Influence of Cascading River–Lake Systems on the Dynamics of Nutrient Circulation in Catchment Areas
Abstract
:1. Introduction
2. Discussion
2.1. The Effect of Water Exchange Intensity on the Nutrient Circulation Dynamics
2.1.1. Lake Basin Shape
2.1.2. Weather Conditions
2.1.3. Global Warming
2.1.4. Hydrological Conditions
2.1.5. Water Retention
2.1.6. Nutrient Exchange
2.1.7. Biogeochemical Barriers
2.1.8. Role of Water Body
2.2. The Role of Bottom Sediments in the Nutrient Cycle in the River–Lake System
2.2.1. Nutrients
2.2.2. Metals
2.2.3. Granulometric Structure
2.3. The Role of Macrophytes in Nutrient Circulation in River–Lake Systems
2.3.1. Species of Macrophytes
2.3.2. Impact of Sediments
2.4. The Role of Bioindicators in Nutrient Circulation in River–Lake Systems
3. Summary
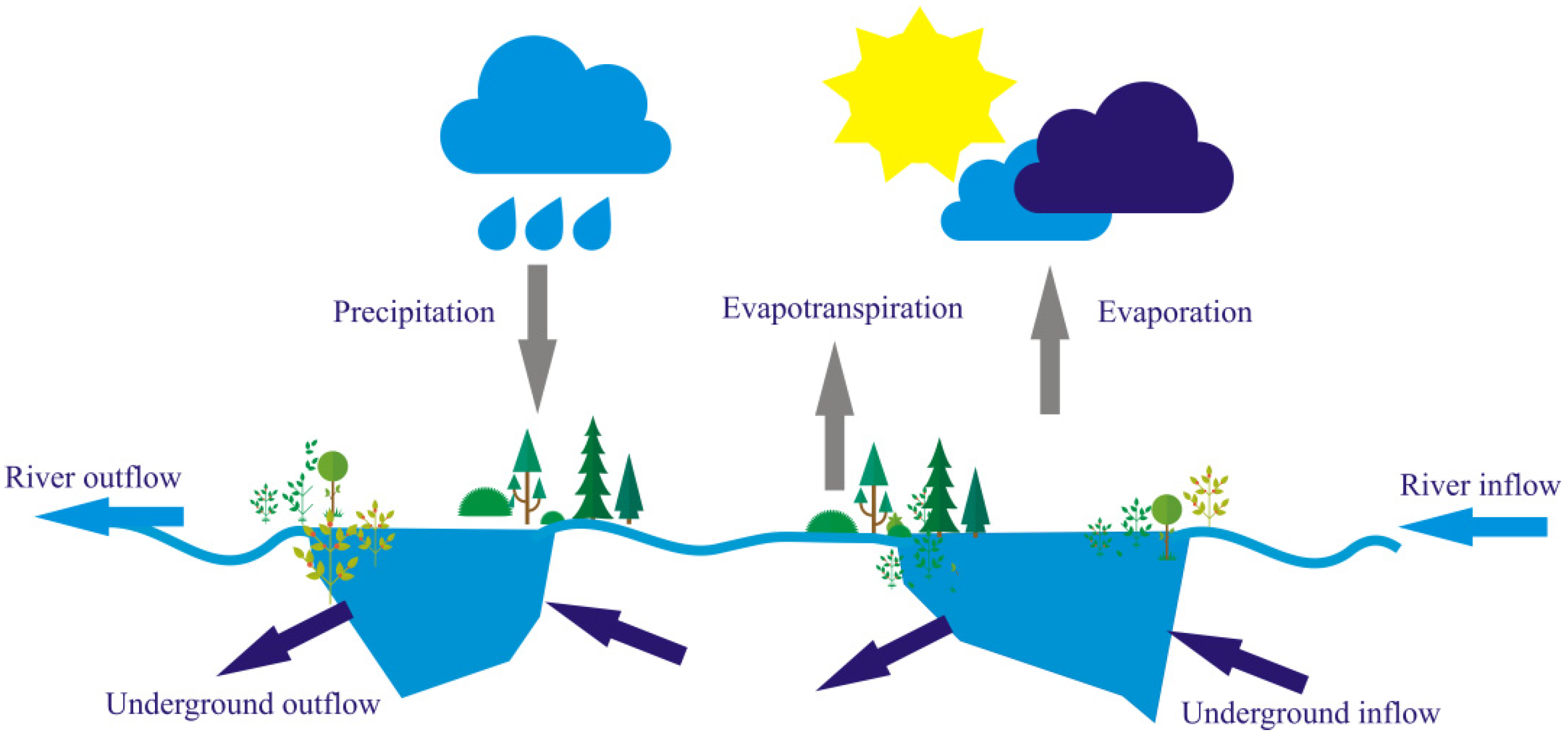
- River–lake systems are characterised by intensive water exchange which is determined by meteorological conditions. In turn, these indirectly affect hydrological conditions (Section 2.1.1 and Section 2.1.2).
- The water exchange is accompanied by the exchange of components between elements of the systems (Section 2.1.6).
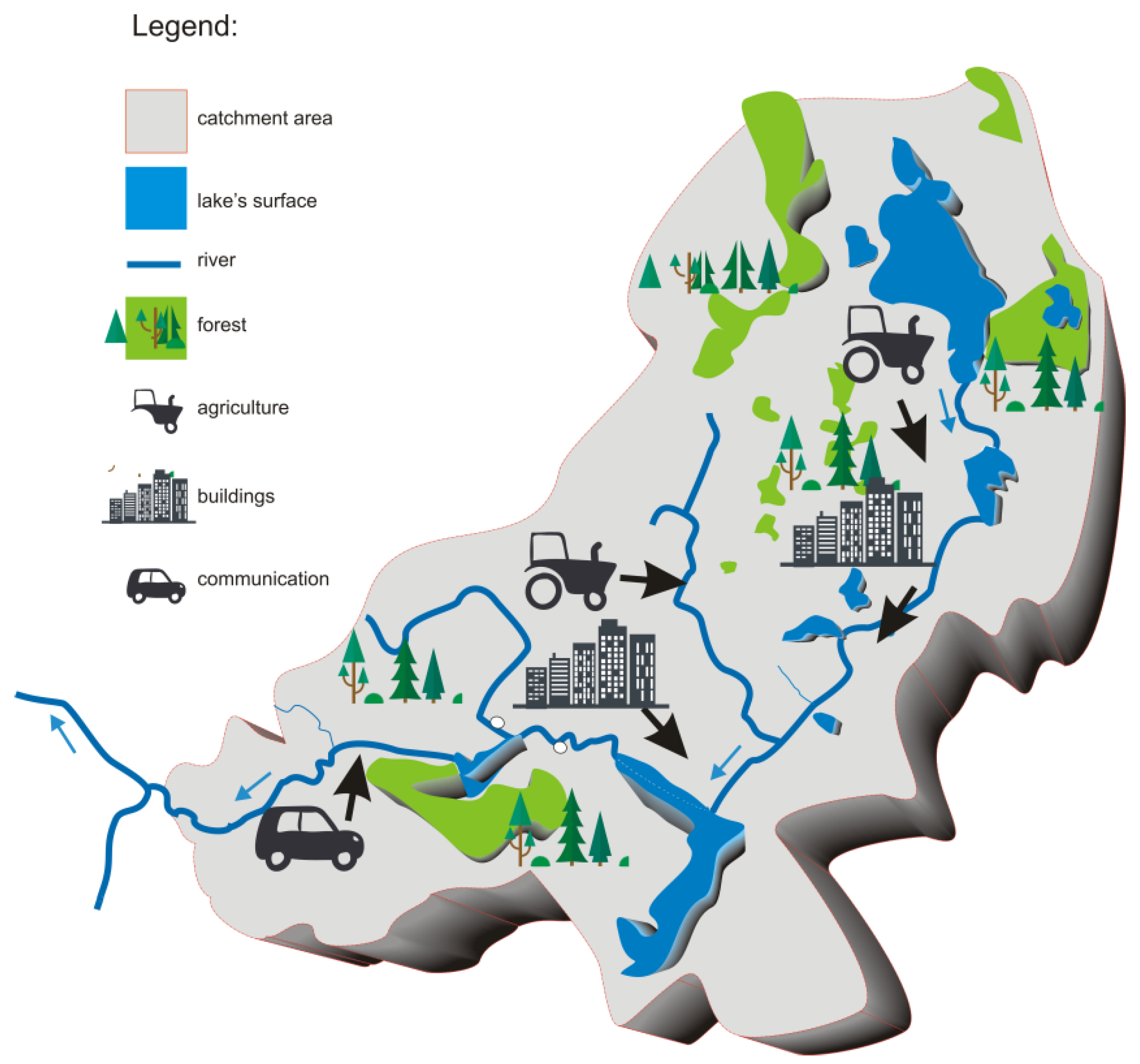
- Rivers flowing through water bodies affect their morphometric conditions. The deposition of various rock-debris fractions affects the lake bed shaping and renders them a barrier to the matter which is temporarily deposited in bottom deposits found in the contact zone between rivers and water bodies (Section 2.2.3).
- The structure of deposits and their granulometric composition have an effect on the component content in macrophyte tissues. The assimilation of pollutants by the tissues of plants growing in river–lake systems is determined by their abundance in the substrate (Section 2.3.2).
- River–lake systems act as natural biogeochemical barriers, thus limiting the transport of pollutants outside the ecosystem; this, however, results in the degradation of the water bodies (Section 2.1.7).
- Not only is the role of water bodies being part of a river–lake system determined by the size of its external load of components originating from the catchment area but also by morphometric, hydrological and meteorological conditions (Section 2.1.8).
- Rivers feeding lakes of a river–lake system have a significant effect on the quality of water in the water bodies themselves as well as on the distribution of pollutants in their bottom deposits. Agricultural development and unregulated water and wastewater management within the catchment area contributes to an increase in the transport of components along with their waters (Section 2.2.1, Section 2.2.2, and Section 2.2.3).
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saunders, D.I.; Kalff, J. Nitrogen retention in wetlands, lakes and rivers. Hydrobiologia 2001, 443, 205–212. [Google Scholar] [CrossRef]
- Schoen, M.E.; Xue, X.; Wood, A.; Hawkins, T.R.; Garland, J.; Ashbolt, N.J. Cost, energy, global warming, eutrophication and local human health impacts of community water and sanitation service options. Water Res. 2017, 109, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Schewe, J.; Heinke, J.; Gerten, D.; Haddeland, I.; Arnell, N.W.; Clar, D.B. Multimodel assessment of water scarcity under climate change. Proc. Natl. Acad. Sci. USA 2014, 111, 3245–3250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okoński, B.; Miler, A.T.; Panfil, M. The dynamic of climate elements and land drainage impact on hydrological conditions in lakeland blind drainage area. J. Water Land Dev. 2009, 13, 225–238. [Google Scholar] [CrossRef]
- Haddeland, I.; Heinke, J.; Biemans, M.; Eisner, S.; Flörke, M.; Hanasaki, N.; Konzmann, M.; Ludwig, L.; Masaki, Y.; Schewe, J.; et al. Global water resources affected by human interventions and climate change. Proc. Natl. Acad. Sci. USA 2014, 111, 3251–3256. [Google Scholar] [CrossRef] [Green Version]
- Wen, L.; Lv, S.; Li, Z.; Zhao, L.; Nagabhatla, N. Impacts of the two biggest lakes on local temperature and precipitation in the Yellow River source region of the Tibetan Plateau. Adv. Meteorol. 2015, 10. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Yao, J. Estimation of transport trajectory and residence time in large river–lake systems: Application to Poyang Lake (China) using a combined model approach. Water 2015, 7, 5203–5223. [Google Scholar] [CrossRef] [Green Version]
- Szymczyk, S.; Szyperek, U. Erozja chemiczna gleb obszarów pojeziernych. Cz. 1. Odpływ związków azotu. Acta Agrophysica 2005, 5, 175–183. [Google Scholar]
- Grabińska, B.; Koc, J.; Glińska-Lewczuk, K. Wpływ użytkowania zlewni Narwi na zagrożenia wód związkami fosforu [The effect of land use in the Narew River catchment basin on water pollution by phosphorus compounds]. Nawozy i Nawożenie. Fertilizers and Fertilization 2004, 2, 178–191. [Google Scholar]
- Ryczek, M.; Kruk, E.; Klatka, S.; Malec, M. Modeling evaluation of surface water erosion risk in agricultural mountain basin. Inżynieria Ekologiczna 2017, 18, 9–17. [Google Scholar] [CrossRef] [Green Version]
- Li, F.; Pan, Y.; Xie, Y.; Chen, X.; Deng, Z.; Li, X.; Hou, Z.; Tang, Y. Different roles of three emergent macrophytes in promoting sedimentation in Dongting Lake, China. Aquat. Sci. 2016, 78, 159–169. [Google Scholar] [CrossRef]
- Alifujiang, Y.; Abuduwaili, J.; Ma, L.; Samat, A.; Groll, M. System dynamics modelling of water level variations of lake. Issyk-Kul, Kyrgyzstan. Water 2017, 9, 989. [Google Scholar] [CrossRef] [Green Version]
- Allan, J.D. Landscapes and riverscapes: The influence of land use on stream ecosystems. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 257–284. [Google Scholar] [CrossRef] [Green Version]
- Doody, D.G.; Withers, P.J.; Dils, R.M.; McDowell, R.W.; Smith, V.; McElarney, Y.R.; Dunbar, M.; Daly, D. Optimizing land use for the delivery of catchment ecosystem services. Front. Ecol. Environ. 2016, 14, 325–332. [Google Scholar] [CrossRef] [Green Version]
- Jeppesen, E.; Brucet, S.; Naselli-Flores, L.; Papastergiadou, E.; Stefanidis, K.; Noges, T.; Noges, P.; Attayde, J.L.; Zohary, T.; Coppeens, J.; et al. Ecological impacts of global warming and water abstraction on lakes and reservoirs due to changes in water level and related changes in salinity. Hydrobiologia 2015, 750, 201–227. [Google Scholar] [CrossRef]
- Glińska-Lewczuk, K. Oxbow lakes as biogeochemical filters for nutrient outflow from agricultural areas. In Dynamics and Biogeochemistry of River Corridors and Wetlands. In Proceedings of the Symposium S4 Held during the Seventh IAHS Scientific Assembly, Foz do Iguacu, Brazil, 17 April 2005. [Google Scholar]
- Glińska-Lewczuk, K. Effect of land use and lake presence on chemical diversity of the Łyna river system. Pol. J. Environ. Stud. 2006, 15, 259–269. [Google Scholar]
- Hillbricht-Ilkowska, A.; Kostrzewska-Szlakowska, I. Ocena ładunku i stanu zagrożenia jezior Rzeki Krutyni (Pojezierze Mazurskie) oraz zależności pomiędzy ładunkiem a stężeniem fosforu w jeziorach. In Funkcjonowanie Systemów Rzeczno-Jeziornych w Krajobrazie Pojeziernym: Rzeka Krutynia (Pojezierze Mazurskie); Instytut Ekologii PAN: Dziekanów Leśny, Polska, 1996; ISBN 83-87089-02-8. [Google Scholar]
- Błaszkiewicz, M.; Piotrowski, J.A.; Brauer, A.; Gierszewski, P.; Kordowski, J.; Kramkowski, M.; Lamparski, P.; Lorenz, S.; Noryśkiewicz, A.M.; Ott, F.; et al. Climatic and morphological controls on diachronous postglacial lake and river valley evolution in the area of Last Glaciation, northern Poland. Quat. Sci. Rev. 2015, 109, 13–27. [Google Scholar] [CrossRef]
- Halecki, W.; Kruk, E.; Ryczek, M. Estimations of nitrate nitrogen, total phosphorus flux and suspended sediment concentration (SSC) as indicators of surface-erosion processes using an ANN (Artificial Neural Network) based on geomorphological parameters in mountainous catchments. Ecol. Indic. 2018, 91, 461–469. [Google Scholar] [CrossRef]
- Andersen, J.M. Water quality management in the River Gudenaa, a Danish lake-stream-estuary system. Hydrobiologia 1994, 275, 499–507. [Google Scholar] [CrossRef]
- Bajkiewicz-Grabowska, E. Obieg Materii w Systemach Rzeczno-Jeziornych; Uniwersytet Warszawski: Warsaw, Poland, 2002. [Google Scholar]
- Hillbricht-Ilkowska, A. Shallow lakes in lowland river systems: Role in transport and transformations of nutrients and in biological diversity. In Shallow Lakes’ 98; Springer: Dordrecht, The Netherlands, 1999; pp. 349–358. [Google Scholar]
- Marszałek, H.; Górniak, D. Changes in water chemistry along the newly formed High Arctic fluvial–lacustrine system of the Brattegg Valley (SW Spitsbergen, Svalbard). Environ. Earth Sci. 2017, 76, 449. [Google Scholar] [CrossRef] [Green Version]
- Sojka, M.; Siepak, M.; Jaskuła, J.; Wicher-Dysarz, J. Heavy metal transport in a river-reservoir system: A case study from central Poland. Pol. J. Environ. Stud. 2018, 27, 1725–1734. [Google Scholar] [CrossRef]
- Tong, Y.; Li, J.; Qi, M.; Zhang, X.; Wang, M.; Liu, X.; Zhang, W.; Wang, X.; Liu, Y.; Lin, Y. Impacts of water residence time on nitrogen budget of lakes and reservoirs. Sci. Total Environ. 2019, 646, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Huziy, O.; Sushama, L. Lake–river and lake–atmosphere interactions in a changing climate over Northeast Canada. Clim. Dyn. 2017, 48, 3227–3246. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, Q.; Wan, R.; Lai, X.; Jiang, X.; Li, L.; Lu, Y. Lake hydrology, water quality and ecology impacts of altered river–lake interactions: Advances in research on the middle Yangtze River. Hydrol. Res. 2016, 47, 1–7. [Google Scholar] [CrossRef]
- Kuriata-Potasznik, A.; Szymczyk, S. Variability of the water availability in a river lake system—A case study of Lake Symsar. J. Water Land Dev. 2016, 31, 87–96. [Google Scholar] [CrossRef] [Green Version]
- Harvey, J.; Gooseff, M. River corridor science: Hydrologic exchange and ecological consequences from bedforms to basins. Water Resour. Res. 2015, 51, 6893–6922. [Google Scholar] [CrossRef] [Green Version]
- Herbst, M.; Kappen, L. The ratio of transpiration versus evaporation in a reed belt as influenced by Feather conditions. Aquat. Bot. 1999, 63, 113–125. [Google Scholar] [CrossRef]
- Borsuk, S. Ekologiczno-Geochemiczny System Oceny Jakości Wód Jeziornych. Problemy. Metodologia. Modelowanie [Ecological-Geochemical Assessment System of Lake Water Quality]; Wydawnictwo Uczelniane UTP: Polska, Bydgoszcz, 2014; pp. 1–198. [Google Scholar]
- Cardille, J.; Coe, M.T.; Vano, J.A. Impacts of climate variation and catchment area on water balance and Lake hydrologic type in groundwater-dominated systems: A generic lake model. Earth Interact. 2004, 8, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Fu, B.J.; He, C.S.; Sun, G.; Gao, G.Y. A comparative analysis of forest cover and catchment water yield relationships in northern China. For. Ecol. Manag. 2011, 262, 1189–1198. [Google Scholar] [CrossRef]
- Barroso, G.F.; Goncalves, M.A.; Garcia, F.D.C. The morphometry of Lake Palmas, a deep natural Lake in Brazil. PLoS ONE 2014, 9, e111469. [Google Scholar] [CrossRef]
- Sahoo, G.B.; Schladow, S.G.; Reuter, J.E.; Coats, R.; Dettinger, M.; Riverson, J.; Costa-Cabral, M. The response of Lake Tahoe to climate change. Clim. Chang. 2013, 116, 71–95. [Google Scholar] [CrossRef]
- Hamidi, S.A.; Bravo, H.R.; Klump, J.V.; Waples, J.T. The role of circulation and heat fluxes in the formation of stratification leading to hypoxia in Green Bay, Lake Michigan. J. Great Lakes Res. 2015, 41, 1024–1036. [Google Scholar] [CrossRef]
- Kuriata-Potasznik, A. The functioning of a water body within a fluvio-lacustrine system as an effect of excessive nitrogen loading—the case of Lake Symsar and its drainage area (Northeastern Poland). Water 2018, 10, 1163. [Google Scholar] [CrossRef] [Green Version]
- Schüler, G.; Schobel, S.; Wilkinson, K.; Schultze, B.; Karl, S.; Scherzer, J. The impacts of a changing climate on catchment water balance and forest management. Ecohydrology 2017, 10, e1805. [Google Scholar] [CrossRef]
- Zhang, J.; Feng, L.; Chen, L.; Wang, D.; Dai, M.; Xu, W.; Yan, T. Water Compensation and Its Implication of the Three Gorges Reservoir for the River-Lake System in the Middle Yangtze River, China. Water 2018, 10, 1011. [Google Scholar] [CrossRef] [Green Version]
- Sojka, M.; Jaskuła, J.; Wicher-Dysarz, J.; Dysarz, T. Assessment of dam construction impact on hydrological regime changes in lowland river–A case of study: The Stare Miasto reservoir located on the Powa River. J. Water Land Dev. 2016, 30, 119–125. [Google Scholar] [CrossRef] [Green Version]
- Kuriata-Potasznik, A.; Szymczyk, S.; Pilejczyk, D. Effect of bottom sediments on the nutrient and metal concentration in macrophytes of river-lake systems. Ann. Limnol. Int. J. Limnol. 2018, 54, 1. [Google Scholar] [CrossRef] [Green Version]
- Potasznik, A.; Szymczyk, S.; Sidoruk, M.; Świtajska, I. Role of lake Symsar in the reduction of phosphorus concentration in surface runoff from agricultural lands. J. Water Land Dev. 2014, 2, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Michalak, A.M.; Anderson, E.J.; Beletsky, D.; Boland, S.; Bosch, N.S.; Bridgeman, T.B.; DePinto, J.V. Record-setting algal bloom in Lake Erie caused by agricultural and meteorological trends consistent with expected future conditions. Proc. Natl. Acad. Sci. USA 2013, 110, 6448–6452. [Google Scholar] [CrossRef] [Green Version]
- Iglesias, M.L.; Devesa-Rey, R.; Pérez-Moreira, R.; Díaz-Fierros, F.; Barral, M.T. Phosphorus transfer across boundaries: From basin soils to river bed sediments. J. Soils Sediments 2011, 11, 1125. [Google Scholar] [CrossRef]
- Fraterrigo, J.M.; Downing, J.A. The influence of land use on lake nutrients varies with watershed transport capacity. Ecosystems 2008, 11, 1021–1034. [Google Scholar] [CrossRef]
- Tao, Y.; Dan, D.; Chengda, H.; Qiujin, X.; Fengchang, W. Response of sediment calcium and magnesium species to the regional acid deposition in eutrophic Taihu Lake, China. Environ. Sci. Pollut. Res. 2016, 23, 22489–22499. [Google Scholar] [CrossRef] [PubMed]
- Potasznik, A.; Szymczyk, S. Magnesium and calcium concentrations in the surface water and bottom deposits of a river-lake system. J. Elem. 2015, 20, 677–692. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Gao, Y.; Zhang, H.; Cao, J.; Cai, J.; Kong, X. Water pollution control technology and strategy for river–lake systems: A case study in Gehu Lake and Taige Canal. Ecotoxicology 2011, 20, 1154–1159. [Google Scholar] [CrossRef] [PubMed]
- Kozerski, H.P.; Behrendt, H.; Köhler, J. The N and P budget of the shallow, flushed lake Müggelsee: Retention, external and internal load. In Shallow Lakes’ 98; Springer: Dordrecht, The Netherlands, 1999; pp. 159–166. [Google Scholar]
- Jensen, J.P.; Jeppesen, E.; Kristensen, P.; Christensen, P.B.; Søndergaard, M. Nitrogen loss and denitrification as studied in relation to reductions in nitrogen loading in a shallow, hypertrophic lake (Lake Søbygård, Denmark). Int. Rev. Gesamten Hydrobiol. Hydrogr. 1992, 77, 29–42. [Google Scholar] [CrossRef]
- Jha, P.K.; Minagawa, M. Assessment of denitrification process in lower Ishikari river system, Japan. Chemosphere 2013, 93, 1726–1733. [Google Scholar] [CrossRef]
- Cook, P.L.; Aldridge, K.T.; Lamontagne, S.; Brookes, J.D. Retention of nitrogen, phosphorus and silicon in a large semi-arid riverine lake system. Biogeochemistry 2010, 99, 49–63. [Google Scholar] [CrossRef]
- Lossow, K. Znaczenie jezior w krajobrazie młodoglacjalnym Pojezierza Mazurskiego (Significance of lakes in postglacial landscape of the Masurian Lakeland). Zesz. Probl. Post. Nauk Rol. 1996, 431, 47–59. [Google Scholar]
- Dunalska, J. Influence of limited water flow in a pipeline on the nutrients budget in a lake restored by hypolimnetic withdrawal method. Pol. J. Environ. Stud. 2002, 11, 631–637. [Google Scholar]
- Kneis, D.; Knoesche, R.; Bronstert, B. Analysis and simulation of nutrient retention and management for a lowland river-lake system. Hydrol. Earth Syst. Sci. Discuss. Eur. Geosci. Union 2006, 10, 575–588. [Google Scholar] [CrossRef] [Green Version]
- Maślanka, W.; Jańczak, J. Circulation of biogenic substances in the Kamionka river-lake system. Limnol. Rev. 2010, 10, 95–101. [Google Scholar] [CrossRef] [Green Version]
- Moss, B.; Kosten, S.; Meerhoff, M.; Battarbee, R.W.; Jeppesen, E.; Mazzeo, N.; Paerl, H. Allied attack: Climate change and eutrophication. Inland Waters 2011, 1, 101–105. [Google Scholar] [CrossRef] [Green Version]
- Aighewi, I.T.; Nosakhare, O.K.; Ishaque, A.B. Land use–Land cover changes and sewage loading in the lower eastern shore watersheds and coastal bays of Maryland: Implications for surface water quality. J. Coast. Res. 2013, 29, 1073–1082. [Google Scholar] [CrossRef]
- Jaskuła, J.; Sojka, M.; Wicher-Dysarz, J.; Dysarz, T. Trend of changes in physicochemical state of the river Ner. J. Ecol. Eng. 2016, 17. [Google Scholar] [CrossRef]
- Potasznik, A.; Szymczyk, S. Does inflow of water river shape the nutrient content of lake sediments? J. Elem. 2016, 21, 471–484. [Google Scholar] [CrossRef]
- Han, Q.; Wang, B.; Liu, C.Q.; Wang, F.; Peng, X.; Liu, X.L. Carbon biogeochemical cycle is enhanced by damming in a karst river. Sci. Total Environ. 2018, 616, 1181–1189. [Google Scholar] [CrossRef]
- Karthe, D.; Chalov, S.; Moreido, V.; Pashkina, M.; Romanchenko, A.; Batbayar, G.; Kalugin, A.; Westphal, K.; Malsy, M.; Flörke, M. Assessment of runoff, water and sediment quality in the Selenga River basin aided by a web-based geoservice. Water Resour. 2017, 44, 399–416. [Google Scholar] [CrossRef]
- Wang, A.J.; Bong, C.W.; Xu, Y.H.; Hassan, M.H.A.; Ye, X.; Bakar, A.F.A.; Li, H.; Lai, Z.K.; Xu, J.; Loh, K.H. Assessment of heavy metal pollution in surficial sediments from a tropical river-estuary-shelf system: A case study of Kelantan River, Malaysia. Mar. Pollut. Bull. 2017, 125, 492–500. [Google Scholar] [CrossRef]
- Ciazela, J.; Siepak, M.; Wojtowicz, P. Tracking heavy metal contamination in a complex river-oxbow lake system: Middle Odra Valley, Germany/Poland. Sci. Total Environ. 2018, 616, 996–1006. [Google Scholar] [CrossRef]
- Kuriata-Potasznik, A.; Szymczyk, S.; Skwierawski, A.; Glińska-Lewczuk, K.; Cymes, I. Heavy metal contamination in the surface layer of bottom sediments in a flow-through lake: A case study of Lake Symsar in Northern Poland. Water 2016, 8, 358. [Google Scholar] [CrossRef]
- Tylmann, W.; Łysek, K.; Kinder, M.; Pempkowiak, J. Regional pattern of heavy metal content in lake sediments in northeastern Poland. Water Air Soil Pollut. 2011, 216, 217–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrović, D.; Jančić, D.; Furdek, M.; Mikac, N.; Krivokapić, S. Aquatic plant Trapa natans L. as bioindicator of trace metal contamination in a freshwater lake (Skadar Lake, Montenegro). Acta Bot. Croat. 2016, 75, 75–236. [Google Scholar] [CrossRef] [Green Version]
- Varol, M. Dissolved heavy metal concentrations of the Kralkizi, Dicle and Batman dam reservoirs in the Tigris River basin, Turkey. Chemosphere 2013, 93, 954. [Google Scholar] [CrossRef] [PubMed]
- El-Amier, Y.A.; Elnaggar, A.; El-Alfy, M.A. Evaluation and mapping spatial distribution of bottom sediment heavy metal contamination in Burullus Lake, Egypt. Egypt. J. Basic Appl. Sci. 2017, 4, 55. [Google Scholar] [CrossRef] [Green Version]
- Thorslund, J.; Jarsjö, J.; Wällstedt, T.; Mörth, C.M.; Lychagin, M.Y.; Chalov, S.R. Speciation and hydrological transport of metals in non-acidic river systems of the Lake Baikal basin: Field data and model predictions. Reg. Environ. Chang. 2017, 17, 2007–2021. [Google Scholar] [CrossRef] [Green Version]
- Remor, M.B.; Sampaio, S.C.; de Rijk, S.; Boas, M.A.V.; Gotardo, J.T.; Pinto, E.T.; Schardong, F.A. Sediment geochemistry of the urban Lake Paulo Gorski. Int. J. Sediment Res. 2018. [Google Scholar] [CrossRef]
- Bąkowska, M.; Obolewski, K.; Ryszard, W. Does Dredging of Floodplain Lakes Affects The Structure of The Macrophytes and Epiphytic Fauna Inhabiting Stratiotes Aloides? E3S Web Conf. EDP Sci. 2017, 17, 5. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Bu, X.Q.; Wan, J.Y.; Dong, B.C.; Luo, F.L.; Li, H.L.; Yu, F.H. Impacts of sediment type on the performance and composition of submerged macrophyte communities. Aquat. Ecol. 2017, 51, 167–176. [Google Scholar] [CrossRef]
- Marchetti, Z.Y.; Scarabotti, P.A. Macrophyte assemblages in relation to environmental, temporal and spatial variations in lakes of a subtropical floodplain-river system, Argentina. Flora-Morphol. Distrib. Funct. Ecol. Plants 2016, 225, 82–91. [Google Scholar] [CrossRef]
- Madsen, J.D.; Chambers, P.A.; James, W.F.; Koch, E.W.; Westlake, D.F. The interaction between water movement, sediment dynamics and submersed macrophytes. Hydrobiologia 2001, 444, 71–84. [Google Scholar] [CrossRef]
- Barko, J.W.; Smart, R.M. Sediment-related mechanisms of growth limitation in submersed macrophytes. Ecology 1986, 67, 1328–1340. [Google Scholar] [CrossRef]
- Rovira, A.; Alcaraz, C.; Trobajo, R. Effects of plant architecture and water velocity on sediment retention by submerged macrophytes. Freshw. Biol. 2016, 61, 758–768. [Google Scholar] [CrossRef]
- Hajduk, W.; Kokoszka, P.; Korzec, K.; Kusior, B.; Ryczek, W.; Siwek, K.; Klich, M. Ocena jakości wód górnych partii rzeki Wisłoka (Polska południowa) na podstawie makrozoobentosu. Sci. Technol. Innov. 2018, 2, 19–26. [Google Scholar] [CrossRef]
- Bąkowska, M.; Mrozińska, N.; Szymańska, M.; Kolárová, N.; Obolewski, K. Periphyton Inhabiting Reeds in Polish Water Ecosystems. In Polish River Basins and Lakes–Part II.; Springer: Cham, Switzerland, 2020; pp. 1–25. [Google Scholar] [CrossRef]
- Obolewski, K.; Glińska-Lewczuk, K.; Strzelczak, A. Does hydrological connectivity determine the benthic macroinvertebrate structure in oxbow lakes? Ecohydrology 2015, 8, 1488–1502. [Google Scholar] [CrossRef]
- Lewandowski, K.; Jakubik, B. Mięczaki wodne systemu rzeczno-jeziornego Krutyni; Mazurski Park Krajobrazowy: Warmian-Masurian, Poland, 2019; pp. 62–71. [Google Scholar]
- Li, Q.; Wang, G.; Wang, H.; Shrestha, S.; Xue, B.; Sun, W.; Yu, J. Macrozoobenthos variations in shallow connected lakes under the influence of intense hydrologic pulse changes. J. Hydrol. 2020, 124755. [Google Scholar] [CrossRef]
- Szmigielska, M.; Wróbel, M.; Stojanowska, A.; Rybak, J. Ocena jakości wody (gmina Złoty Stok, Dolny Śląsk) zanieczyszczonej arsenem w oparciu o makrozoobentos. Inżynieria Ekol. 2018, 19. [Google Scholar] [CrossRef]
- Mimier, D.; Godzich, M.; Żbikowski, J. Macrozoobenthos structure in a temperate acid oligotrophic lake. Ecol. Quest. 2017, 27, 97–107. [Google Scholar] [CrossRef] [Green Version]
- Obolewski, K.; Glińska-Lewczuk, K.; Ożgo, M.; Astel, A. Connectivity restoration of floodplain lakes: An assessment based on macroinvertebrate communities. Hydrobiologia 2016, 774, 23–37. [Google Scholar] [CrossRef] [Green Version]
- Jayanti, A.D.; Fachrul, M.F.; Hendrawan, D. Makrozoobentos as Bioindicator Water Quality of Krukut River, Depok, West Java, Indonesia. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2018; Volume 106, p. 012025. [Google Scholar] [CrossRef]
| No. | River–Lake System | Special Features of System | Nutrient Retention (% Per Year) | Literature | |
|---|---|---|---|---|---|
| TN | TP | ||||
| 1. | Marózka River–Lake Mielno | Catchment: arable land, agricultural | 40.5 | 7.7 | [54] |
| 2. | Marózka River–Lake Maróz | Catchment: arable land, agricultural | 11.1 | 9.1 | [54] |
| 3. | Łyna River–Lake Łańskie | Catchment: forests | 23.8 | 17.4 | [54] |
| 4. | Kortówka River–Lake Kortowskie | Arable land, point and non-point sources. Lake area: 89.7 ha with mean depth: 5.9 m | 56.0 | 26.0 | [55] |
| 5. | Havel River–Havel Lakes | Point and non-point sources. Polytrophic, large interconnected shallow lakes with mean depth: 3.5 m | 30.0 | Increase (internal loading) | [56] |
| 6. | Krzemionka River–Lake Wierzchołek | Shallow of lake with area: 17.3 ha and mean depth: 1.8 m, catchment area: 26.5 ha | Increase (internal loading) | 42.0 | [57] |
| 7. | Symsarna River–Lake Symsar | Agro-forestry catchment with area: 129.1 km2.Last lake with area: 1.29 km2 and mean depth: 4.9 m. | 8.8 | 21.6 | [38] |
| 8. | River Gudenaa–Danish system | 149 km, Natura 2000 | 46.0 | 25.0 | [21] |
| No. | River–Lake System | Special Features of System | Metal Content in Sediments (mg kg−1) | Literature | |||
|---|---|---|---|---|---|---|---|
| Ni | Cu | Zn | Pb | ||||
| 1. | Powa River- Stare Miasto Reservoir | Catchment covered of arable land with area: 1.46 km2 | 2.7 | 1.7 | 10.9 | 3.8 | [25] |
| 2. | Lake Jeżewo | Agricultural catchment. Lake area: 73 ha with mean depth: 2.9 m and 72 days of water retention | 5.9 | 10.1 | 903.7 | 17.6 | [41] |
| 3. | Lake Środa | Agricultural catchment. Lake area: 39 ha with mean depth: 2.3 m and 23 days of water retention | 3.5 | 4.6 | 357.5 | 7.4 | |
| 4. | Lake Września | Agricultural catchment. Lake area: 39 ha with mean depth: 0.9 m and 4 days of water retention | 5.5 | 9.4 | 678.4 | 15.2 | |
| 5. | Symsarna River- Lake Symsar | Agro-forestry catchment with area: 129.1 km2.Last lake in system with area: 1.29 km2 and mean depth: 4.9 m. | 32.6 | 19.2 | 120.0 | 84.0 | [66] |
| 6. | Masurian Lakeland | Small surface area of lake below 100 ha with mean depth: 5.1-17.4 m. | 9.1 | 12.8 | 75.5 | 15.6 | [67] |
| 7. | Suwalki Lakeland | 13.7 | 16.9 | 124.7 | 28.9 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuriata-Potasznik, A.; Szymczyk, S.; Skwierawski, A. Influence of Cascading River–Lake Systems on the Dynamics of Nutrient Circulation in Catchment Areas. Water 2020, 12, 1144. https://doi.org/10.3390/w12041144
Kuriata-Potasznik A, Szymczyk S, Skwierawski A. Influence of Cascading River–Lake Systems on the Dynamics of Nutrient Circulation in Catchment Areas. Water. 2020; 12(4):1144. https://doi.org/10.3390/w12041144
Chicago/Turabian StyleKuriata-Potasznik, Angela, Sławomir Szymczyk, and Andrzej Skwierawski. 2020. "Influence of Cascading River–Lake Systems on the Dynamics of Nutrient Circulation in Catchment Areas" Water 12, no. 4: 1144. https://doi.org/10.3390/w12041144
APA StyleKuriata-Potasznik, A., Szymczyk, S., & Skwierawski, A. (2020). Influence of Cascading River–Lake Systems on the Dynamics of Nutrient Circulation in Catchment Areas. Water, 12(4), 1144. https://doi.org/10.3390/w12041144





