An Overview of the Water Remediation Potential of Nanomaterials and Their Ecotoxicological Impacts
Abstract
1. Introduction
2. Nanomaterials for Water Purification
2.1. Nanomaterials for Adsorption and Photodecomposition
2.2. Nanomaterials for Membrane-Based Water Treatment
3. Ecotoxicology of Nanomaterials
3.1. Nanomaterials in Aquatic Systems
3.2. Nanomaterials in Terrestrial Systems
4. Conclusions and Future Outlook
- (1)
- Solar light driven photocatalysis: With respect to photocatalysis-based water treatment, it is of paramount importance to avoid hole-electron recombination in the photocatalyst and also shift the light responsiveness from the UV to the visible solar light range. The latter goal guarrantees a lower energy consumption and wider applicability of photocatalysis for water purification. Research has begun to develop a new generation of solar light responsive doped photocatalysts that assure versatility and energy efficiency of photocatalytic nanoadsorbents.
- (2)
- Aggregation and poor recovery: One important disadvantage regarding the nanoparticulate adsorbents is their aggregation tendency and challenging recovery. In this regard, one optimum solution is deposition of nanoparticles on nanostructured substrates e.g., nanofibers. This hybridization reduces the aggregation tendency and eases recovery of the nanoparticles, while preserving their high exposure to the external water medium.
- (3)
- Photodegradation of polymer hosts: Many nanomaterials in different forms such as nanoparticles, nanotubes, nanofibers, and nanosheets are typically used as coupled with a polymer substrate or host. In case of applying photocatalytic, aggresive nanomaterials, the chance of photodegradation of the encapsulating polymer is considerable. To address this problem, inclusion of photostabilizers could be a main strategy.
- (4)
- Unwanted release of nanomaterials during the water treatment process: Nanomaterials employed in the construction of micro-, ultra-, and nanofiltration membranes can be released into water streams when the membrane is subjected to harsh water streams and their complicated stress patterns. Therefore, primarily stabilization of nanoparticles on/in the membrane structure should be taken into account and secondly, intoxic materials should be employed that impose less hazardous effects on biota.
- (5)
- Long term, realistic testing of novel generation of nanostructured membranes: Nanomaterials in higher dimensionalities such as 1D nanofibers and the 2D graphene family have also been studied for the development of membranes. Electrospun nanofibers have shown a promising potential in size exclusion and also adsorption of water pollutants. Thanks to their tunable pore size, high porosity and interconnected porous structure they can guarrantee a less energy consuming water treatment process. That is why they have found large applicability for building up advanced ultra- and nanofiltration membranes as a porous, robust support for the overlaying selective layer. However, no industrial utility has been reported for nanofibrous membranes. This could arises from the available gaps with respect to reliable testing of such membranes. Nanofibrous membranes must be challenged in long term, and under realistic conditions with real wastewater models and also be exposed to various complicated mechanical stress patterns. Typically, the relevant research experiments done at the lab scale consider only one type of pollutant and ignore co-existence of other dye, ionic, or organic pollutants, as seen in real wastewater, which compete for a limited number of available active/binding sites. Such a perspective was previously taken into account for activated carbon as a commercial adsorbent, and led to its commercialization. Graphene membranes are also a fascinating group of advanced nanomembranes that have shown amazing potentials, particlularly with respect to water permeability, while offering an ionic selectivity comparable to classic NF and ideally RO membranes. Nevertheless, their properties have been mainly theoretically validated rather than experimentally and there is still a large gap ahead till realistic employment of such membranes.
- (6)
- Large scale and economical production of nanostructured membranes and adsorbents: This issue is under extensive investigation. In fact, technical difficulties with respect to scale-up and integration of nanomaterials into a relevant technology, cost effectiveness, and energy-related issue are all hindering concerns that have slowed the marketing trend of such products. For instance, TiO2 nanoparticles and CNTs are among the most widely studied nanomaterials for adsorption of dyes. However, they are toxic and produced in a costly manner involving high temperature and pressure. The former nano-adsorbent needs UV irradiation to photodecompose the dye pollutants that adds to the expenses of the treatment. In fact, it is highly necessary to produce large amounts of such nanomaterials at justifiable costs for water treatments, specific to different categories of wastewaters.
- (7)
- Environmental hazards: This concern will persist in the future. This stems from the reality that many environmental and biological consequences of nanomaterials should be identified in the long term. Short term studies have shown that several nanomaterials are safe to human being, plants and animals. But, there is no certainty about their long term safety. For this reason, establishment of nanomaterial based water treatment systems should be followed with sufficient precautions. Technologically, it is also vital to secure such systems so that the release of nanomaterials into environment would be miminized.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Homaeigohar, S.; Elbahri, M. Nanocomposite Electrospun Nanofiber Membranes for Environmental Remediation. Materials 2014, 7, 1017–1045. [Google Scholar] [CrossRef] [PubMed]
- Homaeigohar, S. The Nanosized Dye Adsorbents for Water Treatment. Nanomaterials 2020, 10, 295. [Google Scholar] [CrossRef] [PubMed]
- Santhosh, C.; Velmurugan, V.; Jacob, G.; Jeong, S.K.; Grace, A.N.; Bhatnagar, A. Role of nanomaterials in water treatment applications: A review. Chem. Eng. J. 2016, 306, 1116–1137. [Google Scholar] [CrossRef]
- Bansal, P.; Chaudhary, G.R.; Mehta, S.K. Comparative study of catalytic activity of ZrO2 nanoparticles for sonocatalytic and photocatalytic degradation of cationic and anionic dyes. Chem. Eng. J. 2015, 280, 475–485. [Google Scholar] [CrossRef]
- Klaine, S.J.; Alvarez, P.J.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. 2008, 27, 1825–1851. [Google Scholar] [CrossRef]
- Shi, J.P.; Evans, D.E.; Khan, A.; Harrison, R.M. Sources and concentration of nanoparticles (<10 nm diameter) in the urban atmosphere. Atmos. Environ. 2001, 35, 1193–1202. [Google Scholar]
- Tan, K.B.; Vakili, M.; Horri, B.A.; Poh, P.E.; Abdullah, A.Z.; Salamatinia, B. Adsorption of dyes by nanomaterials: Recent developments and adsorption mechanisms. Sep. Purif. Technol. 2015, 150, 229–242. [Google Scholar] [CrossRef]
- Shan, G.; Surampalli, R.Y.; Tyagi, R.D.; Zhang, T.C. Nanomaterials for environmental burden reduction, waste treatment, and nonpoint source pollution control: A review. Front. Environ. Sci. Eng. China 2009, 3, 249–264. [Google Scholar] [CrossRef]
- Crini, G. Non-conventional low-cost adsorbents for dye removal: A review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Elbahri, M. An Amphiphilic, Graphitic Buckypaper Capturing Enzyme Biomolecules from Water. Water 2019, 11, 2. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Strunskus, T.; Strobel, J.; Kienle, L.; Elbahri, M. A Flexible Oxygenated Carbographite Nanofilamentous Buckypaper as an Amphiphilic Membrane. Adv. Mater. Interfaces 2018, 5, 1800001. [Google Scholar] [CrossRef]
- Chang, Y.C.; Chen, D.H. Adsorption kinetics and thermodynamics of acid dyes on a carboxymethylated chitosan-conjugated magnetic nano-adsorbent. Macromol. Biosci. 2005, 5, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Garg, V.K.; Kataria, N.; Kadirvelu, K. Applications of Fe3O4@AC nanoparticles for dye removal from simulated wastewater. Chemosphere 2019, 236, 124280. [Google Scholar] [CrossRef] [PubMed]
- Dhananasekaran, S.; Palanivel, R.; Pappu, S. Adsorption of Methylene Blue, Bromophenol Blue, and Coomassie Brilliant Blue by α-chitin nanoparticles. J. Adv. Res. 2016, 7, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Homaeigohar, S.; Zillohu, A.U.; Abdelaziz, R.; Hedayati, M.K.; Elbahri, M. A Novel Nanohybrid Nanofibrous Adsorbent for Water Purification from Dye Pollutants. Materials 2016, 9, 848. [Google Scholar] [CrossRef] [PubMed]
- Hisaindee, S.; Meetani, M.; Rauf, M. Application of LC-MS to the analysis of advanced oxidation process (AOP) degradation of dye products and reaction mechanisms. Trac Trends Anal. Chem. 2013, 49, 31–44. [Google Scholar] [CrossRef]
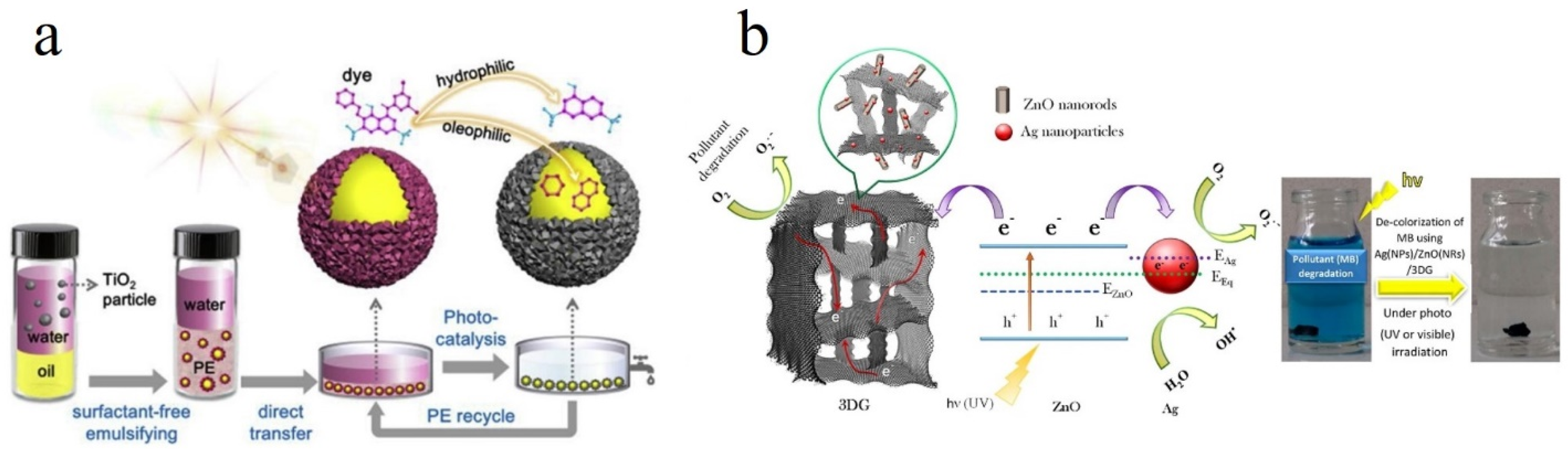
- Li, Q.; Zhao, T.; Li, M.; Li, W.; Yang, B.; Qin, D.; Lv, K.; Wang, X.; Wu, L.; Wu, X.; et al. One-step construction of Pickering emulsion via commercial TiO2 nanoparticles for photocatalytic dye degradation. Appl. Catal. B: Environ. 2019, 249, 1–8. [Google Scholar] [CrossRef]
- Kheirabadi, M.; Samadi, M.; Asadian, E.; Zhou, Y.; Dong, C.; Zhang, J.; Moshfegh, A.Z. Well-designed Ag/ZnO/3D graphene structure for dye removal: Adsorption, photocatalysis and physical separation capabilities. J. Colloid Interface Sci. 2019, 537, 66–78. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Kawi, S.; Ray, M. Photocatalytic degradation of orange II by TiO2 catalysts supported on adsorbents. Catal. Today 2004, 98, 431–439. [Google Scholar] [CrossRef]
- Gao, B.; Yap, P.S.; Lim, T.M.; Lim, T.-T. Adsorption-photocatalytic degradation of Acid Red 88 by supported TiO2: Effect of activated carbon support and aqueous anions. Chem. Eng. J. 2011, 171, 1098–1107. [Google Scholar] [CrossRef]
- Meng, X.; Luo, N.; Cao, S.; Zhang, S.; Yang, M.; Hu, X. In-situ growth of titania nanoparticles in electrospun polymer nanofibers at low temperature. Mater. Lett. 2009, 63, 1401–1403. [Google Scholar] [CrossRef]
- Homaeigohar, S.S.; Mahdavi, H.; Elbahri, M. Extraordinarily water permeable sol gel formed nanocomposite nanofibrous membranes. J. Colloid Interface Sci. 2012, 366, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Homaeigohar, S.; Botcha, N.K.; Zarie, E.S.; Elbahri, M. Ups and Downs of Water Photodecolorization by Nanocomposite Polymer Nanofibers. Nanomaterials 2019, 9, 250. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Choi, W. Solid-phase photocatalytic degradation of PVC–TiO2 polymer composites. J. Photochem. Photobiol. A Chem. 2001, 143, 221–228. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Elbahri, M. Graphene membranes for water desalination. Npg Asia Mater. 2017, 9, e427. [Google Scholar] [CrossRef]
- Abadi, M.B.H.; Ghasemi, I.; Khavandi, A.; Shokrgozar, M.A.; Farokhi, M.; Homaeigohar, S.S.; Eslamifar, A. Synthesis of nano β-TCP and the effects on the mechanical and biological properties of β-TCP/HDPE/UHMWPE nanocomposites. Polym. Compos. 2010, 31, 1745–1753. [Google Scholar] [CrossRef]
- Elbahri, M.; Zillohu, A.U.; Gothe, B.; Hedayati, M.K.; Abdelaziz, R.; El-Khozondar, H.J.; Bawa’aneh, M.; Abdelaziz, M.; Lavrinenko, A.; Zhukovsky, S.; et al. Photoswitchable molecular dipole antennas with tailored coherent coupling in glassy composite. Light Sci. Appl. 2015, 4, e316. [Google Scholar] [CrossRef]
- Homaeigohar, S.S.; Sadi, A.Y.; Javadpour, J.; Khavandi, A. The effect of reinforcement volume fraction and particle size on the mechanical properties of β-tricalcium phosphate–high density polyethylene composites. J. Eur. Ceram. Soc. 2006, 26, 273–278. [Google Scholar] [CrossRef]
- Homaeigohar, S.S.; Shokrgozar, M.A.; Sadi, A.Y.; Khavandi, A.; Javadpour, J.; Hosseinalipour, M. In vitro evaluation of biocompatibility of beta-tricalcium phosphate-reinforced high-density polyethylene; an orthopedic composite. J. Biomed. Mater. Res. Part A 2005, 75A, 14–22. [Google Scholar] [CrossRef]
- Khalil, R.; Homaeigohar, S.; Häußler, D.; Elbahri, M. A shape tailored gold-conductive polymer nanocomposite as a transparent electrode with extraordinary insensitivity to volatile organic compounds (VOCs). Sci. Rep. 2016, 6, 33895. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Elbahri, M. Switchable Plasmonic Nanocomposites. Adv. Opt. Mater. 2019, 7, 1801101. [Google Scholar] [CrossRef]
- Sadi, A.Y.; Homaeigohar, S.S.; Khavandi, A.R.; Javadpour, J. The effect of partially stabilized zirconia on the mechanical properties of the hydroxyapatite–polyethylene composites. J. Mater. Sci. Mater. Med. 2004, 15, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Sadi, A.Y.; Shokrgozar, M.; Homaeigohar, S.S.; Hosseinalipour, M.; Khavandi, A.; Javadpour, J. The effect of partially stabilized zirconia on the biological properties of HA/HDPE composites in vitro. J. Mater. Sci. Mater. Med. 2006, 17, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Yari Sadi, A.; Shokrgozar, M.A.; Homaeigohar, S.S.; Khavandi, A. Biological evaluation of partially stabilized zirconia added HA/HDPE composites with osteoblast and fibroblast cell lines. J. Mater. Sci. Mater. Med. 2008, 19, 2359–2365. [Google Scholar] [CrossRef]
- Homaeigohar, S.S.; Shokrgozar, M.; Javadpour, J.; Khavandi, A.; Sadi, A.Y. Effect of reinforcement particle size on in vitro behavior of β-tricalcium phosphate-reinforced high-density polyethylene: A novel orthopedic composite. J. Biomed. Mater. Res. Part A 2006, 78, 129–138. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Kabir, R.; Elbahri, M. Size-tailored physicochemical properties of Monodisperse polystyrene nanoparticles and the nanocomposites Made thereof. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
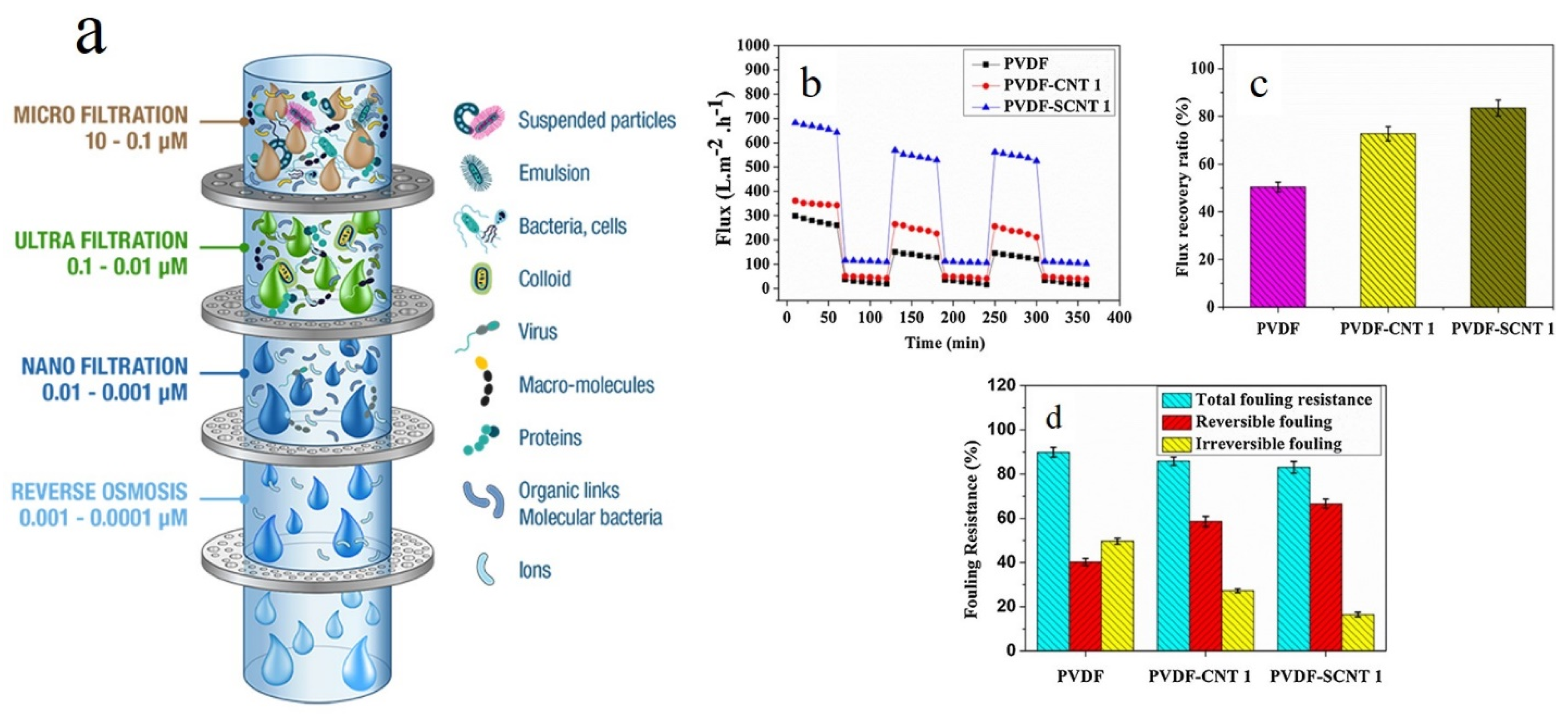
- Rodrigues, R.; Mierzwa, J.C.; Vecitis, C.D. Mixed matrix polysulfone/clay nanoparticles ultrafiltration membranes for water treatment. J. Water Process Eng. 2019, 31, 100788. [Google Scholar] [CrossRef]
- Ayyaru, S.; Pandiyan, R.; Ahn, Y.-H. Fabrication and characterization of anti-fouling and non-toxic polyvinylidene fluoride -Sulphonated carbon nanotube ultrafiltration membranes for membrane bioreactors applications. Chem. Eng. Res. Des. 2019, 142, 176–188. [Google Scholar] [CrossRef]
- Ayyaru, S.; Choi, J.; Ahn, Y.-H. Biofouling reduction in a MBR by the application of a lytic phage on a modified nanocomposite membrane. Environ. Sci. Water Res. Technol. 2018, 4, 1624–1638. [Google Scholar] [CrossRef]
- Zodrow, K.; Brunet, L.; Mahendra, S.; Li, D.; Zhang, A.; Li, Q.; Alvarez, P.J. Polysulfone ultrafiltration membranes impregnated with silver nanoparticles show improved biofouling resistance and virus removal. Water Res. 2009, 43, 715–723. [Google Scholar] [CrossRef]
- Yong, C.W. Study of interactions between polymer nanoparticles and cell membranes at atomistic levels. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140036. [Google Scholar] [CrossRef] [PubMed]
- Akhavan, O.; Ghaderi, E. Toxicity of graphene and graphene oxide nanowalls against bacteria. Acs Nano 2010, 4, 5731–5736. [Google Scholar] [CrossRef] [PubMed]
- Al-Khaldi, F.A.; Abusharkh, B.; Khaled, M.; Atieh, M.A.; Nasser, M.; Saleh, T.A.; Agarwal, S.; Tyagi, I.; Gupta, V.K. Adsorptive removal of cadmium (II) ions from liquid phase using acid modified carbon-based adsorbents. J. Mol. Liq. 2015, 204, 255–263. [Google Scholar]
- Das, R.; Ali, M.E.; Hamid, S.B.A.; Ramakrishna, S.; Chowdhury, Z.Z. Carbon nanotube membranes for water purification: A bright future in water desalination. Desalination 2014, 336, 97–109. [Google Scholar] [CrossRef]
- Chen, C.; Wang, X. Adsorption of Ni (II) from aqueous solution using oxidized multiwall carbon nanotubes. Ind. Eng. Chem. Res. 2006, 45, 9144–9149. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Tsai, T.-Y.; Young, T.-H.; Yang, H.J.; Ji, Y.-R. An electroactive alginate hydrogel nanocomposite reinforced by functionalized graphite nanofilaments for neural tissue engineering. Carbohydr. Polym. 2019, 224, 115112. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Dai, T.; Elbahri, M. Biofunctionalized nanofibrous membranes as super separators of protein and enzyme from water. J. Colloid Interface Sci. 2013, 406, 86–93. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Disci-Zayed, D.; Dai, T.; Elbahri, M. Biofunctionalized nanofibrous membranes mimicking carnivorous plants. Bioinspired Biomim. Nanobiomater. 2013, 2, 186–193. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Davoudpour, Y.; Habibi, Y.; Elbahri, M. The Electrospun Ceramic Hollow Nanofibers. Nanomaterials 2017, 7, 383. [Google Scholar] [CrossRef]
- Homaeigohar, S. Amphiphilic Oxygenated Amorphous Carbon-Graphite Buckypapers with Gas Sensitivity to Polar and Non-Polar VOCs. Nanomaterials 2019, 9, 1343. [Google Scholar] [CrossRef]
- Homaeigohar, S.S.; Buhr, K.; Ebert, K. Polyethersulfone electrsopun nanofibrous composite membrane for liquid filtration. J. Mem. Sci. 2010, 365, 68–77. [Google Scholar] [CrossRef]
- Homaeigohar, S.S.; Elbahri, M. Novel compaction resistant and ductile nanocomposite nanofibrous microfiltration membranes. J. Colloid Interface Sci. 2012, 372, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.; Hsiao, B.S.; Chu, B. Functional nanofibers for environmental applications. J. Mater. Chem. 2008, 18, 5326–5334. [Google Scholar] [CrossRef]
- Song, X.; Liu, Z.; Sun, D.D. Energy recovery from concentrated seawater brine by thin-film nanofiber composite pressure retarded osmosis membranes with high power density. Energy Environ. Sci. 2013, 6, 1199–1210. [Google Scholar] [CrossRef]
- Pan, S.-F.; Ke, X.-X.; Wang, T.-Y.; Liu, Q.; Zhong, L.-B.; Zheng, Y.-M. Synthesis of Silver Nanoparticles Embedded Electrospun PAN Nanofiber Thin-Film Composite Forward Osmosis Membrane to Enhance Performance and Antimicrobial Activity. Ind. Eng. Chem. Res. 2019, 58, 984–993. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Boccaccini, A.R. Antibacterial Biohybrid Nanofibers for Wound Dressings. Acta Biomater. 2020, 107, 25–49. [Google Scholar] [CrossRef]
- Liu, L.; Liu, Z.; Bai, H.; Sun, D.D. Concurrent filtration and solar photocatalytic disinfection/degradation using high-performance Ag/TiO2 nanofiber membrane. Water Res. 2012, 46, 1101–1112. [Google Scholar] [CrossRef]
- Yamanaka, M.; Hara, K.; Kudo, J. Bactericidal actions of a silver ion solution on Escherichia coli, studied by energy-filtering transmission electron microscopy and proteomic analysis. Appl. Environ. Microbiol. 2005, 71, 7589–7593. [Google Scholar] [CrossRef]
- Liu, W.-W.; Chai, S.-P.; Mohamed, A.R.; Hashim, U. Synthesis and characterization of graphene and carbon nanotubes: A review on the past and recent developments. J. Ind. Eng. Chem. 2014, 20, 1171–1185. [Google Scholar] [CrossRef]
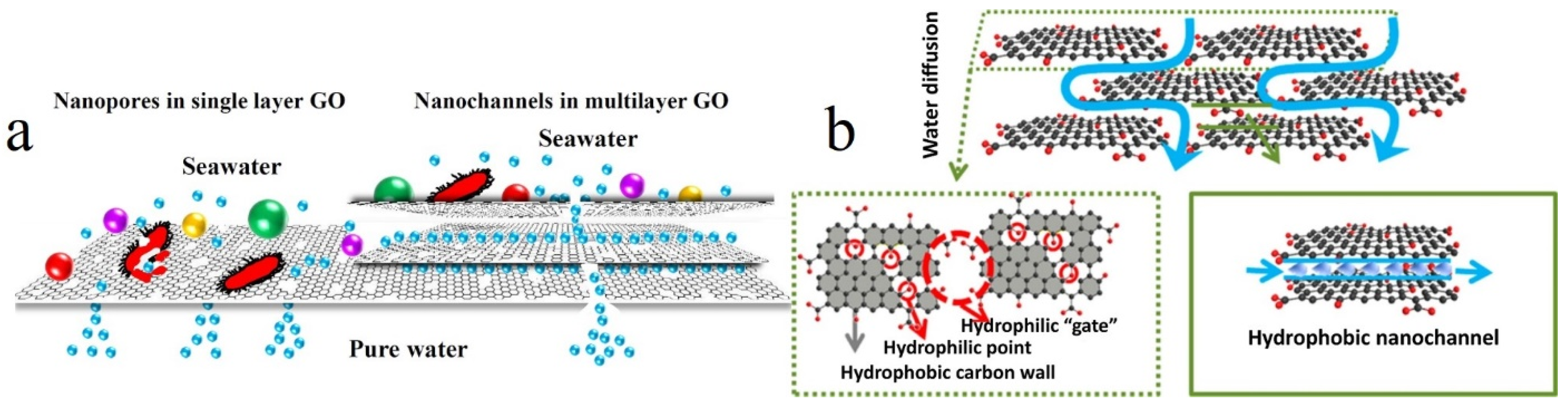
- Han, Y.; Xu, Z.; Gao, C. Ultrathin graphene nanofiltration membrane for water purification. Adv. Funct. Mater. 2013, 23, 3693–3700. [Google Scholar] [CrossRef]
- O’Hern, S.C.; Stewart, C.A.; Boutilier, M.S.; Idrobo, J.-C.; Bhaviripudi, S.; Das, S.K.; Kong, J.; Laoui, T.; Atieh, M.; Karnik, R. Selective molecular transport through intrinsic defects in a single layer of CVD graphene. Acs Nano 2012, 6, 10130–10138. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.; Unnikrishnan, B.; Mao, J.-Y.; Lin, H.-J.; Huang, C.-C. Graphene-based nanofiltration membranes for improving salt rejection, water flux and antifouling–A review. Desalination 2018, 429, 119–133. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, P.; Liang, B.; Liu, Y.; Xu, T.; Wang, L.; Cao, B.; Pan, K. Graphene oxide as an effective barrier on a porous nanofibrous membrane for water treatment. ACS Appl. Mater. Interfaces 2016, 8, 6211–6218. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.; Xu, Z.; Yang, X. Water permeation and ion rejection in layer-by-layer stacked graphene oxide nanochannels: A molecular dynamics simulation. J. Phys. Chem. C 2016, 120, 22585–22596. [Google Scholar] [CrossRef]
- Joshi, R.; Carbone, P.; Wang, F.; Kravets, V.; Su, Y.; Grigorieva, I.; Wu, H.; Geim, A.; Nair, R. Precise and ultrafast molecular sieving through graphene oxide membranes. Science 2014, 343, 752–754. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Q. Health and ecosystem risks of graphene. Chem. Rev. 2013, 113, 3815–3835. [Google Scholar] [CrossRef]
- Bussy, C.; Ali-Boucetta, H.; Kostarelos, K. Safety considerations for graphene: Lessons learnt from carbon nanotubes. Acc. Chem. Res. 2012, 46, 692–701. [Google Scholar] [CrossRef]
- Bianco, A. Graphene: Safe or toxic? The two faces of the medal. Angew. Chem. Int. Ed. 2013, 52, 4986–4997. [Google Scholar] [CrossRef]
- Luan, H.; Teychene, B.; Huang, H. Efficient removal of As(III) by Cu nanoparticles intercalated in carbon nanotube membranes for drinking water treatment. Chem. Eng. J. 2019, 355, 341–350. [Google Scholar] [CrossRef]
- Reddy, C.V.; Reddy, I.N.; Reddy, K.R.; Jaesool, S.; Yoo, K. Template-free synthesis of tetragonal Co-doped ZrO2 nanoparticles for applications in electrochemical energy storage and water treatment. Electrochim. Acta 2019, 317, 416–426. [Google Scholar] [CrossRef]
- Rahdar, S.; Rahdar, A.; Igwegbe, C.A.; Moghaddam, F.; Ahmadi, S. Synthesis and physical characterization of nickel oxide nanoparticles and its application study in the removal of ciprofloxacin from contaminated water by adsorption: Equilibrium and kinetic studies. Desalin. Water Treat. 2019, 141, 386–393. [Google Scholar] [CrossRef]
- Liu, X.; Tian, J.; Li, Y.; Sun, N.; Mi, S.; Xie, Y.; Chen, Z. Enhanced dyes adsorption from wastewater via Fe3O4 nanoparticles functionalized activated carbon. J. Hazard. Mater. 2019, 373, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Cui, J.; Wu, X.; Zhang, X.; Hu, Q.; Hou, X. Rapid in situ microwave synthesis of Fe3O4@MIL-100(Fe) for aqueous diclofenac sodium removal through integrated adsorption and photodegradation. J. Hazard. Mater. 2019, 373, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, N.; Zhang, H.; Baeyens, J. Adsorption of Congo red dye on FexCo3-xO4 nanoparticles. J. Environ. Manag. 2019, 238, 473–483. [Google Scholar] [CrossRef]
- Appiah-Ntiamoah, R.; Baye, A.F.; Gadisa, B.T.; Abebe, M.W.; Kim, H. In-situ prepared ZnO-ZnFe2O4 with 1-D nanofiber network structure: An effective adsorbent for toxic dye effluent treatment. J. Hazard. Mater. 2019, 373, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Gouthaman, A.; Asir, J.A.; Gnanaprakasam, A.; Sivakumar, V.M.; Thirumarimurugan, M.; Ahamed, M.A.R.; Azarudeena, R.S. Enhanced dye removal using polymeric nanocomposite through incorporation of Ag doped ZnO nanoparticles: Synthesis and characterization. J. Hazard. Mater. 2019, 373, 493–503. [Google Scholar] [CrossRef]
- Guo, J.; Khan, S.; Cho, S.-H.; Kim, J. ZnS nanoparticles as new additive for polyethersulfone membrane in humic acid filtration. J. Ind. Eng. Chem. 2019, 79, 71–78. [Google Scholar] [CrossRef]
- Lv, D.; Wang, R.; Tang, G.; Mou, Z.; Lei, J.; Han, J.; De Smedt, S.; Xiong, R.; Huang, C. Ecofriendly electrospun membranes loaded with visible-light-responding nanoparticles for multifunctional usages: Highly efficient air filtration, dye scavenging, and bactericidal activity. Acs Appl. Mater. Interfaces 2019, 11, 12880–12889. [Google Scholar] [CrossRef]
- Farjami, M.; Moghadassi, A.; Vatanpour, V.; Hosseini, S.M.; Parvizian, F. Preparation and characterization of a novel high-flux emulsion polyvinyl chloride (EPVC) ultrafiltration membrane incorporated with boehmite nanoparticles. J. Ind. Eng. Chem. 2019, 72, 144–156. [Google Scholar] [CrossRef]
- Che, W.; Xiao, Z.; Wang, Z.; Li, J.; Wang, H.; Wang, Y.; Xie, Y. Wood-based mesoporous filter decorated with silver nanoparticles for water purification. ACS Sustain. Chem. Eng. 2019, 7, 5134–5141. [Google Scholar] [CrossRef]
- Rowley, J.; Abu-Zahra, N.H. Synthesis and characterization of polyethersulfone membranes impregnated with (3-aminopropyltriethoxysilane) APTES-Fe3O4 nanoparticles for As(V) removal from water. J. Environ. Chem. Eng. 2019, 7, 102875. [Google Scholar] [CrossRef]
- Saraswathi, M.S.S.A.; Rana, D.; Alwarappan, S.; Gowrishankar, S.; Vijayakumar, P.; Nagendran, A. Polydopamine layered poly (ether imide) ultrafiltration membranes tailored with silver nanoparticles designed for better permeability, selectivity and antifouling. J. Ind. Eng. Chem. 2019, 76, 141–149. [Google Scholar] [CrossRef]
- Gopakumar, D.A.; Arumukhan, V.; Gelamo, R.V.; Pasquini, D.; de Morais, L.C.; Rizal, S.; Hermawan, D.; Nzihou, A.; Khalil, H.P.S.A. Carbon dioxide plasma treated PVDF electrospun membrane for the removal of crystal violet dyes and iron oxide nanoparticles from water. Nano-Struct. Nano-Objects 2019, 18, 100268. [Google Scholar] [CrossRef]
- Kadhom, M.; Deng, B. Thin film nanocomposite membranes filled with bentonite nanoparticles for brackish water desalination: A novel water uptake concept. Microporous Mesoporous Mater. 2019, 279, 82–91. [Google Scholar] [CrossRef]
- Liu, X.; Jiang, B.; Yin, X.; Ma, H.; Hsiao, B.S. Highly permeable nanofibrous composite microfiltration membranes for removal of nanoparticles and heavy metal ions. Sep. Purif. Technol. 2020, 233, 115976. [Google Scholar] [CrossRef]
- Kolangare, I.M.; Isloor, A.M.; Karim, Z.A.; Kulal, A.; Ismail, A.F.; Asiri, A.M. Antibiofouling hollow-fiber membranes for dye rejection by embedding chitosan and silver-loaded chitosan nanoparticles. Environ. Chem. Lett. 2019, 17, 581–587. [Google Scholar] [CrossRef]
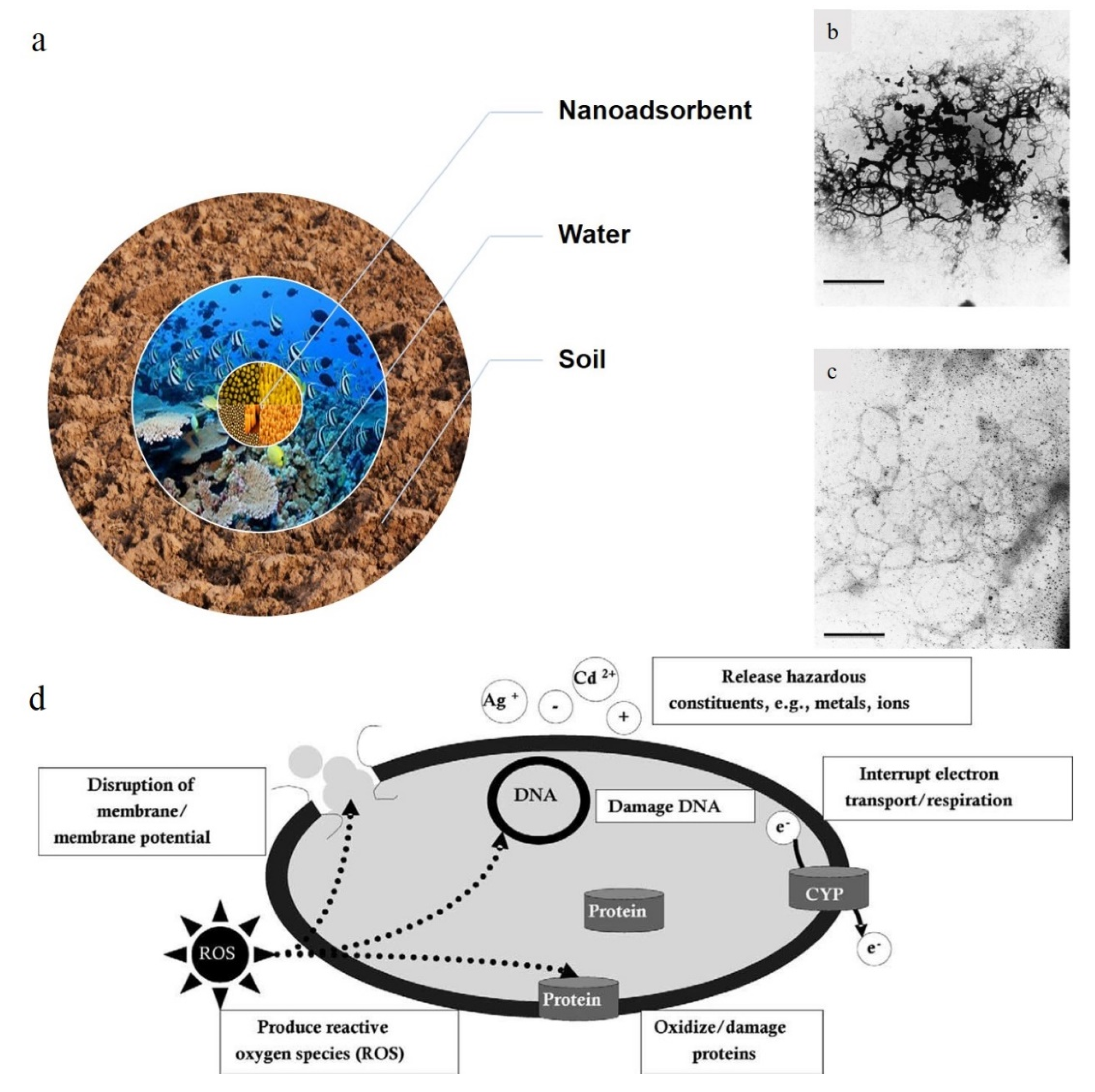
- Boxall, A.B.; Tiede, K.; Chaudhry, Q. Engineered nanomaterials in soils and water: How do they behave and could they pose a risk to human health? Nanomedicine 2007, 2, 919–927. [Google Scholar] [CrossRef]
- Flores-Cervantes, D.X.; Maes, H.M.; Schäffer, A.; Hollender, J.; Kohler, H.-P.E. Slow biotransformation of carbon nanotubes by horseradish peroxidase. Environ. Sci. Technol. 2014, 48, 4826–4834. [Google Scholar] [CrossRef]
- Keller, A.A.; Wang, H.; Zhou, D.; Lenihan, H.S.; Cherr, G.; Cardinale, B.J.; Miller, R.; Ji, Z. Stability and aggregation of metal oxide nanoparticles in natural aqueous matrices. Environ. Sci. Technol. 2010, 44, 1962–1967. [Google Scholar] [CrossRef]
- Adeleye, A.S.; Keller, A.A. Long-term colloidal stability and metal leaching of single wall carbon nanotubes: Effect of temperature and extracellular polymeric substances. Water Res. 2014, 49, 236–250. [Google Scholar] [CrossRef]
- Conway, J.R.; Adeleye, A.S.; Gardea-Torresdey, J.; Keller, A.A. Aggregation, dissolution, and transformation of copper nanoparticles in natural waters. Environ. Sci. Technol. 2015, 49, 2749–2756. [Google Scholar] [CrossRef]
- Wu, B.; Torres-Duarte, C.; Cole, B.J.; Cherr, G.N. Copper oxide and zinc oxide nanomaterials act as inhibitors of multidrug resistance transport in sea urchin embryos: Their role as chemosensitizers. Environ. Sci. Technol. 2015, 49, 5760–5770. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Rico, C.M.; Zhao, L.; Adeleye, A.S.; Keller, A.A.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Toxic effects of copper-based nanoparticles or compounds to lettuce (Lactuca sativa) and alfalfa (Medicago sativa). Environ. Sci. Process. Impacts 2015, 17, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Markus, A.; Parsons, J.; Roex, E.; De Voogt, P.; Laane, R. Modeling aggregation and sedimentation of nanoparticles in the aquatic environment. Sci. Total Environ. 2015, 506, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanopart. Res. 2013, 15, 1692. [Google Scholar] [CrossRef]
- Zhang, W.X.; Elliott, D.W. Applications of iron nanoparticles for groundwater remediation. Remediat. J. 2006, 16, 7–21. [Google Scholar] [CrossRef]
- Elbahri, M.; Homaeigohar, S.; Dai, T.; Abdelaziz, R.; Khalil, R.; Zillohu, A.U. Smart Metal-Polymer Bionanocomposites as Omnidirectional Plasmonic Black Absorbers Formed by Nanofluid Filtration. Adv. Funct. Mater. 2012, 22, 4771–4777. [Google Scholar] [CrossRef]
- Gibson, C.T.; Turner, I.J.; Roberts, C.J.; Lead, J.R. Quantifying the Dimensions of Nanoscale Organic Surface Layers in Natural Waters. Environ. Sci. Technol. 2007, 41, 1339–1344. [Google Scholar] [CrossRef]
- Tipping, E. The adsorption of aquatic humic substances by iron oxides. Geochim. Cosmochim. Acta 1981, 45, 191–199. [Google Scholar] [CrossRef]
- Hyung, H.; Fortner, J.D.; Hughes, J.B.; Kim, J.-H. Natural organic matter stabilizes carbon nanotubes in the aqueous phase. Environ. Sci. Technol. 2007, 41, 179–184. [Google Scholar] [CrossRef]
- Buffle, J.; Wilkinson, K.J.; Stoll, S.; Filella, M.; Zhang, J. A generalized description of aquatic colloidal interactions: The three-colloidal component approach. Environ. Sci. Technol. 1998, 32, 2887–2899. [Google Scholar] [CrossRef]
- Yamashita, Y.; Tsukasaki, A.; Nishida, T.; Tanoue, E. Vertical and horizontal distribution of fluorescent dissolved organic matter in the Southern Ocean. Mar. Chem. 2007, 106, 498–509. [Google Scholar] [CrossRef]
- Wurl, O.; Obbard, J.P. A review of pollutants in the sea-surface microlayer (SML): A unique habitat for marine organisms. Mar. Pollut. Bull. 2004, 48, 1016–1030. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Keskar, G.; Wu, Y.; Wang, X.; Mount, A.S.; Klaine, S.J.; Moore, J.M.; Rao, A.M.; Ke, P.C. Detection of phospholipid-carbon nanotube translocation using fluorescence energy transfer. Appl. Phys. Lett. 2006, 89, 143118. [Google Scholar] [CrossRef]
- Kim, J.S.; Yoon, T.-J.; Yu, K.N.; Kim, B.G.; Park, S.J.; Kim, H.W.; Lee, K.H.; Park, S.B.; Lee, J.-K.; Cho, M.H. Toxicity and tissue distribution of magnetic nanoparticles in mice. Toxicol. Sci. 2005, 89, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Geiser, M.; Rothen-Rutishauser, B.; Kapp, N.; Schürch, S.; Kreyling, W.; Schulz, H.; Semmler, M.; Hof, V.I.; Heyder, J.; Gehr, P. Ultrafine particles cross cellular membranes by nonphagocytic mechanisms in lungs and in cultured cells. Environ. Health Perspect. 2005, 113, 1555–1560. [Google Scholar] [CrossRef]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Kong, J.; Chen, Y. Antibacterial activity of graphite, graphite oxide, graphene oxide, and reduced graphene oxide: Membrane and oxidative stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef]
- Nogueira, V.; Lopes, I.; Rocha-Santos, T.; Santos, A.L.; Rasteiro, G.M.; Antunes, F.; Gonçalves, F.; Soares, A.M.V.M.; Cunha, A.; Almeida, A.; et al. Impact of organic and inorganic nanomaterials in the soil microbial community structure. Sci. Total Environ. 2012, 424, 344–350. [Google Scholar] [CrossRef]
- Jaisi, D.P.; Elimelech, M. Single-walled carbon nanotubes exhibit limited transport in soil columns. Environ. Sci. Technol. 2009, 43, 9161–9166. [Google Scholar] [CrossRef]
- Reddy, P.V.L.; Hernandez-Viezcas, J.A.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Lessons learned: Are engineered nanomaterials toxic to terrestrial plants? Sci. Total Environ. 2016, 568, 470–479. [Google Scholar] [CrossRef]
- Tong, Z.; Bischoff, M.; Nies, L.; Applegate, B.; Turco, R.F. Impact of Fullerene (C60) on a Soil Microbial Community. Environ. Sci. Technol. 2007, 41, 2985–2991. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.; Belozerova, I. Influence of metal nanoparticles on the soil microbial community and germination of lettuce seeds. Water Air Soil Pollut. 2009, 197, 143–148. [Google Scholar] [CrossRef]
- Johansen, A.; Pedersen, A.L.; Jensen, K.A.; Karlson, U.; Hansen, B.M.; Scott-Fordsmand, J.J.; Winding, A. Effects of C60 fullerene nanoparticles on soil bacteria and protozoans. Environ. Toxicol. Chem. Int. J. 2008, 27, 1895–1903. [Google Scholar] [CrossRef]
- Rodrigues, D.F.; Jaisi, D.P.; Elimelech, M. Toxicity of functionalized single-walled carbon nanotubes on soil microbial communities: Implications for nutrient cycling in soil. Environ. Sci. Technol. 2012, 47, 625–633. [Google Scholar] [CrossRef]
- Feng, Y.; Cui, X.; He, S.; Dong, G.; Chen, M.; Wang, J.; Lin, X. The role of metal nanoparticles in influencing arbuscular mycorrhizal fungi effects on plant growth. Environ. Sci. Technol. 2013, 47, 9496–9504. [Google Scholar] [CrossRef] [PubMed]
- Rispail, N.; De Matteis, L.; Santos, R.; Miguel, A.S.; Custardoy, L.; Testillano, P.S.; Risueño, M.C.; Pérez-de-Luque, A.; Maycock, C.; Fevereiro, P. Quantum dot and superparamagnetic nanoparticle interaction with pathogenic fungi: Internalization and toxicity profile. Acs Appl. Mater. Interfaces 2014, 6, 9100–9110. [Google Scholar] [CrossRef]
- Luo, Z.; Qiu, Z.; Chen, Z.; Du Laing, G.; Liu, A.; Yan, C. Impact of TiO2 and ZnO nanoparticles at predicted environmentally relevant concentrations on ammonia-oxidizing bacteria cultures under ammonia oxidation. Environ. Sci. Pollut. Res. 2015, 22, 2891–2899. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zhang, P.; Zhang, Z.; He, X.; Li, Y.; Zhang, J.; Zheng, L.; Chu, S.; Yang, K.; Zhao, Y. Origin of the different phytotoxicity and biotransformation of cerium and lanthanum oxide nanoparticles in cucumber. Nanotoxicology 2015, 9, 262–270. [Google Scholar] [CrossRef]
- Li, K.-E.; Chang, Z.-Y.; Shen, C.-X.; Yao, N. Toxicity of nanomaterials to plants. In Nanotechnology and Plant Sciences; Springer: Berlin/Heidelberg, Germany, 2015; pp. 101–123. [Google Scholar]
- Majumdar, S.; Peralta-Videa, J.R.; Bandyopadhyay, S.; Castillo-Michel, H.; Hernandez-Viezcas, J.-A.; Sahi, S.; Gardea-Torresdey, J.L. Exposure of cerium oxide nanoparticles to kidney bean shows disturbance in the plant defense mechanisms. J. Hazard. Mater. 2014, 278, 279–287. [Google Scholar] [CrossRef]
- Du, W.; Tan, W.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L.; Ji, R.; Yin, Y.; Guo, H. Interaction of metal oxide nanoparticles with higher terrestrial plants: Physiological and biochemical aspects. Plant Physiol. Biochem. 2017, 110, 210–225. [Google Scholar] [CrossRef]
- Holden, P.A.; Klaessig, F.; Turco, R.F.; Priester, J.H.; Rico, C.M.; Avila-Arias, H.; Mortimer, M.; Pacpaco, K.; Gardea-Torresdey, J.L. Evaluation of exposure concentrations used in assessing manufactured nanomaterial environmental hazards: Are they relevant? Environ. Sci. Technol. 2014, 48, 10541–10551. [Google Scholar] [CrossRef] [PubMed]
- Priester, J.H.; Moritz, S.C.; Espinosa, K.; Ge, Y.; Wang, Y.; Nisbet, R.M.; Schimel, J.P.; Susana Goggi, A.; Gardea-Torresdey, J.L.; Holden, P.A. Damage assessment for soybean cultivated in soil with either CeO2 or ZnO manufactured nanomaterials. Sci. Total Environ. 2017, 579, 1756–1768. [Google Scholar] [CrossRef]
- Feizi, H.; Rezvani Moghaddam, P.; Shahtahmassebi, N.; Fotovat, A. Impact of Bulk and Nanosized Titanium Dioxide (TiO2) on Wheat Seed Germination and Seedling Growth. Biol. Trace Elem. Res. 2012, 146, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.H.; Al-Whaibi, M.H. Role of nano-SiO2 in germination of tomato (Lycopersicum esculentum seeds Mill.). Saudi J. Biol. Sci. 2014, 21, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.; Mohamed, M.S.; Gao, W.; Maekawa, T.; Yoshida, Y.; Ajayan, P.M.; Kumar, D.S. Effect of Carbon Nanomaterials on the Germination and Growth of Rice Plants. J. Nanosci. Nanotechnol. 2012, 12, 2212–2220. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012, 2012, 26. [Google Scholar] [CrossRef]
- Gardea-Torresdey, J.L.; Rico, C.M.; White, J.C. Trophic Transfer, Transformation, and Impact of Engineered Nanomaterials in Terrestrial Environments. Environ. Sci. Technol. 2014, 48, 2526–2540. [Google Scholar] [CrossRef]
- Keller, A.A.; Fournier, E.; Fox, J. Minimizing impacts of land use change on ecosystem services using multi-criteria heuristic analysis. J. Environ. Manag. 2015, 156, 23–30. [Google Scholar] [CrossRef]
- McKee, M.S.; Filser, J. Impacts of metal-based engineered nanomaterials on soil communities. Environ. Sci. Nano 2016, 3, 506–533. [Google Scholar] [CrossRef]
- Kwak, J.I.; An, Y.-J. The current state of the art in research on engineered nanomaterials and terrestrial environments: Different-scale approaches. Environ. Res. 2016, 151, 368–382. [Google Scholar] [CrossRef]




| Composition | Structure | Water Pollutant | Removal Mechanism | Nanomaterial Role | Ref |
|---|---|---|---|---|---|
| Cu NP/CNT/PVDF | Nanocomposite film | arsenic | Dynamic adsorption and oxidation | As oxidizer and adsorbent | [69] |
| Co doped ZrO2 | Nanoparticle | MO dye | Visible light photodegradation | As photocatalyst | [70] |
| NiO | Nanoparticle | ciprofloxacin | Adsorption | As adsorbent | [71] |
| Fe3O4 NP/AC | Nanocomposite particle | MO and RhB dye | Adsorption | To enable magnetic recovery and to raise adsorption capacity | [72] |
| Fe3O4@MIL-100(Fe) | Nanocomposite MOF | diclofenac sodium (DCF) | Adsorption and photodegradation | Magnetic recovery | [73] |
| FexCo3−xO4 | Nanoparticle | CR dye | Adsorption | To offer adsorption activity with easy magnetic recovery | [74] |
| ZnO-ZnFe2O4 | Nanofiber | CR dye | Adsorption | To raise adsorption efficiency | [75] |
| Ag-ZnO/PANI | Nanocomposite film | BG dye | Adsorption | To raise adsorption efficiency | [76] |
| ZnS NP/PES | Film membrane | Humic acid | Filtration assisted by the antifoulant NPs | As antifouling agent | [77] |
| ZnO/KGM-PVA | Nanofiber membrane | MO dye | Visible light Photodegradation | To induce photocatalytic and antibacterial activity | [78] |
| Boehmite NP/EPVC | Nanocomposite Film membrane | BSA | Ultrafiltration | To improve hydrophilicity and water flux | [79] |
| Ag NP/wood | Nanocomposite Film membrane | MB dye | physical adsorption and catalytic degradation | Dye adsorption and antibacterial activity | [80] |
| (3-aminopropyl-triethoxysilane) APTES-Fe3O4 NP/PES | Nanocomposite Film membrane | arsenic | Adsorption | Heavy metal ion adsorption | [81] |
| PEI/PD/Ag NP | Nanocomposite Film membrane | BSA/HA/Oil | Ultrafiltration | As anti-fouling and anti-biofouling agent | [82] |
| Carbon dioxide plasma treated PVDF | Nanofiber membrane | CV dye and iron oxide NPs | size exclusion and adsorption | Ionic selectivity | [83] |
| Bentonite NP/PA | Nanocomposite Film membrane | NaCl | Reverse osmosis | To raise water permeability | [84] |
| PVA/PAN | Nanofiber membrane | Nanoparticles and Cr (VI) and Cd (II) ions | Adsorption and microfiltration | PVA nanofibers as the mechanical support and PAN nanofibers for selective adsorption of the ions | [85] |
| Clay NP/mixed matrix PS | Nanocomposite Film membrane | PEG and sodium alginate | Ultrafiltration | To improve antifouling properties, membrane thermal/ mechanical resistance and permeability with minimal loss in rejection | [37] |
| Clay NP/mixed matrix PS | Nanocomposite Film membrane | PEG and sodium alginate | Ultrafiltration | To improve antifouling properties, membrane thermal/ mechanical resistance and permeability with minimal loss in rejection | [37] |
| CS NP&Ag-CS NP/polyphenylsulfone | Nanocomposite Hollow fiber membrane | Reactive black dye | Adsorption | To improve porosity, dye rejection efficiency, hydrophilicity, and antifouling property | [86] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghadimi, M.; Zangenehtabar, S.; Homaeigohar, S. An Overview of the Water Remediation Potential of Nanomaterials and Their Ecotoxicological Impacts. Water 2020, 12, 1150. https://doi.org/10.3390/w12041150
Ghadimi M, Zangenehtabar S, Homaeigohar S. An Overview of the Water Remediation Potential of Nanomaterials and Their Ecotoxicological Impacts. Water. 2020; 12(4):1150. https://doi.org/10.3390/w12041150
Chicago/Turabian StyleGhadimi, Mehrnoosh, Sasan Zangenehtabar, and Shahin Homaeigohar. 2020. "An Overview of the Water Remediation Potential of Nanomaterials and Their Ecotoxicological Impacts" Water 12, no. 4: 1150. https://doi.org/10.3390/w12041150
APA StyleGhadimi, M., Zangenehtabar, S., & Homaeigohar, S. (2020). An Overview of the Water Remediation Potential of Nanomaterials and Their Ecotoxicological Impacts. Water, 12(4), 1150. https://doi.org/10.3390/w12041150






