Planktonic Invertebrates in the Assessment of Long-Term Change in Water Quality of the Sorbulak Wastewater Disposal System (Kazakhstan)
Abstract
:1. Introduction
2. Materials and Methods
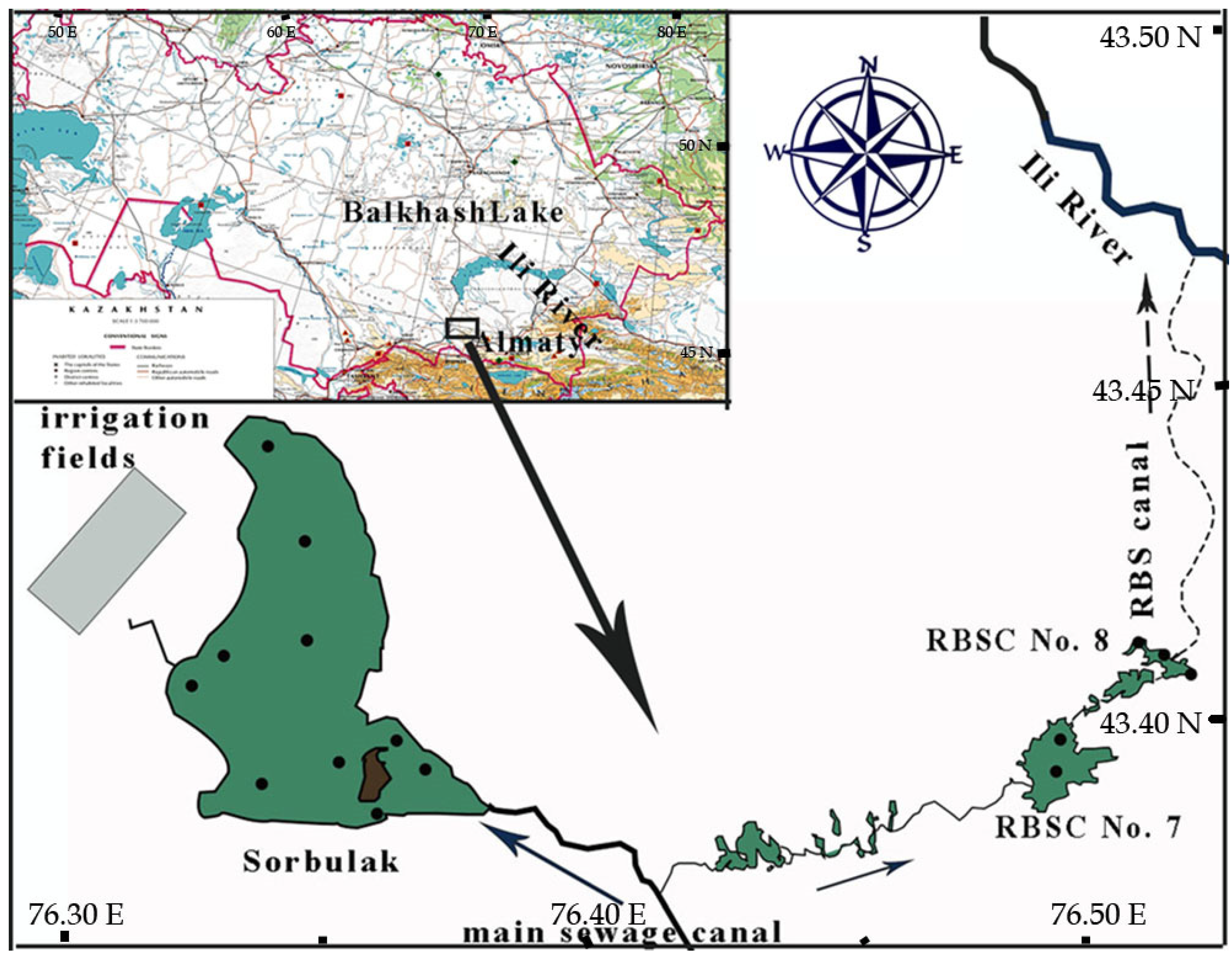
2.1. Description of Study Area
2.1.1. Climate
2.1.2. Physical and Geographical Characteristic of Sorbulak Wastewater Disposal System
2.2. Field Sampling
2.3. Laboratory Analysis
2.4. Statistical Analysis
3. Results
3.1. Hydrochemical and Toxicological Characteristics of the Sorbulak Wastewater Disposal System
3.2. The Structure of Zooplankton Communities
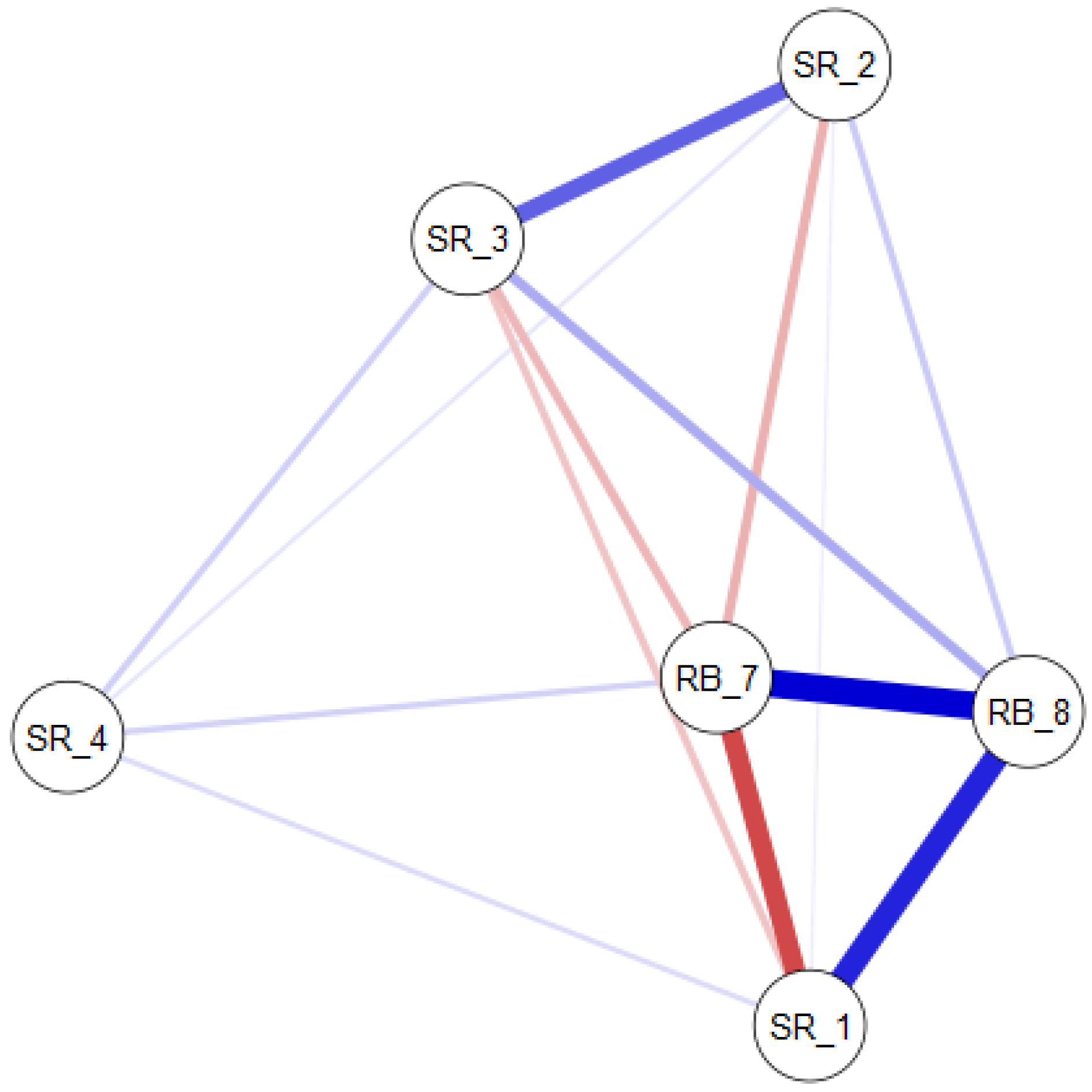
3.3. Nonparametric Correlation Analysis
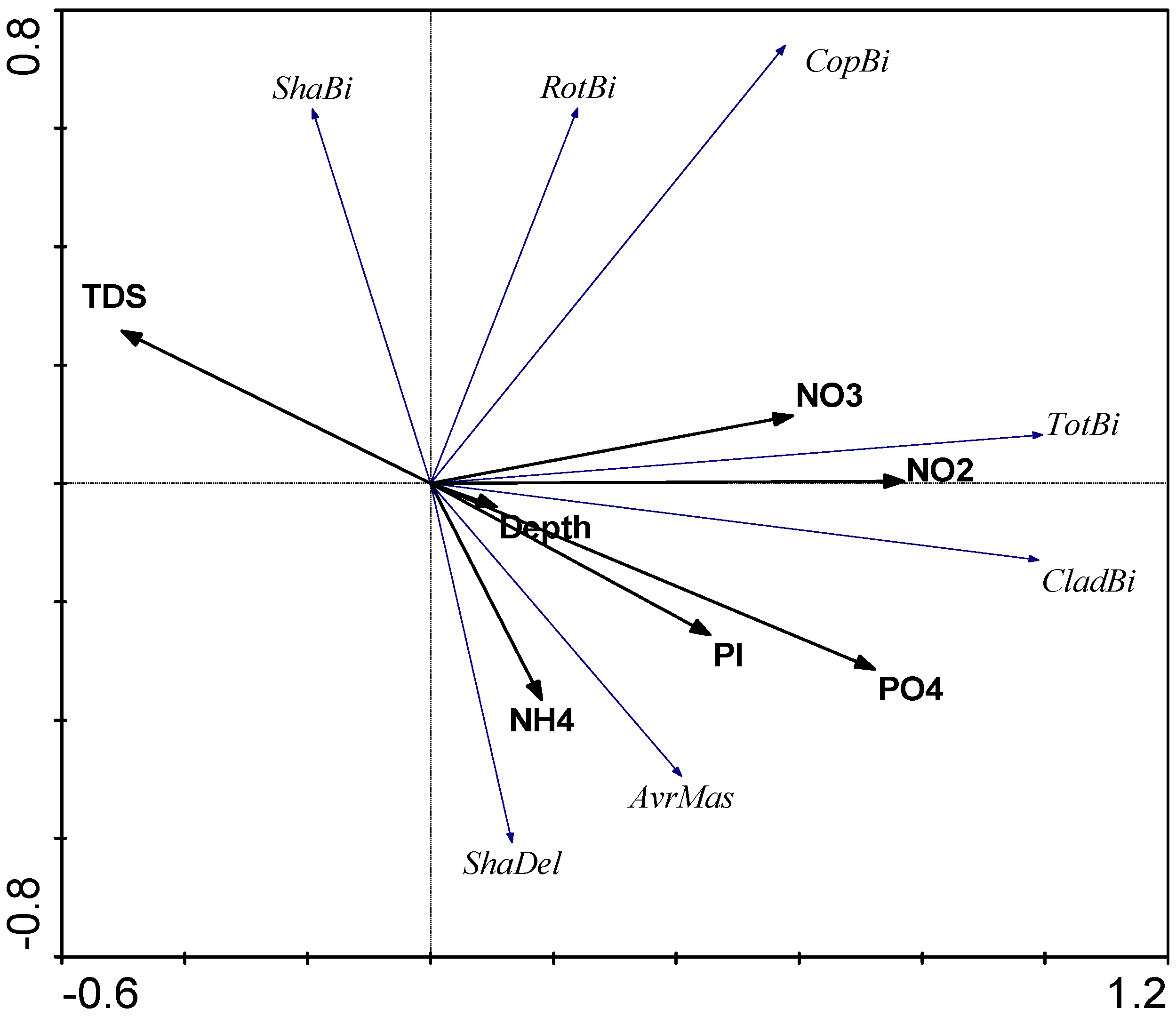
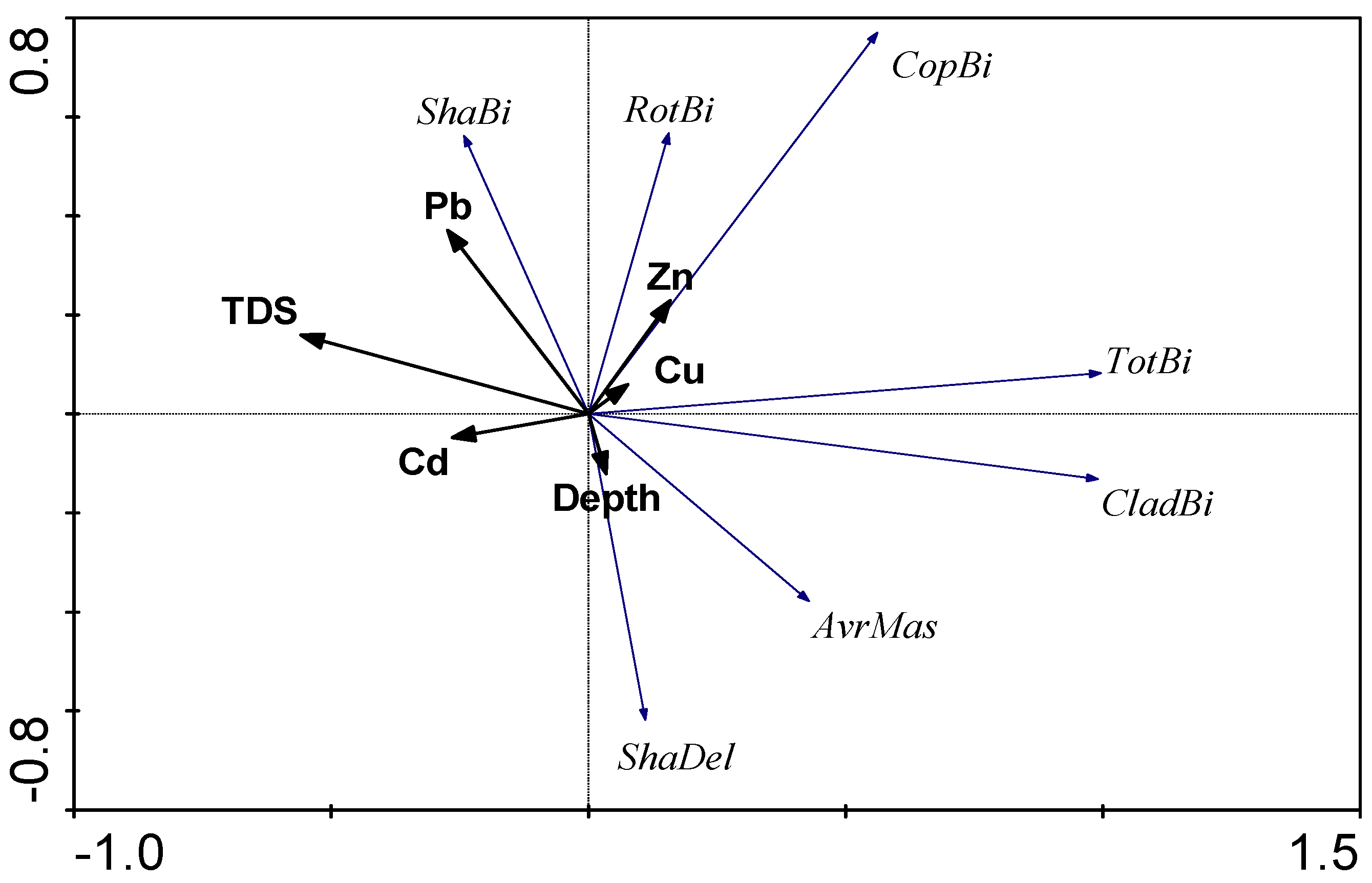
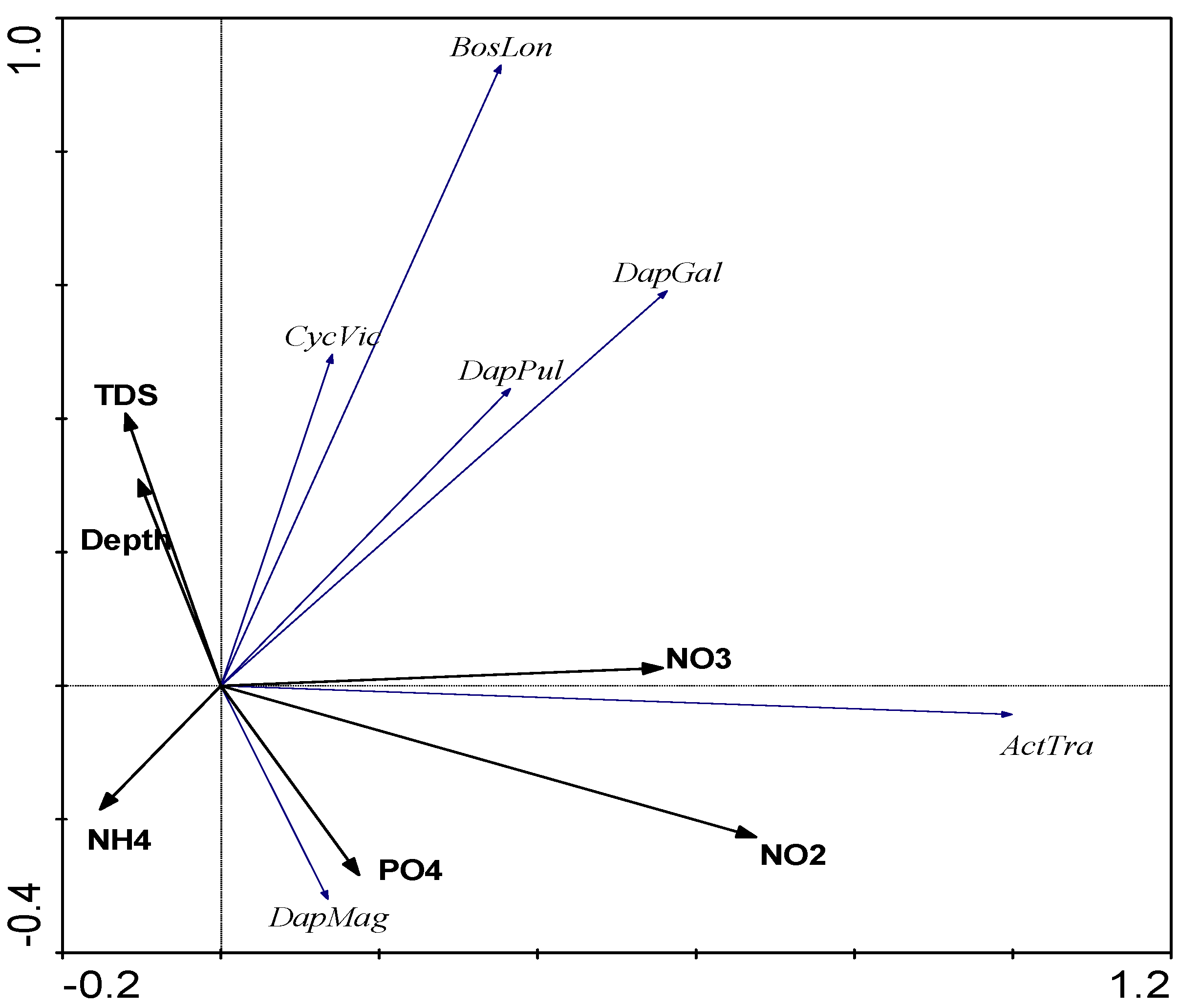
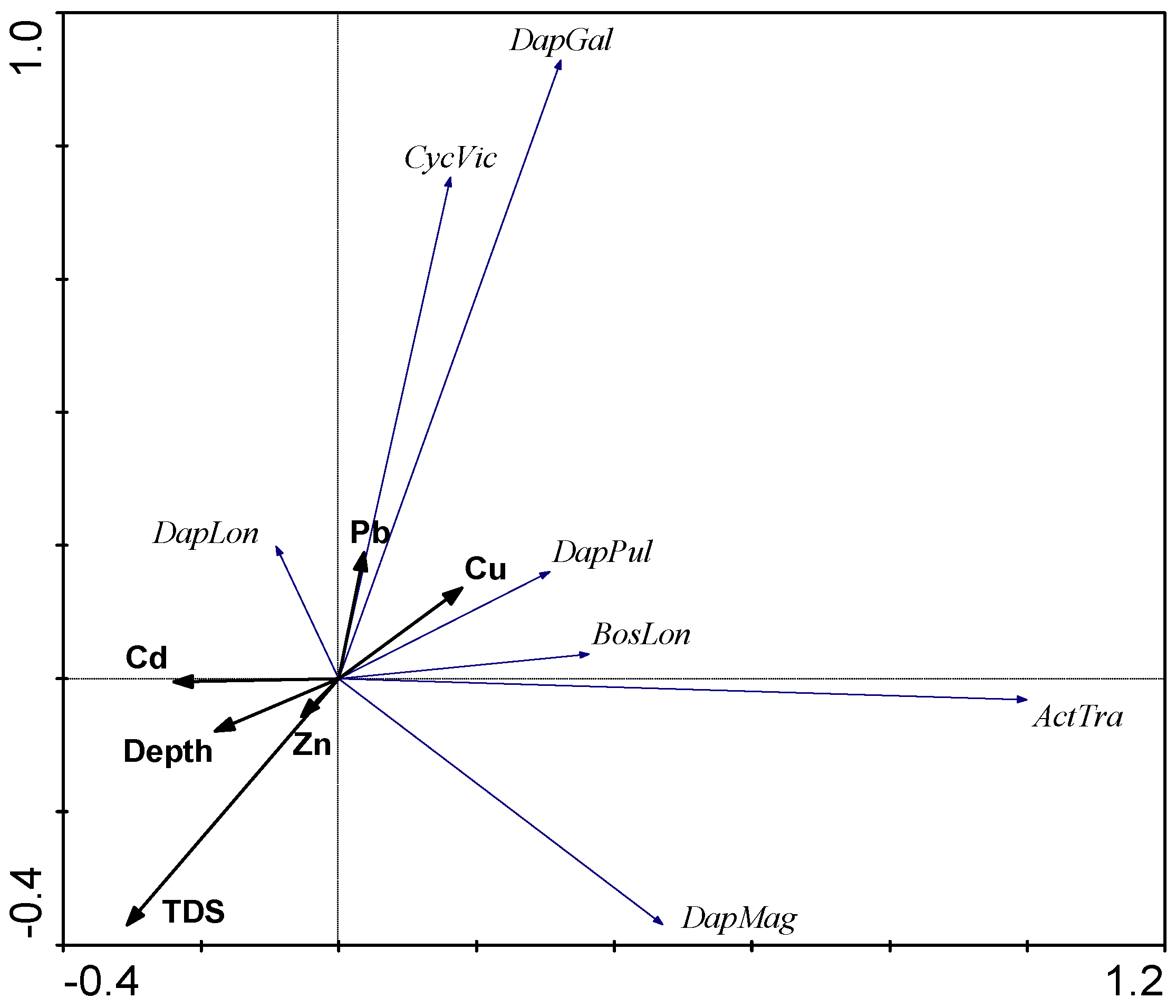
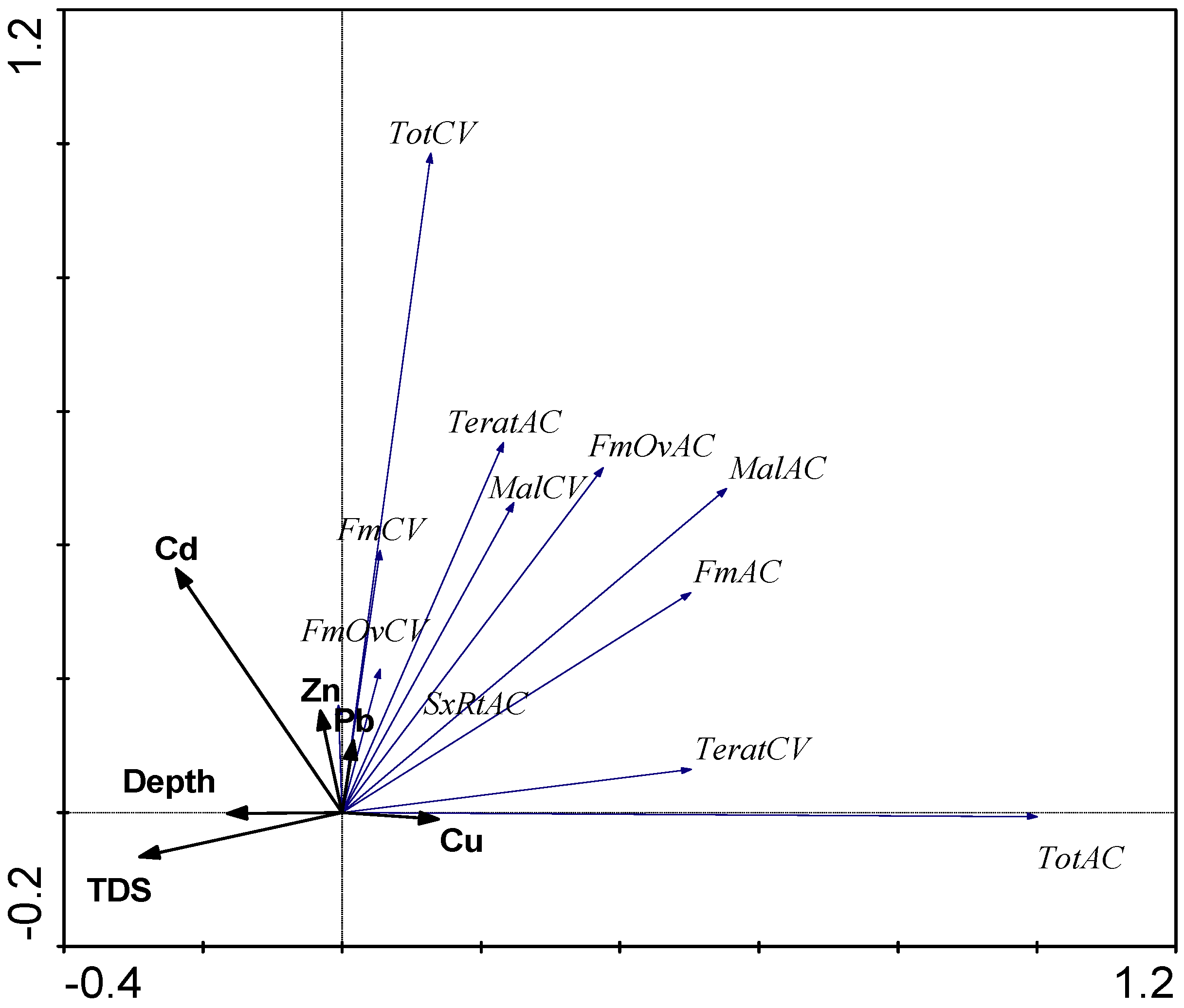
3.4. PCA Analysis
4. Discussion
4.1. Chemical Variables
4.2. Biological Variables
4.2.1. Species Composition and Quantitative Variables
4.2.2. Structural Variables
4.2.3. The Influence of External Factors on the Quantitative Variables and Structure of Zooplankton Communities
4.2.4. Indicator Role of Zooplankton in Assessing the Water Quality of Water Bodies with Multicomponent Pollution
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Drechsel, P.; Evans, A.E.V. Wastewater use in irrigated agriculture. Irrig. Drainage Syst. 2010, 24, 1–3. [Google Scholar] [CrossRef]
- Alfarra, A.; Kemp-Benedict, E.; Hötzl, H.; Sader, N.; Sonneveld, B. A Framework for Wastewater Reuse in Jordan: Utilizing a Modified Wastewater Reuse Index. Water Resour. Manag. 2011, 25, 1153–1167. [Google Scholar] [CrossRef] [Green Version]
- Nan, Z.; Cheng, G. Accumulation of Cd and Pb in spring wheat (Triticum aestivum L.) grown in calcareous soil irrigated with wastewater. Bull. Environ. Contam. Toxicol. 2001, 66, 748–754. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Katnoria, J.K.; Nagpal, A.K. Heavy metals in vegetables: Screening health risks involved in cultivation along wastewater drain and irrigating with wastewater. SpringerPlus 2016, 5, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Kaboosi, K. The assessment of treated wastewater quality and the effects of mid-term irrigation on soil physical and chemical properties (case study: Bandargaz-treated wastewater). Appl. Water Sci. 2017, 7, 2385–2396. [Google Scholar] [CrossRef] [Green Version]
- Kitajima, M.; Rachmadi, A.T.; Iker, B.C.; Haramoto, E.; Pepper, I.L.; Gerba, C.P. Occurrence and genetic diversity of human cosavirus in influent and effluent of wastewater treatment plants in Arizona. Arch. Virol. 2015, 160, 1775–1779. [Google Scholar] [CrossRef] [Green Version]
- Kocak, E. Investigation of potential genotoxic activity using the SOS Chromotest for real paracetamol wastewater and the wastewater treated by the Fenton process. J. Environ. Health Sci. Eng. 2015, 13, 66. [Google Scholar] [CrossRef] [Green Version]
- Matmuratov, S.A.; Bragin, B.I.; Stuge, T.S.; Troshina, T.T.; Akberdina, G.Z.; Krupa, E.G. To the assessment of the ecological situation in the area of the discharges of the Right-Bank Sorbulak Canal in the Ili River. In Geographic Foundations of Sustainable Development of the Republic of Kazakhstan; Kaganat: Almaty, Kazakhstan, 1998; pp. 281–286. (In Russian) [Google Scholar]
- Krupa, E.G.; Amirgaliev, N.A.; Shapovalov, V. Characteristics of the hydrocenosis of the Sorbulak storage pond in terms of toxicological and biological indicators. In Modern Problems of Aquatic Toxicology; Printhous: Borok, Russia, 2002; pp. 166–167. (In Russian) [Google Scholar]
- Berenzen, N.; Hűmmer, S.; Liess, M.; Schulz, R. Pesticide Peak Discharge from Wastewater Treatment Plants into Streams During the Main Period of Insecticide Application: Ecotoxicological Evaluation in Comparison to Run off. Bull. Environ. Contam. Toxicol. 2003, 70, 891–897. [Google Scholar] [CrossRef]
- Ibraev, T.T.; Lee, M.A. Some aspects of the influence of anthropogenic factors on the water resources of Kazakhstan. Water Manag. Kazakhstan 2010, 2, 20. (In Russian) [Google Scholar]
- Moore, J.W.; Ramamoorthy, S. Heavy Metals in Natural Waters Applied Monitoring and Impact Assessment; Springer: New York, NY, USA, 1984; p. 268. [Google Scholar]
- EC. The Directive 2000/60/EP of the European Parliament and of the Council establishing a framework for community action in the field of water policy. Off. J. Eur. Commun. 2000, L327. Available online: https://www.eea.europa.eu/policy-documents/directive-2000-60-ec-of (accessed on 22 December 2000).
- Papaioannou, A.; Mavridou, A.; Hadjichristodoulou, C.; Papastergiou, P.; Pappa, O.; Dovriki, E.; Rigas, I. Application of multivariate statistical methods for groundwater physicochemical and biological quality assessment in the context of public health. Environ. Monit. Assess. 2010, 170, 87–97. [Google Scholar] [CrossRef]
- Seitkaziev, A.S.; Seitkazieva, K.A. Criteria for assessing the quality of aquatic ecosystems in the Republic of Kazakhstan. In Bulletin of the Kazakh-German University; Special issue “Water Resources of Kazakhstan: Current State, Problems, and Ways of Their Solution”; Kazakh-German University: Almaty, Kazakhstan, 2014; Volume 2, pp. 94–101. [Google Scholar]
- Ochocka, A.; Pasztaleniec, A. Sensitivity of plankton indices to lake trophic conditions. Environ. Monit. Assess. 2016, 188, 622. [Google Scholar] [CrossRef] [Green Version]
- Andronikova, I.N. Structural and Functional Organization of Zooplankton of Lake Ecosystems of Various Trophic Types; Nauka: St. Petersburg, Russia, 1996; p. 189. (In Russian) [Google Scholar]
- Alvarado-Flores, J.; Rico-Martı´nez, R.; Adabache-Ortı´z, A.; Silva-Briano, M. Morphological alterations in the freshwater rotifer Brachionus calyciflorus Pallas 1766 (Rotifera: Monogononta) caused by vinclozolin chronic exposure. Ecotoxicology 2015, 24, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Dembowska, E.A.; Mieszczankin, T.; Napiórkowski, P. Changes of the phytoplankton community as symptoms of deterioration of water quality in a shallow lake. Environ. Monit. Assess. 2018, 190, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Svensson, O.; Bellamy, A.S.; Van den Brink, P.J.; Tedengren, M.; Gunnarsson, J.S. Assessing the ecological impact of banana farms on water quality using aquatic macroinvertebrate community composition. Environ. Sci. Pollut. Res. 2018, 25, 13373–13381. [Google Scholar] [CrossRef]
- Dostay, Z.D.; Tyumenev, S.D. Management of Water Supply and Sewerage of the City of Almaty; Euro Print: Almaty, Kazakhstan, 2009; p. 176. (In Russian) [Google Scholar]
- Bekkazinova, D.B.; Aleksanova, A.G. The content of heavy metals (Cd, Zn, Co, Ni, Cu) in the wastewater of the city of Almaty. Bull. Kazakh Natl. Med Univ. 2010, 2, 168–171. (In Russian) [Google Scholar]
- Tyumenev, S.D. Water Resources and Water Supply of the Territory of Kazakhstan; KazNTU: Almaty, Kazakhstan, 2008; p. 267. (In Russian) [Google Scholar]
- Timush, A.V. Seismotectonics of Kazakhstan lithosphere; Institute of Seismology: Almaty, Kazakhstan, 2011; p. 590. (In Russian) [Google Scholar]
- Krupa, E.; Slyvinskiy, G.; Barinova, S. The effect of climatic factors on the long-term dynamics of aquatic ecosystem of the Balkhash Lake (Kazakhstan, Central Asia). Adv. Stud. Biol. 2014, 6, 115–136. [Google Scholar] [CrossRef]
- Kiseleva, V.A. Seasonal dynamics of hydrocenoses of Lake M. Sorbulak (Almaty region) as a probable cause of summer-autumn migration of migratory birds. Selevinia 2014, 1–4, 137–143. (In Russian) [Google Scholar]
- Matmuratov, S.A.; Bragin, B.I.; Troshina, T.T.; Krupa, E.G. Features of the formation of the ecological and toxicological situation in the discharge zone of the Sorbulak canal into the Ili River. In Problems of Protection and Sustainable Use of Biodiversity of Fauna in Kazakhstan; Kaganat: Almaty, Kazakhstan, 1999; pp. 132–133. (In Russian) [Google Scholar]
- Krupa, E.G.; Stuge, T.S.; Akberdina, G.Z. Lower crustaceans from Sorbulak reservoirs under conditions of chronic wastewater impact. In Zoological Research in Kazakhstan; Institute of Zoology: Almaty, Kazakhstan, 2002; pp. 176–177. (In Russian) [Google Scholar]
- Rogovskaya, E.G. (Ed.) Scientific and Applied Reference Book on the Climate of the USSR; Series 3; Gidrometeoizdat: Leningrad, Russia, 1989; Issue 18, p. 440. (In Russian) [Google Scholar]
- Myakisheva, N.V. Multi-Criteria Classification of Lakes; Russian State Hydrometeorological University: St. Petersburg, Russia, 2009; p. 160. [Google Scholar]
- ISO 8467:1993. Water Quality. Determination of Permanganate Index. 1993. Available online: https://www.iso.org/standard/15669.html (accessed on 10 October 2020).
- Kazakhstan Hydrometeorological Service. Surface and Underground Resources Water, Their Use, and Quality (2017 Year); Annual Publication: Almaty, Kazakhstan, 2018. (In Russian) [Google Scholar]
- Romanova, S.M. Non-Drainage Reservoirs of Kazakhstan. In Hydrochemical Regime; Kazakh University: Almaty, Kazakhstan, 2008; Volume 1, p. 250. (In Russian) [Google Scholar]
- Semenov, A.D. Guidance on the Chemical Analysis of Land Surface Water; Gidrometeoizdat: Leningrad, Russia, 1977; p. 541. (In Russian) [Google Scholar]
- Fomin, G.S. Water Control of Chemical, Bacterial, and Radiation Safety According to International Standards; NGO Alternative: Moscow, Russia, 1995; p. 618. (In Russian) [Google Scholar]
- Interstate Standard 31 870-2012. Drinking Water. In Determination of Elements Content by Atomic Spectrometry Methods; Standardinform: Moscow, Russia, 2013. (In Russian) [Google Scholar]
- Kiselev, I.A. Research methods of plankton. In Life of the Fresh Water of the USSR; Pavlovsky, E.N., Zhadin, V.I., Eds.; Academy of Sciences: Moscow/Leningrad, Russia, 1956; pp. 188–253. (In Russian) [Google Scholar]
- Balushkina, E.V.; Vinberg, G.G. The relationship between the length and body weight of planktonic crustaceans. In Experimental and Field Studies of the Biological Foundations of Lake Productivity; Vinberg, G.G., Ed.; Institute of Lake and River Fishery: Leningrad, Russia, 1979; pp. 58–79. (In Russian) [Google Scholar]
- Rylov, V.M. Fauna of the USSR. Crustaceans. Freshwater Cyclopoida; Nauka: Moscow, Russia, 1948; p. 312. (In Russian) [Google Scholar]
- Kutikova, L.A. Rotifers of the Fauna of the USSR; Science: Leningrad, Russia, 1964; p. 744. (In Russian) [Google Scholar]
- Tsalolikhin, S.Y. Key to Freshwater Invertebrates in Russia and Adjacent Territories; Zoological Institute: St. Petersburg, Russia, 1995; Issue 2, p. 628. (In Russian) [Google Scholar]
- Krupa, E.G.; Dobrokhotova, O.V.; Stuge, T.S. Fauna of Calanoida (Crustacea: Copepoda) of Kazakhstan and Adjacent Territories; Etalon Print: Almaty, Kazakhstan, 2016; p. 208. (In Russian) [Google Scholar]
- Magurran, E. Ecological Diversity and its Measurement; Mir: Moscow, Russia, 1998; p. 184. ISBN 5-03-002404-2. (In Russian) [Google Scholar]
- Shitikov, V.K.; Rosenberg, G.S.; Zinchenko, T.D. Quantitative Hydroecology: Methods of Systemic Identification; Institute of Ecology of the Volga Basin of the Russian Academy of Sciences: Togliatti, Russia, 2003; p. 463. ISBN 5-93424-109-5. (In Russian) [Google Scholar]
- Clarke, K.R.; Gorley, R.N. PRIMER v5: User Manual/Tutorial; PRIMER-E Ltd Press: Plymouth, UK, 2001. [Google Scholar]
- Krupa, E.G.; Barinova, S.M.; Romanova, S.M.; Malybekov, A.B. Hydrobiological assessment of the high mountain Kolsay Lakes (Kungey Alatau, Southeastern Kazakhstan) ecosystems in climatic gradient. Br. J. Environ. Clim. Chang. 2016, 6, 259–278. [Google Scholar] [CrossRef]
- Krupa, E.G.; Barinova, S.S. Environmental variables regulating the phytoplankton structure in high mountain lakes. Res. J. Pharm. Biol. Chem. Sci. 2016, 4, 1251–1261. [Google Scholar]
- Krupa, E.G.; Barinova, S.M. The use of structural indicators of hydrocenoses in assessing the ecological state of water bodies in Kazakhstan. In Bioindication in Monitoring Freshwater Ecosystems; Institute of Lake Studies: St. Petersburg, Russia, 2017; pp. 165–170. (In Russian) [Google Scholar]
- Krupa, E.G.; Barinova, S.S.; Romanova, S.M. Zooplankton size structure in the Kolsay Mountain Lakes (Kungei Alatau, Southeastern Kazakhstan) and its relationships with environmental factors. Water Resour. 2019, 46, 403–414. [Google Scholar] [CrossRef]
- Krupa, E.G.; Barinova, S.S.; Isbekov, K.B.; Assylbekova, S.Z. The use of zooplankton distribution maps for assessment of ecological status of the Shardara reservoir (Southern Kazakhstan). Ecohydrol. Hydrobiol. 2018, 1852–1865. [Google Scholar] [CrossRef]
- Barinova, S.; Krupa, E.; Romanova, S. The role of planktonic algae in the ecological assessment of storage-reservoirs of the Ile-Balkhash basin (Kazakhstan). Transylv. Rev. Syst. Ecol. Res. Wetl. Divers. 2018, 20, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Krupa, E.G.; Barinova, S.S.; Assylbekova, S.Z.; Isbekov, K.B. Structural indicators of zooplankton of the Shardara Reservoir (Kazakhstan) and the main influencing factors. Turk. J. Fish. Aquat. Sci. 2018, 18, 659–669. [Google Scholar] [CrossRef]
- Odum, Y. Ecology; Mir: Moscow, Russia, 1981; p. 328. (In Russian) [Google Scholar]
- Love, J.; Selker, R.; Marsman, M.; Jamil, T.; Dropmann, D.; Verhagen, A.J.; Ly, A.; Gronau, Q.F.; Smira, M.; Epskamp, S.; et al. JASP: Graphical statistical software for common statistical designs. J. Stat. Softw. 2019, 88, 1–17. [Google Scholar] [CrossRef] [Green Version]
- STATISTICA (Data Analysis Software System). Version 7.1. StatSoft, Inc.: Tulsa, OK, USA, 2007.
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Microcomputer Power Press: Ithaca, NY, USA, 2002. [Google Scholar]
- Tytła, M.; Kostecki, M. Ecological risk assessment of metals and metalloid in bottom sediments of water reservoir located in the key anthropogenic “hot spot” area (Poland). Environ. Earth Sci. 2019, 78, 179. [Google Scholar] [CrossRef] [Green Version]
- Zubala, T.; Patro, M.; Boguta, P. Variability of zinc, copper and lead contents in sludge of the municipal stormwater treatment plant. Environ. Sci. Pollut. Res. 2017, 24, 17145–17152. [Google Scholar] [CrossRef] [Green Version]
- Krupa, E.G. Zooplankton of limnic and lotic ecosystems in Kazakhstan. In Structure, Patterns of Formation; Palmarium Academic Publishing: Saarbrucken, Germany, 2012. (In Russian) [Google Scholar]
- Zeng, J.; Zhao, D.; Ji, Y.; Wu, Q. Comparison of heavy metal accumulation by a bloom-forming cyanobacterium, Microcystis aeruginosa. Chin. Sci. Bull. 2012, 57, 3790–3797. [Google Scholar] [CrossRef] [Green Version]
- Benfares, R.; Seridi, H.; Belkacem, Y.; Inal, A. Heavy Metal Bioaccumulation in Brown Algae Cystoseira compressa in Algerian Coasts, Mediterranean Sea. Environ. Process. 2015, 2, 429–439. [Google Scholar] [CrossRef] [Green Version]
- Utz, L.R.; Bohrer-Morel, M.B.C. Characterization of the Zooplankton Community of the Secondary Wastewater Treatment System of an Oil Refinery in Southern Brazil. Biocienc. Porto Alegre 2008, 16, 1–14. [Google Scholar]
- Krupa, E.G.; Smirnova, D.A.; Amirgaliev, N.A.; Saduakasova, R.E. Long-term dynamics of zooplankton in the Shardara reservoir. Res. Results 2009, 4, 18–20. (In Russian) [Google Scholar]
- Razak, S.B.A.; Sharip, Z. Spatio-temporal variation of zooplankton community structure in tropical urban waterbodies along trophic and urban gradients. Ecol. Process. 2019, 8, 44. [Google Scholar] [CrossRef]
- Krupa, E.; Romanova, S.; Berkinbaev, G.; Yakovleva, N.; Sadvakasov, E. Zooplankton as Indicator of the Ecological State of Protected Aquatic Ecosystems (Lake Borovoe, Burabay National Nature Park, Northern Kazakhstan). Water 2020, 12, 2580. [Google Scholar] [CrossRef]
- Krupa, E.G.; Matmuratov, S.A. Quantitative development and sex structure of Acanthocyclops robustus (Copepoda, Cyclopoida) populations in water bodies of southeastern Kazakhstan. Tethys Aqua Zool. Res. 2002, 1, 191–194. (In Russian) [Google Scholar]
- Krupa, E.G. Population densities, sex ratios of adults, and occurrence of malformations in three species of Cyclopoid copepods in waterbodies with different degrees of eutrophy and toxic pollution. J. Mar. Sci. Technol. 2005, 13, 226–237. [Google Scholar]
- Leppänen, J.J. An overview of Cladoceran studies conducted in mine water impacted lakes. Int. Aquat. Res. 2018, 10, 207–221. [Google Scholar] [CrossRef] [Green Version]
- Vesela, S.; Vijverberg, J. Effect of body size on toxicity of zinc in neonates of four differently sized Daphnia species. Aquat. Ecol. 2007, 41, 67–73. [Google Scholar] [CrossRef] [Green Version]
- Schwerin, S.; Zeis, B.; Lamkemeyer, T.; Paul, R.J.; Koch, M.; Madlung, J.; Fladerer, C.; Pirow, R. Acclimatory responses of the Daphnia pulex proteome to environmental changes. II. Chronic exposure to different temperatures (10 and 20 °C) mainly affects protein metabolism. BMC Physiol. 2009, 9, 8. [Google Scholar] [CrossRef] [Green Version]
- Stollewerk, A. The Water Flea Daphnia—A ‘New’ Model System for Ecology and Evolution? J. Biol. 2010, 9, 1–4. Available online: http://jbiol.com/content/9/2/21 (accessed on 30 September 2020). [CrossRef] [Green Version]
- Sadeq, S.A.; Beckerman, A.P. The Chronic Effects of Copper and Cadmium on Life History Traits Across Cladocera Species: A Meta‑analysis. Arch. Environ. Contam. Toxicol. 2019, 76, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Sikora, A.; Dawidowicz, P. Breakage of cyanobacterial filaments by small- and large-sized Daphnia: Are there any temperature-dependent differences? Hydrobiologia 2017, 798, 119–126. [Google Scholar] [CrossRef]
- Wejnerowski, L.; Cerbin, S.; Dziuba, M.K. Setae thickening in Daphnia magna alleviates the food stress caused by the filamentous cyanobacteria. Aquat. Ecol. 2017, 51, 485–498. [Google Scholar] [CrossRef] [Green Version]
- Lehman, P.W.; Teh, S.J.; Boyer, G.L.; Nobriga, M.L.; Bass, E.; Hogle, C. Initial impacts of Microcystis aeruginosa blooms on the aquatic food web in the San Francisco Estuary. Hydrobiologia 2010, 637, 229–248. [Google Scholar] [CrossRef] [Green Version]
- Pawlik-Skowrońska, B.; Toporowska, M.; Mazur-Marzec, H. Effects of secondary metabolites produced by different cyanobacterial populations on the freshwater zooplankters Brachionus calyciflorus and Daphnia pulex. Environ. Sci. Pollut. Res. 2019, 26, 11793–11804. [Google Scholar] [CrossRef] [Green Version]
- Peltomaa, E.T.; Aalto, S.L.; Vuorio, K.M.; Taipale, S.J. The importance of phytoplankton biomolecule availability for secondary production. Front. Ecol. Evol. 2017, 5, 128. [Google Scholar] [CrossRef] [Green Version]
- Taipale, S.J.; Aalto, S.L.; Galloway, A.W.; Kuoppamäki, K.; Nzobeuh, P.; Peltomaa, E. Eutrophication and browning influence on Daphnia nutritional ecology. Inland Waters 2019, 9, 374–394. [Google Scholar] [CrossRef]
- Taipale, S.J.; Kuoppamäki, K.; Strandberg, U.; Peltomaa, E.; Vuorio, K. Lake restoration influences nutritional quality of algae and consequently Daphnia biomass. Hydrobiologia 2020, 1–19. [Google Scholar] [CrossRef]
- Jung, S.; Kim, K.; Lee, Y.; Lee, J.; Cheong, Y.; Reza, A.; Kim, J.; Owen, J.S.; Kim, B. The effect of phosphorus removal from sewage on the plankton community in a hypertrophic reservoir. J. Ecol. Environ. 2016, 40, 9. [Google Scholar] [CrossRef] [Green Version]
- Adamczuk, M. Past, present, and future roles of small cladoceran Bosmina longirostris (O. F. Müller, 1785) in aquatic ecosystems. Hydrobiologia 2016, 767, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Krztoń, W.; Kosiba, J.; Pociecha, A.; Wilk‑Woźniak, E. The effect of cyanobacterial blooms on bio‑ and functional diversity of zooplankton communities. Biodivers. Conserv. 2019, 28, 1815–1835. [Google Scholar] [CrossRef] [Green Version]
- Warwick, R.M. A new method for detecting pollution effects on marine macrobenthic communities. Mar. Biol. 1986, 4, 557–562. [Google Scholar] [CrossRef]
- Clarke, K.R. Comparison of dominance curves. J. Exp. Mar. Biol. Ecol. 1990, 138, 143–157. [Google Scholar] [CrossRef]
- Warwick, R.M.; Clarke, K.R. Relearning the ABC: Taxonomic changes and abundance/biomass relationships in disturbed benthic communities. Mar. Biol. 1994, 118, 739–744. [Google Scholar] [CrossRef]
- Moschonas, G.; Gowen, R.J.; Paterson, R.F.; Mitchell, E.; Stewart, B.M.; McNeill, S.; Glibert, P.M.; Davidson, K. Nitrogen dynamics and phytoplankton community structure: The role of organic nutrients. Biogeochemistry 2017, 134, 125–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vuorio, K.; Järvinen, M.; Kotamäki, N. Phosphorus thresholds for bloom-forming cyanobacterial taxa in boreal lakes. Hydrobiologia 2019, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Gasiūnaite, Z.R.; Olenina, I. Zooplankton-phytoplankton interactions: A possible explanation of the seasonal succession in the Kuršiu Marios lagoon. Hydrobiologia 1998, 363, 333–339. [Google Scholar] [CrossRef]
- Pogozhev, P.I.; Gerasimova, T.N. The Role of Zooplankton in the Regulation of Phytoplankton Biomass Growth and Water Transparency in Water Bodies Polluted by Nutrients. Water Resour. 2011, 38, 400–408. [Google Scholar] [CrossRef]
- Hoppenheit, M.; Sperling, K. On the dynamics of exploited populations of Tisbe holothuriae (Copepoda, Harpacticoidae). Helgol. Wiss. Meeresunters 1977, 29, 328–336. [Google Scholar] [CrossRef] [Green Version]
- Offem, B.O.; Ezekiel, O. Ayotunde Toxicity of Lead to Freshwater Invertebrates (Water fleas; Daphnia magna and Cyclop sp.) in Fish Ponds in a Tropical Floodplain. Water Air Soil Pollut. 2008, 192, 39–46. [Google Scholar] [CrossRef]
- Wang, Z.; Yan, C.; Zhang, X. Acute and chronic cadmium toxicity to a saltwater cladoceran Moina monogolica Daday and its relative importance. Ecotoxicology 2009, 18, 47–54. [Google Scholar] [CrossRef]
- Ha, J.-Y.; Kamo, M.; Sakamoto, M. Acute toxicity of copper to Daphnia galeata under different magnesium and calcium conditions. Limnology 2017, 18, 63–70. [Google Scholar] [CrossRef]
- Rodgher, S.; Espíndola, E.L.G.; Lombardi, A.T. Suitability of Daphnia similis as an alternative organism in ecotoxicological tests: Implications for metal toxicity. Ecotoxicology 2010, 19, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Meshkova, T.M. Regularities in the Development of Zooplankton in Lake Sevan; AN ArmSSR: Yerevan, Armenia, 1975; p. 237. (In Russian) [Google Scholar]
- Maier, G. The seasonal cycle of Thermocyclops crassus (Fischer, 1853) (Copepoda: Cyclopoida) in a shallow, eutrophic lake. Hydrobiologia 1989, 178, 43–58. [Google Scholar] [CrossRef]
- Dvoretzky, V.G.; Pakhomova, N.A. Comparative characteristics of the populations of Oithona similis (Claus) in the waters of the Pechora Sea and the coastal zone of East Murman. Vestn. MGTU 2006, 9, 797–804. [Google Scholar]
- Mohamed, H.H.; Salman, S.D.; Abdullah, A.A.M. Some aspects of the biology of two Copepods: Apocyclops dengizicus and Mesocyclops isabellae from a Pool in Garmat—Ali, Basrah, Iraq. Turk. J. Fish. Aquat. Sci. 2008, 8, 239–247. [Google Scholar]
- Kiørboe, T. Sex, sex-ratios, and the dynamics of pelagic copepod populations. Oecologia 2006, 148, 40–50. [Google Scholar] [CrossRef]
- Geodakyan, V.A.; Smirnov, N.N. Sexual dimorphism and evolution of lower crustaceans. In Problems of Evolution; Vorontsov, N.N., Ed.; Nauka: Novosibirsk, Russia, 1968; Volume 1, pp. 30–36. (In Russian) [Google Scholar]
- Ikko, N.V. Features of Biology of Mass Littoral Gammarids (Crustacea, Amphipoda) in the Kola Bay. In Autoreferat Candidate of Biological Sciences: 25.00.28—Oceanology; Murmansk, Russia, 2009; p. 26. Available online: https://www.dissercat.com/content/ekologiya-mnogoshchetinkovykh-chervei-polychaeta-karskogo-morya (accessed on 22 December 2000). (In Russian)
- Krupa, E.G. Sex structure of populations of copepods (Copepoda: Cyclopoida, Calanoida) in water bodies of Kazakhstan. In Fauna of Kazakhstan and Adjacent Territories; Materials of the International Scientific. Conf.: Almaty, Kazakhstan, 2012; pp. 127–128. (In Russian) [Google Scholar]
- Mandil, R.; Prakash, A.; Rahal, A.; Singh, S.P.; Sharma, D.; Kumar, R.; Garg, S.K. In vitro and in vivo effects of flubendiamide and copper on cyto-genotoxicity, oxidative stress and spleen histology of rats and its modulation by resveratrol, catechin, curcumin and α-tocopherol. BMC Pharmacol. Toxicol. 2020, 21, 1–17. [Google Scholar] [CrossRef]
- Wong, C.K.; Pak, A.P. Acute and Subchronic Toxicity of the Heavy Metals Copper, Chromium, Nickel, and Zinc, Individually and in Mixture, to the Freshwater Copepod Mesocyclops pehpeiensis. Bull. Environ. Contam. Toxicol. 2004, 73, 190–196. [Google Scholar] [CrossRef]
- Cerbin, S.; Kraak, M.H.S.; de Voogt, P.; Visser, P.M.; Van Donk, E. Combined and single effects of pesticide carbaryl and toxic Microcystis aeruginosa on the life history of Daphnia pulicaria. Hydrobiologia 2010, 643, 129–138. [Google Scholar] [CrossRef] [Green Version]
- Kayser, H. Effect of zinc sulphate on the growth of mono- and multispecies cultures of some marine plankton algae. Helgolander Wiss. Meeresunters 1977, 30, 682–696. [Google Scholar] [CrossRef] [Green Version]
- Krupa, E.G.; Grishaeva, O.V. The structure of species dominance in the macrozoobenthos of the Small Aral Sea as an indicator of changes in water salinity. In Bioindication in the Monitoring of Freshwater Ecosystems: Abstracts of the 2nd International Conference; Lyubavich: St. Petersburg, Russia, 2011; p. 96. [Google Scholar]
| Variable | Sorbulak | RBSC No. 7 | RBSC No. 8 |
|---|---|---|---|
| altitude above sea level, m | 620 | 618 | 615 |
| water area, km2 | 58 | 3.5 | 0.4 |
| max water volume, km3 | 1000 | 17.5 | 1.2 |
| depth max, m | 20.0 | 6.0 | 7.0 |
| depth average, m | 7.2 | 5.0 | 2.9 |
| transparency, m | 1.5 | 0.2 | 0.5 |
| temperature, °C | 27.0 | 28.5 | 27.8 |
| Variable | Units | Sorbulak | RBSC No. 7 | RBSC No. 8 |
|---|---|---|---|---|
| pH | 9.0 ± 0.6 | 8.5 ± 0.01 | 9.4 ± 0.06 | |
| TDS | mg/dm3 | 1234.3 ± 37.8 | 517.5 ± 20.0 | 584.1 ± 40.1 |
| Hardness | mg-eq. dm3 | 4.9 ± 0.03 | 2.3 ± 0.01 | 2.8 ± 0.3 |
| PI | mg O2 dm3 | 11.2 ± 0.04 | 21.8 ± 0.1 | 10.9 ± 0.9 |
| Si | mg/dm3 | 2.1 ± 0.4 | 8.7 ± 0.1 | 8.3 ± 0.1 |
| N-NO2 | mg/dm3 | 0.012 ± 0.004 | 0.322 ± 0.02 | 0.006 ± 0.002 |
| N-NO3 | mg/dm3 | 0.151 ± 0.016 | 1.046 ± 0.045 | 0.078 ± 0.023 |
| N-NH4 | mg/dm3 | 0.453 ± 0.070 | 0.362 ± 0.045 | 0.275 ± 0.050 |
| PO4 | mg/dm3 | 0.15 ± 0.07 | 0.80 ± 0.27 | 0.28 ± 0.02 |
| Fe | mg/dm3 | 0.70 ± 0.14 | 0.93 ± 0.19 | 1.20 ± 0.40 |
| Mn | mg/dm3 | 0.005 ± 0.001 | 0.0 ± 0.0 | 0.003 ± 0.001 |
| Cd | mg/dm3 | <0.00001 | <0.00001 | <0.00001 |
| Cr | mg/dm3 | 0.007 ± 0.0003 | 0.007 ± 0.0001 | 0.006 ± 0.0006 |
| Cu | mg/dm3 | 0.001 ± 0.0008 | 0.043 ± 0.042 | 0.001 ± 0.0001 |
| Ni | mg/dm3 | 0.0051 ± 0.0003 | 0.006 ± 0.003 | 0.005 ± 0.0001 |
| Pb | mg/dm3 | 0.0001 ± 0.0001 | 0.0001 ± 0.0001 | 0.0001 ± 0.0001 |
| Variables | June–July 2000 | June–July 2001 | June–July 2002 | July–August 2002 |
|---|---|---|---|---|
| TDS | 1079.5 ± 42.7 | 1240.8 ± 5.1 | 1166.5 ± 8.3 | 1129.9 ± 9.0 |
| PI | - | 6.4 ± 0.8 | 5.6 ± 0.6 | 5.7 ± 0.3 |
| N-NO2 | - | 0.150 ± 0.010 | 0.006 ± 0.0001 | 0.036 ± 0.005 |
| N-NO3 | - | 1.400 ± 0.180 | 1.020 ± 0.010 | 0.590 ± 0.030 |
| N-NH4 | - | 0.250 ± 0.020 | 0.070 ± 0.010 | 0.170 ± 0.020 |
| PO4 | - | 0.130 ± 0.010 | 0.070 ± 0.020 | 0.100 ± 0.020 |
| Cd | 0.004 ± 0.0002 | 0.005 ± 0.0001 | 0.078 ± 0.005 | 0.007 ± 0.001 |
| Cu | 0.007 ± 0.001 | 0.005 ± 0.0004 | 0.002 ± 0.0003 | 0.024 ± 0.001 |
| Pb | 0.006 ± 0.001 | 0.030 ± 0.002 | 0.022 ± 0.002 | 0.047 ± 0.006 |
| Zn | 0.047 ± 0.002 | 0.023 ± 0.005 | 0.014 ± 0.003 | 0.030 ± 0.004 |
| Taxon Name | Sorbulak 2000–2002 | 2017 | ||
|---|---|---|---|---|
| Sorbulak | RBSC No. 7 | RBSC No. 8 | ||
| Phylum Rotifera | ||||
| Bdelloida gen.sp. | + | |||
| Notommatidae gen.sp. | + | |||
| Asplanchna girodi (Guerne) | + | + | ||
| Brachionus angularis (Gosse) | + | |||
| Brachionus calyciflorus dorcas Gosse | + | + | + | |
| Brachionus plicatilis longicornis (Fadeev) | + | + | ||
| Brachionus quadridentatus Hermann | + | |||
| Brachionus rubens Ehrenberg | + | |||
| Brachionus urceus (Linnaeus) | + | |||
| Brachionus variabilis (Hempel) | + | |||
| Euchlanis dilatata (Ehrenberg) | + | |||
| Euchlanis sp. | + | |||
| Hexarthra mira (Hudson) | + | + | + | + |
| Keratella quadrata dispersa Carlin | + | + | + | |
| Lecane luna (Muller) | + | |||
| Lecane sp. | + | |||
| Polyarthra major Burckhardt | + | |||
| Polyarthra sp. | + | + | + | |
| Pompholyx sulcata (Hudson) | + | + | ||
| Synchaeta kitina (Roussel.) | + | |||
| Synchaeta stylata (Wierzejski) | + | |||
| Synchaeta sp. | + | |||
| Trichocerca sp. | + | |||
| Phylum: Arthropoda | ||||
| Superorder Cladocera | ||||
| Alona quadrangularis (O.F. Muller) | + | |||
| Alona rectangula (Sars) | + | |||
| Alona sp. | + | |||
| Biapertura affinis (Leydig) | + | |||
| Bosmina (Bosmina) longirostris (O.F. Muller) | + | + | + | |
| Ceriodaphnia pulchella (Sars) | + | |||
| Chydorus sphaericus (O.F. Muller) | + | + | + | + |
| Daphnia (Ctenodaphnia) magna (Straus) | + | + | + | + |
| Daphnia (Daphnia) galeata (G.O. Sars) | + | + | + | |
| Daphnia (Daphnia) pulex (De Geer) | + | + | + | + |
| Daphnia (Daphnia) longispina (O.F. Muller) | + | |||
| Diaphanosoma mongolianum (Veno) | + | + | ||
| Leydigia leydigii (Schoedler) | + | |||
| Macrothrix laticornis (Jurine) | + | |||
| Simocephalus sp. | + | |||
| Subclass Copepoda | ||||
| Acanthocyclops trajani (Mirabdullayev et Defaye) | + | + | + | + |
| Cyclops scutifer (Sars) | + | |||
| Cyclops vicinus (Uljanin) | + | + | + | |
| Cyclopoida gen.sp. | + | |||
| Limnocletodes behningi (Borutzky) | + | |||
| Sinodiaptomus sarsi (Rylov) | + | + | ||
| Acanthodiaptomus denticornis (Wierzejski) | + | |||
| Ergasilus sieboldi Nordmann | + | |||
| Total No. species: | 31 | 29 | 9 | 10 |
| Water Body | Year | Rotifera | Cladocera | Copepoda | Total |
|---|---|---|---|---|---|
| Sorbulak | abundance, thousand ind./m3 | ||||
| 2000 | 73.1 ± 61.1 | 68.4 ± 14.4 | 97.9 ± 20.3 | 190.7 ± 34.3 | |
| 2001 | 174.2 ± 35.8 | 157.8 ± 22.8 | 305.5 ± 47.4 | 637.4 ± 76.9 | |
| 2002 | 49.2 ± 8.3 | 12.9 ± 1.3 | 84.5 ± 34.9 | 146.7 ± 28.6 | |
| 2017 | 175.8 ± 86.6 | 27.7 ± 7.7 | 102.0 ± 22.3 | 305.4 ± 91.8 | |
| RBSC No. 7 | 2017 | 58.0 ± 43.3 | 64.1 ± 11.9 | 535.2 ± 113.8 | 657.4 ± 102.4 |
| RBSC No. 8 | 2017 | 24.0 ± 10.4 | 16.7 ± 2.2 | 146.0 ± 35.9 | 186.7 ± 40.9 |
| Sorbulak | biomass, g/m3 | ||||
| 2000 | 0.10 ± 0.08 | 5.21 ± 1.20 | 1.12 ± 0.38 | 6.41 ± 1.31 | |
| 2001 | 0.49 ± 0.09 | 4.89 ± 0.77 | 1.89 ± 0.27 | 7.31 ± 0.98 | |
| 2002 | 0.10 ± 0.08 | 1.01 ± 0.28 | 0.20 ± 0.05 | 1.29 ± 0.38 | |
| 2017 | 0.18 ± 0.07 | 2.32 ± 0.63 | 0.14 ± 0.02 | 2.65 ± 0.06 | |
| RBSC No. 7 | 2017 | 0.05 ± 0.04 | 15.2 ± 4.85 | 1.78 ± 0.16 | 17.00 ± 5.00 |
| RBSC No. 8 | 2017 | 0.02 ± 0.01 | 5.45 ± 1.56 | 0.64 ± 0.37 | 6.11 ± 1.50 |
| Species Name | Abundance, % | Biomass, % | Species Name | Abundance, % | Biomass, % |
|---|---|---|---|---|---|
| Sorbulak, 2000 | Cyclops vicinus | 29.7 | 13.1 | ||
| Polyarthra sp. | 23.3 | 0.8 | Acanthocyclops trajani | 27.9 | 4.9 |
| Daphnia galeata | 21.9 | 56.9 | Sorbulak, 2017 | ||
| Daphnia longispina | 10.9 | 22.7 | Hexarthra mira | 15.5 | 1.3 |
| Cyclops vicinus | 17.3 | 11.8 | Polyarthra sp. | 12.2 | 0.8 |
| Acanthocyclops trajani | 28.7 | 5.5 | Daphnia magna | 0.3 | 10.6 |
| Sorbulak, 2001 | Daphnia pulex | 4.1 | 72.7 | ||
| Asplanchna girodi | 5.9 | 5.9 | Acanthocyclops trajani | 30.3 | 2.9 |
| Keratella quadrata | 15.4 | 0.8 | RBSC No. 7, 2017 | ||
| Hexarthra mira | 6.4 | 0.3 | Hexarthra mira | 7.1 | 0.3 |
| Bosmina longirostris | 14.5 | 11.1 | Daphnia magna | 2,8 | 76,0 |
| Daphnia galeata | 5.9 | 13.2 | Daphnia pulex | 3.0 | 11.7 |
| Daphnia pulex | 0.1 | 41.1 | Acanthocyclops trajani | 79.7 | 10.3 |
| Acanthocyclops trajani | 42.9 | 23.1 | RBSC No. 8, 2017 | ||
| Sorbulak, 2002 | Hexarthra mira | 6.7 | 0.2 | ||
| Keratella quadrata | 20.4 | 1.4 | Daphnia magna | 4.0 | 67.6 |
| Polyarthra major | 12.2 | 1.3 | Daphnia pulex | 3,8 | 21,2 |
| Daphnia pulex | 5.8 | 66.7 | Acanthocyclops trajani | 77.0 | 10.2 |
| Water Body | Year, Month | Total | ♀♀ | ♀♀ov | ♂♂ | ♂/♀ |
|---|---|---|---|---|---|---|
| Acanthocyclops trajani | ||||||
| Sorbulak | 2000 | 58.7 ± 26.8 | 0.4 ± 0.2 | 0.08 ± 0.03 | 2.8 ± 1.6 | 5.8 |
| 2001 | 360.3 ± 69.1 | 2.6 ± 0.6 | 0.6 ± 0.3 | 14.0 ± 7.8 | 4.4 | |
| 2002 | 75.9 ± 16.6 | 3.8 ± 1.3 | 0.3 ± 0.2 | 8.7 ± 3.0 | 3.3 | |
| 2017 | 92.7 ± 23.0 | 0.05 ± 0.03 | 0.02 ± 0.01 | 0.3 ± 0.1 | 4.3 | |
| RBSC No. 7 | 2017 | 523.9 ± 125.2 | 10.8 ± 1.0 | 0.5 ± 0.4 | 12.8 ± 4.9 | 1.1 |
| RBSC No. 8 | 2017 | 143.6 ± 35.0 | 2.5 ± 2.1 | 0.6 ± 0.5 | 4.6 ± 4.3 | 1.5 |
| Cyclops vicinus | ||||||
| Sorbulak | 2000 | 39.8 ± 14.8 | 0.8 ± 0.5 | 0.6 ± 0.3 | 1.0 ± 0.2 | 0.7 |
| 2001 | 26.8 ± 8.5 | 0.8 ± 0.4 | 0.3 ± 0.1 | 1.2 ± 0.9 | 1.0 | |
| 2002 | 43.6 ± 17.2 | 0.2 ± 0.1 | 0.05 ± 0.01 | 1.1 ± 0.8 | 4.4 | |
| 2017 | 8.1 ± 3.0 | 0.01 ± 0.01 | 0.01 ± 0.01 | 0.008 ± 0.005 | 0.9 | |
| RBSC No.7 | 2017 | 0 | 0 | 0 | 0 | 0 |
| RBSC No.8 | 2017 | 0.01 ± 0.01 | 0 | 0 | 0 | 0 |
| Water Body | Year | Species Number | Average Individual Mass, mg | Shannon Ab | Shannon Bi | Δ Shannon |
|---|---|---|---|---|---|---|
| Sorbulak | 2000 | 8.7 ± 1.5 | 0.0343 ± 0.0047 | 2.18 ± 0.16 | 1.57 ± 0.22 | 0.62 ± 0.19 |
| 2001 | 12.1 ± 2.0 | 0.0117 ± 0.0011 | 2.41 ± 0.18 | 2.09 ± 0.16 | 0.32 ± 0.11 | |
| 2002 | 9.0 ± 1.0 | 0.0096 ± 0.0030 | 2.21 ± 0.13 | 1.70 ± 0.17 | 0.50 ± 0.28 | |
| 2017 | 16.1 ± 0.7 | 0.0182 ± 0.0054 | 2.48 ± 0.07 | 1.77 ± 0.30 | 0.71 ± 0.35 | |
| RBSC No 7 | 2017 | 8.5 ± 1.5 | 0.0253 ± 0.0036 | 1.19 ± 0.26 | 1.16 ± 0.30 | 0.03 ± 0.04 |
| RBSC No 8 | 2017 | 7.7 ± 0.7 | 0.0379 ± 0.0139 | 1.26 ± 0.20 | 1.17 ± 0.31 | 0.10 ± 0.42 |
| Biological Variable | Environmental Variable | rS | Biological Variable | Environmental Variable | rS |
|---|---|---|---|---|---|
| Rotifera Abundance | N-NO2 | 0.356 | Copepoda Biomass | N-NO2 | 0.560 |
| N-NO3 | 0.438 | N-NO3 | 0.380 | ||
| TDS | 0.501 | PO4 | 0.341 | ||
| Cu | −0.621 | Copepoda Biomass | Cu | 0.405 | |
| Zn | −0.516 | Zn | 0.479 | ||
| Cladocera Abundance | TDS | −0.455 | Pb | 0.460 | |
| N-NO2 | 0.600 | Total Abundance | N-NO2 | 0.580 | |
| N-NO3 | 0.379 | N-NO3 | 0.462 | ||
| PO4 | 0.468 | Total Biomass | N-NO2 | 0.589 | |
| Cladocera Biomass | N-NO2 | 0.464 | PO4 | 0.440 | |
| N-NO3 | 0.353 | Si | 0.535 | ||
| PO4 | 0.405 | Cd | −0.389 | ||
| Mn | −0.537 | Average Mass | Depth | 0.362 | |
| N-NH4 | 0.372 | Δ-Shannon | Depth | 0.415 | |
| Cd | −0.435 | Transparency | 0.415 | ||
| Pb | −0.401 | TDS | 0.401 | ||
| Copepoda Abundance | Transparency | −0.351 | Shannon Ab | Depth | 0.466 |
| N-NO2 | 0.630 | Si | −0.709 | ||
| N-NO3 | 0.409 | TDS | 0.705 | ||
| PO4 | 0.475 | Transparency | 0.476 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krupa, E.; Barinova, S.; Romanova, S.; Aubakirova, M.; Ainabaeva, N. Planktonic Invertebrates in the Assessment of Long-Term Change in Water Quality of the Sorbulak Wastewater Disposal System (Kazakhstan). Water 2020, 12, 3409. https://doi.org/10.3390/w12123409
Krupa E, Barinova S, Romanova S, Aubakirova M, Ainabaeva N. Planktonic Invertebrates in the Assessment of Long-Term Change in Water Quality of the Sorbulak Wastewater Disposal System (Kazakhstan). Water. 2020; 12(12):3409. https://doi.org/10.3390/w12123409
Chicago/Turabian StyleKrupa, Elena, Sophia Barinova, Sophia Romanova, Moldir Aubakirova, and Nazia Ainabaeva. 2020. "Planktonic Invertebrates in the Assessment of Long-Term Change in Water Quality of the Sorbulak Wastewater Disposal System (Kazakhstan)" Water 12, no. 12: 3409. https://doi.org/10.3390/w12123409
APA StyleKrupa, E., Barinova, S., Romanova, S., Aubakirova, M., & Ainabaeva, N. (2020). Planktonic Invertebrates in the Assessment of Long-Term Change in Water Quality of the Sorbulak Wastewater Disposal System (Kazakhstan). Water, 12(12), 3409. https://doi.org/10.3390/w12123409






