Recent Developments and Future Challenges of Hydrogels as Draw Solutes in Forward Osmosis Process
Abstract
1. Introduction
2. Critical Parameters Related to Hydrogels as Draw Solutes in FO Process
2.1. Factors Affects the Water Flux
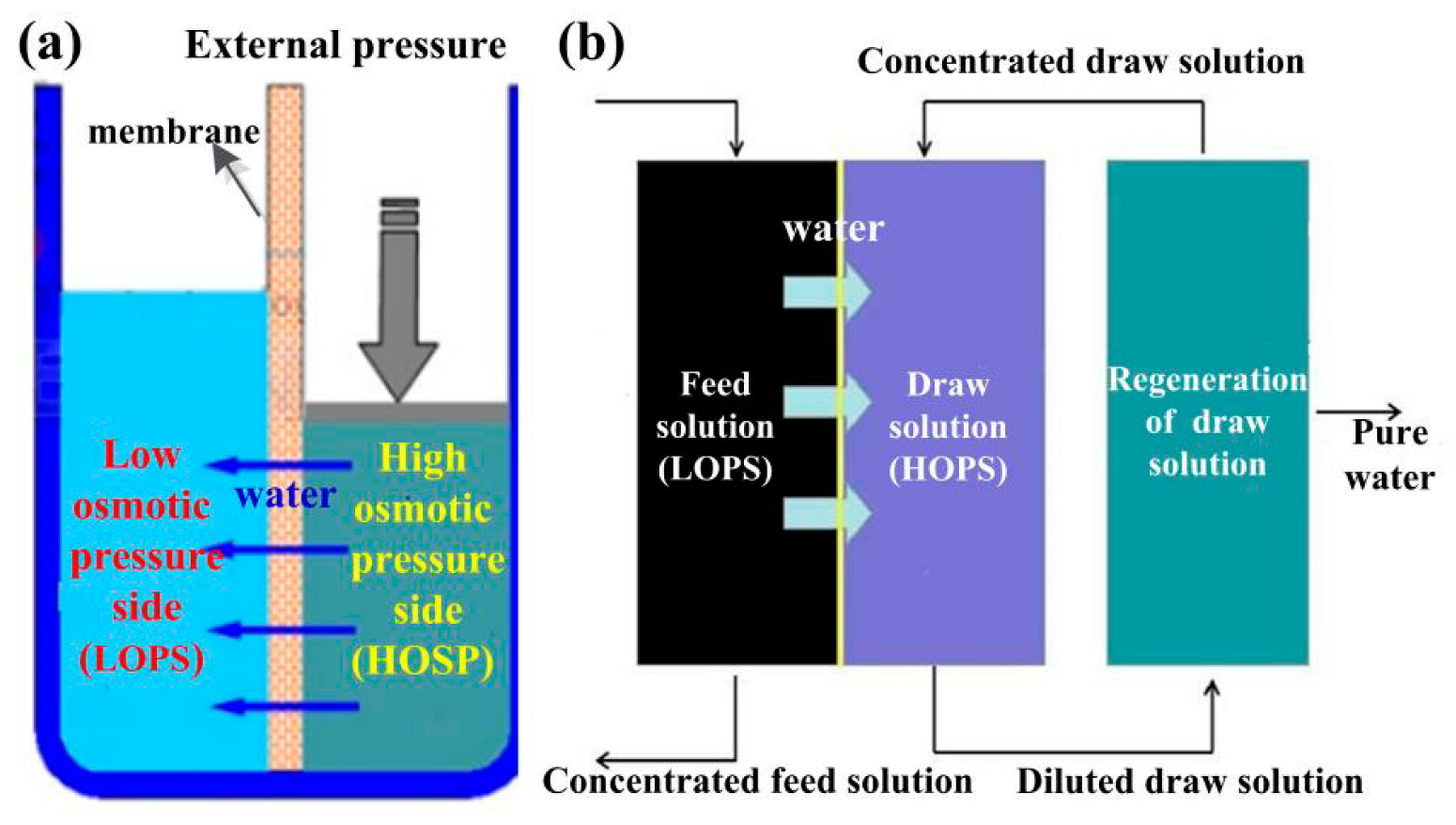
2.1.1. Osmotic Pressure
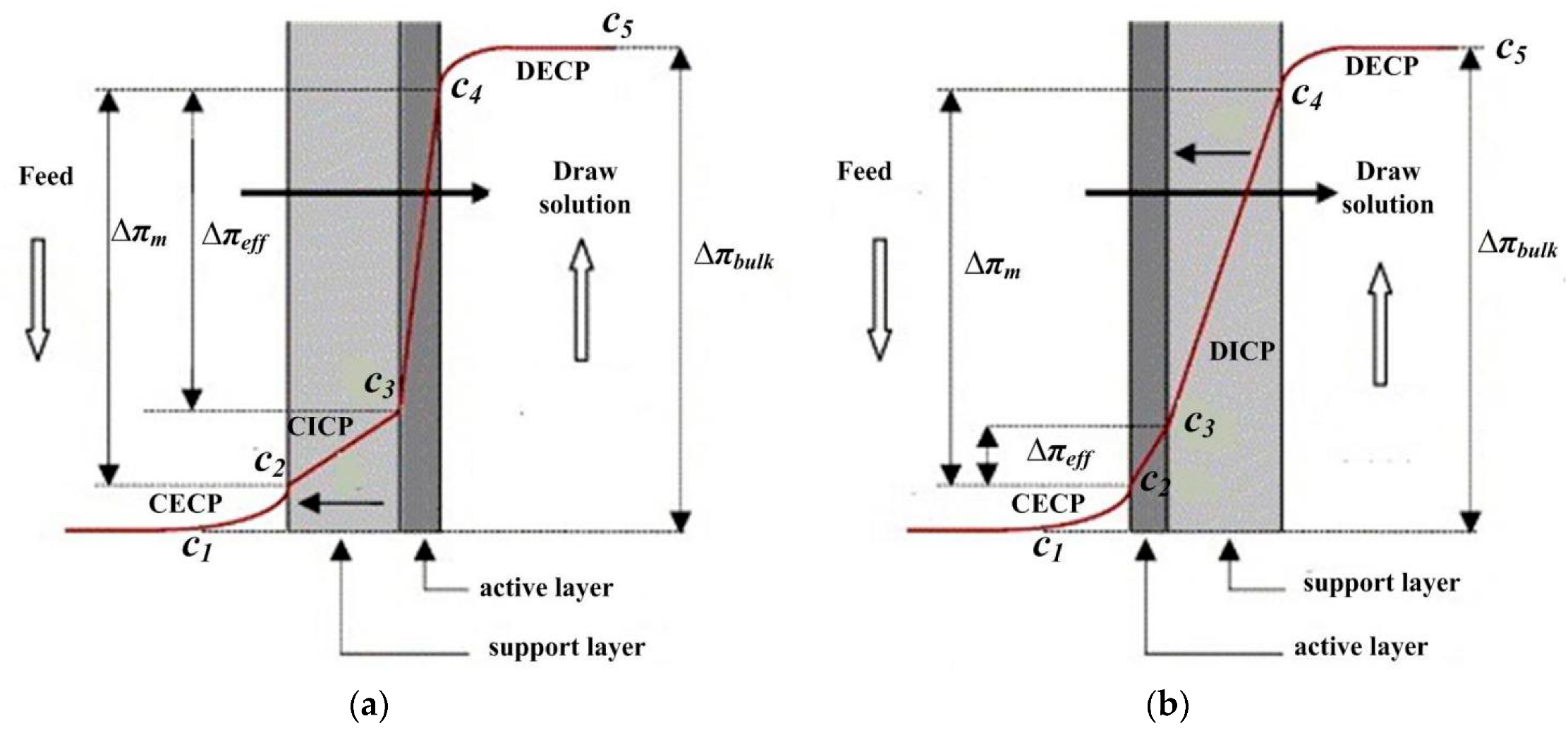
2.1.2. Concentration Polarization
2.2. Reverse Solute Diffusion
2.3. Regeneration of Draw Solutes
2.4. Operation Cost
3. Major Problems Associated with Hydrogels as Draw Solutes in FO Process
4. Recent Development of Hydrogels as Draw Solutes in FO Process
4.1. Enhancing Water Flux by Changing Monomer Composition of Hydrogels
4.2. Enhancing Water Flux by Reducing External Concentration Polarization
4.2.1. Reducing External Concentration Polarization by Introducing Composite Materials
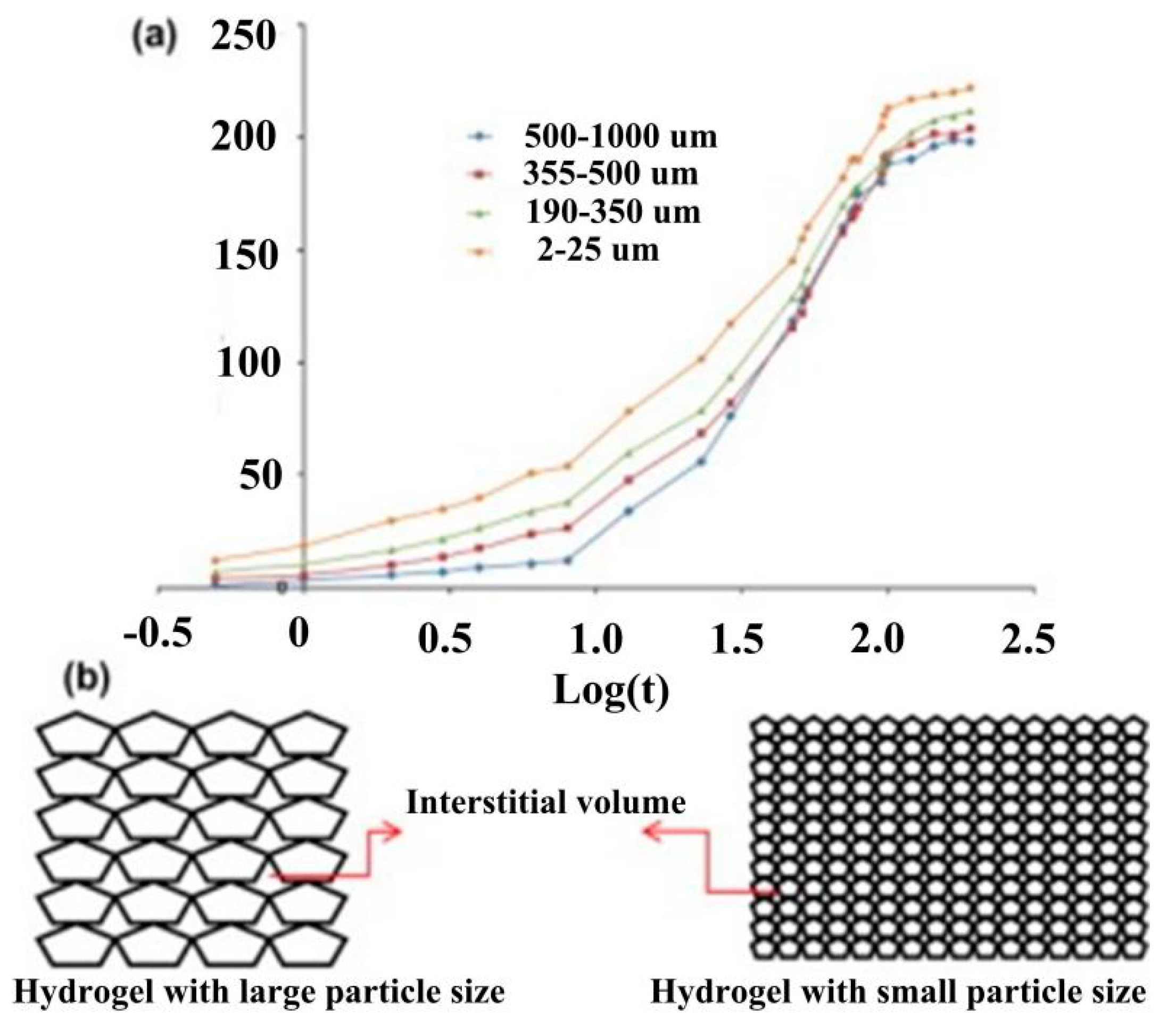
4.2.2. Reducing External Concentration Polarization by Reducing Hydrogel Size
4.3. Various Methods to Realize the Regeneration of Draw Solutes
4.3.1. Enhancing Dewatering Rate by Changing the Network Structure of Hydrogel
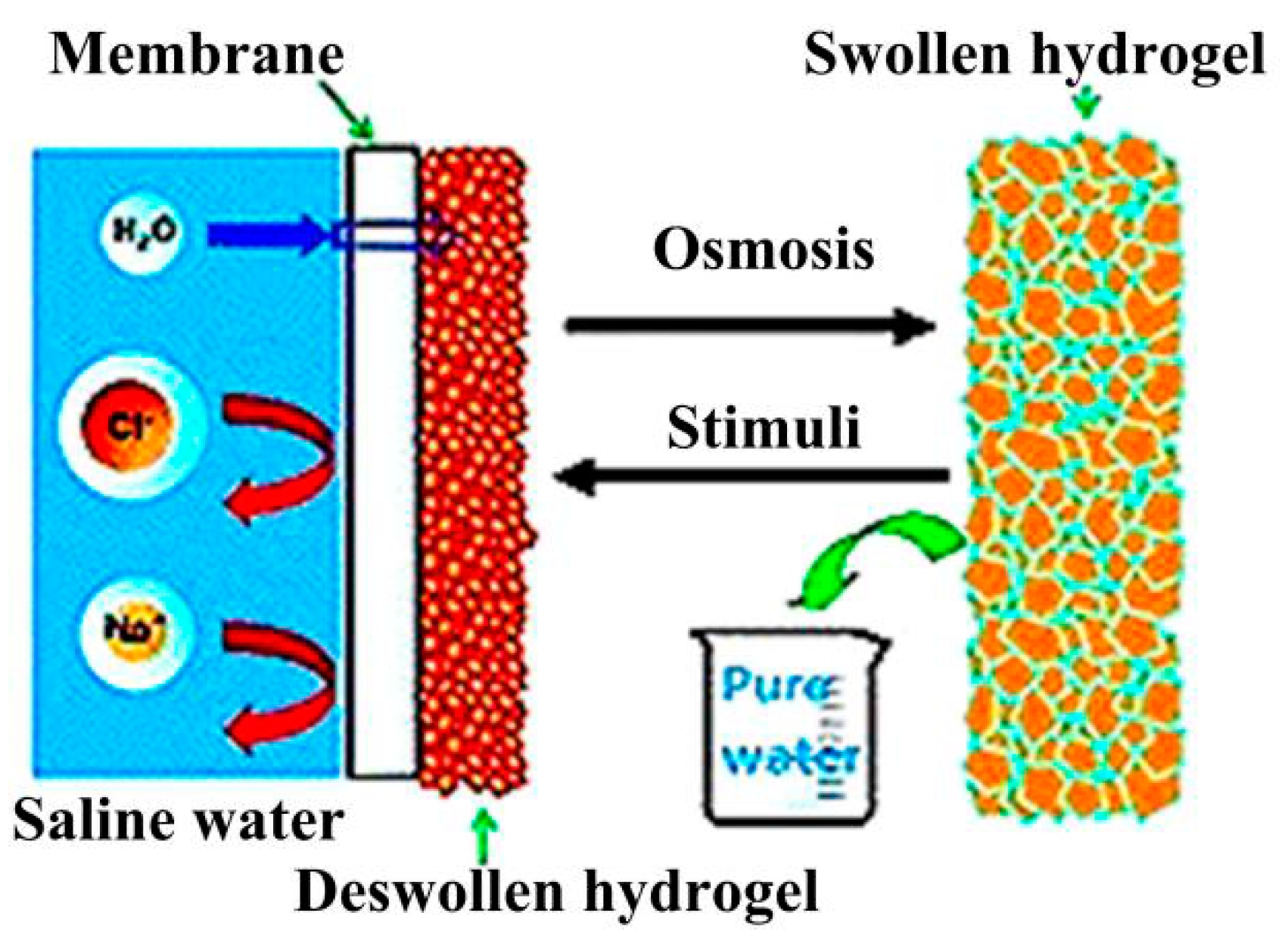
4.3.2. Enhancing Deswelling Rate of Hydrogel by Applying Different External Stimuli
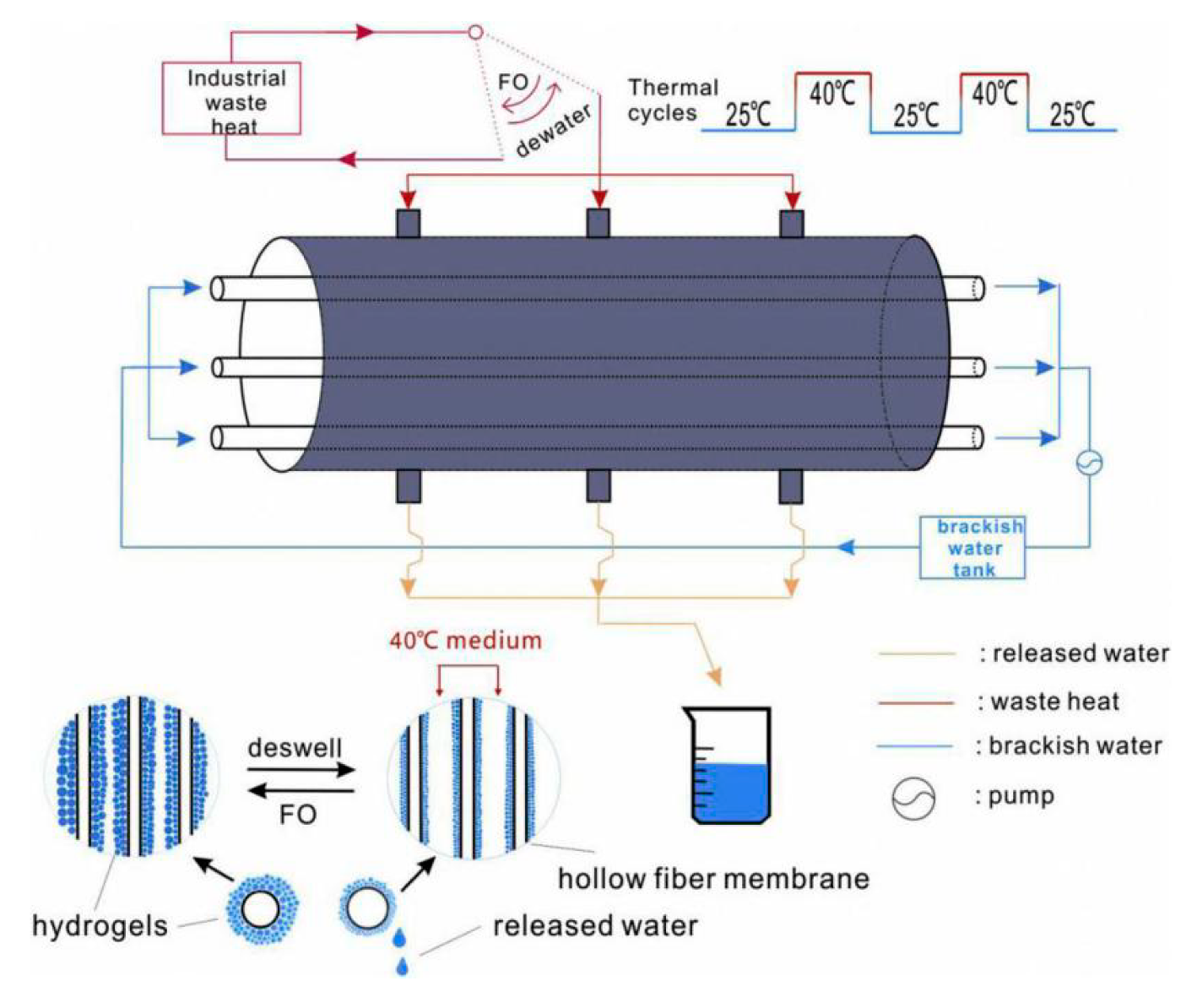
4.4. Designing Continuous Semibatch FO Process to Enhance the Apparent Water Flux
5. Some Important Issues
5.1. What on Earth Is It That Makes Hydrogel Produce Water Flux as the Draw Solute in the FO Process?
5.2. Why the Initial Water Flux of Hydrogel as Draw Solute Is Considered High Whereas the Total Water Flux Is Close to Zero in the 24-h FO process?
5.2.1. The Swelling Procedure and the Principle of Water Absorption by Hydrogel
5.2.2. Factors influencing Swelling Dynamic of Hydrogels
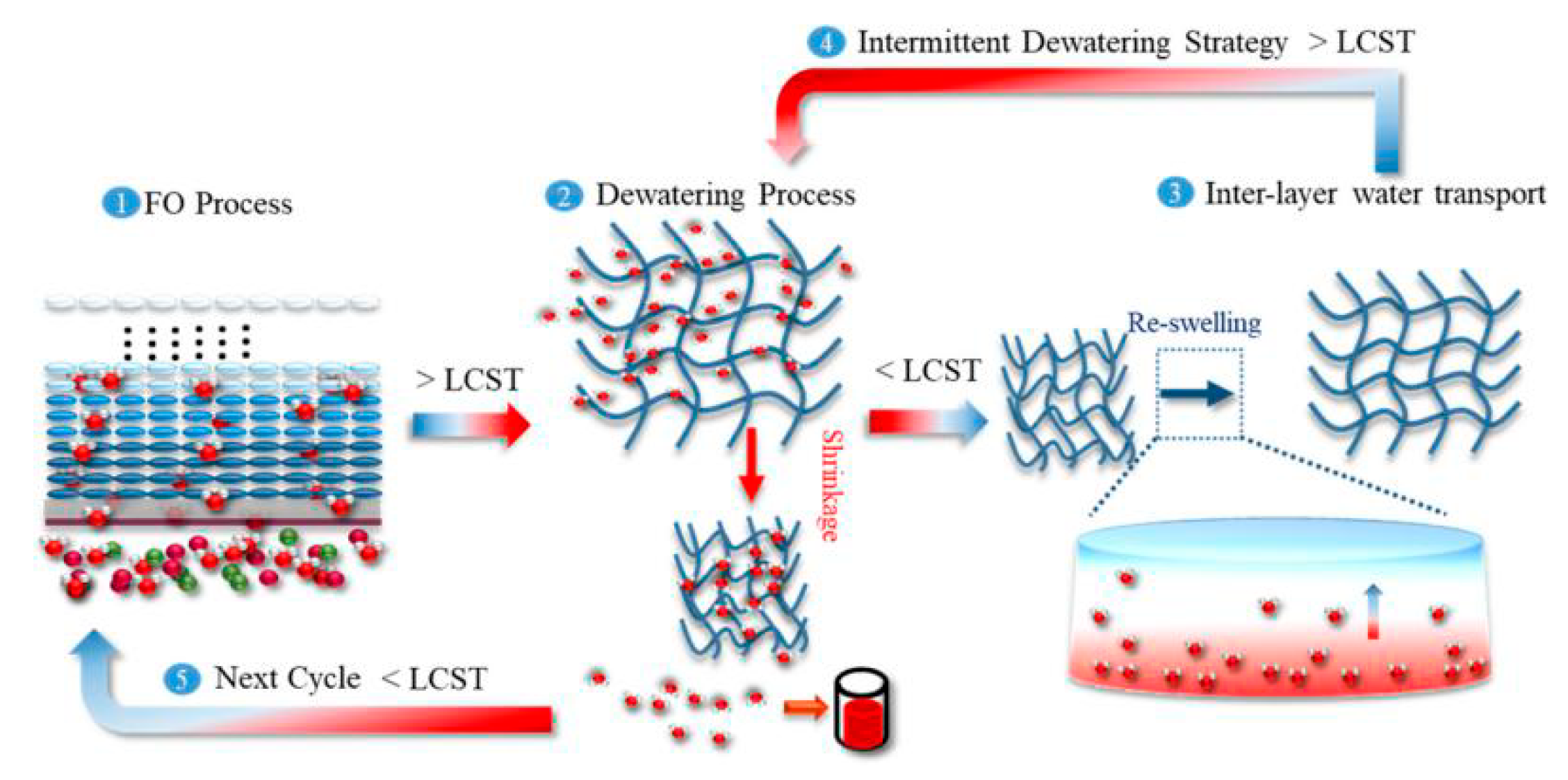
5.2.3. Explaining the High Initial Water Flux but Low Apparent Water Flux
5.3. Hydrogels Draw Solutes Have the Advantages of Intrinsically Zero Draw Solute Reverse Diffusion and There Is No Need to Further Polish the Released Water. Is it Actually True?
6. Challenges and Prospects for the Future
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PAM | poly(acrylamide) |
| PNIPAm | poly(N-isopropylacrylamide) |
| PSA | poly(sodium acrylate) |
| PSA-NIPAm | poly(sodium acrylate)-N-isopropylacrylamide |
| NP | N-isopropylacrylamide |
| NP95-AAm5 | thermo-responsive copolymer microgels (the mass ratio of N-isopropylacrylamide and acrylamide is 95:5) |
| NP95-HEMA5 | thermo-responsive copolymer microgels (the mass ratio of N-isopropylacrylamide and 2-hydroxyethyl methacrylate is 95:5) |
| NP95-PEGA5 | thermo-responsive copolymer microgels (the mass ratio of N-isopropylacrylamide and poly (ethylene glycol) methyl ether acrylate is 95:5) |
| P(MT20EO80) | the copolymer of (2-(methactyloyloxy) ethyl) trimethylammonium chloride (MT) and 2-(2-methoxyethoxy) ethyl methacrylate (EO) (the mass ratio is 20:80) |
| PSSP5 | poly(tetrabuylphosphonium styrenesulfonates)5 |
| TVBP-C6-5/5 | tributyl-4-vinylbenzylphosphonium (TVBP)-C6-5/5 based poly(ionic liquid) hydrogels where six represents the number of carbon atoms in the alkane sulfonate counterion and 5/5 represents the molar ratio of PPG to PEG |
| MCG-NP | microgels-N-isopropylacrylamide |
| MCG-NP-MAA | microgels-N-isopropylacrylamide-methacrylic acid |
| MCG-NP-AA | microgels-N-isopropylacrylamide-acrylic acid |
| MCG-NP-MA | microgels-N-isopropylacrylamide-maleic acid |
| MCG-NP-IA | microgels-N-isopropylacrylamide-itaconic acid |
| MCG-NP-AMPS | microgels-N-isopropylacrylamide-2-acrylamido-2-methyl-1-propanesulfonic acid |
| MCG-NP-DMAEMA | microgels-N-isopropylacrylamide-2-acrylamido-2-methyl-1-propanesulfonic acid |
| MCG-NP-DEAEMA | microgels-N-isopropylacrylamide-2-(diethylamino) ethyl methacrylate |
| MCG-NP-VP | microgels-N-isopropylacrylamide-4-vinylpyridine |
| MCG-NP-VI | microgels-N-isopropylacrylamide-1-vinylimidazole |
| CMC-g-PNaA-co-PNIPAM | carboxymethyl cellulose-graft-poly (sodium acrylate)-copolymerization-N-isopropylacrylamide |
| PSA-C | poly(sodium acrylate)-carbon |
| PNIPAM-C | poly(N-isopropylacrylamide)-carbon |
| PSA- NIPAM-C | poly(sodium acrylate)-N-isopropylacrylamide-carbon |
| PSA-1.2 wt% rGO | poly(sodium acrylate)-1.2wt% reduced graphene oxide |
| PSA-NIPAM-1.2 wt% rGO | Composite polymer hydrogel synthesized by adding 1.2wt% reduced graphene oxide, sodium acrylate and N-isopropylacrylamide |
| PSA-NIPAM-16 wt% γ-Fe2O3 | Composite polymer hydrogel synthesized by adding 16wt% γ-iron(III) oxide nanoparticles, sodium acrylate and N-isopropylacrylamide |
| PNIPAm-PSA-PUF | poly(N-isopropylacrylamide)-poly(sodium acrylate)-polyurethane |
| TPU-PN5S5 | thermoplastic polyurethane-N-isopropylacrylamide-sodium acrylate |
| SI-0.2PSA | semi-IPN-0.2 poly(sodium acrylate) |
| SI-0.5PVA | semi-IPN-0.5 polyvinyl alcohol |
| PEG-PLGA-PEG | poly(ethylene glycol-[DL-lactic acid-co-glycolic acid]-b-ethylene glycol) |
| PEG-PLGA-PEG/GO | poly(ethylene glycol-[DL-lactic acid-co-glycolic acid]-b-ethylene glycol)/Graphene oxide |
| PEG-PLGA-PEG/G | poly(ethylene glycol-[DL-lactic acid-co-glycolic acid]-b-ethylene glycol)/Graphene 5PSA-C-150 |
| PSA-C | poly(sodium acrylate)-carbon |
| MCG-NP-AA | microgel-N-isopropylacrylamide-acid acrylate |
| HA-PVA | Hyaluronic acid-Poly(vinyl alcohol) |
| AMPS/DMAEMA | 2-Acrylamido-2-methyl-1-propanesulfonic acid /2-N,N′-(Dimethylamino)ethyl methacrylate |
| DEAEMA-PEGDA | 2-N,N′-(diethylamino)ethyl methacrylate-poly (ethylene glycol diacrylate) |
| DMAEMA-PEGDA | 2-N,N′-(dimethylamino) ethyl methacrylate (DMAEMA)-poly (ethylene glycol diacrylate) |
| Fe3O4@P(NIPAM-co-AMPS) | Fe3O4@P(N-isopropylacrylamide-copolymerization-sodium 2-acrylamido-2-methylpropane sulfonate) |
| SL | single-layer |
| BL | bi-layer |
| ML | multi-layer |
References
- Klaysom, C.; Cath, T.Y.; Depuydt, T.; Vankelecom, I.F. Forward and pressure retarded osmosis: Potential solutions for global challenges in energy and water supply. Chem. Soc. Rev. 2013, 42, 6959–6989. [Google Scholar] [CrossRef]
- Zhao, D.; Qiu, G.; Li, X.; Wan, C.; Lu, K.; Chung, T.S. Zwitterions coated hollow fiber membranes with enhanced antifouling properties for osmotic power generation from municipal wastewater. Water Res. 2016, 104, 389–396. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen, N.; Zhao, D.; Lu, X. Thermoresponsive magnetic nanoparticles for seawater desalination. ACS Appl. Mater. Interfaces 2013, 5, 11453–11461. [Google Scholar] [PubMed]
- Elimelech, M.; Wiesner, M.R. Membrane Separations in Aquatic Systems. Environ. Eng. Sci. 2002, 19, 341. [Google Scholar] [CrossRef]
- Humplik, T.; Lee, J.; O’Hern, S.C.; Fellman, B.A.; Baig, M.A.; Hassan, S.F.; Atieh, M.A.; Rahman, F.; Laoui, T.; Karnik, R.; et al. Nanostructured materials for water desalination. Nanotechnology 2011, 22, 292001. [Google Scholar] [CrossRef] [PubMed]
- Malaeb, L.; Ayoub, G.M. Reverse osmosis technology for water treatment: State of the art review. Desalination 2011, 267, 1–8. [Google Scholar] [CrossRef]
- Cath, T.; Childress, A.; Elimelech, M. Forward osmosis: Principles, applications, and recent developments. J. Membr. Sci. 2006, 281, 70–87. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Shi, W.; Wang, W.; Yu, H.; Zhu, Z.; Cui, F. Polyamidoamine dendrimer grafted forward osmosis membrane with superior ammonia selectivity and robust antifouling capacity for domestic wastewater concentration. Water Res. 2019, 153, 1–10. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Shi, W.; Wang, W.; Zhu, Z.; Zhang, Z.; Cui, F. Dendritic amine sheltered membrane for simultaneous ammonia selection and fouling mitigation in forward osmosis. J. Membr. Sci. 2019, 584, 9–19. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Tian, J.; Shi, W.; Wang, W.; Zhang, Z.; Cui, F. Fouling mechanism of forward osmosis membrane in domestic wastewater concentration: Role of substrate structures. Chem. Eng. J. 2019, 370, 262–273. [Google Scholar]
- Zhang, X.; Tian, J.; Gao, S.; Shi, W.; Zhang, Z.; Cui, F.; Liu, D. Surface functionalization of TFC FO membranes with zwitterionic polymers: Improvement of antifouling and salt-responsive cleaning properties. J. Membr. Sci. 2017, 544, 368–377. [Google Scholar] [CrossRef]
- Zhang, X.; Tian, J.; Gao, S.; Zhang, Z.; Cui, F.; Tang, C.Y. In situ surface modification of thin film composite forward osmosis membranes with sulfonated poly (arylene ether sulfone) for anti-fouling in emulsified oil/water separation. J. Membr. Sci. 2017, 527, 26–34. [Google Scholar] [CrossRef]
- Zhang, X.; Tian, J.; Ren, Z.; Shi, W.; Zhang, Z.; Xu, Y.; Cui, F. High performance thin-film composite (TFC) forward osmosis (FO) membrane fabricated on novel hydrophilic disulfonated poly (arylene ether sulfone) multiblock copolymer/polysulfone substrate. J. Membr. Sci. 2016, 520, 529–539. [Google Scholar] [CrossRef]
- Qasim, M.; Darwish, N.A.; Sarp, S.; Hilal, N. Water desalination by forward (direct) osmosis phenomenon: A comprehensive review. Desalination 2015, 374, 47–69. [Google Scholar] [CrossRef]
- Werner, C.M.; Logan, B.E.; Saikaly, P.E.; Amy, G.L. Wastewater treatment, energy recovery and desalination using a forward osmosis membrane in an air-cathode microbial osmotic fuel cell. J. Membr. Sci. 2013, 428, 116–122. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; McGinnis, R.L.; Elimelech, M. Desalination by ammonia–carbon dioxide forward osmosis: Influence of draw and feed solution concentrations on process performance. J. Membr. Sci. 2006, 278, 114–123. [Google Scholar] [CrossRef]
- She, Q.; Jin, X.; Tang, C.Y. Osmotic power production from salinity gradient resource by pressure retarded osmosis: Effects of operating conditions and reverse solute diffusion. J. Membr. Sci. 2012, 401–402, 262–273. [Google Scholar] [CrossRef]
- Ling, M.M.; Chung, T.S. Novel dual-stage FO system for sustainable protein enrichment using nanoparticles as intermediate draw solutes. J. Membr. Sci. 2011, 372, 201–209. [Google Scholar] [CrossRef]
- Dova, M.I.; Petrotos, K.B.; Lazarides, H.N. On the direct osmotic concentration of liquid foods. Part I: Impact of process parameters on process performance. J. Food Eng. 2007, 78, 422–430. [Google Scholar] [CrossRef]
- Akther, N.; Sodiq, A.; Giwa, A.; Daer, S.; Arafat, H.A.; Hasan, S.W. Recent advancements in forward osmosis desalination: A review. Chem. Eng. J. 2015, 281, 502–522. [Google Scholar] [CrossRef]
- Phuntsho, S.; Shon, H.K.; Hong, S.; Lee, S.; Vigneswaran, S. A novel low energy fertilizer driven forward osmosis desalination for direct fertigation: Evaluating the performance of fertilizer draw solutions. J. Membr. Sci. 2011, 375, 172–181. [Google Scholar] [CrossRef]
- Petrotos, K.B.; Quantick, P.; Petropakis, H. A study of the direct osmotic concentration of tomato juice in tubular membrane-module configuration. I. The effect of certain basic process parameters on the process performance. J. Membr. Sci. 1998, 150, 99–110. [Google Scholar] [CrossRef]
- Chekli, L.; Phuntsho, S.; Shon, H.K.; Vigneswaran, S.; Kandasamy, J.; Chanan, A. A review of draw solutes in forward osmosis process and their use in modern applications. Desalin. Water Treat. 2012, 43, 167–184. [Google Scholar] [CrossRef]
- Yokozeki, A. Osmotic pressures studied using a simple equation-of-state and its applications. Appl. Energy 2006, 83, 15–41. [Google Scholar] [CrossRef]
- Zhao, S.; Zou, L.; Tang, C.Y.; Mulcahy, D. Recent developments in forward osmosis: Opportunities and challenges. J. Membr. Sci. 2012, 396, 1–21. [Google Scholar] [CrossRef]
- Zydney, A.L. Stagnant film model for concentration polarization in membrane systems. J. Membr. Sci. 1997, 130, 275–281. [Google Scholar] [CrossRef]
- Gray, G.T.; McCutcheon, J.R.; Elimelech, M. Internal concentration polarization in forward osmosis: Role of membrane orientation. Desalination 2006, 197, 1–8. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, Y.N.; Li, W.; Tang, C.Y. Characterization of internal and external concentration polarizations during forward osmosis processes. Desalination 2014, 338, 65–73. [Google Scholar] [CrossRef]
- Phillip, W.A.; Yong, J.S.; Elimelech, M. Reverse draw solute permeation in forward osmosis: Modeling and experiments. Environ. Sci. Technol. 2010, 44, 5170–5176. [Google Scholar] [CrossRef]
- Luo, H.; Wang, Q.; Zhang, T.C.; Tao, T.; Zhou, A.; Chen, L.; Bie, X. A review on the recovery methods of draw solutes in forward osmosis. J. Water Process Eng. 2014, 4, 212–223. [Google Scholar] [CrossRef]
- Gu, B.; Kim, J.H.; Yang, D.R. Theoretical analysis of a seawater desalination process integrating forward osmosis, crystallization, and reverse osmosis. J. Membr. Sci. 2013, 444, 440–448. [Google Scholar] [CrossRef]
- Chung, T.-S.; Li, X.; Ong, R.C.; Ge, Q.; Wang, H.; Han, G. Emerging forward osmosis (FO) technologies and challenges ahead for clean water and clean energy applications. Curr. Opin. Chem. Eng. 2012, 1, 246–257. [Google Scholar] [CrossRef]
- Ron, E.S.; Bromberg, L.E. Temperature-responsive gels and thermogelling polymer matrices for protein and peptide delivery. Adv. Drug Deliv. Rev. 1998, 31, 197–221. [Google Scholar] [PubMed]
- Lin, C.C.; Metters, A.T. Hydrogels in controlled release formulations: Network design and mathematical modeling. Adv. Drug Deliv. Rev. 2006, 58, 1379–1408. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Hu, X.M. A critical review on draw solutes development for forward osmosis. Desalination 2016, 391, 16–29. [Google Scholar] [CrossRef]
- Li, D.; Zhang, X.; Yao, J.; Simon, G.P.; Wang, H. Stimuli-responsive polymer hydrogels as a new class of draw agent for forward osmosis desalination. Chem. Commun. 2011, 47, 1710–1712. [Google Scholar] [CrossRef]
- Qiu, X.P.; Tanaka, F.; Winnik, F.M. Temperature-induced phase transition of well-defined cyclic poly (N-isopropylacrylamide) s in aqueous solution. Macromolecules 2007, 40, 7069–7071. [Google Scholar] [CrossRef]
- Hirose, Y.; Amiya, T.; Hirokawa, Y.; Tanaka, T. Phase transition of submicron gel beads. Macromolecules 1987, 20, 1342–1344. [Google Scholar] [CrossRef]
- Hartanto, Y.; Zargar, M.; Cui, X.; Jin, B.; Dai, S. Non-ionic copolymer microgels as high-performance draw materials for forward osmosis desalination. J. Membr. Sci. 2019, 572, 480–488. [Google Scholar] [CrossRef]
- Kim, J.J.; Chung, J.-S.; Kang, H.; Yu, Y.A.; Choi, W.J.; Kim, H.J.; Lee, J.-C. Thermo-responsive copolymers with ionic group as novel draw solutes for forward osmosis processes. Macromol. Res. 2014, 22, 963–970. [Google Scholar] [CrossRef]
- Kim, J.J.; Kang, H.; Choi, Y.-S.; Yu, Y.A.; Lee, J.C. Thermo-responsive oligomeric poly(tetrabutylphosphonium styrenesulfonate)s as draw solutes for forward osmosis (FO) applications. Desalination 2016, 381, 84–94. [Google Scholar] [CrossRef]
- Fan, X.; Liu, H.; Gao, Y.; Zou, Z.; Craig, V.S.; Zhang, G.; Liu, G. Forward-Osmosis Desalination with Poly(Ionic Liquid) Hydrogels as Smart Draw Agents. Adv. Mater. 2016, 28, 4156–4161. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Shen, W.; Loo, S.L.; Krantz, W.B.; Wang, R.; Fane, A.G.; Hu, X. Towards temperature driven forward osmosis desalination using Semi-IPN hydrogels as reversible draw agents. Water Res. 2013, 47, 3773–3781. [Google Scholar] [CrossRef] [PubMed]
- Hartanto, Y.; Zargar, M.; Wang, H.; Jin, B.; Dai, S. Thermoresponsive Acidic Microgels as Functional Draw Agents for Forward Osmosis Desalination. Environ. Sci. Technol. 2016, 50, 4221–4228. [Google Scholar] [CrossRef] [PubMed]
- Hartanto, Y.; Zargar, M.; Cui, X.; Shen, Y.; Jin, B.; Dai, S. Thermoresponsive cationic copolymer microgels as high performance draw agents in forward osmosis desalination. J. Membr. Sci. 2016, 518, 273–281. [Google Scholar] [CrossRef]
- Gawande, N.; Mungray, A.A. Superabsorbent polymer (SAP) hydrogels for protein enrichment. Sep. Purif. Technol. 2015, 150, 86–94. [Google Scholar] [CrossRef]
- Wei, J.; Low, Z.X.; Ou, R.; Simon, G.P.; Wang, H. Hydrogel-polyurethane interpenetrating network material as an advanced draw agent for forward osmosis process. Water Res. 2016, 96, 292–298. [Google Scholar] [CrossRef]
- Ou, R.; Zhang, H.; Simon, G.P.; Wang, H. Microfiber-polymer hydrogel monolith as forward osmosis draw agent. J. Membr. Sci. 2016, 510, 426–436. [Google Scholar] [CrossRef]
- Li, D.; Zhang, X.; Simon, G.P.; Wang, H. Forward osmosis desalination using polymer hydrogels as a draw agent: Influence of draw agent, feed solution and membrane on process performance. Water Res. 2013, 47, 209–215. [Google Scholar] [CrossRef]
- Razmjou, A.; Simon, G.P.; Wang, H. Effect of particle size on the performance of forward osmosis desalination by stimuli-responsive polymer hydrogels as a draw agent. Chem. Eng. J. 2013, 215–216, 913–920. [Google Scholar] [CrossRef]
- Hartanto, Y.; Yun, S.; Jin, B.; Dai, S. Functionalized thermo-responsive microgels for high performance forward osmosis desalination. Water Res. 2015, 70, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Razmjou, A.; Barati, M.R.; Simon, G.P.; Suzuki, K.; Wang, H. Fast deswelling of nanocomposite polymer hydrogels via magnetic field-induced heating for emerging FO desalination. Environ. Sci. Technol. 2013, 47, 6297–6305. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhang, X.; Yao, J.; Zeng, Y.; Simon, G.P.; Wang, H. Composite polymer hydrogels as draw agents in forward osmosis and solar dewatering. Soft Matter 2011, 7, 10048–10056. [Google Scholar] [CrossRef]
- Zeng, Y.; Qiu, L.; Wang, K.; Yao, J.; Li, D.; Simon, G.P.; Wang, R.; Wang, H. Significantly enhanced water flux in forward osmosis desalination with polymer-graphene composite hydrogels as a draw agent. RSC Adv. 2013, 3, 887–894. [Google Scholar] [CrossRef]
- Debord, J.D.; Lyon, L.A. Synthesis and characterization of pH-responsive copolymer microgels with tunable volume phase transition temperatures. Langmuir 2003, 19, 7662–7664. [Google Scholar] [CrossRef]
- Hirose, H.; Shibayama, M. Kinetics of volume phase transition in poly (N-isopropylacrylamide-co-acrylic acid) gels. Macromolecules 1998, 31, 5336–5342. [Google Scholar] [CrossRef]
- Dragan, E.S. Design and applications of interpenetrating polymer network hydrogels. A review. Chem. Eng. J. 2014, 243, 572–590. [Google Scholar] [CrossRef]
- Pound, G.; Aguesse, F.; McLeary, J.B.; Lange, R.F.M.; Klumperman, B. Xanthate-Mediated Copolymerization of Vinyl Monomers for Amphiphilic and Double-Hydrophilic Block Copolymers with Poly(ethylene glycol). Macromolecules 2007, 40, 8861–8871. [Google Scholar] [CrossRef]
- Nakka, R.; Mungray, A.A. Biodegradable and biocompatible temperature sensitive triblock copolymer hydrogels as draw agents for forward osmosis. Sep. Purif. Technol. 2016, 168, 83–92. [Google Scholar] [CrossRef]
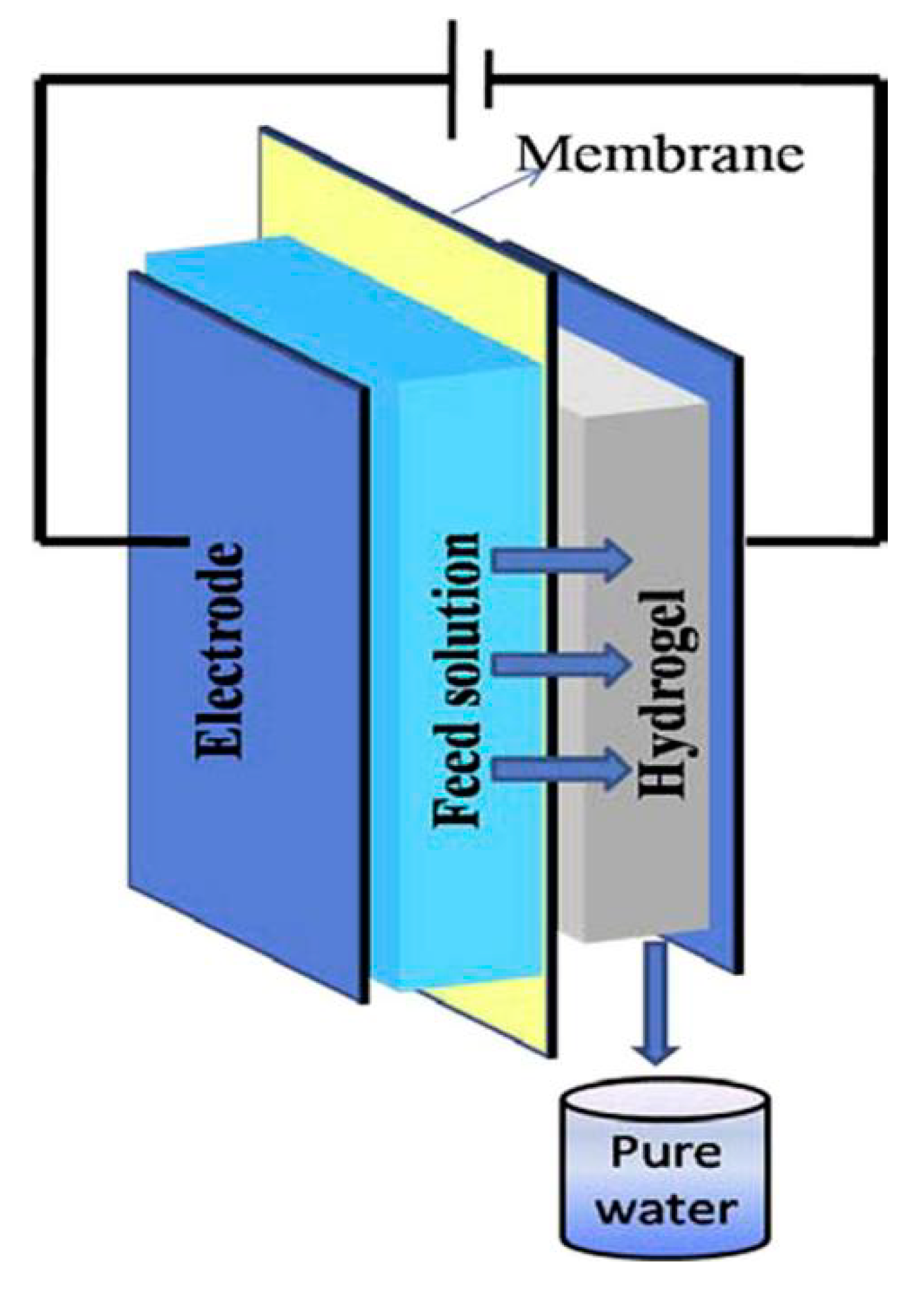
- Zhang, H.; Li, J.; Cui, H.; Li, H.; Yang, F. Forward osmosis using electric-responsive polymer hydrogels as draw agents: Influence of freezing–thawing cycles, voltage, feed solutions on process performance. Chem. Eng. J. 2015, 259, 814–819. [Google Scholar] [CrossRef]
- Juodkazytė, J.; Seniutinas, G.; Šebeka, B. Solar water splitting: Efficiency discussion. Int. J. Hydrogen Energy 2016, 41, 11941–11948. [Google Scholar] [CrossRef]
- Cui, H.; Zhang, H.; Yu, M.; Yang, F. Performance evaluation of electric-responsive hydrogels as draw agent in forward osmosis desalination. Desalination 2018, 426, 118–126. [Google Scholar] [CrossRef]
- Rabiee, H.; Jin, B.; Yun, S.; Dai, S. Gas-responsive cationic microgels for forward osmosis desalination. Chem. Eng. J. 2018, 347, 424–431. [Google Scholar] [CrossRef]
- Zhou, A.; Luo, H.; Wang, Q.; Chen, L.; Zhang, T.C.; Tao, T. Magnetic thermoresponsive ionic nanogels as novel draw agents in forward osmosis. RSC Adv. 2015, 5, 15359–15365. [Google Scholar] [CrossRef]
- Razmjou, A.; Liu, Q.; Simon, G.P.; Wang, H. Bifunctional polymer hydrogel layers as forward osmosis draw agents for continuous production of fresh water using solar energy. Environ. Sci. Technol. 2013, 47, 13160–13166. [Google Scholar] [CrossRef]
- Zeng, J.; Cui, S.; Wang, Q.; Chen, R. Multi-layer temperature-responsive hydrogel for forward-osmosis desalination with high permeable flux and fast water release. Desalination 2019, 459, 105–113. [Google Scholar] [CrossRef]
- Zhao, S. Osmotic pressure versus swelling pressure: Comment on “bifunctional polymer hydrogel layers as forward osmosis draw agents for continuous production of fresh water using solar energy”. Environ. Sci. Technol. 2014, 48, 4212–4213. [Google Scholar] [CrossRef]
- Wang, H.; Wei, J.; Simon, G.P. Response to osmotic pressure versus swelling pressure: Comment on “bifunctional polymer hydrogel layers as forward osmosis draw agents for continuous production of fresh water using solar energy”. Environ. Sci. Technol. 2014, 48, 4214–4215. [Google Scholar] [CrossRef]
- Hildebrand, J.H. Osmotic Pressure. Science 1955, 121, 116–119. [Google Scholar] [CrossRef]
- Wack, H.; Ulbricht, M. Effect of synthesis composition on the swelling pressure of polymeric hydrogels. Polymer 2009, 50, 2075–2080. [Google Scholar] [CrossRef]
- Höhne, P.; Tauer, K. How much weighs the swelling pressure. Colloid Polym. Sci. 2014, 292, 2983–2992. [Google Scholar] [CrossRef][Green Version]
- Horkay, F.; Tasaki, I.; Basser, P.J. Osmotic swelling of polyacrylate hydrogels in physiological salt solutions. Biomacromolecules 2000, 1, 84–90. [Google Scholar] [CrossRef]
- Tanaka, T.; Fillmore, D. Kinetics of swelling of gels. J. Chem. Phys. 1979, 70, 1214–1218. [Google Scholar] [CrossRef]
- Laftah, W.A.; Hashim, S.; Ibrahim, A.N. Polymer Hydrogels: A Review. Polymer-Plast. Technol. Eng. 2011, 50, 1475–1486. [Google Scholar] [CrossRef]
- Fujiyabu, T.; Yoshikawa, Y.; Chung, U.I.; Sakai, T. Structure-property relationship of a model network containing solvent. Sci. Technol. Adv. Mater. 2019, 20, 608–621. [Google Scholar] [CrossRef] [PubMed]
- Mudiyanselage, T.K.; Neckers, D.C. Highly absorbing superabsorbent polymer. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 1357–1364. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, X.; Cheng, S.; Zhuo, R. Strategies to improve the response rate of thermosensitive PNIPAAm hydrogels. Soft Matter. 2008, 4, 385–391. [Google Scholar] [CrossRef]
- Okeyoshi, K.; Abe, T.; Noguchi, Y.; Furukawa, H.; Yoshida, R. Shrinking Behavior of Surfactant-Grafted Thermosensitive Gels and the Mechanism of Rapid Shrinking. Macromol. Rapid Commun. 2008, 29, 897–903. [Google Scholar] [CrossRef]
- Chen, J.; Park, H.; Park, K. Synthesis of superporous hydrogels: Hydrogels with fast swelling and superabsorbent properties. J. Biomed. Mater. Res. 1999, 44, 53–62. [Google Scholar] [CrossRef]
- Lee, W.F.; Yeh, Y.C. Effect of porosigen and hydrophobic monomer on the fast swelling-deswelling behaviors for the porous thermoreversible copolymeric hydrogels. J. Appl. Polym. Sci. 2006, 100, 3152–3160. [Google Scholar] [CrossRef]
- Chirila, T.V.; Constable, I.J.; Crawford, G.J.; Vijayasekaran, S.; Thompson, D.E.; Chen, C.; Fletcher, W.A. Poly (2-hydroxyethyl methacrylate) sponges as implant materials: In vivo and in vitro evaluation of cellular invasion. Biomaterials 1993, 14, 26–38. [Google Scholar] [CrossRef]
- Chang, S.; Kim, M.; Oh, S. Multi-scale characterization of surface-crosslinked superabsorbent polymer hydrogel spheres. Polymer 2018, 145, 174–183. [Google Scholar] [CrossRef]
- Gemeinhart, R.A.; Park, H. Pore structure of superporous hydrogels. Polym. Adv. Technol. 2000, 11, 617–625. [Google Scholar] [CrossRef]
- Khalloufi, S.; Bongers, P. Mathematical investigation of the case hardening phenomenon explained by shrinkage and collapse mechanisms occurring during drying processes. Comput. Aided Chem. Eng. 2012, 30, 1068–1072. [Google Scholar]
- Xie, T. Tunable polymer multi-shape memory effect. Nature 2010, 464, 267–270. [Google Scholar] [CrossRef]
- Shi, Y.; Ma, C.; Peng, L. Conductive “Smart” Hybrid Hydrogels with PNIPAM and Nanostructured Conductive Polymers. Adv. Funct. Mater. 2015, 25, 1219–1225. [Google Scholar] [CrossRef]

| Draw Solution | De-Watering Method | FO Performance | Ref. | |
|---|---|---|---|---|
| Initial Flux (L/m2h) | Water Recovery | |||
| PAM | Heating at 50 °C | 0.36 (1st h) | <10% | [36] |
| PNIPAm | 0.30 (1st h) | 54%–75% | ||
| PSA | 0.96 (1st h) | <5% | ||
| PSA-NIPAm | 0.55 (1st h) | 17% | ||
| NP | Centrifuged at 40 °C and 10,000 rpm for 10 min | 5.1 | 83.3% | [39] |
| NP95-AAm5 | 24.7 | 78.7% | ||
| NP95-HEMA5 | 7.8 | 88.9% | ||
| NP95-PEGA5 | 6.4 | 89.1% | ||
| P(MT20EO80) | Heating at 70 °C followed by microfiltration process | - | 99.7% | [40] |
| PSSP5 | Heating at 60 °C | - | 99.5% | [41] |
| TVBP-C6-5/5 | Heating at 60 °C | < 0.9 | <90% | [42] |
| SI-0.2PSA | Heating at 40 °C for 10 min | 0.18 (1st h) | - | [43] |
| SI-0.5PVA | 0.18 (1st h) | |||
| SI-0.2PVA | 0.12 (1st h) | |||
| MCG-NP | Centrifuged at 40 °C and 10,000 rpm for 10 min | 7.5 (1st min) | 72.1% | [44] |
| MCG-NP-MAA | 10.9 (1st min) | 76.7% | ||
| MCG-NP-AA | 16.7 (1st min) | 55.8% | ||
| MCG-NP-MA | 39.1 (1st min) | 39.1% | ||
| MCG-NP-IA | 44.8 (1st min) | 47.2% | ||
| MCG-NP-AMPS | 42.9 (1st min) | 0% | ||
| MCG-NP-DMAEMA | Centrifuged at 40 °C and 10,000 rpm for 10 min | 33.5 (1st min) | 34.1% | [45] |
| MCG-NP-DEAEMA | 45.6 (1st min) | 44.8% | ||
| MCG-NP-VP | 17.3 (1st min) | 51.9% | ||
| MCG-NP-VI | 28.9 (1st min) | 47.9% | ||
| CMC-g-PNaA-co-PNIPAM | - | - | 70% | [46] |
| PNIPAm-PSA-PUF (mass ration, 8:1) | Under sunlight simulator (2 kw/m2) | 14.8 | 100% | [47] |
| TPU-PN5S5 | Under sunlight simulator (1 kw/m2) | 1.81 (10st min) | - | [48] |
| TPU-PN5S5 | 2.77 (10st min) | |||
| 5PSA-C-150 100–200 um | - | 1.02 | - | [49] |
| PSA-C-150 100–200 um | - | 1.06 | - | |
| PSA-5C-150 100–200 um | - | 1.06 | - | |
| PSA-NIPAm (2–25 um) | N2 gas pressure of 600 kPa for 15 min | - | <7% (swelling ratio 10) | [50] |
| PSA-NIPAm (190–350 um) | - | <5% (swelling ratio 10) | ||
| PSA-NIPAm (500–1000 um) | - | - | ||
| MCG-NP100-AA0 | - | 2 (10st min) | - | [51] |
| MCG-NP50-AA50 | - | 4 (10st min) | - | |
| PSA-NIPAM-16 wt%γ-Fe2O3 | Magnetic AC field (148 kOe and 372 kHz) | <2.5 (1.0st h) | 66% | [52] |
| PSA-C | Under the sunlight simulator (1 kw m−2) | 1.32 (0.5st h) | 70.4% | [53] |
| PNIPAM-C | 0.44 (0.5st h) | 100% | ||
| PSA- NIPAM-C | 0.77 (0.5st h) | 100% | ||
| PSA-1.2 wt% rGO | Under the sunlight simulator (1 kW m−2) for 1h | 3.1 (1.0st h) | 42% | [54] |
| PSA-NIPAM-1.2 wt% rGO | 0.9 (1.0st h) | 45% | ||
| PEG-PLGA-PEG | 13,000 rpm for 10 min | - | 98% | [59] |
| PEG-PLGA-PEG/GO-0.09 wt% | - | 98% | ||
| PEG-PLGA-PEG/G-0.09 wt% | - | 98% | ||
| HA-PVA-5 | Electric field (9 v) | 1.2 | [60] | |
| HA-PVA-7 | 0.91 | |||
| HA-PVA-9 | 0.9 | |||
| AMPS/DMAEMA (0.55/0.45) | Electric field (15 v) for 60 min | 2.09 (1st h) | 67.45% | [62] |
| AMPS/DMAEMA (0.53/0.47) | 1.63 (1st h) | 39.36% | ||
| DEAEMA-PEGDA (1 wt%) | Puring N2 | 56 (10st min) | 55% | [63] |
| DMAEMA-PEGDA (1 wt%) | 41 (10st min) | 44% | ||
| Fe3O4@P(NIPAM-co-AMPS) | 65 °C and magnetic field (250 mT) | 0.26 (20st min) | [64] | |
| PNIPAm-PSA (mass ration, 1:1) | Solar energy of 0.5 kW m−2 for 1 h | 0.25 (20st min) | 7.8% ± 1.5% | [65] |
| SL-2 | Heating at 60 °C f or 120 min | 0.191 (1st h) | [66] | |
| BL-2 | 0.236 (1st h) | 72% | ||
| ML-1 | 0.292 (1st h) | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Gao, S.; Tian, J.; Cui, F.; Shi, W. Recent Developments and Future Challenges of Hydrogels as Draw Solutes in Forward Osmosis Process. Water 2020, 12, 692. https://doi.org/10.3390/w12030692
Wang J, Gao S, Tian J, Cui F, Shi W. Recent Developments and Future Challenges of Hydrogels as Draw Solutes in Forward Osmosis Process. Water. 2020; 12(3):692. https://doi.org/10.3390/w12030692
Chicago/Turabian StyleWang, Jichao, Shanshan Gao, Jiayu Tian, Fuyi Cui, and Wenxin Shi. 2020. "Recent Developments and Future Challenges of Hydrogels as Draw Solutes in Forward Osmosis Process" Water 12, no. 3: 692. https://doi.org/10.3390/w12030692
APA StyleWang, J., Gao, S., Tian, J., Cui, F., & Shi, W. (2020). Recent Developments and Future Challenges of Hydrogels as Draw Solutes in Forward Osmosis Process. Water, 12(3), 692. https://doi.org/10.3390/w12030692





