Impact of Storage Conditions on the Methanogenic Activity of Anaerobic Digestion Inocula
Abstract
1. Introduction
2. Materials and Methods
2.1. Analytical Methods
2.2. Chemical Reagents and Inoculum Source
2.3. Specific Methanogenic Activity (SMA) Test Set-up
2.3.1. Experiment 1: Impact of Storage Temperature on DPM Inoculum
2.3.2. Experiment 2: Impact of Storage Temperature on DSS Inoculum
2.3.3. Experiment 3: Impact of Storage Temperature and Storage Time on DSS Inoculum
2.4. Data Analysis
3. Results
3.1. Impact of Storage Temperature on SMA
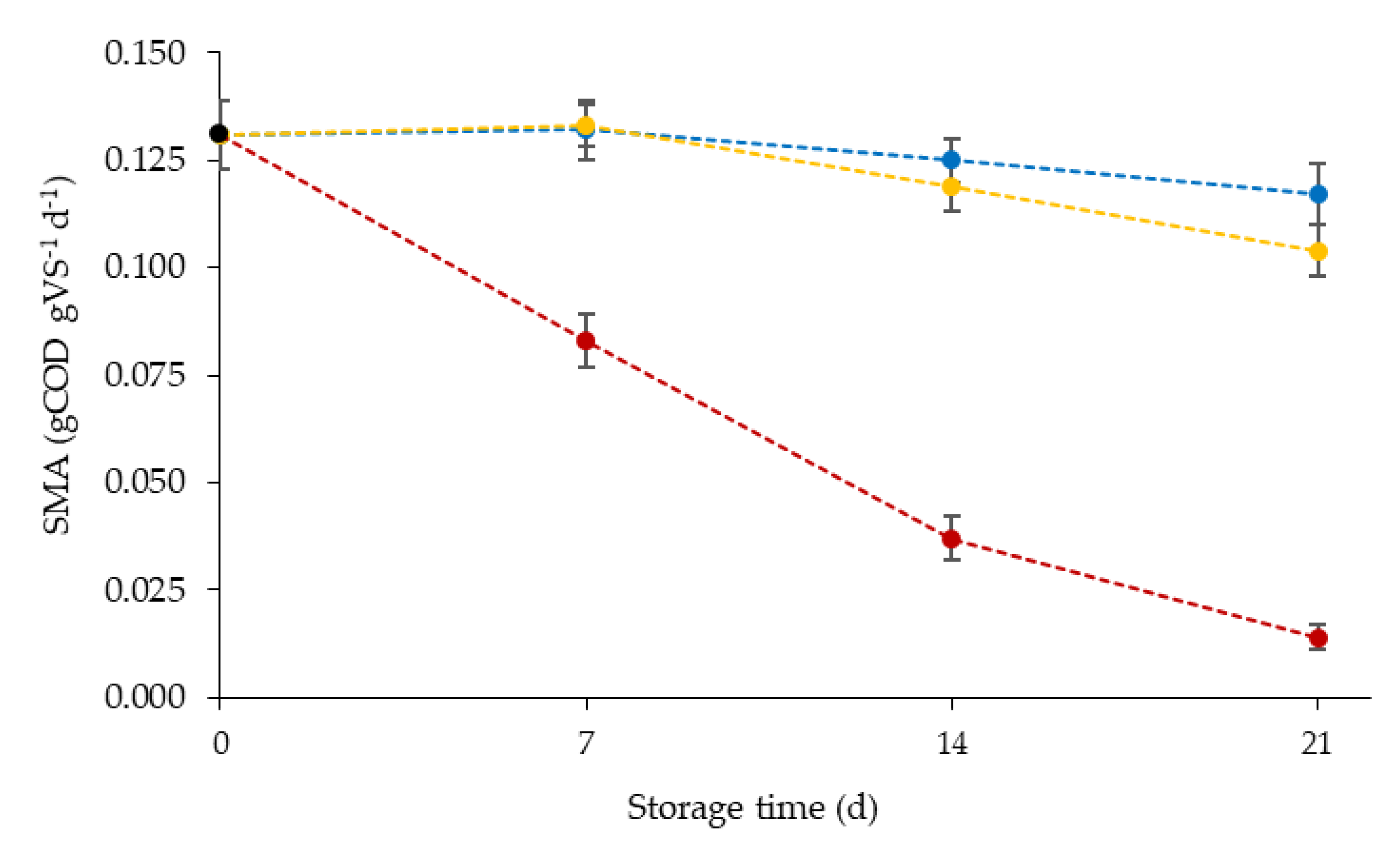
3.2. Impact of Storage Temperature and Storage Time on SMA
4. Discussion
4.1. Impact of Storage Temperature on Inoculum Activity
4.2. Impact of Storage Time on Inoculum Activity
4.3. Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Soto, M.; Mendez, R.; Lema, J.M. Methanogenic and non-methanogenic activity tests: Theoretical basis and experimental set up. Water Res. 1993, 27, 1361–1376. [Google Scholar] [CrossRef]
- Peces, M.; Astals, S.; Jensen, P.; Clarke, W. Deterministic mechanisms define the long-term anaerobic digestion microbiome and its functionality regardless of the initial microbial community. Water Res. 2018, 141, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Angelidaki, I.; Alves, M.; Bolzonella, D.; Borzacconi, L.; Campos, J.L.; Guwy, A.J.; Kalyuzhnyi, S.; Jenicek, P.; van Lier, J.B. Defining the biomethane potential (BMP) of solid organic wastes and energy crops: A proposed protocol for batch assays. Water Sci. Technol. 2009, 59, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Holliger, C.; Alves, M.; Andrade, D.; Angelidaki, I.; Astals, S.; Baier, U.; Bougrier, C.; Buffière, P.; Carballa, M.; de Wilde, V.; et al. Towards a standardization of biomethane potential tests. Water Sci. Technol. 2016, 74, 2515–2522. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, C.; Astals, S.; Peces, M.; Campos, J.L.; Guerrero, L. Biochemical methane potential (BMP) tests: Reducing test time by early parameter estimation. Waste Manag. 2018, 71, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.; Hafner, S.D.; Weinrich, S.; Astals, S.; Holliger, C. Power and Limitations of Biochemical Methane Potential (BMP) Tests. Front. Energy Res. 2020, 8. [Google Scholar] [CrossRef]
- Lindorfer, H.; Pérez López, C.; Resch, C.; Braun, R.; Kirchmayr, R. The impact of increasing energy crop addition on process performance and residual methane potential in anaerobic digestion. Water Sci. Technol. 2007, 56, 55–63. [Google Scholar] [CrossRef]
- Ruile, S.; Schmitz, S.; Mönch-Tegeder, M.; Oechsner, H. Degradation efficiency of agricultural biogas plants—A full-scale study. Bioresour. Technol. 2015, 178, 341–349. [Google Scholar] [CrossRef]
- Owen, W.F.; Stuckey, D.C.; Healy, J.B.; Young, L.Y.; McCarty, P.L. Bioassay for monitoring biochemical methane potential and anaerobic toxicity. Water Res. 1979, 13, 485–492. [Google Scholar] [CrossRef]
- Astals, S.; Batstone, D.J.; Tait, S.; Jensen, P.D. Development and validation of a rapid test for anaerobic inhibition and toxicity. Water Res. 2015, 81, 208–215. [Google Scholar] [CrossRef]
- Elbeshbishy, E.; Nakhla, G.; Hafez, H. Biochemical methane potential (BMP) of food waste and primary sludge: Influence of inoculum pre-incubation and inoculum source. Bioresour. Technol. 2012, 110, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.; Hafner, S.D.; Weinrich, S.; Astals, S. Identification of critical problems in biochemical methane potential (BMP) tests from methane production curves. Fron. Environ. Sci. 2019, 7, 178. [Google Scholar] [CrossRef]
- Castro, H.; Queirolo, M.; Quevedo, M.; Muxi, L. Preservation methods for the storage of anaerobic sludges. Biotechnol. Lett. 2002, 24, 329–333. [Google Scholar] [CrossRef]
- Hagen, L.H.; Vivekanand, V.; Pope, P.B.; Eijsink, V.G.; Horn, S.J. The effect of storage conditions on microbial community composition and biomethane potential in a biogas starter culture. Appl. Microbiol. Biotechnol. 2015, 99, 5749–5761. [Google Scholar] [CrossRef] [PubMed]
- Filer, J.; Ding, H.H.; Chang, S. Biochemical methane potential (BMP) assay method for anaerobic digestion research. Water 2019, 11, 921. [Google Scholar] [CrossRef]
- Raposo, F.; De La Rubia, M.A.; Fernández-Cegrí, V.; Borja, R. Anaerobic digestion of solid organic substrates in batch mode: An overview relating to methane yields and experimental procedures. Renew. Sustain. Energy Rev. 2012, 16, 861–877. [Google Scholar] [CrossRef]
- Ohemeng-Ntiamoah, J.; Datta, T. Perspectives on variabilities in biomethane potential test parameters and outcomes: A review of studies published between 2007 and 2018. Sci. Total Environ. 2019, 664, 1052–1062. [Google Scholar] [CrossRef]
- Wang, B.; Strömberg, S.; Nges, I.A.; Nistor, M.; Liu, J. Impacts of inoculum pre-treatments on enzyme activity and biochemical methane potential. J. Biosci. Bioeng. 2016, 121, 557–560. [Google Scholar] [CrossRef]
- Li, J.; Zicari, S.M.; Cui, Z.; Zhang, R. Processing anaerobic sludge for extended storage as anaerobic digester inoculum. Bioresour. Technol. 2014, 166, 201–210. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef]
- Chen, J.L.; Ortiz, R.; Steele, T.W.J.; Stuckey, D.C. Toxicants inhibiting anaerobic digestion: A review. Biotechnol. Adv. 2014, 32, 1523–1534. [Google Scholar] [CrossRef]
- Rice, E.W.; Baird, R.B.; Eaton, A.D.; Clesceri, L.S. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- Hafner, S.D.; Astals, S.; Buffiere, P.; Løjborg, N.; Holliger, C.; Koch, K.; Weinrich, S. Calculation of Methane Production from Manometric Measurements. Standard BMP Methods document 202, version 2.5. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Hafner, S.D.; Koch, K.; Carrere, H.; Astals, S.; Weinrich, S.; Rennuit, C. Software for biogas research: Tools for measurement and prediction of methane production. SoftwareX 2018, 7, 205–210. [Google Scholar] [CrossRef]
- Lu, Y.; Liaquat, R.; Astals, S.; Jensen, P.; Batstone, D.; Tait, S. Relationship between microbial community, operational factors and ammonia inhibition resilience in anaerobic digesters at low and moderate ammonia background concentrations. New Biotechnol. 2018, 44, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, L.; Veiga, P.; Figueroa, M.; Alonso-Gutierrez, J.; Stams, A.J.; Lema, J.M.; Carballa, M. Relationship between microbial activity and microbial community structure in six full-scale anaerobic digesters. Microbiol. Res. 2012, 167, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Yamazawa, A.; Iikura, T.; Morioka, Y.; Shino, A.; Ogata, Y.; Date, Y.; Kikuchi, J. Cellulose Digestion and Metabolism Induced Biocatalytic Transitions in Anaerobic Microbial Ecosystems. Metabolites 2013, 4, 36–52. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Liu, T.; Müller, B.; Schnürer, A. The microbial community structure in industrial biogas plants influences the degradation rate of straw and cellulose in batch tests. Biotechnol. Biofuels 2016, 9, 128. [Google Scholar] [CrossRef]
- Batstone, D.J.; Keller, J.; Angelidaki, I.; Kalyuzhnyi, S.V.; Pavlostathis, S.G.; Rozzi, A.; Sanders, W.T.; Siegrist, H.; Vavilin, V.A. The IWA Anaerobic Digestion Model No 1 (ADM1). Water Sci. Technol. 2002, 45, 65–73. [Google Scholar] [CrossRef]
- Pavlostathis, S.G.; Giraldo-Gomez, E. Kinetics of anaerobic treatment: A critical review. Crit. Rev. Environ. Control. 1991, 21, 411–490. [Google Scholar] [CrossRef]
- Strübing, D.; Moeller, A.B.; Mößnang, B.; Lebuhn, M.; Drewes, J.E.; Koch, K. Anaerobic thermophilic trickle bed reactor as a promising technology for flexible and demand-oriented H2/CO2 biomethanation. Appl. Energy 2018, 232, 543–554. [Google Scholar] [CrossRef]
- Hülsemann, B.; Zhou, L.; Merkle, W.; Hassa, J.; Müller, J.; Oechsner, H. Biomethane Potential Test: Influence of Inoculum and the Digestion System. Appl. Sci. 2020, 10, 2589. [Google Scholar] [CrossRef]
| Storage Temperature | Storage Time | Test | Inoculum | Substrate | Observations | Ref. |
|---|---|---|---|---|---|---|
| −20 °C, freeze-drying, 4 °C and room temperature | 2 and 5 months | SMA | Psychrophilic anaerobic lagoon 1 | Glucose Acetate |
| [13] |
| 37 °C | 35 days | BMP | Mesophilic DSS 2 | Primary sludge |
| [11] |
| Room temperature | 2 and 4 months | BMP | Thermophilic OFMSW digester 3 | OFMSW Non-fat dry milk |
| [19] |
| −20 °C, 4 °C and room temperature | 1 week; 1, 2, 6 and 11 months | BMP | Mesophilic co-digester (OFMSW and cow manure) 5 | Cellulose |
| [14] |
| 4 °C and 37 °C | 5 days | BMP | Mesophilic DSS | Cellulose Wheat straw |
| [18] |
| 4 °C and 38 °C | 2 weeks | BMP | Mesophilic DSS | Pelleted dog food |
| [12] |
| Parameter | Units | DPM | DSS (Batch 1) | DSS (Batch 2) |
|---|---|---|---|---|
| TS | g TS·kg−1 | 6.5 | 27.0 | 28.6 |
| VS | g VS·kg−1 | 4.2 | 18.8 | 20.5 |
| tCOD | g COD·kg−1 | 6.3 | 27.4 | 31.4 |
| sCOD | g COD·kg−1 | 0.3 | 0.1 | 0.1 |
| pH | - | 7.5 | 7.5 | 7.4 |
| Alkalinity | g CaCO3·L−1 | 1.4 | n.d. | 5.2 |
| VFA | mg VFA·L−1 | 5 | 29 | 23 |
| TAN | mg N·L−1 | 177 | 1105 | 1246 |
| Phosphate | mg P·L−1 | 82 | 183 | 145 |
| Experiment | Inoculum | Storage Temperature | Storage Time |
|---|---|---|---|
| 1 | DPM | Non-stored (control) 1 Refrigerator (4 °C) Refrigerator (4 °C) + 1 day at 37 °C Incubator at 37 °C | 8 days 2 |
| 2 | DSS | Non-stored (control) 1 Refrigerator (4 °C) Refrigerator (4 °C) + 1 day at 37 °C Incubator at 37 °C | 7 days |
| 3 | DSS | Non-stored (control) 1 Refrigerator (4 °C) Room temperature (22 °C) Incubator at 37 °C | 7, 14 and 21 days |
| Experiment | Inoculum | Non-Stored (Control) | Refrigerator (4 °C) | Refrigerator (4 °C) + 1 Day at 37 °C | Incubator (37 °C) |
|---|---|---|---|---|---|
| 1 | DPM | 0.127 ± 0.005 | 0.128 ± 0.005 | 0.123 ± 0.003 | 0.126 ± 0.006 |
| 2 | DSS | 0.128 ± 0.004 | 0.133 ± 0.003 | 0.121 ± 0.004 | 0.103 ± 0.005 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Astals, S.; Koch, K.; Weinrich, S.; Hafner, S.D.; Tait, S.; Peces, M. Impact of Storage Conditions on the Methanogenic Activity of Anaerobic Digestion Inocula. Water 2020, 12, 1321. https://doi.org/10.3390/w12051321
Astals S, Koch K, Weinrich S, Hafner SD, Tait S, Peces M. Impact of Storage Conditions on the Methanogenic Activity of Anaerobic Digestion Inocula. Water. 2020; 12(5):1321. https://doi.org/10.3390/w12051321
Chicago/Turabian StyleAstals, Sergi, Konrad Koch, Sören Weinrich, Sasha D. Hafner, Stephan Tait, and Miriam Peces. 2020. "Impact of Storage Conditions on the Methanogenic Activity of Anaerobic Digestion Inocula" Water 12, no. 5: 1321. https://doi.org/10.3390/w12051321
APA StyleAstals, S., Koch, K., Weinrich, S., Hafner, S. D., Tait, S., & Peces, M. (2020). Impact of Storage Conditions on the Methanogenic Activity of Anaerobic Digestion Inocula. Water, 12(5), 1321. https://doi.org/10.3390/w12051321









