Characteristics, Main Impacts, and Stewardship of Natural and Artificial Freshwater Environments: Consequences for Biodiversity Conservation
Abstract
1. Introduction
2. Ground Water and Groundwater-Dependent Ecosystems (GDEs)
2.1. Ground Water and Groundwater-Dependent Ecosystems
2.2. Springs
3. Running Waters
3.1. Headwaters
3.1.1. Spring-Fed Streams (Crenal)
3.1.2. Glacial Streams (Kryal)
3.2. Streams (Rhithral)
3.3. Large Rivers (Potamal)
4. Lakes
4.1. Ancient and Large Lakes
4.2. High-Mountain Lakes
4.3. Oxbow Lakes
5. Man-Made Freshwater Habitats
5.1. Reservoirs
5.2. Urban (Artificial) Freshwater Habitats
6. Mires (Peatlands): Fens and Bogs
7. Small Standing-Water Ecosystems
8. Cold-Climate Freshwater Habitats (Boreal, Arctic, Antarctic)
8.1. Boreal and Arctic Freshwater Habitats
8.2. Antarctic Freshwater Habitats
9. Mediterranean Freshwater Habitats
9.1. Streams and Rivers
9.2. Ponds
9.3. Near-Natural Lakes (L. Kinneret)
9.4. Reservoirs
10. Tropical Freshwater Habitats
11. Arid-Climate Freshwater Habitats
12. Freshwater Biodiversity Observation Network (FWBON)
13. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sala, O.E.; Stuart Chapin, F., III; Armesto, J.J.; Berlow, E.; Bloomfield, J.; Dirzo, R.; Huber-Sanwald, E.; Huenneke, L.F.; Jackson, R.B.; Kinzig, A.; et al. Global Biodiversity Scenarios for the Year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.H.; Soto, D.; Stiassny, M.L.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef]
- Reid, A.J.; Carlson, A.K.; Creed, I.F.; Eliason, E.J.; Gell, P.A.; Johnson, P.T.J.; Kidd, K.A.; MacCormack, T.J.; Olden, J.D.; Ormerod, S.J.; et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol. Rev. 2019, 94, 849–873. [Google Scholar] [CrossRef]
- Parker, B.R.; Vinebrooke, R.D.; Schindler, D.W. Recent climate extremes alter alpine lake ecosystems. Proc. Natl. Acad. Sci. USA 2008, 105, 12927–12931. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, I.E.; Gleeson, T.; van Beek, L.P.H.; Sutanudjaja, E.H.; Bierkens, M.F.P. Environmental flow limits toglobal groundwater pumping. Nature 2019, 574, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Balian, E.V.; Segers, H.; Lévèque, C.; Martens, K. The Freshwater Animal Diversity Assessment: An overview of the results. Hydrobiologia 2008, 595, 627–637. [Google Scholar] [CrossRef]
- Komárek, J. A polyphasic approach for the taxonomy of cyanobacteria: Principles and applications. Eur. J. Phycol. 2016, 51, 346–353. [Google Scholar] [CrossRef]
- Frassanito, R.; Cantonati, M.; Tardìo, M.; Mancini, I.; Guella, G. On-line identification of secondary metabolites in freshwater microalgae and cyanobacteria by combined liquid chromatography-photodiode array detection-mass spectrometric techniques. J. Chromatogr. A 2005, 1082, 33–42. [Google Scholar] [CrossRef]
- Boon, P.J.; Pringle, C.M. Assessing the Conservation Value of Fresh Waters; Cambridge University Press: Cambridge, UK, 2009; ISBN 9780521848855. [Google Scholar]
- International Union for Conservation of Nature. IUCN Red List of Ecosystems Guidebook: Categories, Criteria and How to Apply Them. Version 1; Ecosystems Red List Thematic Group, Commission on EcosystemManagement (CEM) and Global Ecosystem Management Programme (GEMP); International Union for Conservation of Nature (IUCN): Gland, Switzerland, 2016; ISBN 978-2-8317-1787-6. [Google Scholar]
- International Union for Conservation of Nature. IUCN Red List Categories and Criteria: Version 3.1; Species Survival Commission, World Conservation Union; International Union for Conservation of Nature (IUCN): Gland, Switzerland, 2001; ISBN 2-8317-0633-5. [Google Scholar]
- Jenkins, M. Prospects for Biodiversity. Science 2003, 302, 1175–1177. [Google Scholar] [CrossRef] [PubMed]
- Heilpern, S. Biodiversity: Include freshwater species. Nature 2015, 518, 167. [Google Scholar] [CrossRef]
- Darwall, W.; Bremerich, V.; De Wever, A.; Dell, A.I.; Freyhof, J.; Gessner, M.O.; Grossart, H.P.; Harrison, I.; Irvine, K.; Jänig, S.C.; et al. The Alliance for Freshwater Life: A global call to unite efforts forfreshwater biodiversity science and conservation. Aquat. Conserv. Mar. Freshw. Ecosyst. 2018, 28, 1015–1022. [Google Scholar] [CrossRef]
- Junk, W.J.; An, S.; Finlayson, C.M.; Gopal, B.; Kveˇt, J.; Mitchell, S.A.; Mitsch, W.J.; Robarts, R.D. Current state of knowledge regarding the world’s wetlands and their future under global climate change: A synthesis. Aquat. Sci. 2013, 75, 151–167. [Google Scholar] [CrossRef]
- Davidson, N.C.; Finlayson, C.M. Extent, regional distribution and changes in area of different classes of wetland. Mar. Freshw. Res. 2018, 69, 1525–1533. [Google Scholar] [CrossRef]
- Davidson, N.C. How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar. Freshw. Res. 2014, 65, 936–941. [Google Scholar] [CrossRef]
- Keddy, P.A.; Fraser, L.H.; Solomeshch, A.I.; Junk, W.J.; Campbell, D.R.; Arroyo, M.T.K.; Alho, C.J.R. Wet and Wonderful: The World’s Largest Wetlands Are Conservation Priorities. BioScience 2009, 59, 39–51. [Google Scholar] [CrossRef]
- Wright, S.J. Plant diversity in tropical forests: A review of mechanisms of species coexistence. Oecologia 2002, 130, 1–14. [Google Scholar] [CrossRef]
- Brown, J.H. Why are there so many species in the tropics? J. Biogeogr. 2014, 41, 8–22. [Google Scholar] [CrossRef]
- Fine, P.V.A. Ecological and evolutionary drivers of geographic variation in species diversity. Annu. Rev. Ecol. Syst. 2015, 46, 369–392. [Google Scholar] [CrossRef]
- Heino, J. A macroecological perspective of diversity patterns in the freshwater realm. Freshw. Biol. 2011, 56, 1703–1722. [Google Scholar] [CrossRef]
- Revenga, C.; Kura, Y. Status and Trends of Biodiversity of Inland Water Ecosystems; Technical Series no. 11; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2003; ISBN 92-807-2398-7. [Google Scholar]
- Dudgeon, D. (Ed.) Tropical Stream Ecology; Academic Press: London, UK, 2008; ISBN 978-0-12-088449-0. [Google Scholar]
- Collen, B.; Whitton, F.; Dyer, E.E.; Baillie, J.E.M.; Cumberlidge, N.; Darwall, W.R.T.; Pollock, C.; Richman, N.I.; Soulsby, M.; Böhm, M. Global patterns of freshwater species diversity, threat and endemism. Glob. Ecol. Biogeogr. 2014, 23, 40–51. [Google Scholar] [CrossRef]
- Albert, J.S.; Petry, P.; Reis, R.E. Major Biogeographic and Phylogenetic Patterns. In Historical Biogeography of Neotropical Freshwater Fishes; Albert, J.S., Reis, R.E., Eds.; University of California Press: Berkeley, CA, USA, 2011; pp. 21–57. [Google Scholar] [CrossRef]
- Junk, W.J.; Soares, M.G.M.; Bayley, P.B. Freshwater fishes of the Amazon River basin: Their biodiversity, fisheries, and habitats. Aquat. Ecosyst. Health Manag. 2007, 10, 153–173. [Google Scholar] [CrossRef]
- Albert, J.S.; Carvalho, T.P. Neogene Assembly of Modern Faunas. In Historical Biogeography of Neotropical Freshwater Fishes; Albert, J.S., Reis, R.E., Eds.; University of California Press: Berkeley, CA, USA, 2011; pp. 119–136. [Google Scholar] [CrossRef]
- Albert, J.S.; Carvalho, T.P.; Petry, P.; Holder, M.A.; Maxime, E.L.; Espino, J.; Corahua, I.; Quispe, R.; Rengifo, B.; Ortega, H.; et al. Aquatic Biodiversity in the Amazon: Habitat Specialization and Geographic Isolation Promote Species Richness. Animals 2011, 1, 205–241. [Google Scholar] [CrossRef] [PubMed]
- Morse, J.C. Biodiversity of aquatic insects. In Insect Biodiversity: Science and Society; Foottit, R.G., Adler, P.H., Eds.; Wiley-Blackwell: Chichester, UK, 2009; pp. 165–184. ISBN 978-1-118-94557-5. [Google Scholar]
- Crow, G.E. Species diversity in aquatic angiosperms: Latitudinal patterns. Aquat. Bot. 1993, 44, 229–258. [Google Scholar] [CrossRef]
- Chambers, P.A.; Lacoul, P.; Murphy, K.J.; Thomaz, S.M. Global diversity of aquatic macrophytes in freshwater. Hydrobiologia 2007, 595, 9–26. [Google Scholar] [CrossRef]
- Luize, B.G.; Magalhães, J.L.L.; Queiroz, H.; Lopes, M.A.; Venticinque, E.M.; Leão de Moraes Novo, E.M.; Silva, T.S.F. The tree species pool of Amazonian wetland forests: Which species can assemble in periodically waterlogged habitats? PLoS ONE 2018, 13, e0198130. [Google Scholar] [CrossRef]
- Hillebrand, H.; Azovsky, A.I. Body size determines the strength of the latitudinal diversity gradient. Ecography 2001, 24, 251–256. [Google Scholar] [CrossRef]
- Passy, S.I. A distinct latitudinal gradient of diatom diversity is linked to resource supply. Ecology 2010, 91, 36–41. [Google Scholar] [CrossRef]
- Soininen, J.; Jamoneau, A.; Rosebery, J.; Passy, S.I. Global patterns and drivers of species and trait composition in diatoms. Glob. Ecol. Biogeogr. 2016, 25, 940–950. [Google Scholar] [CrossRef]
- Jyrkänkallio-Mikkola, J.; Siljander, M.; Heikinheimo, V.; Pellikka, P.; Soininen, J. Tropical stream diatom communities—The importance of headwater streams for regional diversity. Ecol. Indic. 2018, 95, 183–193. [Google Scholar] [CrossRef]
- Vyverman, W.; Verleyen, E.; Sabbe, K.; Vanhoutte, K.; Sterken, M.; Hodgson, D.A.; Mann, D.G.; Juggins, S.; Vijver, B.V.; Jones, V.; et al. Historical processes constrain patterns in global diatom diversity. Ecology 2007, 88, 1924–1931. [Google Scholar] [CrossRef]
- Benito, X.; Fritz, S.C.; Steinitz-Kannan, M.; Tapia, P.M.; Kelly, M.A.; Lowell, T.V. Geo-climatic factors drive diatom community distribution in tropical South American freshwaters. J. Ecol. 2018, 106, 1660–1672. [Google Scholar] [CrossRef]
- Taxböck, L.; Linder, H.P.; Cantonati, M. To what extent are Swiss springs refugial habitats for sensitive and endangered diatom taxa? Water 2017, 9, 967. [Google Scholar] [CrossRef]
- Zelnik, I.; Balanč, T.; Toman, M.J. Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors. Water 2018, 10, 361. [Google Scholar] [CrossRef]
- Lai, G.G.; Burato, S.; Padedda, B.M.; Zorza, R.; Pizzul, E.; Delgado, C.; Lugliè, A.; Cantonati, M. Diatom biodiversity in karst springs of Mediterranean geographic areas with contrasting characteristics: Islands vs mainland. Water 2019, 11, 2602. [Google Scholar] [CrossRef]
- Rossini, R.; Fensham, R.; Walter, G. Species-specific environmental matching and patch geometry affect occupancy and persistence of threatened snails in Australian desert springs. Water. under review.
- Stevens, L.; Jenness, J.; Ledbetter, J.D. Geography, Ecology, and Human Impacts on Springs and Springs-dependent Species in the Colorado River Basin, Southwestern North America. Water 2020, 12. in press. [Google Scholar]
- Taxböck, L.; Karger, D.N.; Kessler, M.; Spitale, D.; Cantonati, M. Diatom species richness increases with elevation and habitat complexity in Swiss near-natural springs along elevational gradients. Water 2020, 12. in press. [Google Scholar]
- Richardson, J.S. Biological Diversity in Headwater Streams. Water 2019, 11, 366. [Google Scholar] [CrossRef]
- Segadelli, S.; Grazzini, F.; Adorni, M.; De Nardo, M.T.; Fornasiero, A.; Chelli, A.; Cantonati, M. Towards predicting and understanding extreme-precipitation effects on freshwater biodiversity and ecosystems in small catchments: A case study from the northern Apennines (Italy). Water 2019, 12, 79. [Google Scholar] [CrossRef]
- Füreder, L.; Niedrist, G.H. Glacial stream ecology: Structural and functional assets. Water 2020, 12. in press. [Google Scholar]
- Turak, E.; Bush, A.; DelaCruz, J.; Powell, M. Freshwater Reptile Persistence and Conservation in Cities: Insights From Species Occurrence Records. Water 2020, 12. in press. [Google Scholar]
- Marazzi, L.; Gaiser, E.; Eppinga, M.; Zhai, L.; Sah, J.; Castañeda-Moya, E.; Angelini, C. Why Do We Need to Document and Conserve Foundation Species in Freshwater Wetlands? Water 2019, 11, 265. [Google Scholar] [CrossRef]
- Bolpagni, R.; Poikane, S.; Laini, A.; Bagella, S.; Bartoli, M.; Cantonati, M. Ecological and Conservation Value of Small Standing-Water Ecosystems: A Systematic Review of Current Knowledge and Future Challenges. Water 2019, 11, 402. [Google Scholar] [CrossRef]
- Seeteram, N.A.; Hyera, P.T.; Kaaya, L.T.; Lalika, M.C.S.; Anderson, E.P. Conserving rivers and their biodiversity in Tanzania. Water 2019, 11, 2612. [Google Scholar] [CrossRef]
- Taylor, R.G.; Scanlon, B.; Döll, P.; Rodell, M.; Van Beek, R.; Wada, Y.; Longuevergne, L.; Leblanc, M.; Famiglietti, J.S.; Edmunds, M.; et al. Ground water and climate change. Nat. Clim. Chang. 2013, 3, 322–329. [Google Scholar] [CrossRef]
- Kløve, B.; Ala-Aho, P.; Bertrand, G.; Gurdak, J.J.; Kupfersberge, H.; Kværner, J.; Muotka, T.; Mykrä, H.; Preda, E.; Rossi, P.; et al. Climate change impacts on groundwater and dependent ecosystems. J. Hydrol. 2014, 518, 250–266. [Google Scholar] [CrossRef]
- Steube, C.; Richter, S.; Griebler, C. First attempts towards an integrative concept for the ecological assessment of groundwater ecosystems. Hydrogeol. J. 2009, 17, 23–35. [Google Scholar] [CrossRef]
- Miller, M.P.; Buto, S.G.; Susong, D.D.; Rumsey, C.A. The importance of base flow in sustaining surface water flow in the Upper Colorado River Basin. Water Resour. Res. 2016, 52, 3547–3562. [Google Scholar] [CrossRef]
- Organization for Economic Co-operation and Development—OECD. Groundwater Allocation: Managing Growing Pressures on Quantity and Quality, OECD Studies on Water; OECD Publishing: Paris, France, 2017. [Google Scholar] [CrossRef]
- European Environmental Agency (EEA). 2019. Available online: https://www.eea.europa.eu/data-and-maps/data/waterbase-water-quality-2 (accessed on 21 November 2019).
- Kløve, B.; Ala-Aho, P.; Bertrand, G.; Boukalova, Z.; Ertürk, A.; Goldscheider, N.; Ilmonen, J.; Karakaya, N.; Karakaya, N.; Kupfersberger, H.; et al. Groundwater dependent ecosystems. Part I: Hydroecological status and trends. Environ. Sci. Policy 2011, 14, 770–781. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Galassi, D.M.P. Agricultural impacts on Mediterranean alluvial aquifers: Do invertebrates respond? Fund. Appl. Limnol. 2013, 182, 271–281. [Google Scholar] [CrossRef]
- Richter, B.D.; Postel, S.; Revenga, C.; Scudder, T.; Lehner, B.; Churchill, A.; Chow, M. Lost in development’s shadow: The downstream human consequences of dams. Water Alter. 2010, 3, 14–42. [Google Scholar] [CrossRef]
- Malard, F.; Tockner, K.; Dole-Olivier, M.-J.; Ward, J.V. A landscape perspective of surface–subsurface hydrological exchanges in river corridors. Freshw. Biol. 2002, 47, 621–640. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cifoni, M.; Lombardo, P.; Fiasca, B.; Galassi, D.M.P. Ammonium threshold values for groundwater quality in the EU may not protect groundwater fauna: Evidence from an alluvial aquifer in Italy. Hydrobiologia 2015, 743, 139–150. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Cifoni, M.; Fiasca, B.; Di Cioccio, A.; Galassi, D.M.P. Ecological risk assessment of pesticide mixtures in the alluvial aquifers of central Italy: Toward more realistic scenarios for risk mitigation. Sci. Total Environ. 2018, 644, 161–172. [Google Scholar] [CrossRef]
- Di Marzio, W.D.; Cifoni, M.; Sáenz, M.E.; Galassi, D.M.P.; Di Lorenzo, T. The ecotoxicity of binary mixtures of Imazamox and ionized ammonia on freshwater copepods: Implications for environmental risk assessment in groundwater bodies. Ecotoxicol. Environ. Saf. 2018, 149, 72–79. [Google Scholar] [CrossRef]
- Olisah, C.; Okoh, O.O.; Okoh, A.I. Global evolution of organochlorine pesticides research in biological and environmental matrices from 1992 to 2018: A bibliometric approach. Emerg. Contam. 2019, 5, 157–167. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Sáenz, M.E.; Baratti, M.; Dedonno, A.A.; Iannucci, A.; Cannicci, S.; Messana, G.; Galassi, D.M.P. Sensitivity of hypogean and epigean freshwater copepods to agricultural pollutants. Environ. Sci. Pollut. Res. 2014, 21, 4643–4655. [Google Scholar] [CrossRef]
- Eamus, D.; Froend, R. Groundwater-dependent ecosystems: The where, what and why of GDEs. Aust. J. Bot. 2006, 54, 91–96. [Google Scholar] [CrossRef]
- Environmental Protection Authority—EPA. Guidance for the Assessment of Environmental Factors (In Accordance with the Environmental Protection Act 1986): Consideration of Subterranean Fauna in Groundwater and Caves During Environmental Impact Assessment in Western Australia; Environmental Protection Authority: Perth, Australia, 2004; p. 98.
- Lapworth, D.J.; Baran, N.; Stuart, M.E.; Ward, R.S. Emerging organic contaminants in groundwater: A review of sources, fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef]
- Postigo, C.; Barceló, D. Synthetic organic compounds and their transformation products in groundwater: Occurrence, fate and mitigation. Sci. Total Environ. 2015, 504, 32–47. [Google Scholar] [CrossRef]
- Stuart, M.; Lapworth, D.; Crane, E.; Hart, A. Review of risk from potential emerging contaminants in UK groundwater. Sci. Total Environ. 2012, 416, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, T.; Di Cicco, M.; Di Censo, D.; Galante, A.; Boscaro, F.; Messana, G.; Galassi, D.M.P. Environmental risk assessment of propranolol in the groundwater bodies of Europe. Environ. Pollut. 2019, 255. [Google Scholar] [CrossRef] [PubMed]
- Meffe, R.; Bustamante, I. Emerging organic contaminants in surface water and groundwater: A first overview of the situation in Italy. Sci. Total Environ. 2014, 481, 280–295. [Google Scholar] [CrossRef] [PubMed]
- Jurado, A.; Walther, M.; Diaz-Cruz, S.M. Occurrence, fate and environmental risk assessment of the organic microcontaminants included in the Watch Lists set by EU Decisions 2015/495 and 2018/840 in the groundwater of Spain. Sci. Total Environ. 2019, 663, 258–296. [Google Scholar] [CrossRef] [PubMed]
- Di Marzio, W.D.; Castaldo, D.; Di Lorenzo, T.; Di Cioccio, A.; Sáenz, M.E.; Galassi, D.M.P. Developmental endpoints of chronic exposure to suspected endocrine-disrupting chemicals on benthic and hyporheic freshwater copepods. Ecotoxicol. Environ. Saf. 2013, 96, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Griebler, C.; Avramov, M. Groundwater ecosystem services—A review. Freshw. Sci. 2014, 34, 355–367. [Google Scholar] [CrossRef]
- Galassi, D.M.P.; Lombardo, P.; Fiasca, B.; Di Cioccio, A.; Di Lorenzo, T.; Petitta, M.; Di Carlo, P. Earthquakes trigger the loss of groundwater biodiversity. Sci. Rep. 2014, 4, 6273. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Galassi, D.M.P. Effect of temperature rising on the stygobiotic crustacean species Diacyclops belgicus: Does global warming affect groundwater populations? Water 2017, 9, 951. [Google Scholar] [CrossRef]
- Issartel, J.; Hervant, F.; Voituron, Y.; Renault, D.; Vernon, P. Behavioural, ventilatory and respiratory responses of epigean and hypogean crustaceans to different temperatures. Comp. Biochem. Physiol. 2005, 141, 1–7. [Google Scholar] [CrossRef]
- Colson-Proch, C.; Morales, A.; Hervant, F.; Konecny, L.; Moulin, C.; Douady, C.J. First cellular approach of the effects of global warming on groundwater organisms: A study of the HSP70 gene expression. Cell Stress Chaperon. 2010, 15, 259–270. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Di Marzio, W.D.; Spigoli, D.; Baratti, M.; Messana, G.; Cannicci, S.; Galassi, D.M.P. Metabolic rates of a hypogean and an epigean species of copepod in an alluvial aquifer. Freshw. Biol. 2015, 71, 426–435. [Google Scholar] [CrossRef]
- Kefford, B.J.; Palmer, C.G.; Pakhomova, L.; Nugegoda, D. Comparing test systems to measure the salinity tolerance of freshwater invertebrates. Water Sa 2004, 30, 499–507. [Google Scholar] [CrossRef][Green Version]
- Cantonati, M.; Stevens, L.E.; Segadelli, S.; Springer, A.E.; Goldscheider, N.; Celico, F.; Filippini, M.; Ogata, K.; Gargini, A. Ecohydrogeology: The interdisciplinary convergence needed to improve the study of springs and other groundwater-dependent habitats, biota, and ecosystems. Ecol. Indic. 2020, 110, 105803. [Google Scholar] [CrossRef]
- Tomlinson, M.; Boulton, A.J. Ecology and management of subsurface groundwater dependent ecosystems in Australia—A review. Mar. Freshw. Res. 2010, 61, 936–949. [Google Scholar] [CrossRef]
- Woodward, G.; Perkins, D.M.; Brown, L.E. Climate change and freshwater ecosystems: Impacts across multiple levels of organization. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 2093–2106. [Google Scholar] [CrossRef]
- Stubbington, R.; Wood, P.J.; Boulton, A.J. Low flow controls on benthic and hyporheic macroinvertebrate assemblages during supra-seasonal drought. Hydrol. Proc. 2009, 23, 2253–2263. [Google Scholar] [CrossRef]
- Griebler, C.; Lueders, T. Microbial biodiversity in groundwater ecosystems. Freshw. Biol. 2009, 54, 649–677. [Google Scholar] [CrossRef]
- Mammola, S.; Cardoso, P.; Culver, D.C.; Deharveng, L.; Ferreira, R.L.; Fišer, C.; Galassi, D.M.P.; Griebler, C.; Halse, S.; Humphreys, W.F.; et al. Scientists’ Warning on the Conservation of Subterranean Ecosystems. BioScience 2019, 69, 641–650. [Google Scholar] [CrossRef]
- Humphreys, W.F. Rising from Down Under: Developments in subterranean biodiversity in Australia from a groundwater fauna perspective. Invertebr. Syst. 2008, 22, 85–101. [Google Scholar] [CrossRef]
- Gibert, J.; Culver, D.C. Assessing and conserving groundwater biodiversity: An introduction. Freshw. Biol. 2009, 54, 639–648. [Google Scholar] [CrossRef]
- Gibert, J.; Deharveng, L. Subterranean Ecosystems: A Truncated Functional Biodiversity. BioScience 2002, 52, 473–481. [Google Scholar] [CrossRef]
- Galassi, D.M.P.; Huys, R.; Reid, J.W. Diversity, ecology and evolution of groundwater copepods. Freshw. Biol. 2009, 54, 691–708. [Google Scholar] [CrossRef]
- Fattorini, S.; Fiasca, B.; Di Lorenzo, T.; Di Cicco, M.; Galassi, D.M.P. A new protocol for assessing the conservation priority of groundwater dependent ecosystems. Aquat. Conserv. Mar. Freshw. Ecosyst. under review.
- Galassi, D.M.P.; Fiasca, B.; Di Lorenzo, T.; Montanari, A.; Porfirio, S.; Fattorini, S. Groundwater biodiversity in a chemoautotrophic cave ecosystem: How geochemistry regulates microcrustacean community structure. Aquat. Ecol. 2017, 51, 75–90. [Google Scholar] [CrossRef]
- Malard, F.; Boutin, C.; Camacho, A.I.; Ferreira, D.; Michel, G.; Sket, B.; Stoch, F. Diversity patterns of stygobiotic crustaceans across multiple spatial scales in Europe. Freshw. Biol. 2009, 54, 756–776. [Google Scholar] [CrossRef]
- Stoch, F.; Galassi, D.M.P. Stygobiotic crustacean species richness: A question of numbers, a matter of scale. Hydrobiologia 2010, 653, 217–234. [Google Scholar] [CrossRef]
- Gibert, J.; Culver, D. Diversity patterns in Europe. In Encyclopedia of Caves; Culver, D., White, W.B., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 196–201. ISBN 9780128141243. [Google Scholar]
- Boulton, A.J.; Fenwick, G.D.; Hancock, P.J.; Harvey, M.S. Biodiversity, functional roles and ecosystem services of groundwater invertebrates. Invertebr. Syst. 2008, 22, 103–116. [Google Scholar] [CrossRef]
- Batubara, B.; Batelaan, O.; Quevauviller, P. Science-policy interfacing on the issue of groundwater and groundwater-dependent ecosystems in Europe: Implications for research and policy. WIREs Water 2014, 1, 561–571. [Google Scholar] [CrossRef]
- Tomlinson, M.; Boulton, A.J.; Hancock, P.J.; Cook, P.G. Deliberate omission or unfortunate oversight: Should stygofaunal surveys be included in routine groundwater monitoring programs? Hydrogeol. J. 2007, 15, 1317–1320. [Google Scholar] [CrossRef]
- Cardoso, P.; Erwin, T.L.; Borges, P.A.V. The seven impediments in invertebrate conservation and how to overcome them. Biol. Cons. 2011, 144, 2647–2655. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Canedoli, C.; Stoch, F. The Racovitzan impediment and the hidden biodiversity of unexplored environments. Conserv. Biol. 2019, 33, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Deharveng, L.; Stoch, F.; Gibert, J.; Bedos, A.; Galassi, D.M.P.; Zagmajster, M.; Brancelj, A.; Camacho, A.; Fiers, F.; Martin, P.; et al. Groundwater biodiversity in Europe. Freshw. Biol. 2009, 54, 709–726. [Google Scholar] [CrossRef]
- BioFreshProject. Available online: http://project.freshwaterbiodiversity.eu/ (accessed on 21 November 2019).
- Zagmajster, M.; Eme, D.; Fišer, C.; Galassi, D.M.P.; Marmonier, P.; Stoch, F.; Cornu, J.-F.; Malard, F. Geographic variation in range size and beta diversity of groundwater crustaceans: Insights from habitats with low thermal seasonality. Glob. Ecol. Biogeogr. 2014, 23, 1135–1145. [Google Scholar] [CrossRef]
- Galassi, D.M.P.; Dole-Olivier, M.-J.; De Laurentiis, P. Phylogeny and biogeography of the genus Pseudectinosoma, and description of P. janineae sp. n. (Crustacea: Copepoda, Ectinosomatidae). Zool. Scr. 1999, 28, 289–303. [Google Scholar] [CrossRef]
- Council of the European Communities. Council Directive92⁄43⁄EEC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. Off. J. Eur. Communities 1992, 35, 7–50. [Google Scholar]
- Michel, G.; Malard, F.; Deharveng, L.; Di Lorenzo, T.; Sket, B.; De Boyer, C. Reserve selection for conserving groundwater biodiversity. Freshw. Biol. 2009, 54, 861–876. [Google Scholar] [CrossRef]
- Strona, G.; Fattorini, S.; Fiasca, B.; Di Lorenzo, T.; Di Cicco, M.; Lorenzetti, W.; Boccacci, F.; Galassi, D.M.P. AQUALIFE Software: A New Tool for a Standardized Ecological Assessment of Groundwater Dependent Ecosystems. Water 2019, 11, 2574. [Google Scholar] [CrossRef]
- Margules, C.R.; Pressey, R.L. Systematic conservation planning. Nature 2000, 405, 243–253. [Google Scholar] [CrossRef]
- Linke, S.; Turak, E.; Asmyhr, M.G.; Hose, G. 3D conservation planning: Including aquifer protection in freshwater plans refines priorities without much additional effort. Aquat. Conserv. Mar. Freshw. Ecosys. 2019, 29, 1063–1072. [Google Scholar] [CrossRef]
- Cantonati, M.; Komárek, J.; Montejano, G. Cyanobacteria in ambient springs. Biodivers. Conserv. 2015, 24, 865–888. [Google Scholar] [CrossRef]
- Johnson, R.H.; DeWit, E.; Wirt, L.; Manning, A.H. Using geochemistry to identirfy the source of groundwater to Montezuma Well, a natural springs in central Arizona, USA: Part 2. Envir. Earth Sci. 2012, 67, 1837–1853. [Google Scholar] [CrossRef]
- Glazier, D.S. Springs. In Reference Module in Earth Systems and Environmental Sciences; Elias, S., Ed.; Elsevier: Waltham, MA, USA, 2014; pp. 1–78. ISBN 978-0-12-409548-9. [Google Scholar]
- Stevens, L.E.; Johnson, R.R.; Estes, C. The watershed continuum: A conceptual stream riparian ecosystem model. In Riparian Research and Management: Past, Present, and Future; General Technical Report RMRS-GTR-303; Johnson, R.R., Carothers, S.W., Finch, D.M., Kingsley, K.J., Stanley, J.R.T., Eds.; U.S. Department of Agriculture; Forest Service: Washington, DC, USA; Rocky Mountain Research Station: Fort Collins, CO, USA, 2018; Volume 2, in press. [Google Scholar]
- Springer, A.E.; Stevens, L.E.; Anderson, E.E.; Parnell, R.A.; Kreamer, D.K.; Levin, L.; Flora, S.P. A comprehensive springs classification system: Integrating geomorphic, hydrogeochemical, and ecological criteria. In Aridland Springs in North America: Ecology and Conservation; Stevens, L.E., Meretsky, V.J., Eds.; University of Arizona Press: Tucson, AZ, USA, 2008; pp. 49–75. ISBN 978-0-8165-2645-1. [Google Scholar]
- Spitale, D.; Leira, M.; Angeli, N.; Cantonati, M. Environmental classification of springs of the Italian Alps and its consistency across multiple taxonomic groups. Freshw. Sci. 2012, 31, 563–574. [Google Scholar] [CrossRef]
- Springer, A.E.; Stevens, L.E. Spheres of discharge of springs. Hydrogeol. J. 2009, 17, 83–93. [Google Scholar] [CrossRef]
- Stevens, L.E.; Schenk, E.R.; Springer, A.E. Springs ecosystem classification. Ecol. Appl. in press.
- Stevens, L.E.; Ledbetter, J.D.; Springer, A.E.; Campbell, C.; Misztal, L.; Joyce, M.; Hardwick, G. Arizona Springs Rehabilitation Handbook; Spring Stewardship Institute: Flagstaff, AZ, USA; Sky Island Alliance: Tucson, AZ, USA, 2016. [Google Scholar]
- Shannon, C.E. A Mathematical Theory of Communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Springer, A.E.; Stevens, L.E.; Ledbetter, J.D.; Schaller, E.M.; Gill, K.; Rood, S.B. Ecohydrology and stewardship of Alberta springs ecosystems. Ecohydrology 2015, 8. [Google Scholar] [CrossRef]
- Sinclair, D.A. Springs Geomorphology Influences on Physical and Vegetation Ecosystem Characteristics, Grand Canyon Ecoregion, USA. Master’s Thesis, Northern Arizona University, Flagstaff, AZ, USA, 2018. [Google Scholar]
- Cantonati, M.; Lange-Bertalot, H. Diatom biodiversity of springs in the Berchtesgaden National Park (northern Alps, Germany), with the ecological and morphological characterization of two species new to science. Diatom Res. 2010, 25, 251–280. [Google Scholar] [CrossRef]
- Cantonati, M.; Lange-Bertalot, H.; Scalfi, A.; Angeli, N. Cymbella tridentina sp. nov. (Bacillariophyta), a crenophilous diatom from carbonate springs of the Alps. J. N. Am. Benthol. Soc. 2010, 29, 775–788. [Google Scholar] [CrossRef]
- Cantonati, M.; Füreder, L.; Gerecke, R.; Jüttner, I.; Cox, E.J. Crenic habitats, hotspots for freshwater biodiversity conservation: Toward an understanding of their ecology. Freshw. Sci. 2012, 31, 463–480. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef]
- Huston, M. A general hypothesis of species diversity. Am. Nat. 1979, 113, 81–101. [Google Scholar] [CrossRef]
- Huston, M.A. Biology Diversity: The Coexistence of Species on Changing Landscapes; Cambridge University Press: Cambridge, UK, 1994; ISBN 0-521-36930-4. [Google Scholar]
- Kodric-Brown, A.; Brown, L.H. Native fishes, exotic mammals, and the conservation of desert springs. Front. Ecol. Environ. 2007, 10, 549–553. [Google Scholar] [CrossRef]
- Odum, H.T. Trophic Structure and Productivity of Silver Springs, Florida. Ecol. Monogr. 1957, 27, 55–112. [Google Scholar] [CrossRef]
- Blinn, D.W. The extreme environment, trophic structure, and ecosystem dynamics of a large fishless desert spring: Montezuma Well, Arizona. In Aridland Springs in North America: Ecology and Conservation; Stevens, L.E., Meretsky, V.J., Eds.; University of Arizona Press: Tucson, AZ, USA, 2008; ISBN 978-0-8165-2645-1. [Google Scholar]
- Williams, D.D.; Danks, H.V. Arthropods of springs, with particular reference to Canada. Mem. Entomol. Soc. Can. 1991, 203–217. [Google Scholar] [CrossRef]
- Erman, N.A. Factors determining biodiversity in Sierra Nevada cold spring systems. In The History of Water: Eastern Sierra Nevada, Owens Valley, White-Inyo Mountains; Hall, C.A., Jr., Doyle-Jones, V., Widawski, B., Eds.; University of California White Mountains Research Station: Los Angeles, CA, USA, 1992; pp. 119–127. ISBN 978-1879851047. [Google Scholar]
- Botosaneanu, L. Studies in Crenobiology: The Biology of Springs and Springbrooks; Backhuys: Leiden, The Netherlands, 1998. [Google Scholar] [CrossRef]
- Ponder, W.F. Endemic aquatic macroinvertebrates of artesian springs of the Great Artesian Basin—Progress and future directions. Rec. South Aust. Mus. Monogr. Ser. 2004, 7, 101–110. [Google Scholar]
- Fensham, R.J.; Silcock, J.L.; Kerezsy, A.; Ponder, W. Four desert waters: Setting arid zone wetland conservation priorities. Biol. Conserv. 2011, 144, 2459–2467. [Google Scholar] [CrossRef]
- Unmack, P.J. Australian Desert Fishes Descriptions. 2003. Available online: Desertfishes.org/Australia/index.html (accessed on 5 October 2019).
- Unmack, P.J.; Minckley, W.L. The demise of desert springs. In Aridland Springs in North America: Ecology and Conservation; Stevens, L.E., Meretsky, V.J., Eds.; University of Arizona Press: Tucson, AZ, USA, 2008; pp. 11–34. ISBN 978-0-8165-2645-1. [Google Scholar]
- Martens, A.; Richter, O.; Suhling, F. The relevance of perennial springs for regional biodiversity and conservation. In Biodiversity in Southern Africa 2: Patterns and Processes at Regional Scale; Schmiedel, U., Jürgens, N., Eds.; Klaus Hess Publishers: Göttingen, Germany; Windhoek, Namibia, 2010; pp. 70–74. ISBN 978-3-933117-46-5. [Google Scholar]
- Cuthbert, M.O.; Ashley, G.M. A spring forward for hominin evolution in East Africa. PLoS ONE 2014, 9, e107358. [Google Scholar] [CrossRef]
- Cantonati, M.; Rott, E.; Spitale, D.; Angeli, N.; Komárek, J. Are benthic algae related to spring types? Freshw. Sci. 2012, 31, 481–498. [Google Scholar] [CrossRef]
- Cantonati, M.; Lange-Bertalot, H. Achnanthidium dolomiticum sp. nov. (Bacillariophyta) from oligotrophic mountain springs and lakes fed by dolomite aquifers. J. Phycol. 2006, 42, 1184–1188. [Google Scholar] [CrossRef]
- Cantonati, M.; Lange-Bertalot, H.; Angeli, N. Neidiomorpha gen. nov. (Bacillariophyta): A new freshwater diatom genus separated from Neidium Pfitzer. Bot. Stud. 2010, 51, 195–202. [Google Scholar]
- Rott, E.; Hotzy, R.; Cantonati, M.; Sanders, D. Calcification types of Oocardium stratum Nägeli and microhabitat conditions in springs of the Alps. Freshw. Sci. 2012, 31, 610–624. [Google Scholar] [CrossRef]
- Cantonati, M.; Segadelli, S.; Ogata, K.; Tran, H.; Sanders, D.; Gerecke, R.; Rott, E.; Filippini, M.; Gargini, A.; Celico, F. A global review on ambient Limestone-Precipitating Springs (LPS): Hydrogeological setting, ecology, and conservation. Sci. Total Environ. 2016, 568, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Angeli, N.; Cantonati, M.; Spitale, D.; Lange-Bertalot, H. A comparison between diatom assemblages in two groups of carbonate, low-altitude springs with different levels of anthropogenic disturbances. Fottea 2010, 10, 115–128. [Google Scholar] [CrossRef]
- Cantonati, M.; Ortler, K. Using spring biota of pristine mountain areas for long term monitoring. IAHS Publ. 1998, 248, 379–385. [Google Scholar]
- Ashley, G.M.; de Wet, C.B.; Barboni, D.; Magill, C.R. Subtle signatures of seeps: Record of groundwater in a dryland, DK, Olduvai Gorge, Tanzania. Depos. Rec. 2016, 2, 4–21. [Google Scholar] [CrossRef]
- Haynes, C.V., Jr. Quaternary cauldron springs as paleoecological archives. In Aridland Springs in North America: Ecology and Conservation; Stevens, L.E., Ed.; University of Arizona Press: Tucson, AZ, USA, 2008. [Google Scholar]
- Kreamer, D.K.; Stevens, L.E.; Ledbetter, J.D. Groundwater dependent ecosystems—Science, challenges, and policy directions. In Groundwater; Adelana, S.M., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2015; ISBN 978-1-63321-759-1. [Google Scholar]
- Boschen, R.E.; Rowden, A.A.; Clark, M.R.; Gardner, J.P.A. Mining of deep-sea seafloor massive sulfides: A review of the deposits, their benthic communities, impacts from mining, regulatory frameworks and management strategies. Ocean Coast. Manag. 2013, 84, 54–67. [Google Scholar] [CrossRef]
- De Granade, R.; Stevens, L.E. Desert Oases: Aridland Springs as Diverse Biocultural Ecosystems. In Encyclopedia of the World’s Biomes; Elsevier: Amsterdam, The Netherlands, 2020. (in press)
- Gomi, T.; Sidle, R.; Richardson, J.S. Headwater and channel network -understanding processes and downstream linkages of headwater systems. BioScience 2002, 52, 905–916. [Google Scholar] [CrossRef]
- Strahler, A.N. Quantitative Geomorphology of Drainage Basins and Channel Networks. In Handbook of Applied Hydrology; Chow, V.T., Ed.; McGraw Hill: New York, NY, USA, 1954; ISBN 978-0071835091. [Google Scholar]
- Pitcher, L.H.; Smith, L.C. Supraglacial streams and rivers. Annu. Rev. Earth Planet. Sci. 2019, 47, 421–452. [Google Scholar] [CrossRef]
- Hodson, A.; Anesio, A.M.; Tranter, M.; Fountain, A.; Osborn, M.; Priscu, J.; Laybourn-Parry, J.; Sattler, B. Glacial ecosystems. Ecol. Monogr. 2008, 78, 41–67. [Google Scholar] [CrossRef]
- Ward, J.V. Ecology of alpine streams. Freshw. Biol. 1994, 32, 277–294. [Google Scholar] [CrossRef]
- Brittain, J.E.; Milner, A.M. Ecology of glacier-fed rivers: Current status and concepts. Freshw. Biol. 2001, 46, 1571–1578. [Google Scholar] [CrossRef]
- Whittaker, R.H. Vegetation of the Siskiyou Mountains, Oregon and California. Ecol. Monogr. 1960, 30, 279–338. [Google Scholar] [CrossRef]
- Jacobsen, D.; Milner, A.M.; Brown, L.E.; Dangles, O. Biodiversity under threat in glacier-fed river systems. Nat. Clim. Chang. 2012, 2, 361–364. [Google Scholar] [CrossRef]
- Cantonati, M.; Corradini, G.; Jüttner, I.; Cox, E.J. Diatom assemblages in high mountain streams of the Alps and the Himalaya. Nova Hedwig. 2001, 123, 37–62. [Google Scholar]
- Gesierich, D.; Rott, E. Is diatom richness responding to catchment glaciation? J. Limnol. 2012, 71, 72–83. [Google Scholar] [CrossRef]
- Brown, L.E.; Khamis, K.; Wilkes, M.; Blaen, P.; Brittain, J.E.; Carrivick, J.L.; Fell, S.; Friberg, N.; Füreder, L.; Gislason, G.M.; et al. Functional diversity and community assembly of river invertebrates show globally consistent responses to decreasing glacier cover. Nat. Ecol. Evol. 2018, 2, 325–333. [Google Scholar] [CrossRef]
- Niedrist, G.H.; Cantonati, M.; Füreder, L. Environmental harshness mediates the quality of periphyton and chironomid body mass in alpine streams. Freshw. Sci. 2018, 37, 519–533. [Google Scholar] [CrossRef]
- Vannote, R.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The river continuum concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Frissell, C.A.; Liss, W.J.; Warren, C.E.; Hurley, M.D. A hierarchical framework for stream habitat classification: Viewing streams in a watershed context. Environ. Manag. 1986, 10, 199–214. [Google Scholar] [CrossRef]
- Hayes, J.W.; Goodwin, E.O.; Shearer, K.A.; Hicks, D.M. Relationship between background invertebrate drift concentration and flow over natural flow recession and prediction with a drift transport model. Can. J. Fish. Aquat. Sci. 2019, 76, 871–885. [Google Scholar] [CrossRef]
- Nilsson, C.; Brown, R.L.; Jansson, R.; Merritt, D.M. The role of hydrochory in structuring riparian and wetland vegetation. Biol. Rev. 2010, 85, 837–858. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.R.; Newbold, J.D.; Lin, L. Nutrient Spiraling and Transport in Streams: The Importance of In-Stream Biological Processes to Nutrient Dynamics in Streams. In Stream Ecosystems in a Changing Environment; Jones, J.B.E., Stanley, E., Eds.; Elsevier Academic Press: Cambridge, MA, USA, 2016; pp. 181–199. ISBN 9780124058903. [Google Scholar]
- Wallace, J.B.; Merritt, R.W. Filter-feeding ecology of aquatic insects. Annu. Rev. Entomol. 1980, 25, 103–132. [Google Scholar] [CrossRef]
- Naman, S.M.; Rosenfeld, J.S.; Richardson, J.S. Causes and consequences of invertebrate drift in running waters: From individuals to populations and trophic fluxes. Can. J. Fish. Aquat. Sci 2016, 73, 1292–1305. [Google Scholar] [CrossRef]
- Ward, J.V.; Stanford, J.A. The serial discontinuity concept—Extending the model to floodplain rivers. Regul. Rivers Res. Manag. 1995, 10, 159–168. [Google Scholar] [CrossRef]
- Richardson, J.S.; Mackay, R.J. Lake outlets and the distribution of filter feeders: An assessment of hypotheses. Oikos 1991, 62, 370–380. [Google Scholar] [CrossRef]
- Willis, T.V.; Magnuson, J.J. Patterns in fish species composition across the interface between streams and lakes. Can. J. Fish. Aquat. Sci. 2000, 57, 1042–1052. [Google Scholar] [CrossRef]
- Wotton, R.S.; Malmqvist, B.; Leonardsson, K. Expanding traditional views on suspension feeders—Quantifying their role as ecosystem engineers. Oikos 2003, 101, 441–443. [Google Scholar] [CrossRef]
- Dunn, R.R.; Colwell, R.K.; Nilsson, C. The river domain: Why are there more species halfway up the river? Ecography 2006, 29, 251–259. [Google Scholar] [CrossRef]
- Heino, J.; Paavola, R.; Virtanen, R.; Muotka, T. Searching for biodiversity indicators in running waters: Do bryophytes, macroinvertebrates, and fish show congruent diversity patterns? Biodivers. Conserv. 2005, 14, 415–428. [Google Scholar] [CrossRef]
- Vander Vorste, R.; McElmurray, P.; Bell, S.; Eliason, K.M.; Brown, B.L. Does stream size really explain biodiversity patterns in lotic systems? A call for mechanistic explanations. Diversity 2017, 9, 26. [Google Scholar] [CrossRef]
- Berg, R.; Gaumert, T.; Kämmereit, M.; Klinger, H.; Lemcke, R.; Leuner, E.; Wolter, C.; Dußling, U. The German river typology with respect to fish-faunistic reference conditions. In Handbuch Angewandte Limnologie; Steinberg, C., Calmano, W., Klapper, H., Wilken, R.D., Eds.; Ecomed-Verlag: Landsberg, Germany, 2004; pp. 12–17. ISBN 3-609-75820-1. [Google Scholar]
- Hynes, H.B.N. Keynote Address. In Proceedings of the International Large River Symposium, Honey Harbour, ON, Canada, 14–21 September 1986; Dodge, D.P., Ed.; pp. 5–10. [Google Scholar]
- Church, M. Channel stability: Morphodynamics and the morphology of rivers. In Rivers—Physical, Fluvial and Environmental Processes; Rowiński, P., Radecki-Pawlik, A., Eds.; Springer International: Cham, Switzerland, 2015; pp. 281–321. ISBN 978-3-319-17719-9. [Google Scholar]
- Chetelat, J.; Pick, F.R.; Hamilton, P.B. Potamoplankton size structure and taxonomic composition: Influence of river size and nutrient concentrations. Limnol. Oceanogr. 2006, 51, 681–689. [Google Scholar] [CrossRef]
- Pracheil, B.M.; McIntyre, P.B.; Lyons, J.D. Enhancing conservation of large-river biodiversity by accounting for tributaries. Front. Ecol. Environ. 2013, 11, 124–128. [Google Scholar] [CrossRef]
- He, F.; Zarfl, C.; Bremerich, V.; David, J.N.W.; Hogan, Z.; Kalinkat, G.; Tockner, K.; Jähnig, S.C. The global decline of freshwater megafauna. Glob. Chang. Biol. 2019, 25, 3883–3892. [Google Scholar] [CrossRef] [PubMed]
- Schletterer, M.; Kuzovlev, V.V.; Zhenikov, Y.N.; Tuhtan, J.A.; Haidvogl, G.; Friedrich, T.; Górski, K.; Füreder, L. Fish fauna and fisheries of large European rivers: Examples from the Volga and the Danube. Hydrobiologia 2018, 814, 45–60. [Google Scholar] [CrossRef]
- Nilsson, C.; Reidy, C.A.; Dynesius, M.; Revenga, C. Fragmentation and flow regulation of the world’s large river systems. Science 2005, 308, 405–408. [Google Scholar] [CrossRef]
- Pringle, C.M.; Freeman, M.; Freeman, B. Regional effects of hydrologic alterations on riverine macrobiota in the New World: Tropical-temperate comparisons. BioScience 2000, 50, 807–823. [Google Scholar] [CrossRef]
- Pringle, C.M. Interacting effects of altered hydrology and contaminant: Emerging ecological patterns of global concern. In Achieving Sustainable Freshwater Systems: A Web of Connections; Holland, M., Blood, E., Shaffer, L., Eds.; Island Press: Washington, DC, USA, 2003; pp. 85–107. [Google Scholar]
- Olden, J.D. Challenges and opportunities for fish conservation in dam-impacted waters. In Conservation of Freshwater Fishes; Closs, G., Krkosek, M., Olden, J., Eds.; Cambridge University Press: Cambridge, UK, 2016; pp. 107–148. [Google Scholar] [CrossRef]
- Pringle, C.M.; Withrington, D. Freshwater conservation in action: Contrasting approaches in the US and UK. Chapter 2. In Assessing the Conservation Value of Fresh Waters; Boon, P.J., Pringle, C.M., Eds.; Cambridge University Press: Cambridge, UK, 2009; ISBN 978-0521613224. [Google Scholar]
- Herendorf, C.E. Distribution of the world’s large lakes. In Large Lakes: Ecological Structure and Function; Tilzer, M.M., Serruya, C., Eds.; Springer: Cham, Switzerland, 1990; pp. 3–38. ISBN 978-3-642-84077-7. [Google Scholar]
- Vadeboncoeur, Y.; McIntyre, P.B.; Vander Zanden, M.J. Borders of biodiversity: Life at the edge of the world’s large lakes. BioScience 2011, 61, 526–537. [Google Scholar] [CrossRef]
- Gratton, P.; Allegrucci, G.; Sbordoni, V.; Gandolfi, A. The evolutionary jigsaw puzzle of the surviving trout (Salmo trutta L. complex) diversity in the Italian region: A multilocus Bayesian approach. Evol. Mol. Phylogenet. Evol. 2014, 79, 292–304. [Google Scholar] [CrossRef]
- Kociolek, J.P.; Stoermer, E.F. Oligotrophy: The forgotten end of an ecological spectrum. Acta Bot. Croat. 2009, 68, 465–472. [Google Scholar]
- Stoermer, E.F. Diatoms associated with bryophyte communities growing at extreme depths in Lake Michigan. Proc. Iowa Acad. Sci. 1981, 88, 91–95. [Google Scholar]
- Hampton, S.E.; McGowan, S.; Ozersky, T.; Virdis, S.G.P.; Vu, T.T.; Spanbauer, T.L.; Kraemer, B.M.; Swann, G.; Mackay, A.W.; Powers, S.M.; et al. Recent ecological change in ancient lakes. Limnol. Oceanogr. 2018, 63, 2277–2304. [Google Scholar] [CrossRef]
- Martens, K. Speciation in ancient lakes. Trends Ecol. Evol. 1997, 12, 177–182. [Google Scholar] [CrossRef]
- Nekola, J.C. Paleorefugia and neorefugia: The influence of colonization history on community pattern and process. Ecology 1999, 80, 2459–2473. [Google Scholar] [CrossRef]
- Boxshall, G.A.; Jaume, D. Making Waves: The Repeated Colonization Fresh Water by Copepod Crustaceans. Adv. Ecol. Res. 2000, 31, 61–79. [Google Scholar] [CrossRef]
- Cristescu, M.E.; Adamowicz, S.J.; Vaillant, J.J.; Haffner, G.D. Ancient lakes revisited: From the ecology to the genetics of speciation. Mol. Ecol. 2010, 19, 4837–4851. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.L. Speciation in ancient lakes. Q. Rev. Biol. 1950, 25, 30–176. [Google Scholar] [CrossRef]
- Martens, K.; Coulter, G.; Goddeeris, B. Speciation in ancient lakes—40 years after Brooks. Ergebn. Limnol. 1994, 44, 75–96. [Google Scholar]
- Mackay, W.; Edlund, M.B.; Khursevich, G. Diatoms in ancient lakes. In The Diatoms: Applications for the Environmental and Earth Sciences, 2nd ed.; Smol, J.P., Stoermer, E.F., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 209–228. ISBN 0521509963. [Google Scholar]
- Cohen, A.S.; Stone, J.R.; Beuning, K.R.; Park, L.E.; Reinthal, P.N.; Dettman, D.; Scholz, C.A.; Johnson, T.C.; King, J.W.; Talbot, M.R.; et al. Ecological consequences of early Late Pleistocene megadroughts in tropical Africa. Proc. Natl. Acad. Sci. USA 2007, 104, 16422–16427. [Google Scholar] [CrossRef]
- Johnson, T.C.; Scholz, C.A.; Talbot, M.R.; Kelts, K.; Ricketts, R.D.; Ngobi, G.; Beuning, K.; Ssemmanda, I.I.; McGill, J.W. Late Pleistocene desiccation of Lake Victoria and rapid evolution of cichlid fishes. Science 1996, 273, 1091–1093. [Google Scholar] [CrossRef]
- Fritz, S.C.; Baker, P.A.; Tapia, P.; Spanbauer, T.; Westover, K. Evolution of the Lake Titicaca basin and its diatom flora over the last ~370,000 years. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2012, 317–318, 93–103. [Google Scholar] [CrossRef]
- Hamilton, P.B.; Lavoie, I.; Poulin, M. Spatial, seasonal and inter-annual variability in environmental characteristics and phytoplankton standing stock of the temperate lowland Rideau river, Ontario, Canada. River Res. Appl. 2012, 28, 1551–1566. [Google Scholar] [CrossRef]
- Goulden, C.E.; Tumurtogoo, O.; Karabanov, E.; Mongontsetseg, A. The geological history and geography of Lake Hövsgöl. In The Geology, Biodiversity and Ecology of Lake Hövsgöl (Mongolia); Goulden, C.E., Sitnikova, T., Gelhaus, J., Boldgiv, B., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2006; pp. 1–19. ISBN 90-5782-162-1. [Google Scholar]
- Sabo, E.; Roy, D.; Hamilton, P.B.; Hehanussa, P.E.; McNeely, R.; Haffner, G.D. The plankton community of Lake Matano: Factors regulating plankton composition and relative abundance in an ancient, tropical lake of Indonesia. Hydrobiologia 2008, 615, 225–235. [Google Scholar] [CrossRef]
- Crowe, S.A.; Jones, C.A.; Katsev, S.; Magen, C.; O’Neill, A.H.; Sturm, A.; Canfield, D.E.; Haffner, G.D.; Mucci, A.; Sundby, B.; et al. Photoferrotrophs thrive in an Archean Ocean analogue. Proc. Natl. Acad. Sci. USA 2008, 105, 15938–15943. [Google Scholar] [CrossRef] [PubMed]
- Granina, L. The chemical budget of Lake Baikal: A review. Limnol. Oceanogr. 1997, 42, 373–378. [Google Scholar] [CrossRef]
- Talling, J.F.; Talling, I.B. The chemical composition of African lake waters. Intern. Rev. Gesamten Hydrobiol. Hydrogr. 1965, 50, 421–463. [Google Scholar] [CrossRef]
- Greenwood, P.H. What is a species flock? In Evolution of Fish Species Flocks; Echelle, A.A., Kornfield, I., Eds.; University of Maine Press: Orono, ME, USA, 1984; pp. 13–20. ISBN 0891010572. [Google Scholar]
- Michel, E. Phylogeny of a Gastropod Species Flock: Exploring Speciation in Lake Tanganyika in a Molecular Framework. Adv. Ecol. Res. 2000, 31, 275–302. [Google Scholar] [CrossRef]
- Turner, G.G. The Nature of Species in Ancient Lakes: Perspectives from the Fishes of Lake Malawi. Adv. Ecol. Res. 2000, 31, 39–60. [Google Scholar] [CrossRef]
- Stelbrink, B.; Jovanovska, E.; Levkov, Z.; Ognjanova-Rumenova, O.; Wilke, T.; Albrecht, C. Diatoms do radiate: Evidence for a freshwater species flock. J. Evol. Biol. 2018, 31, 1969–1975. [Google Scholar] [CrossRef]
- Snoeks, J. How well known is the ichthyodiversity of the large East African lakes? Adv. Ecol. Res. 2000, 31, 17–38. [Google Scholar] [CrossRef]
- Shtarkman, Y.M.; Koçer, Z.A.; Edgar, R.; Veerapaneni, R.S.; D’Elia, T.; Morris, P.F.; Rogers, S.O. Subglacial Lake Vostok (Antarctica) Accretion Ice Contains a Diverse Set of Sequences from Aquatic, Marine and Sediment-Inhabiting Bacteria and Eukarya. PLoS ONE 2013, 8, e67221. [Google Scholar] [CrossRef]
- Doran, P.T.; Fritsen, C.H.; McKay, C.P.; Priscu, J.C.; Adams, E.E. Formation and character of an ancient 19-m ice cover and underlying trapped brine in an ‘‘ice-sealed’’ east Antarctic lake. Proc. Natl. Acad. Sci. USA 2003, 100, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Christner, B.C.; Skidmore, M.L.; Priscu, J.C.; Tranter, M.; Foreman, M. Bacteria in subglacial environments. In Psychrophiles: From Biodiversity to Biotechnology; Margesin, R., Schinner, F., Marx, J.-C., Gerday, C., Eds.; Springer Verlag: Berlin, Germany, 2008; pp. 51–71. ISBN 978-3-540-74335-4. [Google Scholar]
- Bramburger, A.J.; Haffner, G.D.; Hamilton, P.B.; Hinz, F.; Hehanussa, P.E. An examination of species within the genus Surirella from the Malili Lakes, Sulawesi Island, Indonesia, with descriptions of 11 new taxa. Diatom Res. 2006, 21, 1–56. [Google Scholar] [CrossRef]
- Kozhova, O.M.; Izmest’eva, L.R. Lake Baikal. Evolution and Biodiversity; Backhuys: Leiden, The Netherlands, 1998; pp. 1–447. ISBN 90-5782-001-3. [Google Scholar]
- Strong, E.E.; Gargominy, O.; Ponder, W.F.; Bouchet, P. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. Hydrobiologia 2008, 595, 149–166. [Google Scholar] [CrossRef]
- Takhev, V.V. Trends in the Evolution of Baikal Amphipods and Evolutionary Parallels with some Marine Malcostracan Faunas. Adv. Ecol. Res. 2000, 31, 197–220. [Google Scholar] [CrossRef]
- Schön, I.; Pieri, V.; Sherbakov, D.Y.; Martens, K. Cryptic diversity and speciation in endemic Cytherissa (Ostracoda, Crustacea) from Lake Baikal. Hydrobiologia 2017, 800, 61–79. [Google Scholar] [CrossRef]
- Stoch, F.; Arteau, M.; Brancelj, A.; Galassi, D.M.P.; Malard, F. Biodiversity indicators in European groundwaters: Towards a predictive model of stygobiotic species richness. Freshw. Biol. 2009, 54, 745–755. [Google Scholar] [CrossRef]
- Meixner, K.J.; Luter, C.; Eckert, C.; Itskovich, V.; Janussen, D.; von Rintelen, T.; Bohne, A.V.; Meixner, J.M.; Hess, W.R. Phylogenetic analysis of freshwater sponges provide evidence for endemism and radiation in ancient lakes. Mol. Phylogenet. Evol. 2007, 35, 875–886. [Google Scholar] [CrossRef]
- Matveyev, A.N.; Samusenok, V.P. The fishes and fishery in Lake Baikal. Aquat. Ecosyst. Health 2015, 18, 134–148. [Google Scholar] [CrossRef]
- Kontula, T.; Kirilchik, S.V.; Väinölä, R. Endemic diversification of the monophyletic cottoid fish species flock in Lake Baikal explored with mtDNA sequencing. Mol. Phylogenet. Evol. 2003, 27, 143–155. [Google Scholar] [CrossRef]
- Herder, F.; Schwarzer, J.; Pfaender, J.; Hadiaty, R.K.; Schliewen, U.K. Preliminary checklist of sailfin silversides (Teleostei: Telmatherinidae) in the Malili Lakes of Sulawesi (Indonesia), with a synopsis of systematics and threats. Verb. Ges. Icthyol. 2006, 5, 139–163. [Google Scholar]
- Roy, D.; Paterson, G.; Hamilton, P.B.; Heath, D.D.; Haffner, G.D. Resource-based adaptive divergence in the freshwater fish Telmatherina from Lake Matano, Indonesia. Mol. Ecol. 2007, 16, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Salzburger, W.; Van Bocxlaer, B.; Cohen, A.S. Ecology and evolution of the African great lakes and their faunas. Annu. Rev. Ecol. Evol. Syst. 2014, 445, 519–545. [Google Scholar] [CrossRef]
- Fryer, G.; Iles, T.D. The Cichlid Fishes of the Great Lakes of Africa: Their Biology and Evolution; Oliver & Boyd: Edinburgh, UK, 1972; pp. 1–641. ISBN 0050023470. [Google Scholar]
- Salzburger, W.; Meyer, A. The species flocks of East African cichlid fishes: Recent advances in molecular phylogenetics and population genetics. Naturwissenschaften 2004, 91, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Jovanovska, E.; Cvetkoska, A.; Hauffe, T.; Levkov, Z.; Wagner, B.; Sulpizio, R.; Francke, A.; Albrecht, C.; Wilke, T. Differential resilience of ancient sister lakes Ohrid and Prespa to environmental disturbances during the Late Pleistocene. Biogeosciences 2016, 13, 1149–1161. [Google Scholar] [CrossRef]
- Pavlov, A.; Levkov, Z.; Williams, D.M.; Edlund, M.B. Observations on Hippodonta (Bacillariophyceae) in selected ancient lakes. Phytotaxa 2013, 90, 1–53. [Google Scholar] [CrossRef]
- Schmieder, K. European lake shores in danger concepts for a sustainable development. Limnologica 2004, 34, 3–14. [Google Scholar] [CrossRef]
- Schindler, D.E.; Scheurell, M. Habitat coupling in lake ecosystems. Oikos 2002, 98, 177–189. [Google Scholar] [CrossRef]
- Beeton, A.M. Large freshwater lakes: Present state, trends and future. Environ. Conserv. 2002, 29, 21–38. [Google Scholar] [CrossRef]
- Albrecht, C.; Wilke, T. Ancient Lake Ohrid: Biodiversity and evolution. Hydrobiologia 2008, 615, 103–140. [Google Scholar] [CrossRef]
- Hsieh, C.H.; Ishikawa, K.; Sakai, Y.; Ishikawa, T.; Ichise, S.; Yamamoto, Y.; Kuo, T.C.; Park, H.D.; Yamamura, N.; Kumagai, M. Phytoplankton community reorganization driven by eutrophication and warming in Lake Biwa. Aquat. Sci. 2010, 72, 467–483. [Google Scholar] [CrossRef]
- Ochumba, P.B.O.; Kibaara, D.I. Observations on blue-green algal blooms in the open waters of Lake Victoria, Kenya. Afr. J. Ecol. 1989, 27, 23–34. [Google Scholar] [CrossRef]
- Hecky, R.E. The eutrophication of Lake Victoria. Verh. Int. Ver. Theor. Angew. Limnol. 1993, 25, 39–48. [Google Scholar] [CrossRef]
- Lazzaro, X.; Gamarra Peralta, C. Fucionamiento limnológico y fotobiologia del Lago Titicaca. In Línea Base De Conocimientos Sobre Los Recursos Hidrológicos e Hidrobiológicos en El Sistema TDPS Con Enfoque en La Cuenca Del Lago Titicaca; Pouilly, M., Lazzaro, X., Point, D., Aguirre, M., Eds.; IRD—UICN: Quito, Ecuador, 2014; pp. 155–217. ISBN 9789997441843. [Google Scholar]
- Kobanova, G.I.; Takhteev, V.V.; Rusanovskaya, O.O.; Timofeyev, M.A. Lake Baikal Ecosystem Faces the Threat of Eutrophication. Acta Oecol. Int. J. Ecol. 2016, 6058082. [Google Scholar] [CrossRef]
- Cohen, A.S.; Gergurich, E.L.; Kraemer, B.M.; McGlue, M.M.; McIntyre, P.B.; Russell, J.M.; Simmons, J.D.; Swarzenski, P.W. Climate warming reduces fish production and benthic habitat in Lake Tanganyika, one of the most biodiverse freshwater ecosystems. Proc. Natl. Acad. Sci. USA 2016, 113, 9563–9568. [Google Scholar] [CrossRef]
- Everett, G.V. The rainbow trout Salmo gairdneri (Rich.) fishery of Lake Titicaca. J. Fish Biol. 1973, 5, 429–440. [Google Scholar] [CrossRef]
- Parenti, L.R. A taxonomic revision of the Andean Killifish Genus Orestias (Cyprinodontiformes, Cyprinodontidae). Bull. Am. Mus. Nat. Hist. 1984, 178, 107–214. [Google Scholar]
- Barel, C.D.N.; Dorit, R.; Greenwood, P.H.; Fryer, G.; Hughes, N.; Jackson, P.B.N.; Kawanabe, H.; Lowe-McConnell, R.H.; Nagoshi, M.; Ribbink, A.J.; et al. Destruction of fisheries in Africa’s lakes. Nature 1985, 315, 19–20. [Google Scholar] [CrossRef]
- Herder, F.; Nolte, A.W.; Pfaender, J.; Schwarzer, J.; Hadiaty, R.K.; Schliewen, U.K. Adaptive radiation and hybridization in Wallace’s dreamponds: Evidence from sailfin silversides in the Malili lakes of Sulawesi. Proc. R. Soc. B 2006, 273, 2209–2217. [Google Scholar] [CrossRef]
- Yin, X.; Nicholson, S.E. The water balance of Lake Victoria. Hydrol. Sci. J. 1998, 43, 789–811. [Google Scholar] [CrossRef]
- Pearce, F. Uganda pulls plug on Lake Victoria. N. Sci. 2006, 2538, 12. [Google Scholar]
- Hassan, A.A.; Jin, S. Lake level change and total water discharge in East Africa Rift Valley from satellite-based observations. Glob. Planet. Chang. 2014, 117, 79–90. [Google Scholar] [CrossRef]
- Peter, H.; Sommaruga, R. Alpine glacier-fed turbid lakes are discontinuous cold polymictic rather than dimictic. Inland Waters 2017, 7, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Tiberti, R.; Buscaglia, F.; Callieri, C.; Rogora, M.; Tartari, G.; Sommaruga, R. Food web complexity of high-mountain lakes is largely affected by glacial retreat. Ecosystems 2019. [Google Scholar] [CrossRef]
- Catalan, J.; Rondón, J.C.D. Perspective for an integrated understanding of tropical and temperate high-mountain lakes. J. Limnol. 2016, 75, 215–234. [Google Scholar] [CrossRef]
- Tiberti, R.; Buscaglia, F.; Armodi, M.; Callieri, C.; Ribelli, F.; Rogora, M.; Tartari, G.; Bocca, M. Mountain lakes of Mont Avic Natural Park: Ecological features and conservation issues. J. Limnol. 2019. [Google Scholar] [CrossRef]
- Hayford, B.L.; Caires, A.M.; Chandra, S. A Tale of Three Lakes: A Comparison of Biodiversity between Lake Tahoe and Crater Lake, USA, and Lake Hövsgöl, Mongolia. Mt. Views 2016, 10, 37–42. [Google Scholar]
- Tolotti, M.; Thies, H.; Cantonati, M.; Tait, D. Flagellate algae (Cryptophyceae, Chrysophyceae, Dinophyceae) in 48 high mountain lakes of the Northern and Southern slope of the Eastern Alps: Diversity, distribution and driving variables. Hydrobiologia 2003, 502, 331–348. [Google Scholar] [CrossRef]
- Kammerlander, B.; Breiner, H.W.; Filker, S.; Sommaruga, R.; Sonntag, B.; Stoeck, T. High diversity of protistan plankton communities in remote high mountain lakes in the European Alps and the Himalayan mountains. FEMS Microbiol. Ecol. 2015, 91. [Google Scholar] [CrossRef]
- Carrillo, P.; Medina-Sánchez, J.M.; Villar-Argaiz, M.; Delgado-Molina, J.A.; Bullejos, F.J. Complex interactions in microbial food webs: Stoichiometric and functional approaches. Limnetica 2006, 25, 189–204. [Google Scholar] [CrossRef]
- Cantonati, M.; Guella, G.; Komárek, J.; Spitale, D. Depth-distribution of epilithic cyanobacteria and pigments in a mountain lake characterized by marked water-level fluctuations. Freshw. Sci. 2014, 33, 537–547. [Google Scholar] [CrossRef]
- Sommaruga-Wögrath, S.; Koinig, K.A.; Schmidt, R.; Sommaruga, R.; Tessadri, R.; Psenner, R. Temperature effects on the acidity of remote alpine lakes. Nature 1997, 387, 64–67. [Google Scholar] [CrossRef]
- Catalán, J.; Ballesteros, E.; Gacia, E.; Palau, A.; Camarero, L. Chemical composition of disturbed and undisturbed high-mountain lakes in the Pyrenees: A reference for acidified sites. Wat. Res. 1993, 27, 133–141. [Google Scholar] [CrossRef]
- Leira, M.; Fillippi, M.L.; Cantonati, M. Diatom community response to extreme water-level fluctuations in two Alpine lakes: A core case study. J. Paleolimnol. 2015, 53, 289–307. [Google Scholar] [CrossRef]
- Tiberti, R.; Splendiani, A. Management of a highly unlikely native fish: The case of arctic charr Salvelinus alpinus from the Southern Alps. Aquatic Conserv. Mar. Freshw. Ecosyst. 2019, 29, 312–320. [Google Scholar] [CrossRef]
- Baron, J. The Fate of Mountain Lakes. Mt. Views 2016, 10, 2–4. [Google Scholar]
- Camarero, L.; Catalan, J.; Boggero, A.; Marchetto, A.; Mosello, R.; Psenner, R. Acidification in high mountain lakes in Central, Southwest and Southeast Europe (Alps, Pyrenees, Pirin). Limnologica 1995, 25, 141–156. [Google Scholar]
- Tait, D.; Thaler, B. Atmospheric deposition and lake chemistry trends at a high mountain site in the eastern Alps. J. Limnol. 2000, 59, 61–71. [Google Scholar] [CrossRef]
- Thies, H.; Nickus, U.; Mair, V.; Tessadri, R.; Tait, D.; Thaler, B.; Psenner, R. Unexpected response of high alpine lake waters to climate warming. Environ. Sci. Technol. 2007, 41, 7424–7429. [Google Scholar] [CrossRef]
- Marnezy, A. Alpine dams. J. Alp. Res. 2008, 96, 103–112. [Google Scholar] [CrossRef]
- Spitale, D.; Angeli, N.; Lencioni, V.; Tolotti, M.; Cantonati, M. Comparison between natural and impacted Alpine lakes six years after hydropower exploitation has ceased. Biologia 2015, 70, 1597–1605. [Google Scholar] [CrossRef]
- Margalef, R. Limnología; Editorial Omega: Barcelona, Spain, 1983; ISBN 8428207143. [Google Scholar]
- Baker, J.A.; Killgore, K.J.; Kasul, R.L. Aquatic habitats and fish communities in the lower Mississippi River. Rev. Aquat. Sci. 1991, 3, 213–356. [Google Scholar]
- Welcomme, R.L. Fisheries Ecology of Floodplain Rivers; Longman: London, UK; New York, NY, USA, 1979; ISBN 0582463106. [Google Scholar]
- Constantine, J.A.; Dunne, T.; Piegay, H.; Kandolf, G.M. Controls on the alluviation of oxbow lakes by bed-material load along the Sacramento River, California. Sedimentology 2010, 57, 389–407. [Google Scholar] [CrossRef]
- Ward, J.V. Riverine landscapes: Biodiversity patterns, disturbance regimes, and aquatic conservation. Biol. Conserv. 1998, 83, 269–278. [Google Scholar] [CrossRef]
- Piégay, H.; Bornette, G.; Citterio, A.; Hérouin, E.; Moulin, B.; Statiotis, C. Channel instability as control factor of silting dynamics and vegetation pattern within perifluvial aquatic zones. Hydrol. Process. 2000, 14, 3011–3029. [Google Scholar] [CrossRef]
- Schramm, H.L., Jr.; Eggleton, M.A.; Minnis, R.B. Spatial Analysis of Floodplain Habitat Critical to Lower Mississippi River Fishes; Mississippi Cooperative Fish and Wildlife Research Unit: Mississippi State, MS, USA, 1999. [Google Scholar]
- Dembowska, E.A.; Kubiak-Wojcicka, K. Influence of water level fluctuations on phytoplankton communities in an oxbow lake. Fundam. Appl. Limnol. 2017, 190, 221–233. [Google Scholar] [CrossRef]
- Naus, C.J.; Reid Adams, S. Fish nursery habitat function of the main channel, floodplain tributaries and oxbow lakes of a medium-sized river. Ecol. Freshw. Fish 2018, 27, 4–18. [Google Scholar] [CrossRef]
- Zeug, S.C.; Winemiller, K.O. Relationships between hydrology, spatial heterogeneity, and fish recruitment dynamics in a temperate floodplain river. River Res. Appl. 2007, 24, 90–102. [Google Scholar] [CrossRef]
- Bolpagni, R.; Piotti, A. Hydro-hygrophilous vegetation diversity and distribution patterns in riverine wetlands in an agricultural landscape: A case study from the Oglio River (Po Plain, Northern Italy). Phytocoenologia 2015, 45, 69–84. [Google Scholar] [CrossRef]
- Bolpagni, R.; Piotti, A. The importance of being natural in a human-altered riverscape: Role of wetland type in supporting habitat heterogeneity and the functional diversity of vegetation. Aquat. Conserv. Mar. Freshw. Ecosys. 2016, 26, 1168–1183. [Google Scholar] [CrossRef]
- Pierobon, E.; Bolpagni, R.; Bartoli, M.; Viaroli, P. Net primary production and seasonal CO2 and CH4 fluxes in a Trapa natans L. meadow. J. Limnol. 2010, 69, 225–234. [Google Scholar] [CrossRef]
- Bolpagni, R.; Pierobon, E.; Longhi, D.; Nizzoli, D.; Bartoli, M.; Tomaselli, M.; Viaroli, P. Diurnal exchanges of CO2 and CH4 across the water-atmosphere interface in a water chestnut meadow (Trapa natans L.). Aquat. Bot. 2007, 87, 43–48. [Google Scholar] [CrossRef]
- Bolpagni, R.; Bartoli, M.; Viaroli, P. Species and functional plant diversity in a heavily impacted riverscape: Implications for threatened hydro-hygrophilous flora conservation. Limnologica 2013, 43, 230–238. [Google Scholar] [CrossRef]
- Racchetti, E.; Bartoli, M.; Soana, E.; Longhi, D.; Christian, R.R.; Pinardi, M.; Viaroli, P. Influence of hydrological connectivity of riverine wetlands on nitrogen removal via denitrification. Biogeochemistry 2011, 103, 335–354. [Google Scholar] [CrossRef]
- Bolpagni, R.; Laini, A.; Mutti, T.; Viaroli, P.; Bartoli, M. Connectivity and habitat typology drive CO2 and CH4 fluxes across land–water interfaces in lowland rivers. Aquat. Sci. 2019, 12, e2036. [Google Scholar] [CrossRef]
- Gore, J.A.; Shields, F.D., Jr. Can large rivers be restored? Bioscience 1995, 45, 142–152. [Google Scholar] [CrossRef]
- Miranda, L.E. Fish Assemblages in Oxbow Lakes Relative to Connectivity with the Mississippi River. T. Am. Fish. Soc. 2011, 134, 1480–1489. [Google Scholar] [CrossRef]
- Pander, J.; Mueller, M.; Geist, J. Habitat diversity and connectivity govern the conservation value of restored aquatic floodplain habitats. Biol. Conserv. 2019, 217, 1–10. [Google Scholar] [CrossRef]
- Lehner, B.; Liermann, C.R.; Revenga, C.; Vörösmarty, C.; Fekete, B.; Crouzet, P.; Döll, P.; Endejan, M.; Frenken, K.; Magome, J.; et al. High-resolution mapping of the world’s reservoirs and dams for sustainable river-flow management. Front. Ecol. Environ. 2011, 9, 494–502. [Google Scholar] [CrossRef]
- Thornton, K.W. Perspectives on reservoir limnology. In Reservoir Limnology: Ecological Perspectives; Thornton, K.W., Kimmel, B.L., Payne, F.E., Eds.; John Wiley & Sons: New York, NY, USA, 1990; pp. 1–13. ISBN 978-0471885016. [Google Scholar]
- Wetzel, R.G. Limnology. Lake and River Ecosystems, 3rd ed.; Academic Press: San Diego, CA, USA, 2001; ISBN 9780127447605. [Google Scholar]
- Cronk, J.K.; Fennessy, M.S. Wetland Plants, Biology and Ecology; CRC Press: Boca Raton, FL, USA, 2001; pp. 1–462. ISBN 9781566703727. [Google Scholar]
- Rørslett, B. An integrated approach to hydropower impact assesment. II. Submerged macrophytes in some Norwegian hydroelectric lakes. Hydrobiologia 1989, 175, 65–82. [Google Scholar] [CrossRef]
- Krolová, M.; Čížková, H.; Hejzlar, J.; Poláková, S. Response of littoral macrophytes to water level fluctuations in a storage reservoir. Knowl. Manag. Aquat. Ec. 2013, 408, 7. [Google Scholar] [CrossRef]
- Mjelde, M.; Hellsten, S.; Ecke, F. A water level drawdown index for aquatic macrophytes in Nordic lakes. Hydrobiologia 2013, 70, 141–151. [Google Scholar] [CrossRef]
- Wilcox, D.A.; Meeker, J.E. Implications for faunal habitat related to altered macrophyte structure in regulated lakes in northern Minnesota. Wetlands 1992, 12, 192–203. [Google Scholar] [CrossRef]
- Poff, N.L.; Olden, J.D.; Merritt, D.M.; Pepin, D.M. Homogenization of regional river dynamics by dams and global biodiversity implications. Proc. Natl. Acad. Sci. USA 2007, 104, 5732–5737. [Google Scholar] [CrossRef] [PubMed]
- Thomaz, S.M.; Cunha, E.R. The role of macrophytes in habitat structuring in aquatic ecosystems: Methods of measurement, causes and consequences on animal assemblages’ composition and biodiversity. Acta Limnol. Bras. 2010, 22, 218–236. [Google Scholar] [CrossRef]
- Krolová, M.; Čížková, H.; Hejzlar, J. Depth limit of littoral vegetation in a storage reservoir: A case study of Lipno Reservoir (Czech Republic). Limnologica 2012, 42, 164–174. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Caraco, N.F.; Correll, D.L.; Howarth, R.W.; Sharpley, A.N.; Smith, V.H. Nonpoint pollution of surface waters with phosphorus and nitrogen. Ecol. Appl. 1998, 8, 559–568. [Google Scholar] [CrossRef]
- Mainstone, C.P.; Parr, W. Phosphorus in Rivers—Ecology and Management. Sci. Total Environ. 2002, 282–283, 25–47. [Google Scholar] [CrossRef]
- Smith, V.H. Eutrophication of freshwater and coastal marine ecosystems—A global problem. Environ. Sci. Pollut. R. 2003, 10, 126–139. [Google Scholar] [CrossRef]
- Hilton, J.; O’Hare, M.; Bowes, M.J.; Jones, J.I. How green is my river? A new paradigm of eutrophication in rivers. Sci. Total Environ. 2006, 365, 66–83. [Google Scholar] [CrossRef]
- Moss, B. Ecology of Freshwaters, Man and Medium, Past to Future, 3rd ed.; Blackwell Science: Oxford, UK, 1998; ISBN 978-1-444-31342-0. [Google Scholar]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Kubečka, J.; Seďa, J.; Duncan, A.; Matěna, J.; Ketelaars, H. Composition and biomass of the fish stocks in various european reservoirs and ecological consequences. Int. Rev. Hydrobiol. 1998, 83, 559–568. [Google Scholar]
- Persson, L.; Greenberg, L.A. Juvenile competitive bottlenecks: The perch (Perca fluviatilis)—Roach (Rutilus rutilus) interaction. Ecology 1990, 71, 44–56. [Google Scholar] [CrossRef]
- Šmejkal, M.; Ricard, D.; Sajdlová, Z.; Čech, M.; Vejřík, L.; Blabolil, P.; Vejříková, I.; Prchalová, M.; Vašek, M.; Souza, A.T.; et al. Can species-specific prey responses to chemical cues explain prey susceptibility to predation? Ecol. Evol. 2018, 8, 4544–4551. [Google Scholar] [CrossRef] [PubMed]
- Poikane, S.; Ritterbusch, D.; Argillier, C.; Białokoz, W.; Blabolil, P.; Breine, J.; Jaarsma, N.G.; Krause, T.; Kubečka, J.; Lauridsen, T.L.; et al. Response of fish communities to multiple pressures: Development of a total anthropogenic pressure intensity index. Sci. Total Environ. 2017, 586, 502–511. [Google Scholar] [CrossRef]
- Draštík, V.; Kubečka, J.; Šovčík, P. Hydrology and angler’s catches in the Czech reservoirs. Ecohydrol. Hydrobiol. 2004, 4, 429–439. [Google Scholar]
- Duncan, A.; Kubečka, J. Land/water ecotone effects in reservoirs on the fish fauna. Hydrobiologia 1995, 303, 11–30. [Google Scholar] [CrossRef]
- Clavero, M.; Hermoso, V.; Aparicio, E.; Godinho, F.N. Biodiversity in heavily modified waterbodies: Native and introduced fish in Iberian reservoirs. Freshw. Biol. 2013, 58, 1190–1201. [Google Scholar] [CrossRef]
- Catford, J.A.; Vesk, P.A.; Richardson, D.M.; Pyšek, P. Quantifying levels of biological invasion: Towards the objective classification of invaded and invasible ecosystems. Glob. Chang. Biol. 2012, 18, 44–62. [Google Scholar] [CrossRef]
- Byers, J.E. Impact of non-indigenous species on natives enhanced by anthropogenic alteration of selection regimes. Oikos 2002, 97, 449–458. [Google Scholar] [CrossRef]
- Pyšek, P.; Richardson, D.M. Invasive species, environmental change and management, and health. Annu. Rev. Env. Resour. 2010, 35, 25–55. [Google Scholar] [CrossRef]
- Johnson, P.T.J.; Olden, J.D.; Vander Zanden, M.J. Dam Invaders: Impoundments facilitate biological invasions into freshwaters. Front. Ecol. Environ. 2008, 6, 357–363. [Google Scholar] [CrossRef]
- Clavero, M.; Hermoso, V. Reservoirs promote the taxonomic homogenization of fish communities within river basins. Biodivers. Conserv. 2011, 20, 41–57. [Google Scholar] [CrossRef]
- Villamagna, A.M.; Murphy, B.R. Ecological and socio-economic impacts of invasive water hyacinth (Eichhornia crassipes): A review. Freshw. Biol. 2010, 55, 282–298. [Google Scholar] [CrossRef]
- Madsen, J.D. Predicting invasion success of Eurasian watermilfoil. J. Aquat. Plant Manag. 1998, 36, 28–32. [Google Scholar]
- Van Zuidam, J.P.; Peeters, E.T.H.M. Occurrence of macrophyte monocultures in drainage ditches relates to phosphorus in both sediment and water. Springer Plus 2013, 2, 564. [Google Scholar] [CrossRef]
- Verdonschot, R.C.M.; Keizer-Vlek, H.E.; Verdonschot, P.F.M. Biodiversity value of agricultural drainage ditches; a comparative analysis of the aquatic invertebrate fauna of ditches and small lakes. Aquat. Conserv. Mar. Freshw. Ecosyst. 2011, 21, 715–727. [Google Scholar] [CrossRef]
- Francis, R.A. Urban rivers: Novel ecosystems, new challenges. WIREs Water 2014, 1, 19–29. [Google Scholar] [CrossRef]
- Booth, D.B.; Roy, A.H.; Smith, B.; Capps, K.A. Global perspectives on the urban stream syndrome. Freshw. Sci. 2016, 35, 412–420. [Google Scholar] [CrossRef]
- Gál, B.; Szivák, I.; Heino, J.; Schmera, D. The effect of urbanization on freshwater macroinvertebrates—Knowledge gaps and future research directions. Ecol. Ind. 2019, 104, 357–364. [Google Scholar] [CrossRef]
- Higgins, S.L.; Thomas, F.; Goldsmith, B.; Brooks, S.J.; Hassall, C.; Harlow, J.; Stone, D.; Völker, S.; White, P. Urban freshwaters, biodiversity, and human health and well-being: Setting an interdisciplinary research agenda. Wiley Interdiscip. Rev. Water 2019, 6, e1339. [Google Scholar] [CrossRef]
- Dearborn, D.C.; Kark, S. Motivations for conserving urban biodiversity. Conserv. Biol. 2019, 24, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Joosten, H.; Clarke, D. Wise Use of Mires and Peatlands. A Framework for Decision-Making; International Mire Conservation Group: Greifswald, Germany; International Peat Society: Jyväskylä, Finland; Saarijärven Offset Oy: Saarijärvi, Finland, 2002; ISBN 951-97744-8-3. [Google Scholar]
- Hájek, M.; Horsák, M.; Hájková, P.; Dítě, D. Habitat diversity of central European fens in relation to environmental gradients and an effort to standardise fen terminology in ecological studies. Perspect. Plant Ecol. 2006, 8, 97–114. [Google Scholar] [CrossRef]
- Dierssen, K.; Dierssen, B. Moore; Ökosysteme Mitteleuropas aus Geobotanischer Sicht: Stuttgart, Germany, 2001; pp. 1–230. ISBN 9783800156436. [Google Scholar]
- Rydin, H.; Jeglum, J.K. The Biology of Peatlands; Oxford University Press: Oxford, UK, 2013; pp. 1–432. ISBN 978-0199603008. [Google Scholar]
- Vitt, D.H. Peatlands: Ecosystems dominated by bryophytes. In Bryophyte Biology; Shaw, A.J., Goffinet, B., Eds.; Cambridge University Press: Cambridge, UK, 2000; pp. 312–343. ISBN 9780511754807. [Google Scholar]
- Wheeler, B.D.; Proctor, M.C.F. Ecological gradients, subdivisions and terminology of north-west European mires. J. Ecol. 2000, 88, 187–203. [Google Scholar] [CrossRef]
- Proctor, M.C.; McHaffie, H.S.; Legg, C.J.; Amphlett, A. Evidence from water chemistry as a criterion of ombrotrophy in the mire complexes of Abernethy Forest, Scotland. J. Veg. Sci. 2009, 20, 160–169. [Google Scholar] [CrossRef]
- Jiroušek, M.; Poulíčková, A.; Kintrová, K.; Opravilová, V.; Hájková, P.; Rybníček, K.; Kočí, M.; Bergová, K.; Hnilica, R.; Mikulášková, E.; et al. Long-term and contemporary environmental conditions as determinants of the species composition of bog organisms. Freshw. Biol. 2013, 58, 2196–2207. [Google Scholar] [CrossRef]
- Lindsay, R. Peatland classification. In The Wetland Book: I: Structure and Function, Management, and Methods; Finlayson, C.M., Everard, M., Irvine, K., McInnes, R.J., Middleton, B.A., van Dam, A.A., Davidson, N.C., Eds.; Springer: Berlin, Germany, 2018; pp. 1515–1528. ISBN 978-94-007-1471-7. [Google Scholar]
- Peterka, T.; Hájek, M.; Jiroušek, M.; Jiménez-Alfaro, B.; Aunina, L.; Bergamini, A.; Dítě, D.; Felbaba-Klushyna, L.; Graf, U.; Hájková, P.; et al. Formalized classification of European fen vegetation at the alliance level. Appl. Veg. Sci. 2017, 20, 124–142. [Google Scholar] [CrossRef]
- Singh, P.; Těšitel, J.; Plesková, Z.; Peterka, T.; Dítě, D.; Hájková, P.; Pawlikowski, P.; Hájek, M. The ratio between bryophyte functional groups impacts vascular plants in rich fens. Appl. Veg. Sci. 2019. [Google Scholar] [CrossRef]
- Horsák, M. Mollusc community patterns and species response curves along a mineral richness gradient: A case study in fens. J. Biogeogr. 2006, 33, 98–107. [Google Scholar] [CrossRef]
- Rozbrojová, Z.; Hájek, M. Changes in nutrient limitation of spring fen vegetation along environmental gradients in the West Carpathians. J. Veg. Sci. 2008, 19, 613–620. [Google Scholar] [CrossRef]
- Vicherová, E.; Hájek, M.; Hájek, T. Calcium intolerance of fen mosses: Physiological evidence, effects of nutrient availability and successional drivers. Perspect. Plant Ecol. 2015, 17, 347–359. [Google Scholar] [CrossRef]
- Bain, J.T.; Proctor, M.C.F. The requirement of aquatic bryophytes for free CO2 as an inorganic carbon source: Some experimental evidence. N. Phytol. 1980, 86, 393–400. [Google Scholar] [CrossRef]
- Patberg, W.; Baaijens, G.J.; Smolders, A.J.; Grootjans, A.P.; Elzenga, J.T.M. The importance of groundwater carbon dioxide in the restoration of small Sphagnum bogs. Preslia 2013, 85, 389–403. [Google Scholar]
- Hájek, M.; Jiroušek, M.; Navrátilová, J.; Horodyská, E.; Peterka, T.; Plesková, Z.; Navrátil, Z.; Hájková, P.; Hájek, T. Changes in the moss layer in Czech fens indicate early succession triggered by nutrient enrichment. Preslia 2015, 87, 279–301. [Google Scholar]
- Hájková, P.; Hájek, M. Species richness and above-ground biomass of poor and calcareous spring fens in the flysch West Carpathians, and their relationships to water and soil chemistry. Preslia 2003, 75, 271–287. [Google Scholar]
- Horsáková, V.; Hájek, M.; Hájková, P.; Dítě, D.; Horsák, M. Principal factors controlling the species richness of European fens differ between habitat specialists and matrix-derived species. Divers. Distrib. 2018, 24, 742–754. [Google Scholar] [CrossRef]
- Dítě, D.; Hájek, M.; Svitková, I.; Košuthová, A.; Šoltés, R.; Kliment, J. Glacial-relict symptoms in the Western Carpathian flora. Folia Geobot. 2018, 53, 277–300. [Google Scholar] [CrossRef]
- Hájková, P.; Štechová, T.; Šoltés, R.; Šmerdová, E.; Plesková, Z.; Dítě, D.; Bradáčová, J.; Mútňanová, M.; Singh, P.; Hájek, M. Using a new database of plant macrofossils of the Czech and Slovak Republics to compare past and present distribution of hypothetically relict fen mosses. Preslia 2018, 90, 367–386. [Google Scholar] [CrossRef]
- Hájek, M.; Horsák, M.; Tichý, L.; Hájková, P.; Dítě, D.; Jamrichová, E. Testing a relict distributional pattern of fen plant and terrestrial snail species at the Holocene scale: A null model approach. J. Biogeogr. 2011, 38, 742–755. [Google Scholar] [CrossRef]
- Horsák, M.; Hájek, M.; Spitale, D.; Hájková, P.; Dítě, D.; Nekola, J.C. The age of island-like habitats impacts habitat specialist species richness. Ecology 2012, 93, 1106–1114. [Google Scholar] [CrossRef]
- Warner, B.G.; Asada, T. Biological diversity of peatlands in Canada. Aquat. Sci. 2006, 68, 240–253. [Google Scholar] [CrossRef]
- Middleton, B.A.; Holsten, B.; van Diggelen, R. Biodiversity management of fens and fen meadows by grazing, cutting and burning. Appl. Veg. Sci. 2006, 9, 307–316. [Google Scholar] [CrossRef]
- Van Diggelen, R.; Middleton, B.; Bakker, J.; Grootjans, A.; Wassen, M. Fens and floodplains of the temperate zone: Present status, threats, conservation and restoration. Appl. Veg. Sci. 2006, 9, 157–162. [Google Scholar] [CrossRef]
- Soomers, H.; Karssenberg, D.; Verhoeven, J.T.; Verweij, P.A.; Wassen, M.J. The effect of habitat fragmentation and abiotic factors on fen plant occurrence. Biodiver. Conserv. 2013, 22, 405–424. [Google Scholar] [CrossRef]
- Buczkó, K. Bryophytic diatoms from Hungary. In Proceedings of the Eighteenth International Diatom Symposium, Miedzyzdroje, Poland, 2–7 September 2004; Witkowski, A., Ed.; Biopress Limited: Bristol, UK, 2006; Volume 2006, pp. 1–15. [Google Scholar]
- Berendse, F.; Breemen, N.; Van Rydin, H.; Buttler, A.; Heijmans, M.; Hoosbeek, M.R.; Lee, J.A.; Mitchell, E.; Saarinen, T.; Vasander, H.; et al. Raised atmospheric CO2 levels and increased N deposition cause shifts in plant species composition and production in Sphagnum bogs. Glob. Chang. Biol. 2001, 7, 591–598. [Google Scholar] [CrossRef]
- Charman, D. Peatlands and Environmental Change; Wiley: Hoboken, NJ, USA, 2002; pp. 1–301. ISBN 978-0-470-84410-6. [Google Scholar]
- Essl, F.; Dullinger, S.; Moser, D.; Rabitsch, W.; Kleinbauer, I. Vulnerability of mires under climate change: Implications for nature conservation and climate change adaptation. Biodivers. Conserv. 2012, 21, 655–669. [Google Scholar] [CrossRef]
- Moradi, H.; Fakheran, S.; Peintinger, M.; Bergamini, A.; Schmid, B.; Joshi, J. Profiteers of environmental change in the Swiss Alps: Increase of thermophilous and generalist plants in wetland ecosystems within the last 10 years. Alp. Bot. 2012, 122, 45–56. [Google Scholar] [CrossRef]
- Herrera-Pantoja, M.; Hiscock, K.M.; Boar, R.R. The potential impact of climate change on groundwater-fed wetlands in eastern England. Ecohydrology 2012, 5, 401–413. [Google Scholar] [CrossRef]
- Jiménez-Alfaro, B.; García-Calvo, L.; García, P.; Acebes, J.L. Anticipating extinctions of glacial relict populations in mountain refugia. Biol. Conserv. 2016, 201, 243–251. [Google Scholar] [CrossRef]
- Page, S.E.; Baird, A.J. Peatlands and global change: Response and resilience. Annu. Rev. Env. Resour. 2016, 41, 35–57. [Google Scholar] [CrossRef]
- Janssen, J.A.M.; Rodwell, J.S.; Criado, M.G.; Arts, G.H.P.; Bijlsma, R.J.; Schaminee, J.H.J. European Red List of Habitats: Part 2. Terrestrial and Freshwater Habitats; Directorate-General for the Environment: Brussels, Belgium, 2016; ISBN 978-92-79-61588-7. [Google Scholar]
- Grootjans, A.P.; Adema, E.B.; Bleuten, W.; Joosten, H.; Madaras, M.; Janáková, M. Hydrological landscape settings of base-rich fen mires and fen meadows: An overview. Appl. Veg. Sci. 2006, 9, 175–184. [Google Scholar] [CrossRef]
- Vicherová, E.; Hájek, M.; Šmilauer, P.; Hájek, T. Sphagnum establishment in alkaline fens: Importance of weather and water chemistry. Sci. Total Environ. 2017, 580, 1429–1438. [Google Scholar] [CrossRef] [PubMed]
- Hájková, P.; Horsák, M.; Hájek, M.; Jankovská, V.; Jamrichová, E.; Moutelíková, J. Using multi-proxy palaeoecology to test a relict status of refugial populations of calcareous-fen species in the Western Carpathians. Holocene 2015, 25, 702–715. [Google Scholar] [CrossRef]
- Bart, D.; Davenport, T.; Yantes, A. Environmental predictors of woody plant encroachment in calcareous fens are modified by biotic and abiotic land-use legacies. J. Appl. Ecol. 2016, 53, 541–549. [Google Scholar] [CrossRef]
- Jamrichová, E.; Gálová, A.; Gašpar, A.; Horsák, M.; Frodlová, J.; Hájek, M.; Hajnalová, M.; Hájková, P. Holocene development of two calcareous spring fens at the Carpathian-Pannonian interface controlled by climate and human impact. Folia Geobot. 2018, 53, 243–263. [Google Scholar] [CrossRef]
- Koch, M.; Jurasinski, G. Four decades of vegetation development in a percolation mire complex following intensive drainage and abandonment. Plant Ecol. Divers. 2015, 8, 49–60. [Google Scholar] [CrossRef]
- Kapfer, J.; Grytnes, J.A.; Gunnarsson, U.; Birks, H.J.B. Fine-scale changes in vegetation composition in a boreal mire over 50 years. J. Ecol. 2011, 99, 1179–1189. [Google Scholar] [CrossRef]
- Pedrotti, E.; Rydin, H.; Ingmar, T.; Hytteborn, H.; Turunen, P.; Granath, G. Fine-scale dynamics and community stability in boreal peatlands: Revisiting a fen and a bog in Sweden after 50 years. Ecosphere 2014, 5, 1–24. [Google Scholar] [CrossRef]
- Seer, F.K.; Schrautzer, J. Status, future prospects, and management recommendations for alkaline fens in an agricultural landscape: A comprehensive survey. J. Nat. Conserv. 2014, 22, 358–368. [Google Scholar] [CrossRef]
- Merriam, K.E.; Markwith, S.H.; Coppoletta, M. Livestock exclusion alters plant species composition in fen meadows. Appl. Veg. Sci. 2018, 21, 3–11. [Google Scholar] [CrossRef]
- Ross, L.C.; Speed, J.D.; Øien, D.I.; Grygoruk, M.; Hassel, K.; Lyngstad, A.; Moen, A. Can mowing restore boreal rich-fen vegetation in the face of climate change? PLoS ONE 2019, 14, e0211272. [Google Scholar] [CrossRef]
- Poulíčková, A.; Hájková, P.; Kintrová, K.; Bat’ková, R.; Czudková, M.; Hájek, M. Tracing decadal environmental change in ombrotrophic bogs using diatoms from herbarium collections and transfer functions. Environ. Pollut. 2013, 179, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Paal, J.; Vellak, K.; Liira, J.; Karofeld, E. Bog recovery in northeastern Estonia after the reduction of atmospheric pollutant input. Restor. Ecol. 2010, 18, 387–400. [Google Scholar] [CrossRef]
- Friberg, N.; Dybkjaer, J.B.; Olafsson, J.S.; Gislason, G.M.; Larsen, S.E.; Lauridsen, T.L. Relationships between structure and function in streams contrasting in temperature. Freshw. Biol. 2009, 54, 2051–2068. [Google Scholar] [CrossRef]
- Gerdol, R.; Petraglia, A.; Bragazza, L.; Iacumin, P.; Brancaleoni, L. Nitrogen deposition interacts with climate in affecting production and decomposition rates in Sphagnum mosses. Glob. Chang. Biol. 2007, 13, 1810–1821. [Google Scholar] [CrossRef]
- Bragazza, L.; Siffi, C.; Iacumin, P.; Gerdol, R. Mass loss and nutrient release during litter decay in peatland: The role of microbial adaptability to litter chemistry. Soil Biol. Biochem. 2007, 39, 257–267. [Google Scholar] [CrossRef]
- Neuhäusl, R. Hochmoore am Teich Velké Dářko; Academia-Verlag: Praha, Czech Republic, 1975; pp. 1–120. [Google Scholar]
- Nørgaard, E. Environment and behaviour of Theridion saxatile. Oikos 1956, 7, 159–192. [Google Scholar] [CrossRef]
- Horsák, M.; Polášková, V.; Zhai, M.; Bojková, J.; Syrovátka, V.; Šorfová, V.; Schenková, J.; Polášek, M.; Peterka, T.; Hájek, M. Spring-fen habitat islands in a warming climate: Partitioning the effects of mesoclimate air and water temperature on aquatic and terrestrial biota. Sci. Total Environ. 2018, 634, 355–365. [Google Scholar] [CrossRef]
- Fernández-Pascual, E.; Jiménez-Alfaro, B.; Hájek, M.; Díaz, T.E.; Pritchard, H.W. Soil thermal buffer and regeneration niche may favour calcareous fen resilience to climate change. Folia Geobot. 2015, 50, 293–301. [Google Scholar] [CrossRef]
- Seymour, K.S.; Christanis, K.; Bouzinos, A.; Papazisimou, S.; Papatheodorou, G.; Moran, E.; Dénès, G. Tephrostratigraphy and tephrochronology in the Philippi peat basin, Macedonia, Northern Hellas (Greece). Quatern. Int. 2004, 121, 53–65. [Google Scholar] [CrossRef]
- Hájek, M.; Hájková, P.; Apostolova, I.; Horsák, M.; Plášek, V.; Shaw, B.; Lazarova, M. Disjunct occurrences of plant species in the refugial mires of Bulgaria. Folia Geobot. 2009, 44, 365–386. [Google Scholar] [CrossRef]
- Vukojičić, S.; Jakovljević, K.; Matevski, V.; Randjelović, V.; Niketić, M.; Lakušić, D. Distribution, Diversity and Conservation of Boreo-Montane Plant Species in the Central Part of the Balkan Peninsula and the Southern Part of the Pannonian Plain. Folia Geobot. 2014, 49, 487–505. [Google Scholar] [CrossRef]
- Hájková, P.; Hájek, M.; Apostolova, I.; Zelený, D.; Dítě, D. Shifts in the ecological behaviour of plant species between two distant regions: Evidence from the base richness gradient in mires. J. Biogeogr. 2008, 35, 282–294. [Google Scholar] [CrossRef]
- Theodoridis, S.; Nogués-Bravo, D.; Conti, E. The role of cryptic diversity and its environmental correlates in global conservation status assessments: Insights from the threatened bird’s-eye primrose (Primula farinosa L.). Divers. Distrib. 2019, 25, 1457–1471. [Google Scholar] [CrossRef]
- Cantonati, M.; Lange-Bertalot, H.; Decet, F.; Gabrieli, J. Diatoms in very-shallow pools of the site of community importance Danta di Cadore Mires (south-eastern Alps), and the potential contribution of these habitats to diatom biodiversity conservation. Nova Hedwig. 2011, 93, 475–507. [Google Scholar] [CrossRef]
- Cantonati, M.; Van de Vijver, B.; Lange-Bertalot, H. Microfissurata gen. nov. (Bacillariophyta), a new diatom genus from dystrophic and intermittently-wet terrestrial habitats. J. Phycol. 2009, 45, 732–741. [Google Scholar] [CrossRef]
- Turetsky, M.R.; Bond-Lamberty, B.; Euskirchen, E.; Talbot, J.; Frolking, S.; McGuire, A.D.; Tuittila, E.S. The resilience and functional role of moss in boreal and arctic ecosystems. N. Phytol. 2012, 196, 49–67. [Google Scholar] [CrossRef]
- Rosset, V.; Angelibert, S.; Arthaud, F.; Bornette, G.; Robin, J.; Wezel, A.; Vallod, D.; Oertli, B. Is eutrophication really a major impairment for small waterbody biodiversity? J. Appl. Ecol. 2014, 51, 415–425. [Google Scholar] [CrossRef]
- Sharpley, A.N.; Bergström, L.; Aronsson, H.; Bechmann, M.; Bolster, C.H.; Börling, K.; Djodjic, F.; Jarvie, H.P.; Schoumans, O.F.; Stamm, C.; et al. Future agriculture with minimized phosphorus losses to waters: Research needs and direction. Ambio 2015, 44, 163–179. [Google Scholar] [CrossRef]
- Wetzel, R.G. Land water interfaces: Metabolic and limnological regulators. Verh. Int. Ver. Limnol. 1990, 24, 6–24. [Google Scholar] [CrossRef]
- Schiemer, F.; Zalewski, M.; Thorpe, J.E. Land/Inland water ecotones: Intermediate habitats critical for conservation and management. Hydrobiologia 1995, 303, 259–264. [Google Scholar] [CrossRef]
- Hill, M.J.; Ryves, D.B.; White, J.C.; Wood, P.J. Macroinvertebrate diversity in urban and rural ponds: Implications for freshwater biodiversity conservation. Biol. Conserv. 2016, 201, 50–59. [Google Scholar] [CrossRef]
- Bagella, S.; Gascón, S.; Filigheddu, R.; Cogoni, A.; Boix, D. Mediterranean Temporary Ponds: New challenges from a neglected habitat. Hydrobiologia 2016, 782, 1–10. [Google Scholar] [CrossRef]
- Hill, M.J.; Wood, P.J. The macroinvertebrate biodiversity and conservation value of garden and field ponds along a rural-urban gradient. Fund. Appl. Limnol. 2014, 185, 107–119. [Google Scholar] [CrossRef]
- Bazzanti, M. Pond macroinvertebrates of the Presidential Estate of Castelporziano (Rome): A review of ecological aspects and selecting indicator taxa for conservation. Rend. Lincei 2015, 26, 337–343. [Google Scholar] [CrossRef]
- Rosset, V.; Simaika, J.P.; Arthaud, F.; Bornette, G.; Vallod, D.; Samways, M.J.; Oertli, B. Comparative assessment of scoring methods to evaluate the conservation value of pond and small lake biodiversity. Aquat. Conserv. Mar. Freshw. Ecosys. 2013, 23, 23–36. [Google Scholar] [CrossRef]
- Fuentes-Rodríguez, F.; Juan, M.; Gallego, I.; Lusi, M.; Fenoy, E.; León, D.; Peñalver, P.; Toja, J.; Casas, J.J. Diversity in Mediterranean farm ponds: Trade-offs and synergies between irrigation modernisation and biodiversity conservation. Freshw. Biol. 2013, 58, 63–78. [Google Scholar] [CrossRef]
- Tóth, A.; Horváth, Z.; Vad, C.F.; Zsuga, K.; Nagy, S.A.; Boros, E. Zooplankton of the european soda pans: Fauna and conservation of a unique habitat type. Int. Rev. Hydrobiol. 2014, 99, 255–276. [Google Scholar] [CrossRef]
- Briers, R.A. Invertebrate communities and environmental conditions in a series of urban drainage ponds in eastern Scotland: Implications for biodiversity and conservation value of SUDS. Clean 2014, 42, 193–200. [Google Scholar] [CrossRef]
- Williams, P.; Whitfield, M.; Biggs, J.; Bray, S.; Fox, G.; Nicolet, P.; Sear, D. Comparative biodiversity of rivers, streams, ditches and ponds in an agricultural landscape in Southern England. Biol. Conserv. 2003, 115, 329–341. [Google Scholar] [CrossRef]
- Hunter, M.L., Jr.; Acuña, V.; Bauer, D.M.; Bell, K.P.; Calhoun, A.J.K.; Felipe-Lucia, M.R.; Fitzsimons, J.A.; González, E.; Kinnison, M.; Lindenmayer, D.; et al. Conserving small natural features with large ecological roles: A synthetic overview. Biol. Conserv. 2017, 211, 88–95. [Google Scholar] [CrossRef]
- Maynou, X.; Martín, R.; Aranda, D. The role of small secondary biotopes in a highly fragmented landscape as habitat and connectivity providers for dragonflies (Insecta: Odonata). J. Insect Conserv. 2017, 21, 517–530. [Google Scholar] [CrossRef]
- Hill, M.J.; Heino, J.; White, J.C.; Ryves, D.B.; Wood, P.J. Environmental factors are primary determinants of different facets of pond macroinvertebrate alpha and beta diversity in a human-modified landscape. Biol. Conserv. 2019, 237, 348–357. [Google Scholar] [CrossRef]
- Oertli, B.; Parris, K.M. Review: Toward management of urban ponds for freshwater biodiversity. Ecosphere 2019, 10, e02810. [Google Scholar] [CrossRef]
- Johansson, F.; Bini, L.M.; Coiffard, P.; Svanbäck, R.; Wester, J.; Heino, J. Environmental variables drive differences in the beta diversity of dragonfly assemblages among urban stormwater ponds. Ecol. Indic. 2019, 106, 105529. [Google Scholar] [CrossRef]
- Hill, M.J.; Biggs, J.; Thornhill, I.; Briers, R.A.; Gledhill, D.G.; White, J.C.; Wood, P.J.; Hassall, C. Urban ponds as an aquatic biodiversity resource in modified landscapes. Glob. Chang. Biol. 2017, 23, 986–999. [Google Scholar] [CrossRef]
- Pielou, E.C. After the Ice Age: The Return of Life to Glaciated North America; University of Chicago Press: Chicago, IL, USA, 1991; ISBN 978-0226668123. [Google Scholar]
- Wrona, F.J.; Reist, J.D. Freshwater ecosystems. In Arctic Biodiversity Assessment. Status and Trends in Arctic Biodiversity; Meltofte, H., Ed.; Conservation of Arctic Flora and Fauna, Arctic Council: Akureyri, Iceland, 2013; pp. 335–377. ISBN 978-9935-431-22-6. [Google Scholar]
- Lento, J.; Goedkoop, W.; Culp, J.; Christoffersen, K.; Fefilova, E.; Guðbergsson, G.; Liljaniemi, P.; Ólafsson, J.S.; Sandøy, S.; Zimmerman, C.; et al. Circumpolar Biodiversity Monitoring Program State of Arctic Freshwater Biodiversity Report; Conservation of Arctic Flora and Fauna, Arctic Council: Akureyri, Iceland, 2019; ISBN 978-9935-431-77-6. [Google Scholar]
- Christiansen, J.S.; Reist, J.D.; Brown, R.J.; Brykov, V.A.; Christensen, G.; Christoffersen, K.; Cott, P.; Crane, P.; Dempson, J.B.; Docker, M.; et al. Fishes. Conservation of Arctic Flora and Fauna. In Arctic Biodiversity Assessment. Status and Trends in Arctic Biodiversity; Meltofte, H., Ed.; Arctic Council: Akureyri, Iceland, 2013; pp. 192–245. ISBN 978-9935-431-22-6. [Google Scholar]
- Jyrkänkallio-Mikkola, J.; Meier, S.; Heino, J.; Laamanen, T.; Pajunen, V.; Tolonen, K.T.; Tolkkinen, M.; Soininen, J. Disentangling multi-scale environmental effects on stream microbial communities. J. Biogeogr. 2017, 44, 1512–1523. [Google Scholar] [CrossRef]
- Soininen, J.; Heino, J.; Kokocinski, M.; Muotka, T. Local-regional diversity relationship varies with spatial scale in lotic diatoms. J. Biogeogr. 2009, 36, 720–727. [Google Scholar] [CrossRef]
- Alahuhta, J. Geographic patterns of lake macrophyte communities and species richness at regional scale. J. Veg. Sci. 2015, 26, 564–575. [Google Scholar] [CrossRef]
- Heino, J.; Tolonen, K.T. Untangling the assembly of littoral macroinvertebrate communities through measures of functional and phylogenetic alpha diversity. Freshw. Biol. 2017, 62, 1168–1179. [Google Scholar] [CrossRef]
- Rocha, M.P.; Bini, L.M.; Domisch, S.; Tolonen, K.T.; Jyrkänkallio-Mikkola, J.; Soininen, J.; Hjort, J.; Heino, J. Local environment and space drive multiple facets of stream macroinvertebrate beta diversity. J. Biogeogr. 2018, 45, 2744–2754. [Google Scholar] [CrossRef]
- Heino, J.; Tolonen, K.T. Ecological drivers of multiple facets of beta diversity in a lentic macroinvertebrate metacommunity. Limnol. Oceanogr. 2017, 62, 2431–2444. [Google Scholar] [CrossRef]
- Erős, T.; Heino, J.; Schmera, D.; Rask, M. Characterising functional trait diversity and trait-environment relationships in fish assemblages of boreal lakes. Freshw. Biol. 2009, 54, 1788–1803. [Google Scholar] [CrossRef]
- Sandin, L. Benthic macroinvertebrates in Swedish streams: Community structure, taxon richness, and environmental relations. Ecography 2003, 26, 269–282. [Google Scholar] [CrossRef]
- Tolonen, K.E.; Leinonen, K.; Marttila, H.; Erkinaro, J.; Heino, J. Environmental predictability of taxonomic and functional community composition in high-latitude streams. Freshw. Biol. 2017, 62, 1–16. [Google Scholar] [CrossRef]
- Johnson, R.K.; Goedkoop, W.; Sandin, L. Spatial scale and ecological relationships between the macroinvertebrate communities of stony habitats of streams and lakes. Freshw. Biol. 2004, 49, 1179–1194. [Google Scholar] [CrossRef]
- Soininen, J.; Kokocinski, M.; Estlander, S.; Kotanen, J.; Heino, J. Neutrality, niches and determinants of plankton metacommunity structure across boreal wetland ponds. Ecoscience 2007, 14, 146–154. [Google Scholar] [CrossRef]
- Grönroos, M.; Heino, J.; Siqueira, T.; Landeiro, V.L.; Kotanen, J.; Bini, L.M. Metacommunity structuring in stream networks: Roles of dispersal mode, distance type and regional environmental context. Ecol. Evol. 2013, 3, 4473–4487. [Google Scholar] [CrossRef]
- Heino, J.; Muotka, T.; Paavola, R. Determinants of macroinvertebrate diversity in headwater streams: Regional and local influences. J. Anim. Ecol. 2003, 72, 425–434. [Google Scholar] [CrossRef]
- Pajunen, V.; Luoto, M.; Soininen, J. Stream diatom assemblages as predictors of climate. Freshw. Biol. 2016, 61, 876–886. [Google Scholar] [CrossRef]
- Culp, J.; Lento, J.; Curry, R.A.; Luiker, E.; Halliwell, D. Arctic biodiversity of stream macroinvertebrates declines in response to latitudinal change in the abiotic template. Freshw. Sci. 2019. [Google Scholar] [CrossRef]
- Toivanen, M.; Hjort, J.; Heino, J.; Tukiainen, H.; Aroviita, J.; Alahuhta, J. Is catchment geodiversity a useful surrogate of aquatic plant species richness? J. Biogeogr. 2019, 46, 1–12. [Google Scholar] [CrossRef]
- Heino, J.; Virkkala, R.; Toivonen, H. Climate change and freshwater biodiversity: Detected patterns, future trends and adaptations in northern regions. Biol. Rev. 2009, 84, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Alahuhta, J.; Heino, J.; Luoto, M. Climate change and the future distribution of aquatic macrophytes across boreal catchments. J. Biogeogr. 2011, 38, 383–393. [Google Scholar] [CrossRef]
- Hildrew, A.G. Freshwater Acidification: Natural History, Ecology and Environmental Policy; International Ecology Institute: Oldendorf, Germany, 2018; ISBN 9783946729273. [Google Scholar]
- Murray, J. British Antarctic Expedition 1907–9, under the Command of Sir E.H. Shackleton. Reports on the Scientific Investigations. Volume 1 Biology; Willian Heinemann: London, UK, 1910; p. 105. [Google Scholar] [CrossRef]
- Laybourn-Parry, J.; Wadham, J.L. Antarctic Lakes; Oxford University Press: Oxford, UK, 2014; pp. 1–215. ISBN 978-0199670505. [Google Scholar]
- Roberts, D.; McMinn, A. A diatom-based palaeosalinity history of Ace Lake, Vestfold Hills, Antarctica. Holocene 1999, 9, 401–408. [Google Scholar] [CrossRef]
- Hawes, I.; Howard-Williams, C.; Fountain, A. Ice-Based freshwater ecosystems. In Polar Lakes and Rivers—Arctic and Antarctic Aquatic Ecosystems; Vincent, W.F., Laybourn-Parry, J., Eds.; Oxford University Press: Oxford, UK, 2008; pp. 103–118. [Google Scholar] [CrossRef]
- Siegert, M.J. A 60-year international history of Antarctic subglacial lake exploration. In Exploration of Subsurface Antarctica: Uncovering Past Changes and Modern Processes; Siegert, M.J., Jamieson, S.S.R., White, D.A., Eds.; Geological Society: London, UK, 2018; Volume 461, pp. 7–22. [Google Scholar] [CrossRef]
- Christner, B.C.; Priscu, J.C.; Achberger, A.M.; Barbante, C.; Carter, S.P.; Christianson, K.; Michaud, A.B.; Mikucki, J.A.; Mitchell, A.C.; Skidmore, M.L.; et al. A microbial ecosystem beneath the West Antarctic ice sheet. Nature 2014, 512, 310–313. [Google Scholar] [CrossRef]
- Howard-Williams, C.; Hawes, I.; Doran, P.; Siegert, M.; Camacho, A.; Kaup, E. Diversity of Antarctic lakes, ponds and streams. Antarctic Environments Portal, Information Summaries. 2019. Available online: https://www.environments.aq/information-summaries/diversity-of-antarctic-lakes-ponds-and-streams/ (accessed on 15 January 2020).
- Obryk, M.K.; Doran, P.T.; Priscu, J.C. Prediction of ice-free conditions for a perennially ice-covered Antarctic Lake. J. Geophys. Res. Earth Surf. 2019, 124, 686–694. [Google Scholar] [CrossRef]
- Quayle, W.C.; Peck, L.S.; Peat, H.; Ellis-Evans, J.C.; Harrigan, P.R. Extreme responses to climate change in Antarctic lakes. Science 2002, 295, 645. [Google Scholar] [CrossRef]
- Gibson, J.A.; Bayly, I.A.E. New insights into the origins of crustaceans of Antarctic lakes. Antarct. Sci. 2007, 19, 157–163. [Google Scholar] [CrossRef]
- Vyverman, W.; Verleyen, E.; Wilmotte, A.; Hodgson, D.A.; Willems, A.; Peeters, K.; Van de Vijver, B.; De Wever, A.; Leliaert, F.; Sabbe, K. Evidence for widespread endemism among Antarctic microorganisms. Polar Sci. 2008, 4, 103–113. [Google Scholar] [CrossRef]
- Pessi, I.G.; Lara, Y.; Durieu, B.; Maalouf, P.D.C.; Verleyen, E.; Wilmotte, A. Community structure and distribution of benthic cyanobacteria in Antarctic lacustrine microbial mats. FEMS Microbiol. Ecol. 2018, 94. [Google Scholar] [CrossRef]
- Jung, P.; Briegel-Williams, L.; Schermer, M.; Büdel, B. Strong in combination: Polyphasic approach enhances arguments for cold-assigned cyanobacterial endemism. MicrobiologyOpen 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Strunecky, O.; Elster, J.; Komárek, J. Molecular clock evidence for survival of Antarctic cyanobacteria (Oscillatoriales, Phormidium autumnale) from Paleozoic times. FEMS Microbiol. Ecol. 2012, 82, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Taton, A.; Grubisic, S.; Balthazart, P.; Hodgson, D.A.; Laybourn-Parry, J.; Wilmotte, A. Biogeographical distribution and ecological range of benthic cyanobacteria in East Antarctic lakes. FEMS Microbiol. Ecol. 2006, 57, 272–289. [Google Scholar] [CrossRef] [PubMed]
- Broady, P.A. Diversity, distribution and dispersal of Antarctic terrestrial algae. Biodiv. Conserv. 1996, 5, 1307–1335. [Google Scholar] [CrossRef]
- Verleyen, E.; Vyverman, W.; Sterken, M.; Hodgson, D.A.; De Wever, A.; Juggins, S.; Van de Vijver, B.; Jones, V.J.; Vanormelingen, P.; Roberts, D.; et al. The importance of dispersal related and local factors in shaping the taxonomic structure of diatom metacommunities. Oikos 2009, 118, 1239–1249. [Google Scholar] [CrossRef]
- Karanovic, T.; Gibson, J.; Hawes, I.; Andersen, D.; Stevens, M. Three new species of Diacyclops (Copepods: Cyclopoida) from continental Antarctica. Antarct. Sci. 2014, 26, 250–260. [Google Scholar] [CrossRef]
- Chown, S.L.; Huiskes, A.H.L.; Gremmen, N.J.M.; Lee, J.E.; Terauds, A.; Crosbie, K.; Frenot, Y.; Hughes, K.A.; Imura, S.; Kiefer, K.; et al. Continent-wide risk assessment for the establishment of nonindigenous species in Antarctica. Proc. Natl. Acad. Sci. USA 2012, 109, 4938–4943. [Google Scholar] [CrossRef]
- Li, S.-P.; Ochyra, R.; Wu, P.-C.; Seppelt, R.D.; Cai, M.-H.; Wang, H.-Y.; Li, C.-S. Drepanocladus longifolius (Amblystegiaceae), an addition to the moss flora of King George Island, South Shetland Islands, with a review of Antarctic benthic mosses. Polar Biol. 2009, 32, 1415–1425. [Google Scholar] [CrossRef]
- Velasco-Castrillón, A.; Gibson, J.A.; Stevens, M.I. A review of current Antarctic limno-terrestrial microfauna. Polar Biol. 2014, 37, 1517–1531. [Google Scholar] [CrossRef]
- Fountain, A.G.; Levy, J.S.; Goosefff, M.N.; Van Horn, D. The McMurdo Dry Valleys: a landscape on the threshold of change. Geomorphology 2014, 225, 25–35. [Google Scholar] [CrossRef]
- Hawes, I.; Sumner, D.Y.; Andersen, D.T.; Mackey, T.J. Legacies of recent environmental change in the benthic communities of Lake Joyce, a perennially ice covered, Antarctic lake. Geobiology 2011, 9, 394–410. [Google Scholar] [CrossRef] [PubMed]
- Jungblut, A.D.; Hawes, I. Using Captain Scott’s Discovery specimens to unlock the past: Has Antarctic cyanobacterial diversity changed over the last 100 years? Proc. Biol. Sci. 2017, 284. [Google Scholar] [CrossRef] [PubMed]
- Naselli-Flores, L. Mediterranean climate and eutrophication of reservoirs: Limnological skills to improve management. In Eutrophication: Causes, Consequences and Control; Ansari, A., Singh, G.S., Lanza, G., Rast, W., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 131–142. [Google Scholar] [CrossRef]
- Bonada, N.; Resh, V.H. Mediterranean-climate streams and rivers: Geographically separated but ecologically comparable freshwater systems. Hydrobiologia 2013, 719, 1–29. [Google Scholar] [CrossRef]
- Cid, N.; Bonada, N.; Carlson, S.M.; Grantham, T.E.; Gasith, A.; Resh, V.H. High Variability Is a Defining Component of Mediterranean-Climate Rivers and Their Biota. Water 2017, 9, 52. [Google Scholar] [CrossRef]
- Carrizo, S.F.; Lengyel, S.; Kapusi, F.; Szabolcs, M.; Kasperidus, H.D.; Scholz, M.; Markovic, D.; Freyhof, J.; Cid, N.; Cardoso, A.C.; et al. Critical catchments for freshwater biodiversity conservation in Europe: identification, prioritisation and gap analysis. J. Appl. Ecol. 2017, 54, 1209–1218. [Google Scholar] [CrossRef]
- Naselli-Flores, L. Man-made lakes in Mediterranean semi-arid climate: The strange case of Dr Deep Lake and Mr Shallow Lake. Hydrobiologia 2003, 506, 13–21. [Google Scholar] [CrossRef]
- Ferreira, M.; Beja, P. Mediterranean amphibians and the loss of temporary ponds: Are there alternative breeding habitats? Biol. Cons. 2013, 165, 179–186. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Marrone, F. Different invasibility of permanent and temporary water bodies in a semi-arid Mediterranean island. Inland Waters 2019, 9, 411–421. [Google Scholar] [CrossRef]
- Barone, R.; Castelli, G.; Naselli-Flores, L. Red sky at night cyanobacteria delight: The role of climate in structuring phytoplankton assemblages in a shallow Mediterranean lake (Biviere di Gela, southeastern Sicily). Hydrobiologia 2010, 639, 43–53. [Google Scholar] [CrossRef]
- Tockner, K.; Ward, J.V.; Arscott, D.B.; Edwards, P.J.; Kollmann, J.; Gurnell, A.M.; Petts, G.E.; Maiolini, B. The Tagliamento River: A model ecosystem of European importance. Aquat. Sci. 2003, 65, 239–253. [Google Scholar] [CrossRef]
- Gallart, F.; Cid, N.; Latron, J.; Llorens, P.; Bonada, N.; Jeuffroy, J.; Jiménez-Argudo, S.-M.; Vega, R.-M.; Solà, C.; Soria, M.; et al. TREHS: An open-access software tool for investigating and evaluating temporary river regimes as a first step for their ecological status assessment. Sci. Total Environ. 2017, 607–608, 519–540. [Google Scholar] [CrossRef] [PubMed]
- Tonkin, J.D.; Bogan, M.T.; Bonada, N.; Rios-Touma, B.; Lytle, D.A. Seasonality and predictability shape temporal species diversity. Ecology 2017, 98, 1201–1216. [Google Scholar] [CrossRef] [PubMed]
- Sabater, S.; Timoner, X.; Borrego, C.; Acuña, V. Stream biofilm responses to flow intermittency: From cells to ecosystems. Front. Environ. Sci. 2016, 4, 1–10. [Google Scholar] [CrossRef]
- Hershkovitz, Y.; Gasith, A. Resistance, resilience, and community dynamics in mediterranean-climate streams. Hydrobiologia 2012, 719, 59–75. [Google Scholar] [CrossRef]
- Bonada, N.; Dolédec, S.; Statzner, B. Taxonomic and biological trait differences of stream macroinvertebrate communities between mediterranean and temperate regions: Implications for future climatic scenarios. Glob. Chang. Biol. 2007, 13, 1658–1671. [Google Scholar] [CrossRef]
- Kerezsy, A.; Gido, K.; Magalhaes, M.F.; Skelton, P.H. The biota of intermittent rivers and ephemeral streams: Fishes. In Intermittent Rivers and Ephemeral Streams; Datry, T., Bonada, N., Boulton, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 273–298. [Google Scholar] [CrossRef]
- Múrria, C.; Bonada, N.; Vellend, M.; Zamora-Muñoz, C.; Alba-Tercedor, J.; Sainz-Cantero, C.E.; Garrido, J.; Acosta, R.; El Alami, M.; Barquín, J.; et al. Local environment rather than past climate determines community composition of mountain stream macroinvertebrates across Europe. Mol. Ecol. 2017, 26, 6085–6099. [Google Scholar] [CrossRef]
- Soria, M.; Leigh, C.; Datry, T.; Bini, L.M.; Bonada, N. Biodiversity in perennial and intermittent rivers: A meta-analysis. Oikos 2017, 126, 1078–1089. [Google Scholar] [CrossRef]
- Soria, M.; Gutiérrez-Cánovas, C.; Bonada, N.; Acosta, R.; Rodríguez-Lozano, P.; Fortuño, P.; Burgazzi, G.; Vinyoles, D.; Gallart, F.; Latron, J.; et al. Natural disturbances can produce misleading bioassessment results: Identifying metrics to detect anthropogenic impacts in intermittent rivers. J. Appl. Ecol. 2019, in press. [Google Scholar] [CrossRef]
- Munné, A.; Prat, N. Effects of Mediterranean climate annual variability on stream biological quality assessment using macroinvertebrate communities. Ecol. Indic. 2011, 11, 651–662. [Google Scholar] [CrossRef]
- Fritz, K.; Cid, N.; Autrey, B. Governance, Legislation, and Protection of Intermittent Rivers and Ephemeral Streams. In Intermittent Rivers and Ephemeral Streams; Datry, T., Bonada, N., Boulton, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 477–507. [Google Scholar] [CrossRef]
- Millán, A.; Velasco, J.; Gutiérrez-Cánovas, C.; Arribas, P.; Picazo, F.; Sánchez-Fernández, D.; Abellán, P. Mediterranean saline streams in southeast Spain: What do we know? J. Arid Environ. 2011, 75, 1352–1359. [Google Scholar] [CrossRef]
- Arribas, P.; Abellán, P.; Velasco, J.; Bilton, D.T.; Millán, A.; Sánchez-Fernández, D. Evaluating drivers of vulnerability to climate change: A guide for insect conservation strategies. Glob. Chang. Biol. 2012, 18, 2135–2146. [Google Scholar] [CrossRef]
- Döll, P.; Schmied, H.M. How is the impact of climate change on river flow regimes related to the impact on mean annual runoff? A global-scale analysis. Environ. Res. Lett. 2012, 7, 014037. [Google Scholar] [CrossRef]
- Datry, T.; Bonada, N.; Heino, J. Towards understanding the organisation of metacommunities in highly dynamic ecological systems. Oikos 2016, 125, 149–159. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Barone, R. Phytoplankton dynamics and structure: A comparative analysis in natural and man-made water bodies of different trophic state. Hydrobiologia 2000, 438, 65–74. [Google Scholar] [CrossRef]
- Jeppesen, E.; Brucet, S.; Naselli-Flores, L.; Papastergiadou, E.; Stefanidis, K.; Nõges, T.; Nõges, P.; Attayde, J.L.; Zohary, T.; Coppens, J.; et al. Ecological impacts of global warming and water abstraction on lakes and reservoirs due to change in water level and related changes in salinity. Hydrobiologia 2015, 750, 201–227. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Barone, R. Mixotrophic phytoplankton dynamics in a shallow Mediterranean water body: How to make a virtue out of necessity. Hydrobiologia 2019, 831, 33–41. [Google Scholar] [CrossRef]
- García-Berthou, E.; Moreno-Amich, R. Introduction of exotic fish into a Mediterranean lake over a 90-year period. Arch. Hydrobiol. 2000, 149, 271–284. [Google Scholar] [CrossRef]
- Gómez-Rodríguez, C.; Díaz-Paniagua, C.; Serrano, L.; Florencio, M.; Portheault, A. Mediterranean temporary ponds as amphibians breeding habitats: The importance of preserving pond networks. Aquat. Ecol. 2009, 43, 1179–1191. [Google Scholar] [CrossRef]
- Griffiths, R.A. Temporary ponds as amphibian habitat. Aquat. Conserv. Mar. Freshw. Ecosys. 1997, 7, 119–126. [Google Scholar] [CrossRef]
- Incagnone, G.; Marrone, F.; Barone, R.; Robba, L.; Naselli-Flores, L. How do freshwater organisms cross the “dry ocean”? A review on passive dispersal and colonization processes with a special focus on temporary ponds. Hydrobiologia 2015, 750, 103–123. [Google Scholar] [CrossRef]
- Boix, D.; Kneitel, J.; Robson, B.J.; Duchet, C.; Zúñiga, L.; Day, J.; Gascón, S.; Sala, J.; Quintana, X.D.; Blaustein, L. Invertebrates of temporary ponds in Mediterranean climates. In Invertebrates in Freshwater Wetlands. An International Perspective on Their Ecology; Batzer, D., Boix, D., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 141–189. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Termine, R.; Barone, R. Phytoplankton colonization patterns. Is species richness depending on distance among freshwaters and on their connectivity? Hydrobiologia 2016, 764, 103–113. [Google Scholar] [CrossRef]
- Stoch, F.; Korn, M.; Turki, S.; Naselli-Flores, L.; Marrone, F. The role of spatial environmental factors as determinants of large branchiopod distribution in Tunisian temporary ponds. Hydrobiologia 2016, 782, 37–51. [Google Scholar] [CrossRef]
- Sahuquillo, M.; Miracle, M.R. The role of historic and climatic factors in the distribution of crustacean communities in Iberian Mediterranean ponds. Freshw. Biol. 2013, 58, 1251–1266. [Google Scholar] [CrossRef]
- Downing, J.A. Emerging global role of small lakes and ponds: Little things mean a lot. Limnetica 2010, 29, 9–24. [Google Scholar]
- Bolgovics, A.; Viktoria, B; Várbíró, G.; Krasznai, E.A.; Ács, É.; Kiss, K.T.; Borics, G. Groups of small lakes maintain larger microalgal diversity than large ones. Sci. Total Environ. 2019, 678, 162–172. [Google Scholar] [CrossRef]
- Bagella, S.; Gascón, S.; Caria, M.C.; Sala, J.; Boix, D. Cross-taxon congruence in Mediterranean temporary wetlands: Vascular plants, crustaceans, and coleopterans. Community Ecol. 2011, 12, 40–50. [Google Scholar] [CrossRef]
- Marrone, F.; Alfonso, G.; Stoch, F.; Pieri, V.; Alonso, M.; Dretakis, M.; Naselli-Flores, L. An account on the non-malacostracan crustacean fauna from the inland waters of Crete, Greece, with the synonymization of Arctodiaptomus piliger Brehm, 1955 with Arctodiaptomus alpinus (Imhof, 1885). Limnetica 2019, 38, 167–187. [Google Scholar] [CrossRef]
- Marrone, F.; Barone, R.; Naselli-Flores, L. Cladocera (Branchiopoda: Anomopoda, Ctenopoda and Onychopoda) from Sicilian inland waters: An updated inventory. Crustaceana 2005, 78, 1025–1039. [Google Scholar] [CrossRef]
- Marrone, F.; Castelli, G.; Barone, R.; Naselli-Flores, L. Ecology and distribution of calanoid copepods in Sicilian inland waters (Italy). Verh. Internat. Verein. Limnol. 2006, 29, 2150–2156. [Google Scholar] [CrossRef]
- Bagella, S.; Caria, M.C. Diversity and ecological characteristics of vascular flora in Mediterranean temporary pools. C. R. Biol. 2012, 335, 69–76. [Google Scholar] [CrossRef]
- Sciandrello, S.; Privitera, M.; Puglisi, M.; Minissale, P. Diversity and spatial patterns of plant communities in volcanic temporary ponds of Sicily (Italy). Biologia 2016, 71, 793–803. [Google Scholar] [CrossRef]
- Cogoni, A.; Filippino, G.; Marignani, M. Small-scale pattern of bryoflora in Mediterranean temporary ponds: Hints for monitoring. Hydrobiologia 2016, 782, 81–95. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Barone, R. Phytoplankton dynamics in permanent and temporary Mediterranean waters: Is the game hard to play because of hydrological disturbance? Hydrobiologia 2012, 698, 147–159. [Google Scholar] [CrossRef]
- Hutchinson, G.E. Homage to Santa Rosalia or why are there so many kinds of animals? Am. Nat. 1959, 93, 145–159. [Google Scholar] [CrossRef]
- Zohary, T.; Yacobi, Y.Z.; Alster, A.; Fishbein, T.; Lippman, S.; Tibor, G. Phytoplankton. In Lake Kinneret: Ecology and Management; Chapter 10; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 161–190. [Google Scholar] [CrossRef]
- Nishri, A.; Leibovici, E. External sources of nutrients. In Lake Kinneret: Ecology and Management; Chapter 19; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 329–346. [Google Scholar] [CrossRef]
- Zohary, T.; Sukenik, A.; Berman, T.; Nishri, A. Lake Kinneret: Ecology and Management; Springer: Heidelberg, Germany, 2014; p. 683. [Google Scholar] [CrossRef]
- Ostrovsky, I.; Goren, M.; Shapiro, J. Fish Biology and Ecology. In Lake Kinneret: Ecology and Management; Chapter 16; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 273–292. [Google Scholar] [CrossRef]
- Tsurnamal, M. The biology and ecology of the blind prawn, Typhlocaris galilea Calman (Decapoda, Caridea). Crustaceana 1978, 34, 195–213. [Google Scholar] [CrossRef]
- Berman, T.; Zohary, T.; Nishri, A.; Sukenik, A. General background. In Lake Kinneret: Ecology and Management; Chapter 1; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 1–15. [Google Scholar] [CrossRef]
- Markel, D.; Shamir, U.; Green, P. Operational Management of Lake Kinneret and its watershed. In Lake Kinneret: Ecology and Management; Chapter 31; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 541–560. [Google Scholar] [CrossRef]
- Sukenik, A.; Zohary, T.; Markel, D. The monitoring program. In Lake Kinneret: Ecology and Management; Chapter 32; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 561–575. [Google Scholar] [CrossRef]
- Zohary, T.; Ostrovsky, I. Ecological impacts of excessive water level fluctuations in stratified freshwater lakes. Inland Water 2011, 1, 47–59. [Google Scholar] [CrossRef]
- Dreizin, Y.; Tenne, A.; Hoffman, D. Integrating large scale seawater desalination plants within Israel’s water supply system. Desalination 2008, 220, 132–149. [Google Scholar] [CrossRef]
- Rimmer, A.; Gal, G.; Opher, T.; Lechinsky, Y.; Yacobi, Y.Z. Mechanisms of long-term variations of the thermal structure in a warm lake. Limnol. Oceanogr. 2011, 56, 974–988. [Google Scholar] [CrossRef]
- Pollingher, U. Phytoplankton periodicity in a subtropical lake (Lake Kineret, Israel). Hydrobiologia 1986, 138, 127–138. [Google Scholar] [CrossRef]
- Zohary, T. Changes to the phytoplankton assemblage of Lake Kinneret after decades of a predictable, repetitive pattern. Freshw. Biol. 2004, 49, 1355–1371. [Google Scholar] [CrossRef]
- Zohary, T.; Sukenik, A.; Nishri, A. Overview. In Lake Kinneret: Ecology and Management; Chapter 37; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 657–671. [Google Scholar] [CrossRef]
- Sukenik, A.; Hadas, O.; Kaplan, A. Cyanobacteria. In Lake Kinneret: Ecology and Management; Chapter 12; Zohary, T., Sukenik, A., Berman, T., Nishri, A., Eds.; Springer: Heidelberg, Germany, 2014; pp. 213–226. [Google Scholar] [CrossRef]
- Zohary, T.; Alster, A.; Hadas, O.; Obertegger, U. There to stay: Opportunistic and versatile filamentous green alga Mougeotia in Lake Kinneret, Israel. Hydrobiologia 2019, 831, 87–100. [Google Scholar] [CrossRef]
- Heller, J.; Dolev, A.; Zohary, T.; Gal, G. Invasion dynamics of the snail Pseudoplotia scabra in Lake Kinneret. Biological Invasions 2014, 16, 7–12. [Google Scholar] [CrossRef]
- Moss, B. The kingdom of the shores: Achievement of good ecological potential in reservoirs. Freshw. Rev. 2008, 1, 29–42. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Barone, R. Water-level fluctuations in Mediterranean reservoirs: Setting a dewatering threshold as a management tool to improve water quality. Hydrobiologia 2005, 548, 85–99. [Google Scholar] [CrossRef]
- Naselli-Flores, L. Morphological analysis of phytoplankton as a tool to assess ecological state of aquatic ecosystems: The case of Lake Arancio, Sicily, Italy. Inland Waters 2014, 4, 15–26. [Google Scholar] [CrossRef]
- Gownaris, N.J.; Rountos, K.J.; Kaufman, L.; Kolding, J.; Lwiza, K.M.M.; Pikitch, E.K. Water level fluctuations and the ecosystem functioning of lakes. J. Great Lakes Res. 2018, 44, 1154–1163. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Barone, R. Steady-state assemblages in a Mediterranean hypertrophic reservoir. The role of Microcystis ecomorphological variability in maintaining an apparent equilibrium. Hydrobiologia 2003, 502, 133–143. [Google Scholar] [CrossRef]
- Moss, B. The Water Framework Directive: Total environment or political compromise? Sci. Total Environ. 2008, 400, 32–41. [Google Scholar] [CrossRef]
- Oertli, B. Editorial: Freshwater biodiversity conservation: The role of artificial ponds in the 21st century. Aquat. Conserv. Mar. Freshw. Ecosys. 2018, 28, 264–269. [Google Scholar] [CrossRef]
- Junk, W.; Bayley, P.B.; Sparks, R.E. The flood pulse concept in river-floodplain systems. Can. Spec. Publ. Fish. Aquat. Sci. 1989, 106, 110–127. [Google Scholar]
- Aselmann, I.; Crutzen, P.J. Global distribution of natural freshwater wetlands and rice paddies, their net primary productivity, seasonality and possible methane emissions. J. Atmos. Chem. 1989, 8, 307–358. [Google Scholar] [CrossRef]
- Fraser, L.H.; Keddy, P.A. The World’s Largest Wetlands: Ecology and Conservation. Ecoscience 2005, 13, 559–560. [Google Scholar] [CrossRef]
- Wantzen, K.M.; Junk, W.J. The importance of stream-wetland-systems for biodiversity: A tropical perspective. In Biodiversity in Wetlands: Assessment, Function and Conservation; Gopal, B., Junk, W.J., Davies, J.A., Eds.; Backhuys: Leiden, The Netherlands, 2000; pp. 11–34. ISBN 90-5782-059-5. [Google Scholar]
- Reis, V.; Hermoso, V.; Hamilton, S.K.; Bunn, S.E.; Fluet-Chouinard, E.; Venables, B.; Linke, S. Characterizing seasonal dynamics of Amazonian wetlands for conservation and decision making. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 29, 1073–1082. [Google Scholar] [CrossRef]
- Ramsar Convention Secretariat. The Ramsar Convention Manual: A Guide to the Convention on Wetlands (Ramsar, Iran, 1971), 6th ed.; Ramsar Convention Secretariat: Gland, Switzerland, 2013. [Google Scholar]
- Sieben, E.J.J.; Khubeka, S.P.; Sithole, S.; Job, N.M.; Kotze, D.C. The classification of wetlands: Integration of top-down and bottom-up approaches and their significance for ecosystem service determination. Wetl. Ecol. Manag. 2017, 26, 441–458. [Google Scholar] [CrossRef]
- Gumbricht, T.; Roman-Cuesta, R.M.; Verchot, L.; Herold, M.; Wittmann, F.; Householder, F.; Herold, N.; Murdiyarso, D. An expert system model for mapping tropical wetlands and peatlands reveals South America as the largest contributor. Glob. Chang. Biol. 2017, 23, 3581–3599. [Google Scholar] [CrossRef]
- Page, S.E.; Rieley, J.O.; Banks, C.J. Global and regional importance of the tropical peatland carbon pool. Glob. Chang. Biol. 2011, 17, 798–818. [Google Scholar] [CrossRef]
- Nilssen, J.P. Tropical lakes—Functional ecology and future development. The need for a process-orientated approach. Hydrobiologia 1984, 113, 231–242. [Google Scholar] [CrossRef]
- Jenkins, M.W.; McCord, S.; Edebe, J. Sustaining Lake Levels in Lake Nakuru, Kenya: Development of a Water Balance Mode for Decision Making. Research Brief 09-05-SUMAWA; University of California Global Livestock Collaborative Research Support Program: Davis, CA, USA, 2009; p. 4. [Google Scholar]
- Junk, W.J. Long-term environmental trends and the future of tropical wetlands. Environ. Conserv. 2002, 29, 414–435. [Google Scholar] [CrossRef]
- Gopal, B.; Junk, W.J.; Finlayson, C.M.; Breen, C.M. Present state and future of tropical wetlands. In Aquatic Ecosystems; Polunin, N., Ed.; Cambridge University Press: Cambridge, UK, 2008; pp. 142–154. ISBN 9780521833271. [Google Scholar]
- Oviedo, G.; Kenza, A.M. Indigenous Peoples, Local Communities and Wetland Conservation; Ramsar Convention Secretariat: Gland, Switzerland, 2018. [Google Scholar]
- Clarkson, R.B.E.; Ausseil, A.; Gerbeaux, P. Wetland ecosystem services. In Ecosystem Services in New Zealand; Dymond, J.R., Ed.; Manaaki Whenua Press: Lincoln, New Zealand, 2014. [Google Scholar]
- Mitsch, W.J.; Blanca Bernal, B.; Hernandez, M.E. Ecosystem services of wetlands. IJBESM 2015, 11, 1–4. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Knight, R.L. Treatment Wetlands; Lewis: New York, NY, USA, 1996; ISBN 978-1-56670-526-4. [Google Scholar]
- Mitsch, W.J.; Day, J.W., Jr.; Gilliam, J.W.; Groffman, P.M.; Hey, D.L.; Randall, G.W.; Wang, N. Reducing nitrogen loading to the Gulf of Mexico from the Mississippi River Basin: Strategies to counter a persistent ecological problem. BioScience 2001, 51, 373–388. [Google Scholar] [CrossRef]
- Day, J.W.; Boesch, D.F.; Clairain, E.J.; Kemp, G.P.; Laska, S.B.; Mitsch, W.J.; Orth, K.; Mashriqui, H.S.; Reed, D.J.; Shabman, L.A.; et al. Restoration of the Mississippi Delta: Lessons from Hurricanes Katrina and Rita. Science 2007, 315, 1679–1684. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.; Jackson, R. Hydrology of wetlands. In Freshwaters of New Zealand; Harding, J., Mosley, P., Pearson, C., Sorrell, B., Eds.; Caxton Press for the New Zealand Hydrological Society and New Zealand Limnological Society: Christchurch, New Zealand, 2004; pp. 1–20. ISBN 0-476-00708-9. [Google Scholar]
- Béné, C. Contribution of inland fisheries to rural livelihoods and food security in Africa: An overview. In Freshwater Ecoregions of Current State of Knowledge Regarding the World’s Wetlands 165 Africa and Madagascar: A Conservation Assessment; Thieme, M.L., Abell, R., Stiassny, M.L.J., Skelton, P., Eds.; Island Press: Washington, DC, USA, 2005; pp. 6–11. ISBN 9781559633659. [Google Scholar]
- Matthews, E.; Fung, I. Methane emissions from natural wetlands: Global distribution, area, and environmental characteristics of sources. Glob. Biogeochem. Cycles 1987, 1, 61–86. [Google Scholar] [CrossRef]
- Petrescu, A.M.; van Beek, L.; van Huissteden, J.; Prigent, C.; Sachs, T.; Corradi, C.; Parmentier, F.J.V.; Dolman, A.J. Modelling regional to global CH4 emissions from boreal and arctic wetlands. Glob. Biogeochem. Cycles 2010, 24, GB4009. [Google Scholar] [CrossRef]
- Petrescu, A.M.; Lohila, A.; Tuovinen, J.; Baldocchi, D.D.; Desai, A.R.; Roulet, N.T.; Vesala, T.; Dolman, A.J.; Oechel, W.C.; Marcolla, B.; et al. The uncertain climate footprint of wetlands under human pressure. Proc. Natl. Acad. Sci. USA 2015, 112, 4594–4599. [Google Scholar] [CrossRef] [PubMed]
- Lal, R. Carbon sequestration. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 815–830. [Google Scholar] [CrossRef] [PubMed]
- Lähteenoja, O.; Ruokoleinen, K.; Schulman, L.; Oinonen, M. Amazonian peatlands: An ignored C sink and potential source. Glob. Chang. Biol. 2009, 15, 2311–2320. [Google Scholar] [CrossRef]
- Sjögersten, S.; Black, C.R.; Evers, S.; Hoyos-Santillan, J.; Wright, E.L.; Turner, B.L. Tropical wetlands: A missing link in the global carbon cycle? Glob. Biogeochem. Cycles 2014, 28, 1371–1386. [Google Scholar] [CrossRef]
- Gumbricht, T. Mapping Global Tropical Wetlands from Earth Observing Satellite Imagery; Working Paper 103; CIFOR: Bogor, Indonesia, 2012. [Google Scholar] [CrossRef]
- Wohl, E.; Barros, A.; Brunsell, N.; Chappell, N. The hydrology of the humid tropics. Nat. Clim. Chang. 2012, 2, 655–662. [Google Scholar] [CrossRef]
- Castello, L.; Macedo, M.N. Large-scale degradation of Amazonian freshwater ecosystems. Glob. Chang. Biol. 2016, 22, 990–1007. [Google Scholar] [CrossRef]
- Mergou, F.E.; Lazaridou-Dimitriadou, M.; Albanakis, K. The effets of dams on rivers “continuum”. Conference paper. In Proceedings of the Protection and Restoration of the Environment XI, Thessaloniki, Greece, 3–6 July 2012. [Google Scholar]
- Childs, M. Literature Survey: The Impacts of Dams on River Channel Geomorphology; MSc research at The University of Hull; The University of Hull: Hull, UK, 2010. [Google Scholar]
- Winton, R.S.; Calamita, E.; Wehrli, B. Reviews and syntheses: Dams, water quality and tropical reservoir stratification. Biogeosciences 2019, 16, 1657–1671. [Google Scholar] [CrossRef]
- Zarfl, C.; Lumsdon, A.E.; Berlekamp, J.; Tydecks, L.; Tockner, K. A global boom in hydropower dam construction. Aquat. Sci. 2014, 77, 161–170. [Google Scholar] [CrossRef]
- Anderson, E.P.; Jenkins, C.N.; Heilpern, S.; Maldonado-Ocampo, J.A.; Carvajal-Vallejos, F.M.; Encalada, A.C.; Rivadeneira, J.F.; Hidalgo, M.; Cañas, C.M.; Ortega, H.; et al. Fragmentation of Andes-to-Amazon connectivity by hydropower dams. Sci. Adv. 2018, 4. [Google Scholar] [CrossRef] [PubMed]
- Lees, A.C.; Peres, C.A.; Fearnside, P.M.; Schneider, M.; Zuanon, J.A.S. Hydropower and the future of Amazonian biodiversity. Biodivers. Conserv. 2016, 25, 451–466. [Google Scholar] [CrossRef]
- Winemiller, K.O.; McIntyre, P.B.; Castello, L.; Fluet-Chouinard, E.; Giarrizzo, T.; Nam, S.; Baird, I.G.; Darwall, W.; Lujan, N.K.; Harrison, I.; et al. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science 2016, 351, 128–129. [Google Scholar] [CrossRef]
- Roberts, T.R. On the river of no returns: Thailand’s Pak Mun Dam and its fish ladder. Nat. Hist. Bull. Siam. Soc. 2001, 49, 189–230. [Google Scholar]
- Dudgeon, D. Threats to freshwater biodiversity globally and in the Indo-Burma Biodiversity Hotspot. In The Status and Distribution of Freshwater Biodiversity in Indo-Burma; Allen, D.J., Smith, K.G., Darwall, W.R.T., Eds.; IUCN: Cambridge, UK; Gland, Switzerland, 2012; pp. 1–28. ISBN 978-2-8317-1424-0. [Google Scholar]
- Millennium Ecosystem Assessment. Ecosystem and Human Well-Being: Biodiversity Synthesis; World Resources Institute: Washington, DC, USA, 2005; ISBN 9781569735886. [Google Scholar]
- Lodge, D.M. Lakes. In Global Biodiversity in a Changing Environment: Scenarios for the 21st Century; Chapin, F.S., III, Sala, O.E., Huber-Sannwald, E., Eds.; Springer: New York, NY, USA, 2001; pp. 277–313. ISBN 978-1-4613-0157-8. [Google Scholar]
- Craig, J.F. Human induced changes in the composition of fish communities in the African Great Lakes. Rev. Fish Biol. Fisher. 1992, 2, 93–124. [Google Scholar] [CrossRef]
- Lowe-McConnell, R.H. Fish faunas of the African Great Lakes: Origins, diversity, and vulnerability. Conserv. Biol. 1993, 7, 634–643. [Google Scholar] [CrossRef]
- Vimos, D.J.; Encalada, A.C.; Ríos-Touma, B.; Suárez, E.; Prat, N. Effects of exotic trout on benthic communities in high-Andean tropical streams. Freshw. Sci. 2015, 34, 770. [Google Scholar] [CrossRef]
- Strayer, D.L. Alien species in fresh waters: Ecological effects, interactions with other stressors, and prospects for the future. Freshw. Biol. 2010, 55, 152–174. [Google Scholar] [CrossRef]
- Carlsson, N.O.L.; Brönmark, C.; Hansson, L. Invading herbivory: The golden apple snail alters ecosystem functioning in Asian wetlands. Ecology 2004, 85, 1575–1580. [Google Scholar] [CrossRef]
- Global Invasive Species Database. Species Profile: Eichhornia crassipes. 2019. Available online: http://www.iucngisd.org/gisd/species.php?sc=70 (accessed on 3 September 2019).
- Dos Santos, J.R.; Galvão, L.S.; Oliveira, L.E.; de Aragão, C. Remote sensing of amazonian forests: Monitoring structure, phenology and responses to environmental changes. Rev. Bras. Cartogr. 2014, 3, 1413–1436. [Google Scholar]
- Miettinen, J.; Hooijer, A.; Shi, C.; Tollenaar, D.; Vernimmen, R.; Liew, S.C.; Malins, C.; Page, S.E. Extent of industrial plantations on Southeast Asian peatlands in 2010 with analysis of historical expansion and future projections. GCB Bioenergy 2012, 4, 908–918. [Google Scholar] [CrossRef]
- Miettinen, J.; Shi, C.; Liew, S.C. Land cover distribution in the peatlands of Peninsular Malaysia, Sumatra and Borneo in 2015 with changes since 1990. Glob. Ecol. Conserv. 2016, 6, 67–78. [Google Scholar] [CrossRef]
- Langner, A.; Miettinen, J.; Siegert, F. Land cover change 2002–2005 in Borneo and the role of fire derived from MODIS imagery. Glob. Chang. Biol. 2007, 13, 2329–2340. [Google Scholar] [CrossRef]
- Hooijer, A.; Page, S.; Jauhiainen, J.; Lee, W.A.; Lu, X.X.; Idris, A.; Anshari, G. Subsidence and carbon loss in drained tropical peatlands. Biogeosciences 2012, 9, 1053–1071. [Google Scholar] [CrossRef]
- Harrison, M.E.; Ripoll Capilla, B.; Thornton, S.A.; Cattau, M.E.; Page, S.E. Impacts of the 2015 fire season on peat-swamp forest biodiversity in Indonesian Borneo. In Peatlands in Harmony—Agriculture, Industry & Nature: Oral Presentations, Proceedings of the 15th International Peat Congress, Sarawak, Malaysia, 15–19 August 2016; International Peat Society and Malaysian Peat Society: Kuching, Malaysia, 2016; pp. 713–717. [Google Scholar]
- Page, S.E.; Siegert, F.; Rieley, J.O.; Boehm, H.D.V.; Jaya, A.; Limin, S. The amount of carbon released from peat and forest fires in Indonesia during 1997. Nature 2002, 420, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Segah, H.; Harada, T.; Limin, S.; June, T.; Hirata, R.; Osaki, M. Carbon dioxide balance of a tropical peat swamp forest in Kalimantan, Indonesia. Glob. Chang. Biol. 2006, 13, 412–425. [Google Scholar] [CrossRef]
- Moore, S.; Evans, C.D.; Page, S.E.; Garnett, M.H.; Jones, T.G.; Freeman, C.; Hooijer, A.; Wiltshire, A.J.; Limin, S.H.; Gauci, V. Deep instability of deforested tropical peatlands revealed by fluvial organic carbon fluxes. Nature 2013, 493, 660–663. [Google Scholar] [CrossRef]
- Mitchell, S.A. The status of wetlands, threats and the predicted effect of global climate change: The situation in Sub-Saharan Africa. Aquat. Sci. 2013, 75, 95–112. [Google Scholar] [CrossRef]
- Thieme, M.L.; Lehner, B.; Abell, R.; Matthews, J. Exposure of Africa’s freshwater biodiversity to a changing climate. Conserv. Lett. 2010, 3, 324–331. [Google Scholar] [CrossRef]
- Barros, D.F.; Albernaz, A.L.M. Possible impacts of climate change on wetlands and its biota in the Brazilian Amazon. Braz. J. Biol. 2014, 74, 810–820. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Grumbine, R.E.; Shrestha, A.; Eriksson, M.; Yang, X.; Wang, Y.; Wilkes, A. The melting Himalayas: Cascading effects of climate change on water, biodiversity, and livelihoods. Conserv. Biol. 2009, 23, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Gopal, B. Future of wetlands in tropical and subtropical Asia, especially in the face of climate change. Aquat. Sci. 2013, 75, 39–61. [Google Scholar] [CrossRef]
- Jafari, M.; Tavili, A.; Panahi, F.; Esfahan, E.Z.; Ghorbani, M. Characteristics of arid and desert ecosystems. In Reclamation of Arid Lands; Jafari, M., Tavili, A., Panahi, F., Esfahan, E.Z.M., Ghorbani, M., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 21–91. [Google Scholar] [CrossRef]
- Tydecks, L.; Jeschke, J.M.; Zarfl, C.; Böhning-Gaese, K.; Schütt, B.; Bremerich, V.; Tockner, K. Oases in the Sahara Desert—Linking Biological and Cultural Diversity. People Nat. 2020, in press. [Google Scholar]
- Holzapfel, C. Deserts. In Encyclopedia of Ecology; Jørgensen, S.E., Fath, B.D., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2008; pp. 879–898. [Google Scholar]
- Fisher, S.G.; Gray, L.J.; Grimm, N.B.; Busch, D.E. Temporal succession in a desert stream ecosystem following flash flooding. Ecol. Monogr. 1982, 52, 93–110. [Google Scholar] [CrossRef]
- Khozin-Goldberg, I.; Cohen, Z. The effect of phosphate starvation on the lipid and fatty acid composition of the freshwater eustigmatophyte Monodus subterraneus. Phytochemistry 2006, 67, 696–701. [Google Scholar] [CrossRef]
- Grimm, N.B.; Petrone, K.C. Nitrogen fixation in a desert stream ecosystem. Biogeochemistry 1997, 37, 33–61. [Google Scholar] [CrossRef]
- Scott, J.T.; Marcarelli, A.M. Cyanobacteria in freshwater benthic environments. In Ecology of Cyanobacteria II: Their Diversity in Space and Time; Whitton, B.A., Ed.; Springer Science+Business Media: Dordrecht, The Netherlands, 2012; pp. 271–289. [Google Scholar] [CrossRef]
- Harms, T.K.; Sponseller, R.A.; Grimm, N.B. Desert Streams. In Encyclopedia of Ecology; Jørgensen, S.E., Fath, B.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 871–879. ISBN 9780444520333. [Google Scholar]
- Saber, A.A.; Mareš, J.; Guella, G.; Anesi, A.; Štenclová, L.; Cantonati, M. Polyphasic approach to a characteristic Ulva population from a limno-rheocrenic, mineral (chloride, sodium, sulphate) spring in the Siwa Oasis (Western Desert of Egypt). Fottea 2018, 18, 227–242. [Google Scholar] [CrossRef]
- Green, A.J.; Jenkins, K.M.; Bells, D.; Morris, P.J.; Kingsford, R.T. The potential role of waterbirds in dispersing invertebrates and plants in arid Australia. Freshw. Biol. 2008, 53, 380–392. [Google Scholar] [CrossRef]
- Keleher, M.J.; Rader, R.B. Dispersal limitations and history explain community composition of metaphyton in desert springs of the Bonneville Basin, Utah: A multiscale analysis. Limnol. Oceanogr. 2008, 53, 1604–1613. [Google Scholar] [CrossRef]
- Foissner, W. Biogeography and dispersal of microorganisms: A review emphasizing protists. Acta Protozool. 2006, 45, 111–136. [Google Scholar]
- Witt, J.D.; Threloff, D.L.; Hebert, P.D. DNA barcoding reveals extraordinary cryptic diversity in an amphipod genus: Implications for desert spring conservation. Mol. Ecol. 2006, 15, 3073–3082. [Google Scholar] [CrossRef] [PubMed]
- Sluys, R.; Grant, L.J.; Blair, D. Freshwater planarians from artesian springs in Queensland, Australia (Platyhelminthes, Tricladida, Paludicola). Contrib. Zool. 2007, 76, 9–19. [Google Scholar] [CrossRef]
- Brown, C.M. 1965. Freshwater algae of the Central Death Valley Desert. Ohio J. Sci. 1965, 65, 11–28. [Google Scholar]
- Shaaban, A.S.; Mansour, H.A.; Saber, A.A. Unveiling algal biodiversity of El-Farafra Oasis (Western Desert, Egypt) and potential relevance of its use in water bio-assessment: Special interest on springs and drilled wells. Egypt. J. Phycol. 2015, 16, 47–75. [Google Scholar]
- Garcia-Pichel, F.; Wade, B.D.; Farmer, J.D. Jet-suspended, calcite-ballasted cyanobacterial waterwarts in a desert spring. J. Phycol. 2002, 38, 420–428. [Google Scholar] [CrossRef]
- Saber, A.A.; Ballot, A.; Schneider, S.; Cantonati, M. Morphological and molecular features of a Chara vulgaris population from desert springs on the Sinai Peninsula (Springs of Moses, Egypt). Bot. Lett. 2018, 165, 77–89. [Google Scholar] [CrossRef]
- Dvořák, P.; Casamatta, D.A.; Hašler, P.; Jahodářová, E.; Norwich, A.R.; Poulíčková, A. Diversity of the cyanobacteria. In Modern topics in the phototrophic prokaryotes: Environmental and applied Aspects; Hallenbeck, P.C., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 3–46. [Google Scholar]
- Minckley, T.A.; Turner, D.S.; Weinstein, S.R. The relevance of wetland conservation in arid regions: A re-examination of vanishing communities in the American Southwest. J. Arid Environ. 2013, 88, 213–221. [Google Scholar] [CrossRef]
- Rossini, R.A.; Fensham, R.J.; Stewart-Koster, B.; Gotch, T.; Kennard, M.J. Biogeographical patterns of endemic diversity and its conservation in Australia’s artesian desert springs. Divers. Distrib. 2018, 24, 1199–1216. [Google Scholar] [CrossRef]
- Rossini, R.A.; Tibbetts, H.L.; Fensham, R.J.; Walter, G.H. Can environmental tolerances explain convergent patterns of distribution in endemic spring snails from opposite sides of the Australian arid zone? Aquat. Ecol. 2017, 51, 605–624. [Google Scholar] [CrossRef]
- Holste, D.R.; Inoue, K.; Lang, B.K.; Berg, D.J. Identification of microsatellite loci and examination of genetic structure for the endangered springsnails Juturnia kosteri and Pyrgulopsis roswellensis in the Chihuahuan Desert. Aquat. Conserv. Mar. Freshw. Ecosys. 2016, 26, 715–723. [Google Scholar] [CrossRef]
- Stanislawczyk, K.; Walters, A.D.; Haan, T.J.; Sei, M.; Lang, B.K.; Berg, D.J. Variation among macroinvertebrate communities suggests the importance of conserving desert springs. Aquat. Conserv. Mar. Freshw. Ecosys. 2018, 28, 944–953. [Google Scholar] [CrossRef]
- Murphy, N.P.; Pavlova, A.L.; Thompson, R.; Davis, J.; Sunnucks, P. Swimming through sand: Connectivity of aquatic fauna in deserts. Ecol. Evol. 2015, 5, 5252–5264. [Google Scholar] [CrossRef] [PubMed]
- Saber, A.A.; Kouwets, F.A.C.; Haworth, E.Y.; Cantonati, M. A new Euastrum species (Conjugatophyceae, Streptophyta) from the Western Desert of Egypt. Cryptogam. Algol. 2018, 39, 215–226. [Google Scholar] [CrossRef]
- Navarro, L.M.; Fernández, N.; Guerra, C.; Guralnick, R.; Kissling, W.D.; Londoño, M.C.; Muller-Karger, F.; Turak, E.; Balvanera, P.; Costello, M.J.; et al. Monitoring biodiversity change through effective global coordination. Curr. Opin. Env. Sust. 2017, 29, 158–169. [Google Scholar] [CrossRef]
- Pereira, H.M.; Ferrier, S.; Walters, M.; Geller, G.N.; Jongman, R.H.; Scholes, R.J.; Bruford, M.W.; Brummitt, N.; Butchart, S.H.; Cardoso, A.C.; et al. Essential biodiversity variables. Science 2013, 339, 277–278. [Google Scholar] [CrossRef]
- Turak, E.; Harrison, I.; Dudgeon, D.; Abell, R.; Bush, A.; Darwall, W.; Finlayson, C.M.; Ferrier, S.; Freyhof, J.; Hermoso, V.; et al. Essential Biodiversity Variables for measuring change in global freshwater biodiversity. Biol. Conserv. 2017, 213, 272–279. [Google Scholar] [CrossRef]
- Kissling, W.D.; Ahumada, J.A.; Bowser, A.; Fernandez, M.; Fernández, N.; García, E.A.; Guralnick, R.P.; Isaac, N.J.; Kelling, S.; Los, W.; et al. Building essential biodiversity variables (EBV s) of species distribution and abundance at a global scale. Biol. Rev. 2018, 93, 600–625. [Google Scholar] [CrossRef]
- Jetz, W.; McGeoch, M.; Guralnick, R.; Ferrier, S.; Beck, J.; Costello, M.; Fernandez, M.; Geller, G.; Keil, P.; Cory Merow, C.; et al. Essential biodiversity variables for mapping and monitoring species populations. Nat. Ecol. Evol. 2019, 3. [Google Scholar] [CrossRef]
- Richardson, J.S.; Sato, T. Resource flows across freshwater-terrestrial boundaries and influence on processes linking adjacent ecosystems. Ecohydrology 2015, 8, 406–415. [Google Scholar] [CrossRef]
- Soininen, J.; Bartels, P.; Heino, J.; Luoto, M.; Hillebrand, H. Toward more integrated ecosystem research in aquatic and terrestrial environments. BioScience 2015, 65, 174–182. [Google Scholar] [CrossRef]
- Tonkin, J.D.; Altermatt, F.; Finn, D.; Heino, J.; Olden, J.D.; Pauls, S.U.; Lytle, D.A. The role of dispersal in river network metacommunities: Patterns, processes, and pathways. Freshw. Biol. 2018, 63, 141–163. [Google Scholar] [CrossRef]
- Hughes, J.M.R.; Bond, H.; Knight, C.; Stanley, K. Challenges for Freshwater Ecosystems. In Water Science, Policy, and Management; Dadson, S.J., Garrick, D.E., Penning-Rowsell, C., Hall, J.W., Hope, R., Hughes, J., Eds.; John Wiley & Sons Ltd: Hoboken, NJ, USA, 2019. [Google Scholar] [CrossRef]
- Strayer, D.L.; Dudgeon, D. Freshwater biodiversity conservation: Recent progress and future challenges. J. N. Am. Benthol. Soc. 2010, 29, 344–358. [Google Scholar] [CrossRef]
- Downing, J.A. Limnology and oceanography: Two estranged twins reuniting by global change. Inland Waters 2014, 4, 215–232. [Google Scholar] [CrossRef]
- Allan, J.D.; Flecker, A.S. Biodiversity conservation in running waters. BioScience 1993, 43, 32–43. [Google Scholar] [CrossRef]
- Stendera, S.; Adrian, R.; Bonada, N.; Cañedo-Argüelles, M.; Hugueny, B.; Januschke, K.; Plettenbuer, F.; Hering, D. Drivers and stressors of freshwater biodiversity patterns across different ecosystems and scales: A review. Hydrobiologia 2012, 696, 1–28. [Google Scholar] [CrossRef]
- Cañedo-Argüelles, M.; Hawkins, C.P.; Kefford, B.J.; Schäfer, R.B.; Dyack, B.J.; Brucet, S.; Buchwalter, D.; Dunlop, J.; Frör, O.; Lazorchak, J.; et al. Saving freshwater from salts. Science 2016, 351, 914–916. [Google Scholar] [CrossRef]
- Kernan, M.; Battarbee, R.W.; Moss, B. Climate Change, Freshwater Ecosystems. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2013. [Google Scholar] [CrossRef]
- Richter, B.D.; Powell, E.M.; Lystash, T.; Faggert, M. Protection and Restoration of Freshwater Ecosystems. In Water Policy and Planning in a Variable and Changing Climate; Miller, K.A., Hamlet, A.F., Kenney, D.S., Redmond, K.T., Eds.; Taylor & Francis Group, LLC: London, UK, 2016; pp. 82–103. ISBN 9781138490864. [Google Scholar]
- Maliva, R.G. ASR and Aquifer Recharge Using Wells. In Anthropogenic Aquifer Recharge; Maliva, R.G., Ed.; Springer: Berlin, Germany, 2020. [Google Scholar] [CrossRef]
- Poikane, S.; Kelly, M.G.; Cantonati, M. Benthic algal assessment of ecological status in European lakes and rivers: Challenges and opportunities. Sci. Total Environ. 2016, 568, 603–613. [Google Scholar] [CrossRef]
- Cardinale, B.J.; Srivastava, D.S.; Duffy, J.E.; Wright, J.P.; Downing, A.L.; Sankaran, M.; Jouseau, C. Effects of biodiversity on the functioning of trophic groups and ecosystems. Nature 2006, 443, 989–992. [Google Scholar] [CrossRef]
- Loreau, M.; Naeem, S.; Inchausti, P.; Bengtsson, J.; Grime, J.P.; Hector, A.; Hooper, D.U.; Huston, M.A.; Raffaelli, D.; Schmid, B.; et al. Biodiversity and ecosystem functioning: Current knowledge and future challenges. Science 2001, 294, 804–808. [Google Scholar] [CrossRef]
- Tilman, D.; Downing, J.A. Biodiversity and stability in grasslands. Nature 1994, 367, 363–365. [Google Scholar] [CrossRef]
- Covich, A.P.; Austen, M.C.; Bärlocher, F.; Chauvet, E.; Cardinale, B.J.; Biles, C.L.; Inchausti, P.; Dangles, O.; Solan, M.; Gessner, M.O.; et al. The role of biodiversity in the functioning of freshwater and marine benthic ecosystems. BioScience 2004, 54, 767–775. [Google Scholar] [CrossRef]
- Strayer, D.; Downing, J.A.; Haag, W.R.; King, T.L.; Layer, J.B.; Newton, T.J.; Nichols, S. Changing perspectives on pearly mussels, North America’s most imperiled animals. BioScience 2004, 54, 429–439. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Reconciliation ecology and the future of species diversity. Oryx 2003, 37, 194–205. [Google Scholar] [CrossRef]
- Palmer, M.A.; Hondula, K.L.; Koch, B.J. Ecological Restoration of Streams and Rivers: Shifting Strategies and Shifting Goals. Annu. Rev. Ecol. Evol. S. 2014, 45, 247–269. [Google Scholar] [CrossRef]
- Nilsson, C.; Polvi, L.E.; Gardeström, J.; Hasselquist, E.M.; Lind, L.; Sarneel, J.M. Riparian and in-stream restoration of boreal streams and rivers: Success or failure? Ecohydrology 2015, 8, 753–764. [Google Scholar] [CrossRef]
- Boon, P.J.; Davies, B.R.; Petts, G.E. Global Perspectives on River Conservation: Science, Policy and Practice; John Wiley: Chichester, UK, 2000; p. 548. ISBN 978-0-471-96062-1. [Google Scholar]
- Moss, B. Biodiversity in fresh waters: An issue of species preservation or system functioning? Environ. Conserv. 2000, 27, 1–4. [Google Scholar] [CrossRef]
- Herbert, M.E.; Mcintyre, P.B.; Doran, P.J.; Allan, J.D.; Abell, R. Terrestrial Reserve Networks Do Not Adequately Represent Aquatic Ecosystems. Conserv. Biol. 2010, 24, 1002–1011. [Google Scholar] [CrossRef]
- Poff, N.L.; Allan, J.D.; Palmer, M.A.; Hart, D.D.; Richter, B.D.; Arthington, A.H.; Rogers, K.H.; Meyer, J.L.; Stanford, J.A. River flows and water wars: Emerging science for environmental decision-making. Front. Ecol. Environ. 2003, 1, 298–306. [Google Scholar] [CrossRef]
- Postel, S.L.; Daily, G.C.; Ehrlich, P.R. Human appropriation of renewable fresh water. Science 1996, 271, 785–788. [Google Scholar] [CrossRef]
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Reidy Liermann, C.; et al. Global threats to human water security and river biodiversity. Nature 2010, 467, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Baron, J.S.; LeRoy Poff, N.; Angermeier, P.L.; Dahm, C.N.; Gleick, P.H.; Hairston, N.G., Jr.; Jackson, R.B.; Johnston, C.A.; Richter, B.D.; Steinman, A.D. Meeting ecological and societal needs for freshwater. Ecol. Appl. 2002, 12, 1247–1260. [Google Scholar] [CrossRef]






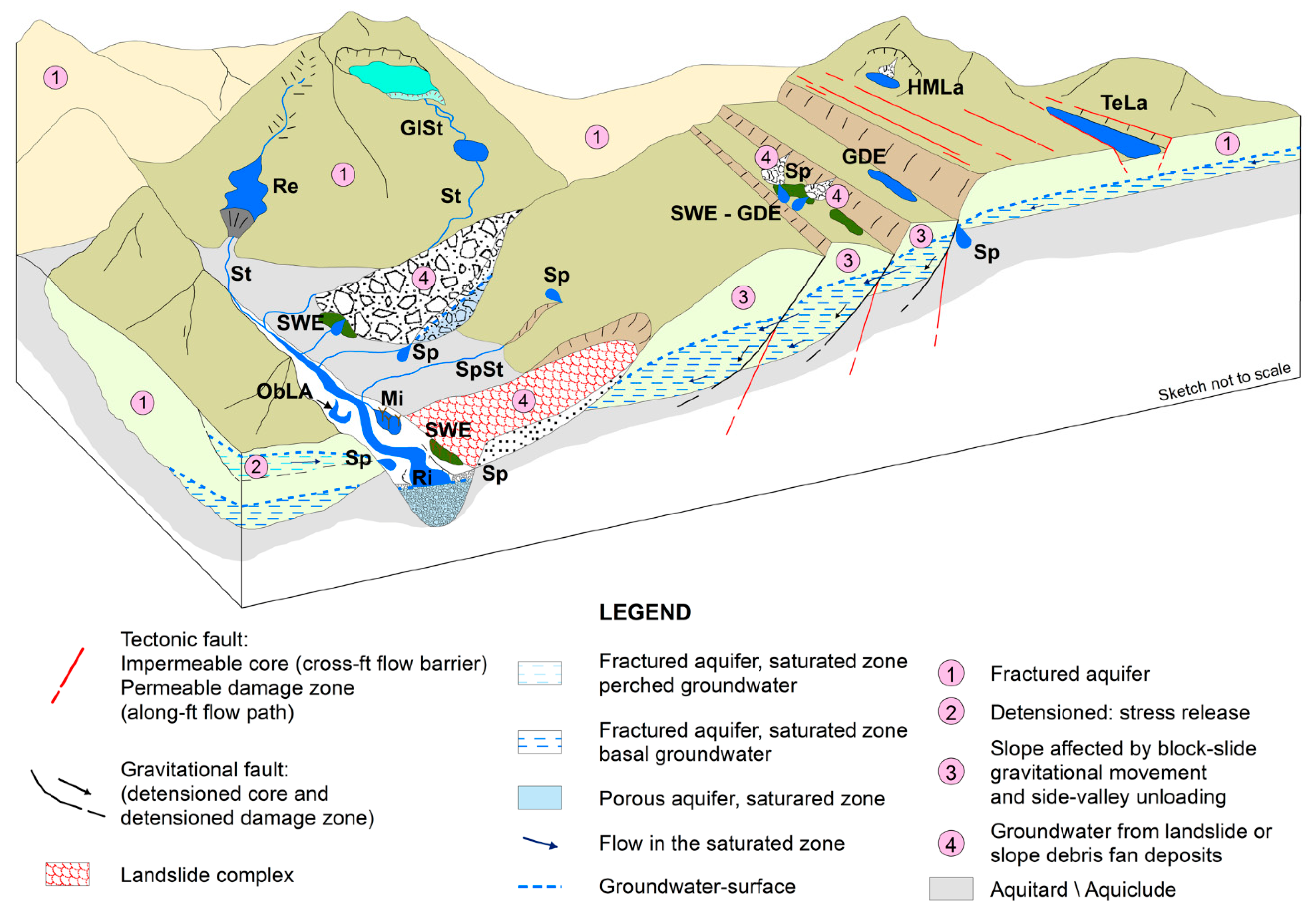
| Freshwater Habitat Type | Biodiversity and Ecological Features | Main Impacts | Conservation Issues |
|---|---|---|---|
| Groundwater (GW) and Dependent Ecosystems [47] | Rare, narrow endemics, sensitive | GW overexploitation, pollution, global change, alteration in flow regime, disconnection between SW bodies and GW | Biodiversity decreasing in both abundance and species richness, narrow endemics at high risk of extinction |
| Springs and Spring-fed Streams (Crenal) [40,41,42,43,44,45,46,47] | Exclusive, rare, threatened, sensitive species (LIHRe) | Water abstraction, habitat destruction (tapping), livestock watering practices | Near-natural springs are vanishing |
| Glacial Streams (Kryal) [48] | Extremely harsh and selective environments due to high turbidity and low temperatures | Global warming causing glacier retreat and disappearance | Glaciers are disappearing not only from isolated tropical summits but also from mid-latitude mountain ranges |
| Streams (Rhithral) [46,47] | Dynamic core section of the River Continuum, unidirectional flow, nutrient spiraling, patchiness | Hydraulic modifications, water abstraction, diking, damming, contamination, pollution | Severe constraints to natural seasonal-flow variability and channel-morphology rearrangement |
| Large Rivers (Potamal) | Naturally turbid, floodplains, meanders, potamoplankton, large-bodied species | Hydroelectric power production, used as transportation corridors, diked to protect infrastructure, drained for irrigation, introduction of non-native species | Cumulative effects of dams involve declines in native freshwater biodiversity at regional scales |
| Ancient and Large Lakes | Large L.: Mainly postglacial colonizers but endemic species in some. Ancient L.: High endemism and diversity. Substantial depth variation in benthic communities | Eutrophication, climate warming, overfishing, invasive non-native species, hydrological alterations | Strict protection of endemic species is necessary. Safeguarding water levels is of great importance |
| High-mountain lakes [47] | Often isolated (local genotypes), phytoplankton dominated by flagellate mixotrophic algae which control bacterioplankton, substantial depth variation in benthic communities | Environmental & climate change, diffuse airborne pollution, introduction of non-native fish species, exploitation for hydroelectric purposes or artificial-snow making, eutrophication, acidification, grazing, tourism | Conservation measures especially in natural preserves: fish-stocking ban, fish eradication, protection against eutrophication |
| Oxbow Lakes | Threatened, sensitive species | Alteration in flow regime, water abstraction, damming, flood control infrastructures, global change | Natural river dynamic processes are largely impaired, increasing water turbidity, spread of alien species |
| Reservoirs | Abundant, diverse, reduced littoral, often productivity gradients increasing diversity | Water-level fluctuations, eutrophication, floods, fishery/recreation, invasive species | Mesotrophic/oligotrophic species, species dependent on stable water level, phytophilous flora and fauna |
| Urban Freshwater Habitats [49] | Typically low biodiversity, but there are also examples of higher species richness compared with similar non-urban habitats | Pollution, habitat destruction, invasive species | Reference conditions for novel ecosystems, identification of specific conservation goals, matching biodiversity indicators to the conservation objectives |
| Mires [47,50] | Semi-zonal at North, rare and relict at South, diverse, endangered, sensitive | Drainage, hydrological alterations, eutrophication, land-use change, GW overexploitation and pollution, nitrogen deposition, climate change | Rapidly declining biodiversity (e.g., microorganisms, invertebrates, plants) especially in base-rich fens, succession to less-unique ecosystems, endangered worldwide, complex inter-specific interactions |
| Small Wetland Ecosystems [51] | Threatened, rare, sensitive species | Land reclamation, drainage, water overexploitation, pollution, global change | Abandonment of traditional agronomic practices, water turbidity, alien species |
| Boreal and Arctic Freshwater Habitats | Relatively low levels of endemism but some unique cold-adapted species | Climate warming (melting permafrost), land-use change, hydromorphological alteration, long-range pollution | Concomitant reduction of multiple stressors exacerbated by climate change |
| Antarctic Freshwater Habitats | High levels of endemism, depauperate | Alteration of habitat through changing climate/hydrology | Protection from invasive taxa |
| Mediterranean Streams | High species richness, high levels of endemism, high temporal turnover in species composition | Climate change (i.e., increased frequency and severity of droughts), dam construction, water abstraction, pollution, invasive species | Biodiversity decline, high extinction risk (especially strictly aquatic organisms, such as fish and mollusks), many protected areas do not cover critical areas of biodiversity |
| Mediterranean Reservoirs | Low biodiversity, lack of littoral species, high risk for non-native-species propagation | No or very low threats, favor non-native species | No particular conservation issues exist |
| Mediterranean Temp. and Perm. Ponds | Very high biodiversity, high number of exclusive species (temp. ponds), high risk of extinction | Temp. ponds: Industrial agriculture (water over-exploitation, crop-extension increases, transformation into perm. water bodies), used as dumpsites or filled to create infrastructures | Exclusive species at risk of extinction, biodiversity lowering due to decreased species richness |
| Mediterranean Large, Near-Natural Lakes (L. Kinneret) | Endangered native aquatic fauna and flora, long-term changes in phytoplankton species composition from a stable community until mid 1990s to an unpredictable community with toxic cyanobacteria | Great efforts made to minimize influence of effluents, dust is a major phosphate source (especially in summer), man-made manipulation of water levels at an amplitude much greater than natural | Shift from using lake water to using desalinized water as a major source of drinking water to stop the large year-to-year fluctuations in water level, non-native fish stocking ban |
| Tropical Freshwater Habitats [52] | Flood pulse, intermittent wetlands, biggest river basins, hotspot of mega-biodiversity (i.e., fishes, amphibians) | Hydrological alteration by dam construction and habitat destruction for cultivation and urbanization, non-native species introduction | Increase of economic activities in developing countries, low knowledge about species and their ecology; scarce environmental protection legislation |
| Arid-Climate Freshwater Habitats | Harsh conditions due to variable hydrologic regime (periods of drying, flash floods) and high temperatures | Diverse human pressures, land reclamation, GW deterioration, climatic changes | Conservation of rare and threatened species, ecosystem protection from urbanization |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantonati, M.; Poikane, S.; Pringle, C.M.; Stevens, L.E.; Turak, E.; Heino, J.; Richardson, J.S.; Bolpagni, R.; Borrini, A.; Cid, N.; et al. Characteristics, Main Impacts, and Stewardship of Natural and Artificial Freshwater Environments: Consequences for Biodiversity Conservation. Water 2020, 12, 260. https://doi.org/10.3390/w12010260
Cantonati M, Poikane S, Pringle CM, Stevens LE, Turak E, Heino J, Richardson JS, Bolpagni R, Borrini A, Cid N, et al. Characteristics, Main Impacts, and Stewardship of Natural and Artificial Freshwater Environments: Consequences for Biodiversity Conservation. Water. 2020; 12(1):260. https://doi.org/10.3390/w12010260
Chicago/Turabian StyleCantonati, Marco, Sandra Poikane, Catherine M. Pringle, Lawrence E. Stevens, Eren Turak, Jani Heino, John S. Richardson, Rossano Bolpagni, Alex Borrini, Núria Cid, and et al. 2020. "Characteristics, Main Impacts, and Stewardship of Natural and Artificial Freshwater Environments: Consequences for Biodiversity Conservation" Water 12, no. 1: 260. https://doi.org/10.3390/w12010260
APA StyleCantonati, M., Poikane, S., Pringle, C. M., Stevens, L. E., Turak, E., Heino, J., Richardson, J. S., Bolpagni, R., Borrini, A., Cid, N., Čtvrtlíková, M., Galassi, D. M. P., Hájek, M., Hawes, I., Levkov, Z., Naselli-Flores, L., Saber, A. A., Cicco, M. D., Fiasca, B., ... Znachor, P. (2020). Characteristics, Main Impacts, and Stewardship of Natural and Artificial Freshwater Environments: Consequences for Biodiversity Conservation. Water, 12(1), 260. https://doi.org/10.3390/w12010260













