Preliminary Analysis of the Diet of Triturus carnifex and Pollution in Mountain Karst Ponds in Central Apennines
Abstract
1. Introduction
2. Materials and Methods
2.1. Target Species
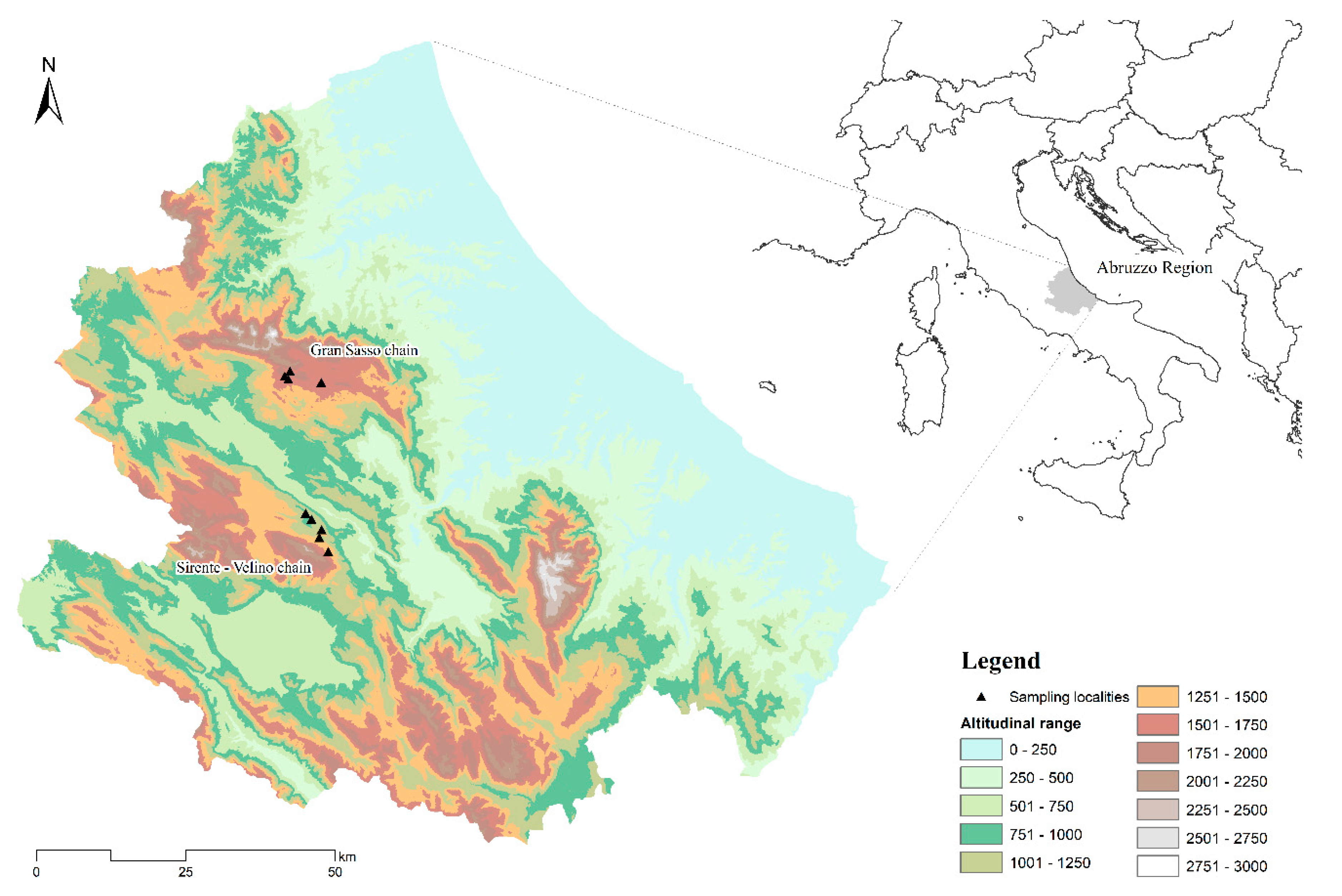
2.2. Study Area
2.3. Sampling Methods
2.4. Diet Analysis
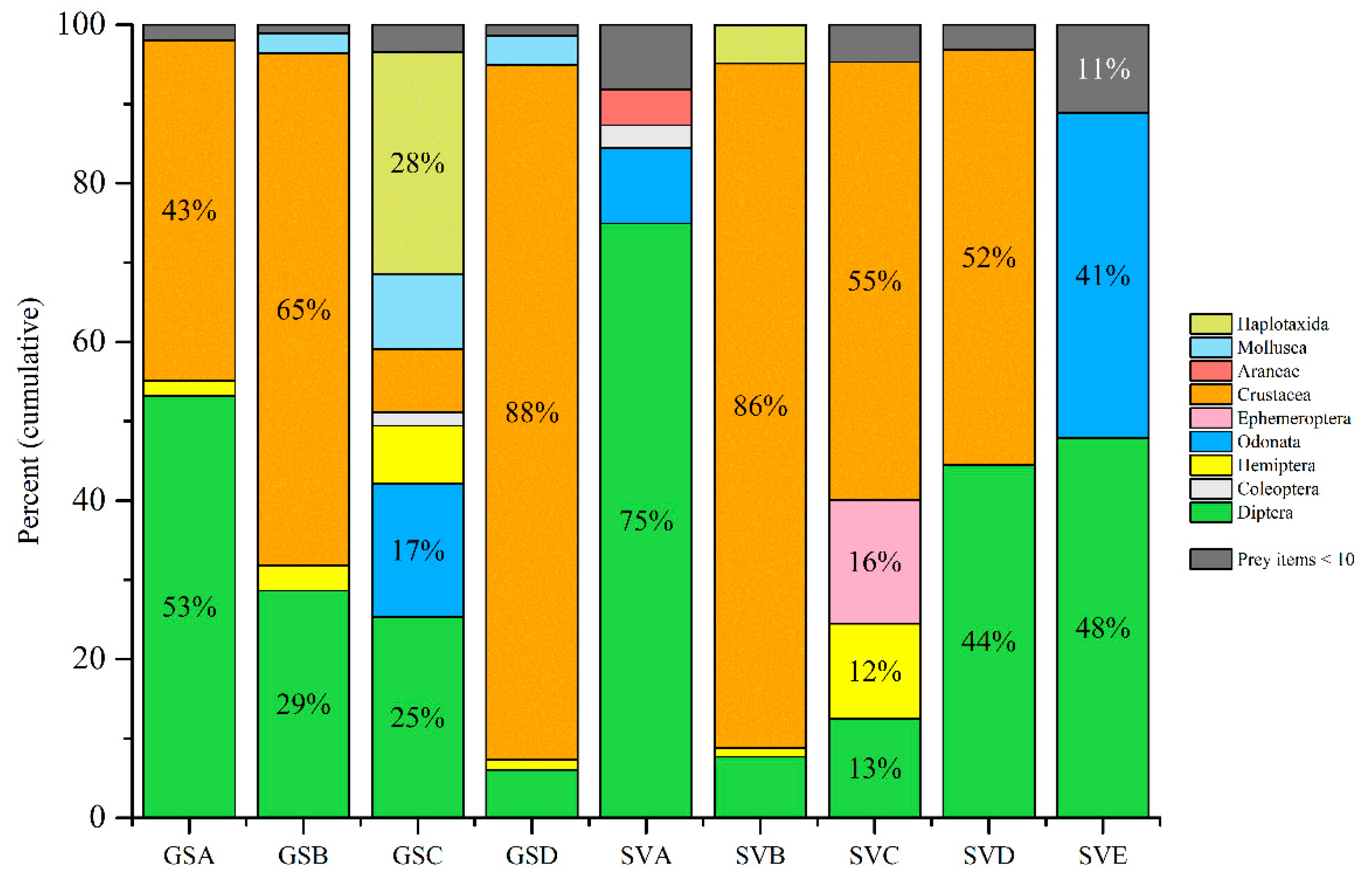
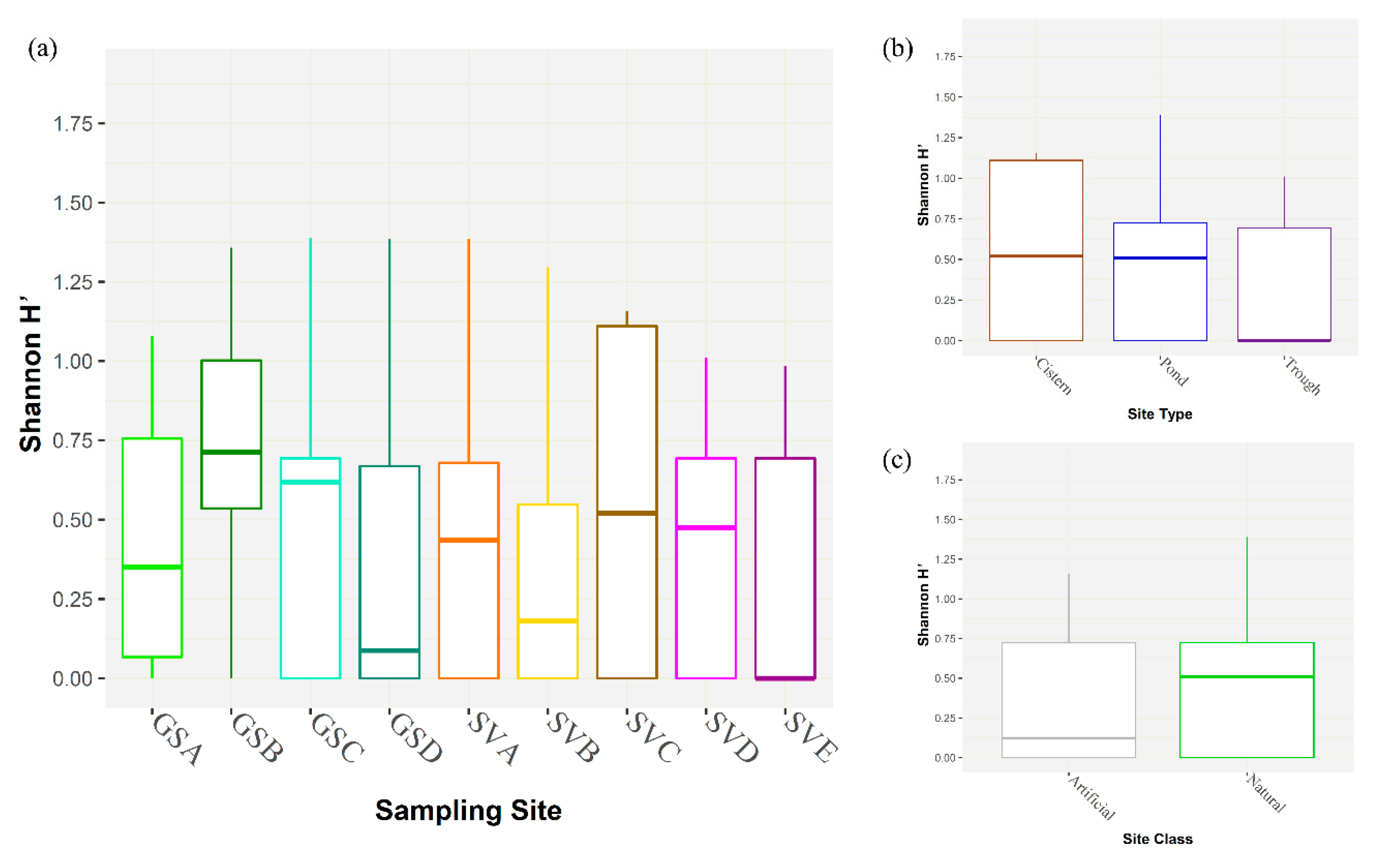
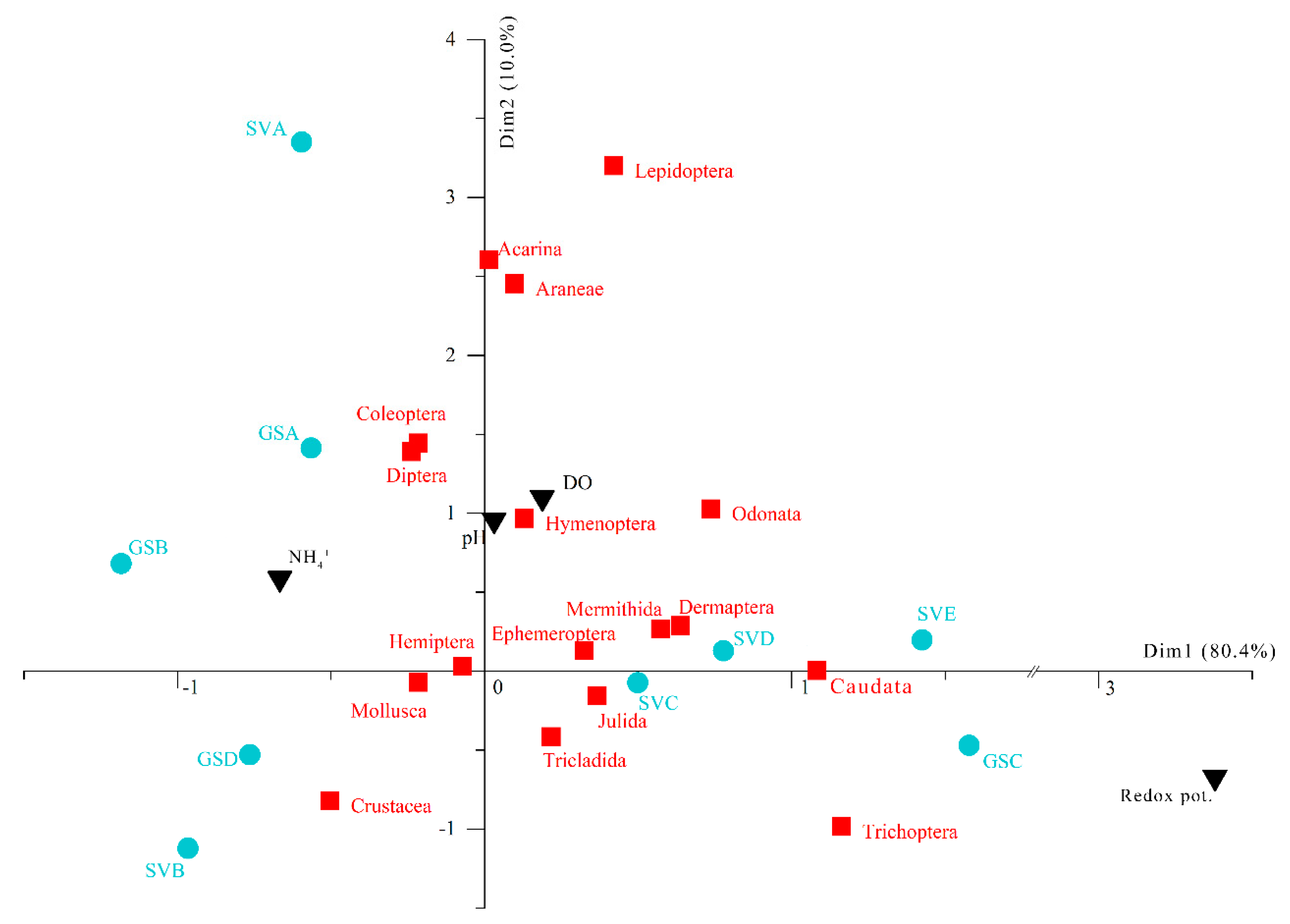
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rogora, M.; Frate, L.; Carranza, M.; Freppaz, M.; Stanisci, A.; Bertani, I.; Bottarin, R.; Brambilla, A.; Canullo, R.; Carbognani, M.; et al. Assessment of climate change effects on mountain ecosystems through a cross-site analysis in the Alps and Apennines. Sci. Total Environ. 2018, 624, 1429–1442. [Google Scholar] [CrossRef]
- Dirnböck, T.; Essl, F.; Rabitsch, W. Disproportional risk for habitat loss of high-altitude endemic species under climate change. Glob. Chang. Biol. 2011, 17, 990–996. [Google Scholar] [CrossRef]
- Brambilla, M.; Pedrini, P.; Rolando, A.; Chamberlain, D.E. Climate change will increase the potential conflict between skiing and high-elevation bird species in the Alps. J. Biogeogr. 2016, 43, 2299–2309. [Google Scholar] [CrossRef]
- Di Musciano, M.; Carranza, M.; Frate, L.; Di Cecco, V.; Di Martino, L.; Frattaroli, A.; Stanisci, A. Distribution of plant species and dispersal traits along environmental gradients in central Mediterranean summits. Diversity 2018, 10, 58. [Google Scholar] [CrossRef]
- Cerasoli, F.; Iannella, M.; Biondi, M. Between the hammer and the anvil: How the combined effect of global warming and the non-native common slider could threaten the European pond turtle. Manag. Biol. Invasions 2019, 10, 428. [Google Scholar] [CrossRef]
- Chamberlain, D.; Brambilla, M.; Caprio, E.; Pedrini, P.; Rolando, A. Alpine bird distributions along elevation gradients: The consistency of climate and habitat effects across geographic regions. Oecologia 2016, 181, 1139–1150. [Google Scholar] [CrossRef] [PubMed]
- Cerasoli, F.; Thuiller, W.; Guéguen, M.; Renaud, J.; D’Alessandro, P.; Biondi, M. The role of climate and biotic factors in shaping current distributions and potential future shifts of European Neocrepidodera (Coleoptera, Chrysomelidae). Insect Divers. Conserv. 2019. [Google Scholar] [CrossRef]
- Brunetti, M.; Magoga, G.; Iannella, M.; Biondi, M.; Montagna, M. Phylogeography and species distribution modelling of Cryptocephalus barii (Coleoptera: Chrysomelidae): Is this alpine endemic species close to extinction? ZooKeys 2019, 856, 3. [Google Scholar] [CrossRef] [PubMed]
- Wiens, J.J. Climate-Related local extinctions are already widespread among plant and animal species. PLoS Biol. 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Oertli, B.; Biggs, J.; Céréghino, R.; Grillas, P.; Joly, P.; Lachavanne, J.B. Conservation and monitoring of pond biodiversity: Introduction. Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 15, 535–540. [Google Scholar] [CrossRef]
- Niedrist, G.; Psenner, R.; Sommaruga, R. Climate warming increases vertical and seasonal water temperature differences and inter-annual variability in a mountain lake. Clim. Chang. 2018, 151, 473–490. [Google Scholar] [CrossRef]
- Sommaruga, R. The role of solar UV radiation in the ecology of alpine lakes. J. Photochem. Photobiol. B Biol. 2001, 62, 35–42. [Google Scholar] [CrossRef]
- Hinden, H.; Oertli, B.; Menetrey, N.; Sager, L.; Lachavanne, J.B. Alpine pond biodiversity: What are the related environmental variables? Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 15, 613–624. [Google Scholar] [CrossRef]
- Miaud, C.; Guyetant, R.; Faber, H. Age, size, and growth of the alpine newt, Triturus alpestris (Urodela: Salamandridae), at high altitude and a review of life-history trait variation throughout its range. Herpetologica 2000, 56, 135–144. [Google Scholar]
- Alonso, M.; Garcia-De-Lomas, J. Systematics and ecology of Linderiella baetica n. sp. (Crustacea, Branchiopoda, Anostraca, Chirocephalidae), a new species from southern Spain. Zoosystema 2009, 31, 807–828. [Google Scholar] [CrossRef]
- Mushet, D.M.; Euliss Jr, N.H.; Chen, Y.; Stockwell, C.A. Complex spatial dynamics maintain northern leopard frog (Lithobates pipiens) genetic diversity in a temporally varying landscape. Herpetol. Conserv. Biol. 2013, 8, 163–175. [Google Scholar]
- Lund, J.O.; Wissinger, S.A.; Peckarsky, B.L. Caddisfly behavioral responses to drying cues in temporary ponds: Implications for effects of climate change. Freshw. Sci. 2016, 35, 619–630. [Google Scholar] [CrossRef]
- Catorci, A.; Gatti, R.; Vitanzi, A. Relationship between phenology and above-ground phytomass in a grassland community in central Italy. In Nature Conservation. Environmental Science and Engineering (Environmental Science); Gafta, D., Akeroyd, J., Eds.; Springer: Heidelberg, Germany, 2006; pp. 309–327. [Google Scholar]
- Filibeck, G.; Cancellieri, L.; Sperandii, M.G.; Belonovskaya, E.; Sobolev, N.; Tsarevskaya, N.; Becker, T.; Berastegi, A.; Bückle, C.; Che, R. Biodiversity patterns of dry grasslands in the Central Apennines (Italy) along a precipitation gradient: Experiences from the 10th EDGG Field Workshop. Bull. Eurasian Dry Grassl. Group 2018, 36, 25–41. [Google Scholar]
- Primi, R.; Filibeck, G.; Amici, A.; Bückle, C.; Cancellieri, L.; Di Filippo, A.; Gentile, C.; Guglielmino, A.; Latini, R.; Mancini, L.D. From Landsat to leafhoppers: A multidisciplinary approach for sustainable stocking assessment and ecological monitoring in mountain grasslands. Agric. Ecosyst. Environ. 2016, 234, 118–133. [Google Scholar] [CrossRef]
- Ruggiero, A.; Solimini, A.G.; Carchini, G. Limnological aspects of an Apennine shallow lake. Ann. Limnol. Int. J. Limnol. 2004, 40, 89–99. [Google Scholar] [CrossRef]
- Solimini, A.G.; Bazzanti, M.; Ruggiero, A.; Carchini, G. Developing a multimetric index of ecological integrity based on macroinvertebrates of mountain ponds in central Italy. In Pond Conservation in Europe; Oertli, B., Céréghino, R., Biggs, J., Declerck, S., Hull, A., Miracle, M.R., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 109–123. [Google Scholar]
- Calhoun, A.J.; Mushet, D.M.; Bell, K.P.; Boix, D.; Fitzsimons, J.A.; Isselin-Nondedeu, F. Temporary wetlands: Challenges and solutions to conserving a ‘disappearing’ecosystem. Biol. Conserv. 2017, 211, 3–11. [Google Scholar] [CrossRef]
- Hunter, M.L., Jr.; Acuña, V.; Bauer, D.M.; Bell, K.P.; Calhoun, A.J.; Felipe-Lucia, M.R.; Fitzsimons, J.A.; González, E.; Kinnison, M.; Lindenmayer, D. Conserving small natural features with large ecological roles: A synthetic overview. Biol. Conserv. 2017, 211, 88–95. [Google Scholar] [CrossRef]
- Ruggiero, A.; Solimini, A.; Carchini, G. The alternative stable state concept and the management of Apennine mountain ponds. Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 15, 625–634. [Google Scholar] [CrossRef]
- Solimini, A.G.; Ruggiero, A.; Anello, M.; Mutschlechner, A.; Carchini, G. The benthic community structure in mountain ponds affected by livestock watering in nature reserves of Central Italy. Int. Ver. Theor. Angew. Limnol. Verh. 2000, 27, 501–505. [Google Scholar] [CrossRef]
- Ruggiero, A.; Solimini, A.; Carchini, G. Pratica dell’alpeggio e conservazione ambientale negli stagni montani dell’Appennino Centrale. In Proceedings of the Atti del Convegno Nazionale Conservazione Dell’ambiente e Rischio Idrogeologico, Perugia, Italy, 11–12 December 2002; pp. 687–694. [Google Scholar]
- Wissinger, S.A.; Oertli, B.; Rosset, V. Invertebrate communities of alpine ponds. In Invertebrates in Freshwater Wetlands; Batzer, D., Boix, D., Eds.; Springer: Cham, Switzerland, 2016; pp. 55–103. [Google Scholar]
- Howell, H.J.; Mothes, C.C.; Clements, S.L.; Catania, S.V.; Rothermel, B.B.; Searcy, C.A. Amphibian responses to livestock use of wetlands: New empirical data and a global review. Ecol. Appl. 2019. [Google Scholar] [CrossRef]
- Iannella, M.; Cerasoli, F.; Biondi, M. Unraveling climate influences on the distribution of the parapatric newts Lissotriton vulgaris meridionalis and L. italicus. Front. Zool. 2017, 14, 55. [Google Scholar] [CrossRef]
- Iannella, M.; D’Alessandro, P.; Biondi, M. Evidences for a shared history for spectacled salamanders, haplotypes and climate. Sci. Rep. 2018, 8, 16507. [Google Scholar] [CrossRef]
- Hayes, T.B.; Stuart, A.A.; Mendoza, M.; Collins, A.; Noriega, N.; Vonk, A.; Johnston, G.; Liu, R.; Kpodzo, D. Characterization of atrazine-induced gonadal malformations in African clawed frogs (Xenopus laevis) and comparisons with effects of an androgen antagonist (cyproterone acetate) and exogenous estrogen (17β-estradiol): Support for the demasculinization/feminization hypothesis. Environ. Health Perspect 2006, 114, 134–141. [Google Scholar]
- Buck, J.C.; Scheessele, E.A.; Relyea, R.A.; Blaustein, A.R. The effects of multiple stressors on wetland communities: Pesticides, pathogens and competing amphibians. Freshw. Biol. 2012, 57, 61–73. [Google Scholar] [CrossRef]
- Patar, A.; Giri, A.; Boro, F.; Bhuyan, K.; Singha, U.; Giri, S. Cadmium pollution and amphibians–Studies in tadpoles of Rana limnocharis. Chemosphere 2016, 144, 1043–1049. [Google Scholar] [CrossRef]
- Ferreira, M.; Beja, P. Mediterranean amphibians and the loss of temporary ponds: Are there alternative breeding habitats? Biol. Conserv. 2013, 165, 179–186. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Rondinini, C.; Bonardi, A.; Baisero, D.; Padoa-Schioppa, E. Habitat availability for amphibians and extinction threat: A global analysis. Divers. Distrib. 2015, 21, 302–311. [Google Scholar] [CrossRef]
- Stuart, S.N.; Chanson, J.S.; Cox, N.A.; Young, B.E.; Rodrigues, A.S.; Fischman, D.L.; Waller, R.W. Status and trends of amphibian declines and extinctions worldwide. Science 2004, 306, 1783–1786. [Google Scholar] [CrossRef] [PubMed]
- May, R.M. Ecological science and tomorrow’s world. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 41–47. [Google Scholar] [CrossRef][Green Version]
- Best, M.L.; Welsh, J.; Hartwell, H. The trophic role of a forest salamander: Impacts on invertebrates, leaf litter retention, and the humification process. Ecosphere 2014, 5, 1–19. [Google Scholar] [CrossRef]
- Davic, R.D.; Welsh, H.H. On the ecological roles of salamanders. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 405–434. [Google Scholar] [CrossRef]
- Salvidio, S.; Costa, A.; Crovetto, F. Individual trophic specialization in the Alpine newt increases with increasing resource diversity. Ann. Zool. Fenn. 2019, 56, 17–24. [Google Scholar] [CrossRef]
- Vignoli, L.; Bombi, P.; D’Amen, M.; Bologna, M.A. Seasonal variation in the trophic niche of a heterochronic population of Triturus alpestris apuanus from the south-western Alps. Herpetol. J. 2007, 17, 183–191. [Google Scholar]
- Schabetsberger, R.; Jehle, R.; Maletzky, A.; Pesta, J.; Sztatecsny, M. Delineation of terrestrial reserves for amphibians: Post-breeding migrations of Italian crested newts (Triturus c. carnifex) at high altitude. Biol. Conserv. 2004, 117, 95–104. [Google Scholar] [CrossRef]
- Vanni, S.; Andreone, F.; Tripepi, S. Istituto Superiore per La Protezione e La Ricerca Ambientale; Anfibi d’Italia. Lanza, B., Nistri, A., Vanni, S., Eds.; Ministero dell’Ambiente e della Tutela del Territorio e del Mare: Rome, Italy, 2007; Volume 29.
- Naviglio, L. Aspetti naturalistici del Lago Pantaniello. Nat. Montagna. 1984, 31, 49–57. [Google Scholar]
- Ferri, V.; Di Tizio, L.; Pellegrini, M. Atlante Degli Anfibi d’Abruzzo; Ianieri–Talea: Pescara, Italy, 2007; p. 199. [Google Scholar]
- Andreone, F.; Corti, C.; Ficetola, F.; Razzetti, E.; Romano, A.; Sindaco, R. Triturus carnifex. The IUCN Red List of Threatened Species. 2009: e.T59474A11947714. Available online: https://www.iucnredlist.org/species/59474/11947714 (accessed on 5 December 2018).
- Beebee, T.J.C.; Griffiths, R.A. The amphibian decline crisis: A watershed for conservation biology? Biol. Conserv. 2005, 125, 271–285. [Google Scholar] [CrossRef]
- D’Amen, M.; Bombi, P. Global warming and biodiversity: Evidence of climate-linked amphibian declines in Italy. Biol. Conserv. 2009, 142, 3060–3067. [Google Scholar] [CrossRef]
- Collins, J.P.; Crump, M.L.; Lovejoy Iii, T.E. Extinction in Our Times: Global Amphibian Decline; Oxford University Press: New York, NY, USA, 2009. [Google Scholar]
- Solé, M.; Rödder, D. Dietary assessments of adult amphibians. In Amphibian Ecology and Conservation: A Handbook of Techniques; Oxford University Press: New York, NY, USA, 2010; pp. 167–184. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.; Simpson, G.L.; Solymos, P. 2019. Vegan: Community Ecology Package. R package version 2.5-4. Available online: https://cran.r-project.org/web/packages/vegan (accessed on 1 September 2019).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: http://www. R-project. org (accessed on 15 June 2017).
- Nenadic, O.; Greenacre, M. Correspondence Analysis in R, with two-and three-dimensional graphics: The ca package. J. Stat. Softw. 2007, 20, 1–13. [Google Scholar]
- Costa, A.; Salvidio, S.; Posillico, M.; Matteucci, G.; De Cinti, B.; Romano, A. Generalisation within specialization: Inter-individual diet variation in the only specialized salamander in the world. Sci. Rep. 2015, 5, 13260. [Google Scholar] [CrossRef]
- Vignoli, L.; Bologna, M.A. Dietary patterns and overlap in an amphibian assemblage at a pond in Mediterranean central Italy. Vie Milieu-Life Environ. 2009, 59, 47–57. [Google Scholar]
- Fasola, M.; Canova, L. Feeding habits of Triturus vulgaris, T. cristatus and T. alpestris (Amphibia, Urodela) in the northern Apennines (Italy). Ital. J. Zool. 1992, 59, 273–280. [Google Scholar]
- Regio Decreto Legislativo. Riordinamento E Riforma Della Legislazione in Materia di Boschi E di Terreni Montani; Italy, Ed.; NORMATTIVA: Rome, Italy, 1923. [Google Scholar]
- Buono, V.; Bissattini, A.M.; Vignoli, L. Can a cow save a newt? The role of cattle drinking troughs in amphibian conservation. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 29, 964–975. [Google Scholar] [CrossRef]
- Schmutzer, A.C.; Gray, M.J.; Burton, E.C.; Miller, D.L. Impacts of cattle on amphibian larvae and the aquatic environment. Freshw. Biol. 2008, 53, 2613–2625. [Google Scholar] [CrossRef]
- Hoverman, J.T.; Gray, M.J.; Miller, D.L.; Haislip, N.A. Widespread occurrence of ranavirus in pond-breeding amphibian populations. Eco. Health 2012, 9, 36–48. [Google Scholar] [CrossRef]
- Badillo-Saldaña, L.M.; Ramírez-Bautista, A.; Wilson, L.D. Effects of establishment of grazing areas on diversity of amphibian communities in tropical evergreen forests and mountain cloud forests of the Sierra Madre Oriental. Rev. Mex. Biodivers. 2016, 87, 133–139. [Google Scholar] [CrossRef]
- Moreira, L.F.; Moura, R.G.; Maltchik, L. Stop and ask for directions: Factors affecting anuran detection and occupancy in Pampa farmland ponds. Ecol. Res. 2016, 31, 65–74. [Google Scholar] [CrossRef]
- Howard, A.; Munger, J.C. Effects of Livestock Grazing on the Invertebrate Prey Base and on the Survival and Growth of Larvae of the Columbia Spotted Frog, Rana Luteiventris; Bureau of Land Management, Idaho State Office: Boise, ID, USA, 2003.
- Zelnik, I.; Gregorič, N.; Tratnik, A. Diversity of macroinvertebrates positively correlates with diversity of macrophytes in karst ponds. Ecol. Eng. 2018, 117, 96–103. [Google Scholar] [CrossRef]
- Byrne, P.G.; Silla, A.J. Testing the effect of dietary carotenoids on larval survival, growth and development in the critically endangered southern corroboree frog. Zoo. Biol. 2017, 36, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Clugston, R.D.; Blaner, W.S. Vitamin A (retinoid) metabolism and actions: What we know and what we need to know about amphibians. Zoo. Biol. 2014, 33, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Szuroczki, D.; Koprivnikar, J.; Baker, R.L. Dietary antioxidants enhance immunocompetence in larval amphibians. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2016, 201, 182–188. [Google Scholar] [CrossRef]
- Arribas, R.; Díaz-Paniagua, C.; Caut, S.; Gomez-Mestre, I. Stable isotopes reveal trophic partitioning and trophic plasticity of a larval amphibian guild. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the marine environment: A review of the methods used for identification and quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef]
- Döring, B.; Mecke, S.; Kieckbusch, M.; O’Shea, M.; Kaiser, H. Food spectrum analysis of the Asian toad, Duttaphrynus melanostictus (Schneider, 1799) (Anura: Bufonidae), from Timor Island, Wallacea. J. Nat. Hist. 2017, 51, 607–623. [Google Scholar] [CrossRef]
- Hu, L.; Chernick, M.; Hinton, D.E.; Shi, H. Microplastics in small waterbodies and tadpoles from Yangtze River Delta, China. Environ. Sci. Technol. 2018, 52, 8885–8893. [Google Scholar] [CrossRef]
- De Felice, B.; Bacchetta, R.; Santo, N.; Tremolada, P.; Parolini, M. Polystyrene microplastics did not affect body growth and swimming activity in Xenopus laevis tadpoles. Environ. Sci. Pollut. Res. 2018, 25, 34644–34651. [Google Scholar] [CrossRef] [PubMed]
- Da Costa Araújo, A.P.; de Melo, N.F.S.; de Oliveira Junior, A.G.; Rodrigues, F.P.; Fernandes, T.; de Andrade Vieira, J.E.; Rocha, T.L.; Malafaia, G. How much are microplastics harmful to the health of amphibians? A study with pristine polyethylene microplastics and Physalaemus cuvieri. J. Hazard. Mater. 2020, 382, 121066. [Google Scholar] [CrossRef] [PubMed]

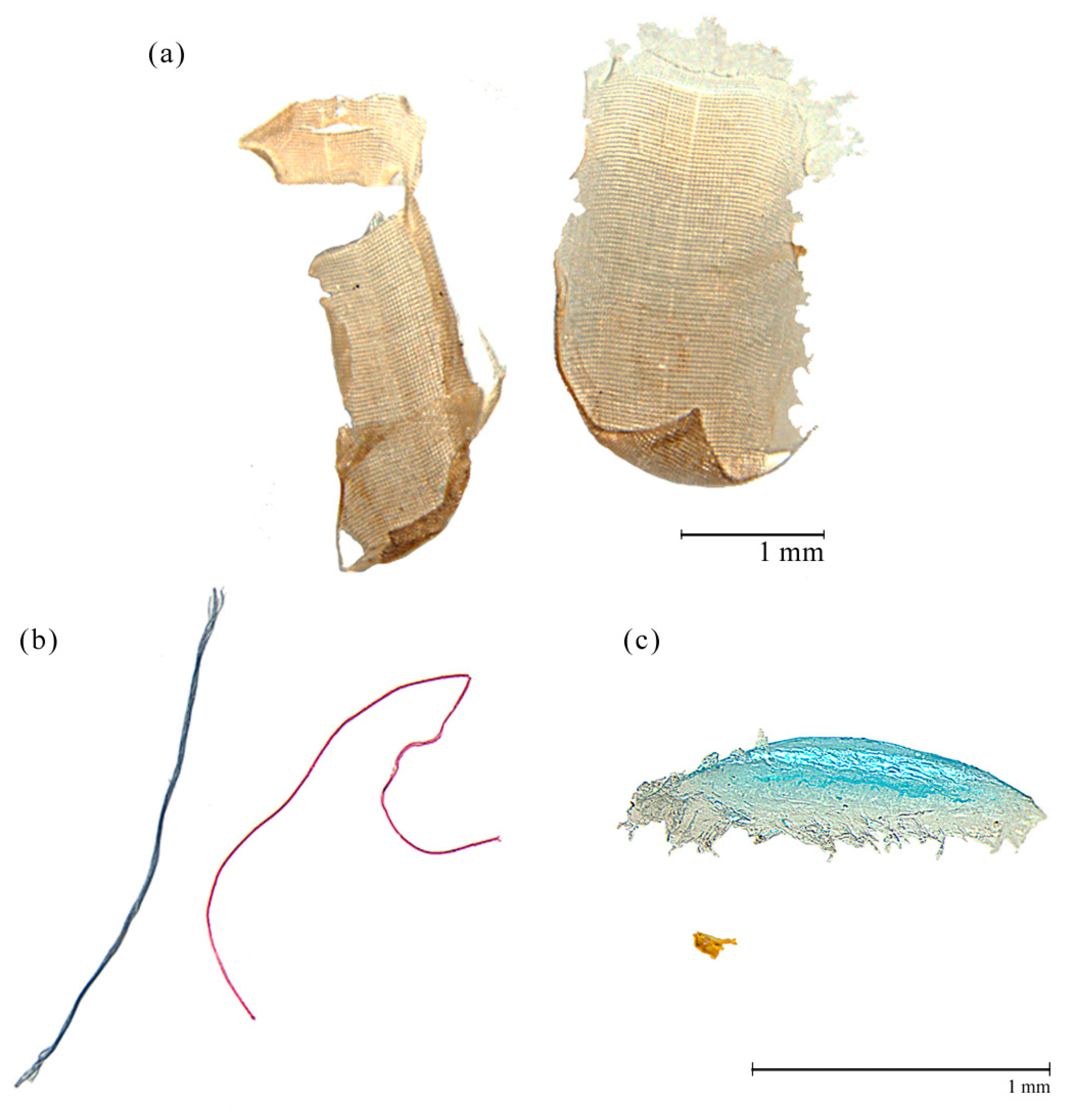
| Name | Code | Type | Area (m2) | Max. Depth (m) | Latitude | Longitude | Elevation (m a.s.l.) |
|---|---|---|---|---|---|---|---|
| Fossetta A | GSA | Pond | 1870 | 1.2 | 42°24′17″ N | 13°35′14″ E | 1658 |
| Fossetta B | GSB | Pond | 358 | 1.2 | 42°24′19″ N | 13°35′13″ E | 1658 |
| Fossa di Paganica | GSC | Pond | 4500 | 1.5 | 42°24′55″ N | 13°39′27″ E | 1680 |
| Racollo | GSD | Pond | 7000 | 0.8 | 42°23′37″ N | 13°39′27″ E | 1570 |
| Tempra | SVA | Pond | 870 | 1.5 | 42°10′22″ N | 13°38′30″ E | 1183 |
| Piane di Iano | SVB | Pond | 780 | 1.2 | 42°11′21″ N | 13°37′22″ E | 960 |
| Pagliare di Tione | SVC | Cistern | 125 | 2.5 | 42°11′55″ N | 13°36′41″ E | 1080 |
| Piano Canale | SVD | Trough | 15 | 0.5 | 42°08′25″ N | 13°39′17″ E | 1342 |
| Fonte dell′Acqua | SVE | Trough | 26 | 0.5 | 42°08′21″ N | 13°39′10″ E | 1167 |
| Name | Code | pH | Redox Potential (mV) | DO (mg/L) (DO (%sat), Temp. (°C)) | NH4+ (mg/L) |
|---|---|---|---|---|---|
| Fossetta A | GSA | 7.3 | −6.9 | 3.0 (38%, 20.8 °C) | 0.12 |
| Fossetta B | GSB | 7.6 | −17.7 | 3.6 (47%, 21.1 °C) | 0.17 |
| Fossa di Paganica | GSC | 8.3 | 38.1 | 4.5 (62%, 24.0 °C) | 0.00 |
| Racollo | GSD | 7.6 | −7.9 | 2.3 (32%, 23.2 °C) | 0.03 |
| Tempra | SVA | 9.5 | −5.1 | 7.0 (90%, 24.2 °C) | 0.06 |
| Piane di Iano | SVB | 7.5 | −28.7 | 3.7 (45%, 17.9 °C) | 0.26 |
| Pagliare di Tione | SVC | 8.5 | 16.2 | 7.2 (87%, 21.4 °C) | 0.00 |
| Piano Canale | SVD | 8.1 | 27.2 | 7.8 (96%, 20.3 °C) | 0.00 |
| Fonte dell’Acqua | SVE | 7.9 | 24.7 | 7.4 (93%, 23.4 °C) | 0.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iannella, M.; Console, G.; D’Alessandro, P.; Cerasoli, F.; Mantoni, C.; Ruggieri, F.; Di Donato, F.; Biondi, M. Preliminary Analysis of the Diet of Triturus carnifex and Pollution in Mountain Karst Ponds in Central Apennines. Water 2020, 12, 44. https://doi.org/10.3390/w12010044
Iannella M, Console G, D’Alessandro P, Cerasoli F, Mantoni C, Ruggieri F, Di Donato F, Biondi M. Preliminary Analysis of the Diet of Triturus carnifex and Pollution in Mountain Karst Ponds in Central Apennines. Water. 2020; 12(1):44. https://doi.org/10.3390/w12010044
Chicago/Turabian StyleIannella, Mattia, Giulia Console, Paola D’Alessandro, Francesco Cerasoli, Cristina Mantoni, Fabrizio Ruggieri, Francesca Di Donato, and Maurizio Biondi. 2020. "Preliminary Analysis of the Diet of Triturus carnifex and Pollution in Mountain Karst Ponds in Central Apennines" Water 12, no. 1: 44. https://doi.org/10.3390/w12010044
APA StyleIannella, M., Console, G., D’Alessandro, P., Cerasoli, F., Mantoni, C., Ruggieri, F., Di Donato, F., & Biondi, M. (2020). Preliminary Analysis of the Diet of Triturus carnifex and Pollution in Mountain Karst Ponds in Central Apennines. Water, 12(1), 44. https://doi.org/10.3390/w12010044







