Evaluation of Legionella pneumophila Decrease in Hot Water Network of Four Hospital Buildings after Installation of Electron Time Flow Taps
Abstract
1. Introduction
2. Materials and Methods
2.1. Settings
2.2. Water Disinfection
2.3. Hot Water Samplings and Legionella spp. Detection
- One week before TFT installation (Time 0 or T0);
- The day after TFT installation (Time 1 or T1);
- Seven days after TFT installation (Time 2 or T2);
- Fifteen days after TFT installation (Time 3 or T3);
- One month after TFT installation, and then regularly on a monthly basis (Time 4 or T4, Time 5 or T5, etc.).
2.4. Statistical Analysis
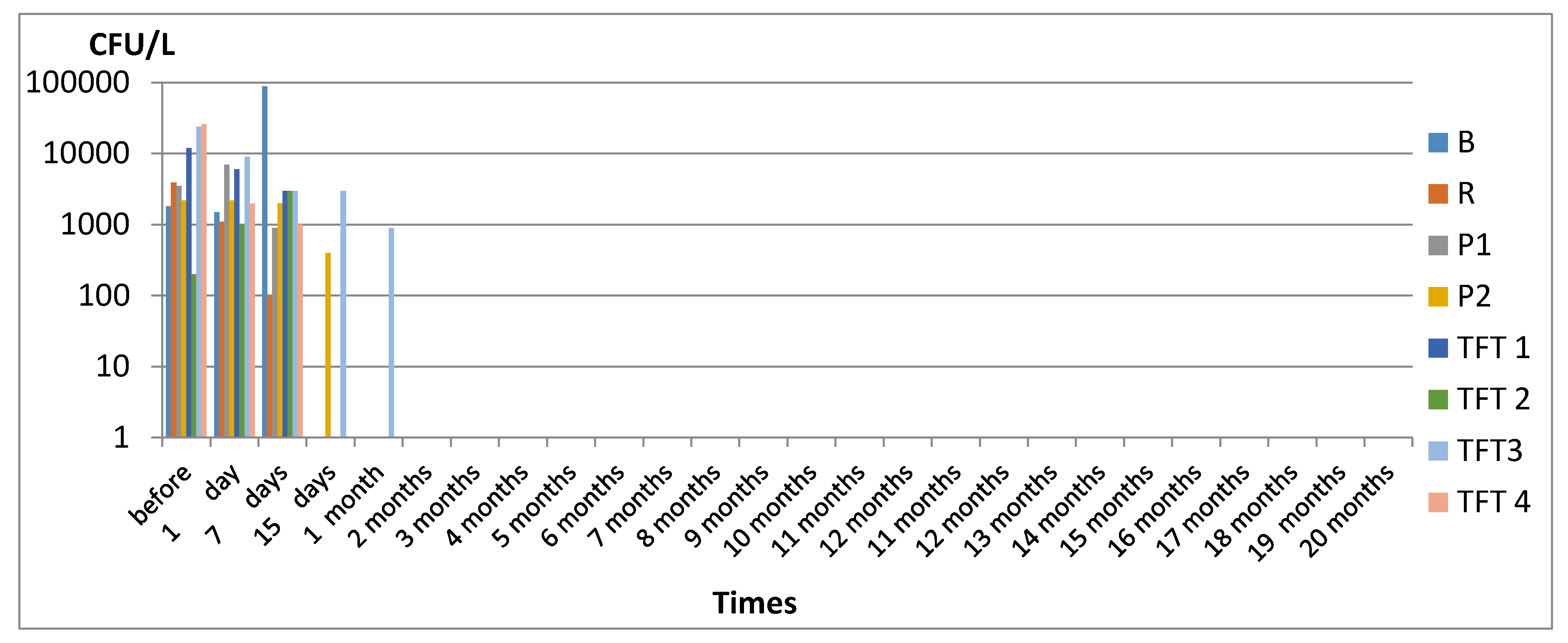
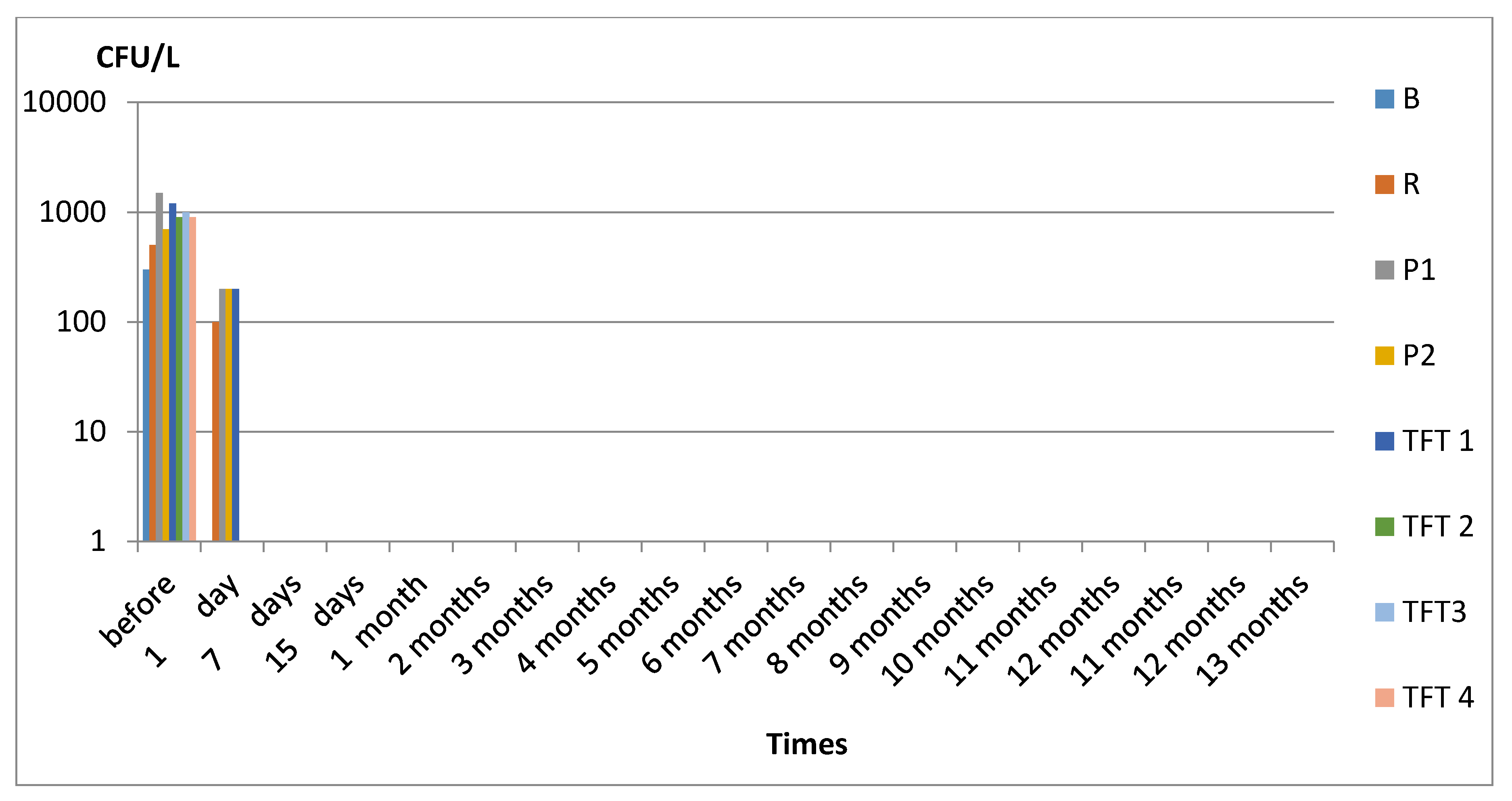
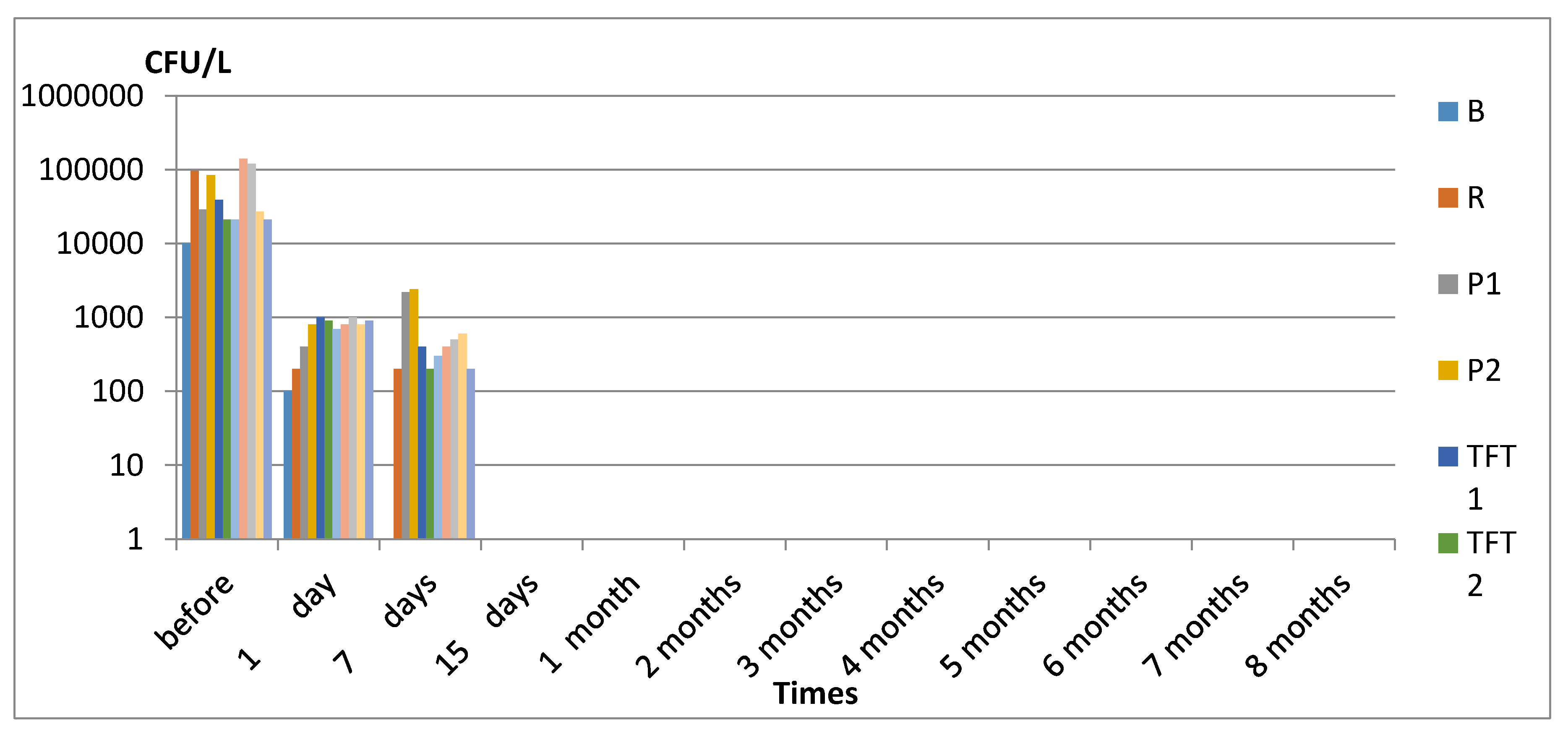
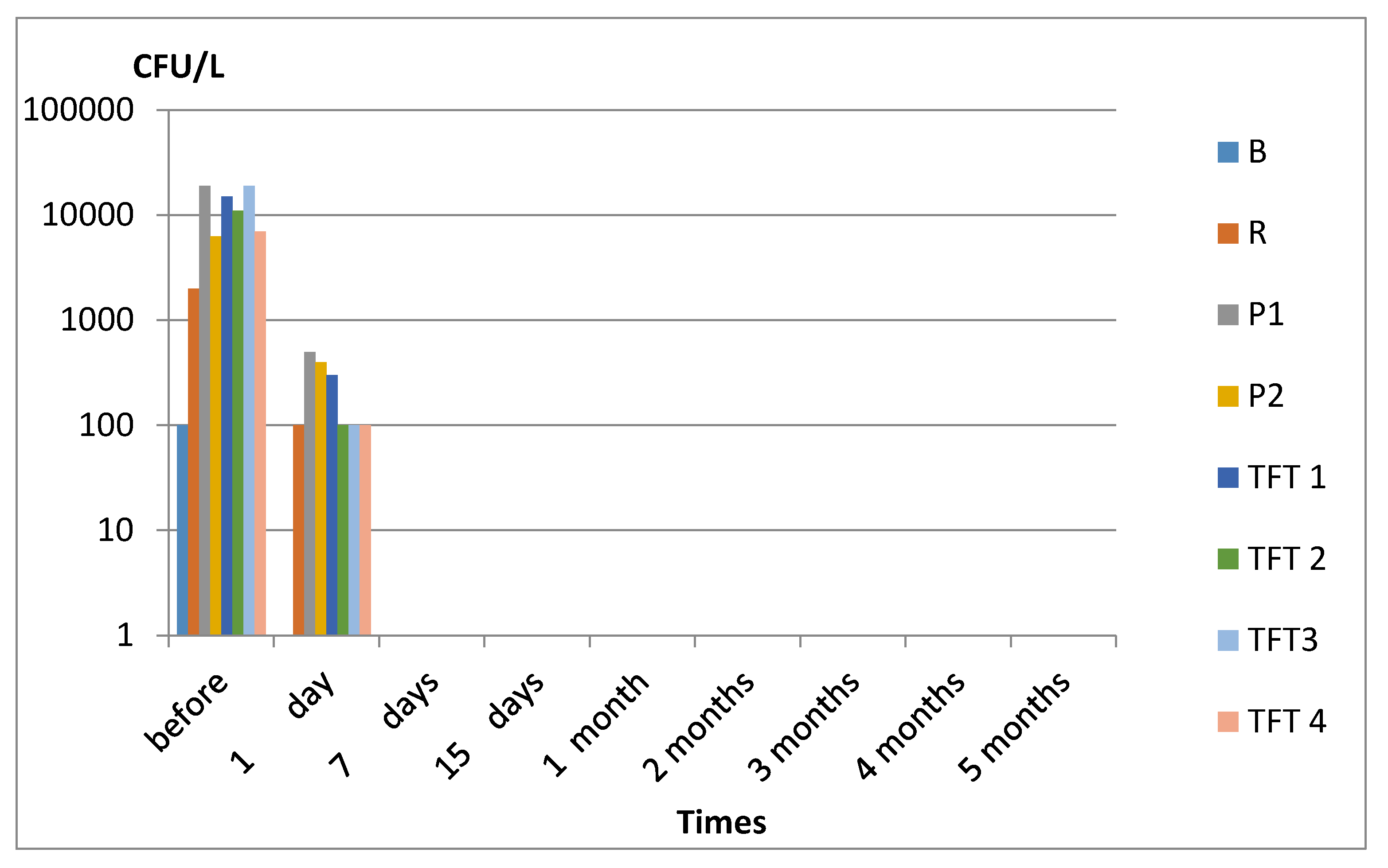
3. Results
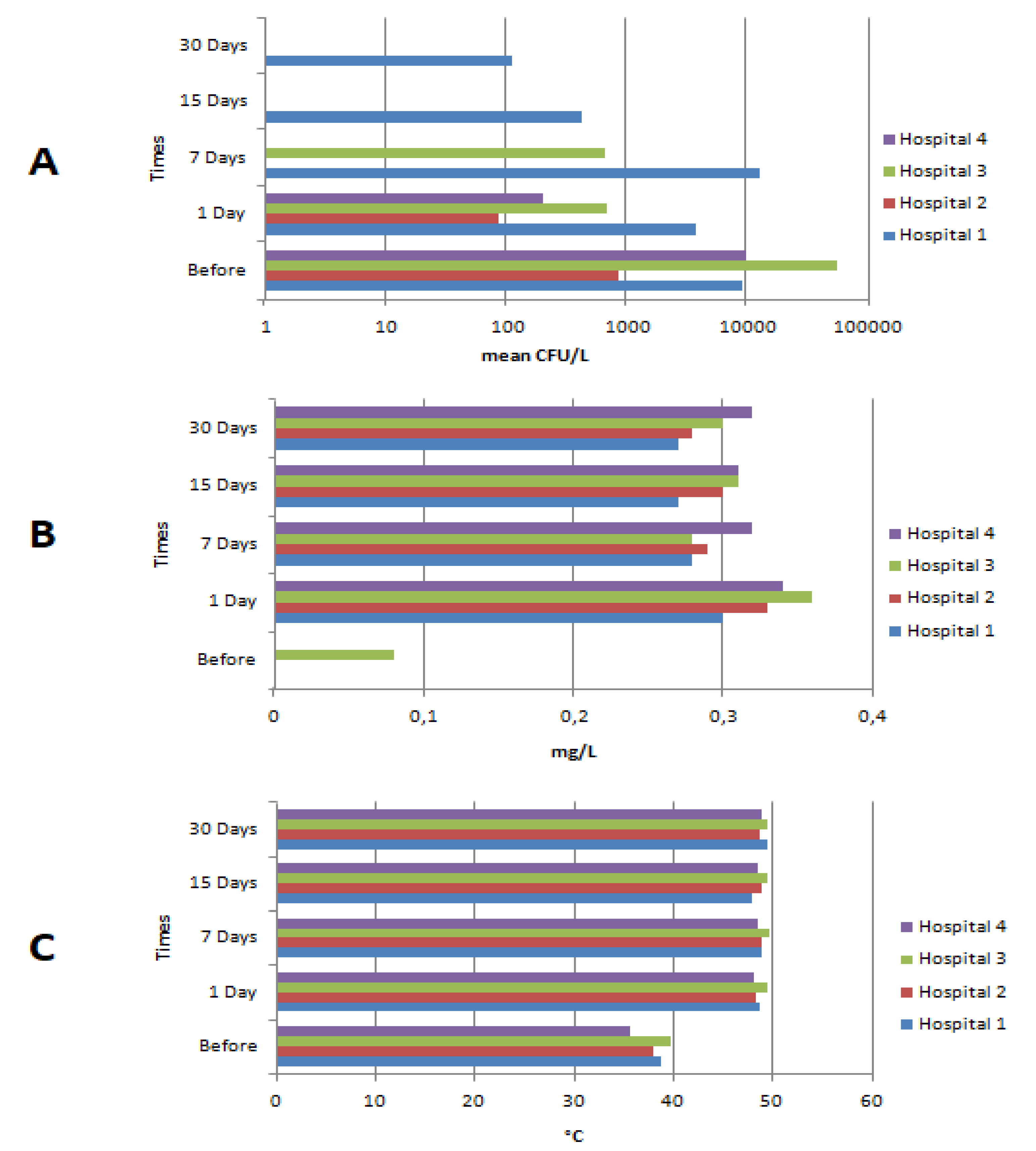
3.1. Legionella spp.
3.2. Physical–Chemical Parameters
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Oda, N.; Hirahara, T.; Fujioka, Y.; Mitani, R.; Takata, I. Legionella Pneumonia Following the Heavy Rain Event of July 2018 in Japan. Intern Med. 2019, 58, 2831–2834. [Google Scholar] [CrossRef] [PubMed]
- Muldrow, L.L.; Tyndall, R.L.; Fliermans, C.B. Application of flow cytometry to studies of pathogenic free-living amoebae. Appl. Environ. Microbiol. 1982, 44, 1258–1269. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Legionnaires’ Disease. Annual Epidemiological Report for 2017; European Centre for Disease Prevention and Control Stockholm: Solna, Sweden, 2019; pp. 1–6. Available online: https://www.ecdc.europa.eu/en/publications-data/legionnaires-disease-annual-epidemiological-report-2017 (accessed on 12 November 2019).
- Alexandropoulou, I.G.; Ntougias, S.; Konstantinidis, T.G.; Parasidis, T.A.; Panopoulou, M.; Constantinidis, T.C. Environmental surveillance and molecular epidemiology of waterborne pathogen Legionella pneumophila in health-care facilities of Northeastern Greece: A 4-year survey. Environ. Sci. Pollut. Res. Int. 2015, 22, 7628–7640. [Google Scholar] [CrossRef] [PubMed]
- Superior Institute of Health. Rapporto Annuale Sulla Legionellosi in Italia Nel 2017; Superior Institute of Health: Rome, Italy, 2018; Volume 31, pp. 1–24. Available online: http://www.legionellaonline.it/notiziari_ISS.htm (accessed on 12 November 2019).
- World Health Organization. Legionella and the Prevention of Legionellosis; World Health Organization: Geneva, Switzerland, 2007; pp. 1–276. Available online: https://www.who.int/water_sanitation_health/publications/legionella/en/ (accessed on 12 November 2019).
- Superior Institute of Health. Linee guida per la prevenzione ed il controllo della legionellosi; Superior Institute of Health: Rome, Italy, 2015; pp. 1–149. Available online: http://www.salute.gov.it/portale/documentazione/p6_2_2_1.jsp?id=2362 (accessed on 12 November 2019).
- Whiley, H.; Hinds, J.; Xi, J.; Bentham, R. Real-Time Continuous Surveillance of Temperature and Flow Events Presents a Novel Monitoring Approach for Hospital and Healthcare Water Distribution Systems. Int. J. Environ. Res. Public Health 2019, 16, 1332. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, W.J.; Pruden, A.; Edwards, M.A. Interactive Effects of Corrosion, Copper, and Chloramines on Legionella and Mycobacteria in Hot Water Plumbing. Environ. Sci. Technol. 2017, 51, 7065–7075. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Manuel, C.M.; Nunes, O.C.; Melo, L.F. Dynamics of drinking water biofilm in flow/non-flow conditions. Water Res. 2007, 41, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Totaro, M.; Valentini, P.; Costa, A.L.; Giorgi, S.; Casini, B.; Baggiani, A. Rate of Legionella pneumophila colonization in hospital hot water network after time flow taps installation. J. Hosp. Infect. 2018, 98, 60–63. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. ISO 11731 Water quality—Enumeration of Legionella; International Organization for Standardization: Geneva, Switzerland, 2017; pp. 1–38. Available online: https://www.iso.org/standard/61782.html (accessed on 19 November 2019).
- The Council of the European Union. Council Directive 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption; The Council of the European Union: Bruxelles, Belgium, 1998; pp. 1–23. Available online: https://op.europa.eu/en/publication-detail/-/publication/880a8575-7ee4-438e-b953-567df6719e26 (accessed on 19 November 2019).
- Montagna, M.T.; De Giglio, O.; Cristina, M.L.; Napoli, C.; Pacifico, C.; Agodi, A.; Baldovin, T.; Casini, B.; Coniglio, M.A.; D’Errico, M.M.; et al. Evaluation of Legionella Air Contamination in Healthcare Facilities by Different Sampling Methods: An Italian Multicenter Study. Int. J. Environ. Res. Public Health 2017, 14, 670. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, H.; Weber, D.J.; Rutala, W.A. Healthcare Outbreaks Associated with a Water Reservoir and Infection Prevention Strategies. Clin. Infect. Dis. 2016, 62, 1423–1435. [Google Scholar] [CrossRef] [PubMed]
- Totaro, M.; Casini, B.; Valentini, P.; Miccoli, M.; Giorgi, S.; Porretta, A.; Privitera, G.; Lopalco, P.L.; Baggiani, A. Evaluation and control of microbial and chemical contamination in dialysis water plants of Italian nephrology wards. J. Hosp. Infect. 2017, 97, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Scaturro, M.; Dell’eva, I.; Helfer, F.; Ricci, M.L. Persistence of the same strain of Legionella pneumophila in the water system of an Italian hospital for 15 years. Infect. Control Hosp. Epidemiol. 2007, 28, 1089–1092. [Google Scholar] [CrossRef] [PubMed]
- Grayman, W.M. A quarter of a century of water quality modeling in distribution systems. In Eighth Annual Water Distribution Systems Analysis Symposium (WDSA); American Society of Civil Engineers: Cincinnati, OH, USA, 2012; Available online: https://ascelibrary.org/doi/abs/10.1061/40941%28247%294 (accessed on 19 November 2019).
- Gavaldà, L.; Garcia-Nuñez, M.; Quero, S.; Gutierrez-Milla, C.; Sabrià, M. Role of hot water temperature and water system use on Legionella control in a tertiary hospital: An 8-year longitudinal study. Water Res. 2019, 149, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Bédard, E.; Laferrière, C.; Déziel, E.; Prévost, M. Impact of stagnation and sampling volume on water microbial quality monitoring in large buildings. PLoS ONE 2018, 13, e0199429. [Google Scholar] [CrossRef] [PubMed]
- Fish, K.; Osborn, A.M.; Boxall, J.B. Biofilm structures (EPS and bacterial communities) in drinking water distribution systems are conditioned by hydraulics and influence discolouration. Sci. Total Environ. 2017, 593, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Totaro, M.; Valentini, P.; Casini, B.; Miccoli, M.; Costa, A.L.; Baggiani, A. Experimental comparison of point-of-use filters for drinking water ultrafiltration. J. Hosp. Infect. 2017, 96, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Szabo, J.; Minamyer, S. Decontamination of biological agents from drinking water infrastructure: A literature review and summary. Environ. Int. 2014, 72, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Manuel, C.M.; Nunes, O.C.; Melo, L.F. Unsteady state flow and stagnation in distribution systems affect the biological stability of drinking water. Biofouling 2010, 26, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Totaro, M.; De Vita, E.; Mariotti, T.; Bisordi, C.; Giorgi, S.; Gallo, A.; Costa, A.L.; Casini, B.; Valentini, P.; Privitera, G.; et al. Cost analysis for electron time-flow taps and point of use filters: A comparison of two methods for Legionnaires’ disease prevention in hospital water networks. J. Hosp. Infect. 2019, 103, 231–232. [Google Scholar] [CrossRef] [PubMed]
| BEFORE TFT INSTALLATION (September 2017) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0 | 48.4 ± 2.8 | 6.52 ± 0.31 | 52 ± 4.5 | 67 ± 2.6 | <3 |
| R | 0 | 38.8 ± 1.9 | 6.50 ± 0.12 | 69 ± 4.3 | 73 ± 1.9 | <3 |
| P1 | 0 | 38.8 ± 2.1 | 6.60 ± 0.16 | 57 ± 3.9 | 82 ± 3.8 | <3 |
| P2 | 0 | 36.4 ± 1.9 | 6.57 ± 0.11 | 98 ± 2.6 | 79 ± 6.2 | <3 |
| TFT 1 | 0 | 39.5 ± 1.6 | 6.58 ± 0.09 | 74 ± 5.9 | 65 ± 7.1 | <3 |
| TFT 2 | 0 | 37.7 ± 1.7 | 6.70 ± 0.31 | 61 ± 6.6 | 81 ± 6.0 | <3 |
| TFT 3 | 0 | 38.1 ± 2.5 | 6.60 ± 0.09 | 69 ± 5.7 | 83 ± 5.7 | <3 |
| TFT 4 | 0 | 37.6 ± 2.2 | 6.52 ± 0.06 | 70 ± 4.2 | 77 ± 6.4 | <3 |
| AFTER TFT INSTALLATION (September 2017–July 2019) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0.31 ± 0.06 | 53.2 ± 1.6 | 6.59 ± 0.18 | 59 ± 3.1 | 74 ± 3 | <3 |
| R | 0.23 ± 0.04 | 49.6 ± 2.1 | 6.60 ± 0.11 | 77 ± 3.6 | 81 ± 4.1 | <3 |
| P1 | 0.29 ± 0.09 | 47.4 ± 1.2 | 6.56 ± 0.09 | 63 ± 2.9 | 79 ± 3.3 | <3 |
| P2 | 0.27 ± 0.09 | 45.5 ± 3.2 | 6.5 ± 0.08 | 77 ± 4.2 | 66 ± 4.2 | <3 |
| TFT 1 | 0.26 ± 0.08 | 48.6 ± 2.5 | 6.58 ± 0.13 | 68 ± 3.8 | 78 ± 2.6 | <3 |
| TFT 2 | 0.24 ± 0.04 | 49.8 ± 1.5 | 6.51 ± 0.10 | 71 ± 4.6 | 82 ± 5.2 | <3 |
| TFT 3 | 0.26 ± 0.05 | 48.3 ± 2.1 | 6.61 ± 0.07 | 74 ± 2.3 | 84 ± 3.9 | <3 |
| TFT 4 | 0.26 ± 0.08 | 47.3 ± 2.2 | 6.58 ± 0.09 | 75 ± 3.3 | 79 ± 3.6 | <3 |
| BEFORE TFT INSTALLATION (April 2018) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0 | 43.5 ± 8.4 | 6.72 ± 0.18 | 66 ± 2.2 | 64 ± 1.1 | <3 |
| R | 0 | 37.6 ± 9.2 | 6.70 ± 0.19 | 68 ± 2.1 | 66 ± 1.6 | <3 |
| P1 | 0 | 39.2 ± 5.4 | 6.74 ± 0.21 | 61 ± 1.9 | 64 ± 1.0 | <3 |
| P2 | 0 | 35.8 ± 3.9 | 6.80 ± 0.20 | 78 ± 1.1 | 80 ± 0.9 | <3 |
| TFT 1 | 0 | 38.8 ± 4.8 | 6.78 ± 0.16 | 79 ± 1.8 | 85 ± 0.7 | <3 |
| TFT 2 | 0 | 39.4 ± 5.9 | 6.78 ± 0.19 | 65 ± 2.0 | 87 ± 1.2 | <3 |
| TFT 3 | 0 | 39.1 ± 4.6 | 6.76 ± 0.16 | 69 ± 2.2 | 86 ± 1.3 | <3 |
| TFT 4 | 0 | 36.9 ± 6.9 | 6.80 ± 0.25 | 73 ± 2.1 | 79 ± 1.0 | <3 |
| AFTER TFT INSTALLATION (April 2018–July 2019) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0.35 ± 0.07 | 51.4 ± 1.7 | 6.79 ± 0.11 | 74 ± 0.19 | 63 ± 0.19 | <3 |
| R | 0.26 ± 0.06 | 47.6 ± 1.9 | 6.70 ± 0.10 | 69 ± 0.15 | 68 ± 0.13 | <3 |
| P1 | 0.24 ± 0.09 | 47.6 ± 1.4 | 6.60 ± 0.10 | 63 ± 0.15 | 66 ± 0.14 | <3 |
| P2 | 0.23 ± 0.08 | 46.6 ± 2.1 | 6.62 ± 0.09 | 75 ± 0.11 | 81 ± 0.18 | <3 |
| TFT 1 | 0.25 ± 0.06 | 47.5 ± 2.1 | 6.68 ± 0.11 | 79 ± 0.17 | 84 ± 0.17 | <3 |
| TFT 2 | 0.27 ± 0.05 | 47.9 ± 1.6 | 6.61 ± 0.14 | 70 ± 0.12 | 82 ± 0.12 | <3 |
| TFT 3 | 0.26 ± 0.03 | 48.4 ± 1.7 | 6.64 ± 0.11 | 74 ± 0.17 | 85 ± 0.19 | <3 |
| TFT 4 | 0.27 ± 0.05 | 46.9 ± 1.8 | 6.78 ± 0.11 | 76 ± 0.18 | 81 ± 0.11 | <3 |
| BEFORE TFT INSTALLATION (November 2018) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0.10 ± 0.02 | 40.6 ± 3.1 | 6.80 ± 0.21 | 55 ± 0.8 | 54 ± 1.6 | <3 |
| R | 0.05 ± 0.01 | 35.8 ± 2.5 | 6.78 ± 0.20 | 52 ± 0.9 | 61 ± 2.1 | <3 |
| P1 | 0.1 ± 0.05 | 40.1 ± 1.8 | 6.76 ± 0.15 | 51 ± 1.1 | 53 ± 2.2 | <3 |
| P2 | 0.0 ± 0.0 | 42.6 ± 1.6 | 6.78 ± 0.17 | 60 ± 0.8 | 54 ± 2.5 | <3 |
| TFT 1 | 0.05 ± 0.01 | 41.1 ± 1.5 | 6.78 ± 0.17 | 61 ± 0.9 | 61 ± 2.6 | <3 |
| TFT 2 | 0.05 ± 0.01 | 41.9 ± 2.2 | 6.75 ± 0.15 | 56 ± 1.2 | 58 ± 3.6 | <3 |
| TFT 3 | 0.05 ± 0.01 | 42.6 ± 3.6 | 6.82 ± 0.20 | 55 ± 1.9 | 57 ± 3.4 | <3 |
| TFT 4 | 0.05 ± 0.01 | 39.8 ± 3.2 | 6.82 ± 0.19 | 56 ± 2.2 | 55 ± 5.0 | <3 |
| TFT 5 | 0.05 ± 0.02 | 39.5 ± 3.0 | 6.79 ± 0.12 | 55 ± 2.6 | 60 ± 2.9 | <3 |
| TFT 6 | 0.05 ± 0.03 | 39.1 ± 2.9 | 6.80 ± 0.16 | 54 ± 2.0 | 59 ± 2.3 | <3 |
| TFT 7 | 0.05 ± 0.01 | 32.8 ± 1.8 | 6.80 ± 0.09 | 55 ± 1.8 | 56 ± 3.3 | <3 |
| AFTER TFT INSTALLATION (November 2018–July 2019) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0.40 ± 0.01 | 50.7 ± 1.6 | 6.79 ± 0.11 | 59 ± 0.12 | 55 ± 0.12 | <3 |
| R | 0.25 ± 0.10 | 44.8 ± 1.8 | 6.70 ± 0.10 | 59 ± 0.16 | 57 ± 0.12 | <3 |
| P1 | 0.31 ± 0.10 | 46.5 ± 1.8 | 6.60 ± 0.10 | 61 ± 0.18 | 55 ± 0.11 | <3 |
| P2 | 0.33 ± 0.11 | 49.8 ± 2.2 | 6.62 ± 0.09 | 65 ± 0.13 | 63 ± 0.19 | <3 |
| TFT 1 | 0.28 ± 0.12 | 48.8 ± 2.0 | 6.68 ± 0.11 | 61 ± 0.11 | 62 ± 0.15 | <3 |
| TFT 2 | 0.30 ± 0.06 | 49.1 ± 1.8 | 6.61 ± 0.14 | 62 ± 0.10 | 68 ± 0.14 | <3 |
| TFT 3 | 0.29 ± 0.05 | 48.8 ± 1.6 | 6.64 ± 0.11 | 63 ± 0.11 | 74 ± 0.10 | <3 |
| TFT 4 | 0.28 ± 0.03 | 47.6 ± 1.6 | 6.78 ± 0.11 | 66 ± 0.13 | 61 ± 0.09 | <3 |
| TFT 5 | 0.32 ± 0.04 | 48.6 ± 1.2 | 6.80 ± 0.10 | 61 ± 0.09 | 69 ± 0.11 | <3 |
| TFT 6 | 0.29 ± 0.06 | 47.6 ± 1.4 | 6.69 ± 0.10 | 59 ± 0.13 | 64 ± 0.11 | <3 |
| TFT 7 | 0.30 ± 0.02 | 47.2 ± 1.3 | 6.72 ± 0.11 | 59 ± 0.12 | 66 ± 0.08 | <3 |
| BEFORE TFT INSTALLATION (February 2019) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0 | 39.9 ± 3.2 | 6.81 ± 0.10 | 77 ± 0.50 | 69 ± 0.18 | <3 |
| R | 0 | 37.1 ± 2.5 | 6.78 ± 0.12 | 81 ± 0.60 | 68 ± 0.20 | <3 |
| P1 | 0 | 35.4 ± 2.9 | 6.76 ± 0.13 | 76 ± 0.62 | 64 ± 0.16 | <3 |
| P2 | 0 | 34.6 ± 2.6 | 6.80 ± 0.09 | 74 ± 0.41 | 78 ± 0.17 | <3 |
| TFT 1 | 0 | 33.2 ± 1.8 | 6.79 ± 0.18 | 75 ± 0.52 | 74 ± 0.24 | <3 |
| TFT 2 | 0 | 32.4 ± 4.2 | 6.78 ± 0.20 | 71 ± 0.19 | 79 ± 0.20 | <3 |
| TFT 3 | 0 | 33.3 ± 3.6 | 6.77 ± 0.15 | 79 ± 0.20 | 75 ± 0.19 | <3 |
| TFT 4 | 0 | 34.5 ± 2.7 | 6.74 ± 0.11 | 81 ± 0.16 | 76 ± 0.11 | <3 |
| AFTER TFT INSTALLATION (February 2019–July 2019) | ||||||
| SAMPLING POINT | TOTAL CHLORINE (Mean ± SD mg/L) | TEMPERATURE (Mean ± SD °C) | pH (Mean ± SD) | IRON IONS (Mean ± SD µg/L) | ZINC IONS (Mean ± SD µg/L) | THMs (µg/L) |
| B | 0.36 ± 0.04 | 50.1 ± 1.9 | 6.83 ± 0.08 | 80 ± 0.14 | 73 ± 0.17 | <3 |
| R | 0.30 ± 0.04 | 46.4 ± 2.2 | 6.79 ± 0.08 | 83 ± 0.12 | 71 ± 0.15 | <3 |
| P1 | 0.34 ± 0.07 | 48.5 ± 1.3 | 6.79 ± 0.09 | 79 ± 0.09 | 72 ± 0.18 | <3 |
| P2 | 0.29 ± 0.09 | 49.9 ± 2.0 | 6.78 ± 0.09 | 76 ± 0.10 | 80 ± 0.19 | <3 |
| TFT 1 | 0.31 ± 0.07 | 48.9 ± 2.1 | 6.81 ± 0.06 | 79 ± 0.15 | 80 ± 0.19 | <3 |
| TFT 2 | 0.32 ± 0.04 | 48.2 ± 1.9 | 6.70 ± 0.08 | 76 ± 0.14 | 79 ± 0.18 | <3 |
| TFT 3 | 0.29 ± 0.02 | 47.9 ± 2.3 | 6.72 ± 0.09 | 83 ± 0.19 | 78 ± 0.16 | <3 |
| TFT 4 | 0.28 ± 0.06 | 48.2 ± 2.2 | 6.79 ± 0.08 | 76 ± 0.18 | 77 ± 0.19 | <3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Totaro, M.; Mariotti, T.; Bisordi, C.; De Vita, E.; Valentini, P.; Costa, A.L.; Casini, B.; Privitera, G.; Baggiani, A. Evaluation of Legionella pneumophila Decrease in Hot Water Network of Four Hospital Buildings after Installation of Electron Time Flow Taps. Water 2020, 12, 210. https://doi.org/10.3390/w12010210
Totaro M, Mariotti T, Bisordi C, De Vita E, Valentini P, Costa AL, Casini B, Privitera G, Baggiani A. Evaluation of Legionella pneumophila Decrease in Hot Water Network of Four Hospital Buildings after Installation of Electron Time Flow Taps. Water. 2020; 12(1):210. https://doi.org/10.3390/w12010210
Chicago/Turabian StyleTotaro, Michele, Tommaso Mariotti, Costanza Bisordi, Erica De Vita, Paola Valentini, Anna Laura Costa, Beatrice Casini, Gaetano Privitera, and Angelo Baggiani. 2020. "Evaluation of Legionella pneumophila Decrease in Hot Water Network of Four Hospital Buildings after Installation of Electron Time Flow Taps" Water 12, no. 1: 210. https://doi.org/10.3390/w12010210
APA StyleTotaro, M., Mariotti, T., Bisordi, C., De Vita, E., Valentini, P., Costa, A. L., Casini, B., Privitera, G., & Baggiani, A. (2020). Evaluation of Legionella pneumophila Decrease in Hot Water Network of Four Hospital Buildings after Installation of Electron Time Flow Taps. Water, 12(1), 210. https://doi.org/10.3390/w12010210






