Effects of Crucian Carp (Carassius auratus) on Water Quality in Aquatic Ecosystems: An Experimental Mesocosm Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Material
2.2. Experimental Design
2.3. Sampling
2.4. Statistical Analyses
3. Results
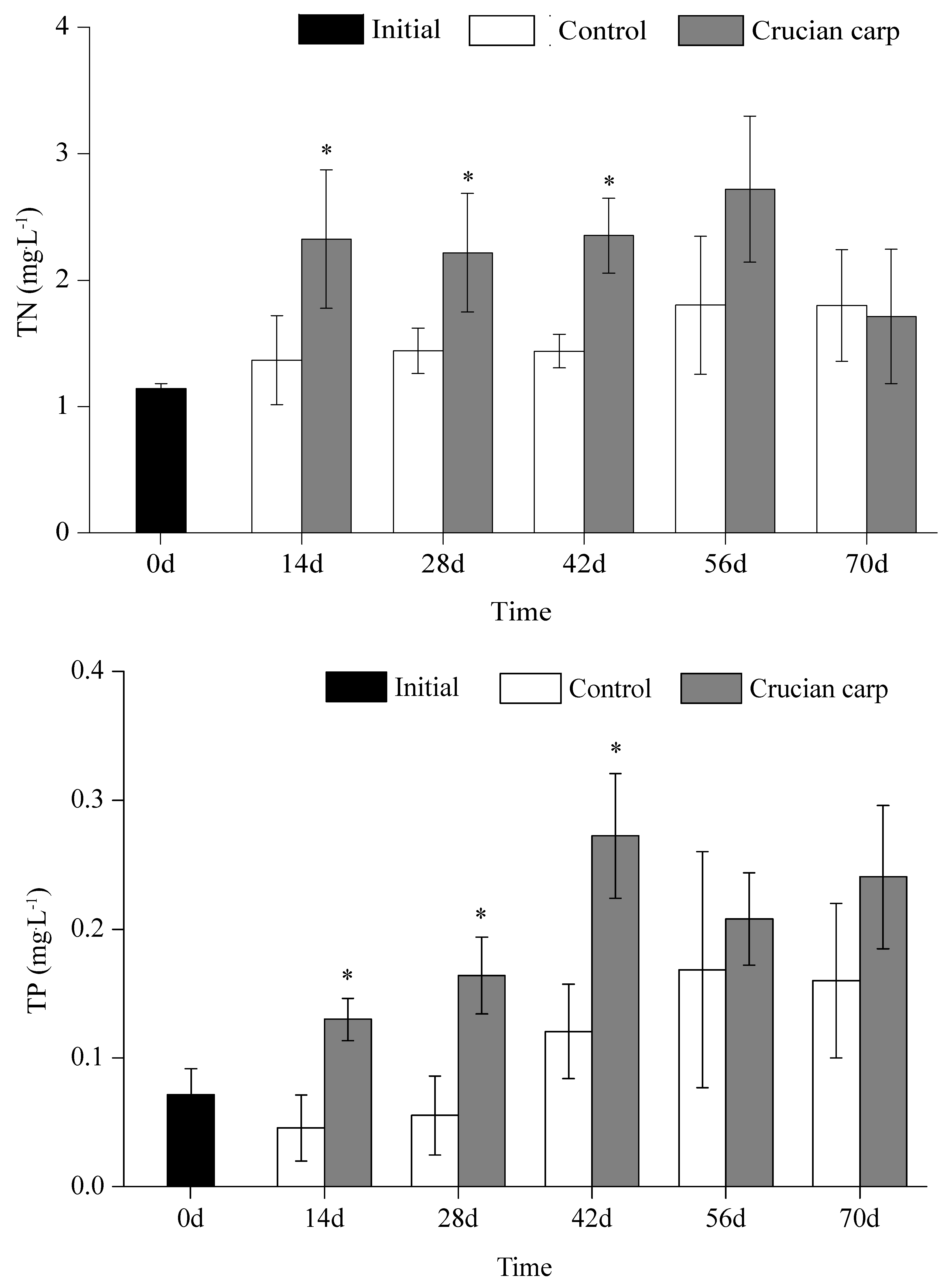
3.1. TN and TP
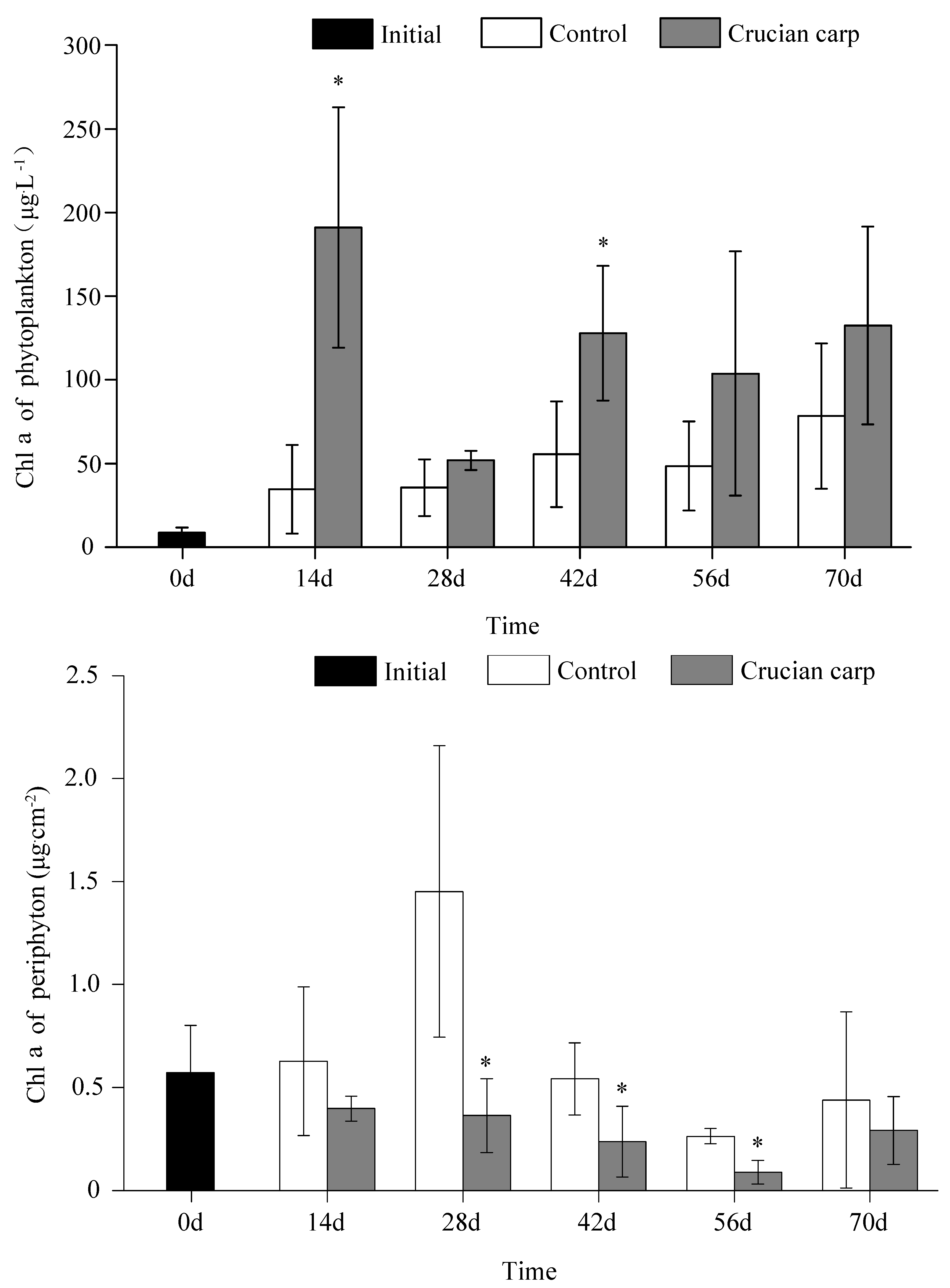
3.2. Phytoplankton and Periphyton Biomass
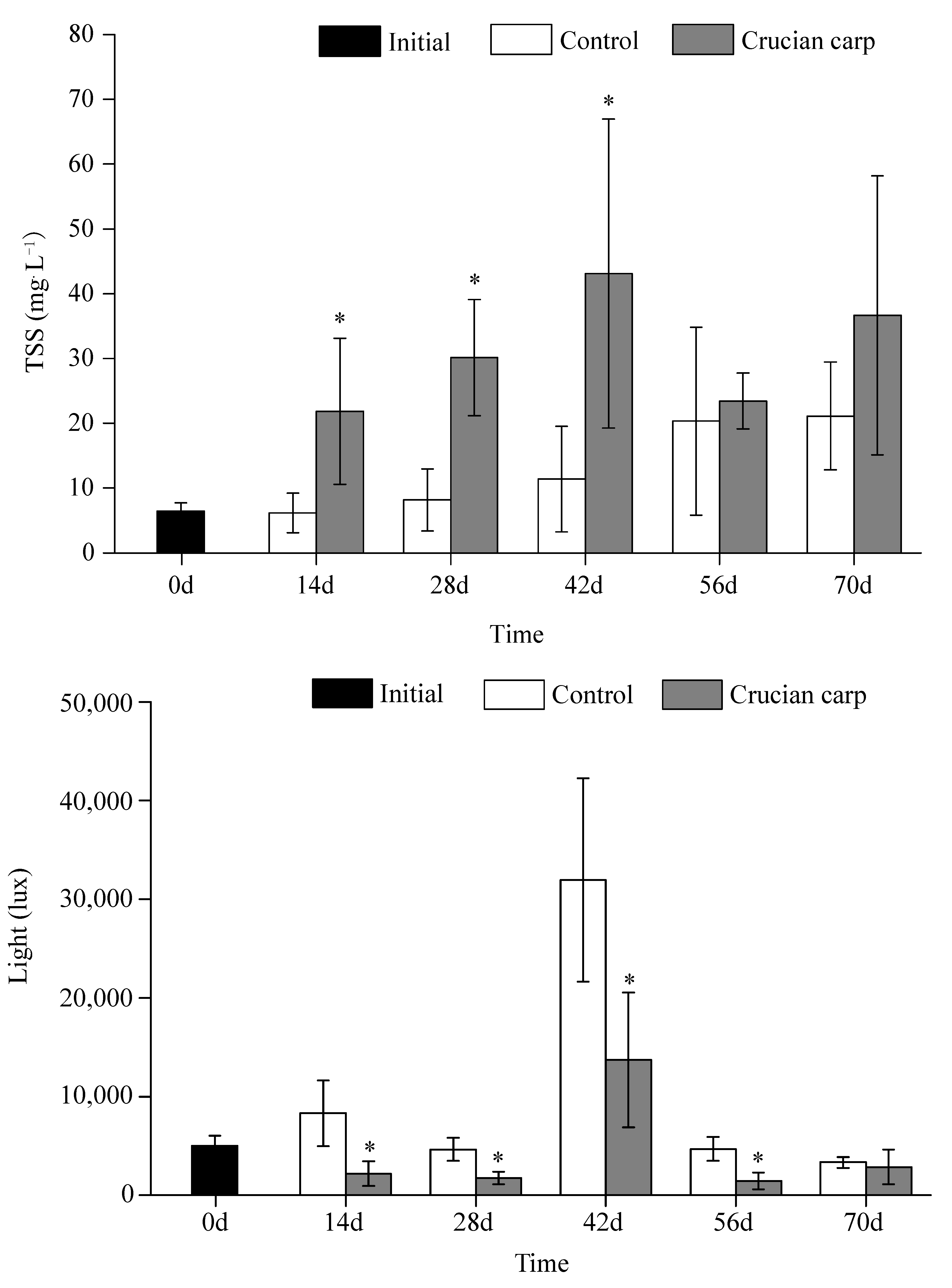
3.3. TSS and Light Intensity
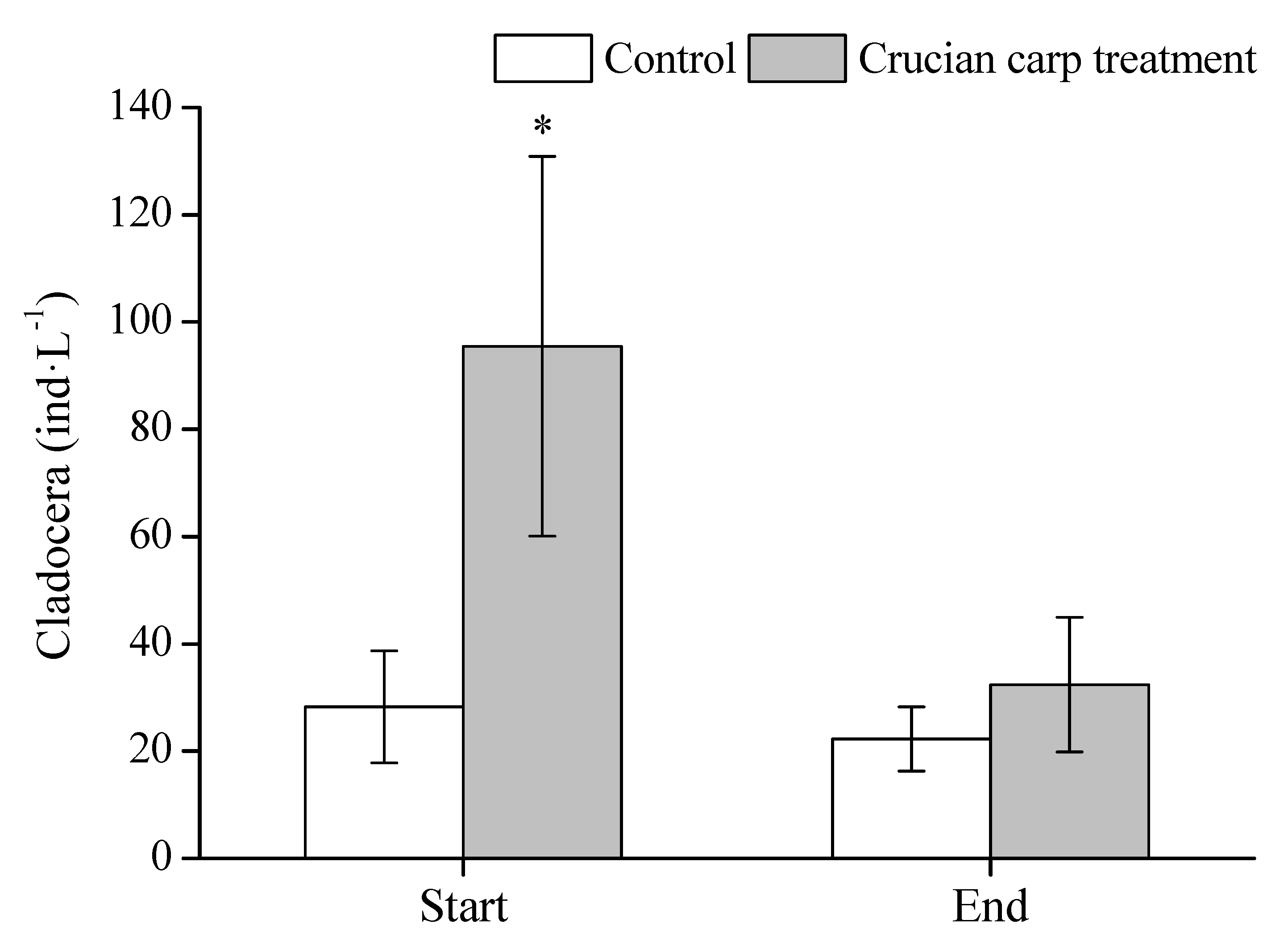
3.4. Zooplankton
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andersson, G.; Berggren, H.; Cronberg, G.; Gelin, C. Effects of planktivorous and benthivorous fish on organisms and water chemistry in eutrophic lakes. Hydrobiologia 1978, 59, 9–15. [Google Scholar] [CrossRef]
- Menezes, R.F.; Attayde, J.L.; Vasconcelos, F.R. Effects of omnivorous filter-feeding fish and nutrient enrichment on the plankton community and water transparency of a tropical reservoir. Freshw. Biol. 2010, 55, 767–779. [Google Scholar] [CrossRef]
- Zambrano, L.; Hinojosa, D. Direct and indirect effects of carp (Cyprinus carpio L.) on macrophyte and benthic communities in experimental shallow ponds in central Mexico. Hydrobiologia 1999, 408, 131–138. [Google Scholar] [CrossRef]
- Vanni, M.J.; Arend, K.K.; Bremigan, M.T.; Bunnell, D.B.; Garvey, J.E.; González, M.J.; Renwick, W.H.; Soranno, P.A.; Stein, R.A. Linking landscapes and food webs: Effects of omnivorous fish and watersheds on reservoir ecosystems. Bioscience 2005, 55, 155–167. [Google Scholar] [CrossRef]
- FAO. Cultured Aquatic Species Information Programme: Carassius carassius. Available online: http://www.fao.org/fishery/culturedspecies/Carassius_carassius/en (accessed on 9 January 2020).
- Gao, J.; Liu, Z.; Jeppesen, E. Fish community assemblages changed but biomass remained similar after lake restoration by biomanipulation in a Chinese tropical eutrophic lake. Hydrobiologia 2014, 724, 127–140. [Google Scholar] [CrossRef]
- He, H.; Hu, E.; Yu, J.; Luo, X.; Li, K.; Jeppesen, E.; Liu, Z. Does turbidity induced by Carassius carassius limit phytoplankton growth? A mesocosm study. Environ. Sci. Pollut. Res. 2017, 24, 5012–5018. [Google Scholar] [CrossRef]
- Richardson, M.J.; Whoriskey, F.G.; Roy, L.H. Turbidity generation and biological impacts of an exotic fish Carassius auratus, introduced into shallow seasonally anoxic ponds. J. Fish. Biol. 1995, 47, 576–585. [Google Scholar] [CrossRef]
- Breukelaar, A.W.; Lammens, E.H.R.R.; Breteler, J.G.P.K.; Tátrai, I. Effects of benthivorous bream (Abramis brama) and carp (Cyprinus carpio) on sediment resuspension and concentrations of nutrients and chlorophyll a. Freshw. Biol. 1994, 32, 113–121. [Google Scholar] [CrossRef]
- Scheffer, M.; Portielje, R.; Zambrano, L. Fish facilitate wave resuspension of sediment. Limnol. Oceanogr. 2003, 48, 1920–1926. [Google Scholar] [CrossRef]
- Weber, M.J.; Brown, M.L. Effects of common carp on aquatic ecosystems 80 years after “carp as a dominant”: Ecological insights for fisheries management. Rev. Fish. Sci. 2009, 17, 524–537. [Google Scholar] [CrossRef]
- Zhang, X.; Mei, X.; Gulati, R.D. Effects of omnivorous tilapia on water turbidity and primary production dynamics in shallow lakes: Implications for ecosystem management. Rev. Fish. Biol. Fish. 2017, 27, 245–254. [Google Scholar] [CrossRef]
- Zambrano, L.; Perrow, M.R.; Macías-García, C.; Aguirre-Hidalgo, V. Impact of introduced carp (Cyprinus carpio) in subtropical shallow ponds in Central Mexico. J. Aquat. Ecosystem. Stress Recov. 1998, 6, 281–288. [Google Scholar] [CrossRef]
- Chumchal, M.M.; Drenner, R.W. Interrelationships between phosphorus loading and common carp in the regulation of phytoplankton biomass. Arch. Hydrobiol. 2004, 161, 147–158. [Google Scholar] [CrossRef]
- Adámek, Z.; Maršálek, B. Bioturbation of sediments by benthic macroinvertebrates and fish and its implication for pond ecosystems: A review. Aquac. Int. 2013, 21, 1–17. [Google Scholar] [CrossRef]
- Attayde, J.L.; Hansson, L.A. Fish-mediated nutrient recycling and the trophic cascade in lakes. Can. J. Fish. Aquat. Sci. 2001, 58, 1924–1931. [Google Scholar] [CrossRef]
- Matsuzaki, S.S.; Usio, N.; Takamura, N.; Washitani, I. Effects of common carp on nutrient dynamics and littoral community composition: Roles of excretion and bioturbation. Fundam. Appl. Limnol. Arch. Hydrobiol. 2007, 168, 27–38. [Google Scholar] [CrossRef]
- Fischer, J.R.; Krogman, R.M.; Quist, M.C. Influences of native and non-native benthivorous fishes on aquatic ecosystem degradation. Hydrobiologia 2013, 711, 187–199. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Meerhoff, M.; Lauridsen, T.L.; Jensen, J.P. Shallow lake restoration by nutrient loading reduction-some recent findings and challenges ahead. Hydrobiologia 2007, 584, 239–252. [Google Scholar] [CrossRef]
- He, H.; Han, Y.; Li, Q.; Jeppesen, E.; Li, K.; Yu, J.; Liu, Z. Crucian carp (Carassius carassius) Strongly affect C/N/P stoichiometry of suspended particulate matter in shallow warm water eutrophic lakes. Water 2019, 11, 524. [Google Scholar] [CrossRef]
- Iglesias, C.; Mazzeo, N.; Meerhoff, M.; Lacerot, G.; Clemente, J.; Scasso, F.; Kruk, C.; Goyenola, G.; García-Alonso, J.; Amsinck, S.L.; et al. High predation is the key factor for dominance of small-bodied zooplankton in warm lakes-evidence from lakes, fish exclosures and surface sediment. Hydrobiologia 2011, 667, 133–147. [Google Scholar] [CrossRef]
- Qiu, X.; Mei, X.; Razlutskij, V.; Rudstam, L.G.; Liu, Z.; Tong, C.; Zhang, X. Effects of common carp (Cyprinus carpio) on water quality in aquatic ecosystems dominated by submerged plants: A mesocosm study. Knowl. Manag. Aquat. Ecosyst. 2019, 420, 28. [Google Scholar] [CrossRef]
- Roberts, J.; Chick, A.; Oswald, L.; Thompson, P. Effect of carp, Cyprinus carpio L., an exotic benthivorous fish, on aquatic plants and water quality in experimental ponds. Mar. Freshw. Res. 1995, 46, 1171–1180. [Google Scholar] [CrossRef]
- Fletcher, A.R.; Morison, A.K.; Hume, D.J. Effects of carp, Cyprinus carpio L., on communities of aquatic vegetation and turbidity of water bodies in the Lower Goulburn River Basin. Mar. Freshw. Res. 1985, 36, 311–327. [Google Scholar] [CrossRef]
- Havens, K.E. Fish-induced sediment resuspension: Effects on phytoplankton biomass and community structure in a shallow hypereutrophic lake. J. Plankton Res. 1991, 13, 1163–1176. [Google Scholar] [CrossRef]
- Yi, C.; Guo, L.; Ni, L.; Luo, C. Silver carp exhibited an enhanced ability of biomanipulation to control cyanobacteria bloom compared to bighead carp in hypereutrophic Lake Taihu mesocosms. Ecol. Eng. 2016, 89, 7–13. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Z.; Jeppesen, E.; Taylor, W.D.; Rudstam, L.G. Effects of benthic-feeding common carp and filter-feeding silver carp on benthic-pelagic coupling: Implications for shallow lake management. Ecol. Eng. 2016, 88, 256–264. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Davidson, T.A.; Liu, Z.; Mazzeo, N.; Trochine, C.; Özkan, K.; Jensen, H.S.; Trolle, D.; et al. Biomanipulation as a restoration tool to combat eutrophication: Recent advances and future challenges. Adv. Ecol. Res. 2012, 47, 411–487. [Google Scholar]
- Zhang, X.; Liu, Z.; Jeppesen, E.; Taylor, W.D. Effects of deposit-feeding tubificid worms and filter-feeding bivalves on benthic-pelagic coupling: Implications for the restoration of eutrophic shallow lakes. Water Res. 2014, 50, 135–146. [Google Scholar] [CrossRef]
- Penghan, L.Y.; Cao, Z.D.; Fu, S.J. Effect of temperature and dissolved oxygen on swimming performance in crucian carp. Aquat. Biol. 2014, 21, 57–65. [Google Scholar] [CrossRef][Green Version]
- Jespersen, A.M.; Christoffersen, K. Measurements of chlorophyll a from phytoplankton using ethanol as extraction solvent. Arch. Hydrobiol. 1987, 109, 445–454. [Google Scholar]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Chiang, S.C.; Du, N.S. Fauna Sinica: Crustacea: Freshwater Cladocera; Science Press: Beijing, China, 1979. (In Chinese) [Google Scholar]
- Diana, J.S.; Dettweiler, D.J.; Lin, C.K. Effect of Nile tilapia (Oreochromis niloticus) on the ecosystem of aquaculture ponds, and its significance to the trophic cascade hypothesis. Can. J. Fish. Aquat. Sci. 1991, 48, 183–190. [Google Scholar] [CrossRef]
- Attayde, J.L.; Hansson, L.A. Effects of nutrient recycling by zooplankton and fish on phytoplankton communities. Oecologia 1999, 121, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Glaholt, J.R.; Vanni, M.J. Ecological responses to simulated benthic-derived nutrient subsidies mediated by omnivorous fish. Freshw. Biol. 2005, 50, 1864–1881. [Google Scholar] [CrossRef]
- Wahl, D.H.; Wolfe, M.D.; Santucci, V.J.; Santucci Jr, V.J.; Freedman, J.A. Invasive carp and prey community composition disrupt trophic cascades in eutrophic ponds. Hydrobiologia 2011, 678, 49–63. [Google Scholar] [CrossRef]
- Liu, Z.; Hu, J.; Zhong, P.; Zhang, X.; Ning, J.; Larsen, S.E.; Chen, D.; Gao, Y.; He, H.; Jeppesen, E. Successful restoration of a tropical shallow eutrophic lake: Strong bottom-up but weak top-down effects recorded. Water Res. 2018, 146, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Driver, P.D.; Closs, G.P.; Koen, T. The effects of size and density of carp (Cyprinus carpio L.) on water quality in an experimental pond. Arch. Hydrobiol. 2005, 163, 117–131. [Google Scholar] [CrossRef]
- Brabrand, Å.; Faafeng, B.A.; Moritz Nilssen, J.P. Relative importance of phosphorus supply to phytoplankton production: Fish excretion versus external loading. Can. J. Fish. Aquat. Sci. 1990, 47, 364–372. [Google Scholar] [CrossRef]
- Persson, A. Effects of fish predation and excretion on the configuration of aquatic food webs. Oikos 1997, 79, 137–146. [Google Scholar] [CrossRef]
- Shin-ichiro, S.M.; Usio, N.; Takamura, N.; Washitani, I. Contrasting impacts of invasive engineers on freshwater ecosystems: An experiment and meta-analysis. Oecologia 2009, 158, 673–686. [Google Scholar]
- Guo, X.T.; Liu, F.; Wang, F. Carbon, nitrogen, and phosphorus stoichiometry of three freshwater cultured fishes in growth stage. Turk. J. Fish Aquat. Sci. 2018, 18, 239–245. [Google Scholar] [CrossRef]
- Gu, J.; Jin, H.; He, H.; Ning, X.; Yu, J.; Tan, B.; Jeppesen, E.; Li, K. Effects of small-sized crucian carp (Carassius carassius) on the growth of submerged macrophytes: Implications for shallow lake restoration. Ecol. Eng. 2016, 95, 567–573. [Google Scholar] [CrossRef]
- Hansson, L.A. Effects of competitive interactions on the biomass development of planktonic and periphytic algae in lakes. Limnol. Oceanogr. 1988, 33, 121–128. [Google Scholar] [CrossRef]
- van Dam, A.A.; Beveridge, M.C.M.; Azim, M.E.; Verdegem, M.C.J. The potential of fish production based on periphyton. Rev. Fish. Biol. Fish. 2002, 12, 1–31. [Google Scholar] [CrossRef]
- Huchette, M.H.; Beveridge, M.C.M.; Baird, D.J.; Irelandb, M. The impacts of grazing by tilapias (Oreochromis niloticus L.) on periphyton communities growing on artificial substrate in cages. Aquaculture 2000, 186, 45–60. [Google Scholar] [CrossRef]
- Azim, M.E.; Verdegem, M.C.J.; Singh, M.; Van Dam, A.A.; Beveridge, M.C.M. The effects of periphyton substrate and fish stocking density on water quality, phytoplankton, periphyton and fish growth. Aquac. Res. 2003, 34, 685–695. [Google Scholar] [CrossRef]
- Liboriussen, L.; Jeppesen, E.; Bramm, M.E.; Lassen, M.F. Periphyton-macroinvertebrate interactions in light and fish manipulated enclosures in a clear and a turbid shallow lake. Aquat. Ecol. 2005, 39, 23–39. [Google Scholar] [CrossRef]
- Kerfoot, W.C.; Levitan, C.; DeMott, W.R. Daphnia-phytoplankton interactions: Density-dependent shifts in resource quality. Ecology 1988, 69, 1806–1825. [Google Scholar] [CrossRef]
- Elser, J.J. Phytoplankton dynamics and the role of grazers in Castle Lake, California. Ecology 1992, 73, 887–902. [Google Scholar] [CrossRef]
- Vanni, M.J. Effects of food availability and fish predation on a zooplankton community. Ecol. Monogr. 1987, 57, 61–88. [Google Scholar] [CrossRef]
- Dejenie, T.; Asmelash, T.; Rousseaux, S.; Gebregiorgis, T.; Gebrekidan, A.; Teferi, M.; Nyssen, J.; Deckers, J.; Van Der Gucht, K.; Vyverman, W.; et al. Impact of the fish Garra on the ecology of reservoirs and the occurrence of Microcystis blooms in semi-arid tropical highlands: An experimental assessment using enclosures. Freshw. Biol. 2009, 54, 1605–1615. [Google Scholar] [CrossRef]
- Suresh, A.V.; Lin, C.K. Tilapia culture in saline waters: A review. Aquaculture 1992, 106, 201–226. [Google Scholar] [CrossRef]
- Covich, A.P.; Palmer, M.A.; Crowl, T.A. The role of benthic invertebrate species in freshwater ecosystems: Zoobenthic species influence energy flows and nutrient cycling. BioScience 1999, 49, 119–127. [Google Scholar] [CrossRef]
- Lacerot, G.; Kruk, C.; Lürling, M.; Scheffer, M. The role of subtropical zooplankton as grazers of phytoplankton under different predation levels. Freshw. Biol. 2013, 58, 494–503. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.; Mei, X.; Rudstam, L.G.; Taylor, W.D.; Urabe, J.; Jeppesen, E.; Liu, Z.; Zhang, X. Effects of Crucian Carp (Carassius auratus) on Water Quality in Aquatic Ecosystems: An Experimental Mesocosm Study. Water 2020, 12, 1444. https://doi.org/10.3390/w12051444
Huang Y, Mei X, Rudstam LG, Taylor WD, Urabe J, Jeppesen E, Liu Z, Zhang X. Effects of Crucian Carp (Carassius auratus) on Water Quality in Aquatic Ecosystems: An Experimental Mesocosm Study. Water. 2020; 12(5):1444. https://doi.org/10.3390/w12051444
Chicago/Turabian StyleHuang, Yehui, Xueying Mei, Lars G. Rudstam, William D. Taylor, Jotaro Urabe, Erik Jeppesen, Zhengwen Liu, and Xiufeng Zhang. 2020. "Effects of Crucian Carp (Carassius auratus) on Water Quality in Aquatic Ecosystems: An Experimental Mesocosm Study" Water 12, no. 5: 1444. https://doi.org/10.3390/w12051444
APA StyleHuang, Y., Mei, X., Rudstam, L. G., Taylor, W. D., Urabe, J., Jeppesen, E., Liu, Z., & Zhang, X. (2020). Effects of Crucian Carp (Carassius auratus) on Water Quality in Aquatic Ecosystems: An Experimental Mesocosm Study. Water, 12(5), 1444. https://doi.org/10.3390/w12051444







