Influence of Physical and Chemical Characteristics of Sediment on Macroinvertebrate Communities in Agricultural Headwater Streams
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Sediment Sampling and Measurement of Physical and Chemical Characteristics
2.3. Macroinvertebrate Sampling
2.4. Statistical Analyses
3. Results
3.1. Physical and Chemical Results
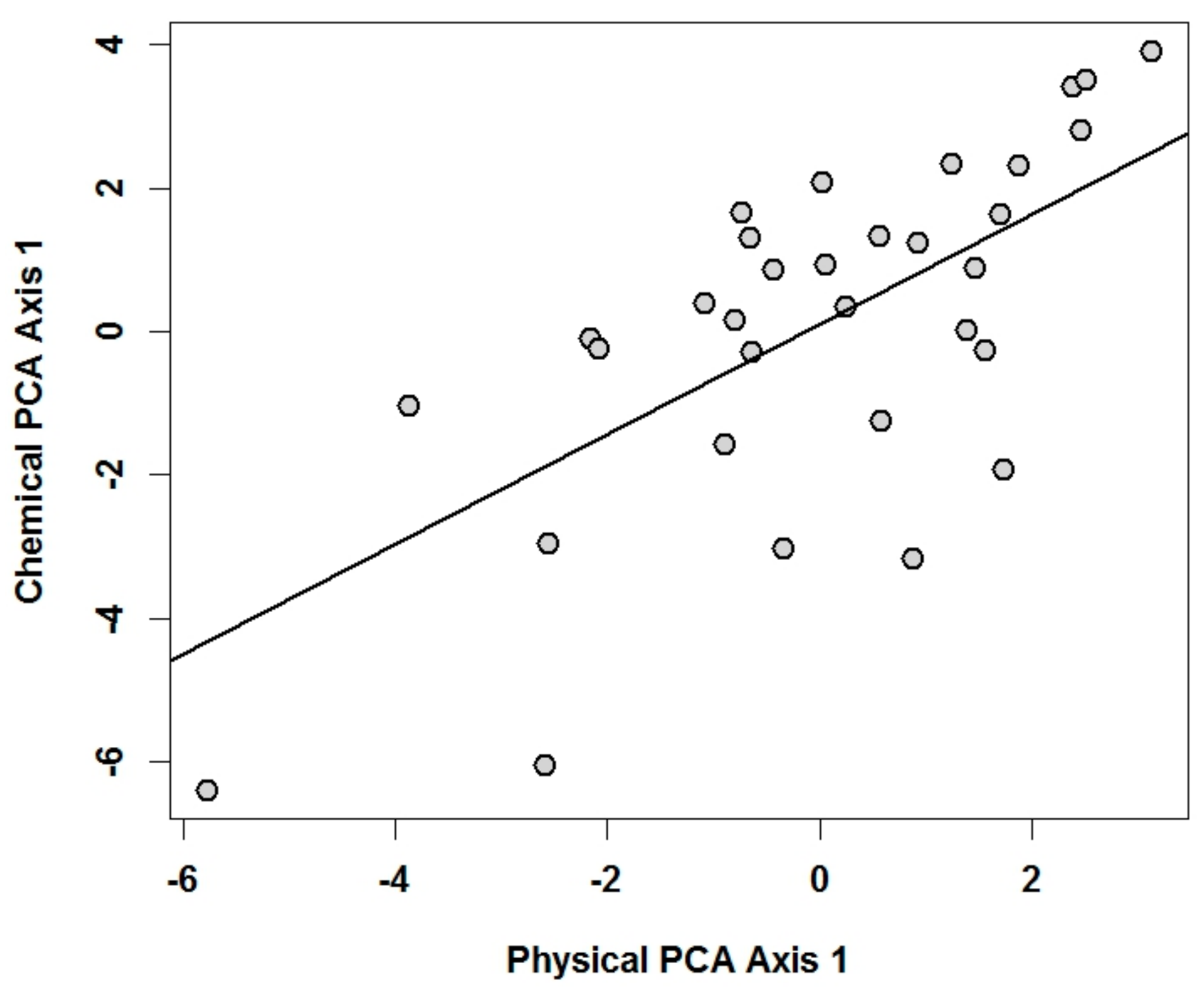
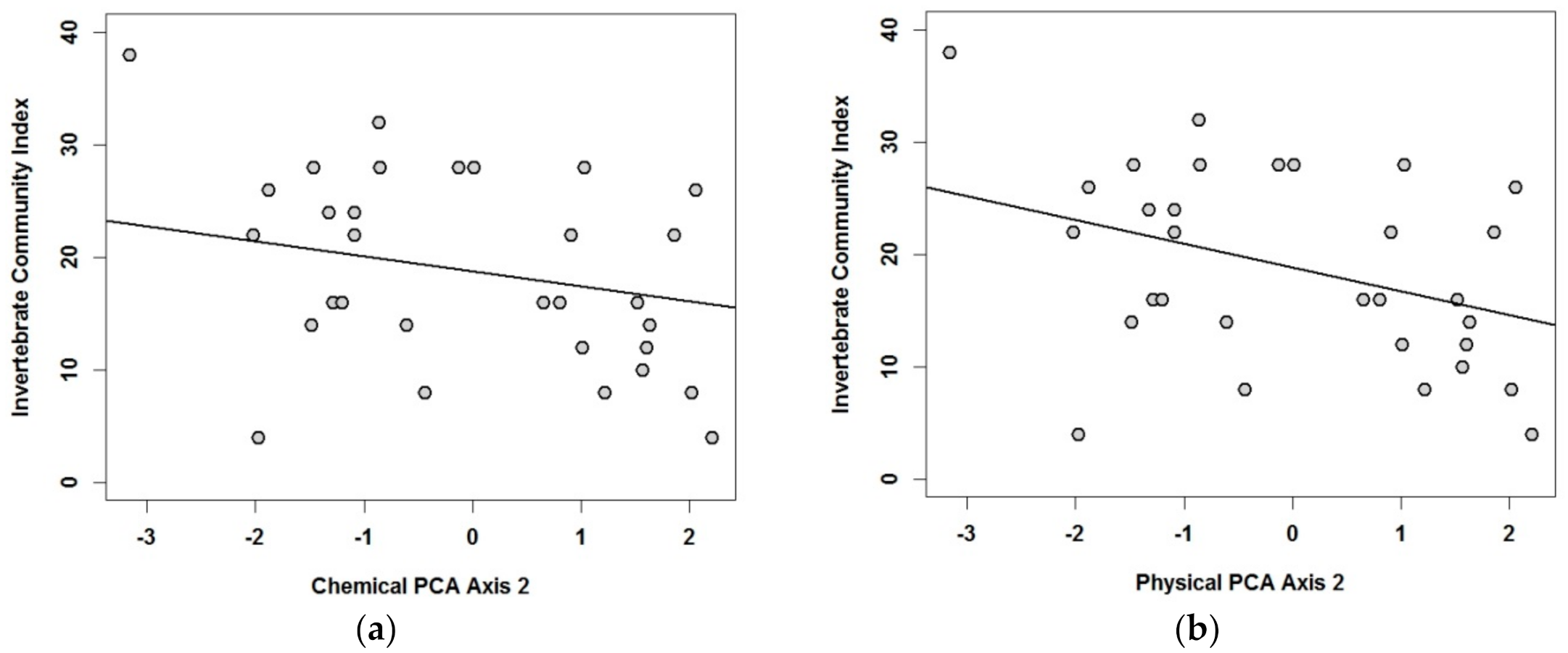
3.2. Relationships of Macroinvertebrate Communities with Sediment’s Physical and Chemical Characteristics
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vinson, M.R.; Hawkins, C.P. Biodiversity of stream insects: Variation at local, basin, and regional scales. Annu. Rev. Entomol. 1998, 43, 271–293. [Google Scholar] [CrossRef]
- Turley, M.D.; Bilotta, G.S.; Chadd, R.P.; Extence, C.A.; Brazier, R.E.; Burnside, N.G.; Pickwell, A.G.G. A sediment-specific family level biomonitoring tool to identify the impacts of fine sediment in temperate rivers and streams. Ecol. Indic. 2016, 70, 151–165. [Google Scholar] [CrossRef]
- Cummins, K.W.; Lauff, G.H. The influence of particle size on the microdistribution of stream macrobenthos. Hydrobiologia 1969, 34, 145–181. [Google Scholar] [CrossRef]
- Southwood, T.R.E. Habitat, the templet for ecological strategies. J. Anim. Ecol. 1977, 46, 337–365. [Google Scholar] [CrossRef]
- Minshall, G.W. Aquatic insect substratum relationships. In The Ecology of Aquatic Insects; Resh, V.H., Rosenberg, D.M., Eds.; Praeger: New York, NY, USA, 1984; pp. 358–400. [Google Scholar]
- Jowett, I. A method for objectively identifying pool, run, and riffle habitats from physical measurements. N. Zealand J. Mar. Freshw. Res. 1993, 27, 241–248. [Google Scholar] [CrossRef]
- Haeur, C.; Leitner, P.; Unfer, G.; Pulg, U.; Habersack, H.; Graf, W. The role of sediment and sediment dynamics in the aquatic environment. In Riverine Ecosystem Management; Schumutz, S., Sendzimir, J., Eds.; Springer: Cham, Switzerland, 2018; pp. 151–169. [Google Scholar]
- Townsend, C.R.; Scarsbrook, M.R. Quantifying disturbance in streams: Alternative measures of disturbance in relation to macroinvertebrate species traits and species richness. J. N. Am. Benthol. Soc. 1997, 16, 531–544. [Google Scholar] [CrossRef]
- Lepori, F.; Hjerdt, N. Disturbance and aquatic biodiversity: Reconciling contrasting views. Bioscience 2006, 56, 809–818. [Google Scholar] [CrossRef]
- Burdon, F.J.; McIntosh, A.R.; Harding, J.S. Habitat loss drives threshold response of benthic invertebrate communities to deposited sediment in agricultural streams. Ecol. Appl. 2013, 23, 1036–1047. [Google Scholar] [CrossRef]
- Wood, P.J.; Armitage, P.D. Biological effects of fine sediment in the lotic environment. Environ. Manag. 1997, 21, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.L.; Murphy, J.F.; Collins, A.L.; Sear, D.A.; Naden, P.S.; Armitage, P.D. The impact of fine sediment on macro-invertebrates. River Res. Appl. 2012, 28, 1055–1071. [Google Scholar] [CrossRef]
- Lemly, A.D. Modification of benthic insect communities in polluted streams: Combined effects of sedimentation and nutrient enrichment. Hydrobiologia 1982, 87, 229–245. [Google Scholar] [CrossRef]
- Megahan, W.F. Sediment pollution. In Environmental Geology. Encyclopedia of Earth Science; Springer: Dordrecht, The Netherlands, 1999. [Google Scholar]
- Moran, P.W.; Nowell, L.H.; Kemble, N.E.; Mahler, B.J.; Waite, I.R.; Van Metre, P.C. Influence of sediment chemistry and sediment toxicity on macroinvertebrate communities across 99 wadeable streams of the Midwestern USA. Sci. Total Environ. 2017, 2017, 1469–1478. [Google Scholar] [CrossRef] [PubMed]
- Moran, P.W.; Kemble, N.E.; Waite, I.R.; Mahler, B.J.; Nowell, L.H.; Van Metre, P.C. Legacy and current-use contaminants in sediments alter macroinvertebrate communities in southeastern US streams. Environ. Toxicol. Chem. 2020, 39, 1219–1232. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.A., Jr. Sediment quality criteria in use around the world. Limnology 2002, 3, 65–75. [Google Scholar] [CrossRef]
- Dewey, S.L. Effects of the herbicide atrazine on aquatic insect community structure and emergence. Ecology 1986, 67, 148–162. [Google Scholar] [CrossRef]
- Huber, W. Ecotoxicological relevance of atrazine in aquatic systems. Environ. Toxicol. Chem. 1993, 12, 1865–1881. [Google Scholar] [CrossRef]
- Riseng, C.M.; Wiley, M.J.; Black, R.W.; Munn, M.D. Impacts of agricultural land use on biological integrity: A causal analysis. Ecol. Appl. 2011, 21, 3128–3146. [Google Scholar] [CrossRef]
- Clements, W.H.; Kahsian, D.R.; Kiffney, P.M.; Zuellig, R.E. Perspectives on the context-dependency of stream community responses to contaminant. Freshw. Biol. 2016, 61, 2162–2170. [Google Scholar] [CrossRef]
- Wohl, E.; Bledsoe, B.P.; Jacobson, R.B.; Poff, N.L.; Rathburn, S.L.; Walters, D.M.; Wilcox, A.C. The natural sediment regime in rivers: Broadening the foundation for ecosystem management. BioScience 2015, 65, 358–371. [Google Scholar] [CrossRef]
- Smiley, P.C., Jr.; Gillespie, R.B. Influence of physical habitat and agricultural contaminants on fishes within agricultural drainage ditches. In Agricultural Drainage Ditches: Mitigation Wetlands for the 21st Century; Moore, M.T., Kroger, R., Eds.; Research Signpost: Kerala, India, 2010; pp. 37–73. [Google Scholar]
- Smiley, P.C., Jr.; Gillespie, R.B.; King, K.W.; Huang, C.H. Contribution of habitat and water quality to the integrity of fish communities in agricultural drainage ditches. J. Soil Water Conserv. 2008, 63, 218A–219A. [Google Scholar] [CrossRef]
- Jordan, M.A.; Castañeda, A.; Smiley, P.C., Jr.; Gillespie, R.B.; Smith, D.R.; King, K.W. Influence of instream habitat and water chemistry on amphibians in channelized agricultural headwater streams. Agric. Ecosyst. Environ. 2016, 230, 87–97. [Google Scholar] [CrossRef]
- Sanders, K.E.; Smiley, P.C., Jr.; Gillespie, R.B.; King, K.W.; Smith, D.R.; Pappas, E.A. Conservation implications of fish-habitat relationships in channelized agricultural headwater streams. J. Environ. Qual. 2020. [Google Scholar] [CrossRef]
- Wood, T.C.; Smiley, P.C., Jr.; Gillespie, R.B.; Gonzalez, J.M.; King, K.W. Injury frequency and severity in crayfish communities as indicators of physical habitat quality and water quality within agricultural headwater streams. Environ. Monit. Assess. 2020, 192, 227. [Google Scholar] [CrossRef] [PubMed]
- St Joseph River Watershed Initiative. St. Joseph River Watershed Management Plan; St. Joseph River Watershed Initiative: Fort Wayne, IN, USA, 2006. [Google Scholar]
- Ohio EPA. Total Maximum Daily Loads for the Big Walnut Creek Watershed; Final Report; Division of Surface Water, State of Ohio Environmental Protection Agency: Columbus, OH, USA, 2005. [Google Scholar]
- Miller, B.A.; Schaetzl, R.J. Precision of soil particle size analysis using laser diffractometry. Soil Sci. Soc. Am. J. 2012, 76, 1719–1727. [Google Scholar] [CrossRef]
- Magurran, A.E. Ecological Diversity and Its Measurement; Croom Helm: London, UK, 1988. [Google Scholar]
- Hofer, S. Determination of Ammonia (Salicylate) in 2 M KCl Soil Extracts by Flow Injection Analysis; QuikChem Method 12-107-06-2-A; Lachat Instruments: Loveland, CO, USA, 2003. [Google Scholar]
- Knepel, K. Determination of Nitrate in 2M KCl Soil Extracts by Flow Injection Analysis; QuikChem Method 12-107-04-1-B; Lachat Instruments: Loveland, CO, USA, 2003. [Google Scholar]
- Mehlich, A. Mehlich 3 soil test extractant: A modification of Mehlich 2 extractant. Commun. Soil Sci. Plant Anal. 1984, 15, 1409–1416. [Google Scholar] [CrossRef]
- Vonberg, D.; Hofmann, D.; Vanderborght, J.; Lelickens, A.; Köppchen, S.; Pütz, T.; Burauel, P.; Vereecken, H. Atrazine soil core residue analysis from an agricultural field 21 years after its ban. J. Environ. Qual. 2014, 43, 1450–1459. [Google Scholar] [CrossRef] [PubMed]
- Brinkhurst, C.; McCormick, S.; Williamson, S. The Bug Book: A Guide to the Identification of Common Aquatic Benthic Macroinvertebrate Families of California and Western North America; Friends of Deer Creek: Nevada City, CA, USA, 2009. [Google Scholar]
- Merrit, R.W.; Cummins, K.W. An Introduction to the Aquatic Insects of North America; Kendall/Hunt: Dubuque, IA, USA, 1996. [Google Scholar]
- Merritt, R.W.; Cummins, K.W.; Berg, M.B. An Introduction to the Aquatic Insects of North America; Kendall/Hunt Publishing Company: Dubuque, IA, USA, 2008. [Google Scholar]
- Thorp, J.H.; Rogers, D.C. Field Guide to Freshwater Invertebrates of North America; Academic Press: Oxford, UK, 2011. [Google Scholar]
- Hilsenhoff, W.L. Rapid field assessment of organic pollution with a family-level biotic index. J. N. Am. Benthol. Soc. 1988, 7, 65–68. [Google Scholar] [CrossRef]
- Ohio EPA. Biological Criteria for the Protection of Aquatic Life. Volume II; State of Ohio Environmental Protection Agency: Columbus, OH, USA, 1988. [Google Scholar]
- McCune, B.; Mefford, M.J. PC-ORD. Multivariate Analysis of Ecological Data; Version 5.0 for Windows; MjM Software: Gleneden Beach, OR, USA, 2006. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 6, 1–48. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest package: Tests in linear mixed effects models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression; Sage: Thousand Oaks, CA, USA, 2019. [Google Scholar]
- RStudio Team. RStudio: Integrated Development for R; RStudio, PBC: Boston, MA, USA, 2020. [Google Scholar]
- Carmago, J.A.; Alonso, A. Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Environ. Int. 2006, 32, 831–849. [Google Scholar]
- Indiana Legislative Services Agency. Indiana Administrative Code. 327 IAC 2-1.5-8. 2020. Available online: http://www.in.gov/legislative/iac/ (accessed on 2 September 2020).
- US EPA. Aquatic Life Ambient Water Quality Criteria for Ammonia—Freshwater; Report Number EPA-822-R-13-001; U.S. Environmental Protection Agency: Washington, DC, USA, 2013.
- Carmago, J.A.; Alonso, A.; Salamanca, A. Nitrate toxicity to aquatic animals: A review with new data for freshwater invertebrates. Chemosphere 2005, 38, 1255–1267. [Google Scholar]
- MacDonald, D.D.; Ingersoll, C.G.; Berger, T.A. Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch. Environ. Contam. Toxicol. 2000, 39, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Pappas, E.A.; Smith, D.R. Effects of dredging an agricultural drainage ditch on water column herbicide concentration, as predicted by fluvarium techniques. J. Soil Water Conserv. 2007, 62, 262–268. [Google Scholar]
- Johnson, W.W.; Finley, M.T. Handbook of Acute Toxicity of Chemicals to Fish and Aquatic Invertebrates; Summaries of Toxicity Tests Conducted at Columbia National Fisheries Research Laboratory, 1965–1978; United States Department of the Interior, Fish and Wildlife Service: Washington, DC, USA, 1980.
- Gunasekara, A.S.; Troiano, J.; Goh, K.S.; Tjeerdema, R.S. Chemistry and fate of simazine. Rev. Environ. Contam. Toxicol. 2007, 189, 1–23. [Google Scholar] [PubMed]
- US EPA. Aquatic Life Benchmarks and Ecological Risk Assessments for Registered Pesticides. 2020. Available online: https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/aquatic-life-benchmarks-and-ecological-risk#aquatic-benchmarks (accessed on 20 August 2020).
- Clark, E.W. A review of literature on calcium and magnesium in insects. Ann. Entomol. Soc. Am. 1958, 51, 142–154. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R. Calcium in plants. Ann. Bot. 2003, 92, 487–511. [Google Scholar] [CrossRef] [PubMed]
- Stone, M.L.; Whiles, M.R.; Webber, J.A.; Williard, W.J.; Reeve, J.D. Macroinvertebrate communities in agriculturally impacted southern Illinois streams: Patterns with riparian vegetation, water quality, and in-stream habitat quality. J. Environ. Qual. 2005, 34, 907–917. [Google Scholar] [CrossRef]
- Hutchens, J.J., Jr.; Schuldt, J.A.; Richards, C.; Johnson, L.B.; Host, G.E.; Breneman, D.H. Multi-scale mechanistic indicators of midwestern USA stream macroinvertebrates. Ecol. Indic. 2009, 9, 1138–1150. [Google Scholar] [CrossRef]
- Herringshaw, C.J.; Stewart, T.W.; Thompson, J.R.; Anderson, P.F. Land use, stream habitat, and benthic invertebrate assemblages in a highly altered Iowa watershed. Am. Midl. Nat. 2011, 165, 274–293. [Google Scholar] [CrossRef]
- Hrodey, P.J.; Sutton, T.M.; Frimpong, E.A. Land-use impacts on watershed health and integrity in Indiana warmwater streams. Am. Midl. Nat. 2009, 161, 76–95. [Google Scholar] [CrossRef]
- Pilière, A.; Schipper, A.M.; Breure, T.M.; Posthuma, L.; de Zwart, D.; Dyer, S.D.; Huijbregts, M.A.J. Unraveling the relationships between freshwater invertebrate assemblages and interacting environmental factors. Freshw. Sci. 2014, 33, 1148–1158. [Google Scholar]
- Longing, S.D.; Voshell, J.R., Jr.; Dolloff, C.A.; Roghair, C.N. Relationship of sedimentation and benthic macroinvertebrate assemblages in headwater streams using systematic longitudinal sampling at the reach scale. Environ. Monit. Assess. 2010, 161, 517–530. [Google Scholar] [CrossRef]
- Lamberti, G.A.; Berg, M.B. Invertebrates and other benthic features as indicators of environmental change in Juday Creek, Indiana. Nat. Area. J. 1995, 15, 249–258. [Google Scholar]
- Bossley, J.P.; Smiley, P.C., Jr. Impact of student-induced disturbance on stream macroinvertebrates differs among habitat types. Sci. Rep. 2019, 9, 1447. [Google Scholar] [CrossRef]
| Site | Watershed | Watershed Size (km2) | Percent Agriculture | Percent Canopy Cover | Stream Type |
|---|---|---|---|---|---|
| EBSJ | SJR | 21.6 | 29.3 | 21.3 | Unchannelized |
| ALG | SJR | 20.4 | 60.7 | 0.0 | Channelized |
| BLG | SJR | 13.8 | 69.0 | 0.0 | Channelized |
| CLG | SJR | 14.0 | 71.5 | 0.0 | Channelized |
| C1 | UBWC | 4.4 | 55.7 | 86.5 | Unchannelized |
| MS1 | UBWC | 9.7 | 63.5 | 1.7 | Channelized |
| B1 | UBWC | 3.8 | 75.1 | 0.3 | Channelized |
| NR1 | UBWC | 7.0 | 86.9 | 13.3 | Channelized |
| Site | % Large Gravel | % Small Gravel | % Sand | %Silt | % Clay | LOI %OC | Grain Size Richness | GSD |
|---|---|---|---|---|---|---|---|---|
| EBSJ | 21.61 (4.70–38.00) | 30.73 (19.84–38.58) | 39.64 (24.16–62.66) | 6.67 (3.65–10.75) | 1.36 (0.86–2.05) | 2.38 (1.30–4.80) | 5.0 (5–5) | 1.22 (1.08–1.33) |
| ALG | 5.43 (0.00–10.33) | 28.05 (17.20–34.44) | 35.64 (27.72–46.83) | 22.67 (18.16–26.11) | 8.2 (6.31–12.64) | 3.8 (2.40–6.00) | 4.8 (4–5) | 1.38 (1.27–1.45) |
| BLG | 0.86 (0.00–2.93) | 13.83 (6.77–19.8) | 46.84 (41.48–49.15) | 26.82 (23.05–32.40) | 11.64 (10.98–12.53) | 2.63 (1.90–3.10) | 4.5 (4–5) | 1.25 (1.16–1.31) |
| CLG | 1.06 (0.00–2.63) | 28.19 (23.07–38.14) | 43.08 (39.44–47.64) | 19.17 (13.17–24.14) | 8.49 (5.36–11.48) | 1.78 (1.10–2.20) | 4.8 (4–5) | 1.27 (1.21–1.33) |
| C1 | 19.56 (13.54–25.2) | 38.33 (30.75–51.65) | 16.85 (10.83–23.04) | 16.35 (7.90–22.21) | 8.91 (3.87–11.89) | 2.33 (1.80–2.90) | 5.0 (5–5) | 1.46 (1.28–1.54) |
| MS1 | 0.20 (0.00–0.81) | 14.7 (0.00–24.44) | 24.31 (21.48–27.55) | 38.83 (33.04–49.82) | 21.96 (20.60–23.90) | 7.48 (6.40–8.90) | 4.0 (3–5) | 1.28 (1.04–1.37) |
| B1 | 16.72 (13.54–23.55) | 37.40 (34.08–39.57) | 12.78 (3.98–22.99) | 19.43 (15.23–23.86) | 13.67 (7.26–20.79) | 3.75 (3.40–4.20) | 5.0 (5–5) | 1.48 (1.44–1.54) |
| NR1 | 7.13 (0.00–17.57) | 20.58 (4.06–41.88) | 15.56 (10.72–17.78) | 31.82 (21.32–42.07) | 24.91 (16.56–36.09) | 3.68 (3.20–3.90) | 4.8 (4–5) | 1.40 (1.17–1.54) |
| Site | Conductivity (dS/m) | pH | % TC | % TN | NH3–N (mg/kg) | NO3–N (mg/kg) | Ca (mg/kg) | K (mg/kg) | P (mg/kg) | S (mg/kg) |
|---|---|---|---|---|---|---|---|---|---|---|
| EBSJ | 0.261 (0.221–0.316) | 7.70 (7.55–8.04) | 1.71 (1.29–2.35) | 0.10 (0.02–0.18) | 6.77 (3.45–10.39) | 0.07 (0.01–0.25) | 4060 (3460–4890) | 30 (20–30) | 20 (20–20) | 50 (40–60) |
| ALG | 0.486 (0.306–0.710) | 7.58 (7.30–7.77) | 4.00 (2.82–4.57) | 0.14 (0.09–0.18) | 43.57 (26.59–60.89) | 0.34 (0.01–0.73) | 4410 (3870–5380) | 70 (0.05–0.09) | 30 (20–40) | 180 (60–410) |
| BLG | 0.479 (0.247– 0.721) | 8.18 (7.25–9.98) | 3.00 (2.77–3.27) | 0.10 (0.04–0.17) | 15.59 (3.62–21.55) | 0.23 (0.01–0.61) | 3450 (3090–4080) | 60 (50–80) | 40 (20–50) | 130 (50–210) |
| CLG | 0.472 (0.272–0.659) | 7.59 (7.28–7.9) | 2.60 (2.15–3.29) | 0.10 (0.05–0.13) | 15.37 (1.36–30.35) | 0.01 (0.01–0.01) | 3560 (3000–4000) | 60 (40–70) | 30 (20–50) | 110 (60–160) |
| C1 | 0.189 (0.086–0.344) | 8.05 (7.65–8.41) | 1.58 (1.10–1.87) | 0.08 (0.00–0.16) | 9.25 (5.56–16.70) | 0.35 (0.01–0.76) | 2150 (1360–3220) | 50 (30–70) | 10 (10–20) | 20 (0–60) |
| MS1 | 0.616 (0.433–0.736) | 6.94 (6.74–7.11) | 3.2 (2.15–3.95) | 0.21 (0.11–0.31) | 39.26 (17.7–62.23) | 0.21 (0.01–0.80) | 2890 (1590–4230) | 110 (60–170) | 20 (20–20) | 140 (90–210) |
| B1 | 0.432 (0.199–0.709) | 7.69 (7.47–8.15) | 2.98 (2.06–3.58) | 0.14 (0.05–0.19) | 11.70 (5.28–22.11) | 0.75 (0.01–2.09) | 3150 (1850–3760) | 90 (50–120) | 20 (10–30) | 110 (30–220) |
| NR1 | 0.263 (0.213–0.350) | 7.91 (7.62–8.21) | 1.74 (1.51–1.93) | 0.08 (0.06–0.11) | 9.02 (7.90–10.71) | 0.45 (0.01–1.02) | 2480 (1720–3370) | 80 (60–100) | 10 (0–20) | 40 (10–90) |
| Site | Mg (mg/kg) | Al (mg/kg) | Cu (mg/kg) | Fe (mg/kg) | Mn (mg/kg) | Zn (mg/kg) | Atrazine (µg/kg) | 2hyat (µg/kg) | Simazine (µg/kg) | Meta (µg/kg) |
|---|---|---|---|---|---|---|---|---|---|---|
| EBSJ | 160 (130–200) | 30 (10–60) | 0 (0–0) | 590 (460–820) | 200 (90–270) | 0 (0–0) | 0.77 (0.35–1.6) | 3.69 (2.71–4.69) | 0.05 (0.00–0.16) | 0.35 (0.00–0.59) |
| ALG | 200 (140–280) | 60 (10–140) | 0 (0–0) | 650 (530–810) | 160 (110–240) | 10 (0–10) | 6.04 (3.78–9.49) | 21.94 (13.2–27.54) | 0.58 (0.34–0.72) | 1.81 (1.18–2.77) |
| BLG | 140 (100–180) | 70 (10–160) | 0 (0–0) | 1450 (860–1840) | 110 (50–170) | 0 (0–10) | 8.26 (4.89–9.97) | 16.28 (12.73–21.72) | 0.49 (0.36–0.59) | 1.76 (0.86–2.98) |
| CLG | 150 (110–180) | 70 (30–120) | 0 (0–0) | 1240 (490–2290) | 100 (40–120) | 0 (0–10) | 5.82 (2.1–8.03) | 14.38 (9.68–23.67) | 0.39 (0.3–0.62) | 2.16 (0.27–5.59) |
| C1 | 200 (130–240) | 130 (70–190) | 0 (0–10) | 300 (170–410) | 110 (60–170) | 0 (0–0) | 6.07 (2.95–8.03) | 21.5 (14.41–28.21) | 1.27 (0.75–1.85) | 5.59 (2.21–9.21) |
| MS1 | 350 (180–480) | 340 (190–460) | 0 (0–0) | 2200 (1110–3430) | 90 (40–160) | 0 (0–10) | 11.64 (7.05–16.06) | 52.52 (44.52–62.48) | 0.63 (0.28–1.23) | 8.33 (5.75–13.96) |
| B1 | 300 (180–460) | 160 (50–230) | 0 (0–0) | 880 (630–1120) | 90 (30–130) | 0 (0–10) | 14.08 (9.22–18.61) | 53.18 (33.41–77.05) | 0.38 (0.23–0.72) | 15.13 (10.02–24.68) |
| NR1 | 290 (220–380) | 260 (130–380) | 0 (0–0) | 520 (430–570) | 140 (110–180) | 0 (0–10) | 12.75 (7.40–18.47) | 64.22 (26.95–99.95) | 2.69 (1.60–4.79) | 8.39 (0.00–13.46) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shuman, T.C.; Smiley, P.C., Jr.; Gillespie, R.B.; Gonzalez, J.M. Influence of Physical and Chemical Characteristics of Sediment on Macroinvertebrate Communities in Agricultural Headwater Streams. Water 2020, 12, 2976. https://doi.org/10.3390/w12112976
Shuman TC, Smiley PC Jr., Gillespie RB, Gonzalez JM. Influence of Physical and Chemical Characteristics of Sediment on Macroinvertebrate Communities in Agricultural Headwater Streams. Water. 2020; 12(11):2976. https://doi.org/10.3390/w12112976
Chicago/Turabian StyleShuman, Tyler C., Peter C. Smiley, Jr., Robert B. Gillespie, and Javier M. Gonzalez. 2020. "Influence of Physical and Chemical Characteristics of Sediment on Macroinvertebrate Communities in Agricultural Headwater Streams" Water 12, no. 11: 2976. https://doi.org/10.3390/w12112976
APA StyleShuman, T. C., Smiley, P. C., Jr., Gillespie, R. B., & Gonzalez, J. M. (2020). Influence of Physical and Chemical Characteristics of Sediment on Macroinvertebrate Communities in Agricultural Headwater Streams. Water, 12(11), 2976. https://doi.org/10.3390/w12112976





