Performance of Field-Scale Phosphorus Removal Structures Utilizing Steel Slag for Treatment of Subsurface Drainage
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Characteristics
2.2. Slag Characterization
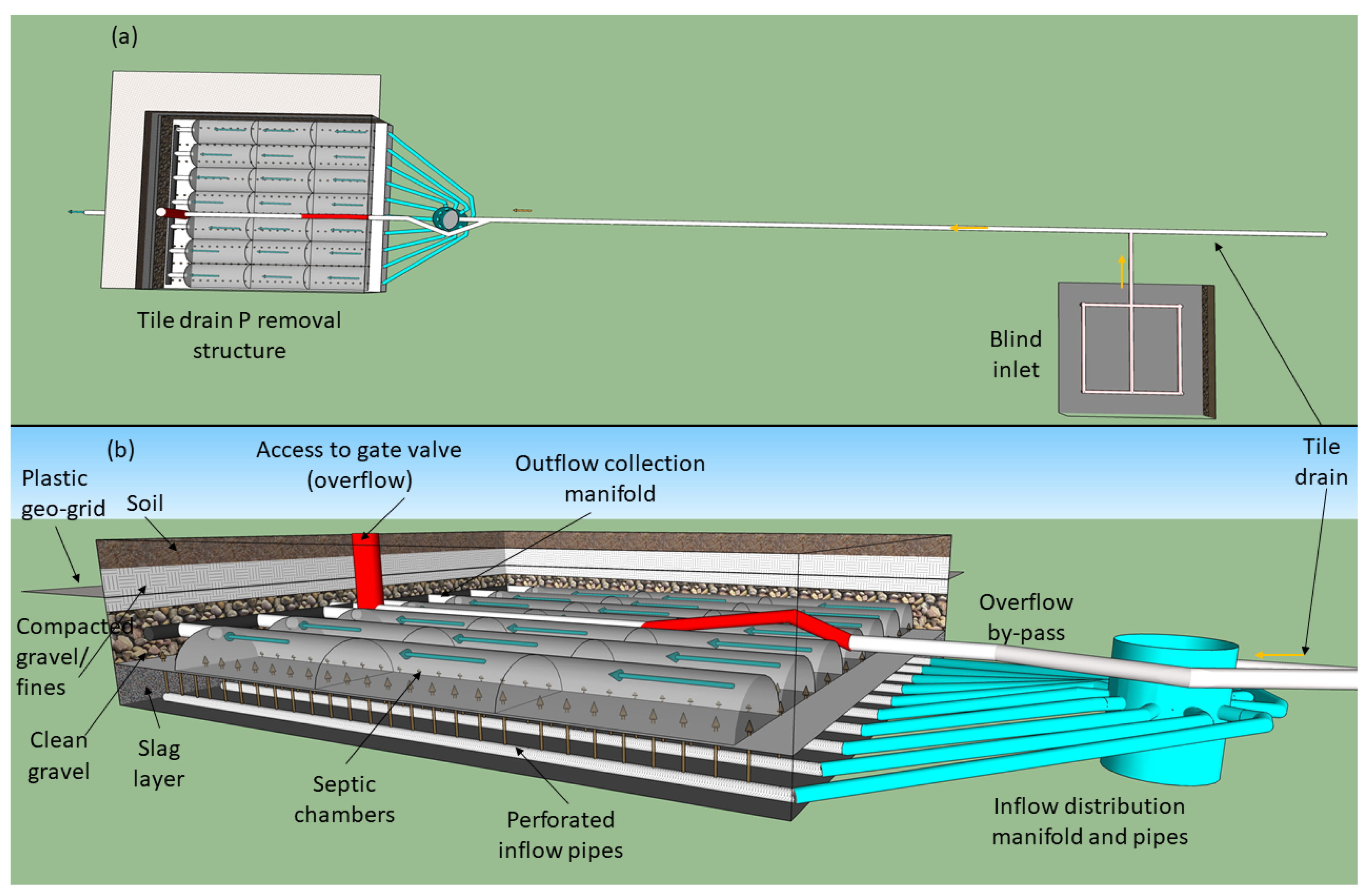
2.3. P Removal Structure Construction
2.3.1. Subsurface P Removal Structure
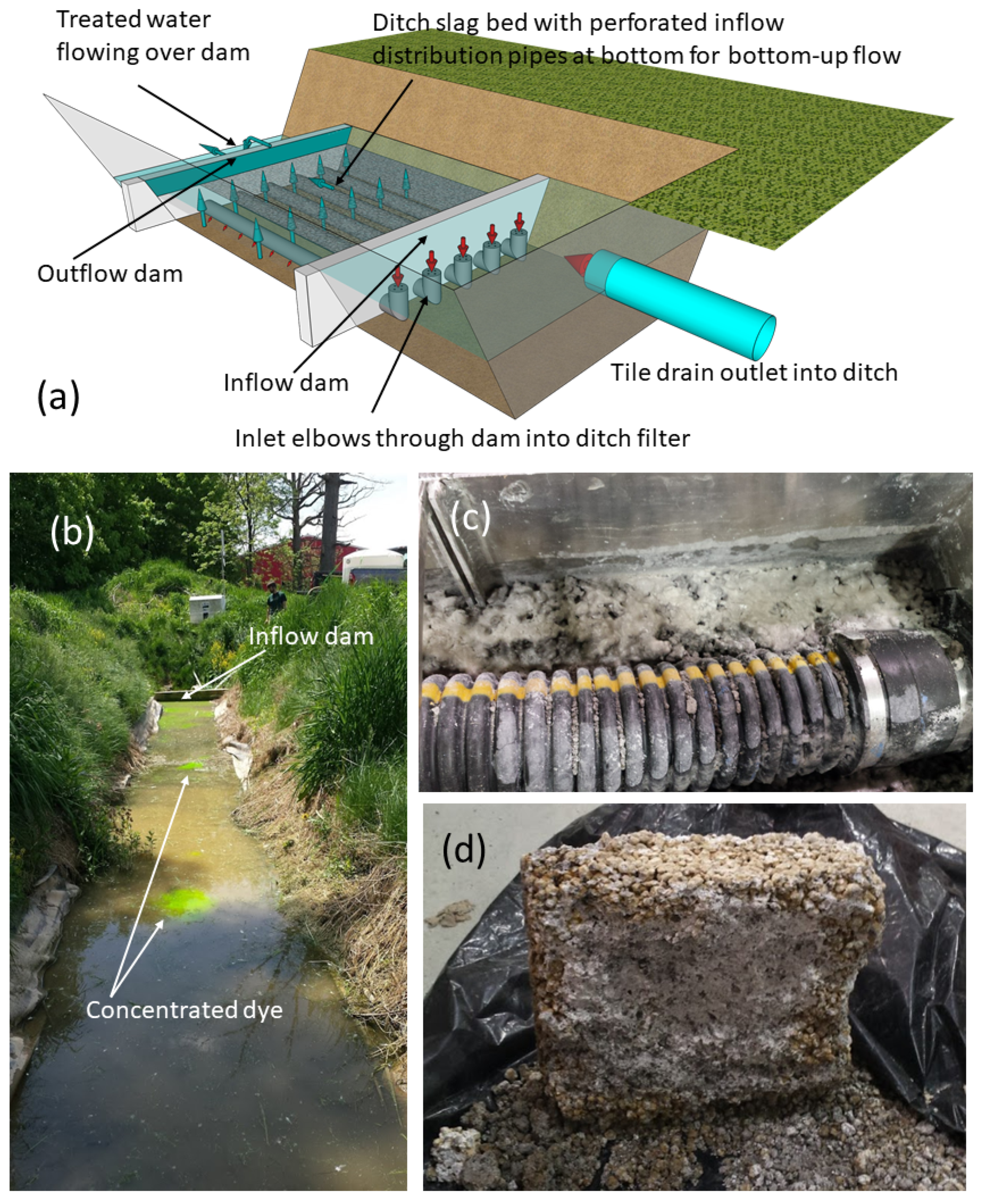
2.3.2. Ditch P Removal Structure for Treating Tile Drainage
2.4. Monitoring and Analysis
3. Results and Discussion
3.1. Slag Characterization
3.2. Performance of the Tile Drain Phosphorus Removal Structure
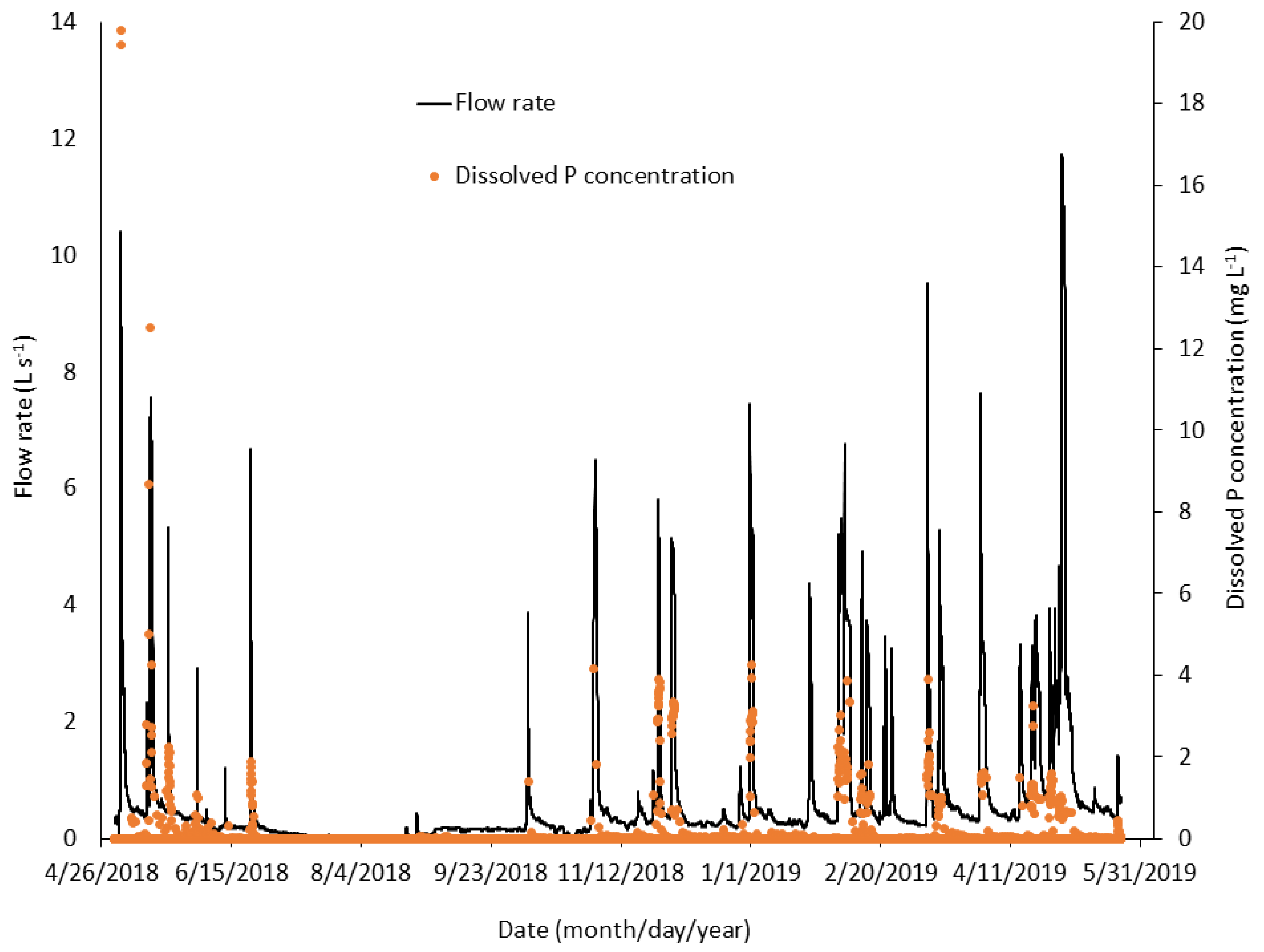
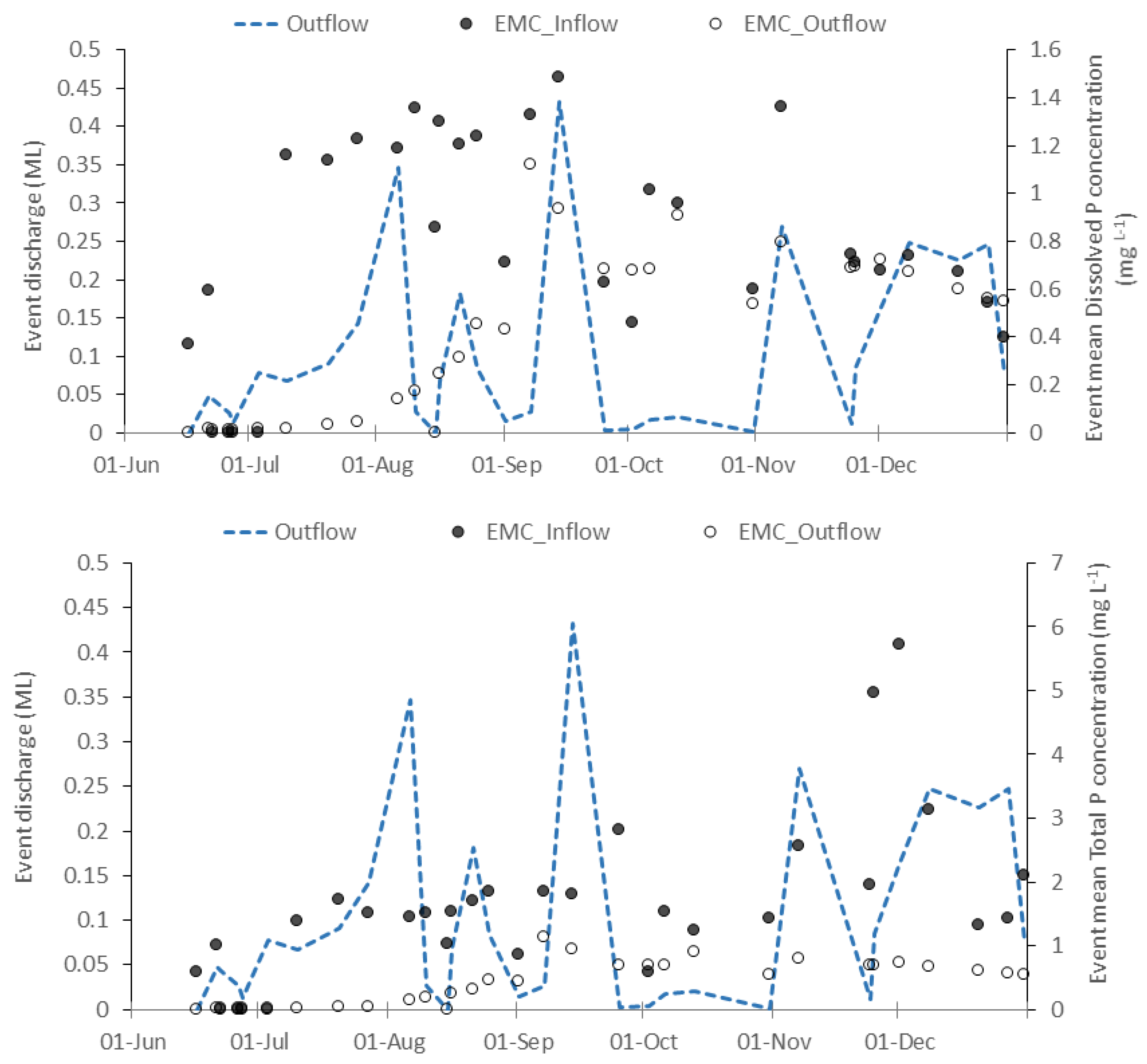
3.2.1. Inflow Rates, Concentrations, and Loads
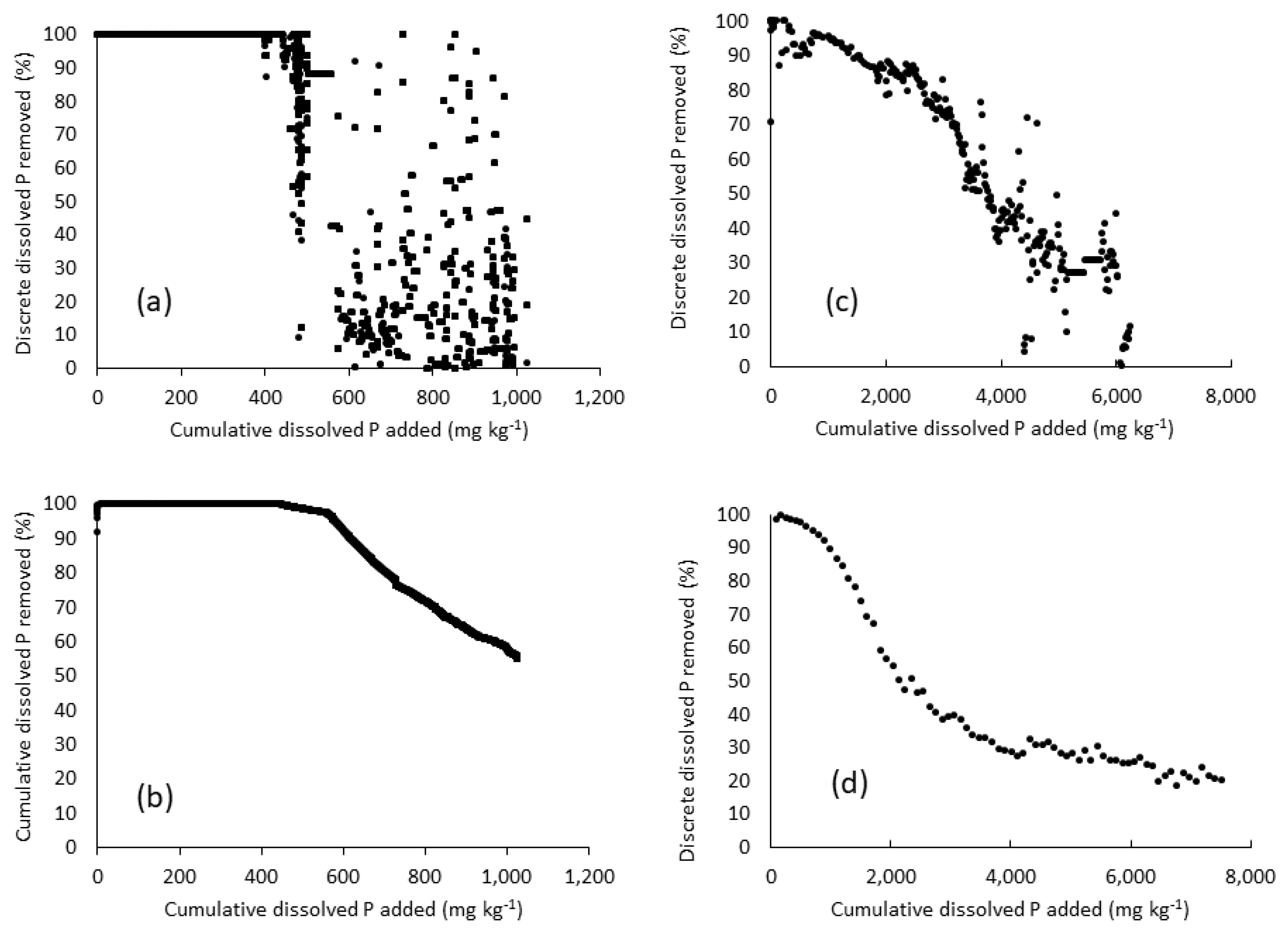
3.2.2. Phosphorus Removal, pH, and Metals
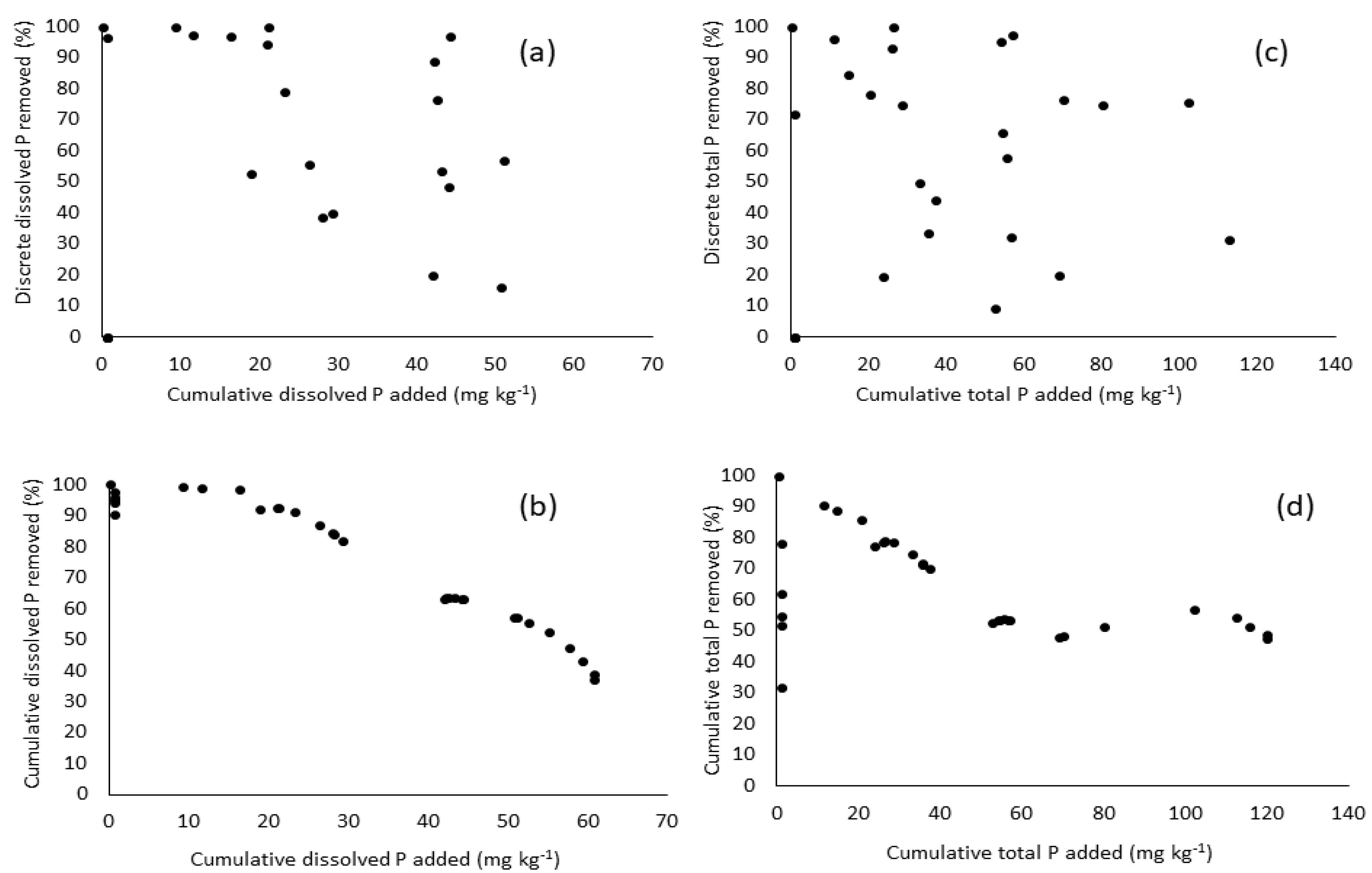
3.3. Performance of Ditch-Style Phosphorus Removal Structure
3.4. Potential Explantions for Underperformance of the Phosphorus Removal Structures
3.5. Economics of Dissolved Phosphorus Removal
4. Summary and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baker, D.B.; Confesor, R.; Ewing, D.E.; Johnson, L.T.; Kramer, J.W.; Merryfield, B.J. Phosphorus loading to Lake Erie from the Maumee, Sandusky and Cuyahoga rivers: The importance of bioavailability. J. Great Lakes Res. 2014, 40, 502–517. [Google Scholar]
- Kane, D.D.; Conroy, J.D.; Richards, R.P.; Baker, D.B.; Culver, D.A. Re-eutrophication of Lake Erie: Correlations between tributary nutrient loads and phytoplankton biomass. J. Great Lakes Res. 2014, 40, 496–501. [Google Scholar] [CrossRef]
- Scavia, D.; Allan, J.D.; Arend, K.K.; Bartell, S.; Beletsky, D.; Bosch, N.S.; Brandt, S.B.; Briland, R.D.; Daloğlu, I.; DePinto, J. V Assessing and addressing the re-eutrophication of Lake Erie: Central basin hypoxia. J. Great Lakes Res. 2014, 40, 226–246. [Google Scholar] [CrossRef]
- Turner, R.E.; Rabalais, N.N.; Justic, D. Predicting summer hypoxia in the northern Gulf of Mexico: Riverine, N., P., and Si loading. Mar. Pollut. Bull. 2006, 52, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Vadas, P.A.; Fiorellino, N.M.; Coale, F.J.; Kratochvil, R.; Mulkey, A.S.; McGrath, J.M. Estimating legacy soil phosphorus impacts on phosphorus loss in the Chesapeake Bay watershed. J. Environ. Qual. 2018, 47, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, R.P.; Johnson, L.T.; Wynne, T.T.; Baker, D.B. Forecasting annual cyanobacterial bloom biomass to inform management decisions in Lake Erie. J. Great Lakes Res. 2016, 42, 1174–1183. [Google Scholar] [CrossRef]
- Matisoff, G.; Kaltenberg, E.M.; Steely, R.L.; Hummel, S.K.; Seo, J.; Gibbons, K.J.; Bridgeman, T.B.; Seo, Y.; Behbahani, M.; James, W.F. Internal loading of phosphorus in western Lake Erie. J. Great Lakes Res. 2016, 42, 775–788. [Google Scholar] [CrossRef]
- Preedy, N.; McTiernan, K.; Matthews, R.; Heathwaite, L.; Haygarth, P. Rapid incidental phosphorus transfers from grassland. J. Environ. Qual. 2001, 30, 2105–2112. [Google Scholar] [CrossRef]
- McDowell, R.W.; Catto, W. Alternative fertilisers and management to decrease incidental phosphorus loss. Environ. Chem. Lett. 2005, 2, 169–174. [Google Scholar] [CrossRef]
- Penn, C.J.; Mullins, G.L.; Zelazny, L.W.; Warren, J.G.; McGrath, J.M. Surface runoff losses of phosphorus from Virginia soils amended with Turkey manure using phytase and high available phosphorus corn diets. J. Environ. Qual. 2004, 33, 1431–1439. [Google Scholar]
- Haygarth, P.M.; Sharpley, A.N. Terminology for phosphorus transfer. J. Environ. Qual. 2000, 29, 10–15. [Google Scholar] [CrossRef]
- Haygarth, P.M.; Jarvie, H.P.; Powers, S.M.; Sharpley, A.N.; Elser, J.J.; Shen, J.; Peterson, H.M.; Chan, N.-I.; Howden, N.J.K.; Burt, T. Sustainable Phosphorus Management and the Need for a Long-Term Perspective: The Legacy Hypothesis; ACS: Washington, DC, USA, 2014. [Google Scholar]
- Jarvie, H.P.; Sharpley, A.N.; Spears, B.; Buda, A.R.; May, L.; Kleinman, P.J.A. Water Quality Remediation Faces Unprecedented Challenges from Legacy Phosphorus; ACS: Washington, DC, USA, 2013. [Google Scholar]
- Qin, Z.; Shober, A. The Challenges of Managing Legacy Phosphorus Losses from Manure-Impacted Agricultural Soils. Curr. Pollut. Rep. 2018, 4, 265–276. [Google Scholar] [CrossRef]
- Ross, J.A.; Herbert, M.E.; Sowa, S.P.; Frankenberger, J.R.; King, K.W.; Christopher, S.F.; Tank, J.L.; Arnold, J.G.; White, M.J.; Yen, H. A synthesis and comparative evaluation of factors influencing the effectiveness of drainage water management. Agric. Water Manag. 2016, 178, 366–376. [Google Scholar] [CrossRef]
- Williams, M.R.; King, K.W.; Fausey, N.R. Drainage water management effects on tile discharge and water quality. Agric. Water Manag. 2015, 148, 43–51. [Google Scholar] [CrossRef]
- Penn, C.J.; Bryant, R.B.; Callahan, M.P.; Mcgrath, J.M. Use of industrial by-products to sorb and retain phosphorus. Commun. Soil Sci. Plant Anal. 2011, 42, 633–644. [Google Scholar] [CrossRef]
- Penn, C.J.; Bryant, R.B. Application of phosphorus sorbing materials to streamside cattle loafing areas. J. Soil Water Conserv. 2006, 61, 303–310. [Google Scholar]
- Inamdar, S.P.; Mostaghimi, S.; Mcclellan, P.W.; Brannan, K.M. BMP impacts on sediment and nutrient yields from an agricultural watershed in the Coastal Plain Region. Trans. ASAE 2001, 44, 1191–1200. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Courcelles, B.; Comeau, Y. Phosphorus removal by steel slag filters: Modeling dissolution and precipitation kinetics to predict longevity. Environ. Sci. Technol. 2014, 48, 7486–7493. [Google Scholar] [CrossRef]
- Allred, B.J.; Martinez, L.R.; Gamble, D.L. Phosphate removal from agricultural drainage water using an iron oxyhydroxide filter material. Water Air Soil Pollut. 2017, 228, 240. [Google Scholar] [CrossRef]
- Sibrell, P.L.; Kehler, T. Phosphorus removal from aquaculture effluents at the Northeast Fishery Center in Lamar, Pennsylvania using iron oxide sorption media. Aquac. Eng. 2016, 72, 45–52. [Google Scholar] [CrossRef]
- Groenenberg, J.E.; Chardon, W.J.; Koopmans, G.F. Reducing phosphorus loading of surface water using iron-coated sand. J. Environ. Qual. 2013, 42, 250–259. [Google Scholar] [CrossRef] [PubMed]
- McDowell, R.W.; Sharpley, A.N.; Bourke, W. Treatment of drainage water with industrial by-products to prevent phosphorus loss from tile-drained land. J. Environ. Qual. 2008, 37, 1575–1582. [Google Scholar] [CrossRef] [PubMed]
- Penn, C.; Bowen, J.; McGrath, J.; Nairn, R.; Fox, G.; Brown, G.; Wilson, S.; Gill, C. Evaluation of a universal flow-through model for predicting and designing phosphorus removal structures. Chemosphere 2016, 151, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Penn, C.J.; Bryant, R.B.; Kleinman, P.J.A.; Allen, A.L. Removing dissolved phosphorus from drainage ditch water with phosphorus sorbing materials. J. Soil Water Conserv. 2007, 62, 269–276. [Google Scholar]
- Penn, C.; Chagas, I.; Klimeski, A.; Lyngsie, G. A review of phosphorus removal structures: How to assess and compare their performance. Water 2017, 9, 583. [Google Scholar] [CrossRef]
- Weber, D.; Drizo, A.; Twohig, E.; Bird, S.; Ross, D. Upgrading constructed wetlands phosphorus reduction from a dairy effluent using electric arc furnace steel slag filters. Water Sci. Technol. 2007, 56, 135–143. [Google Scholar] [CrossRef]
- Erickson, A.; Gulliver, J.; Weiss, P. Phosphate removal from agricultural tile drainage with iron enhanced sand. Water 2017, 9, 672. [Google Scholar] [CrossRef]
- Penn, C.J.; Bowen, J.M. Design and Construction of Phosphorus Removal Structures for Improving Water Quality; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 3319586580. [Google Scholar]
- Drizo, A.; Forget, C.; Chapuis, R.P.; Comeau, Y. Phosphorus removal by electric arc furnace steel slag and serpentinite. Water Res. 2006, 40, 1547–1554. [Google Scholar] [CrossRef]
- Karczmarczyk, A.; Bus, A.; Baryła, A. Filtration curtains for phosphorus harvesting from small water bodies. Ecol. Eng. 2016, 86, 69–74. [Google Scholar] [CrossRef]
- Penn, C.; McGrath, J.; Bowen, J.; Wilson, S. Phosphorus removal structures: A management option for legacy phosphorus. J. Soil Water Conserv. 2014, 69, 51A–56A. [Google Scholar] [CrossRef]
- Klimeski, A.; Uusitalo, R.; Turtola, E. Screening of Ca-and Fe-rich materials for their applicability as phosphate-retaining filters. Ecol. Eng. 2014, 68, 143–154. [Google Scholar] [CrossRef]
- Canga, E.; Heckrath, G.J.; Kjaergaard, C. Agricultural drainage filters. II. Phosphorus retention and release at different flow rates. Water Air Soil Pollut. 2016, 227, 276. [Google Scholar] [CrossRef]
- Penn, C.J.; McGrath, J.M.; Rounds, E.; Fox, G.; Heeren, D. Trapping phosphorus in runoff with a phosphorus removal structure. J. Environ. Qual. 2012, 41, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Bell, G.E.; Penn, C.J.; Moss, J.Q.; Payton, M.E. Phosphorus reduction in turfgrass runoff using a steel slag trench filter system. Crop Sci. 2014, 54, 1859–1867. [Google Scholar] [CrossRef]
- Shilton, A.N.; Elmetri, I.; Drizo, A.; Pratt, S.; Haverkamp, R.G.; Bilby, S.C. Phosphorus removal by an ‘active’slag filter—A decade of full scale experience. Water Res. 2006, 40, 113–118. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Lida, F.; Comeau, Y. Improving phosphorus removal of conventional septic tanks by a recirculating steel slag filter. Water Qual. Res. J. 2015, 50, 211–218. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Wallace, S.; Comeau, Y. Removal of phosphorus, fluoride and metals from a gypsum mining leachate using steel slag filters. Water Res. 2013, 47, 1512–1520. [Google Scholar] [CrossRef]
- Vohla, C.; Kõiv, M.; Bavor, H.J.; Chazarenc, F.; Mander, Ü. Filter materials for phosphorus removal from wastewater in treatment wetlands—A review. Ecol. Eng. 2011, 37, 70–89. [Google Scholar] [CrossRef]
- Williams, M.R.; Livingston, S.J.; Penn, C.J.; Smith, D.R.; King, K.W.; Huang, C.-H. Controls of event-based nutrient transport within nested headwater agricultural watersheds of the western Lake Erie basin. J. Hydrol. 2018, 559, 749–761. [Google Scholar] [CrossRef]
- Smith, D.R.; King, K.W.; Johnson, L.; Francesconi, W.; Richards, P.; Baker, D.; Sharpley, A.N. Surface runoff and tile drainage transport of phosphorus in the midwestern United States. J. Environ. Qual. 2015, 44, 495–502. [Google Scholar] [CrossRef]
- Kõiv, M.; Liira, M.; Mander, Ü.; Mõtlep, R.; Vohla, C.; Kirsimäe, K. Phosphorus removal using Ca-rich hydrated oil shale ash as filter material–the effect of different phosphorus loadings and wastewater compositions. Water Res. 2010, 44, 5232–5239. [Google Scholar] [CrossRef] [PubMed]
- Dobbie, K.E.; Heal, K.V.; Aumonier, J.; Smith, K.A.; Johnston, A.; Younger, P.L. Evaluation of iron ochre from mine drainage treatment for removal of phosphorus from wastewater. Chemosphere 2009, 75, 795–800. [Google Scholar] [CrossRef] [PubMed]
- Klimeski, A.; Uusitalo, R.; Turtola, E. Variations in phosphorus retention by a solid material while scaling up its application. Environ. Technol. Innov. 2015, 4, 285–298. [Google Scholar] [CrossRef]
- Kirkkala, T.; Ventelä, A.-M.; Tarvainen, M. Long-term field-scale experiment on using lime filters in an agricultural catchment. J. Environ. Qual. 2012, 41, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; Francesconi, W.; Livingston, S.J.; Huang, C. Phosphorus losses from monitored fields with conservation practices in the Lake Erie Basin, USA. AMBIO 2015, 44, 319–331. [Google Scholar] [CrossRef]
- Feyereisen, G.W.; Francesconi, W.; Smith, D.R.; Papiernik, S.K.; Krueger, E.S.; Wente, C.D. Effect of replacing surface inlets with blind or gravel inlets on sediment and phosphorus subsurface drainage losses. J. Environ. Qual. 2015, 44, 594–604. [Google Scholar] [CrossRef]
- Smith, D.R.; Livingston, S.J. Managing farmed closed depressional areas using blind inlets to minimize phosphorus and nitrogen losses. Soil Use Manag. 2013, 29, 94–102. [Google Scholar] [CrossRef]
- Gonzalez, J.M.; Smith, D.R.; Livingston, S.; Warnemuende-Pappas, E.; Zwonitzer, M. Blind inlets: Conservation practices to reduce herbicide losses from closed depressional areas. J. Soils Sediments 2016, 16, 1921–1932. [Google Scholar] [CrossRef]
- Sims, J.T.; Maguire, R.O.; Leytem, A.B.; Gartley, K.L.; Pautler, M.C. Evaluation of Mehlich 3 as an agri-environmental soil phosphorus test for the Mid-Atlantic United States of America. Soil Sci. Soc. Am. J. 2002, 66, 2016–2032. [Google Scholar] [CrossRef]
- Duncan, E.W.; King, K.W.; Williams, M.R.; LaBarge, G.; Pease, L.A.; Smith, D.R.; Fausey, N.R. Linking soil phosphorus to dissolved phosphorus losses in the Midwest. Agric. Environ. Lett. 2017, 2. [Google Scholar] [CrossRef]
- USEPA. Acid Digestion of Sediments, Sludges, and Soils (Method 3050B). In Test Methods Eval. Solid Waste, Physic Methods; USEPA: Washington, DC, USA, 1996. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Stoner, D.; Penn, C.; McGrath, J.; Warren, J. Phosphorus removal with by-products in a flow-through setting. J. Environ. Qual. 2012, 41, 654–663. [Google Scholar] [CrossRef]
- Nelson, N.S. An acid-persulfate digestion procedure for determination of phosphorus in sediments. Commun. Soil Sci. Plant Anal. 1987, 18, 359–369. [Google Scholar] [CrossRef]
- Penn, C.J.; McGrath, J.M. Predicting phosphorus sorption onto steel slag using a flow-through approach with application to a pilot scale system. J. Water Resour. Prot. 2011, 3, 235. [Google Scholar] [CrossRef]
- National Slag Association. A Guide For The Use Of Steel Slag in Agriculture and for Reclamation of Acid Lands; National Slag Association: Pleasant Grove, UT, USA, 2019. [Google Scholar]
- Yildirim, I.Z.; Prezzi, M. Chemical, mineralogical, and morphological properties of steel slag. Adv. Civ. Eng. 2011, 2011. [Google Scholar] [CrossRef]
- Zuo, M.; Renman, G.; Gustafsson, J.P.; Klysubun, W. Phosphorus removal by slag depends on its mineralogical composition: A comparative study of AOD and EAF slags. J. Water Process Eng. 2018, 25, 105–112. [Google Scholar] [CrossRef]
- Drizo, A.; Comeau, Y.; Forget, C.; Chapuis, R.P. Phosphorus saturation potential: A parameter for estimating the longevity of constructed wetland systems. Environ. Sci. Technol. 2002, 36, 4642–4648. [Google Scholar] [CrossRef]
- Bowden, L.I.; Jarvis, A.P.; Younger, P.L.; Johnson, K.L. Phosphorus removal from waste waters using basic oxygen steel slag. Environ. Sci. Technol. 2009, 43, 2476–2481. [Google Scholar] [CrossRef]
- Eveborn, D.; Gustafsson, J.P.; Hesterberg, D.; Hillier, S. XANES speciation of P in environmental samples: An assessment of filter media for on-site wastewater treatment. Environ. Sci. Technol. 2009, 43, 6515–6521. [Google Scholar] [CrossRef]
- Usepa, A. Plain English Guide to the EPA Part 503 Biosolids Rule. U.S. Patent 93003A, 04 Febuary 2020. [Google Scholar]
- King, K.W.; Williams, M.R.; Johnson, L.T.; Smith, D.R.; LaBarge, G.A.; Fausey, N.R. Phosphorus availability in Western Lake Erie Basin drainage waters: Legacy evidence across spatial scales. J. Environ. Qual. 2017, 46, 466–469. [Google Scholar] [CrossRef]
- Sharpley, A.N.; Kleinman, P.J.A.; Heathwaite, A.L.; Gburek, W.J.; Folmar, G.J.; Schmidt, J.P. Phosphorus loss from an agricultural watershed as a function of storm size. J. Environ. Qual. 2008, 37, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.; Broch, K.; Andersen, M.R. First flush effects in an urban catchment area in Aalborg. Water Sci. Technol. 1998, 37, 251–257. [Google Scholar] [CrossRef]
- Kato, T.; Kuroda, H.; Nakasone, H. Runoff characteristics of nutrients from an agricultural watershed with intensive livestock production. J. Hydrol. 2009, 368, 79–87. [Google Scholar] [CrossRef]
- Bertrand-Krajewski, J.-L.; Chebbo, G.; Saget, A. Distribution of pollutant mass vs volume in stormwater discharges and the first flush phenomenon. Water Res. 1998, 32, 2341–2356. [Google Scholar] [CrossRef]
- Pionke, H.B.; Gburek, W.J.; Sharpley, A.N. Critical source area controls on water quality in an agricultural watershed located in the Chesapeake Basin. Ecol. Eng. 2000, 14, 325–335. [Google Scholar] [CrossRef]
- Williams, M.R.; King, K.W.; Duncan, E.W.; Pease, L.A.; Penn, C.J. Fertilizer placement and tillage effects on phosphorus concentration in leachate from fine-textured soils. Soil Tillage Res. 2018, 178, 130–138. [Google Scholar] [CrossRef]
- Williams, M.R.; King, K.W.; Ford, W.; Buda, A.R.; Kennedy, C.D. Effect of tillage on macropore flow and phosphorus transport to tile drains. Water Resour. Res. 2016, 52, 2868–2882. [Google Scholar] [CrossRef]
- Pease, L.A.; King, K.W.; Williams, M.R.; LaBarge, G.A.; Duncan, E.W.; Fausey, N.R. Phosphorus export from artificially drained fields across the Eastern Corn Belt. J. Great Lakes Res. 2018, 44, 43–53. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Wallace, S.; Comeau, Y. Model of phosphorus precipitation and crystal formation in electric arc furnace steel slag filters. Environ. Sci. Technol. 2012, 46, 1465–1470. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Courcelles, B.; Pasquier, P.; Comeau, Y. Numerical simulations with the P-Hydroslag model to predict phosphorus removal by steel slag filters. Water Res. 2017, 126, 421–432. [Google Scholar] [CrossRef]
- Claveau-Mallet, D.; Boutet, É.; Comeau, Y. Steel slag filter design criteria for phosphorus removal from wastewater in decentralized applications. Water Res. 2018, 143, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Shober, A.L.; Scheckel, K.G.; Penn, C.J.; Turner, K.C. Mechanisms of Phosphorus Removal by Phosphorus Sorbing Materials. J. Environ. Qual. 2018, 47, 1232–1241. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, W.L. Chemical Equilibria in Soils.; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 1979; ISBN 0471027049. [Google Scholar]
- Dunets, C.S.; Zheng, Y.; Dixon, M. Use of phosphorus-sorbing materials to remove phosphate from greenhouse wastewater. Environ. Technol. 2015, 36, 1759–1770. [Google Scholar] [CrossRef]
- Kostura, B.; Kulveitova, H.; Leško, J. Blast furnace slags as sorbents of phosphate from water solutions. Water Res. 2005, 39, 1795–1802. [Google Scholar] [CrossRef] [PubMed]
- Liira, M.; Kõiv, M.; Mander, Ü.; Mõtlep, R.; Vohla, C.; Kirsimäe, K. Active filtration of phosphorus on Ca-rich hydrated oil shale ash: Does longer retention time improve the process? Environ. Sci. Technol. 2009, 43, 3809–3814. [Google Scholar] [CrossRef] [PubMed]
- Omernik, J.M.; Powers, C.F. Total alkalinity of surface waters—A national map. Ann. Assoc. Am. Geogr. 1983, 73, 133–136. [Google Scholar] [CrossRef]
- Fausey, N.R.; Brown, L.C.; Belcher, H.W.; Kanwar, R.S. Drainage and water quality in Great Lakes and cornbelt states. J. Irrig. Drain. Eng. 1995, 121, 283–288. [Google Scholar] [CrossRef]
- Jones, C.S.; Kult, K.J. Use alkalinity monitoring to optimize bioreactor performance. J. Environ. Qual. 2016, 45, 855–865. [Google Scholar] [CrossRef]
- Olsen, S.R. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; US Department of Agriculture: Washington, DC, USA, 1954.
- Jiang, F.; Beck, M.B.; Cummungs, R.G.; Rowles, K.; Russell, D.L. Estimation of costs of phosphorus removal in wastewater treatment facilities: Adaptation of existing facilities. Water Policy Work. Pap. 2005, 11, 1–45. [Google Scholar]


| Parameter | Units | Value |
|---|---|---|
| Physical properties | ||
| >4.8 mm | % | 43.3 |
| 2–4.8 mm | % | 35.5 |
| 1–2 mm | % | 17.6 |
| 0.5 to 1 mm | % | 0.77 |
| 0.2 to 0.5 mm | % | 0.62 |
| 0.15 to 0.2 mm | % | 0.33 |
| 0.12 to 0.15 mm | % | 0.26 |
| <0.12 mm | % | 1.7 |
| Bulk density | g cm−3 | 1.69 |
| Chemical properties | ||
| pH | 11.53 | |
| Buffer index | eq L−1 | 0.104 |
| Electrical conductivity | mS cm−1 | 3.40 |
| Ca | g kg−1 | 185 |
| Mg | g kg−1 | 29.7 |
| Fe | g kg−1 | 110 |
| Al | g kg−1 | 9.49 |
| Mn | g kg−1 | 14.6 |
| P | g kg−1 | 2.32 |
| K | mg kg−1 | 226 |
| Cr | mg kg−1 | 1727 |
| Ni | mg kg−1 | 464 |
| Pb | mg kg−1 | 3.03 |
| Zn | mg kg−1 | 362 |
| B | mg kg−1 | 75.7 |
| S | mg kg−1 | 345 |
| Flow | Inflow Dissolved P | Dissolved P Removed | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Event | Date | Peak Flow Rate | Total Volume | Peak Concentration | Mean Flow-Weighted Concentration | Load | % of Total | Load | Discrete % | Minimum RT | |
| L s−1 | ML | % of total | mg L−1 | mg L−1 | kg | kg | minutes | ||||
| Storm 1 | 5/2/2018 | 10.4 | 949 | 4.32 | 19.80 | 14.4 | 13.69 | 39.0 | 13.7 | 100 | 13 |
| Storm 2 | 5/13/2018 | 2.3 | 81 | 0.37 | 2.78 | 1.72 | 0.14 | 0.40 | 0.13 | 97 | 58 |
| Storm 3 | 5/14/2018 | 7.2 | 161 | 0.73 | 12.53 | 7.95 | 1.28 | 3.65 | 1.28 | 100 | 19 |
| Storm 4 | 5/15/2018 | 7.6 | 625 | 2.85 | 4.26 | 1.52 | 0.95 | 2.71 | 0.83 | 87 | 18 |
| Storm 5 | 5/21/2018 | 5.3 | 172 | 0.78 | 2.25 | 1.31 | 0.22 | 0.64 | 0.21 | 96 | 25 |
| Storm 6 | 6/1/2018 | 2.9 | 96 | 0.44 | 1.06 | 0.29 | 0.03 | 0.08 | 0.03 | 91 | 46 |
| Storm 7 | 6/12/2018 | 1.2 | 40 | 0.18 | 0.33 | 0.13 | 0.01 | 0.01 | 0.01 | 100 | 110 |
| Storm 8 | 6/22/2018 | 6.7 | 263 | 1.20 | 1.89 | 0.78 | 0.21 | 0.59 | 0.17 | 83 | 20 |
| Storm 9 | 10/6/2018 | 3.9 | 351 | 1.60 | 1.39 | 0.55 | 0.19 | 0.55 | 0.18 | 91 | 35 |
| Storm 10 | 11/1/2018 | 6.5 | 900 | 4.10 | 4.15 | 2.72 | 2.45 | 6.98 | 1.92 | 78 | 21 |
| Storm 11 | 11/25/2018 | 11.7 | 540 | 2.46 | 3.90 | 2.47 | 1.89 | 5.38 | 0.26 | 13 | 12 |
| Storm 12 | 12/1/2018 | 5.2 | 764 | 3.48 | 3.36 | 2.47 | 1.89 | 5.38 | 0.26 | 14 | 26 |
| Storm 13 | 12/31/2018 | 7.4 | 739 | 3.37 | 4.27 | 2.64 | 1.95 | 5.57 | 0.15 | 8 | 18 |
| Storm 14 | 2/3/2019 | 6.8 | 1721 | 7.84 | 3.85 | 1.89 | 3.25 | 9.25 | 0.65 | 20 | 20 |
| Storm 15 | 2/12/2019 | 4.9 | 234 | 1.06 | 1.58 | 0.96 | 0.22 | 0.64 | 0.00 | 1 | 27 |
| Storm 16 | 2/14/2019 | 3.7 | 364 | 1.66 | 1.82 | 0.96 | 0.35 | 1.00 | −0.03 | −9 | 36 |
| Storm 17 | 2/20/2019 | 1.9 | 32 | 0.15 | 1.30 | 1.29 | 0.04 | 0.12 | 0.02 | 59 | 70 |
| Storm 18 | 2/21/2019 | 3.5 | 144 | 0.65 | 1.50 | 0.90 | 0.13 | 0.37 | −0.03 | −20 | 39 |
| Storm 19 | 2/24/2019 | 3.3 | 132 | 0.60 | 1.70 | 1.66 | 0.22 | 0.62 | 0.18 | 83 | 41 |
| Storm 20 | 3/9/2019 | 9.5 | 1444 | 6.58 | 3.90 | 1.12 | 1.62 | 4.61 | 0.04 | 2 | 14 |
| Storm 21 | 3/30/2019 | 7.6 | 642 | 2.92 | 1.62 | 1.20 | 0.77 | 2.20 | −0.03 | −3 | 18 |
| Storm 22 | 4/18/2019 | 3.8 | 891 | 4.06 | 3.24 | 1.08 | 0.97 | 2.75 | 0.22 | 23 | 35 |
| Storm 23 | 4/25/2019 | 11.7 | 2918 | 13.3 | 1.58 | 0.64 | 1.86 | 5.29 | −0.45 | −24 | 12 |
| Storm 24 | 5/22/2019 | 1.4 | 86 | 0.39 | 0.44 | 0.13 | 0.01 | 0.03 | 0.00 | −18 | 95 |
| Baseflow | NA | NA | 7673 | 34.9 | NA | 0.17 | 1.28 | 3.66 | −0.40 | −31 | NA |
| Storm Event Total | NA | 14,289 | 65.1 | NA | 2.37 | 33.8 | 96.3 | 19.6 | 58 | NA | |
| TOTAL | NA | 21,962 | NA | NA | 1.60 | 35.1 | NA | 19.2 | 55 | NA | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Penn, C.; Livingston, S.; Shedekar, V.; King, K.; Williams, M. Performance of Field-Scale Phosphorus Removal Structures Utilizing Steel Slag for Treatment of Subsurface Drainage. Water 2020, 12, 443. https://doi.org/10.3390/w12020443
Penn C, Livingston S, Shedekar V, King K, Williams M. Performance of Field-Scale Phosphorus Removal Structures Utilizing Steel Slag for Treatment of Subsurface Drainage. Water. 2020; 12(2):443. https://doi.org/10.3390/w12020443
Chicago/Turabian StylePenn, Chad, Stan Livingston, Vinayak Shedekar, Kevin King, and Mark Williams. 2020. "Performance of Field-Scale Phosphorus Removal Structures Utilizing Steel Slag for Treatment of Subsurface Drainage" Water 12, no. 2: 443. https://doi.org/10.3390/w12020443
APA StylePenn, C., Livingston, S., Shedekar, V., King, K., & Williams, M. (2020). Performance of Field-Scale Phosphorus Removal Structures Utilizing Steel Slag for Treatment of Subsurface Drainage. Water, 12(2), 443. https://doi.org/10.3390/w12020443








