Managing Water Quality in Intermittent Supply Systems: The Case of Mukono Town, Uganda
Abstract
1. Introduction
2. Materials and Methods
2.1. Current State of Site and Its Water Distribution System
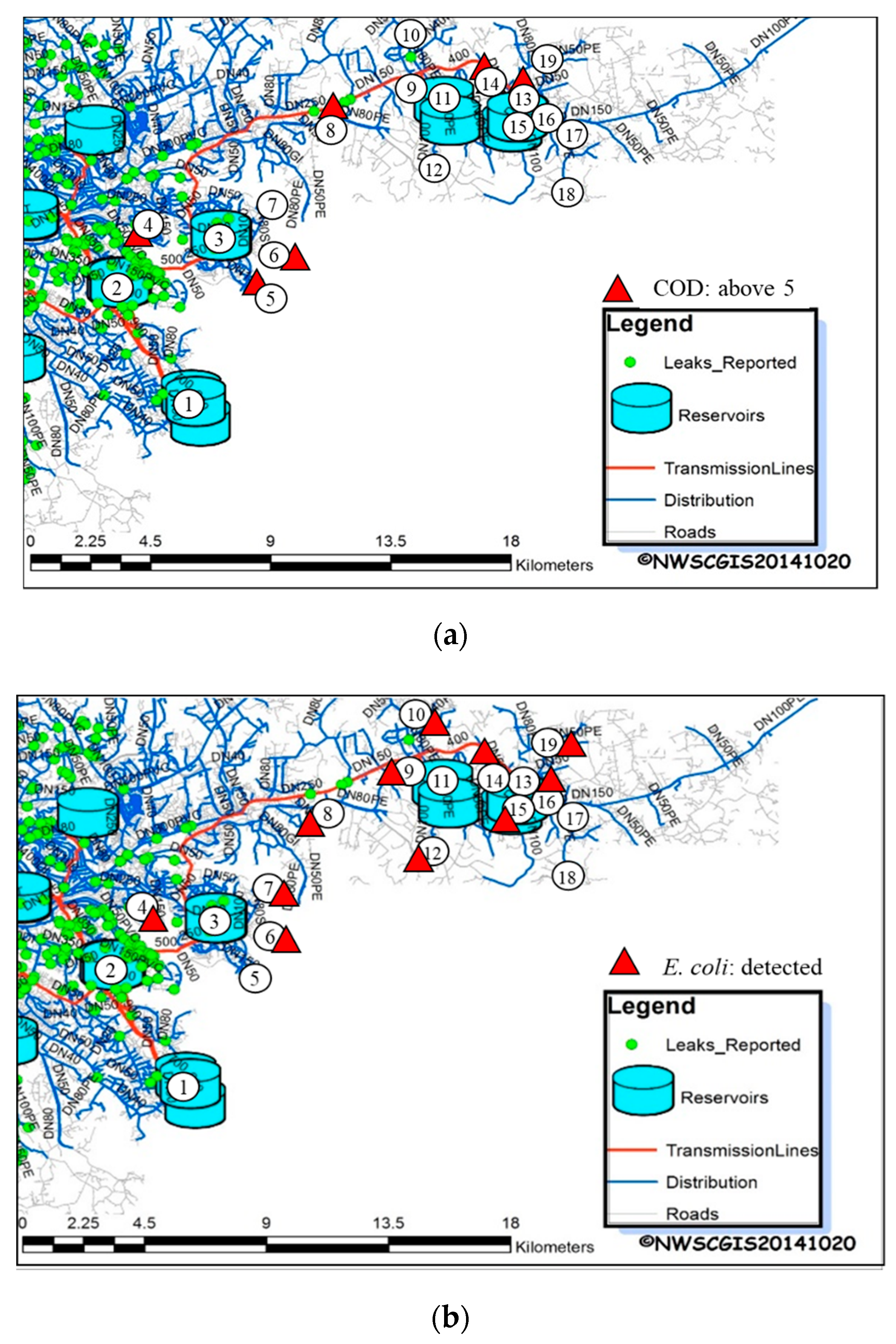
2.2. Sampling Locations
2.3. Choice of Water Quality Parameters
2.4. Sampling Methods, Measurement Items and Analysis
2.5. Water Quality Modelling
2.5.1. Headloss in Pipes (Hazen–Williams Formula)
2.5.2. Chlorine Decay Kinetics
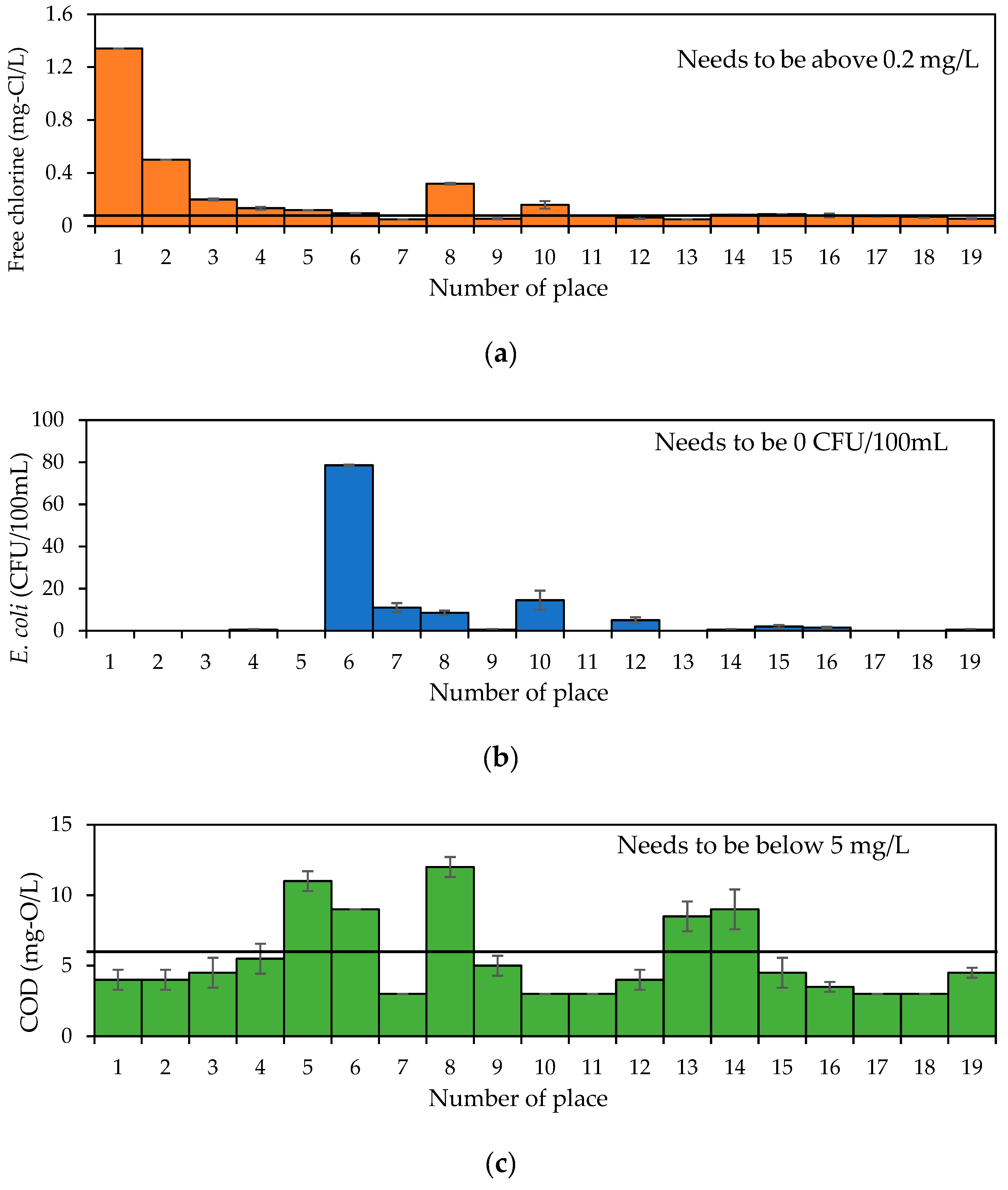
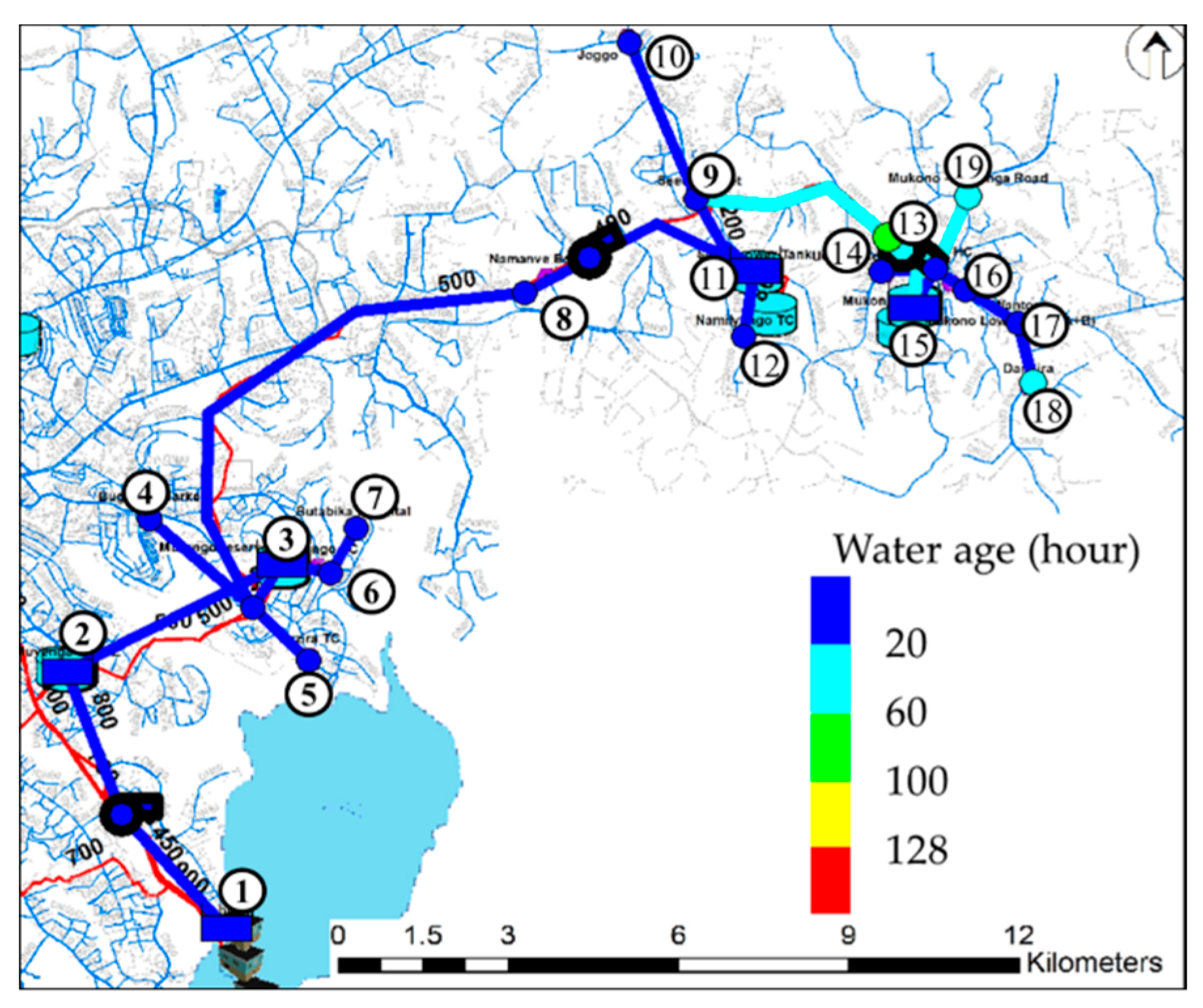
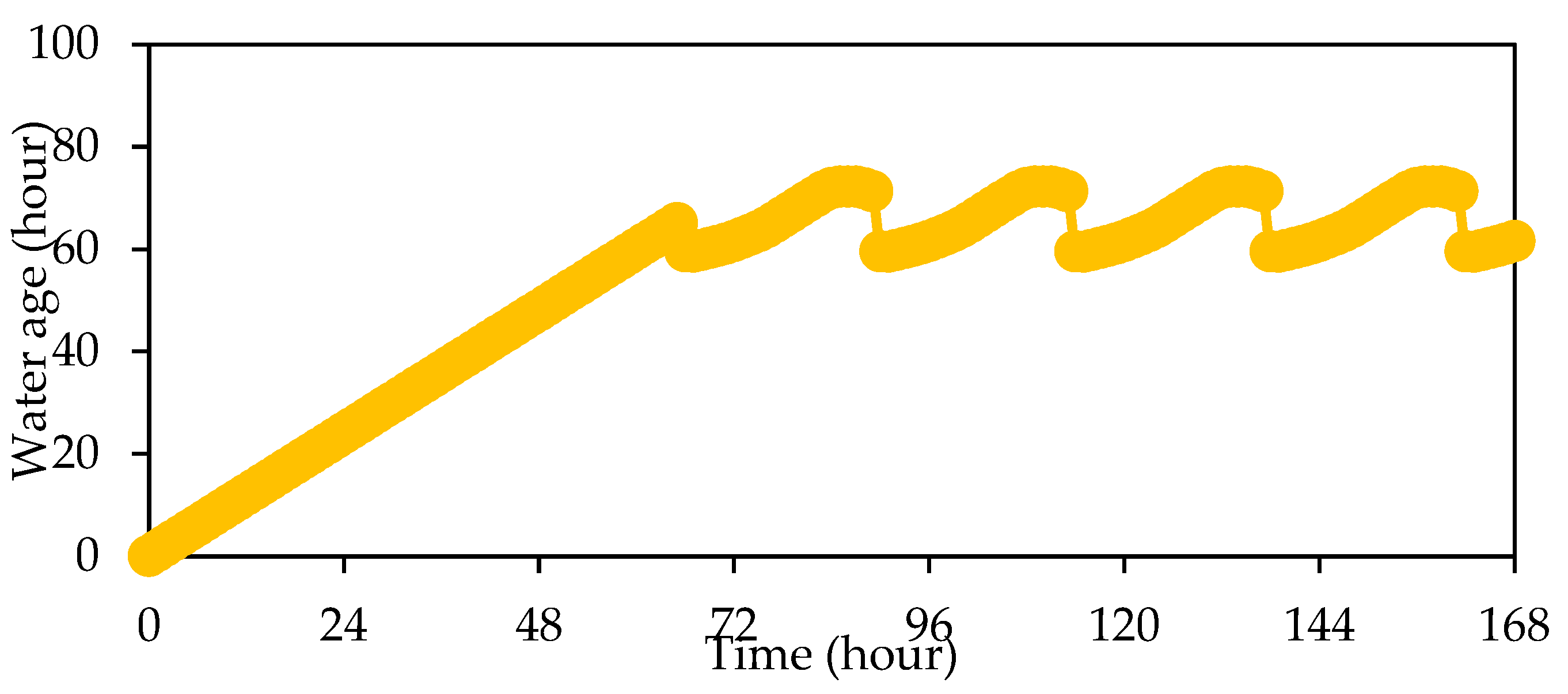
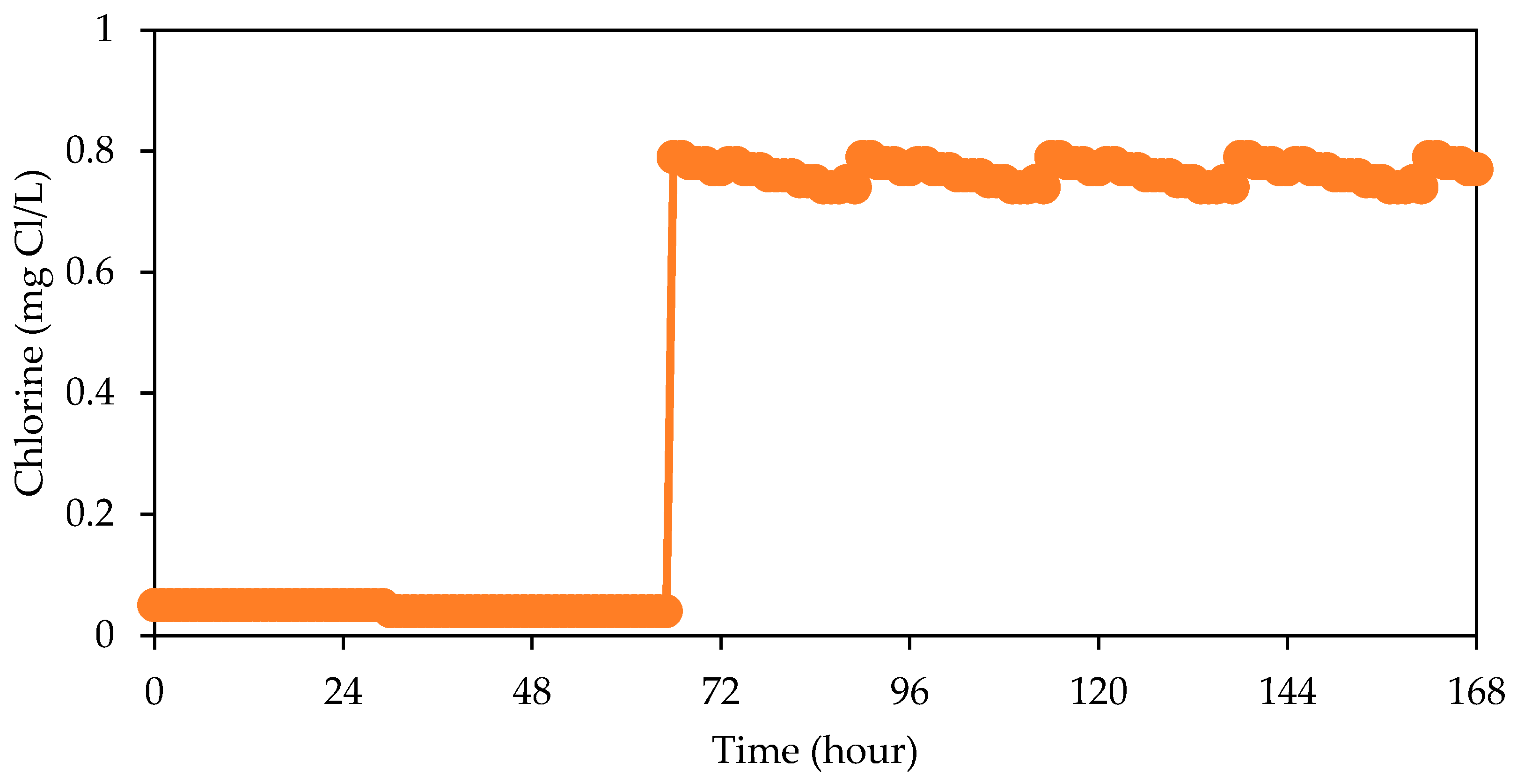
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Charalambous, B.; Liemberger, R. Dealing with the Complex Interrelation of Intermittent Supply and Water Losses; International Water Association: London, UK, 2017; Available online: https://www.iwapublishing.com/books/9781780407067/dealing-complex-interrelation-intermittent-supply-and-water-losses (accessed on 12 January 2019).
- Van den Berg, C.; Danilenko, A. The IBNET Water Supply and Sanitation Performance Blue Book; The World Bank: Washington, DC, USA, 2011; Available online: http://documents.worldbank.org/curated/en/420251468325154730/The-IBNET-water-supply-and-sanitation-performance-blue-book (accessed on 22 February 2019).
- World Health Organization; UNICEF. Global Water Supply and Sanitation Assessment 2000 Report; World Health Organization: Geneva, Switzerland; UNICEF: New York, NY, USA, 2000; Available online: https://www.who.int/water_sanitation_health/monitoring/jmp2000.pdf (accessed on 12 January 2019).
- Government of Uganda Ministry of Water and Environment. Water and Sanitation Sector Performance Report 2006; Government of Uganda: Kampala, Uganda, 2006. Available online: https://docs.google.com/file/d/0BwhPKU71ZwQDSDc3eTdXMlpLV1U/edit (accessed on 22 June 2019).
- Intermittent Water Supply—A Paradigm Shift is Imperative. Available online: https://iwa-network.org/intermittent-water-supply-a-paradigm-shift-is-imperative/ (accessed on 25 June 2019).
- Vairavamoorthy, K.; Gorantiwar, S.D.; Mohan, S. Intermittent water supply under water scarcity situations. Water Int. 2007, 32, 121–132. [Google Scholar] [CrossRef]
- Barwick, R.S.; Uzicanin, A.; Lareau, S.; Malakmadze, N.; Imnadze, P.; Iosava, M.; Ninashvili, N.; Wilson, M.; Hightower, A.W.; Johnston, S.; et al. Outbreak of Amebiasis in Tbilisi, republic of Georgia, 1998. Am. Trop. Med. Hyg. 2002, 67, 623–631. [Google Scholar] [CrossRef]
- Kumpel, E.; Nelson, K.L. Mechanisms affecting water quality in an intermittent piped water supply. Environ. Sci. Technol. 2014, 48, 2766–2775. [Google Scholar] [CrossRef] [PubMed]
- Mutikanga, H.E.; Sharma, S.K.; Vairavamoorthy, K. Assessment of apparent losses in urban water systems. Water Environ. J. 2011, 25, 327–335. [Google Scholar] [CrossRef]
- Haward, G.; Pedley, S.; Tibatemwa, S. Quantitative microbial risk assessment to estimate health risks attributable to water supply: Can the technique be applied in developing countries with limited data? J. Water Health 2006, 4, 49–65. [Google Scholar] [CrossRef]
- IWA Connect. Available online: https://iwa-connect.org/group/intermittent-water-supply-iws/about?view=public (accessed on 26 June 2019).
- Klingel, P.; Nestmann, F. From intermittent to continuous water distribution: A proposed conceptual approach and a case study of Béni Abbès (Algeria). Urban Water J. 2014, 11, 240–251. [Google Scholar] [CrossRef]
- National Planning Authority (NPA), Government of Uganda. Uganda Vision 2040; NPA: Kampala, Uganda, 2013. Available online: https://www.greengrowthknowledge.org/sites/default/files/downloads/policy-database/UGANDA%29%20Vision%202040.pdf (accessed on 8 January 2019).
- The Republic of Uganda, Ministry of Water and Environment. Framework and Guidelines for Water Source Protection Volume 2: Guidelines for Protecting Water Sources for Piped Water Supply Systems; The Republic of Uganda Ministry of Water and Environment: Kampala, Uganda, 2013. Available online: https://www.mwe.go.ug/sites/default/files/library/Vol.%202%20-%20Guidelines%20for%20Protecting%20Piped%20Water%20Sources%20-%20FINAL.pdf (accessed on 10 January 2019).
- Ecuru, J.; Okumu, O.J.; Okurut, O.T. Monitoring residual chlorine decay and coliform contamination in water distribution network of Kampala, Uganda. Afr. J. Online 2011, 15, 167–173. [Google Scholar] [CrossRef]
- Rossman, L. EPANET 2 User’s Manual; Risk Reduction Engineering Laboratory, U.S. Environmental Protection Agency: Cincinnati, OH, USA, 2000.
- Uganda Bureau of Statistics Population and Censuses, Population Projections 2018. Available online: https://www.ubos.org/explore-statistics/20/ (accessed on 30 June 2019).
- The Republic of Uganda Ministry of water and environment. Water Supply Design Manual Second Edition; The Republic of Uganda Ministry of Water and Environment: Kampala, Uganda, 2013. Available online: https://www.mwe.go.ug/sites/default/files/library/Water%20Supply%20Design%20Manual%20v.v1.1.pdf (accessed on 12 January 2019).
- Kalibbala, H.M.; Nalubega, M.; Wahlberg, O.; Hultman, B. Performance Evaluation of Drinking Water Treatment Plants in Kampala—Case of Ggaba II. In Proceedings of the 32nd WEDC International Conference, Colombo, Sri Lanka, 13–17 November 2006; pp. 373–376. [Google Scholar]
- Environmental Protection Agency. Quick Guide to Drinking Water Sample Collection; Environmental Protection Agency: Washington, DC, USA, 2016. Available online: https://www.epa.gov/sites/production/files/2015-11/documents/drinking_water_sample_collection.pdf (accessed on 22 February 2019).
- Blokker, M.; Furnass, W.; Machell, J.; Mounce, S.; Schaap, P.G.; Boxall, J. Boxall. Relating Water Quality and age in drinking water distribution systems using self-organising maps. Environments 2016, 3, 10. [Google Scholar] [CrossRef]
- Minnesota Pollution Control Agency. Turbidity: Description, Impact on Water Quality, Sources, Measures—A General Overview; Minnesota Pollution Control Agency: St Paul, MN, USA, 2008. Available online: https://www.pca.state.mn.us/sites/default/files/wq-iw3-21.pdf (accessed on 20 February 2019).
- World Health Organization. pH in Drinking-Water Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Cotruvo, J.A. 2017 WHO guidelines for drinking water quality: First addendum to the fourth edition. J. Am. Water Works Assoc. 2017, 109, 44–51. [Google Scholar] [CrossRef]
- Ainsworth, R. Safe Piped Water: Managing Microbial Water Quality in Piped Distribution Systems; World Health Organization: Geneva, Switzerland, 2004; Available online: https://apps.who.int/iris/handle/10665/42785 (accessed on 12 January 2019).
- Geldreich, E. Microbial Quality of Water Supply in Distribution Systems; CRC Lewis Publishers: Boca Raton, FL, USA, 1996. [Google Scholar]
- William, D.H.; Robert, S.S.; Elston, S.; Sharon, C.M.; Marjorie, G.B.; Barbara, G.S.; Susan, N.P. Intestinal effects of sulphate in drinking water on normal human subjects. Dig. Dis. Sci. 1997, 42, 1055–1061. [Google Scholar] [CrossRef]
- Prest, E.I.; Hammes, F.; van Loosdrecht, M.C.; Vrouwenvelder, J.S. Biological stability of drinking water: Controlling factors, methods, and challenges. Front. Microbiol. 2016, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Ammonia in drinking-Water Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2001; Available online: https://www.who.int/water_sanitation_health/water-quality/guidelines/chemicals/ammonia.pdf?ua=1 (accessed on 12 January 2019).
- World Health Organization. Nitrate and Nitrite in Drinking-Water Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2016; Available online: https://apps.who.int/iris/handle/10665/75380 (accessed on 12 January 2019).
- Ministry of the Environment Government of Japan. Available online: https://www.env.go.jp/en/index.html (accessed on 12 January 2019).
- Wang, H.; Masters, S.; Hong, Y.; Stallings, J.; Falkinham, J.O., III; Edwards, M.A.; Pruden, A. Effect of disinfectant, water age, and pipe material on occurrence and persistence of Legionella, mycobacteria, Pseudomonas aeruginosa, and two amoebas. Environ. Sci. Technol. 2012, 46, 11566–11574. [Google Scholar] [CrossRef] [PubMed]
- Tadeusz, J. Swierzawski. Piping Handbook 7th Edition, Flow and fluids Chapter B8; MCGRAW-HILL: New York, NY, USA, 2000; Available online: https://engineeringdocu.files.wordpress.com/2011/12/piping-handbook.pdf (accessed on 23 February 2019).
- Ozdemir, O.N.; Ucak, A. Simulation of chlorine decay in drinking-water distribution systems. J. Environ. Eng. 2002, 128, 31–39. [Google Scholar] [CrossRef]
- Vieira, P.; Coelho, S.T.; Loureiro, D. Accounting for the influence of initial chlorine concentration, TOC, iron and temperature when modeling chlorine decay in water supply. J. Water Supply Res. Technol. AQUA 2004, 53, 453–467. [Google Scholar] [CrossRef]
- Al-Jasser, A.O. Chlorine Decay in Drinking-Water Transmission and distribution systems: Pipe service age effect. Water Res. 2007, 41, 387–396. [Google Scholar] [CrossRef]
- Housing and Building National Research Centre. Egyptian Code for Design and Implementation of Pipeline Networks for Drinking Water and Sanitation, 10th ed.; Housing and Building National Research Centre: Cairo, Egypt, 2007. [Google Scholar]
- Helbling, D.E.; Van Briesen, J.M. Modeling residual chlorine response to a microbial contamination event in drinking water distribution systems. J. Environ. Eng. 2009, 135, 918–927. [Google Scholar] [CrossRef]
- The Republic of Uganda, Ministry of Water and Environment. Uganda Water and Environment Sector Performance Report 2017; The Republic of Uganda Ministry of Water and Environment: Kampala, Uganda, 2017. Available online: https://www.mwe.go.ug/sites/default/files/library/SPR%202017%20Final.pdf (accessed on 20 February 2020).
- LeChevallier, M.W.; Babcock, T.M.; Lee, R.G. Examination and characterization of distribution system biofilms. Appl. Environ. Microbiol. 1987, 53, 2714–2724. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Percival, S.; Walker, J.T. Contamination potential of biofilms in water distribution systems. Water Sci. Technol. Water Supply 2002, 2, 271–280. [Google Scholar] [CrossRef]
- Norton, C.D.; LeChevallier, M.W. Chloramination: Its effect on distribution system water quality. Am. Water Works Assoc. J. 1997, 89, 66–77. [Google Scholar] [CrossRef]
- Kumpel, E.; Nelson, K.L. Intermittent water supply: Prevalence, practice, and microbial water quality. Environ. Sci. Technol. 2015, 50, 542–553. [Google Scholar] [CrossRef]
- Xin, K.; Zhou, X.; Qian, H.; Yan, H.; Tao, T. Chlorine-age based booster chlorination optimization in water distribution network considering the uncertainty of residuals. Water Supply 2018, 19, 796–807. [Google Scholar] [CrossRef]
- Tryby, M.E.; Boccelli, D.L.; Uber, J.G.; Rossman, L.A. Facility location model for booster disinfection of water supply networks. J. Water Resourc. Plan. Manag. 2002, 128, 322–333. [Google Scholar] [CrossRef]
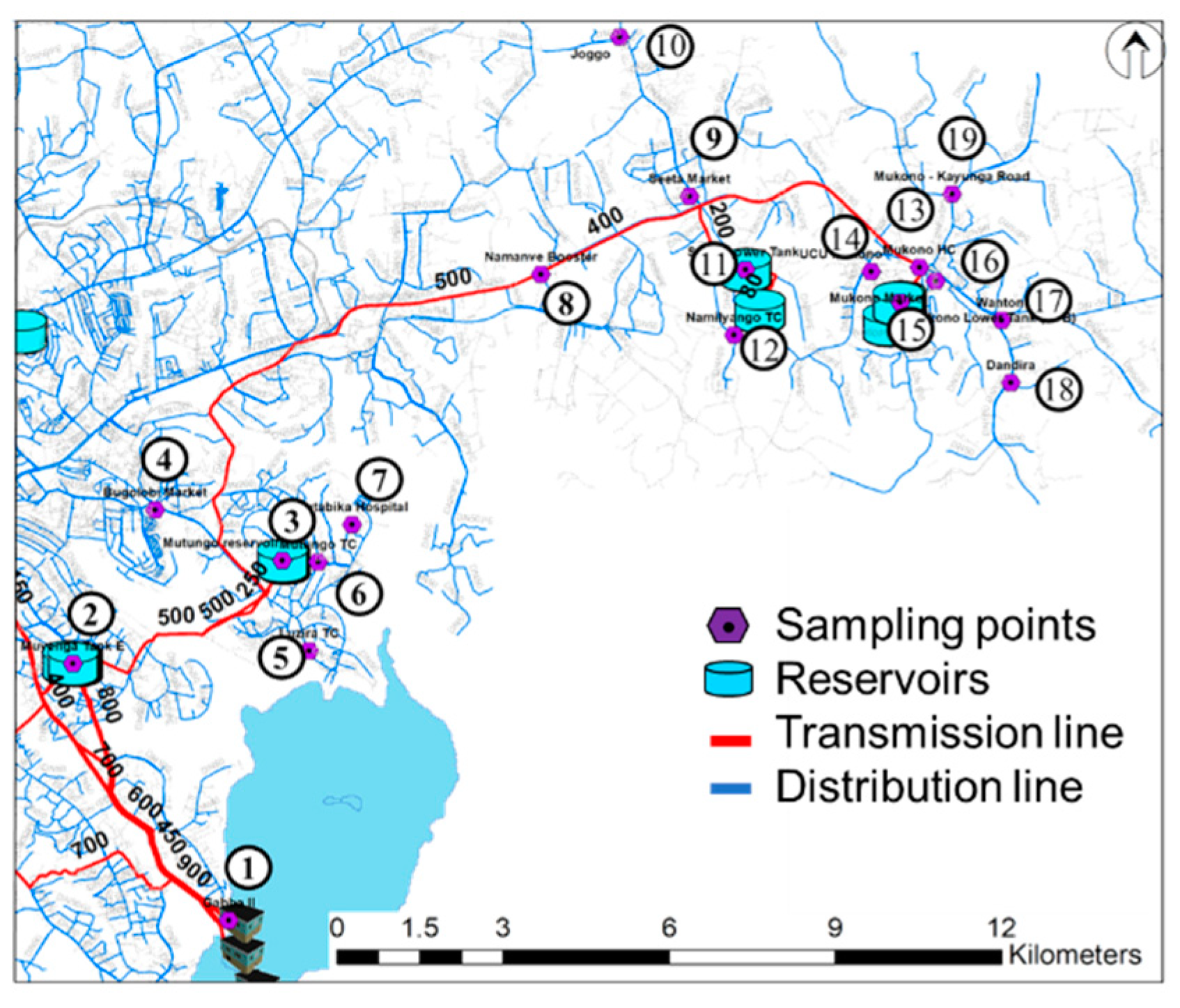
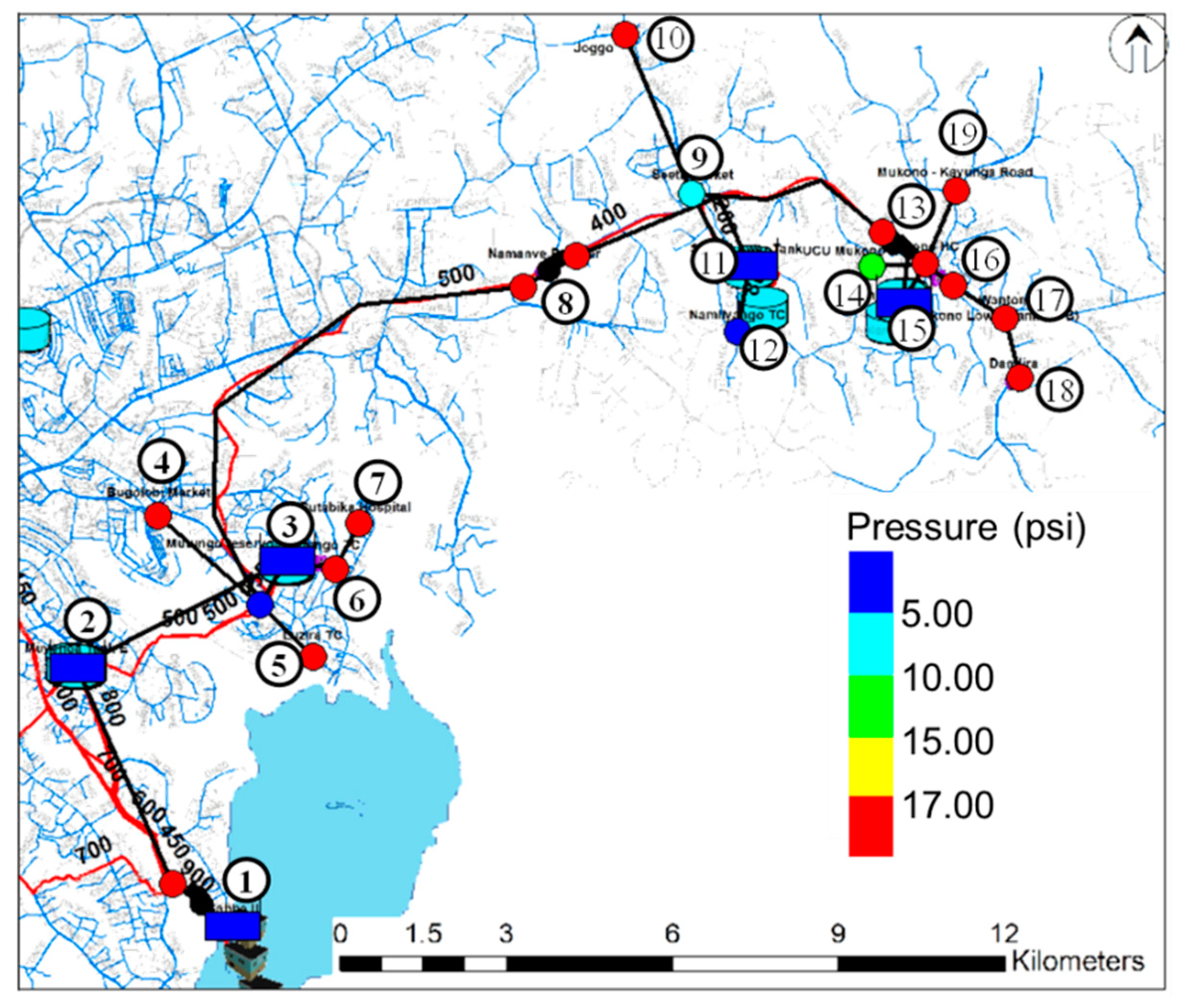
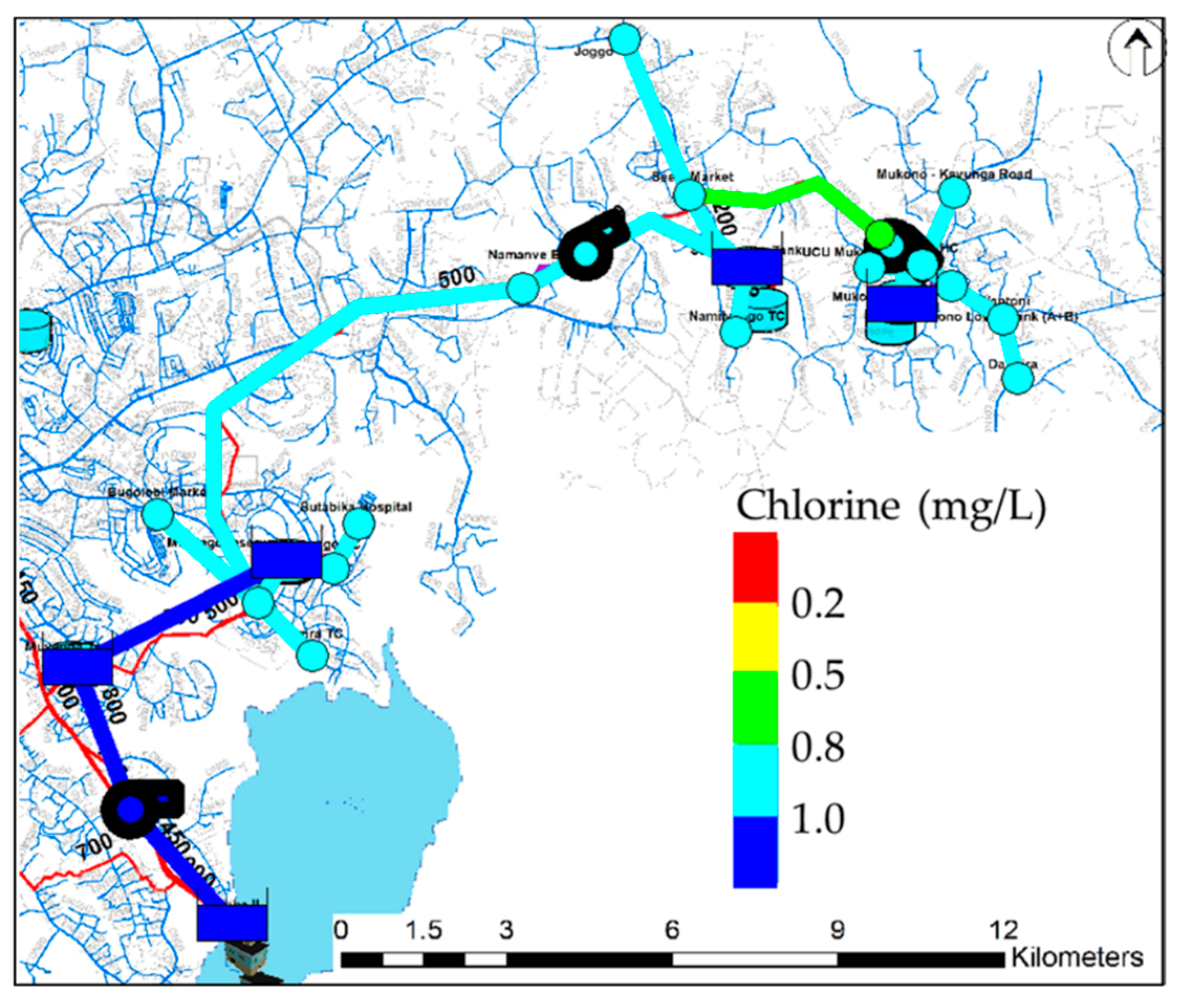
| Number | Name of Place | Elevation (m) | Number | Name of Place | Elevation (m) |
|---|---|---|---|---|---|
| 1 | Ggaba II | 1126 | 11 | Seeta tank lower | 1170 |
| 2 | Muyunga Tank E | 1232 | 12 | Namilyango Trading center | 1196 |
| 3 | Mutungo reserver | 1228 | 13 | Mukono Health Center | 1168 |
| 4 | Bugorobi Market | 1172 | 14 | Mukono UCU | 1256 |
| 5 | Luzira Trading Center | 1146 | 15 | Mukono tank A+B | 1164 |
| 6 | Mutungo Trading center | 1168 | 16 | Mukono Market | 1240 |
| 7 | Butabika Hospital | 1168 | 17 | Mukono Wantoni | 1176 |
| 8 | Namanve Booster | 1134 | 18 | Mukono Dandira | 1160 |
| 9 | Seeta market | 1182 | 19 | Mukono Kayunga Road | 1172 |
| 10 | Bukerere Joggo | 1202 |
| Measurement Items | Measurement Devices |
|---|---|
| E. coli, Total coliform | Compact Dry EC Nissui for Coliform and E. coli |
| Free chlorine, COD, Ammonia, Nitrate, Nitrite, Sulphate, Turbidity | Photometer PF-12Plus and NANOCOLOR tube test |
| Measurement Items | Measurement Standard | Range of Photometer |
|---|---|---|
| Free chlorine | 0.5 mg/L (0.2–1.0) [24] | - |
| E. coli | 0 (CFU/100mL) [24] | - |
| Total coliform | 0 (CFU/100mL) [24] | - |
| COD | ≤5 mg/L [31] | 3 to 150 mg/L |
| Ammonia | ≤1.5 mg/L [24] | 0.04 to 2.30 mg/L |
| Nitrite | ≤3 mg/L [24] | 0.3 to 22.0 mg/L |
| Nitrate | ≤50 mg/L [24] | 0.1 to 4.0 mg/L |
| Sulphate | ≤250 mg/L [24] | 40 to 400 mg/L |
| Turbidity | ≤5 NTU [24] | 1 to 1000 NTU |
| pH | 6 ≤ pH ≤ 9 [24] | - |
| Water age | ≤5.7 days (136.8 h) [32] | - |
| Water pressure | ≥17 psi [8] | - |
| Pipe Name (from node No. x to y) | Pipe Length (m) | Pipe Diameter (mm) | Pipe Roughness |
|---|---|---|---|
| 1 to 2 | 5260 | 800 | 140 |
| 2 to 3 | 4210 | 500 | 140 |
| 3 to 4 | 2700 | 100 | 140 |
| 3 to 5 | 1740 | 100 | 140 |
| 3 to 6 | 660 | 100 | 140 |
| 6 to 7 | 1340 | 100 | 140 |
| 3 to 8 | 11700 | 500 | 140 |
| 8 to 11 | 4710 | 400 | 140 |
| 11 to 9 | 1680 | 200 | 140 |
| 11 to 12 | 1230 | 100 | 140 |
| 9 to 10 | 3260 | 100 | 140 |
| 9 to15 | 6920 | 400 | 140 |
| 15 to 13 | 1220 | 100 | 140 |
| 15 to 14 | 710 | 100 | 140 |
| 15 to 16 | 1530 | 100 | 140 |
| 16 to 17 | 1100 | 100 | 140 |
| 17 to 18 | 860 | 100 | 140 |
| 13 to 16 | 1240 | 100 | 140 |
| 13 to 19 | 1100 | 100 | 140 |
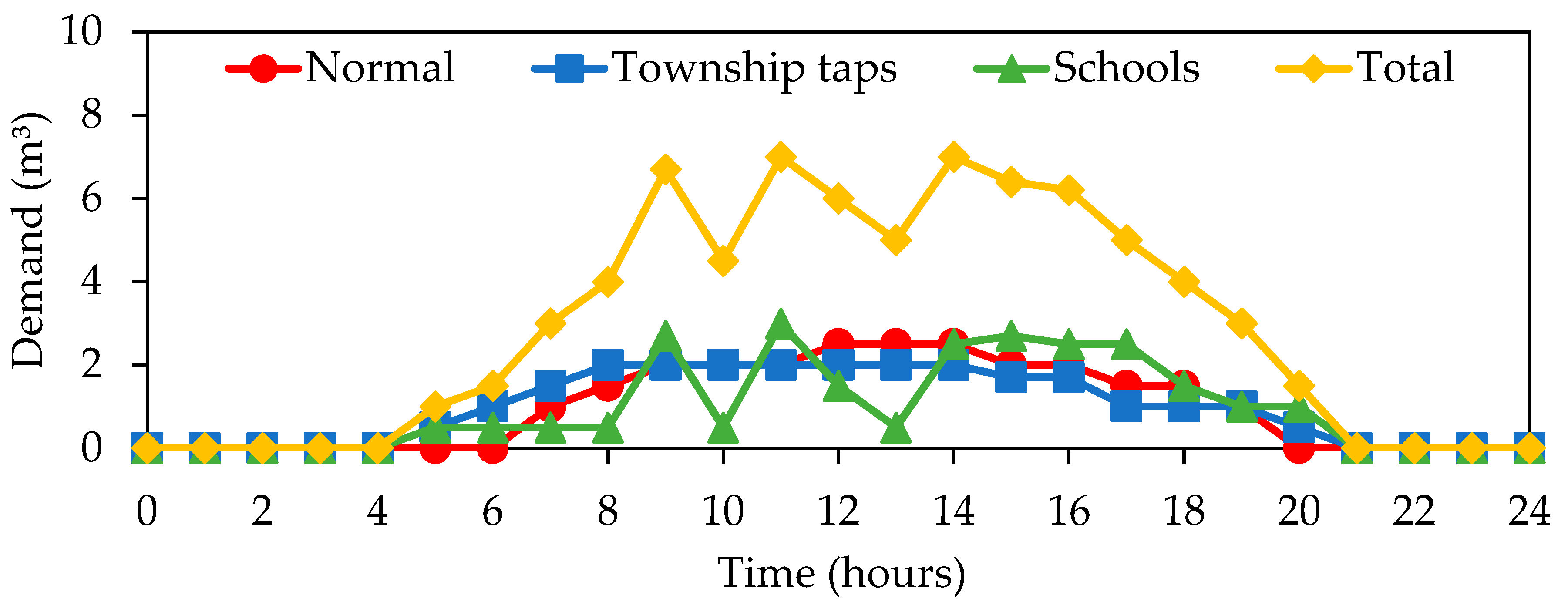
| No. | Pattern | No. | Pattern |
|---|---|---|---|
| 1 | - | 11 | - |
| 2 | - | 12 | Township taps |
| 3 | - | 13 | Normal |
| 4 | Township taps | 14 | School |
| 5 | Township taps | 15 | - |
| 6 | Township taps | 16 | Township taps |
| 7 | Township taps | 17 | Normal |
| 8 | - | 18 | Normal |
| 9 | Township taps | 19 | Normal |
| 10 | Normal |
| No. | Ammonia (mg N/L) | Nitrite (mg N/L) | Nitrate (mg N/L) | Sulphate (1st, 2nd) (mg S/L) | Turbidity (NTU) |
|---|---|---|---|---|---|
| 1 | - | - | - | - | - |
| 2 | - | - | - | - | - |
| 3 | - | - | - | - | - |
| 4 | - | - | - | 60, 40 | - |
| 5 | - | - | - | - | - |
| 6 | - | - | - | - | - |
| 7 | - | - | - | - | - |
| 8 | - | - | - | 54, 40 | - |
| 9 | - | - | - | - | - |
| 10 | - | - | - | 46, 47 | - |
| 11 | - | - | - | 48, 40 | - |
| 12 | - | - | - | - | - |
| 13 | - | - | - | - | - |
| 14 | - | - | - | - | - |
| 15 | - | - | - | - | - |
| 16 | - | - | - | 40, 48 | - |
| 17 | - | - | - | 73, 40 | - |
| 18 | - | - | - | - | - |
| 19 | - | - | - | 42, 46 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakomoto, T.; Lutaaya, M.; Abraham, E. Managing Water Quality in Intermittent Supply Systems: The Case of Mukono Town, Uganda. Water 2020, 12, 806. https://doi.org/10.3390/w12030806
Sakomoto T, Lutaaya M, Abraham E. Managing Water Quality in Intermittent Supply Systems: The Case of Mukono Town, Uganda. Water. 2020; 12(3):806. https://doi.org/10.3390/w12030806
Chicago/Turabian StyleSakomoto, Takuya, Mahmood Lutaaya, and Edo Abraham. 2020. "Managing Water Quality in Intermittent Supply Systems: The Case of Mukono Town, Uganda" Water 12, no. 3: 806. https://doi.org/10.3390/w12030806
APA StyleSakomoto, T., Lutaaya, M., & Abraham, E. (2020). Managing Water Quality in Intermittent Supply Systems: The Case of Mukono Town, Uganda. Water, 12(3), 806. https://doi.org/10.3390/w12030806






