Quantifying Escherichia coli and Suspended Particulate Matter Concentrations in a Mixed-Land Use Appalachian Watershed
Abstract
1. Introduction
2. Methods
2.1. Study Site Description
2.2. Data Collection
2.3. Data Analysis
3. Results
3.1. Climate during Study
3.2. Suspended Particulate Matter Concentrations and Escherichia coli
3.3. Non-Parametric Statistical Analysis
4. Discussion
4.1. E. coli and SPM Concentrations
4.2. Non-Parametric Statistical Analysis
4.3. Study Considerations and Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oliver, D.M.; Clegg, C.D.; Heathwaite, A.L.; Haygarth, P.M. Preferential attachment of Escherichia coli to different particle size fractions of an agricultural grassland soil. Water Air Soil Pollut. 2007, 185, 369–375. [Google Scholar] [CrossRef]
- Samie, A. Escherichia coli—Recent Advances on Physiology, Pathogenesis and Biotechnological Applications; IntechOpen Seattle: Washington, WA, USA, 2017. [Google Scholar]
- Mahmoud, M.A.; Abdelsalam, M.; Mahdy, O.A.; El Miniawy, H.M.F.; Ahmed, Z.A.M.; Osman, A.H.; Mohamed, H.M.H.; Khattab, A.M.; Zaki Ewiss, M.A. Infectious bacterial ogens, parasites and pathological correlations of sewage pollution as an important threat to farmed fishes in Egypt. Environ. Pollut. 2016, 219, 939–948. [Google Scholar] [CrossRef]
- Bain, R.; Cronk, R.; Hossain, R.; Bonjour, S.; Onda, K.; Wright, J.; Yang, H.; Slaymaker, T.; Hunter, P.; Prüss-Ustün, A.; et al. Global assessment of exposure to faecal contamination through drinking water based on a systematic review. Trop. Med. Int. Health 2014, 19, 917–927. [Google Scholar] [CrossRef]
- Bain, R.; Cronk, R.; Wright, J.; Yang, H.; Slaymaker, T.; Bartram, J. Fecal contamination of drinking-water in low- and middle-income countries: A systematic review and meta-analysis. PLoS Med. 2014, 11. [Google Scholar] [CrossRef]
- WHO World Water Day Report. Available online: https://www.who.int/water_sanitation_health/takingcharge.html (accessed on 12 December 2019).
- Benedict, K.M. Surveillance for waterborne disease outbreaks associated with drinking water—United States, 2013–2014. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1216. [Google Scholar] [CrossRef]
- Olsen, C.R.; Cutshall, N.H.; Larsen, I.L. Pollutant—Particle associations and dynamics in coastal marine environments: A review. Mar. Chem. 1982, 11, 501–533. [Google Scholar] [CrossRef]
- Jamieson, R.; Joy, D.M.; Lee, H.; Kostaschuk, R.; Gordon, R. Transport and deposition of sediment-associated Escherichia coli in natural streams. Water Res. 2005, 39, 2665–2675. [Google Scholar] [CrossRef]
- Amalfitano, S.; Corno, G.; Eckert, E.; Fazi, S.; Ninio, S.; Callieri, C.; Grossart, H.-P.; Eckert, W. Tracing particulate matter and associated microorganisms in freshwaters. Hydrobiologia 2017, 800, 145–154. [Google Scholar] [CrossRef]
- Jeng, H.C.; England, A.J.; Bradford, H.B. Indicator organisms associated with stormwater suspended particles and estuarine sediment. J. Environ. Sci. Health 2005, 40, 779–791. [Google Scholar] [CrossRef]
- Hassard, F.; Gwyther, C.L.; Farkas, K.; Andrews, A.; Jones, V.; Cox, B.; Brett, H.; Jones, D.L.; McDonald, J.E.; Malham, S.K. Abundance and distribution of enteric bacteria and viruses in coastal and estuarine sediments—A review. Front. Microbiol. 2016, 7, 1692. [Google Scholar] [CrossRef]
- Rieck, A.; Herlemann, D.P.; Jürgens, K.; Grossart, H.-P. Particle-associated differ from free-living bacteria in surface waters of the Baltic Sea. Front. Microbiol. 2015, 6, 1297. [Google Scholar] [CrossRef]
- Grossart, H.-P. Ecological consequences of bacterioplankton lifestyles: Changes in concepts are needed. Environ. Microbiol. Rep. 2010, 2, 706–714. [Google Scholar] [CrossRef]
- Drummond, J.D.; Davies-Colley, R.J.; Stott, R.; Sukias, J.P.; Nagels, J.W.; Sharp, A.; Packman, A.I. Microbial transport, retention, and inactivation in streams: A combined experimental and stochastic modeling approach. Environ. Sci. Technol. 2015, 49, 7825–7833. [Google Scholar] [CrossRef]
- Simon, M.; Grossart, H.-P.; Schweitzer, B.; Ploug, H. Microbial ecology of organic aggregates in aquatic ecosystems. Aquat. Microb. Ecol. 2002, 28, 175–211. [Google Scholar] [CrossRef]
- Bidle, K.D.; Flecthcer, M. Comparison of free-living and particle-associated bacterial communities in the Chesapeake Bay by stable low-molecular-weight Rna analysis. Appl. Environ. Microbiol. 1995, 61, 944–952. [Google Scholar] [CrossRef]
- Allen, H.K.; Donato, J.; Wang, H.H.; Cloud-Hansen, K.A.; Davies, J.; Handelsman, J. Call of the wild: Antibiotic resistance genes in natural environments. Nat. Rev. Microbiol. 2010, 8, 251. [Google Scholar] [CrossRef]
- Corno, G.; Coci, M.; Giardina, M.; Plechuk, S.; Campanile, F.; Stefani, S. Antibiotics promote aggregation within aquatic bacterial communities. Front. Microbiol. 2014, 5, 297. [Google Scholar] [CrossRef]
- Madoux-Humery, A.-S.; Dorner, S.; Sauvé, S.; Aboulfadl, K.; Galarneau, M.; Servais, P.; Prévost, M. The effects of combined sewer overflow events on riverine sources of drinking water. Water Res. 2016, 92, 218–227. [Google Scholar] [CrossRef]
- Jett, B.D.; Huycke, M.M.; Gilmore, M.S. Virulence of enterococci. Clin. Microbiol. Rev. 1994, 7, 462–478. [Google Scholar] [CrossRef]
- Anastasi, E.M.; Matthews, B.; Gundogdu, A.; Vollmerhausen, T.L.; Ramos, N.L.; Stratton, H.; Ahmed, W.; Katouli, M. Prevalence and persistence of Escherichia coli strains with uropathogenic virulence characteristics in sewage treatment plants. Appl. Environ. Microbiol. 2010, 76, 5882–5886. [Google Scholar] [CrossRef]
- Abia, A.L.K.; Ubomba-Jaswa, E.; Genthe, B.; Momba, M.N.B. Quantitative microbial risk assessment (QMRA) shows increased public health risk associated with exposure to river water under conditions of riverbed sediment resuspension. Sci. Total Environ. 2016, 566, 1143–1151. [Google Scholar] [CrossRef]
- Characklis, G.W.; Dilts, M.J.; Simmons III, O.D.; Likirdopulos, C.A.; Krometis, L.-A.H.; Sobsey, M.D. Microbial partitioning to settleable particles in stormwater. Water Res. 2005, 39, 1773–1782. [Google Scholar] [CrossRef]
- Hubbart, J.A. Using sediment particle size class analysis to better understand urban land-use effects. Int. J. Appl. 2012, 2, 12–27. [Google Scholar]
- Kellner, E.; Hubbart, J.A. Improving understanding of mixed-land-use watershed suspended sediment regimes: Mechanistic progress through high-frequency sampling. Sci. Total Environ. 2017, 598, 228–238. [Google Scholar] [CrossRef]
- Kellner, E.; Hubbart, J.A. Flow class analyses of suspended sediment concentration and particle size in a mixed-land-use watershed. Sci. Total Environ. 2019, 648, 973–983. [Google Scholar] [CrossRef]
- Kellner, E.; Hubbart, J.A. Quantifying urban land-use impacts on suspended sediment particle size class distribution: A method and case study. Stormwater J. 2014, 15, 40–50. [Google Scholar]
- Chen, H.J.; Chang, H. Response of discharge, TSS, and E. coli to rainfall events in urban, suburban, and rural watersheds. Environ. Sci. Process. Impacts 2014, 16, 2313–2324. [Google Scholar] [CrossRef]
- Cantor, J.; Krometis, L.-A.; Sarver, E.; Cook, N.; Badgley, B. Tracking the downstream impacts of inadequate sanitation in central Appalachia. J. Water Health 2017, 15, 580–590. [Google Scholar] [CrossRef]
- Koppen, W.D. Das geographische system der klimate. Handb. Klimatol. 1936, 46, 1–44. [Google Scholar]
- Central Appalachian Broadleaf Forest—Coniferous Forest—Meadow Province. Available online: https://www.fs.fed.us/land/ecosysmgmt/colorimagemap/images/m221.html (accessed on 12 December 2019).
- Arcipowski, E.; Schwartz, J.; Davenport, L.; Hayes, M.; Nolan, T. Clean water, clean life: Promoting healthier, accessible water in rural Appalachia. J. Contemp. Water Res. Educ. 2017, 161, 1–18. [Google Scholar] [CrossRef]
- Kellner, E.; Hubbart, J.; Stephan, K.; Morrissey, E.; Freedman, Z.; Kutta, E.; Kelly, C. Characterization of sub-watershed-scale stream chemistry regimes in an Appalachian mixed-land-use watershed. Environ. Monit. Assess. 2018, 190, 586. [Google Scholar] [CrossRef]
- Petersen, F.; Hubbart, J.A.; Kellner, E.; Kutta, E. Land-use-mediated Escherichia coli concentrations in a contemporary Appalachian watershed. Environ. Earth Sci. 2018, 77, 754. [Google Scholar] [CrossRef]
- The West Virginia Water Research Institute, The West Run Watershed Association. (WVWRI) Watershed Based Plan for West Run of the Monongahela River 2008; The West Virginia Water Research Institute, The West Run Watershed Association: Morgantown, WV, USA, 2008. [Google Scholar]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. Discuss. 2007, 4, 439–473. [Google Scholar] [CrossRef]
- Arguez, A.; Durre, I.; Applequist, S.; Squires, M.; Vose, R.; Yin, X.; Bilotta, R. NOAA’s US climate normals (1981–2010). NOAA Natl. Cent. Environ. Inf. 2010, 10, V5PN93JP. [Google Scholar]
- Myers, M.D. National field manual for the collection of water-quality data. US Geol. Surv. Tech. Water Resour. Investig. Book 2006, 9. [Google Scholar] [CrossRef]
- Hubbart, J.A.; Kellner, E.; Hooper, L.W.; Zeiger, S. Quantifying loading, toxic concentrations, and systemic persistence of chloride in a contemporary mixed-land-use watershed using an experimental watershed approach. Sci. Total Environ. 2017, 581, 822–832. [Google Scholar] [CrossRef]
- Kellner, E.; Hubbart, J. Advancing understanding of the surface water quality regime of contemporary mixed-land-use watersheds: An application of the experimental watershed method. Hydrology 2017, 4, 31. [Google Scholar] [CrossRef]
- Zeiger, S.J.; Hubbart, J.A. Quantifying flow interval–pollutant loading relationships in a rapidly urbanizing mixed-land-use watershed of the Central USA. Environ. Earth Sci. 2017, 76, 484. [Google Scholar] [CrossRef]
- Zeiger, S.J.; Hubbart, J.A. Nested-scale nutrient flux in a mixed-land-use urbanizing watershed. Hydrol. Process. 2016, 30, 1475–1490. [Google Scholar] [CrossRef]
- Crump, B.C.; Armbrust, E.V.; Baross, J.A. Phylogenetic analysis of particle-attached and free-living bacterial communities in the Columbia River, its estuary, and the adjacent coastal ocean. Appl. Environ. Microbiol. 1999, 65, 3192–3204. [Google Scholar] [CrossRef]
- Riemann, L.; Winding, A. Community dynamics of free-living and particle-associated bacterial assemblages during a freshwater phytoplankton bloom. Microb. Ecol. 2001, 42, 274–285. [Google Scholar] [CrossRef]
- Ortega-Retuerta, E.; Joux, F.; Jeffrey, W.H.; Ghiglione, J.-F. Spatial variability of particle-attached and free-living bacterial diversity in surface waters from the Mackenzie River to the Beaufort Sea (Canadian Arctic). Biogeosciences 2013, 10, 2747–2759. [Google Scholar] [CrossRef]
- Wentworth, C.K. A scale of grade and class terms for clastic sediments. J. Geol. 1922, 30, 377–392. [Google Scholar] [CrossRef]
- IDEXX Laboratories Colilert Procedure Manual. Available online: https://www.idexx.com/files/colilert-procedure-en.pdf (accessed on 4 April 2019).
- Cummings, D. The Fecal Coliform Test Compared to Specific Tests For Escherichia Coli. Available online: https://www.idexx.com/resource-library/water/water-reg-article9B.pdf (accessed on 24 September 2019).
- Yazici, B.; Yolacan, S. A comparison of various tests of normality. J. Stat. Comput. Simul. 2007, 77, 175–183. [Google Scholar] [CrossRef]
- Stokes, M.E.; Davis, C.S.; Koch, G.G. Categorical Data Analysis Using SAS; SAS institute: Cary, NC, USA, 2012. [Google Scholar]
- United States Climate Data (USCD). Available online: https://www.usclimatedata.com/climate/morgantown/west-virginia/united-states/uswv0507/2012/7 (accessed on 28 September 2019).
- Stein, E.D.; Tiefenthaler, L.; Schiff, K. Comparison of stromwater pollutant loading by land use type. Proc. Water Environ. Fed. 2007, 2007, 700–722. [Google Scholar] [CrossRef]
- Bro, R.; Smilde, A.K. Principal component analysis. Anal. Methods 2014, 6, 2812–2831. [Google Scholar] [CrossRef]
- Jeloudar, F.T.; Sepanlou, M.G.; Emadi, S.M. Impact of land use change on soil erodibility. Glob. J. Environ. Sci. Manag. 2018, 4, 59–70. [Google Scholar]
- Tong, S.T.; Chen, W. Modeling the relationship between land use and surface water quality. J. Environ. Manag. 2002, 66, 377–393. [Google Scholar] [CrossRef]
- Wilson, C.; Weng, Q. Assessing surface water quality and its relation with urban land cover changes in the Lake Calumet Area, Greater Chicago. Environ. Manag. 2010, 45, 1096–1111. [Google Scholar] [CrossRef]
- Causse, J.; Billen, G.; Garnier, J.; Henri-des-Tureaux, T.; Olasa, X.; Thammahacksa, C.; Latsachak, K.O.; Soulileuth, B.; Sengtaheuanghoung, O.; Rochelle-Newall, E. Field and modelling studies of Escherichia coli loads in tropical streams of montane agro-ecosystems. J. Hydro Environ. Res. 2015, 9, 496–507. [Google Scholar] [CrossRef]
- Rochelle-Newall, E.J.; Ribolzi, O.; Viguier, M.; Thammahacksa, C.; Silvera, N.; Latsachack, K.; Dinh, R.P.; Naporn, P.; Sy, H.T.; Soulileuth, B. Effect of land use and hydrological processes on Escherichia coli concentrations in streams of tropical, humid headwater catchments. Sci. Rep. 2016, 6, 32974. [Google Scholar] [CrossRef]
- Dong, H.; Onstott, T.C.; DeFlaun, M.F.; Fuller, M.E.; Scheibe, T.D.; Streger, S.H.; Rothmel, R.K.; Mailloux, B.J. Relative dominance of physical versus chemical effects on the transport of adhesion-deficient bacteria in intact cores from South Oyster, Virginia. Environ. Sci. Technol. 2002, 36, 891–900. [Google Scholar] [CrossRef]
- Levy, J.; Sun, K.; Findlay, R.H.; Farruggia, F.T.; Porter, J.; Mumy, K.L.; Tomaras, J.; Tomaras, A. Transport of Escherichia coli bacteria through laboratory columns of glacial-outwash sediments: Estimating model parameter values based on sediment characteristics. J. Contam. Hydrol. 2007, 89, 71–106. [Google Scholar] [CrossRef]
- Mallin, M.A.; Johnson, V.L.; Ensign, S.H. Comparative impacts of stormwater runoff on water quality of an urban, a suburban, and a rural stream. Environ. Monit. Assess. 2009, 159, 475–491. [Google Scholar] [CrossRef]
- Gilley, J.E.; Bartelt-Hunt, S.L.; Eskridge, K.M.; Li, X.; Schmidt, A.M.; Snow, D.D. Setback distance requirements for removal of swine slurry constituents in runoff. Trans. ASABE 2017, 60, 1885–1894. [Google Scholar] [CrossRef]
- Sutherland, D.G.; Ball, M.H.; Hilton, S.J.; Lisle, T.E. Evolution of a landslide-induced sediment wave in the Navarro River, California. GSA Bull. 2002, 114, 1036–1048. [Google Scholar] [CrossRef]
- Gotkowska-Plachta, A.; Golaś, I.; Korzeniewska, E.; Koc, J.; Rochwerger, A.; Solarski, K. Evaluation of the distribution of fecal indicator bacteria in a river system depending on different types of land use in the southern watershed of the Baltic Sea. Environ. Sci. Pollut. Res. 2016, 23, 4073–4085. [Google Scholar] [CrossRef]
- Davies, C.M.; Bavor, H.J. The fate of stormwater-associated bacteria in constructed wetland and water pollution control pond systems. J. Appl. Microbiol. 2000, 89, 349–360. [Google Scholar] [CrossRef]
- Muirhead, R.W.; Collins, R.P.; Bremer, P.J. Interaction of Escherichia coli and soil particles in runoff. Appl. Environ. Microbiol. 2006, 72, 3406–3411. [Google Scholar] [CrossRef]
- Guber, A.K.; Pachepsky, Y.A.; Shelton, D.R.; Yu, O. Effect of bovine manure on fecal coliform attachment to soil and soil particles of different sizes. Appl. Environ. Microbiol. 2007, 73, 3363–3370. [Google Scholar] [CrossRef]
- Southard, J. Introduction to Fluid Motions, Sediment Transport, and Current-Generated Sedimentary Structures Course Textbook; MIT OpenCourseWare, Massachusetts Institute of Technology: Cambridge, MA, USA, 2006. [Google Scholar]
- Multivariate Analysis: Principal Component Analysis: Biplots-9.3. Available online: http://support.sas.com/documentation/cdl/en/imlsug/64254/HTML/default/viewer.htm#imlsug_ugmultpca_sect003.htm (accessed on 23 December 2019).
- Presser, K.A.; Ratkowsky, D.A.; Ross, T. Modelling the growth rate of Escherichia coli as a function of pH and lactic acid concentration. Appl. Environ. Microbiol. 1997, 63, 2355–2360. [Google Scholar] [CrossRef]
- Noble, R.T.; Lee, I.M.; Schiff, K.C. Inactivation of indicator micro-organisms from various sources of faecal contamination in seawater and freshwater. J. Appl. Microbiol. 2004, 96, 464–472. [Google Scholar] [CrossRef]
- Helsel, D.R.; Hirsch, R.M. Statistical Methods in Water Resources, 1st ed.; Elsevier: Amsterdam, The Netherlands, 1992; Volume 49, ISBN 978-0-08-087508-8. [Google Scholar]
- Wagner, M.; Lambert, S. Freshwater Microplastics: Emerging Environmental Contaminants; The Handbook of Environmental Chemistry; Barcelo, D., Kostianoy, A.G., Eds.; Springer International Publishing: Cham, Switzerland, 2018; ISBN 978-3-319-61614-8. [Google Scholar]
- Rillig, M.C. Microplastic in terrestrial ecosystems and the soil? Environ. Sci. Technol. 2012, 46, 6453–6454. [Google Scholar] [CrossRef]
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef]
- Lithner, D.; Damberg, J.; Dave, G.; Larsson, Å. Leachates from plastic consumer products—Screening for toxicity with Daphnia magna. Chemosphere 2009, 74, 1195–1200. [Google Scholar] [CrossRef]
- Bejgarn, S.; MacLeod, M.; Bogdal, C.; Breitholtz, M. Toxicity of leachate from weathering plastics: An exploratory screening study with Nitocra spinipes. Chemosphere 2015, 132, 114–119. [Google Scholar] [CrossRef]
- Pandey, P.K.; Soupir, M.L. Assessing linkages between E. coli levels in streambed sediment and overlying water in an agricultural watershed in Iowa during the first heavy rain event of the season. Trans. ASABE 2014, 57, 1571. [Google Scholar]
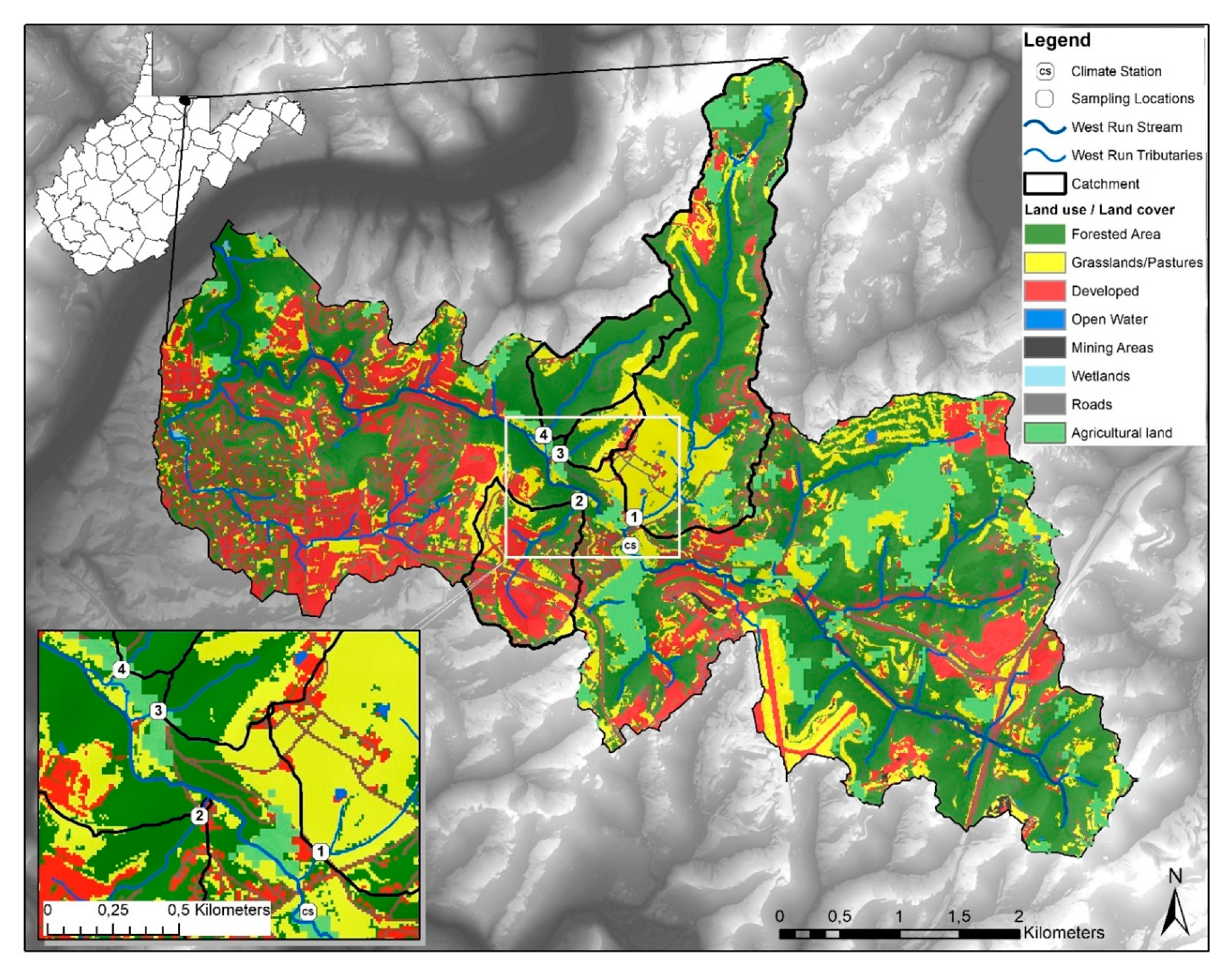
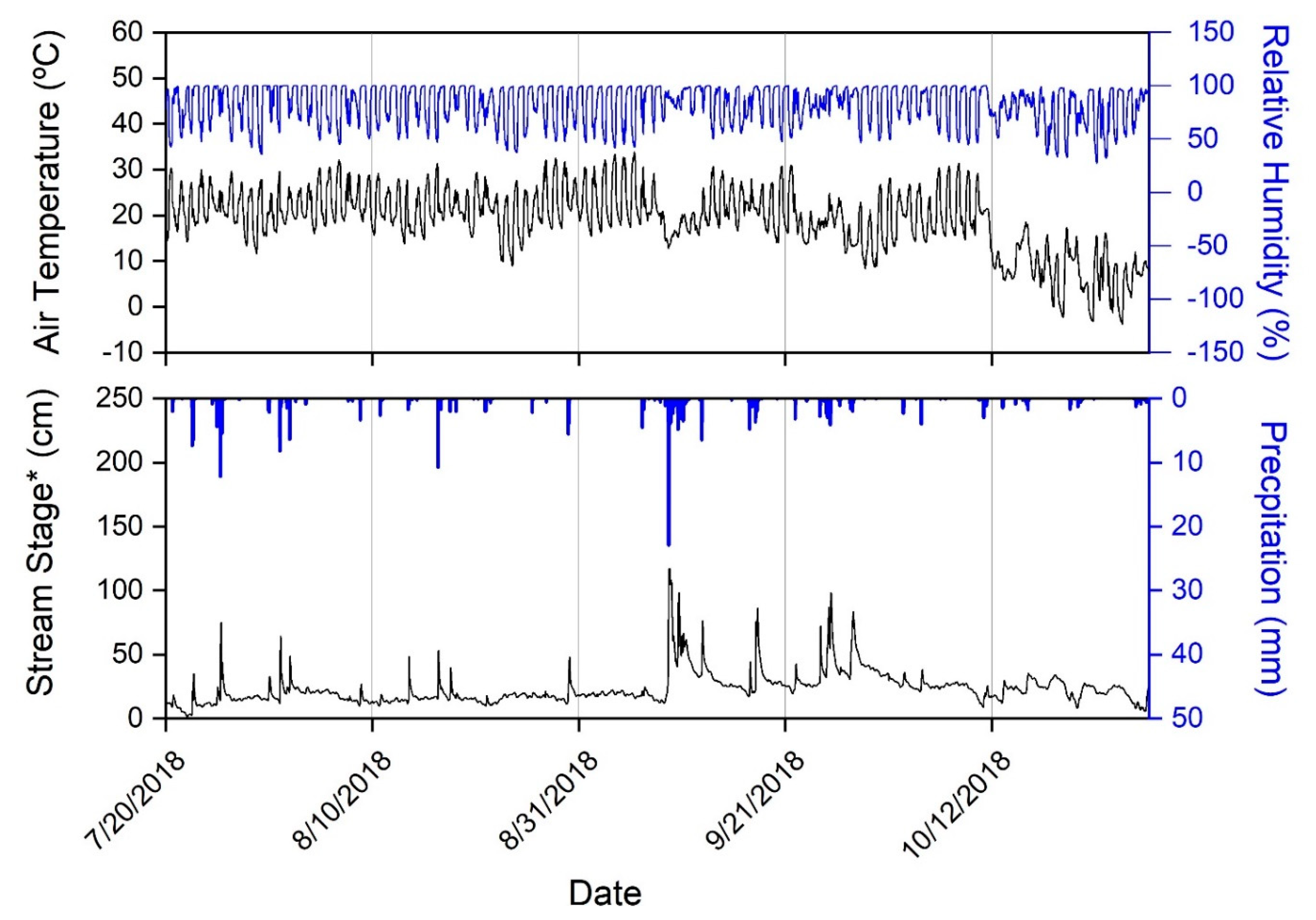
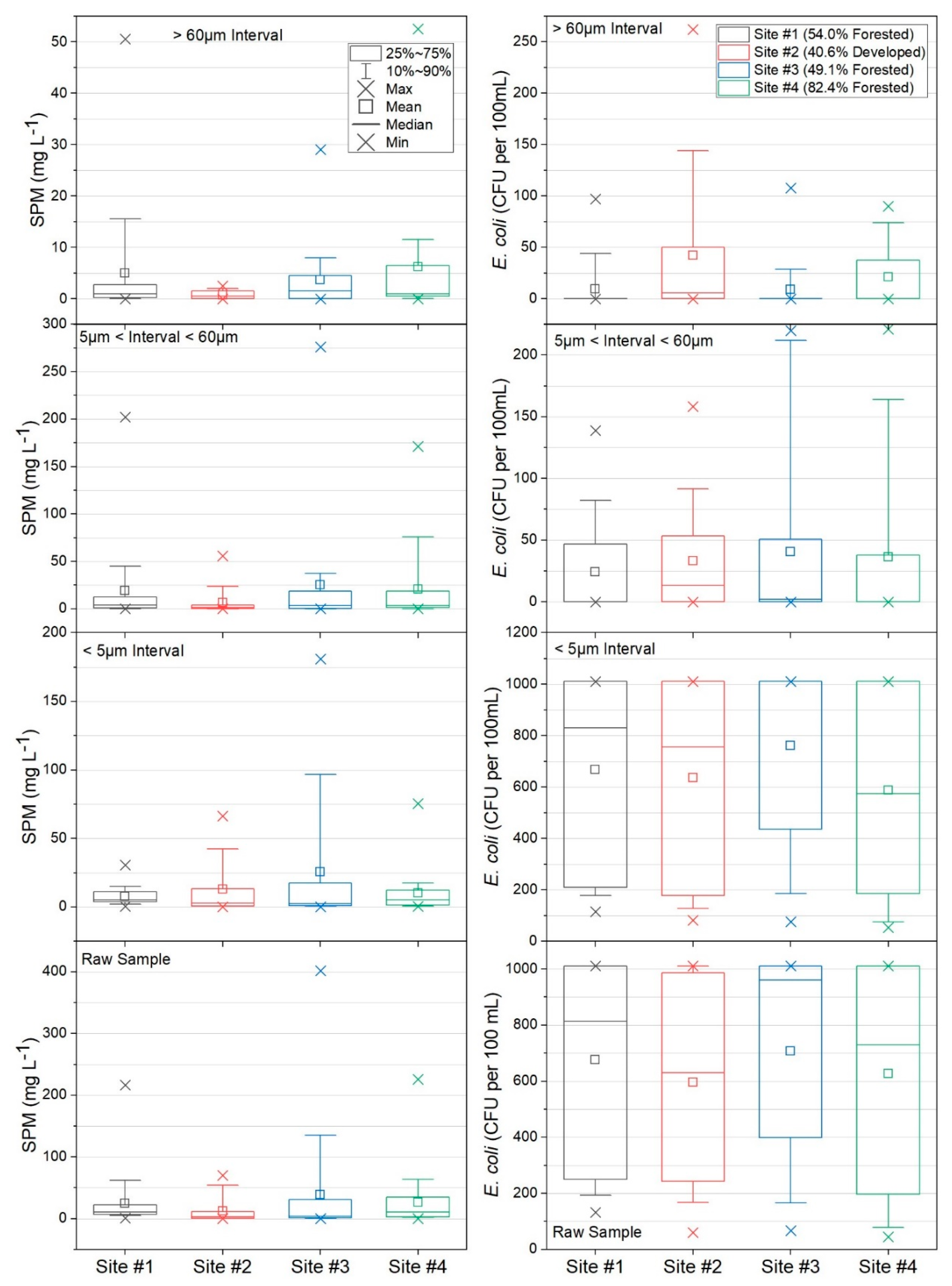
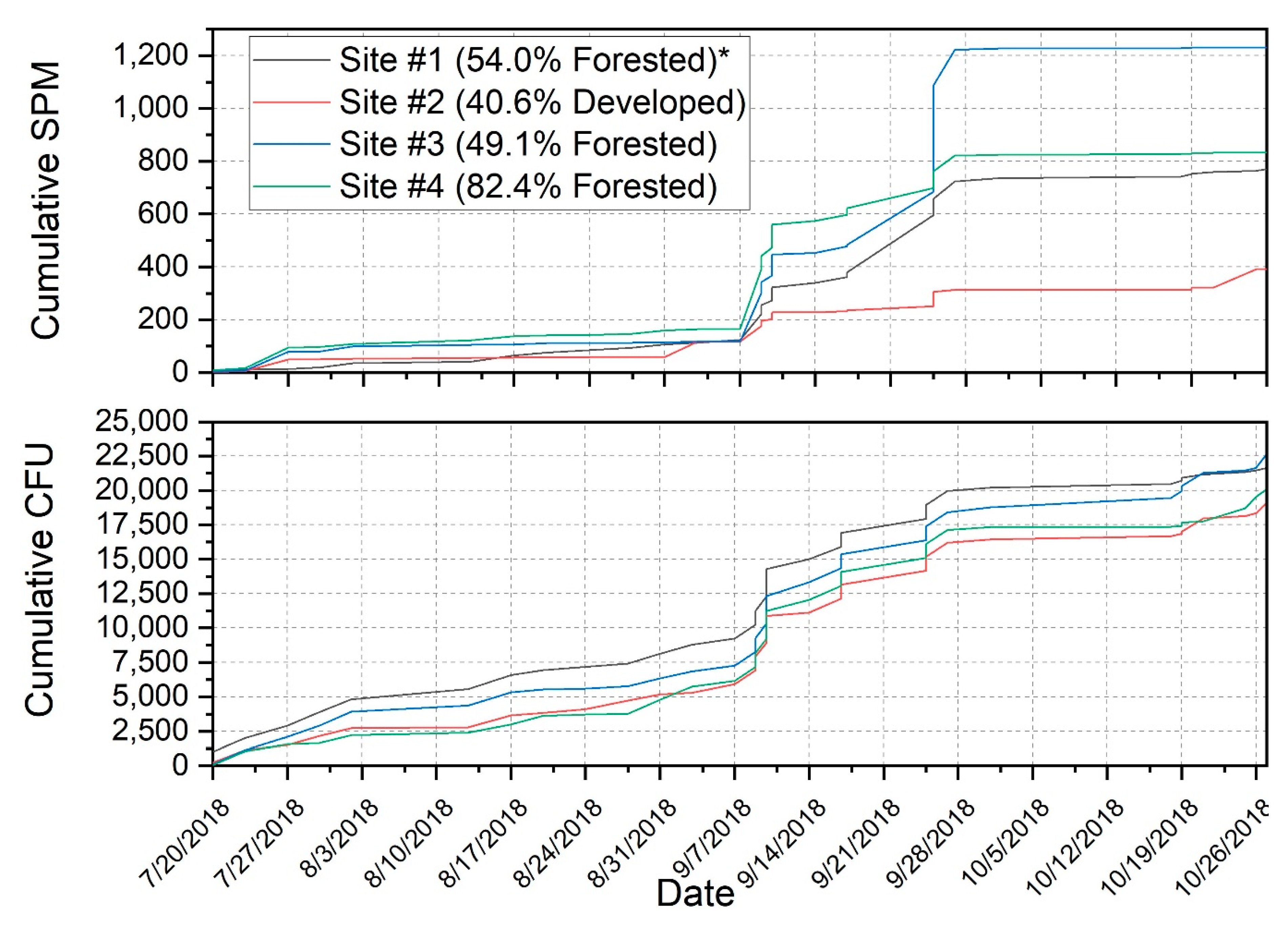
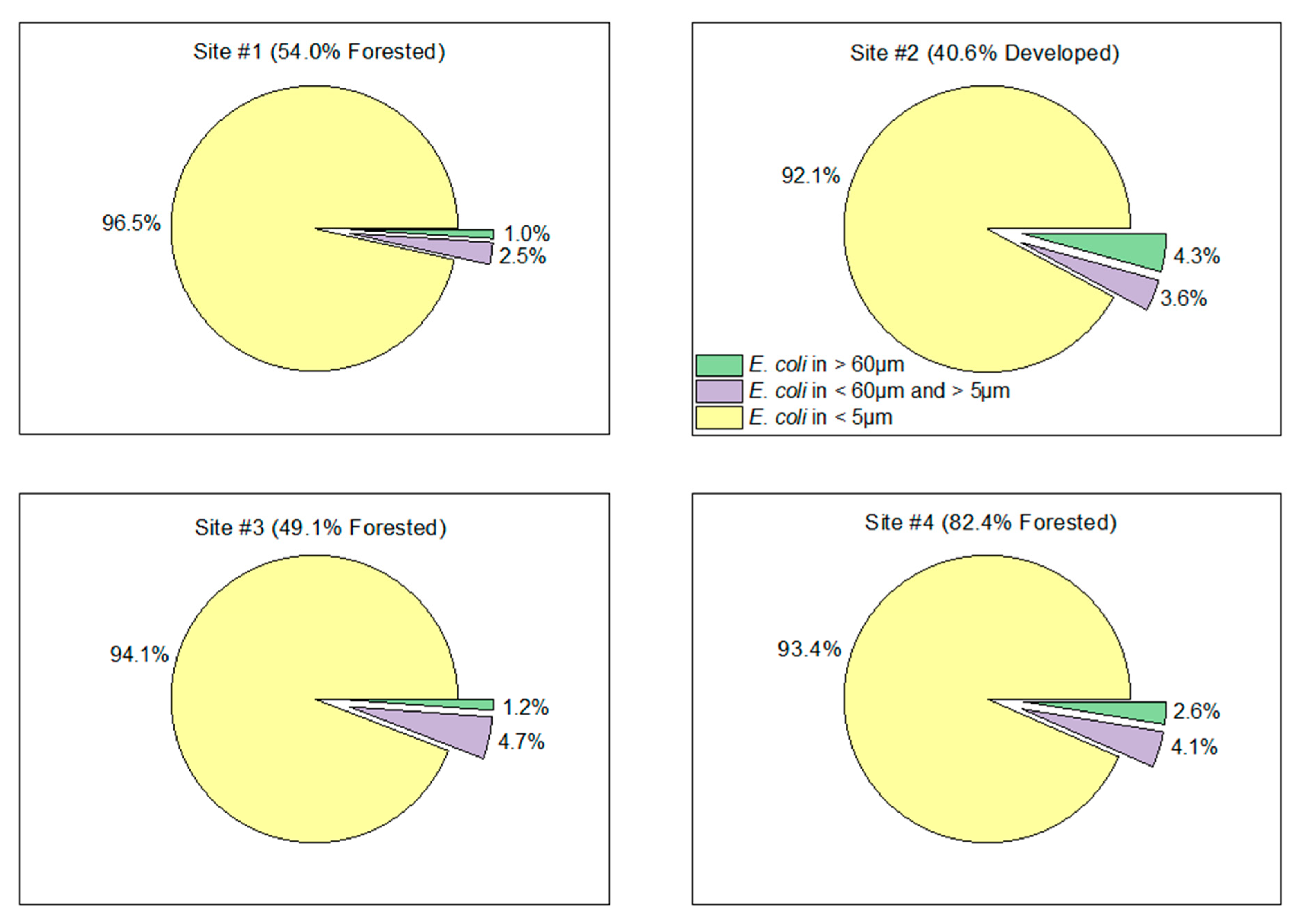

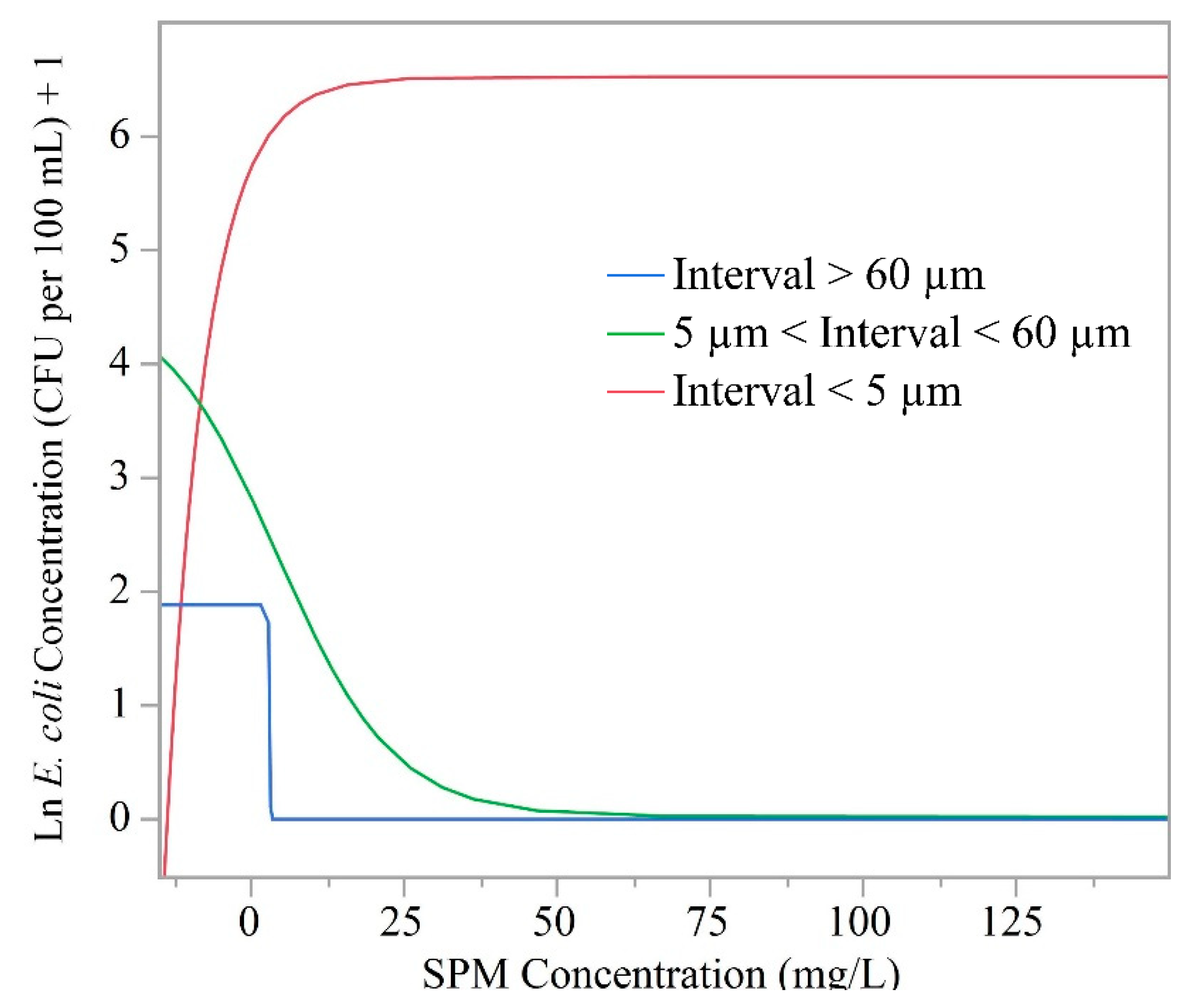
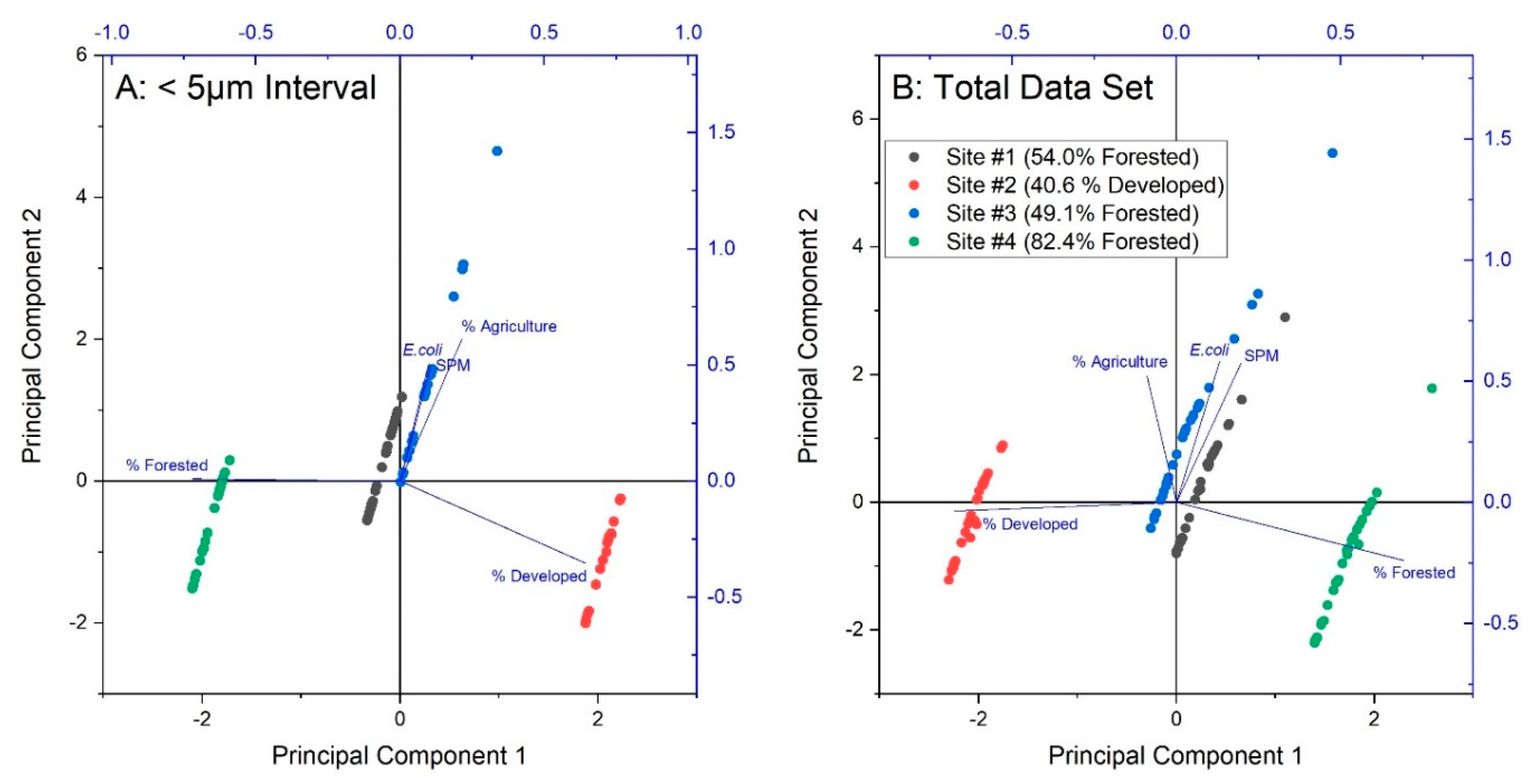
| Site Number and General Description | Forest (%) | Agriculture (%) | Developed (%) | Drainage Area (km2) |
|---|---|---|---|---|
| #1 (54.0% Forested) | 54.0 | 31.8 | 7.6 | 3.3 |
| #2 (40.6% Developed) | 27.6 | 20.8 | 40.6 | 1.0 |
| #3 (49.1% Forested) | 49.1 | 42.1 | 6.6 | 0.2 |
| #4 (82.4% Forested) | 82.4 | 13.3 | 0.8 | 0.7 |
| WRW (Total) | 50.1 | 22.6 | 19.0 | 23.3 |
| Variable | Eigenvalue | Percentage of Variance | Cumulative Variance |
|---|---|---|---|
| Smallest Interval (<5 µm) | |||
| E. coli concentration | 1.89 | 38% | 38% |
| SPM concentration | 1.35 | 27% | 65% |
| % Agriculture | 0.91 | 18% | 83% |
| % Forested | 0.84 | 17% | 100% |
| % Developed | 0 | 0 | 100% |
| Total Data Set | |||
| E. coli concentration | 1.90 | 38% | 38% |
| SPM concentration | 1.41 | 28% | 66% |
| % Agriculture | 1.01 | 20% | 86% |
| % Forested | 0.67 | 14% | 100% |
| % Developed | 0 | 0% | 100% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petersen, F.; Hubbart, J.A. Quantifying Escherichia coli and Suspended Particulate Matter Concentrations in a Mixed-Land Use Appalachian Watershed. Water 2020, 12, 532. https://doi.org/10.3390/w12020532
Petersen F, Hubbart JA. Quantifying Escherichia coli and Suspended Particulate Matter Concentrations in a Mixed-Land Use Appalachian Watershed. Water. 2020; 12(2):532. https://doi.org/10.3390/w12020532
Chicago/Turabian StylePetersen, Fritz, and Jason A. Hubbart. 2020. "Quantifying Escherichia coli and Suspended Particulate Matter Concentrations in a Mixed-Land Use Appalachian Watershed" Water 12, no. 2: 532. https://doi.org/10.3390/w12020532
APA StylePetersen, F., & Hubbart, J. A. (2020). Quantifying Escherichia coli and Suspended Particulate Matter Concentrations in a Mixed-Land Use Appalachian Watershed. Water, 12(2), 532. https://doi.org/10.3390/w12020532






