Hydrochemical Characteristics and Water Quality Evaluation of Rivers in Different Regions of Cities: A Case Study of Suzhou City in Northern Anhui Province, China
Abstract
:1. Introduction
2. Materials and Methods
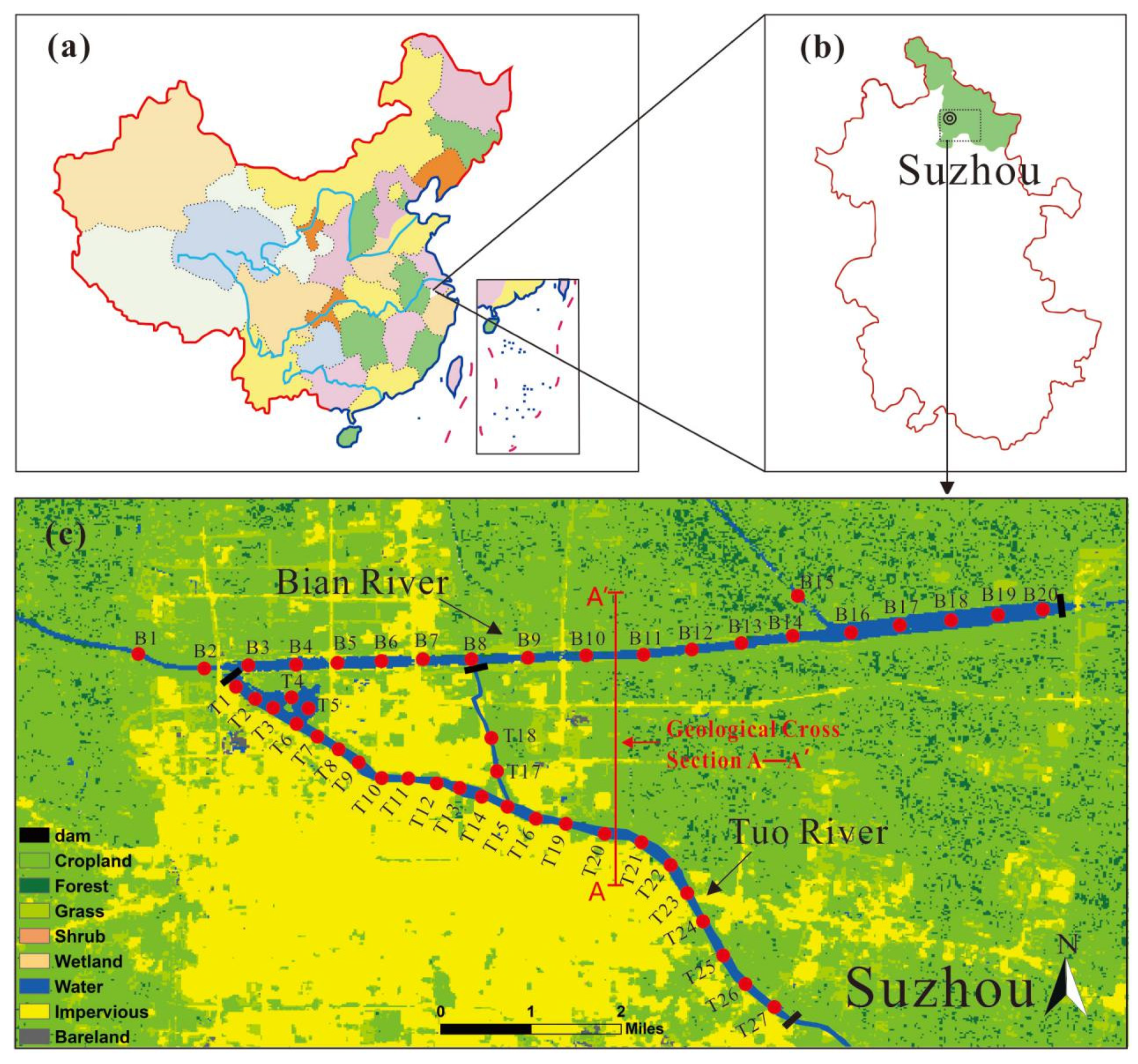
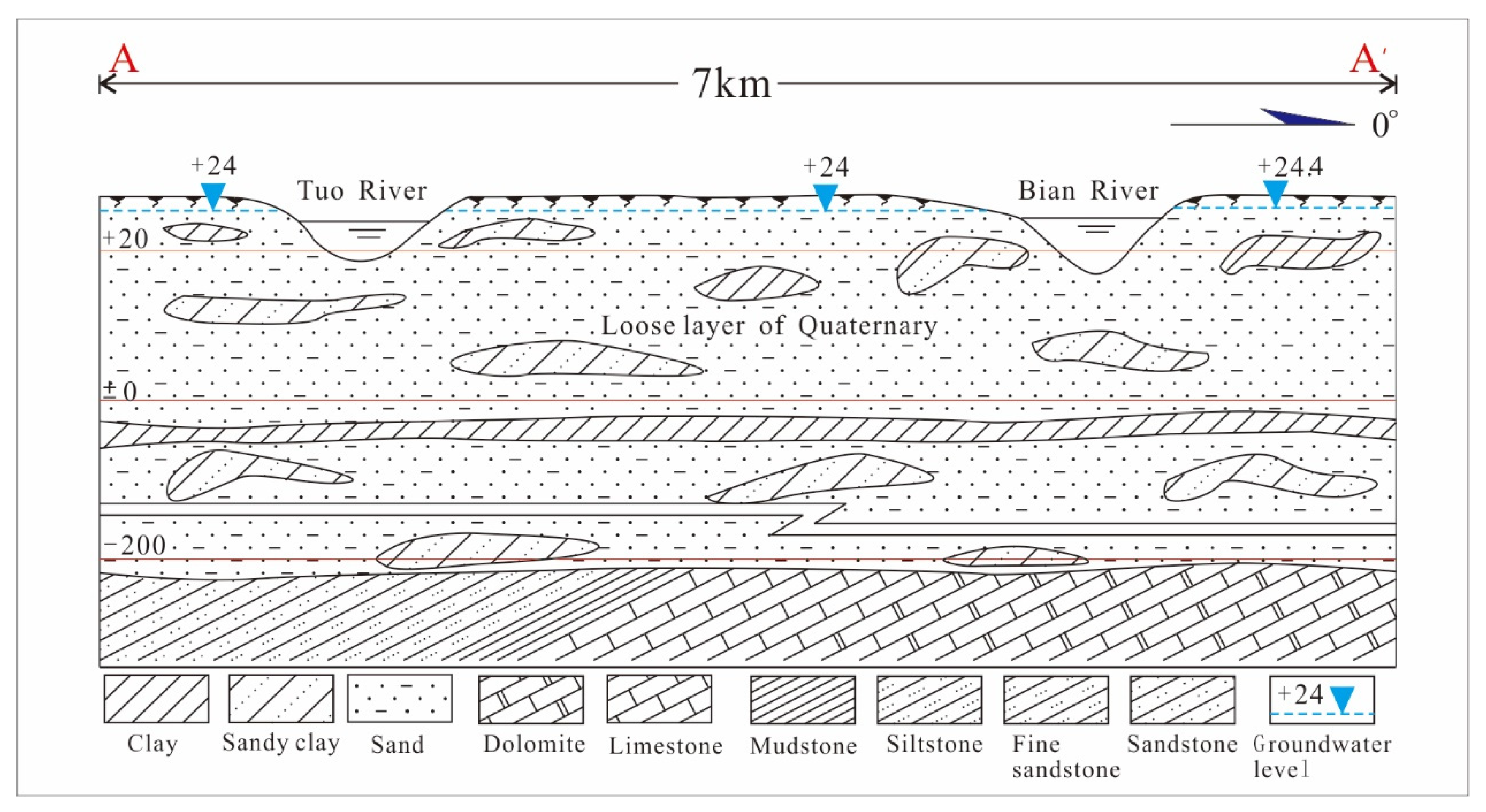
2.1. Study Area
2.2. Sampling and Testing
2.3. Software
2.4. Improved Fuzzy Comprehensive Evaluation
- Class I:
- Class II–IV:
- Class V
2.5. Water Quality Evaluation of Irrigation Water
3. Results
4. Discussion
4.1. Sources and Influencing Factors for Major Ions
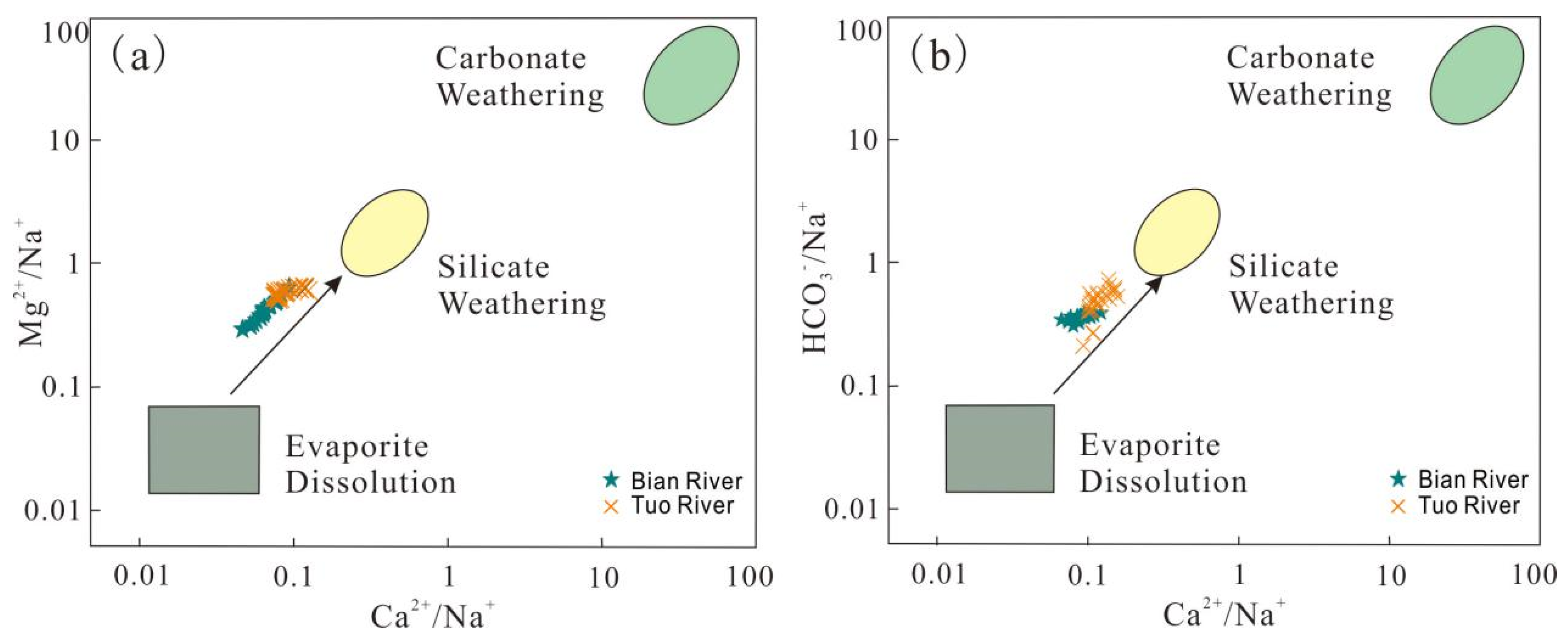
4.1.1. Processes Controlling River Solute
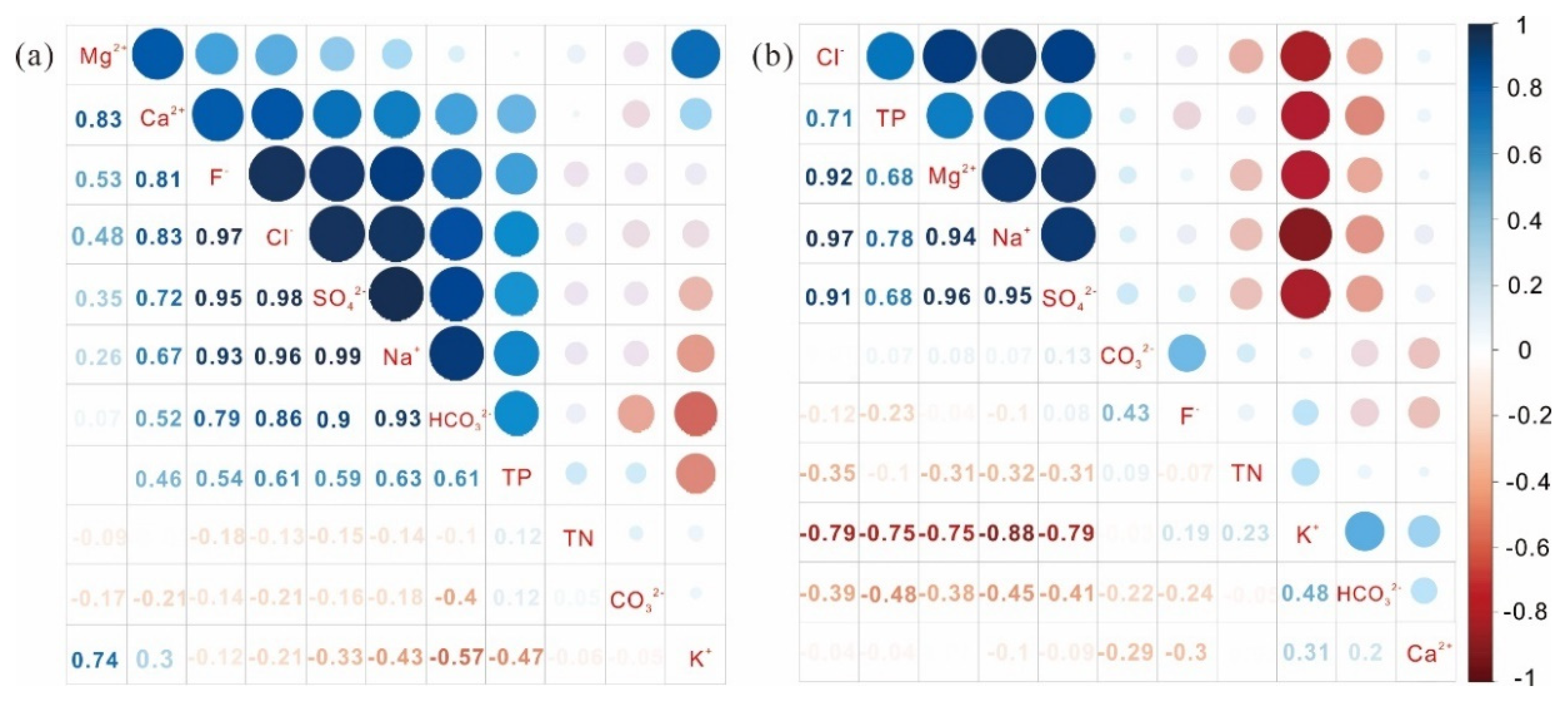
4.1.2. Correlation Analysis of Water Quality Parameters
4.1.3. Analysis of River Replenishment Sources
4.2. Water Quality Evaluation of the Study Area
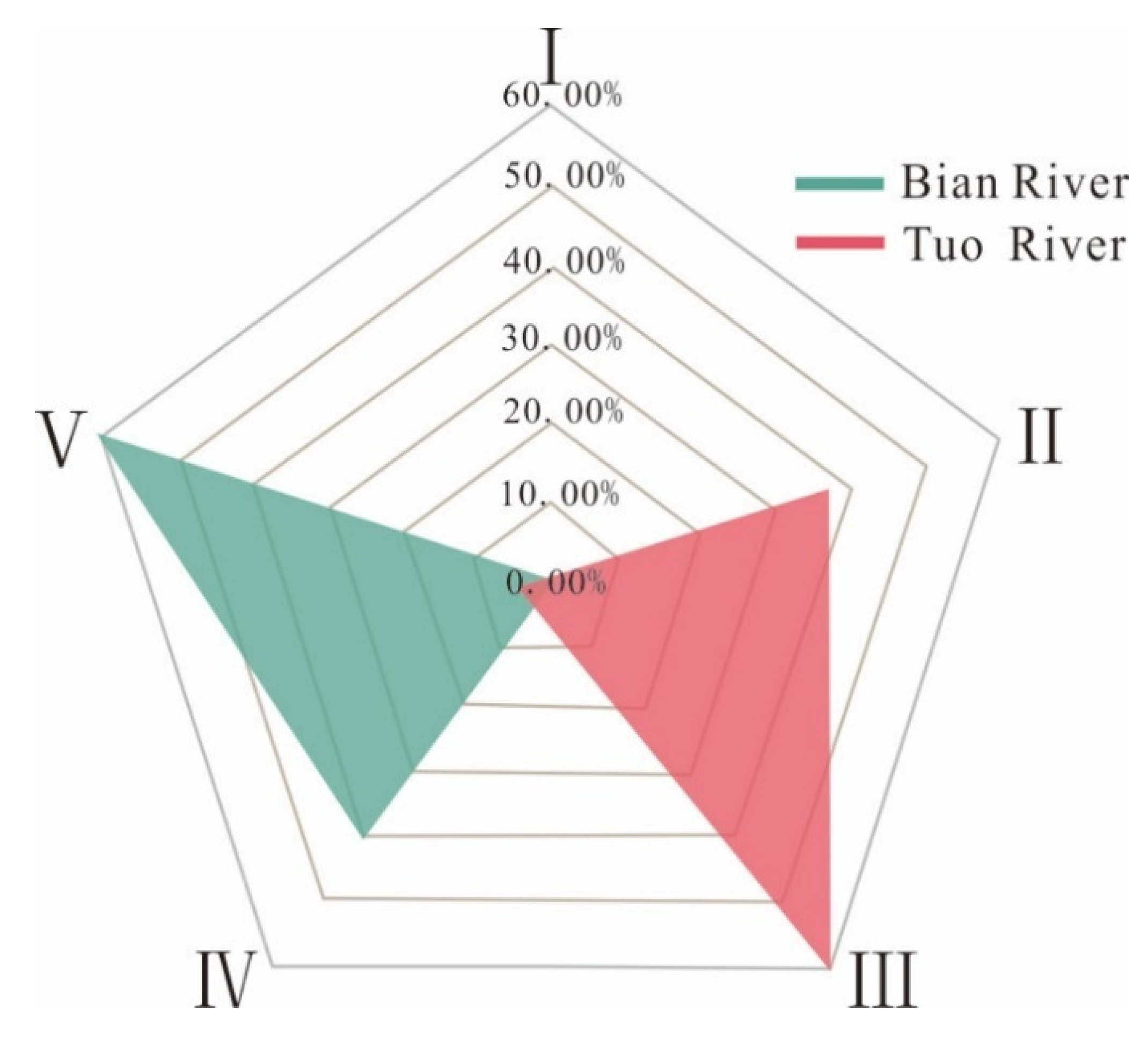
4.2.1. Water Quality Grade Evaluation
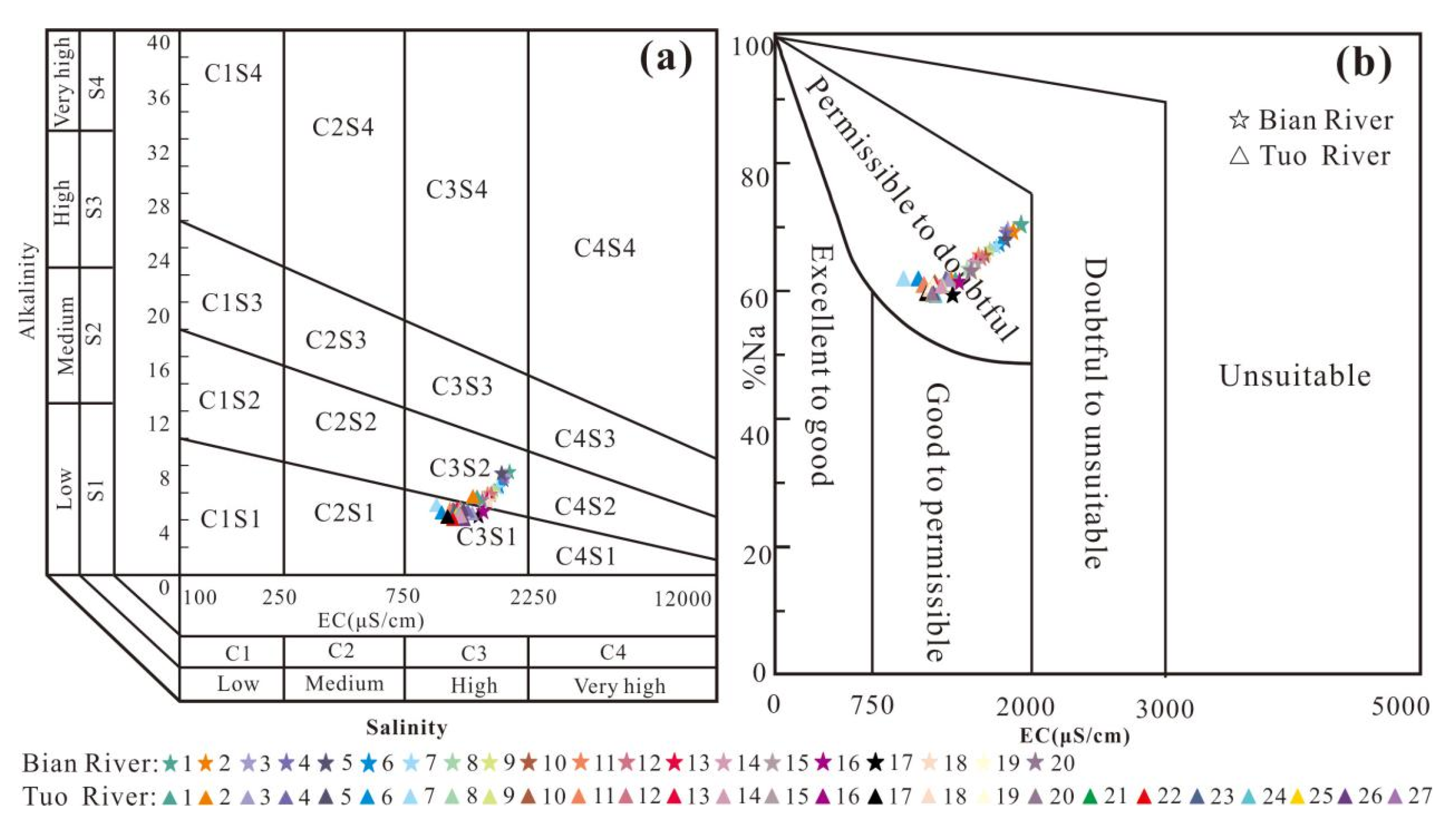
4.2.2. Water Quality Evaluation of Irrigation Water
4.3. Implications for Water Resource Management
5. Conclusions
- (1)
- The content of cations in the Tuo River and Bian River of the study area changed to Na+ > Mg2+ > K+ > Ca2+, and the content of anions changed to SO42− > HCO3− > Cl− > CO32− > F−. The contents of main ions and nutrition indexes in the Bian River are higher than those in the Tuo River, especially the contents of Na+, K+, SO42−, Cl−, Ca2+, and TP (ANOVA, p < 0.001).
- (2)
- The hydrochemical types of the two rivers were the SO4-Cl-Na type. The chemical composition of the two rivers was mainly mainly affected by silicate weathering; there is a partial influence of evaporate dissolution. The results of correlation analysis show that Bian River is greatly affected by agricultural non-point source pollution. The analysis of hydrogen and oxygen isotopes shows that precipitation is the supply source of the Bian River and Tuo River.
- (3)
- The results of fuzzy comprehensive evaluation showed that the water quality of the Tuo River water sample was good, which was suitable for fish breeding, landscape entertainment and industrial and agricultural water; while that of the Bian River water sample was poor, which may only be suitable for general agricultural irrigation or need to be selected or treated according to the purpose of water use. The evaluation results of irrigation water quality showed that the water samples of the Tuo River were high in salt and low in alkali. When the soil leaching conditions were good, they could be used for irrigation, while the water samples of the Bian River were high in salt and alkali, suitable for irrigating plants with strong salt tolerance.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vorosmarty, C.J. Global Water Resources: Vulnerability from Climate Change and Population Growth. Science 2000, 289, 284–288. [Google Scholar] [CrossRef] [Green Version]
- Jasechko, S.; Perrone, D.; Befus, K.M.; Cardenas, M.B.; Ferguson, G.; Gleeson, T.; Luijendijk, E.; McDonnell, J.J.; Taylor, R.G.; Wada, Y.; et al. Global aquifers dominated by fossil groundwaters but wells vulnerable to modern contamination. Nat. Geosci. 2017, 10, 425–429. [Google Scholar] [CrossRef] [Green Version]
- Meng, Z.; Yang, Y.; Qin, Z.; Huang, L. Evaluating Temporal and Spatial Variation in Nitrogen Sources along the Lower Reach of Fenhe River (Shanxi Province, China) Using Stable Isotope and Hydrochemical Tracers. Water 2018, 10, 231. [Google Scholar] [CrossRef] [Green Version]
- Torres Lopez, S.; de los Angeles Barrionuevo, M.; Rodriguez-Labajos, B. Water accounts in decision-making processes of urban water management: Benefits, limitations and implications in a real implementation. Sustain. Cities Soc. 2019, 50, 101676. [Google Scholar] [CrossRef]
- Chen, J.; Wang, F.; Xia, X.; Zhang, L. Major element chemistry of the Changjiang (Yangtze River). Chem. Geol. 2002, 187, 231–255. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, Y.; Li, Y.; Zwahlen, F.; Boillat, J. Hydrogeochemical characteristics of central Jianghan Plain, China. Environ. Earth Sci. 2013, 68, 765–778. [Google Scholar]
- Fu, C.; Li, X.; Ma, J.; Liu, L.; Gao, M.; Bai, Z. A hydrochemistry and multi-isotopic study of groundwater origin and hydrochemical evolution in the middle reaches of the Kuye River basin. Appl. Geochem. 2018, 98, 82–93. [Google Scholar] [CrossRef]
- Kanduc, T.; Kocman, D.; Ogrinc, N. Hydrogeochemical and stable isotope characteristics of the River Idrijca (Slovenia), the boundary watershed between the Adriatic and Black Seas. Aquat. Geochem. 2008, 14, 239–262. [Google Scholar] [CrossRef]
- Rashid, I.; Romshoo, S.A. Impact of anthropogenic activities on water quality of Lidder River in Kashmir Himalayas. Environ. Monit. Assess. 2013, 185, 4705–4719. [Google Scholar] [CrossRef]
- Misaghi, F.; Delgosha, F.; Razzaghmanesh, M.; Myers, B. Introducing a water quality index for assessing water for irrigation purposes: A case study of the Ghezel Ozan River. Sci. Total Environ. 2017, 589, 107–116. [Google Scholar] [CrossRef]
- Hua, K.; Xiao, J.; Li, S.; Li, Z. Analysis of hydrochemical characteristics and their controlling factors in the Fen River of China. Sustain. Cities Soc. 2020, 52, 101827. [Google Scholar] [CrossRef]
- Rose, S. The effects of urbanization on the hydrochemistry of base flow within the Chattahoochee River Basin (Georgia, USA). J. Hydrol. 2007, 341, 42–54. [Google Scholar] [CrossRef]
- Lu, X.; Li, L.Y.; Lei, K.; Wang, L.; Zhai, Y.; Zhai, M. Water quality assessment of Wei River, China using fuzzy synthetic evaluation. Environ. Earth Sci. 2010, 60, 1693–1699. [Google Scholar] [CrossRef]
- Qin, H.; Gao, B.; He, L.; Hu, X.; Dong, L.; Sanjay, D.; Dong, A.; Sun, Z.; Wan, W. Hydrogeochemical Characteristics and Controlling Factors of the Lhasa River under the Influence of Anthropogenic Activities. Water 2019, 11, 948. [Google Scholar] [CrossRef] [Green Version]
- Qiu, H.; Gui, H.; Cui, L.; Pan, Z.; Lu, B. Hydrogeochemical characteristics and water quality assessment of shallow groundwater: A case study from Linhuan coal-mining district in northern Anhui Province, China. Water Sci. Technol.-Water Supply 2019, 19, 1572–1578. [Google Scholar] [CrossRef]
- Qiu, H.; Gui, H. Heavy metals contamination in shallow groundwater of a coal-mining district and a probabilistic assessment of its human health risk. Hum. Ecol. Risk Assess. Int. J. 2019, 25, 548–563. [Google Scholar] [CrossRef]
- Wang, Y.; Sheng, D.; Wang, D.; Ma, H.; Wu, J.; Xu, F. Variable Fuzzy Set Theory to Assess Water Quality of the Meiliang Bay in Taihu Lake Basin. Water Resour. Manag. 2014, 28, 867–880. [Google Scholar] [CrossRef]
- Dahiya, S.; Singh, B.; Gaur, S.; Garg, V.K.; Kushwaha, H.S. Analysis of groundwater quality using fuzzy synthetic evaluation. J. Hazard. Mater. 2007, 147, 938–946. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, B.; Teng, D.; Yang, W.; Qiu, D. Comprehensive evaluation method of groundwater environment in a mining area based on fuzzy set theory. Geosyst. Eng. 2018, 21, 103–112. [Google Scholar] [CrossRef]
- Masoud, A.A.; El-Horiny, M.M.; Atwia, M.G.; Gemail, K.S.; Koike, K. Assessment of groundwater and soil quality degradation using multivariate and geostatistical analyses, Dakhla Oasis, Egypt. J. Afr. Earth Sci. 2018, 142, 64–81. [Google Scholar] [CrossRef]
- Jalali, M. Groundwater geochemistry in the Alisadr, Hamadan, western Iran. Environ. Monit. Assess. 2010, 166, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xiao, J.; Evaristo, J.; Li, Z. Spatiotemporal variations in the hydrochemical characteristics and controlling factors of streamflow and groundwater in the Wei River of China. Environ. Pollut. 2019, 254, 113006. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011; pp. 104–108. [Google Scholar]
- Liu, S.; Ryu, D.; Webb, J.A.; Lintern, A.; Waters, D.; Guo, D.; Western, A.W. Characterisation of spatial variability in water quality in the Great Barrier Reef catchments using multivariate statistical analysis. Mar. Pollut. Bull. 2018, 137, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Bu, H.; Song, X.; Zhang, Y. Using multivariate statistical analyses to identify and evaluate the main sources of contamination in a polluted river near to the Liaodong Bay in Northeast China. Environ. Pollut. 2019, 245, 1058–1070. [Google Scholar] [CrossRef]
- Rodrigues, V.; Estrany, J.; Ranzini, M.; de Cicco, V.; Tarjuelo Martin-Benito, J.M.; Hedo, J.; Lucas-Borja, M.E. Effects of land use and seasonality on stream water quality in a small tropical catchment: The headwater of Corrego Agua Limpa, Sao Paulo (Brazil). Sci. Total Environ. 2018, 622, 1553–1561. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Mei, K.; Dahlgren, R.A.; Wang, T.; Gong, J.; Zhang, M. Impacts of land use and population density on seasonal surface water quality using a modified geographically weighted regression. Sci. Total Environ. 2016, 572, 450–466. [Google Scholar] [CrossRef] [Green Version]
- Adimalla, N. Groundwater Quality for Drinking and Irrigation Purposes and Potential Health Risks Assessment: A Case Study from Semi-Arid Region of South India. Expo. Health 2019, 11, 109–123. [Google Scholar] [CrossRef]
- Ma, L.; Abuduwaili, J.; Li, Y.; Uulu, S.A.; Mu, S. Hydrochemical Characteristics and Water Quality Assessment for the Upper Reaches of Syr Darya River in Aral Sea Basin, Central Asia. Water 2019, 11, 1893. [Google Scholar] [CrossRef] [Green Version]
- Reeburgh, W.S. Global environment: Water, air, and geochemical cycles (BR). Int. J. Environ. Stud. 1996, 70, 155–156. [Google Scholar]
- Gibbs, R.J. Mechanisms Controlling World Water Chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef]
- Neogi, B.; Singh, A.K.; Pathak, D.D.; Chaturvedi, A. Hydrogeochemistry of coal mine water of North Karanpura coalfields, India: Implication for solute acquisition processes, dissolved fluxes and water quality assessment. Environ. Earth Sci. 2017, 76, 489. [Google Scholar] [CrossRef]
- Marghade, D.; Malpe, D.B.; Zade, A.B. Major ion chemistry of shallow groundwater of a fast growing city of Central India. Environ. Monit. Assess. 2012, 184, 2405–2418. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Rishi, M.S.; Herojeet, R.; Kaur, L.; Priyanka; Sharma, K. Multivariate analysis and geochemical signatures of groundwater in the agricultural dominated taluks of Jalandhar district, Punjab, India. J. Geochem. Explor. 2020, 208, 106395. [Google Scholar] [CrossRef]
- Pan, G.; Li, X.; Zhang, J.; Liu, Y.; Liang, H. Groundwater-flow-system characterization with hydrogeochemistry: A case in the lakes discharge area of the Ordos Plateau, China. Hydrogeol. J. 2019, 27, 669–683. [Google Scholar] [CrossRef]
- Manoj, S.; Thirumurugan, M.; Elango, L. Hydrogeochemical modelling to understand the surface water–groundwater interaction around a proposed uranium mining site. J. Earth Syst. Sci. 2019, 128, 49. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.K.; Sarin, M.M.; France-Lanord, C. Chemical erosion in the eastern Himalaya: Major ion composition of the Brahmaputra and δ13C of dissolved inorganic carbon. Geochim. Cosmochim. Acta 2005, 69, 3573–3588. [Google Scholar] [CrossRef]
- Hydrogeological and hydrogeochemical characterization of a karstic mountain region. Environ. Geol. 2008, 54, 309–310. [CrossRef] [Green Version]
- Wang, M.; Gui, H.; Hu, R.; Zhao, H.; Li, J.; Yu, H.; Fang, H. Hydrogeochemical Characteristics and Water Quality Evaluation of Carboniferous Taiyuan Formation Limestone Water in Sulin Mining Area in Northern Anhui, China. Int. J. Environ. Res. Public. Health 2019, 16, 2512. [Google Scholar] [CrossRef] [Green Version]
- Zong-Jie, L.; Zong-Xing, L.; Ling-Ling, S.; Jin-Zhu, M.; Yong, S. Environment significance and hydrochemical characteristics of supra-permafrost water in the source region of the Yangtze River. Sci. Total Environ. 2018, 644, 1141–1151. [Google Scholar] [CrossRef]
- Tarawaneh, M.S.M.; Janardhana, M.R.; Ahmed, M.M. Hydrochemical processes and groundwater quality assessment in North eastern region of Jordan valley, Jordan. HydroResearch 2019, 2, 129–145. [Google Scholar] [CrossRef]
- Yu, Y.; Yang, J. Health risk assessment of fluorine in fertilizers from a fluorine contaminated region based on the oral bioaccessibility determined by Biomimetic Whole Digestion-Plasma in-vitro Method (BWDPM). J. Hazard. Mater. 2020, 383, 121124. [Google Scholar] [CrossRef] [PubMed]
- Machavaram, M.V.; Whittemore, D.O.; Conrad, M.E.; Miller, N.L. Precipitation induced stream flow: An event based chemical and isotopic study of a small stream in the Great Plains region of the USA. J. Hydrol. 2006, 330, 470–480. [Google Scholar] [CrossRef]
- Craig, H. Isotopic Variations in Meteoric Waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.K.; Solomon, A.M.; Brown, S.; Houghton, R.A.; Trexier, M.C.; Wisniewski, J. Carbon Pools and Flux of Global Forest Ecosystems. Science 1994, 263, 185–190. [Google Scholar] [CrossRef]
- Ehya, F.; Mosleh, A. Hydrochemistry and quality assessment of groundwater in Basht Plain, Kohgiluyeh-va-Boyer Ahmad Province, SW Iran. Environ. Earth Sci. 2018, 77, 164. [Google Scholar] [CrossRef]
- Zheng, L.; Chen, X.; Dong, X.; Wei, X.; Jiang, C.; Tang, Q. Using δ34S–SO4 and δ18O–SO4 to trace the sources of sulfate in different types of surface water from the Linhuan coal-mining subsidence area of Huaibei, China. Ecotoxicol. Environ. Saf. 2019, 181, 231–240. [Google Scholar] [CrossRef]
- Tang, J.; Wang, W.; Yang, L.; Qiu, Q.; Lin, M.; Cao, C.; Li, X. Seasonal variation and ecological risk assessment of dissolved organic matter in a peri-urban critical zone observatory watershed. Sci. Total Environ. 2020, 707, 136093. [Google Scholar] [CrossRef]




| Grade | Classification | Parameters(Unit: mg/L) | ||||
|---|---|---|---|---|---|---|
| TN | TP | COD | SO42− | Cl− | ||
| Ⅰ | Excellent suitable for drinking water | 0.2 | 0.01 | 15 | ||
| Ⅱ | Good suitable for drinking water | 0.5 | 0.025 | 15 | ||
| Ⅲ | Moderate suitable for drinking water | 1 | 0.05 | 20 | 250 | 250 |
| Ⅳ | Poor suitable for drinking water | 1.5 | 0.1 | 30 | ||
| Ⅴ | Unsuitable suitable for drinking water | 2 | 0.2 | 40 | ||
| Samples of Bian River | Na+ (mg/L) | K+ (mg/L) | Mg2+ (mg/L) | Ca2+ (mg/L) | F− (mg/L) | Cl− (mg/L) | SO42− (mg/L) | HCO3− (mg/L) | CO32− (mg/L) | pH - | TDS (mg/L) | EC (μS/cm) | TN (mg/L) | TP (mg/L) | COD (mg/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B1 | 294.85 | 24.83 | 57.05 | 17.07 | 1.06 | 163.30 | 359.43 | 260.64 | 33.69 | 8.76 | 953 | 1910 | 1.84 | 0.12 | 20.57 |
| B2 | 282.28 | 25.43 | 57.69 | 17.68 | 1.07 | 163.97 | 347.61 | 250.62 | 29.36 | 8.52 | 929 | 1833 | 1.80 | 0.15 | 15.05 |
| B3 | 284.28 | 25.13 | 57.46 | 17.88 | 1.01 | 162.05 | 347.32 | 243.41 | 36.84 | 8.58 | 921 | 1848 | 2.15 | 0.17 | 20.57 |
| B4 | 273.04 | 25.08 | 56.99 | 17.63 | 1.00 | 158.68 | 327.15 | 253.43 | 28.57 | 8.61 | 891 | 1818 | 1.72 | 0.16 | 18.06 |
| B5 | 316.12 | 30.45 | 68.46 | 21.10 | 1.20 | 186.40 | 388.43 | 254.63 | 28.96 | 8.57 | 875 | 1788 | 2.27 | 0.16 | 18.06 |
| B6 | 254.01 | 25.87 | 57.28 | 17.74 | 0.99 | 155.44 | 313.13 | 239.81 | 31.72 | 8.63 | 862 | 1752 | 3.10 | 0.17 | 21.11 |
| B7 | 251.35 | 26.14 | 57.81 | 17.40 | 1.00 | 154.98 | 314.89 | 230.39 | 35.07 | 8.57 | 845 | 1724 | 2.42 | 0.18 | 22.61 |
| B8 | 245.15 | 26.16 | 57.63 | 17.56 | 0.96 | 151.41 | 308.93 | 222.18 | 35.07 | 8.67 | 832 | 1674 | 5.17 | 0.17 | 25.65 |
| B9 | 239.27 | 26.41 | 58.05 | 17.25 | 0.95 | 151.14 | 303.67 | 220.17 | 32.71 | 8.62 | 819 | 1646 | 4.56 | 0.13 | 28.10 |
| B10 | 233.49 | 26.59 | 57.99 | 17.04 | 0.96 | 148.05 | 295.64 | 221.38 | 30.74 | 8.72 | 810 | 1634 | 4.32 | 0.14 | 30.62 |
| B11 | 234.23 | 26.82 | 58.40 | 16.75 | 0.95 | 147.14 | 297.69 | 197.94 | 37.83 | 8.75 | 795 | 1600 | 2.02 | 0.13 | 31.18 |
| B12 | 234.25 | 27.30 | 59.23 | 16.91 | 0.94 | 144.98 | 300.21 | 219.57 | 30.93 | 8.60 | 780 | 1614 | 2.10 | 0.13 | 34.62 |
| B13 | 223.18 | 26.85 | 58.04 | 16.86 | 0.91 | 143.89 | 285.64 | 219.57 | 28.96 | 8.60 | 776 | 1576 | 3.23 | 0.13 | 25.59 |
| B14 | 221.43 | 27.46 | 57.98 | 16.83 | 0.88 | 143.97 | 286.91 | 219.97 | 26.80 | 8.56 | 766 | 1549 | 1.68 | 0.13 | 30.11 |
| B15 | 220.52 | 27.12 | 58.10 | 16.95 | 0.93 | 144.70 | 286.75 | 201.74 | 36.25 | 8.65 | 771 | 1579 | 1.60 | 0.15 | 29.10 |
| B16 | 196.32 | 28.62 | 58.78 | 16.83 | 0.90 | 136.78 | 266.75 | 186.72 | 38.22 | 8.69 | 732 | 1437 | 2.75 | 0.12 | 32.66 |
| B17 | 177.92 | 29.83 | 58.94 | 16.50 | 0.88 | 130.81 | 249.97 | 176.10 | 32.12 | 8.76 | 682 | 1375 | 2.06 | 0.09 | 33.65 |
| B18 | 214.72 | 28.36 | 60.06 | 17.63 | 0.94 | 145.53 | 285.71 | 198.14 | 35.47 | 8.57 | 745 | 1530 | 1.71 | 0.12 | 32.12 |
| B19 | 216.48 | 27.99 | 59.54 | 17.62 | 0.92 | 143.58 | 286.07 | 206.95 | 35.07 | 8.76 | 747 | 1522 | 3.31 | 0.15 | 36.14 |
| B20 | 215.15 | 27.68 | 58.97 | 17.38 | 0.91 | 144.03 | 284.39 | 214.96 | 27.98 | 8.47 | 738 | 1506 | 3.79 | 0.11 | 31.11 |
| Min | 177.92 | 24.83 | 56.99 | 16.50 | 0.88 | 130.81 | 249.97 | 176.10 | 26.80 | 8.47 | 682 | 1375 | 1.60 | 0.09 | 15.05 |
| Max | 316.12 | 30.45 | 68.46 | 21.10 | 1.20 | 186.40 | 388.43 | 260.64 | 38.22 | 8.76 | 953 | 1910 | 5.17 | 0.18 | 36.14 |
| Mean | 241.40 | 27.01 | 58.72 | 17.43 | 0.97 | 151.04 | 306.81 | 221.92 | 32.62 | 8.63 | 813 | 1646 | 2.60 | 0.14 | 26.83 |
| SV(%) | 13.94 | 5.48 | 4.04 | 5.31 | 7.64 | 7.74 | 10.56 | 10.28 | 10.48 | 0.96 | 8.45 | 8.60 | 38.89 | 16.74 | 22.81 |
| T1 | 200.42 | 19.55 | 56.78 | 16.74 | 0.89 | 120.85 | 261.57 | 250.83 | 32.12 | 8.96 | 715 | 1445 | 2.38 | 0.11 | 22.59 |
| T2 | 198.22 | 19.01 | 56.99 | 16.79 | 0.92 | 120.50 | 259.44 | 142.24 | 25.61 | 9.02 | 709 | 1407 | 2.78 | 0.12 | 24.61 |
| T3 | 195.08 | 19.19 | 57.10 | 16.34 | 0.96 | 119.42 | 258.81 | 139.44 | 18.52 | 8.85 | 661 | 1398 | 2.21 | 0.09 | 21.57 |
| T4 | 194.30 | 19.28 | 56.99 | 15.99 | 0.94 | 118.88 | 258.12 | 226.38 | 31.52 | 8.72 | 666 | 1379 | 2.80 | 0.09 | 23.58 |
| T5 | 193.09 | 19.12 | 56.78 | 15.61 | 0.96 | 116.27 | 257.97 | 100.17 | 27.39 | 9.14 | 646 | 1343 | 2.90 | 0.11 | 20.57 |
| T6 | 192.99 | 19.06 | 56.63 | 15.55 | 0.95 | 118.37 | 256.81 | 211.96 | 46.11 | 8.92 | 634 | 1138 | 1.76 | 0.09 | 28.38 |
| T7 | 193.84 | 19.21 | 56.79 | 15.52 | 0.95 | 119.72 | 258.28 | 194.73 | 30.74 | 8.24 | 624 | 1023 | 2.41 | 0.08 | 21.08 |
| T8 | 189.64 | 21.72 | 57.43 | 15.84 | 0.98 | 113.92 | 249.67 | 196.53 | 53.40 | 8.45 | 678 | 1345 | 2.31 | 0.09 | 20.32 |
| T9 | 179.54 | 20.89 | 55.52 | 16.00 | 0.93 | 112.50 | 238.36 | 238.00 | 22.86 | 8.76 | 649 | 1316 | 2.19 | 0.09 | 15.80 |
| T10 | 187.91 | 22.57 | 57.53 | 15.15 | 1.11 | 115.65 | 287.44 | 195.93 | 45.71 | 8.30 | 610 | 1246 | 2.29 | 0.08 | 20.56 |
| T11 | 176.33 | 21.83 | 54.76 | 14.26 | 0.88 | 112.70 | 232.39 | 239.41 | 20.69 | 8.65 | 632 | 1184 | 3.00 | 0.07 | 26.60 |
| T12 | 176.28 | 22.09 | 54.91 | 14.84 | 0.95 | 111.33 | 231.03 | 211.16 | 36.45 | 8.92 | 638 | 1270 | 2.43 | 0.10 | 21.07 |
| T13 | 177.31 | 21.98 | 55.25 | 15.78 | 0.92 | 112.81 | 235.55 | 243.01 | 21.08 | 8.54 | 631 | 1263 | 2.25 | 0.08 | 19.56 |
| T14 | 175.30 | 21.94 | 54.63 | 15.74 | 0.94 | 111.47 | 232.40 | 199.94 | 44.73 | 8.71 | 633 | 1295 | 2.39 | 0.09 | 23.08 |
| T15 | 182.24 | 25.84 | 57.93 | 20.63 | 0.89 | 117.64 | 253.89 | 236.20 | 23.05 | 8.53 | 605 | 1238 | 2.28 | 0.08 | 21.06 |
| T16 | 165.91 | 23.63 | 53.92 | 15.46 | 0.93 | 107.83 | 219.44 | 202.14 | 38.42 | 8.58 | 622 | 1248 | 2.29 | 0.08 | 19.56 |
| T17 | 161.41 | 24.30 | 52.93 | 15.70 | 0.96 | 106.02 | 207.19 | 218.37 | 31.92 | 8.88 | 598 | 1202 | 2.96 | 0.06 | 21.07 |
| T18 | 164.69 | 24.72 | 52.25 | 14.91 | 0.98 | 110.07 | 212.14 | 180.91 | 44.73 | 8.92 | 614 | 1239 | 2.35 | 0.07 | 22.58 |
| T19 | 173.08 | 26.20 | 54.74 | 14.94 | 1.00 | 115.03 | 221.58 | 213.16 | 35.07 | 8.92 | 614 | 1234 | 2.13 | 0.08 | 21.98 |
| T20 | 158.99 | 25.15 | 52.47 | 16.31 | 0.96 | 104.52 | 200.53 | 197.53 | 42.76 | 8.88 | 608 | 1231 | 2.99 | 0.08 | 29.15 |
| T21 | 158.90 | 25.01 | 52.71 | 17.17 | 0.96 | 105.58 | 204.54 | 222.58 | 33.30 | 8.68 | 615 | 1222 | 2.72 | 0.07 | 19.50 |
| T22 | 158.85 | 25.55 | 52.51 | 17.33 | 0.97 | 105.76 | 197.77 | 228.59 | 30.54 | 8.80 | 604 | 1218 | 2.03 | 0.06 | 28.63 |
| T23 | 157.78 | 25.29 | 51.93 | 17.21 | 0.94 | 105.33 | 201.53 | 235.60 | 25.61 | 8.94 | 621 | 1260 | 1.22 | 0.06 | 18.06 |
| T24 | 159.78 | 25.75 | 52.40 | 16.37 | 0.98 | 106.93 | 203.79 | 247.62 | 21.08 | 8.78 | 630 | 1261 | 2.09 | 0.07 | 16.55 |
| T25 | 158.06 | 25.67 | 52.21 | 16.07 | 0.95 | 104.10 | 200.59 | 269.86 | 14.78 | 8.63 | 621 | 1233 | 2.10 | 0.07 | 17.31 |
| T26 | 158.74 | 25.19 | 51.85 | 16.44 | 0.95 | 104.90 | 204.04 | 219.37 | 35.07 | 8.92 | 611 | 1246 | 2.98 | 0.07 | 26.86 |
| T27 | 161.59 | 25.47 | 52.45 | 17.60 | 0.96 | 106.65 | 206.21 | 235.60 | 29.55 | 9.11 | 618 | 1243 | 2.77 | 0.08 | 27.37 |
| Min | 157.78 | 19.01 | 51.85 | 14.26 | 0.88 | 104.10 | 197.77 | 100.17 | 14.78 | 8.24 | 598 | 1023 | 1.22 | 0.06 | 15.80 |
| Max | 200.42 | 26.20 | 57.92 | 20.63 | 1.11 | 120.85 | 287.44 | 269.86 | 53.40 | 9.14 | 715 | 1445 | 3.00 | 0.12 | 29.15 |
| Mean | 175.93 | 22.79 | 54.76 | 16.16 | 0.95 | 112.03 | 231.52 | 211.01 | 31.96 | 8.78 | 634 | 1264 | 2.41 | 0.08 | 22.19 |
| SV(%) | 8.34 | 11.33 | 3.78 | 7.36 | 4.37 | 4.97 | 10.92 | 17.24 | 30.03 | 2.50 | 4.62 | 6.72 | 16.9 | 16.74 | 16.32 |
| Parameters | Unit | Tuo River | Bian River | ||||
|---|---|---|---|---|---|---|---|
| Min | Max | Mean | Min | Max | Mean | ||
| δD | ‰ | −45.72 | −30.94 | −37.72 | −33.94 | −29.19 | −31.59 |
| δ18O | ‰ | −5.55 | −2.40 | −3.78 | −3.76 | −2.32 | −3.20 |
| d-excess | ‰ | −12.18 | −1.36 | −7.52 | −11.56 | −0.93 | −5.97 |
| Sample | Ⅰ | Ⅱ | Ⅲ | Ⅳ | Ⅴ | Water Quality Level |
|---|---|---|---|---|---|---|
| B1 | 0.0000 | 0.1222 | 0.2124 | 0.3001 | 0.3652 | Ⅴ |
| B2 | 0.0000 | 0.3793 | 0.0267 | 0.3535 | 0.2404 | Ⅱ |
| B3 | 0.0000 | 0.1240 | 0.2156 | 0.3195 | 0.3409 | Ⅴ |
| B4 | 0.0000 | 0.2299 | 0.2141 | 0.3397 | 0.2163 | Ⅳ |
| B5 | 0.0000 | 0.1908 | 0.2108 | 0.0908 | 0.5076 | Ⅴ |
| B6 | 0.0000 | 0.1333 | 0.0760 | 0.3618 | 0.4289 | Ⅴ |
| B7 | 0.0000 | 0.1340 | 0.0721 | 0.3003 | 0.4936 | Ⅴ |
| B8 | 0.0000 | 0.1390 | 0.0781 | 0.1860 | 0.5968 | Ⅴ |
| B9 | 0.0000 | 0.1394 | 0.0875 | 0.5111 | 0.2620 | Ⅳ |
| B10 | 0.0027 | 0.1382 | 0.1008 | 0.2793 | 0.4789 | Ⅴ |
| B11 | 0.0040 | 0.1369 | 0.0970 | 0.2490 | 0.5130 | Ⅴ |
| B12 | 0.0071 | 0.1339 | 0.0923 | 0.3180 | 0.4487 | Ⅴ |
| B13 | 0.0086 | 0.1324 | 0.1561 | 0.4622 | 0.2408 | Ⅳ |
| B14 | 0.0112 | 0.1325 | 0.1169 | 0.5367 | 0.2054 | Ⅳ |
| B15 | 0.0075 | 0.1335 | 0.1172 | 0.5876 | 0.1542 | Ⅳ |
| B16 | 0.0186 | 0.1223 | 0.1543 | 0.2196 | 0.4851 | Ⅴ |
| B17 | 0.0271 | 0.1140 | 0.2505 | 0.1676 | 0.4409 | Ⅴ |
| B18 | 0.0063 | 0.1347 | 0.1192 | 0.4635 | 0.2764 | Ⅳ |
| B19 | 0.0091 | 0.1319 | 0.1185 | 0.1949 | 0.5456 | Ⅴ |
| B20 | 0.0084 | 0.1326 | 0.1216 | 0.4505 | 0.2869 | Ⅳ |
| Sample | Ⅰ | Ⅱ | Ⅲ | Ⅳ | Ⅴ | Water Quality Level |
|---|---|---|---|---|---|---|
| T1 | 0.1054 | 0.2562 | 0.2196 | 0.2801 | 0.1387 | Ⅳ |
| T2 | 0.1067 | 0.2550 | 0.2097 | 0.2290 | 0.1997 | Ⅱ |
| T3 | 0.1106 | 0.2510 | 0.3394 | 0.1764 | 0.1226 | Ⅲ |
| T4 | 0.1125 | 0.2491 | 0.2879 | 0.2279 | 0.1226 | Ⅲ |
| T5 | 0.1220 | 0.2396 | 0.2912 | 0.2138 | 0.1333 | Ⅲ |
| T6 | 0.1144 | 0.2472 | 0.2695 | 0.2170 | 0.1519 | Ⅲ |
| T7 | 0.1095 | 0.2521 | 0.3391 | 0.1767 | 0.1226 | Ⅲ |
| T8 | 0.1305 | 0.2319 | 0.3827 | 0.1323 | 0.1226 | Ⅲ |
| T9 | 0.1356 | 0.3413 | 0.2751 | 0.1254 | 0.1226 | Ⅱ |
| T10 | 0.1242 | 0.2374 | 0.2912 | 0.2246 | 0.1226 | Ⅲ |
| T11 | 0.1349 | 0.2675 | 0.2839 | 0.1743 | 0.1394 | Ⅲ |
| T12 | 0.1398 | 0.2657 | 0.2994 | 0.1724 | 0.1226 | Ⅲ |
| T13 | 0.1345 | 0.2699 | 0.3513 | 0.1218 | 0.1226 | Ⅲ |
| T14 | 0.1393 | 0.2630 | 0.2745 | 0.2005 | 0.1226 | Ⅲ |
| T15 | 0.1170 | 0.2446 | 0.3918 | 0.1240 | 0.1226 | Ⅲ |
| T16 | 0.1525 | 0.2845 | 0.3509 | 0.0895 | 0.1226 | Ⅲ |
| T17 | 0.1590 | 0.3017 | 0.3696 | 0.0471 | 0.1226 | Ⅲ |
| T18 | 0.1444 | 0.3049 | 0.3050 | 0.1231 | 0.1226 | Ⅲ |
| T19 | 0.1265 | 0.3010 | 0.3175 | 0.1324 | 0.1226 | Ⅲ |
| T20 | 0.1645 | 0.3117 | 0.1851 | 0.1110 | 0.2277 | Ⅱ |
| T21 | 0.1606 | 0.3062 | 0.2338 | 0.0716 | 0.2277 | Ⅱ |
| T22 | 0.1600 | 0.3226 | 0.2396 | 0.0646 | 0.2133 | Ⅱ |
| T23 | 0.1615 | 0.3531 | 0.3162 | 0.1155 | 0.0537 | Ⅱ |
| T24 | 0.1557 | 0.3854 | 0.2646 | 0.0716 | 0.1226 | Ⅱ |
| T25 | 0.1660 | 0.3666 | 0.2803 | 0.0645 | 0.1226 | Ⅱ |
| T26 | 0.1631 | 0.3049 | 0.2218 | 0.1469 | 0.1632 | Ⅱ |
| T27 | 0.1567 | 0.3063 | 0.2018 | 0.1561 | 0.1791 | Ⅱ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Y.; Gui, H.; Yu, H.; Wang, M.; Fang, H.; Wang, C.; Chen, C.; Zhang, Y.; Huang, Y. Hydrochemical Characteristics and Water Quality Evaluation of Rivers in Different Regions of Cities: A Case Study of Suzhou City in Northern Anhui Province, China. Water 2020, 12, 950. https://doi.org/10.3390/w12040950
Jiang Y, Gui H, Yu H, Wang M, Fang H, Wang C, Chen C, Zhang Y, Huang Y. Hydrochemical Characteristics and Water Quality Evaluation of Rivers in Different Regions of Cities: A Case Study of Suzhou City in Northern Anhui Province, China. Water. 2020; 12(4):950. https://doi.org/10.3390/w12040950
Chicago/Turabian StyleJiang, Yaqi, Herong Gui, Hao Yu, Meichen Wang, Hongxia Fang, Chunlei Wang, Chen Chen, Yaru Zhang, and Yiheng Huang. 2020. "Hydrochemical Characteristics and Water Quality Evaluation of Rivers in Different Regions of Cities: A Case Study of Suzhou City in Northern Anhui Province, China" Water 12, no. 4: 950. https://doi.org/10.3390/w12040950
APA StyleJiang, Y., Gui, H., Yu, H., Wang, M., Fang, H., Wang, C., Chen, C., Zhang, Y., & Huang, Y. (2020). Hydrochemical Characteristics and Water Quality Evaluation of Rivers in Different Regions of Cities: A Case Study of Suzhou City in Northern Anhui Province, China. Water, 12(4), 950. https://doi.org/10.3390/w12040950





