Assessing the Ecological Water Level: The Case of Four Mediterranean Lakes
Abstract
1. Introduction
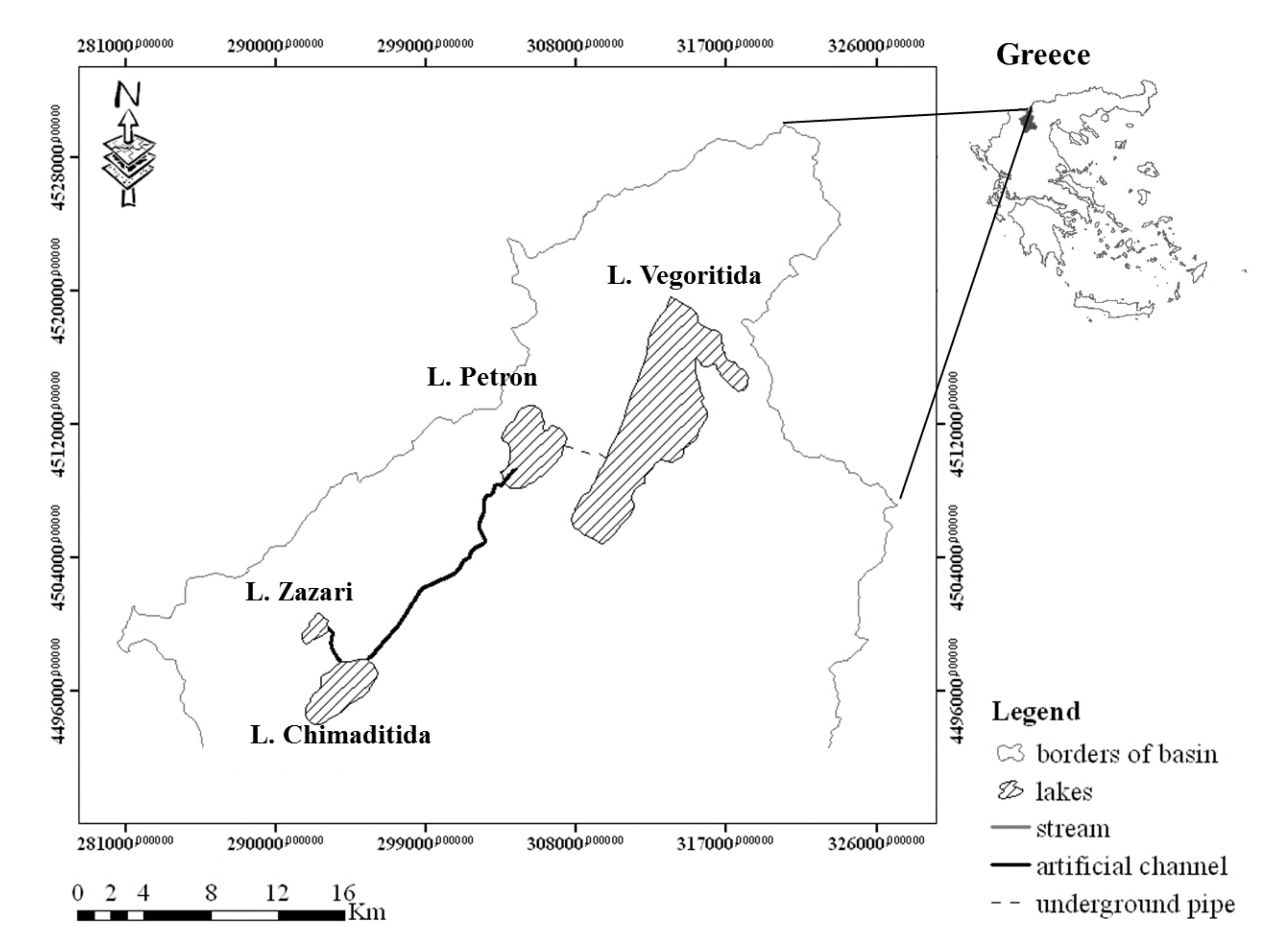
2. Study Area
3. Materials and Methods
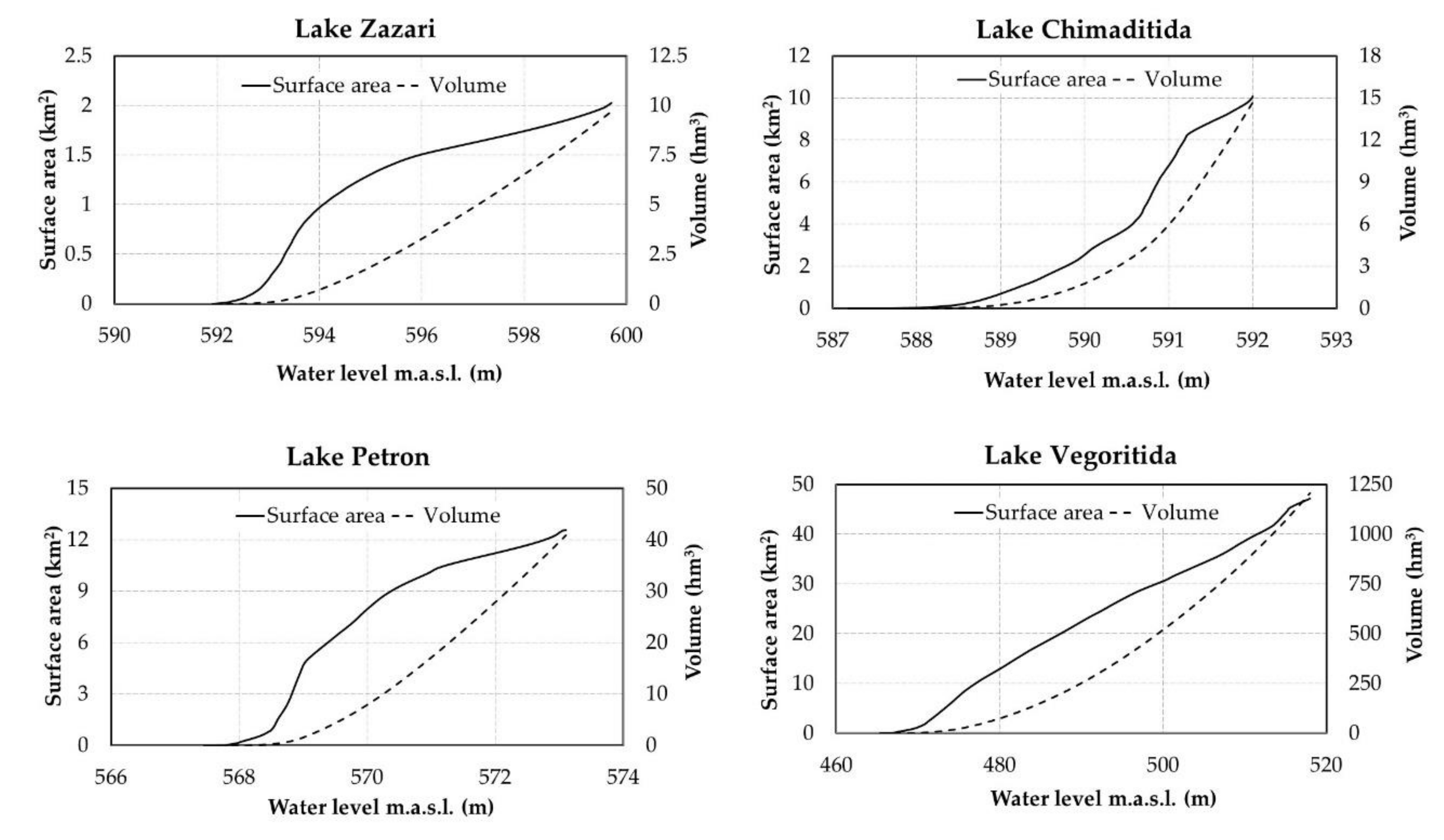
3.1. Assessing the Maximum Ecological Water Level
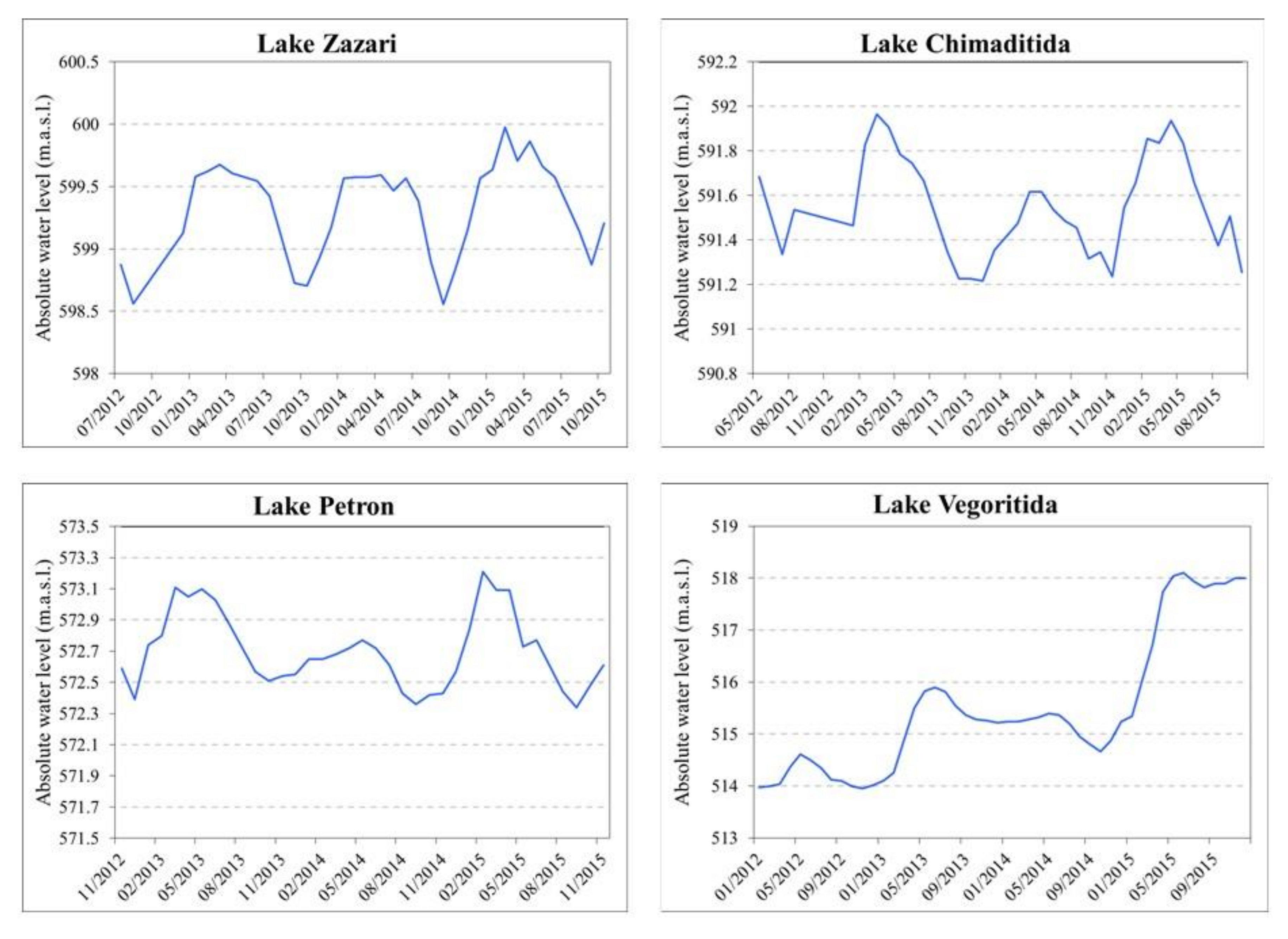
3.2. Hydromorphological Analysis
3.3. Macrophytes
3.4. Fish
3.5. Ecological Water Level Assessment
4. Results and Discussion
4.1. Morphological Analysis
4.2. Macrophytes
4.3. Fish Fauna
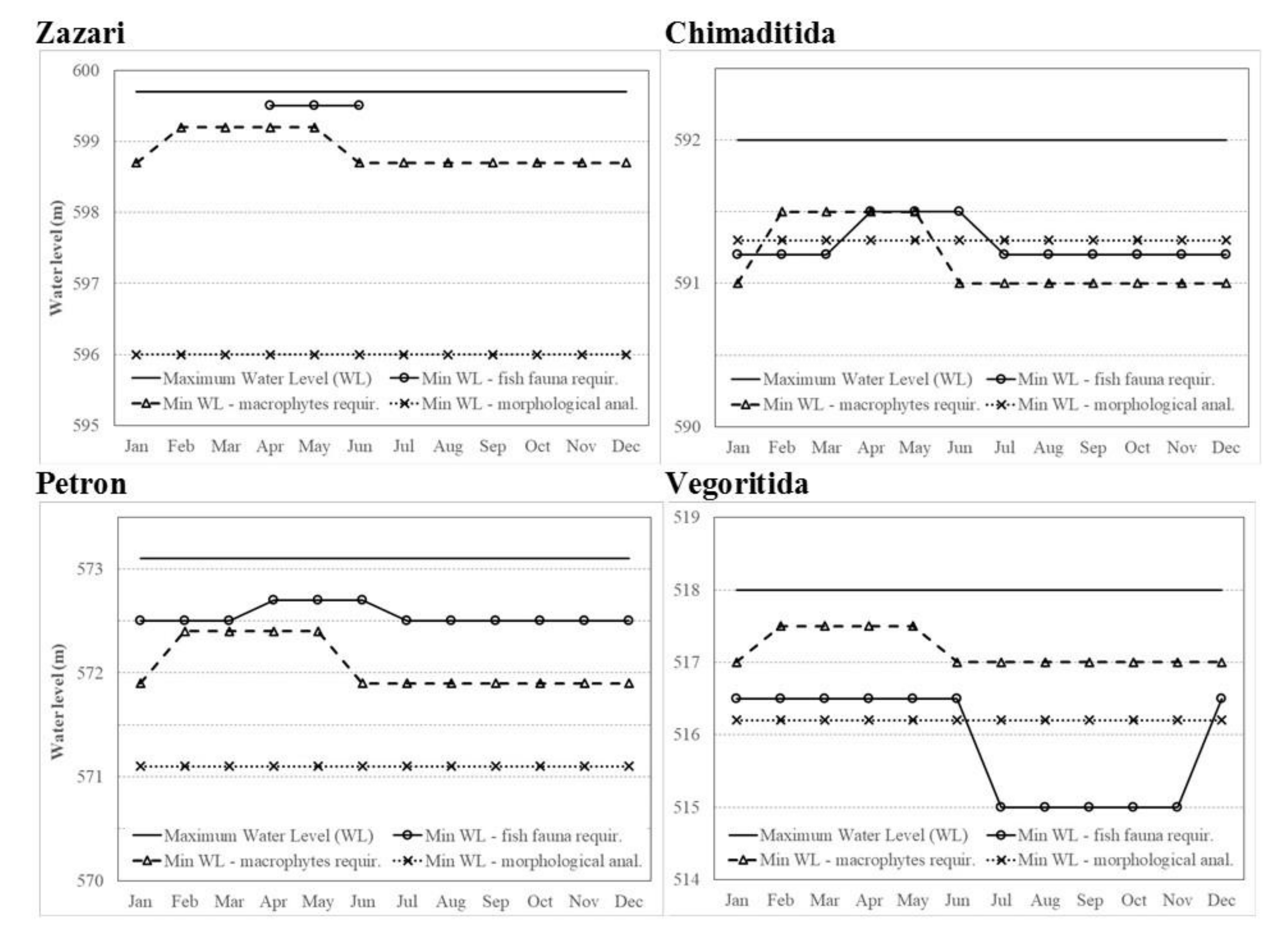
4.4. Ecological Water Level
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Phragmites australis |
|
| Typha spp. |
|
| Schoenoplectus lacustris |
|
| Juncus subnodulosus |
|
| Alisma plantago-aquatica |
|
| Myriophyllum spicatum |
|
| Ranunculus trichophyllus |
|
| Alisma gramineum |
|
| Zannichellia palustris |
|
| Parameter | Altitude (m.a.s.l.) | Surface Area (km2) | Volume (106 m3) | Surface Area (%) | Stored Volume (%) |
|---|---|---|---|---|---|
| Lake Zazari | |||||
| Max WL | 599.7 | 2.03 | 9.71 | ||
| Min WL—fish fauna—April–June | 599.5 | 1.98 | 9.31 | −2.5% | −4.1% |
| Min WL—fish fauna—July–March | 599.5 | ||||
| Min WL—macrophytes—June–January | 598.7 | 1.83 | 7.79 | −9.9% | −19.8% |
| Min WL—macrophytes—February–May | 599.2 | ||||
| Min WL—morphological analysis | 596.0 | 1.51 | 3.31 | −25.6% | −65.9% |
| Lake Chimaditida | |||||
| Max WL | 592.0 | 10.08 | 14.70 | ||
| Min WL—fish fauna—April–June | 591.5 | ||||
| Min WL—fish fauna—July–March | 591.2 | 8.10 | 7.45 | −19.6% | −49.3% |
| Min WL—macrophytes—June–January | 591.0 | 6.79 | 5.97 | −32.6% | −59.4% |
| Min WL—macrophytes—February–May | 591.5 | ||||
| Min WL—morphological analysis | 591.3 | 8.36 | 7.95 | −17.1% | −45.9% |
| Lake Petron | |||||
| Max WL | 573.1 | 12.56 | 40.83 | ||
| Min WL—fish fauna—April–June | 572.7 | ||||
| Min WL—fish fauna—July–March | 572.5 | 11.68 | 33.58 | −7.0% | −17.8% |
| Min WL—macrophytes—June–January | 571.9 | 11.13 | 26.74 | −11.4% | −34.5% |
| Min WL—macrophytes—February–May | 572.4 | ||||
| Min WL—morphological analysis | 571.1 | 10.34 | 18.14 | −17.7% | −55.6% |
| Lake Vegoritida | |||||
| Max WL | 518.0 | 47.15 | 1206.16 | ||
| Min WL—fish fauna—April–June * | 515.0 | 44.27 | 1068.14 | −6.1% | −11.4% |
| Min WL—fish fauna—July–March * | 515.0 | ||||
| Min WL—macrophytes—June–January | 517.0 | 46.45 | 1159.37 | −1.5% | −3.9% |
| Min WL—macrophytes—February–May | 517.5 | ||||
| Min WL—morphological analysis | 516.2 | 45.89 | 1123.81 | −2.7% | −6.8% |
References
- Naselli-Flores, L.; Barone, R. Water-level fluctuations in Mediterranean reservoirs: Setting a dewatering threshold as a management tool to improve water quality. Hydrobiologia 2005, 548, 85–99. [Google Scholar] [CrossRef]
- Zohary, T.; Ostrovsky, I. Ecological impacts of excessive water level fluctuations in stratified freshwater lakes. Inland Waters 2011, 1, 47–59. [Google Scholar] [CrossRef]
- Cott, P.A.; Sibley, P.K.; Somers, W.M.; Lilly, M.R.; Gordon, A.M. A Review of Water Level Fluctuations on Aquatic Biota with an Emphasis on Fishes in Ice-Covered Lakes. J. Am. Water Resour. Assoc. 2008, 44, 343–359. [Google Scholar] [CrossRef]
- Wantzen, K.M.; Junk, W.J.; Rothhaupt, K.O. An extension of the floodpulse concept (FPC) for lakes. Hydrobiologia 2008, 613, 151–170. [Google Scholar] [CrossRef]
- Aroviita, J.; Hämäläinen, H. The impact of water-level regulation on littoral macroinvertebrate assemblages in boreal lakes. Hydrobiologia 2008, 613, 45–56. [Google Scholar] [CrossRef]
- Evtimova, V.V.; Donohue, I. Water-level fluctuations regulate the structure and functioning of natural lakes. Freshwater Biol. 2016, 61, 251–264. [Google Scholar] [CrossRef]
- Cui, B.; Tang, N.; Zhao, X.; Bai, J. A management-oriented valuation method to determine ecological water requirement for wetlands in the Yellow River Delta of China. J. Nat. Conserv. 2009, 17, 129–141. [Google Scholar] [CrossRef]
- Cui, B.; Hua, Y.; Wang, C.; Liao, X.; Tan, X.; Tao, W. Estimation of ecological water requirements based on habitat response to water level in Huanghe River Delta, China. Chin. Geogr. Sci. 2010, 20, 318–329. [Google Scholar] [CrossRef]
- Becht, R.; Harper, D.M. Towards an understanding of human impact upon the hydrology of lake Naivasha, Kenya. In Lake Naivasha, Kenya; Springer: Dordrecht, The Netherlands, 2002; pp. 1–11. [Google Scholar]
- Legesse, D.; Vallet-Coulomb, C.; Gasse, F. Analysis of the hydrological response of a tropical terminal lake, Lake Abiyata (Main Ethiopian Rift Valley) to changes in climate and human activities. Hydrol. Process. 2004, 18, 487–504. [Google Scholar] [CrossRef]
- Kummu, M.; Tes, S.; Yin, S.; Adamson, P.; Józsa, J.; Koponen, J.; Richey, J.; Sarkkula, J. Water balance analysis for the Tonle Sap Lake–floodplain system. Hydrol. Process. 2014, 28, 1722–1733. [Google Scholar] [CrossRef]
- Benson, L.V.; Paillet, F.L. The use of total lake-surface area as an indicator of climatic change: Examples from the Lahontan Basin. Quat. Res. 1989, 32, 262–275. [Google Scholar] [CrossRef]
- Browne, R.A. Lakes as islands: Biogeographic distribution, turnover rates, and species composition in the lakes of central New York. J. Biogeogr. 1981, 8, 75–83. [Google Scholar] [CrossRef]
- Shang, S.H. Lake surface area method to define minimum ecological lake level from level–area–storage curves. J. Arid Land 2013, 5, 133–142. [Google Scholar] [CrossRef]
- Gippel, C.J.; Stewardson, M.J. Use of wetted perimeter in defining minimum environmental flows. River Regul. 1998, 14, 53–67. [Google Scholar] [CrossRef]
- Krstolic, J.L.; Hayes, D.C.; Ruhl, P.H. Physical Habitat Classification and Instream Flow Modeling to Determine Habitat Availability during Low-Flow Periods, North Fork Shenandoah River, Virginia. In U.S. Geological Survey Scientific Investigations Report; USGS: Reston, VA, USA, 2006; Volume 5025, p. 63. [Google Scholar]
- Leira, M.; Cantonati, M. Effects of water-level fluctuations on lakes: An annotated bibliography. Hydrobiologia 2008, 6, 171–184. [Google Scholar] [CrossRef]
- Poff, N.L.; Zimmerman, J.K. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ. Manag. 2002, 30, 492–507. [Google Scholar]
- Poff, N.L.; Zimmerman, J.K. Ecological responses to altered flow regimes: A literature review to inform the science and management of environmental flows. Freshwater Biol. 2010, 55, 194–205. [Google Scholar] [CrossRef]
- Haxton, T.J.; Findlay, C.S. Meta-analysis of the impacts of water management on aquatic communities. Can. J. Fish Aquat. Sci. 2008, 65, 437–447. [Google Scholar] [CrossRef]
- Gafny, S.; Gasith, A.; Goren, M. Effect of water level fluctuation on shore spawning of Mirogrex terraesanctae (Steinitz), (Cyprinidae) in Lake Kinneret, Israel. J. Fish Biol. 1992, 41, 863–871. [Google Scholar] [CrossRef]
- Coops, H.; van der Velde, G. Impact of hydrodynamic changes on the zonation of helophytes. Neth. J. Aquat. Ecol. 1996, 30, 165–176. [Google Scholar] [CrossRef]
- Fischer, P.; Öhl, U. Effects of water-level fluctuations on the littoral benthic fish community in lakes: A mesocosm experiment. Behav. Ecol. 2005, 16, 741–746. [Google Scholar] [CrossRef]
- Cook, C.D.K. Aquatic Plant Book; SPB Academic Publishing: The Hague, The Netherlands, 1990. [Google Scholar]
- Armstrong, W.; Brandle, R.; Jackson, M.B. Mechanisms of flood tolerance in plants. Acta Bot. Neerl. 1994, 43, 307–358. [Google Scholar] [CrossRef]
- Keddy, P.A. Wetland Ecology: Principles and Conservation; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Van der Valk, A.G.; Welling, C.H. Diversity and pattern in plant communities. In The Development of Zonation in Freshwater Wetlands: An Experimental Approach; During, J.H., Werger, M.J.A., Willems, H.J., Eds.; SPB Academic Publishing: The Hague, The Netherlands, 1988; p. 14. [Google Scholar]
- Middelboe, A.L.; Markager, S. Depth limits and minimum light requirements of freshwater macrophytes. Freshwater Biol. 1997, 37, 553–568. [Google Scholar] [CrossRef]
- Kolada, A.; Hellsten, S.; Sondergaard, M.; Mjelde, M.; Dudley, B.; Van Geest, G.; Goldsmith, B.; Davidson, T.; Bennion, H.; Noges, P.; et al. Deliverable D3.2-3: Report on the Most Suitable Lake Macrophyte Based Assessment Methods for Impacts of Eutrophication and Water Level Fluctuations; WISER, Institute of Environmental Protection (IEP): Warsaw, Poland, 2011; Available online: http://www.wiser.eu/results/deliverables/.2011 (accessed on 26 January 2018).
- Hudon, C. Impact of water level fluctuations on St. Lawrence River aquatic vegetation. Can. J. Fish Aquat. Sci. 1997, 54, 2853–2865. [Google Scholar] [CrossRef]
- Hill, N.M.; Keddy, P.A.; Wisheu, I.C. A hydrological model for predicting the effects of dams on the shoreline vegetation of lakes and reservoirs. Environ. Manag. 1998, 22, 723–736. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kohmatsu, Y.; Yuma, M. Effects of summer drawdown on cyprinid fish larvae in Lake Biwa, Japan. Limnology 2006, 7, 75–82. [Google Scholar] [CrossRef]
- De Nie, H.W.; European Inland Fisheries Advisory Commission. The Decrease in Aquatic Vegetation in Europe and Its Consequences for Fish Populations; Fisheries Department of the Netherlands Ministry of Agriculture and Fisheries: The Hague, The Netherlands, 1987. [Google Scholar]
- Savino, J.F.; Stein, R.A. Behavioural interactions between fish predators and their prey: Effects of plant density. Anim. Behav. 1989, 37, 311–321. [Google Scholar] [CrossRef]
- Kahl, U.; Hülsmann, S.; Radke, R.J.; Benndorf, J. The impact of water level fluctuations on the year class strength of roach: Implications for fish stock management. Limnologica 2008, 38, 258–268. [Google Scholar] [CrossRef]
- Stefanidis, K.; Papastergiadou, E. Relationships between lake morphometry, water quality, and aquatic macrophytes, in greek lakes. Fresen. Environ. Bull. 2012, 21, 3018–3026. [Google Scholar]
- Doulgeris, C.; Papadimos, D.; Katsavouni, S.; Argyroudi, A. Investigation of the Spatial Distribution of Water Depth and Quality of Lakes Zazari, Cheimaditida, Petron and Vegoritida (D.A1.3); Greek Biotope/Wetland Centre (EKBY): Thermi, Greece, 2017. [Google Scholar]
- Shang, S.H. A multiple criteria decision-making approach to estimate minimum environmental flows based on wetted perimeter. River Res. Appl. 2008, 24, 54–67. [Google Scholar] [CrossRef]
- CEN. Water Quality—Guidance Standard for the Surveying of Macrophytes in Lakes; I.S. EN 15460: 2007; European Committee for Standardization (CEN): Brussels, Belgium, 2007. [Google Scholar]
- CEN. Water Quality—Sampling of Macrophyte Communities in Water Bodies; XP T90-328: 2010; European Committee for Standardization (CEN): Brussels, Belgium, 2010. [Google Scholar]
- CEN. Guidance Standard for the Surveying of Aquatic Macrophytes in Lakes; EN 14184; Rep. CEN/TC 230/WG2/TG3:N55; European Committee for Standardization (CEN): Brussels, Belgium, 2003. [Google Scholar]
- Kolada, A.; Hellsten, S.; Kanninen, A.; Søndergaard, M.; Dudley, B.; Nõges, P.; Ott, I.; Ecke, F.; Mjedle, M.; Bertrin, V.; et al. Deliverable D3.2-1: Overview and Comparison of Macrophyte Survey Methods Used in European Countries and a Proposal of Harmonized Common Sampling Protocol to Be Used for WISER Uncertainty Exercise Including a Relevant Common Species List; WISER, Institute of Environmental Protection (IEP): Warsaw, Poland, 2009. [Google Scholar]
- Jensen, S. An objective method for sampling the macrophyte vegetation in lakes. Vegetatio 1977, 33, 107–118. [Google Scholar] [CrossRef]
- Janauer, G.A. Guidance on the Assessment of Aquatic Macrophytes in Lakes Under the Conditions of the Monitoring for the Water Framework Directive/EU; Monograph; University of Vienna: Vienna, Austria, 2002. [Google Scholar]
- Palmer, M.A.; Bell, S.L.; Butterfield, I.A. A botanical classification of standing waters in Britain: Application for conservation and monitoring. Aquat. Conserv. 1992, 2, 125–143. [Google Scholar] [CrossRef]
- Fasset, N.C. A Manual of Aquatic Plants; The University of Wisconsin Press: Madison, WI, USA, 1957. [Google Scholar]
- Ascherson, P.; Graebner, P. Potamogetonaceae. In Das Pflanzenreich, Regni Vegetabilis Conspectus; Verlag von J. Cramer, H.R. Engelmann: Weinheim, Germany, 1959; p. 81. [Google Scholar]
- Wood, R.D.; Imahori, K. Iconograph of the Characeae; Verlag von J. Cramer: Weinheim, Germany, 1964. [Google Scholar]
- Wood, R.D.; Imahori, K. Monograph of the Characeae; Verlag von J. Cramer: Weinheim, Germany, 1965. [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea; Cambridge University Press: Cambridge, UK, 1968; Volume 2–4. [Google Scholar]
- Tutin, T.G.; Burges, N.A.; Charter, A.O.; Edmondson, J.R.; Heywood, V.H.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea, 2nd. ed.; Cambridge University Press: Cambridge, UK, 1993. [Google Scholar]
- Correll, D.S.; Correll, H.B. Correll, Aquatic and Wetland Plants of Southwestern United States; Environmental Protection Agency: Washington, DC, USA, 1972; Volume 1. [Google Scholar]
- Casper, S.J.; Krausch, H.D. Pteridophyta und Anthophyta 1. In Subwasserflora von Mitteleuropa; Gustav Fischer Verlag: Stuttgart, Germany, 1980; Volume 23. [Google Scholar]
- Casper, S.J.; Krausch, H.D. Pteridophyta und Anthophyta 2. In Subwasserflora von Mitteleuropa; Gustav Fischer Verlag: Stuttgart, Germany, 1981; Volume 24. [Google Scholar]
- Krause, W. Charales (Charophyceae). In Subwasserflora von Mitteleuropa Bd 18; Spektrum Akademischer Verlag: Heidelberg, Germany, 1997. [Google Scholar]
- Strid, A.; Tan, K. Flora Hellenica; Koeltz Scientific Books: Königstein, Germany, 1997; Volume 1. [Google Scholar]
- Strid, A.; Tan, K. Flora Hellenica; Koeltz Scientific Books: Ruggell, Liechtenstein, 2002; Volume 2. [Google Scholar]
- Crow, G.E.; Hellquist, C.B. Crow, Aquatic and Wetland Plants of Northeastern North America; The University of Wisconsin Press: Madison, WI, USA, 2000. [Google Scholar]
- Pedrotti, C.C. Flora Dei Muschi d’6 Italia; Parte I; Delfino, A., Ed.; Libro Universitario: Roma, Italy, 2001. [Google Scholar]
- Pedrotti, C.C. Flora Dei Muschi d’6 Italia; Parte II; Delfino, A., Ed.; Libro Universitario: Roma, Italy, 2006. [Google Scholar]
- Schumacker, R.; Vana, J. Identification Keys to the Liverworts and Hornworts of Europe and Macaronesia; Ed. Sorus: Poznan, Poland, 2005. [Google Scholar]
- Froese, R.; Pauly, D. (Eds.) FishBase: World Wide Web Electronic Publication. Available online: www.fishbase.org (accessed on 10 February 2018).
- Hartog, C.; Segal, S. A new classification of the water-plant communities. Acta Bot. Neerl. 1964, 13, 367–393. [Google Scholar] [CrossRef]
- Economidis, P.S.; Dimitriou, E.; Pagoni, R.; Michaloudi, E.; Natsis, L. Introduced and translocated fish species in the inland waters of Greece. Fish. Manag. Ecol. 2000, 7, 239–250. [Google Scholar] [CrossRef]
- CEN. Water Quality—Sampling of Fish with Multimesh Gillnets; I.S. EN 14757: 2005; European Committee for Standardization (CEN): Brussels, Belgium, 2005. [Google Scholar]
- Kiernan, J.D.; Moyle, P.B.; Crain, P.K. Restoring native fish assemblages to a regulated California stream using the natural flow regime concept. Ecol. Appl. 2012, 22, 1472–1482. [Google Scholar] [CrossRef] [PubMed]

| Hydromorphological Characteristics | Zazari | Chimaditida | Petron | Vegoritida |
|---|---|---|---|---|
| Max water level (m a.s.l.) | 599.7 * | 592.0 * | 573.1 * | 518.0 |
| Surface area (km2) | 2.0 | 10.1 | 12.6 | 47.2 |
| Stored volume (106 m3) | 9.7 | 14.7 | 40.8 | 1206.2 |
| Mean depth (m) | 5.0 | 1.5 | 3.5 | 26.0 |
| Maximum depth (m) | 7.6 | 4.8 | 5.5 | 52.6 |
| Trophic level | EU | EU | EU | ME–EU |
| Scenario of Morphological Analysis | Min WL (m.a.s.l.) | |||
|---|---|---|---|---|
| Zazari | Chimaditida | Petron | Vegoritida | |
| envScenario (w1 = 30%, w2 = 70%) | 596.0 | 591.3 | 571.1 | 516.2 |
| eqScenario (w1 = w2 = 50%) | 594.9 | 591.2 | 570.3 | 490.9 |
| wuScenario (w1 = 70%, w2 = 30%) | 594.0 | 589.2 | 569.1 | 477.9 |
| Taxa | Life Form | Sensitive to HA | Z | C | P | V |
|---|---|---|---|---|---|---|
| Phragmites australis (Cav.) Steud. | Hel | YES * | X | X | X | X |
| Typha spp. L. | Hel | YES * | X | X | X | |
| Paspalum distichum L. | Hel | X | X | |||
| Rumex palustris Sm. | Hel | X | X | |||
| Lycopus europaeus L. | Hel | X | ||||
| Chaerophyllum bulbosum L. | Hel | X | ||||
| Mentha aquatica L. | Hel | X | X | X | ||
| Schoenoplectus lacustris (L.) Palla | Hel | YES * | X | X | ||
| Eleocharis mitracarpa Steud. | Hel | YES | X | |||
| Schoenoplectus litoralis (Schrad.) Palla | Hel | YES | X | |||
| Stachys palustris L. | Hel | X | X | |||
| Rorippa amphibia (L.) Besser | Hel | X | X | |||
| Alisma plantago-aquatica L. | Hel | YES* | X | |||
| Carex pseudocyperus L. | Hel | YES | X | |||
| Juncus subnodulosus Schrank | Hel | YES * | X | |||
| Cyperus longus L. | Hel | YES | X | |||
| Eleocharis palustris (L.) R. Br. | Hel | YES | X | |||
| Cladophora spp. Kutz. | Hyd | X | X | X | X | |
| Ceratophyllum demersum L. | Hyd | X | X | X | ||
| Ceratophyllum submersum L. | Hyd | X | X | |||
| Potamogeton perfoliatus L. | Hyd | X | X | X | ||
| Stuckenia pectinata (L.) Borner | Hyd | X | X | |||
| Myriophyllum spicatum L. | Hyd | YES * | X | X | ||
| Vallisneria spiralis L. | Hyd | X | X | |||
| Nitella furcata (Roxb. Ex Bruz.) Ag. | Hyd | X | X | |||
| Chara tomentosa L. | Hyd | X | X | |||
| Chara vulgaris L. | Hyd | X | X | |||
| Lemna minor L. | Hyd | X | X | |||
| Azolla filliculoides Lam. | Hyd | X | X | |||
| Spirodella polyrhiza (L.) Scheid. | Hyd | X | X | |||
| Ranunculus trichophyllus Chaix | Hyd | YES * | X | X | ||
| Lemna gibba L. | Hyd | X | ||||
| Persicaria amphibia (L.) S. F. Gray | Hyd | X | ||||
| Potamogeton trichoides Cham. & Schltdl. | Hyd | YES | X | |||
| Alisma gramineum Lej. | Hyd | YES * | X | |||
| Utricularia vulgaris L. | Hyd | X | X | |||
| Najas marina L. | Hyd | X | ||||
| Chara hispida L. | Hyd | X | ||||
| Zannichellia palustris L. | Hyd | YES * | X | |||
| Nitellopsis οbtusα (Desv. In Lois.) J. Gr. | Hyd | X | ||||
| Hydrocharis morsus-ranae L. | Hyd | X | ||||
| Lemna trisulca L. | Hyd | X | ||||
| Total | 42 | 15 | 9 | 19 | 21 | 24 |
| Taxa | Zazari | Chimaditida | Petron | Vegoritida | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 4 | |
| P. australis | A | A | F | A | A | R | A | F | - | O | R | O | O |
| Typha spp. | - | - | - | O | O | - | R | R | - | O | R | - | - |
| S. lacustris | - | - | - | O | - | - | O | R | - | - | - | - | - |
| A. plantago-aquatica | - | - | - | R | - | - | - | - | - | - | - | - | - |
| J. subnodulosus | - | - | - | R | - | - | - | - | - | - | - | - | - |
| M. spicatum | - | - | - | - | - | - | R | R | R | R | O | F | O |
| R. trichophyllus | R | - | - | - | - | - | - | - | - | R | R | R | - |
| A. gramineum | - | - | - | - | - | - | - | - | - | R | - | - | - |
| Z. palustris | - | - | - | - | - | - | R | - | - | - | - | - | - |
| Family/Species | Z | C | P | V | Reproductive Substrate | Τrophic Guild | Habitat | Reproductive Period | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | ||||||||
| Centrarchidae | |||||||||||||||||||
| L. gibbosus | I | I | I | LITH | INV | WCOL | + | + | + | ||||||||||
| Cyprinidae | |||||||||||||||||||
| A. thessalicus | Ν | N | PHLI | PLAN | WCOL | (+) | (+) | (+) | (+) | ||||||||||
| B. balcanicus * | N | N | N | LITH | INV | WCOL | + | + | + | ||||||||||
| C. gibelio | I | I | I | I | PHYT | OMNI | BENT | + | + | + | |||||||||
| C. carpio | N | Ν | N | N | PHYT | OMNI | BENT | + | + | + | |||||||||
| G. bulgaricus * | N | PSAM | INV | WCOL | + | + | |||||||||||||
| P. macedonicum | N | N | N | N | PHLI | INV | WCOL | + | + | ||||||||||
| P. parva | I | PHLI | OMNI | WCOL | + | + | + | ||||||||||||
| R. meridionalis | N | Ν | N | N | OSTR | OMNI | WCOL | (+) | (+) | (+ | (+) | (+) | |||||||
| R. rutilus | N | Ν | Ν | N | PHLI | OMNI | WCOL | + | + | ||||||||||
| S. erythrophthalmus ** | N | PHYT | OMNI | WCOL | + | + | + | ||||||||||||
| S. vardarensis | N | LITH | OMNI | WCOL | (+) | (+) | (+) | ||||||||||||
| T. tinca | N | Ν | Ν | N | PHYT | OMNI | BENT | + | + | + | + | ||||||||
| Esocidae | |||||||||||||||||||
| E. lucius | N | N | Ν | N | PHYT | PISC | WCOL | + | + | + | + | + | |||||||
| Percidae | |||||||||||||||||||
| P. fluviatilis | N | N | N | N | PHLI | INV/PISC | WCOL | + | + | + | + | + | + | ||||||
| Poeciliidae | |||||||||||||||||||
| G. holbrooki | I | I | I | OVI | INV | WCOL | + | + | + | + | + | + | + | ||||||
| Salmonidae | |||||||||||||||||||
| C. cf lavaretus | I | LITH | INV | WCOL | + | + | + | ||||||||||||
| O. kisutch * | I | - | - | - | |||||||||||||||
| O. mykiss * | I | I | - | - | - | ||||||||||||||
| S. fontinalis * | I | - | - | - | |||||||||||||||
| S. cf. trutta * | I | - | - | - | |||||||||||||||
| Siluridae | - | - | - | ||||||||||||||||
| S. glanis | N | PHYT | PISC | WCOL | + | + | + | ||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petriki, O.; Zervas, D.; Doulgeris, C.; Bobori, D. Assessing the Ecological Water Level: The Case of Four Mediterranean Lakes. Water 2020, 12, 2977. https://doi.org/10.3390/w12112977
Petriki O, Zervas D, Doulgeris C, Bobori D. Assessing the Ecological Water Level: The Case of Four Mediterranean Lakes. Water. 2020; 12(11):2977. https://doi.org/10.3390/w12112977
Chicago/Turabian StylePetriki, Olga, Dimitrios Zervas, Charalampos Doulgeris, and Dimitra Bobori. 2020. "Assessing the Ecological Water Level: The Case of Four Mediterranean Lakes" Water 12, no. 11: 2977. https://doi.org/10.3390/w12112977
APA StylePetriki, O., Zervas, D., Doulgeris, C., & Bobori, D. (2020). Assessing the Ecological Water Level: The Case of Four Mediterranean Lakes. Water, 12(11), 2977. https://doi.org/10.3390/w12112977







