Effect of Species Invasion on Transport of Solutes at Different Levels of Soft Sediment Macrofauna Diversity: Results from an Experimental Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Sediment and Macrofauna Sampling
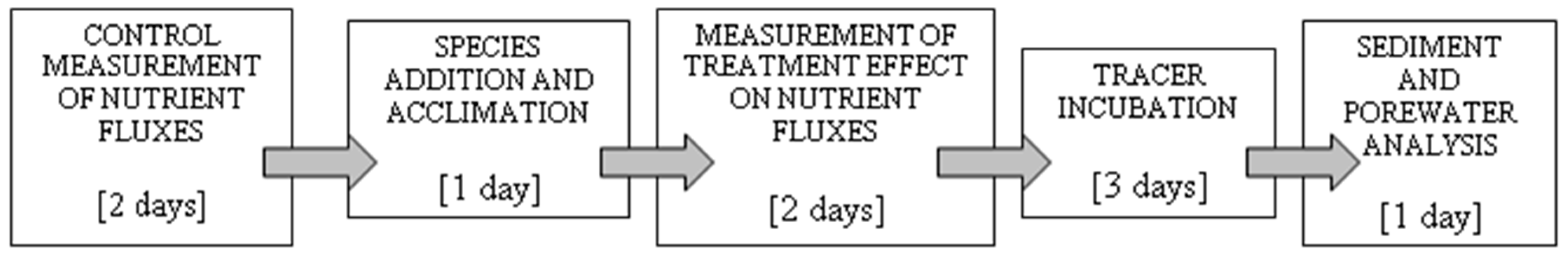
2.2. Experimental Design
2.3. Processing of Samples and Calculation of Solute Fluxes
2.4. Data Analysis
3. Results
3.1. Macrofauna and Sediment Characteristics
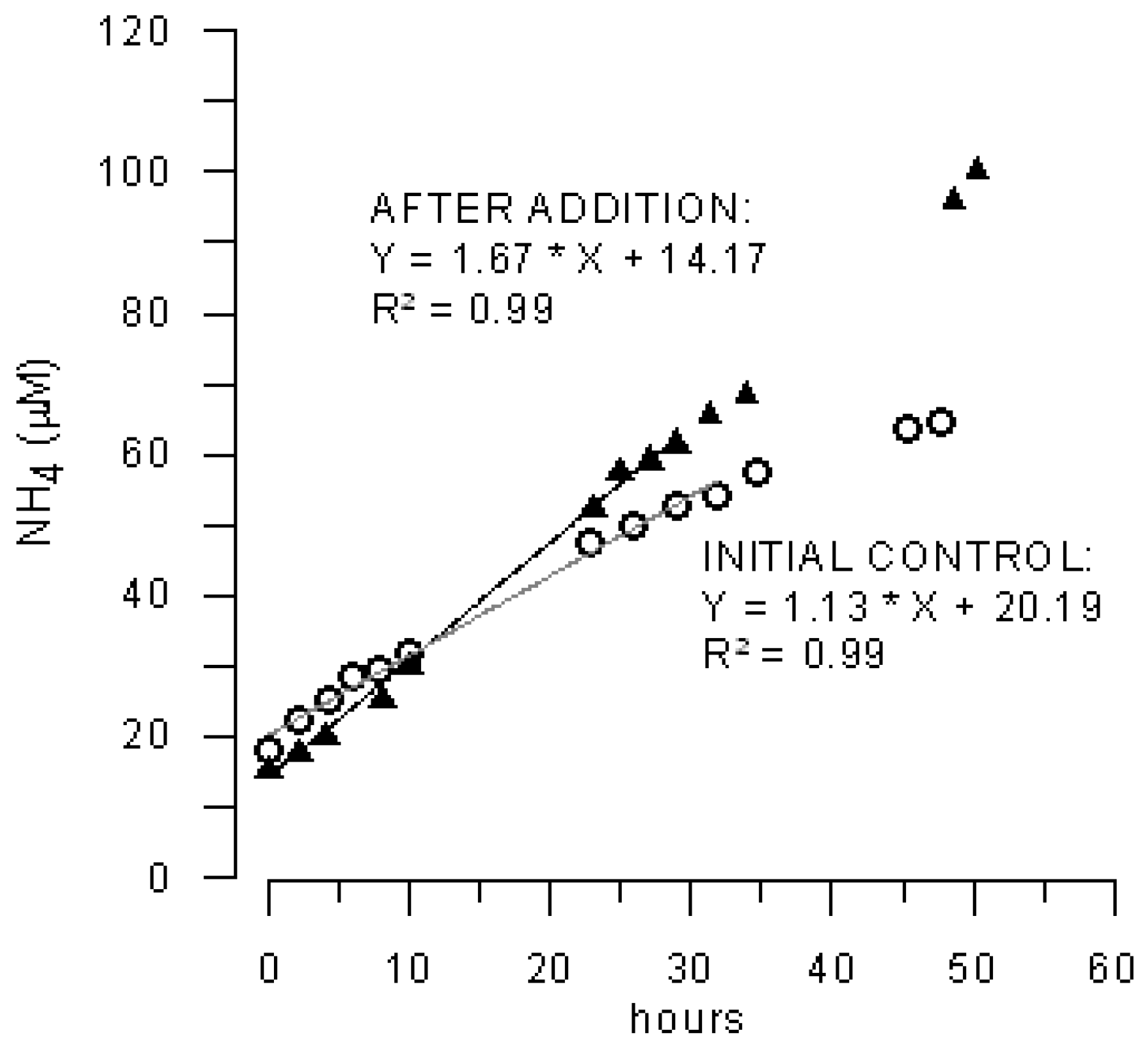
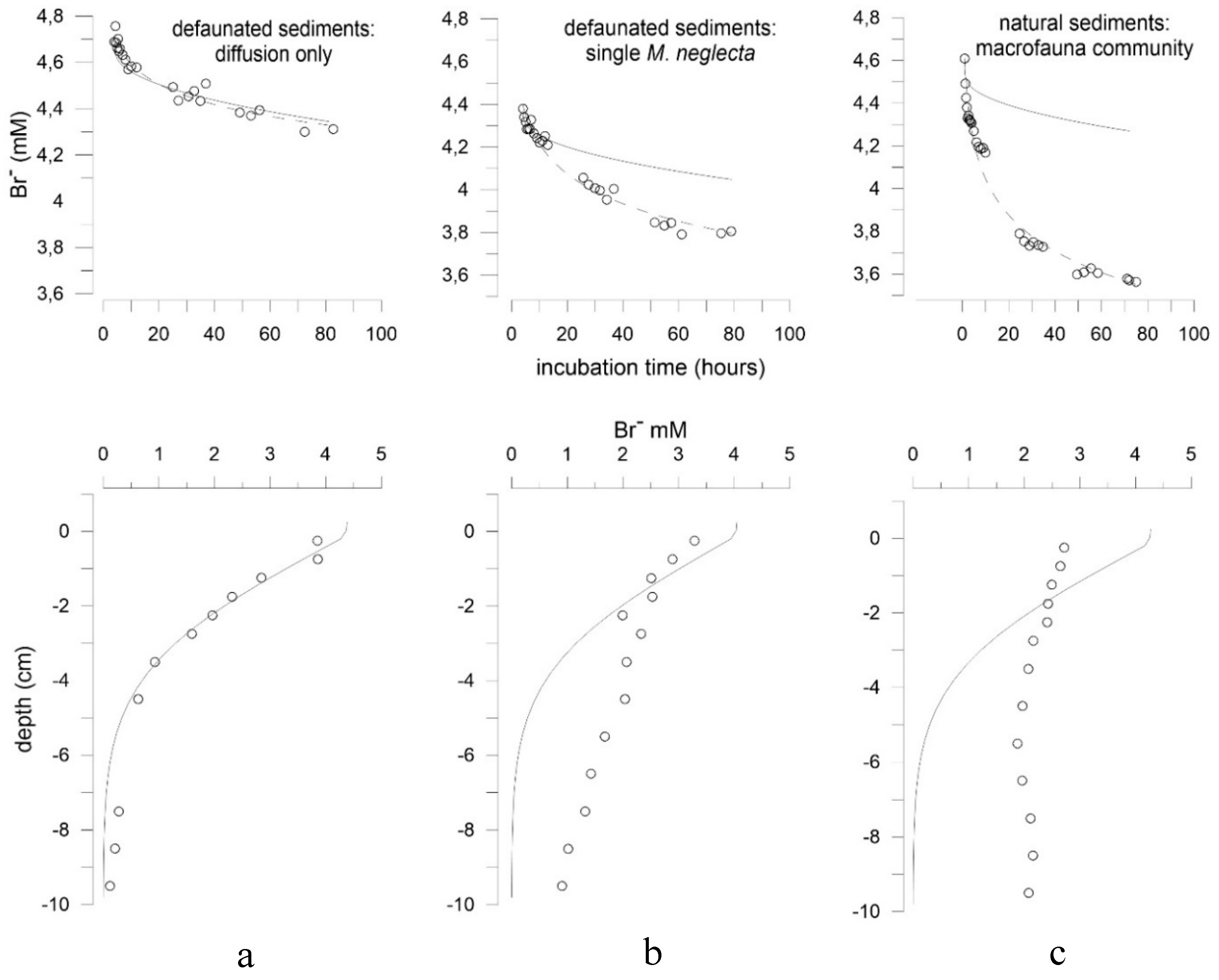
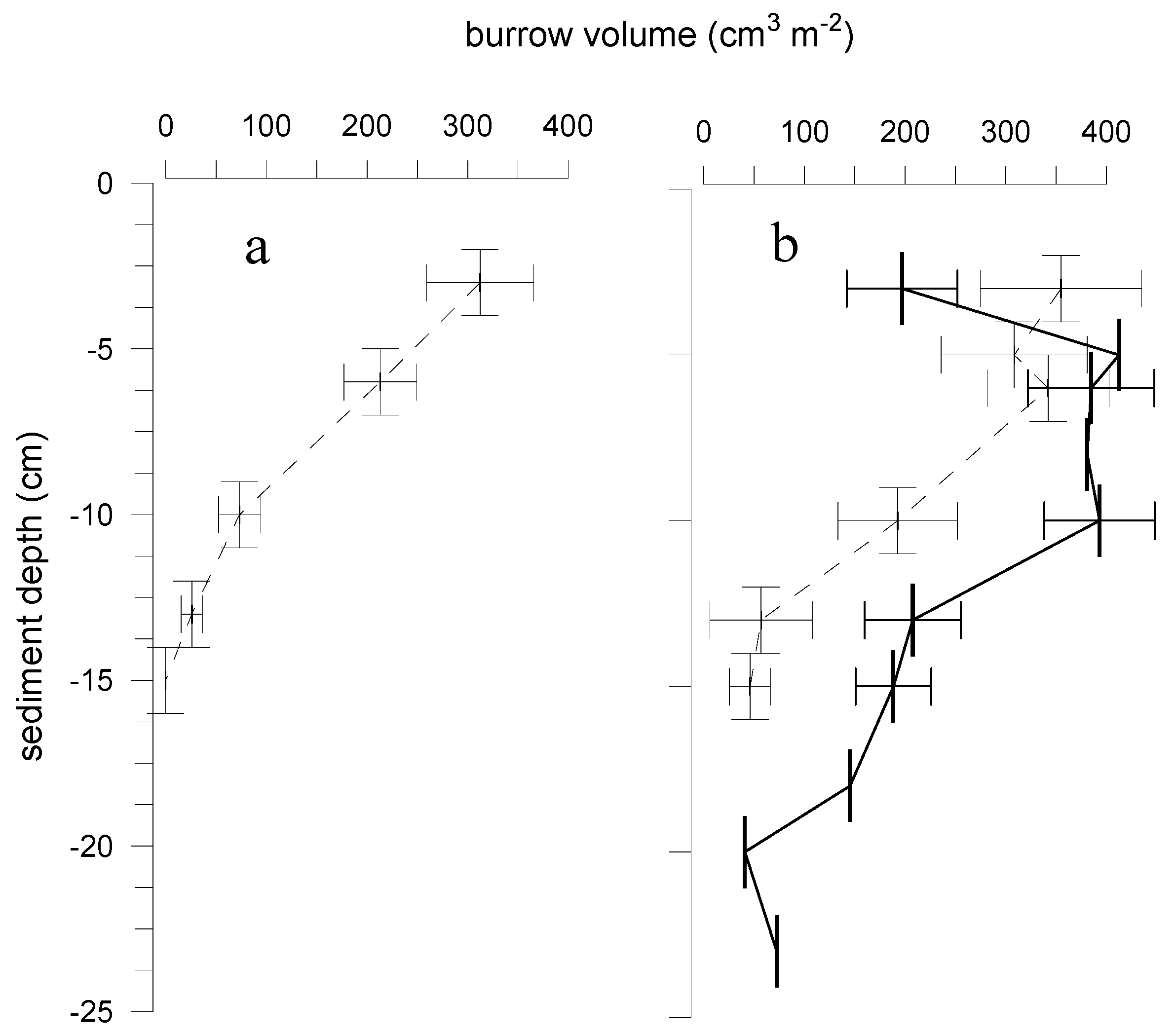
3.2. Effects of Marenzelleria neglecta on Bromide Fluxes in Defaunated Sediment (Experiment 1)
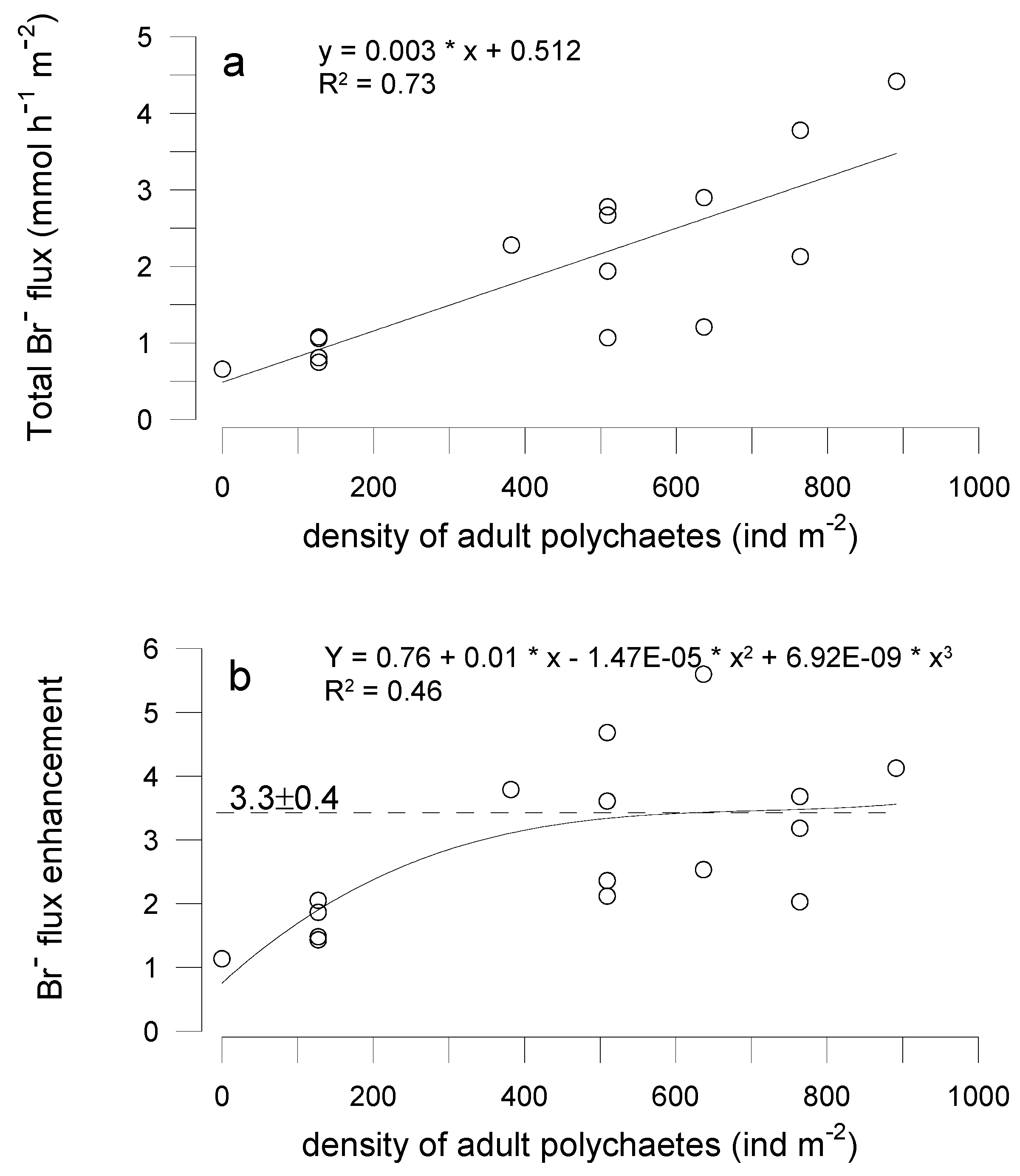
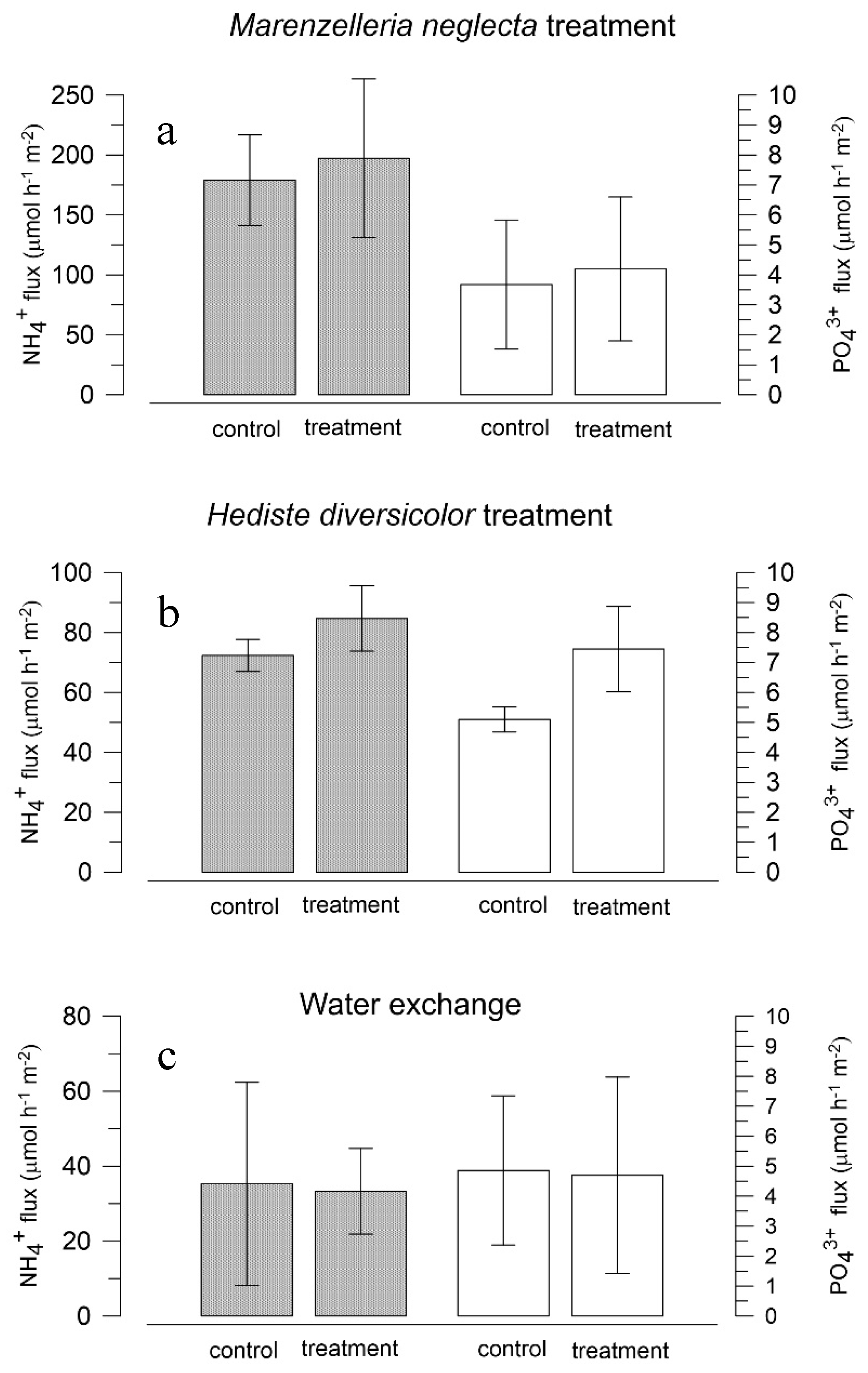
3.3. Effects of Polychaetes on Nutrient and Bromide Fluxes in Natural Sediment (Experiment 2)
4. Discussion
4.1. Effects of Polychaetes on Nutrient and Bromide Fluxes
4.2. Implications for Experiments on New Species Impacts
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carlton, J.T.; Geller, J.B. Ecological roulette: The global transport of nonindigenous marine organisms. Science 1993, 261, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Carlton, J.T. Pattern, process and prediction in marine invasion ecology. Biol. Conserv. 1996, 78, 97–106. [Google Scholar] [CrossRef]
- Mack, R.N.; Occhipinti, A. Biotic invasion: A global perspective and ecology of invasion: Patterns and perspectives. In Perspectives in Ecology; Farina, A., Ed.; Backhuys Publishers: Leiden, The Netherlands, 1999; pp. 67–74. [Google Scholar]
- Leppäkoski, E.; Gollasch, S.; Olenin, S. Alien species in European waters. In Aquatic Invasive Species of Europe—Distribution, Impacts and Management; Leppäkoski, E., Gollasch, S., Olenin, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 1–6. [Google Scholar]
- Norkko, J.; Reed, D.C.; Timmermann, K.; Norkko, A.; Gustafsson, B.G.; Bonsdorff, E.; Slomp, C.P.; Carstensen, J.; Conley, D.J. A welcome can of worms? Hypoxia mitigation by an invasive species. Glob. Chang. Biol. 2012, 18, 422–434. [Google Scholar] [CrossRef]
- Hewitt, J.E.; Norkko, J.; Kauppi, L.; Villnäs, A.; Norkko, A.; Peters, D.P.C. Species and functional trait turnover in response to broad-scale change and an invasive species. Ecosphere 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Parker, I.M.; Simberloff, D.; Lonsdale, W.M.; Goodell, K.; Wonham, M.; Kareiva, P.M.; Williamson, M.H.; Von Holle, B.; Moyle, P.B.; Byers, J.E.; et al. Impact: Toward a framework for understanding the ecological effects of invaders. Biol. Inv. 1999, 1, 3–19. [Google Scholar] [CrossRef]
- Ruiz, G.M.; Fofonoff, P.; Hines, A.H.; Grosholz, E.D. Non-indigenous species as stressors in estuarine and marine communities: Assessing invasion impacts and interactions. Limnol. Oceanogr. 1999, 44, 950–972. [Google Scholar] [CrossRef]
- Carlton, J.T. Bioinvasion ecology: Assessing invasion impact and scale. In Aquatic Invasive Species of Europe—Distribution, Impacts and Management; Leppäkoski, E., Gollasch, S., Olenin, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 7–19. [Google Scholar]
- Renz, J.; Forster, S. Effects of bioirrigation by the three sibling species of Marenzelleria spp. on solute fluxes and porewater nutrient profiles. Mar. Ecol. Prog. Ser. 2014, 505, 145–159. [Google Scholar] [CrossRef]
- Quintana, C.O.; Raymond, C.; Nascimento, F.; Bonaglia, S.; Forster, S.; Gunnarsson, J.S.; Kristensen, E. Functional performance of three invasive Marenzelleria species under contrasting ecological conditions within the Baltic Sea. Estuaries Coast. 2018, 41, 1766–1781. [Google Scholar] [CrossRef]
- Carstensen, J.; Conley, D.J.; Bonsdorff, E.; Gustafsson, B.G.; Hietanen, S.; Janas, U.; Jilbert, T.; Maximov, A.; Norkko, A.; Norkko, J.; et al. Hypoxia in the Baltic Sea: Biogeochemical cycles, benthic fauna, and management. AMBIO 2014, 43, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Lodge, D.M. Biological invasions: Lessons for ecology. Trends Ecol. Evol. 1993, 8, 133–137. [Google Scholar] [CrossRef]
- Levine, J.M.; D’Antonio, C.M. Elton revisited: A review of evidence linking diversity and invisibility. Oikos 1999, 87, 15–26. [Google Scholar] [CrossRef]
- Ruiz, G.M.; Hewitt, C.L. Toward understanding patterns of coastal marine invasions: A prospectus. In Aquatic Invasive Species of Europe—Distribution, Impacts and Management; Leppäkoski, E., Gollasch, S., Olenin, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 529–547. [Google Scholar]
- Maximov, A.; Bonsdorff, E.; Eremina, T.; Kauppi, L.; Norkko, A.; Norkko, J. Context-dependent consequences of Marenzelleria spp. (Spionidae: Polychaeta) invasion for nutrient cycling in the Northern Baltic Sea. Oceanologia 2015, 57, 342–348. [Google Scholar] [CrossRef]
- Levin, L.A.; Boesh, D.F.; Covich, A.; Dahm, C.; Erseus, C.H.; Ewel, K.C.; Kneib, R.T.; Moldenke, A.; Palmer, M.A.; Snelgrove, P.; et al. The function of marine critical transition zones and the importance of sediment biodiversity. Ecosystems 2001, 4, 430–450. [Google Scholar] [CrossRef]
- Stachowicz, J.J.; Fried, H.; Osman, R.W.; Whitlatch, R.B. Biodiversity, invasion resistance, and marine ecosystem function: Reconciling pattern and process. Ecology 2002, 83, 2575–2590. [Google Scholar] [CrossRef]
- Naeem, S.; Knops, J.M.H.; Tilman, D.; Howe, K.M.; Kennedy, T.; Gale, S. Plant diversity increases resistance to invasion in the absence of covarying extrinsic factors. Oikos 2000, 91, 97–108. [Google Scholar] [CrossRef]
- Wardle, D.A.; Bonner, K.I.; Barker, G.M. Stability of ecosystem properties in response to above-ground functional group richness and composition. Oikos 2000, 89, 11–23. [Google Scholar] [CrossRef]
- Hammond, D.E.; Fuller, C. The use of radon-222 to estimate benthic exchange and atmospheric exchange rates in San Francisco Bay. In San Francisco Bay: The Urbanized Estuary; Conomos, T.J., Ed.; AAAS: San Francisco, CA, USA, 1979; pp. 213–230. [Google Scholar]
- Callender, E.; Hammond, D.E. Nutrient exchange across the sediment-water interface in the Potomac River Estuary. Estuar. Coast. Shelf. Sci. 1982, 15, 395–413. [Google Scholar] [CrossRef]
- Clavero, V.; Niell, F.X.; Fernandez, J.A. Effects of Nereis diversicolor O.F. Müller abundance on the dissolved phosphate exchange between sediment and overlying water in Palmones River Estuary (southern Spain). Estuar. Coast. Shelf Sci. 1991, 33, 193–202. [Google Scholar] [CrossRef]
- Forja, J.M.; Gomez-Parra, A. Measuring nutrient fluxes across the sediment-water interface using benthic chambers. Mar. Ecol. Prog. Ser. 1998, 164, 95–105. [Google Scholar] [CrossRef]
- Forster, S.; Glud, R.N.; Gundersen, J.K.; Huettel, M. In situ study of bromide tracer and oxygen flux in coastal sediments. Estuar. Coast. Shelf. Sci. 1999, 49, 813–827. [Google Scholar] [CrossRef]
- Henriksen, K.; Hansen, J.I.; Blackburn, T.H. The influence of benthic infauna on exchange rates of inorganic nitrogen between sediment and water. Ophelia 1980, 1, 249–256. [Google Scholar]
- Clavero, V.; Niell, F.X.; Fernandez, J.A. An experimental approach to quantify the influence of Nereis diversicolor in the interchange of phosphate between sediment and water. J. Exp. Mar. Biol. Ecol. 1994, 176, 257–267. [Google Scholar] [CrossRef]
- Pelegri, S.P.; Nielsen, L.P.; Blackburn, T.H. Denitrification in estuarine sediment stimulated by the irrigation activity of the amphipod Corophium volutator. Mar. Ecol. Prog. Ser. 1994, 105, 285–290. [Google Scholar] [CrossRef]
- Matisoff, G.; Wang, X. Solute transport in sediment by freshwater infaunal bioirigators. Limnol. Oceanogr. 1998, 43, 1487–1499. [Google Scholar] [CrossRef]
- Nizzoli, D.; Bartoli, M.; Cooper, M.; Welsh, D.T.; Underwood, G.J.C.; Viaroli, P. Implications for oxygen, nutrient fluxes and denitrification rates during the early stage of sediment colonisation by the polychaete Nereis spp. in four estuaries. Est. Coast. Shelf Sci. 2007, 75, 125–134. [Google Scholar] [CrossRef]
- Essink, K.; Kleef, H.L. Marenzelleria viridis (Verrill, 1873) (Polychaeta: Spionidae): A new record from Ems estuary (The Netherlands/Federal Republic of Germany). Zool. Bijdr. 1988, 38, 1–13. [Google Scholar]
- McLusky, D.S.; Hull, S.C.; Elliott, M. Variations in the intertidal and subtidal macrofauna and sediments along a salinity gradient in the upper Forth Estuary. Neth. J. Aquat. Ecol. 1993, 27, 101–107. [Google Scholar] [CrossRef]
- Bick, A.; Burckhardt, R. Erstnachweis von Marenzelleria viridis (Polychaeta, Spionidae) für den Ostseeraum, mit einem Bestimmungsschlüssel der Spioniden der Ostsee. Mitt. Zool. Mus. Berl. 1989, 65, 237–247. [Google Scholar] [CrossRef]
- Bochert, R.; Fritzsche, D.; Burckhardt, R. Influence of salinity and temperature on growth and survival of the planktonic larvae of Marenzelleria viridis (Polychaeta, Spionidae). J. Plankton Res. 1996, 18, 1239–1251. [Google Scholar] [CrossRef]
- Bochert, A.; Richard, D.; Bochert, R. Marenzelleria cf. viridis and the sulphide regime. Aquat. Ecol. 1997, 31, 223–231. [Google Scholar] [CrossRef]
- Schiedek, D. Marenzelleria cf. viridis (Polychaeta: Spionidae)—Ecophysiological adaptations to a life in the coastal waters of the Baltic Sea. Aquat. Ecol. 1997, 31, 199–210. [Google Scholar] [CrossRef]
- Zettler, M.L. Population dynamics, growth and production of the neozoon Marenzelleria cf. viridis (Verrill, 1873) (Polychaeta: Spionidae) in a coastal water of the southern Baltic Sea. Aquat. Ecol. 1997, 31, 177–186. [Google Scholar] [CrossRef]
- Zettler, M.L.; Daunys, D.; Kotta, J.; Bick, A. History and success of an invasion into the Baltic Sea: The polychaete Marenzelleria cf. viridis development and strategies. In Aquatic Invasive Species of Europe—Distribution, Impacts and Management; Leppäkoski, E., Gollasch, S., Olenin, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 66–75. [Google Scholar]
- Essink, K. Dispersal and development of Marenzelleria spp. (Polychaeta, Spionidae) populations in NW Europe and the Netherlands. Helgol. Meeresunters. 1999, 52, 367–372. [Google Scholar] [CrossRef]
- Zettler, M.L. Successful establishment of the spionid polychaete Marenzelleria viridis (Verrill 1873), in the Darss-Zingst estuary (southern Baltic) and its influence on the indigenous macrozoobenthos. Arch. Fish. Mar. Res. 1996, 43, 273–284. [Google Scholar]
- Burckhardt, R.; Schumann, R.; Bochert, R. Feeding biology of the pelagic larvae of Marenzelleria cf. viridis (Polychaeta: Spionidae) from the Baltic Sea. Aquat. Ecol. 1997, 31, 149–162. [Google Scholar] [CrossRef]
- Winkler, H.M.; Debus, L. Is the polychaete Marenzelleria viridis an important food item for fish? In Proceedings of the 13th BMB Symposium; Andrushaitis, A., Ed.; Institute of Aquatic Ecology, University of Latvia: Riga, Latvia, 1993; pp. 147–151. [Google Scholar]
- Kotta, J.; Orav, H.; Sandberg-Kilpi, E. Ecological consequence of the introduction of the polychaete Marenzelleria viridis into a shallow water biotope of the northern Baltic Sea. J. Sea Res. 2001, 46, 273–280. [Google Scholar] [CrossRef]
- Leppäkoski, E. Assessment of degree of pollution on the basis of macrozoobenthos in marine and brackish-water environments. Acta Academia Aboensis B 1975, 35, 37–47. [Google Scholar]
- Rumohr, H.; Bonsdorff, E.; Pearson, T.H. Zoobenthos succession in Baltic sedimentary habitats. Arch. Fish. Mar. Res. 1996, 44, 179–214. [Google Scholar]
- Zettler, M.L. Untersuchungen zum Makrozoobenthos des Breitlings (südliche Ostsee) unter besonderer Berücksichtigung der Crustacea. Rostocker Meeresbiol. Beitr. 1999, 7, 79–90. [Google Scholar]
- Schiedek, D. Ecophysiological capability of Marenzelleria populations inhabiting North Sea estuaries: An overview. Helgol. Meeresunter. 1999, 52, 373–382. [Google Scholar] [CrossRef]
- Saager, P.M.; Sweerts, J.-P.; Ellermeijer, H.J. A simple pore-water sampler for coarse, sandy sediments of low porosity. Limnol. Oceanogr. 1990, 35, 747–751. [Google Scholar] [CrossRef]
- Zettler, M.L. Ökologische Untersuchungen am Neozoon Marenzelleria viridis (Verrill, 1873) (Polychaeta: Spionidae) in Einem Küstengewässer der Südlichen Ostsee. Ph.D. Thesis, Rostock University, Rostock, Germany, 1996; p. 149. [Google Scholar]
- Daunys, D.; Schiedek, D.; Olenin, S. Species strategy near its boundary: The Marenzelleria cf. viridis (Polychaeta, Spionidae) case in the south-eastern Baltic Sea. Internat. Rev. Hydrobiol. 2000, 85, 639–651. [Google Scholar] [CrossRef]
- Koroleff, F.; Grasshoff, K. Determination of nutrients. In Methods of Seawater Analyses; Grasshoff, K., Ed.; Wiley: Hoboken, NY, USA, 1983; pp. 125–188. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Analyt. Chim. Acta. 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Glud, R.N.; Forster, S.; Huettel, M. Influence of radial pressure gradients on solute exchange in stirred benthic chambers. Mar. Ecol. Prog. Ser. 1996, 141, 303–311. [Google Scholar] [CrossRef]
- Ullman, W.J.; Aller, R.C. Diffusion coefficients in nearshore marine sediments. Limnol. Oceanogr. 1985, 27, 552–556. [Google Scholar] [CrossRef]
- Banta, G.T.; Holmer, M.; Jensen, M.H.; Kristensen, E. Effects of two polychaete worms, Nereis diversicolor and Arenicola marina, on aerobic and anaerobic decomposition in a sandy marine sediments. Aquat. Microb. Ecol. 1999, 19, 189–204. [Google Scholar] [CrossRef]
- Aller, R.C.; Aller, J.Y. Meiofauna and solute transport in marine muds. Limnol. Oceanogr. 1992, 37, 1018–1033. [Google Scholar] [CrossRef]
- Ekeroth, N.; Blomqvist, S.; Hall, P.O.J. Nutrient fluxes from reduced Baltic Sea sediment: Effects of oxygenation and macrobenthos. Mar. Ecol. Prog. Ser. 2016, 544, 77–92. [Google Scholar] [CrossRef]
- Hahlbeck, E.; Arndt, C.; Schiedek, D. Sulphide detoxification in Hediste diversicolor and Marenzelleria viridis, two dominant polychaete worms within the shallow coastal waters of the southern Baltic Sea. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2000, 125, 457–471. [Google Scholar] [CrossRef]
- Zilius, M.; Bartoli, M.; Bresciani, M.; Katarzyte, M.; Ruginis, T.; Petkuviene, J.; Lubiene, I.; Giardino, C.; Bukaveckas, P.A.; De Wit, R.; et al. Feedback mechanisms between cyanobacterial blooms, transient hypoxia, and benthic phosphorus regeneration in shallow coastal environments. Estuar. Coast. 2014, 37, 680–694. [Google Scholar] [CrossRef]
- Kristensen, E.; Hansen, T.; Delefosse, M.; Banta, G.T. Quintana CO Contrasting effects of the polychaetes Marenzelleria viridis and Nereis diversicolor on benthic metabolism and solute transport in sandy coastal sediment. Mar. Ecol. Prog. Ser. 2011, 425, 125–139. [Google Scholar] [CrossRef]
- Bonaglia, F.J.; Nascimento, A.; Bartoli, M.; Klawonn, I.; Brüchert, V. Meiofauna increases bacterial denitrification in marine sediments. Nat. Commun. 2014, 5, 5133. [Google Scholar] [CrossRef]
- Miron, G.; Kristensen, E. Factors influencing the distribution of nereid polychaetes: The sulfide aspect. Mar. Ecol. Prog. Ser. 1993, 93, 143. [Google Scholar] [CrossRef]
- Martin, W.R.; Banta, G.T. The measurement of sediment irrigation rates: A comparison of the Br- tracer and 222Rn/226Ra disequilibrium techniques. J. Mar. Res. 1992, 50, 125–154. [Google Scholar] [CrossRef]
- Rysgaard, S.; Christensen, P.B.; Sorensen, M.V.; Funch, P.; Berg, P. Marine meiofauna, carbon and nitrogen mineralization in sandy and soft sediments of Disko Bay, West Greenland. Aq. Microb. Ecol. 2000, 21, 59–71. [Google Scholar] [CrossRef]
- Nielsen, L.P. Denitrification in sediment determined from nitrogen isotope pairing. FEMS Microbiol. Lett. 1992, 86, 357–362. [Google Scholar] [CrossRef]
- Fritzsche, D.; von Oertzen, J.-A. Metabolic responses to changing environmental conditions in the brackish water polychaetes Marenzelleria viridis and Hediste diversicolor. Mar. Biol. 1995, 121, 693–699. [Google Scholar] [CrossRef]
- Kristensen, E. Ventilation and oxygen uptake by three species of Nereis (Annelida: Polychaeta). II. Effects of temperature and salinity changes. Mar. Ecol. Prog. Ser. 1983, 12, 299–306. [Google Scholar] [CrossRef]
- Davey, J.T. The architecture of the burrow of Nereis diversicolor and its quantification in relation to sediment-water exchange. J. Exp. Mar. Biol. Ecol. 1994, 179, 115–129. [Google Scholar] [CrossRef]
- Zettler, M.L.; Bick, A.; Bochert, R. Röhrenbau und Vertikalverteilung von Marenzelleria viridis (Polychaeta: Spionidae) in einem inneren Küstengewässer der südlichen Ostsee. Rostocker Meeresbiol. Beitr. 1994, 2, 215–225. [Google Scholar]
- Renz, J.; Forster, S. Are similar worms different? A comparative tracer study on bioturbation in the three sibling species Marenzelleria arctia, M. viridis, and M. neglecta from the Baltic Sea. Limnol. Oceanogr. 2013, 58, 2046–2058. [Google Scholar] [CrossRef]
- Dauer, D.M.; Ewing, R.M.; Tourtellotte, G.H.; Barker, H.R., Jr. Nocturnal swimming of Scolecolepides viridis (Polychaeta: Spionidae). Estuaries 1980, 3, 148—149. [Google Scholar] [CrossRef]
- Dauer, D.M.; Ewing, R.M.; Sourbeer, J.W.; Harlan, W.T.; Stokes, T.L. Nocturnal movements of the macrobenthos of the Lafayette River, Virginia. Int. Rev. Ges. Hydrobiol. 1982, 67, 761—775. [Google Scholar]
- Reible, D.D.; Popov, V.; Valsaraj, K.T.; Thibodeaux, L.J.; Lin, F.; Dikshit, M.; Todaro, M.A.; Fleeger, J.W. Contaminant fluxes from sediment due to tubificid oligochaete bioturbation. Water Res. 1996, 30, 704–714. [Google Scholar] [CrossRef]
- Yu, N.; Culver, D.A. Effective clearance rate and refiltration estimation of zebra mussels (Dreissena polymorpha) in a stratified reservoir. Fresh. Biol. 1999, 41, 481–492. [Google Scholar] [CrossRef]
- Aller, R.C. The effects of macrobenthos on chemical properties of marine sediment and overlying water. In Animal-Sediment Relations; McCall, P.L., Tevesz, M.J.S., Eds.; Plenum Press: New York, NY, USA, 1982; pp. 53–102. [Google Scholar]
- Naeem, S.; Loreau, M.; Inchausti, P. Biodiversity and ecosystem functioning: The emergence of a synthetic ecological framework. In Biodiversity and Ecosystem Functioning; Loreau, M., Naeem, S., Inchausti, P., Eds.; Oxford University Press: Oxford, UK, 2002; pp. 3–11. [Google Scholar]
- Daunys, D. Patterns of the bottom macrofauna variability and its role in the shallow coastal lagoon. Ph.D. Thesis, Klaipeda University, Klaipeda, Lithuania, 2001; 85p. [Google Scholar]
| T A X A | D E N S I T Y | B I O M A S S | ||
|---|---|---|---|---|
| Average | Min | Max | Average | |
| Ind m−2 | Ind m−2 | Ind m−2 | g AFDW m−2 | |
| Mollusca | ||||
| Mya arenaria | 425 | 127 | 637 | 55.3 |
| Cerastoderma glaucum | 127 | 0 | 255 | 0.2 |
| Peringia ulvae | 297 | 0 | 510 | 0.16 |
| Ecrobia ventrosa | 12,357 | 11,592 | 12,866 | 4.7 |
| Potamopyrgus antipodarum | 8705 | 2930 | 20,127 | 2.89 |
| Oligochaeta | ||||
| Baltidrilus costatus | 3,482 | 2293 | 5350 | 0.18 |
| Polychaeta | ||||
| Hediste diversicolor | 10,870 | 8408 | 12,357 | 36.2 |
| Polydora cornuta | 255 | 0 | 510 | 0.08 |
| Streblospio shrubsolii | 849 | 0 | 1401 | 0.07 |
| Crustacea | ||||
| Corophium volutator | 764 | 0 | 1656 | 0.22 |
| Cyathura carinata | 552 | 255 | 764 | 0.26 |
| Lekanesphaera hookeri and rugicauda | 254 | 127 | 382 | 0.09 |
| Rest | 100 | 0 | 127 | 0.05 |
| TOTAL: | 39,122 | 33,503 | 49,172 | 100.4 |
| Incubation Type | Diffusive | Total | Ratio |
|---|---|---|---|
| Experiment 1: | |||
| Defaunated sediment | 0.58 | 0.66 | 1.14 |
| Defaunated sediment (+ 1 M. neglecta) | 0.54 ± 0.01 | 0.87 ± 0.09 | 1.60 ± 0.14 |
| Experiment 2: | |||
| Natural community (+ 3 M. neglecta) | 0.76 ± 0.26 | 2.33 ± 0.79 | 3.00 ± 0.46 |
| Natural community (+ 3 H. diversicolor) | 0.53 ± 0.01 | 2.42 ± 0.39 | 3.75 ± 1.03 |
| Natural community | 0.74 ± 0.12 | 2.41 ± 0.50 | 3.26 ± 0.44 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daunys, D.; Forster, S.; Schiedek, D.; Olenin, S.; Zettler, M.L. Effect of Species Invasion on Transport of Solutes at Different Levels of Soft Sediment Macrofauna Diversity: Results from an Experimental Approach. Water 2019, 11, 1544. https://doi.org/10.3390/w11081544
Daunys D, Forster S, Schiedek D, Olenin S, Zettler ML. Effect of Species Invasion on Transport of Solutes at Different Levels of Soft Sediment Macrofauna Diversity: Results from an Experimental Approach. Water. 2019; 11(8):1544. https://doi.org/10.3390/w11081544
Chicago/Turabian StyleDaunys, Darius, Stefan Forster, Doris Schiedek, Sergej Olenin, and Michael L. Zettler. 2019. "Effect of Species Invasion on Transport of Solutes at Different Levels of Soft Sediment Macrofauna Diversity: Results from an Experimental Approach" Water 11, no. 8: 1544. https://doi.org/10.3390/w11081544
APA StyleDaunys, D., Forster, S., Schiedek, D., Olenin, S., & Zettler, M. L. (2019). Effect of Species Invasion on Transport of Solutes at Different Levels of Soft Sediment Macrofauna Diversity: Results from an Experimental Approach. Water, 11(8), 1544. https://doi.org/10.3390/w11081544






