In Situ Rates of Carbon and Nitrogen Uptake by Phytoplankton and the Contribution of Picophytoplankton in Kongsfjorden, Svalbard
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling
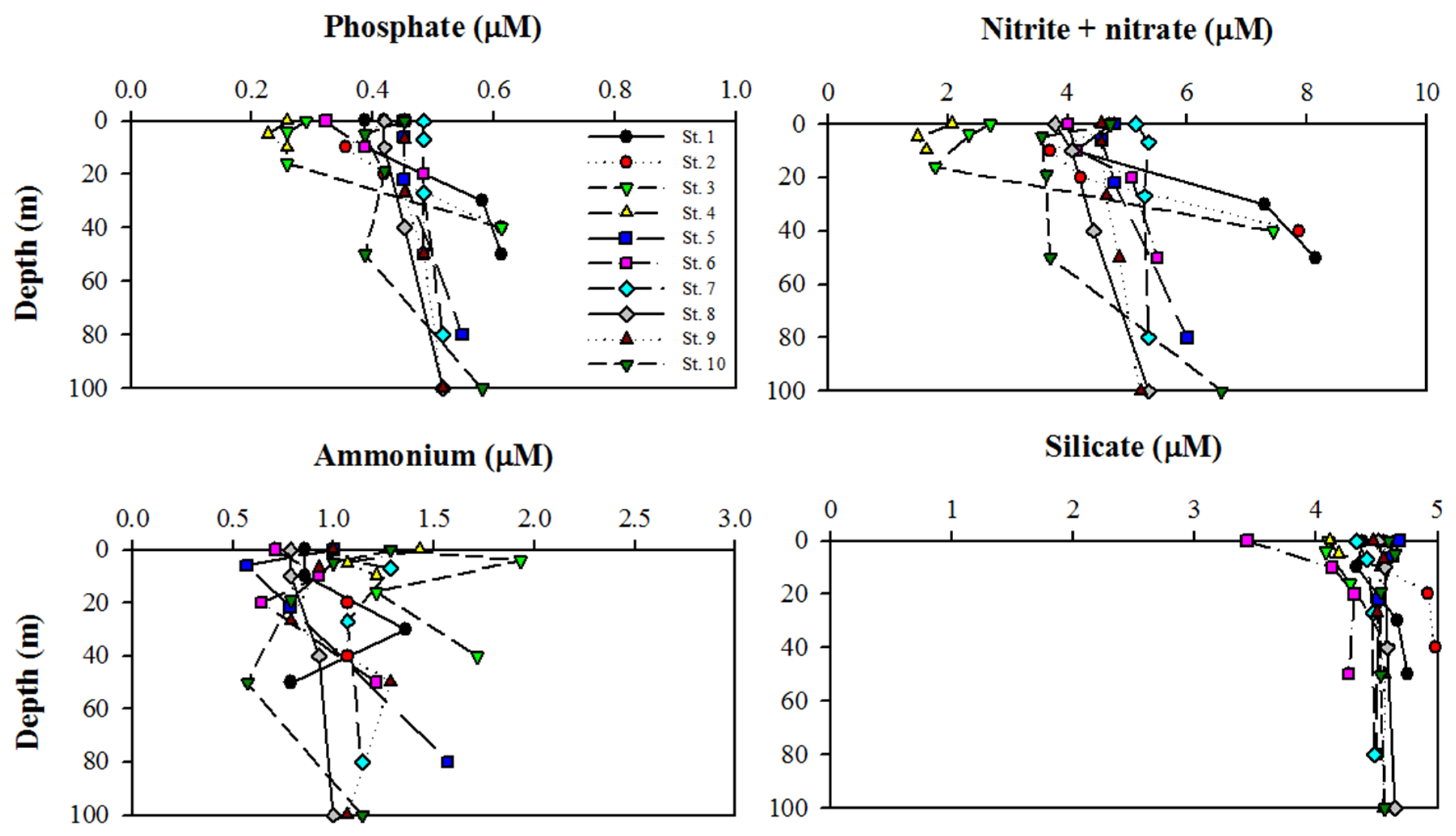
2.2. Major Inorganic Nutrient Analysis
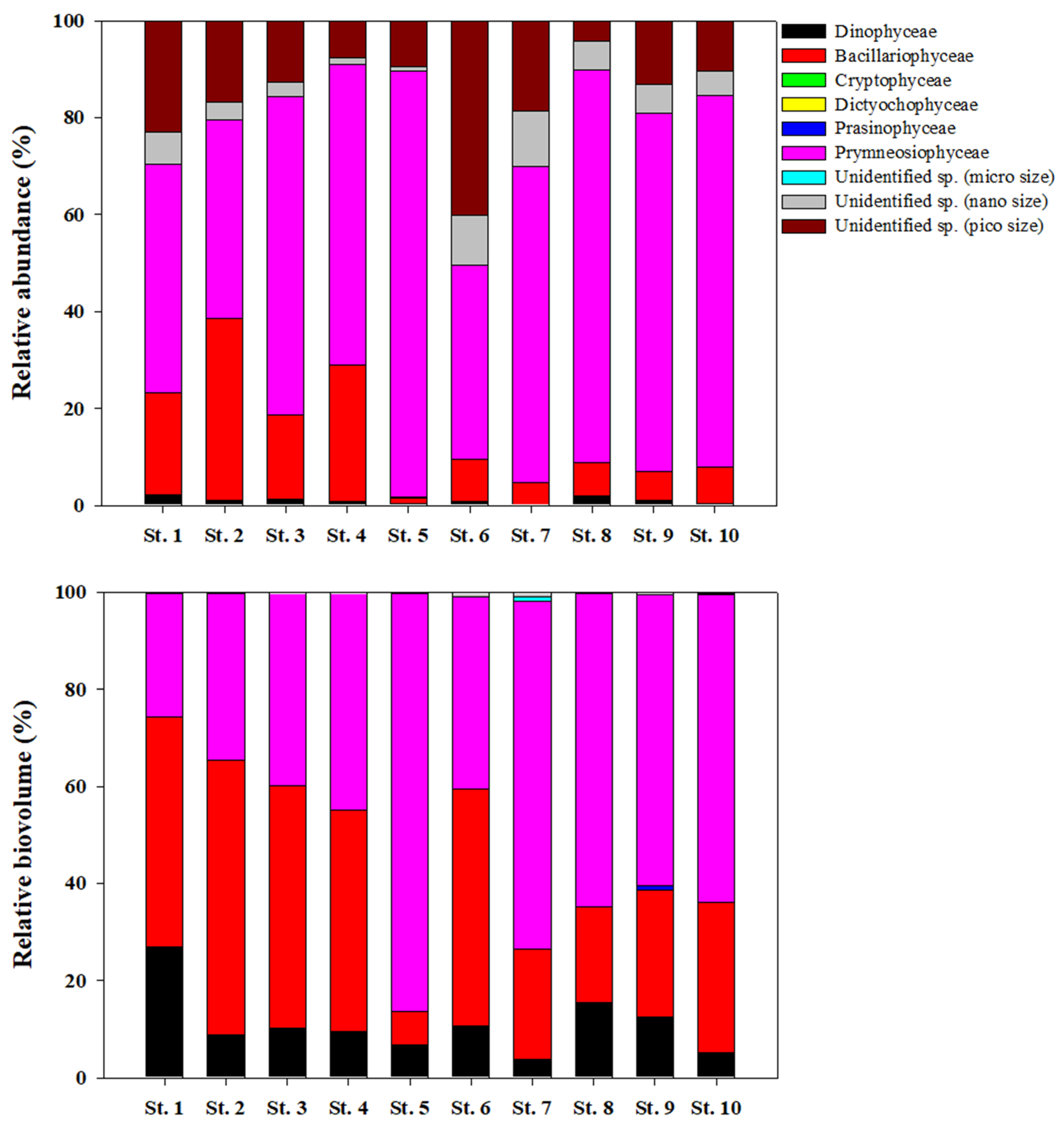
2.3. Chlorophyll a, Identification and Counts of Phytoplankton
2.4. Carbon and Nitrogen Uptake Experiments
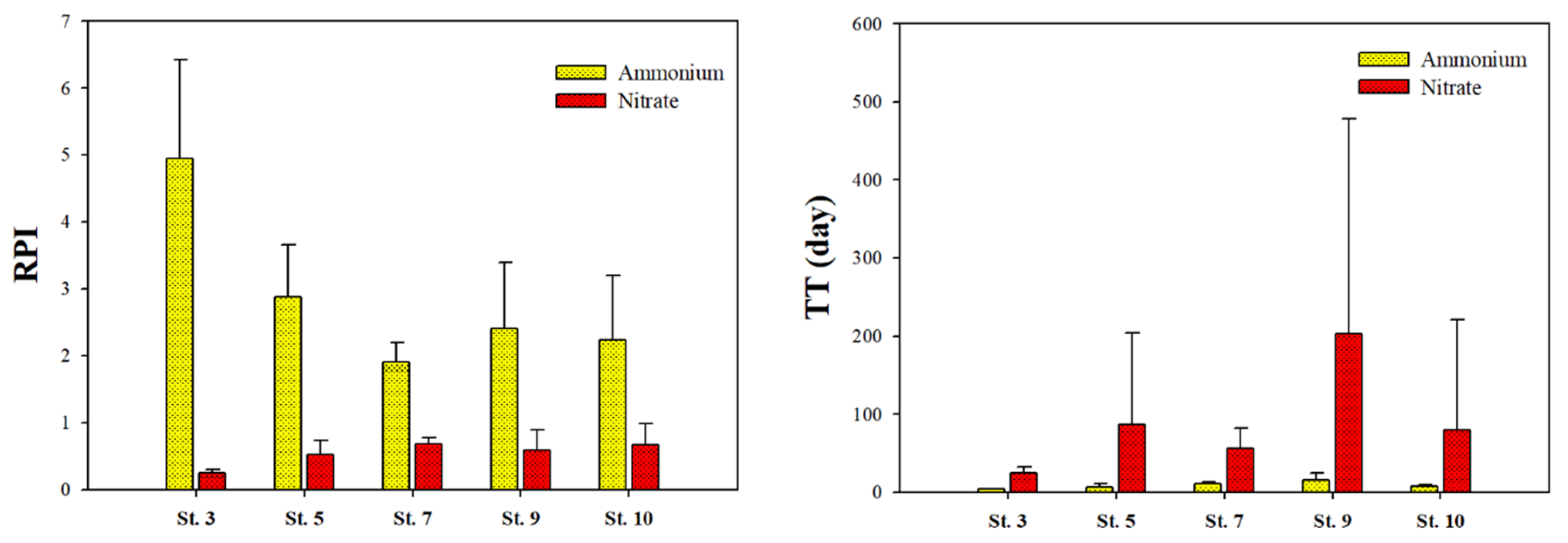
2.5. Relative Preference Index and Turnover Time of Picophytoplankton
2.6. Statistical Analysis
3. Results
3.1. Physical and Nutrient Properties in Kongsfjorden
3.2. Chlorophyll a, Size Distribution, and Community of Phytoplankton
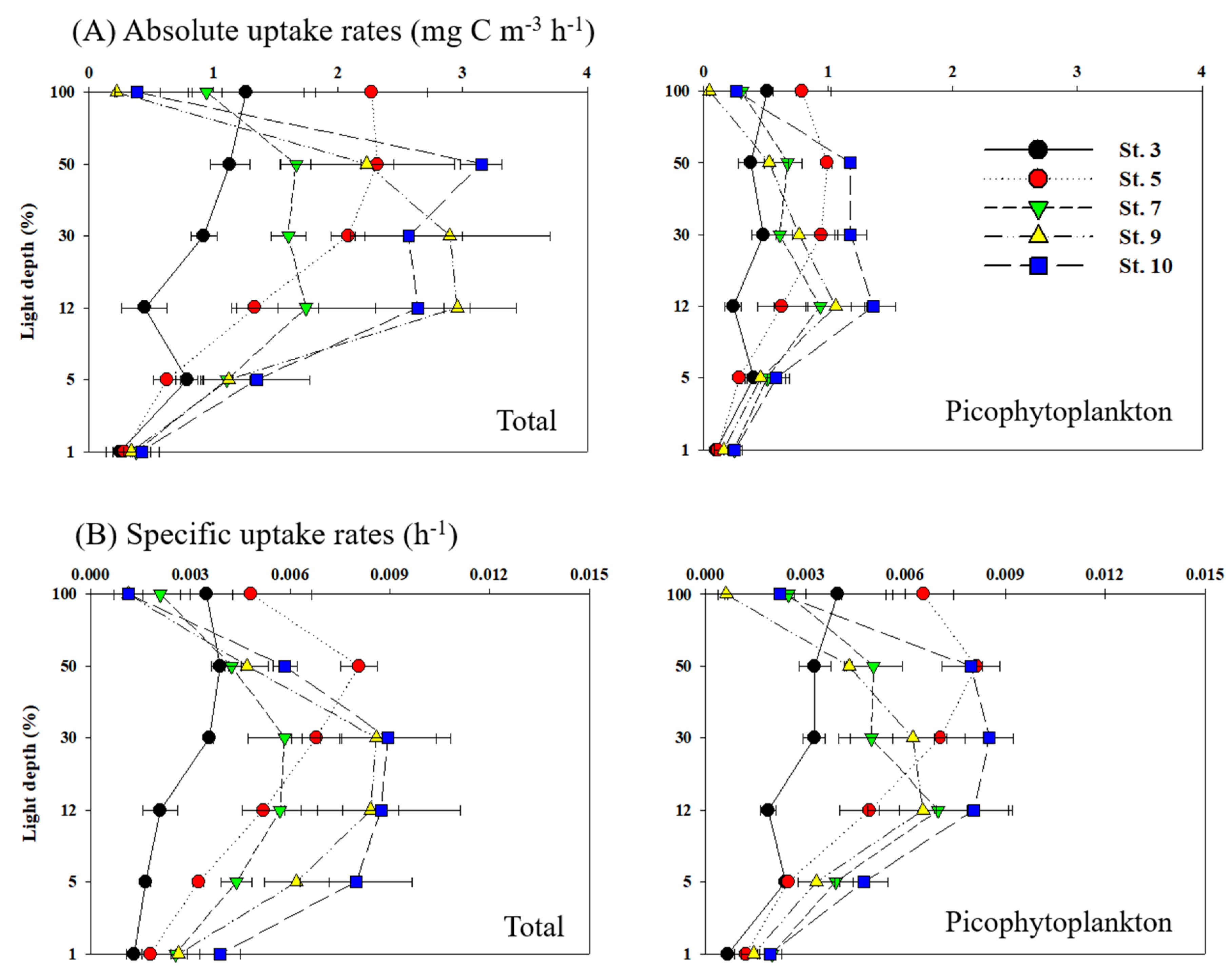
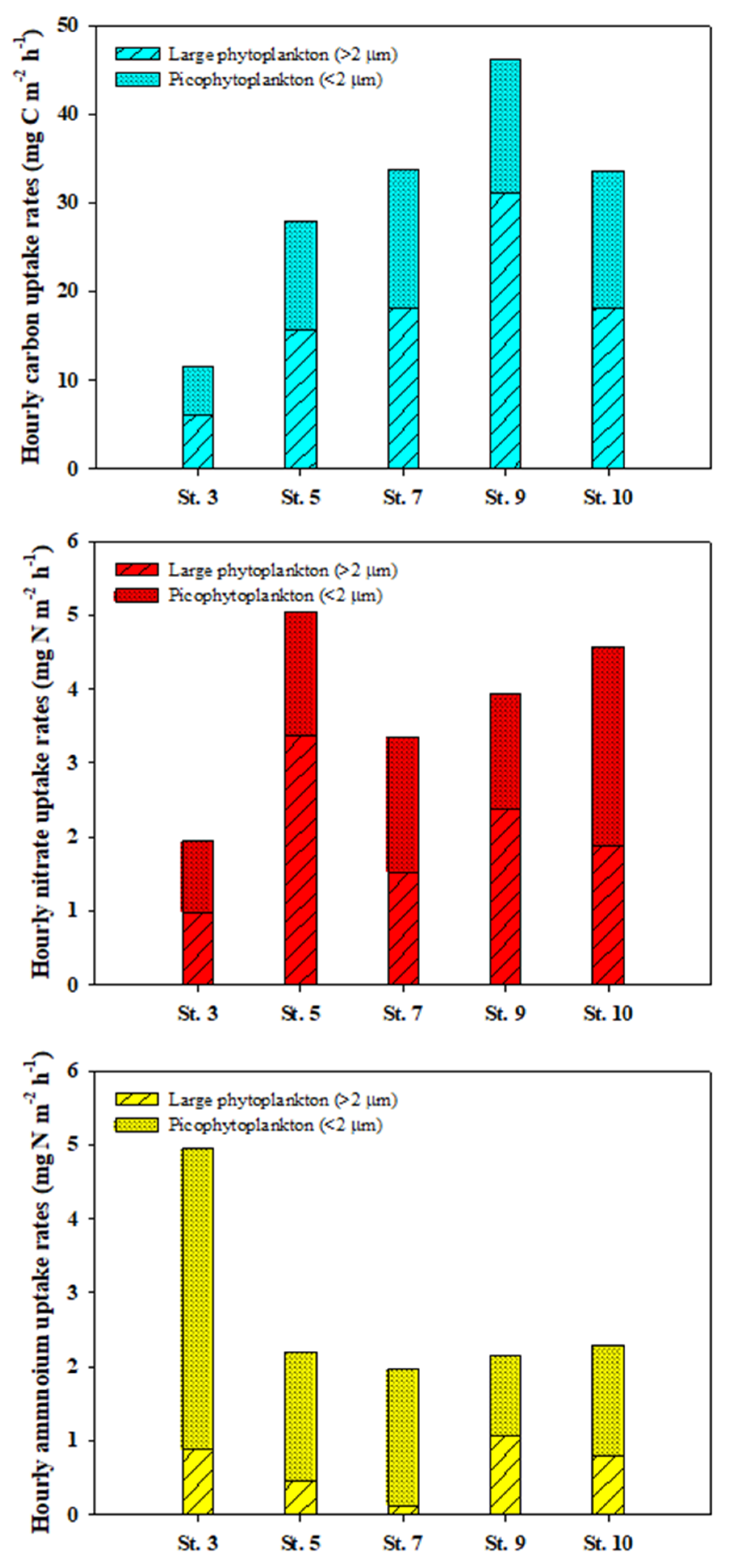
3.3. Carbon Uptake Rates of Phytoplankton
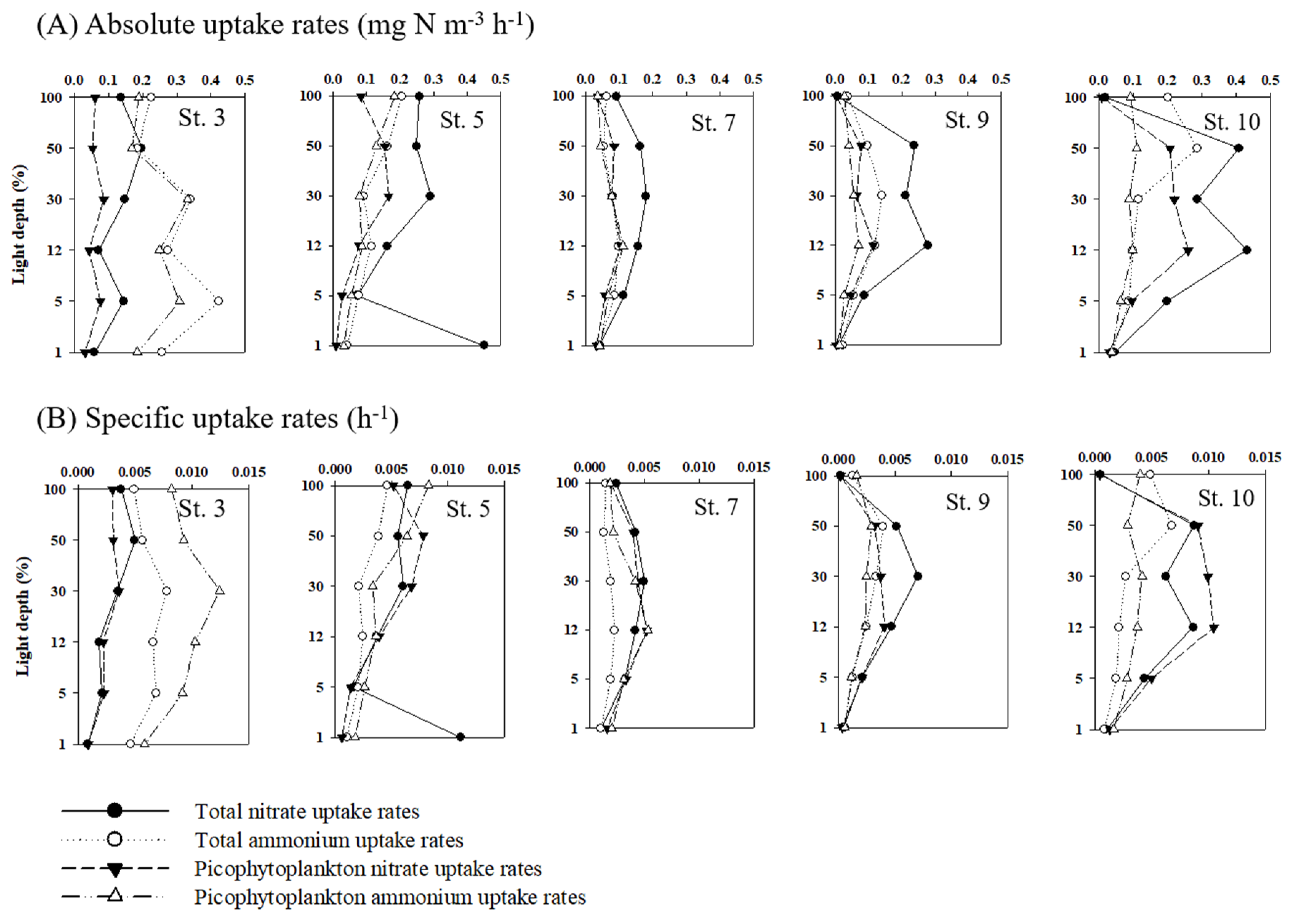
3.4. Nitrogen Uptake Rates of Phytoplankton
4. Discussion
4.1. Environmental and Phytoplankton Community Characteristics of Early Springtime in Kongsfjorden
4.2. Total Carbon and Nitrogen Uptake Rates of Phytoplankton
4.3. Contribution of Picophytoplankton to Carbon and Nitrogen Uptake Rates
5. Summary and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Azam, F. Oceanography: Microbial Control of Oceanic Carbon Flux: The Plot Thickens. Science 1998, 280, 694–696. [Google Scholar] [CrossRef]
- Arrigo, K.R. Marine microorganisms and global nutrient cycles. Nature 2005, 437, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Falkowski, P.G.; Raven, J.A. Aquatic Photosynthesis, 2nd ed.; Princeton University Press: Princeton, NJ, USA, 2007. [Google Scholar]
- Dugdale, R.C.; Goering, J.J. Uptake of new and regenerated forms of nitrogen in primary productivity. Limnol. Oceanogr. 1967, 12, 196–206. [Google Scholar] [CrossRef]
- Eppley, R.W.; Peterson, B.J. Particulate organic matter flux and planktonic new production in the deep ocean. Nature 1979, 282, 677–680. [Google Scholar] [CrossRef]
- Ducklow, H.; Steinberg, D.; Buesseler, K. Upper Ocean Carbon Export and the Biological Pump. Oceanography 2001, 14, 50–58. [Google Scholar] [CrossRef]
- Wassmann, P.; Slagstad, D.; Riser, C.W.; Reigstad, M. Modelling the ecosystem dynamics of the Barents Sea including the marginal ice zone. J. Mar. Syst. 2006, 59, 1–24. [Google Scholar] [CrossRef]
- Olli, K.; Wassmann, P.; Reigstad, M.; Ratkova, T.N.; Arashkevich, E.; Pasternak, A.; Matrai, P.A.; Knulst, J.; Tranvik, L.J.; Klais, R.; et al. The fate of production in the central Arctic Ocean—top–down regulation by zooplankton expatriates? Prog. Oceanogr. 2007, 72, 84–113. [Google Scholar] [CrossRef]
- Richardson, T.L.; Jackson, G.A. Small Phytoplankton and Carbon Export from the Surface Ocean. Science 2007, 315, 838–840. [Google Scholar] [CrossRef]
- Henson, S.A.; Le Moigne, F.A.C.; Giering, S.L.C. Drivers of Carbon Export Efficiency in the Global Ocean. Glob. Biogeochem. Cycles 2019, 33, 891–903. [Google Scholar] [CrossRef]
- Li, W.K.W.; McLaughlin, F.A.; Lovejoy, C.; Carmack, E.C. Smallest Algae Thrive as the Arctic Ocean Freshens. Science 2009, 326, 539. [Google Scholar] [CrossRef]
- Comiso, J.C.; Hall, D.K. Climate trends in the Arctic as observed from space. Wiley Interdiscip. Rev. Clim. Chang. 2014, 5, 389–409. [Google Scholar] [CrossRef] [PubMed]
- Morán, X.A.G.; López-Urrutia, Á.; Calvo-Díaz, A.; Li, W.K.W. Increasing importance of small phytoplankton in a warmer ocean. Glob. Chang. Biol. 2010, 16, 1137–1144. [Google Scholar] [CrossRef]
- Lee, S.H.; Yun, M.S.; Kim, B.K.; Joo, H.; Kang, S.-H.; Chang-Keun, K.; Whitledge, T.E. Contribution of small phytoplankton to total primary production in the Chukchi Sea. Cont. Shelf Res. 2013, 68, 43–50. [Google Scholar] [CrossRef]
- Hopkins, T.S. The GIN Sea—A synthesis of its physical oceanography and literature review 1972–1985. Earth Sci. Rev. 1991, 30, 175–319. [Google Scholar] [CrossRef]
- Hop, H.; Pearson, T.; Hegseth, E.N.; Kovacs, K.M.; Wiencke, C.; Kwasniewski, S.; Eiane, K.; Mehlum, F.; Gulliksen, B.; Wlodarska-Kowalczuk, M.; et al. The marine ecosystem of Kongsfjorden, Svalbard. Polar Res. 2002, 21, 167–208. [Google Scholar] [CrossRef]
- Payne, C.M.; Roesler, C. Characterizing the influence of Atlantic water intrusion on water mass formation and phytoplankton distribution in Kongsfjorden, Svalbard. Cont. Shelf Res. 2019, 191, 104005. [Google Scholar] [CrossRef]
- Svendsen, H.; Beszczynska-Møller, A.; Hagen, J.O.; Lefauconnier, B.; Tverberg, V.; Gerland, S.; Ørbæk, J.B.; Bischof, K.; Papucci, C.; Zajaczkowski, M.; et al. The physical environment of Kongsfjorden-Krossfjorden, an Arctic fjord system in Svalbard. Polar Res. 2002, 21, 133–166. [Google Scholar]
- Stempniewicz, L.; Błachowiak-Samołyk, K.; Węsławski, J.M. Impact of climate change on zooplankton communities, seabird populations and arctic terrestrial ecosystem—A scenario. Deep Sea Res. Part II 2007, 54, 2934–2945. [Google Scholar] [CrossRef]
- Iversen, K.R.; Seuthe, L. Seasonal microbial processes in a high-latitude fjord (Kongsfjorden, Svalbard): I. Heterotrophic bacteria, picoplankton and nanoflagellates. Polar Biol. 2011, 34, 731–749. [Google Scholar] [CrossRef]
- Eilertsen, H.C.; Taasen, J.P.; Weslawski, J.M. Phytoplankton studies in the fjords of West Spitzbergen: Physical environment and production in spring and summer. J. Plankton Res. 1989, 11, 1245–1260. [Google Scholar] [CrossRef]
- Richardson, A.J.; Schoeman, D.S. Climate impact on plankton ecosystems in the Northeast Atlantic. Science 2004, 305, 1609–1612. [Google Scholar] [CrossRef] [PubMed]
- Trudnowska, E.; Basedow, S.L.; Blachowiak-Samolyk, K. Mid-summer mesozooplankton biomass, its size distribution, and estimated production within a glacial Arctic fjord (Hornsund, Svalbard). J. Mar. Syst. 2014, 137, 55–66. [Google Scholar] [CrossRef]
- Stroeve, J.; Markus, T.; Meier, W.N.; Miller, J. Recent changes in the Arctic melt season. Ann. Glaciol. 2006, 44, 367–374. [Google Scholar] [CrossRef][Green Version]
- Serreze, M.C.; Holland, M.M.; Stroeve, J. Perspectives on the Arctic’s Shrinking Sea-Ice Cover. Science 2007, 315, 1533–1536. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.Z.; Guo, C.Y.; Luo, W.; Cai, M.H.; He, J.F. The distribution of picoplankton and nanoplankton in Kongsfjorden, Svalbard during late summer 2006. Polar Biol. 2009, 32, 1233–1238. [Google Scholar] [CrossRef][Green Version]
- Sinha, R.K.; Krishnan, K.P.; Hatha, A.A.M.; Rahiman, M.; Thresyamma, D.D.; Kerkar, S. Diversity of retrievable heterotrophic bacteria in Kongsfjorden, an Arctic fjord. Environ. Microbiol. 2017, 48, 51–61. [Google Scholar] [CrossRef]
- Piwosz, K.; Walkusz, W.; Hapter, R.; Wieczorek, P.; Hop, H.; Wiktor, J. Comparison of productivity and phytoplankton in a warm (Kongsfjorden) and a cold (Hornsund) Spitsbergen fjord in mid-summer 2002. Polar Biol. 2009, 32, 549–559. [Google Scholar] [CrossRef]
- Poole, H.H.; Atkins, W.R.G. Photo-electric measurements of submarine illumination throughout the year. J. Mar. Biol. Assoc. UK 1929, 16, 297–324. [Google Scholar] [CrossRef]
- Parsons, T.R.; Maita, Y.; Lalli, C.M. A Manual of Chemical and Biological Methods for Seawater Analysis; Pergamon Press: Oxford, UK, 1984; 173p. [Google Scholar]
- Bérard-Therriault, L.; Poulin, M.; Bossé, L. Guide D’identification du Phytoplancton Marin de L’estuaire et du Golfe du Saint-Laurent Incluantégalement Certains Protozoaires; Conseil National de Researches du Canada: Ottawa, ON, Canada, 1999.
- Booth, B.C. Estimating cell concentration and biomass of autotrophic plankton using microscopy. In Handbook of Methods in Aquatic Microbial Ecology; Kemp, P.F., Sherr, B.F., Sherr, E.B., Cole, J.J., Eds.; Lewis Publishers: Boca Raton, FL, USA, 1993; pp. 199–205. [Google Scholar]
- Tomas, C.R. Identifying Marine Phytoplankton; Academic Press: San Diego, CA, USA, 1997. [Google Scholar]
- Kang, S.H.; Fryxell, G.A.; Roelke, D.L. Fragilariopsis cylindrus compared with other species of the diatom family Bacillariaceae in Antarctic marginal ice-edge zones. Nova Hedwig. Beih. 1993, 106, 335–352. [Google Scholar]
- Garneau, M.-È.; Gosseling, M.; Klein, B.; Tremblay, J.-È.; Fouilland, E. New and regenerated production during a late summer bloom in an Arctic polynya. Mar. Ecol. Prog. Ser. 2007, 345, 13–26. [Google Scholar] [CrossRef]
- Dugdale, R.C.; Wilkerson, F.P. The use of 15N to measure nitrogen uptake in eutrophic ocean; experimental considerations. Limnol. Oceanogr. 1986, 31, 637–689. [Google Scholar] [CrossRef]
- Hama, T.; Miyazaki, T.; Ogawa, Y.; Iwakuma, T.; Takahashi, M. Measurement of Photosynthetic Production of a Marine Phytoplankton Population Using a STable 13C Isotope. Mar. Biol. 1983, 73, 31–36. [Google Scholar] [CrossRef]
- Hodal, H.; Kristiansen, S. The importance of small-called phytoplankton in spring blooms at the marginal ice zone in the northern Barents Sea. Deep Sea Res. Part II 2008, 55, 2176–2185. [Google Scholar] [CrossRef]
- McCarthy, J.J.; Taylor, W.R.; Taft, J.L. Nitrogenous nutrition of the plankton in the Chesapeake Bay. I. Nutrient availability and phytoplankton preferences. Limnol. Oceanogr. 1977, 22, 996–1011. [Google Scholar] [CrossRef]
- Gu, B.; Alexander, V. Dissolved nitrogen uptake by a Cyanobacterial bloom (Anabaena flos-aquae) in a subarctic lake. J. Appl. Environ. Microbiol. 1993, 59, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Hodal, H.; Falk-Petersen, S.; Hop, H.; Kristiansen, S.; Reigstad, M. Spring bloom dynamics in Kongsfjorden, Svalbard: Nutrients, phytoplankton, protozoans and primary production. Polar Biol. 2012, 35, 191–203. [Google Scholar] [CrossRef]
- Hegseth, E.N.; Tverberg, V. Effect of Atlantic water inflow on timing of the phytoplankton spring bloom in a high Arctic fjord (Kongsfjorden, Svalbard). J. Mar. Syst. 2013, 113–114, 94–105. [Google Scholar] [CrossRef]
- von Quillfeldt, C.H. Common Diatom Species in Arctic Spring Blooms: Their Distribution and Abundance. Bot. Mar. 2000, 43, 499–516. [Google Scholar] [CrossRef]
- Piquet, A.M.-T.; van de Poll, W.H.; Visser, R.J.W.; Wiencke, C.; Bolhuis, H.; Buma, A.G.J. Springtime phytoplankton dynamics in Arctic Krossfjorden and Kongsfjorden (Spitsbergen) as a function of glacier proximity. Biogeosciences 2014, 11, 2263–2279. [Google Scholar] [CrossRef]
- Redifield, A.C. The biological control of chemical factors in the environment. Am. Sci. 1958, 46, 205–221. [Google Scholar]
- Thingstad, T.F.; Sakshaug, E. Control of phytoplankton growth in nutrient recycling ecosystems. Theory and Terminology. Mar. Ecol. Prog. Ser. 1990, 63, 261–272. [Google Scholar] [CrossRef]
- van De Poll, W.H.; Maat, D.S.; Fischer, P.; Rozema, P.D.; Daly, O.B.; Koppelle, S.; Visser, R.J.W.; Buma, A.G.J. Atlantic advection driven changes in glacial meltwater: Effects on phytoplankton chlorophyll a and taxonomic composition in Kongsfjorden, Spitsbergen. Front. Mar. Sci. 2016, 3, 200. [Google Scholar] [CrossRef]
- Probyn, T.A. Nitrogen uptake by size-fractionated phytoplankton populations in the southern Benguela upwelling system. Mar. Ecol. Prog. Ser. 1985, 22, 249–258. [Google Scholar] [CrossRef]
- Koike, I.; Holm-Hansen, O.; Biggs, D.C. Inorganic nitrogen metabolism by Antarctic phytoplankton with special reference to ammonium cycling. Mar. Ecol. Prog. Ser. 1986, 30, 105–116. [Google Scholar] [CrossRef]
- Dugdale, R.C.; Wilkerson, F.P.; Hogue, V.E.; Marchi, A. The role of ammonium and nitrate in spring bloom development in San Francisco Bay. Estuar. Coast. Shelf Sci. 2007, 73, 17–29. [Google Scholar] [CrossRef]
- Keck, A.; Wiktor, J.; Hapter, R.; Nilsen, R. Phytoplankton assemblages related to physical gradients in an arctic, glacier-fed fjord in summer. ICES J. Mar. Sci. 1999, 56, 203–214. [Google Scholar] [CrossRef]
- Hop, H.; Falk-Petersen, S.; Svendsen, H.; Kwasniewski, S.; Pavlov, V.; Pavlova, O.; Søreide, J.E. Physical and biological characteristics of the pelagic system across Fram Strait to Kongsfjorden. Prog. Oceanogr. 2006, 71, 182–231. [Google Scholar] [CrossRef]
- Van de Poll, W.H.; Kulk, G.; Rozema, P.D.; Brussaard, C.P.D.; Visser, R.J.W.; Buma, A.G.J. Contrasting glacial meltwater effects on post-bloom phytoplankton on temporal and spatial scales in Kongsfjorden, Spitsbergen. Elem. Sci. Anthr. 2018, 6, 50. [Google Scholar] [CrossRef]
- Raven, J.A. The twelfth Tansley Lecture. Small is beautiful: The picophytoplankton. Funct. Ecol. 1998, 12, 503–513. [Google Scholar] [CrossRef]
- Callieri, C. Picophytoplankton in freshwater ecosystems: The importance of small-sized phototrophs. Freshw. Rev. 2008, 1, 1–28. [Google Scholar] [CrossRef]
- Finkel, Z.V.; Beardall, J.; Flynn, K.J.; Quigg, A.; Rees, T.A.V.; Raven, J.A. Phytoplankton in a changing world: Cell size and elemental stoichiometry. J. Plankton Res. 2010, 32, 119–137. [Google Scholar] [CrossRef]
- Lourey, M.J.; Thompson, P.A.; McLaughlin, M.J.; Bonham, P.; Feng, M. Primary production and phytoplankton community structure during a winter shelf-scale phytoplankton bloom off Western Australia. Mar. Biol. 2013, 160, 335–369. [Google Scholar] [CrossRef]
- Huot, Y.; Babin, M.; Bruyant, F.; Grob, C.; Twardowski, M.; Claustre, H. Relationship between photosynthetic parameters and different proxies of phytoplankton biomass in the subtropical ocean. Biogeosciences 2017, 4, 853–868. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, B.K.; Yun, M.S.; Joo, H.; Yang, E.J.; Kim, Y.N.; Shin, H.C.; Lee, S. Spatial distribution of phytoplankton productivity in the Amundsen Sea, Antarctica. Polar Biol. 2012, 35, 1721–1733. [Google Scholar] [CrossRef]
- Gaard, E.; Norði, Á.G.; Simonsen, K. Environmental effects on phytoplankton production in a Northeast Atlantic fjord, Faroe Islands. J. Plankton Res. 2011, 33, 947–959. [Google Scholar] [CrossRef]
- Lee, S.H.; Whitledge, T.E. Primary and new production in the deep Canada Basin during summer 2002. Polar Biol. 2005, 28, 190–197. [Google Scholar] [CrossRef]
- Lee, S.H.; Whitledge, T.E.; Kang, S.-H. Recent carbon and nitrogen uptake rates of phytoplankton in Bering Strait and the Chukchi Sea. Cont. Shelf Res. 2007, 27, 2231–2249. [Google Scholar] [CrossRef]
- Yun, M.S.; Chung, K.H.; Zimmermann, S.; Zhao, J.; Joo, H.M.; Lee, S.H. Phytoplankton productivity and its response to higher light levels in the Canada Basin. Polar Biol. 2012, 35, 257–268. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, J.H.; Lee, H.; Kang, J.J.; Lee, J.H.; Lee, D.; An, S.H.; Stockwell, D.A.; Whitledge, T.E. Field-obtained carbon and nitrogen uptake rates of phytoplankton in the Laptev and East Siberian seas. Biogeosci. Discuss. 2017, 1–32. [Google Scholar] [CrossRef]
- Harrison, W.G.; Cota, G.F. Primary production in polar waters: Relation to nutrient availability. Polar Res. 1991, 10, 87–104. [Google Scholar] [CrossRef]
- Willis, K.J.; Cottier, F.R.; Kwasniewski, S. Impact of warm water advection on the winter zooplankton community in an Arctic fjord. Polar Biol. 2008, 31, 475–481. [Google Scholar] [CrossRef]
- Walkusz, W.; Kwasniewski, S.; Falk-Petersen, S.; Hop, H.; Tverberg, V.; Wieczorek, P.; Weslawski, J.M. Seasonal and spatial changes in zooplankton composition in the glacially influenced Kongsfjorden, Svalbard. Polar Res. 2009, 28, 254–281. [Google Scholar] [CrossRef]
- Halbach, L.; Vihtakari, M.; Duarte, P.; Everett, A.; Granskog, M.A.; Hop, H.; Kauko, H.M.; Kristiansen, S.; Myhre, P.I.; Pavlov, A.K.; et al. Tidewater Glaciers and Bedrock Characteristics Control the Phytoplankton Growth Environment in a Fjord in the Arctic. Front. Mar. Sci. 2019, 6, 254. [Google Scholar] [CrossRef]
- Partensky, F.; Garczarek, L. Prochlorococcus: Advantages and limits of minimalism. Annu. Rev. Mar. Sci. 2010, 2, 305–331. [Google Scholar] [CrossRef]
- Poulton, A.J.; Holligan, P.M.; Hickman, A.; Kim, Y.-N.; Adey, T.R.; Stinchcombe, M.C.; Holeton, C.; Root, S.; Woodward, E.M.S. Phytoplankton carbon fixation, chlorophyll-biomass and diagnostic pigments in the Atlantic Ocean. Deep Sea Res. Part II 2006, 53, 1593–1610. [Google Scholar] [CrossRef]
- Bhavya, P.S.; Lee, J.H.; Lee, H.W.; Kang, J.J.; Lee, J.H.; Lee, D.; An, S.H.; Stockwell, D.A.; Whitledge, T.E.; Lee, S.H. First in situ estimations of small phytoplankton carbon and nitrogen uptake rates in the Kara, Laptev, and East Siberian seas. Biogeosciences 2018, 15, 5503–5517. [Google Scholar] [CrossRef]
- Tremblay, G.; Belzile, C.; Gosselin, M.; Poulin, M.; Roy, S.; Tremblay, J.-É. Late summer phytoplankton distribution along a 3500 km transect in Canadian Arctic waters: Strong numerical dominance by picoeukaryotes. Aquat. Microb. Ecol. 2009, 54, 55–70. [Google Scholar] [CrossRef]
- Ardyna, M.; Gosselin, M.; Michel, C.; Poulin, M.; Tremblay, J.-É. Environmental forcing of phytoplankton community structure and function in the Canadian High Arctic: Contrasting oligotrophic and eutrophic regions. Mar. Ecol. 2011, 442, 37–57. [Google Scholar] [CrossRef]
- Metfies, K.; von Appen, W.-J.; Kilias, E.; Nicolaus, A.; Nöthig, E.-M. Biogeography and Photosynthetic Biomass of Arctic Marine Pico-Eukaroytes during Summer of the Record Sea Ice Minimum 2012. PLoS ONE 2016, 11, e0148512. [Google Scholar] [CrossRef]
- Yun, M.S.; Kim, B.K.; Joo, H.T.; Yang, E.J.; Nishino, S.; Chung, K.H.; Kang, S.-H.; Lee, S.H. Regional productivity of phytoplankton in the Western Arctic Ocean during summer in 2010. Deep Sea Res. Part II 2015, 120, 61–71. [Google Scholar] [CrossRef]
- Tremblay, J.-É.; Legendre, L.; Klein, B.; Therriault, J.-C. Size-differential uptake of nitrogen and carbon in a marginal sea (Gulf of St. Lawrence, Canada): Significance of diel periodicity and urea uptake. Deep Sea Res. Part II 2000, 47, 489–518. [Google Scholar] [CrossRef]
- Goeyens, L.; Tréguer, P.; Baumann, M.E.M.; Baeyens, W.; Dehairs, F. The leading role of ammonium in the nitrogen uptake regime of Southern Ocean marginal ice zones. J. Mar. Syst. 1995, 6, 345–361. [Google Scholar] [CrossRef]
- Xu, J.; Glibert, P.M.; Liu, H.; Yin, K.; Yuan, X.; Chen, M.; Harrision, P. Nitrogen sources and rates of phytoplankton uptake in different regions of Hong Kong Waters in summer. Estuar. Coast. 2012, 35, 559–571. [Google Scholar] [CrossRef]
- Kim, B.K.; Joo, H.T.; Song, H.J.; Yang, E.J.; Lee, S.H.; Hahm, D.; Rhee, T.S.; Lee, S.H. Large seasonal variation in phytoplankton production in the Amundsen Sea. Polar Biol. 2015, 38, 319–331. [Google Scholar] [CrossRef]
- MacIsaac, J.J.; Dugdale, R.C. The kinetics of nitrate and ammonia uptake by natural populations of marine phytoplankton. Deep Sea Res. 1969, 16, 45–57. [Google Scholar] [CrossRef]
- Paasche, E.; Kristiansen, S. Nitrogen nutrition of the phytoplankton in the Oslofjord. Estuar. Coast. Shelf Sci. 1982, 14, 237–249. [Google Scholar] [CrossRef]
- Dortch, Q. The interaction between ammonium and nitrate uptake in phytoplankton. Mar. Ecol. Prog. Ser. 1990, 61, 183–201. [Google Scholar] [CrossRef]
- Cochlan, W.P.; Harrison, P.J. Uptake of nitrate, ammonium, and urea by nitrogen-starved cultures of Micromonas pusilla (Prasinophyceae): Transient responses. J. Phycol. 1991, 27, 673–679. [Google Scholar] [CrossRef]
- L’Helguen, S.; Maguer, J.-F.; Caradec, J. Inhibition kinetics of nitrate uptake by ammonium in size-fractionated oceanic phytoplankton communities: Implications for new production and f-ratio estimates. J. Plankton Res. 2008, 30, 1179–1188. [Google Scholar] [CrossRef]
- Wada, E.; Terazaki, M.; Kabaya, Y.; Nemoto, T. 15N and 13C abundances in the Antarctic Ocean with emphasis on the biogeochemical structure of the food web. Deep Sea Res. Part A 1987, 34, 829–841. [Google Scholar] [CrossRef]
- Corbisier, T.N.; Petti, M.A.V.; Skowronski, R.S.P.; Brito, T.A.S. Trophic relationships in the nearshore zone of Martel Inlet (King George Island, Antarctica): δ13C stable-isotope analysis. Polar Biol. 2004, 27, 75–82. [Google Scholar] [CrossRef]
- Kim, B.K.; Lee, J.H.; Joo, H.T.; Song, H.J.; Yang, E.J.; Lee, S.H.; Lee, S.H. Macromolecular compositions of phytoplankton in the Amundsen Sea, Antarctica. Deep Sea Res. Part II 2016, 123, 42–49. [Google Scholar] [CrossRef]
- Baumann, M.E.M.; Lancelot, C.; Brandini, F.P.; Sakshaug, E.; John, D.M. The taxonomic identity of the cosmopolitan prymnesiophyte Phaeocystis: A morphological and ecophysiological approach. J. Mar. Syst. 1994, 5, 5–22. [Google Scholar] [CrossRef]
- Kendall, C.; Silva, S.R.; Kelly, V.J. Carbon and nitrogen isotopic compositions of particulate organic matter in four large river systems across the United States. Hydrol. Process. 2001, 15, 1301–1346. [Google Scholar] [CrossRef]
- Schoemann, V.; Becquevort, S.; Stefels, J.; Rousseau, V.; Lancelot, C. Phaeocystis blooms in the global ocean and their controlling mechanisms: A review. J. Sea Res. 2005, 53, 43–66. [Google Scholar] [CrossRef]
- Fouilland, E.; Gosselin, M.; Rivkin, R.B.; Vasseur, C.; Mostajir, B. Nitrogen uptake by heterotrophic bacteria and phytoplankton in Arctic surface waters. J. Plankton Res. 2007, 29, 369–376. [Google Scholar] [CrossRef]
- Kirchman, D.L.; Ducklow, H.W.; McCarthy, J.J.; Garside, C. Biomass and nitrogen uptake by heterotrophic bacteria during the spring phytoplankton bloom in the North Atlantic Ocean. Deep Sea Res. Part I 1994, 41, 879–895. [Google Scholar] [CrossRef]
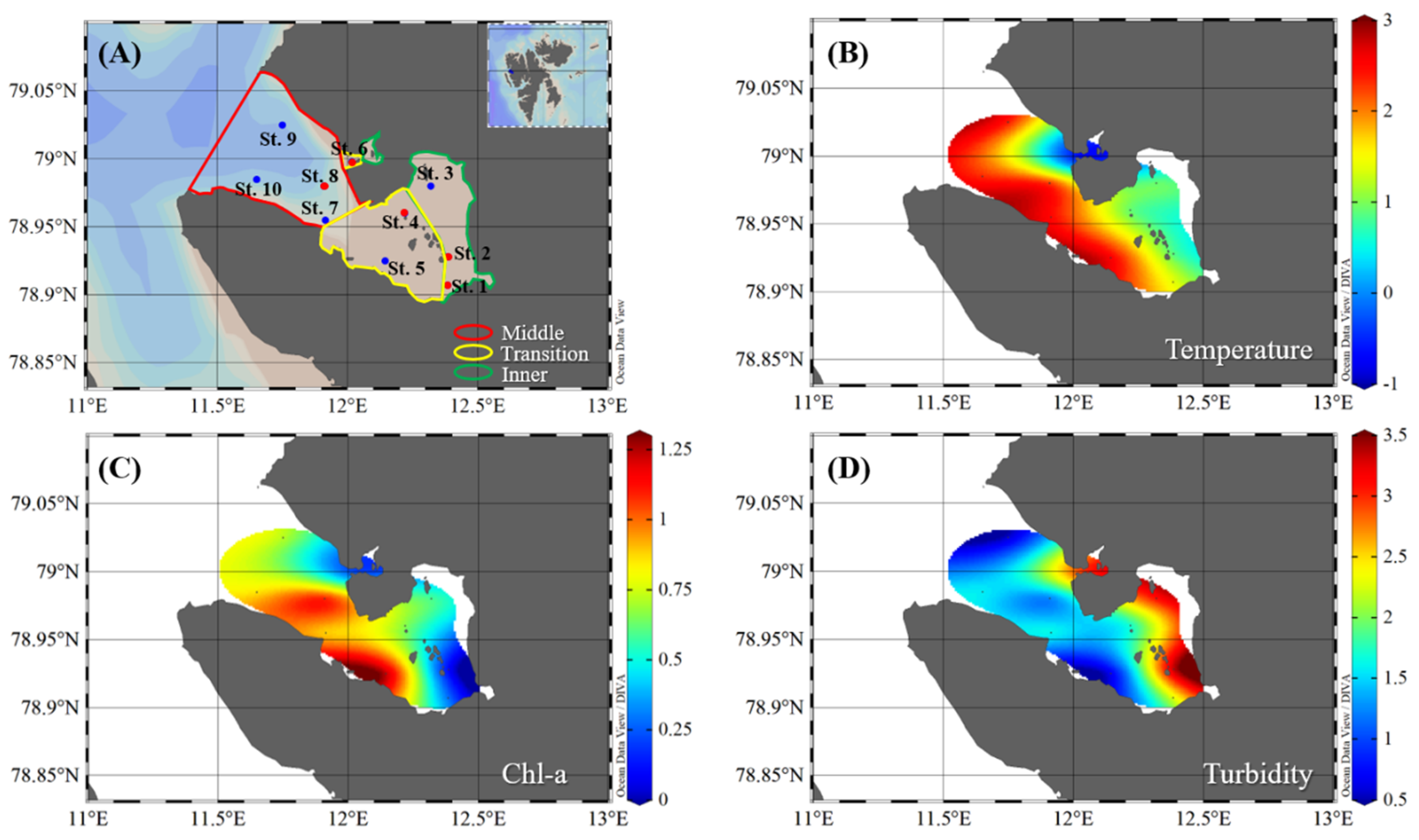

| Station | Latitude (°N) | Longitude (°E) | Date (Day, Month Year) | Bottom Depth (m) | Euphotic Depth (m) | Sampling Depth (m) | Zone |
|---|---|---|---|---|---|---|---|
| St. 1 | 78.91 | 12.39 | 4 May 2017 | 63 | - | 0, 10, 30, 50 | Inner |
| St. 2 | 78.93 | 12.39 | 4 May 2017 | 47 | - | 0, 10, 20, 40 | Inner |
| St. 3 | 78.98 | 12.32 | 7 May 2017 | 55 | 16 | 0, 4, 16, 40 | Inner |
| St. 4 | 78.96 | 12.22 | 7 May 2017 | 14 | - | 0, 5, 10 | Transition |
| St. 5 | 78.93 | 12.15 | 4 May 2017 | 110 | 22 | 0, 6, 22, 80 | Transition |
| St. 6 | 79 | 12.02 | 7 May 2017 | 63 | - | 0, 10, 20, 50 | Transition |
| St. 7 | 78.96 | 11.92 | 7 May 2017 | 357 | 27 | 0, 7, 27, 80 | Middle |
| St. 8 | 78.98 | 11.91 | 8 May 2017 | 226 | - | 0, 10, 40, 100 | Middle |
| St. 9 | 79.03 | 11.75 | 8 May 2017 | 211 | 27 | 0, 7, 27, 50, 100 | Middle |
| St. 10 | 78.99 | 11.65 | 8 May 2017 | 287 | 19 | 0, 5, 19, 50, 100 | Middle |
| Station | Depth (m) | Microphytoplankton (%) | Nanophytoplankton (%) | Picophytoplankton (%) |
|---|---|---|---|---|
| St. 1 | 0 | 38.1 | 37.6 | 24.2 |
| 10 | 54.0 | 26.0 | 20.0 | |
| 30 | 71.6 | 13.5 | 14.9 | |
| 50 | 80.0 | 11.2 | 8.8 | |
| St. 2 | 0 | 9.7 | 53.5 | 36.8 |
| 10 | 48.3 | 21.3 | 30.3 | |
| 20 | 79.2 | 5.7 | 15.1 | |
| 40 | 73.8 | 12.7 | 13.5 | |
| St. 3 | 0 | 38.2 | 35.4 | 26.4 |
| 4 | 52.7 | 28.6 | 18.8 | |
| 16 | 61.1 | 21.9 | 17.0 | |
| 40 | 57.0 | 38.7 | 4.3 | |
| St. 4 | 0 | 64.1 | 19.3 | 16.6 |
| 5 | 75.5 | 10.8 | 13.7 | |
| 10 | 73.6 | 14.8 | 11.6 | |
| St. 5 | 0 | 78.8 | 6.5 | 14.7 |
| 6 | 82.1 | 7.3 | 10.6 | |
| 22 | 81.5 | 5.4 | 13.1 | |
| 80 | 57.8 | 9.8 | 32.4 | |
| St. 6 | 0 | 29.5 | 48.8 | 21.7 |
| 10 | 55.1 | 37.0 | 7.9 | |
| 20 | 71.5 | 9.9 | 18.6 | |
| 50 | 53.1 | 24.2 | 22.7 | |
| St. 7 | 0 | 80.1 | 7.8 | 12.1 |
| 7 | 68.1 | 9.5 | 22.4 | |
| 27 | 69.6 | 7.1 | 23.3 | |
| 80 | 52.7 | 11.2 | 36.0 | |
| St. 8 | 0 | 86.5 | 4.4 | 9.1 |
| 10 | 57.0 | 9.6 | 33.4 | |
| 40 | 47.3 | 10.3 | 42.3 | |
| 100 | 60.5 | 13.0 | 26.5 | |
| St. 9 | 0 | 81.4 | 4.2 | 14.5 |
| 7 | 61.1 | 9.5 | 29.4 | |
| 27 | 75.1 | 6.2 | 18.7 | |
| 50 | 74.4 | 7.5 | 18.1 | |
| 100 | 74.3 | 6.5 | 19.2 | |
| St. 10 | 0 | 66.6 | 11.2 | 22.2 |
| 5 | 68.6 | 10.5 | 20.9 | |
| 19 | 56.3 | 13.5 | 30.3 | |
| 50 | 53.8 | 13.3 | 32.9 | |
| 100 | 75.9 | 7.7 | 16.4 |
| Temperature | Phosphate | Nitrite + Nitrate | Silicate | Ammonium | Specific Carbon | Absolute Carbon | Specific Nitrate | Absolute Nitrate | Specific Ammonium | Absolute Ammonium | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature | 1 | ||||||||||
| Phosphate | 0.868 ** | 1 | |||||||||
| Nitrite + Nitrate | 0.797 ** | 0.974 ** | 1 | ||||||||
| Silicate | 0.795 ** | 0.706 ** | 0.598 * | 1 | |||||||
| Ammonium | −0.664 ** | −0.630 * | −0.503 | −0.651 ** | 1 | ||||||
| Specific Carbon | 0.200 | 0.140 | 0.140 | 0.353 | −0.130 | 1 | |||||
| Absolute Carbon | 0.165 | 0.101 | 0.102 | 0.326 | −0.109 | 0.998 ** | 1 | ||||
| Specific Nitrate | 0.232 | 0.116 | 0.161 | 0.248 | −0.187 | 0.567 * | 0.566 * | 1 | |||
| Absolute Nitrate | 0.234 | 0.069 | 0.102 | 0.272 | −0.209 | 0.525 * | 0.531 * | 0.979 ** | 1 | ||
| Specific Ammonium | −0.698 ** | −0.716 ** | −0.621 * | −0.469 | 0.795 ** | 0.183 | 0.207 | 0.026 | 0.026 | 1 | |
| Absolute Ammonium | −0.732 ** | −0.784 ** | −0.701 ** | −0.501 | 0.768 ** | 0.141 | 0.167 | 0.006 | 0.015 | 0.987 ** | 1 |
| Sampling Period | Daily Carbon Uptake Rate (g C m−2 day−1) | Reference | Remarks |
|---|---|---|---|
| July 1979 | 0.128 | Eilertsen et al. [21] | Daily mean production rate 76% of noon; calculated by Hop et al. [16] |
| July 1980 | 0.024 | ||
| July 1996 | 0.8–1.4 | Hop et al. [16] and references therein | Night production 32% of daytime production; calculated by Hop et al. [16] |
| 1–22 May 2002 | 0.466 | Hodal et al. [41] | Integrated to 30 m |
| 23–27 July 2002 | 0.088 | Piwosz et al. [28] | Integrated to 30 m, daily production was calculated based on 24 h |
| 18 March 2006 | 0.004 | Iversen and Seuthe [20] | Integrated to 50 m |
| 25 April 2006 | 0.405 | ||
| 30 May 2006 | 0.445 | ||
| 4 July 2006 | 0.155 | ||
| 16 September 2006 | 0.080 | ||
| Jun 2015 | 0.528 | van De Poll et al. [53] | Integrated to euphotic zone (defined as the depth interval down to 0.1% irradiance) |
| 4–8 May 2017 | 0.733 | This study | Integrated to 1% light depth |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, B.K.; Joo, H.M.; Jung, J.; Lee, B.; Ha, S.-Y. In Situ Rates of Carbon and Nitrogen Uptake by Phytoplankton and the Contribution of Picophytoplankton in Kongsfjorden, Svalbard. Water 2020, 12, 2903. https://doi.org/10.3390/w12102903
Kim BK, Joo HM, Jung J, Lee B, Ha S-Y. In Situ Rates of Carbon and Nitrogen Uptake by Phytoplankton and the Contribution of Picophytoplankton in Kongsfjorden, Svalbard. Water. 2020; 12(10):2903. https://doi.org/10.3390/w12102903
Chicago/Turabian StyleKim, Bo Kyung, Hyoung Min Joo, Jinyoung Jung, Boyeon Lee, and Sun-Yong Ha. 2020. "In Situ Rates of Carbon and Nitrogen Uptake by Phytoplankton and the Contribution of Picophytoplankton in Kongsfjorden, Svalbard" Water 12, no. 10: 2903. https://doi.org/10.3390/w12102903
APA StyleKim, B. K., Joo, H. M., Jung, J., Lee, B., & Ha, S.-Y. (2020). In Situ Rates of Carbon and Nitrogen Uptake by Phytoplankton and the Contribution of Picophytoplankton in Kongsfjorden, Svalbard. Water, 12(10), 2903. https://doi.org/10.3390/w12102903





