Improving Inter-Laboratory Reproducibility in Measurement of Biochemical Methane Potential (BMP)
Abstract
1. Introduction
- Quantify and partition observed variability in BMP measured using a standardized protocol;
- Assess possible sources of error in BMP measurement, including inocula, calculation errors, and systematic measurement biases;
- Test and revise BMP validation criteria based on a quantitative analysis of collected data.
2. Materials and Methods
2.1. Project Structure
2.2. Participating Laboratories
2.3. Test Substrates
2.4. BMP Measurement
2.4.1. First Study (S1)
2.4.2. Second Study (S2)
2.4.3. Measurement Methods
2.5. Data Submission
2.6. Data Analysis and Calculations
2.6.1. Data Processing
2.6.2. Data Subsets
- A.
- Reported BMP. For S1, submitted standardized CH4 volume at the duration identified by participants (1 value per bottle) was used to calculate BMP along with data on bottle contents and laboratory-measured VS concentrations. For S2, participating laboratories provided BMP directly in spreadsheet templates. For both studies, participants identified the time that met the requested duration criterion based on their own calculations.
- B.
- Reported BMP under reference conditions. Thus, a subset of A. For 5 laboratories that used multiple biogas measurement methods, only results from their typical method were retained (with the exception of one laboratory that used both manometric and gravimetric methods, because of a dearth of gravimetric results). ISR was limited to values given in the study protocol (Section 2.4.1 and Section 2.4.2), and mean BMP values based on less than 3 substrate bottles or less than 3 blanks were excluded. However, the use of multiple inocula per laboratory was permitted, and therefore, there was an unequal number of BMP values among laboratories. Because of apparent difficulty in exactly matching requested ISR values, limits were somewhat lenient: nominal ISR was taken as 2 for 1.5 < ISR < 2.5, and 4 for 3 < ISR < 5. For CEL, ISR > 0.7 was accepted.
- C.
- Calculated BMP at reported durations. Mean BMP values (n = 3 substrate bottles) were calculated from submitted raw data at durations that exactly matched those in set A, using laboratory-measured VS concentrations. These values were aligned (merged) with reported values to assess calculation and data entry errors by participants. Values that could not be matched and any with missing replicates were dropped.
- D.
- Calculated BMP at various durations. BMP values were calculated from all submitted raw data and median substrate VS concentrations for various relative and fixed durations. This subset was used to compare duration criteria.
- E.
- Calculated BMP under reference conditions. A subset of D, but with the same constraints as in subset B. Only 1% net 3 d (E1) and final (latest available) durations (E2) were used.
- F.
- Calculated BMP with laboratory-measured VS. As with E1 but calculated with laboratory-measured substrate VS concentrations. This set was compared to E1 to assess the contribution of VS measurement error to inter-laboratory variability in BMP.
- G.
- Calculated BMP from inoculum comparisons. A subset of D from trials where inocula were compared. BMP was taken as 1% net values. This set was divided into two for analysis based on how the inoculum exchange was carried out: a single inoculum shared among more than two labs in each country (G1) or exchange of two inocula between two labs per country (G2) (Section 2.4.2).
- H.
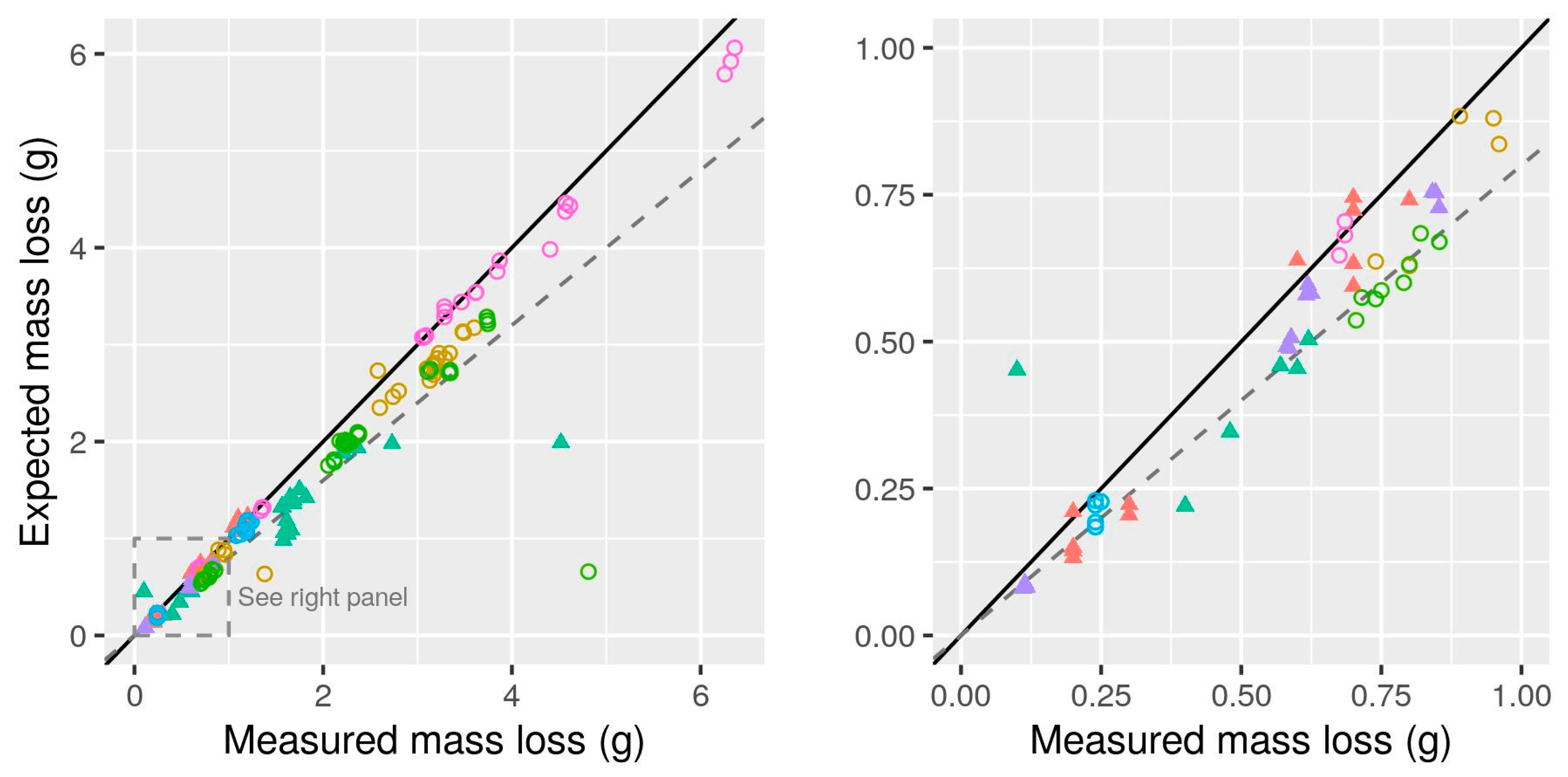
- Calculated CH4 production with gravimetric evaluation. Results from tests where the initial and final mass of each bottle was determined by participating laboratories. Only the final (latest) values were used in order to evaluate measurements using total mass loss over the duration of the trial. Unlike the other subsets, the response variable of interest was total CH4 production, calculated for each bottle.
- I.
- Calculated BMP from 3 laboratories which compared measurement methods. Unlike the other subsets, 1% net 3 d BMP was calculated separately for each bottle so methods could be compared. One laboratory compared three methods, while the others used two. Four methods were used in total: automated volumetric (AMPTS II and a different system) and manual volumetric, manometric, and gravimetric.
- J.
- Calculated BMP at the 1% net 3 d duration from 14 laboratories that varied ISR within BMP tests. A subset of D.
2.6.3. Data Analysis and Display
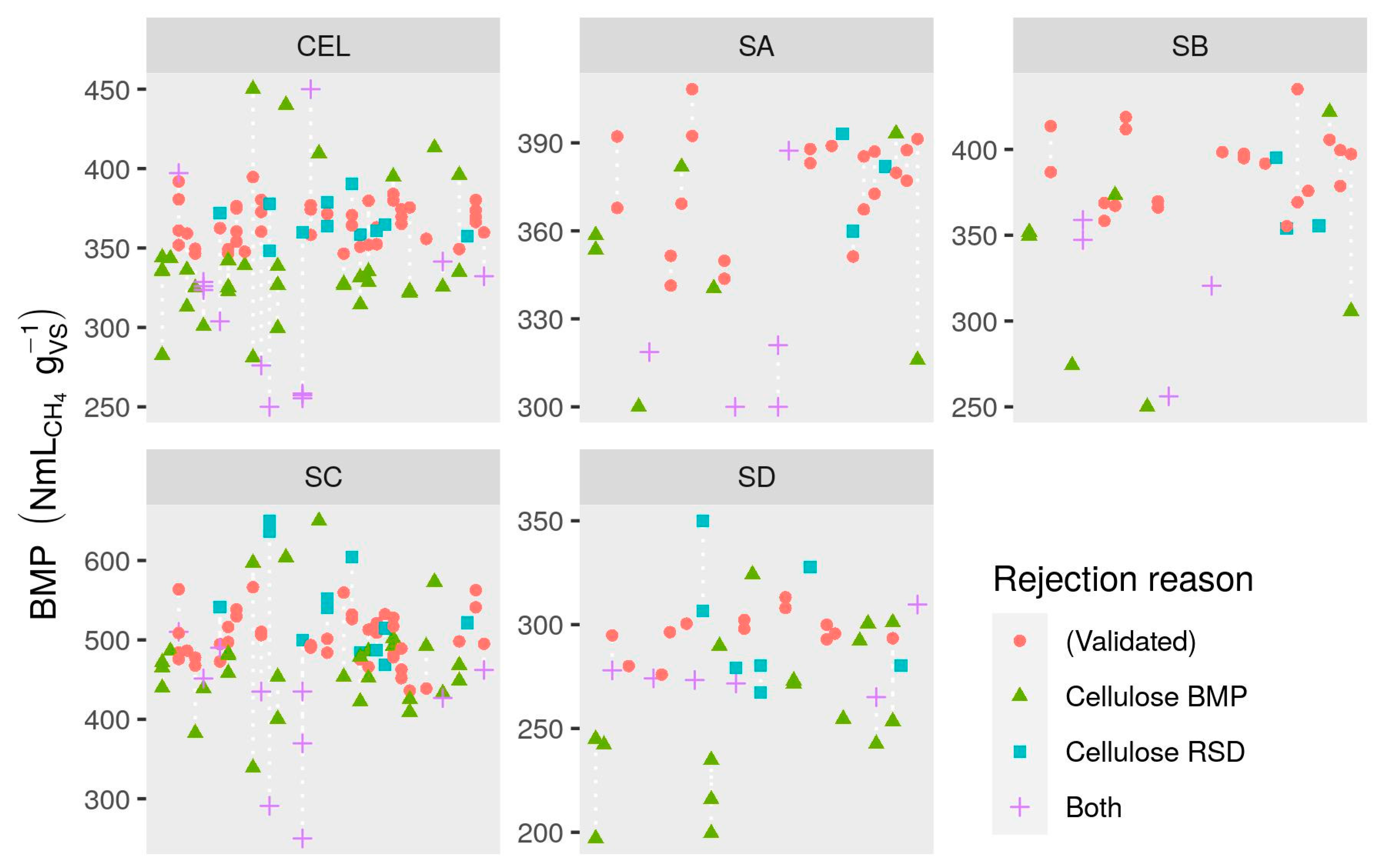
2.6.4. Validation Criteria
- i.
- Duration at least 1% net 3 d;
- ii.
- Blank SMP RSD ≤ 5%;
- iii.
- Cellulose RSDB ≤ 5%;
- iv.
- Substrate RSDB ≤ 5%;
- v.
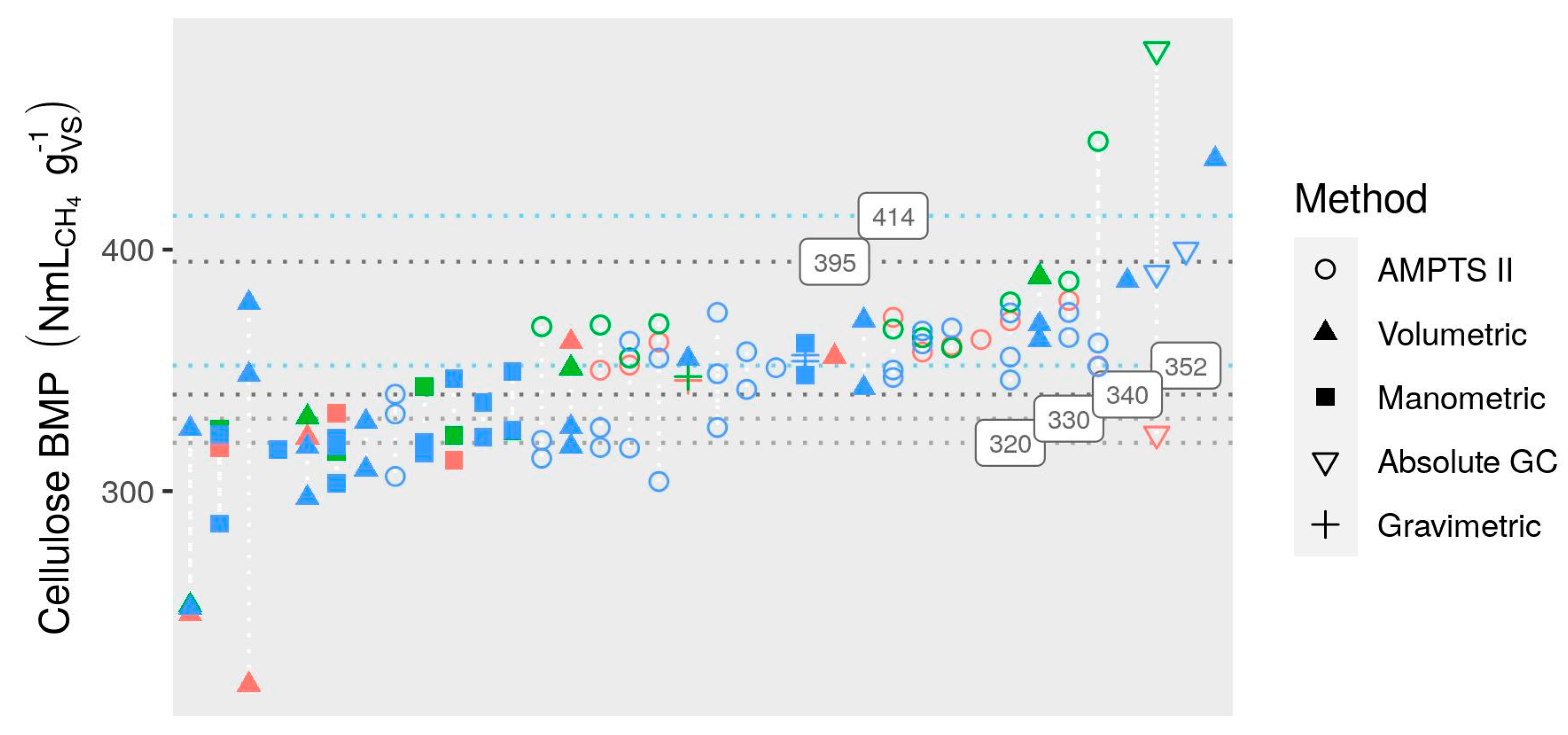
- Cellulose BMP between 352 and 414 NmLCH4 gVS−1.
- i.
- Duration at least 1% net 3 d;
- ii.
- Cellulose RSDB ≤ 6%;
- iii.
- Cellulose BMP between 320 and 414 NmLCH4 gVS−1.
- i.
- Duration at least 1% net 3 d;
- ii.
- Cellulose RSDB ≤ 6%;
- iii.
- Cellulose BMP between 340 and 395 NmLCH4 gVS−1.
3. Results and discussion
3.1. Data Set Overview
3.1.1. Data Set Size
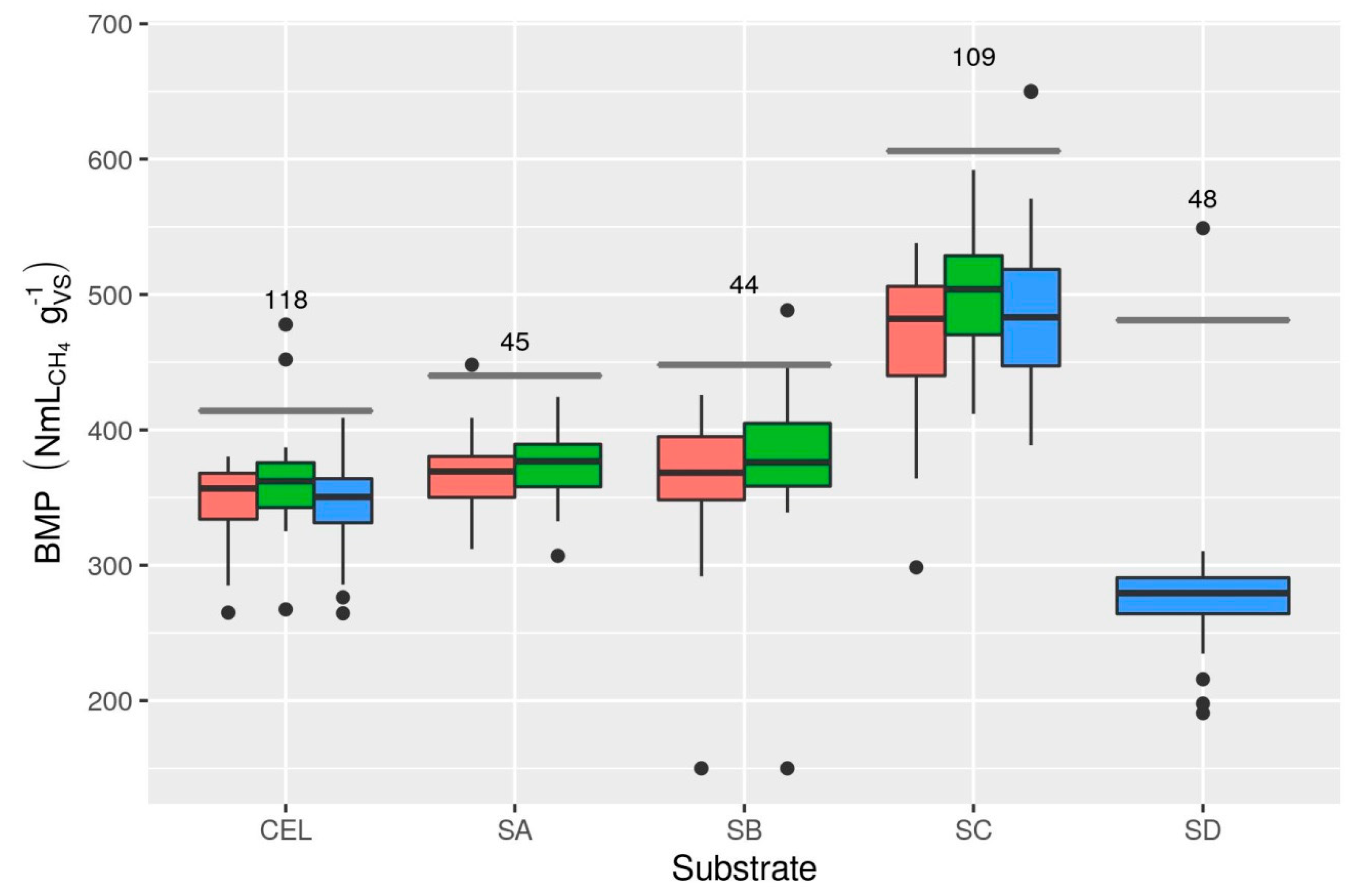
3.1.2. BMP Reproducibility
3.2. Evaluation of Sources of Error
3.2.1. Volatile Solids Measurement
3.2.2. Data Processing
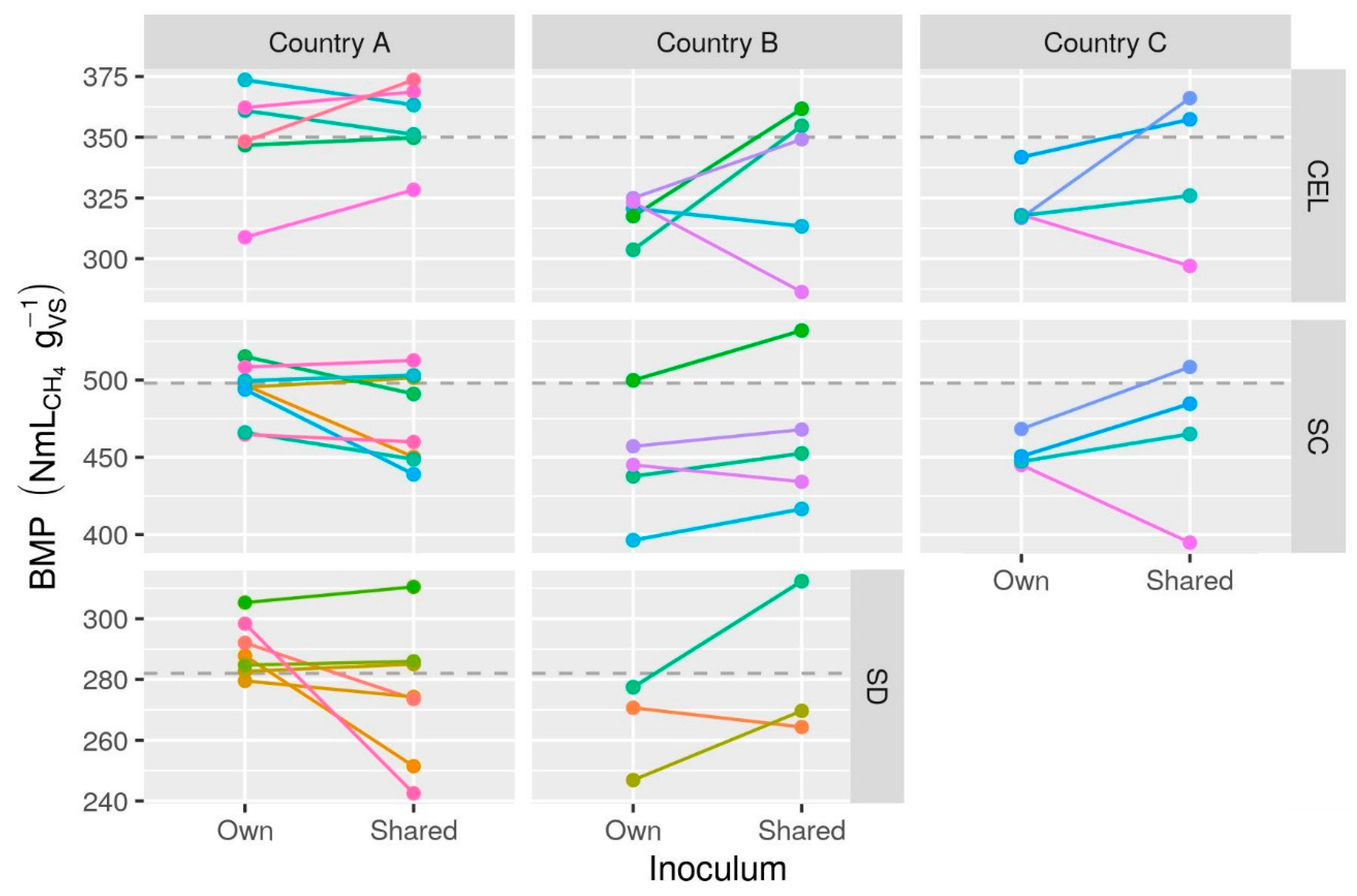
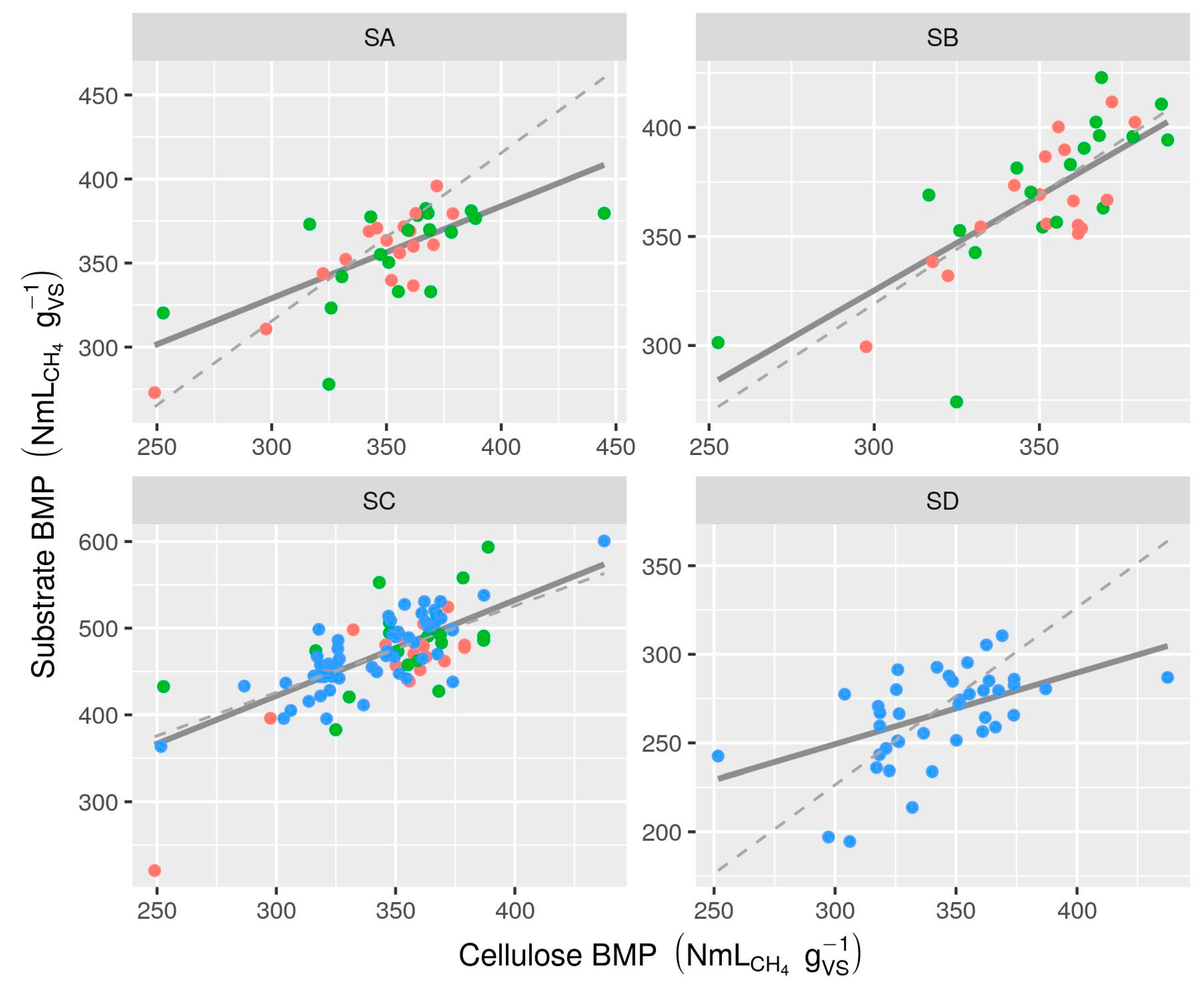
3.2.3. Inoculum Effects
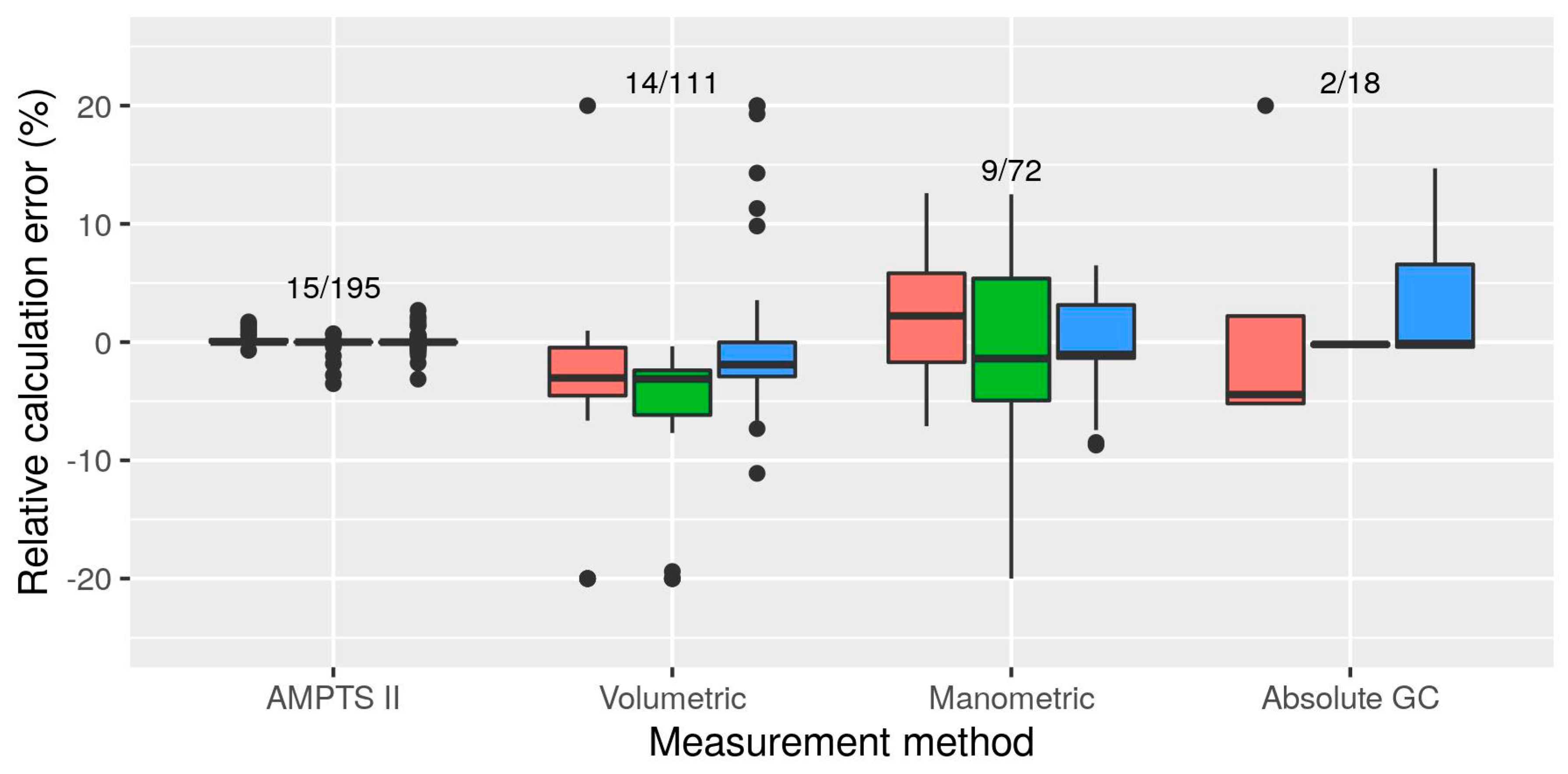
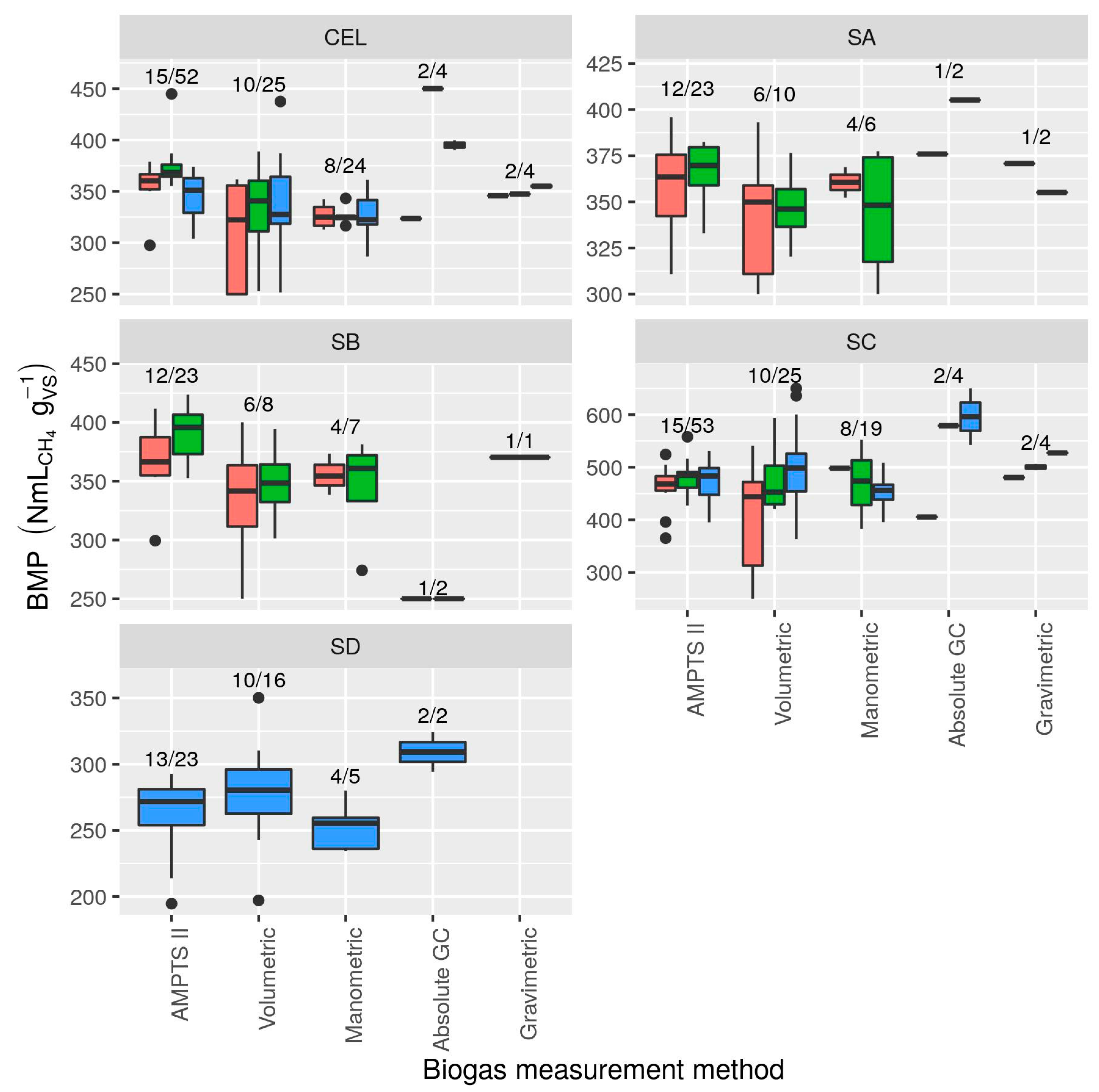
3.2.4. Measurement Methods
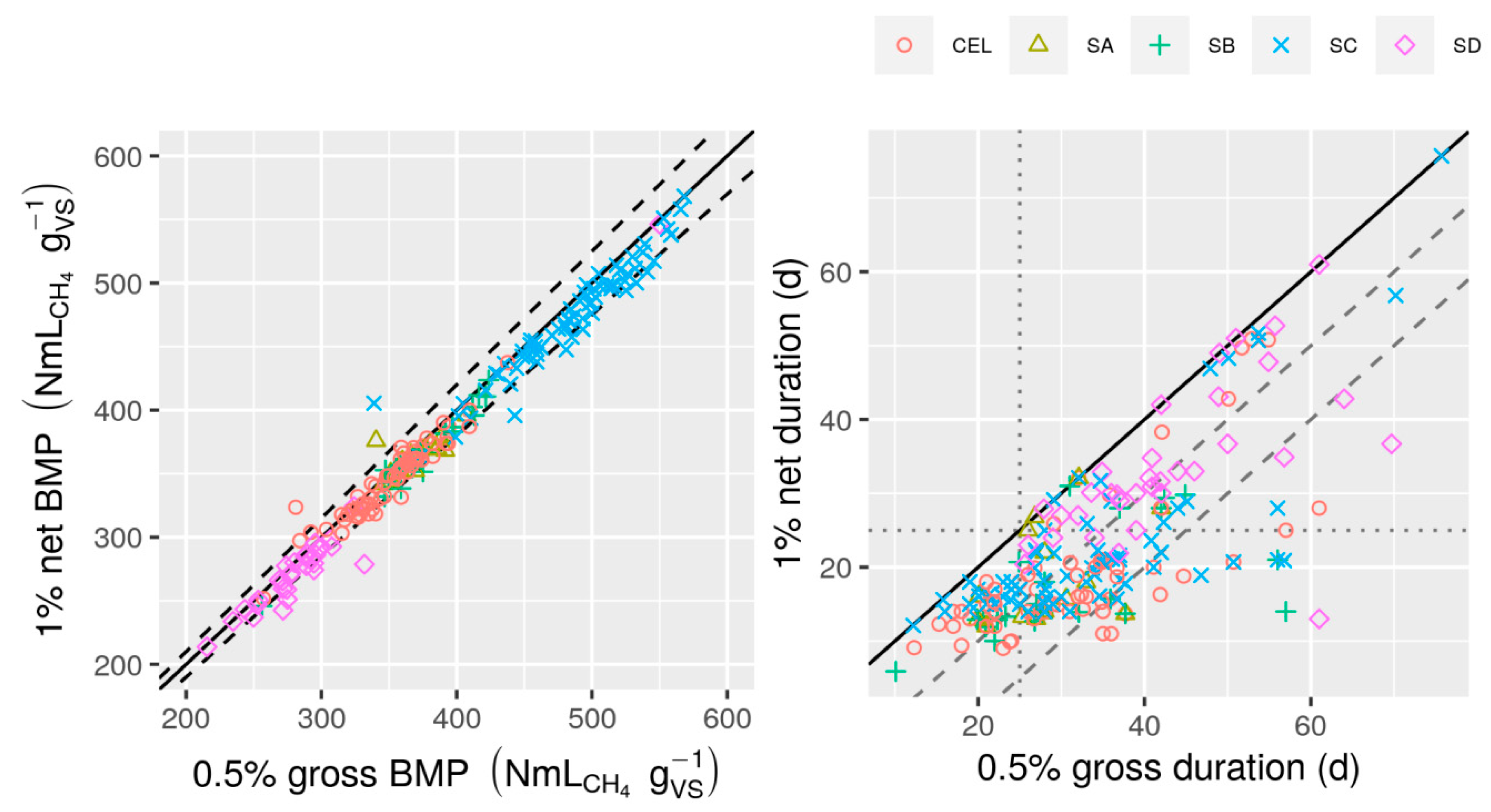
3.3. Evaluation of Duration Criteria
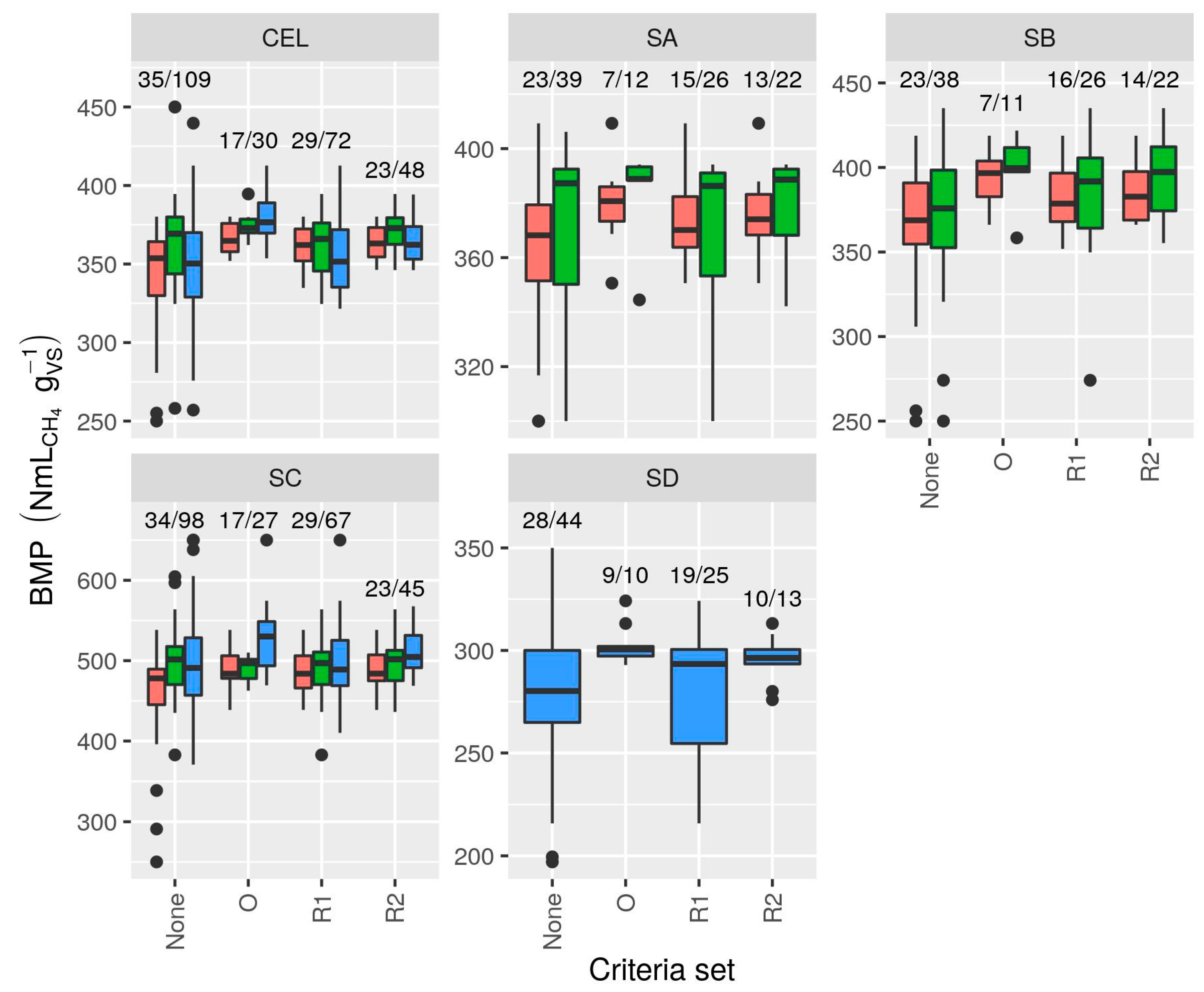
3.4. Validation Criteria
3.4.1. Cellulose BMP Limits
3.4.2. Random Error in Cellulose BMP (RSDB)
3.4.3. Random Error in Blanks and Substrate BMP
3.4.4. Evaluation of Validation Criteria
3.5. Recommendations
3.5.1. BMP Method Standardization
3.5.2. BMP Measurement Methods
3.5.3. Data Processing and Reporting
- i.
- Duration at least 1% net 3 d (by substrate);
- ii.
- Cellulose BMP RSDB ≤ 6%;
- iii.
- Cellulose BMP between 340 and 395 NmLCH4 gVS−1.
4. Summary and Conclusions
- Even with the use of a single protocol, inter-laboratory variability was a significant problem, inflated by a small number of extreme values. Relative standard deviation among laboratories (RSDR) was as high as 24%, and relative range 130%.
- The validation criteria proposed by Holliger et al. [19], based on duration, mean cellulose BMP and variability in methane production from blanks, cellulose, and substrate were together effective in substantially reducing inter-laboratory variability. However, the majority of all BMP values were rejected by application of these criteria, including many that were apparently accurate.
- Errors in data processing calculations or data entry (which are difficult to separate) were moderate, or in some cases, major sources of error in BMP. Additionally, calculation of relative standard deviation for BMP values was done inconsistently among laboratories. Use of standardized approaches for data processing, as well as checking of calculations using standardized software, is strongly recommended.
- There was evidence of differences among measurement methods even after re-calculation of all BMP values from original measurements: manual manometric and manual volumetric methods had a tendency to result in slightly (if not consistently) lower BMP values (as much as 14% below mean AMPTS II results). Evaluation of some manual methods based on mass loss measurement showed that negative bias was common (10% on average). Assessment of measurement biases, e.g., by comparing to gravimetric measurements, is recommended. Moderate correlation between substrate BMP and BMP of cellulose in the same test suggests that specific practices of laboratories or even technicians may be an underlying cause of observed inter-lab variability and that cellulose BMP is a promising indicator for validation. However, correlation is only moderate and cellulose results should not be used to “correct” potentially biased BMP measurements.
- There was virtually no evidence of consistent effects of inoculum source on BMP, and any potential effects were much smaller than variation among labs. It is unlikely that inoculum source contributed substantially to observed inter-lab variation, suggesting that selection of a suitable inoculum was not a challenge for the participating laboratories. Large effects of inoculum source found in some other studies may be due to unusually ineffective inocula or insufficient duration and therefore may not be representative of typical BMP tests.
- The best BMP duration criterion of those considered was when the daily net (after subtracting estimated inoculum contribution) CH4 production (or production rate in mL d−1) drops below 1% of cumulative net CH4 production for at least 3 consecutive days (the “1% net 3 d” duration). Resulting BMP values were close to those from more stringent criteria, but duration was usually much shorter.
- Based on evaluation of calculated BMP values, new validation criteria were proposed, and are recommended for use in all BMP tests:
- a.
- Duration at least 1% net 3 d;
- b.
- Cellulose RSDB ≤ 6%;
- c.
- Cellulose BMP between 340 and 395 NmLCH4 gVS−1.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carrere, H.; Antonopoulou, G.; Affes, R.; Passos, F.; Battimelli, A.; Lyberatos, G.; Ferrer, I. Review of feedstock pretreatment strategies for improved anaerobic digestion: From lab-scale research to full-scale application. Bioresour. Technol. 2016, 199, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Ariunbaatar, J.; Panico, A.; Esposito, G.; Pirozzi, F.; Lens, P.N.L. Pretreatment methods to enhance anaerobic digestion of organic solid waste. Appl. Energy 2014, 123, 143–156. [Google Scholar] [CrossRef]
- Holliger, C.; Fruteau de Laclos, H.; Hack, G. Methane production of full-scale anaerobic digestion plants calculated from substrate’s biomethane potentials compares well with the one measured on-site. Front. Energy Res. 2017, 5. [Google Scholar] [CrossRef]
- Li, C.; Nges, I.A.; Lu, W.; Wang, H. Assessment of the degradation efficiency of full-scale biogas plants: A comparative study of degradation indicators. Bioresour. Technol. 2017, 244, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Lindorfer, H.; Pérez López, C.; Resch, C.; Braun, R.; Kirchmayr, R. The impact of increasing energy crop addition on process performance and residual methane potential in anaerobic digestion. Water Sci. Technol. 2007, 56, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Ruile, S.; Schmitz, S.; Mönch-Tegeder, M.; Oechsner, H. Degradation efficiency of agricultural biogas plants—A full-scale study. Bioresour. Technol. 2015, 178, 341–349. [Google Scholar] [CrossRef]
- Owen, W.F.; Stuckey, D.C.; Healy, J.B., Jr.; Young, L.Y.; McCarty, P.L. Bioassay for monitoring biochemical methane potential and anaerobic toxicity. Water Res. 1979, 13, 485–492. [Google Scholar] [CrossRef]
- Hansen, T.L.; Schmidt, J.E.; Angelidaki, I.; Marca, E.; Jansen, J.L.C.; Mosbæk, H.; Christensen, T.H. Method for determination of methane potentials of solid organic waste. Waste Manag. 2004, 24, 393–400. [Google Scholar] [CrossRef]
- Justesen, C.G.; Astals, S.; Mortensen, J.R.; Thorsen, R.; Koch, K.; Weinrich, S.; Triolo, J.M.; Hafner, S.D. Development and validation of a low-cost gas density method for measuring biochemical methane potential (bmp). Water 2019, 11, 2431. [Google Scholar] [CrossRef]
- Pabón Pereira, C.P.; Castañares, G.; Van Lier, J.B. An OxiTop® protocol for screening plant material for its biochemical methane potential (BMP). Water Sci. Technol. 2012, 66, 1416–1423. [Google Scholar] [CrossRef]
- Verein Deutsch er Ingenieure e.V. Fermentation of Organic Materials: Characterisation of the Substrate, Sampling, Collection of Material Data, Fermentation Tests; Verein Deutsch er Ingenieure e.V.: Düsseldorf, Germany, 2016. [Google Scholar]
- Hafner, S.D.; Rennuit, C.; Triolo, J.M.; Richards, B.K. Validation of a simple gravimetric method for measuring biogas production in laboratory experiments. Biomass Bioenergy 2015, 83, 297–301. [Google Scholar] [CrossRef]
- Strömberg, S.; Nistor, M.; Liu, J. Towards eliminating systematic errors caused by the experimental conditions in Biochemical Methane Potential (BMP) tests. Waste Manag. 2014, 34, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Cresson, R.; Pommier, S.; Beline, F.; Bouchez, T.; Bougrier, C.; Buffière, P.; Pauss, A.; Pouech, P.; Preys, S.; Ribeiro, T. Results from a French Inter-Laboratory Campaign on the Biological Methane Potential of Solid Substrates. Available online: https://hal.archives-ouvertes.fr/hal-01947463 (accessed on 17 June 2020).
- Raposo, F.; Fernandez-Cegri, V.; De la Rubia, M.A.; Borja, R.; Beline, F.; Cavinato, C.; Demirer, G.; Fernandez, B.; Fernandez-Polanco, M.; Frigon, J.C.; et al. Biochemical methane potential (BMP) of solid organic substrates: Evaluation of anaerobic biodegradability using data from an international interlaboratory study. J. Chem. Technol. Biotechnol. 2011, 86, 1088–1098. [Google Scholar] [CrossRef]
- Weinrich, S.; Schäfer, F.; Liebetrau, J.; Bochmann, G.; Paterson, M.; Oechsner, H.; Tillmann, P. Value of Batch Tests for Biogas Potential Analysis: Method Comparison and Challenges of Substrate and Efficiency Evaluation of Biogas Plants; IEA Bioenergy: Paris, France, 2018; ISBN 978-1-910154-49-6. [Google Scholar]
- Angelidaki, I.; Alves, M.; Bolzonella, D.; Borzacconi, L.; Campos, J.L.; Guwy, A.J.; Kalyuzhnyi, S.; Jenicek, P.; Van Lier, J.B. Defining the biomethane potential (BMP) of solid organic wastes and energy crops: A proposed protocol for batch assays. Water Sci. Technol. 2009, 59, 927–934. [Google Scholar] [CrossRef]
- Verein Deutsch er Ingenieure e.V. Fermentation of Organic Materials; Verein Deutsch er Ingenieure e.V.: Düsseldorf, Germany, 2006. [Google Scholar]
- Holliger, C.; Alves, M.; Andrade, D.; Angelidaki, I.; Astals, S.; Baier, U.; Bougrier, C.; Buffière, P.; Carballa, M.; De Wilde, V.; et al. Towards a standardization of biomethane potential tests. Water Sci. Technol. 2016, 74, 2515–2522. [Google Scholar] [CrossRef] [PubMed]
- Filer, J.; Ding, H.H.; Chang, S. Biochemical Methane Potential (BMP) Assay Method for Anaerobic Digestion Research. Water 2019, 11, 921. [Google Scholar] [CrossRef]
- Pearse, L.F.; Hettiaratchi, J.P.; Kumar, S. Towards developing a representative biochemical methane potential (BMP) assay for landfilled municipal solid waste—A review. Bioresour. Technol. 2018, 254, 312–324. [Google Scholar] [CrossRef]
- Raposo, F.; De la Rubia, M.A.; Fernandez-Cegri, V.; Borja, R. Anaerobic digestion of solid organic substrates in batch mode: An overview relating to methane yields and experimental procedures. Renew. Sustain. Energy Rev. 2012, 16, 861–877. [Google Scholar] [CrossRef]
- Koch, K.; Lippert, T.; Drewes, J.E. The role of inoculum’s origin on the methane yield of different substrates in biochemical methane potential (BMP) tests. Bioresour. Technol. 2017, 243, 457–463. [Google Scholar] [CrossRef]
- De Vrieze, J.; Raport, L.; Willems, B.; Verbrugge, S.; Volcke, E.; Meers, E.; Angenent, L.T.; Boon, N. Inoculum selection influences the biochemical methane potential of agro-industrial substrates. Microb. Biotechnol. 2015, 8, 776–786. [Google Scholar] [CrossRef]
- Hülsemann, B.; Zhou, L.; Merkle, W.; Hassa, J.; Müller, J.; Oechsner, H. Biomethane Potential Test: Influence of Inoculum and the Digestion System. Appl. Sci. 2020, 10, 2589. [Google Scholar] [CrossRef]
- Dechrugsa, S.; Kantachote, D.; Chaiprapat, S. Effects of inoculum to substrate ratio, substrate mix ratio and inoculum source on batch co-digestion of grass and pig manure. Bioresour. Technol. 2013, 146, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Reilly, M.; Dinsdale, R.; Guwy, A. The impact of inocula carryover and inoculum dilution on the methane yields in batch methane potential tests. Bioresour. Technol. 2016, 208, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Hagen, L.H.; Vivekanand, V.; Pope, P.B.; Eijsink, V.G.H.; Horn, S.J. The effect of storage conditions on microbial community composition and biomethane potential in a biogas starter culture. Appl. Microbiol. Biotechnol. 2015, 99, 5749–5761. [Google Scholar] [CrossRef] [PubMed]
- Elbeshbishy, E.; Nakhla, G.; Hafez, H. Biochemical methane potential (BMP) of food waste and primary sludge: Influence of inoculum pre-incubation and inoculum source. Bioresour. Technol. 2012, 110, 18–25. [Google Scholar] [CrossRef]
- Astals, S.; Koch, K.; Weinrich, S.; Hafner, S.D.; Tait, S.; Peces, M. Impact of Storage Conditions on the Methanogenic Activity of Anaerobic Digestion Inocula. Water 2020, 12, 1321. [Google Scholar] [CrossRef]
- Fabbri, A.; Serranti, S.; Bonifazi, G. Biochemical methane potential (BMP) of artichoke waste: The inoculum effect. Waste Manag. Res. 2014, 32, 207–214. [Google Scholar] [CrossRef]
- Raposo, F.; Banks, C.J.; Siegert, I.; Heaven, S.; Borja, R. Influence of inoculum to substrate ratio on the biochemical methane potential of maize in batch tests. Process Biochem. 2006, 41, 1444–1450. [Google Scholar] [CrossRef]
- Rodriguez-Chiang, L.M.; Dahl, O.P. Effect of inoculum to substrate ratio on the methane potential of microcrystalline cellulose production wastewater. BioResources 2015, 10, 898–911. [Google Scholar] [CrossRef]
- Himanshu, H.; Voelklein, M.A.; Murphy, J.D.; Grant, J.; O’Kiely, P. Factors controlling headspace pressure in a manual manometric BMP method can be used to produce a methane output comparable to AMPTS. Bioresour. Technol. 2017, 238, 633–642. [Google Scholar] [CrossRef]
- Valero, D.; Montes, J.A.; Rico, J.L.; Rico, C. Influence of headspace pressure on methane production in Biochemical Methane Potential (BMP) tests. Waste Manag. 2016, 48, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Hafner, S.D.; Astals, S. Systematic error in manometric measurement of biochemical methane potential: Sources and solutions. Waste Manag. 2019, 91, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Kleinheinz, G.; Hernandez, J. Comparison of two laboratory methods for the determination of biomethane potential of organic feedstocks. J. Microbiol. Methods 2016, 130, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Pham, C.H.; Triolo, J.M.; Cu, T.T.T.; Pedersen, L.; Sommer, S.G. Validation and recommendation of methods to measure biogas production potential of animal manure. Asian Australas. J. Anim. Sci. 2013, 26, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Richards, B.K.; Cummings, R.J.; White, T.E.; Jewell, W.J. Methods for kinetic-analysis of methane fermentation in high solids biomass digesters. Biomass Bioenergy 1991, 1, 65–73. [Google Scholar] [CrossRef]
- Hafner, S.D.; Rennuit, C.; Olsen, P.J.; Pedersen, J.M. Quantification of leakage in batch biogas assays. Water Pract. Technol. 2018, 13, 52–61. [Google Scholar] [CrossRef]
- Svensson, K.; Kjørlaug, O.; Horn, S.J.; Agger, J.W. Comparison of approaches for organic matter determination in relation to expression of bio-methane potentials. Biomass Bioenergy 2017, 100, 31–38. [Google Scholar] [CrossRef]
- Fanelli, D. Do pressures to publish increase scientists’ bias? An empirical support from US states data. PLoS ONE 2010, 5. [Google Scholar] [CrossRef]
- International Organization for Standardization. Solid Biofuels—Determination of Total Content of Carbon, Hydrogen and Nitrogen (iso 16948:2015); International Organization for Standardization: Geneva, Switzerland, 2015. [Google Scholar]
- International Organization for Standardization. Solid Biofuels—Conversion of Analytical Results from One Basis to Another (iso 16993:2016); International Organization for Standardization: Geneva, Switzerland, 2016. [Google Scholar]
- Hafner, S.D.; Koch, K.; Carrere, H.; Astals, S.; Weinrich, S.; Rennuit, C. Software for biogas research: Tools for measurement and prediction of methane production. SoftwareX 2018, 7, 205–210. [Google Scholar] [CrossRef]
- Hafner, S.; Rennuit, C.; Justesen, C.G.; Løjborg, N.; Mortensen, J.R.; Biogas Package v. 1.24.3. Available online: https://github.com/sashahafner/biogas (accessed on 7 April 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Rittmann, B.E.; McCarty, P.L. Environmental Biotechnology: Principles and Applications; McGraw-Hill: Boston, MA, USA, 2001; ISBN 0-07-234553-5. [Google Scholar]
- Environmental Protection Agency. Method 1684 Total, Fixed, and Volatile Solids in Water, Solids, and Biosolids; U.S. Environmental Protection Agency, Office of Water, Office of Science and Technology Engineering and Analysis Division (4303): Washington, DC, USA, 2001.
- American Public Health Association, American Water Works Associate, and Water Environment Federation. Standard Methods for the Examination of Water and Wastewater, 21st ed.; APHA-AWWA-WEF: Washington, DC, USA, 2005; ISBN 978-0-87553-047-5. [Google Scholar]
- BioProcess Control. AMPTS II—Methane Potential Analysis Tool. Available online: https://www.bioprocesscontrol.com/products/ampts-ii (accessed on 19 June 2020).
- Rozzi, A.; Remigi, E. Methods of assessing microbial activity and inhibition under anaerobic conditions: A literature review. Rev. Environ. Sci. Biotechnol. 2004, 3, 93–115. [Google Scholar] [CrossRef]
- Hafner, S.D.; Astals, S.; Holliger, C.; Koch, K.; Weinrich, S. Calculation of Biochemical Methane Potential (BMP). Standard BMP Methods Document 200, Version 1.6. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Hafner, S.D.; Løjborg, N.; Astals, S.; Holliger, C.; Koch, K.; Weinrich, S. Calculation of Methane Production from Volumetric Measurements. Standard BMP Methods Document 201, Version 1.5. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Hafner, S.D.; Astals, S.; Buffiere, P.; Løjborg, N.; Holliger, C.; Koch, K.; Weinrich, S. Calculation of Methane Production from Manometric Measurements. Standard BMP Methods Document 202, Version 2.5. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Hafner, S.D.; Richards, B.K.; Astals, S.; Holliger, C.; Koch, K.; Weinrich, S. Calculation of Methane Production from Gravimetric Measurements. Standard BMP Methods Document 203, Version 1.0. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Hafner, S.D.; Justesen, C.; Thorsen, R.; Astals, S.; Holliger, C.; Koch, K.; Weinrich, S. Calculation of Methane Production from Gas Density-Based Measurements. Standard BMP Methods Document 204, Version 1.5. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Crowder, M. Interlaboratory comparisons: Round robins with random effects. J. R. Stat. Soc. Ser. C (Appl. Stat.) 1992, 41, 409–425. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis, 4th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999; ISBN 0-13-081542-X. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Faraway, J.J. Extending the Linear Model with R: Generalized Linear, Mixed Effects and Nonparametric Regression Models, 1st ed.; Chapman and Hall/CRC: Boca Raton, FL, USA, 2006; ISBN 1-58488-424-X. [Google Scholar]
- Faraway, J.J. Linear Models with R; Texts in statistical science; Chapman & Hall/CRC: Boca Raton, FL, USA, 2005; ISBN 1-58488-425-8. [Google Scholar]
- Rosenbaum, P.R. Design of Observational Studies; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-1-4419-1213-8. [Google Scholar]
- Bauer, D.F. Constructing Confidence Sets Using Rank Statistics. J. Am. Stat. Assoc. 1972, 67, 687–690. [Google Scholar] [CrossRef]
- Venables, W.N. Modern Applied Statistics with S Statistics and Computing, 4th ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95457-0. [Google Scholar]
- Kreuger, E.; Nges, I.A.; Bjornsson, L. Ensiling of crops for biogas production: Effects on methane yield and total solids determination. Biotechnol. Biofuels 2011, 4, 44. [Google Scholar] [CrossRef]
- Weissbach, F.; Strubelt, C. Correcting the dry matter content of maize silages as a substrate for biogas production. Landtechnik 2008, 63, 82–83. [Google Scholar]
- Raposo, F.; Borja, R.; Ibelli-Bianco, C. Predictive regression models for biochemical methane potential tests of biomass samples: Pitfalls and challenges of laboratory measurements. Renew. Sustain. Energy Rev. 2020, 127, 109890. [Google Scholar] [CrossRef]
- Wang, B.; Strömberg, S.; Nges, I.A.; Nistor, M.; Liu, J. Impacts of inoculum pre-treatments on enzyme activity and biochemical methane potential. J. Biosci. Bioeng. 2016, 121, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.; Hafner, S.D.; Weinrich, S.; Astals, S. Identification of critical problems in biochemical methane potential (BMP) tests from methane production curves. Front. Environ. Sci. 2019, 7, 178. [Google Scholar] [CrossRef]
- Weinrich, S. Praxisnahe Modellierung von Biogasanlagen: Systematische Vereinfachung des Anaerobic Digestion Model No. 1 (ADM1); Universität Rostock: Rostock, Germany, 2018. [Google Scholar]
- Weinrich, S.; Nelles, M. Critical comparison of different model structures for the applied simulation of the anaerobic digestion of agricultural energy crops. Bioresour. Technol. 2015, 178, 306–312. [Google Scholar] [CrossRef]
- Batstone, D.J.; Keller, J.; Angelidaki, I.; Kalyuzhnyi, S.V.; Pavlostathis, S.G.; Rozzi, A.; Sanders, W.; Siegrist, H.; Vavilin, V. Anaerobic Digestion Model No. 1 (ADM1), Report no. 13; International Water Association: London, UK, 2002. [Google Scholar]
- Mata-Alvarez, J. (Ed.) Fundamentals of the Anaerobic Digestion Process. In Biomethanization of the Organic Fraction of Municipal Solid Wastes; International Water Associate: London, UK, 2005; Volume 4, pp. 1–20. ISBN 978-1-78040-299-4. [Google Scholar]
- Amodeo, C.; Hafner, S.D.; Franco, R.T.; Benbelkacem, H.; Moretti, P.; Bayard, R.; Buffière, P. How different are manometric, gravimetric and automated volumetric BMP results? Water 2020. in preparation. [Google Scholar]
- Holliger, C.; Fruteau de Laclos, H.; Hafner, S.D.; Koch, K.; Weinrich, S.; Astals, S.; Alves, M.; Andrade, D.; Angelidaki, I.; Appels, L.; et al. Requirements for Measurement of Biochemical Methane Potential (BMP). Standard BMP Methods Document 100, Version 1.3. Available online: https://www.dbfz.de/en/BMP (accessed on 19 April 2020).
- Shah, T.A.; Ullah, R. Pretreatment of wheat straw with ligninolytic fungi for increased biogas productivity. Int. J. Environ. Sci. Technol. 2019, 16, 7497–7508. [Google Scholar] [CrossRef]
| Substrate Key | Tests | Description | TS (% FM) * | VS (% TS) † | Chemical Formula ‡ | Theoretical Max. BMP (NmLCH4 gVS−1) ¶ |
|---|---|---|---|---|---|---|
| SA | T1 | Animal feed | 88.8 | 93.3 | C17H30O12N | 440 |
| SB | T1 | Animal feed | 89.1 | 97.2 | C23H37O16N | 448 |
| SC | T1, T2 | Animal feed | 92.8 | 87.9 | C18H31O8N | 606 |
| SD | T2 | Wheat straw | 92.2 | 93.8 | C59H88O38N | 481 |
| CEL | T1, T2 | Microcrystalline cellulose | 94.9/99.0 ‖ | 100.0 | C6H10O5 | 414 |
| Subset | No. Labs | No. Countries | No. BMP Tests | No. Observations * |
|---|---|---|---|---|
| A | 37 | 15 | 124 | 444 |
| B | 36 | 14 | 117 | 359 |
| C † | 37 | 15 | 122 | 410 |
| D ‡ | 37 | 15 | 123 | 412 |
| E ¶ and F | 36 | 14 | 116 | 344 |
| G1 | 15 | 3 | 30 | 84 |
| G2 | 6 | 3 | 12 | 32 |
| H | 7 | 7 | 14 | 154 |
| I | 3 | 3 | 12 | 109 |
| J | 14 | 9 | 22 | 66 |
| Study | Test | Substrate | No. Labs | No. Obs. | No. Extreme * | Mean BMP (NmLCH4 gVS−1) | RSDR (%) † | RR (%) ‡ |
|---|---|---|---|---|---|---|---|---|
| S1 | T1 | CEL | 23 | 23 | 0 | 346 | 9.0 | 32 |
| S1 | T1 | SA | 22 | 22 | 0 | 368 | 8.9 | 37 |
| S1 | T1 | SB | 21 | 21 | 1 | 353 | 24.3 | 114 |
| S1 | T1 | SC | 20 | 21 | 1 | 465 | 12.9 | 50 |
| S1 | T2 | CEL | 22 | 22 | 2 | 365 | 11.4 | 58 |
| S1 | T2 | SA | 22 | 22 | 0 | 374 | 7.5 | 31 |
| S1 | T2 | SB | 22 | 22 | 2 | 372 | 22.6 | 121 |
| S1 | T2 | SC | 20 | 22 | 0 | 500 | 9.5 | 36 |
| S2 | T1 | CEL | 36 | 72 | 0 | 346 | 7.7 | 41 |
| S2 | T1 | SC | 35 | 65 | 3 | 494 | 13.1 | 81 |
| S2 | T1 | SD | 29 | 47 | 3 | 279 | 17.3 | 128 |
| Study | Test | Substrate | Validated * (%) | Mean BMP (NmLCH4 gVS−1) | RSDR (%) | RR (%) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| O | R2 | O | R2 | O | R2 | O | R2 | |||
| S1 | T1 | CEL | 41 | 55 | 367 | 363 | 2.9 | 3.1 | 8 | 9 |
| S1 | T1 | SA | 37 | 53 | 380 | 375 | 4.7 | 4.6 | 15 | 16 |
| S1 | T1 | SB | 32 | 53 | 394 | 385 | 4.8 | 4.8 | 13 | 14 |
| S1 | T1 | SC | 47 | 63 | 486 | 487 | 6.2 | 5.7 | 21 | 21 |
| S1 | T2 | CEL | 29 | 57 | 375 | 371 | 3.0 | 3.9 | 9 | 13 |
| S1 | T2 | SA | 25 | 60 | 382 | 378 | 5.5 | 5.3 | 13 | 13 |
| S1 | T2 | SB | 26 | 63 | 398 | 394 | 6.1 | 6.4 | 16 | 20 |
| S1 | T2 | SC | 30 | 65 | 490 | 501 | 3.8 | 7.1 | 10 | 25 |
| S2 | T1 | CEL | 23 | 36 | 380 | 365 | 4.5 | 3.6 | 16 | 13 |
| S2 | T1 | SC | 20 | 34 | 542 | 512 | 14.2 | 5.5 | 55 | 20 |
| S2 | T1 | SD | 23 | 30 | 303 | 296 | 3.1 | 3.4 | 10 | 13 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hafner, S.D.; Fruteau de Laclos, H.; Koch, K.; Holliger, C. Improving Inter-Laboratory Reproducibility in Measurement of Biochemical Methane Potential (BMP). Water 2020, 12, 1752. https://doi.org/10.3390/w12061752
Hafner SD, Fruteau de Laclos H, Koch K, Holliger C. Improving Inter-Laboratory Reproducibility in Measurement of Biochemical Methane Potential (BMP). Water. 2020; 12(6):1752. https://doi.org/10.3390/w12061752
Chicago/Turabian StyleHafner, Sasha D., Hélène Fruteau de Laclos, Konrad Koch, and Christof Holliger. 2020. "Improving Inter-Laboratory Reproducibility in Measurement of Biochemical Methane Potential (BMP)" Water 12, no. 6: 1752. https://doi.org/10.3390/w12061752
APA StyleHafner, S. D., Fruteau de Laclos, H., Koch, K., & Holliger, C. (2020). Improving Inter-Laboratory Reproducibility in Measurement of Biochemical Methane Potential (BMP). Water, 12(6), 1752. https://doi.org/10.3390/w12061752








