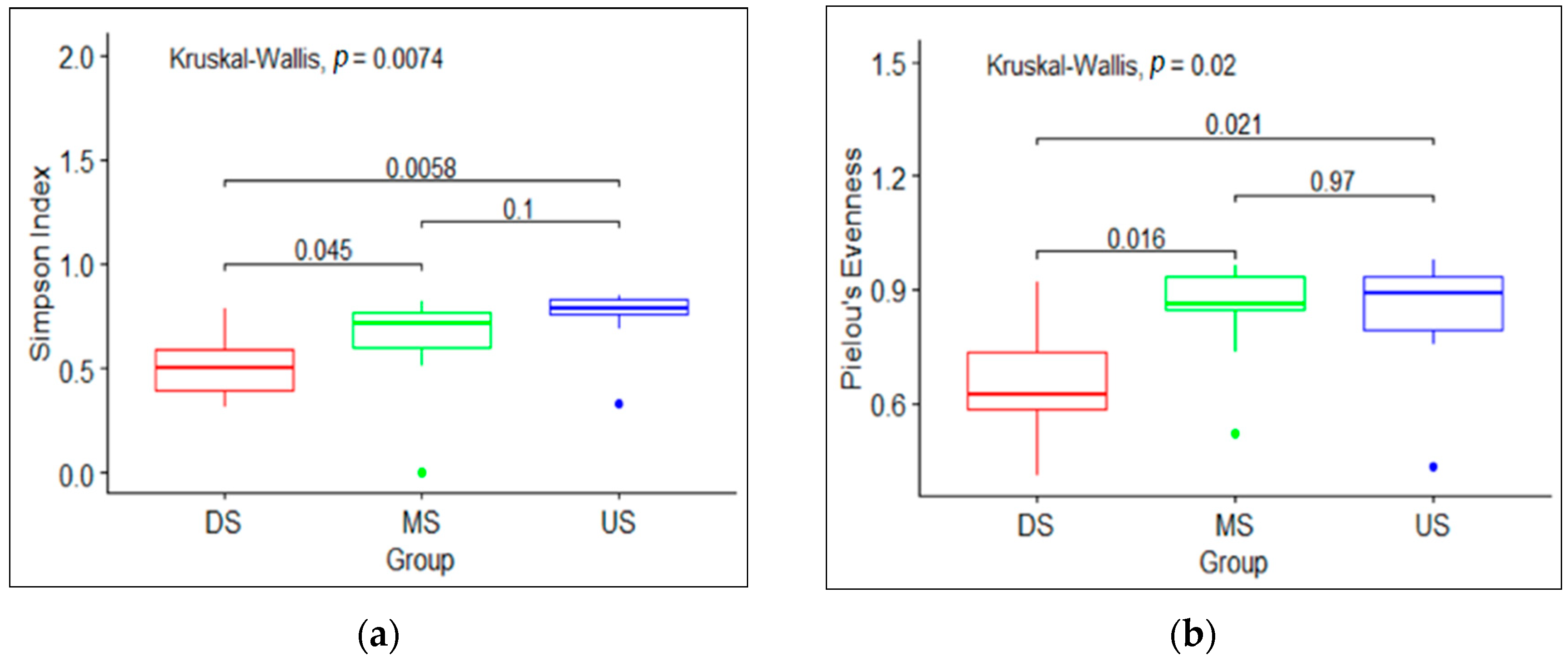
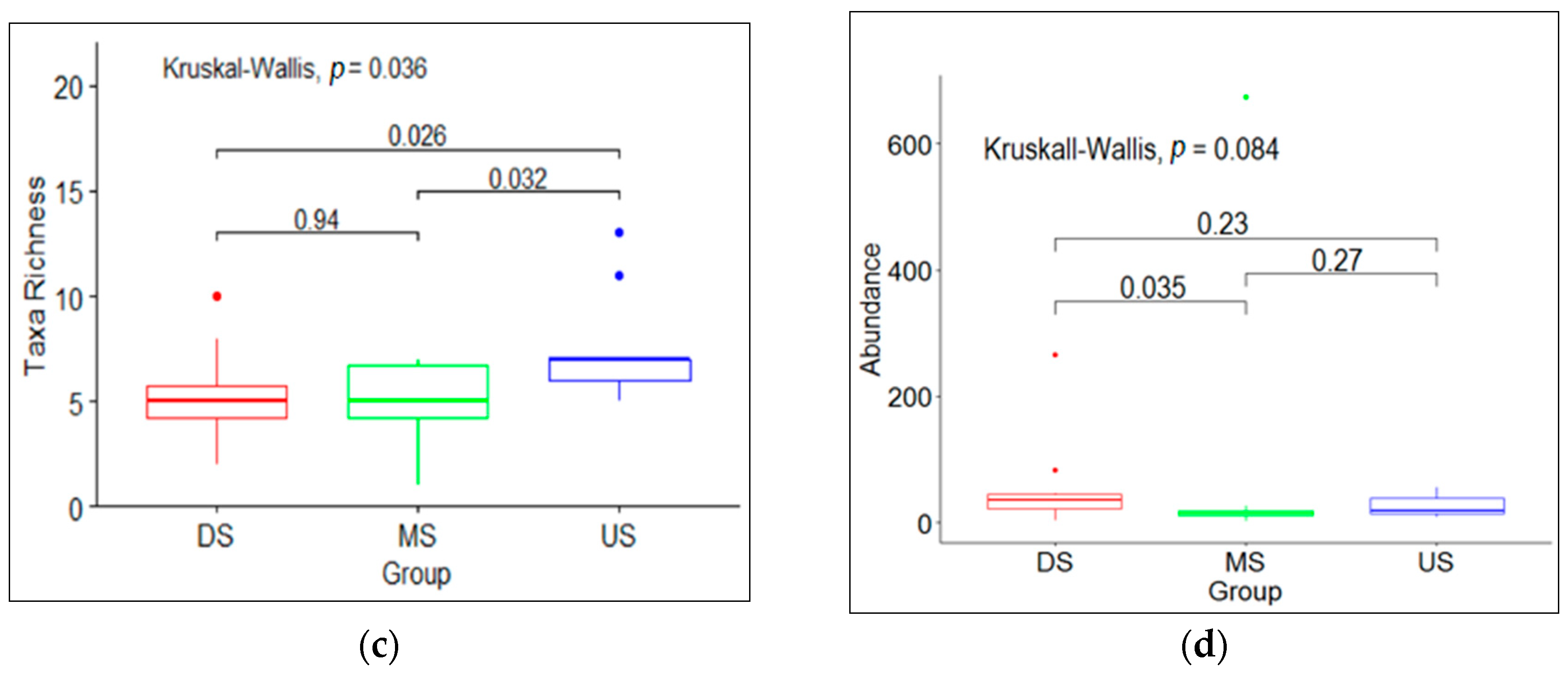
3.1. Macroinvertebrate Community Diversity
3.2. Indicator Taxa Analysis
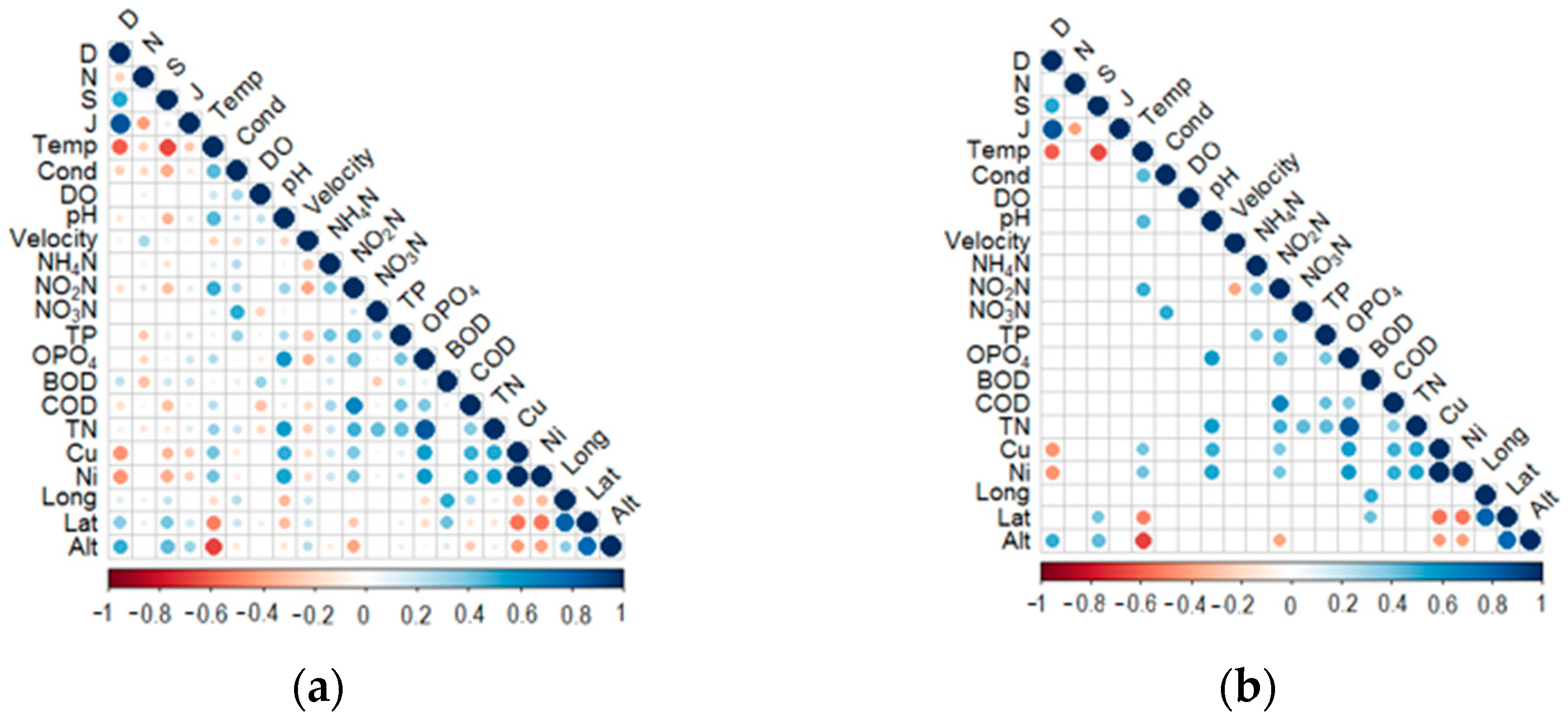
3.3. Relationships between Diversity Metrics and Environmental Variables
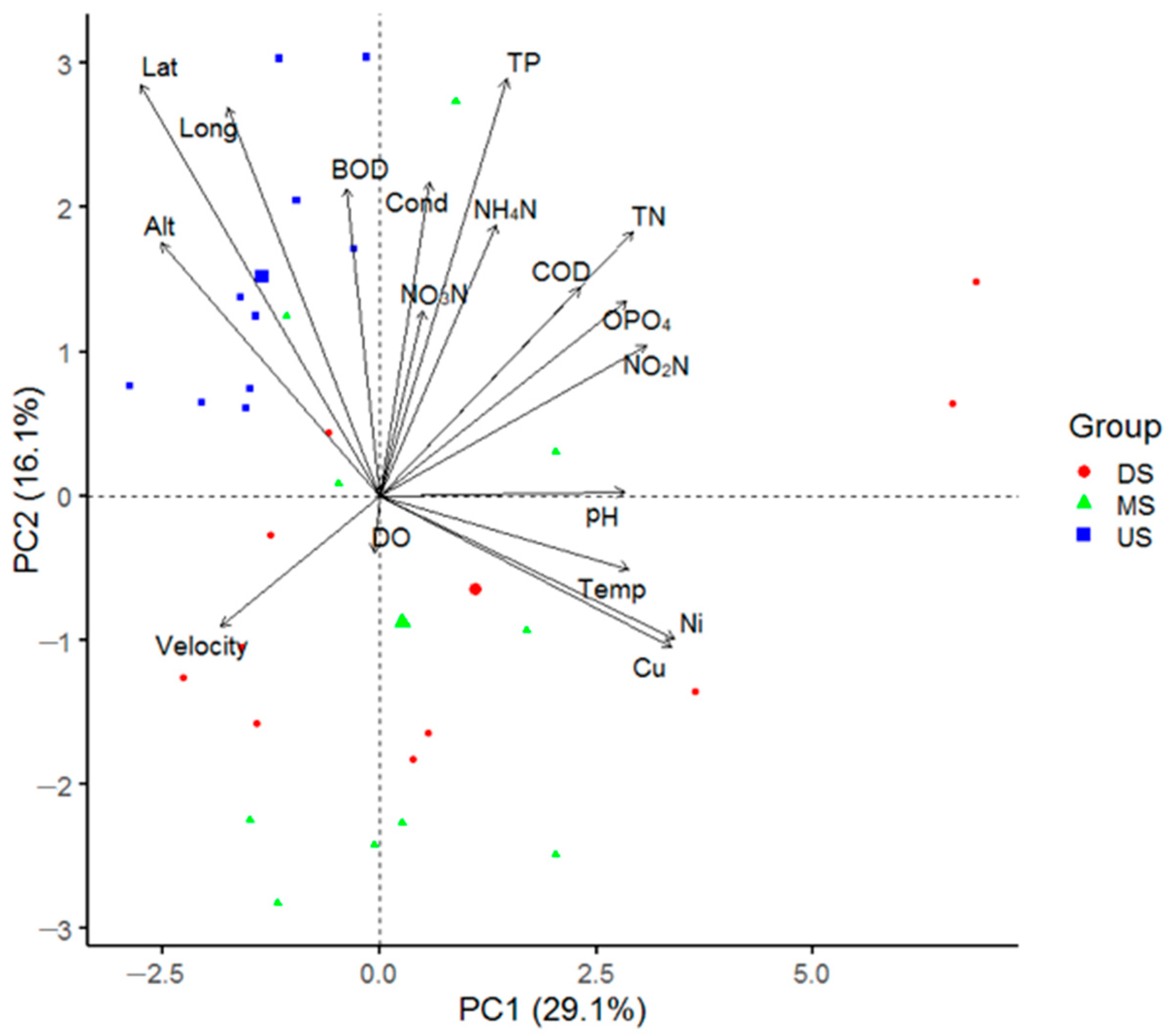
3.4. Principle Component Analysis (PCA) Ordination
3.5. Selection of Variables (Predictors) for Macroinvertebrate Community Diversity
4. Discussion
4.1. Influence of Geographical Variables on Macroinvertebrate Diversity
4.2. Indicator Families and Macroinvertebrate Diversity
4.3. Physicochemical Variables Influence on Macroinvertebrate Diversity
4.4. Future Research and Implementation of Insights in Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wittebolle, L.; Marzorati, M.; Clement, L.; Balloi, A.; Daffonchio, D.; Heylen, K.; De Vos, P.; Verstraete, W.; Boon, N. Initial community evenness favours functionality under selective stress. Nature 2009, 458, 623. [Google Scholar] [CrossRef] [PubMed]
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Liermann, C.R. Global threats to human water security and river biodiversity. Nature 2010, 467, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Collen, B.; Whitton, F.; Dyer, E.E.; Baillie, J.E.M.; Cumberlidge, N.; Darwall, W.R.T.; Pollock, C.; Richman, N.I.; Soulsby, A.M.; Böhm, M. Global patterns of freshwater species diversity, threat and endemism. Glob. Ecol. Biogeogr. 2014, 23, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Omann, I.; Stocker, A.; Jäger, J. Climate change as a threat to biodiversity: An application of the dpsir approach. Ecol. Econ. 2009, 69, 24–31. [Google Scholar] [CrossRef]
- Midgley, G.; Hannah, L.; Millar, D.; Rutherford, M.; Powrie, L. Assessing the vulnerability of species richness to anthropogenic climate change in a biodiversity hotspot. Glob. Ecol. Biogeogr. 2002, 11, 445–451. [Google Scholar] [CrossRef]
- Plumptre, A.J.; Davenport, T.R.; Behangana, M.; Kityo, R.; Eilu, G.; Ssegawa, P.; Ewango, C.; Meirte, D.; Kahindo, C.; Herremans, M. The biodiversity of the albertine rift. Biol. Conserv. 2007, 134, 178–194. [Google Scholar] [CrossRef]
- Burgess, N.D.; Balmford, A.; Cordeiro, N.J.; Fjeldså, J.; Küper, W.; Rahbek, C.; Sanderson, E.W.; Scharlemann, J.P.; Sommer, J.H.; Williams, P.H. Correlations among species distributions, human density and human infrastructure across the high biodiversity tropical mountains of africa. Biol. Conserv. 2007, 134, 164–177. [Google Scholar] [CrossRef]
- Aynekulu, E.; Aerts, R.; Moonen, P.; Denich, M.; Gebrehiwot, K.; Vågen, T.-G.; Mekuria, W.; Boehmer, H.J. Altitudinal variation and conservation priorities of vegetation along the great rift valley escarpment, northern ethiopia. Biodivers. Conserv. 2012, 21, 2691–2707. [Google Scholar] [CrossRef]
- Roller, S.; Hornung, J.; Hinderer, M.; Ssemmanda, I. Middle miocene to pleistocene sedimentary record of rift evolution in the southern albert rift (uganda). Int. J. Earth Sci. 2010, 99, 1643–1661. [Google Scholar] [CrossRef]
- Xue, L.; Gani, N.D.; Abdelsalam, M.G. Geomorphologic proxies for bedrock rivers: A case study from the rwenzori mountains, east african rift system. Geomorphology 2017, 285, 374–398. [Google Scholar] [CrossRef]
- Musonge, P.S.; Boets, P.; Lock, K.; Ambarita, N.M.D.; Forio, M.A.E.; Verschuren, D.; Goethals, P.L. Baseline assessment of benthic macroinvertebrate community structure and ecological water quality in rwenzori rivers (albertine rift valley, uganda) using biotic-index tools. Limnologica 2019, 75, 1–10. [Google Scholar] [CrossRef]
- Orlove, B.; Wiegandt, E.; Luckman, B.H. The Place of Glaciers in Natural and Cultural Landscapes; University of California Press: Berkeley, CA, USA, 2008; pp. 3–19. [Google Scholar]
- Olson, D.M.; Dinerstein, E. The global 200: Priority ecoregions for global conservation. Ann. Mo. Bot. Gard. 2002, 89, 199–224. [Google Scholar] [CrossRef]
- Russell, J.M.; Johnson, T.C.; Kelts, K.R.; Lærdal, T.; Talbot, M.R. An 11 000-year lithostratigraphic and paleohydrologic record from equatorial africa: Lake edward, uganda–congo. Palaeogeogr. Palaeoclim. Palaeoecol. 2003, 193, 25–49. [Google Scholar] [CrossRef]
- Eggermont, H.; Van Damme, K.; Russell, J.M. Rwenzori mountains (mountains of the moon): Headwaters of the white nile. In The Nile; Springer: Berlin/Heidelberg, Germany, 2009; pp. 243–261. [Google Scholar]
- Natamba, L.; Weihua, Z.; Sissoko, F. Fertilizer application and non-point source pollution problems between Uganda and China-review. Adv. Soc. Sci. Res. J. 2016, 3, 133–146. [Google Scholar]
- Abraham, M.R.; Susan, T.B. Water contamination with heavy metals and trace elements from kilembe copper mine and tailing sites in western uganda; implications for domestic water quality. Chemosphere 2017, 169, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Darwall, W.; Smith, K.; Allen, D.; Holland, R.; Harrison, I.; Brooks, E. The Diversity of Life in African Freshwaters: Underwater, Under Threat: An Analysis of the Status and Distribution of Freshwater Species Throughout Mainland Africa; The World Conservation Union (IUCN): Gland, Switzerland, 2011; Available online: http://cmsdata.iucn.org/downloads/iucn_africa_freshwaters_2011.pdf (accessed on 10 June 2020).
- Edwards, D.P.; Sloan, S.; Weng, L.; Dirks, P.; Sayer, J.; Laurance, W.F. Mining and the african environment. Conserv. Lett. 2014, 7, 302–311. [Google Scholar] [CrossRef]
- Smith, H. The Overlap between Conservation and Development Organisations in the Albertine Rift, Western Uganda; The Poverty and Conservation Learning Group Discussion Paper; International Institute for Environment and Development (IIED): London, UK, 2012. [Google Scholar]
- Tumusiime, D.M.; Mawejje, J.; Byakagaba, P. Discovery of oil: Community perceptions and expectations in uganda’s albertine region. J. Sustain. Dev. 2016, 9, 1. [Google Scholar] [CrossRef]
- Muwanga, A. Environmental Impacts of Copper Mining at Kilembe, Uganda: A Geochemical Investigation of Heavy Metal Pollution of Drainage Waters, Stream Sediments, and Soils in the Kilembe Valley in Relation to Mine Waste Disposal; Inst. für Geowiss. der Techn. Univ: Braunschweig, Germany, 1997. [Google Scholar]
- Denny, P.; Bailey, R.; Tukahirwa, E.; Mafabi, P. Heavy metal contamination of lake george (uganda) and its wetlands. Hydrobiologia 1995, 297, 229–239. [Google Scholar] [CrossRef]
- Hare, L. Aquatic insects and trace metals: Bioavailability, bioaccumulation, and toxicity. Crit. Rev. Toxicol. 1992, 22, 327–369. [Google Scholar] [CrossRef] [PubMed]
- Goretti, E.; Pallottini, M.; Pagliarini, S.; Catasti, M.; La Porta, G.; Selvaggi, R.; Gaino, E.; Di Giulio, A.M.; Ali, A. Use of larval morphological deformities in chironomus plumosus (chironomidae: Diptera) as an indicator of freshwater environmental contamination (Lake Trasimeno, Italy). Water 2020, 12, 1. [Google Scholar] [CrossRef]
- Bozanic, M.; Markovic, Z.; Zivic, M.; Dojčinović, B.; Perić, A.; Stankovic, M.; Zivic, I. Mouthpart deformities of chironomus plumosus larvae caused by increased concentrations of copper in sediment from carp fish pond. Turk. J. Fish. Aquat. Sci. 2019, 19, 251–259. [Google Scholar] [CrossRef]
- McClanahan, Y.; McClanahan, S.C.Z.T. East. African Ecosystems and Their Conservation; Oxford University Press: New York, NY, USA, 1996. [Google Scholar]
- Taylor, R.G.; Mileham, L.; Tindimugaya, C.; Majugu, A.; Muwanga, A.; Nakileza, B. Recent glacial recession in the rwenzori mountains of east africa due to rising air temperature. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Mölg, T.; Georges, C.; Kaser, G. The contribution of increased incoming shortwave radiation to the retreat of the rwenzori glaciers, east africa, during the 20th century. Int. J. Clim. 2003, 23, 291–303. [Google Scholar] [CrossRef]
- Taylor, R.G.; Mileham, L.; Tindimugaya, C.; Mwebembezi, L. Recent glacial recession and its impact on alpine riverflow in the rwenzori mountains of uganda. J. Afr. Earth Sci. 2009, 55, 205–213. [Google Scholar] [CrossRef]
- Maiolini, B.; Lencioni, V. Longitudinal distribution of macroinvertebrate assemblages in a glacially influenced stream system in the italian alps. Freshw. Biol. 2001, 46, 1625–1639. [Google Scholar] [CrossRef]
- Brown, L.E.; Hannah, D.M.; Milner, A.M. Vulnerability of alpine stream biodiversity to shrinking glaciers and snowpacks. Glob. Chang. Biol. 2007, 13, 958–966. [Google Scholar] [CrossRef]
- Milner, A.M.; Brown, L.E.; Hannah, D.M. Hydroecological response of river systems to shrinking glaciers. Hydrol. Process. 2009, 23, 62–77. [Google Scholar] [CrossRef]
- Finn, D.S.; Raesaenen, K.; Robinson, C.T. Physical and biological changes to a lengthening stream gradient following a decade of rapid glacial recession. Glob. Chang. Biol. 2010, 16, 3314–3326. [Google Scholar] [CrossRef]
- Dallas, H.F.; Rivers-Moore, N.A. Critical thermal maxima of aquatic macroinvertebrates: Towards identifying bioindicators of thermal alteration. Hydrobiologia 2012, 679, 61–76. [Google Scholar] [CrossRef]
- Stewart, B.A.; Close, P.G.; Cook, P.A.; Davies, P.M. Upper thermal tolerances of key taxonomic groups of stream invertebrates. Hydrobiologia 2013, 718, 131–140. [Google Scholar] [CrossRef]
- Cadbury, S.; Hannah, D.; Milner, A.; Pearson, C.; Brown, L. Stream temperature dynamics within a new zealand glacierized river basin. River Res. Appl. 2008, 24, 68–89. [Google Scholar] [CrossRef]
- Scotti, A.; Jacobsen, D.; Tappeiner, U.; Bottarin, R. Spatial and temporal variation of benthic macroinvertebrate assemblages during the glacial melt season in an italian glacier-fed stream. Hydrobiologia 2019, 827, 123–139. [Google Scholar] [CrossRef]
- Docherty, C.; Hannah, D.; Riis, T.; Leth, S.R.; Milner, A.M. Longitudinal distribution of macroinvertebrates in snowmelt streams in northeast greenland: Understanding biophysical controls. Polar Biol. 2018, 41, 1567–1580. [Google Scholar] [CrossRef] [PubMed]
- Kock Laursen, S.; Hamerlik, L.; Moltesen, K.; Seestern Christoffersen, K.; Jacobsen, D. Diversity and composition of macroinvertebrate assemblages in high-altitude tibetan streams. Inland Waters 2015, 5, 263–274. [Google Scholar] [CrossRef]
- Cauvy-Fraunié, S.; Espinosa, R.; Andino, P.; Jacobsen, D.; Dangles, O. Invertebrate metacommunity structure and dynamics in an andean glacial stream network facing climate change. PLoS ONE 2015, 10, e0136793. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, J.; Andino, P.; Calvez, R.; Espinosa, R.; Hamerlik, L.; Vie, S.; Dangles, O.; Jacobsen, D. Spatial variability in macroinvertebrate assemblages along and among neighbouring equatorial glacier-fed streams. Freshw. Biol. 2011, 56, 2226–2244. [Google Scholar] [CrossRef]
- Snook, D.L.; Milner, A.M. The influence of glacial runoff on stream macroinvertebrate communities in the taillon catchment, french pyrénées. Freshw. Biol. 2001, 46, 1609–1623. [Google Scholar] [CrossRef]
- Milner, A.M.; Brittain, J.E.; Castella, E.; Petts, G.E. Trends of macroinvertebrate community structure in glacier-fed rivers in relation to environmental conditions: A synthesis. Freshw. Biol. 2001, 46, 1833–1847. [Google Scholar] [CrossRef]
- Brown, L.E.; Milner, A.M.; Hannah, D.M. Groundwater influence on alpine stream ecosystems. Freshw. Biol. 2007, 52, 878–890. [Google Scholar] [CrossRef]
- Fjellheim, A.; Raddum, G.G. Growth and voltinism in the aquatic insects of a regulated river subject to groundwater inflows. River Res. Appl. 2008, 24, 710–719. [Google Scholar] [CrossRef]
- Jacobsen, D.; Cauvy-Fraunie, S.; Andino, P.; Espinosa, R.; Cueva, D.; Dangles, O. Runoff and the longitudinal distribution of macroinvertebrates in a glacier-fed stream: Implications for the effects of global warming. Freshw. Biol. 2014, 59, 2038–2050. [Google Scholar] [CrossRef]
- Jacobsen, D.; Dangles, O.; Andino, P.; Espinosa, R.; Hamerlik, L.; Cadier, E. Longitudinal zonation of macroinvertebrates in an ecuadorian glacier-fed stream: Do tropical glacial systems fit the temperate model? Freshw. Biol. 2010, 55, 1234–1248. [Google Scholar] [CrossRef]
- Fox, J.F. Intermediate-disturbance hypothesis. Science 1979, 204, 1344–1345. [Google Scholar] [CrossRef] [PubMed]
- Townsend, C.R.; Scarsbrook, M.R.; Dolédec, S. The intermediate disturbance hypothesis, refugia, and biodiversity in streams. Limnol. Oceanogr. 1997, 42, 938–949. [Google Scholar] [CrossRef]
- Death, R. The effect of habitat stability on benthic invertebrate communities: The utility of species abundance distributions. Hydrobiologia 1996, 317, 97–107. [Google Scholar] [CrossRef]
- Winterbourn, M. New zealand mountain stream communities: Stable yet disturbed? In Evolutionary Ecology of Freshwater Animals; Springer: Berlin/Heidelberg, Germany, 1997; pp. 31–54. [Google Scholar]
- Richards, C.; Johnson, L.B.; Host, G. Landscape Influences on Habitat, Water Chemistry, and Macroinvertebrate Assemblages In Midwestern Stream Ecosystems; University of Minnesota Duluth: Duluth, MN, USA, 1993. [Google Scholar]
- Aghaindum, A.G.; Achuo, E.D.; Menbohan, F. Chironomid bio-ecology in an anthropisized aquatic ecosystem in yaounde (cameroon): Community diversity and relationship with the environmental variables of the water system. Entomol. Appl. Sci. Lett. 2016, 3, 196–203. [Google Scholar]
- Dalu, T.; Wasserman, R.J.; Tonkin, J.D.; Alexander, M.E.; Dalu, M.T.; Motitsoe, S.N.; Manungo, K.I.; Bepe, O.; Dube, T. Assessing drivers of benthic macroinvertebrate community structure in african highland streams: An exploration using multivariate analysis. Sci. Total Environ. 2017, 601, 1340–1348. [Google Scholar] [CrossRef]
- Múrria, C.; Bonada, N.; Vellend, M.; Zamora-Muñoz, C.; Alba-Tercedor, J.; Sainz-Cantero, C.E.; Garrido, J.; Acosta, R.; El Alami, M.; Barquín, J. Local environment rather than past climate determines community composition of mountain stream macroinvertebrates across europe. Mol. Ecol. 2017, 26, 6085–6099. [Google Scholar] [CrossRef]
- Nautiyal, P.; Mishra, A.S.; Verma, J.; Agrawal, A. River ecosystems of the central highland ecoregion: Spatial distribution of benthic flora and fauna in the plateau rivers (tributaries of the yamuna and ganga) in central india. Aquat. Ecosyst. Health Manag. 2017, 20, 43–58. [Google Scholar] [CrossRef]
- Windsor, F.; Grocott, M.; Milner, A. An inter-catchment assessment of macroinvertebrate communities across groundwater-fed streams within denali national park, interior alaska. Hydrobiologia 2017, 785, 373–384. [Google Scholar] [CrossRef]
- Kasangaki, A.; Chapman, L.J.; Balirwa, J. Land use and the ecology of benthic macroinvertebrate assemblages of high-altitude rainforest streams in uganda. Freshw. Biol. 2008, 53, 681–697. [Google Scholar] [CrossRef]
- Kasangaki, A.; Babaasa, D.; Efitre, J.; McNeilage, A.; Bitariho, R. Links between anthropogenic perturbations and benthic macroinvertebrate assemblages in afromontane forest streams in Uganda. Hydrobiologia 2006, 563, 231–245. [Google Scholar] [CrossRef]
- Pabire Ghandi, W.; Ndawula, L. The Diversity of Macro Invertebrates in the Victoria and Kyoga Lake Basin and Their Relationship to Ecosystem Functioning; National Fisheries Resources Research Institute: Jinja, Uganda, 2000; Available online: http://aquaticcommons.org/id/eprint/20234 (accessed on 10 June 2020).
- Salt, G. Insects and other invertebrate animals collected at high altitudes in the ruwenzori and on mount Kenya. Afr. J. Ecol. 1987, 25, 95–106. [Google Scholar] [CrossRef]
- Halls, A. Wetlands, Biodiversity and the Ramsar Convention: The Role of the Convention on Wetlands in the Conservation and Wise use of Biodiversity; Ramsar Convention Bureau: Gland, Switzerland, 1997. [Google Scholar]
- BakamaNume, B.B. A Contemporary Geography of Uganda. In African Books Collective; Mukuku na Nyota Publishers Ltd.: Dare salaam, Tanzania, 2010. [Google Scholar]
- Diem, J.E.; Hartter, J.; Ryan, S.J.; Palace, M.W. Validation of satellite rainfall products for western Uganda. J. Hydrometeorol. 2014, 15, 2030–2038. [Google Scholar] [CrossRef]
- Mabidi, A.; Bird, M.S.; Perissinotto, R. Distribution and diversity of aquatic macroinvertebrate assemblages in a semi-arid region earmarked for shale gas exploration (eastern cape karoo, South Africa). PLoS ONE 2017, 12, e0178559. [Google Scholar] [CrossRef] [PubMed]
- Lencioni, V.; Marziali, L.; Rossaro, B. Chironomids as bioindicators of environmental quality in mountain springs. Freshw. Sci. 2012, 31, 525–541. [Google Scholar] [CrossRef]
- Chapman, D.V. Water Quality Assessments: A Guide to the use of Biota, Sediments, and Water in Environmental Monitoring, 2nd ed.; Published on behalf of UNESCO; WHO and UNEP by Chapman and Hall: London, UK, 1996. [Google Scholar]
- Metzeling, L.; Chessman, B.; Hardwick, R.; Wong, V. Rapid assessment of rivers using macroinvertebrates: The role of experience, and comparisons with quantitative methods. Hydrobiologia 2003, 510, 39–52. [Google Scholar] [CrossRef]
- Lock, K.; Adriaens, T.; Goethals, P. Effect of water quality on blackflies (diptera: Simuliidae) in flanders (Belgium). Limnol. Ecol. Manag. Inland Waters 2014, 44, 58–65. [Google Scholar] [CrossRef]
- Damanik-Ambarita, M.N.; Everaert, G.; Forio, M.A.E.; Nguyen, T.H.T.; Lock, K.; Musonge, P.L.S.; Suhareva, N.; Dominguez-Granda, L.; Bennetsen, E.; Boets, P. Generalized linear models to identify key hydromorphological and chemical variables determining the occurrence of macroinvertebrates in the guayas river basin (Ecuador). Water 2016, 8, 297. [Google Scholar] [CrossRef]
- Gabriels, W.; Lock, K.; De Pauw, N.; Goethals, P.L.M. Multimetric macroinvertebrate index flanders (MMIF) for biological assessment of rivers and lakes in flanders (Belgium). Limnol. Ecol. Manag. Inland Waters 2010, 40, 199–207. [Google Scholar] [CrossRef]
- Nguyen, H.H.; Everaert, G.; Gabriels, W.; Hoang, T.H.; Goethals, P.L.M. A multimetric macroinvertebrate index for assessing the water quality of the cau river basin in Vietnam. Limnol. Ecol. Manag. Inland Waters 2014, 45, 16–23. [Google Scholar] [CrossRef]
- Ambelu, A.; Mekonen, S.; Koch, M.; Addis, T.; Boets, P.; Everaert, G.; Goethals, P. The application of predictive modelling for determining bio-environmental factors affecting the distribution of blackflies (diptera: Simuliidae) in the gilgel gibe watershed in southwest Ethiopia. PLoS ONE 2014, 9, e112221. [Google Scholar] [CrossRef] [PubMed]
- Gerber, A.; Gabriel, M.J.M. Aquatic Invertebrates of South African Rivers Field Guide; Resource Water Quality Services, Department of Water Affairs and Forestry: Pretoria, South Africa, 2002. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; ISBN 3-900051-07-0. Available online: https://www.R-project.org (accessed on 10 June 2020).
- Yang, G.; He, D.; Wang, C.; Miao, Y.; Yu, H. Study on the biological oceanography characteristics of planktonic copepods in the waters north of taiwan, ii. Community characteristics. Acta Oceanol. Sin. 1999, 21, 72–80. [Google Scholar]
- De Cáceres, M.; Legendre, P.; Moretti, M. Improving indicator species analysis by combining groups of sites. Oikos 2010, 119, 1674–1684. [Google Scholar] [CrossRef]
- Hawksworth, D.L.; Bull, A.T. Marine, Freshwater, and Wetlands Biodiversity Conservation; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; Volume 4. [Google Scholar]
- Dufrêne, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- Cáceres, M.D.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef] [PubMed]
- Kruskal, W.H.; Wallis, W.A. Use of ranks in one-criterion variance analysis. J. Am. Stat. Assoc. 1952, 47, 583–621. [Google Scholar] [CrossRef]
- Husson, F.; Josse, J.; Le, S.; Mazet, J.; Husson, M.F. Package ‘factominer’. Obtenido de Multivariate Exploratory Data Analysis and Data Mining. 2018. Available online: http://cran/.r-project.org/web/packages/FactoMineR/FactoMineR (accessed on 10 June 2020).
- Friedman, J.; Hastie, T.; Tibshirani, R. Glmnet: Lasso and Elastic-Net Regularized Generalized Linear Models; R Package Version; Department of Statistics, Stanford University: San Francisco, CA, USA, 2009; Volume 1. [Google Scholar]
- Tibshirani, R. Regression shrinkage and selection via the lasso: A retrospective. J. R. Stat. Soc. Ser. B (Stat. Methodol.) 2011, 73, 273–282. [Google Scholar] [CrossRef]
- Tibshirani, R. Regression shrinkage and selection via the lasso. J. R. Stat. Soc. Ser. B (Methodol.) 1996, 58, 267–288. [Google Scholar] [CrossRef]
- Henriques-Oliveira, A.L.; Nessimian, J.L. Aquatic macroinvertebrate diversity and composition in streams along an altitudinal gradient in southeastern brazil. Biota Neotrop. 2010, 10, 115–128. [Google Scholar] [CrossRef]
- Xu, M.; Wang, Z.; Pan, B.; Yu, G. The assemblage characteristics of benthic macroinvertebrates in the yalutsangpo river, the highest major river in the world. Front. Earth Sci. 2014, 8, 351–361. [Google Scholar] [CrossRef]
- Fjellheim, A.; Raddum, G.G.; Vandvik, V.; Cogalniceanu, D.; Boggero, A.; Brancelj, A.; Galas, J.; Šporka, F.; Vidinova, Y.; Bitušík, P. Diversity and distribution patterns of benthic invertebrates along alpine gradients. A study of remote european freshwater lakes. Adv. Limnol. 2009, 62, 167–190. [Google Scholar]
- Li, J.; Herlihy, A.; Gerth, W.; Kaufmann, P.; Gregory, S.; Urquhart, S.; Larsen, D.P. Variability in stream macroinvertebrates at multiple spatial scales. Freshw. Biol. 2001, 46, 87–97. [Google Scholar] [CrossRef]
- Passy, S.I.; Bode, R.W.; Carlson, D.M.; Novak, M.A. Comparative environmental assessment in the studies of benthic diatom, macroinvertebrate, and fish communities. Int. Rev. Hydrobiol. A J. Cover. All Asp. Limnol. Mar. Biol. 2004, 89, 121–138. [Google Scholar] [CrossRef]
- Mykrä, H.; Heino, J.; Muotka, T. Scale-related patterns in the spatial and environmental components of stream macroinvertebrate assemblage variation. Glob. Ecol. Biogeogr. 2007, 16, 149–159. [Google Scholar] [CrossRef]
- Heino, J.; Muotka, T.; Paavola, R.; Hämäläinen, H.; Koskenniemi, E. Correspondence between regional delineations and spatial patterns in macroinvertebrate assemblages of boreal headwater streams. J. N. Am. Benthol. Soc. 2002, 21, 397–413. [Google Scholar] [CrossRef]
- Buytaert, W.; Cuesta-Camacho, F.; Tobón, C. Potential impacts of climate change on the environmental services of humid tropical alpine regions. Glob. Ecol. Biogeogr. 2011, 20, 19–33. [Google Scholar] [CrossRef]
- Schneider, S.; Hornung, J.; Hinderer, M.; Garzanti, E. Petrography and geochemistry of modern river sediments in an equatorial environment (rwenzori mountains and albertine rift, Uganda)—Implications for weathering and provenance. Sediment. Geol. 2016, 336, 106–119. [Google Scholar] [CrossRef]
- Jacobsen, D.; Schultz, R.; Encalada, A. Structure and diversity of stream invertebrate assemblages: The influence of temperature with altitude and latitude. Freshw. Biol. 1997, 38, 247–261. [Google Scholar] [CrossRef]
- Domisch, S.; Jähnig, S.C.; Haase, P. Climate-change winners and losers: Stream macroinvertebrates of a submontane region in central europe. Freshw. Biol. 2011, 56, 2009–2020. [Google Scholar] [CrossRef]
- Skoulikidis, N.T.; Amaxidis, Y.; Bertahas, I.; Laschou, S.; Gritzalis, K. Analysis of factors driving stream water composition and synthesis of management tools—A case study on small/medium Greek catchments. Sci. Total Environ. 2006, 362, 205–241. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, D. Tropical high-altitude streams. In Tropical Stream Ecology; Elsevier: Amsterdam, The Netherlands, 2008; pp. 219–256. [Google Scholar]
- Tomanova, S.; Tedesco, P.A.; Campero, M.; Van Damme, P.A.; Moya, N.; Oberdorff, T. Longitudinal and altitudinal changes of macroinvertebrate functional feeding groups in neotropical streams: A test of the river continuum concept. Fundam. Appl. Limnol./Arch. Für Hydrobiol. 2007, 170, 233–241. [Google Scholar] [CrossRef]
- Jacobsen, D.; Rostgaard, S.; Vásconez, J.J. Are macroinvertebrates in high altitude streams affected by oxygen deficiency? Freshw. Biol. 2003, 48, 2025–2032. [Google Scholar] [CrossRef]
- Hoback, W.W.; Stanley, D.W. Insects in hypoxia. J. Insect Physiol. 2001, 47, 533–542. [Google Scholar] [CrossRef]
- Chapman, L.J.; Schneider, K.R.; Apodaca, C.; Chapman, C.A. Respiratory ecology of macroinvertebrates in a swamp-river system of east africa. Biotropica 2004, 36, 572–585. [Google Scholar]
- Jacobsen, D. Low oxygen pressure as a driving factor for the altitudinal decline in taxon richness of stream macroinvertebrates. Oecologia 2008, 154, 795–807. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, D.; Dangles, O. Ecology of High Altitude Waters; Oxford University Press: New York, NY, USA, 2017. [Google Scholar]
- Suren, A.M. Macroinvertebrate communities of streams in western Nepal: Effects of altitude and land use. Freshw. Biol. 1994, 32, 323–336. [Google Scholar] [CrossRef]
- Jansson, A.; Reavell, P. North american species of trichocorixa (heteroptera: Corixidae) introduced into Africa. Afr. Entomol. 1999, 7, 295–297. [Google Scholar]
- Shull, D.R.; Stewart, R.L., Jr.; Hurd, T.M.; Light, T. A case for unique habitat selection by sigara mathesoni (hemiptera: Corixidae) in south-central pennsylvania. Northeast. Nat. 2016, 23, 174–183. [Google Scholar] [CrossRef]
- Ribera, I.; Barraclough, T.; Vogler, A. The effect of habitat type on speciation rates and range movements in aquatic beetles: Inferences from species-level phylogenies. Mol. Ecol. 2001, 10, 721–735. [Google Scholar] [CrossRef] [PubMed]
- White, D. Coleoptera (Beetles) in Aquatic Ecosystems; Encyclopedia of Inland Waters; Elsevier: Amsterdam, The Netherlands, 2009; pp. 144–156. [Google Scholar] [CrossRef]
- Nachtigall, W. Swimming, lake insects. In Encyclopedia of Insects, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 972–975. [Google Scholar]
- Nilsson, A.N. Dytiscidae (Coleoptera); Apollo Books: Stenstrup, Denmark, 2001; Volume 3. [Google Scholar]
- Stoian, L.C.; Gagyi-Palffy, A.; Stan, G. Preliminary aspects regarding the use of some invertebrate bioindicator species in the ecological study of an aquatic lotic ecosystem. Aquac. Aquar. Conserv. Legis. Int. J. Bioflux Soc. (AACL Bioflux) 2009, 2, 331–338. [Google Scholar]
- Owor, M.; Hartwig, T.; Muwanga, A.; Zachmann, D.; Pohl, W. Impact of tailings from the kilembe copper mining district on lake george, uganda. Environ. Geol. 2007, 51, 1065–1075. [Google Scholar] [CrossRef]
- Lwanga, M.S.; Kansiime, F.; Denny, P.; Scullion, J. Heavy metals in lake george, uganda, with relation to metal concentrations in tissues of common fish species. Hydrobiologia 2003, 499, 83–93. [Google Scholar] [CrossRef]
- Kiffney, P.M.; Clements, W.H. Effects of metals on stream macroinvertebrate assemblages from different altitudes. Ecol. Appl. 1996, 6, 472–481. [Google Scholar] [CrossRef]
- Malmqvist, B.; Hoffsten, P.-O. Influence of drainage from old mine deposits on benthic macroinvertebrate communities in central swedish streams. Water Res. 1999, 33, 2415–2423. [Google Scholar] [CrossRef]
- Loayza-Muro, R.A.; Elías-Letts, R.; Marticorena-Ruíz, J.K.; Palomino, E.J.; Duivenvoorden, J.F.; Kraak, M.H.; Admiraal, W. Metal-induced shifts in benthic macroinvertebrate community composition in andean high altitude streams. Environ. Toxicol. Chem. 2010, 29, 2761–2768. [Google Scholar] [CrossRef]
- Hogsden, K.L.; Harding, J.S. Consequences of acid mine drainage for the structure and function of benthic stream communities: A review. Freshw. Sci. 2011, 31, 108–120. [Google Scholar] [CrossRef]
- Winner, R.; Boesel, M.; Farrell, M. Insect community structure as an index of heavy-metal pollution in lotic ecosystems. Can. J. Fish. Aquat. Sci. 1980, 37, 647–655. [Google Scholar] [CrossRef]
- Archaimbault, V.; Usseglio-Polatera, P.; Garric, J.; Wasson, J.G.; Babut, M. Assessing pollution of toxic sediment in streams using bio-ecological traits of benthic macroinvertebrates. Freshw. Biol. 2010, 55, 1430–1446. [Google Scholar] [CrossRef]
- Pallottini, M.; Cappelletti, D.; Fabrizi, A.; Gaino, E.; Goretti, E.; Selvaggi, R.; Céréghino, R. Macroinvertebrate functional trait responses to chemical pollution in agricultural–industrial landscapes. River Res. Appl. 2017, 33, 505–513. [Google Scholar] [CrossRef]
- Gheorghe, S.; Stoica, C.; Vasile, G.G.; Nita-Lazar, M.; Stanescu, E.; Lucaciu, I.E. Metals toxic effects in aquatic ecosystems: Modulators of water quality. In Water Quality; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar]
- Glazier, D.S. Temperature affects food-chain length and macroinvertebrate species richness in spring ecosystems. Freshw. Sci. 2012, 31, 575–585. [Google Scholar] [CrossRef]
- Jyväsjärvi, J.; Marttila, H.; Rossi, P.M.; Ala-Aho, P.; Olofsson, B.; Nisell, J.; Backman, B.; Ilmonen, J.; Virtanen, R.; Paasivirta, L. Climate-induced warming imposes a threat to north european spring ecosystems. Glob. Chang. Biol. 2015, 21, 4561–4569. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, C.; Meerhoff, M.; Johansson, L.S.; González-Bergonzoni, I.; Mazzeo, N.; Pacheco, J.P.; Teixeira-de Mello, F.; Goyenola, G.; Lauridsen, T.L.; Søndergaard, M. Stable isotope analysis confirms substantial differences between subtropical and temperate shallow lake food webs. Hydrobiologia 2017, 784, 111–123. [Google Scholar] [CrossRef]
- Wilhelm, L.; Singer, G.A.; Fasching, C.; Battin, T.J.; Besemer, K. Microbial biodiversity in glacier-fed streams. ISME J. 2013, 7, 1651. [Google Scholar] [CrossRef] [PubMed]
- Slemmons, K.E.; Saros, J.E.; Simon, K. The influence of glacial meltwater on alpine aquatic ecosystems: A review. Environ. Sci. Process. Impacts 2013, 15, 1794–1806. [Google Scholar] [CrossRef] [PubMed]
- Flemming, C.; Trevors, J. Copper toxicity and chemistry in the environment: A review. WaterAirSoil Pollut. 1989, 44, 143–158. [Google Scholar] [CrossRef]
- Förstner, U.; Wittmann, G.T. Metal Pollution in the Aquatic Environment; Springer Science & Business Media: Berlin, Germany, 2012. [Google Scholar]
- Viers, J.; Dupré, B.; Gaillardet, J. Chemical composition of suspended sediments in world rivers: New insights from a new database. Sci. Total Environ. 2009, 407, 853–868. [Google Scholar] [CrossRef] [PubMed]
- Gíslason, G.M.; Ad✗alsteinsson, H.; Hansen, I.; Ólafsson, J.S.; Svavarsdóttir, K. Longitudinal changes in macroinvertebrate assemblages along a glacial river system in central iceland. Freshw. Biol. 2001, 46, 1737–1751. [Google Scholar] [CrossRef]
- Smith, A.J.; Bode, R.W.; Kleppel, G.S. A nutrient biotic index (nbi) for use with benthic macroinvertebrate communities. Ecol. Indic. 2007, 7, 371–386. [Google Scholar] [CrossRef]
- Everall, N.C.; Johnson, M.F.; Wood, P.; Paisley, M.F.; Trigg, D.J.; Farmer, A. Macroinvertebrate community structure as an indicator of phosphorus enrichment in rivers. Ecol. Indic. 2019, 107, 105619. [Google Scholar] [CrossRef]
- Peralta, E.M.; Batucan, L.S., Jr.; De Jesus, I.B.B.; Triño, E.M.C.; Uehara, Y.; Ishida, T.; Kobayashi, Y.; Ko, C.-Y.; Iwata, T.; Borja, A.S. Nutrient loadings and deforestation decrease benthic macroinvertebrate diversity in an urbanised tropical stream system. Limnologica 2020, 80, 125744. [Google Scholar] [CrossRef]
- Horak, C.N.; Assef, Y.A.; Miserendino, M.L. Assessing effects of confined animal production systems on water quality, ecological integrity, and macroinvertebrates at small piedmont streams (patagonia, argentina). Agric. Water Manag. 2019, 216, 242–253. [Google Scholar] [CrossRef]
- Basset, Y.; Missa, O.; Alonso, A.; Miller, S.E.; Curletti, G.; De Meyer, M.; Eardley, C.; Lewis, O.T.; Mansell, M.W.; Novotny, V. Changes in arthropod assemblages along a wide gradient of disturbance in Gabon. Conserv. Biol. 2008, 22, 1552–1563. [Google Scholar] [CrossRef] [PubMed]
- Madsen, P.B.; Morabowen, A.; Andino, P.; Espinosa, R.; Cauvy-Fraunié, S.; Dangles, O.; Jacobsen, D. Altitudinal distribution limits of aquatic macroinvertebrates: An experimental test in a tropical alpine stream. Ecol. Entomol. 2015, 40, 629–638. [Google Scholar] [CrossRef]
- Loayza-Muro, R.A. Life at the Edge: Benthic Invertebrates in High Altitude Andean Streams. Ph.D. Thesis, Universiteit van Amsterdam, Amsterdam, The Netherlands, 2013. [Google Scholar]
- Lencioni, V. Glacial influence and stream macroinvertebrate biodiversity under climate change: Lessons from the southern alps. Sci. Total Environ. 2018, 622, 563–575. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, D.; Milner, A.M.; Brown, L.E.; Dangles, O. Biodiversity under threat in glacier-fed river systems. Nat. Clim. Chang. 2012, 2, 361. [Google Scholar] [CrossRef]
- Finn, D.S.; Khamis, K.; Milner, A.M. Loss of small glaciers will diminish beta diversity in pyrenean streams at two levels of biological organization. Glob. Ecol. Biogeogr. 2013, 22, 40–51. [Google Scholar] [CrossRef]
- Danladi Bello, A.-A.; Hashim, N.B.; Mohd Haniffah, M.R. Predicting impact of climate change on water temperature and dissolved oxygen in tropical rivers. Climate 2017, 5, 58. [Google Scholar] [CrossRef]
| Taxa | |
|---|---|
| DS (n = 5) | Corixidae *, Belostomatidae, Leptophlebiidae, Thiaridae, Tricorythidae |
| MS (n = 6) | Chironomidae, Lumbricidae, Ecnomidae, Gerridae, Perlidae, Simuliidae |
| US (n = 11) | Dytiscidae *, Elmidae, Lepidostomatidae, Hydraenidae, Hydrophilidae, Limoniidae, Dugesiidae, Erpobdellidae, Limoniidae, Sphaeriidae, Staphylinidae |
| DS + MS (n = 5) | Baetidae, Hydropsychidae, Physidae, Psychodidae, Atyidae |
| DS + US (n = 7) | Coenagrionidae, Caenidae, Leptoceridae, Planorbidae, Veliidae, Hydracarina, Tubificidae |
| MS + US (n = 3) | Gomphidae, Gyrinidae, Tipulidae |
| Title Variable | All Sites (N = 30) | US (N = 10) | MS (N = 10) | DS (N = 10) |
|---|---|---|---|---|
| (Intercept) | 11.678 | 651.61 | 5.282 | 12.518 |
| Temperature | −0.269 | −0.553 | −0.292 | |
| Nickel | −1.733 | −4.012 | ||
| Conductivity | −0.3571 | −0.653 | ||
| Longitude | −233.122 | |||
| Total phosphorus | −0.642 | |||
| Nitrite nitrogen | −1.271 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musonge, P.S.L.; Boets, P.; Lock, K.; Goethals, P.L.M. Drivers of Benthic Macroinvertebrate Assemblages in Equatorial Alpine Rivers of the Rwenzoris (Uganda). Water 2020, 12, 1668. https://doi.org/10.3390/w12061668
Musonge PSL, Boets P, Lock K, Goethals PLM. Drivers of Benthic Macroinvertebrate Assemblages in Equatorial Alpine Rivers of the Rwenzoris (Uganda). Water. 2020; 12(6):1668. https://doi.org/10.3390/w12061668
Chicago/Turabian StyleMusonge, Peace S.L., Pieter Boets, Koen Lock, and Peter L.M. Goethals. 2020. "Drivers of Benthic Macroinvertebrate Assemblages in Equatorial Alpine Rivers of the Rwenzoris (Uganda)" Water 12, no. 6: 1668. https://doi.org/10.3390/w12061668
APA StyleMusonge, P. S. L., Boets, P., Lock, K., & Goethals, P. L. M. (2020). Drivers of Benthic Macroinvertebrate Assemblages in Equatorial Alpine Rivers of the Rwenzoris (Uganda). Water, 12(6), 1668. https://doi.org/10.3390/w12061668







