Partition and Fate of Phthalate Acid Esters (PAEs) in a Full-Scale Horizontal Subsurface Flow Constructed Wetland Treating Polluted River Water
Abstract
1. Introduction
2. Materials and Methods
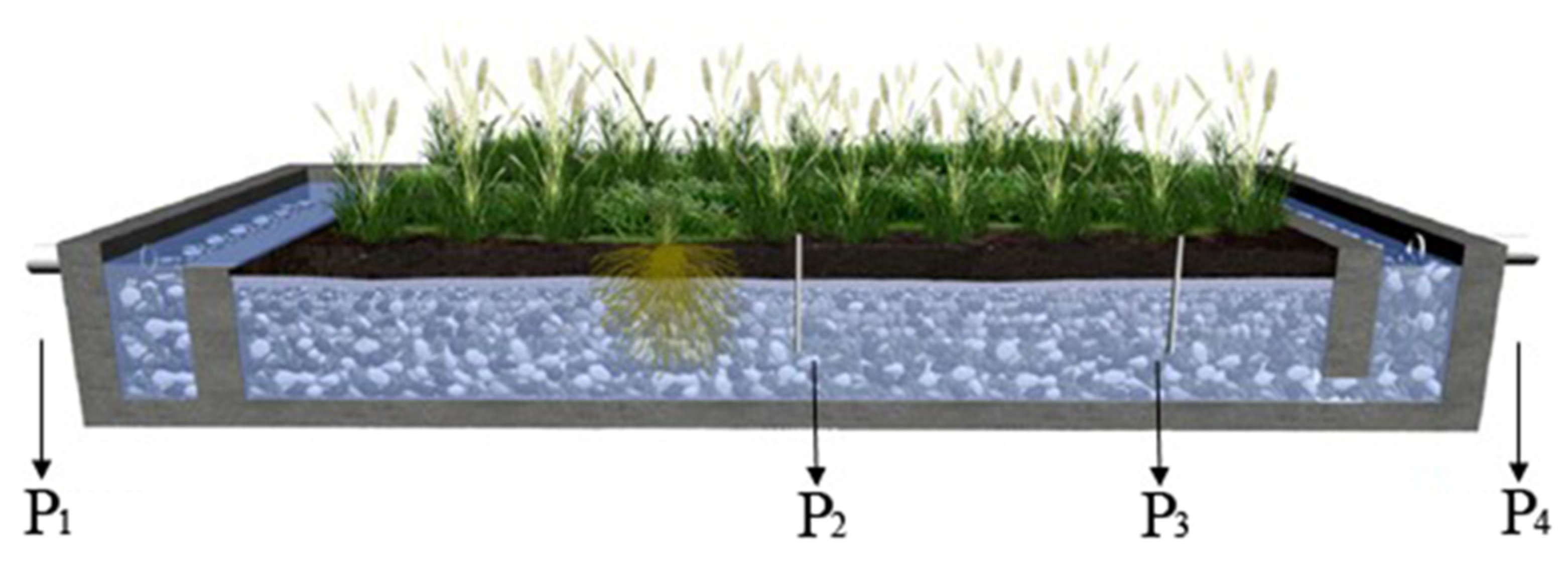
2.1. Field Site
2.2. Sampling
2.3. Sample Pretreatment
2.4. PAEs Analysis
2.5. Data Analysis
3. Results and Discussion
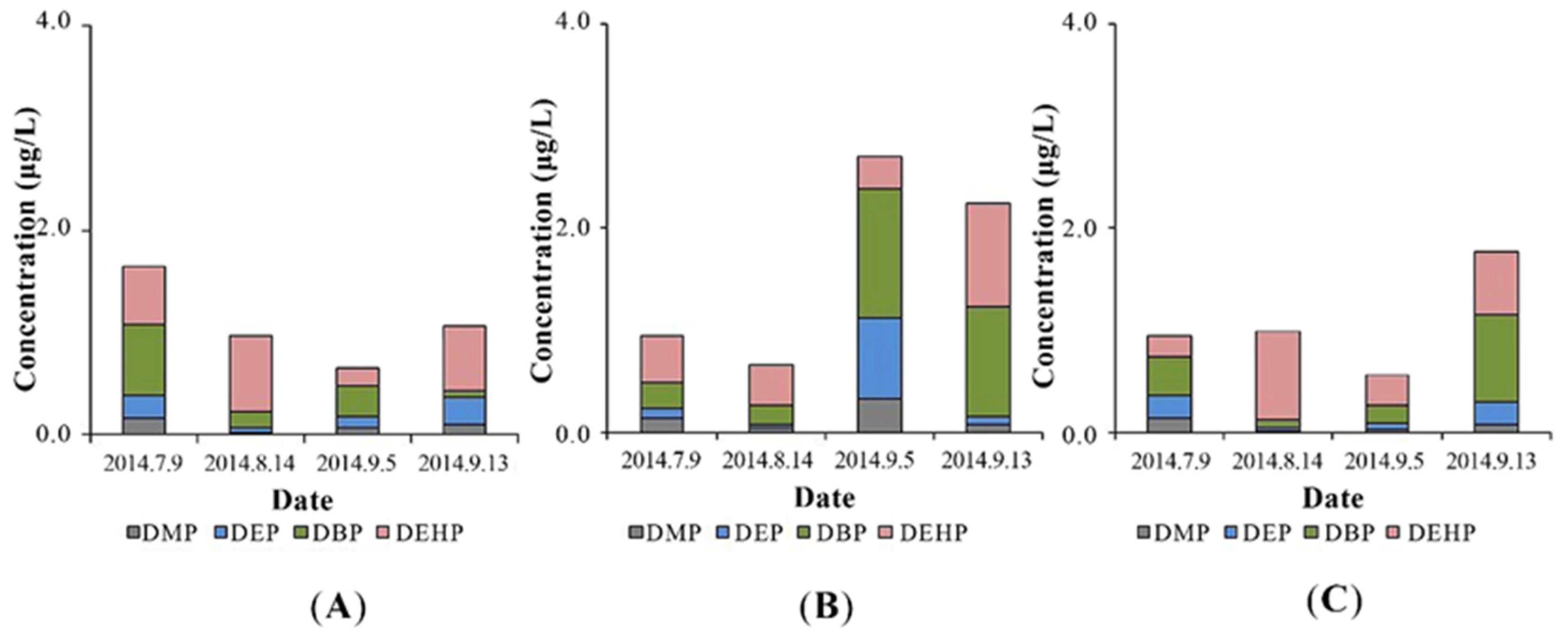
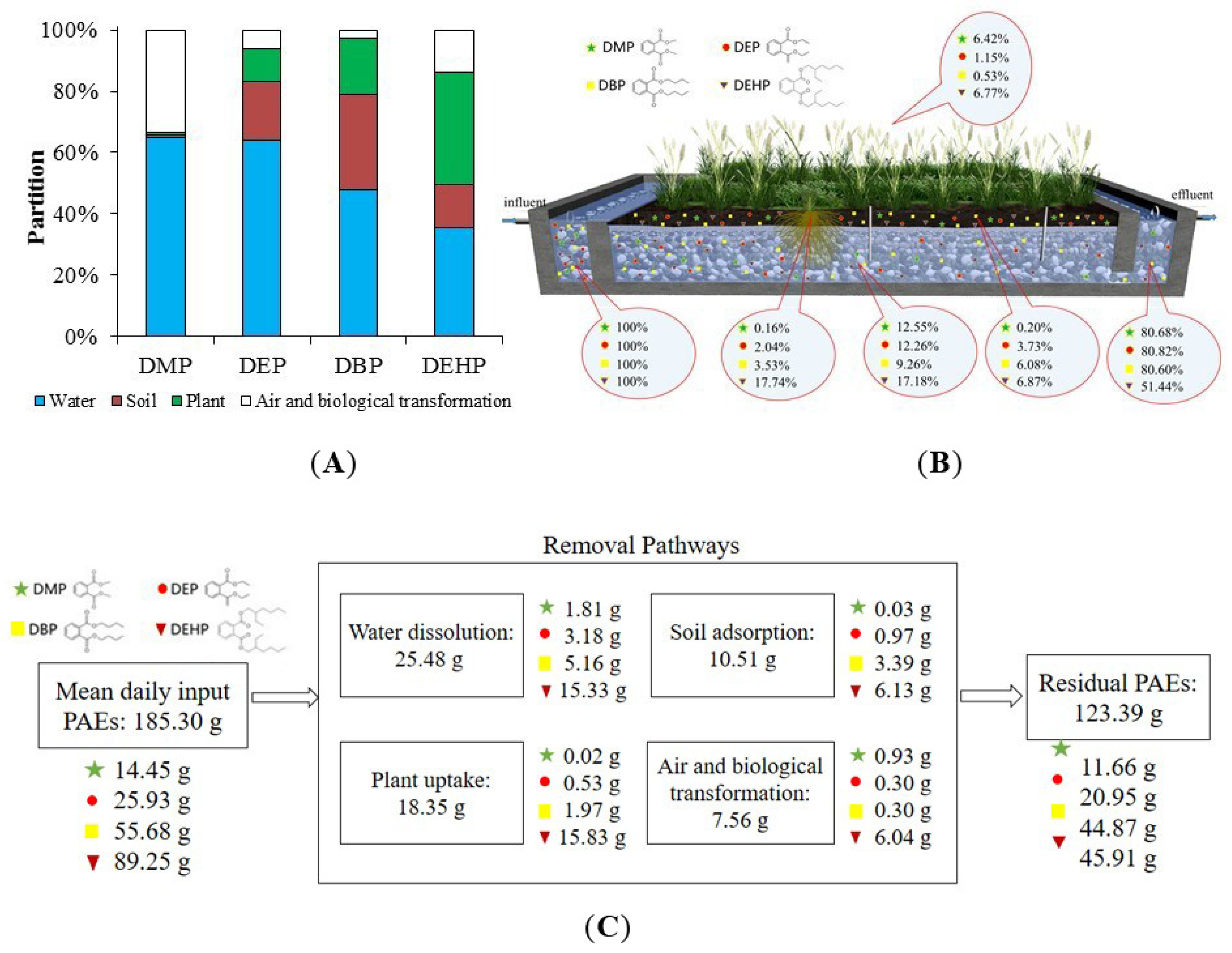
3.1. PAEs Removal in Constructed Wetland
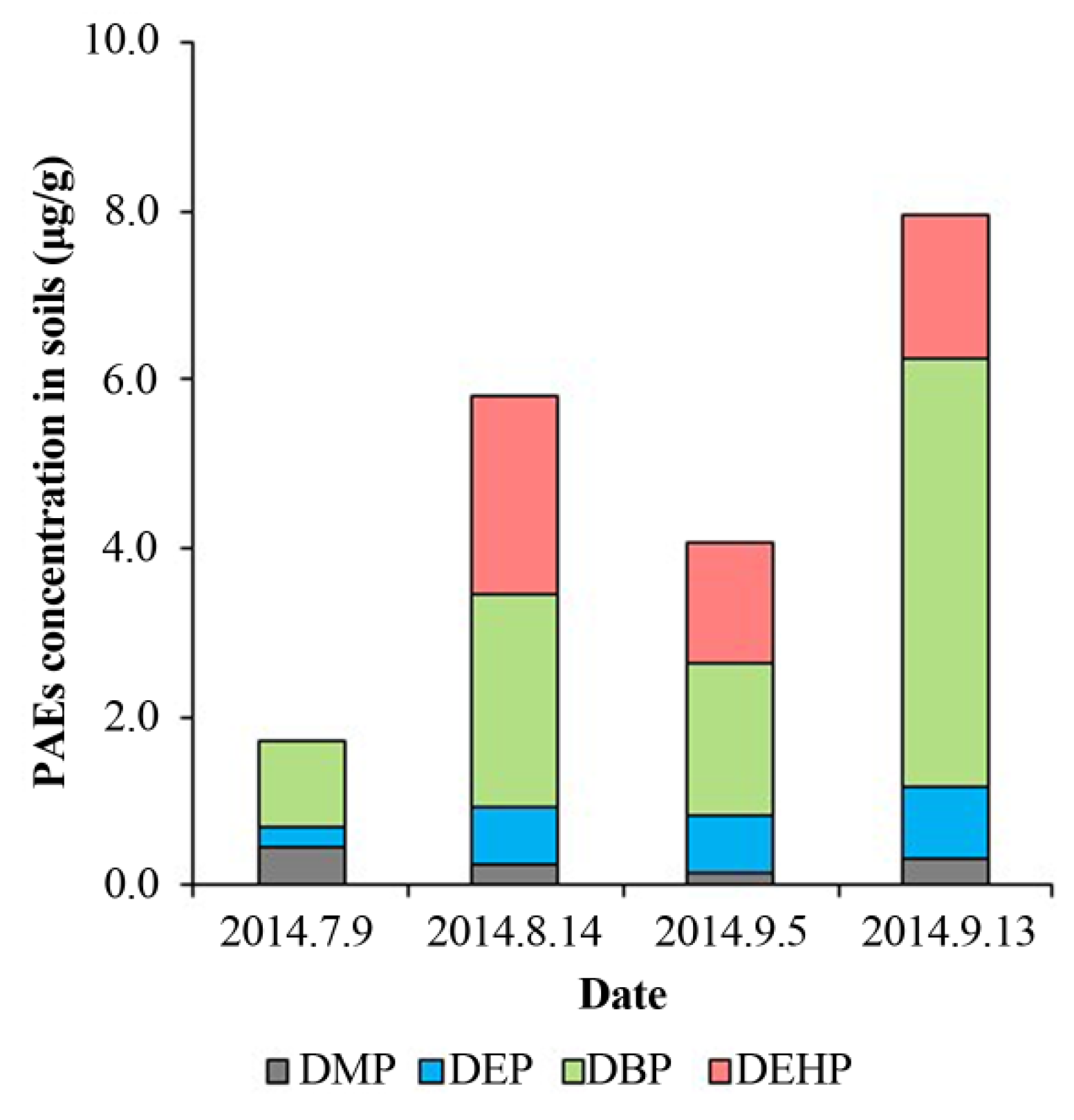
3.2. PAEs Absorption in Soils
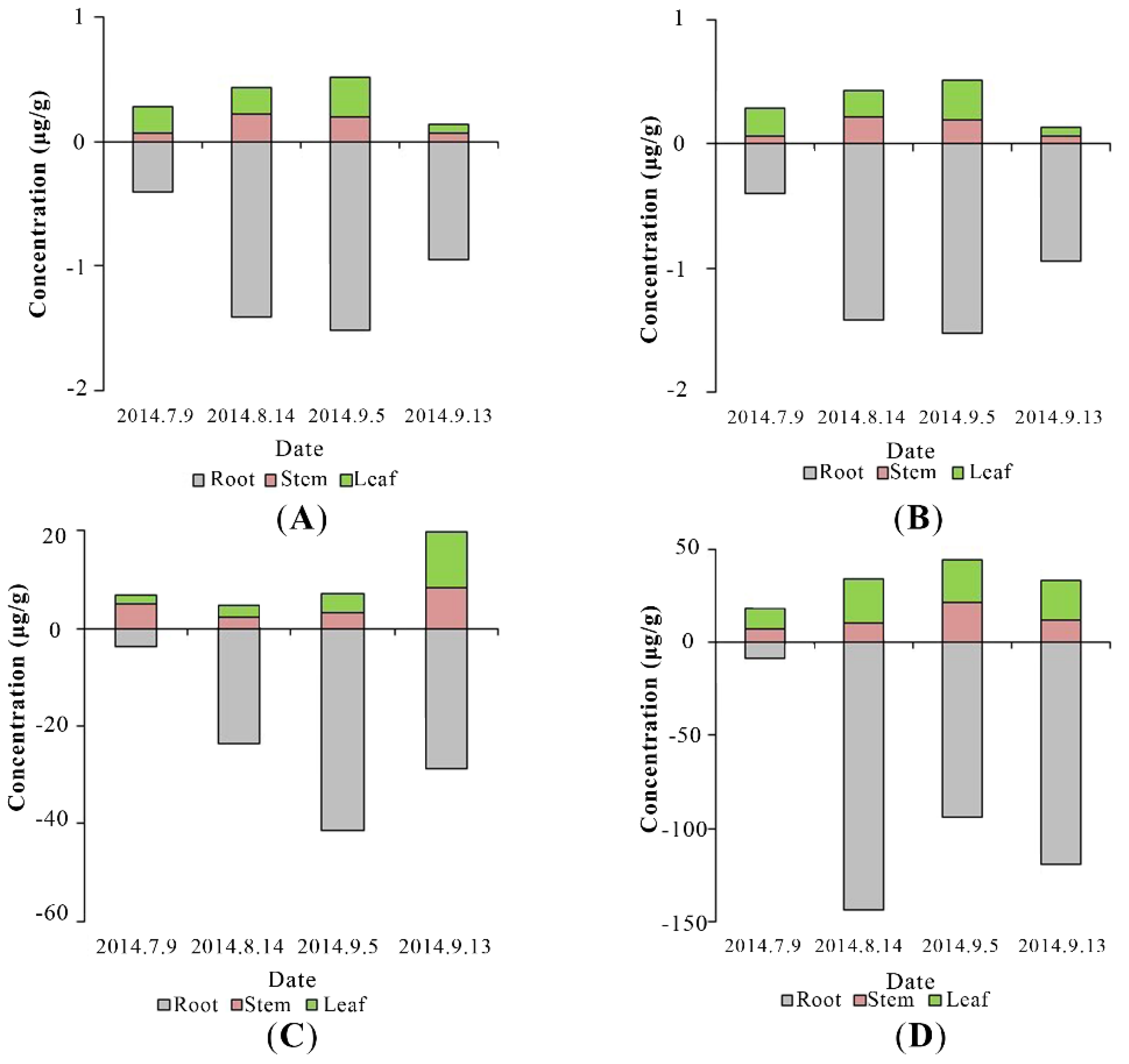
3.3. PAEs Uptake by Plants
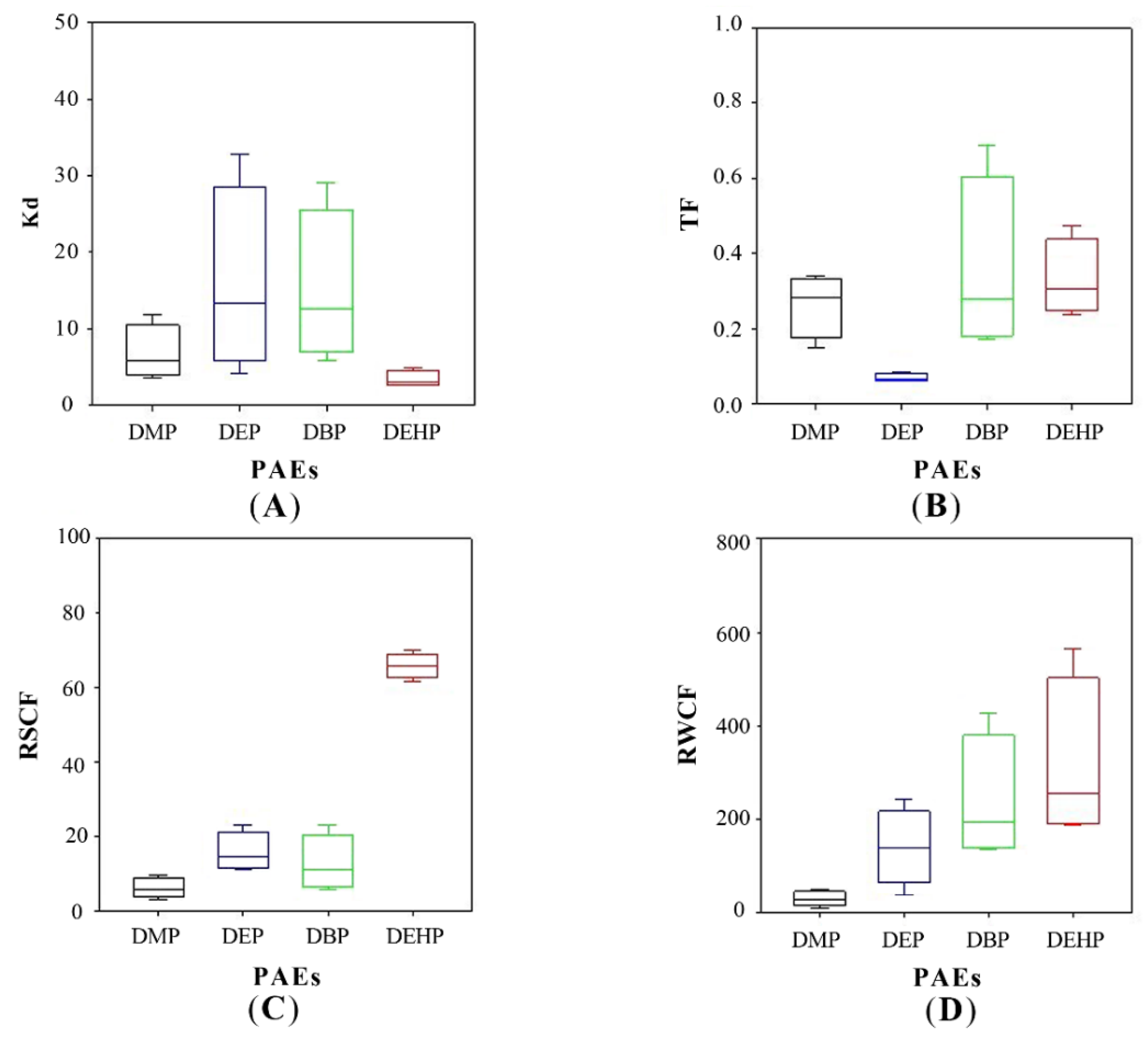
3.4. PAEs Distribution and Fate
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gao, D.; Li, Z.; Wen, Z.; Ren, N. Occurrence and fate of phthalate esters in full-scale domestic wastewater treatment plants and their impact on receiving waters along the Songhua River in China. Chemosphere 2014, 95, 24–32. [Google Scholar] [CrossRef]
- Koniecki, D.; Wang, R.; Moody, R.P.; Zhu, J. Phthalates in cosmetic and personal care products: Concentrations and possible dermal exposure. Environ. Res. 2011, 111, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Ueda, K.; Kojima, N. Potential risks of phthalate esters: Acquisition of endocrine-disrupting activity during environmental and metabolic processing. J. Health Sci. 2011, 57, 497–503. [Google Scholar] [CrossRef]
- Cho, E.; Khim, J.; Chung, S.; Seo, D.; Son, Y. Occurrence of micropollutants in four major rivers in Korea. Sci. Total Environ. 2014, 491–492, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Cui, K.; Zeng, F.; Chen, L.; Cheng, Y.; Li, H.; Li, S.; Zhou, X.; Zhu, F.; Ouyang, G.; et al. Occurrence and distribution of phthalate esters in riverine sediments from the Pearl River Delta region, South China. Mar. Pollut. Bull. 2014, 83, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wu, X.; Gan, J. Uptake and metabolism of phthalate esters by edible plants. Environ. Sci. Technol. 2015, 49, 8471–8478. [Google Scholar] [CrossRef] [PubMed]
- Bergé, A.; Gasperi, J.; Rocher, V.; Gras, L.; Coursimault, A.; Moilleron, R. Phthalates and alkylphenols in industrial and domestic effluents: Case of Paris conurbation (France). Sci. Total Environ. 2014, 488–489, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Man, Y.; Cheung, K.; Wong, M. Risk assessment of human exposure to bioaccessible phthalate esters via indoor dust around the Pearl River Delta. Environ. Sci. Technol. 2012, 46, 8422–8430. [Google Scholar] [CrossRef]
- Joel, D.B. Ultra-trace determination of phthalate ester metabolites in seawater, sediments, and biota from an urbanized marine inlet by LC/ESI-MS/MS. Environ. Sci. Technol. 2009, 16, 6262–6268. [Google Scholar]
- Hanno, C.E. Leaching of the plasticizer di(2-ethylhexyl)phthalate (DEHP) from plastic containers and the question of human exposure. Appl. Microbiol. Biot. 2014, 24, 9967–9981. [Google Scholar]
- He, W.; Qin, N.; Kong, X.; Liu, W.; He, Q.; Ouyang, H.; Yang, C.; Jiang, Y.; Wang, Q.; Yang, B.; et al. Spatio-temporal distributions and the ecological and health risks of phthalate esters (PAEs) in the surface water of a large, shallow Chinese lake. Sci. Total Environ. 2013, 461–462, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Ventrice, P.; Ventrice, D.; Russo, E.; Sarro, G. Phthalates: European regulation, chemistry, pharmacokinetic and related toxicity. Environ. Toxicol. Phar. 2013, 36, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Dargnat, C.; Teil, M.-J.; Chevreuil, M.; Blanchard, M. Phthalate removal throughout wastewater treatment plant: Case study of Marne Aval station (France). Sci. Total Environ. 2009, 407, 1235–1244. [Google Scholar] [CrossRef]
- Adhoum, N.; Monser, L. Removal of phthalate on modified activated carbon: Application to the treatment of industrial wastewater. Sep. Purif. Technol. 2004, 38, 233–239. [Google Scholar] [CrossRef]
- Gültekin, I.; Ince, N.H. Synthetic endocrine disruptors in the environment and water remediation by advanced oxidation processes. J. Environ. Manage. 2007, 85, 816–832. [Google Scholar] [CrossRef] [PubMed]
- Zolfaghari, M.; Drogui, P.; Seyhi, B.; Brar, K.; Buelna, G.; Dubé, R. Occurrence, fate and effects of di (2-ethylhexyl) phthalate in wastewater treatment plants: A review. Environ. Pollut. 2014, 194, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Kivaisi, A.K. The potential for constructed wetlands for wastewater treatment and reuse in developing countries: A review. Ecol. Eng. 2001, 16, 545–560. [Google Scholar] [CrossRef]
- Ye, J.; Xu, Z.X.; Li, H.Z. Dynamic rule of organic matter removal in vertical-flow constructed wetland. Environ. Sci. 2008, 29, 2166–2171. [Google Scholar]
- Gruchlik, Y.; Linge, K.; Joll, C. Removal of organic micropollutants in waste stabilisation ponds: A review. J. Environ. Manage. 2018, 206, 202–214. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, H. The fate and risk of selected pharmaceutical and personal care products in wastewater treatment plants and a pilot-scale multistage constructed wetland system. Environ. Sci. Pollut. R. 2014, 21, 1466–1479. [Google Scholar] [CrossRef]
- Stales, C.A.; Peterson, D.R.; Parkerton, T.; Adams, W. The environmental fate of phthalate esters: A literature review. Chemosphere 1997, 35, 667–749. [Google Scholar] [CrossRef]
- Tang, X.; Suyu, W.; Yang, Y.; Ran, T.; Dai, Y.-N.; Dan, A.; Li, L. Removal of six phthalic acid esters (PAEs) from domestic sewage by constructed wetlands. Chem. Eng. J. 2015, 275, 198–205. [Google Scholar]
- Wania, F.; Mackay, D. A global distribution model for persistent organic chemicals. Sci. Total. Environ. 1995, 160–161, 0–232. [Google Scholar] [CrossRef]
- Xu, F.L.; Qin, N.; Zhu, Y.; He, W.; Kong, X.Z.; Barbour, M.; He, Q.S.; Wang, Y.; Ouyang, H.; Tao, S. Multimedia fate modeling of polycyclic aromatic hydrocarbons (PAHs) in Lake Small Baiyangdian, Northern China. Ecol. Model. 2013, 252, 246–257. [Google Scholar] [CrossRef]
- Wania, F.; Dugani, S. Assessing the long-range transport potential of polybrominated diphenyl ethers: A comparison of four multimedia models. Environ. Toxicol. Chem. 2003, 22, 1252–1261. [Google Scholar] [CrossRef]
- Zhu, Y.; Tao, S.; Price, O.; Shen, H.; Jones, K.; Sweetman, A. Environmental distributions of benzo[a]pyrene in China: Current and future emission reduction scenarios explored using a spatially explicit multimedia fate model. Environ. Sci. Technol. 2015, 49, 13868–13877. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Price, O.; Tao, S.; Jones, K.; Sweetman, A. A new multimedia contaminant fate model for China: How important are environmental parameters in influencing chemical persistence and long-range transport potential? Environ. Int. 2014, 6, 18–27. [Google Scholar] [CrossRef]
- Hsu, F.C.; Marxmiller, R.L.; Yang, A.Y. Study of root uptake and xylem translocation of cinmethylin and related compounds in detopped soybean roots using a pressure chamber technique. Plant. Physiol. 1990, 93, 1573–1578. [Google Scholar] [CrossRef]
- Tian, F.; Liu, Y.; Liu, C.; Gu, H.; Liu, H. Pollution Status and multimedia fate simulation of phthalate acid esters (PAEs) in an arid city. Pol. J. Environ. Stud. 2016, 25, 325–331. [Google Scholar]
- Simon, S.; Graham, J.C.; Terp, S.D. Uptake of 40K and 137Cs in native plants of the Marshall Islands. J. Environ. Radioactiv. 2002, 59, 223–243. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, B.T.; Teng, Y. Distribution of phthalate acid esters in lakes of Beijing and its relationship with anthropogenic activities. Sci. Total Environ. 2014, 476, 107–113. [Google Scholar] [CrossRef]
- Marttinen, S.; Kettunen, R.H.; Sormunen, K.; Rintala, J. Removal of bis (2-ethylhexyl) phthalate at a sewage treatment plant. Water. Res. 2003, 37, 1385–1393. [Google Scholar] [CrossRef]
- Oliver, R.; May, E.; Williams, J. The occurrence and removal of phthalates in a trickle filter STW. Water Res. 2005, 39, 4436–4444. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Hawker, D.W.; Mueller, J.F.; Leusch, F.D.; Tremblay, L.A.; Chapman, H.F. Modelling of the fate of selected endocrine disruptors in a municipal wastewater treatment plant in South East Queensland, Australia. Chemosphere 2007, 69, 644–654. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Deng, J.-q.; Zhan, F.C.; Wu, Z.B. Effects of constructed wetland system on the removal of dibutyl phthalate (DBP). Microbiol. Res. 2009, 164, 206–211. [Google Scholar] [CrossRef]
- Yu Zhao, W.; Bin Wu, Z.; Hong Zhou, Q.; Ping Cheng, S.; Ping Fu, G.; He, F. Removal of dibutyl phthalate by a staged, vertical-flow constructed wetland. Wetlands 2004, 24, 202. [Google Scholar] [CrossRef]
- Guo, X.; Wang, L.; Wang, X.; Liu, H. Occurrence and environmental risk assessment of PAEs in Weihe River near Xi’an City, China. Water Sci. Technol. 2013, 67, 948–958. [Google Scholar] [CrossRef]
- Wu, X.; Hong, H.; Liu, X.; Guan, W.; Meng, L.; Ye, Y.; Ma, Y. Graphene-dispersive solid-phase extraction of phthalate acid esters from environmental water. Sci. Total Environ. 2013, 444, 224–230. [Google Scholar] [CrossRef]
- Teil, M.-J.; Blanchard, M.; Moreau-Guigon, E.; Dargnat, C.; Alliot, F.; Bourges, C.; Desportes, A.; Chevreuil, M. Phthalate fate in the hydrographic network of the river Seine basin (France) under contrasted hydrological conditions. Water Air Soil Poll. 2013, 224, 1592–1606. [Google Scholar] [CrossRef]
- Liang, D.-W.; Zhang, T.; Fang, H.; He, J. Phthalates biodegradation in the environment. Appl. Microbiol. Biot. 2008, 80, 183. [Google Scholar] [CrossRef]
- Julinová, M.; Slavík, R. Removal of phthalates from aqueous solution by different adsorbents: A short review. J. Environ. Manage. 2012, 94, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Yuwatini, E.; Hata, N.; Kuramitz, H.; Taguchi, S. Effect of salting-out on distribution behavior of di (2-ethylhexyl) phthalate and its analogues between water and sediment. SpringerPlus 2013, 2, 422. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-W.; Chen, C.-F.; Dong, C.-D. Distribution of phthalate esters in sediments of Kaohsiung Harbor, Taiwan. Soil Sediment. Contam 2013, 22, 119–131. [Google Scholar] [CrossRef]
- Sun, H.; An, T.; Li, G.; Qiao, M.; Wei, D. Distribution, possible sources, and health risk assessment of SVOC pollution in small streams in Pearl River Delta, China. Environ. Sci. Pollut. Res. 2014, 21, 10083–10095. [Google Scholar] [CrossRef]
- Zeng, F.; Cui, K.; Xie, Z.; Liu, M.; Li, Y.; Lin, Y.; Zeng, Z.; Li, F. Occurrence of phthalate esters in water and sediment of urban lakes in a subtropical city, Guangzhou, South China. Environ. Int. 2008, 34, 372–380. [Google Scholar] [CrossRef]
- Sangster, J. Octanol-water partition coefficients of simple organic compounds. J. Phys. Chem. Ref. Data 1989, 18, 1111–1229. [Google Scholar] [CrossRef]
- Jackson Ellington, J. Octanol/water partition coefficients and water solubilities of phthalate esters. J. Chem. Eng. Data 1999, 44, 1414–1418. [Google Scholar] [CrossRef]
- Li, Y.-W.; Cai, Q.-Y.; Mo, C.-H.; Zeng, Q.-Y.; Lü, H.; Li, Q.-S.; Xu, G.-S. Plant uptake and enhanced dissipation of di(2-Ethylhexyl) phthalate (DEHP) in spiked soils by different plant species. Int. J. Phytoremediat. 2014, 16, 609–620. [Google Scholar] [CrossRef]
- Yin, R.; Lin, X.G.; Wang, S.G.; Zhang, H.Y. Effect of DBP/DEHP in vegetable planted soil on the quality of capsicum fruit. Chemosphere 2003, 50, 801–805. [Google Scholar] [CrossRef]
- Chi, J.; Gao, J. Effects of Potamogeton crispus L.–bacteria interactions on the removal of phthalate acid esters from surface water. Chemosphere 2015, 119, 59–64. [Google Scholar] [CrossRef]
- Dodgen, L.K.; Li, J.; Parker, D.; Gan, J.J. Uptake and accumulation of four PPCP/EDCs in two leafy vegetables. Environ. Pollut. 2013, 182, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Namiki, S.; Otani, T.; Seike, N. Fate and plant uptake of persistent organic pollutants in soil. Soil. Sci. Plant. Nut. 2013, 59, 669–679. [Google Scholar] [CrossRef]
- Macherius, A.; Eggen, T.; Lorenz, W.; Moeder, M.; Ondruschka, J.; Reemtsma, T. Metabolization of the bacteriostatic agent triclosan in edible plants and its consequences for plant uptake assessment. Environ. Sci. Technol. 2012, 46, 10797–10804. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.; Blake-Kalff, M.; Davies, E. Detoxification of xenobiotics by plants: Chemical modification and vacuolar compartmentation. Trends Plant. Sci. 1997, 2, 144–151. [Google Scholar] [CrossRef]
- Mikeš, O.; Cupr, P.; Trapp, S.; Klanova, J. Uptake of polychlorinated biphenyls and organochlorine pesticides from soil and air into radishes (Raphanus sativus). Environ. Pollut. 2009, 157, 488–496. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, L.; Liu, T.; Xie, E.; Liu, M.; Ding, A.; Zhang, B.-T.; Li, X.; Zhang, D. Partition and Fate of Phthalate Acid Esters (PAEs) in a Full-Scale Horizontal Subsurface Flow Constructed Wetland Treating Polluted River Water. Water 2020, 12, 865. https://doi.org/10.3390/w12030865
Zheng L, Liu T, Xie E, Liu M, Ding A, Zhang B-T, Li X, Zhang D. Partition and Fate of Phthalate Acid Esters (PAEs) in a Full-Scale Horizontal Subsurface Flow Constructed Wetland Treating Polluted River Water. Water. 2020; 12(3):865. https://doi.org/10.3390/w12030865
Chicago/Turabian StyleZheng, Lei, Tingting Liu, En Xie, Mingxue Liu, Aizhong Ding, Bo-Tao Zhang, Xiaoyan Li, and Dayi Zhang. 2020. "Partition and Fate of Phthalate Acid Esters (PAEs) in a Full-Scale Horizontal Subsurface Flow Constructed Wetland Treating Polluted River Water" Water 12, no. 3: 865. https://doi.org/10.3390/w12030865
APA StyleZheng, L., Liu, T., Xie, E., Liu, M., Ding, A., Zhang, B.-T., Li, X., & Zhang, D. (2020). Partition and Fate of Phthalate Acid Esters (PAEs) in a Full-Scale Horizontal Subsurface Flow Constructed Wetland Treating Polluted River Water. Water, 12(3), 865. https://doi.org/10.3390/w12030865




