Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media
Abstract
1. Introduction
1.1. Challenges and Opportunities Associated with Produced Water
1.2. Research Objective
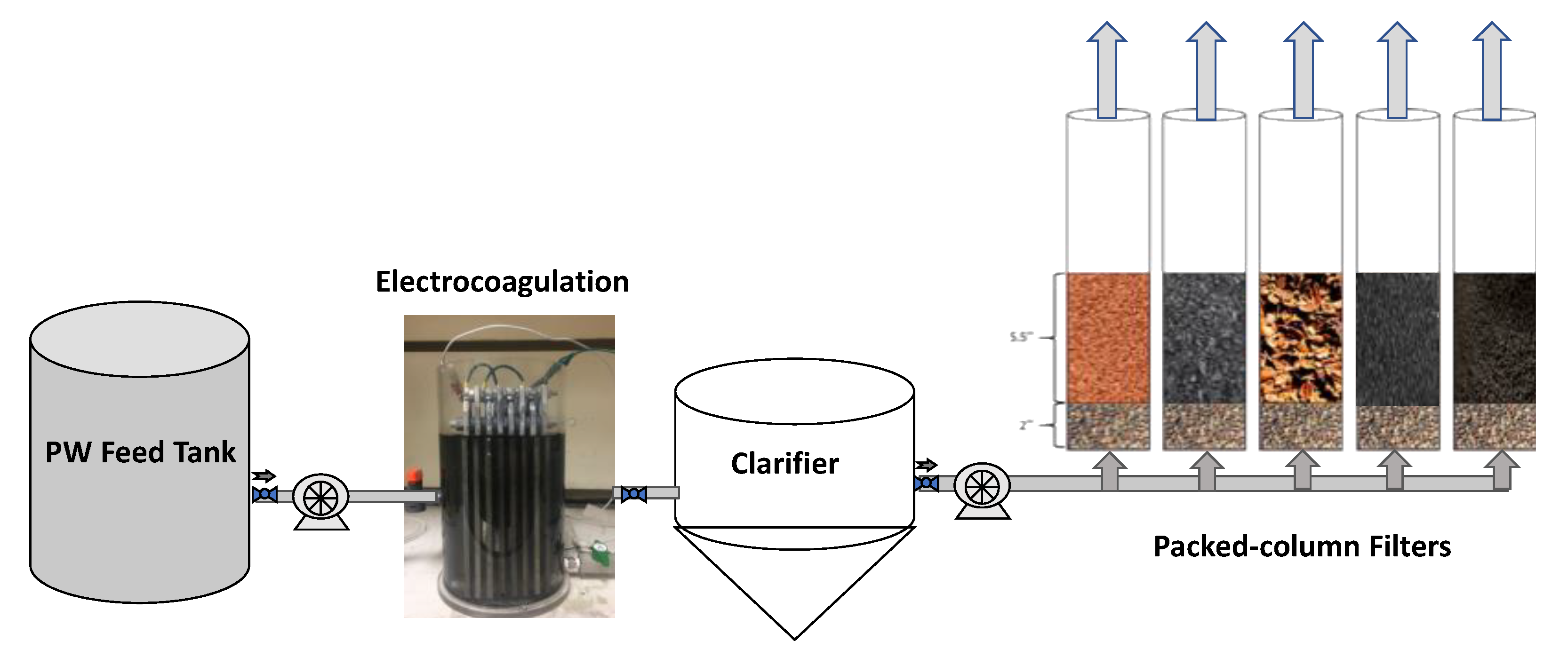
2. Materials and Methods
2.1. Produced Water
2.2. Produced Water Treatment
2.2.1. Chemical coagulation (CC)
2.2.2. Electrocoagulation (EC)
2.2.3. Filtration
2.3. Continuous-Flow Experiments
3. Results
3.1. Chemical Coagulation (CC)
3.2. Electrocoagulation (EC)
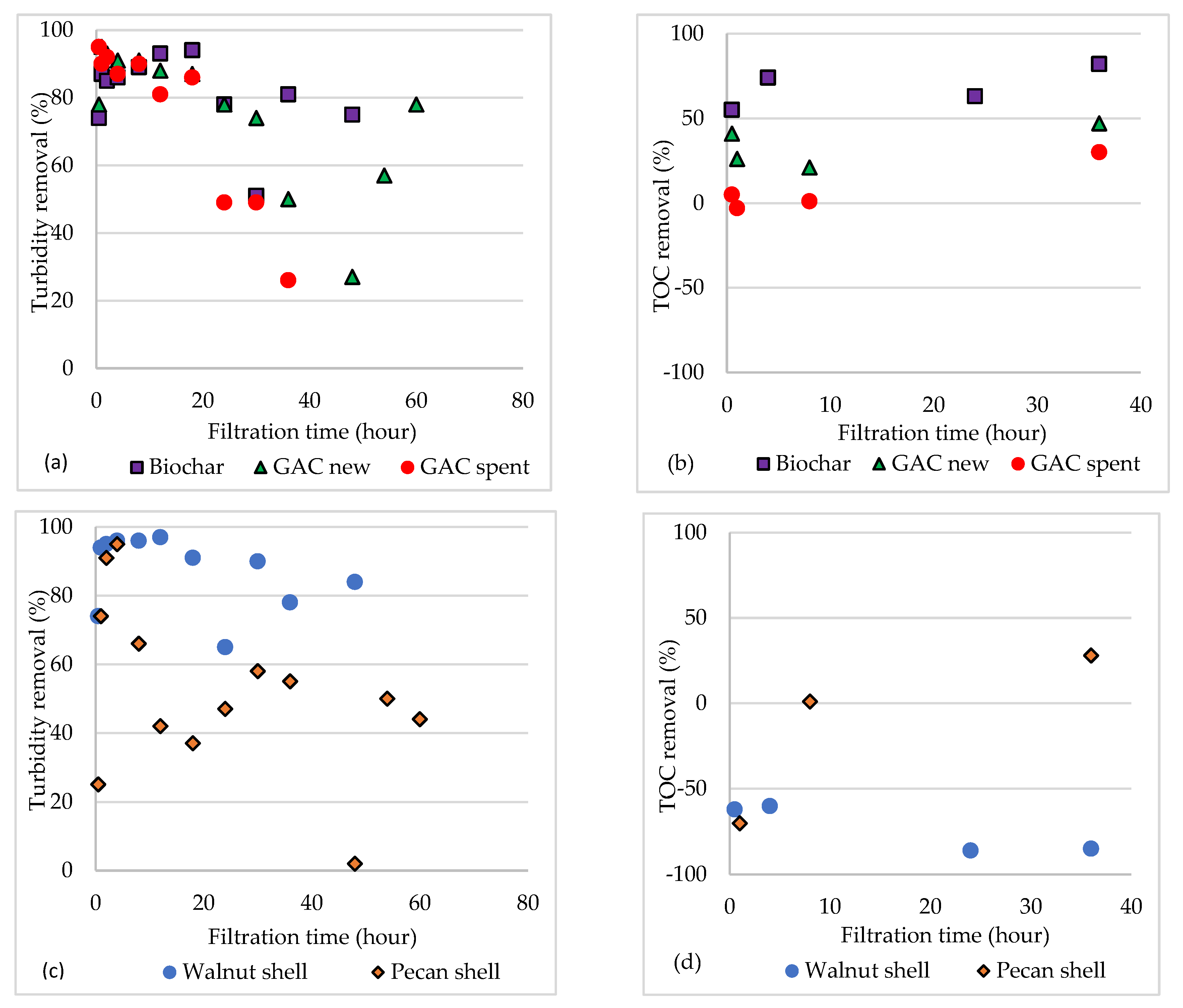
3.3. Continuous-Flow Filtration
3.3.1. New and Spent GAC
3.3.2. Walnut and Pecan Shell
3.3.3. Biochar
3.3.4. Removal of metals
3.4. Potential for Onsite Produced Water Reuse
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Veil, J.U.S. Produced Water Volumes and Management Practices in 2012; Veil Environmental, LLC: Annapolis, MD, USA, 2015. [Google Scholar]
- Arthur, J.D.; Dillon, L.W.; Frazan, D.J.; Hochheiser, W.H. Management of Produced Water from Oil and Gas Wells; National Petroleum Council: Washington, DC, USA, 2011. [Google Scholar]
- Jiménez, S.; Micó, M.M.; Arnaldos, M.; Medina, F.; Contreras, S. State of the art of produced water treatment. Chemosphere 2018, 192, 186–208. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A.; Engle, M.; Dungan, B.; Holguin, F.O.; Xu, P.; Carroll, K.C. Volatile-organic molecular characterization of shale-oil produced water from the Permian Basin. Chemosphere 2016, 148, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Dahm, K.G.; Guerra, K.L.; Xu, P.; Drewes, J.E. Composite geochemical database for coalbed methane produced water quality in the Rocky Mountain region. Environ. Sci. Technol. 2011, 45, 7655–7663. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Drewes, J.E.; Heil, D. Beneficial use of co-produced water through membrane treatment: Technical-economic assessment. Desalination 2008, 225, 139–155. [Google Scholar] [CrossRef]
- Chaudhary, B.K.; Sabie, R.; Engle, M.A.; Xu, P.; Willman, S.; Carroll, K.C. Produced Water Quality Spatial Variability and Alternative-Source Water Analysis Applied to the Permian Basin, USA. Hydrogeol. J. 2020, 27, 2889–2905. [Google Scholar] [CrossRef]
- USEPA. U.S. Environmental Protection Agency. Study of Oil and Gas Extraction Wastewater Management Under the Clean Water Act. EPA-821-R19-001. Available online: https://www.epa.gov/sites/production/files/2019-05/documents/oil-and-gas-study_draft_05-2019.pdf (accessed on 29 January 2019).
- Hu, L.; Yu, J.; Luo, H.; Wang, H.; Xu, P.; Zhang, Y. Simultaneous recovery of ammonium, potassium and magnesium from produced water by struvite precipitation. Chem. Eng. J. 2020, 382, 123001. [Google Scholar] [CrossRef]
- Liden, T.; Santos, I.C.; Hildenbrand, Z.L.; Schug, K.A. Treatment modalities for the reuse of produced waste from oil and gas development. Sci. Total Environ. 2018, 643, 107–118. [Google Scholar] [CrossRef]
- Guglielmi, Y.; Cappa, F.; Avouac, J.P.; Henry, P.; Elsworth, D. Induced seismicity. Seismicity triggered by fluid injection-induced aseismic slip. Science 2015, 348, 1224–1226. [Google Scholar] [CrossRef]
- Scanlon, B.R.; Reedy, R.C.; Xu, P.; Engle, M.; Nicot, J.P.; Yoxtheimer, D.; Yang, Q.; Ikonnikova, S. Can we beneficially reuse produced water from oil and gas extraction in the U.S.? Sci. Total Environ. 2020, 717, 137085. [Google Scholar] [CrossRef]
- Reyes, F.R.; Engle, M.; Lixin, J.; Jacobs, M.A.; Konter, J.G. Hydrogeochemical controls on brackish groundwater and its suitability for use in hydraulic fracturing: The Dockum Aquifer, Midland Basin, Texas. Environ. Geosci. 2018, 25, 37–63. [Google Scholar] [CrossRef]
- Wang, M.; Wang, M.; Chen, D.; Gong, Q.; Yao, S.; Jiang, W.; Chen, Y. Evaluation of pre-treatment techniques for shale gas produced water to facilitate subsequent treatment stages. J. Environ. Chem. Eng. 2019, 7, 102878. [Google Scholar] [CrossRef]
- Dastgheib, S.A.; Knutson, C.; Yang, Y.; Salih, H.H. Treatment of produced water from an oilfield and selected coal mines in the Illinois Basin. Int. J. Greenh. Gas Control 2016, 54, 513–523. [Google Scholar] [CrossRef]
- Younker, J.M.; Walsh, M.E. Bench-scale investigation of an integrated adsorption–coagulation–dissolved air flotation process for produced water treatment. J. Environ. Chem. Eng. 2014, 2, 692–697. [Google Scholar] [CrossRef]
- Hakizimana, J.N.; Gourich, B.; Chafi, M.; Stiriba, Y.; Vial, C.; Drogui, P.; Naja, J. Electrocoagulation process in water treatment: A review of electrocoagulation modeling approaches. Desalination 2017, 404, 1–21. [Google Scholar] [CrossRef]
- Kuokkanen, V.; Kuokkanen, T.; Rämö, J.; Lassi, U. Recent Applications of Electrocoagulation in Treatment of Water and Wastewater—A Review. Green Sustain. Chem. 2013, 3, 89–121. [Google Scholar] [CrossRef]
- Valero, D.; Ortiz, J.M.; Expósito, E.; Montiel, V.; Aldaz, A. Electrocoagulation of a synthetic textile effluent powered by photovoltaic energy without batteries: Direct connection behaviour. Sol. Energy Mater. Sol. Cells 2008, 92, 291–297. [Google Scholar] [CrossRef]
- Jain, P.; Sharma, M.; Dureja, P.; Sarma, P.M.; Lal, B. Bioelectrochemical approaches for removal of sulfate, hydrocarbon and salinity from produced water. Chemosphere 2017, 166, 96–108. [Google Scholar] [CrossRef]
- Millar, G.J.; Lin, J.; Arshad, A.; Couperthwaite, S.J. Evaluation of electrocoagulation for the pre-treatment of coal seam water. J. Water Process Eng. 2014, 4, 166–178. [Google Scholar] [CrossRef]
- Zhao, S.; Huang, G.; Cheng, G.; Wang, Y.; Fu, H. Hardness, COD and turbidity removals from produced water by electrocoagulation pretreatment prior to Reverse Osmosis membranes. Desalination 2014, 344, 454–462. [Google Scholar] [CrossRef]
- CleanWave-FactSheet. CleanWaveSM Water Treatment Service. In Mobile Service for Produced and Flow Water; Halliburton: Houston, TX, USA, 2017. [Google Scholar]
- Mousa, I.E. Total petroleum hydrocarbon degradation by hybrid electrobiochemical reactor in oilfield produced water. Mar. Pollut. Bull. 2016, 109, 356–360. [Google Scholar] [CrossRef]
- Camarillo, M.K.; Domen, J.K.; Stringfellow, W.T. Physical-chemical evaluation of hydraulic fracturing chemicals in the context of produced water treatment. J. Environ. Manag. 2016, 183, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.; Cocke, D.; Das, K.; Guttula, M.; Tran, D.; Beckman, J. Treatment of produced water by electrocoagulation. In Proceedings of the PD Congress 2009: Proceedings of sessions and symposia held during TMS 2009 Annual Meeting & Exhibition, San Francisco, CA, USA, 15–19 February 2009; pp. 459–466. [Google Scholar]
- Ezechi, E.H.; Isa, M.H.; Kutty, S.R.M.; Yaqub, A. Boron removal from produced water using electrocoagulation. Process Saf. Environ. Prot. 2014, 92, 509–514. [Google Scholar] [CrossRef]
- Shamaei, L.; Khorshidi, B.; Perdicakis, B.; Sadrzadeh, M. Treatment of oil sands produced water using combined electrocoagulation and chemical coagulation techniques. Sci. Total Environ. 2018, 645, 560–572. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Du, X.; Carlson, K.H.; Robbins, C.A.; Tong, T. Effective treatment of shale oil and gas produced water by membrane distillation coupled with precipitative softening and walnut shell filtration. Desalination 2019, 454, 82–90. [Google Scholar] [CrossRef]
- Kusworo, T.D.; Aryanti, N.; Qudratun; Utomo, D.P. Oilfield produced water treatment to clean water using integrated activated carbon-bentonite adsorbent and double stages membrane process. Chem. Eng. J. 2018, 347, 462–471. [Google Scholar] [CrossRef]
- Hosny, R.; Fathy, M.; Ramzi, M.; Abdel Moghny, T.; Desouky, S.E.M.; Shama, S.A. Treatment of the oily produced water (OPW) using coagulant mixtures. Egypt. J. Pet. 2016, 25, 391–396. [Google Scholar] [CrossRef]
- Lin, L.; Jiang, W.; Xu, P. Comparative study on pharmaceuticals adsorption in reclaimed water desalination concentrate using biochar: Impact of salts and organic matter. Sci. Total Environ. 2017, 601, 857–864. [Google Scholar] [CrossRef]
- Chang, H.; Liu, B.; Yang, B.; Yang, X.; Guo, C.; He, Q.; Liang, S.; Chen, S.; Yang, P. An integrated coagulation-ultrafiltration-nanofiltration process for internal reuse of shale gas flowback and produced water. Sep. Purif. Technol. 2019, 211, 310–321. [Google Scholar] [CrossRef]
- Nadella, M.; Sharma, R.; Chellam, S. Fit-for-purpose treatment of produced water with iron and polymeric coagulant for reuse in hydraulic fracturing: Temperature effects on aggregation and high-rate sedimentation. Water Res. 2020, 170, 115330. [Google Scholar] [CrossRef]
- Cañizares, P.; Martínez, F.; Jiménez, C.; Sáez, C.; Rodrigo, M.A. Technical and economic comparison of conventional and electrochemical coagulation processes. J. Chem. Technol. Biotechnol. 2009, 84, 702–710. [Google Scholar] [CrossRef]
- Gu, Z.; Liao, Z.; Schulz, M.; Davis, J.R.; Baygents, J.C.; Farrell, J. Estimating Dosing Rates and Energy Consumption for Electrocoagulation Using Iron and Aluminum Electrodes. Ind. Eng. Chem. Res. 2009, 48, 3112–3117. [Google Scholar] [CrossRef]
- Picard, T.; Cathalifaud-Feuillade, G.; Mazet, M.; Vandensteendam, C. Cathodic dissolution in the electrocoagulation process using aluminium electrodes. J. Environ. Monit. 2000, 2, 77–80. [Google Scholar] [CrossRef] [PubMed]
- EPA, U.S. Granular Activated Carbon. Available online: https://iaspub.epa.gov/tdb/pages/treatment/treatmentOverview.do?treatmentProcessId=2074826383#content (accessed on 29 January 2019).
- Ahmad, M.; Lee, S.S.; Dou, X.; Mohan, D.; Sung, J.-K.; Yang, J.E.; Ok, Y.S. Effects of pyrolysis temperature on soybean stover-and peanut shell-derived biochar properties and TCE adsorption in water. Bioresour. Technol. 2012, 118, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Tong, X.J.; Li, J.Y.; Yuan, J.H.; Xu, R.K. Adsorption of Cu(II) by biochars generated from three crop straws. Chem. Eng. J. 2011, 172, 828–834. [Google Scholar] [CrossRef]
- Whitfield, S. Permian, Bakken Operators Face Produced Water Challenges. J. Pet. Technol. 2017, 69, 48–51. [Google Scholar] [CrossRef]



| Ref. | HRT (min) | Experimental Set up/Arrangement | Current Density (mA/cm2) | Produced Water Characteristics |
|---|---|---|---|---|
| [26] | 4 | Continuous flow goes 4 times through the reactor (each time 1 min), Al and Fe electrodes | 10 and 20 | Conductivity 150–160 mS/cm, COD 27 mg/L, 68% COD removal |
| 45 | Batch operation PW 400 mL, Al and Fe electrodes | 13.9 | Conductivity 160–167 mS/cm, COD 32 mg/L, 67% COD removal | |
| [22] | 30 | Continuous flow, pilot scale PW 80 L Fe electrodes | 5.56 | COD 280 mg/L, Turbidity 135 NTU; Max. turbidity removal 93.8% and 67% COD removal |
| [21] | 1 | Continuous flow at 1.14 L/min Al electrodes Coal seam PW | 0.8 | Turbidity 8.5 NTU, DOC 6.4 mg/L; 54% DOC removal Significant removal in scale formation species |
| [27] | 15–90 | Batch operation PW 500 mL Aluminum electrodes | 1.2–3.6 | TDS 15 g/L, boron 10mg/L 15 min removes 60% boron; 90 min removes 98% boron |
| [28] | 30–90 | Batch operation, Volume of oil sands PW 1.5 L Al and Fe electrodes; AlCl3 added | 6–30 | TDS 1.7 g/L TOC 475–720 mg/L 39% TOC removal |
| Parameter | Unit | Value |
|---|---|---|
| pH | 7.30 ± 0.21 | |
| Alkalinity | mg/L as CaCO3 | 2345 ± 329 |
| Electrical conductivity | mS/cm | 201.2 ± 13.3 |
| Total dissolved solids | g/L | 129.3 ± 8.5 |
| Total organic carbon | mg/L | 83.1 ± 30.8 |
| Total phosphorus | mg/L | <0.1 |
| Turbidity | NTU | 53.4 ± 5.0 |
| Ammonium | mg/L | 655 ± 77 |
| Arsenic | mg/L | 1.1 ± 0.0 |
| Barium | mg/L | 1.0 ± 0.0 |
| Bromide | mg/L | 591 ± 16 |
| Calcium | mg/L | 4247 ± 752 |
| Chloride | mg/L | 65,800 ± 1600 |
| Iron | mg/L | 11 ± 9 |
| Lithium | mg/L | 18.8 ± 0.3 |
| Magnesium | mg/L | 727 ± 54 |
| Manganese | mg/L | 0.66 ± 0.02 |
| Nickel | mg/L | 0.02 ± 0.004 |
| Potassium | mg/L | 805 ± 230 |
| Silica | mg/L | 32 ± 2 |
| Sodium | mg/L | 42,720 ± 2093 |
| Strontium | mg/L | 257 ± 20 |
| Sulfate | mg/L | 1010 ± 9 |
| Water Sample | Time (Hours) | As (mg/L) | Ba (mg/L) | Fe (mg/L) | Ca (mg/L) | Mg (mg/L) | SiO2 (mg/L) | Na (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Filter influent | 0 | 1.12 | 1.03 | 13.4 | 3799 | 529 | 34.7 | 33,190 |
| Biochar | 0.25 | 0.98 | 2.18 | ND | 2943 | 448 | 14.3 | 26,840 |
| 0.5 | 1.04 | 1.25 | ND | 3682 | 527 | 13.2 | 33,840 | |
| 36 | 1.04 | 0.95 | ND | 3907 | 539 | 18.4 | 34,840 | |
| Walnut | 0.25 | 1.03 | 1.29 | ND | 3807 | 555 | 16.9 | 33,990 |
| 0.5 | 1.15 | 1.01 | ND | 3837 | 550 | 13.7 | 34,310 | |
| 36 | 1.01 | 0.94 | 0.3 | 3933 | 553 | 17.9 | 35,140 | |
| MDL | 0.1 | 0.001 | 0.0287 | 0.016 | 0.24 | 0.015 | 0.0483 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez, A.Z.; Wang, H.; Hu, L.; Zhang, Y.; Xu, P. Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media. Water 2020, 12, 770. https://doi.org/10.3390/w12030770
Rodriguez AZ, Wang H, Hu L, Zhang Y, Xu P. Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media. Water. 2020; 12(3):770. https://doi.org/10.3390/w12030770
Chicago/Turabian StyleRodriguez, Alfredo Zendejas, Huiyao Wang, Lei Hu, Yanyan Zhang, and Pei Xu. 2020. "Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media" Water 12, no. 3: 770. https://doi.org/10.3390/w12030770
APA StyleRodriguez, A. Z., Wang, H., Hu, L., Zhang, Y., & Xu, P. (2020). Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media. Water, 12(3), 770. https://doi.org/10.3390/w12030770









