Monitoring of Cyanobacteria in Water Using Spectrophotometry and First Derivative of Absorbance
Abstract
1. Introduction
2. Materials and Methods
2.1. Cultivation of Cyanobacteria
2.2. Preparation of Cyanobacteria Samples for Spectrophotometric Analysis
2.3. Absorbance Measurements
2.4. First Derivative Of Absorbance
2.5. Savitzky-Golay First Derivative of Absorbance
2.6. Method Detection Limit
3. Results
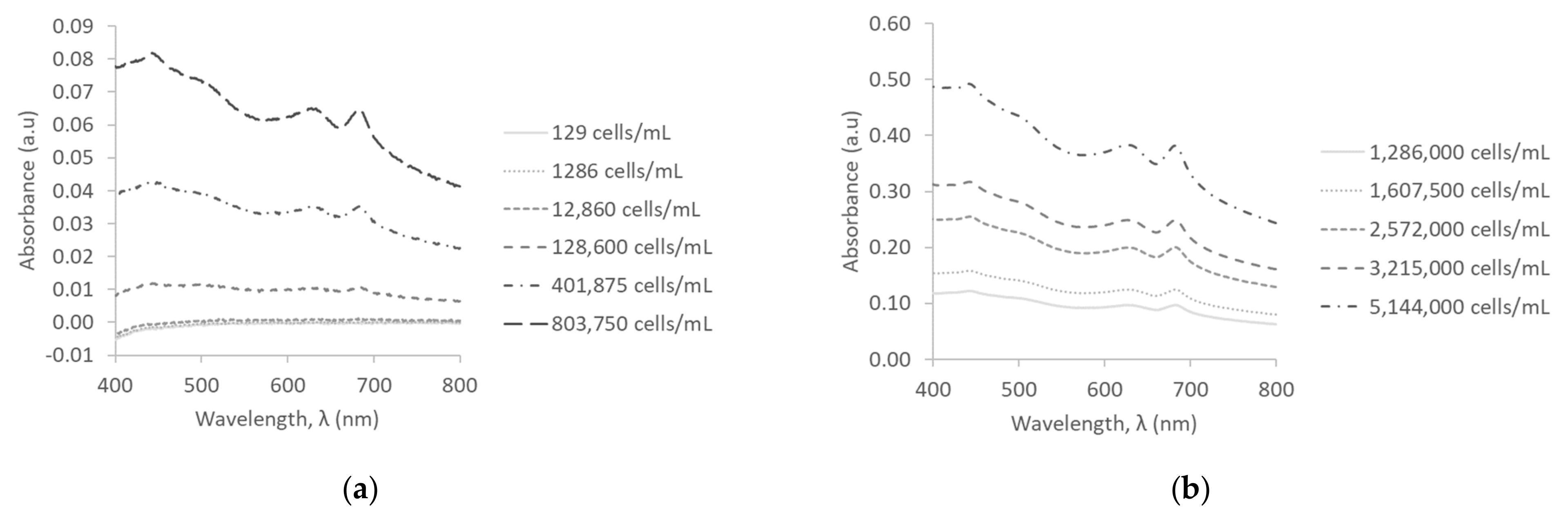
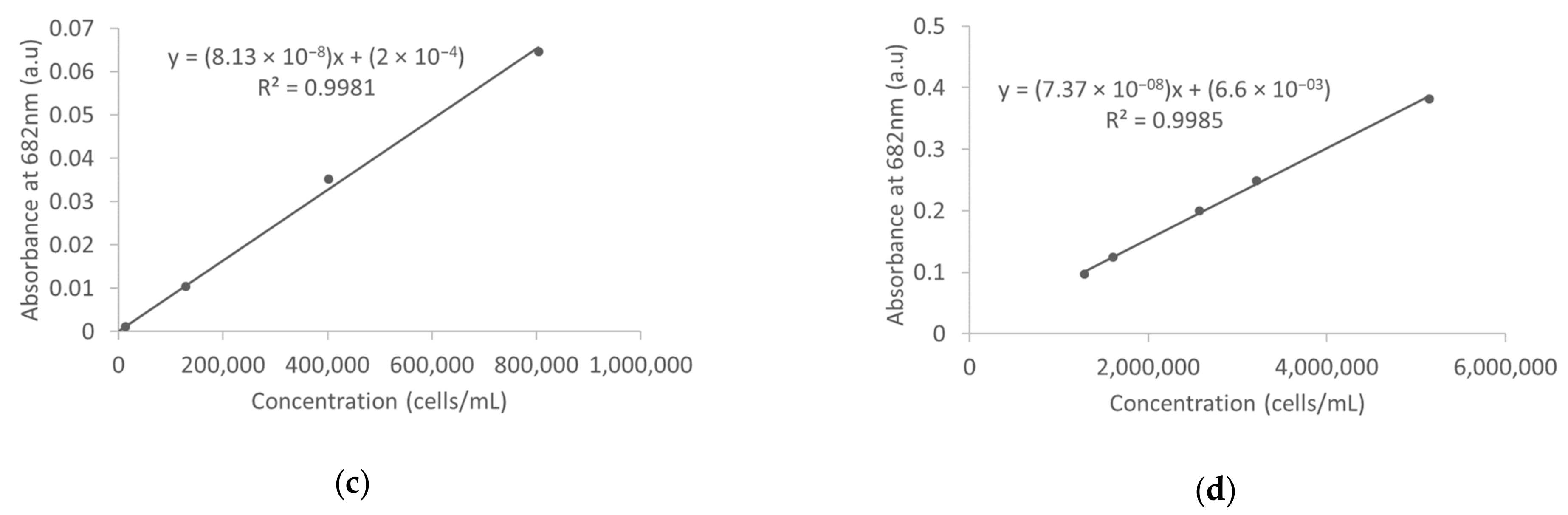
3.1. Absorbance Measurements in Deionized Water
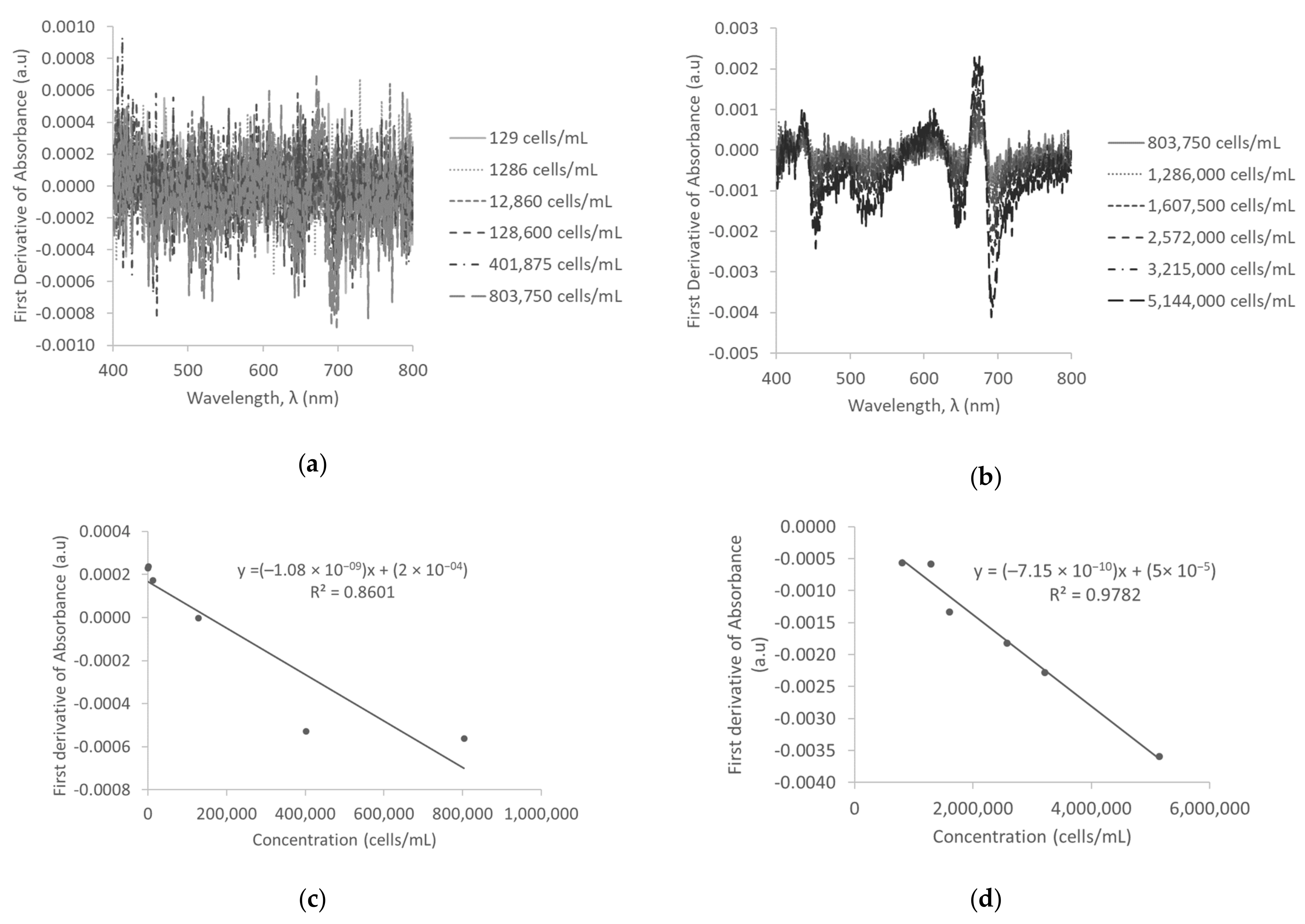
3.2. First Derivative of Absorbance in D.I. Water
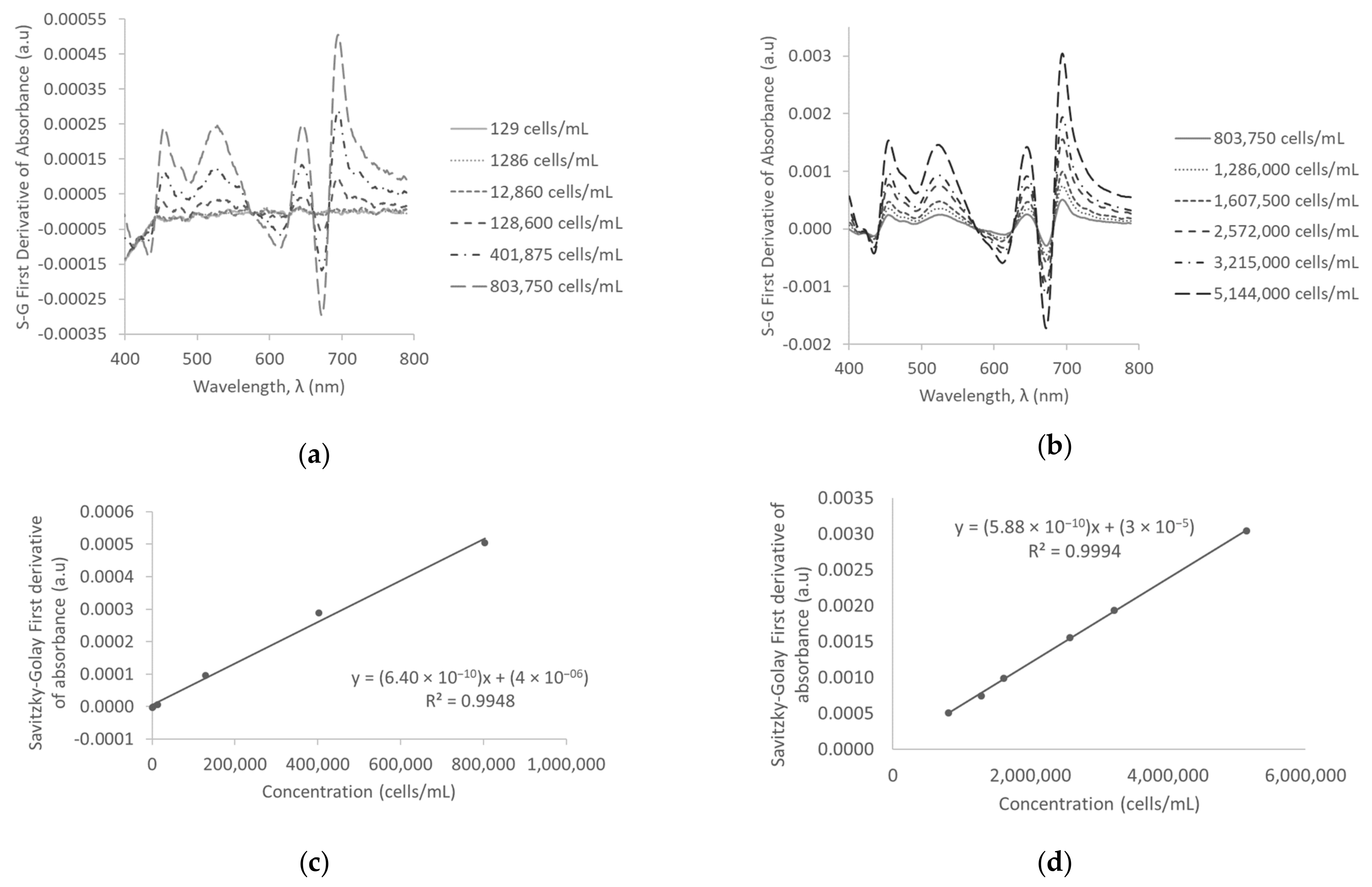
3.3. Savitzky-Golay First Derivative of Absorbance in D.I. Water
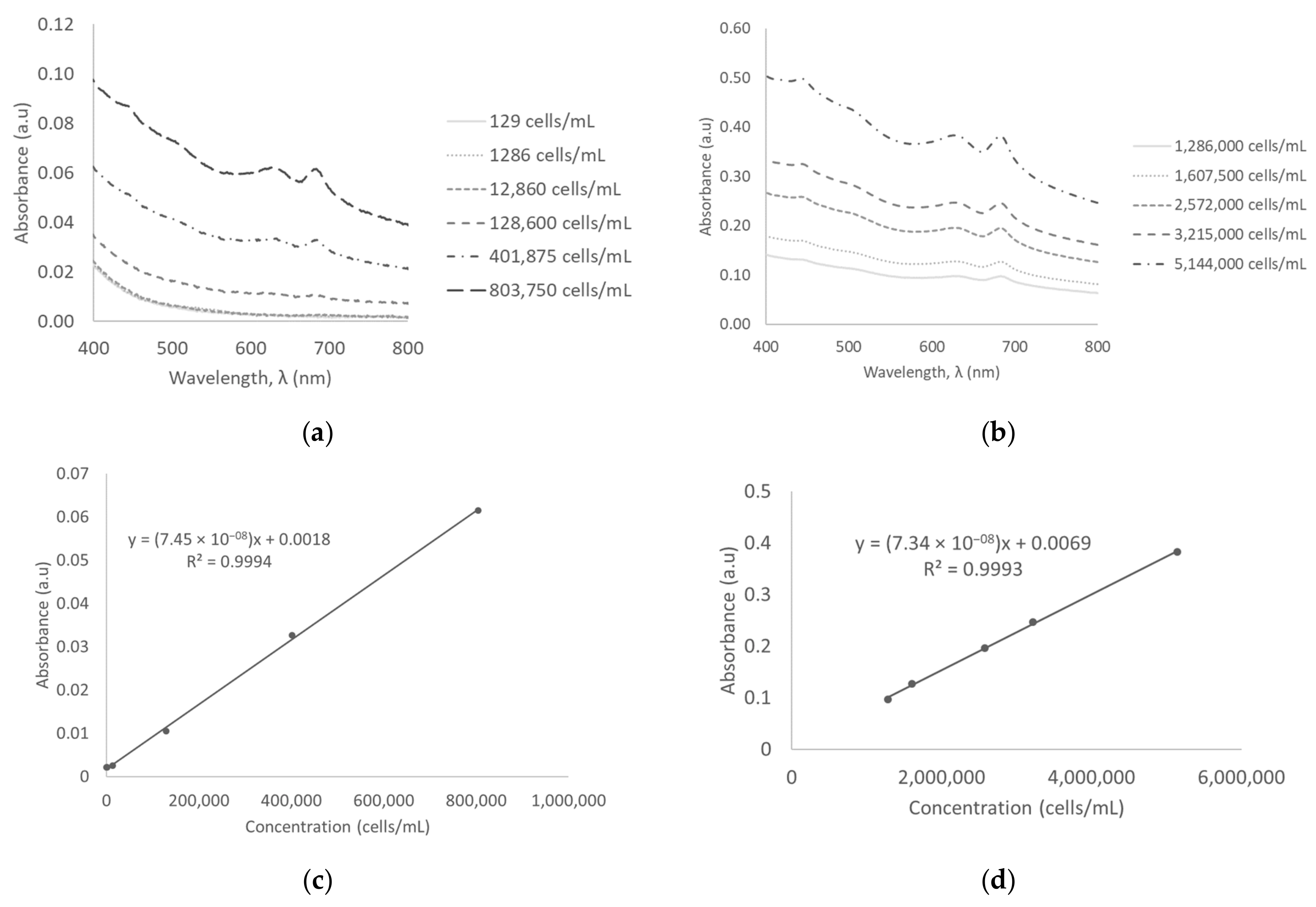
3.4. Absorbance Measurements in Surface Water
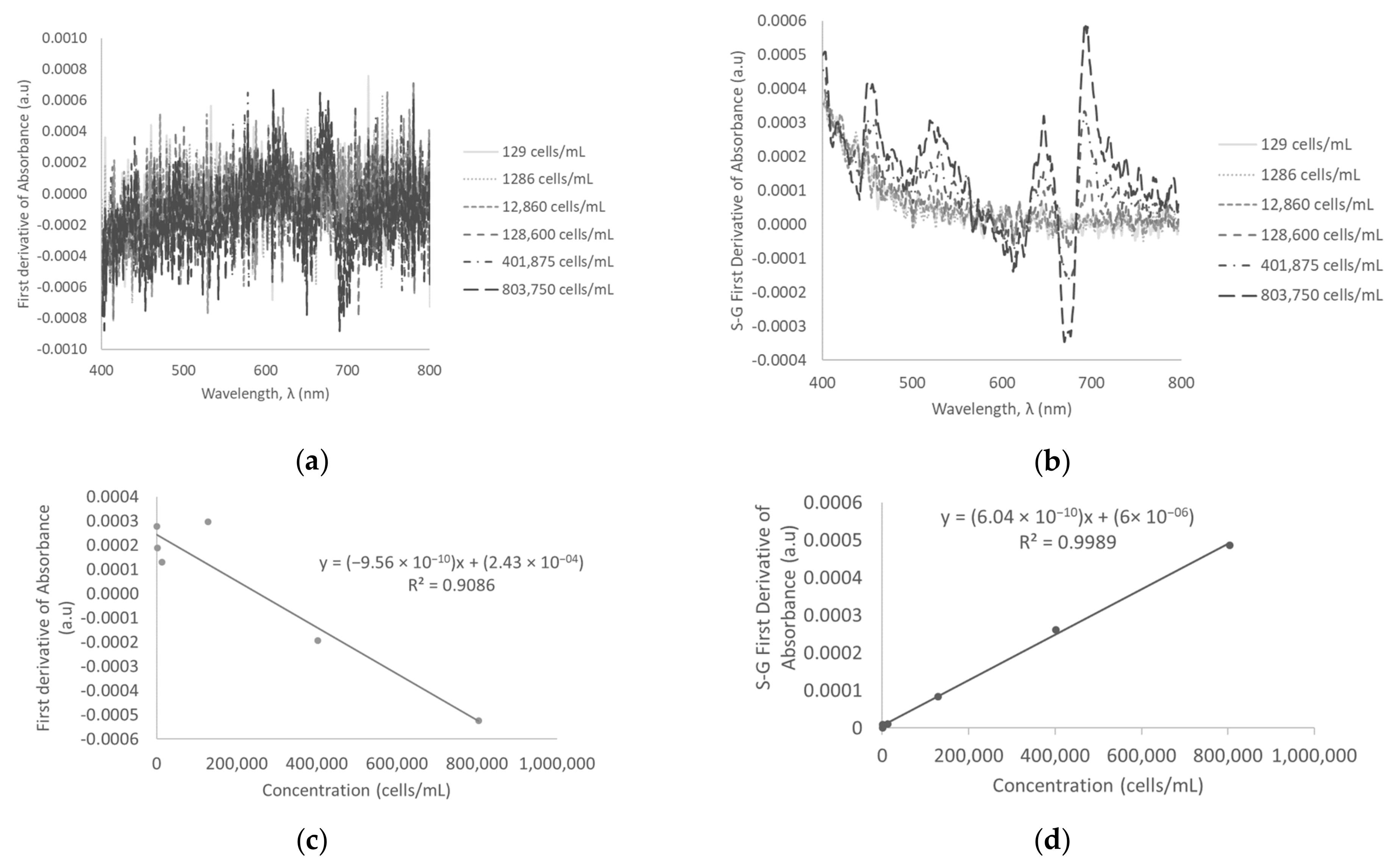
3.5. First Derivative of Absorbance and Savitzky-Golay First Derivative of Absorbance in Surface Water
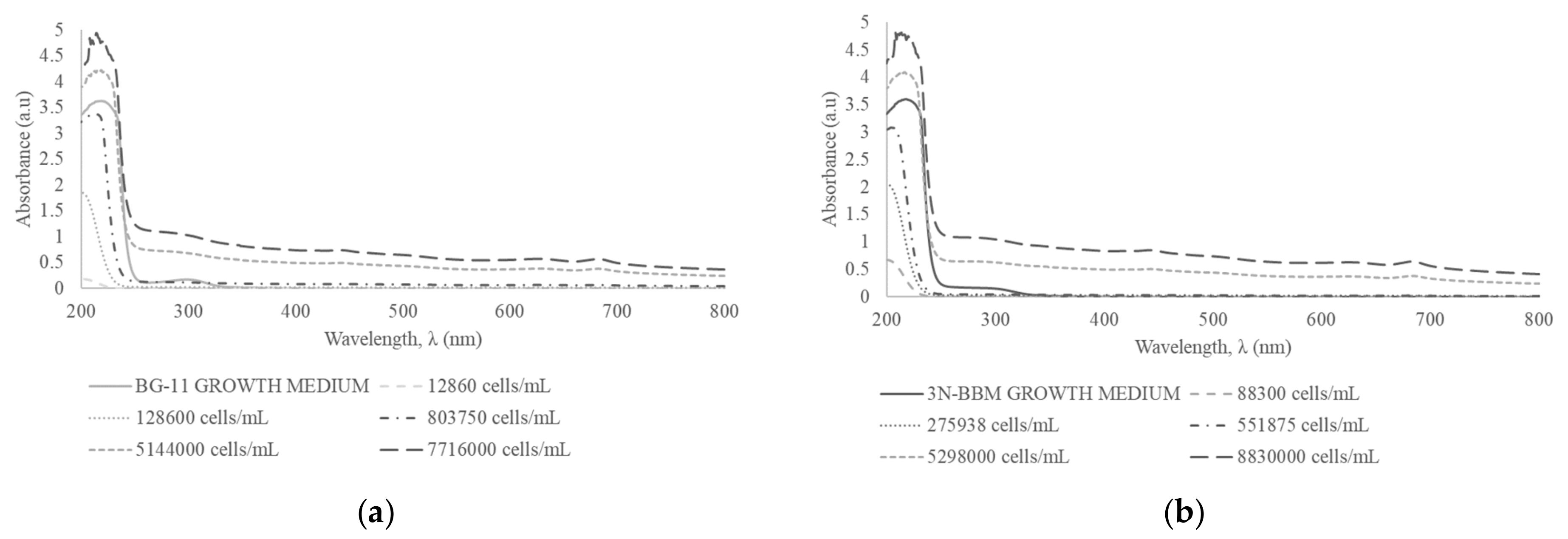
3.6. Comparison of M. aeruginosa CPCC 299 and M. aeruginosa CPCC 632 Spectra
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Falconer, I.R. An Overview of problems caused by toxic blue–green algae (cyanobacteria) in drinking and recreational water. Environ. Toxicol. 1999, 14, 5–12. [Google Scholar] [CrossRef]
- Carmichael, W.W.; Boyer, G.L. Health impacts from cyanobacteria harmful algae blooms: Implications for the North American Great Lakes. Harmful Algae 2016, 54, 194–212. [Google Scholar] [CrossRef]
- Kenefick, S.L.; Hrudey, S.E.; Prepas, E.E.; Motkosky, N.; Peterson, H.G. Odorous Substances and Cyanobacterial Toxins in Prairie Drinking Water Sources. Water Sci. Technol. 1992, 25, 147–154. [Google Scholar] [CrossRef]
- Sivonen, K.; Niemelä, S.I.; Niemi, R.M.; Lepistö, T.; Luoma, T.H.; Räsänen, L.A. Toxic cyanobacteria (blue-green algae) in Finnish fresh and coastal waters. Hydrobiologia 1990, 190, 267–275. [Google Scholar] [CrossRef]
- Bláha, L.; Babica, P.; Maršalek, B. Toxins produced in cyanobacterial water blooms-toxicity and risks. Interdiscip. Toxicol. 2009, 2, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Kaebernick, M.; Neilan, B.A. Ecological and molecular investigations of cyanotoxin production. FEMS Microbiol. Ecol. 2001, 35, 1–9. [Google Scholar] [CrossRef]
- Chorus, I.; Bartram, J. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; E & FN Spon, Published on Behalf of the World Health Organization: New York, NY, USA, 1999. [Google Scholar]
- Bullerjahn, G.S.; McKay, R.M.; Davis, T.W.; Baker, D.B.; Boyer, G.L.; D’Anglada, L.V.; Doucette, G.J.; Ho, J.C.; Irwin, E.G.; Kling, C.L.; et al. Global solutions to regional problems: Collecting global expertise to address the problem of harmful cyanobacterial blooms. A Lake Erie case study. Harmful Algae 2016, 54, 223–238. [Google Scholar] [CrossRef]
- Chaffin, J.D.; Kane, D.D.; Stanislawczyk, K.; Parker, E.M. Accuracy of data buoys for measurement of cyanobacteria, chlorophyll, and turbidity in a large lake (Lake Erie, North America): Implications for estimati.on of cyanobacterial bloom parameters from water quality sonde measurements. Environ. Sci. Pollut. Res. 2018, 25, 25175–25189. [Google Scholar] [CrossRef]
- Read, J.; Klump, V.; Johengen, T.; Schwab, D.; Paige, K.; Eddy, S.; Anderson, E.; Manninen, C. Working in freshwater: The Great Lakes observing system contributions to regional and national observations, data infrastructure, and decision support. Mar. Technol. Soc. J. 2010, 44, 84–98. [Google Scholar] [CrossRef]
- Srivastava, A.; Singh, S.; Ahn, C.-Y.; Oh, H.-M.; Asthana, R.K. Monitoring Approaches for a Toxic Cyanobacterial Bloom. Environ. Sci. Technol. 2013, 47, 8999–9013. [Google Scholar] [CrossRef]
- Moreira, C.; Ramos, V.; Azevedo, J.; Vasconcelos, V. Methods to detect cyanobacteria and their toxins in the environment. Appl. Microbiol. Biotechnol. 2014, 98, 8073–8082. [Google Scholar] [CrossRef] [PubMed]
- Zamyadi, A.; Choo, F.; Newcombe, G.; Stuetz, R. A review of monitoring technologies for real-time management of cyanobacteria: Recent advances and future direction. Trends Anal. Chem. 2016, 85, 83–96. [Google Scholar] [CrossRef]
- Falkowski, P.; Kiefer, D.A. Chlorophyll a fluorescence in phytoplankton: Relationship to photosynthesis and biomass. J. Plankton Res. 1985, 7, 715–731. [Google Scholar] [CrossRef]
- Gan, F.; Shen, G.; Bryant, D.A. Occurrence of Far-Red Light Photoacclimation (FaRLiP) in Diverse Cyanobacteria. Life 2015, 5, 4–24. [Google Scholar] [CrossRef] [PubMed]
- Kulasooriya, S.A. Cyanobacteria: Pioneers of Planet Earth. Ceylon J. Sci. (Bio. Sci.) 2011, 40, 71–88. [Google Scholar] [CrossRef]
- Conroy, J.D.; Kane, D.D.; Dolan, D.M.; Edwards, W.J.; Charlton, M.N.; Culver, D.A. Temporal trends in Lake Erie plankton biomass: Roles of external phosphorus loading and Dreissenid mussels. J. Great Lakes Res. 2005, 31 (Suppl. S2), 89–110. [Google Scholar] [CrossRef]
- Almomani, F.A.; Örmeci, B. Monitoring and measurement of microalgae using the first derivative of absorbance and comparison with chlorophyll extraction method. Environ. Monit. Assess. 2018, 190, 90. [Google Scholar] [CrossRef]
- Kuś, S.; Marczenko, Z.; Obarski, N. Derivative UV-VIS Spectrophotometry in Analytical Chemistry. Chem. Anal. (Wars.) 1996, 41, 899–927. [Google Scholar]
- Patel, K.N.; Patel, J.K.; Rajput, G.C.; Rajgor, N.B. Derivative spectrophotometry method for chemical analysis: A review. Sch. Res. Libr. 2010, 2, 139–150. [Google Scholar]
- Beversdorf, L.J.; Miller, T.R.; McMahon, K.D. The Role of Nitrogen Fixation in Cyanobacterial Bloom Toxicity in a Temperate, Eutrophic Lake. PLoS ONE 2013, 8, e56103. [Google Scholar] [CrossRef]
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, M.J.E. Smoothing and Differentiation of Data by Simplified Least Squares. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Fastner, J.; Erhard, M.; von Döhren, H. Determination of oligopeptide diversity within a natural population of Microcystis spp. (Cyanobacteria) by typing single colonies by matrix-assisted laser desorption ionization-time of flight mass spectrometry. Appl. Environ. Microbiol. 2001, 67, 5069–5076. [Google Scholar] [CrossRef] [PubMed]
- Gan, N.; Xiao, Y.; Zhu, L.; Wu, Z.; Liu, J.; Hu, C.; Song, L. The role of microcystins in maintaining colonies of bloom forming Microcystis spp. Environ. Microbiol. 2012, 14, 730–742. [Google Scholar] [CrossRef] [PubMed]
- Sobiechowska-Sasim, M.; Stoń-Egiert, J.; Kosakowska, A. Quantitative analysis of extracted phycobilin pigments in cyanobacteria-an assessment of spectrophotometric and spectrofluorometric methods. J. Appl. Phycol. 2014, 26, 2065–2074. [Google Scholar] [CrossRef]
- Dos Santos, A.C.; Calijuri, M.C.; Moraes, E.M.; Adorno, M.A.; Falco, P.B.; Carvalho, D.P.; Deberdt, G.L.; Benassi, S.F. Comparison of three methods for Chlorophyll determination: Spectrophotometry and Fluorimetry in samples containing pigment mixtures and spectrophotometry in samples with separate pigments through High Performance Liquid Chromatography. Acta Limnol. Bras. 2003, 15, 7–18. [Google Scholar]
| Cyanobacteria Cell Concentration (cells/ml) | Probability of Adverse Health Effects |
|---|---|
| <20,000 | Relatively mild and/or low |
| 20,000–100,000 | Moderate |
| 100,000–10,000,000 | High |
| >10,000,000 | Very high |
| Water Sample | Test | Concentration Range (cells/mL) | Slope | R2 | Detection Limit (cells/mL) |
|---|---|---|---|---|---|
| Deionized water | Absorbance | 129–803,750 | 8.13208 × 10−08 ± 1.75287 × 10−09 | 0.9981 | 338,950 |
| 1,286,000–5,144,000 | 7.36864 × 10−08 ± 1.67453 × 10−09 | 0.9985 | |||
| 5,144,000–10,288,000 | 6.99701 × 10−08 ± 8.81363 × 10−10 | 0.9997 | |||
| First derivative of absorbance | 129–803,750 | −1.07539 × 10−09 ± 2.16885 × 10−10 | 0.8601 | ||
| 1,286,000–5,144,000 | −7.14544 × 10−10 ± 5.33754 × 10−11 | 0.9782 | |||
| 5,144,000–10,288,000 | −7.50473 × 10−10 ± 1.23489 × 10−11 | 0.9995 | |||
| Savitzky-Golay first derivative of absorbance | 129–803,750 | 6.39754 × 10−10 ± 2.32216 × 10−11 | 0.9948 | 41,802 | |
| 1,286,000–5,144,000 | 5.88213 × 10−10 ± 7.20283 × 10−12 | 0.9994 | |||
| 5,144,000–10,288,000 | 6.26434 × 10−10 ± 3.44903 × 10−12 | 0.9999 | |||
| Surface water | Absorbance | 129–803,750 | 7.45411 × 10−08 ± 9.27447 × 10−10 | 0.9994 | 392,982 |
| 1,286,000–5,144,000 | 7.33732 × 10−08 ± 1.1411 × 10−09 | 0.9993 | |||
| 5,144,000–10,288,000 | 7.37948 × 10−08 ± 2.32234 × 10−09 | 0.9980 | |||
| First derivative of absorbance | 129–803,750 | −9.5609 × 10−10 ± 1.51611 × 10−10 | 0.9086 | ||
| 1,286,000–5,144,000 | −7.3683 × 10−10 ± 3.83158 × 10−11 | 0.9920 | |||
| 5,144,000–10,288,000 | −7.3868 × 10−10 ± 2.82227 × 10−11 | 0.9971 | |||
| Savitzky-Golay first derivative of absorbance | 129–803,750 | 6.04214 × 10−10 ± 1.03161 × 10−11 | 0.9989 | 90,231 | |
| 1,286,000–5,144,000 | 5.8526 × 10−10 ± 1.0753 × 10−11 | 0.9990 | |||
| 5,144,000–10,288,000 | 6.20429 × 10−10 ± 2.0855 × 10−11 | 0.9977 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agberien, A.V.; Örmeci, B. Monitoring of Cyanobacteria in Water Using Spectrophotometry and First Derivative of Absorbance. Water 2020, 12, 124. https://doi.org/10.3390/w12010124
Agberien AV, Örmeci B. Monitoring of Cyanobacteria in Water Using Spectrophotometry and First Derivative of Absorbance. Water. 2020; 12(1):124. https://doi.org/10.3390/w12010124
Chicago/Turabian StyleAgberien, Adogbeji Valentine, and Banu Örmeci. 2020. "Monitoring of Cyanobacteria in Water Using Spectrophotometry and First Derivative of Absorbance" Water 12, no. 1: 124. https://doi.org/10.3390/w12010124
APA StyleAgberien, A. V., & Örmeci, B. (2020). Monitoring of Cyanobacteria in Water Using Spectrophotometry and First Derivative of Absorbance. Water, 12(1), 124. https://doi.org/10.3390/w12010124





