Effect of Zr Impregnation on Clay-Based Materials for H2O2-Assisted Photocatalytic Wet Oxidation of Winery Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Winery Wastewater Sampling
2.2. Clay Mineral
2.3. Analytical Techniques
2.4. Catalysts Preparation
2.5. Catalysts Characterization
2.6. Adsorption Tests
2.7. Photocatalytic Experiments and Kinetic Modelling
3. Results and Discussion
3.1. Catalysts Characterization
3.2. Adsorption vs. Reaction
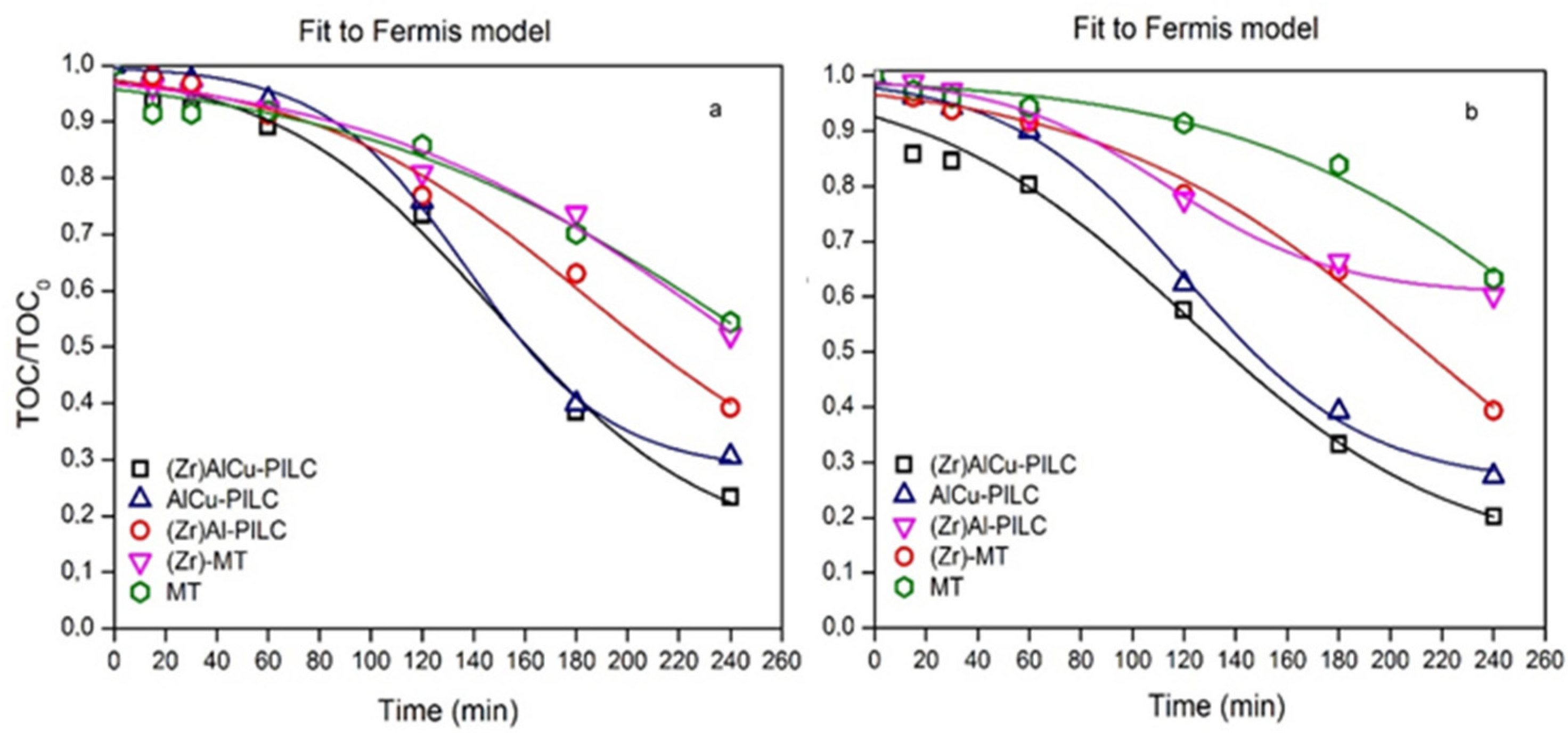
3.3. Kinetic Study
3.4. Catalyst Regeneration
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amor, C.; Marchão, L.; Lucas, M.S.; Peres, J.A. Application of Advanced Oxidation Processes for the Treatment of Recalcitrant Agro-Industrial Wastewater: A Review. Water 2019, 11, 205. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, L.C.; Fernandes, J.R.; Rodríguez-Chueca, J.; Peres, J.A.; Lucas, M.S.; Tavares, P.B. Photocatalytic degradation of an agro-industrial wastewater model compound using a UV LEDs system: Kinetic study. J. Environ. Manag. 2020, 269, 110740. [Google Scholar] [CrossRef]
- Noukeu, N.A.; Gouado, I.; Priso, R.J.; Ndongo, D.; Taffouo, V.D.; Dibong, S.D.; Ekodeck, G.E. Characterization of effluent from food processing industries and stillage treatment trial with Eichhornia crassipes (Mart.) and Panicum maximum (Jacq.). Water Resour. Ind. 2016, 16, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Sousa, R.M.O.F.; Amaral, C.; Fernandes, J.M.C.; Fraga, I.; Semitela, S.; Braga, F.; Coimbra, A.M.; Dias, A.A.; Bezerra, R.M.; Sampaio, A. Hazardous impact of vinasse from distilled winemaking by-products in terrestrial plants and aquatic organisms. Ecotoxicol. Environ. Saf. 2019, 183, 109493. [Google Scholar] [CrossRef]
- Al Bsoul, A.; Hailat, M.; Abdelhay, A.; Tawalbeh, M.; Jum’h, I.; Bani-Melhem, K. Treatment of olive mill effluent by adsorption on titanium oxide nanoparticles. Sci. Total Environ. 2019, 688, 1327–1334. [Google Scholar] [CrossRef]
- Chen, B.; Jiang, C.; Yu, D.; Wang, Y.; Xu, T. Design of an alternative approach for synergistic removal of multiple contaminants: Water splitting coagulation. Chem. Eng. J. 2020, 380, 122531. [Google Scholar] [CrossRef]
- Candia-Onfray, C.; Espinoza, N.; Sabino da Silva, E.B.; Toledo-Neira, C.; Espinoza, L.C.; Santander, R.; García, V.; Salazar, R. Treatment of winery wastewater by anodic oxidation using BDD electrode. Chemosphere 2018, 206, 709–717. [Google Scholar] [CrossRef]
- Aziz, H.A.; Abu Amr, S.S. Advanced Oxidation Processes (AOPs) in Water and Wastewater Treatment; IGI Global: Hershey, PA, USA, 2019. [Google Scholar] [CrossRef]
- Trapido, M.; Tenno, T.; Goi, A.; Dulova, N.; Kattel, E.; Klauson, D.; Klein, K.; Tenno, T.; Viisimaa, M. Bio-recalcitrant pollutants removal from wastewater with combination of the Fenton treatment and biological oxidation. J. Water Process Eng. 2017, 16, 277–282. [Google Scholar] [CrossRef]
- Amor, C.; Rodríguez-Chueca, J.; Fernandes, J.L.; Domínguez, J.R.; Lucas, M.S.; Peres, J.A. Winery wastewater treatment by sulphate radical based-advanced oxidation processes (SR-AOP): Thermally vs UV-assisted persulphate activation. Process Saf. Environ. Prot. 2019, 122, 94–101. [Google Scholar] [CrossRef]
- Lucas, M.S.; Peres, J.A. Removal of Emerging Contaminants by Fenton and UV-Driven Advanced Oxidation Processes. Water Air Soil Pollut. 2015, 226, 273. [Google Scholar] [CrossRef]
- Rodríguez-Chueca, J.; Amor, C.; Mota, J.; Lucas, M.S.; Peres, J.A. Oxidation of winery wastewater by sulphate radicals: Catalytic and solar photocatalytic activations. Environ. Sci. Pollut. Res. 2017, 24, 22414–22426. [Google Scholar] [CrossRef]
- Lucas, M.S.; Peres, J.A.; Li Puma, G. Treatment of winery wastewater by ozone-based advanced oxidation processes (O3, O3/UV and O3/UV/H2O2) in a pilot-scale bubble column reactor and process economics. Sep. Purif. Technol. 2010, 72, 235–241. [Google Scholar] [CrossRef]
- Amor, C.; Lucas, M.S.; García, J.; Dominguez, J.R.; De Heredia, J.B.; Peres, J.A. Combined treatment of olive mill wastewater by Fenton’s reagent and anaerobic biological process. J. Environ. Sci. Health Part A 2015, 50, 161–168. [Google Scholar] [CrossRef]
- Rodríguez-Chueca, J.; Amor, C.; Fernandes, J.R.; Tavares, P.B.; Lucas, M.S.; Peres, J.A. Treatment of crystallized-fruit wastewater by UV-A LED photo-Fenton and coagulation–flocculation. Chemosphere 2016, 145, 351–359. [Google Scholar] [CrossRef] [Green Version]
- Brink, A.; Sheridan, C.; Harding, K. Combined biological and advance oxidation processes for paper and pulp effluent treatment. South Afr. J. Chem. Eng. 2018, 25, 116–122. [Google Scholar] [CrossRef]
- M’Arimi, M.M.; Mecha, C.A.; Kiprop, A.K.; Ramkat, R. Recent trends in applications of advanced oxidation processes (AOPs) in bioenergy production: Review. Renew. Sustain. Energy Rev. 2020, 121, 109669. [Google Scholar] [CrossRef]
- Luo, H.; Zeng, Y.; He, D.; Pan, X. Application of iron-based materials in heterogeneous advanced oxidation processes for wastewater treatment: A review. Chem. Eng. J. 2020. [Google Scholar] [CrossRef]
- Rueda Márquez, J.J.; Levchuk, I.; Sillanpää, M. Application of Catalytic Wet Peroxide Oxidation for Industrial and Urban Wastewater Treatment: A Review. Catalysts 2018, 8, 673. [Google Scholar] [CrossRef] [Green Version]
- Gil, A.; Galeano, L.A.; Vicente, M.Á. Applications of Advanced Oxidation Processes (AOPs) in Drinking Water Treatment; Springer International Publishing: New York, NY, USA, 2018. [Google Scholar] [CrossRef]
- Ameta, S.C.; Ameta, R. Advanced Oxidation Processes for Wastewater Treatment: Emerging Green Chemical Technology; Elsevier Science: Amsterdam, The Netherlands, 2018; ISBN 9780128105252. [Google Scholar]
- Santos Silva, A.; Seitovna Kalmakhanova, M.; Kabykenovna Massalimova, B.G.; Sgorlon, J.; Jose Luis, D.T.; Gomes, H.T. Wet Peroxide Oxidation of Paracetamol Using Acid Activated and Fe/Co-Pillared Clay Catalysts Prepared from Natural Clays. Catalysts 2019, 9, 705. [Google Scholar] [CrossRef] [Green Version]
- Ormad, M.P.; Mosteo, R.; Ibarz, C.; Ovelleiro, J.L. Multivariate approach to the photo-Fenton process applied to the degradation of winery wastewaters. Appl. Catal. B Environ. 2006, 66, 58–63. [Google Scholar] [CrossRef]
- Khare, P.; Patel, R.K.; Sharan, S.; Shankar, R. 8—Recent trends in advanced oxidation process for treatment of recalcitrant industrial effluents. In Advanced Oxidation Processes for Effluent Treatment Plants; Shah, M.P., Ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 137–160. [Google Scholar] [CrossRef]
- Jaén-Gil, A.; Buttiglieri, G.; Benito, A.; Mir-Tutusaus, J.A.; Gonzalez-Olmos, R.; Caminal, G.; Barceló, D.; Sarrà, M.; Rodriguez-Mozaz, S. Combining biological processes with UV/H2O2 for metoprolol and metoprolol acid removal in hospital wastewater. Chem. Eng. J. 2021, 404, 126482. [Google Scholar] [CrossRef]
- Giannakis, S.; Jovic, M.; Gasilova, N.; Pastor Gelabert, M.; Schindelholz, S.; Furbringer, J.-M.; Girault, H.; Pulgarin, C. Iohexol degradation in wastewater and urine by UV-based Advanced Oxidation Processes (AOPs): Process modeling and by-products identification. J. Environ. Manag. 2017, 195, 174–185. [Google Scholar] [CrossRef] [Green Version]
- Guimarães, V.; Teixeira, A.R.; Lucas, M.S.; Silva, A.M.T.; Peres, J.A. Pillared interlayered natural clays as heterogeneous photocatalysts for H2O2-assisted treatment of a winery wastewater. Sep. Purif. Technol. 2019, 228, 115768. [Google Scholar] [CrossRef]
- Guimarães, V.; Lucas, M.S.; Peres, J.A. Combination of adsorption and heterogeneous photo-Fenton processes for the treatment of winery wastewater. Environ. Sci. Pollut. Res. 2019, 26, 31000–31013. [Google Scholar] [CrossRef]
- Domínguez, C.M.; Quintanilla, A.; Casas, J.A.; Rodriguez, J.J. Treatment of real winery wastewater by wet oxidation at mild temperature. Sep. Purif. Technol. 2014, 129, 121–128. [Google Scholar] [CrossRef]
- Mosteo, R.; Ormad, P.; Mozas, E.; Sarasa, J.; Ovelleiro, J.L. Factorial experimental design of winery wastewaters treatment by heterogeneous photo-Fenton process. Water Res. 2006, 40, 1561–1568. [Google Scholar] [CrossRef]
- Tyagi, B.; Sidhpuria, K.; Shaik, B.; Jasra, R.V. Synthesis of Nanocrystalline Zirconia Using Sol−Gel and Precipitation Techniques. Ind. Eng. Chem. Res. 2006, 45, 8643–8650. [Google Scholar] [CrossRef]
- Samadi, S.; Yousefi, M.; Khalilian, F.; Tabatabaee, A. Synthesis, characterization, and application of Nd, Zr–TiO2/SiO2 nanocomposite thin films as visible light active photocatalyst. J. Nanostruct. Chem. 2015, 5, 7–15. [Google Scholar] [CrossRef] [Green Version]
- Sayagués, M.J.; Avilés, M.A.; Córdoba, J.M.; Gotor, F.J. Self-propagating combustion synthesis via an MSR process: An efficient and simple method to prepare (Ti, Zr, Hf)B2–Al2O3 powder nanocomposites. Powder Technol. 2014, 256, 244–250. [Google Scholar] [CrossRef] [Green Version]
- Teymourian, H.; Salimi, A.; Firoozi, S.; Korani, A.; Soltanian, S. One-pot hydrothermal synthesis of zirconium dioxide nanoparticles decorated reduced graphene oxide composite as high performance electrochemical sensing and biosensing platform. Electrochim. Acta 2014, 143, 196–206. [Google Scholar] [CrossRef]
- Rajabi, M.; Khodai, M.M.; Askari, N. Microwave-assisted sintering of Al–ZrO2 nano-composites. J. Mater. Sci. Mater. Electron. 2014, 25, 4577–4584. [Google Scholar] [CrossRef]
- Sannino, D.; Vaiano, V.; Ciambelli, P.; Isupova, L.A. Mathematical modelling of the heterogeneous photo-Fenton oxidation of acetic acid on structured catalysts. Chem. Eng. J. 2013, 224, 53–58. [Google Scholar] [CrossRef]
- He, J.; Yang, X.; Men, B.; Wang, D. Interfacial mechanisms of heterogeneous Fenton reactions catalyzed by iron-based materials: A review. J. Environ. Sci. 2016, 39, 97–109. [Google Scholar] [CrossRef]
- Kalmakhanova, M.S.; Diaz de Tuesta, J.L.; Massalimova, B.K.; Gomes, H.T. Pillared clays from natural resources as catalysts for catalytic wet peroxide oxidation: Characterization and kinetic insights. Environ. Eng. Res. 2020, 25, 186–196. [Google Scholar] [CrossRef] [Green Version]
- Rache, M.L.; García, A.R.; Zea, H.R.; Silva, A.M.T.; Madeira, L.M.; Ramírez, J.H. Azo-dye orange II degradation by the heterogeneous Fenton-like process using a zeolite Y-Fe catalyst—Kinetics with a model based on the Fermi’s equation. Appl. Catal. B Environ. 2014, 146, 192–200. [Google Scholar] [CrossRef] [Green Version]
- Ghime, D.; Ghosh, P. Decolorization of diazo dye trypan blue by electrochemical oxidation: Kinetics with a model based on the Fermi’s equation. J. Environ. Chem. Eng. 2020, 8, 102792. [Google Scholar] [CrossRef]
- Herney-Ramirez, J.; Silva, A.M.T.; Vicente, M.A.; Costa, C.A.; Madeira, L.M. Degradation of Acid Orange 7 using a saponite-based catalyst in wet hydrogen peroxide oxidation: Kinetic study with the Fermi’s equation. Appl. Catal. B Environ. 2011, 101, 197–205. [Google Scholar] [CrossRef]
- Silva, A.M.T.; Herney-Ramirez, J.; Söylemez, U.; Madeira, L.M. A lumped kinetic model based on the Fermi’s equation applied to the catalytic wet hydrogen peroxide oxidation of Acid Orange 7. Appl. Catal. B Environ. 2012, 121–122, 10–19. [Google Scholar] [CrossRef]
- Chapman, H.D. Cation exchange capacity. Methods Soil Anal. Chem. Microbiol. Prop. 1965, 9, 891–901. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem 1951, 193, 265–275. [Google Scholar]
- Molina, C.B.; Casas, J.A.; Zazo, J.A.; Rodríguez, J.J. A comparison of Al-Fe and Zr-Fe pillared clays for catalytic wet peroxide oxidation. Chem. Eng. J. 2006, 118, 29–35. [Google Scholar] [CrossRef]
- Guimarães, V.; Rodríguez-Castellón, E.; Algarra, M.; Rocha, F.; Bobos, I. Influence of pH, layer charge location and crystal thickness distribution on U(VI) sorption onto heterogeneous dioctahedral smectite. J. Hazard. Mater. 2016, 317, 246–258. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, W.; Wang, A. Efficient adsorption of methylene blue on an alginate-based nanocomposite hydrogel enhanced by organo-illite/smectite clay. Chem. Eng. J. 2013, 228, 132–139. [Google Scholar] [CrossRef]
- Hou, B.; Han, H.; Jia, S.; Zhuang, H.; Xu, P.; Wang, D. Heterogeneous electro-Fenton oxidation of catechol catalyzed by nano-Fe3O4: Kinetics with the Fermi’s equation. J. Taiwan Inst. Chem. Eng. 2015, 56, 138–147. [Google Scholar] [CrossRef]
- Zhou, J.; Wu, P.; Dang, Z.; Zhu, N.; Li, P.; Wu, J.; Wang, X. Polymeric Fe/Zr pillared montmorillonite for the removal of Cr(VI) from aqueous solutions. Chem. Eng. J. 2010, 162, 1035–1044. [Google Scholar] [CrossRef]
- Carriazo, J.G.; Guelou, E.; Barrault, J.; Tatibouët, J.M.; Moreno, S. Catalytic wet peroxide oxidation of phenol over Al–Cu or Al–Fe modified clays. Appl. Clay Sci. 2003, 22, 303–308. [Google Scholar] [CrossRef]
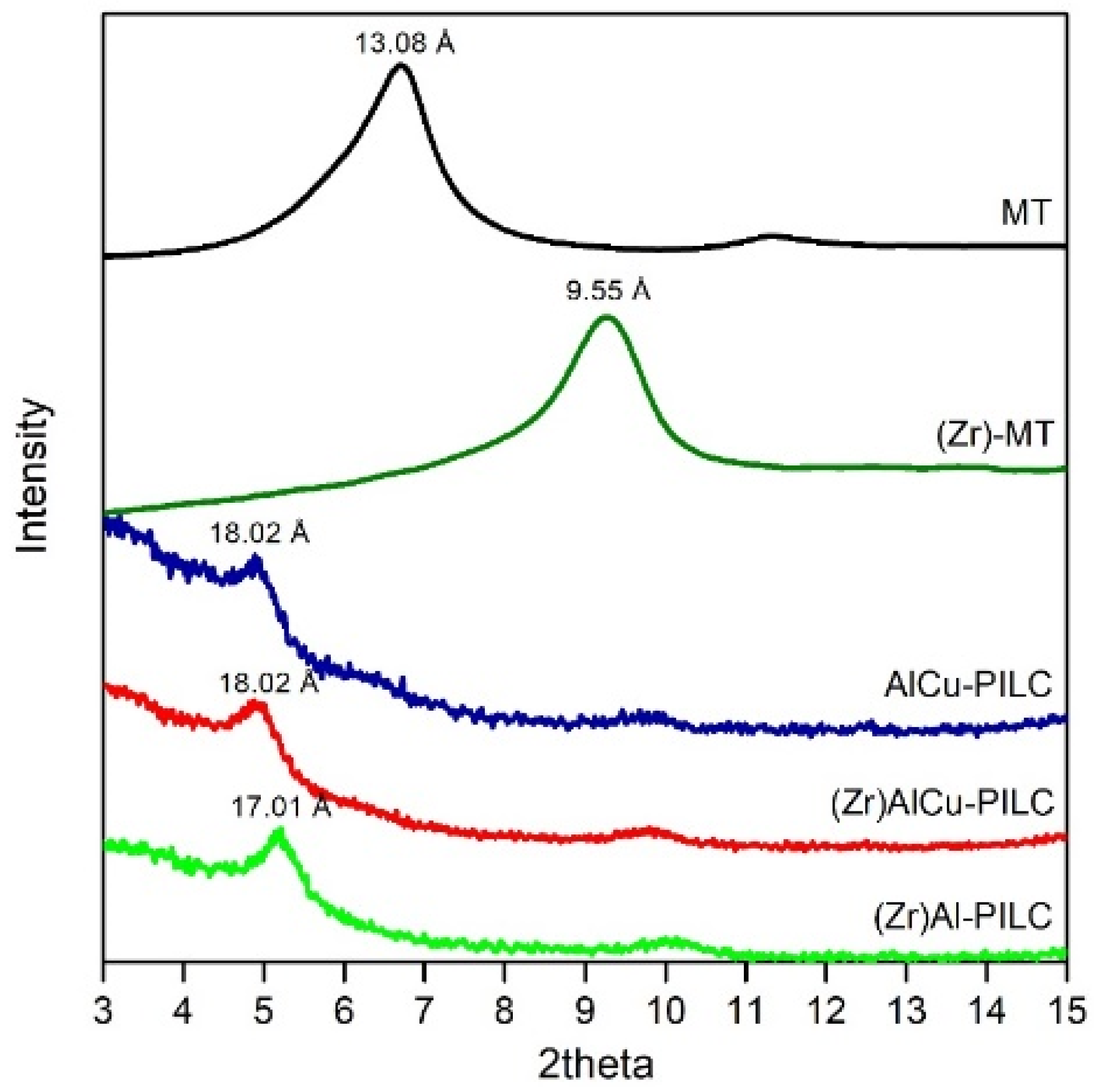
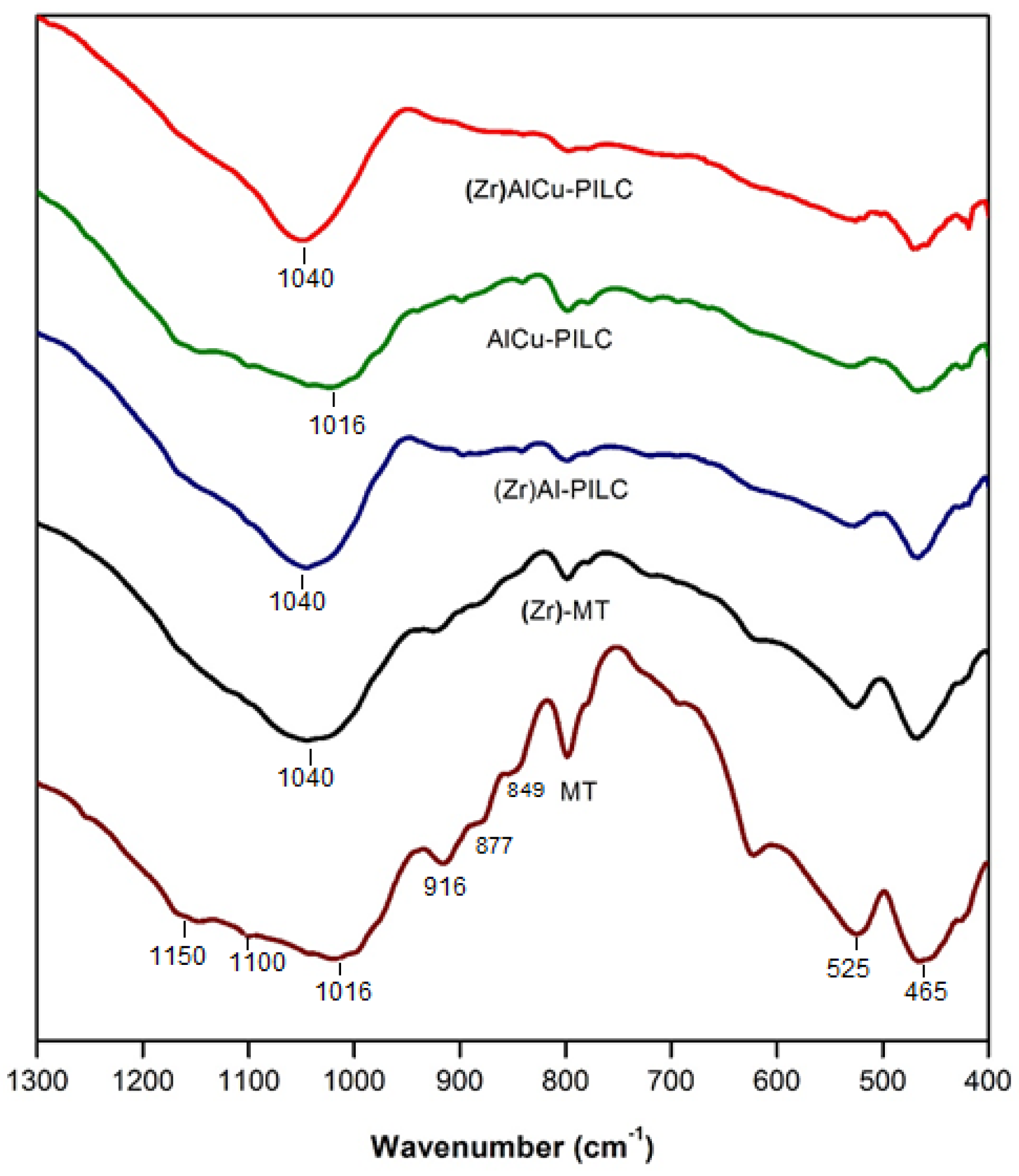
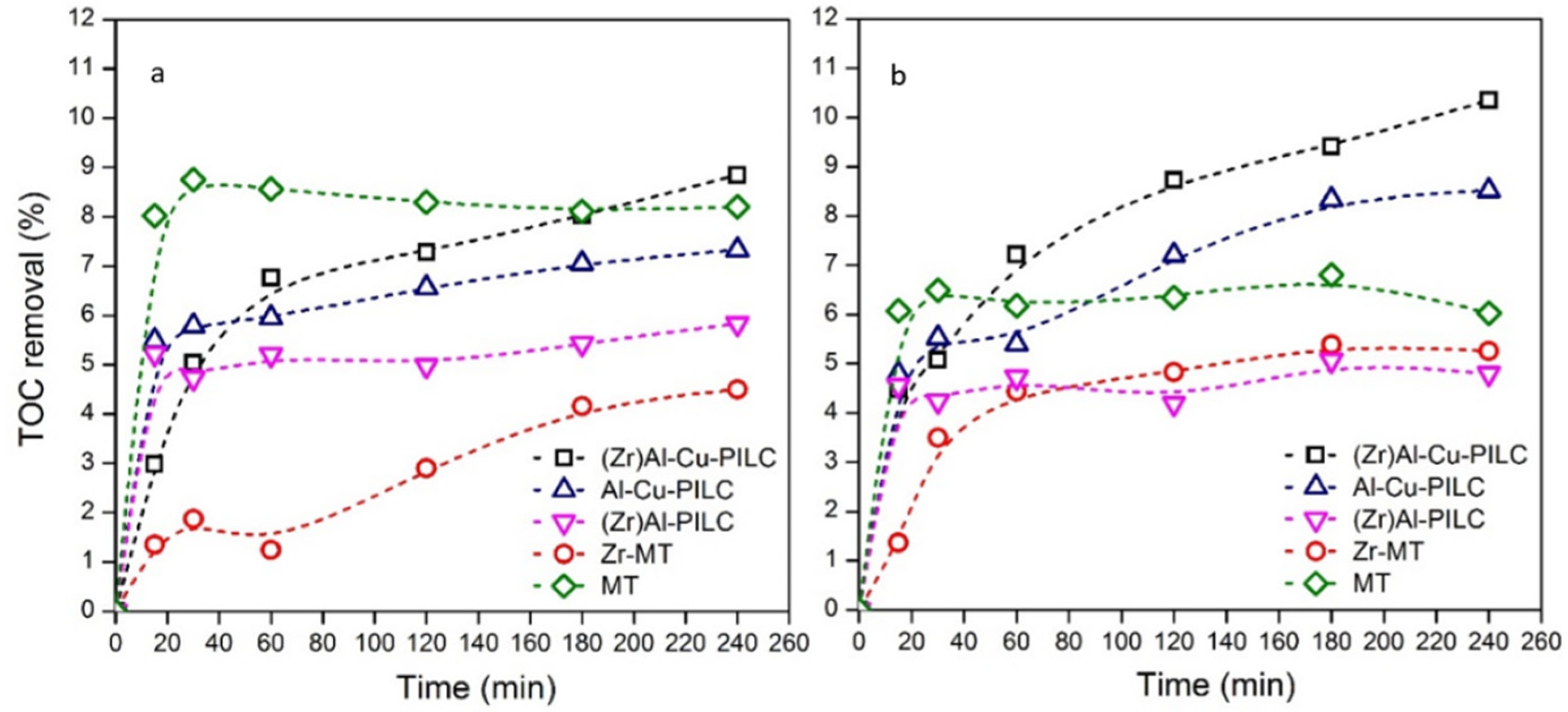
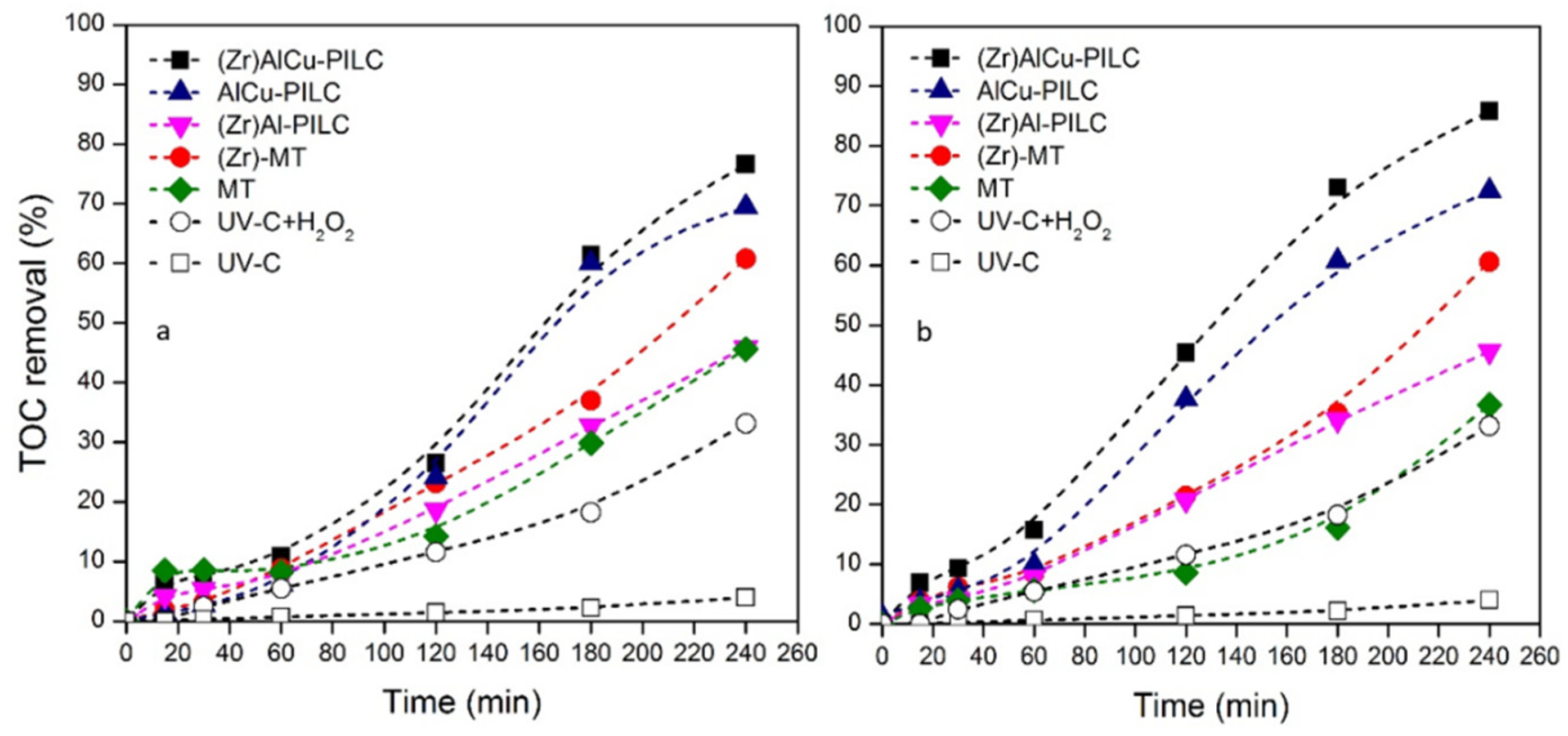
| Parameter | Value |
|---|---|
| pH | 3.8 ± 0.1 |
| Chemical Oxygen Demand (mg O2/L) | 1420 ± 45 |
| Biochemical Oxygen Demand (mg O2/L) | 610 ± 15 |
| Total Organic Carbon (mg C/L) | 500 ± 12 |
| Total Polyphenols (mg gallic acid/L) | 105 ± 3 |
| Phosphates (mg P2O5/L) | 2.7 ± 0.2 |
| Sulphates (mg SO42−/L) | 17.8 ± 1.0 |
| Total Iron (mg Fe/L) | 0.45 ± 0.02 |
| Aluminium (µg Al/L) | 17.5 ± 0.9 |
| Cadmium (µg Cd/L) | 2.1 ± 0.1 |
| Copper (µg Cu/L) | 400 ± 18 |
| Chromium (µg Cr/L) | 0.05 ± 0.003 |
| Manganese (µg Mn/L) | 29 ± 1.4 |
| Zinc (µg Zn/L) | 4200 ± 200 |
| Sample | SiO2 (%) | Al2O3 (%) | Fe2O3 (%) | MgO (%) | Na2O (%) | CaO (%) | K2O (%) | CuO (%) | ZrO2 (%) | Al/Si | Zr/Si | CEC (meq/g) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MT | 68.80 | 21.97 | 1.58 | 3.13 | 2.54 | 0.95 | 0.31 | ‒ | ‒ | 0.32 | ‒ | 0.61 |
| Zr-MT | 63.35 | 20.09 | 1.50 | 2.90 | 2.90 | 0.85 | 0.31 | ‒ | 5.54 | 0.32 | 11.44 | 0.22 |
| AlCu-PILC | 64.15 | 26.36 | 1.36 | 2.53 | 0.97 | 0.26 | 0.37 | 1.32 | ‒ | 0.41 | ‒ | 0.23 |
| (Zr)Al-PILC | 60.66 | 25.62 | 1.69 | 2.15 | 1.17 | 0.56 | 0.6 | ‒ | 5.14 | 0.42 | 11.80 | 0.21 |
| (Zr)AlCu-PILC | 60.46 | 25.64 | 1.03 | 1.97 | 1.02 | 0.24 | 0.23 | 1.30 | 5.41 | 0.42 | 11.18 | 0.22 |
| Sample | SBET (m2/g) | Vtotal pore (cm3/g) |
|---|---|---|
| MT | 8.5 | 0.047 |
| Zr-MT | 65 | 0.109 |
| Al-Cu-PILC | 107 | 0.202 |
| (Zr)Al-PILC | 81 | 0.146 |
| (Zr)Al-Cu-PILC | 118 | 0.217 |
| Heterogeneous Catalyst | Variables | Kinetic Parameters | |||
|---|---|---|---|---|---|
| kTOC (min−1) | (min) | xTOC | r2 | ||
| (Zr)AlCu-PILC | pH 4.0 | 2.35 × 10−2 | 148 | 0.14 | 0.990 |
| pH 7.0 | 2.58 × 10−2 | 119 | 0.11 | 0.996 | |
| AlCu-PILC | pH 4.0 | 3.54 × 10−2 | 137 | 0.28 | 0.998 |
| pH 7.0 | 2.87 × 10−2 | 121 | 0.26 | 0.997 | |
| (Zr)Al-PILC | pH 4.0 | 1.90 × 10−2 | 158 | 0.45 | 0.992 |
| pH 7.0 | 2.16 × 10−2 | 140 | 0.50 | 0.993 | |
| Zr-MT | pH 4.0 | 1.83 × 10−2 | 183 | 0.19 | 0.983 |
| pH 7.0 | 1.56 × 10−2 | 214 | 0.0 | 0.990 | |
| MT | pH 4.0 | 1.23 × 10−2 | 253 | 0.0 | 0.960 |
| pH 7.0 | 1.50 × 10−2 | 279 | 0.0 | 0.980 | |
| Experiment | Time (min) | H2O2 Removal (%) | ||
|---|---|---|---|---|
| Blank (H2O2 Only) | (Zr)AlCu-PILC | AlCu-PILC | ||
| pH 4.0 | 0 | 0.00 | 0.00 | 0.00 |
| 15 | 2.03 | 14.03 | 11.18 | |
| 30 | 9.03 | 10.88 | 9.73 | |
| 60 | 20.2 | 54.38 | 35.63 | |
| 120 | 38.3 | 47.39 | 41.89 | |
| 180 | 51.1 | 79.04 | 83.58 | |
| 240 | 62.0 | 96.86 | 93.13 | |
| pH 7.0 | 0 | 0.00 | 0.0 | 0.00 |
| 15 | 12.7 | 16.0 | 12.06 | |
| 30 | 8.61 | 12.9 | 10.05 | |
| 60 | 16.3 | 8.19 | 49.30 | |
| 120 | 28.5 | 45.6 | 53.29 | |
| 180 | 37.0 | 69.3 | 83.07 | |
| 240 | 46.0 | 88.4 | 94.69 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guimarães, V.; Teixeira, A.R.; Lucas, M.S.; Peres, J.A. Effect of Zr Impregnation on Clay-Based Materials for H2O2-Assisted Photocatalytic Wet Oxidation of Winery Wastewater. Water 2020, 12, 3387. https://doi.org/10.3390/w12123387
Guimarães V, Teixeira AR, Lucas MS, Peres JA. Effect of Zr Impregnation on Clay-Based Materials for H2O2-Assisted Photocatalytic Wet Oxidation of Winery Wastewater. Water. 2020; 12(12):3387. https://doi.org/10.3390/w12123387
Chicago/Turabian StyleGuimarães, Vanessa, Ana R. Teixeira, Marco S. Lucas, and José A. Peres. 2020. "Effect of Zr Impregnation on Clay-Based Materials for H2O2-Assisted Photocatalytic Wet Oxidation of Winery Wastewater" Water 12, no. 12: 3387. https://doi.org/10.3390/w12123387
APA StyleGuimarães, V., Teixeira, A. R., Lucas, M. S., & Peres, J. A. (2020). Effect of Zr Impregnation on Clay-Based Materials for H2O2-Assisted Photocatalytic Wet Oxidation of Winery Wastewater. Water, 12(12), 3387. https://doi.org/10.3390/w12123387







