Phosphorus and Nitrogen Adsorption Capacities of Biochars Derived from Feedstocks at Different Pyrolysis Temperatures
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Adsorption Kinetics Experiments
2.3. Adsorption Isotherm Experiment
2.4. Adsorption Thermodynamics
2.5. Analysis Method
2.6. Statistical Analysis
3. Results and Discussion
3.1. Characteristics of Biochar
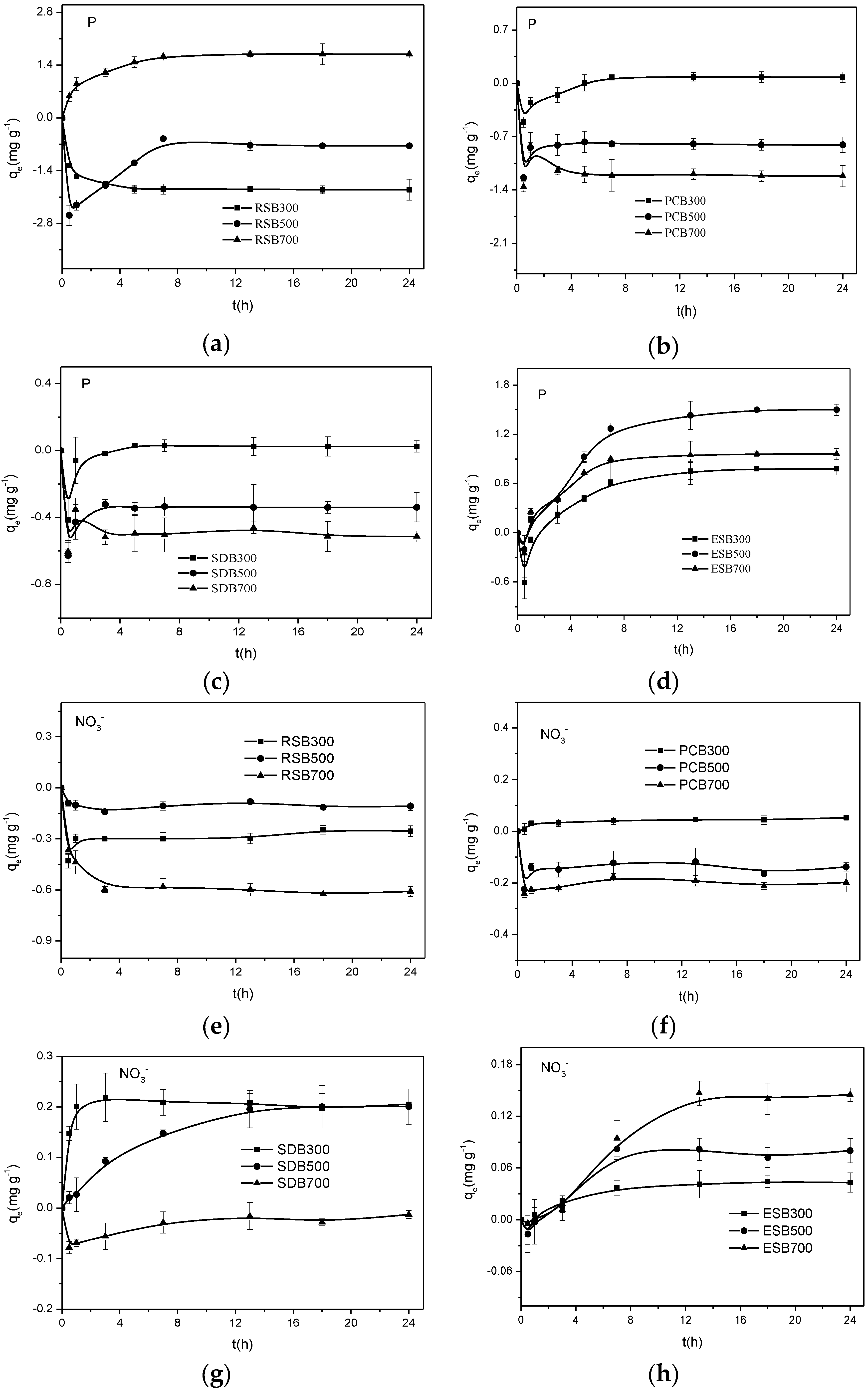
3.2. Sorption Kinetics of Biochar
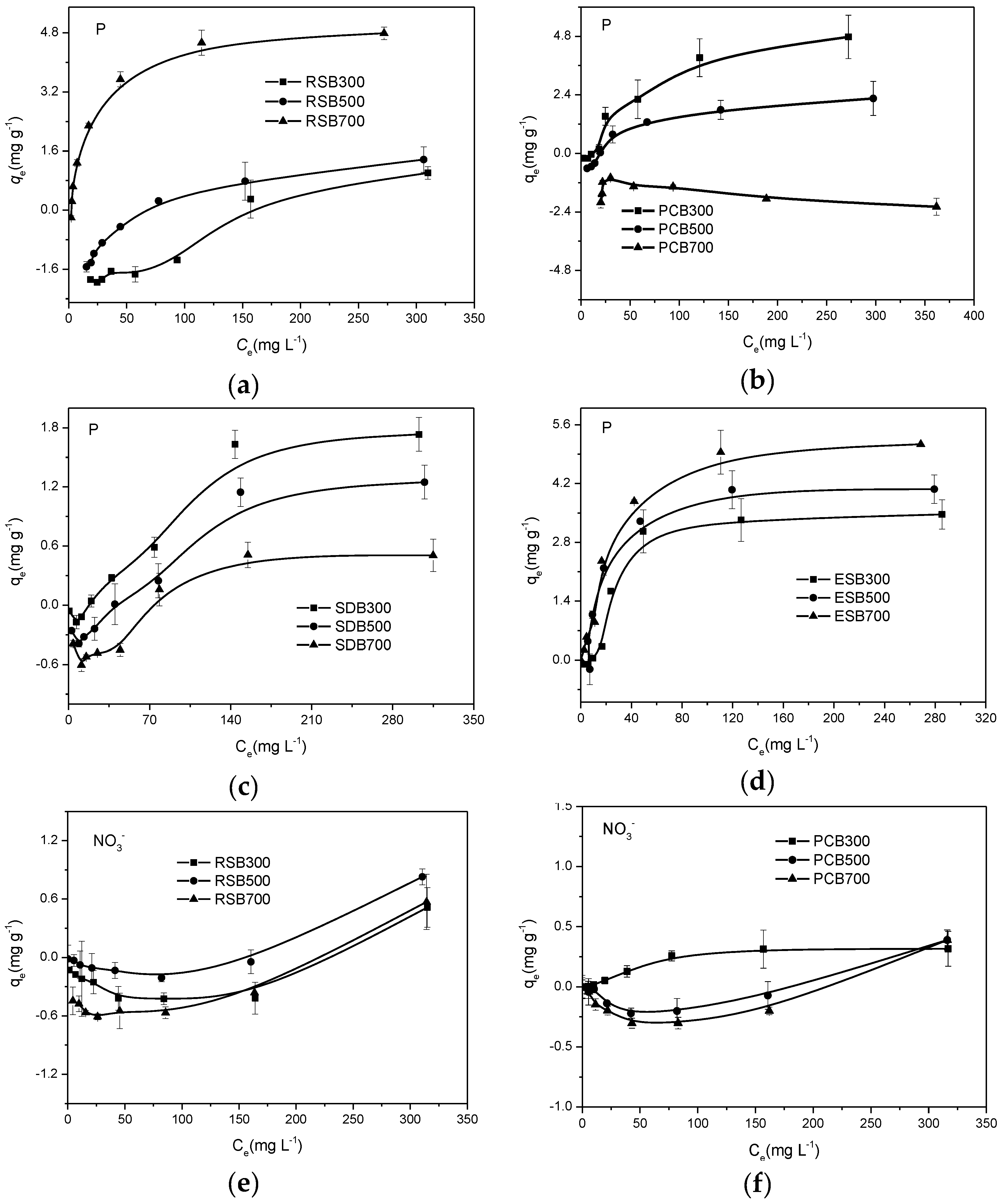
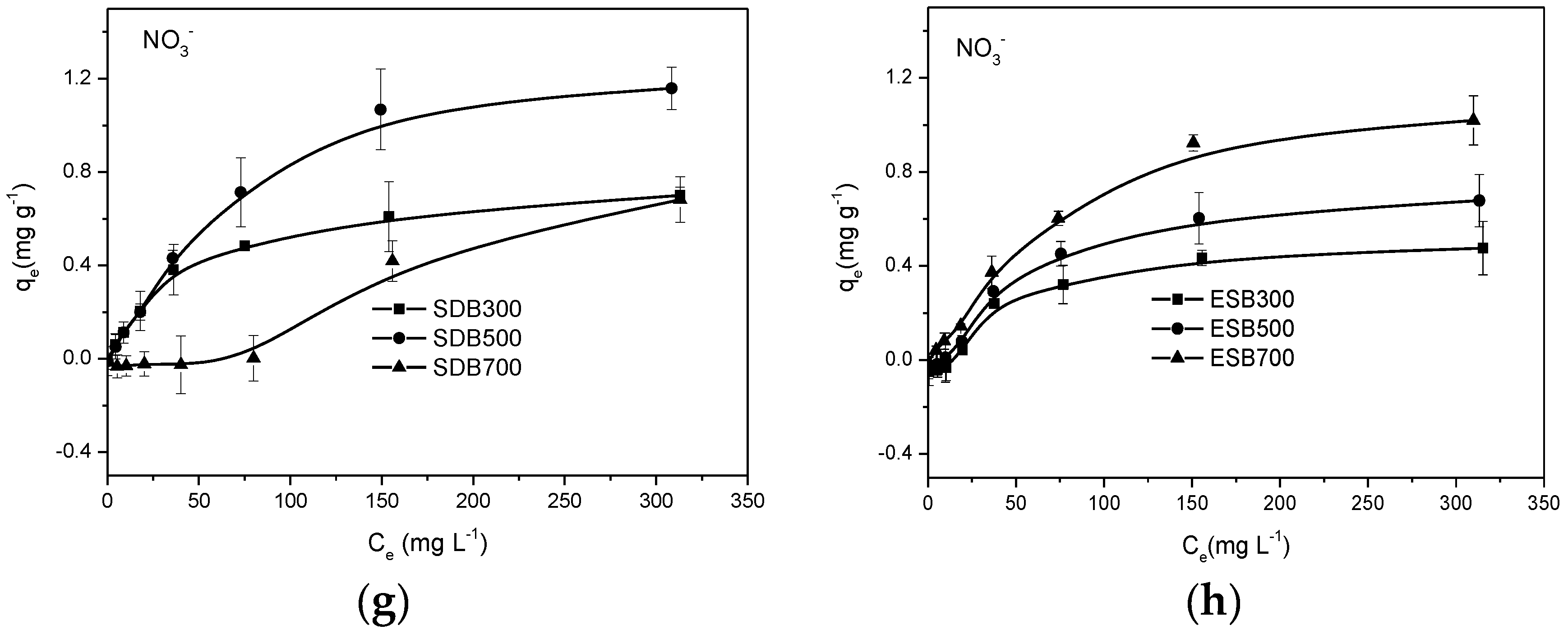
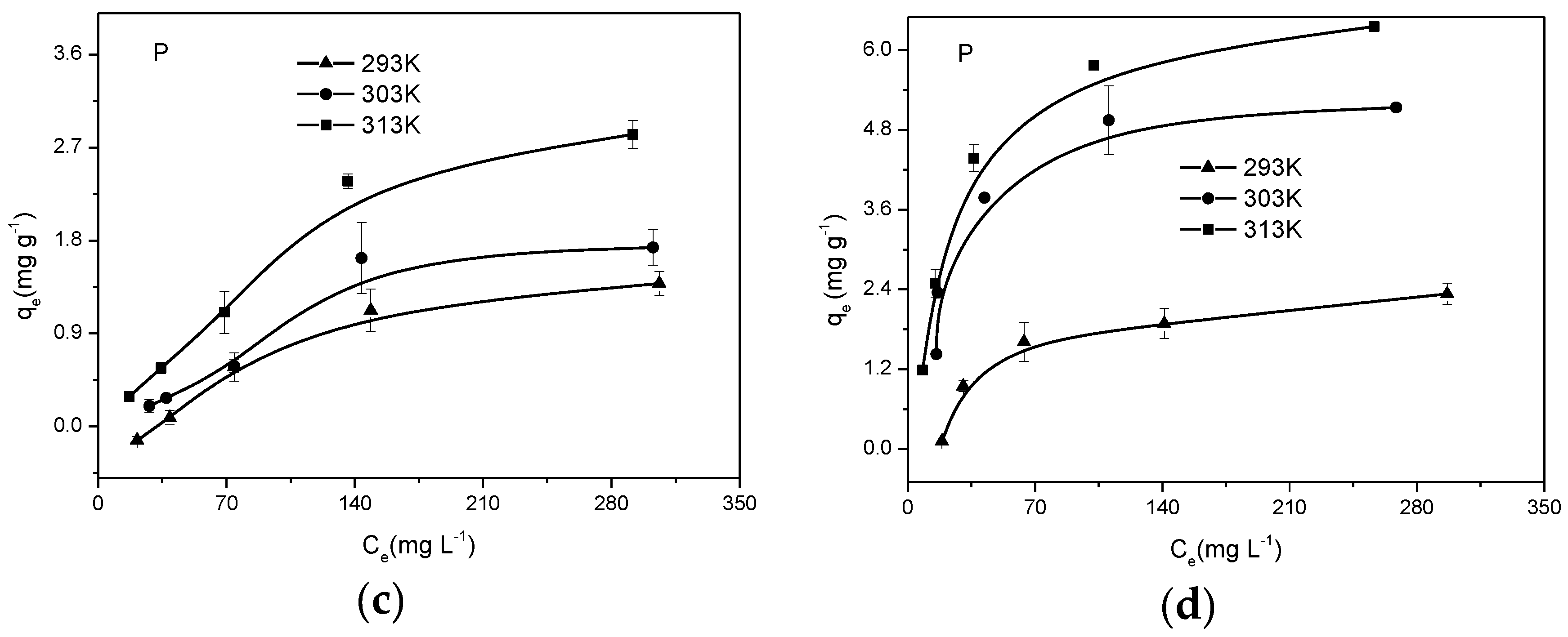
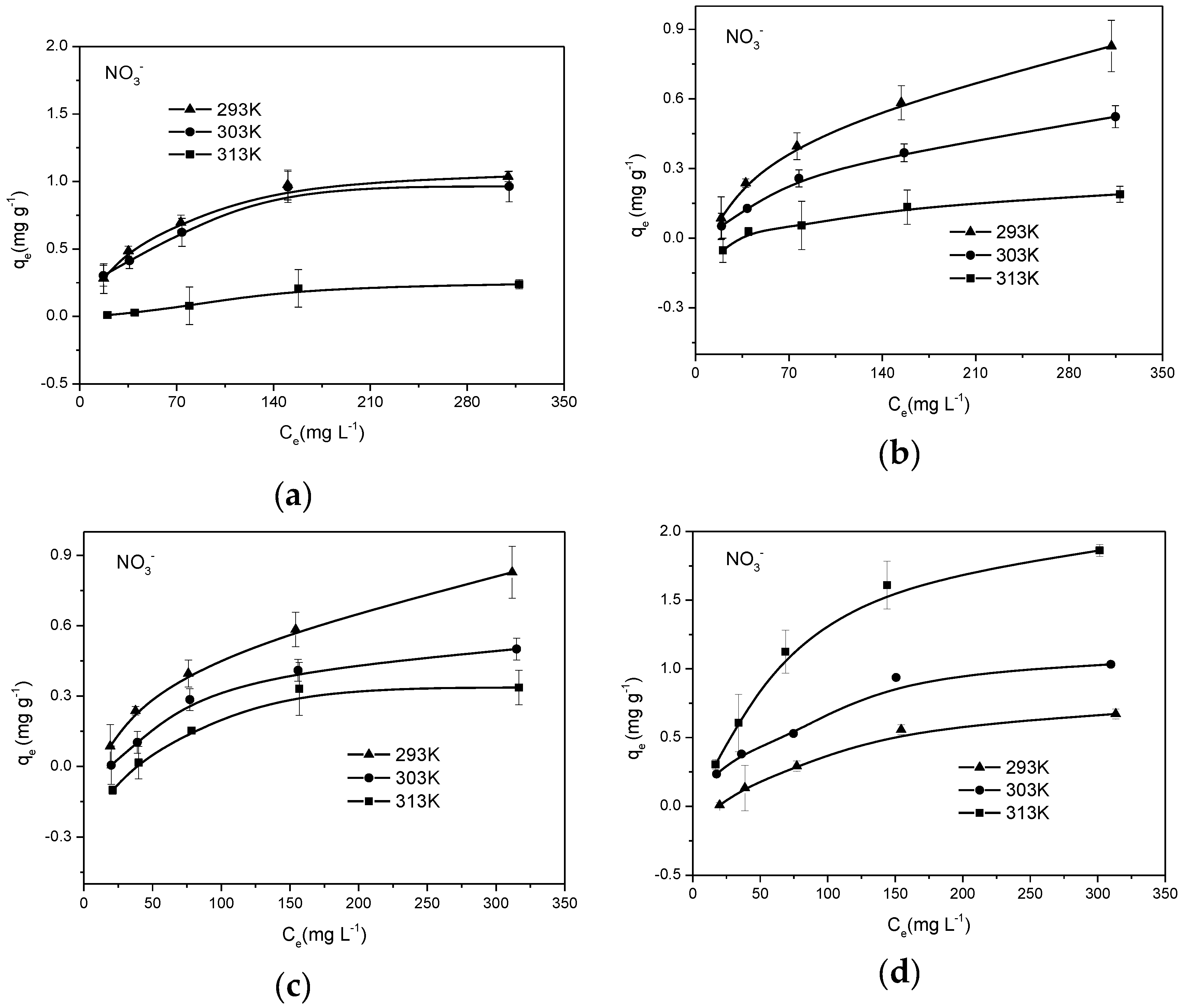
3.3. Sorption Isotherms of Biochar
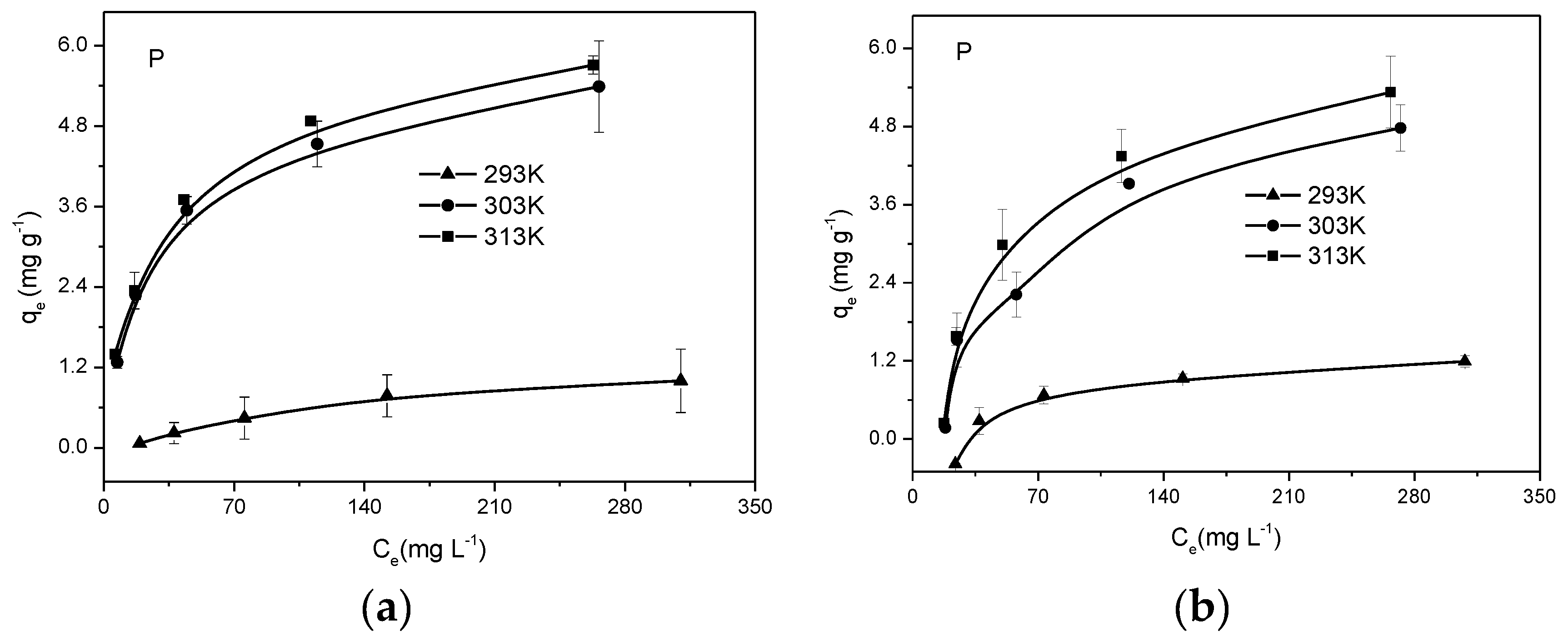
3.4. Adsorption Thermodynamics of Biochars
3.5. Effect of Characteristic of Biochar on Capacity of Nitrate and Phosphate Adsorption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barcellos, D.; Queiroz, H.M.; Nóbrega, G.N.; De Oliveira Filho, R.L.; Santaella, S.T.; Otero, X.L.; Ferreira, T.O. Phosphorus enriched effluents increase eutrophication risks for mangrove systems in northeastern Brazil. Mar. Pollut. Bull. 2019, 142, 58–63. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Chen, J.F.; Tong, T.L.; Li, B.Q.; He, T.; Liu, Y.; Xie, S.G. Eutrophication influences methanotrophic activity, abundance and community structure in freshwater lakes. Sci. Total Environ. 2019, 662, 863–872. [Google Scholar] [CrossRef]
- Karaca, S.; Gurses, A.; Ejder, M.; Acikyildiz, M. Kinetic modeling of liquid phase adsorption of phosphate on dolomite. J. Colloid Interface Sci. 2004, 277, 257–263. [Google Scholar] [CrossRef]
- Yin, Q.; Zhang, B.; Wang, R.; Zhao, Z. Biochar as an adsorbent for inorganic nitrogen and phosphorus removal from water: A review. Environ. Sci. Pollut. Res. 2017, 24, 26297–26309. [Google Scholar] [CrossRef]
- Wang, D.B.; Yang, G.J.; Li, X.M.; Zheng, W.; Wu, Y.; Yang, Q.; Zeng, G.M. Inducing mechanism of biological phosphorus removal driven by the aerobic/extended-idle regime. Biotechnol. Bioeng. 2012, 109, 2798–2807. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.D.; Pullammanappallil, P.; Yang, L.Y. Removal of phosphate from aqueous solution by biochar derived from anaerobically digested sugar beet tailings. J. Hazard. Mater. 2011, 190, 501–507. [Google Scholar] [CrossRef]
- Neufeld, R.D.; Thodos, G. Removal of orthophosphates from aqueous solutions with activated alumina. Environ. Sci. Technol. 1969, 3, 661–667. [Google Scholar] [CrossRef]
- Hossein, K.; Masoud, S.N.; Hamed, S.; Hossein, S.H. Facile reduction of graphene using urea in solid phase and surface modification by N-doped graphene quantum dots for adsorption of organic dyes. Diam Relat. Mater. 2017, 79, 133–144. [Google Scholar]
- Bhatnagar, A.; Sillanpää, M. A review of emerging adsorbents for nitrate removal from water. Chem. Eng. J. 2011, 168, 493–504. [Google Scholar] [CrossRef]
- Doula, M.K.; Sarris, A.; Hliaoutakis, A.; Kydonakis, A.; Papadopoulos, N.S.; Argyriou, L. Building a strategy for soil protection at local and regional scale-the case of agricultural wastes land spreading. Environ. Monit. Assess. 2016, 188, 114. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, X.; Chen, X.; Dai, C.; Zhang, J.; Zhang, Y. Biosorption of clofibric acid and carbamazepine in aqueous solution by agricultural waste rice straw. J. Environ. Sci. 2013, 25, 2384–2395. [Google Scholar] [CrossRef]
- He, J.W.; Yang, S.M.; Zhang, G.Y. Recent studies on eggshell as adsorption material. Trans. Chin. Soc. Agric. Eng. 2016, 32, 297–303. [Google Scholar]
- Badr, A.M.; Naoko, E.; Chang, S.K.; Bi, X.T. The role of tailored biochar in increasing plant growth, and reducing bioavailability, phytotoxicity, and uptake of heavy metals in contaminated soil. Environ. Pollut. 2017, 230, 329–338. [Google Scholar]
- Yao, Y.; Gao, B.; Zhang, M.; Inyang, M.; Zimmerman, A.R. Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 2012, 89, 146–1471. [Google Scholar] [CrossRef]
- Hale, S.E.; Alling, V.; Martinsen, V.; Mulder, J.; Breedveld, G.D.; Cornelissen, G. The sorption and desorption of phosphate-P ammonium-N and nitrate-N in cacao shell and corn cob biochars. Chemosphere 2013, 91, 1612–1619. [Google Scholar] [CrossRef]
- Jung, K.W.; Kyu-Hong, A. Fabrication of porosity-enhanced MgO/biochar for removal of phosphate from aqueous solution: Application of a novel combined electrochemical modification method. Bioresour. Technol. 2016, 200, 1029–1032. [Google Scholar] [CrossRef]
- Michalekova-Richveisova, B.; Fristak, V.; Pipiska, M.; Duriska, L.; Moreno-Jimenez, E.; Soja, G. Iron-impregnated biochars as effective phosphate sorption materials. Environ. Sci. Pollut. Res. Int. 2017, 24, 463–475. [Google Scholar] [CrossRef]
- Novais, S.V.; Zenero, M.D.O.; Barreto, M.S.C.; Montes, C.R.; Cerri, C.E.P. Phosphorus removal from eutrophic water using modified biochar. Sci. Total Environ. 2018, 633, 825–835. [Google Scholar] [CrossRef]
- Zhang, H.Z.; Chen, C.R.; Evan, M.G.; Sue, E.B.; Yang, H.; Zhang, D.K. Roles of biochar in improving phosphorus availability in soils: A phosphate adsorbent and a source of available phosphorus. Geoderma 2016, 276, 1–6. [Google Scholar] [CrossRef]
- Zhang, H.; Voroney, R.P.; Price, G.W. Effects of temperature and activation on biochar chemical properties and their impact on ammonium, nitrate, and phosphate sorption. J. Environ. Qual. 2017, 46, 889–896. [Google Scholar] [CrossRef]
- Kameyama, K.; Miyamoto, T.; Shiono, T.; Shinogi, Y. Influence of sugarcane bagasse-derived biochar application on nitrate leaching in calcaric dark red soil. J. Environ. Qual. 2012, 41, 1131–1137. [Google Scholar] [CrossRef]
- Chintala, R.; Schumacher, T.E.; McDonald, L.M.; Clay, D.E.; Malo, D.D.; Papiernik, S.K.; Clay, S.A.; Julson, J.L. Phosphorus sorption and availability from biochars and soil/biochar mixtures. Clean Soil Air Water 2014, 42, 626–634. [Google Scholar] [CrossRef]
- Novais, S.V.; Zenero, M.D.O.; Tronto, J.; Conz, R.F.; Cerri, C.E.P. Poultry manure and sugarcane straw biochars modified with MgCl2 for phosphorus adsorption. J. Environ. Manag. 2018, 214, 36–44. [Google Scholar] [CrossRef]
- Schneider, F.; Haderlein, S.B. Potential effects of biochar on the availability of phosphorus-mechanistic insights. Geoderma 2016, 277, 83–90. [Google Scholar] [CrossRef]
- Elsa, A.; Mohan, V.J.; Graham, B.; Philip, A.S. Isotherms, kinetics and mechanism analysis of phosphorus recovery from aqueous solution by calcium-rich biochar produced from biosolids via microwave pyrolysis. J. Environ. Chem. Eng. 2018, 6, 395–403. [Google Scholar]
- Yin, Q.Q.; Wang, R.K.; Zhao, Z.H. Application of Mg-Al-modified biochar for simultaneous removal of ammonium, nitrate, and phosphate from eutrophic water. J. Clean Prod. 2018, 176, 230–240. [Google Scholar] [CrossRef]
- Wang, S.D.; Kong, L.J.; Long, J.Y.; Su, M.H.; Diao, Z.H.; Chang, X.Y.; Chen, D.Y.; Song, G.; Kaimin, S. Adsorption of phosphorus by calcium-flour biochar: Isotherm, kinetic and transformation studies. Chemosphere 2018, 195, 666–672. [Google Scholar] [CrossRef]
- Tan, I.A.; Ahmad, A.L.; Hameed, B.H. Adsorption of basic dye on high-surface-area activated carbon prepared from coconut husk: Equilibrium, kinetic and thermodynamic studies. J. Hazard. Mater. 2008, 154, 337–346. [Google Scholar] [CrossRef]
- Hema, M.; Arivoli, S. Comparative study on the adsorption kinetics and thermodynamics of dyes onto acid activated low cost carbon. Int. J. Phys. Sci. 2007, 2, 10–17. [Google Scholar]
- Zhang, T.; Xu, H.Y.; Li, H.H.; He, X.Y.; Shi, Y.J.; Andrea, K. Microwave digestion-assisted HFO/Biochar adsorption to recover phosphorus from swine manure. Sci. Total Environ. 2018, 621, 1512–1526. [Google Scholar] [CrossRef]
- Qu, B.; Zhou, J.; Xiang, X.; Zheng, C.; Zhao, H.; Zhou, X. Adsorption behavior of Azo Dye, CI Acid Red 14 in aqueous solution on surface soils. J. Environ. Sci. 2008, 20, 704–709. [Google Scholar] [CrossRef]
- Simon, K.; Wu, S.B.; Kirui, W.K.; Lei, M.; Lu, Q.M.; Bah, H.D.; Dong, R.J. Evaluation of slow pyrolyzed wood and rice husks biochar for adsorption of ammonium nitrogen from piggery manure anaerobic digestate slurry. Sci. Total Environ. 2015, 505, 102–112. [Google Scholar]
- Chen, Y.C.; Lu, C. Kinetics, thermodynamics and regeneration of molybdenum adsorption in aqueous solutions with NaOCl-oxidized multiwalled carbon nanotubes. J. Ind. Eng. Chem. 2014, 20, 2517–2521. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, B.; Yao, Y.; Xue, Y.W.; Inyang, M. Synthesis of porous MgO-biochar nanocomposites for removal of phosphate and nitrate from aqueous solutions. Chem. Eng. J. 2012, 210, 26–32. [Google Scholar] [CrossRef]
- Zhang, Z.R.; Yan, L.G.; Yu, H.Q.; Yan, T.; Li, X.G. Adsorption of phosphate from aqueous solution by vegetable biochar/layered double oxides: Fast removal and mechanistic studies. Bioresour. Technol. 2019, 284, 65–71. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, X.L.; Luo, W.; Sun, J.; Xu, Q.X.; Chen, F.; Zhao, J.W.; Wang, S.N.; Yao, F.B.; Wang, D.B.; et al. Effectiveness and mechanisms of phosphate adsorption on iron-modified biochars derived from waste activated sludge. Bioresour. Technol. 2018, 247, 537–544. [Google Scholar] [CrossRef]
- Yin, Q.Q.; Ren, H.P.; Wang, R.K.; Zhao, Z.H. Evaluation of nitrate and phosphate adsorption on Al-modified biochar: Influence of Al content. Sci. Total Environ. 2018, 631, 895–903. [Google Scholar] [CrossRef]
- Iida, T.; Amano, Y.; Machida, M.; Imazeki, F. Effect of surface property of activated carbon on adsorption of nitrate ion. Chem. Pharm. Bull. 2013, 61, 1173–1177. [Google Scholar] [CrossRef]
- Wang, Z.H.; Guo, H.Y.; Shen, F.; Yang, G.; Zhang, Y.Z.; Zeng, Y.M.; Wang, L.L.; Xiao, H.; Deng, S.H. Biochar produced from oak sawdust by Lanthanum (La)-involved pyrolysis for adsorption of ammonium (NH4+), nitrate (NO3−), and phosphate (PO43−). Chemosphere 2015, 119, 646–653. [Google Scholar] [CrossRef]
- Park, J.H.; Ok, Y.S.; Kim, S.H.; Cho, J.S.; Heo, J.S.; Delaune, R.D.; Seo, D.C. Evaluation of phosphorus adsorption capacity of sesame straw biochar on aqueous solution: Influence of activation methods and pyrolysis temperatures. Environ. Geochem. Health 2015, 37, 969–983. [Google Scholar] [CrossRef]
- Ketcha, M.J.; Manga, N.H.; Daouda, K.; Tchoua, N.P.H. Kinetic and equilibrium studies of the adsorption of nitrates ions in aqueous solutions by activated carbons and zeolite. Res. J. Chem. Environ. 2007, 11, 47–51. [Google Scholar]


| Sample | Total Volume (cm3 g−1) | Specific Surface Area (m2 g−1) | pH | Zeta Potential (mV) |
|---|---|---|---|---|
| RSB300 | 0.022 ± 0.003 a | 5.9 ± 0.6 a | 6.61 ± 0.05 a | −30.50 ± 0.70 a |
| RSB500 | 0.072 ± 0.008 b | 34.0 ± 3.6 b | 9.28 ± 0.07 b | −41.90 ± 1.05 b |
| RSB700 | 0.189 ± 0.002 c | 122.6 ± 14.3 c | 10.06 ± 0.1 c | −19.35 ± 1.92 c |
| PCB300 | 0.008 ± 0.0009 a | 3.5 ± 0.4 a | 6.43 ± 0.03 a | −39.30 ± 0.76 a |
| PCB500 | 0.106 ± 0.002 b | 131.5 ± 14.3 b | 6.82 ± 0.02 b | −30.33 ± 2.01 b |
| PCB700 | 0.415 ± 0.05 c | 441.7 ± 45.6 c | 9.42 ± 0.04 c | −23.41 ± 0.70 c |
| SDB300 | 0.006 ± 0.0007 a | 2.9 ± 0.4 a | 4.55 ± 0.14 a | −25.63 ± 0.54 a |
| SDB500 | 0.233 ± 0.03 b | 378.7 ± 39.6 b | 6.03 ± 0.02 b | −39.90 ± 1.20 b |
| SDB700 | 0.278 ± 0.04 b | 594.9 ± 60.3 c | 7.88 ± 0.08 c | −16.60 ± 0.29 c |
| ESB300 | 0.004 ± 0.0005 a | 2.0 ± 0.2 a | 7.89 ± 0.04 a | −9.99 ± 0.92 a |
| ESB500 | 0.006 ± 0.0007 ab | 3.7 ± 0.4 ab | 8.02 ± 0.11 a | −4.94 ± 1.63 b |
| ESB700 | 0.009 ± 0.0008 b | 5.3 ± 0.5 b | 9.46 ± 0.07 b | −2.99 ± 1.37 b |
| Sample | Pseudo-First-Order | Pseudo-Second-Order | Intra-particle Diffusion | ||||||
|---|---|---|---|---|---|---|---|---|---|
| K1 (h−1) | qe (mg g−1) | R2 | K2 (g mg−1 h−1) | qe (mg g−1) | R2 | K3 (mg g−1 h−0.5) | C (mg g−1) | R2 | |
| RSB700 | 0.005 | 0.998 | 0.865 | 0.009 | 1.790 | 0.999 | 0.032 | 0.703 | 0.778 |
| PCB300 | 0.006 | 0.430 | 0.868 | 0.622 | 0.084 | 0.998 | 0.016 | −0.392 | 0.706 |
| SDB300 | 0.008 | 0.100 | 0.800 | 4.480 | 0.024 | 0.994 | 0.002 | −0.046 | 0.601 |
| ESB300 | 0.006 | 1.879 | 0.966 | 0.012 | 0.827 | 0.951 | 0.038 | −0.411 | 0.800 |
| ESB500 | 0.004 | 2.027 | 0.995 | 0.020 | 1.514 | 0.919 | 0.054 | −0.231 | 0.893 |
| ESB700 | 0.006 | 1.534 | 0.984 | 0.011 | 1.026 | 0.947 | 0.033 | −0.053 | 0.753 |
| Sample | Pseudo-First-Order | Pseudo-Second-Order | Intra-Particle Diffusion | ||||||
|---|---|---|---|---|---|---|---|---|---|
| K1 (h−1) | qe (mg g−1) | R2 | K2 (g mg−1 h−1) | qe (mg g−1) | R2 | K3 (mg g−1 h−0.5) | C (mg g−1) | R2 | |
| PCB300 | 0.001 | 0.028 | 0.764 | 0.153 | 0.053 | 0.984 | 0.001 | 0.015 | 0.771 |
| SDB300 | 0.001 | 0.023 | 0.700 | 0.287 | 0.216 | 0.992 | 0.001 | 0.177 | 0.328 |
| SDB500 | 0.006 | 0.300 | 0.968 | 0.010 | 0.267 | 0.974 | 0.006 | −0.002 | 0.928 |
| ESB300 | 0.004 | 0.047 | 0.970 | 0.449 | 0.044 | 0.966 | 0.002 | −0.009 | 0.804 |
| ESB500 | 0.008 | 0.127 | 0.906 | 0.097 | 0.05 | 0.878 | 0.003 | −0.021 | 0.814 |
| ESB700 | 0.003 | 0.193 | 0.989 | 0.024 | 0.112 | 0.935 | 0.005 | −0.036 | 0.914 |
| Sample | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| qmax (mg g−1) | KL (L mg−1) | R2 | KF (mg g−1) | n | R2 | |
| RSB700 | 5.407 | 0.039 | 0.981 | 0.611 | 2.558 | 0.876 |
| PCB300 | 7.747 | 0.007 | 0.942 | 0.125 | 1.501 | 0.897 |
| PCB500 | 3.270 | 0.008 | 0.933 | 0.026 | 1.246 | 0.760 |
| SDB300 | 3.859 | 0.003 | 0.903 | 0.019 | 1.230 | 0.872 |
| SDB500 | 3.395 | 0.002 | 0.911 | 0.002 | 0.871 | 0.728 |
| ESB300 | 4.538 | 0.018 | 0.852 | 0.244 | 2.009 | 0.760 |
| ESB500 | 4.922 | 0.030 | 0.920 | 0.482 | 2.458 | 0.800 |
| ESB700 | 6.084 | 0.031 | 0.973 | 0.598 | 2.542 | 0.855 |
| Sample | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| qmax (mg g−1) | KL (L mg−1) | R2 | KF (mg g−1) | n | R2 | |
| PCB300 | 0.433 | 0.012 | 0.955 | 0.021 | 2.006 | 0.876 |
| SDB300 | 0.804 | 0.021 | 0.996 | 0.070 | 2.416 | 0.949 |
| SDB500 | 1.574 | 0.011 | 0.989 | 0.067 | 1.941 | 0.941 |
| ESB300 | 0.671 | 0.010 | 0.919 | 0.020 | 1.735 | 0.850 |
| ESB500 | 0.937 | 0.010 | 0.963 | 0.032 | 1.810 | 0.901 |
| ESB700 | 1.426 | 0.010 | 0.987 | 0.051 | 1.855 | 0.941 |
| P | NO3− | |||||||
|---|---|---|---|---|---|---|---|---|
| T(K) | Sample | ∆H0 (kJ mol−1) | ∆S0 (kJ mol−1K−1) | G0 (kJ mol−1) | Sample | ∆H0 (kJ mol−1) | ∆S0 (kJ mol−1 K−1) | ∆G0 (kJ mol−1) |
| 293 | RSB700 | 83.54 | 0.33 | −11.95 | RSB500 | −70.80 | −0.19 | −13.47 |
| 303 | −17.70 | −12.69 | ||||||
| 313 | −18.41 | −9.73 | ||||||
| 293 | PCB300 | 44.63 | 0.19 | −11.27 | PCB300 | −25.37 | −0.05 | −10.79 |
| 303 | −13.92 | −10.70 | ||||||
| 313 | −15.07 | −9.73 | ||||||
| 293 | SDB300 | 27.53 | 0.13 | −10.48 | SDB500 | −27.44 | −0.06 | −10.79 |
| 303 | −11.99 | −10.70 | ||||||
| 313 | −13.07 | −8.67 | ||||||
| 293 | ESB700 | 39.68 | 0.19 | −14.53 | ESB300 | 39.20 | 0.17 | −9.81 |
| 303 | −17.47 | −12.45 | ||||||
| 313 | −18.20 | −13.11 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.; Xu, D.; Li, Y.; Pan, Q.; Wang, J.; Xue, L.; Howard, A. Phosphorus and Nitrogen Adsorption Capacities of Biochars Derived from Feedstocks at Different Pyrolysis Temperatures. Water 2019, 11, 1559. https://doi.org/10.3390/w11081559
Zhou L, Xu D, Li Y, Pan Q, Wang J, Xue L, Howard A. Phosphorus and Nitrogen Adsorption Capacities of Biochars Derived from Feedstocks at Different Pyrolysis Temperatures. Water. 2019; 11(8):1559. https://doi.org/10.3390/w11081559
Chicago/Turabian StyleZhou, Lei, Defu Xu, Yingxue Li, Qianchen Pan, Jiajun Wang, Lihong Xue, and Alan Howard. 2019. "Phosphorus and Nitrogen Adsorption Capacities of Biochars Derived from Feedstocks at Different Pyrolysis Temperatures" Water 11, no. 8: 1559. https://doi.org/10.3390/w11081559
APA StyleZhou, L., Xu, D., Li, Y., Pan, Q., Wang, J., Xue, L., & Howard, A. (2019). Phosphorus and Nitrogen Adsorption Capacities of Biochars Derived from Feedstocks at Different Pyrolysis Temperatures. Water, 11(8), 1559. https://doi.org/10.3390/w11081559





