Assessing the Cadmium Effects on the Benthic Foraminifer Ammonia cf. parkinsoniana: An Acute Toxicity Test
Abstract
1. Introduction
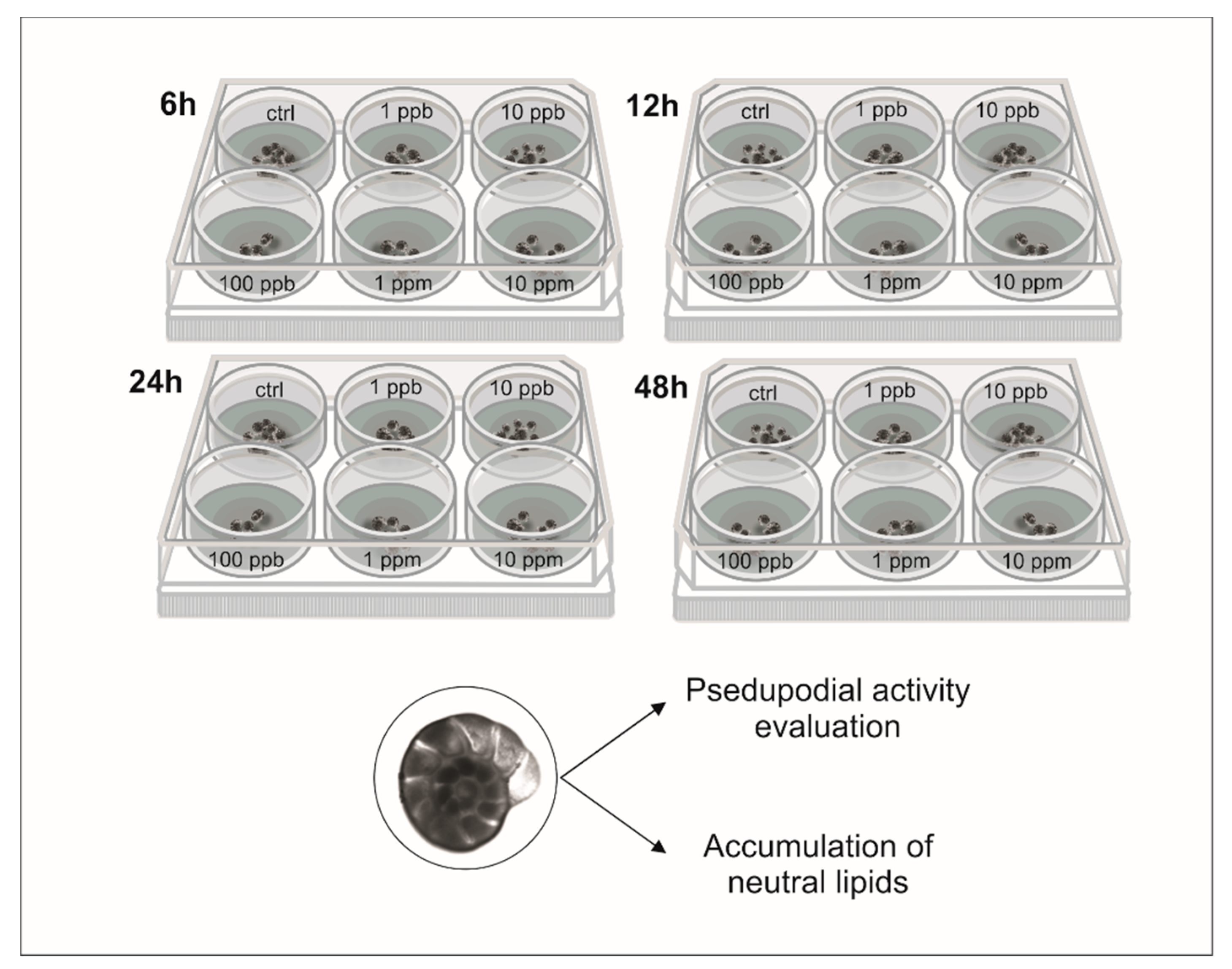
2. Materials and Methods
3. Results
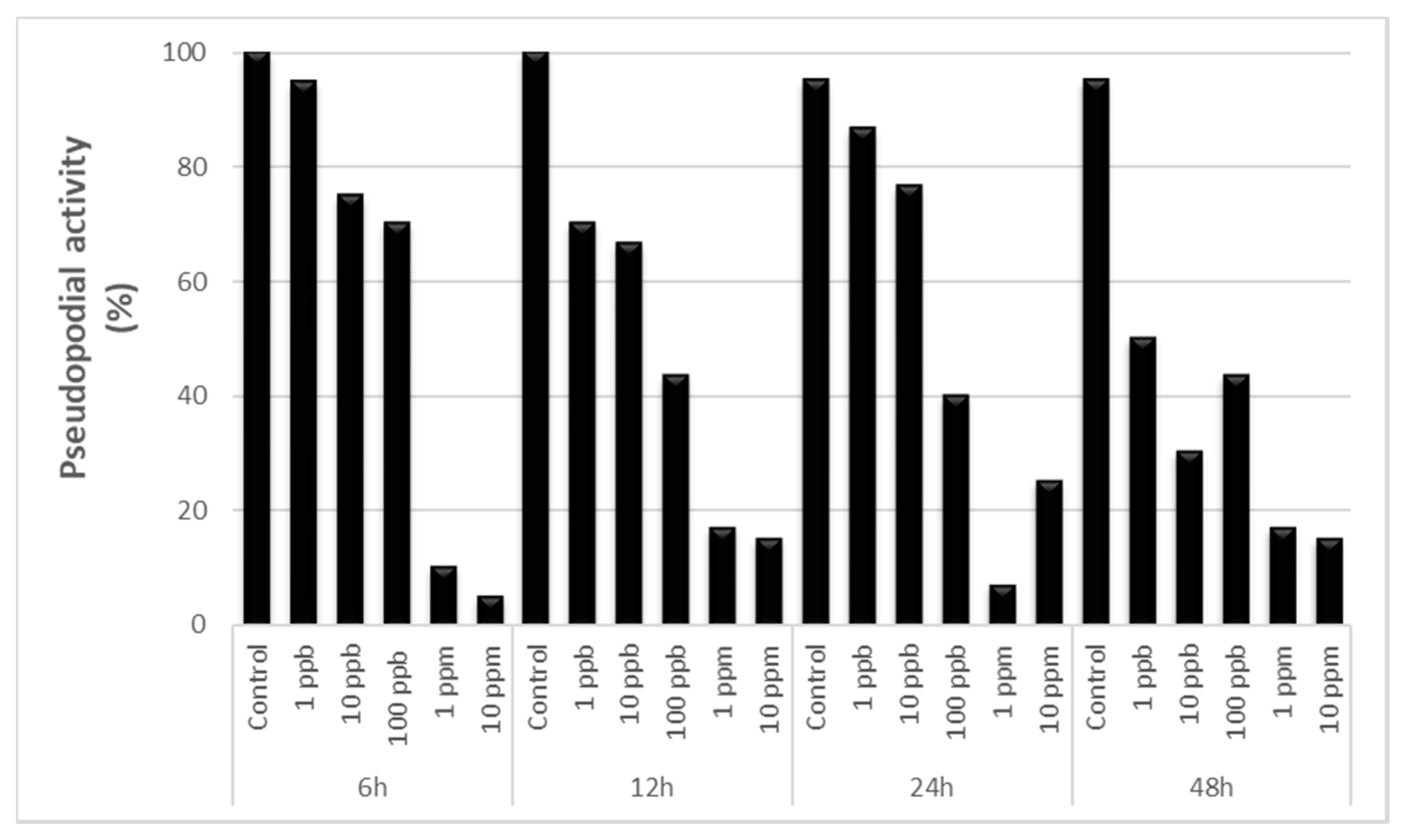
3.1. Pseudopodial Activity
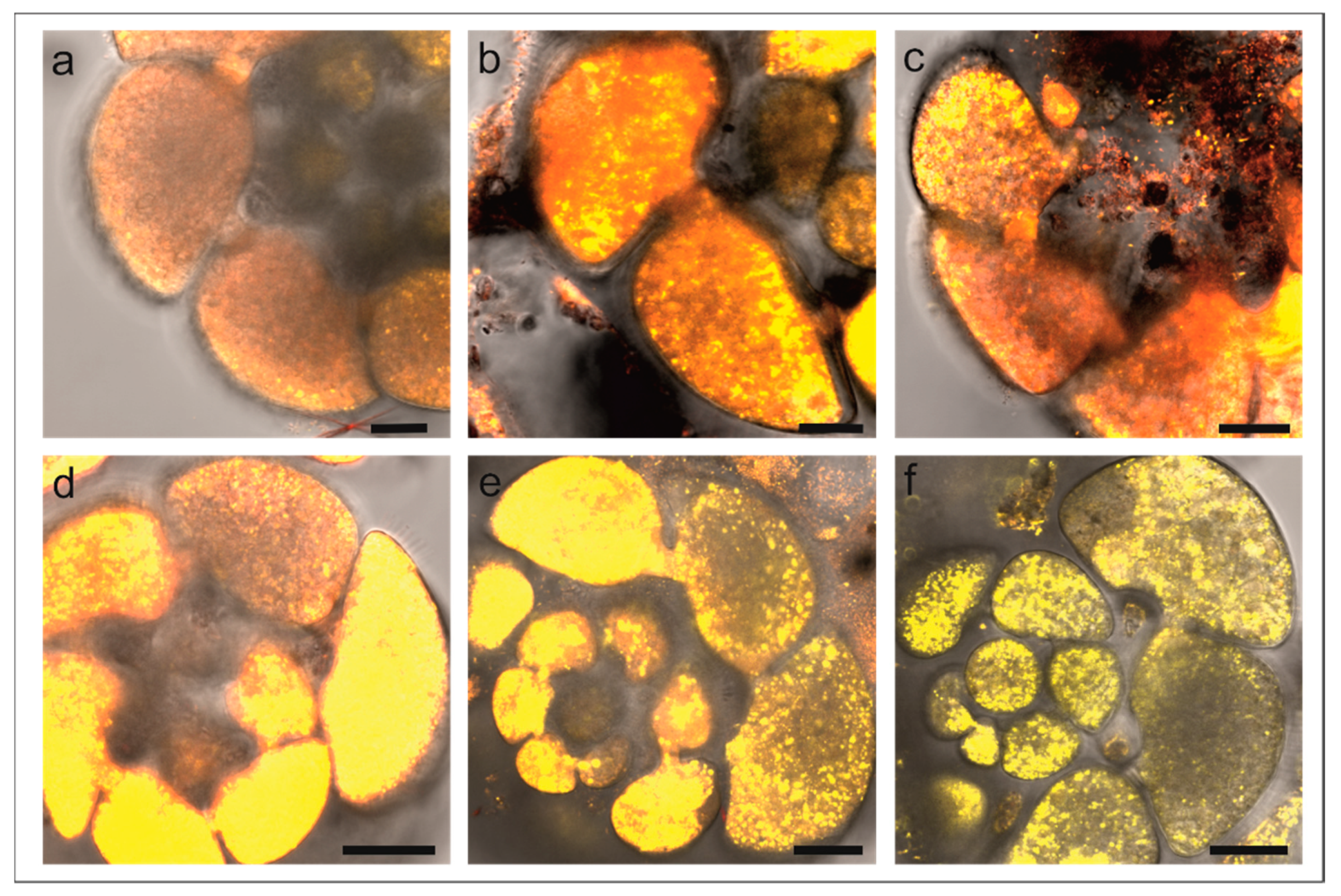
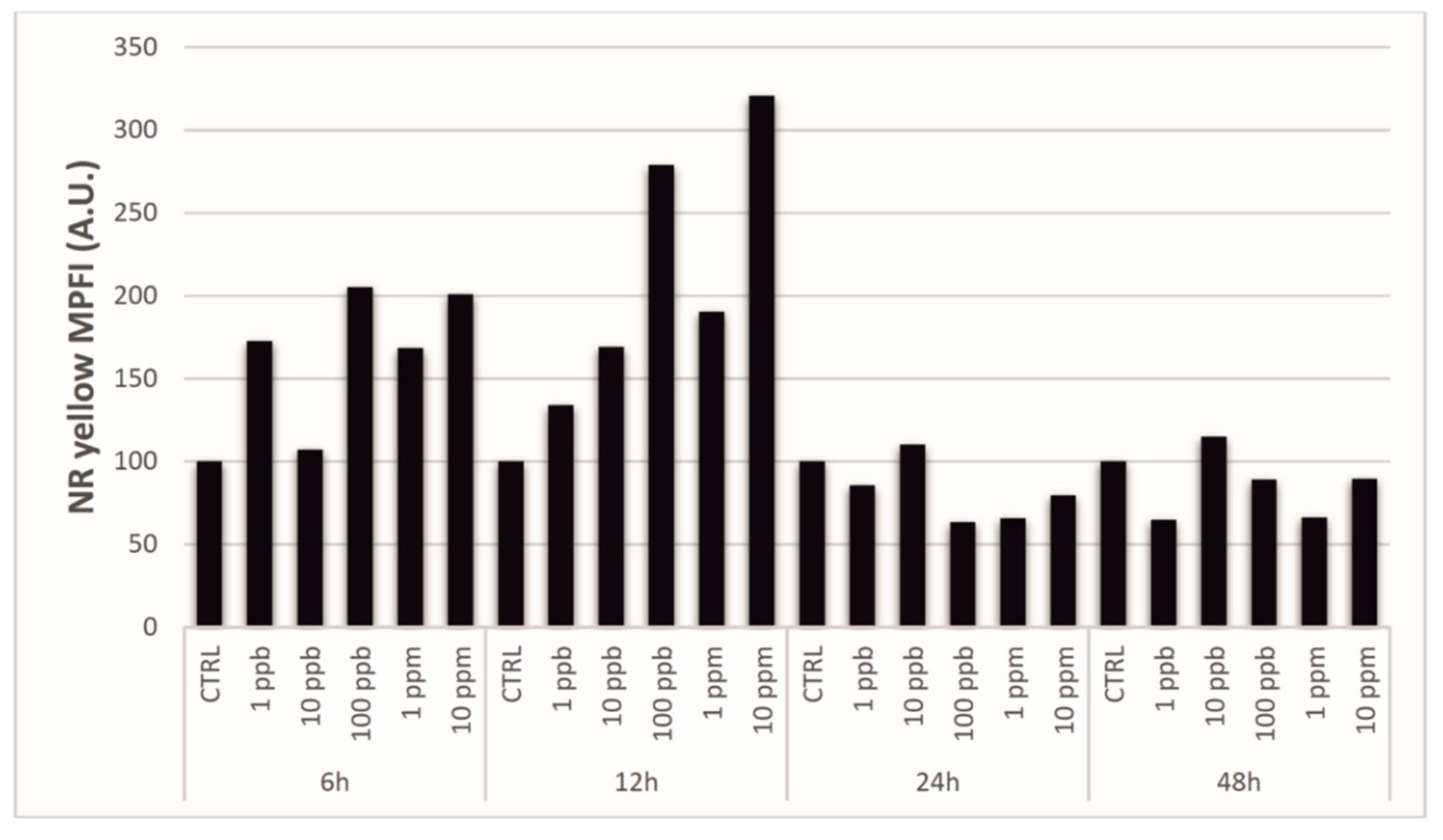
3.2. Laser Confocal Scan Microscopy
4. Discussion
4.1. Effects of Cd on Benthic Foraminifera
4.2. Heavy Metals Effects on Benthic Foraminifera
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Borja, A.; Elliott, M.; Andersen, J.H.; Cardoso, A.C.; Carstensen, J.; Ferreira, J.; Heiskanen, A.-S.; Marques, J.C.; Neto, J.; Teixeira, H.; et al. Good Environmental Status of marine ecosystems: What is it and how do we know when we have attained it? Mar. Pollut. Bull. 2013, 76, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Foster, W.J.; Armynot Du Châtelet, E.; Rogerson, M. Testing benthic foraminiferal distributions as a contemporary quantitative approach to biomonitoring estuarine heavy metal pollution. Mar. Pollut. Bull. 2012, 64, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Gallego, A.; Martín-González, A.; Ortega, R.; Gutierrez, J.C. Flow cytometry assessment of cytotoxicity and reactive oxygen species generation by single and binary mixtures of cadmium, zinc and copper on populations of the ciliated protozoan Tetrahymena thermophila. Chemosphere 2007, 68, 647–661. [Google Scholar] [CrossRef] [PubMed]
- Linshy, V.N.; Saraswat, R.; Kurtarkar, S.R.; Nigam, R. Experiment to decipher the effect of heavy metal cadmium on coastal benthic foraminifera Pararotalia nipponica (Asano). J. Paleontolog. Soc. Ind. 2013, 58, 205–211. [Google Scholar]
- Li, T.; Xiang, R.; Li, T. Influence of trace metals in recent benthic foraminifera distribution in the Pearl River Estuary. Mar. Micropaleontol. 2014, 108, 13–27. [Google Scholar] [CrossRef]
- Stankovic, S.; Stankovic, A.R. Bioindicators of toxic metals. In Green Materials for Energy, Products and Depollution; Lichtfouse, E., Schwarzbauer, J., Robert, D., Eds.; Springer Science+Business Media: Dordrecht, The Netherlands, 2013; pp. 151–217. [Google Scholar]
- Lesser, M.P. Oxidative stress in marine environments: Biochemistry and physiological ecology. Annu. Rev. Physiol. 2006, 68, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Eisler, R. Cadmium hazards to fish, wildlife, and invertebrates: A synoptic review. US Fish Wildl. Serv. Biol. Rep. 1985, 85, 46. [Google Scholar]
- Eisler, R.; Zaroogian, G.E.; Hennekey, R.J. Cadmium Uptake by Marine Organisms. J. Fish. Res. Board Can. 1972, 29, 1367–1369. [Google Scholar] [CrossRef]
- Šmirjákova, S.; Ondrašovičová, O.; Kašková, A.; Laktičová, K. The effect of cadmium and lead pollution on human and animal health. Folia Vet. 2005, 49, 31–32. [Google Scholar]
- Ognjanović, B.I.; Marković, S.D.; Pavlović, S.Z.; Žikić, R.V.; Štajn, A.Š.; Saicić, Z.S. Effect of chronic cadmium exposure on antioxidant defense system in some tissues of rats: Protective effect of selenium. Physiol. Res. 2007, 57, 403–411. [Google Scholar]
- Nordberg, G.F. Health hazards of environmental cadmium pollution. Ambio 1974, 3, 55–66. [Google Scholar]
- Satarug, S.; Baker, J.R.; Urbenjapol, S.; Hasweel-Elkins, M.; Reilly, P.E.B.; Williams, D.J.; Moore, M.R. A global perspective on cadmium pollution and toxicity in non-ocupationally exposed population. Toxicol. Lett. 2003, 137, 65–83. [Google Scholar] [CrossRef]
- Bertin, G.; Averbeck, D. Cadmium: Cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie 2006, 88, 1549–1559. [Google Scholar] [CrossRef] [PubMed]
- Olabarrieta, I.; L’Azou, B.; Yuric, S.; Cambar, J.; Cajaraville, M.P. In vitro effects of cadmium on two different animal cell models. Toxicol. Vitr. 2001, 15, 511–517. [Google Scholar] [CrossRef]
- Waisberg, M.; Joseph, P.; Hale, B.; Beyersmann, D. Molecular and cellular mechanisms of cadmium carcinogenesis. Toxicology 2003, 192, 95–117. [Google Scholar] [CrossRef]
- Stanković, S.; Kalaba, P.; Stankovic, A.R. Biota as toxic metal indicators. Environ. Chem. Lett. 2013, 12, 63–84. [Google Scholar] [CrossRef]
- Frontalini, F.; Coccioni, R. Benthic foraminifera as bioindicators of pollution: A review of Italian research over the last three decades. Revue de Micropaléontologie 2011, 54, 115–127. [Google Scholar] [CrossRef]
- Frontalini, F.; Greco, M.; Di Bella, L.; Lejzerowicz, F.; Reo, E.; Caruso, A.; Cosentino, C.; Maccotta, A.; Scopelliti, G.; Nardelli, M.P.; et al. Assessing the effect of mercury pollution on cultured benthic foraminifera community using morphological and eDNA metabarcoding approaches. Mar. Pollut. Bull. 2018, 129, 512–524. [Google Scholar] [CrossRef]
- Frontalini, F.; Nardelli, M.P.; Curzi, D.; Martín-González, A.; Sabbatini, A.; Negri, A.; Losada, M.; Gobbi, P.; Coccioni, R.; Bernhard, J. Benthic foraminiferal ultrastructural alteration induced by heavy metals. Mar. Micropaleontol. 2018, 138, 83–89. [Google Scholar] [CrossRef]
- Saraswat, R.; Kurtarkar, S.R.; Mazumder, A.; Nigam, R. Foraminifers as indicators of marine pollution: A culture experiment with Rosalina leei. Mar. Pollut. Bull. 2004, 48, 91–96. [Google Scholar] [CrossRef]
- Le Cadre, V.; Debenay, J.-P. Morphological and cytological responses of Ammonia (foraminifera) to copper contamination: Implication for the use of foraminifera as bioindicators of pollution. Environ. Pollut. 2006, 143, 304–317. [Google Scholar] [CrossRef] [PubMed]
- Nigam, R.; Linshy, V.N.; Kurtarkar, S.; Saraswat, R. Effects of sudden stress due to heavy metal mercury on benthic foraminifer Rosalina leei: Laboratory culture experiment. Mar. Pollut. Bull. 2009, 59, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Denoyelle, M.; Geslin, E.; Jorissen, F.; Cazes, L.; Galgani, F. Innovative use of foraminifera in ecotoxicology: A marine chronic bioassay for testing potential toxicity of drilling muds. Ecol. Indic. 2012, 12, 17–25. [Google Scholar] [CrossRef]
- Frontalini, F.; Curzi, D.; Giordano, F.; Bernhard, J.; Falcieri, E.; Coccioni, R. Effects of Lead Pollution on Ammonia parkinsoniana (Foraminifera): Ultrastructural and Microanalytical Approaches. Eur. J. Histochem. 2015, 59, 2460. [Google Scholar] [CrossRef] [PubMed]
- Frontalini, F.; Curzi, D.; Cesarini, E.; Canonico, B.; Giordano, F.M.; De Matteis, R.; Bernhard, J.M.; Pieretti, N.; Gu, B.; Eskelsen, J.; et al. Mercury-Pollution Induction of Intracellular Lipid Accumulation and Lysosomal Compartment Amplification in the Benthic Foraminifer Ammonia parkinsoniana. PLoS ONE 2016, 11, e0162401. [Google Scholar] [CrossRef] [PubMed]
- Frontalini, F.; Losada, M.T.; Toyofuku, T.; Tyszka, J.; Goleń, J.; De Nooijer, L.; Canonico, B.; Cesarini, E.; Nagai, Y.; Bickmeyer, U.; et al. Foraminiferal Ultrastructure: A perspective From Fluorescent and Fluorogenic Probes. J. Geophys. Res. Biogeosciences 2019, 124, 2823–2850. [Google Scholar] [CrossRef]
- Ciacci, C.; Grimmelpont, M.V.; Corsi, I.; Bergami, E.; Curzi, D.; Burini, D.; Bouchet, V.M.P.; Ambrogini, P.; Gobbi, P.; Ujiié, Y.; et al. Nanoparticle-Biological Interactions in a Marine Benthic Foraminifer. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Al-Zamel, A.Z.; Al-Sarawi, M.A.; Khader, S.R.; Al-Rifaiy, I.A. Benthic foraminifera from polluted marine environment of Sulaibikhat Bay (Kuwait). Environ. Monit. Assess. 2008, 149, 395–409. [Google Scholar] [CrossRef]
- Cherif, O.H.; Al-Ghadban, A.-N.; Al-Rifaiy, I.A. Distribution of Foraminifera in the Arabian Gulf. Micropaleontol. 1997, 43, 253. [Google Scholar] [CrossRef]
- Greenspan, P.; Fowler, S.D. Spectrofluorometric studies of the lipid probe, nile red. J. Lipid Res. 1985, 26, 781–789. [Google Scholar]
- Sackett, D.L.; Wolff, J. Nile red as a polarity-sensitive fluorescent probe of hydrophobic protein surfaces. Anal. Biochem. 1987, 167, 228–234. [Google Scholar] [CrossRef]
- Greenspan, P.; Mayer, E.P.; Fowler, S.D. Nile red: A selective fluorescent stain for intracellular lipid droplets. J. Cell Boil. 1985, 100, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Diaz, G.; Melis, M.; Batetta, B.; Angius, F.; Falchi, A.M. Hydrophobic characterization of intracellular lipids in situ by Nile Red red/yellow emission ratio. Micron 2008, 39, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Bresler, V.; Yanko, v. Acute toxicity of heavy metals for benthic epiphytic foraminifera Pararotalia spinigera (Le Calvez) and influence of seaweed-derived doc. Environ. Toxicol. Chem. 1995, 14, 1687. [Google Scholar] [CrossRef]
- European Chemicals Agency. Available online: https://echa.europa.eu/registration-dossier/-/registered-dossier/14430/6/1 (accessed on 25 March 2020).
- Acquatic Life Ambient Water Quality Criteria Cadmium; EPA-820-R-16-002; United States Environmental Protection Agency, 2016. Available online: https://www.epa.gov/sites/production/files/2016-03/documents/cadmium-final-report-2016.pdf (accessed on 15 March 2020).
- Prazeres, M.D.F.; Martins, S.E.; Bianchini, A. Biomarkers response to zinc exposure in the symbiont-bearing foraminifer Amphistegina lessonii (Amphisteginidae, Foraminifera). J. Exp. Mar. Boil. Ecol. 2011, 407, 116–121. [Google Scholar] [CrossRef]
- Nardelli, M.P.; Sabbatini, A.; Negri, A. Experimental chronic exposure of the foraminifer Pseudotriloculina rotunda to zinc. Acta Protozool. 2013, 52, 193–202. [Google Scholar]
- Díaz, S.; Martín-González, A.; Gutiérrez, J.C. Evaluation of heavy metal acute toxicity and bioaccumulation in soil ciliated protozoa. Environ. Int. 2006, 32, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Madoni, P.; Davoli, D.; Gorbi, G.; Vescovi, L. Toxic effect of heavy metals on the activated sludge protozoan community. Water Res. 1996, 30, 135–141. [Google Scholar] [CrossRef]
- Martín-González, A.; Díaz, S.; Borniquel, S.; Gallego, A.; Gutiérrez, J.C. Cytotoxicity and bioaccumulation of heavy metals by ciliated protozoa isolated from urban wastewater treatment plants. Res. Microbiol. 2006, 157, 108–118. [Google Scholar] [CrossRef]
- Madoni, P.; Romeo, M.G. Acute toxicity of heavy metals towards freshwater ciliated protists. Environ. Pollut. 2006, 141, 1–7. [Google Scholar] [CrossRef]
- Nalecz-Jawecki, G.; Sawicki, J. Toxicity of inorganic compound in Spirotox test—A miniaturised version of the Spirostomum ambiguum test. Arch. Environ. Cont. Tox. 1998, 34, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Morvan, J.; Le Cadre, V.; Jorissen, F.; Debenay, J.-P. Foraminifera as potential bio-indicators of the “Erika” oil spill in the Bay of Bourgneuf: Field and experimental studies. Aquat. Living Resour. 2004, 17, 317–322. [Google Scholar] [CrossRef]
- Frontalini, F.; Semprucci, F.; Di Bella, L.; Caruso, A.; Cosentino, C.; Maccotta, A.; Scopelliti, G.; Sbrocca, C.; Bucci, C.; Balsamo, M.; et al. The response of cultured meiofaunal and benthic foraminiferal communities to lead contamination: Results from mesocosm experiments. Environ. Toxicol. Chem. 2018, 37, 2439–2447. [Google Scholar] [CrossRef] [PubMed]
- Artal, M.; Holtz, R.D.; Kummrow, F.; Alves, O.L.; Umbuzeiro, G. The role of silver and vanadium release in the toxicity of silver vanadate nanowires toward Daphnia similis. Environ. Toxicol. Chem. 2013, 32, 908–912. [Google Scholar] [CrossRef]
- Murphy, G.; Rouse, R.L.; Polk, W.W.; Henk, W.G.; Barker, S.A.; Boudreaux, M.J.; Floyd, E.; Penn, A.L. Combustion-Derived Hydrocarbons Localize to Lipid Droplets in Respiratory Cells. Am. J. Respir. Cell Mol. Boil. 2008, 38, 532–540. [Google Scholar] [CrossRef]
- Rowan-Carroll, A.; Halappanavar, S.; Williams, A.; Somers, C.M.; Yauk, C. Mice exposed in situ to urban air pollution exhibit pulmonary alterations in gene expression in the lipid droplet synthesis pathways. Environ. Mol. Mutagen. 2013, 54, 240–249. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losada Ros, M.T.; Al-Enezi, E.; Cesarini, E.; Canonico, B.; Bucci, C.; Alves Martins, M.V.; Papa, S.; Frontalini, F. Assessing the Cadmium Effects on the Benthic Foraminifer Ammonia cf. parkinsoniana: An Acute Toxicity Test. Water 2020, 12, 1018. https://doi.org/10.3390/w12041018
Losada Ros MT, Al-Enezi E, Cesarini E, Canonico B, Bucci C, Alves Martins MV, Papa S, Frontalini F. Assessing the Cadmium Effects on the Benthic Foraminifer Ammonia cf. parkinsoniana: An Acute Toxicity Test. Water. 2020; 12(4):1018. https://doi.org/10.3390/w12041018
Chicago/Turabian StyleLosada Ros, María Teresa, Eqbal Al-Enezi, Erica Cesarini, Barbara Canonico, Carla Bucci, Maria Virgínia Alves Martins, Stefano Papa, and Fabrizio Frontalini. 2020. "Assessing the Cadmium Effects on the Benthic Foraminifer Ammonia cf. parkinsoniana: An Acute Toxicity Test" Water 12, no. 4: 1018. https://doi.org/10.3390/w12041018
APA StyleLosada Ros, M. T., Al-Enezi, E., Cesarini, E., Canonico, B., Bucci, C., Alves Martins, M. V., Papa, S., & Frontalini, F. (2020). Assessing the Cadmium Effects on the Benthic Foraminifer Ammonia cf. parkinsoniana: An Acute Toxicity Test. Water, 12(4), 1018. https://doi.org/10.3390/w12041018






