Spatial Variation of Metallic Contamination and Its Ecological Risk in Sediment and Freshwater Mollusk: Melanoides tuberculata (Müller, 1774) (Gastropoda: Thiaridae)
Abstract
1. Introduction
2. Materials and Methods
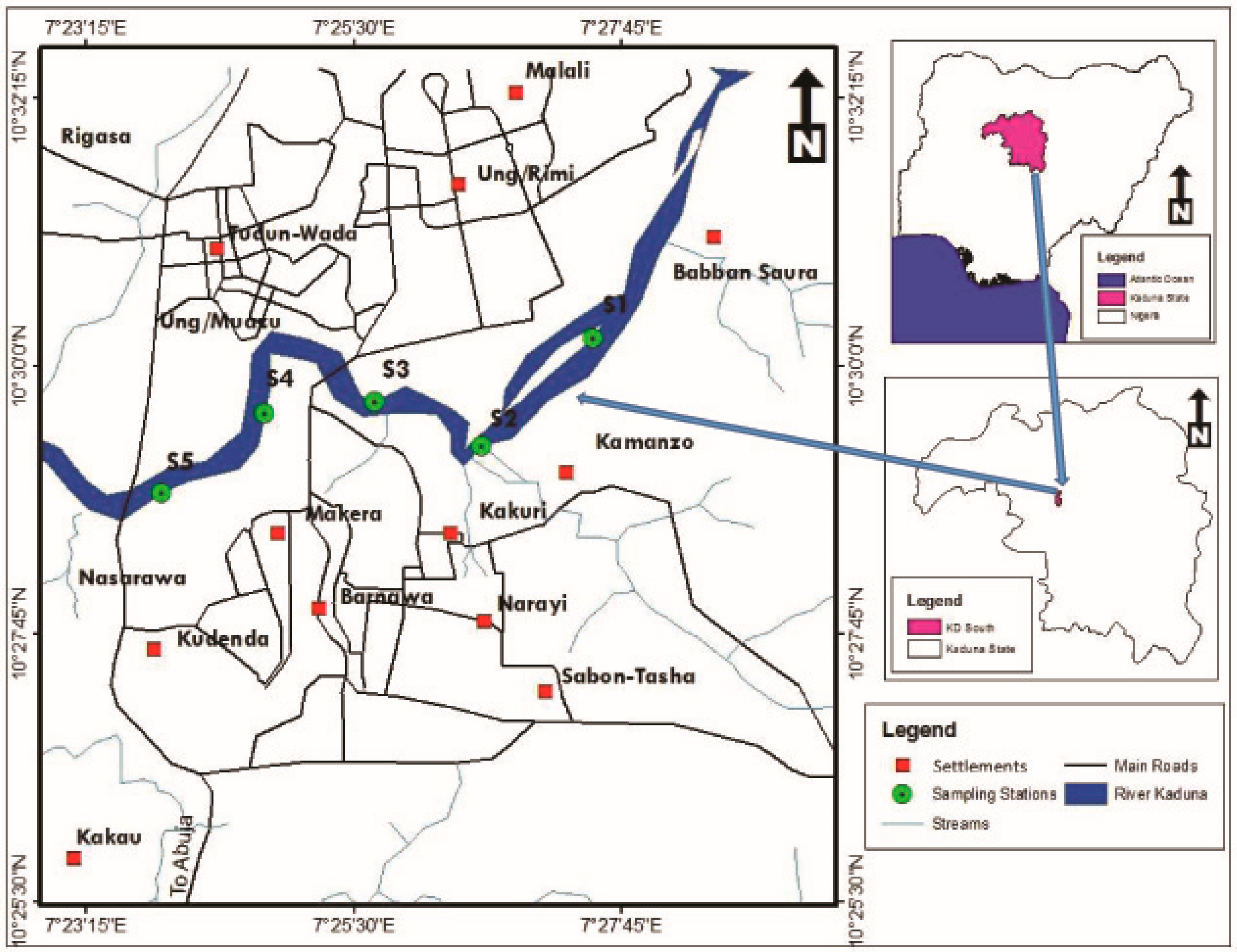
2.1. Study Area
2.2. Sample Collection
2.3. Determination of Metallic Pollutants (MPs)
2.4. Determination of Antioxidant Enzymes Activities
2.4.1. Determination of Catalase (CAT) Activity
2.4.2. Determination of AChE Activity
2.4.3. Determination of Glutathione-S-Transferase (GST)
2.5. Sediment Pollution and Potential Ecological Risk
2.6. Data Analysis
3. Results
3.1. Spatial Distribution of Metallic Pollutants in Sediment and Freshwater Mollusc
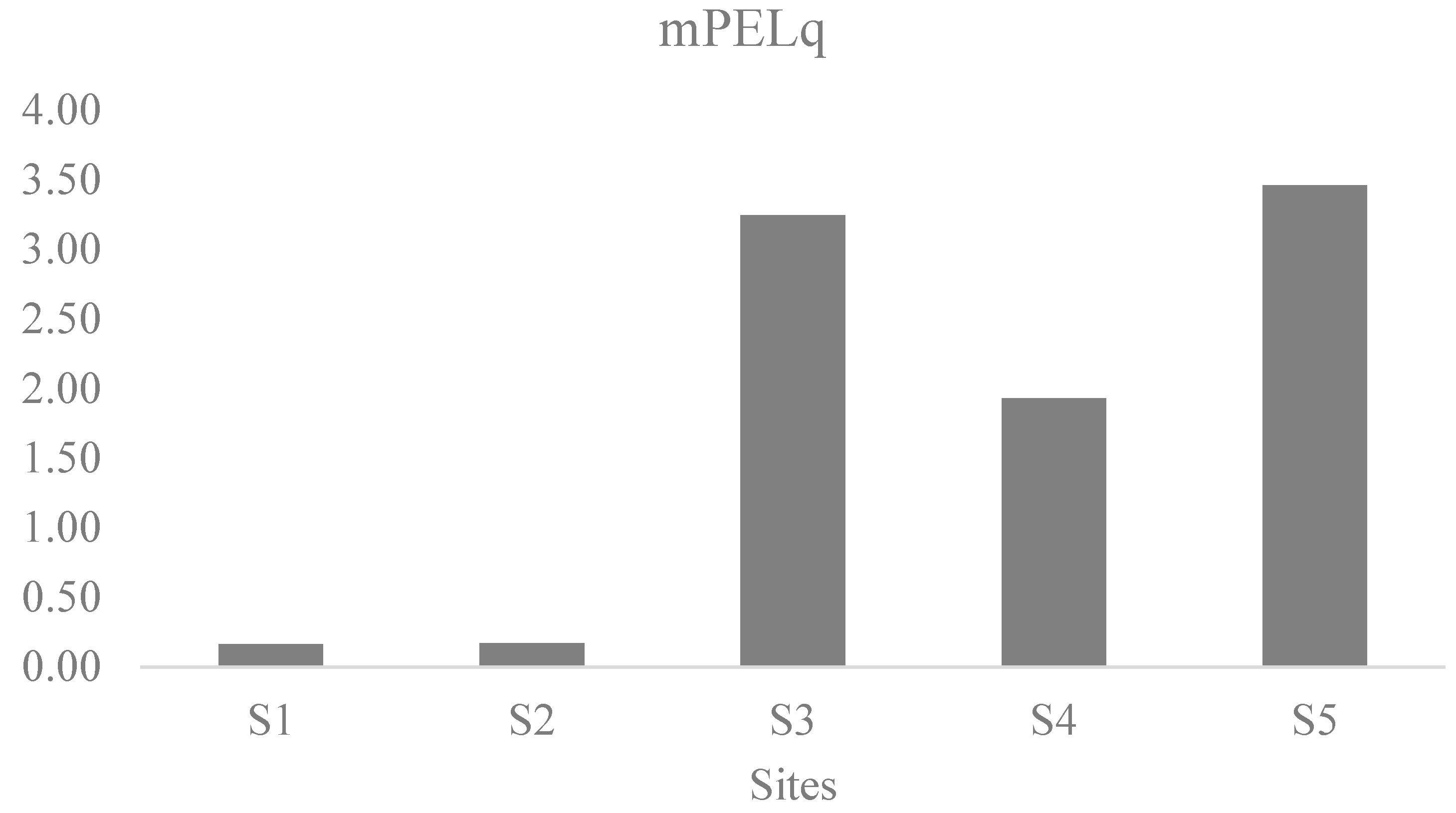
3.2. Enrichment Factors of MPs
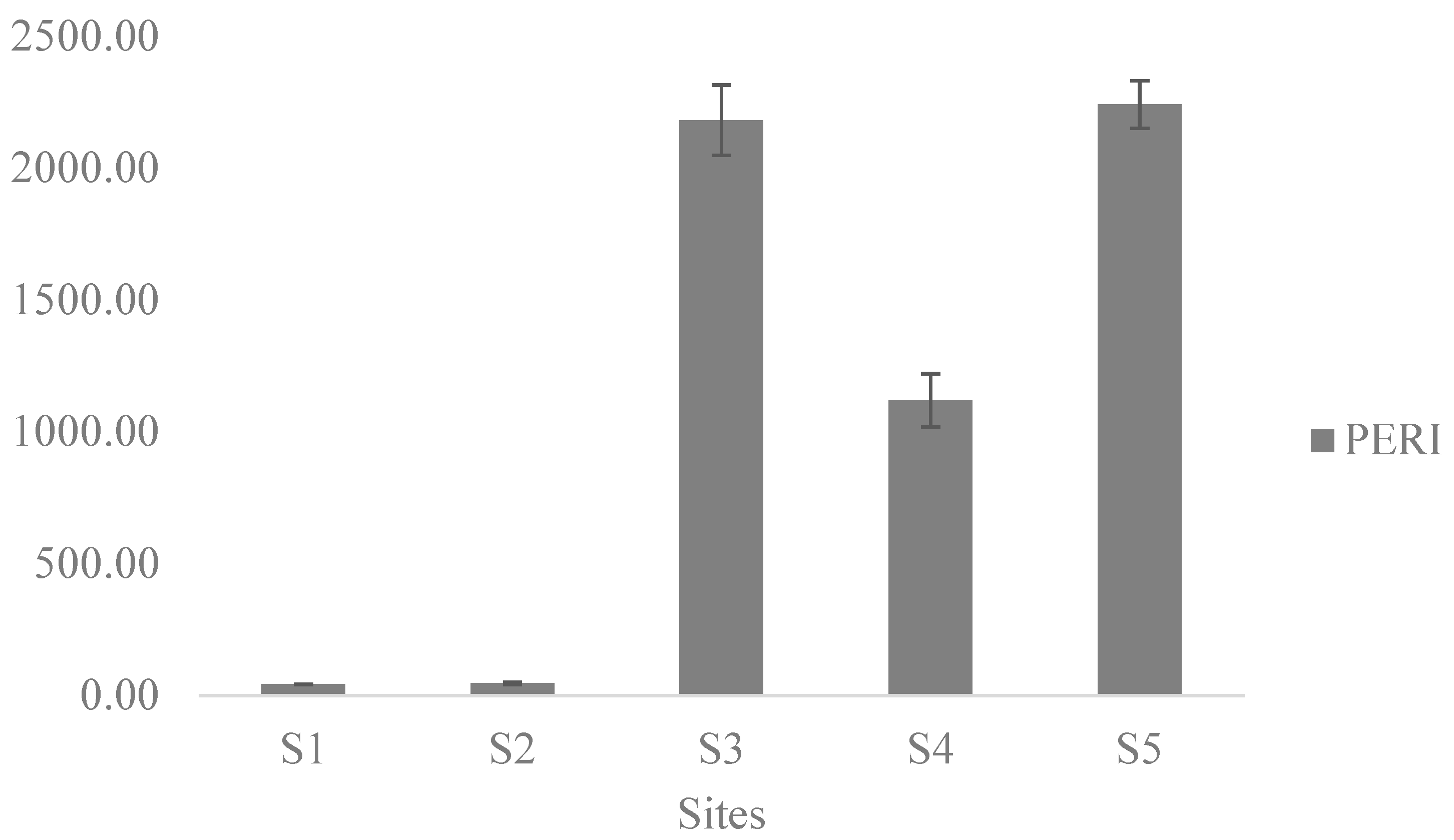
3.3. Potential Ecological Risk Assessment of PHTEs
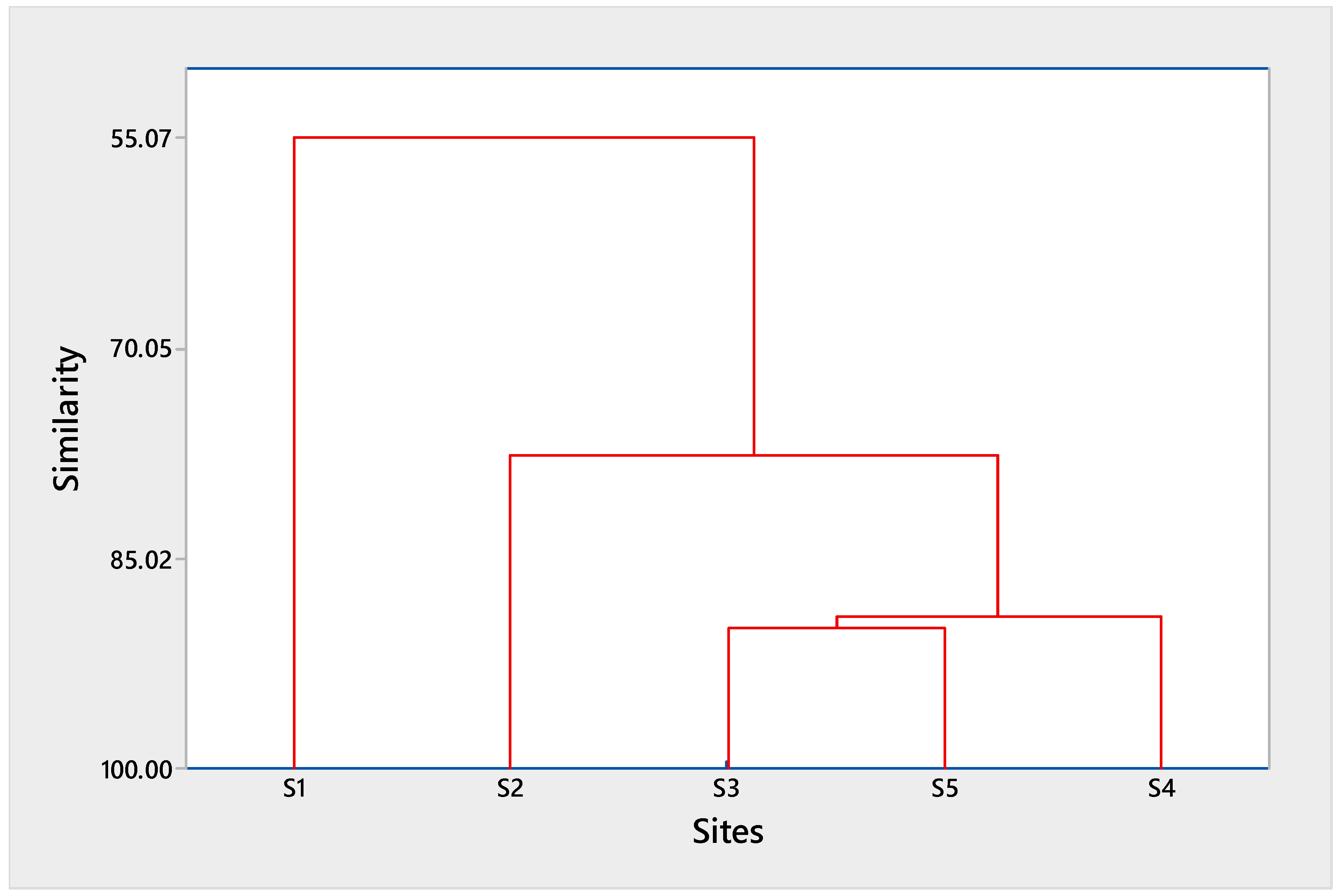
3.4. Identification of Pollution Sources
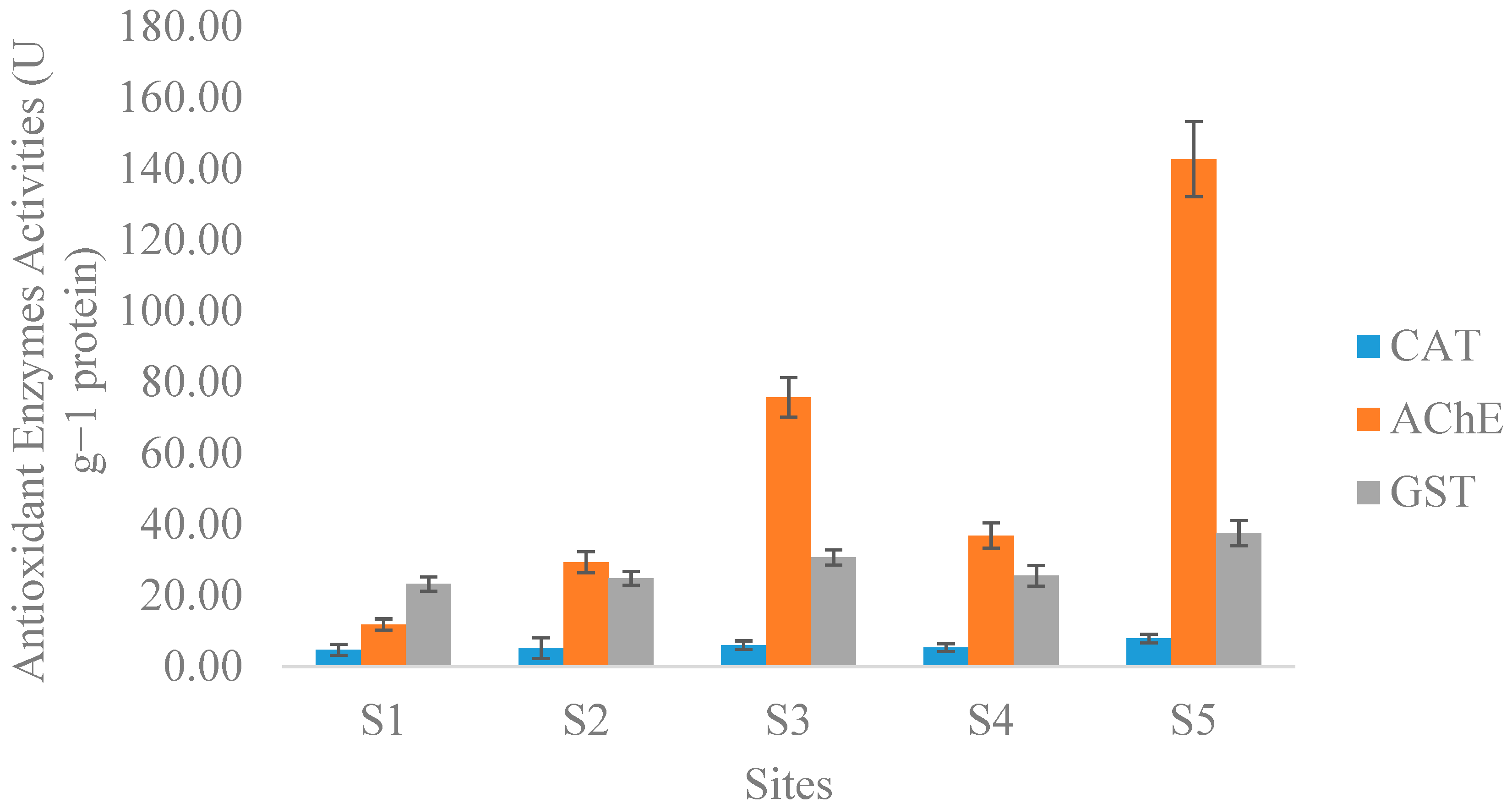
3.5. Biomarkers of Metal Pollution in Freshwater Mollusc
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- El Zrelli, R.; Courjault-Radé, P.; Rabaoui, L.; Castet, S.; Michel, S.; Bejaoui, N. Heavy metal contamination and ecological risk assessment in the surface sediments of the coastal area surrounding the industrial complex of Gabes city, Gulf of Gabes, SE Tunisia. Mar. Pollut. Bull. 2015, 101, 922–929. [Google Scholar] [CrossRef]
- Xu, M.; Sun, W.; Wang, R. Spatial Distribution and Ecological Risk Assessment of Potentially Harmful Trace Elements in Surface Sediments from Lake Dali, North China. Water 2019, 11, 2544. [Google Scholar] [CrossRef]
- Gao, X.; Chen, C.T.A. Heavy metal pollution status in surface sediments of the coastal Bohai Bay. Water Res. 2012, 46, 1901–1911. [Google Scholar] [CrossRef]
- Maanan, M.; Saddik, M.; Maanan, M.; Chaibi, M.; Assobhei, O.; Zourarah, B. Environmental and ecological risk assessment of heavy metals in sediments of Nador lagoon, Morocco. Ecol. Indic. 2015, 48, 616–626. [Google Scholar] [CrossRef]
- Bastami, K.D.; Neyestani, M.R.; Shemirani, F.; Soltani, F.; Haghparast, S.; Akbari, A. Heavy metal pollution assessment in relation to sediment properties in the coastal sediments of the southern Caspian Sea. Mar. Pollut. Bull. 2015, 92, 237–243. [Google Scholar] [CrossRef]
- Islam, M.S.; Ahmed, M.K.; Raknuzzaman, M.; Habibullah-Al-Mamun, M.; Islam, M.K. Heavy metal pollution in surface water and sediment: a preliminary assessment of an urban river in a developing country. Ecol. Indic. 2015, 48, 282–291. [Google Scholar] [CrossRef]
- Yu, R.; Yuan, X.; Zhao, Y.; Hu, G.; Tu, X. Heavy metal pollution in intertidal sediments from Quanzhou Bay, China. J. Environ. Sci. 2008, 20, 664–669. [Google Scholar] [CrossRef]
- Li, C.; Song, C.; Yin, Y.; Sun, M.; Tao, P.; Shao, M. Spatial distribution and risk assessment of heavy metals in sediments of Shuangtaizi estuary, China. Mar. Pollut. Bull. 2015, 98, 358–364. [Google Scholar] [CrossRef]
- Bian, B.; Zhou, Y.; Fang, B.B. Distribution of heavy metals and benthic macroinvertebrates: Impacts from typical inflow river sediments in the Taihu Basin, China. Ecol. Indic. 2016, 69, 348–359. [Google Scholar] [CrossRef]
- Brady, J.P.; Ayoko, G.A.; Martens, W.N.; Goonetilleke, A. Enrichment, distribution and sources of heavy metals in the sediments of Deception Bay, Queensland, Australia. Mar. Pollut. Bull. 2014, 81, 248–255. [Google Scholar] [CrossRef]
- Dutta, S.M.; Mustafi, S.B.; Raha, S.; Chakraborty, S.K. Biomonitoring role of some cellular markers during heat stress-induced changes in highly representative freshwater mollusc, Bellamya bengalensis: Implication in climate change and biological adaptation. Ecotoxicol. Environ. Saf. 2018, 157, 482–490. [Google Scholar] [CrossRef]
- Lushchak, V.I. Environmentally induced oxidative stress in aquatic animals. Aquat. Toxicol. 2011, 101, 13–30. [Google Scholar] [CrossRef]
- Bai, J.; Cui, B.; Chen, B.; Zhang, K.; Deng, W.; Gao, H.; Xiao, R. Spatial distribution and ecological risk assessment of heavy metals in surface sediments from a typical plateau lake wetland, China. Ecol. Model. 2011, 222, 301–306. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, L.; Kong, L.; Liu, E.; Wang, L.; Zhu, J. Spatial distribution, ecological risk assessment and source identification for heavy metals in surface sediments from Dongping Lake, Shandong, East China. Catena 2015, 125, 200–205. [Google Scholar] [CrossRef]
- Ayanda, I.O.; Ekhator, U.I.; Bello, O.A. Determination of selected heavy metal and analysis of proximate composition in some fish species from Ogun River, Southwestern Nigeria. Heliyon 2019, 5, e02512. [Google Scholar] [CrossRef]
- Kolo, R.; Oladimeji, A. Water quality and some nutrient levels in Shiroro Lake, Niger State, Nigeria. J. Aquat. Sci. 2004, 19, 99–106. [Google Scholar]
- Arimoro, F.O.; Auta, Y.I.; Odume, O.N.; Keke, U.N.; Mohammed, A.Z. Mouthpart deformities in Chironomidae (Diptera) as bioindicators of heavy metals pollution in Shiroro Lake, Niger State, Nigeria. Ecotoxicol. Environ. Saf. 2018, 149, 96–100. [Google Scholar] [CrossRef]
- NIMET. Nigeria Meteorological Agency. Nigeria, Kaduna, Kaduna, Nigeria: 2010; NIMET: Kaduna, Nigeria, 2010.
- KEPA. Kaduna State Environmental Protection Authority, Revised in 1998:1998, KEPA: Kaduna, Nigeria, 1998.
- Brown, D.; Kristensen, T. A Field Guide to African Freshwater Snails, I. West African Species; Danish Bilharziasis Laboratory: Charlottenlund, Denmark, 1993; p. 32. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Method 3051 A. Microwave assisted acid digestion of sediments, sludge’s, soils and oils. Test. Methods Eval. Solid Waste 2007, 1–30. Available online: http://www.epa.gov/SW-846/pdfs/3051a.pdf (accessed on 26 June 2019).
- Bakshi, M.; Ghosh, S.; Chakraborty, D.; Hazra, S.; Chaudhuri, P. Assessment of potentially toxic metal (PTM) pollution in mangrove habitats using biochemical markers: A case study on Avicennia officinalis L. in and around Sundarban, India. Mar. Pollut. Bull. 2018, 133, 157–172. [Google Scholar] [CrossRef]
- Banaee, M.; Sureda, A.; Taheri, S.; Hedayatzadeh, F. Sub-lethal effects of dimethoate alone and in combination with cadmium on biochemical parameters in freshwater snail, Galba truncatula. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019, 220, 62–70. [Google Scholar] [CrossRef]
- Chance, B.; Maehly, A. [136] Assay of catalases and peroxidases. Methods Enzymol. 1955, 2, 764–775. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar]
- Abrahim, G.; Parker, R. Assessment of heavy metal enrichment factors and the degree of contamination in Mar. sediments from Tamaki Estuary, Auckland, New Zealand. Environ. Monit. Assess. 2008, 136, 227–238. [Google Scholar] [CrossRef]
- Hakanson, L. An ecological risk index for aquatic pollution control. A sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Long, E.; MacDonald, D. Recommended uses of empirically derived, sediment quality guidelines for Mar. and estuarine ecosystems. Hum. Ecol. Risk Assess. 1998, 4, 1019–1039. [Google Scholar] [CrossRef]
- MacDonald, D.D.; Ingersoll, C.G.; Berger, T. Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch. Environ. Contam. Toxicol. 2000, 39, 20–31. [Google Scholar] [CrossRef]
- Long, E.R.; Field, L.J.; MacDonald, D.D. Predicting toxicity in Mar. sediments with numerical sediment quality guidelines. Environ. Toxicol. Chem. Int. J. 1998, 17, 714–727. [Google Scholar] [CrossRef]
- Long, E.R.; MacDonald, D.D.; Severn, C.G.; Hong, C.B. Classifying probabilities of acute toxicity in Mar. sediments with empirically derived sediment quality guidelines. Environ. Toxicol. Chem. Int. J. 2000, 19, 2598–2601. [Google Scholar] [CrossRef]
- Sutherland, R. Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environ. Geol. 2000, 39, 611–627. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, J.; Xiong, K.; Huang, X.; Duan, S. Distribution of heavy metals in core sediments from Baihua Lake. Procedia Environ. Sci. 2012, 16, 51–58. [Google Scholar] [CrossRef]
- Ke, X.; Gui, S.; Huang, H.; Zhang, H.; Wang, C.; Guo, W. Ecological risk assessment and source identification for heavy metals in surface sediment from the Liaohe River protected area, China. Chemosphere 2017, 175, 473–481. [Google Scholar] [CrossRef]
- Kükrer, S.; Şeker, S.; Abacı, Z.T.; Kutlu, B. Ecological risk assessment of heavy metals in surface sediments of northern littoral zone of Lake Çıldır, Ardahan, Turkey. Environ. Monit. Assess. 2014, 186, 3847–3857. [Google Scholar] [CrossRef]
- Asaeda, T.; Barnuevo, A. Oxidative stress as an indicator of niche-width preference of mangrove Rhizophora stylosa. Forest Ecol. Manag. 2019, 432, 73–82. [Google Scholar] [CrossRef]
- Bakshi, M.; Ram, S.; Ghosh, S.; Chakraborty, A.; Sudarshan, M.; Chaudhuri, P. Micro-spatial variation of elemental distribution in estuarine sediment and their accumulation in mangroves of Indian Sundarban. Environ. Monit. Assess. 2017, 189, 221. [Google Scholar] [CrossRef]
- Ghosh, S.; Ram, S.S.; Bakshi, M.; Chakraborty, A.; Sudarshan, M.; Chaudhuri, P. Vertical and horizontal variation of elemental contamination in sediments of Hooghly Estuary, India. Mar. Pollut. Bull. 2016, 109, 539–549. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, S.; Xie, Y.; Wang, X.; Sheng, X.; Chen, J. Distribution and pollution character of heavy metals in the surface sediments of Liao River. Huanjing Kexue 2008, 29, 2413–2418. [Google Scholar]
- Lin, Q.; Liu, E.; Zhang, E.; Nath, B.; Shen, J.; Yuan, H.; Wang, R. Reconstruction of atmospheric trace metals pollution in Southwest China using sediments from a large and deep alpine lake: historical trends, sources and sediment focusing. Sci. Total Environ. 2018, 613, 331–341. [Google Scholar] [CrossRef]
- Wan, D.; Song, L.; Mao, X.; Yang, J.; Jin, Z.; Yang, H. One-century sediment records of heavy metal pollution on the southeast Mongolian Plateau: Implications for air pollution trend in China. Chemosphere 2019, 220, 539–545. [Google Scholar] [CrossRef]
- Zeng, H.; Wu, J.; Liu, W. Two-century sedimentary record of heavy metal pollution from Lake Sayram: A deep mountain lake in central Tianshan, China. Quat. Int. 2014, 321, 125–131. [Google Scholar] [CrossRef]
- Wan, D.; Song, L.; Yang, J.; Jin, Z.; Zhan, C.; Mao, X.; Liu, D.; Shao, Y. Increasing heavy metals in the background atmosphere of central North China since the 1980s: Evidence from a 200-year lake sediment record. Atmos. Environ. 2016, 138, 183–190. [Google Scholar] [CrossRef]
- Antizar-Ladislao, B.; Mondal, P.; Mitra, S.; Sarkar, S.K. Assessment of trace metal contamination level and toxicity in sediments from coastal regions of West Bengal, eastern part of India. Mar. Pollut. Bull. 2015, 101, 886–894. [Google Scholar] [CrossRef]
- Cheng, H.; Li, M.; Zhao, C.; Yang, K.; Li, K.; Peng, M.; Yang, Z.; Liu, F.; Liu, Y.; Bai, R. Concentrations of toxic metals and ecological risk assessment for sediments of major freshwater lakes in China. J. Geochem. Explor. 2015, 157, 15–26. [Google Scholar] [CrossRef]
- Zuykov, M.; Pelletier, E.; Harper, D.A. Bivalve molluscs in metal pollution studies: from bioaccumulation to biomonitoring. Chemosphere 2013, 93, 201–208. [Google Scholar] [CrossRef]
- Chowdhury, R.; Favas, P.J.; Pratas, J.; Jonathan, M.; Ganesh, P.S.; Sarkar, S.K. Accumulation of trace metals by mangrove plants in Indian Sundarban Wetland: prospects for phytoremediation. Int. J. Phytoremediation 2015, 17, 885–894. [Google Scholar] [CrossRef]
- Rahman, M.S.; Hossain, M.B.; Babu, S.O.F.; Rahman, M.; Ahmed, A.S.; Jolly, Y.; Choudhury, T.; Begum, B.; Kabir, J.; Akter, S. Source of metal contamination in sediment, their ecological risk, and phytoremediation ability of the studied mangrove plants in ship breaking area, Bangladesh. Mar. Pollut. Bull. 2019, 141, 137–146. [Google Scholar] [CrossRef]
- Chung, C.-Y.; Chen, J.-J.; Lee, C.-G.; Chiu, C.-Y.; Lai, W.-L.; Liao, S.-W. Integrated estuary management for diffused sediment pollution in Dapeng Bay and neighboring rivers (Taiwan). Environ. Monit. Assess. 2011, 173, 499–517. [Google Scholar] [CrossRef]
- Varol, M.; Şen, B. Assessment of nutrient and heavy metal contamination in surface water and sediments of the upper Tigris River, Turkey. Catena 2012, 92, 1–10. [Google Scholar] [CrossRef]
- Gawad, S.S.A. Concentrations of heavy metals in water, sediment and mollusc gastropod, Lanistes carinatus from Lake Manzala, Egypt. Egypt. J. Aquat. Res. 2018, 44, 77–82. [Google Scholar] [CrossRef]

| Fe | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co | |
|---|---|---|---|---|---|---|---|---|---|
| Sediment | |||||||||
| S1 | 34.54 ± 9.78 | 6.36 ± 0.28 | 1.63 ± 0.17 | 4.50 ± 0.45 | 0.96 ± 0.06 | 16.09 ± 3.15 | 39.43 ± 3.88 | 2.64 ± 0.86 | 11.42 ± 2.76 |
| S2 | 58.95 ± 7.76 | 10.85 ± 2.21 | 6.52 ± 0.83 | 5.34 ± 0.06 | 0.68 ± 0.11 | 21.49 ± 3.94 | 38.59 ± 3.49 | 4.05 ± 0.56 | 62.10 ± 0.34 |
| S3 | 103.95 ± 12.26 | 73.86 ± 11.53 | 59.01 ± 0.17 | 33.53 ± 2.91 | 57.60 ± 0.45 | 72.68 ± 2.36 | 95.12 ± 3.32 | 31.95 ± 2.01 | 32.40 ± 2.88 |
| S4 | 91.35 ± 9.23 | 41.40 ± 4.31 | 30.26 ± 2.69 | 62.89 ± 0.90 | 29.42 ± 2.74 | 43.93 ± 2.74 | 69.13 ± 2.18 | 59.57 ± 0.28 | 4.61 ± 0.58 |
| S5 | 165.32 ± 15.69 | 78.98 ± 6.41 | 59.01 ± 0.17 | 61.48 ± 0.06 | 58.84 ± 0.45 | 79.20 ± 3.60 | 96.08 ± 3.60 | 60.08 ± 1.80 | 62.61 ± 2.31 |
| ERL | 70 | 120 | 5 | 80 | 35 | 30 | |||
| ERM | 390 | 270 | 9 | 145 | 110 | 50 | |||
| TEL | 35.7 | 123 | 0.596 | 37.3 | 35 | 18 | |||
| PEL | 197 | 315 | 3.53 | 90 | 91.3 | 36 | |||
| Snail | |||||||||
| S1 | 7.61 ± 1.24 | 1.24 ± 0.11 | 0.34 ± 0.01 | 1.04 ± 0.03 | 0.16 ± 0.01 | 3.58 ± 0.70 | 7.85 ± 0.05 | 0.41 ± 0.02 | 1.30 ± 0.07 |
| S2 | 19.54 ± 1.29 | 1.47 ± 0.01 | 2.39 ± 0.99 | 1.61 ± 0.04 | 3.21 ± 0.05 | 4.78 ± 0.88 | 9.36 ± 0.26 | 2.39 ± 0.13 | 2.54 ± 0.06 |
| S3 | 30.49 ± 2.76 | 15.34 ± 1.49 | 13.09 ± 0.01 | 13.76 ± 0.12 | 12.91 ± 0.15 | 15.89 ± 0.26 | 20.01 ± 0.61 | 13.36 ± 0.19 | 15.53 ± 0.35 |
| S4 | 24.09 ± 1.74 | 10.28 ± 1.70 | 7.73 ± 1.43 | 7.44 ± 0.34 | 8.15 ± 0.52 | 10.89 ± 0.75 | 14.95 ± 0.56 | 7.16 ± 0.61 | 7.20 ± 0.65 |
| S5 | 35.30 ± 3.93 | 20.55 ± 1.43 | 17.11 ± 0.04 | 19.93 ± 0.25 | 18.84 ± 0.14 | 18.74 ± 0.63 | 26.01 ± 0.14 | 15.35 ± 0.40 | 22.91 ± 0.51 |
| Fe | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co | |
|---|---|---|---|---|---|---|---|---|---|
| Sediment | |||||||||
| F | 6.933 | 4.260 | 4.519 | 4.929 | 5.024 | 4.767 | 4.750 | 4.811 | 5.059 |
| p Value | 0.028 | 0.033 | 0.023 | 0.023 | 0.013 | 0.049 | 0.047 | 0.044 | 0.042 |
| Snail | |||||||||
| F | 4.259 | 4.122 | 5.758 | 5.052 | 6.451 | 3.318 | 6.607 | 4.746 | 4.079 |
| p Value | 0.046 | 0.045 | 0.041 | 0.037 | 0.033 | 0.015 | 0.031 | 0.044 | 0.049 |
| Antioxidant Enzymes | |||||||||
| CAT | AChE | GST | |||||||
| F | 3630.706 | 17393.953 | 3650.209 | ||||||
| p Value | 0.000 | 0.000 | 0.000 | ||||||
| S1 | S2 | S3 | S4 | S5 | Mean | |
|---|---|---|---|---|---|---|
| Mn | 1.82 | 3.12 | 4.31 | 3.78 | 6.37 | 3.88 |
| Cu | 15.85 | 73.09 | 108.02 | 90.80 | 172.64 | 92.08 |
| Zn | 37.45 | 35.96 | 96.49 | 179.11 | 75.55 | 84.91 |
| Cd | 34.42 | 17.81 | 404.33 | 329.92 | 632.76 | 283.85 |
| Cr | 437.22 | 407.82 | 397.24 | 371.26 | 582.32 | 439.17 |
| Pb | 127.74 | 111.92 | 62.31 | 77.62 | 98.66 | 95.65 |
| Ni | 29.92 | 29.01 | 109.70 | 197.55 | 103.53 | 93.94 |
| Co | 43.47 | 59.84 | 489.61 | 63.33 | 366.61 | 204.57 |
| Mean | 90.99 | 92.32 | 209.00 | 164.17 | 254.80 |
| Mn_Sd | Cu_Sd | Zn_Sd | Cd_Sd | Cr_Sd | Pb_Sd | Ni_Sd | Co_Sd | Fe_S | Mn_S | Cu_S | Zn_S | Cd_S | Cr_S | Pb_S | Ni_S | Co_S | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mn_Sd | 1 | ||||||||||||||||
| Cu_Sd | 0.984 ** | 1 | |||||||||||||||
| Zn_Sd | 0.651 * | 0.571 | 1 | ||||||||||||||
| Cd_Sd | 0.980 ** | 0.995 ** | 0.587 | 1 | |||||||||||||
| Cr_Sd | 0.964 ** | 0.984 ** | 0.562 | 0.991 ** | 1 | ||||||||||||
| Pb_Sd | 0.965 ** | 0.991 ** | 0.576 | 0.994 ** | 0.980 ** | 1 | |||||||||||
| Ni_Sd | 0.533 | 0.584 | 0.587 | 0.609 | 0.612 | 0.634 * | 1 | ||||||||||
| Co_Sd | 0.282 | 0.211 | 0.147 | 0.169 | 0.233 | 0.129 | −0.234 | 1 | |||||||||
| Fe_S | 0.596 | 0.659 * | 0.502 | 0.658 * | 0.695 * | 0.686 * | 0.608 | 0.244 | 1 | ||||||||
| Mn_S | 0.990 ** | 0.981 ** | 0.661 * | 0.985 ** | 0.970 ** | 0.972 ** | 0.602 | 0.202 | 0.584 | 1 | |||||||
| Cu_S | 0.604 | 0.601 | 0.622 | 0.617 | 0.619 | 0.611 | 0.621 | 0.196 | 0.777 ** | 0.567 | 1 | ||||||
| Zn_S | 0.981 ** | 0.994 ** | 0.587 | 0.999 ** | 0.994 ** | 0.990 ** | 0.595 | 0.196 | 0.671 * | 0.984 ** | 0.619 | 1 | |||||
| Cd_S | 0.971 ** | 0.985 ** | 0.622 | 0.981 ** | 0.978 ** | 0.975 ** | 0.635 * | 0.235 | 0.728 * | 0.971 ** | 0.660 * | 0.985 ** | 1 | ||||
| Cr_S | 0.975 ** | 0.984 ** | 0.596 | 0.989 ** | 0.989 ** | 0.976 ** | 0.597 | 0.196 | 0.632 | 0.988 ** | 0.555 | 0.993 ** | 0.981 ** | 1 | |||
| Pb_S | 0.634 * | 0.626 | 0.643 * | 0.639 * | 0.639 * | 0.633 * | 0.604 | 0.235 | 0.765 ** | 0.592 | 0.994 ** | 0.638 * | 0.671 * | 0.572 | 1 | ||
| Ni_S | 0.974 ** | 0.982 ** | 0.577 | 0.988 ** | 0.990 ** | 0.972 ** | 0.583 | 0.241 | 0.672 * | 0.978 ** | 0.622 | 0.993 ** | 0.988 ** | 0.992 ** | 0.634 * | 1 | |
| Co_S | 0.982 ** | 0.998 ** | 0.545 | 0.994 ** | 0.986 ** | 0.988 ** | 0.586 | 0.223 | 0.640 * | 0.982 ** | 0.585 | 0.993 ** | 0.983 ** | 0.987 ** | 0.608 | 0.986 ** | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljahdali, M.O.; Alhassan, A.B. Spatial Variation of Metallic Contamination and Its Ecological Risk in Sediment and Freshwater Mollusk: Melanoides tuberculata (Müller, 1774) (Gastropoda: Thiaridae). Water 2020, 12, 206. https://doi.org/10.3390/w12010206
Aljahdali MO, Alhassan AB. Spatial Variation of Metallic Contamination and Its Ecological Risk in Sediment and Freshwater Mollusk: Melanoides tuberculata (Müller, 1774) (Gastropoda: Thiaridae). Water. 2020; 12(1):206. https://doi.org/10.3390/w12010206
Chicago/Turabian StyleAljahdali, Mohammed Othman, and Abdullahi Bala Alhassan. 2020. "Spatial Variation of Metallic Contamination and Its Ecological Risk in Sediment and Freshwater Mollusk: Melanoides tuberculata (Müller, 1774) (Gastropoda: Thiaridae)" Water 12, no. 1: 206. https://doi.org/10.3390/w12010206
APA StyleAljahdali, M. O., & Alhassan, A. B. (2020). Spatial Variation of Metallic Contamination and Its Ecological Risk in Sediment and Freshwater Mollusk: Melanoides tuberculata (Müller, 1774) (Gastropoda: Thiaridae). Water, 12(1), 206. https://doi.org/10.3390/w12010206






