Evaluation of Dispersed Alkaline Substrate and Diffusive Exchange System Technologies for the Passive Treatment of Copper Mining Acid Drainage
Abstract
1. Introduction
2. Materials and Methods
2.1. Reactor Set Up and Operation
2.2. Water Sampling and Analysis
2.3. Solids Sampling and Mineralogy
2.4. Microbiological and Molecular Analysis
3. Results
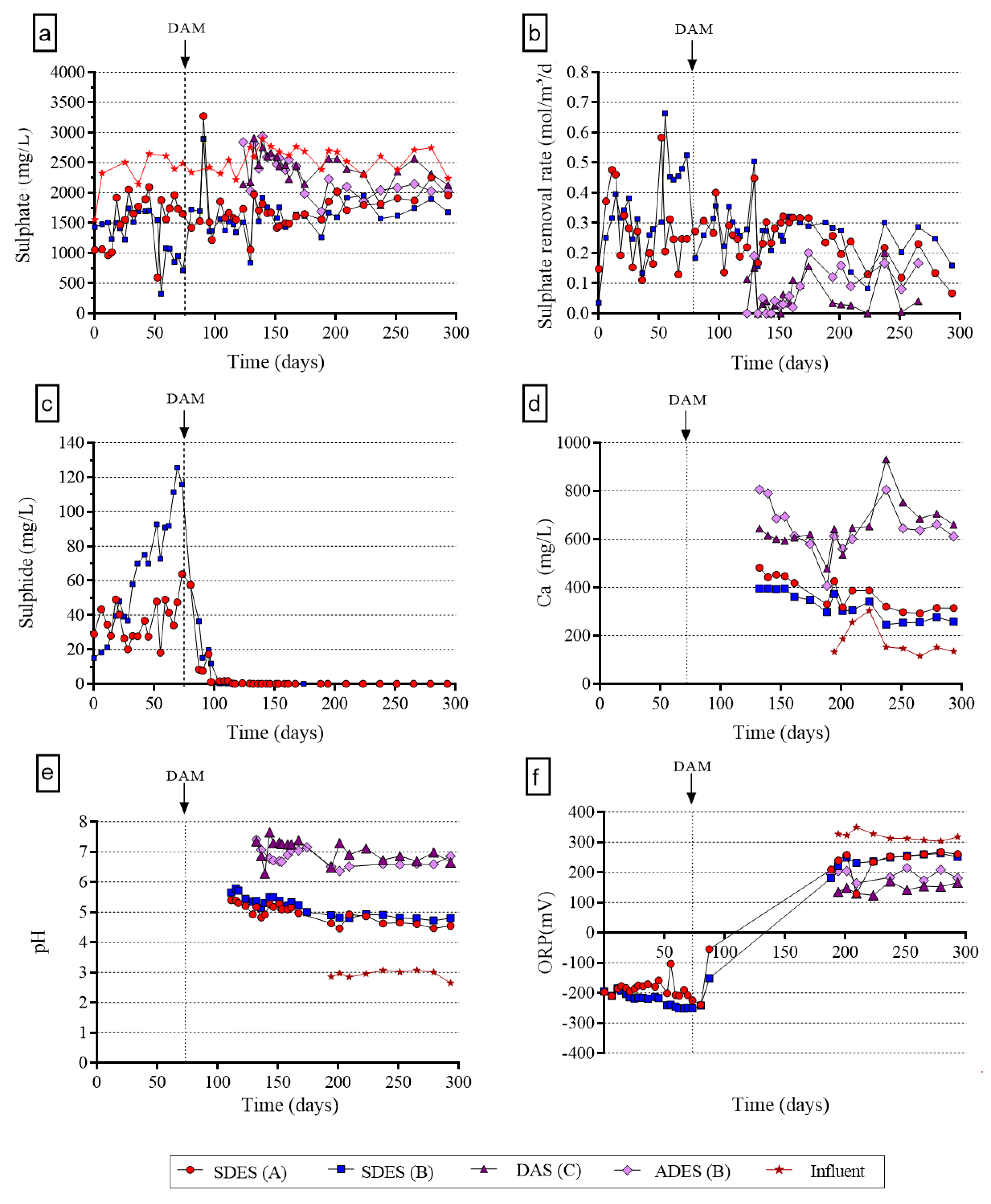
3.1. Effluent Hydrochemistry
3.1.1. Sulfate Removal
3.1.2. Chemical and Biological Process Indicators (ORP, pH, Sulfide, Calcium)
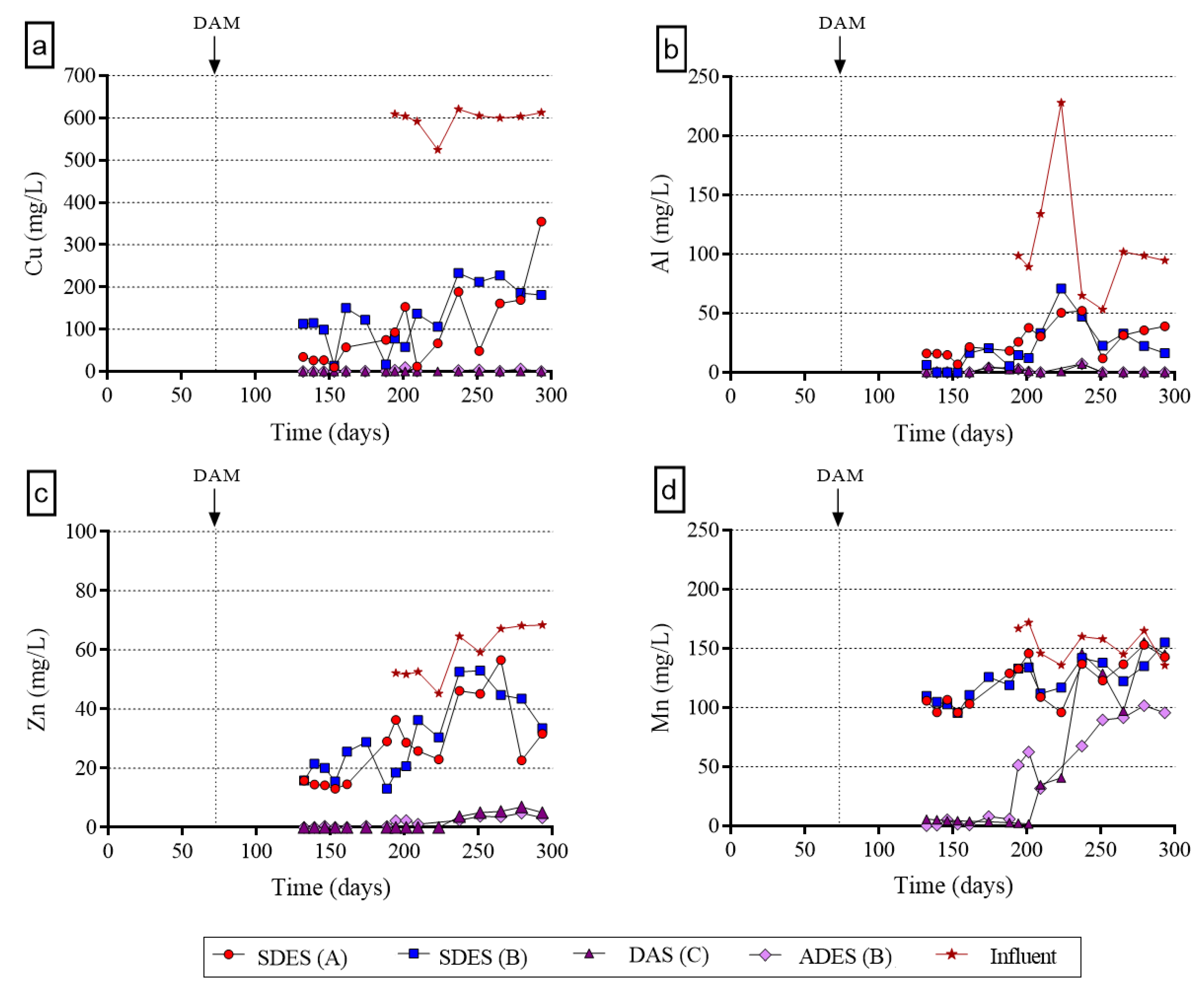
3.1.3. Metal Removal
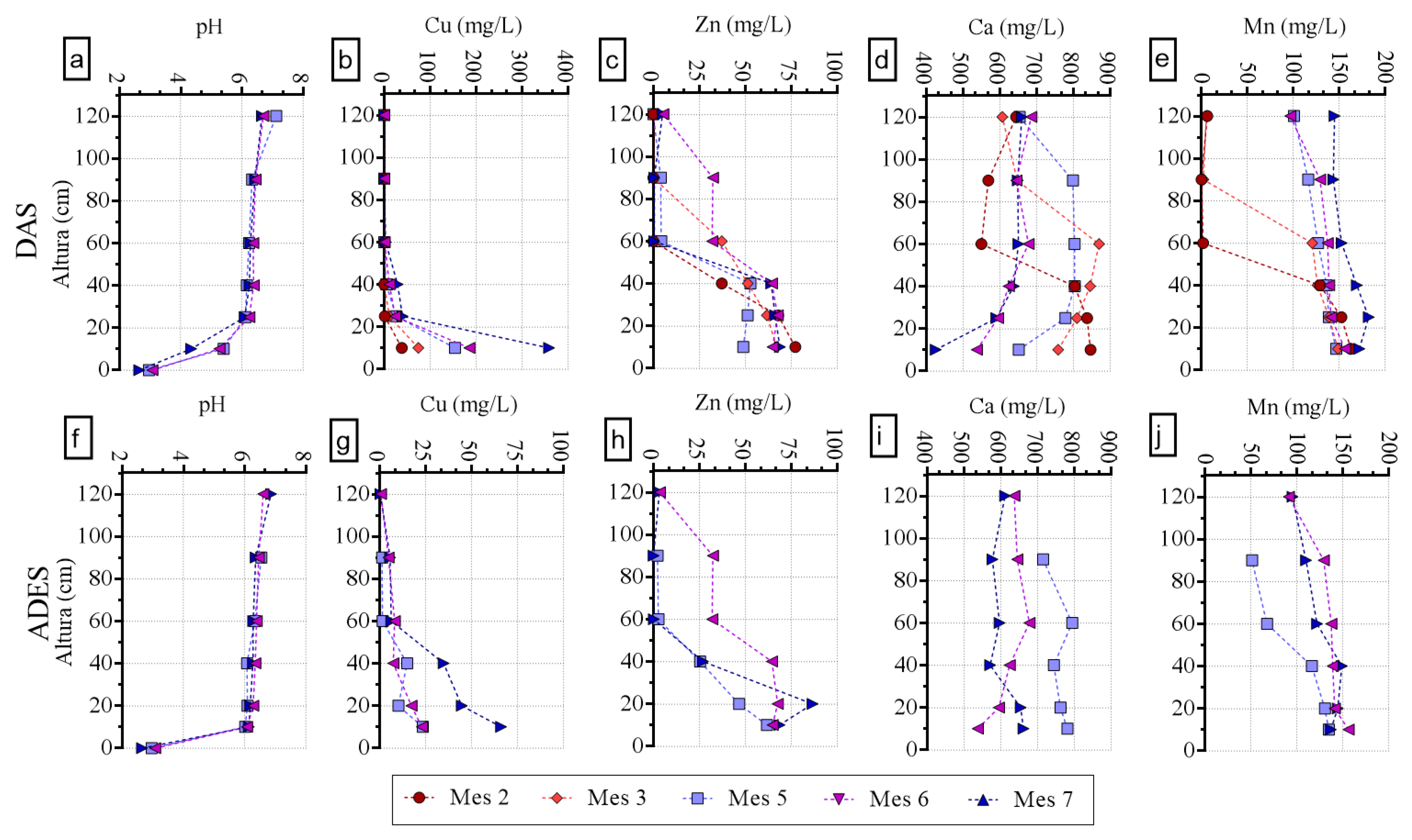
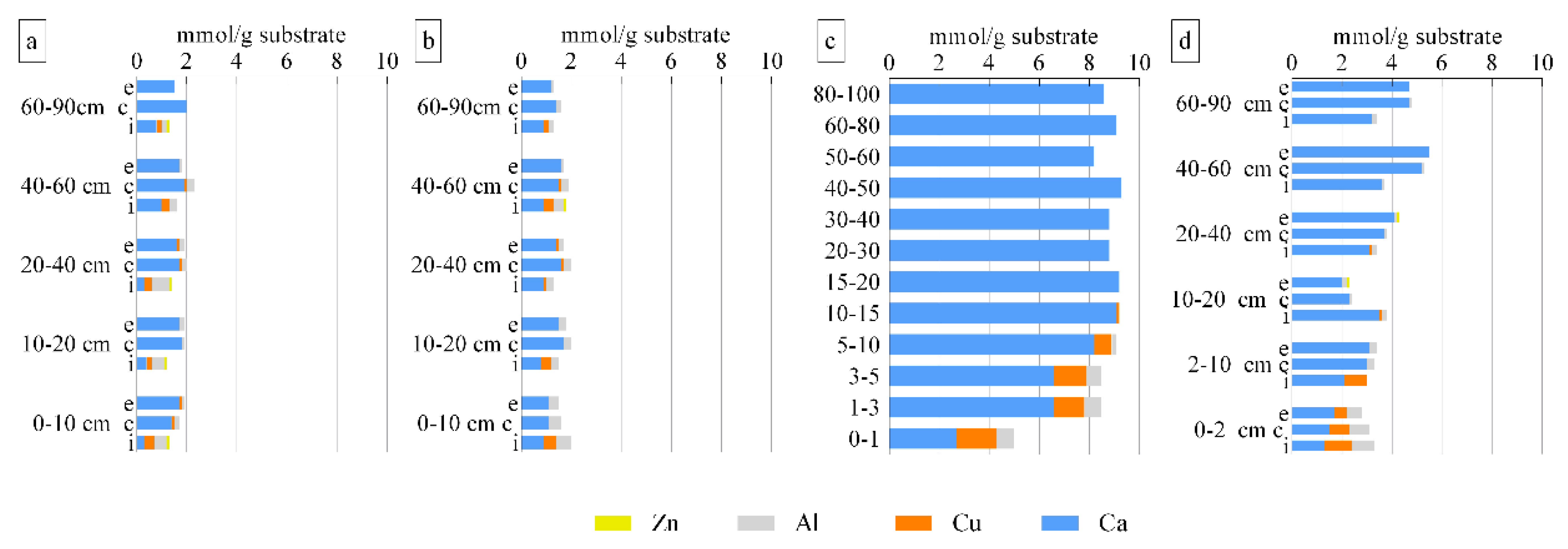
3.2. Reactor Hydrochemical Profiles (DAS and ADES Only)
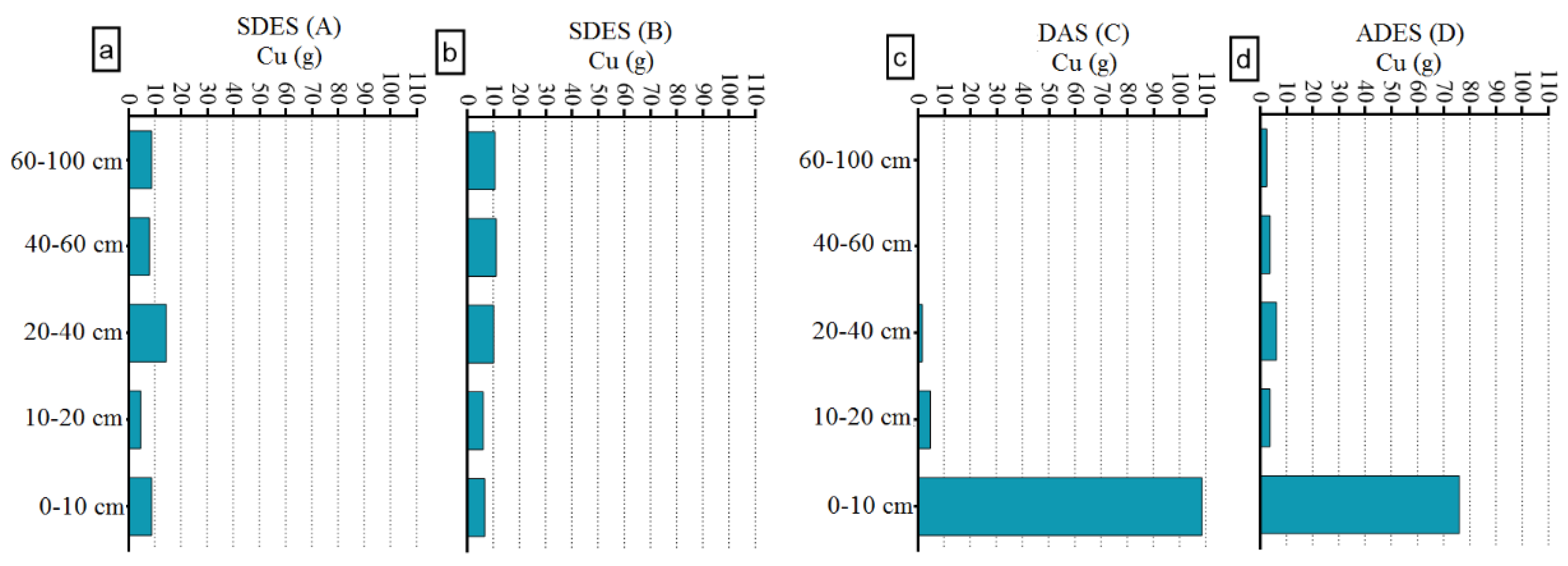
3.3. Metal Accumulation in Reactors
3.4. Post-Treatment Mineralogy (XRD)
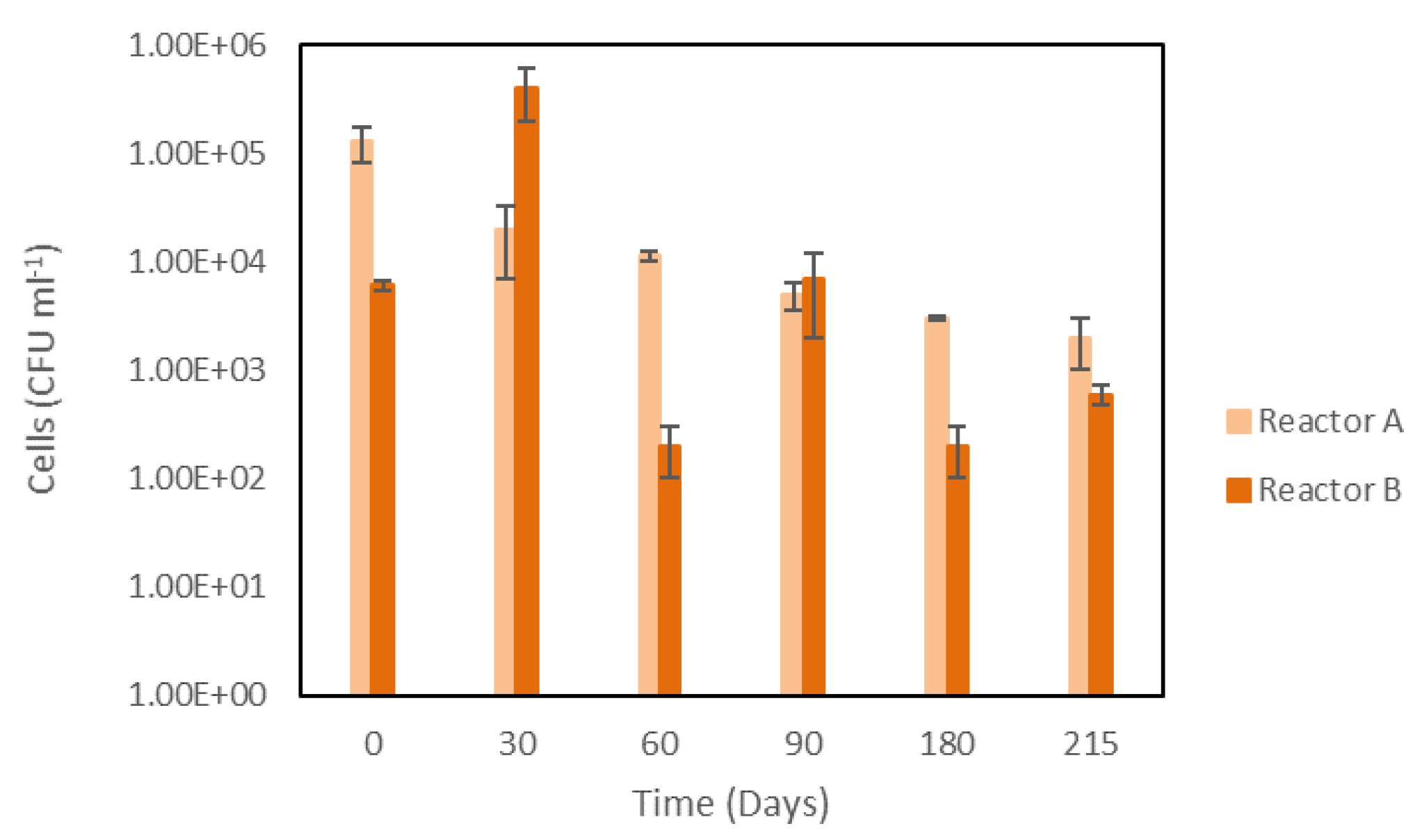
3.5. Microbiological Changes in the Bioreactors
3.5.1. Evolution of CFU Counts
3.5.2. Identification of Bacterial Isolates
4. Conclusions
- The DES technology showed a good performance in the chemical and biological treatment of AMD, eliminating clogging problems, contributing to increasing the useful life of the ADES reactor, and controlling the harmful effects of metals, favoring the establishment of a diverse community of microorganisms in the SDES reactors.
- Due to the high reactivity of its substrate, the DAS reactor accumulated the metals in a lower reactor volume, allowing to conclude that it requires the lowest HRT and smallest substrate volume among all reactor types.
- A side effect of this concentration of precipitates in the DAS, however, was the creation of a hardpan layer that caused a significant loss of hydraulic conductivity. Periodic harvesting (removal) of the hardpan could facilitate metal recovery and restore substrate permeability.
- The study indicates that the limitations of each treatment could be overcome by combining their strengths in a combined or serial treatment, such as introducing screen tubes in DAS reactors to overcome clogging problems.
Author Contributions
Funding
Conflicts of Interest
References
- Ayora, C.; Nieto, J.-M.; Rötting, T.S.; Caraballo, M.A.; Carrera, J.; Macias, F. Acid mine drainage in the Iberian Pyrite Belt: 2. Lessons learned from recent passive remediation experiences. Environ. Sci. Pollut. Res. 2013, 20, 7837–7853. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, K.B. New perspectives in acid mine drainage microbiology. Hydrometallurgy 2010, 104, 448–453. [Google Scholar] [CrossRef]
- Simate, G.S.; Ndlovu, S. Acid mine drainage: Challenges and opportunities. J. Environ. Chem. Eng. 2014, 2, 1785–1803. [Google Scholar] [CrossRef]
- Rakotonimaro, T.V.; Neculita, C.M.; Bussière, B.; Zagury, G.J. Effectiveness of various dispersed alkaline substrates for the pre-treatment of ferriferous acid mine drainage. Appl. Geochem. 2016, 73, 13–23. [Google Scholar] [CrossRef]
- Pérez, N.; Schwarz, A.; Sanhueza, P.; Chaparro, G. Performance of three bench-scale diffusive exchange systems during treatment of acid mine drainage with high copper concentration. Desalin. Water Treat. 2017, 64, 21–30. [Google Scholar] [CrossRef]
- Pérez, N.; Schwarz, A.; Barahona, E.; Sanhueza, P.; Diaz, I.; Urrutia, H. Performance of two differently designed permeable reactive barriers with sulfate and zinc solutions. Sci. Total Environ. 2018, 642, 894–903. [Google Scholar] [CrossRef]
- Pino, L.; Vargas, C.; Schwarz, A.; Borquez, R. Influence of operating conditions on the removal of metals and sulfate from copper acid mine drainage by nanofiltration. Chem. Eng. J. 2018, 345, 114–125. [Google Scholar] [CrossRef]
- Andalaft, J.; Schwarz, A.; Pino, L.; Fuentes, P.; Bórquez, R.; Aybar, M. Assessment and modeling of nanofiltration of acid mine drainage. Ind. Eng. Chem. Res. 2018, 57, 14727–14739. [Google Scholar] [CrossRef]
- Torregrosa, M.; Schwarz, A.; Nancucheo, I.; Balladares, E. Evaluation of the bio-protection mechanism in diffusive exchange permeable reactive barriers for the treatment of acid mine drainage. Sci. Total Environ. 2019, 655, 374–383. [Google Scholar] [CrossRef]
- Johnson, D.B. Biomining-biotechnologies for extracting and recovering metals from ores and waste materials. Curr. Opin. Biotechnol. 2014, 30, 24–31. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Kalin, M.; Fyson, A.; Wheeler, W.N. The chemistry of conventional and alternative treatment systems for the neutralization of acid mine drainage. Sci. Total Environ. 2006, 366, 395–408. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, M.A.; Macías, F.; Rötting, T.S.; Nieto, J.M.; Ayora, C. Long term remediation of highly polluted acid mine drainage: A sustainable approach to restore the environmental quality of the Odiel river basin. Environ. Pollut. 2011, 159, 3613–3619. [Google Scholar] [CrossRef] [PubMed]
- Neculita, C.M.; Zagury, G.J.; Bussière, B. Effectiveness of sulfate-reducing passive bioreactors for treating highly contaminated acid mine drainage: II. Metal removal mechanisms and potential mobility. Appl. Geochemistry 2008, 23, 3545–3560. [Google Scholar] [CrossRef]
- ITRC (Interstate Technology & Regulatory Council). Biochemical Reactors for Mining Influenced Water. BCR-1; Biochemical Reactors for Mining-Influenced Waste Team: Washington, DC, USA, 2013. [Google Scholar]
- Jage, C.R.; Zipper, C.E.; Noble, R. Factors affecting alkalinity generation by successive alkalinity-producing systems. J. Environ. Qual. 2010, 30, 1015–1022. [Google Scholar] [CrossRef]
- Neculita, C.M.; Zagury, G.J. Biological treatment of highly contaminated acid mine drainage in batch reactors: Long-term treatment and reactive mixture characterization. J. Hazard. Mater. 2008, 157, 358–366. [Google Scholar] [CrossRef]
- Vasquez, Y.; Neculita, C.M.; Roldan, F.; Escobar, M.C.; Arbeli, Z. Selection of reactive mixture for biochemical passive treatment of acid mine drainage. Environ. Earth Sci. 2016, 75, 576–587. [Google Scholar] [CrossRef]
- Zagury, G.J.; Kulnieks, V.I.; Neculita, C.M. Characterization and reactivity assessment of organic substrates for sulfate-reducing bacteria in acid mine drainage treatment. Chemosphere 2006, 64, 944–954. [Google Scholar] [CrossRef]
- Neculita, C.-M.; Zagury, G.J.; Bussière, B. Passive treatment of acid mine drainage in bioreactors using sulfate-reducing bacteria. J. Environ. Qual. 2007, 36, 1–16. [Google Scholar] [CrossRef]
- USEPA (United States Environmental Protection Agency). Reference Guide to Treatment Technologies for Mining-Influenced Water. EPA 542-R-14-001; Office of Superfund Remediation and Technology Innovation, 2014. Available online: https://www.epa.gov/remedytech/reference-guide-treatment-technologies-mining-influenced-water (accessed on 12 January 2020).
- Drennan, D.M.; Almstrand, R.; Lee, I.; Landkamer, L.; Figueroa, L.; Sharp, J.O. Organoheterotrophic bacterial abundance associates with zinc removal in lignocellulose-based sulfate-reducing systems. Environ. Sci. Technol. 2016, 50, 378–387. [Google Scholar] [CrossRef]
- Vasquez, Y.; Neculita, C.M.; Roldan, F.; Escobar, M.C.; Saenz, J.S.; Quiceno-Vallejo, M.F.; Arbeli, Z. Effect of hydraulic retention time on microbial community in biochemical passive reactors during treatment of acid mine drainage. Bioresour. Technol. 2017, 247, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Rezadehbash, M.; Baldwin, S.A. Core sulphate-reducing microorganisms in metal-removing semi-passive biochemical reactors and the co-occurrence of methanogens. Microorganisms 2018, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Torres, E.; Castillo, J.; Macías, F.; Torres, E.; Ayora, C.; Lozano, A.; Gomez-Arias, A. Passive elimination of sulfate and metals from acid mine drainage using combined limestone and barium carbonate systems. J. Clean. Prod. 2018, 182, 114–123. [Google Scholar] [CrossRef]
- Schwarz, A.O.; Rittmann, B.E. The diffusion-active permeable reactive barrier. J. Contam. Hydrol. 2010, 112, 155–162. [Google Scholar] [CrossRef]
- Caraballo, M.A.; Macías, F.; Nieto, J.M.; Castillo, J.; Quispe, D.; Ayora, C. Hydrochemical performance and mineralogical evolution of a dispersed alkaline substrate (DAS) remediating the highly polluted acid mine drainage in the full-scale passive treatment of Mina Esperanza (SW Spain). Am. Mineral. 2011, 96, 1270–1277. [Google Scholar] [CrossRef]
- Lozano, A.; Ayora, C.; Macias, F.; Nieto, J.M.; Gomez-Arias, A.; Castillo, J.; Heerden, E. Sulfate removal from acid mine drainage: Evaluation of granular BaCO3 with column experiments. Macla 2014, 19, 83–84. [Google Scholar]
- Rötting, T.S.; Caraballo, M.A.; Serrano, J.A.; Ayora, C.; Carrera, J. Field application of calcite Dispersed Alkaline Substrate (calcite-DAS) for passive treatment of acid mine drainage with high Al and metal concentrations. Appl. Geochem. 2008, 23, 1660–1674. [Google Scholar] [CrossRef]
- Soler, J.M.; Mogollón, J.L.; Tamura, N.; Cama, J.; Kunz, M.; Nico, P.S.; Boi, M.; Ayora, C. The passivation of calcite by acid mine water. Column experiments with ferric sulfate and ferric chloride solutions at pH 2. Appl. Geochem. 2008, 23, 3579–3588. [Google Scholar] [CrossRef]
- Rötting, T.S.; Thomas, R.C.; Ayora, C.; Carrera, J. Passive treatment of acid mine drainage with high metal concentrations using dispersed alkaline substrate. J. Environ. Qual. 2008, 37, 1741–1751. [Google Scholar] [CrossRef]
- Macías, F.; Caraballo, M.A.; Rötting, T.S.; Pérez-López, R.; Nieto, J.M.; Ayora, C. From highly polluted Zn-rich acid mine drainage to non-metallic waters: Implementation of a multi-step alkaline passive treatment system to remediate metal pollution. Sci. Total Environ. 2012, 433, 323–330. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Water Works Association/American Public Health Association/Water Environment Federation: Denver, CO, USA, 2005. [Google Scholar]
- Rowe, O.F.; Sánchez-España, J.; Hallberg, K.B.; Johnson, D.B. Microbial communities and geochemical dynamics in an extremely acidic, metal-rich stream at an abandoned sulfide mine (Huelva, Spain) underpinned by two functional primary production systems. Environ. Microbiol. 2007, 9, 1761–1771. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.J. 16S/23S rRNA sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Goodfellow, M., Stackebrandt, E., Eds.; Wiley Publishing: New York, NY, USA, 1991; pp. 115–175. [Google Scholar]
- Marchesi, J.R.; Wade, J.R.; Sato, T.; Weightman, A.J.; Martin, T.A.; Fry, J.C.; Hiom, S.J.; Dymock, D.; Wade, W.G. Design and evaluation of useful bacterium-specific PCR primers that amplify genes coding for bacterial 16S rRNA. Appl. Environ. Microbiol. 1998, 64, 795–799. [Google Scholar] [CrossRef] [PubMed]
- González, D.; Liu, Y.; Villa Gomez, D.; Southam, G.; Hedrich, S.; Galleguillos, P.; Colipai, C.; Nancucheo, I. Performance of a sulfidogenic bioreactor inoculated with indigenous acidic communities for treating an extremely acidic mine water. Miner. Eng. 2019, 131, 370–375. [Google Scholar] [CrossRef]
- Caraballo, M.A.; Rötting, T.S.; Silva, V. Implementation of an MgO-based metal removal step in the passive treatment system of Shilbottle, UK: Column experiments. J. Hazard. Mater. 2010, 181, 923–930. [Google Scholar] [CrossRef]
- Gibert, O.; De Pablo, J.; Cortina, J.L.; Ayora, C. Municipal compost-based mixture for acid mine drainage bioremediation: Metal retention mechanisms. Appl. Geochem. 2005, 20, 1648–1657. [Google Scholar] [CrossRef]
- Caraballo, M.A.; Rötting, T.S.; Nieto, J.M.; Ayora, C. Sequential extraction and DXRD applicability to poorly crystalline Fe- and Al-phase characterization from an acid mine water passive remediation system. Am. Mineral. 2009, 94, 1029–1038. [Google Scholar] [CrossRef]
- Kaksonen, A.H.; Puhakka, J.A. Sulfate reduction based bioprocesses for the treatment of acid mine drainage and the recovery of metals. Eng. Life Sci. 2007, 7, 541–564. [Google Scholar] [CrossRef]
- Utgikar, V.P.; Harmon, S.M.; Tabak, H.H.; Govind, R.; Chaudhary, N.; Haines, J.R. Inhibition of sulfate-reducing bacteria by metal sulfide formation in bioremediation of acid mine drainage. Environ. Toxicol. 2002, 17, 40–48. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Chang, J.; Quan, X.; Li, Q. Biological sulfate reduction in the acidogenic phase of anaerobic digestion under dissimilatory Fe (III)—Reducing conditions. Water Res. 2013, 47, 2033–2040. [Google Scholar] [CrossRef]
- Zhao, Y.; Fu, Z.; Chen, X.; Zhang, G. Bioremediation process and bioremoval mechanism of heavy metal ions in acidic mine drainage. Chem. Res. Chin. Univ. 2018, 34, 33–38. [Google Scholar] [CrossRef]
- Dar, S.A.; Bijmans, M.F.M.; Dinkla, I.J.T.; Geurkink, B.; Lens, P.N.L.; Dopson, M. Population Dynamics of a Single-Stage Sulfidogenic Bioreactor Treating Synthetic Zinc-Containing Waste Streams. Microb. Ecol. 2009, 58, 529–537. [Google Scholar] [CrossRef]
- Briones, A.M.; Daugherty, B.J.; Angenent, L.T.; Rausch, K.D.; Tumbleson, M.E.; Raskin, L. Microbial diversity and dynamics in multi- and single-compartment anaerobic bioreactors processing sulfate-rich waste streams. Environ. Microbiol. 2007, 9, 93–106. [Google Scholar] [CrossRef]
- Logan, M.V.; Reardon, K.F.; Figueroa, L.A.; McLain, J.E.; Ahmann, D.M. Microbial community activities during establishment, performance, and decline of bench-scale passive treatment systems for mine drainage. Water Res. 2005, 39, 4537–4551. [Google Scholar] [CrossRef] [PubMed]
- Gibiino, G.; Binda, C.; Lopetuso, L.R.; Rizzatti, G.; Gasbarrini, A.; Scaldaferri, F. Exploring Bacteroidetes: Metabolic key points and immunological tricks of our gut commensals. Dig. Liver Dis. 2018, 50, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, S.A.; Sanei, H.; Hallam, S.; Rezadehbashi, M.; Khoshnoodi, M.; Taupp, M.; Mattes, A. The microbial community of a passive biochemical reactor treating arsenic, zinc, and sulfate-rich seepage. Front. Bioeng. Biotechnol. 2015, 3, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, C.W.; Beveridge, T.J.; Hellstrom, A. Cellulase and xylanase release from Bacteroides succinogenes and its importance in the rumen environment. Appl. Environ. Microbiol. 1981, 42, 886–896. [Google Scholar] [CrossRef]
- Hiibel, S.R.; Reisman, D.J.; Pruden, A.; Breazeal, M.V.R.; Reardon, K.F.; Pereyra, L.P. Effect of organic substrate on the microbial community structure in pilot-scale sulfate-reducing biochemical reactors treating mine drainage. Environ. Eng. Sci. 2011, 28, 563–572. [Google Scholar] [CrossRef]
- Chamkh, F.; Qatibi, A.-I.; Besson, S.; El Asli, A.-G.; Sproer, C.; Lemos, P.C.; Reis, M.; Bennisse, R.; Labat, M. Desulfovibrio marrakechensis sp. nov, a 1,4-tyrosol-oxidizing, sulfate-reducing bacterium isolated from olive mill wastewater. Int. J. Syst. Evol. Microbiol. 2009, 59, 936–942. [Google Scholar] [CrossRef][Green Version]
- Zhu, B.; Macleod, L.C.; Kitten, T.; Xu, P. Streptococcus sanguinis biofilm formation & interaction with oral pathogens. Future Microbiol. 2018, 13, 915–932. [Google Scholar] [CrossRef]
- Abraham, W.R.; Stroempl, C.; Meyer, H.; Lindholst, S.; Moore, E.R.; Christ, R.; Vancanneyt, M.; Tindall, B.J.; Bennasar, A.; Smit, J.; et al. Brevundimonas mediterranea sp. nov, a non-stalked species from the Mediterranean Sea. Int. J. Syst. Bacteriol. 1999, 49, 1053–1073. [Google Scholar] [CrossRef]
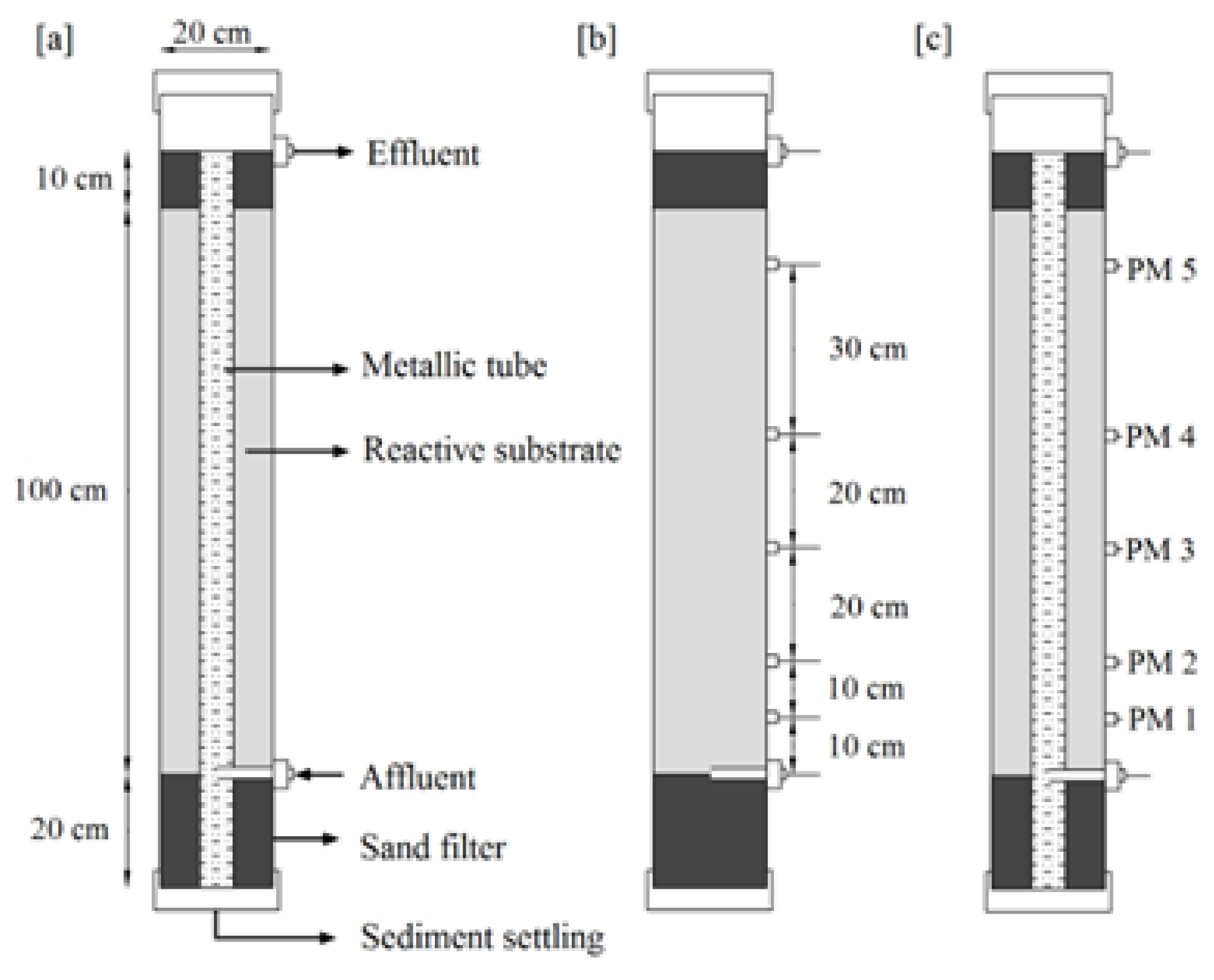



| Substrate | Reactor A (SDES) | Reactor B (SDES) | Reactor C (DAS) | Reactor D (ADES) |
|---|---|---|---|---|
| Fibers of forest origin | 4.48 kg | 4.05 kg | - | - |
| Leaf compost | 2.84 kg | 2.57 kg | - | - |
| Cow manure | 1.63 kg | 1.48 kg | - | - |
| Pulverized calcite | 3.15 kg | 2.85 kg | 14.9 kg | - |
| Bacterial inoculum | 8.17 L | 7.38 L | - | - |
| Calcite sand (1–4.75 mm) | - | - | - | 20.05 kg |
| Wood chips | - | - | 1.95 kg | - |
| Coarse sand (1–4.75 mm) | - | - | - | 17.38 kg |
| Reactor | Cu | Al | Zn | Mn | Total |
|---|---|---|---|---|---|
| Rates (mmol m−3 day−1) | |||||
| SDES A | 213.3 ± 40.7 | 76.7 ± 32.8 | 14.1 ± 5.0 | 22.5 ± 11.4 | 326.6 ± 35.3 |
| SDES B | 197.1 ± 24.2 | 79.9 ± 26.5 | 12.8 ± 5.1 | 21.0 ± 7.9 | 310.8 ± 22.9 |
| DAS C | 221.2 ± 19.8 | 88.1 ± 30.3 | 21.0 ± 3.0 | 48.3 ± 24.6 | 378.6 ± 21.9 |
| ADES D | 245.9 ± 19.2 | 97.2 ± 32.2 | 23.3 ± 3.4 | 55.0 ± 17.0 | 421.4 ± 21.0 |
| Total removed (moles) and efficiencies (in parenthesis) | |||||
| SDES A | 1.26 (87%) | 0.47 (80%) | 0.09 (66%) | 0.19 (45%) | 2.02 (80%) |
| SDES B | 1.18 (82%) | 0.47 (82%) | 0.08 (60%) | 0.18 (41%) | 1.91 (77%) |
| DAS C | 1.41 (100%) | 0.57 (99%) | 0.14 (98%) | 0.31 (73%) | 2.42 (95%) |
| ADES D | 1.43 (100%) | 0.57 (99%) | 0.14 (98%) | 0.32 (76%) | 2.46 (96%) |
| Isolate | Main Physiological Characteristics | Amplified Product Length (nt) (16S rRNA gene) | Closest Relative | Similarity (%) | GenBank Accession Number |
|---|---|---|---|---|---|
| UDEC_USS ASMT5 16S | Strictly anaerobic, non-motile and Gram-negative [52] | 1384 | Desulfovibrio marrakechensis strain EMSSDQ4 [52] | 99 | MK225576 |
| UDEC_USS ASMT6 16S | Gram-positive, nonspore-forming and facultative anaerobe [53] | 1363 | Streptococcus sanguinis strain: JCM 5708 | 99 | MK226162 |
| UDEC_USS ASMT7 16S | Gram-negative, rod-shaped, non-spore-forming [54] | 1302 | Brevundimonas mediterranea strain V4.BO [54] | 99 | MK226198 |
| UDEC_USS ASMT8 16S | Gram-negative [48] | 1361 | Bacteroidetes bacterium | 94 | MK228862 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwarz, A.; Nancucheo, I.; Gaete, M.A.; Muñoz, D.; Sanhueza, P.; Torregrosa, M.; Rötting, T.; Southam, G.; Aybar, M. Evaluation of Dispersed Alkaline Substrate and Diffusive Exchange System Technologies for the Passive Treatment of Copper Mining Acid Drainage. Water 2020, 12, 854. https://doi.org/10.3390/w12030854
Schwarz A, Nancucheo I, Gaete MA, Muñoz D, Sanhueza P, Torregrosa M, Rötting T, Southam G, Aybar M. Evaluation of Dispersed Alkaline Substrate and Diffusive Exchange System Technologies for the Passive Treatment of Copper Mining Acid Drainage. Water. 2020; 12(3):854. https://doi.org/10.3390/w12030854
Chicago/Turabian StyleSchwarz, Alex, Iván Nancucheo, Maria A. Gaete, Diego Muñoz, Pamela Sanhueza, Martin Torregrosa, Tobias Rötting, Gordon Southam, and Marcelo Aybar. 2020. "Evaluation of Dispersed Alkaline Substrate and Diffusive Exchange System Technologies for the Passive Treatment of Copper Mining Acid Drainage" Water 12, no. 3: 854. https://doi.org/10.3390/w12030854
APA StyleSchwarz, A., Nancucheo, I., Gaete, M. A., Muñoz, D., Sanhueza, P., Torregrosa, M., Rötting, T., Southam, G., & Aybar, M. (2020). Evaluation of Dispersed Alkaline Substrate and Diffusive Exchange System Technologies for the Passive Treatment of Copper Mining Acid Drainage. Water, 12(3), 854. https://doi.org/10.3390/w12030854





